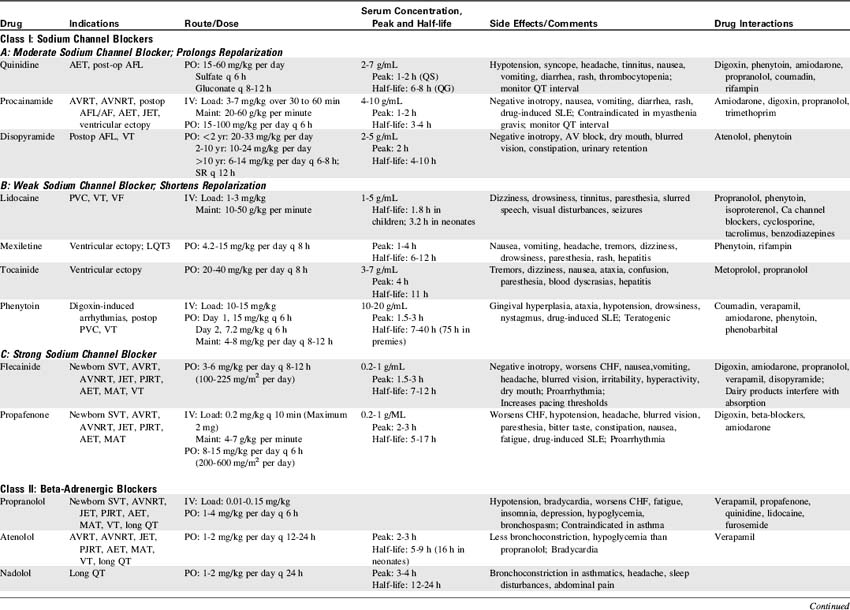
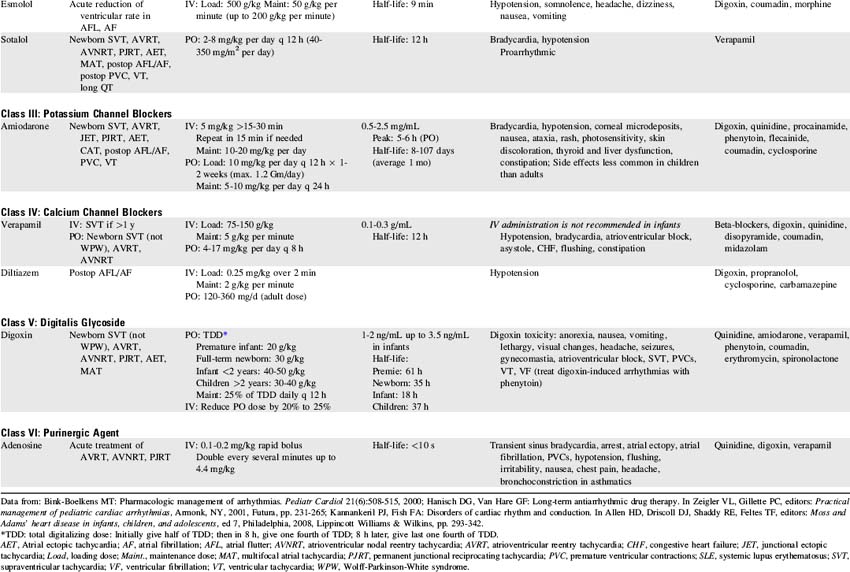
Management
Care of the child with congestive heart failure targets improvement of cardiac function and elimination of excess intravascular fluid. In addition, oxygen delivery must be supported and oxygen demands controlled or minimized. Reversible causes of congestive heart failure also must be treated.
Improvement in Cardiac Function: Digitalis Derivatives
Digitalis may be extremely effective in the treatment of congestive heart failure. It must, however, be used appropriately with careful monitoring for therapeutic and side and toxic effects.
Therapeutic Effects
Digitalis (administered predominantly as digoxin in children) continues to be used in the treatment of congestive heart failure in children, although evidence of its efficacy in pediatric patients is controversial.435 The presumed effect of digoxin in older children is inotropic, improving ventricular contractility. Digoxin also affects the excitability of myocardial cells and slows the heart rate. Pediatric digoxin therapy (particularly during infancy) also appears to relieve some symptoms of congestive heart failure through effects on oxygen consumption.
The inotropic effects of digoxin result from inhibition of the sodium-potassium pump, so that sodium accumulates intracellularly. Sodium then competes with calcium for sites on the sodium-calcium exchange mechanism, raising intracellular calcium levels and improving myocardial contractility. Digitalis also interacts with the sarcolemma to sequester calcium, so that intracellular calcium is increased further.355,841
Digoxin also slows the heart rate by lowering the resting membrane potential, increasing parasympathetic sensitivity, and reducing sensitivity to norepinephrine. It slows conduction velocity and increases recovery time of the atrioventricular (AV) node. These effects result in a decrease in heart rate but also may contribute to increased ectopic activity.
Digitalis has some direct and indirect peripheral and coronary vasoconstrictive effects, resulting in increased systemic vascular resistance.355,735 When digitalis is administered to patients with congestive heart failure, it appears to antagonize peripheral effects of catecholamines, thereby reducing oxygen consumption.
The clinical effects of digoxin administration in infants may be variable,473,789 and positive effects of digoxin may be related to effects on oxygen supply and demand rather than to inotropic effects. Digoxin administration to the premature neonate may not produce significant improvement in contractility, and the incidence of clinical toxicity is significant in these patients.435 For this reason, digoxin may not be useful in the treatment of congestive heart failure in premature neonates.
Digoxin administration often fails to produce measurable echocardiographic evidence of improvement in cardiac contractility in full-term infants with congestive heart failure caused by a ventricular septal defect. However, demonstrable clinical improvement often is noted; this improvement probably is related to a significant fall in oxygen consumption.435 Studies of digoxin's effects on animals during periods of induced anemia and reduced oxygen transport have documented reduced oxygen consumption and improvement in the match between oxygen supply and oxygen demand.769
Dose
When digoxin therapy is initiated, several loading (“digitalizing”) doses typically are administered to provide therapeutic serum levels of the drug. Once these levels are achieved, maintenance doses of digoxin are administered to replace the child's estimated daily renal excretion of the drug, so that therapeutic levels are maintained (Table 8-7).
Table 8-7 Pediatric Digoxin Dose507
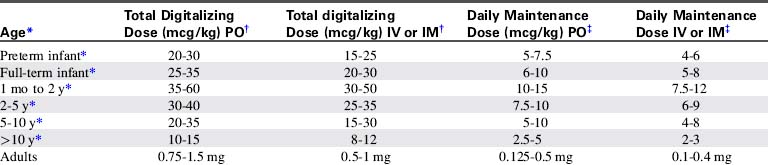
Digoxin doses must be modified based on the child's age and clinical condition. Because digoxin is excreted through the kidneys, neonates who have limited renal tubular function and children with renal failure require reduced dosing. Premature neonates have limited renal tubular function; they are more sensitive to digoxin and require smaller digoxin doses than full-term neonates or older children.355 For this reason the risk of digoxin toxicity is highest in premature neonates.
Infants have a larger volume of digoxin distribution than older children because infants have more red blood cell binding sites for digoxin than do older patients. For this reason, higher loading and maintenance doses of digoxin were recommended for neonates and young infants in the past. Although young infants apparently tolerate high doses and high serum digoxin levels better than adults, it is not clear that the higher dosing is beneficial during infancy. Beyond the neonatal period patients normally excrete approximately one third of the daily administered oral dose.507
Whenever digoxin is administered to a critically ill child, careful consideration of dose and careful monitoring for evidence of toxicity is required. In any patient the appropriate dose of digoxin is the minimum dose necessary to produce therapeutic effects. There is a very small difference between optimal therapeutic levels of digoxin and toxic levels of the drug.
Children with inflammatory cardiac diseases (e.g., myocarditis or cardiomyopathy), chronic hypoxemia, and postoperative cardiovascular patients may demonstrate increased sensitivity to the drug.355 When providing digoxin therapy for the first time to these patients it is prudent to consider providing lower doses of digoxin, or more gradual loading of the drug. Maintenance doses of digoxin may be administered without previous loading doses; this will result in achievement of therapeutic digoxin levels over 4 to 5 days.841
The total oral digitalizing dose for children is approximately 25 to 60 mcg/kg (or 0.025-0.060 mg/kg), and the higher doses in these ranges usually are administered to children 2 years of age or less. The digitalizing or loading dose usually is administered in two to four divided oral doses over 24 to 48 hours.
The maintenance digoxin dose is approximately one eighth (12%) of the total loading dose, administered twice a day. The child receives approximately one fourth of the loading dose daily.
Administration
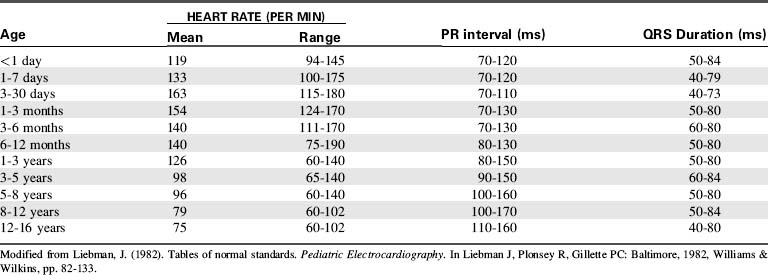
Before administration of the final loading dose, an electrocardiogram or rhythm strip is typically ordered to ensure that arrhythmias have not developed. Because digoxin slows conduction through the AV node, an increased P-R interval often is present following establishment of a therapeutic serum digoxin level. However, second- or third-degree heart block or ectopy should be reported to a physician or the on-call provider before the next dose of digoxin is administered.
In the hospital the child's heart rate usually is checked before administration of any digoxin. If bradycardia is detected the dose usually is held until an electrocardiographic rhythm strip is obtained and toxic heart block or arrhythmias are ruled out. When attempting to determine if digoxin toxicity is present, a particular heart rate is usually less important than assessment of the child's systemic perfusion and evaluation of the electrocardiogram for evidence of heart block (see section, Digoxin Levels).
If the child vomits after administration of an oral dose of digoxin in the hospital, a physician or on-call provider should be consulted before the dose is readministered. It is often difficult to determine how much of the dose was lost, so a repeat dose may result in elevation of serum digoxin levels. In addition, vomiting may be a sign of digoxin toxicity.
It is imperative that the written order for and preparation of the digitalizing doses be double checked before administration because it is very easy to make an error by a factor of 10 or 100 when working with micrograms and milligrams. The order for the digoxin dose should be written (verbal orders should not be used) to avoid miscommunication. The order should be written in both milligrams and micrograms.
Digoxin should be administered with caution to children with decreased renal function, and the dose should be reduced accordingly. Hypokalemia can contribute to the development of clinical signs of digoxin toxicity even in the presence of relatively low serum digoxin levels, so the serum potassium should be monitored and potassium supplementation provided as needed. Hypomagnesemia and hypercalcemia also may aggravate digitalis toxicity, and quinidine may potentiate digoxin toxicity.355 This is especially pertinent as many patients receiving digoxin are also on concomitant diuretics, which may cause electrolyte disturbances.
Digoxin Levels
A serum digoxin level may be monitored when digoxin therapy is instituted (during the “loading phase”), when the child's response to therapy is suboptimal, when toxicity is suspected, or when the drug dose is changed. Therapeutic serum digoxin levels vary from institution to institution but are in the range of 1.1 to 2.2 ng/mL (nontoxic levels in infants may be as high as 3.5 ng/mL). Serum digoxin levels exceeding 3.5 ng/mL are generally considered toxic.
Blood sampling for serum digoxin levels should be performed at prescribed intervals following digoxin administration (consult with the child's physician or ordering provider and hospital clinical laboratory). These levels must be interpreted with caution. As noted, hypokalemia, hypomagnesemia, and hypercalcemia can aggravate digoxin cardiotoxicity even in the presence of “normal” digoxin levels.735 Some children exhibit endogenous digitalis-like substances that can influence serum digoxin levels. In addition, premature neonates may demonstrate bradyarrhythmias even in the presence of “therapeutic” levels of digoxin. For these reasons the presence of clinical symptoms compatible with digoxin toxicity usually is interpreted more strongly than the serum digoxin level alone.
Several common critical care drugs are known to affect digoxin levels. Amiodarone, verapamil, diltiazem, spironolactone, carvedilol and indomethacin all increase serum digoxin levels.735 The digoxin dose should be reduced during concurrent administration of these drugs, and serum digoxin levels should be monitored.
Digoxin Toxicity
The most serious toxic effects of digoxin in children are arrhythmias, which may be observed in the absence of other clinical signs of toxicity. The most common arrhythmia in young children is bradycardia, although heart block and atrial and ventricular premature contractions (ectopy), and ventricular fibrillation have been reported. Virtually any new arrhythmia appearing after initiation of digoxin therapy may be caused by digoxin toxicity.735
Less specific and less common signs of digoxin toxicity in children include anorexia, nausea, vomiting, and diarrhea. Drowsiness and lethargy are common signs of toxicity in infants and young children.356
If digoxin toxicity is suspected, the physician or on-call provider should be notified; further digoxin usually is held pending the results of serum digoxin-level testing. A blood specimen should be drawn for laboratory analysis. Toxicity may be present at levels as low as 2.5 ng/mL or lower; the serum digoxin level must be evaluated in light of the patient's clinical condition.355
If toxicity is discovered in the asymptomatic child, electrocardiographic monitoring should be instituted and the child should be observed closely for the development of arrhythmias. If large amounts of oral digoxin have recently been ingested or administered, induced emesis soon after the ingestion may succeed in recovering 35% to 40% of the ingested drug. Vomiting should only be induced if the child is alert and demonstrates a cough and gag reflex. If massive amounts of digoxin have been ingested, the insertion of temporary transvenous pacing wires is recommended before the development of symptoms.
Treatment of symptomatic digoxin toxicity requires support of cardiovascular function (including treatment of arrhythmias), prevention of further drug absorption, and enhancement of digoxin excretion.356 Bradycardia usually is treated with atropine or pacemaker therapy. Phenytoin (2-5 mg/kg IV) often is effective in the treatment of digoxin-induced arrhythmias because it increases the sinoatrial node conduction rate and reduces automaticity. Lidocaine does not affect atrial activity but will suppress ventricular automaticity, and so may be effective in the treatment of ventricular tachyarrhythmias.735,841 Synchronized cardioversion may convert ventricular tachycardia to refractory fibrillation,365 so it should not be performed.
When renal function is reduced, digoxin excretion is impaired and toxicity may develop more readily. The digoxin level may remain elevated long after the digoxin therapy is stopped in these patients, so support of cardiorespiratory function may be required for several days.
Digoxin excretion is not improved by the administration of furosemide (or other diuretics), exchange transfusion, or dialysis. Hemoperfusion using activated charcoal has had limited effect because the digoxin usually is distributed and bound extensively in tissue.356,365
Life-threatening digoxin toxicity associated with malignant arrhythmias, hypotension, and poor systemic perfusion is treated with digoxin-specific Fab antibody fragments.507 This antibody binds serum digoxin, rendering it inactive.365 The dose of Fab provided is determined by the total body exposure to digoxin, which can be estimated from the digoxin level or the amount of digoxin ingested (for formulas, see Table 8-7 or package insert). In general, approximately 40 mg of purified digoxin-specific Fab will bind approximately 0.5 mg of digoxin.507 Note that digoxin elixir is considered to be absorbed totally, while digoxin tablets generally are calculated to be 80% absorbed.
Parent Instruction
If the parents will be administering digoxin at home, the parents must be taught how to administer the drug. In addition, the parents must be taught what to do if a dose is omitted or if the child vomits after the medication is administered. It is usually helpful to provide the parents with a specific approximate administration schedule; for example, the digoxin may be given at 8 AM and 8 PM. If the morning or evening dose is forgotten, but remembered by 12 noon or midnight, respectively, it may be given. However, if the drug is forgotten and not remembered until after 12 noon or midnight, that dose should be omitted and should not be “made up” in subsequent doses. If the parents are unsure whether a specific dose was administered, that dose should be omitted. If the child vomits after receiving the digoxin, the dose probably should not be repeated because it is difficult to predict how much of the drug was absorbed before regurgitation.
Parents should be taught to contact the child's physician or on-call provider if more than one dose of digoxin is omitted or if the child appears ill for any reason, because digoxin toxicity may be present. Parents are typically not taught to count the child's pulse routinely before administration of digoxin doses, because this focuses attention on specific numbers rather than on overall assessment of the child, and it can increase the parents' anxiety. Monitoring of heart rate does not ensure better detection of digoxin toxicity than that resulting from general evaluation of the child's condition.
The parents should be aware that a digoxin overdose may cause serious arrhythmias or death. Digoxin must be kept out of reach of children, and the medication bottle should have a “child-proof” cap.
Improvement in Cardiac Function: Additional Inotropic Agents
Several inotropic agents may effectively improve myocardial contractility during the treatment of congestive heart failure. Dopamine, dobutamine, and epinephrine are adrenergic agonists that may be titrated to provide beta-1 sympathomimetic effects (increased heart rate, atrioventricular conduction velocity, and ventricular contractility). Each of these drugs also may produce peripheral vascular effects that must be considered during drug selection and administration. An additional nonadrenergic inodilator, milrinone, improves myocardial contractility by inhibition of phosphodiesterase, so that intracellular effects of circulating catecholamines are prolonged. (For further information see Shock, Chapter 6.)
Vasodilator Therapy
Vasodilator therapy may improve myocardial function by altering both ventricular preload and afterload. Ventricular preload is reduced as a result of venodilation and displacement of blood volume into venous capacitance vessels. Ventricular afterload is reduced as a result of arterial dilation; in addition, ventricular wall stress decreases when ventricular chamber size is reduced. When vasodilator therapy is provided, the patient's ventricular compliance curve is altered, so that higher ventricular end-diastolic volume (and ultimately, stroke volume) is present at lower ventricular end-diastolic pressure (see Fig. 8-13 earlier in chapter).
Obviously the beneficial effects of vasodilators must be balanced with the potential detrimental effects of reduction in venous return and the potential fall in blood pressure. The hypovolemic patient is particularly likely to become hypotensive during vasodilator therapy. Volume expanders always should be readily available during the initial administration of these drugs.
No vasodilator is a pure arterial or venous dilator. However, these drugs often are classified by their primary sites of action. Vasodilators that dilate both arteries and veins include nitroprusside, phentolamine, prazosin, captopril, and nifedipine. Predominant arterial dilators include hydralazine and minoxidil. The most common venodilator is nitroglycerin. For further information about the dose, administration, and effects of these vasodilators the reader is referred to Shock in Chapter 6.
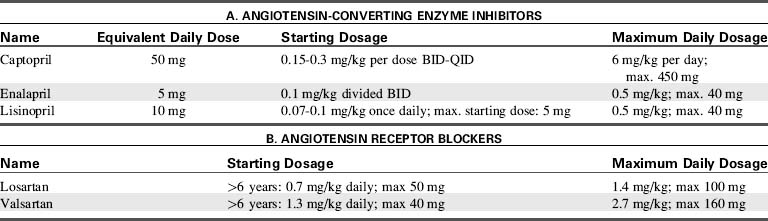
Angiotensin Converting Enzyme (ACE) Inhibitors
Angiotensin-converting-enzyme (ACE) inhibitors block the conversion of angiotensin I to angiotensin II, resulting in potent vasodilation. ACE inhibitors also block the breakdown of bradykinin, a powerful vasodilator, so it prolongs bradykinin action and augments afterload reduction (Fig. 8-14). In patients with left-to-right shunt lesions, reduction in systemic vascular resistance promotes blood flow into the systemic circulation rather than shunting to the pulmonary vascular bed.552
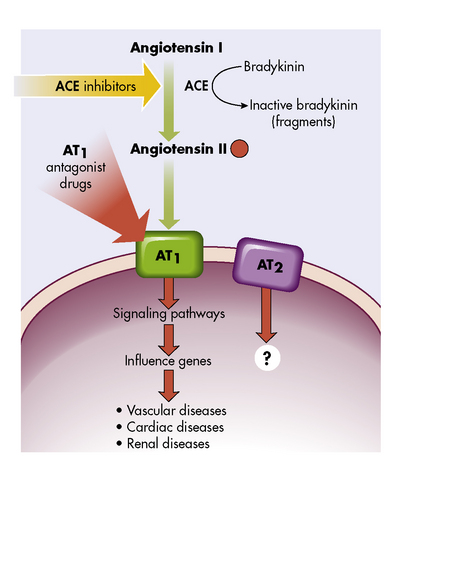
Fig. 8-14 Angiotensins and their receptors, AT1 and AT2. Blocking the angiotensin-converting enzyme (ACE) with ACE inhibitors decreases the amount of angiotensin II. Blocking the receptor AT1 with drugs (AT1 antagonists) blocks the attachment of angiotensin II to the cell, preventing the cellular effects and decreasing the vascular, cardiac, and renal effects.
(From McCance KL, Huether SE: Pathophysiology: The biologic basis for disease in adults and children, ed 6. St Louis, Mosby, 2009.)
ACE inhibitors also inhibit ventricular remodeling modulated by angiotensin II, thereby preventing the development of ventricular hypertrophy.697 Ultimately, these drugs have been shown to reduce mortality in adults with CHF and in children with dilated cardiomyopathy.530,829,850,861 Side effects include hypotension, cough, hyperkalemia, headache, dizziness, fatigue, nausea, and renal impairment.
Careful monitoring of hemodynamic status should be carried out in all patients with CHF when ACE inhibitors are started, because compromised cardiac output increases the risk of significant hypotension. Initial doses of ACE inhibitors are purposely low and then titrated upward to achieve maximal therapeutic effect.
Angiotensin Receptor Blockers
Because of other conversion pathways, ACE inhibitors do not completely prevent the formation of angiotensin II. Therefore, angiotensin receptor blockers (ARBs) are useful for patients who develop side effects from ACE inhibitors. Specifically, the cough associated with ACE inhibitors is caused by elevated bradykinin levels. Because ARBs do not affect bradykinin metabolism, ARBs are a logical choice when this particular side effect is observed (see Table 8-8). Although the benefits of ARBs in adults with heart failure are well described, there is limited information regarding use of ARBs in children with heart failure. Losartan has been used safely in children with Marfan syndrome113 and is the subject of a current multicenter trial.
Reduction in Intravascular Volume: Diuretic Therapy
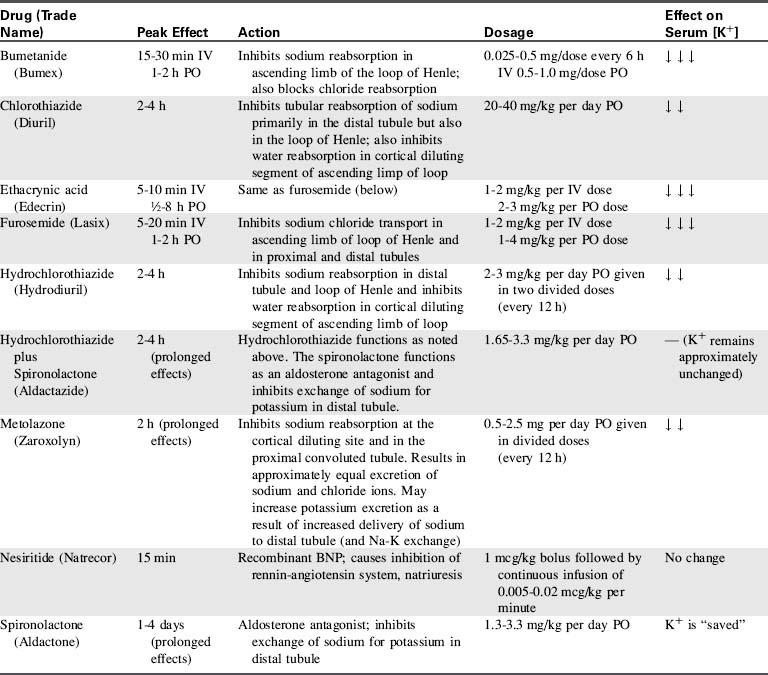
Limitation of fluid intake and improvement in systemic perfusion and blood distribution may increase renal perfusion sufficiently in the child with congestive heart failure to prompt a diuresis. However, administration of diuretics is often necessary to aid in the elimination of excess intravascular fluid (Table 8-9).
The most common diuretics used in children are the loop diuretics. These drugs block sodium and chloride reabsorption in the ascending limb of the loop of Henle, so that diuresis occurs. However, the increased sodium and chloride excretion may produce hyponatremia and hypochloremia. Potassium excretion in the distal nephron is also typically enhanced by these diuretics, so hypokalemia also may develop.
Hypochloremic or hypokalemic metabolic alkalosis is a significant potential complication of loop-diuretic therapy because either hypokalemia or hypochloremia will enhance renal hydrogen ion excretion and bicarbonate reabsorption. Significant metabolic alkalosis is treated by replacement of potassium and chloride losses. Hypochloremia must be treated effectively because it will prevent sodium excretion and compromise the effectiveness of diuretic therapy. If metabolic alkalosis persists, administration of ammonium chloride (75 mg/kg per day in divided doses) or acetazolamide (Diamox—a carbonic anhydrase inhibitor, administered 5 mg/kg PO or IV once daily) may be indicated.668
Electrolyte and acid-base balance must be monitored closely during diuretic therapy. Concurrent administration of a potassium-sparing diuretic may prevent hypokalemia.
Ototoxicity is a potential complication of these diuretics. In addition, the child's renal function should be monitored closely; these drugs usually are not administered if the blood urea nitrogen (BUN) and creatinine levels rise significantly.
Furosemide (Lasix)
Furosemide is the most popular loop diuretic. It acts rapidly when administered intravenously (within 5-10 min), and it usually results in significant diuresis. Generally an intravenous dose of 1 mg/kg is effective, although the dose may be doubled (or more) in children with severe heart failure who require chronic diuretic therapy. Furosemide also may be administered intramuscularly for rapid action, provided the child's systemic perfusion is adequate. Oral furosemide is administered in doses of 1 to 2 mg/kg when less acute diuresis is required (peak action: 1-2 h). This drug should not be administered to children who are allergic to sulfonamides.
Bumetanide (Bumex)
This diuretic is extremely potent at doses much smaller (approximately 0.025-0.5 mg/kg IV, PO, or IM every 12 hours) than those required for furosemide. It has a relatively rapid onset of action (approximately 10 minutes), and it has actions similar to furosemide at the loop of Henle. In addition, bumetanide may produce renal and peripheral vasodilation, so that the glomerular filtration rate increases temporarily. This drug also may cause ototoxicity, and diuretic effects can be blunted with concomitant administration of indomethacin. Cross-sensitivity to bumetanide may occur in patients with sulfonamide allergy.
Ethacrynic Acid (Edecrin)
This drug is similar in action to furosemide, with a rapid onset of action (1-2 mg/kg IV, slowly). However, it is prescribed less commonly for children because it is associated with a high incidence of gastrointestinal side effects. There is a significant incidence of ototoxicity with pediatric administration of this drug.
Thiazides and Chlorothiazide
Additional diuretics act at the cortical diluting segment, preventing sodium chloride (and water) reabsorption. These drugs include the thiazides, which may be utilized for less acute diuresis. Potassium loss and hypokalemia may result from these diuretics, but not to the degree seen with loop diuretics.
Chlorothiazide (Diuril) is the most popular of the pediatric thiazide diuretics. It is administered orally (20-40 mg/kg over 24 h in divided doses) and has a peak effect within 2 to 4 hours.
Metolazone (Zaroxolyn)
This drug also works by blocking sodium chloride and water reabsorption at the cortical diluting segments. It may be particularly effective when administered with furosemide. Relatively small doses (0.5-2.5 mg/kg per day, orally) are usually effective, with a rapid (2-h) onset of action. This drug may produce hepatic dysfunction.
Aldosterone Inhibitors
Aldosterone inhibition also produces diuresis. This prevents sodium reabsorption and inhibits potassium and hydrogen ion loss. These drugs also are known as “potassium-sparing” drugs because potassium loss is minimal; they should not be administered to patients with hyperkalemia. The effects of these drugs are gradual and do not peak for several days; for this reason it is important to anticipate the need for dosage adjustments. If the child is discharged during diuretic therapy, fluid and electrolyte balances should be well regulated before discharge.
Spironolactone (Aldactone) may be administered once daily (1.3-3.3 mg/kg per day orally) and will produce effective diuresis within 1 to 4 days. It is most effective when administered in conjunction with another diuretic that has a different site of renal action. It is often used to counteract the potassium loss brought on by loop diuretics. In addition to its diuretic properties, it is known to have a protective effect for adults with severe congestive heart failure, reducing mortality by up to 30%, and hospitalization rates by up to 35%.704
Hydrochlorothiazide and spironolactone (Aldactazide) is a combination of diuretics with differing sites of action that may provide extremely effective diuresis on a chronic basis. The potassium-sparing properties of the spironolactone component may prevent the development of hypokalemia. The dose is similar to spironolactone (1.65-3.3 mg/kg per day). Because this drug has a gradual onset and provides diuresis only several days after beginning therapy, it may be necessary to taper concurrent administration of other (short-acting) drugs.
Nesiritide (Natrecor)
Nesiritide is the recombinant form of BNP, the 32 amino acid polypeptide, released from ventricular myocardium. It produces vasodilation and diuresis. Because of reports of increased renal dysfunction and mortality in adult patients, its use is restricted to intravenous administration for patients in decompensated, severe heart failure with dyspnea at rest. It appears to be well-tolerated in pediatric patients, though hypotension and arrhythmias can occur.555
Nursing Implications
When the child receives diuretics it is important for the nurse to monitor the effectiveness of therapy and assess the child carefully for evidence of complications. The precise time of diuretic administration should be noted on the child's medication record and flow sheet, and the timing and quantity of the child's diuretic response also must be noted. It may be helpful to highlight the diuretic response in the nursing record so that it is identified easily. The physician or on-call care provider should be notified immediately if the child fails to respond to a previously effective dose because this may indicate worsening of heart failure (or low cardiac output) or development of renal failure.
Throughout diuretic therapy the child's fluid balance and hydration must be monitored closely. If the child's cardiovascular function is extremely unstable, diuresis may produce acute hypovolemia and a compromise in systemic perfusion. In addition, aggressive diuretic therapy may result in undesirable hemoconcentration.
When the child's congestive heart failure is severe, absorption of and response to oral diuretics may not be satisfactory. It may be necessary to switch to parenteral administration of the drugs until systemic perfusion and gastrointestinal function improve.
Electrolyte balance, particularly serum potassium and chloride ion concentrations, must be monitored during diuretic therapy. Because hypokalemia can potentiate digoxin toxicity, it should be prevented in these children. Potassium replacement of 1 to 4 mEq/kg per day should be sufficient to maintain serum potassium levels of 3.5 to 4.5 mEq/L, despite increased urinary potassium loss. As noted above, potassium-sparing drugs should not be administered in the presence of hyperkalemia, and potassium supplementation should be tapered accordingly when these drugs are added. Acid-base balance also should be monitored, and metabolic alkalosis should be prevented or treated.
If diuretics are administered late in the evening, diuresis may result in sleep disruption (either the child awakens to void, or awakens during diaper or bed-linen change) unless a urinary catheter is in place. Therefore, unless the child's heart failure is severe, some adjustment in scheduling of the evening diuretic dose should be made so that the child experiences diuresis before bedtime.
Parental teaching is required if the child is to receive diuretic therapy at home. Such information should include the technique of administration, potential effects of drug toxicity, flexibility (or lack of it) in administration schedule, and indications for contacting a healthcare provider. If supplementary potassium chloride administration is required the importance of the supplement must be emphasized.
Beta-Adrenergic Blockade
Activation of the sympathetic nervous system and release of catecholamines normally increases cardiac output in acutely ill patients. However, in the setting of chronic congestive heart failure, chronic adrenergic stimulation leads to alterations in myocardial excitation-contraction, gene expression, and eventually, ventricular remodeling and fibrosis.109,441,748 Beta-blockers prevent these maladaptive changes, while also having antiarrhythmic properties, inducing coronary vasodilation and slowing heart rate.121,792
Metoprolol
Metoprolol, a selective beta-1 receptor antagonist, has been shown to be very effective in adults with congestive heart failure.377,794 Metoprolol improves ejection fraction in children with dilated cardiomyopathy, but sometimes is not well tolerated.261
Carvedilol
Carvedilol is a nonselective beta-blocker and alpha-1 blocker. It is gaining acceptance as first line beta-blockade therapy in children with dilated cardiomyopathy. It, too, has been shown to be effective in adults with congestive heart failure,678 as well as in small case series of pediatric patients. A recent randomized controlled trial showed no difference between carvedilol and placebo, but the study may have been underpowered to show a difference.793 Subgroup analyses found that single ventricle patients of left ventricular morphology respond better than those of non-LV morphology. A distinguishing feature of carvedilol is its antioxidant activity, which limits both apoptosis (cell death) and cell proliferation (hypertrophy).32 Similar to ACE inhibitors, carvedilol can cause hypotension and so is started at low doses and cautiously titrated upward. Initial dosing is 0.05 to 0.1 mg/kg per dose given twice daily and is gradually adjusted upward to 0.2 to 0.4 mg/kg per dose twice/day, with a maximum dose of 25 mg.
Fluid Therapy and Nutrition
Accurate measurement and recording of the child's daily weight and intake and output is imperative when congestive heart failure is present. The child should be weighed on the same scale (or in the bed) at the same time of day (preferably by the same nurse) so that weight gain or loss can be evaluated. Significant weight changes (greater than 50 g/24 h in infants, 200 g/24 h in children, or 500 g/24 h in adolescents) should be verified and reported to the on-call provider.
Normal urine output in children should average 1.0 to 2.0 mL/kg body weight per hour if fluid intake is adequate. Sources of fluid loss that are not measured, such as excessive diaphoresis during fever or periods of increased respiratory rate, also should be considered. If a urinary catheter is not in place, all diapers and draw-sheets or pads must be weighed before and after use. One gram of weight increase resulting from urine is counted as 1 mL of urine output.
All sources of fluid intake and output must be totaled to evaluate the child's fluid status and the effectiveness of diuresis. If IV catheters are in place, total IV and oral fluid intake must be considered. Fluids required to flush IV or arterial catheters, to dilute medications, or to obtain cardiac output measurements are often sources of unrecognized fluid intake for the child.
During diuretic therapy the nurse must assess clinical signs of the child's fluid balance. The hypovolemic child characteristically demonstrates urine output of less than 0.5 mL/kg body weight per hour and has dry skin and mucous membranes, a flat or sunken fontanelle (in infants less than 18 months of age), and decreased or normal tearing; the child may demonstrate weight loss. The child's central venous or pulmonary artery wedge pressure is usually low when hypovolemia is present, although congestive heart failure or cardiac dysfunction may cause increased systemic and pulmonary venous pressures.
The child with hypervolemia usually demonstrates signs and symptoms of systemic and/or pulmonary venous congestion. The central venous and/or pulmonary artery wedge pressure is elevated, and the child usually gains weight. In addition, the child's mucous membranes are moist, and periorbital edema and hepatomegaly usually are noted. If an endotracheal tube is in place, it may be necessary to suction the child's airway more frequently as a result of copious pulmonary secretions.
Infants with congestive heart failure often do not tolerate oral feedings. Small, frequent feedings are usually more successful than infrequent, larger ones. If the infant is breathing faster than 60 times/min or is requiring nearly an hour to ingest 1 to 2 oz of formula, it may be better to provide tube feedings (see section, Altered Nutrition and Potential Gastrointestinal Complications in this chapter and section, Enteral and Parenteral Alimentation in Chapter 14) until the heart failure has improved; continued attempts at oral feedings may cause the infant to use more calories breathing and feeding than the child can possibly ingest. The child's daily caloric maintenance requirements should be calculated (Table 8-10), and the nurse should consult with the on-call provider or nutrition therapist if the child's caloric intake is inadequate.
Table 8-10 Calculation of Pediatric Daily Caloric Requirements
| Age | Daily Requirements* (kcal/kg) |
| High-risk neonate | 120-150 |
| Normal neonate | 100-120 |
| 1-2 y | 90-100 |
| 2-6 y | 80-90 |
| 7-9 y | 70-80 |
| 10-12 y | 50-60 |
* Ill children (with disease, surgery, fever, or pain) may require additional calories above the maintenance value, and comatose children may require fewer calories (because of lack of movement).
Restriction of fluid intake is often required if heart failure is severe (see Table 8-11 for the formulas necessary for the estimation of daily fluid requirements). If an infant is vigorously demanding more oral fluids than the amount allowed, the nurse should discuss with the healthcare team the possibility of increasing the oral intake and diuretic therapy proportionally.
Table 8-11 Formulas for Estimating Daily Maintenance Fluid and Electrolyte Requirements for Children
| Daily Requirements | Hourly Requirements | |
| Fluid Requirements Estimated from Weight* | ||
| Newborn (up to 72 hr after birth) | 60-100 mL/kg (newborns are born with excess body water) | – |
| Up to 10 kg | 100 mL/kg (can increase up to 150 mL/kg to provide caloric requirements if renal and cardiac function are adequate) | 4 mL/kg |
| 11-20 kg | 1000 mL for the first 10 kg + 50 mL/kg for each kg over 10 kg | 40 mL for first 10 kg + 2 mL/kg for each kg over 10 kg |
| 21-30 kg | 1500 mL for the first 20 kg + 25 mL/kg for each kg over 20 kg | 60 mL for first 20 kg + 1 mL/kg for each kg over 20 kg |
| Fluid Requirements Estimated from Body Surface Area (BSA) | ||
| Maintenance | 1500 mL/m2 BSA | – |
| Insensible losses | 300-400 mL/m2 BSA | – |
| Electrolytes | ||
| Sodium (Na) | 2-4 mEq/kg | – |
| Potassium (K) | 1-2 mEq/kg | – |
| Chloride (Cl) | 2-3 mEq/kg | – |
| Calcium (Ca) | 0.5-3 mEq/kg | – |
| Phosphorous (Phos) | 0.5-2 mmol/kg | – |
| Magnesium (Mg) | 0.4-0.9 mEq/kg | – |
* The “maintenance” fluids calculated by these formulas must only be used as a starting point to determine the fluid requirements of an individual patient. If intravascular volume is adequate, children with cardiac, pulmonary, or renal failure or increased intracranial pressure should generally receive less than these calculated “maintenance” fluids. The formula utilizing body weight generally results in a generous “maintenance” fluid total.
If congestive heart failure is severe or chronic, consult with the dietician when providing instructions for nutrition after discharge. Low-sodium infant formulas (such as Similac PM 60/40) are available, but their increased cost should be considered when deciding if the infant requires the formula for home care. The child with chronic congestive heart failure should avoid excessively salty foods, such as bacon, ham, sausage, potato chips, and some soft drinks are to be avoided if the child is requiring diuretic therapy. If a low-sodium diet is absolutely necessary for an older child, the child and the child's primary caretaker must be included in the dietary planning.
Comfort Measures and Thermoregulation
The child with congestive heart failure usually is most comfortable if placed in the semi-Fowler or sitting position so that abdominal contents can drop away from the diaphragm; this allows maximal diaphragm excursion and lung expansion. In addition, placement of a small linen roll under the child's shoulders extends the child's airway and may help the child to breathe with less difficulty.
The child's environment should be kept as quiet as possible to reduce stimulation and encourage rest. The nurse must decide when and how to consolidate nursing care so that the child is allowed periods of uninterrupted sleep yet excessive stimulation is avoided.
Premature infants and neonates with little subcutaneous fat have more difficulty maintaining body temperature when environmental temperature is low. In addition, the neonate's oxygen requirements are increased when the environmental temperature is excessively warm or cold. The “neutral thermal environment” is that environmental temperature at which the neonate maintains a rectal temperature of 37° C with the lowest oxygen consumption. In the critical care unit, a warm environmental temperature is maintained with overbed warmers.
The nurse is responsible for maintaining an appropriate environmental temperature while the infant is in the unit or during diagnostic tests or transport. When an overbed warmer is used, the infant's insensible fluid loss is increased by approximately 40% to 50%.
Transfusion Therapy to Treat Severe Anemia
If severe congestive heart failure is produced by anemia, improvement in arterial oxygen-carrying capacity through transfusion is usually necessary. This transfusion therapy improves arterial oxygen content, so that oxygen transport can be maintained without the need for an extremely high cardiac output. However, transfusion therapy must be performed with caution when anemia is profound or compensated because hypervolemia may develop and worsen symptoms of congestive heart failure.
Packed red blood cells usually are administered to children with chronic severe anemia at a rate of approximately 3 mL/kg per hour. This transfusion rate should be sufficiently gradual so that hypervolemia and worsening of congestive heart failure are avoided. Concurrent administration of a diuretic usually is required. If severe congestive heart failure is already present, a partial exchange transfusion will enable simultaneous removal of red cell-poor blood and replacement with packed red blood cells. Immune-mediated hemolytic anemia may not respond to transfusion therapy; steroid administration or splenectomy may be necessary for these patients (see Anemia, in Chapter 15).
Evaluation of Therapy
The nurse must be aware of the signs and symptoms of increasing heart failure, including continued tachycardia, increased peripheral vasoconstriction, decreased urine output, increased hepatomegaly, and increased respiratory rate and effort. Some of these symptoms may be noted easily by monitoring trends in the vital signs and record of intake and output. However, hepatomegaly and respiratory distress may be described less specifically. It is helpful to mark the edge of the liver at the beginning of the day (with another nurse or provider present to validate) so that changes in liver size can be recognized easily throughout the day. Location and severity of any existing retractions always should be recorded with the vital signs so that an increase in respiratory distress will be apparent to even a new nurse caring for the child.
Care of the child with congestive heart failure requires careful monitoring of clinical condition and careful titration of therapy to maximize therapeutic effects and minimize side effects. Advanced concepts in management of congestive heart failure are included in Box 8-10.
Box 8-10 Advanced Concepts: Resynchronization Therapy for the Treatment of CHF
Biventricular pacing and cardiac resynchronization therapy have been shown to be effective in adults with heart failure and prolonged QRS duration of left bundle branch block morphology.963 The purpose is to restore a more synchronized, “efficient” cardiac contraction in those patients in whom interruption of the ventricular conduction system has caused asynchronous activation and uncoordinated beating of the heart. In pediatric patients, cardiac resynchronization can be successful in decreasing QRS duration, and improving ejection fraction, and ultimately improving NYHA heart failure classification.
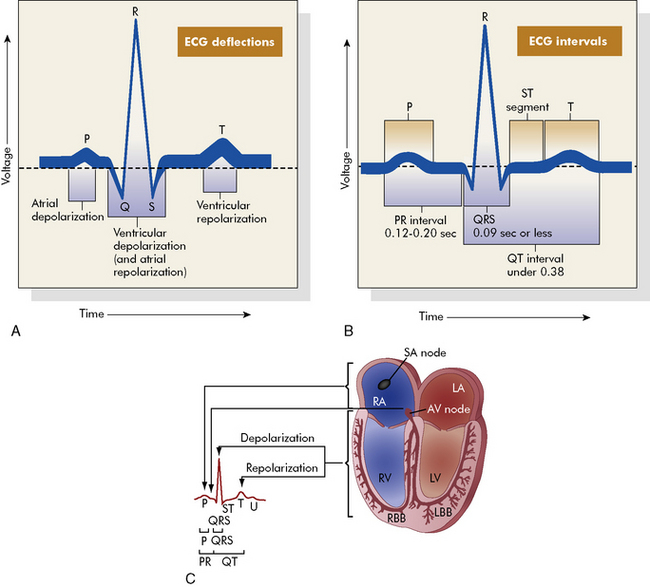
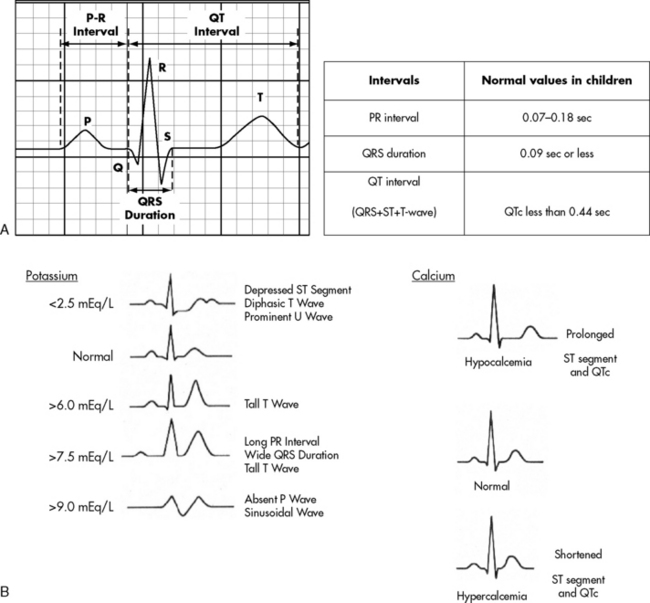
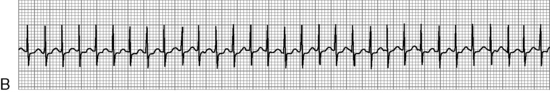
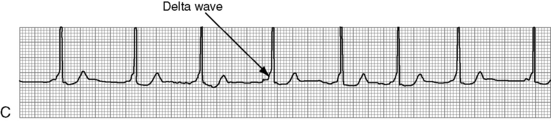
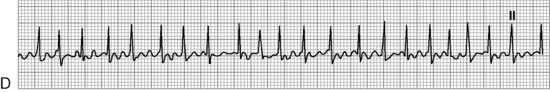
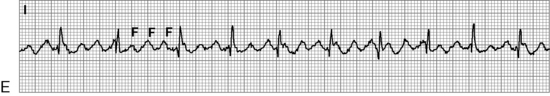
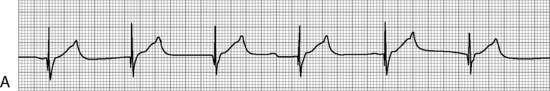
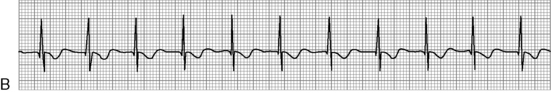
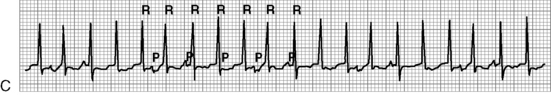
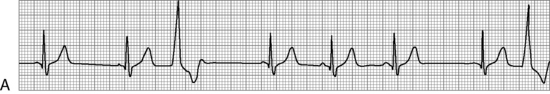
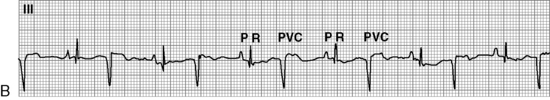
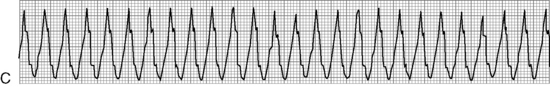
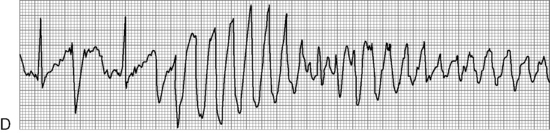
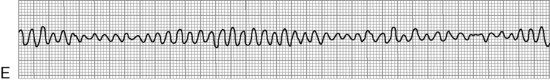
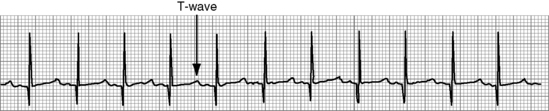
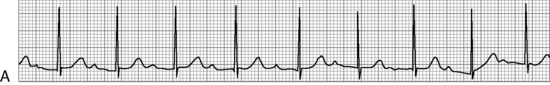
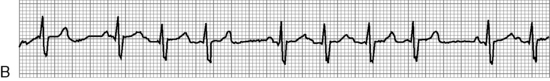
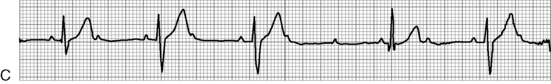
 . B, ECG changes caused by electrolyte imbalance.
. B, ECG changes caused by electrolyte imbalance.