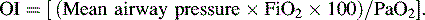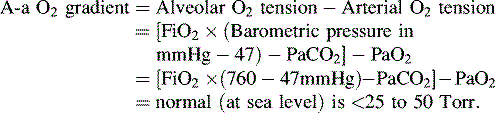7 Mechanical Support of Cardiopulmonary Function
Extracorporeal Membrane Oxygenation, Ventricular Assist Devices, and the Intraaortic Balloon Pump
 Be sure to check out the supplementary content available at http://evolve.elsevier.com/Hazinski.
Be sure to check out the supplementary content available at http://evolve.elsevier.com/Hazinski.
Pearls
• Signs of critically low cardiac output requiring mechanical circulatory support include: mixed venous oxygen saturation less than 40%, increased ventricular end-diastolic filling pressure, cardiac index less than 2 L/min per m2 body surface area (BSA), persistent metabolic acidosis, oliguria, poor peripheral perfusion, increased inspired oxygen requirements, and poor ventricular function by echocardiogram.
• Mechanical circulatory support (MCS) is indicated when low cardiac output persists despite maximal medical treatment, including afterload reduction combined with inotropes/catecholamines, diuretics, a phosphodiesterase-III inhibitor, fluid and transfusion management, and mechanical ventilation.
• In the pediatric population, ventricular assist devices (VADs) are used to support the failing heart as a “bridge to recovery” or “bridge to transplant.”
• MCS should be anticipated and every attempt should be made to initiate support before the development of end-organ dysfunction or circulatory collapse.
• Persistent low filling of a left ventricular assist device (LVAD) pump with rising right atrial pressure is an indication of right-sided heart failure. An echocardiogram is indicated to evaluate ventricular function and check for a pericardial effusion.
• The oxygen index (OI) is often used to identify possible indication for extracorporeal membrane oxygenation (ECMO) for children with respiratory failure.
OI greater than 35 on three or more postductal arterial blood gases 30 to 60 minutes apart is an indication for ECMO support.
• Alveolar-arterial (A-a) gradient of 600 to 624 mm Hg is a clinical indication for ECMO. This is computed as follows (NOTE: 47 mm Hg = partial pressure of water vapor):
• Signs to change venovenous (VV) ECMO to venoarterial (VA) ECMO include: persistent hypoxia in the presence of hypotension and poor cardiac output on maximal ventilator and inotropic support, nonperfusing arrhythmias, or cardiac arrest.
Introduction
The child with life-threatening cardiac or respiratory failure that is refractory to maximal medical support may require MCS. Over the last few years, substantial advances in pediatric MCS have occurred, with expanding indications for use, greater availability of devices suitable for pediatrics, and improved outcomes.16,20
ECMO and centrifugal ventricular assist devices remain the mainstay of MCS for children.23 MCS is not a treatment but rather a therapeutic support to provide adequate tissue oxygen delivery and maintain end-organ perfusion and function while the reversible disease responds to treatment. This support has many potential complications and should be used in appropriate patients with specific criteria for initiation.
Use of MCS in the pediatric population requires knowledge of the pathophysiology and treatment of cardiac and respiratory failure (see Chapters 6 to 9). In addition, the critical care nurse and the ECMO specialist require advanced training in MCS, because they must be familiar with the function of the support device, components of the circuit or console, potential complications, and troubleshooting for the device.
The purpose of this chapter is to provide an overview of MCS options in the pediatric population, with an emphasis on ECMO and VAD physiology, patient selection, indications and contraindications, complications, and postoperative nursing care.
Device Selection
The selection of MCS device is influenced by the indication for the device, the duration of support anticipated, and the devices available. Devices are categorized for short- or long-term support. ECMO, centrifugal VADs, and intraaortic balloon pumps (IABPs) are considered short-term modalities. Pulsatile and rotary-axial devices are typically considered long-term modalities.11
Pediatric MCS offers a potential “bridge to transplant” or a “bridge to recovery.” The term destination therapy is used to describe long-term support for patients who are not heart transplant candidates and those whose heart function is unlikely to recover. Currently, no devices are approved by the U.S. Food and Drug Administration (FDA) for destination therapy in children. Three devices approved by the FDA for destination therapy in adults are the HeartMate VE/XVE LVAS and HeartMate II (Thoratec Corporation, Pleasanton, Calif.), and the AbioCor Implantable replacement heart (Abiomed, Danvers, Mass.).11,55
Extracorporeal membrane oxygenation
ECMO remains the most common form of MCS in the pediatric population. ECMO support evolved directly from cardiopulmonary bypass devices beginning with development of the membrane oxygenator in the 1950s.13,41 In the 1970s, intraoperative cardiopulmonary bypass was successfully used for infants during surgical correction of congenital heart disease.3 In 1972, successful ECMO support of a 24-year-old trauma victim with acute respiratory distress syndrome by Hill et al.37 led to a multicenter National Institutes of Health clinical trial of ECMO versus medical management of adult acute respiratory distress syndrome that failed to show a survival advantage with ECMO support, but did lead to widespread improvement in mechanical ventilation in adults.
In 1976, Bartlett et al.6 reported the first successful use of ECMO for treatment of severe neonatal respiratory distress. Since that time, ECMO has been used successfully in the treatment of neonates with meconium aspiration, congenital diaphragmatic hernia, pneumonia, sepsis, or persistent pulmonary hypertension.57 ECMO is now used as rescue therapy for pediatric patients with severe refractory respiratory or heart failure unresponsive to maximal conventional treatment.
In 1989, the ECMO centers formed a national organization, the Extracorporeal Life Support Organization to coordinate clinical research, develop ECMO guidelines, and maintain a national registry of all ECMO centers and cases. ECMO follow-up data have documented the cost effectiveness of ECMO compared with conventional support and improved patient outcomes with the use of ECMO.49
ECMO Terminology
Bypass therapy outside of the operating suite has been called extracorporeal carbon dioxide removal, extracorporeal heart assist, extracorporeal lung assist, ECMO, and most recently, extracorporeal life support (ECLS). These terms are often used interchangeably, but the term ECMO will be used in this chapter.
ECMO therapy supports cardiopulmonary function through the use of external cardiopulmonary bypass with an oxygenator. ECMO is designed to deliver well-oxygenated blood to an artery or vein, remove carbon dioxide, and normalize acid-base balance and metabolic processes. This system provides temporary pulmonary and possibly cardiac support for infants or children with severe refractory respiratory failure, heart failure, or cardiorespiratory failure, so the failing organs have time to recover or the child is supported until transplantation.
The two types of ECMO support are VA and VV ECMO. VA ECMO provides both cardiac and respiratory support, whereas VV ECMO provides only pulmonary support. The differences between the VA and VV ECMO are summarized in Table 7-1.
Table 7-1 Differences Between VA and VV ECMO
| VA ECMO | VV ECMO |
| Achieves higher PaO2 | Achieves lower PaO2 |
| Requires lower perfusion rates | Requires higher perfusion rates |
| Bypasses pulmonary circulation | Maintains pulmonary blood flow |
| Decreases pulmonary artery pressures | Elevates mixed venous PO2 |
| Provides cardiac support to assist systemic circulation | Does not provide cardiac support to assist systemic circulation |
| Requires arterial cannulation | Requires only venous cannulation |
ECMO, Extracorporeal membrane oxygenation; PaO2, partial pressure of arterial oxygenation; PO2, partial pressure of oxygen; VA, venoarterial; VV, venovenous.
The appropriate ECMO circuit size is determined by the weight of the child; the larger the circuit membrane surface area, the greater the potential gas exchange. However, the larger the surface area of the membrane, the higher the platelet consumption and the larger the circuit priming volume. Because the circuit is not typically primed with blood, a high priming volume relative to the child’s circulating blood volume will result in hemodilution.
Venoarterial ECMO
Circuit
The VA ECMO circuit is composed of polyvinyl chloride tubing attached to venous and arterial cannulas (Fig. 7-1). The venous cannula is inserted into the internal jugular vein and advanced through the superior vena cava and right atrium to the tricuspid valve. The arterial cannula is placed into the right common carotid artery with the tip of the cannula advanced to the innominate artery. Postoperative cannulation immediately after cardiac surgery is often accomplished through the median sternotomy incision with direct cannulation of the right atrial appendage and the aorta. In adolescent or adult patients, the femoral artery and vein are often used for VA ECMO cannulation.
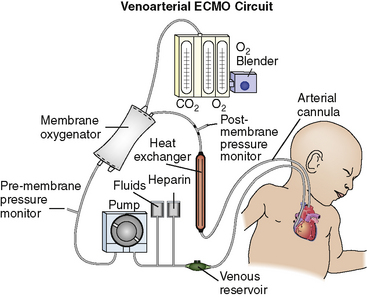
Fig. 7-1 Venoarterial extracorporeal membrane oxygenation circuit.
(From Biddle M, Gulanick M, Berra K: Interdisciplinary team in cardiac rehabilitation. In Moser DK, Riegel B, editors: Cardiac nursing: a companion to Braunwald’s heart disease. Philadelphia, 2008, Saunders.)
The bridge connects the arterial and venous lines and is located near the cannula. The bridge is routinely clamped during ECMO support with brief periods of unclamping to prevent clot formation caused by stagnation of blood in the bridge. If the patient must be separated from ECMO for mechanical complications or for trial periods off ECMO support, the open bridge isolates the patient from the circuit while allowing blood flow to continue through the circuit to prevent stagnation and circuit thrombosis.
Blood from the venous cannula drains passively into a small reservoir called the bladder. The ECMO pump draws blood from the bladder, which works like the right atrium. The function of this bladder is to prevent negative pressure from pulling the vessel wall into the cannula, so it reduces risk of damage to the vena cava. The bladder is connected to a servo regulator mechanism that reduces or stops the pump flow if venous return decreases below a minimum threshold.
The tubing from the bladder joins to the ECMO pump, which is either a roller pump or a centrifugal pump. For the past few decades, the roller pump has been used most commonly for ECMO. However, centrifugal pumps are becoming more popular, especially in cardiac ECMO programs and for extracorporeal cardiopulmonary resuscitation. The roller pump functions on the principles of compression and displacement; blood is displaced as rollers travel the length of the raceway (the segment of tubing contained within the pump head). A strong polymer tubing, called Tygon, is used in the raceway because it is resistant to creasing and erosion.
Blood leaving the pump enters the membrane oxygenator. The membrane consists of a thin silicon rubber sheath with a plastic screen spacer inside to create a semipermeable membrane separating the gas compartment from the compartment containing the patient’s blood. As the patient’s blood flows past one side of the membrane, oxygen diffuses from the gas side across the membrane into the blood, and carbon dioxide from the blood diffuses through the membrane into the gas compartment. The carbon dioxide is then removed, or “swept out” of the system, by the ventilating gas. This ventilating gas is also referred to as the sweep gas, and it is regulated by a flow meter from an oxygen blender. The amount of oxygen in the sweep gas and the available surface area for diffusion determines the amount of oxygen delivered to the patient’s blood as it flows through the oxygenator. The rate of carbon dioxide removal is also dependent on the amount of sweep gas flow and the surface area. The movement of both oxygen and carbon dioxide are dependent on the pressure gradients for the gases across the membrane.
As the blood moves through the ECMO circuit, heat is lost, so ECMO systems use a heat exchanger to keep the blood warm. The heat exchanger is located either between the oxygenator and the patient or integrated into the oxygenator. When the heat exchanger is placed between the oxygenator and the patient, it also serves as a bubble trap.
The blood returns to the patient from the heat exchanger through an arterial cannula inserted into the right common carotid artery. The tip of the cannula is just proximal to the junction of the brachiocephalic artery and the aorta. With this type of cannulation, ECMO becomes essentially a cardiopulmonary bypass system. The greater the ECMO pump flow, the greater the oxygen delivery.
Venoarterial ECMO Flow and Function
In VA ECMO, blood is drained from the right side of the heart and is returned to the arterial side of the circulation, so that it bypasses the heart. Highly oxygenated blood is returned to the arterial circuit, where it mixes with the blood that the native heart is ejecting into the arterial circulation. Ventilator support is minimal, so blood returning from the lungs is relatively desaturated. As ECMO flow increases, the relative percentage of highly oxygenated blood in the arterial circulation is increased. Therefore, the patient’s partial pressure of arterial oxygen (PaO2) increases with increased ECMO flow. With inadequate ECMO flow, the relative percentage of desaturated blood is increased, so the PaO2 is low.
Flow from the ECMO circuit delivered into the aorta is continuous and nonpulsatile. As a result, as ECMO flow increases the patient’s native (intrinsic) cardiac output decreases and the systemic arterial blood pressure waveform is dampened by the nonpulsatile blood flow into the aorta. At 100% ECMO flow, the arterial pressure waveform is flat with only an occasional pulse that may result when slow ventricular filling triggers occasional ventricular systole. Such complete bypass is not typically provided if the patient has some cardiac function.
Typically VA ECMO is run at 80% bypass, so approximately 80% of the patient’s total cardiac output is diverted to the ECMO circuit and then returned to the arterial circulation. At this level of flow, the blood pressure waveform is dampened but preserved, with a pulse pressure of approximately 10 to 15 mmHg. At 80% bypass, the resulting admixture of saturated blood (80% of normal cardiac output) and desaturated blood (from intrinsic cardiac activity) will create sufficient arterial oxygen tension (PaO2), hemoglobin saturation and oxygen delivery. The PaO2 can be expected to be normal or even high.
One method to assess adequacy of oxygen delivery is monitoring of the oxygen saturation of blood in the venous side of the circuit; this oxygen saturation is the effective mixed venous oxygen saturation (SvO2). An SvO2 of approximately 70% to 75% indicates that the ECMO flow is providing sufficient oxygen delivery. Because the normal range of cardiac output varies widely between and within pediatric age ranges, the typical ECMO flow rate varies widely. Approximations of flow are 100 for infants, 80 for children, and 50 mL/kg per minute for adults.
Hemodynamic Changes During Venoarterial ECMO
During VA ECMO, the patient’s PaO2, SvO2, and blood pressure will be affected by changes in the ECMO flow and the patient’s intrinsic (native) cardiac output and differences between the two.
Extremely High PaO2 or Fall in Native Cardiac Output
During ECMO support, an extremely elevated PaO2 typically indicates deterioration rather than improvement in the patient’s cardiac output. Any decrease in native cardiac output is likely to increase the relative percentage of arterial flow that consists of highly oxygenated blood from the ECMO circuit. As a result, the PaO2 rises.
The patient’s cardiac output may fall as the result of hypovolemia and decreased preload, hypertension, and increased afterload or myocardial dysfunction. Hypovolemia can result from hemorrhage, frequent blood sampling that is not replaced, diuresis, and capillary leak (e.g., with septic shock). Hypertension and increased afterload can also worsen myocardial dysfunction and reduce native cardiac output, so these conditions can also cause the PaO2 to rise substantially. If hypertension develops during VA ECMO, aggressive treatment with vasodilators is indicated because hypertension increases the risk of intracranial hemorrhage.
Myocardial dysfunction can cause a fall in native cardiac output. An extreme form of myocardial dysfunction during VA ECMO is known as cardiac stun. When cardiac stun develops, the ventricles barely contract. The electrocardiogram (ECG) and heart rate may be normal, but with little contractility observed by echocardiogram. The blood pressure waveform is severely dampened and pulse pressure approaches zero because the mean arterial pressure is generated almost completely from the ECMO flow.
The etiology of cardiac stun is multifactorial, including increased ventricular wall stress, inadequate coronary perfusion, coronary hypoxia from poorly oxygenated left ventricular output during partial ECMO flow, reperfusion injury, and potential hypocalcemia from the use of older or citrated blood products to prime the ECMO circuit. Calcium is precipitated by the phosphate in the citrate-phosphate-dextrose anticoagulant in bank blood. Cardiac stun is usually reversible with time and adequate VA support. ECMO flow must be adjusted to maintain adequate mean arterial pressure while reversible causes such as acid-base and electrolyte imbalances are corrected and myocardial function recovers.
Native cardiac output can also be reduced by cardiac tamponade, hemothorax, or pneumothorax. These problems decrease venous return to the heart and reduce pulmonary blood flow and native cardiac output so the patient’s PaO2 rises because most flow in the aorta is directly from the ECMO pump. If venous return to the right atrium is severely compromised, drainage to the ECMO circuit will drop; if the condition develops suddenly, the fall in drainage to the circuit will be precipitous. The ECMO circuit contains a safety mechanism (either a mechanical bladder box or pressure modulation) that will stop the pump when venous drainage is inadequate to maintain flow through the circuit. In circuits with pressure control modules, a decrease in the venous pressure indicates a fall in venous return.
If ECMO flow is not increased to compensate for a fall in native cardiac output, oxygen delivery will be compromised and the SvO2 will fall. Systemic venous oxygen saturation can also fall if oxygen consumption is increased by conditions such as pain, anxiety, fever, and seizures.
Falling PaO2 or Increase in Native Cardiac Output
The PaO2 will fall during VA ECMO if the patient’s pulmonary blood flow and cardiac output increase, because mechanical ventilation support is minimal and the lungs do not oxygenate the blood effectively. Thus, pulmonary venous return and blood ejected by the left ventricle will be relatively hypoxemic or desaturated. Anything that increases pulmonary blood flow and native cardiac output will increase the proportion of aortic flow that is provided by this relatively hypoxemic/desaturated blood, so the PaO2 falls. Hyperdynamic heart function and hypervolemia secondary to increased volume administration or decreased urine output can increase cardiac output and pulmonary blood flow.
When the PaO2 falls, ECMO flow is usually increased to maintain systemic oxygen delivery while hypervolemia is treated with diuretic therapy or hemofiltration as appropriate. If the ECMO flow is increased appropriately, the systemic venous oxygen saturation should return to previous levels.
ECMO Support with Single Ventricle Physiology
In patients with single ventricle physiology or shunt-dependent pulmonary blood flow, controversy exists about management of the shunt during MCS. There is debate regarding whether the shunt should be completely closed, partially closed, or left open during MCS. The challenge is to maintain balance between systemic and pulmonary circulation. If the shunt is completely closed during support, there is a risk of pulmonary infarction. If the shunt is open during ECMO, increased ECMO flow may be needed to maintain adequate pulmonary and systemic blood flow and systemic oxygen delivery.53
There are limited data regarding the role of ECMO after stage 1 palliation (and its variants) for hypoplastic left heart syndrome. ECMO is a useful tool for treatment of potentially reversible conditions, such as acute shunt thrombosis and transient depressed ventricular function53; ECMO may be used electively in the immediate postoperative period. However, the use of ECMO after the Norwood procedure remains controversial because the infant’s anatomy is complicated, the therapy is expensive, and survival rate is low.18,51,53,57
Venovenous ECMO
Circuit
VV ECMO is an alternative to VA ECMO that may be preferred for treatment of respiratory failure. In VV ECMO, venous blood is drained, sent through the circuit for oxygenation and carbon dioxide removal (ventilation), and returned to the venous side of the circulation.
The VV ECMO circuit has the same major components and flow pattern as the VA ECMO circuit (described previously), but blood is infused back into the venous circulation by a second venous cannula or a double-lumen single venous cannula. In infants, a double-lumen cannula is placed in the right internal jugular vein and blood is withdrawn through this catheter and returned to the right atrium. In older patients, two or more venous cannulas are placed, often in the right internal jugular vein, the femoral vein, or both.
Venovenous ECMO Flow and Function
Because VV ECMO delivers highly oxygenated/saturated blood to the venous side of the heart, the right heart cannula must be carefully positioned and rotated, and native cardiac function must be adequate. The range of PaO2 typical during VV ECMO is 50 to 70 mmHg, lower than during VA ECMO.
Because both the venous drainage and reinfusion cannula are in the venous system, some blood will be cycled through the ECMO circuit over and over again. This phenomenon is known as recirculation. As ECMO flow increases, so does the percentage of blood that will be recirculated. The greater the volume of blood that is recirculated, the lower the amount of blood flow that is sent forward through the tricuspid valve, into the lungs, left heart, and to the body, and the lower the efficiency of gas transfer between the oxygenator and the patient. The degree of recirculation is monitored by comparing the oxygen saturation of the venous drainage (SvO2) with the arterial saturation (SaO2). If the SvO2 is higher than normal and the SaO2 is low, the volume of recirculation is excessive and either the blood flow rate or cannula placement requires adjustment.
Hemodynamic Changes during Venovenous ECMO
Venous cannula position can impede forward flow, as can right or left atrial volume, and poor contractility. If forward flow is impeded, PaO2, cardiac output and oxygen delivery fall.
If the PaO2 is low or falls during VV ECMO, the cannula position should be checked and the physician should be notified to make any adjustments. In addition, therapy to improve cardiac output may include volume administration with packed red blood cells (to maximize oxygen-carrying capacity) and initiation of inotropic support. Occasionally, conversion from VV ECMO to VA ECMO is needed if hypoxia persists, particularly in the presence of hypotension and poor cardiac output despite maximal ventilator and inotropic support. VA ECMO is also considered if the child develops nonperfusing arrhythmias or cardiac arrest.
Although there is no difference in the rate of survival, or rates of intracranial hemorrhage and infarction, seizures and brain death in pediatric respiratory patients for VV versus VA ECMO, VV ECMO is associated with significantly more cannula problems, higher incidence of CPR on ECMO, and use of inotropic support. In addition, there is also a trend toward more renal failure and use of hemofiltration on VV ECMO.62
Advantages and Disadvantages of VA and VV ECMO
The major advantage of VA ECMO is that it provides both cardiac and pulmonary support. The disadvantages of VA ECMO are: the need to ligate the right common carotid artery and internal jugular vein after decannulation, that the blood flow it provides to body organs is nonpulsatile, that it may result in lower oxygen delivery to the coronary arteries, and that there is a potential for emboli directly into the arterial circulation.
The advantages of VV ECMO are that the carotid artery is spared, pulsatile flow is maintained, and potential emboli from the circuit are trapped in the pulmonary vascular bed. The major disadvantage of VV ECMO is lack of cardiac support. However, the most common cause of myocardial dysfunction in the neonate is respiratory failure. VV ECMO delivers high concentrations of oxygen to both the lungs and the coronary arteries. The high oxygen levels can produce pulmonary vasodilation, decreasing pulmonary hypertension and reversing hypoxic myocardial dysfunction.
Indications and Contraindications of ECMO Support
The indications and contraindications differ for neonatal, pediatric, and cardiac ECMO. Although specific inclusion and exclusion criteria exist, the use of ECMO remains a center specific decision and has to be considered on a case-by-case basis. The inclusion criteria for neonatal and pediatric ECMO are summarized in Box 7-1.
Box 7-1 Inclusion Criteria for Neonatal and Pediatric Respiratory ECMO Support
Pediatric
• Potentially reversible etiology for pulmonary failure
• Oxygenation index >40 and worsening respiratory failure despite maximal ventilator support
• Ventilator support for <14 days
• No other major contraindication to ECMO support such as severe central nervous system abnormality, ongoing hemorrhagic condition or coagulopathy, or multiple organ system failure in >3 organ systems
ECMO, Extracorporeal membrane oxygenation; FiO2, fraction inspired oxygen; PaO2, arterial partial pressure of oxygen.
Neonatal Respiratory Failure
Neonatal ECMO has generally been considered an invasive rescue therapy with an identified set of risks; it is reserved for patients with a high predicted mortality who fail to respond to optimal conventional therapies. In newborns, a predicted mortality rate of 80% or greater was historically the main indication for ECMO. Much work was done in the early years of ECMO therapy to establish criteria for 80% predicted mortality. The most commonly used variable is the OI. An OI greater than 35 on three or more postductal arterial blood gases, 30 to 60 minutes apart, is consistent with an 80% predicted mortality. In addition, a single OI of 60 is a significant predictor of mortality.
Some centers used the A-a oxygen difference (A-a DO2), also known as the A-a O2 gradient. An A-a difference or gradient of more than 600 for a period between 4 and 12 hours is an accepted indication for ECMO support. However, some centers simply report the use of ECMO in patients with severe hypoxemia (PaO2 <50 mm Hg), severe acidosis (pH <7.25), and acute deterioration resulting in a PaO2 of approximately 30 to 40 mm Hg; these are all considered indications for ECMO.
ECMO may be initiated for patients with 50% to 80% predicted mortality if ECMO offers a higher potential for survival than conventional therapy. Examples of such patients include those with meconium aspiration and congenital diaphragmatic hernia with an OI of 25 to 40. The inclusion criteria of a minimum gestational age greater than 34 weeks’ gestation and weight greater than 2 kg is somewhat arbitrary, but is based on the increased risk of intraventricular hemorrhage (IVH) and difficult intravenous access in premature neonates.
Contraindications for neonatal ECMO include the presence of irreversible and uncontrolled bleeding or coagulopathy, IVH greater than grade II, birth weight <2 kg, or gestational age <34 weeks, lethal chromosomal abnormality or congenital malformation incompatible with life, and duration of mechanical ventilation >14 days.43
With more extensive use of therapies such as high-frequency ventilation, surfactant, and nitric oxide, there has been a marked decline in the need for neonatal ECMO.38 If patients do not respond to these therapies, ECMO should be offered in a timely manner.
Pediatric Respiratory Failure
There is less consensus regarding indications for ECMO support for pediatric patients than for neonates with respiratory failure. Severe respiratory failure in children has many etiologies. A child is generally considered to be a candidate for ECMO if death is believed to be nearly certain despite maximal conventional therapy, and the lung disease is believed to be reversible and other organ systems are intact.29 In children, the OI has not been shown to be as predictive of mortality as it is in neonates. Many centers use the criteria of OI >40 and rising, hypercarbia and pH <7.1, a PaO2:FiO2 ratio less than 100 and falling, ventilator support for less than 14 days, and no contraindications such as significant neurologic or hemorrhagic conditions, or failure of more than three organ systems.43 With the use of lung protective strategies and adjunctive therapies such as high-frequency ventilation, surfactant, and nitric oxide, it has become harder to establish criteria for initiation of ECMO support. Contraindications for pediatric ECMO are similar to those in the neonatal period; these include diagnoses incompatible with life, intractable hemorrhage or coagulopathy, or severe central nervous system abnormality.
Cardiac Failure
Cardiac ECMO support has steadily increased over the past decade.24 Although isolated left ventricular failure is relatively rare in children, right ventricular failure, pulmonary hypertension, and hypoxemia are often associated with circulatory collapse in children with congenital heart disease. The most common causes of circulatory failure in infants and children are cardiovascular surgery (postcardiotomy), end-stage cardiomyopathy, and acute myocarditis. The most common indications for ECMO in cardiac patients are severe hypoxia, severe pulmonary artery hypertension, cardiogenic shock, cardiac arrest, and failure to wean from cardiopulmonary bypass after surgical repair.
With ECMO support, postcardiotomy myocardial recovery should occur in approximately 72 hours to 7 days. If there are no signs of myocardial recovery during this time, a cardiac catheterization is needed to assess for residual structural defects. If none exist, the child is listed as a candidate for cardiac transplant. If residual structural defects are identified, surgical reintervention is scheduled.
Contraindications for the use of cardiac ECMO include incurable malignancy, advanced and presumed irreversible multisystem organ failure, extreme prematurity, and severe central nervous system abnormality or hemorrhage.59 In addition, if a patient will not be a transplant candidate, the patient should be carefully evaluated before ECMO. During the past 10 years, many contraindications have been removed from the list or labeled as relative rather than absolute contraindications.
Weaning from ECMO
There are two basic approaches to weaning patients from VA ECMO. Neither method has been shown to be superior to the other.32
In the gradual approach, as lung function improves ECMO is slowly withdrawn and ventilator support is slowly increased. Weaning with this approach may take a period of several days to reach idling flow, or approximately 20 mL/kg per minute. Clinical improvements in pulmonary function and chest radiograph, and loss of edema with a return to pre-ECMO weight faciitate the weaning process.
In the second, more abrupt approach to weaning, ECMO is maintained at full flow of 100 mL/kg per minute with minimal ventilator support until moments before the ECMO circuit is clamped. At that time, the ECMO flow is decreased over a few minutes while ventilator support is simultaneously increased. The circuit is then clamped off and blood gases are obtained to assess pulmonary function. The rationale for this more abrupt termination of ECMO support is that it allows a longer period of low ventilator support to maximize the resting time for lungs to heal.
To discontinue ECMO therapy, the patient must be able to maintain adequate blood pressure, perfusion, acid-base balance, and oxygenation with acceptable ventilator settings without ECMO support. Hypoxia, acidosis, increased serum lactate, decreased cardiac function, and hypotension may be indications to resume ECMO support. During the weaning process, it is not uncommon to increase the ECMO flow or to have more than one unsuccessful attempt to remove ECMO before successful weaning.
Weaning VV ECMO is slightly different than weaning VA ECMO. When weaning VV ECMO, after the ventilator settings are increased, both of the membrane oxygenator gas ports are isolated from ambient air. Eventually, the blood entering and exiting the membrane oxygenator is in equilibrium and reflects typical venous values. This eliminates the need to clamp the venous cannula and allows a longer trial off VV ECMO.32
Complications of ECMO
ECMO has historically been reserved for those with the highest predicted mortality, so its use has a favorable benefit-to-risk ratio. There are a number of identified complications of ECMO therapy. Some of these complications are related to the use of ECMO and some are related to the complications of the initial disease process. With any cause of severe cardiorespiratory failure there is the antecedent injury associated with hypoxemia, acidosis, and shock; if ECMO is provided, there are additional potential complications related to altered flow dynamics of ECMO cannulation and bypass. In addition, there are issues of reperfusion injury and heparinization. It can become difficult to separate the complications actually related to ECMO from those related to the initial illness.
The two most common complications of ECMO are bleeding and clot formation in the circuit. Bleeding and hemorrhage are related to the use of heparin for anticoagulation of the circuit; this heparin then anticoagulates the patient too. As blood flows through the circuit, coagulation factors and platelets are activated and clot formation is triggered in the circuit. A consumptive coagulopathy can develop, causing bleeding complications. The tubing and the oxygenator trap platelets, causing thrombocytopenia. A large percentage of the circulating platelets will have reduced function.
During ECMO, patients requiring any surgical intervention (even just chest tube placement) are at increased risk for bleeding at incision sites. Gastrointestinal hemorrhage and IVH are known ECMO complications.
Because the clotting cascade is activated, clots can develop in the ECMO circuit. These clots can obstruct flow if they are sufficiently large. When clots develop in the oxygenator they can cause oxygenator failure.
Once ECMO is initiated, antithrombin III (AT III) replacement is necessary. AT III acts in concert with heparin to provide anticoagulation. Without adequate levels of AT III, the heparin infusing in the patients during ECMO cannot be truly effective as an anticoagulant. Levels of AT III have been shown to be low in most patients requiring ECMO.
The art of managing patients during ECMO involves assessing each patient’s activated clotting time (ACT) and other measures of coagulation including AT III levels, platelets, prothrombin time (PT)/activated partial thromboplastin time (aPTT) and international normalized ratio (INR) values, anti Xa heparin activity, and thromboelastrography. Heparin and blood product administration and factor replacement should be tailored to patient needs.
Other complications of ECMO include neurologic, cardiovascular, and renal complications in addition to infectious and metabolic problems. Neurologic complications include seizures, IVH, cerebral infarction, and the potential for brain death. It is important to note that even before ECMO is initiated, the patient suffers an insult to the central nervous system secondary to hypoxia and poor perfusion. Cannulation for ECMO alters perfusion and can also contribute to reperfusion injury to the brain. It is therefore difficult to identify whether a neurologic complication is related to the use of ECMO, the antecedent injury suffered before ECMO, or a combination of factors.
Cardiovascular complications of ECMO therapy include arrhythmias, hypotension, hypertension, tamponade, and cardiac stun. In addition, newborns are at risk for maintenance or persistence of a patent ductus arteriosus.
Pulmonary complications include pneumothorax or other air leaks as well as pulmonary hemorrhage and hemothoraces. Pneumothoraces are often minimized by the use of rest ventilator settings. However, when increased settings are used to try to wean ECMO support, a spontaneous pneumothorax may develop.
Infectious complications include culture-proven infections and signs of inflammation, such as neutropenia and elevated C-reactive protein that suggest infection. Attachment to an extracorporeal circuit for an extended period provides a portal for potential infection, but these patients are also intubated and have many catheters and tubes that contribute to infection risk. Meticulous attention to aseptic care of the ECMO circuit can diminish the risks of an ECMO-related infection.
Finally, there are a number of metabolic complications of ECMO therapy, including glucose, electrolyte, and acid-base abnormalities. Banked blood may contain high quantities of glucose and citrate, so glucose and calcium imbalances can be problematic. Older blood has a high potassium concentration. In addition, patients may have a high bilirubin level or high plasma-free hemoglobin, indicating significant hemolysis secondary to ECMO.
ECMO Troubleshooting
ECMO Circuit Emergencies
There are many types of ECMO circuit emergencies, so ECMO staff and the bedside nurse must be constantly vigilant to prevent a circuit problem and rapidly respond to and correct any circuit emergency. The following circuit components need to be rapidly assessed in an ECMO emergency:
• Is blood ejecting from the circuit?
• Is air being pumped in the circuit?
• Is the pump delivering forward flow?
• Does the post-oxygenator blood appear red?
• Is the sweep gas being delivered and vented from the oxygenator?
The circuit check can direct the ECMO specialist’s immediate response to the emergency. If there is blood ejecting from the circuit and air being pumped, then immediate removal from ECMO is required before the problem can be repaired. The ECMO specialist must identify and fix the pump problem while the patient’s nurse and additional critical care unit (PCCU) staff support the patient off ECMO. Immediately clamp the venous line, open the bridge, and clamp the arterial line to remove the patient from the ECMO circuit. Because the patient is dependent on the ventilator, provide ventilation with 100% oxygen or shift the patient back to pre-ECMO ventilator settings. In addition, increased volume and inotropic support may be required, and full cardiopulmonary resuscitation may be needed.
If there is no squirting blood or air being pumped, the patient can be maintained on ECMO while the problem is corrected. ECMO circuit emergencies and problem troubleshooting are summarized in Table 7-2.
Table 7-2 Troubleshooting the ECMO Circuit
| Problem | Signs and Symptoms | Response |
| Clots in circuit | Dark zones or streaks seen; can cause bleeding due to coagulopathy (decreased platelets or fibrinogen unresponsive to transfusion) | Monitor circuit, monitor for coagulopathy, change circuit |
| Oxygenator failure | Failure to remove CO2 or add adequate levels of oxygen in spite of increasing sweep gases and FiO2; may see blood or serum leaking from gas exhaust port | Remove air and debubble circuit if present; check that all gas lines are intact and not leaking to rule out gas line failure; replace oxygenator if it has failed |
| Air in venous side of circuit | Bubbles seen | Correct source of problem, remove air |
| Air in arterial side of circuit | Bubbles seen | Remove patient from ECMO by clamping arterial and venous lines to patient; place patient in Trendelenburg position; replace/repair the component identified as cause; remove all air, recirculate, and then return to ECMO |
| Power failure | Pump stops with no AC power | Plug into emergency hospital power; use battery or UPS; hand crank |
| Accidental decannulation | With partial removal on venous side, air entrainment can be seen; with complete decannulation, cannula will be out of the body with bleeding from cannulation site and possible pumping of air or blood | Cease ECMO and stop pump; put direct pressure on site; call surgeon for immediate replacement of cannula and to control bleeding; replace volume losses with available blood products and crystalloid |
| Water heater failure | Patient exhibits hypothermia; bradycardia with reflex hypertension may be seen with pallor | Obtain new water heater and connect to circuit |
| Raceway rupture | Blood spurts out of damaged tubing in pump head | Cease ECMO; turn pump off; replace raceway segment with new tubing; clean pump head; place new raceway segment into the pump head and recirculate; return to ECMO |
| Cracks in tubing or connectors or loose stopcocks | On venous side [negative pressure side] air will entrain into the circuit. On positive pressure side of the circuit, blood will leak out | Tighten connections or replace cracked tubing or connectors; cease ECMO; stop pump; cut out and replace the damaged segments |
AC, Alternating current; ECMO, extracorporeal membrane oxygenation; UPS, universal power source.
Ventricular assist devices
The development and clinical application of circulatory support devices have closely paralleled the development of cardiac transplantation. Denton Cooley and colleagues achieved the first successful bridge to cardiac transplantation with an artificial heart in the late 1960s. Routine bridging to transplantation began in 1984 with the short-term use of the total artificial heart by Jack Copeland, the Pierce-Donachy (Thoratec) VAD by Donald Hill and colleagues, and the Novacor electrical VAD by Oyer and colleagues.
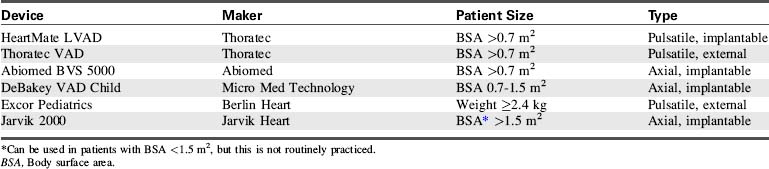
The options for MCS for infants and children with cardiac failure are limited. There are currently no devices approved by the FDA for specific use in infant patients. However, several devices have been used successfully to support infants and children through the following: off-label use of devices approved by the FDA for use in adults, compassionate use of investigational adult VADs, the use of devices that have received an FDA Humanitarian Device Exemption, the use of devices fabricated from FDA-approved components (such as ECMO circuits), and devices that are approved on a case-by-case basis by the FDA for emergency use.7,8,12,20-22,27,28,31,44,56 Table 7-3 lists ventricular assist devices currently being used in the pediatric population under various exemptions or adult approved devices used in older children.2,26,30,35,42,45,46,61 Additional information regarding VADs used in children is included in Evolve Table 7-1 in the Chapter 7 Supplement on the Evolve Website.
Components and Function of VAD Support
A VAD is a heart pump that can be used to support the right ventricle (called a right ventricular assist device [RVAD]), the left ventricle (called an LVAD), or both ventricles (called a BiVAD). Most VADs have three major components: a pump (located inside or outside the body), a control system, and an energy source. The control system and energy source are found outside the body. The energy source can be a battery or compressed air (pneumatic).23
In the pediatric population, most VAD pumps are extracorporeal (outside the body) and connected to inflow and outflow cannulae (Fig. 7-2). The critical care nurse should understand the components of the VAD and VAD function.
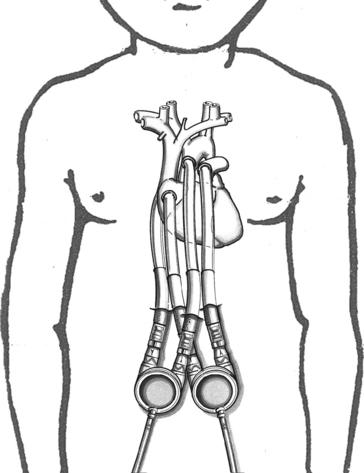
Fig. 7-2 Extracorporeal pneumatic biventricular assist device (Berlin Heart EXCOR).
(Redrawn from an illustration of the Berlin EXCOR by Berlin Heart, Berlin, Germany.)
The placement of the VAD cannula differs if the device is a bridge to recovery, compared with a bridge to transplantation. If the VAD support is serving as a bridge to recovery, the inflow cannula is often connected to the patient’s atrium.19 This cannulation is technically easier and spares the ventricle further injury. However, ventricular cannulation enables higher flow rates, so this form of cannulation is used when VAD support is used as a bridge to transplant, because ventricular injury is not a concern.
When an LVAD is used, the inflow cannula (carrying blood from the patient to the pump) is inserted in either the left atrium or the left ventricle. The outflow cannula (carrying blood from the pump to the patient) is inserted in the ascending aorta. When an RVAD is used, the inflow cannula is inserted in either the right atrium or right ventricle, and the outflow cannula is inserted in the pulmonary artery.
Ventricular Assist Device Flow and Function
Most VAD pumps used in children are either displacement pumps (pulsatile or pneumatic devices) or rotary pumps (continuous flow devices). The pulsatile or pneumatic pumps mimic the natural contraction (pumping action) of the heart. Flow rates depend on preload and the size of the external pump. Average flows for an infant-sized pump (12 or 15 mL) are 0.5 to 1.3 L/min and for a child-sized pump (25 or 30 mL) are 1.3 to 3.3 L/min.4 The external pump size can be changed to accommodate the child’s growth and increased stroke volume.
The most common continuous flow devices are axial or centrifugal pumps. Both types have a central rotor containing permanent magnets. Controlled electric currents that run through coils in the pump housing apply forces to the magnets causing the rotors to turn. Axial flow rates vary depending on the size of the device implanted. The child-sized device has been reported to provide an average flow of 0.3 to 2.5 L/min.4 The pediatric centrifugal Bio-Medicus Bio-pump (Medtronic, Inc. Minneapolis, MN) has both a 50- and 80-mL pump head size to accommodate infants <10 kg and children >10 kg, respectively. The 50-mL pump can provide flow up to 1.5 L/min and the 80-mL pump can provide >2 L/min.27
The VAD console and energy source varies depending on the VAD type. Most pneumatic consoles display both left- and right-sided heart support, the pump rate in beats per minute, systolic and diastolic or fill pressures, and vacuum drive pressures. The consoles also have backup units in case of malfunction. These backup units can be automatically or manually converted, depending on the device. In addition, the consoles can be operated by external electrical power as well as internal batteries. Battery life varies by device with an average of 1 to 2 hours. External backup pumping devices should be attached to the console in case of an emergency.
The console of the centrifugal pump displays the speed and blood flow rate, which are manually adjusted by the operator. Inlet and outlet pressure monitors can be used to guide pump speed and prevent tubing collapse. External electrical power and internal batteries operate the console if transport is needed.
All VADs are preload dependent—the amount of blood returning to the heart is the amount of blood pumped to the body. The VAD is sensitive to impedance to flow, so hypertension and mechanical obstruction must be corrected. Both the LVAD and RVAD allow blood to bypass the failing ventricle. This decompresses that ventricle, decreases myocardial work, and reduces oxygen demand while maintaining adequate systemic perfusion to sustain end-organ function. VAD support has been shown to improve myocardial contractility. It also reverses beta receptor downregulation (documented to occur with heart failure), restoring myocardial response to the inotropic effects of adrenergic stimulation.48 VAD support can also normalize chamber geometry and reduce myocardial fibrosis, hypertrophy, and disruption in cytoskeletal proteins.10,34
Types of Ventricular Assist Device Pumps
Several VAD pumps made for adult patients are available for clinical use. Some are implanted internally and others are external pumps. The VAD pumps can be broadly subdivided into continuous flow (nonpulsatile) and pulsatile pumps. Both types have a central rotor containing permanent magnets; the magnets cause the rotors to turn.
Continuous Flow Pumps
The continuous flow (nonpulsatile) VADs use either centrifugal or axial flow pumps. Each has advantages and disadvantages.
Centrifugal Pump
The centrifugal pump is external, requires cannulation via a thoracotomy or sternotomy, and can be used for single or biventricular support. The most common centrifugal pump is the Bio-Medicus Bio-Pump (Medtronic, Inc. Minneapolis, Minn). The Bio-Pump uses two magnetically coupled, polycarbonate rotator cones that spin to create centrifugal force along a vertical axis.11 The rotors are shaped to accelerate the blood circumferentially and thus cause it to move toward the outer rim of the pump. The constrained vortex pump design creates subatmospheric pressure at the tip of the cone, establishing suction in the venous cannula.40 Blood enters at the apex of the cone and is ejected tangentially at the base of the cone (Fig. 7-3). The cone design retains any small air bubbles.
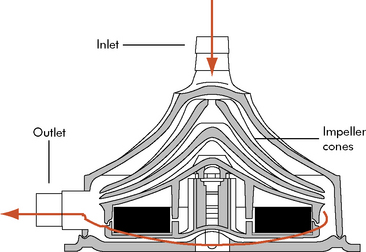
Fig. 7-3 Bio-Medicus centrifugal pump.
(From Karl TR, Horton SB, and Brizard C: Postoperative support with the centrifugal pump ventricular assist device (VAD). Seminars in Thoracic and Cardiovascular Surgery: Pediatric Cardiac Surgery Annual 9:83–91, 2006.)
The pump output is proportional to revolutions per minute and is adjusted according to the venous return. Spins averaging 10,000 to 20,000 rpm will create a blood flow of 5 to 6 L/min in larger pumpheads.36 This type of pump can support neonates and older children with postoperative cardiac failure but competent lung function.36,58
The advantages of the centrifugal pump include: no need for an oxygenator, low priming volume (pediatrics, 50 mL), low requirements for heparin and little hemolysis, adequate decompression of the left ventricle, easy transport, and low cost. However, adequate pulmonary function is required and the chest must remain open, as with ECMO.
The centrifugal VADs include Bio-Medicus Bio-pump, Levitronix CentriMag (Levotronix, Waltham, Mass.), RotaFlow (Jostra, Hirrlingen, Germany), and the Capiox Terumo (Terumo Cardiovascular Systems, Ann Arbor, Mich.). The Levitronix CentriMag is marketed in Europe, but available in the United Stated only as an investigational device.11 The advantage to this device is that it can be attached to cardiopulmonary bypass cannula already in place. However, cannula adaptors may be needed for smaller patients.
Axial Flow Pump
Axial flow pumps reflect recent industry efforts to develop smaller, lighter, quieter, implantable pediatric VADs. Axial flow pumps are designed to assist the left ventricle as a bridge to transplant or destination therapy. The pump has a magnetically levitated impeller that rotates in a cylindrical chamber so that blood accelerates toward the rotor’s axis, usually 7 to 11.5 cm in length, with spins of 7500 to 12,000 rpm (Fig. 7-4). A flexible motor cable supplies power to the implanted pump. The axial flow pumps have been used successfully in 5 to 16 year olds with BSA ≥0.7 m2.
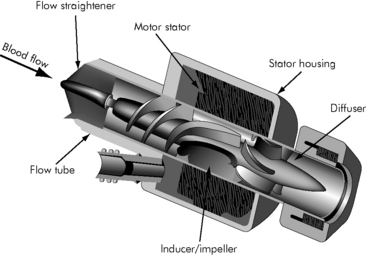
Fig. 7-4 Axial continuous flow pump
(Redrawn from an illustration of the Axial Flow Pump, Micromed DeBakey VAD, by Micromed Technology, Houston, Texas.)
Advantages of the axial pumps are small size, ease of implantation and explantation, low rates of infection, and minimal formation of thrombus.16 The disadvantages are nonpulsatile flow, limited sizes for children, and need for ventricular apical cannulation.16
The MicroMed DeBakey VAD/MicroMed DeBakey VAD Child (MicroMed Cardiovascular, Houston, Texas), HeartMate II LVAS (Thoratec, Pleasanton, Calif.), and Jarvik 2000 (Jarvik Heart, New York, N.Y.) are currently used axial flow VADs for adults and selected children with a BSA >0.7 m2. An infant (3-15 kg) and child (15-25 kg) Jarvik 2000 are being developed through the National Heart, Lung and Blood Institute (NHLBI) Circulatory Assist Program.4
Pulsatile Pumps
Pulsatile VADs contain a reservoir. Blood is ejected by the pump either electronically through the movement of pusher plates or with compressed air movement of the bladder. These devices propel blood in synchrony with the patient’s ventricular ejection, producing pulsatile arterial blood flow. These devices are paracorporeal systems and consist of a pneumatic compressor-operated diaphragm pump with inflow and outflow valves. A transparent polyurethane pump housing allows inspection for potential thrombus development. The external pump position enables fast and safe pump changes if required.54
The Berlin Heart Excor Pediatric VAD (Berlin Heart, Berlin, Germany), Medos HIA-VAD (Medos Medizintechnik, Aachen, Germany) and the Thoratec VAD (Thoratec) provide both single and biventricular support. The Berlin Heart and Medos HIA-VAD are available in the United States under compassionate use appeal to the FDA. The Thoratec device can only be used in patients with BSA >0.8 m2.11
Adult pulsatile devices used in older children include the HeartMate XVE LVAS (Thoratec), Abiomed BVS 5000 (Abiomed, Danvers, Mass.), and Novacor LVAS (WorldHeart Corp. Oakland, CA). These devices are suitable for patients with BSA >1.2 m2 and who require pump flows of more than 2 L/min.
The advantages of pulsatile devices are feasibility for long-term support, potential use without the need for mechanical ventilation, mobility, ability to transition out of the ICU, and need for only low-dose anticoagulation. Disadvantages include thromboembolic complications, infection, and cost.
Indications and Contraindications for Ventricular Assist Device Support
The clinical indications for pediatric VAD support are severe ventricular failure, shocklike state, or the progression of multiorgan failure resulting from acute fulminant myocarditis, cardiomyopathy, postcardiotomy failure, posttransplantation graft failure, and end-stage congenital heart disease.19 Signs of critically low cardiac output or ventricular failure include: mixed venous oxygen saturation (SvO2) <40%, increased ventricular end-diastolic filling pressure, cardiac index <2 L/min per m2 BSA, persistent metabolic acidosis, oliguria, poor peripheral perfusion, increasing FiO2 requirement, signs of beginning renal and hepatic failure, and significantly impaired ventricular function by echocardiogram (Box 7-2). VAD support is indicated when all medical treatment options have been exhausted, including afterload reduction combined with catecholamines, diuretics, a phosphodiesterase-III inhibitor, fluid and transfusion management, and mechanical ventilation. In addition, any residual correctable contributing lesion should be ruled out in patients with congenital heart disease.
Box 7-2 Clinical Indications for VAD Support
• Mixed venous saturation <40%
• Cardiac index <2.0 L/min per m2
• Increased filling pressures (CVP, RA)
• Persistent metabolic acidosis
• Increasing FiO2 requirements
• Signs of beginning renal and hepatic failure (elevated LFT results and BUN/creatinine levels)
BUN, Blood urea nitrogen; CVP, central venous pressure; LFT, liver function test; RA, right atrium; VAD, ventricular assist device.
Before device application, the patient’s neurologic status should be evaluated by clinical and cranial ultrasound examination (in neonates and infants) to rule out intracranial bleeding and cerebral damage. However, such evaluation may not be possible when VAD support is initiated in the operating room after cardiotomy. The decision for device application should then be based on the patient’s preoperative status and intraoperative course.
Patients are evaluated on an individual basis for VAD support. The overall decision for VAD support should be made earlier rather than later to improve chances for end-organ recovery and survival. Contraindications to VAD support include extreme prematurity, irreversible multisystem organ failure, incurable malignancy, and severe central nervous system damage.20
Weaning From VAD Support
Weaning from VAD support is planned for patients with temporary myocardial dysfunction, most commonly associated with acute myocarditis. Before weaning, ventricular recovery and function are assessed.
During use of the centrifugal VAD, the appearance of a pulsatile systemic arterial pressure waveform during full flow is an early sign of improved ventricular function. Transesophageal echocardiogram assessment is helpful to evaluate ventricular contractility and response to volume loading. Once ventricular ejection is verified, the device flow is gradually reduced to a minimum of 150 mL/min. Additional heparin may be required at lower flows, and it may be necessary to flush the cannulae with heparin. Inotropic and pulmonary support should be initiated to maintain adequate perfusion and ventilation during weaning. The device is typically removed in the operating room with sternal closure, if warranted.
Before weaning a pulsatile pneumatic device, improvement in systolic myocardial function is verified by transthoracic echocardiogram or cardiac catheterization. Weaning protocols including the need for additional anticoagulation are device-specific and should be reviewed before initiating the weaning process. The device is removed in the operating suite, with or without the use of temporary cardiopulmonary bypass support. Support can be converted to ECMO support or to a centrifugal VAD pump with later weaning as described previously.1
Termination or withdrawal of VAD support may be indicated if myocardial function does not recover. Withdrawal of support requires careful communication with family members (see Chapters 2 and 3).
Complications of Ventricular Assist Device Support
Potential complications of VAD support include: bleeding requiring reoperation, embolism (clot or air), hemolysis, infection, and mechanical failure.19 Bleeding and embolism are the most common complications following VAD insertion. Mild to moderate bleeding is common and is most often caused by anticoagulation, a coagulopathy, or surgical bleeding. Excessive bleeding requiring massive transfusions often results in pulmonary and multiorgan dysfunction and can be fatal.
Neurologic events such as intracranial hemorrhage and cerebral emboli can result in significant long-term neurologic deficits. Such events are among the major indications for VAD support termination.
Troubleshooting Ventricular Assist Device Support
Troubleshooting VAD support requires knowledge of the type of device (continuous flow or pulsatile), type of support (single or biventricular support), and make of device. Common problems and potential causes are listed in Table 7-4 and are addressed under Nursing Care.
Table 7-4 Potential Problems and Causes During LVAD Support
| Problem | Potential Cause |
| Low LA pressure | |
| High LA pressure | |
| Low RA pressure | |
| High RA pressure | |
| Inability to maintain adequate pump flow | |
| Metabolic acidosis | |
| Hypoxia | |
| Excessive bleeding |
ASD, Atrial septal defect; DIC, disseminated intravascular coagulation; HIT, heparin-induced thrombocytopenia; LA, left atrium; LVAD, left ventricular assist device; PFO, patent foramen ovale; RA, right atrium; RV, right ventricle.
(From Reddy M and Hanley FL. Mechanical support of the myocardium. In Chang AC et al., editors. Pediatric cardiac intensive care. Baltimore, 1998, Williams and Wilkins, pp. 345–349.)
The paracorporeal pneumatic VADs with small pump volumes, designed specifically for infants and children, have been used in Europe since 1992. The Berlin Heart Excor VAD is an extracorporeal device made in a wide range of pump sizes from 10 to 80 mL; it can provide medium- to long-term circulatory support for pediatric patients ranging from 2.5-kg infants to adolescents (see Fig. 7-2).
The Berlin Heart VAD is intended for patients with severe ventricular failure resulting from acute fulminant myocarditis, cardiomyopathy, postcardiotomy failure, posttransplantation graft failure, and end-stage congenital heart disease. In 2007, the FDA approved the use of the Berlin Heart EXCOR Pediatric VAD under a limited conditional investigational device exemption in the United States. The results of the first multisite pediatric clinical trial have been submitted to the FDA. An announcement of approval is pending.
In 2002, the NHLBI recognized the limitations of circulatory support devices in small children. The NHLBI awarded contracts to five research institutes to develop and evaluate circulatory assist devices for children.4 Information about the devices and program locations is available in Evolve Table 7-1 in the Chapter 7 Supplement on the Evolve Website.
Intraaortic balloon pump
IABP counterpulsation is frequently used in adults for managing acute left ventricular dysfunction after myocardial infarction or cardiac surgery.50 The first reported use of an IABP in pediatrics was in 1980.52 Support for the failing pediatric myocardium has primarily focused on the use of ECMO and VAD. Despite the availability of pediatric-sized catheters, the use of IABP in infants and children is not widespread, with use at a limited number of centers.14,39,50 The use of IABP in infants and children remains limited for a variety of reasons, including technical difficulty inserting the catheters in the infant or small child, the limited availability of smaller volume catheters, the greater distensibility of the pediatric vasculature, and the difficulty in balloon timing with rapid heart rates in pediatric patients.
Components and Function
The IABP is commonly used for managing acute postoperative or ischemic left ventricular dysfunction in adult patients. However, its use in infants and small children remains limited because of difficulty in catheter insertion and synchronization of the device with the child’s rapid heart rate.
The IABP catheter is inserted through a vertical groin incision that provides direct visualization of the common femoral artery. The balloon is inserted until the tip is above the renal artery but distal to (approximately 2 cm below) the origin of the left subclavian artery.
The IABP augments cardiac output by inflating during diastole and by deflating immediately before systole. As the balloon inflates, it displaces a volume equal to the balloon volume, producing augmentation of the diastolic pressure and increased coronary artery perfusion. Counterpulsation requires precise balloon inflation at the onset of diastole and aortic valve closure, to augment diastolic and coronary flow, and rapid balloon deflation at the onset of systole and aortic valve opening so that it produces a fall in left ventricular impedance/afterload as the left ventricle begins to eject (Fig. 7-5).
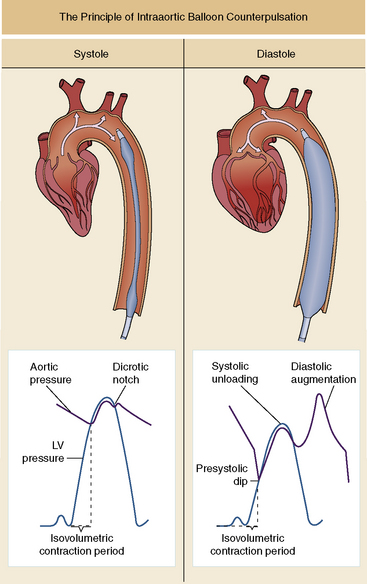
Fig. 7-5 Intraaortic balloon pump. This device includes an elongated balloon that is inserted into the descending aorta. If possible, the tip of the balloon should be distal to the left subclavian artery, to avoid occlusion of the aortic arch vessels. This illustration depicts two counterpulsation cycles. The balloon is rapidly inflated at the beginning of diastole, just after aortic valve closure or at the diacrotic notch, which is timed with the ECG. Balloon inflation augments the aortic diastolic pressure; as a result, arterial diastolic pressure may be higher than arterial systolic pressure (see top right waveform). Rapid deflation of the balloon occurs just before ventricular ejection, producing a systolic unloading effect to augment ventricular ejection.
(From Cercek B, Shah PK: Complications of acute myocardial infarction. In Crawford MH, editor: Cardiology, ed 3, St Louis, 2009, Mosby.)
Counterpulsation increases cardiac output while decreasing myocardial work and oxygen consumption. Other physiologic benefits of IABP include a reduction in left ventricular end-diastolic pressure, left atrial pressure, and pulmonary artery pressure. To optimize beneficial effects, the balloon size must be optimal and balloon inflation and deflation must be synchronized to the patient’s cardiac cycle.
Indications and Contraindications
Indications and contraindications for IABP use have been well established for adults. However, balloon insertion is more invasive in children and requires more consideration and preparation. IABP use is most common in children with left ventricular dysfunction after cardiac surgery or associated with myocarditis, cardiomyopathy, Kawasaki disease, persistent ventricular arrhythmias, or severe sepsis.33
Contraindications for IABP therapy in children include the presence of a patent ductus arteriosus, recent coarctation or aortic arch augmentation, significant aortic valve insufficiency, and when left ventricular failure is unlikely to be reversed.
Technique of Balloon Placement
Pediatric intraaortic balloon catheters are generally inserted through a vertical groin incision that provides direct visualization of the common femoral artery. The balloon can also be inserted directly into the aortic arch if the chest is open.
A wide range of pediatric balloon sizes are available. The standard balloon volumes for children are 2.5 to 20 mL (adult capacity, 40 mL), mounted on 4.5- to 7-Fr catheters. The balloon catheter is placed through a 1- to 2-cm section of polytetrafluoroethylene graft material. The balloon is aspirated to remove air, lubricated, and threaded into position in the descending aorta. The balloon is positioned above the renal artery and distal to (approximately 2 cm below) the origin of the left subclavian artery. After insertion, placement is verified by chest radiograph, fluoroscopy, or transesophageal echocardiogram. On the chest radiograph, the tip is roughly at the left second intercostal space. Once placement is confirmed, the polytetrafluoroethylene graft is sutured to the arteriotomy.
Ideally, the balloon occludes 85% to 90% of the aortic lumen during inflation; it should not completely occlude the aorta. Total occlusion could result in aortic wall injury and damage to red blood cells and platelets. After hemostasis is achieved, a heparin infusion is started to maintain an aPTT of 40 to 60 seconds.
Timing of Inflation and Deflation
The IABP uses helium or carbon dioxide to rapidly inflate and deflate the balloon. The balloon inflates at the onset of cardiac diastole and deflates at the onset of systole. The standard method of balloon inflation timing is by triggering the R wave from the ECG while monitoring the arterial waveform pressure. The inflation and deflation points are adjusted to maximize afterload reduction and diastolic augmentation. It is helpful to record an arterial waveform tracing during a 1:2 heart rate-to-counterpulsation ratio, comparing the assisted and unassisted pulse to assess the effectiveness of the timing of counterpulsation.50 Balloon inflation and deflation can also be timed using M-mode echocardiography.50 The transducer is positioned parasternal to obtain simultaneous images of both the aortic valve and the balloon. During imaging, the balloon inflation and deflation points are adjusted to coincide with aortic valve closure and opening, respectively.
Weaning
The weaning of the IABP usually begins once systemic perfusion is adequate after pharmacologic support has been substantially reduced or removed. A two-dimensional echocardiogram is often used to aid in the decision to wean. Parameters for weaning generally include improvement in the left ventricular ejection phase indices, such as shortening or ejection fraction. Weaning may occur over a 24- to 48-hour period. The pump is systematically reduced from a 1:1 ratio to 1:2 and then 1:3 over the weaning period.
The IABP catheter is removed at the patient’s bedside. Embolectomy catheters may be used once the balloon is removed to evacuate any clot that might remain in the common femoral artery before closure of the artery. Distal pulses should be confirmed before closure of the groin incision and monitored following closure.
Complications
Complications reported during pediatric IABP therapy include limb ischemia, infection, balloon rupture, vessel perforation, thrombocytopenia, aortic dissection, and mesenteric, renal, or cerebral ischemia.33,50
The IABP should never remain dormant for longer than 15 minutes, because blood trapped in the folds of the deflated balloon can trigger clot formation. If the IABP is dormant for longer than 15 minutes, manual inflation and deflation is required every 5 minutes until the problem is resolved.
Blood in the driveline or tubing can indicate balloon rupture. Immediately notify a physician if balloon rupture is suspected and prepare for IABP removal. Systemic arterial emboli from the balloon catheter or graft can develop.
Nursing Considerations
The pediatric nurse should be trained and familiar with the IABP console and how to make adjustments to improve timing. Initially, two nurses are usually needed to provide nursing care; one for the care of the patient and one devoted to running the balloon pump. When the patient’s condition stabilizes, the critical care nurse will be responsible for both the patient and the IABP.
During IABP therapy, hemodynamic monitoring should be consistent with that required for any patient with LV dysfunction. The nurse may note absent or diminished pulse in the involved extremity following balloon insertion. The nurse should frequently evaluate lower extremity perfusion (including color, temperature, and capillary refill) and notify a provider immediately if evidence of limb ischemia and coolness appears.
Nursing care of the pediatric patient requiring MCS
The purpose of this section is to present essential concepts for the nursing care of the postoperative pediatric MCS support patient. General principles of care are similar for all pediatric patients requiring MCS. Most MCS devices should be initiated before the presence of end-organ dysfunction or circulatory collapse. Recently, ECMO or ECLS has been used for rapid resuscitation for children with cardiac arrest. The goal of ECMO or VAD support is the recovery of cardiopulmonary function or provision of MCS as a bridge to transplant.
The multidisciplinary team should prepare the child (if possible) and family regarding the child’s clinical condition, expected prognosis, and anticipated support duration (bridge to transplant or bridge to recovery). Although rehabilitation will likely be required for all children after MCS, VAD support requires rehabilitation during therapy. After the patient is stabilized with pneumatic VAD support, progression of early extubation, invasive line removal, nutritional support, and cardiac rehabilitation are facilitated to minimize the risk of infection and optimize the patient condition before transplant or device removal.
The nursing care plan in Box 7-3 is designed for pediatric patients on ECMO and VAD support. Interventions are separated if they differ for ECMO or VAD support.
Box 7-3 Nursing Care of the Pediatric Patient During Mechanical Circulatory Support
Potential for Hemorrhage and other Complications Related to MCS Malfunction
• ECMO device failure/disruption can result from raceway rupture, inadvertent decannulation, power failure
• VAD device failure/disruption can result from console malfunction or power failure
Nursing Interventions
• Frequently inspect circuits for cracks or stress on the raceway and make planned repairs or replacements.
• Frequently inspect and test alarms and pressure monitors.
• Assess electrical outlets and connections on a routine basis (per hospital protocol).
• Keep emergency resuscitation medications at bedside.
• Keep appropriate emergency equipment (e.g., metal tubing clamps, hand crank, bulb syringes) available
• Have backup ECMO circuit/pump and VAD console available and charged.
Potential Inadquate Cardiac Output Related to
• Tamponade or pericardial effusion
• Myocardial dysfunction (cardiac stun)
Nursing Interventions
• Assess child’s systemic perfusion, including temperature, color of mucous membranes, nail beds, skin, quality of peripheral pulses, and capillary refill time.
• Notify provider of signs of poor systemic perfusion.
• Monitor for evidence of bleeding: excessive chest tube output (>3 mL/kg per hour), oozing from cannulation site and sternal patch (if closure delayed).
• Monitor for signs of coagulopathies.
 Draw blood samples for coagulation studies (ACT, PLT, PT, aPTT) immediately after ECMO/VAD insertion and then as ordered.
Draw blood samples for coagulation studies (ACT, PLT, PT, aPTT) immediately after ECMO/VAD insertion and then as ordered.• Monitor for signs of cardiac tamponade in patients receiving MCS:
 Rising central venous pressure (CVP) right atrial (RA) and pulmonary artery end-diastolic pressures, tachycardia, hypotension, decreased SvO2 or NIRS, decreased MCS device filling, decreased VAD output, and widened mediastinum on chest radiograph.
Rising central venous pressure (CVP) right atrial (RA) and pulmonary artery end-diastolic pressures, tachycardia, hypotension, decreased SvO2 or NIRS, decreased MCS device filling, decreased VAD output, and widened mediastinum on chest radiograph. If tamponade is suspected, notify physician immediately and prepare for emergency chest exploration and evacuation of clot, or prepare for the patient to return to the operating suite per institutional policy.
If tamponade is suspected, notify physician immediately and prepare for emergency chest exploration and evacuation of clot, or prepare for the patient to return to the operating suite per institutional policy.• Measure and record hourly urine output; report output <0.5 mL/kg per hour.
• Monitor heart rate and rhythm; ensure that heart rate is appropriate for the patient’s age and condition. Report clinically significant arrhythmias to the provider.
• Monitor patient’s arterial blood pressure; report hypotension or hypertension.
• Monitor cardiac index; report decrease and any cardiac index <2.5 L/min per m2 BSA.
• Monitor arterial blood gases and serum lactate; report acidosis or abnormalities.
• During ECMO, obtain simultaneous arterial and SvO2 measurements. If difference is increasing, the child’s cardiac output is probably falling; if difference is decreasing, the child’s cardiac output probably is rising. Adjust ECMO flow per protocol or order.
• Monitor patient for normal MAP (per age). If SVR or MAP is elevated:
 Administer continuous vasodilators, such as nitroglycerin or sodium nitroprusside, to keep MAP within normal limits.
Administer continuous vasodilators, such as nitroglycerin or sodium nitroprusside, to keep MAP within normal limits. Ensure adequate intravascular volume before initiation of vasodilators to minimize risk of hypotension.
Ensure adequate intravascular volume before initiation of vasodilators to minimize risk of hypotension.Potential for Inadequate Intravascular Volume Related to
Expected Outcome
• Patient demonstrates adequate intravascular volume with adequate ECMO/VAD filling
• Hematocrit within normal limits per age and orders or protocol
• Adequate CVP or RA pressure and pulmonary capillary wedge pressure (PCWP) or left atrial (LA) pressure
• Urine output 0.5-2.0 mL/kg per hour
• Chest tube output <3 mL/kg per hour
• Adequate systemic perfusion, moist mucous membranes, and good skin turgor
Nursing Intervention
• Record and calculate total fluid intake and output, including blood drawn for laboratory analysis.
• Measure CVP/RA and PCWP/LA pressures and support to optimize perfusion per orders.
• Draw blood sample for Hct immediately after ECMO/VAD insertion and then per orders. If Hct is low, administer packed red blood cells (PRBCs) per order.
• Draw blood samples for coagulation studies (ACT, PLT, PT, aPTT) immediately after ECMO/VAD insertion and then per orders. Adjust heparin infusion per standing orders for VAD and ECMO device. If platelet count is low, replace with platelets per orders. Keep ACT between 140 and 180 sec for VAD devices and between 200 and 240 sec for ECMO.
• Milk chest tubes gently and tap firmly to keep free of clots. Notify the provider if chest tube output is >3 mL/kg per hour for 3 h or 5 mL/kg per hour for 1 h.
• For patients with delayed sternal closure, notify the provider if the sternal patch fills or bulges.
• Discuss with a provider the possibility of surgical bleeding in the presence of excessive chest tube output and the absence of coagulopathy.
• Assess patient’s hydration and report signs of inadequate hydration. Signs of adequate hydration are
• Avoid any nonessential invasive procedures (e.g., peripheral IV removal) while the patient is receiving heparin therapy, because the procedure may cause uncontrolled bleeding at site.
Potential Impairment in Gas Exchange Related to
Expected Outcomes
• Patient will demonstrate adequate gas exchange as evidenced by satisfactory clinical appearance and arterial blood gases, appropriate pulse oximetry and exhaled carbon dioxide, and normal lung compliance with full lung expansion and no evidence of atelectasis.
• For the patient requiring VAD/ECMO support, SvO2 of 70-75% and PaO2 of 50-70 mmHg will be maintained.
Nursing Interventions
• Monitor for signs of hypoxemia, including tachycardia, increased spontaneous respiratory rate, compromise in systemic perfusion, deterioration in level of consciousness, decreased hemoglobin saturation, rise in exhaled CO2, deterioration in PaO2 or pH.
• Long-term VAD use should be weaned to extubation once the patient’s condition is stable, with regular pulmonary toilet incorporated into daily routine.
• Draw arterial, venous, and pump (for ECMO) blood gases immediately after ECMO/VAD insertion and then per orders. Venous and arterial pH, PaO2, and PaCO2 should be within normal limits.
• Adjust sweep gas and ECMO flow per protocol or MD order. Increasing the sweep gas decreases PaCO2 levels, and increasing the ECMO flow rate elevates the PaO2 levels.
• Auscultate lung sounds and monitor chest expansion.
• Verify appropriate endotracheal tube depth of insertion or adjust if needed.
• Monitor for and notify provider of any change in PIP, PEEP, and exhaled tidal volume.
 Loss of PEEP can occur with spontaneous inspiration, a leak around the endotracheal tube, or a leak in the system (see Chapters 9 and 21).
Loss of PEEP can occur with spontaneous inspiration, a leak around the endotracheal tube, or a leak in the system (see Chapters 9 and 21).• Suction via endotracheal tube per unit protocol and as needed. Monitor color, consistency and quantity of secretions.
• Change the patient’s body position every 1-2 h as tolerated.
Potential Alteration in Cerebral Perfusion and Function Related to
Nursing Interventions
• Assess neurologic function before and immediately after initiation of MCS.
• Correct hypotension, hypoxemia, and acidosis as soon as possible.
• Monitor NIRS cerebral tissue oxygenation (normal range >50%). Notify provider of decrease.
• Verify performance and report results of cranial ultrasound examinations daily for first 3 days of ECMO (or per protocol).
• Maintain ACT in desired range for VAD (140-180 sec) or ECMO (200-240 sec)
• Monitor ECMO/VAD circuit and tubing for clot or air formation; notify provider if present.
• Check coagulation studies and administer anticoagulation therapy as indicated for MCS.
Potential Alteration in Renal Function and Urine Output Related to
Nursing Intervention
• Measure urine output every hour and notify provider if hourly total is < 0.5 mL/kg per hour
 Initiate fluid restriction and diuretics per order if urine output remains inadequate despite normal to high CVP.
Initiate fluid restriction and diuretics per order if urine output remains inadequate despite normal to high CVP.• If urine is red or rust color, provide intravenous fluids to maintain urine flow and prevent RBC accumulation in glomeruli causing acute tubular necrosis or renal failure.
• If hyperkalemia develops during VAD, administer calcium, glucose, and insulin, or sodium polystyrene sulfonate (Kayexalate) enema per orders (see Chapter 12 and Table 12-4).
• Prepare for peritoneal dialysis, hemodialysis, or modified hemofiltration (depending on circulatory support) per orders as indicated (see Chapter 13).
• If renal impairment is suspected, closely monitor serum creatinine, BUN, and K+ concentrations.
• If renal impairment is present, evaluate doses of drugs that require renal excretion.
Potential Infection Related to
Nursing Interventions
• Ensure that every provider washes hands before and after patient contact.
• Ensure appropriate sterile and aseptic techniques for procedures or dressing changes.
• Maintain occlusive dressings over all invasive monitoring catheters and an open chest.
• Change dressings and intravenous tubing per unit policy.
• Monitor patient for signs and symptoms of infection.
• Administer antibiotics as ordered.
• If infection is present, monitor for evidence of sepsis (tachycardia, blood pressure instability, thrombocytopenia, temperature instability).
Potential Child and Family Knowledge Deficit Related to
• Child’s cardiovascular disease
• Initiation and requirement of mechanical circulatory support device
Nursing Interventions
• Ascertain what the patient and family have been told about the disease, therapy, and prognosis.
• Ask parents how they think child can best be prepared for MCS (as appropriate).
• Assess and document the child’s and the family’s learning needs and barriers to learning.
• Prepare the child and family for clinical course of mechanical circulatory support including:
• Encourage the child and the family to ask questions and participate in care when appropriate.
• Record specific teaching information (including terminology used) so that the healthcare team can use consistent terms and reinforce identical information.
Potential Child and Family Anxiety Related to
Nursing Interventions
• Maintain open communication with patient and family.
• Answer all questions honestly at the appropriate level for patient and family understanding.
• Prepare patient (as age appropriate) and family for procedures. Include a child life specialist as appropriate for the child’s age and clinical condition.
• Encourage family participation in patient care as appropriate.
• Encourage the child’s expression of feelings and emphasize acceptability of expression.
• Assess the family’s resources and refer to appropriate hospital support services (See Chapter 2)
Potential Additional Patient Care Priorities
Pain or discomfort related to
ACT, Activated clotting time; aPTT, activated partial thromboplastin time; BSA, body surface area; BUN, blood urea nitrogen; CVP, central venous pressure; ECMO, extracorporeal membrane oxygenation; Hct, hematocrit; IV, intravenous; LA, left atrium; MAP, mean arterial pressure; MCS, mechanical circulatory support; NIRS, near-infrared spectroscopy; PCWP, pulmonary capillary wedge pressure; PEEP, positive end-expiratory pressure; PIP, peak inspiratory pressure; PLT, platelet count; PRBC, packed red blood cells; PT, prothrombin time; PVR, pulmonary vascular resistance; RA, right atrium; SvO2, venous oxygen saturation; VAD, ventricular assist device.
Preparation of the Child and Family
If the child deteriorates rapidly, preparation of the child for MCS support may be limited. This time is stressful for the family when making life saving decisions. Parents need hope within reason. Honest and open communication between the nurse and family is central to developing a trusting relationship. Before the family visits the child for the first time after mechanical support placement, the nurse should meet with family members away from the bedside and explain to them what they are about to see. Furthermore, the nurse should encourage the parents to visit, talk to, and touch their child when possible. The social worker and chaplain are often important members of the multidisciplinary team and should be available to the family for support before the initiation of mechanical support.
Pediatric Critical Care Unit (PCCU) Setup
The PCCU bedside setup for MCS is the same for a postoperative cardiac surgery patient (see Chapter 8). The bed selection, bed location, and equipment needed (IV pumps, ventilator) should be prearranged, if possible, to optimize space for easy movement during care of the patient. Although equipment for MCS can vary depending on the selected device, all extension cords and drive lines must be well secured to minimize accidental falls or disconnection.
Emergency equipment that should be present at the bedside at all times includes heavy metal tubing clamps, emergency equipment to manually hand crank centrifugal devices, and bulb suction equipment to manually pump the pneumatic device. A backup ECMO machine or VAD console should be plugged, tested, and ready for use if any malfunction requires a circuit or console change. Blood products should be readily available during MCS support.
Admission or Initiation of MCS and Staffing
ECMO support can be initiated in the operating suite, or cannulation can be performed at the bedside. Although there are many variations in staffing, in most ECMO centers two providers are assigned to the patient: a critical care nurse provides all patient care and an ECMO specialist monitors and cares for the ECMO circuit. ECMO specialists are specially trained nurses, respiratory care practitioners, or perfusionists who have attended an ECMO training class. The ELSO published guidelines for the training of these specialists, including detailed knowledge of the pathophysiology of common diseases, ECMO physiology, and immediate treatment for patient events and ECMO emergencies. (For more information, see http://www.elso.med.umich.edu/)
VADs are placed in the operating suite. Two nurses are initially required to accept the child to the ICU after VAD placement: one nurse cares for the patient and a second nurse monitors the VAD. Once the patient is stabilized, one nurse can care for both the patient and the VAD pneumatic device, provided the nurse is trained in the device type and its operation and components, troubleshooting, and response to emergencies. Some centers that use centrifugal pumps may require a perfusionist to manage the VAD.
Postoperative Care
The postoperative nursing care of patients receiving MCS involves thorough and repeated assessment and support of every organ system, device management and troubleshooting, monitoring of laboratory values, and anticipation and treatment of any complications. Upon initial cannulation or placement of the MCS device, the nurse should perform a primary assessment to provide a baseline to identify any clinical changes.
Neurologic Assessment
The pediatric patient receiving MCS is at risk for cerebral embolism and bleeding, so frequent neurologic assessment is required. Routine cranial ultrasound examinations are performed to assess for the presence of IVH in neonates and infants with an open fontanelle. Clinical neurologic assessment is particularly challenging in patients with centrifugal VAD or ECMO support with open chest cannulation; such cannulation typically requires that patients be sedated with possible neuromuscular blockade. In patients receiving neuromuscular blockade, the only clinical signs of seizures may be abrupt changes in heart rate, blood pressure, skin perfusion, and pupil size. If seizures are suspected, a cranial ultrasound examination or head computed tomography scan will be obtained to look for evidence of bleeding. An electroencephalogram will be required to assess for the presence of seizures and cerebral cortical activity.
When ECMO cannulation uses the internal jugular vein and right common carotid artery, the head is maintained in the midline position to avoid obstruction of the vein or artery. Such compression could compromise cerebral venous drainage or perfusion.
Near-Infrared Spectroscopy (NIRS)
NIRS provides continuous, real-time data regarding tissue oxygenation that can be used in conjunction with clinical and laboratory assessment to evaluate systemic oxygenation and perfusion. The NIRS system uses adhesive sensors placed on the forehead and over the abdomen. Each adhesive sensor contains a light source and two optodes (fiberoptic bundles), often placed at angles from one another to evaluate light absorption and reflection of different tissue depths. As light passes through the skin—and in the case of the brain, through the skull—and into tissue, oxygenated and deoxygenated hemoglobin and oxygenated cell mitochondria affect light absorption and reflection. The NIRS devices calculate the average oxygen saturation of underlying arteries, capillaries, and veins, to reflect a tissue or regional tissue oxygen saturation (rSO2) that is similar to a mixed venous oxygen saturation. This tissue oxygen saturation is digitally and continuously displayed.
Evaluation of both cerebral and somatic (typically evaluated over the abdomen) oxygenation can help identify changes in regional oxygenation and blood flow. A change in NIRS saturation can indicate declining or inadequate cerebral or somatic oxygenation and allow corrective action to prevent hypoxic injury.
The cerebral rSO2 value will vary depending on the level of sedation or agitation and the location of the sensor. It will also be affected by factors influencing cerebral blood flow such as increased intracranial pressure (can decrease blood flow), changes in PaCO2 (hypocarbia produces cerebral vasoconstriction and decreased cerebral blood flow, and hypercarbia produces cerebral vasodilation and increased blood flow) and severe hypoxemia (produces cerebral artery dilation and attempts to increase cerebral blood flow). The target cerebral rSO2 is greater than 50%.25
The somatic tissue oxygenation should be higher than cerebral tissue oxygenation, because the kidneys receive 25% of cardiac output at any time. The target difference between somatic and cerebral rSO2 should be greater than 10%, with somatic readings approximately 10% to 15% points higher than cerebral readings.25
NIRS monitoring can be a valuable tool during ECMO support to identify a compromise in tissue oxygenation that may result from reversible problems with cannula placement, and it can assist in the evaluation of systemic oxygen delivery and readiness for decannulation. If a change in cannula position or a circuit malfunction occurs, the flow deficit will produce a fall in rSO2. Furthermore, if the rSO2 falls significantly during weaning and clamping of the ECMO circuit, then additional time on ECMO will be needed. If the rSO2 remains stable in a patient with other signs of adequate systemic perfusion, the patient is likely to be sufficiently stable for decannulation.25
Sedation and Analgesia
Sedation and analgesia administration after ECMO or VAD placement will vary depending whether the patient is cannulated through an open or closed chest, and must be titrated to patient response. Continuous sedation and analgesia is maintained with fentanyl (0.5-2 mcg/kg per hour) or morphine sulfate (0.02 mg/kg per hour) and midazolam (0.05-0.1 mg/kg per hour) infusions. Intermittent doses can be provided for agitation, painful procedures, or unexplained hypertension and tachycardia.
Neuromuscular blockade may be needed for the hemodynamically unstable patient receiving MCS. Pancuronium (0.1 mg/kg per dose) or a Cis-atracurium infusion (0.2 mg/kg per hour) can be used. Minimal use of neuromuscular blockade is advantageous to allow spontaneous movement and assess for seizure activity. For further information, see Chapter 5.
Respiratory Assessment
All patients receiving MCS are initially intubated and their lungs are mechanically ventilated. Lung sounds should be auscultated and chest expansion assessed during hand ventilation (with bag). The depth of endotracheal tube insertion should be adjusted if breath sounds are decreased on the left side or if placement is too low or too high on chest radiograph. Patients receiving only left ventricular support via centrifugal devices require full ventilator support. Patients requiring more long-term pneumatic assistance with a VAD should be weaned to extubation once stable.
During VA ECMO, mechanical ventilation settings are generally reduced to an inspired oxygen of 21% to 40%, peak inspiratory pressure of 20 to 25 cm H2O, positive end-expiratory pressure of 4 to 10 cm H2O, and respiratory rate of 5 to 10 breaths per minute. The goal of the resting ventilator settings is to maintain functional residual capacity and prevent barotrauma. In patients with minimal lung disease and native cardiac output, gas exchange will then occur within the lungs and through the membrane oxygenator.
Daily chest radiographs and frequent arterial and venous blood gas samples are used to monitor respiratory function. Bilateral opacification of lung fields is generally apparent on chest radiograph within the first 24 hours of ECMO, resulting from a reduction of positive airway pressure and complement activation leading to capillary leak and pulmonary edema. Patients with persistent pulmonary air leaks while receiving ECMO may require very low positive airway pressure to allow the lungs to heal. Pulmonary care should include chest vibration and suctioning of the endotracheal tube as needed to clear secretions. Limitation of suctioning may be needed if bleeding is exacerbated by the procedure.
Potential respiratory complications include pneumothorax or hemothorax, pleural effusions, and pulmonary hemorrhage. Needle aspiration or chest tube insertion are options for emergent decompression of a pneumothorax or hemothorax or drainage of a pleural effusion. If pulmonary hemorrhage develops, management consists of minimizing the target ACT, increasing the platelet count, and minimizing suctioning. If bleeding becomes more severe, epinephrine (0.1 mL/kg of 10,000 dilution) may be instilled into the trachea and positive end expiratory pressure instituted. See Chapter 9 for information regarding mechanical ventilation.
Cardiac Assessment and Support
The patient’s cardiac output, hemodynamics, and device flow must be monitored continuously to ensure adequate systemic perfusion. The critical care nurse must understand the device operation and components to interpret hemodynamic changes and ensure effective MCS. Inadequate systemic perfusion can result from inadequate intravascular volume, inappropriate vascular resistance, (native) myocardial dysfunction, arrhythmias, or tamponade.
Inadequate Intravascular Volume
Inadequate intravascular volume can result from hemorrhage, excessive diuresis, or inadequate fluid administration. In the early postoperative period, bleeding following MCS placement can be life-threatening and increase demands on blood resources. Risk factors for bleeding include: low preoperative platelet count, long cardiopulmonary bypass time, hypothermia, preoperative anticoagulation, and hepatic dysfunction. Intraoperative prevention and management require meticulous hemostasis and reversing heparin. Anticoagulation, if indicated for the device, should not be initiated until bleeding from the chest tubes has decreased. Maintaining high filling pressures and treating coagulopathies with blood component therapies (packed red blood cells, fresh frozen plasma, or platelets) is essential. Volume status can be assessed by monitoring central venous pressure, mean arterial or pulse pressure, and indirect indicators such as urine output.
The nurse needs to monitor for excessive fluid loss leading to hypovolemia and inadequate VAD or ECMO filling. If the patient’s hematocrit is stable and within normal range for age, colloid or crystalloid solutions can be administered to maintain adequate circulating volume. Maintenance intravenous fluids may need to be added or adjusted until urine output has normalized at 0.5 to 2 mL/kg per hour.
Inappropriate Vascular Resistance
Elevated systemic vascular resistance can result from hypervolemia or from the initiation of MCS. Nonpulsatile device flow leads to the release of renin, which stimulates the renin-angiotensin system, causing arteriolar constriction.47 Hypertension can cause bleeding in the patient receiving anticoagulation. Vigilant blood pressure control is essential to reduce the incidence of IVH. In addition, the nurse must assess for and treat agitation and pain as potential causes of hypertension.
Neonatal pulmonary hypertension or postoperative elevated pulmonary vascular resistance that is unresponsive to mechanical ventilation, intravenous pulmonary vasodilators, or inhaled nitric oxide may require ECMO support for patient survival, particularly if the increased pulmonary vascular resistance is associated with low cardiac output and acidosis. ECMO can interrupt the cycle of pulmonary hypertension and provides support until the underlying condition resolves or pulmonary vascular resistance falls.
Native Myocardial Dysfunction
Native cardiac output should be optimized on VV ECMO or LVAD support as a bridge to recovery. During VV ECMO, which does not support cardiac function, and occasionally during LVAD support, right ventricular failure develops and requires inotropic support. Inotropic agents such as milrinone, dopamine, and dobutamine are avoided if possible because they increase myocardial oxygen consumption. However, drugs such as epinephrine and norepinephrine may ultimately be required in patients with more severe myocardial dysfunction or overwhelming sepsis.
Native myocardial function will often improve with afterload reduction, particularly when hypertension is present. The most effective antihypertensive drugs are nitroprusside, nitroglycerin, prostaglandin E1, or hydralazine. Before initiation of these vasodilators, volume status is optimized to minimize the risk of hypotension, and abnormal electrolyte concentrations are corrected.
During VA ECMO, the pump provides nonpulsatile flow, so systemic perfusion will improve if the native myocardium ejects some blood.60 Left ventricular ejection facilitates decompression of the left heart, reducing the need for left atrial venting, and may potentially reduce the risk of intracardiac thrombosis formation from blood stasis. It is challenging to identify the optimal pump flow rate to optimize cardiac output in the presence of right ventricular failure caused by hypoxia and pulmonary hypertension. Before weaning of ECMO support, the native myocardium may require inotropic support.
Arrhythmias
Arrhythmias during MCS can result from hypoxia, from electrolyte imbalance, from irritability caused by atrial cannula position, or as a complication of cardiac surgery. In patients requiring VA ECMO or BiVAD support, atrioventricular synchrony is not crucial because the body is perfused, despite the presence of what are normally nonperfusing arrhythmias. However, synchrony will be critical during weaning from MCS when myocardial function is limited. After cardiotomy, patients with atrial and ventricular epicardial pacing wires may benefit from cardiac pacing if indicated. Antiarrhythmic medication, radiofrequency catheter ablation, or recovery of the underlying myocardial disease may be required before ECMO removal in patients receiving ECMO secondary to lethal arrhythmias.
Tamponade
Pericardial or mediastinal tamponade can develop during MCS. Tamponade inhibits venous return to the MCS device and decreases cardiac output. Signs of cardiac tamponade include a convex or bulging sternotomy patch (if open sternum), increasing right and left atrial pressures with decrease in mean arterial pressure, tachycardia, decreased VAD (cardiac) output, a widened heart size or mediastinum on chest radiograph, cessation of chest tube drainage (if present), or a pericardial effusion with ventricular compression noted on an echocardiogram. The echocardiogram is the definitive diagnostic test, but treatment should not await the study. Treatments include volume administration, inotropic agents, milking or suctioning (with sterile technique) of chest tubes (if present) to remove clot formation, or immediate reexploration of the chest in the operating suite.
VADs must receive blood from the native heart, so any condition that alters this flow of blood to the device will affect VAD performance and the patient’s clinical status. For example, patients receiving LVAD support may have decreased VAD flow despite volume replacement. To improve LVAD flow, the cause of decreased flow (e.g., increased pulmonary vascular resistance, pericardial effusion, decreased native cardiac output) must be identified and treated.
Troubleshooting VAD Pump and Console
As more VADs are placed in children as a bridge to transplant, longer support durations are inevitable. Consequently, VAD dysfunction may become more prevalent. The VAD-trained critical care nurse should examine all external device components such as cannulas, drive lines, battery support, and all mechanical and electrical connections. The nurse should be familiar with the VAD alarms and troubleshooting techniques (see Table 7-4).
The Bio-Medicus centrifugal console has low- and high-flow alarms. Flow can frequently change in response to blood pressure or vascular resistance. Constant visual, auditory, and tactile monitoring is required. Visual and tactile checks of the circuit can detect line “chattering” that would indicate inadequate preload or cannula obstruction. Other visual checks include examination of lines, connector sites, and the centrifugal head for changes in color that could indicate fibrin clot formation. In addition, auditory and tactile checks are needed to detect vibration of the centrifugal head.
The pneumatic console delivers air pressure to compress and empty the blood pump sac delivering pulsatile flow. Each blood pump must fill and eject completely to give the patient an adequate cardiac output. Cardiac output is optimized by adjusting the pressure and vacuum values on the console. Most pneumatic consoles have low-fill alarms, indicating inadequate pump preload before ejection. The nurse should assess the patient’s volume status for any changes or signs of right-sided heart failure during single LVAD support.
Use a flashlight daily to assess external pumps for thrombi formation and complete VAD emptying. Illuminate the pump at an angle to verify transmission of a white light through to the opposite side. If a pink light is noted, then the VAD is not completely emptying. The physician should be notified immediately whenever clot formation is noted or suspected in the circuit or pump. The use of heparin-coated pumps and circuits will hopefully reduce the incidence of thromboembolic complications in the future.
Hematologic Assessment
Thrombosis and bleeding are the most common complications of MCS.60 Patients receiving MCS are susceptible to bleeding from all surgical sites and should be monitored closely (e.g., cannula sites, incisions, chest tubes). All saturated dressings should be measured and included in the total output calculation. All bodily fluids such as stool, urine, gastric drainage, and sputum should be checked for blood. The nurse should monitor the child’s abdominal girth and assess for increased abdominal distension and firmness. In addition, the nurse should try to avoid any procedure that may produce bleeding such as heel sticks, venipunctures or injections, insertion of a nasogastric tube, or urinary catheter or rectal probe insertion.
The patient’s hematocrit, fibrinogen, and platelet counts are routinely monitored, and appropriate blood products are administered as needed. Blood product replacement protocols may vary among institutions. In general, the target platelet count is >100,000/mm3, fibrinogen 100 mg/dL, and hematocrit >35%. If possible, blood transfusions should be limited for patients awaiting transplant, because frequent exposure can increase the panel reactive antibody, which can complicate matching for a future transplantation.11 Blood products should be irradiated, depleted of leukocytes, and negative for cytomegalovirus.11 If excessive nonsurgical bleeding is encountered, aminocaproic acid, aprotinin, and recombinant factor VII may be administered.
Anticoagulation
Anticoagulation management can be an extremely challenging part of management in patients receiving MCS. The goal of anticoagulation therapy is a balance of preventing pump thrombosis formation and bleeding. Blood exposure to an artificial circuit triggers the coagulation cascade and causes clots to form in the circuit. Anticoagulation is therefore necessary to prevent clot formation and the resultant consumptive coagulopathy that would occur as procoagulants are depleted by the clotting process.
Most anticoagulation regimens begin with intravascular heparin sodium infusion generally titrated in the range of 20 to 60 units/kg per hour to achieve a desired aPTT of 60 to 80 sec or an ACT of 140 to 180 sec (VAD) or 200 to 240 sec (ECMO). These ranges can vary depending on the type of support, the technology used to measure them (e.g., Hemochrons, I Stats), the patient’s risk for bleeding, and whether the device circuit is coated with heparin or another bioactive surface. The heparin infusion is usually started once chest tube output has decreased. Patients receiving long-term VAD support can eventually transition to oral anticoagulation, such as warfarin or antiplatelet therapy. Protocols to guide anticoagulation therapy are typically available for specific devices, with recommended INR ranges.
The control of the coagulation system on ECMO is complex. The ACT is followed every hour with titration of the heparin dose. However, AT III levels are low in many patients receiving ECMO, and the combination of AT III and a heparin cofactor is needed for anticoagulant effects. The combination of AT III and a heparin cofactor stops the conversion of fibrinogen to fibrin and ultimately prevents clot formation. A deficiency in AT III can cause heparin to be ineffective, leading to excessive heparin use and the potential for clotting of the circuit. Replacement of AT III or fresh frozen plasma is necessary to restore adequate AT III levels and the anticoagulant effects of heparin.
Patients receiving ECMO also have thrombocytopenia, because platelets are deposited onto artificial surfaces and lost to clot formation in the circuit. In addition, after exposure to the circuit, many remaining platelets are nonfunctional. Thrombocytopenia will ultimately lead to bleeding. Platelet transfusions are given to reverse thrombocytopenia and reduce the risk of hemorrhage.
Consumptive Coagulopathy
Consumptive coagulopathy can develop secondary to clotting in the circuit and inadequate replacement of clotting factors, creating risk of both thromboembolism and risk of bleeding. Replacement with platelet and cryoprecipitate transfusions or use of replacement products, such as factor VII, can precipitate clotting in the circuit. In addition, the use of aminocaproic acid (Amicar) or other fibrinolytic system stabilizers increases the risk of thrombus formation in the device circuit. Fibrinolytics are used before and after surgery in patients receiving ECMO to decrease bleeding from activation of fibrinolysis (clot breakdown), which can increase bleeding. Although stopping lysis of clots is beneficial to control bleeding, the risk is that clot formation is encouraged within the circuit. Monitoring of the circuit and careful titration of heparin can reduce this risk. Patient disorders that cause hypercoagulation or hypocoagulation states can also complicate anticoagulation management.
Heparin-Induced Thrombocytopenia (HIT)
A potential problem with the use of heparin is HIT. HIT occurs in approximately 5% of patients who receive heparin.15 HIT is an immune response with an immune complex (called heparin-platelet factor IV complex) formed between administered heparin and platelet factor IV on the surfaces of platelets. The immune system responds to this complex as a foreign substance and forms antibodies, causing platelet aggregation, platelet damage, and ultimately thrombotic complications and thrombocytopenia. Testing for HIT is done by checking the platelet count and serologic or functional assays for heparin-platelet factor 4 (PF 4) antibodies.
The treatment for HIT is removal of all sources of heparin. Because most patients receiving MCS will require anticoagulation, a direct thrombin inhibitor such as argatroban or lepirudin can be used in place of heparin.5
Fluid and Electrolyte Replacement and Balance
Patients typically are quite edematous during MCS because they often require fluid resuscitation before device insertion, and often develop capillary leak before and after device insertion. Aggressive diuresis can lead to intravascular volume depletion and cardiac compromise. Diuretic therapy is initiated early to promote the removal of edema fluid. A furosemide infusion (0.2 to 0.3 mg/kg per hour) is commonly used to promote diuresis. Chlorothiazide and bumetanide can be added to promote diuresis, or renal dose dopamine infusion can enhance renal perfusion and glomerular filtration. When the child receives diuretics and requires frequent blood replacement, the child’s serum potassium, calcium, and other electrolytes should be closely monitored and replaced as needed.
Renal and Hepatic Function
During the first 24 to 48 hours of MCS therapy, patients often develop acute tubular necrosis with oliguria and rising blood urea nitrogen (BUN) and creatinine. The kidneys and liver may be compromised by low cardiac output, prolonged period of hypoxia or hypotension before initiation of support, prolonged cardiopulmonary bypass (if postoperative), multiple blood transfusions, thromboembolism, antibiotics or other toxic medications, and severe hemolysis.
Monitoring of urine output, BUN and creatinine levels, and liver function studies for worsening or improvement in end-organ perfusion is imperative. Once adequate cardiac output has been restored through the use of MCS, renal and hepatic function should improve. Interventions to minimize renal and hepatic dysfunction include minimizing blood transfusions, providing early nutritional support, monitoring drug levels, and discontinuing of renal or hepatic toxic medications if possible.
If urine output does not improve, the patient may have an open patent ductus arteriosus (PDA) or a combination of inadequate flow, low cardiac output, and low mean arterial pressure compromising renal blood flow. Surgical ligation of the PDA may be warranted if a PDA is documented by echocardiogram.
In the case of rising creatinine levels, hyperkalemia, persistent oliguria, and maximal diuretic support, it may be necessary to perform dialysis via peritoneal dialysis, perform ultrafiltration via the ECMO, or centrifuge VAD circuit. Ultrafiltration is accomplished through the use of a hemofilter connected to the ECMO or centrifuge VAD circuit; water passes through the filter from the plasma, reducing hypervolemia. Ultrafiltration may need to be stopped in the case of low atrial pressure, hypotension, or circuit shutdown from low volume detected by the bladder box.
Nutrition
Infants and children receiving MCS typically have compromised nutritional status secondary to presupport clinical decompensation. Parenteral nutrition and intralipid therapy are usually started within 48 to 72 hours after initiation of MCS with gradual increase to achieve a caloric goal of 80 to 100 kcal/kg per day. Lipid intake should not exceed 1 g/kg per day, or it should be infused through the bladder port to prevent accumulation and embolism within the ECMO circuit. In addition, ranitidine is added in the parenteral nutrition or as a supplementary intravenous medication to inhibit gastric secretions secondary to stress.
Once inotropic support is minimal and bowel sounds have resumed, enteral feedings can be started slowly through a nasogastric tube. Oral feedings can be attempted if the child is extubated. In patients with poor systemic perfusion before MCS, mesenteric artery blood flow is compromised, so feedings should be started slowly with careful monitoring for signs of feeding intolerance such as abdominal distension, high gastric residuals, no stools, or bloody stools. These signs may also be associated with necrotizing enterocolitis in the infant and may not be present until feeding is initiated. The nutritional support service should be involved in the child’s care to determine the current nutritional status, recommend age-appropriate diet or formula and caloric goals, and monitor progress through laboratory values such as total protein and albumin.
Infection
Patients receiving MCS support are at high risk for nosocomial infection secondary to surgical incisions or open chest, cannulae sites, invasive monitoring catheters, drainage tubes, mechanical ventilation, and compromised nutrition and immune status. Early indicators of infection may absent or difficult to detect during MCS. Thrombocytopenia results from platelet destruction by the ECMO circuit or VAD pump. In addition, core body temperature can be affected by the ECMO heat exchanger.47 The white blood cell count with differential should be obtained daily, and routine blood, urine, and endotracheal tube cultures should be obtained to monitor for infection. Patients with unexplained hemodynamic instability, coagulopathy, elevated white blood cell count, or fever should be promptly treated for suspected sepsis pending culture results (see Chapter 6).60
Prophylactic broad-spectrum intravenous antibiotics are administered before mechanical support and are continued after insertion. Cannulation through an open sternum poses a greater risk for mediastinitis. Therefore, in patients with an open sternum, antibiotic coverage is broadened to include a second- or third-generation cephalosporin in combination with a course of vancomycin or oxacillin.9,60
The nurse should monitor the patient closely for signs and symptoms of infection during MCS. In addition, the nurse should use meticulous aseptic and sterile technique when required for procedures or direct patient care.
Skin Assessment
Children receiving MCS are at high risk for developing skin breakdown related to suboptimal nutritional status and immobility. Furthermore, the patient with a pneumatic VAD will have external pumps on the surface of the abdomen, and cannulas, and drive line connections may be a potential source for the development of skin breakdown. VAD site dressing care (either the pump or percutaneous components) is performed according to manufacturer’s recommendations. Strict sterile technique is preferred until the skin has adhered to the Dacron on the cannulae. The nurse should carefully remove encrustations that result from wound secretions to prevent skin necrosis at the cannulae exit sites.
Exit sites should be covered at all times, and the use of age-appropriate binders or pouches is recommended to stabilize pumps or components. Several VADs use proprietary materials with guidelines regarding wound care. Many companies do not recommend oil-based products (ointments), alcohol, or acetone that might degrade the pump case or cannula. (e.g., the Thoratec VAD outer pump casing is sensitive to acetone).
Frequent skin assessment is critical to early detection and prevention of skin breakdown. Early initiation of enteral nutrition and mobility (if requiring long-term use of a pneumatic VAD) can prevent the development of skin breakdown. However, infants or children receiving ECMO might not tolerate position changes. In such patients, low air-flow beds can be used to maintain skin integrity. A gel pillow under the occiput is often used to prevent skin breakdown. Pressure points should be assessed routinely, and passive range-of-motion exercises should be performed to maintain skin integrity and prevent muscle contractures.
Activities of Daily Living and Rehabilitation
For children receiving ECMO or centrifugal VAD support, activities of daily living are limited to passive range-of-motion exercises throughout the duration of support. For the child requiring long-term use of a pneumatic VAD, the goal is extubation and rehabilitation. The primary nurse and multidisciplinary care team facilitate early extubation, discontinuation of invasive catheters, and early ambulation. Patient, parent, and physical therapist involvement are crucial to successful rehabilitation. Allowing the patient to gain independence in activities of daily living and device management (if age appropriate) encourages both functional and psychological well-being. The patient’s family should participate in the rehabilitation phase of recovery.
The multidisciplinary team should develop and follow a daily schedule of activity or exercise plan, rest periods, and sleep during the night. If the child is of school age, daily class work is incorporated into the schedule with input from the hospital or local school district teacher.
Psychological Support
One of the most stressful experiences for any parent is a child’s critical illness. Psychological support in addition to patient and family education are essential components of postoperative MCS care. If emergent VAD placement occurs, patients may wake from anesthesia unaware of the device, and the family often has no time to prepare.
Parents often face overwhelming feelings of anxiety, fear, and helplessness, yet can remain hopeful for successful therapy.17 Age-appropriate explanations of components and functions can gradually introduce the device to the patient and family. The nurse also provides crucial emotional support during the recovery period.
When the child’s condition is stable, visits from classmates and friends are encouraged, provided that visitors are screened for infection. Visits from other VAD recipients can be a positive experience for the patients requiring support as a bridge to transplant and for their families.
Commonly used drugs
The most commonly used drugs during MCS are ones that optimize cardiac and pulmonary function, such as inotropes, antihypertensive or afterload reducing agents, and pulmonary vasodilators. A balance between hemostasis and anticoagulation is required after initiation of MCS. Multiple antifibrinolytics and anticoagulants can be used depending on postinsertion bleeding, development of coagulopathies, and device-specific coagulation to prevent thrombus formation. Other commonly used drugs during MCS include analgesics, sedatives, antibiotics, diuretics, electrolyte replacement, and resuscitation drugs (in case of mechanical failure and emergent removal from ECMO). Table 7-5 includes a list of commonly used drugs with doses and cautions. See Chapter 6 for further description of resuscitation.
Table 7-5 Drugs Commonly Used During MCS
| Drug | Dose | Caution |
| Anticoagulation* | ||
| Heparin sodium | IV infusion: 20 units/kg per hour | May cause thrombocytopenia or HIT; hemorrhage is the most common complication; monitor platelet count, PTT, hematocrit, and ACT |
| Adjust dose by 2-4 units/kg per hour every 4-8 h as required | ||
| Aspirin | For Blalock-Taussig (subclavian to pulmonary artery) shunts: 3-5 mg/kg per day up to 40-81 mg/day | At high serum concentrations, may cause GI intolerance, respiratory alkalosis, and pulmonary edema |
| For prosthetic heart valves: 6-20 mg/kg per day | ||
| Aminocaproic acid (Amicar) | IV infusion: 33.3 mg/kg per hour | May cause hypotension, bradycardia, and arrhythmias |
| Argroban | Initial dose: 2 mcg/kg per minute | May be used for anticoagulation in patients diagnosed with HIT; may cause hypotension, ventricular tachycardia, and bradycardia |
| Measure aPTT after 2 h; adjust dose until aPTT is 1.5-3 times baseline | ||
| Recombinant activated factor VII (rFVIIa; NovoSeven) | 90 mcg/kg q2 h until hemostasis is achieved or judged ineffective | May cause hypertension or hemorrhage; monitor for signs/symptoms of thrombosis |
| Warfarin sodium (Coumadin) | Maintenance dose: 0.05-0.34 mg/kg per day | For long-term use in stable patient with a VAD; monitor INR or PT and signs/symptoms of hemorrhage |
| Titrate based on INR | ||
| Aprotinin (Trasylol) | 40 mL/m2 per hour | May cause renal impairment, heart attack, and stroke |
| Inotropes† | ||
| Dopamine (Inotropin) | 2-20 mcg/kg per minute (titrate to desired effect) | May produce extreme tachycardia or result in increased pulmonary artery pressure |
| Dobutamine (Dobutrex) | 2-20 mcg/kg per minute (titrate to desired effect) | May produce extreme tachycardia and pulmonary vasoconstriction; patients with atrial fibrillation or flutter are at risk of developing a rapid ventricular response |
| Epinephrine (Adrenalin) | 0.2-0.3 mcg/kg per minute | May produce tachycardia, hypertension, cardiac arrhythmias, increased myocardial oxygen consumption, decreased renal, and splenic blood flow |
| Norepinephrine | 0.05-1 mcg/kg per minute | May produce arrhythmias, bradycardia, tachycardia, hypertension, or organ ischemia caused by vasoconstriction of renal and mesenteric arteries |
| Vasopressin (Pitressin) | 0.2 to 2 milliunits/kg per minute (0.0002-0.002 units/kg per minute | Can produce venous thrombosis, vasoconstriction, bradycardia, heart block, and distal limb ischemia |
| Phosphodiesterase-III inhibitor‡ | ||
| Milrinone (Primacor) | Loading dose of 0.05 mg/kg (50 mcg/kg) slowly (over 10-60 min). Infusion dose: 0.25-0.75 mcg/kg per minute | Can produce ventricular and supraventricular arrhythmias and bronchospasm |
| Vasodilators/Antihypertensives§ | ||
| Nitropresside (Nipride) | IV: 0.3-8 mcg/kg per minute | Can produce excessive hypotensive response, tachycardia, and elevated serum creatinine |
| Enalapril (Vasotec) | PO: 0.1-0.5 mg/kg per day in 1-2 doses | Can produce hypotension, angioedema, anaphylactic reactions, neutropenia, proteinuria, and hepatic failure |
| IV: 5-10 mcg/kg per dose q8-24 h | ||
| Captopril (Capoten) | PO: 0.1-0.5 mg/kg per dose q8-24 h | Can produce hypotension hepatic failure, anaphylactoid reactions, agranulocytosis, proteinuria |
| Maximum: 6 mg/kg per day | ||
| Esmolol (Brevibloc) | IV: 75-150 mcg/kg per minute | May cause bradycardia, hypotension, peripheral ischemia |
| Titrate dose by 50 mcg/kg per min q10 min | ||
| Maximum dose: 1000 mcg/kg per minute | ||
| Propranolol (Inderal) | PO: 0.5 mg/kg per day | May cause hypotension and bradycardia. |
| IV: 0.15-0.25 mg/kg | ||
| Pulmonary Vasodilators|| | ||
| Inhaled Nitric Oxide (INOmax) | In the ventilated patient: 5-20 ppm | Abrupt discontinuation may lead to worsening hypotension, oxygenation, and increasing pulmonary artery pressure; doses >20 ppm increase the risk of methemoglobinemia and elevated nitrogen dioxide levels |
| Sildenafil (Viagra) | PO: 0.25-2 mg/kg per dose q4-8 h | May cause hypotension, tachycardia, ventricular arrhythmia, cerebrovascular hemorrhage, pulmonary hemorrhage, sudden hearing loss |
| Diuretics¶ | ||
| Furosemide (Lasix) | PO, IV: 1-2 mg/kg per dose q4-12 h | May cause hypokalemia and hyponatremia necessitating electrolyte monitoring |
| IV continuous infusion: 0.1-0.4 mg/kg per hour | ||
| Chlorothiazide (Diuril) | PO: 10-40 mg/kg per day divided in 2 doses | May cause hypokalemia, hyperglycemia, and hyperuricemia |
| IV: 2-8 mg/kg per day divided into 1-2 doses | ||
| Spironolactone (Aldactone) | PO: 1-3.5 mg/kg per day; may be given as single dose or total dose can be divided into 2 or 4 doses (given twice a day or every 6 h) | Potassium-sparing diuretic; may cause hyponatremia, hyperkalemia, and dehydration |
| Bumetanide (Bumex) | PO: 0.04-0.8 mg/kg per day; dose is divided and given every 6-8 h | Can have profound diuresis with fluid and electrolyte loss; close supervision and dose evaluation are required. |
| Electrolyte Replacement# | ||
| Potassium chloride | Normal requirement: 2-3 mEq/kg per day | May cause cardiac arrhythmias, heart block, hypotension, and bradycardia; patients have a cardiac monitor during intermittent infusions |
| Prevention of hypokalemia: 1-2 mEq/kg per day above maintenance | ||
| IV intermittent infusion for severe depletion: 0.5-1 mEq/kg per dose, typically over 1-3 hours | ||
| Calcium chloride | IV: 10-20 mg/kg per dose | May cause vasodilation, hypotension, bradycardia, and cardiac arrhythmias; avoid extravasation |
| Repeat q4-6 h if needed | ||
| Magnesium sulfate | PO: 100-200 mg/kg per dose q6 h | May cause hypotension, circulatory collapse, depression of cardiac function, and heart block |
| IV: 25-50 mg/kg per dose q4-6 h | ||
ACT, Activated clotting time; aPTT, activated partial thromboplastin time; GI, gastrointestinal; HIT, heparin-induced thrombocytopenia; INR, international normalized ratio; IV, intravenous; PO, by mouth; PT, prothrombin time; PTT, partial prothrombin time; VAD, ventricular assist device.
* Primary concern related to blood-surface interaction with artificial materials, increasing the risk of clot in the mechanical circulatory support device.
† Indicated for poor systemic perfusion and myocardial function with appropriate oxygenation, ventilation, heart rate, and intravascular volume.
‡ Inotropic, chronotropic, and vasodilatory effects.
§ Dilate arteries and veins to reduce preload, reduce systemic vascular resistance, and improve ventricular compliance, thus improving cardiac output.
|| To aid in oxygenation and decrease right ventricular afterload.
¶ For treatment of oliguria and hypervolemia associated with poor systemic perfusion and CHF as the result of low cardiac output.
# Electrolyte imbalances alter myocardial transmembrane potential, depolarization, and repolarization, thus affecting the excitability of myocardial tissue and conduction of electrical impulses. To ensure optimal myocardial function and cardiac output, electrolytes must be closely monitored and replaced as necessary.
MCS for specific diseases
The Chapter 7 Supplement on the Evolve Website provides detailed information about common diseases requiring MCS support. Many of the specifics can be reviewed in more depth in Chapters 6 to 9. It is important to remember that the specific diseases discussed are at end-stage and require MCS for survival.
Diagnostic tests during MCS
The following diagnostic tests are the most commonly used to assess cardiac and respiratory function and to evaluate for possible postoperative MCS complications. Refer to Chapters 8 to 10 for more detailed descriptions of the tests.
Echocardiogram
The echocardiogram is often used during ECMO and VAD support to assess for myocardial recovery (by measuring the ejection fraction) and to check cannula placement. The ECMO and VAD flows are reduced to allow the heart to eject blood while receiving minimal inotropic support to best assess readiness for ECMO decannulation or VAD removal.
Cardiac Catheterization
The use of cardiac catheterization is rare during mechanical support because of the risk of bleeding. However, select cases may require cardiac catheterization to obtain muscle biopsy for rejection status in the patient requiring mechanical support after heart transplantation, before heart or lung transplant to assess pulmonary artery pressure, or after a cardiotomy to identify any residual defects that can be corrected surgically to improve ventricular function.
Chest Radiograph
The chest radiograph is frequently used to assess endotracheal tube, invasive catheter, chest tube, and ECMO/VAD cannula placement. Chest radiographs should be evaluated for clinical deterioration or unexplained changes in the patient’s condition caused by pleural effusion, hemothorax, pneumothorax, or lobe collapse. General opacification of lung fields within the first 24 hours of ECMO is common and results from complement activation, leading to capillary leak syndrome and withdrawal of distending airway pressure.
Arterial and Venous Blood Gas Assessment
Arterial and venous blood samples are used to assess oxygenation and mixed venous saturation. The preoxygenator blood gas samples are from the right atrium and considered the venous blood gas. The postoxygenator blood gas assesses the oxygenator function, and the child’s blood gas represents the arterial blood gas sample. Venous and arterial pH, PaO2 and PaCO2 should be within normal limits. Increasing the sweep gas decreases PaCO2 levels and increasing the ECMO flow rate elevates the PaO2 levels.
Cranial Ultrasound
IVH is a possible complication of anticoagulation associated with MCS. The risk of IVH is higher in the neonate and infant because of the continued development of the cerebral vasculature and germinal matrix that are highly sensitive to changes in pH, PaO2, and intracranial pressure. In patients with an open fontanelle, cranial ultrasound examination is routinely performed before support is initiated, and routine surveillance is per institutional protocol. Development of a large IVH may preclude continuation of ECMO or VAD support.
1 Alexi-Meskishvili V., et al. The use of the Berlin heart in children. In: Duncan B.W., editor. Mechanical support for cardiac and respiratory failure in pediatric patients. New York: Marcel Dekker, 2001.
2 Ashton R.C.Jr, et al. Left ventricular assist device options in pediatric patients. ASAIO J. 1995;41:M277-M280.
3 Baffes T.G., et al. Extracorporeal circulation for support of palliative cardiac surgery in infants. Ann Thorac Surg. 1970;10:354-363.
4 Baldwin J.T., et al. The national heart, lung, and blood institute pediatric circulatory support program. Circulation. 2006;113:147-155.
5 Baroletti S.A., Goldhaber S.Z. Heparin-induced thrombocytopenia. Circulation. 2006;114:355-356.
6 Bartlett R.H., et al. Extracorporeal membrane oxygenation (ECMO) cardiopulmonary support in infancy. Trans Am Soc Artif Intern Organs. 1976;22:80-93.
7 Boneva R.S., et al. Mortality associated with congenital heart defects in the United States: trends and racial disparities, 1979-1997. Circulation. 2001;103:2376-2381.
8 Boucek M.M., et al. Registry for the International Society for Heart and Lung Transplantation: seventh official pediatric report: 2004. J Heart Lung Transplant. 2004;23:933-947.
9 Brown K.L., et al. Healthcare-associated infection in pediatric patients on extracorporeal life support: the role of multidisciplinary surveillance. Pediatr Crit Care Med. 2006;7:546-550.
10 Bruckner B.A., et al. Regression of fibrosis and hypertrophy in failing myocardium following mechanical circulatory support. J Heart Lung Transplant. 2001;20:457.
11 Carberry K.E., et al. Mechanical circulatory support for the pediatric patient. Crit Care Nurs Q. 2007;30(2):121-142.
12 Chen Y.S., et al. Experience and result of extracorporeal membrane oxygenation in treating fulminant myocarditis with shock: what mechanical support should be considered first? J Heart Lung Transplant. 2005;24:81-87.
13 Clowes G.H., Hopkins A.L., Neville W.E. An artificial lung dependent upon diffusion of oxygen and carbon dioxide through plastic membranes. J Thorac Cardiovasc Surg. 1956;32:630-637.
14 Collison P.S., Dager S.K. The role of the Intra-aortic balloon pump in supporting children with acute cardiac failure. Postgrad Med J. 2007;83:308-311.
15 Comunale M.E., Vancott E.M. Heparin induced thrombocytopenia. Int Anesthesiol Clin. 2004;42(3):27-43.
16 Cooper D.S., et al. Cardiac extracorporeal life support: state of the art in 2007. Cardiol Young. 2007;17(Suppl. 2):104-115.
17 Curley M.A., Meyer E.C. Parental experience of highly technical therapy: survivors and nonsurvivors of extracorporeal membrane oxygenation support. Pediatr Crit Care Med. 2003;4(2):214-219.
18 De Oliveira N.C., et al. Prevention of sudden circulatory collapse after the Norwood operation. Circulation. 2004;110:II133-II138.
19 Dickerson H.A., Chang A.C. Perioperative management of ventricular assist devices in children and adolescents. Semin Thorac Cardiovasc Surg Pediatr Card Surg Annu. 2006;9:128-139.
20 Duncan B.W., et al. Mechanical circulatory support for the treatment of children with acute fulminant myocarditis. J Thorac Cardiovasc Surg. 2001;122:440-448.
21 Duncan B.W. Mechanical circulatory support for infants and children with cardiac disease. Ann Thorac Surg. 2002;73:1670-1677.
22 Duncan B.W. Mechanical circulatory support in infants and children with cardiac disease. In Zwischenberger J.B., Bartlett R.H., editors: ECMO Extracorporeal Cardiopulmonary Support in Critical Care, ed 2, Ann Arbor, MI: Extracorporeal Life Support Organization, 2000.
23 Duncan B.W. Pediatric mechanical circulatory support in the United States: past, present, and future. ASAIO J. 2006;52:525-529.
24 : ECMO Registry of the Extracorporeal Life Support Organization (ELSO). January 2004 Ann Arbor, Michigan
25 Fenton K.N., et al. The significance of baseline cerebral oxygen saturation in children undergoing congenital heart surgery. Am J Surg. 2005;190(2):260-263.
26 Frazier O.H., et al. Research and development of an implantable axial-flow left ventricular assist device: the Jarvik 2000 Heart. Ann Thorac Surg. 2001;71:S125-S132.
27 Fuchs A., Netz H. Ventricular assist devices in pediatrics. Images Paediatr Cardiol. 2002;9:24-54.
28 Goldman A.P., et al. The waiting game: bridging to paediatric heart transplantation. Lancet. 2003;362:1967-1970.
29 Goodman D.M., Green T.P. Extracorporeal life support for children with acute respiratory distress syndrome. In: Zwischenberger J.B., Steinhorn R.H., Bartlett R.H., editors. ECMO: extracorporeal cardiopulmonary support in critical care. Ann Arbor, MI: Extracorporeal Life Support Organization; 2000:451-460.
30 Griffith B.P., et al. HeartMate II left ventricular assist system: from concept to first clinical use. Ann Thorac Surg. 2001;71:S116-S120.
31 Grinda J.M., et al. Fulminant myocarditis in adults and children: bi-ventricular assist device for recovery. Eur J Cardiothorac Surg. 2004;26:1169-1173.
32 Hansell D.R. Extracorporeal membrane oxygenation for perinatal and pediatric patients. Respir Care. 2003;48(4):352-362.
33 Hawkin J.A., Minich L.L. Intraaortic balloon counterpulsation for children with cardiac disease. In: Duncan B.W., editor. Mechanical support for cardiac and respiratory failure in pediatric patients. New York: Marcel Dekker, 2001.
34 Heerdt P.M., et al. Chronic unloading by left ventricular assist device reverses contractile dysfunction and alters gene expression in end-stage heart failure. Circulation. 2000;102:2713.
35 Helman D.N., et al. Implantable left ventricular assist devices can successfully bridge adolescent patients to transplant. J Heart Lung Transplant. 2000;19:121-126.
36 Hetzer R., Stiller B. Ventricular assist device for children. Nat Clin Pract Cardiovasc Med. 2006;3(7):377-386.
37 Hill J.D., et al. Prolonged extracorporeal oxygenation for acute post-traumatic respiratory failure (shock-lung syndrome). N Engl J Med. 1972;286:629-634.
38 Hintz S.R., et al. Decreased use of neonatal extracorporeal membrane oxygenation (ECMO): how new treatment modalities have affected ECMO utilization. Pediatrics. 2000;106(6):1339-1343.
39 Kalavrouziotis G., Karunaratne A., Raja S. Intraaortic balloon pumping in children undergoing cardiac surgery: an update on the Liverpool experience. J Thorac Cardiovasc Surg. 2006;131:1382-1389.
40 Karl T.R., Horton S.B. Centrifugal pump ventricular assist device in pediatric cardiac surgery. In: Duncan B.W., editor. Mechanical support for cardiac and respiratory failure in pediatric patients. New York: Marcel Dekker, 2001.
41 Kolobow T., Bowman R.L. Construction and evaluation of an alveolar membrane artificial heart-lung. Trans Am Soc Artif Intern Organs. 1963;9:238-243.
42 Korfer R., et al. Single-center experience with the Thoratec ventricular assist device. J Thorac Cardiovasc Surg. 2000;119:596-600.
43 Lequier L. Extracorporeal life support in pediatric and neonatal critical care: a review. J Intensive Care Med. 2004;19:243-258.
44 Lipshultz S.E., et al. The incidence of pediatric cardiomyopathy in two regions of the United States. N Engl J Med. 2003;348:1647-1655.
45 McBride L.R., et al. Clinical experience with 111 Thoratec ventricular assist devices. Ann Thorac Surg. 1999;67:1233-1239.
46 Morales D.L., et al. Lessons learned from the first application of the DeBakey VAD Child: an intracorporeal ventricular assist device for children. J Heart Lung Transplant. 2005;24:331-337.
47 Moynihan P.J., et al. Nursing management of children with cardiac disease on mechanical circulatory support. In: Duncan B.W., editor. Mechanical support for cardiac and respiratory failure in pediatric patients. New York: Marcel Dekker, 2001.
48 Ogletree-Hughes M.L., et al. Mechanical unloading restores beta-adrenergic responsiveness and reverses receptor down regulation in the failing human heart. Circulation. 2001;104:881.
49 Petrou S., et al. Cost-effectiveness of neonatal extracorporeal membrane oxygenation based on 7-year results from the United Kingdom Collaborative ECMO Trial. Pediatrics. 2006;117(5):1640-1649.
50 Pinkney K.A., et al. Current results with intraaortic balloon pumping in infants and children. Ann Thorac Surg. 2002;73:887-891.
51 Pizarro C., et al. Is there a role for extracorporeal life support after stage I Norwood? Eur J Cardiothorac Surg. 2001;19:294-301.
52 Pollock J., Charlton M.C., Williams W.G. Intraaortic balloon pumping in children. Ann Thorac Surg. 1980;29:522-528.
53 Ravishankar C., et al. Extracorporeal membrane oxygenation after staged I reconstruction for hypoplastic left heart syndrome. Pediatr Crit Care Med. 2006;7:319-323.
54 Schmid C., et al. Pediatric assist with Medos and Excor systems in small children. ASAIO J. 2006;52:505-508.
55 Slaughter MS, et al. Advanced heart failure treated with continuous-flow left ventricular assist device. N Engl J Med. 2009;361:2241-2251.
56 Throckmorton A.L., et al. Pediatric circulatory support systems. ASAIO J. 2002;48:216-221.
57 UK Collaborative ECMO Trial Group. UK collaborative randomised trial of neonatal extracorporeal membrane oxygenation. Lancet. 1996;348:75-82.
58 Ungerleider R.M., et al. Routine mechanical ventricular assist following the Norwood procedure—improved neurologic outcome and excellent hospital survival. Ann Thorac Surg. 2004;77:18-22.
59 Van Meurs K.P., et al, editors. ECMO: extracorporeal cardiopulmonary support in critical care, ed 3, Ann Arbor, MI: Extracorporeal Life Support Organization, 2005.
60 Wessel D.L., Almodovar M.C., Laussen P.C. Intensive care management of cardiac patients on extracorporeal membrane oxygenation. In: Duncan B.W., editor. Mechanical support for cardiac and respiratory failure in pediatric patients. New York: Marcel Dekker, 2001.
61 Wieselthaler G.M., et al. First clinical experience with the DeBakey VAD continuous-axial-flow pump for bridge to transplantation. Circulation. 2000;101:356-359.
62 Zahraa J.N., et al. Venovenous versus venoarterial extracorporeal life support for pediatric respiratory failure: are there differences in survival and acute complications? Crit Care Med. 2000;28(2):521-525.