A summary of the neurological examination and extending the neurological examination
HANDEDNESS, ORIENTATION AND SPEECH
Ask the patient if he or she is right- or left-handed. As a screening assessment, ask for the patient’s name, present location and the date. Next ask the patient to name an object pointed at and have him or her point to a named object in the room, to test for dysphasia. Ask the patient to say ‘British Constitution’ to test for dysarthria.
NECK STIFFNESS AND KERNIG’S SIGN
Ask the patient to lie flat and attempt gently to flex the patient’s head by placing a hand under the occiput. Flex the patient’s hip with the knee bent and then attempt to straighten the leg.
CRANIAL NERVES
The patient should sit over the edge of the bed if possible. Begin by general inspection of the head and neck looking for craniotomy scars, neurofibromas, facial asymmetry, ptosis, proptosis, skew deviation of the eyes or inequality of the pupils.
The second nerve
Test visual acuity with the patient wearing his or her spectacles. Each eye is tested separately, while the other is covered with a small card.
Examine the visual fields by confrontation using a hat pin or fingers. Your head should be level with the patient’s head. Each eye is tested separately. If visual acuity is very poor, the fields are mapped using the fingers.
The third, fourth and sixth nerves
Look at the pupils, noting the shape, relative sizes and any associated ptosis. Use a pocket torch and shine the light from the side to gauge the reaction of the pupils to light. Assess quickly both the direct and the consensual responses. Look for an afferent pupillary defect by moving the torch in an arc from pupil to pupil. Test accommodation by asking the patient to look into the distance and then at the hat pin or your finger held about 30 centimetres from the patient’s nose.
Assess eye movements with both eyes first, getting the patient to follow the pin in each direction. Look for failure of movement and for nystagmus. Ask about diplopia in each direction.
The fifth nerve
Test the corneal reflexes gently and ask the patient if he or she can feel the touch of the cottonwool on the cornea. The sensory component of this reflex is V and the motor component VII.
Test facial sensation in the three divisions: ophthalmic, maxillary and mandibular. Test pain sensation with the pin first and map any area of sensory loss from dull to sharp. Test light touch as well so that sensory dissociation can be detected if present.
Examine the motor division of the fifth nerve by asking the patient to clench the teeth while you feel the masseter muscles. Then get the patient to open the mouth while you attempt to force it closed; this is not possible if the pterygoid muscles are working. A unilateral lesion causes the jaw to deviate towards the weak (affected) side.
Test the jaw jerk. This is increased in cases of pseudobulbar palsy.
The seventh nerve
Test the muscles of facial expression. Ask the patient to look up and wrinkle the forehead. Look for loss of wrinkling and feel the muscle strength by pushing down on each side. This is preserved in upper motor neurone lesions because of bilateral cortical representation of these muscles.
Next ask the patient to shut the eyes tightly and compare the two sides. Tell the patient to grin and compare the nasolabial grooves.
The eighth nerve
Ask the patient to repeat a number you have softly whispered 60 centimetres away from each ear. Examine the external auditory canals and the eardrums if this is indicated.
The ninth and tenth nerves
Look at the palate and note any uvular displacement. Ask the patient to say ‘Ah’ and look for symmetrical movement of the soft palate. A unilateral lesion causes the uvula to be drawn towards the unaffected (normal) side. Test gently for sensation on the palate (the ninth nerve). Ask the patient to speak to assess hoarseness, and to cough. A bovine cough suggests bilateral recurrent laryngeal nerve lesions.
The twelfth nerve
While examining the mouth, inspect the tongue for wasting and fasciculation. Next ask the patient to protrude the tongue. With a unilateral lesion the tongue deviates towards the weaker (affected) side.
The eleventh nerve
Look for torticollis and test the sternocleidomastoid and trapezius muscles. Ask the patient to shrug the shoulders and feel the trapezius as you push the shoulders down. Then ask the patient to turn the head against resistance and also feel the bulk of the sternocleidomastoid. Then examine the skull and auscultate for carotid bruits.
UPPER LIMBS
Shake the patient’s hand firmly. Ask him or her to sit over the side of the bed facing you, if possible.
Examine the motor system systematically every time. Inspect first for wasting (both proximally and distally) and fasciculations. Do not forget to include the shoulder girdle in your inspection.
Ask the patient to hold both hands out (palms up) with the arms extended and close the eyes. Look for drifting of one or both arms (upper motor neurone weakness, cerebellar lesion or posterior column loss). Also note any tremor or pseudoathetosis due to proprioceptive loss.
Feel the muscle bulk next, both proximally and distally, and note any muscle tenderness.
Test tone at the wrists and elbows by passively moving the joints at varying velocities.
Assess power at the shoulders, elbows, wrists and fingers.
If indicated, test for an ulnar nerve lesion (Froment’s sign) and a median nerve lesion (pen-touching test).
Examine the reflexes: biceps (C5, C6), triceps (C7, C8) and brachioradialis (C5, C6).
Assess coordination with finger–nose testing and look for dysdiadochokinesis and rebound.
Motor weakness can be due to an upper motor neurone lesion, a lower motor neurone lesion or a myopathy. If there is evidence of a lower motor neurone lesion, consider anterior horn cell, nerve root or brachial plexus lesions, peripheral nerve lesions or a motor peripheral neuropathy.
Examine the sensory system after motor testing, because this can be time-consuming.
First test the spinothalamic pathway (pain and temperature). Demonstrate to the patient the sharpness of a pin on the anterior chest wall or forehead. Then ask him or her to close the eyes and tell you if the sensation is sharp or dull. Start proximally and test each dermatome. Map the abnormal area. As you are assessing, try to fit any sensory loss into dermatomal (cord or nerve root lesion), peripheral nerve, peripheral neuropathy (glove) or hemisensory (cortical or cord) distribution. It is not usually necessary to test temperature.
Next test the posterior column pathway (vibration and proprioception). Use a 128 Hz tuning fork to assess vibration sense. Place the vibrating fork on a distal interphalangeal joint when the patient has the eyes closed and ask whether it can be felt. If so, ask the patient to tell you when the vibration ceases and then, after a short wait, stop the vibrations. If the patient has deficient sensation, test at the wrist, then at the elbow and then at the shoulder.
Examine proprioception with the distal interphalangeal joint of the little finger. When the patient has the eyes open, grasp the distal phalanx from the sides and move it up and down to demonstrate, then ask the patient to close the eyes and repeat the manoeuvre. Normally, movement through even a few degrees is detectable, and the patient can tell whether it is up or down. If there is an abnormality, test larger movements and then proceed to test the wrist and elbows similarly if necessary.
Test light touch with cottonwool. Touch the skin lightly (do not stroke) in each dermatome.
Feel for thickened nerves—the ulnar at the elbow, the median at the wrist and the radial at the wrist—and feel the axillae if there is evidence of a proximal lesion. Note any scars, and finally examine the neck if relevant.
LOWER LIMBS
Test the stance and gait first if possible. Then put the patient in bed with the legs entirely exposed. Place a towel over the groin—note whether a urinary catheter is present.
Look for muscle wasting and fasciculations. Note any tremor. Feel the muscle bulk of the quadriceps and run your hand up each shin, feeling for wasting of the anterior tibial muscles.
Test tone at the knees and ankles. Test clonus at this time. Push the lower end of the quadriceps sharply down towards the knee. Sustained rhythmical contractions indicate an upper motor neurone lesion. Also test the ankle by sharply dorsiflexing the foot with the knee bent and the thigh externally rotated.
Assess power next at the hips, knees and ankles.
Elicit the reflexes: knee (L3, L4), ankle (S1, S2) and plantar response (L5, S1, S2).
Test coordination with the heel–shin test, toe–finger test and tapping of the feet.
Examine the sensory system as for the upper limbs: pinprick, then vibration and proprioception, and then light touch. If there is a peripheral sensory loss, attempt to establish a sensory level by moving the pin up the leg and onto the abdomen and, if necessary, onto the chest. Examine sensation in the saddle region and test the anal reflex (S2, S3, S4).
Go to the back. Look for deformity, scars and neurofibromas. Palpate for tenderness over the vertebral bodies and auscultate for bruits. Perform the straight leg-raising test.
Diagnostic testing
Neurological diagnosis depends on the history and examination. Imaging can confirm the location of the lesion and the likely pathology. Special testing is guided by the clinical setting.
LUMBAR PUNCTURE
If acute meningitis is suspected, or less often if a CT of the brain is negative but you clinically suspect a subarachnoid haemorrhage, a lumbar puncture (LP) is indicated. An LP may also help in the diagnosis of certain neurological diseases, such as carcinomatous meningitis, tuberculous meningitis, central nervous system syphilis or vasculitis, idiopathic intracranial hypertension (pseudotumour cerebri) or normal-pressure hydrocephalus. Less often, an LP may be helpful in diagnosing multiple sclerosis, Guillain-Barré syndrome or a paraneoplastic syndrome.
If there is papilloedema on fundoscopy or focal neurological signs (e.g. one side is weak), a computed tomography (CT) or magnetic resonance imaging (MRI) scan of the brain should be undertaken first to exclude raised intracranial pressure (because of the risk of cerebral herniation after an LP in this setting).
The cerebrospinal fluid (CSF) is sent for Gram staining and culture as well as cell count, protein and glucose testing. Special tests may include fungal serology, syphilis (VDRL), acid fast smear and culture (TB), cryptococcal antigen test and fungal serology.
Remember, if acute bacterial meningitis is suspected, start the patient on antibiotics as soon as the CSF from the LP and the blood cultures have been collected—do not wait.
NEUROLOGICAL IMAGING
CT and MRI scans have revolutionised neurological investigations. These techniques are generally non-invasive, although CT scans expose patients to moderately large doses of radiation and both types of scans are often used with contrast agents, which can affect some patients. MRI scans cannot be performed on patients with magnetic metal implants (e.g. most pacemakers).
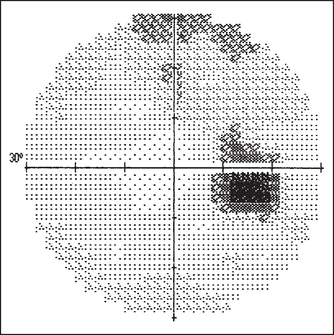
Figure 36.9 Visual field charts Both eyes superimposed. The blind spot is indicated in a darker colour. Stamper RL, Visual field interpretation. Becker-Shaffer’s Diagnosis and Therapy of the Glaucomas. 2009: Elsevier.
Interpretation of the scans often requires the assessment of multiple planes and slices or of three-dimensional reconstructions.
Some common examples of important diagnostic scans are shown in Figures 36.2–36.8 and 36.10.
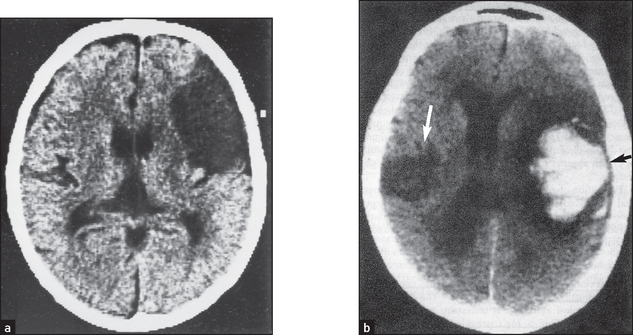
Figure 36.2 Embolic stroke These CT scans show a large cortical infarct in the middle cerebral artery in a patient with atrial fibrillation. (Crawford. Cardiology, 3rd edn. Mosby, 2009.)
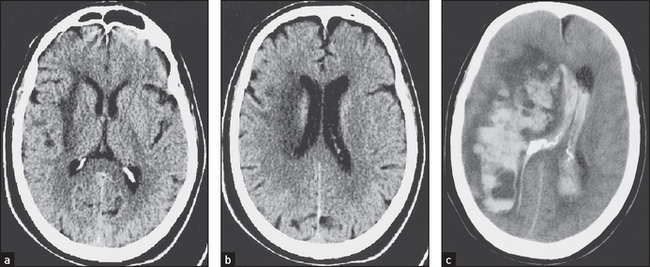
Figure 36.3 Haemorrhagic stroke These CT scans without contrast injection ((a) and (b)) show low-density changes confined to the deep subcortical regions in a patient with acute left-sided weakness. The image in (c) taken two days later shows extensive haemorrhage within the area of infarction, which has ruptured into the ventricular system. (Haaga J. CT and MRI of the whole body, 5th edn. Mosby, 2008.)
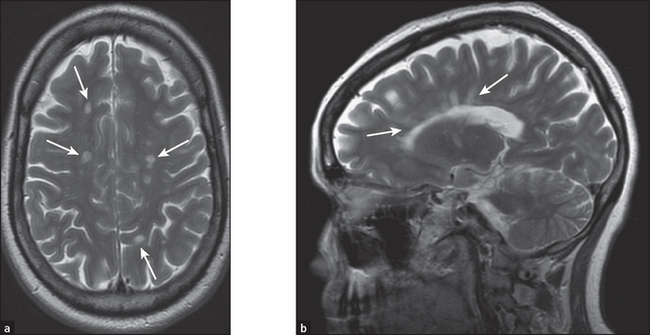
Figure 36.4 Multiple sclerosis The lesions of multiple sclerosis on MRI tend to involve the periventricular area, the corpus callosum and the optic nerves (solid white arrows). (a) The lesions produce discrete, globular foci of high-signal intensity (white) on T2-weighted MRI scans. (b) Ovoid lesions with their long axis perpendicular to the ventricular surface are called Dawson’s fingers (solid white arrows). (Herring W. Learning radiology: recognising the basics, 2dn edn. Saunders, 2011.)
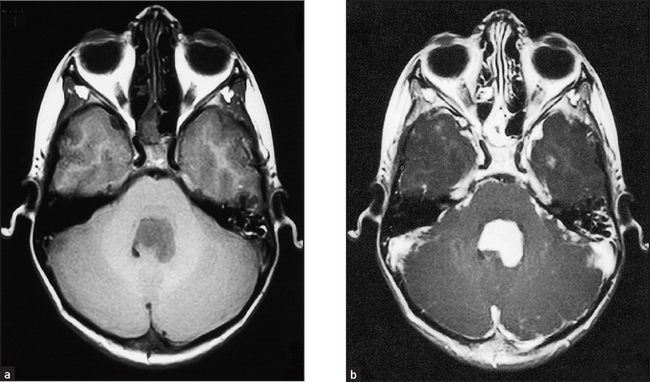
Figure 36.5 Cerebellar tumour This is the typical appearance of a brain tumour on MRI, in this case a cerebellar medulloblastoma. (a) The tumour has a low signal on spin-echo T1-weighted images. (b) After injection of gadolinium (a contrast agent) the tumour enhances avidly, indicating a breakdown of the blood–brain barrier. (Adam A. Grainger & Allison’s diagnostic radiology, 5th edn. Churchill Livingstone, 2008.) Churchill Livingstone
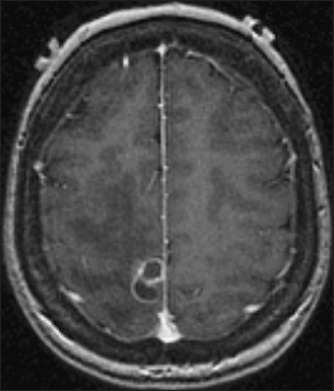
Figure 36.6 Brain abscess This gadolinium-enhanced MRI scan shows a multi-loculated ring-enhancing lesion caused by Nocardiainfection. (Goldman L. Goldman’s Cecil medicine, 24th edn. Saunders, 2011.)
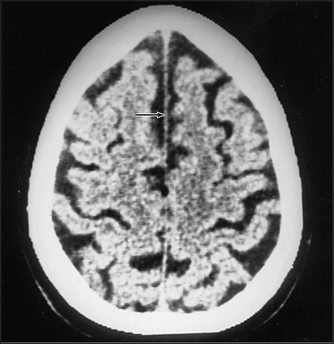
Figure 36.7 Cerebral atrophy In this CT scan, note the expanded sulci, the shrunken gyri and the retraction of the cerebral cortex from the inner table of the skull. The ventricles have expanded. (Kaufman D. Clinical neurology for psychiatrists, 6th edn. Saunders, 2006.)
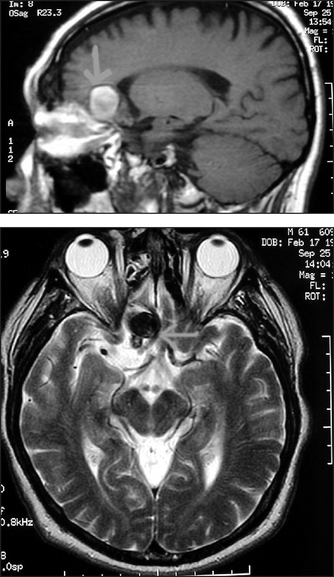
Figure 36.8 Cerebral aneurysm MRI scan showing a partly thrombosed giant aneurysm. (Layon AJ. Textbook of neurointensive care, 1st edn. Saunders, 2003.)
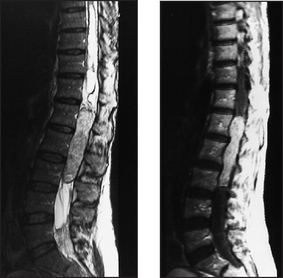
Figure 36.10 Spinal cord tumour An MRI scan showing an intradural and partly extradural right C3 schwannoma. Axial (a) and coronal (b) T1-weighted images with gadolinium contrast. Note the large lobular tumour extending into the spinal canal and compressing the spinal cord (arrow). (Adam A. Grainger & Allison’s diagnostic radiology, 5th edn. Churchill Livingstone, 2008.) Churchill Livingstone
