1 Abella B.S., et al. Quality of cardiopulmonary resuscitation during in-hospital cardiac arrest. J Am Med Assoc. 2005;293(3):305-310.
2 Abman S.H. Pulmonary hypertension in children: a historical overview. Pediatr Crit Care Med. 2010;11(Suppl.):S4-S9.
3 Absi M.A., Lutterman J., Wetzel G.T. Noninvasive cardiac output monitoring in the pediatric cardiac intensive care unit. Curr Opin Cardiol. 2010;25:77-79.
4 Adams J.E.I.I.I., et al. Cardiac troponin I: a marker with high specificity for cardiac injury. Circulation. 1993;88:101-106.
5 Adelson P.D., et al. Guidelines for the acute management of severe traumatic brain injury in infants, children, and adolescents. Pediatr Crit Care Med. 2003;4(3 Suppl.):S12-S18.
6 Adrie C., et al. Successful cardiopulmonary resuscitation after cardiac arrest as a “sepsis-like” syndrome. Circulation. 2002;106(5):562-568.
7 Albert N.M., et al. Use of aldosterone antagonists in heart failure. J Am Med Assoc. 2009;302(15):1658-1665.
8 Amaral A., Opal S.M., Vincent J.L. Coagulation in sepsis. Intensive Care Med. 2004;30(6):1032-1040.
9 American College of Chest Physicians, and Society of Critical Care Medicine Consensus Conference. Definitions of sepsis and organ failure and guidelines for the use of innovative therapies in sepsis. Crit Care Med. 1992;20:864-875.
10 Annane D., et al. Effect of treatment with low doses of hydrocortisone and fludrocortisone on mortality in patients with septic shock. J Am Med Assoc. 2002;288:862-867.
11 Annane D., Bellissant E., Cavaillon J. Septic shock. Lancet. 2005:365-378.
12 Annane D., et al. Norepinephrine plus dobutamine versus epinephrine alone for management of septic shock: a randomized trial. Lancet. 2007;360:767-784.
13 Annane D., et al. Corticosteroids in the treatment of severe sepsis and septic shock in adults: a systematic review. J Am Med Assoc. 2009;301(22):2362-2375.
14 Atabaki S.M. Pediatric head injury. Ped Rev. 2007;28(6):215-223.
15 Atkins D.L., et al. Epidemiology and outcomes from out-of-hospital cardiac arrest in children: the Resuscitation Outcomes Consortium Epistry-Cardiac Arrest. Circulation. 2009;119(11):1484-1491.
16 Aurigemma G.P., Gaasch W.H. Diastolic heart failure. N Engl J Med. 2004;351(11):1097-1105.
17 Azzopardi D.V., et al. Moderate hypothermia to treat perinatal asphyxial encephalopathy. N Engl J Med. 2009;361(14):1349-1358.
18 Babyn P.S., Gahunia H.K., Massicote P. Pulmonary thromboembolism in children. Pediatr Radiol. 2005;35:258-274.
19 Baden H.P. Does nesiritide offer something new in the management of pediatric heart failure? Pediatric Crit Care Med. 2005;6:613.
20 Baird J.S., Greene A., Schleien C.L. Massive pulmonary embolus without hypoxemia. Pediatr Crit Care Med. 2005;6(5):602-603.
21 Bakker J., et al. Blood lactate levels are superior to oxygen-derived variables in predicting outcome in human septic shock. Chest. 1991;99:956-962.
22 Bakker J., et al. Serial blood lactate levels can predict the development of multiple organ failure following septic shock. Am J Surg. 1996;171:221-226.
23 Baraff L.J. Capillary refill: is it a useful clinical sign? Pediatrics. 1993;92:723-724.
24 Barrett L.K., Singer M., Clapp L.H. Vasopressin: mechanisms of action on the vasculature in health and in septic shock. Crit Care Med. 2007;35(1):33-40.
25 Barton P., et al. Hemodynamic effects of intravenous milrinone lactate in pediatric patients with septic shock. Chest. 1996;109:1302-1312.
26 Berg M., et al. Part 13, pediatric basic life support. In 2010 American Heart Association Guidelines for Cardiopulmonary Resuscitation and Emergency Cardiovascular Care. Circulation. 2010;122(Suppl. 3):S862-S875.
27 Berg R.A., et al. Adverse hemodynamic effects of interrupting chest compressions for rescue breathing during cardiopulmonary resuscitation for ventricular fibrillation cardiac arrest. Circulation. 2001;104(20):2465-2470.
28 Berkowitz I.D., et al. Blood flow during cardiopulmonary resuscitation with simultaneous compression and ventilation in infant pigs. Pediatr Res. 1989;26:558.
29 Bernard S.A., et al. Treatment of comatose survivors of out-of-hospital cardiac arrest with induced hypothermia. N Engl J Med. 2002;346(8):557-563.
30 Bernus A., et al. Brain natriuretic peptide levels in managing pediatric patients with pulmonary atrial hypertension. Chest. 2009;135:745-751.
31 Bersin R.M., Arieff A.I. Use of sodium salts in the treatment of hypoxia-induced acidosis. Circulation. 1988;77:227.
32 Bettendorf M., et al. Tri-iodothyronine treatment in children after cardiac surgery: a double-blind, randomized, placebo-controlled study. Lancet. 2000;356:529-534.
33 Bissler J.J., Welch T.R., Loggie J.M.H. Paradoxical hypertension in hypovolemic children. Pediatr Emerg Care. 1991;7:350-352.
34 Boehmer J.P., Popjes E. Cardiac failure: mechanical support strategies. Crit Care Med. 2006;24(9):S268-S277.
35 Bratton S.L. Patterns of septic shock in children: warm versus cold shock. AAP Grand Rounds. 2009;21(2):16.
36 Braun J.P., et al. Successful treatment of dilative cardiomyopathy in a 12-year-old-girl using the calcium sensitizer levosimendan after weaning from mechanical biventricular assist support. J Cardiothorac Vasc Anesth. 2004;18:772-774.
37 Brierley J., Peters M.J. Distinct hemodynamic patterns of septic shock at presentation to pediatric intensive care. Pediatrics. 2008;122(4):752-759.
38 Brierley J., Carcillo J.A., Choong K., Cornell T., et al. Clinical practice parameters for hemodynamic support of pediatric and neonatal septic shock: 2007 update from the American College of Critical Care Medicine. Crit Care Med. 2009;37:666-688.
39 Brilli R.J., Goldstein B. Pediatric sepsis definitions: past, present and future. Pediatr Crit Care Med. 2005;6(3):S6-S8.
39a Brocklehurst P. and the Neonatal Immunotherapy Study (INIS) Collaborative Group. Treatment of neonatal sepsis with intravenous immune globulin. N Engl J Med. 2011;365:1201-1211.
40 Bromberg B.I., et al. Recognition and management of nonpenetrating cardiac trauma in children. J Pediatr. 1996;128:536-541.
41 Bronicki R.A., Anas N.G. Cardiopulmonary interaction. Pediatr Crit Care Med. 2009;10:313-322.
42 Brown C.G., Dzwonczyk R. Signal analysis of the human electrocardiogram during ventricular fibrillation: frequency and amplitude parameters as predictors of successful countershock. Ann Emerg Med. 1996;27(2):184-188.
43 Brown N.J., Vaughan D.E. Angiotensin-converting enzyme inhibitors. Circulation. 1998;97:1411-1420.
44 Bunnell E., Parrillo J.E. Cardiac dysfunction during septic shock. Clin Chest Med. 1996;17:237-248.
45 Burch M. Heart failure in the young. Heart. 2002;88:198-202.
46 Burns J.P. Septic shock in the pediatric patient: pathogenesis and novel treatments. Pediatr Emerg Care. 2003;19(2):112-115.
47 Burton A.L. The law of the heart. In: Burton A.C., editor. Physiology and biophysics of the circulation. Chicago: Year Book Medical Publishers, 1972.
48 Cameron J.L., Fontanarosa P.B., Passalaqua A.M. A comparative study of peripheral to central circulation delivery times between intraosseous and intravenous injection using a radionuclide technique in normovolemic and hypovolemic canines. J Emerg Med. 1989;7(2):123-127.
49 Canter C.E., Strauss A.W. Cardiomyopathies—when to think of congenital causes. Contemp Pediatr. 1995;12:25-40.
50 Carcillo J.A., David A.L., Zaritsky A. Role of early fluid resuscitation in pediatric septic shock. J Am Med Assoc. 1991;266:1242-1245.
51 Carcillo J.A., Cunnion R.E. Septic shock. Crit Care Clinics. 1997;13:553-574.
52 Carcillo J.A., et al. Clinical practice parameters for hemodynamic support of pediatric and neonatal patients in septic shock. Crit Care Med. 2002;30:1365-1378.
53 Carcillo J.A., et al. Shock: an overview. In: Wheeler D.S., et al, editors. Resuscitation and stabilization of the critically ill child. London: Springer-Verlag; 2009:89-113.
54 Carter B.G., Butt W., Taylor A. ICP and CPP: excellent predictors of long term outcome in severely brain injured children. Childs Nerv Syst. 2008;24:245-251.
55 Casella E.S., Rogers M.L., Zahka K.G. Developmental physiology of the cardiovascular system. In: Rogers M.C., editor. Textbook of pediatric intensive care. Baltimore: Williams and Wilkins, 1987.
56 Ceneviva G., et al. Hemodynamic support in fluid-refractory pediatric septic shock. Pediatrics. 1998;102:e19.
57 Chameides L., editor. Textbook of pediatric advanced life support. Dallas: American Heart Association, 1988.
57a Chameides L., Samson R., Schexnayder S., Hazinski M.F., editors. Pediatric advanced life support provider manual. Dallas: American Heart Association, 2011.
58 Checchia P.A., Dietrich A., Perkin R.M. Current concepts in the recognition and management of pediatric cardiogenic shock and congestive heart failure. Ped Emerg Med Reports. 2000;5(5):41-60.
59 Chen D., et al. Cytokines and acute heart failure. Crit Care Med. 2008;26(Suppl.):S9-S16.
60 Chesnut R.M. Avoidance of hypotension: Condition sine qua non of successful severe head-injury management. J Trauma. 1997;42:S4-S9.
61 Choi P., et al. Crystalloids vs. colloids in fluid resuscitation: a systematic review. Crit Care Med. 1999;27:200-210.
62 Choong K., Kisson N. Vasopressin in pediatric shock and cardiac arrest. Pediatr Crit Care Med. 2008;9:372-379.
63 Cobb J.P. Use of nitric oxide synthase inhibitors to treat septic shock: the light has changed from yellow to red. Crit Care Med. 1999;27:855-856.
64 Costello J.M., Goodman D.M., Green T.P. A review of the natriuretic hormone system’s diagnostic and therapeutic potential in critically ill children. Pediatr Crit Care Med. 2006;7(4):308-318.
65 Critchley L., Peng Z., Fok B. Testing the reliability of a new ultrasound cardiac output monitor, the USCOM, by using aortic flow probes in anesthetized dogs. Anesth Analg. 2005;100(3):748-753.
66 Crookes B.A., et al. Noninvasive muscle oxygenation to guide fluid resuscitation after traumatic shock. Surgery. 2004;135:662-670.
67 de Jonge R.C.J., Polderman K.H., Gemke R. Central venous catheter use in the pediatric patient: Mechanical and infectious complications. Pediatr Crit Care Med. 2005;6:329-339.
68 De Kleijn E.D., et al. Low serum cortisol in combination with high adrenocorticotrophic hormone concentrations is associated with poor outcome in children with severe meningococcal disease. Pediatr Infect Dis J. 2002;21:330-336.
69 Dean J.M., et al. Age-related changes in chest geometry during cardiopulmonary resuscitation. J Appl Physiol. 1987;62(6):2212-2219.
70 DeBacker D., et al. Comparison of dopamine and norepinephrine in the treatment of shock. N Engl J Med. 2010;362:779-789.
71 Dellinger R.P., et al. Surviving sepsis campaign: international guidelines for management of severe sepsis and septic shock. Crit Care Med. 2008;36(1):196-327.
72 deOliveira C.F., et al. ACCM/PALS haemodynamic support guidelines for paediatric septic shock: an outcomes comparison with and without monitoring central venous oxygen saturation. Intensive Care Med. 2008;34:1065-1075.
73 Diana M.C., et al. Sudden death in an infant revealing atypical Kawasaki disease. Pediatr Emerg Care. 2006;22(1):35-37.
74 Didwania A., et al. Effect of intravenous lactated Ringer’s solution infusion on the circulating lactate concentration. Crit Care Med. 1997;25:1851-1854.
75 Domico M., et al. Elevation of brain natriuretic peptide level in children with septic shock. Pediatr Crit Care Med. 2008;9:478-483.
76 Dominquez S.R., et al. Kawasaki disease in a pediatric intensive care unit: a case-control study. Pediatrics. 2008;122:e786-e790.
77 Donoghue A., et al. Bradycardia in children. Pediatrics. 2009;124(6):1541-1548. Epub 2009 Nov 16
78 Dowd M.D., Krug S. Pediatric blunt cardiac injury: epidemiology, clinical features, and diagnosis. J Trauma. 1996;40:61-67.
79 Driscoll D.J. Left-to-right shunt lesions. Pediatr Clin North Am. 1999;46:355-368.
80 Dudley N.C., et al. The effect of family presence on the efficiency of pediatric trauma resuscitations. Ann Emerg Med. 2009;53(6):777-784. e773
81 Dueck M.H., et al. Trends but not individual values of central venous oxy gen saturation agree with mixed venous oxygen saturation during varying hemodynamic conditions. Anesthesiology. 2005;103(2):249-257.
82 Dung N.M., et al. Fluid replacement in dengue shock syndrome: a randomized, double-blind comparison of four intravenous-fluid regimens. Clin Infect Dis. 1999;29:787-794.
83 Duser M.W., et al. Ischemic skin lesions as a complication of continuous vasopressin infusion in catecholamine-resistant vasodilatory shock: incidence and risk factors. Crit Care Med. 2003;31:1394-1398.
84 Eftestol T., Sunde K., Steen P.A. Effects of interrupting precordial compressions on the calculated probability of defibrillation success during out-of-hospital cardiac arrest. Circulation. 2002;105(19):2270-2273.
85 Eisenberg M., Bergner L., Halstrom A. Epidemiology of cardiac arrest and resuscitation in children. Ann Emerg Med. 1983;12:672.
86 Ellis D.Y., et al. Cricoid pressure in emergency department rapid sequence tracheal intubations: a risk-benefit analysis. Ann Emerg Med. 2007;50(6):653-665.
87 El-Nawawy A., et al. Intravenous polyclonal immunoglobulin administration to sepsis syndrome patients: a prospective study in a pediatric intensive care unit. J Trop Pediatr. 2005;51:271-278.
88 Emory E.F., Greenough A. Efficacy of low-dose dopamine infusion. Acute Paediatr. 1993;82:430-432.
89 Ernest D., Belzberg A.S., Dodek P.M. Distribution of normal saline and 5% albumin infusion in septic patients. Crit Care Med. 1999;27:46-50.
90 Fakler U., Pauli C., Balling G., Lorenz H.P., Eicken A., Hennig M., et al. Cardiac index monitoring by pulse contour analysis and thermodilution after pediatric cardiac surgery. J Thorac Cardiovasc Surg. 2007;133(1):224-228.
91 Fadel P.J., et al. Noninvasive assessment of sympathetic vasoconstriction in human and rodent skeletal muscle using near-infrared spectroscopy and Doppler ultrasound. J Appl Physiol. 2004;96:1323-1330.
92 Falk J.L., O’Brien J.F., Kerr R. Fluid resuscitation in traumatic hemorrhagic shock. Crit Care Clin. 1992;8:323-340.
93 Faustino E.V., Apkon M. Persistent hyperglycemia in critically ill children. J pediatr. 2005;146:30-34.
94 Fedderly R.T. Left ventricular outflow obstruction. Pediatr Clin North Am. 1999;46:369-383.
95 Feneley M.P., et al. Influence of compression rate on initial success of resuscitation and 24 hour survival after prolonged manual cardiopulmonary resuscitation in dogs. Circulation. 1988;77(1):240-250.
96 Finfer S., et al. A comparison of albumin and saline for fluid resuscitation in the intensive care unit. N Engl J Med. 2004;350(22):2247-2256.
97 Fried I., et al. Comparison of N-terminal Pro-B-Type natriuretic peptide levels in critically ill children with sepsis versus acute left ventricular dysfunction. Pediatrics. 2006;118:e1165.
98 Friedman W.F., George B.L. Treatment of congestive heart failure by altering loading conditions of the heart. J Pediatr. 1985;106:697.
99 Gaines B.A., Ford H.R. Abdominal and pelvic trauma in children. Crit Care Med. 2002;30(11 Suppl.):S416-S423.
100 Gausch W.H. Diagnosis and treatment of heart failure based on left ventricular systolic or diastolic dysfunction. J Am Med Assoc. 1994;271:1276-1280.
101 Gazmuri R.J., et al. Cardiac effects of carbon dioxide-consuming and carbon dioxide-generating buffers during cardiopulmonary resuscitation. J Am Coll Cardiol. 1990;15(2):482-490.
102 Gazmuri R., et al. Arterial PCO2 as an indicator of systemic perfusion during CPR. Crit Care Med. 1989;17:237.
103 Gazmuri R., et al. Cardiac effects of carbon dioxide-consuming and carbon dioxide-generating buffers during cardiopulmonary resuscitation. J Am Coll Cardiol. 1990;15:482.
104 Gillis J., et al. Results of inpatient pediatric resuscitation. Crit Care Med. 1986;14:469.
105 Gluckman P.D., et al. Selective head cooling with mild systemic hypothermia after neonatal encephalopathy: multicentre randomised trial. Lancet. 2005;365(9460):663-670.
106 Goetting M.G., Paradis N.A. High dose epinephrine in refractory pediatric cardiac arrest. Crit Care Med. 1989;17:1258.
107 Goetting M.G., Paradis N.A. High-dose epinephrine improves outcome from pediatric cardiac arrest. Ann Emerg Med. 1990;20:22.
108 Goldman A.P., et al. Extracorporeal support for intractable cardiorespiratory failure due to meningococcal disease. Lancet. 1997;349:466-469.
109 Goldstein B., et al. International pediatric sepsis consensus conference: definitions for sepsis and organ dysfunction in pediatrics. Pediatr Crit Care Med. 2005;6:2-8.
110 Goldstein B., et al. ENHANCE: Results of a global open-label trial of Drotrecogin alfa (activated) in children with severe sepsis. Pediat Crit Care Med. 2006;7:200-211.
111 Goodfriend T.L., Elliot M.E., Catt K.J. Angiotensin receptors and their antagonists. N Engl J Med. 1996;334:1649-1654.
112 Gorelick M.H., Shaw K.N., Baker M.D. Effect of ambient temperature on capillary refill in healthy children. Pediatrics. 1993;92:699-702.
113 Gorelick M.H., Shaw K.N., Murphy K.O. Validity and reliability of clinical signs in the diagnosis of dehydration in children. Pediatrics. 1997;99:e6.
114 Greyson C.R. Pathophysiology of right ventricular failure. Crit Care Med. 2008;36(Suppl.):S57-S65.
115 Grossman W. Diastolic dysfunction in CHF. N Engl J Med. 1991;325:1557-1564.
116 Guay J., Lortie L. An evaluation of pediatric in-hospital advanced life support interventions using the pediatric Utstein guidelines: a review of 203 cardiorespiratory arrests. Can J Anaesth. 2004;51(4):373-378.
117 HACA (HACASG). Mild therapeutic hypothermia to improve the neurologic outcome after cardiac arrest. N Engl J Med. 2002;346(8):549-556.
118 Haites N.E., et al. Assessment of cardiac output by Doppler ultrasound technique alone. Br Heart J. 1985;53(2):123-129.
119 Han Y.Y., et al. Early reversal of pediatric-neonatal septic shock by community physicians is associated with improved outcome. Pediatrics. 2003;112:793-799.
120 Haque I.U., Zaritsky A.L. Analysis of the evidence for the lower limit of systolic and mean arterial pressure in children. Pediatr Crit Care Med. 2007;8(2):138-144.
121 Hartsilver E.L., Vanner R.G. Airway obstruction with cricoid pressure. Anaesthesia. 2000;55(3):208-211.
122 Haupt M.T. Impaired oxygen extraction in sepsis: is supranormal oxygen delivery helpful? Crit Care Med. 1997;25:904-905.
123 Haupt M.T., Kaufman B.S., Carlson R.W. Fluid resuscitation in patients with increased vascular permeability. Crit Care Clin. 1992;8:341-353.
124 Hayes M.A., et al. Oxygen transport patterns in patients with sepsis syndrome or septic shock: influence of treatment and relationship to outcome. Crit Care Med. 1997;25:926-936.
125 Hayes M.A., et al. Symmetrical peripheral gangrene: association with noradrenaline administration. Intensive Care Med. 1992;18:433-436.
126 Hazinski M.F., Nolan J.N. Part 1: executive summary. In 2010 International Consensus on CPR Science with Treatment Recommendations. Circulation. 2010;122(Suppl. 2):S250-S275.
127 Hazinski M.F. Sudden cardiac death in children. Crit Care Q. 1984;7:59.
128 Heerman W., et al. Accuracy of non-invasive cardiac monitoring (USCOM). Crit Care Med. 2006;34(12 Suppl. 2):A61.
129 Hickey R.W., et al. Hypothermia and hyperthermia in children after resuscitation from cardiac arrest. Pediatrics. 2000;106(pt 1)(1):118-122.
130 Hinkle A.J. A rapid and reliable method of selecting endotracheal tube size in children. Anesth Analg. 1988;67:S-592. (abstract)
131 Hirsch R., et al. Cardiac troponin I in pediatrics: normal values and potential use in the assessment of cardiac injury. J Pediatr. 1997;130:872-877.
132 Hoffman G.M., Ghanayem N.S., Tweddell J.S. Noninvasive assessment of cardiac output. Semin Thorac Cardiovasc Surg Pediatr Card Surg Ann. 2005;8:12-21.
133 Hoffman G.M., et al. Changes in cerebral and somatic oxygenation during stage 1 palliation of hypoplastic left heart syndrome using continuous regional cerebral perfusion. J Thorac Cardiovasc Surg. 2004;127:223-233.
134 Hoffman T.M., et al. Efficacy and safety of milrinone in preventing low cardiac output syndrome in infants and children after corrective surgery for congenital heart disease. Circulation. 2003;107:996-1002.
135 Hotchkiss R.S., Karl I.E. The pathophysiology and treatment of sepsis. N Engl J Med. 2003;348:138-150.
136 Hunter J.J., Chien K.R. Signaling pathways for cardiac hypertrophy and failure. N Engl J Med. 1999;341:1276-1283.
137 Inwald D.P., et al. Emergency management of children with severe sepsis in the United Kingdom: the results of the Paediatric Intensive Care Society sepsis audit. Arch Dis Child. 2009;94:348-353.
138 Jacobs B.R. Central venous catheter occlusion and thrombosis. Crit Care Clin. 2003;19:489-514.
139 Jezierski M. Infant death: guidelines for support of parents in the emergency department. J Emerg Nurs. 1989;15(6):475-476.
140 Kahn J.M., Kress J.P., Hall J.B. Skin necrosis after extravasation of low-dose vasopressin administered for septic shock. Crit Care Med. 2002;30:1899-1901.
141 Kallen R.J. The management of diarrheal dehydration in infants using parenteral fluids. Pediatr Clin North Am. 1990;37:265.
142 Kanaan U.S., Chiang V.W. Cardiac troponins in pediatrics. Pediatr Emerg Care. 2004;20(5):323-329.
143 Kanegaye J.T., et al. Recognition of a Kawasaki disease shock syndrome. Pediatrics. 2009;123(5):e783-e789.
144 Kanter R.K., et al. Pediatric emergency intravenous access: evaluation of a protocol. Am J Dis Child. 1986;140:132.
145 Kellum J.A., Decker J. Use of dopamine in acute renal failure: a meta-analysis. Crit Care Med. 2001;29:1526-1531.
146 Kern K.B., et al. Importance of continuous chest compressions during cardiopulmonary resuscitation: improved outcome during a simulated single lay-rescuer scenario. Circulation. 2002;105(5):645-649.
147 Kern K.B., et al. A study of chest compression rates during cardiopulmonary resuscitation in humans. The importance of rate-directed chest compressions. Arch Intern Med. 1992;152(1):145-149.
148 Kitamura T., et al. Conventional and chest-compression-only cardiopulmonary resuscitation by bystanders for children who have out-of-hospital cardiac arrests: a prospective, nationwide, population-based cohort study. Lancet. 2010;375(9723):1347-1354.
149 Kleinman M., et al. Part 14, pediatric advanced life support in 2010 American heart association guidelines for cardiopulmonary resuscitation and emergency cardiovascular care. Circulation. 2010;122(Suppl. 3):S876-S908s.
150 Klemperer J.D., et al. Thyroid hormone treatment after coronary-artery bypass surgery. N Engl J Med. 1995;333:1522-1527.
151 Klinzing S., et al. High dose vasopressin is not superior to norepinephrine in septic shock. Crit Care Med. 2003;31:2646-2650.
152 Knapp J.F. Practical issues in the care of pediatric trauma patients. Curr Probl Pediatr. 1998;28(10):305-328.
153 Knoester H., et al. Cardiac function in pediatric septic shock survivors. Arch Pediatr Adolesc Med. 2008;162(12):1164-1168.
154 Konstantinides S. Acute pulmonary embolism. N Engl J Med. 2008;359(26):2804-2813.
155 Korth S.K. Unexpected pediatric death in the emergency department: supporting the family. J Emerg Nurs. 1988;14:302.
156 Kouwenhoven W.B., Jude J.R., Knickerbocker G.G. Closed-chest cardiac massage. J Am Med Assoc. 1960;173:1064-1067.
157 Krivitski N.M., Kislukhin W., Thuramalla N.V. Theory and in-vitro validation of a new extracorporeal AV loop approach for hemodynamic assessment in pediatric and neonatal ICU patients. Pediatr Crit Care Med. 2008;9(4):423-428.
158 Kurth C.D., Levy W.J., McCann J. Near-infrared spectroscopy cerebral oxygen saturation thresholds for hypoxia-ischemia in piglets. J Cereb Blood Flow Metab. 2002;22:335-341.
159 Lacroix J., Cotting J. Severity of illness and organ dysfunction scoring in children. Pediatr Crit Care Med. 2005;6(3):S126-S134.
160 Lacroix J., et al. Transfusion strategies for patients in pediatric intensive care units. N Engl J Med. 2007;356:1609-1619.
161 Lambermont B., et al. Effects of endotoxic shock on right ventricular systolic function and mechanical efficiency. Cardiovasc Res. 2003;59:412-418.
162 Landry D.W., Oliver J.A. The pathogenesis of vasodilatory shock. N Engl J Med. 2001;345(8):588-595.
163 Laurent I., et al. Reversible myocardial dysfunction in survivors of out-of-hospital cardiac arrest. J Am Coll Cardiol. 2002;40(12):2110-2116.
164 Leclere F., et al. Cumulative influence of organ dysfunctions and septic state on mortality of critically ill children. Am J Respir Crit Care Ced. 2005;171:348-353.
165 Levi M. Disseminated intravascular coagulation. Crit Care Med. 2007;35:2191-2195.
166 Levi M., vander Poll T. Inflammation and coagulation. Crit Care Med. 2010;38(Suppl.):S26-S34.
167 Levin E.R. Endothelins. N Engl J Med. 1995;333:356-363.
168 Levin E.R., Gardner D.G., Samson W.K. Natriuretic peptides. N Engl J Med. 1998;339:321-328.
169 Levine H.F., Gausch W.H. Diastolic compliance of the left ventricle. Med Concepts Cardiovasc Dis. 1978;47:95.
170 Levy J.H. Treating shock—old drugs, new ideas. N Engl J Med. 2010;362(9):841-842.
171 Lewis J.K., et al. Outcome of pediatric resuscitation. Ann Emerg Med. 1983;12:297.
172 Liang K.V., et al. Acute decompensation heart failure and the cardiorenal syndrome. Crit Care Med. 2008;36(Suppl.):S75-S88.
173 Liedel J.L., et al. Use of vasopressin in refractory hypotension in children with vasodilatory shock: five cases and a review of the literature. Pediatr Crit Care Med. 2002;3(1):15-18.
174 Lindsay C.A., et al. Pharmacokinetics and pharmodynamics of milrinone lactate in pediatric patients with septic shock. J Pediatr. 1998;132:329-334.
175 Little W.C., Freeman G.L. Pericardial disease. Circulation. 2006;113:1622-1632.
176 Lodha R., et al. Serial circulating vasopressin levels in children with septic shock. Pediatric Crit Care Med. 2006;7:220-224.
177 Lopez-Herce J., et al. Characteristics and outcome of cardiorespiratory arrest in children. Resuscitation. 2004;63(3):311-320.
178 Lopez-Herce J.C., et al. Outcome of out-of-hospital cardiorespiratory arrest in children. Pediatr Emerg Care. 2005;21(12):807-815.
179 Lubitz D.S., et al. A rapid method for estimating weight and resuscitation drug dosages from length in the pediatric age group. Ann Emerg Med. 1988;17:576.
180 Lucking S.E., et al. Dependence of oxygen consumption on oxygen delivery in children with hyperdynamic septic shock and low oxygen extraction. Crit Care Med. 1990;18:1316-1318.
181 Luckner G., et al. Arginine vasopressin in 316 patients with advanced vasodilatory shock. Crit Care Med. 2005;33:2659-2666.
182 Luscher T.F. The endothelium and cardiovascular disease—a complex relation. N Engl J Med. 1994;330:1081-1083.
183 Maar S.P. Searching for the Holy Grail: a review of markers of tissue perfusion in pediatric critical care. Pediatr Emerg Care. 2008;24(12):883-887.
184 Macicek S.M., et al. Acute heart failure syndromes in the pediatric emergency department. Pediatrics. 2009;124:e898-e904.
185 MacLeod J., et al. Predictors of mortality in trauma patients. Am Surg. 2004;70:805-810.
186 Mahar K.O., et al. B-type natriuretic peptide in the emergency diagnosis of critical heart disease in children. Pediatrics. 2008;121:e1484-e1488.
187 Mahle W.T. A dangerous case of colic: anomalous left coronary artery presenting with paroxysms of irritability. Pediatr Emerg Care. 1998;14:24-27.
188 Mahle W.T., et al. Nesiritide in infants and children with congestive heart failure. Pediatr Crit Care Med. 2005;6:543-546.
189 Maier G.W., et al. The physiology of external cardiac massage: high-impulse cardiopulmonary resuscitation. Circulation. 1984;70(1):86-101.
190 Marik P.E. Critical illness-related corticosteroid insufficiency. Chest. 2009;135:181-193.
191 Marik P.E., Varon J. The hemodynamic derangements in sepsis: implications for treatment strategies. Chest. 1998;114:854-860.
192 Markovitz B.P., et al. A retrospective cohort study of prognostic factors associated with outcome in pediatric severe sepsis: what is the role of steroids? Pediatr Crit Care Med. 2005;6:270-274.
193 Marshall J.C., Charbonney E., Gonzako P.D. The immune system in critical illness. Clin Chest Med. 2008;29:605-616.
194 Martin J.B., Wheeler A.P. Approach to the patient with sepsis. Clin Chest Med. 2009;30:1-16.
195 Martinot A., et al. Sepsis in neonates and children: definitions, epidemiology, and outcome. Pediatr Emerg Care. 1997;13:277-281.
196 Marx G., Reinhart K. Venous oximetry. Curr Opin Crit Care. 2006;12(3):263-268.
197 Matok I., et al. Terlipressin after children with extremely low cardiac output after open heart surgery. Ann Pharmacother. 2009;43:423-429.
198 Mazzoni M.L., et al. Lumenal narrowing and endothelial cell swelling in skeletal muscle capillaries during hemorrhagic shock. Circ Shock. 1989;29:27-39.
199 Mazzoni M.L., et al. Capillary narrowing in hemorrhagic shock is rectified by hyperosmotic saline-dextron reinfusion. Circ Shock. 1990;31:407-418.
200 McKiernan C.A., Liberman S.A. Circulatory shock in children: an overview. Pediatr Rev. 2005;26(12):451-459.
201 McLean A.S., et al. Prognostic values of B-type natriuretic peptide in severe sepsis and septic shock. Crit Care Med. 2007;35:1019-1026.
202 Meadows D., et al. Reversal of intractable septic shock with norepinephrine therapy. Crit Care Med. 1988;16:663-666.
203 Meaney P.A., et al. Higher survival rates among younger patients after pediatric intensive care unit cardiac arrests. Pediatrics. 2006;118(6):2424-2433.
204 Mettauer N., Brierley J. A novel use of etomidate for intentional adrenal suppression to control severe hypercortisolemia of childhood. Pediatr Crit Care Med. 2009;10:e37-e40.
205 Meyer D.M., Jessen M.E. Results of extracorporeal membrane oxygenation in children with sepsis: the Extracorporeal Life Support Organization. Ann Thorac Surg. 1997;63:756-761.
206 Meyer R.A. Echocardiography. In: Admas F.H., Emmanouilides G.C., Riemenschneider S., editors. Moss’ heart disease in infants, children and adolescents. Baltimore: Williams and Wilkins, 1989.
207 Meyer R.J., et al. Post-resuscitation right ventricular dysfunction: delineation and treatment with dobutamine. Resuscitation. 2002;55(2):187-191.
208 Michael J.R., et al. Mechanisms by which epinephrine augments cerebral and myocardial perfusion during cardiopulmonary resuscitation in dogs. Circulation. 1984;69:822.
209 Moffett B.S., Price J.F. Evaluation of sodium nitroprusside toxicity in pediatric cardiac surgical patients. Ann Pharmacother. 2008;42:1600-1604.
210 Moncada S., Higgs A. The L-arginine-nitric oxide pathway. N Engl J Med. 1993;329:2002-2012.
211 Morris M.C., Wernovsky G., Nadkarni V.M. Survival outcomes after extracorporeal cardiopulmonary resuscitation instituted during active chest compressions following refractory in-hospital pediatric cardiac arrest. Pediatr Crit Care Med. 2004;5(5):440-446.
212 Munt B., et al. Diastolic filling in human severe sepsis: an echocardiographic study. Crit Care Med. 1998;26:1829-1833.
213 Nadel S., et al. Drotrecogin alpha (activated) in children with severe sepsis: a multicentre phase III randomized controlled trial. Lancet. 2007;369:836-843.
214 Nadkarni V.M., et al. First documented rhythm and clinical outcome from in-hospital cardiac arrest among children and adults. J Am Med Assoc. 2006;295(1):50-57.
215 Namachivayam P., et al. Early experience with Levosimendan in children with ventricular dysfunction. Pediatr Crit Care Med. 2006;7:445-448.
216 Narula J., Kharbandu S., Khan B. Apoptosis and the heart. Chest. 1997;112:1358-1362.
217 Nasser N., Bar-Oz B., Nir A. Natriuretic peptides and heart disease in infants and children. J Pediatr. 2005:248-253.
218 Newburger J.W., et al. Diagnosis, treatment, and long-term management of Kawasaki disease: a statement for health professionals from the committee on rheumatic fever, endocarditis, Kawasaki disease, Council on Cardiovascular Disease in the Young, American Heart Association. Pediatrics. 2004;114(6):1708-1733.
219 Newth C.J., et al. The use of cuffed versus uncuffed endotracheal tubes in pediatric intensive care. J Pediatr. 2004;144(3):333-337.
220 Ngo N.T., et al. Acute management of dengue shock syndrome: a randomized double-blind comparison of 4 intravenous fluid regimens in the first hour. Clin Infect Dis. 2001;32:204-213.
221 Nichols D.G., et al. Factors influencing outcome of cardiopulmonary arrest in children. Pediatr Emerg Care. 1986;2:1.
222 Niemann J.T., et al. Milrinone facilitates resuscitation from cardiac arrest and attenuates postresuscitation myocardial dysfunction. Circulation. 2003;108(24):3031-3035.
223 Niemann J.T., Stratton S.J. Endotracheal versus intravenous epinephrine and atropine in out-of-hospital “primary” and postcountershock asystole. Crit Care Med. 2000;28(6):1815-1819.
224 Niemann J.T., et al. Pressure-synchronized cine-angiography during experimental cardiopulmonary resuscitation. Circulation. 1981;64:985.
225 Nieves J., Kohr L. Nursing considerations in the care of patients with pulmonary hypertension. Pediatr Crit Care Med. 2010;11(Suppl.):S74-S78.
226 Nimah M., Brilli R.J. Coagulation dysfunction in sepsis and multiple organ system failure. Crit Care Clin. 2003;19:441-458.
227 Ninis N., et al. The role of healthcare delivery in the outcome of meningococcal disease in children: case-control study of fatal and non-fatal cases. BMJ. 2005;330:1475.
228 Nollert G., Jonas R.A., Reichart B. Optimizing cerebral oxygenation during cardiac surgery: a review of experimental and clinical investigations with near infrared spectrophotometry. Thorac Cardiovasc Surg. 2000;48:247-253.
229 O’Connor C.M., Gattis W.A., Swedberg K. Current and novel pharmacologic approaches in advanced heart failure. Heart Lung. 1999;28:227-239.
230 Olasveengen T.M., et al. Intravenous drug administration during out-of-hospital cardiac arrest: a randomized trial. J Am Med Assoc. 2009;302(20):2222-2229.
231 Opal S.M. Concept of PIRO as a new conceptual framework to understand sepsis. Pediatr Crit Care Med. 2005;6(3):S55-S60.
232 Opal S.M., Cohen J. Clinical gram-positive sepsis: does it fundamentally differ from gram-negative bacterial sepsis? Crit Care Med. 1999:1608-1616.
233 Orlowski J.P. Optimum position for external cardiac compression in infants and young children. Ann Emerg Med. 1986;15:667.
234 O’Rourke P.P. Outcome of children who are apneic and pulseless in the emergency room. Crit Care Med. 1986;14:466.
235 Pacher R., et al. Prognostic impact of big endothelin-1 plasma concentrations compared with invasive hemodynamic evaluation in severe heart failure. J Am Coll Cardiol. 1996;27:633-641.
236 Paradis N.A., et al. Coronary perfusion pressure and the return of spontaneous circulation in human cardiopulmonary resuscitation. J Am Med Assoc. 1990;263(8):1106-1113.
237 Parasuraman S., Goldhabar S.Z. Venous thromboembolism in children. Circulation. 2006;113(2):e12-e16.
238 Parker M.M. Pathophysiology of cardiovascular dysfunction in septic shock. New Horizons. 1998;6:130-138.
239 Parker M.M. Pediatric definitions for sepsis: it’s about time. Pediatr Crit Care Med. 2005;6(1):83.
240 Parrillo J.E. Septic shock—vasopressin, norepinephrine, and urgency. N Engl J Med. 2008;358(9):954-955.
241 Pathan N., Faust S.N., Levin M. Pathophysiology of meningococcal meningitis and septicaemia. Arch Dis Child. 2003;88:601-607.
242 Patterson S.W., Starling E.H. On the mechanical factors which determine the output of the ventricle. J Physiol. 1914;48:357.
243 Perkin R.M. Current concepts in the recognition and management of pediatric hypovolemic and septic shock. Pediatr Emerg Med Rep. 1999;4(10):95-114.
244 Perkin R.M., Anas N.G. Nonsurgical contractility manipulation of the failing circulation. In: Swellow D.B., Raphaely R.L., editors. Cardiovascular problems in pediatric critical care. New York: Churchhill Livingstone; 1986:229-256.
245 Perkin R.M. Levin DL. Shock in the pediatric patient. Part I. J Pediatr. 1982;101:163-169.
246 Perkin R.M., Levin D.L. Shock in the pediatric patient. Part II. Therapy. J Pediatr. 1982;101:319-332.
247 Perkin R.M., et al. Dobutamine: a hemodynamic evaluation in children with shock. J Pediatr. 1982;100:977-983.
248 Perondi M.B., et al. A comparison of high-dose and standard-dose epinephrine in children with cardiac arrest. N Engl J Med. 2004;350(17):1722-1730.
249 Petre R., Chilcott M. Blunt trauma to the heart and great vessels. N Engl J Med. 1997;336:626-632.
250 Pitt B. Blockade of the renin-angiotensin system. Cardiol Clin. 1994;12:101-113.
251 Pitt B., et al. The effect of spironolactone on morbidity and mortality in patients with severe heart failure. N Engl J Med. 1999;334:709-717.
252 Pizarro C.F., et al. Absolute and relative adrenal insufficiency in children with septic shock. Crit Care Med. 2005;33:855-859.
253 Pollack M.M., Fields A.I., Ruttimann U.E. Sequential cardiopulmonary variables of infants and children in septic shock. Crit Care Med. 1984;12:554-559.
254 Pollack M.M., Fields A.I., Ruttiman U.E. Distributions of cardiopulmonary variables in pediatric survivors and non-survivors of septic shock. Crit Care Med. 1985;13:454-459.
255 Pytte M., et al. Haemodynamic effects of adrenaline (epinephrine) depend on chest compression quality during cardiopulmonary resuscitation in pigs. Resuscitation. 2006;71(3):369-378.
256 Raffini L., et al. Dramatic increase in venous thromboembolism in Children’s Hospitals in the United States from 2001-2007. Pediatrics. 2009;124:1001-1008.
257 Rajagopal S.K., et al. Extracorporeal membrane oxygenation for the support of infants, children, and young adults with acute myocarditis: a review of the Extracorporeal Life Support Organization registry. Crit Care Med. 2010;38:382-387.
258 Ralston D.R., St. John R.C. Immunotherapy for sepsis. Clin Chest Med. 1996;17:307-317.
259 Ralston M., et al, editors. PALS provider manual. Dallas; American Heart Association:2006:86
260 Rhodes A., Bennett E.D. Early goal-directed therapy: an evidence-based review. Crit Care Med. 2004;32(Suppl.):S448-S450.
261 Riordan F.A., et al. Admission cortisol and adrenocorticotrophic hormone levels in children with meningococcal disease: evidence of adrenal insufficiency? Crit Care Med. 1999;27:2257-2261.
262 Rivers E., et al. Early goal-directed therapy in the treatment of severe sepsis and septic shock. N Engl J Med. 2001;345:1368-1377.
263 Rosenweig E.B., et al. Intravenous arginine-vasopressin in children with vasodilatory shock after cardiac surgery. Circulation. 1999;100(19 Suppl.):II182-II486.
264 Rudiger A., Singer M. Mechanisms of sepsis-induced cardiac dysfunction. Crit Care Med. 2007;35(6):1599-1608.
265 Russell J.A. Management of sepsis. N Engl J Med. 2006;355:1699-1713.
266 Russell J.A., et al. Vasopressin versus norepinephrine infusion in patients with septic shock. N Engl J Med. 2008;358(9):877-887.
267 Samson R.A., et al. Outcomes of in-hospital ventricular fibrillation in children. N Engl J Med. 2006;354(22):2328-2339.
268 Schlant R.C., Sonnenblick E.H., Gorlin R. Normal physiology of the cardiovascular system. In: Hurst J.W., editor. The heart, arteries, and veins. New York: McGraw-Hill, 1990.
269 Schrier R.W., Abraham W.T. Hormones and hemodynamics in heart failure. N Engl J Med. 1999;341:577-585.
270 Schubert S., Schmitz T., Weiss M., Nagdyman N., et al. Continuous non-invasive techniques to determine cardiac output in children after cardiac surgery:evaluatin of transesophageal Doppler and electric velocimetry. J Clin Monit comput. 2008;22:299-307.
271 Sebat F., et al. Effect of a rapid response system for patients in shock on time to treatment and mortality during 5 years. Crit Care Med. 2007;35:2568-2575.
272 Seri I. Cardiovascular, renal, and endocrine actions of dopamine in neonates and children. J Pediatr. 1995;126:333-344.
273 Shaddy R.E. Optimizing treatment for chronic congestive heart failure in children. Pediatr Crit Care Med. 2001;2(4 Suppl.):S69-S72.
274 Shapiro N.I., et al. Isolated prehospital hypotension after traumatic injuries: a predictor of mortality? J Emerg Med. 2003;25:175-179.
275 Shippy C.R., Appel P.L., Shoemaker W.C. Reliability of clinical monitoring to assess blood volume in critically ill patients. Crit Care Med. 1984;12:107.
276 Shoemaker W.C. Relation of oxygen transport patterns to the pathophysiology and therapy of shock states. Intensive Care Med. 1987;13:231-234.
277 Shulman D.I., et al. Adrenal insufficiency still a cause of morbidity and death in childhood. Pediatrics. 2007;119(2):e484-e494.
278 Slaughter M.S., et al. Advanced heart failure treated with continuous-flow left ventricular assist device. N Engl J Med. 2009;361:2241-2251.
279 Smith B.T., Rea T.D., Eisenberg M.S. Ventricular fibrillation in pediatric cardiac arrest. Acad Emerg Med. 2006;13:525-529.
280 Solaro R.J., Rarick H.M. Troponin and tropomyosin: proteins that switch on and tune in the activity of cardiac myofilaments. Circ Res. 1998;83:471-480.
281 Spivey W.H.L. Intraosseous infusions. J Pediatr. 1987;111:639.
282 Spodick D.H. Acute cardiac tamponade. N Engl J Med. 2003;349(7):684-690.
283 Sprung C.L., et al. Hydrocortisone therapy for patients with septic shock. N Engl J Med. 2008;358:111-124.
284 Srinivasan V., et al. Association of timing, duration, and intensity of hyperglycemia with intensive care unit mortality in critically ill children. Pediatr Crit Care Med. 2004;5:329-336.
285 Stein P.D., Kayali F., Olson R.E. Incidence of venous thromboembolism in infants and children: data from the National Hospital Discharge Survey. J Pediatr. 2004;145:563-565.
286 Strohmenger H.U., et al. Effects of epinephrine and vasopressin on median fibrillation frequency and defibrillation success in a porcine model of cardiopulmonary resuscitation. Resuscitation. 1996;31(1):65-73.
287 Sunde K., et al. Implementation of a standardised treatment protocol for post resuscitation care after out-of-hospital cardiac arrest. Resuscitation. 2007;73(1):29-39.
288 Sutton R.M., et al. Quantitative analysis of CPR quality during in-hospital resuscitation of older children and adolescents. Pediatrics. 2009;124(2):494-499.
289 Symeonides S., Balk R.A. Nitric oxide in the pathogenesis of sepsis. Infect Dis Clin North Am. 1999;13:449-463.
290 Synder J.D. Use and misuse of oral therapy for diarrhea: comparison of U.S. practices with American Academy of Pediatrics recommendations. Pediatrics. 1991;87:28-33.
291 Tabbutt S. Heart failure in pediatric septic shock: utilizing inotropic support. Crit Care Med. 2001;29(10 Suppl.):S231-S236.
292 Task Force of the American College of Critical Care Medicine and Society of Critical Care Medicine. Practice parameters for hemodynamic support of sepsis in adult patients. Crit Care Med. 1999;27:639-660.
293 Taylor M.D., Laussen P.C. Fundamentals of management of acute postpartum pulmonary hypertension. Pediatr Crit Care Med. 2010;11(Suppl.):S27-S29.
294 Thiagarajan R.R., et al. Extracorporeal membrane oxygenation to aid cardiopulmonary resuscitation in infants and children. Circulation. 2007;116(15):1693-1700.
295 Thomas N.J., Carcillo J.A. Hypovolemic shock in pediatric patients. New Horiz. 1998;6:120-129.
296 Tibballs J., Kinney S. Reduction of hospital mortality and of preventable cardiac arrest and death on introduction of a pediatric medical emergency team. Pediatr Crit Care Med. 2009;10(3):306-312.
297 Tibby S.M., Hatherill M., Murdoch I.A. Capillary refill and core-peripheral temperature gap as indicators of hemodynamic status in pediatric intensive care patients. Arch Dis Child. 1999;80:163-166.
298 Tinsley C., et al. Experience of families during cardiopulmonary resuscitation in a pediatric intensive care unit. Pediatrics. 2008;122(4):e799-e804.
299 Topalian S., Ginsberg F., Parillo J.E. Cardiogenic shock. Crit Care Med. 2008;36(Suppl.):S66-S74.
300 Truitt A.K., et al. Pulmonary embolism: which pediatric trauma patients are at risk? J Pediatr Surg. 2005;40:124-127.
301 Turanlahti M., et al. Pharmacokinetics of Levosimendan in pediatric patients evaluated for cardiac surgery. Pediatr Crit Care Med. 2004;5:457-462.
302 Udomphorn Y., Armstead W.M., Vavilala M.S. Cerebral blood flow and autoregulation after pediatric traumatic brain injury. Pediatr Neurol. 2008;38:225-234.
303 Upadhyay M. Randomized evaluation of fluid resuscitation with crystalloid (saline) and colloid (polymer from degraded Gelatin) in pediatric septic shock. Indian Pediatr. 2005;42(3):223-231.
304 Van den Berghe G., De Zeghar F., Lauwers P. Dopamine suppresses pituitary function in infants and children. Crit Care Med. 1994;22:1747-1753.
305 Van der Poll T., van Deventer S.J.H. Cytokines and anticytokines in the pathogenesis of sepsis. Infect Dis Clin North Am. 1999;13:413-426.
306 van Haren F.M., Rozendaal F.W., vander Hoevan J.G. The effect of vasopressin on gastric perfusion in catecholamine-dependent patients in septic shock. Chest. 2003;124:2256-2260.
307 Vinclair M., et al. Duration of adrenal inhibition following a single dose of etomidate in critically ill patients. Intensive Care Med. 2008;34:714-719.
308 Von Seggern K., Egar M., Fuhrman B.P. Cardiopulmonary resuscitation in a pediatric ICU. Crit Care Med. 1986;14:275.
309 Walsh C.K., Krongrad E. Terminal cardiac electrical activity in pediatric patients. Am J Cardiol. 1983;51:557.
310 Weber K.T. Aldosterone and spironolactone in heart failure. N Engl J Med. 1999;341:753-755.
311 Weiler H. Regulation of inflammation by the protein C system. Crit Care Med. 2010;38(2 Suppl.):S18-S25.
312 Weinberger H.D. Diagnosis and treatment of diastolic heart failure. Hosp Pract. 1999;34:115-126.
313 Wessel D.L. Managing low cardiac output syndrome after congenital heart surgery. Pediatr Crit Care Med. 2001;2(Suppl.):S52-S62.
314 Wessel D.L. Testing new drugs for heart failure in children. Pediatr Crit Care Med. 2006;7:493-494.
315 Westaby S., Franklin O., Burch M. New developments in the treatment of cardiac failure. Arch Dis Child. 1999;81:276-277.
316 Wheeler A.P., Bernard G.R. Treating patients with severe sepsis. N Engl J Med. 1999;340:207-214.
317 Wik L., et al. Quality of cardiopulmonary resuscitation during out-of-hospital cardiac arrest. J Am Med Assoc. 2005;293(3):299-304.
318 Williams R.S. Apoptosis and heart failure. N Engl J Med. 1999;341:759-760.
319 Wills B.A., et al. Comparison of three fluid solutions for resuscitation in Dengue shock syndrome. N Engl J Med. 2008;353(9):877-889.
320 Wong H.R., et al. Increased serum nitrite and nitrate concentrations in children with sepsis syndrome. Crit Care Med. 1995;23:835-842.
321 Woodward G.A., et al. Sepsis, septic shock, acute abdomen? The ability of cardiac disease to mimic other medical illnesses. Pediatr Emerg Care. 1996;12:317-324.
322 Young K.D., Seidel J.S. Pediatric cardiopulmonary resuscitation: a collective review. Ann Emerg Med. 1999;33(2):195-205.
323 Yunge M., Petros A. Angiotensin for septic shock unresponsive to noradrenaline. Arch Dis Child. 2000;82:388-389.
324 Zaritsky A. Controversial issues in pediatric cardio-pulmonary resuscitation. Crit Care Clin. 1989;4:735.
324a Zaritsky A., Chernow B. Use of catecholamines in children. J Pediatr. 1984;105:341-348.
325 Zaritsky A., et al. CPR in children. Ann Emerg Med. 1987;16(10):1107-1111.
326 Zaritsky A., Subcommittee on Pediatric Resuscitation. Approach to drug therapy in pediatric advanced life support, pediatric advanced life support, course material. Dallas: American Heart Association; 1990.
327 Zimmerman J.J. A history of adjunctive glucocorticoid treatment for pediatric sepsis: moving beyond steroid pulp fiction toward evidence-based medicine. Pediatr Crit Care Med. 2007;8:530-539.
328 Zuercher M., et al. Leaning during chest compression impairs cardiac output and left ventricular myocardial blood flow in piglet cardiac arrest. Crit Care Med. 2010;38(4):1141-1146.
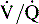 mismatching is compounded by the accompanying fall in CO that results from massive PE, leading to mixed venous desaturation. There is some evidence that inhibition of cyclooxygenase may improve ventilation-perfusion mismatch in patients with PE.20
mismatching is compounded by the accompanying fall in CO that results from massive PE, leading to mixed venous desaturation. There is some evidence that inhibition of cyclooxygenase may improve ventilation-perfusion mismatch in patients with PE.20