Specific diseases
Head Trauma
Etiology
CNS injury is the leading cause of death in children. In children younger than 2 years, serious head injuries (traumatic brain injuries [TBIs]) are commonly intentional (also called inflicted or non-accidental trauma). Children older than 3 years of age sustain head injuries secondary to falls or to motor vehicle, all terrain vehicle, bicycle, and pedestrian collisions. In high school-aged adolescents, football is the team sport most commonly linked with head injury.
Each year 1.5 million head injuries occur in the United States. Of this number approximately 300,000 children are hospitalized with head trauma. Nearly 90% of injury-related deaths are associated with head trauma. Males are twice as likely as females to sustain head trauma. Severe TBI or associated cardiorespiratory arrest can cause death within a few hours of injury.20
Pathophysiology
The rigid cranium and the CSF cushion can protect the child's brain from injury during minor trauma. However, if distortion of the skull, shear injury, actual tissue damage, intracranial hemorrhage, or cerebral edema develop, the injury is likely to be complicated by increased ICP. Therefore, the child with head trauma requires assessment and treatment of the primary (or direct) injury, as well as careful assessment and treatment of secondary complications.
The types of cerebral injuries occurring with head trauma include concussions, contusions, skull fractures, vascular injuries, diffuse axonal injuries, penetrating injuries, and cerebral edema. Each lesion is summarized below.
Concussion
A concussion is associated with mild to moderate cerebral injury. It results from a blow to the head or a shearing rotational injury of the brain within the skull that produces no structural brain damage. The concussion is more likely to occur if the head moves freely after impact; an acceleration-deceleration injury produces shearing stresses on the brainstem and results in injury to the reticular activating system. Many patients with concussion experience loss of consciousness for a few seconds or several hours, although some may never lose consciousness.
After the impact, the CSF pressure rises transiently, and electroencephalographic evidence of slow brain wave activity has been documented.62 Infants may exhibit less specific signs than those of older children; the infant may develop seizures, nausea, emesis, and lethargy, but usually no loss of consciousness. Older children may complain of headache, dizziness, fatigue, and amnesia. Generally, symptoms of a concussion will resolve in approximately 1 week. However, some symptoms may persist for up to 1 year. The diagnosis of concussion is generally made based on a history of a temporary loss of consciousness with no other findings.50,78
Contusion
A cerebral contusion is a localized brain injury that consists of bruising, hemorrhage, and cerebral edema. The hemorrhage may be epidural, subdural, or subarachnoid (see the section on vascular injuries that follows), and it can produce an increase in ICP or loss of consciousness.
The injury can occur directly beneath the site of impact (the coup injury) or on the side of the brain opposite the impact (the contrecoup injury). The contrecoup injury is thought to occur as the brain strikes the skull on the side of the head opposite the initial impact. The severity of the cerebral contusion is determined by the amount of direct tissue injury, bleeding, and edema that result.50
Posttraumatic seizures occur in approximately 10% of children with cerebral contusions. Prophylactic anticonvulsant drugs are of little benefit in preventing seizures. These delayed effects of head injury can be classified as early or late seizures. Early seizures typically occur within the first week after injury, usually seen within the first 24 hours. Factors associated with increased risk include age <5 years, prolonged course of posttraumatic amnesia, intracranial (particularly intraventricular) hemorrhage, and compound depressed skull fractures.
Late seizures develop in approximately 5% of children with TBI, typically developing between the first week and 1 year after injury. Risk factors include early seizures, intracranial hemorrhage, and compound depressed skull fractures.78
Skull Fractures
A skull fracture is a break in the continuity of the cranial bones that may or may not be associated with displacement of the bone fragments. Skull fractures are present in approximately one fourth of all patients hospitalized with head injury and can be further classified as simple (closed injury) or compound (open injury). Compound fractures are frequently associated with dural tears; these increase the risk of abscess formation and meningitis.78 The fracture itself may be benign, but is often associated with injury to the underlying meninges or vasculature.
Approximately three fourths of all skull fractures in children are simple or linear skull fractures. In this form of skull fracture, the bone fragments remain approximated and the dura mater is not penetrated.
A depressed skull fracture is present when one or more bone fragments are indented below the normal contour or table of the cranium. As a result, the skull is indented, and the brain tissue below the fracture is injured. A hematoma may cover the area of injury, and a cerebral contusion may be present below the fracture. The dura usually is not penetrated when a depressed skull fracture is present.
A compound skull fracture exists when a scalp laceration and depressed skull fracture are present, allowing direct communication from the scalp through the skull and into the cranium. The dura often is penetrated when a compound fracture is present, and the skull fragment can be displaced into the brain tissue.
Basilar skull fractures are those that involve a break in the posteroinferior portion of the skull. These fractures do not typically produce cerebral tissue damage, but they frequently produce dural tears. As a result, basilar skull fractures are commonly associated with leak of CSF. When a dural tear and CSF leak are present, contamination of the CSF by ascending upper respiratory tract infection is also possible and can result in the development of meningitis.
Basilar skull fractures can occur over the paranasal sinuses of the frontal bone, over the temporal bone, or over the entrance of the internal carotid artery into the skull. A fracture over the internal carotid artery can result in hemorrhage, aneurysm, or a fistula.62,78
Petrous bone fractures can involve injury of cranial nerves V, VII, and VIII and sometimes are associated with injury to the nerve, cochlea, or ossicles. Cranial nerve involvement can be acute or delayed. Treatment may involve the administration of steroids, surgical decompression, or both. Acute presentations are usually associated with poor outcome, whereas children with delayed presentation usually recover. Neuronal deafness is often permanent, but conductive deafness usually improves gradually.78
Vascular Injuries and Hemorrhage
Vascular injuries resulting from head trauma can produce epidural hematoma, subdural hematoma, or a subarachnoid hemorrhage. Each of these vascular injuries is discussed separately.
An epidural hematoma (EDH) results from hemorrhage into the extradural space secondary to a tear in the middle meningeal artery or dural veins. Arterial bleeding is the most common source of bleeding and is responsible for 85% of EDHs; the remaining 15% of EDHs arise from the middle meningeal vein or dural sinus. An EDH usually results from direct trauma in the region of the temporal bone and approximately half of patients with EDH have an associated with a skull fracture. Because the hematoma often develops from the artery, blood can rapidly accumulate between the skull and the dura. For this reason EDHs are considered neurosurgical emergencies.
The child with EDH may demonstrate a lucid period that can last minutes to several days after the head injury. As the hematoma expands it compresses the temporal lobe, leading to an acute rise in ICP. The child may abruptly lose consciousness and demonstrate ipsilateral pupil dilation and contralateral hemiparesis. Left untreated, uncal herniation and death will result. Because an EDH is usually detected on an initial CT scan, the classic presentation is somewhat uncommon. Significant mortality and morbidity results if recognition or treatment is delayed.50,125
A subdural hematoma (SDH) is a collection of blood between the dura and the arachnoid membranes, resulting from tearing of bridging veins that drain the cortex.125 An SDH is usually associated with some damage to the underlying brain tissue from the initial injury.
An SDH can be classified as acute, subacute, or chronic. An acute subdural hematoma usually develops after severe head injury or cerebral laceration and results in accumulation of blood within hours of the injury. A subacute subdural hematoma occurs early after a less severe cerebral contusion and usually produces a rise in ICP that prevents the patient from regaining consciousness after the head injury.62 A chronic subdural hematoma develops weeks or months after a relatively minor head injury. The injury typically produces a venous tear, and blood slowly accumulates in the subdural space. Subdural hematomas are often present bilaterally, and they are frequently present in victims of inflicted trauma under the age of 2 years (see Chapter 19).
Children with subdural hematomas will often exhibit symptoms of increased ICP. These symptoms are secondary to direct injury to the brain tissue, often associated with vasogenic cerebral edema. Many patients who sustain subdural hematomas develop early or late seizures as a result of the injury.50,83
A subarachnoid hemorrhage (SAH) results from a severe head injury. Hemorrhage occurs when shear forces producing the massive head injury tear the subarachnoid vessels. Children with an SAH can demonstrate seizures or rapid development of increased ICP. Because subarachnoid hemorrhages frequently are present in children with inflicted trauma, the presence of an SAH warrants careful examination for evidence of other injuries, including healed fractures or retinal hemorrhages (see Inflicted Injuries in Chapter 19).
Diffuse Axonal Injury
Diffuse axonal injury results from an acceleration-deceleration brain injury. This movement creates pressure that begins at the brain surface, and as greater force is transmitted it can extend deep into the brain. This pressure creates a shearing force that tears the axons (long projections of the cell bodies that carry nerve impulses). Traumatic diffuse axonal injury (TDAI) appears to be age independent and may be devastating to the developing brain. It appears to be more widespread in the victim of inflicted trauma.
Traumatic diffuse axonal injury can be further categorized into three subsets: grade 1, microscopic damage to the axons without gross hemorrhage; grade 2, microscopic damage to axons with hemorrhage in the corpus callosum; and grade 3, microscopic damage to axons with hemorrhage in the dorsal aspect of the brainstem. Hemorrhages associated with shearing are often initially microscopic (measuring approximately 1 mm); they can be described as streak or punctuate hemorrhages. However, as the bleeding continues over several days the size can easily reach several centimeters. Hemorrhage is rarely seen in infants or younger children because the cerebral vasculature is relatively elastic. The result of this elasticity is that vessels do not appear to tear as easily as the axons and surrounding tissue. Treatment is largely supportive with intensive long-term rehabilitation.30,122
Penetrating Injuries
A small percentage of pediatric TBI is related to penetrating head trauma. Although injuries from motor vehicle crashes and falls are still leading causes of TBI and death in children, there are other common mechanisms of TBI. These mechanisms include but are not limited to bullets, pellets, knives, pencils, sticks, coat hangers, glass, and objects propelled from lawnmowers.
Initial management for penetrating head trauma is the same as for any TBI: assess and support airway, breathing, and circulation. Once initial vital functions are supported, a CT scan may be obtained to document the involved areas and assist the surgeon in making operative decisions. Any object protruding from the skull should not be removed before surgery—support with gauze and tape will keep it in place. Care should be taken to avoid dislodging the object.
Management usually requires a multidisciplinary approach. Depending on the area of involvement, the surgical team may be composed of specialists in neurosurgery, plastic surgery, maxillofacial surgery, otolaryngology, ophthalmology, and vascular surgery. A medical examiner may be helpful to determine patterns of injury.
Postsurgical management is dependent on the area of injury. Penetrating trauma is often associated with skull fractures, pneumocephalus, SDH, intraventricular hemorrhage, and intraparenchymal hemorrhage. These associated injuries can produce traumatic seizures, cerebral edema, increased ICP, meningitis, or intracranial abscess formation.
BB guns are generally thought to fire with low velocity that will likely cause only superficial injury. As a result, children with these injuries may be discharged from the emergency department without evaluation for intracranial injury. Because serious intracranial injuries (including intraparenchymal hemorrhage) have been documented hours after BB gun injury, it is reasonable to admit children with these injuries for short-term observation.69,74,92,116
Inflicted (Intentional/Non-accidental) Injuries and Shaken Baby Syndrome
Shaking injury is the most common form of inflicted (intentional/non-accidental) brain injury in infants up to 12 months of age. The infant's head is large in proportion to the body, and the muscles and ligaments in the neck are immature and weak. Shaken baby syndrome results from a violent whiplash acceleration-deceleration of the head that causes significant brain injury. In some instances there is additional impact injury from striking the infant's head on a hard surface. Brain injuries include subdural or subarachnoid hemorrhages or chronic subdural fluid collections (see Vascular Injuries and Hemorrhage).
Infants with inflicted trauma (including shaken baby syndrome) may present with what appears to be new onset seizures, sudden infant death syndrome, or an apparent life-threatening event (ALTE). The provider should be suspicious of inflicted injuries if the history given by the caretaker does not match the presentation or if there are suspicious findings such as retinal hemorrhages, bilateral chronic subdural hematomas, skull fractures, and significant neurologic injury without external signs of trauma. Many victims die from uncontrollable intracranial hypertension, and survivors may be neurologically devastated.50,122
Cerebral Edema
Cerebral edema is an immediate consequence of TBI, intracerebral hemorrhage, or other brain insult. This increase in brain volume can develop up to 72 hours after the injury. Cerebral edema can be categorized into four subsets: cytotoxic, vasogenic, osmotic and interstitial.83,108 Children may develop any or a combination of these types of cerebral edema following direct cerebral or vascular injury or secondary hypoxia. The pathophysiology of cerebral edema is reviewed in detail in the Increased Intracranial Pressure section earlier in this chapter.
Clinical Signs and Symptoms
The child with head injury requires frequent neurologic examinations and monitoring of neurologic function and responsiveness. Recently, evaluation of cerebral biomarkers has been shown to be helpful in the identification of head injury.
This section describes the use of serum biomarkers and the clinical signs and symptoms of each of the major forms of head injury. A review of the initial assessment of children with head trauma is included in the following section, Management.
Cerebral Biomarkers
Cerebral biomarkers are proteins that are normally present in low concentrations in the serum and cerebral spinal fluid (CSF), but concentrations increase following specific types of brain injury. Evaluation of these biomarkers can be helpful in establishing the diagnosis and prognosis of pediatric traumatic brain injury (TBI), hypoxic-ischemic encephalopathy (HIE), and TBI from inflicted (intentional/non-accidental) trauma.72b
Three cerebral biomarkers shown to be useful in the care of pediatric patients with CNS insults are: neuron specific enolase (NSE), S100-Beta (S100B), and myelin basic protein (MBP).21b,72a,110a NSE is a marker of neuronal death, S100B is a marker of astrocyte injury or death and MBP marks axonal injury.72a
NSE is only released during cell destruction, and shows different patterns in children with inflicted TBI and HIE versus children with non-inflicted TBI.72a The NSE serum concentration peaks early (within 12 hours) after TBI but the rise in serum concentration is delayed (3-5 days) in HIE and inflicted trauma. Bell & Kochanek21b suggest that this delay in the rise in NSE could be related to additional apoptosis that occurs following HIE and inflicted trauma. Higher concentrations of NSE are associated with worse outcome following TBI in children.72a
MBP is elevated in patients with TBI and inflicted trauma. HIE does not appear to produce elevated levels of MBP.21b This is likely related to the fact that the white matter is resistant to ischemic injury while there is greater axonal injury seen in TBI.21b,110a
S100Beta (S100B) is a calcium-binding protein molecule that is primarily found in the cytoplasm of cells in the CNS, although it may be present in some non-CNS cells. S100B crosses into the systemic circulation when there is a breech in the blood-brain barrier. It is metabolized by the renal system and excreted in the urine.110a The sensitivity and specificity of S100B in separating inflicted versus non-inflected TBI and in differentiating severe from mild TBI has varied in the relatively small pediatric case series published to date.72a In a study by Bechtel et al,21a S100B levels were higher in pediatric patients with than without closed head injury (CHI) if the child with CHI presented to the emergency department (ED) within 6 hours of the injury. Although there was a relationship between elevated S100B levels and CHI, S100B may not be the most sensitive and specific screening marker in CHI because the half-life of the protein is approximately 6 hours, an elevated S100B may also be associated with long bone fractures (so it is less useful in patients with multiple trauma), and non-Caucasian children normally have higher levels of S100B proteins than Caucasian children.21a, 72a
Biomarkers can be helpful in screening patients with TBI. In combination with the clinical examination, these markers may be useful in establishing the prognosis of head injuries. However, further studies in pediatric patients are needed. If these markers are monitored, the provider must be familiar with the laboratory normal ranges for the biomarker concentrations, and potential causes of elevations in these biomarkers.110a Assays for cerebral biomarkers may not be available in all institutions.
Concussion
The patient who sustains a concussion loses consciousness for a variable period of time. This loss of consciousness typically is associated with a brief slowing of respirations (possibly accompanied by apnea), bradycardia, and hypotension. All reflexes should be present after the injury, but some (e.g., corneal or gag reflex) may initially be depressed. The patient may demonstrate reduced response to painful stimuli.83
Upon waking, the patient slowly becomes oriented to surroundings (over a period of hours or days) and is gradually able to respond to questions and follow commands. Patients often experience delayed effects of head injury and may suffer temporary memory loss, called posttraumatic amnesia. After a concussion, there is no evidence of further neurologic injury unless a second impact occurs within a few days or weeks (see Contusion). Patients occasionally complain of headache, malaise, vertigo, anxiety, or fatigue for several days or weeks following a concussion. These symptoms, known as postconcussion syndrome, may require several weeks to resolve. Injury to the vestibular region may be responsible for symptoms of dizziness and vertigo.78,125
Contusion
The clinical signs and symptoms that result from a cerebral contusion are dependent on the extent of the cranial injury, the volume of bleeding present, and the amount of cerebral edema that develops. The associated hemorrhage may be epidural, subdural, or subarachnoid (see Vascular Injuries and Hemorrhage). The resultant cerebral edema can produce increased ICP (see Increased Intracranial Pressure).
Patients who suffer a cerebral contusion may or may not lose consciousness, and they may demonstrate mild motor and sensory weakness or coma. Children may demonstrate a vacant stare or puzzled expression, slurred or incoherent speech, memory deficits, or difficulty focusing, and they may be slow to answer questions or follow commands. Because at least 10% of children with cerebral contusion develop posttraumatic seizures, most are hospitalized for observation after the injury.50,78
Second impact syndrome is a rare condition described primarily in athletes who sustain a second head injury after a recent (generally within 7-10 days) concussion. During the days after the concussion, the brain is particularly vulnerable, and repeated injury can cause malignant cerebral edema, hyperemia, intracranial hypertension, and herniation50 that often occur within minutes and are refractory to most therapy. The mortality of second impact is extremely high. The classic presentation of second impact injury is the athlete who walks off the field without assistance and then abruptly deteriorates into a coma within a few minutes. Attempts to prevent these second impact injuries have led to playing restrictions for athletes who sustain concussions.
Skull Fractures
The clinical signs and symptoms associated with any skull fracture will depend on the location of the fracture and on the extent of the underlying cranial injury. Most skull fractures are diagnosed by radiographic examination rather than by clinical examination, because the vast majority of skull fractures are linear (i.e., the bone fragments remain approximated).
A basilar skull fracture is often not detectable on a radiograph, unless a blood-air level develops in the sphenoid sinus. A CT scan may be required to identify a basilar skull fracture. Approximately 40% of patients with EDH have no identified fractures.50
Depressed or compound skull fractures should be suspected whenever the contour of the patient's head is altered or whenever an obvious indentation in the skull is observed or palpated. If a depressed skull fracture is located over the sagittal or lateral sinus, profuse bleeding can develop from injury to these venous channels, and hypovolemic shock can result.
If a basilar skull fracture is present, the patient can develop a CSF leak from the floor of the brain into the nose or ears. Although the CSF leak itself is not harmful, it indicates that communication is present between the upper respiratory tract and the subarachnoid space; this communication increases risk of CNS infection either in the form of meningitis or an intracranial abscess.
Detection of a CSF leak is extremely difficult; various bedside techniques have been described, but none are reliable and all can provide false-positive results. For example, if a yellow halo forms around serosanguineous drainage that has been collected from the nose or ear, the halo is thought to be produced by CSF; however, plasma frequently can produce a similar halo. Nasal or ear drainage can be tested for glucose, because theoretically the presence of glucose in the drainage indicates that CSF is present; however, nasal drainage also can contain glucose. If confirmation of CSF drainage is desired, a sample of the fluid can be sent to the laboratory for a beta-2 transferrin (a protein found only in CSF and perilymph fluid) level. This beta-2 transferrin has >94% sensitivity and 98% specificity, and it has become the gold standard test for detection of CSF leak.57
In general, a CSF leak may be present in any child with a basilar skull fracture, so the child with such a skull fracture usually is admitted to the hospital for observation. Signs of deterioration in clinical status or signs of CNS infection (e.g., fever, irritability, nuchal rigidity, leukocytosis) must be reported to an on-call provider immediately. Other signs associated with basilar skull fracture include the presence of ecchymotic lesions over the mastoid (Battle's sign) or around the eyes (raccoon sign), bleeding at the tympanic membrane, and palsies of the first, seventh, and eighth cranial nerves (see Table 11-2).50,78
Vascular Injuries and Hemorrhage
The classic presentation of an EDH includes brief loss of consciousness after an injury, regaining consciousness with a lucid interval lasting minutes to several hours, and then abruptly developing a headache, obtundation, ipsilateral pupil dilation, and contralateral hemiparesis. If EDH goes unrecognized and untreated, the child will experience decerebrate posturing, signs of increased ICP, and death. (see the Increased Intracranial Pressure section of this chapter).35,50 However, fewer than one third of the children sustaining an epidural hematoma lose consciousness and do not awaken spontaneously, and 60% of children with EDH never lose consciousness. Thus, the classic presentation of an EDH occurs infrequently.50
The healthcare team should suspect an EDH in any child who develops headache, a rapid deterioration in level of consciousness, a decrease in the hematocrit by 10%, and unilateral pupil dilation. The pupil usually dilates on the same side (ipsilateral) as the injury. However, because an EDH can result from a contrecoup injury, the pupil contralateral to the initial side of impact may dilate. A fever also may be present.74 As the child's symptoms progress, decerebrate posturing may be observed, and approximately half of involved children develop hemiparesis (usually on the side contralateral to that of pupil dilation). If immediate surgical decompression of the hematoma is not provided, the child will develop bilateral pupil dilation, respiratory depression, bradycardia, apnea, and death from increased ICP.50,125
The best diagnostic test to confirm the presence of an EDH is a CT scan. Because the hematoma often is located directly under the skull fracture, plain skull radiographs may be adequate to localize the hematoma in a severely ill child. Most EDHs have the classic biconvex appearance on CT. A small percentage of EDHs have a crescent shape, and resemble an SDH. However, the edges of an EDH are usually sharp and the EDH has uniform density. Because the hematoma develops rapidly, there is almost always mass effect apparent on the CT scan. In rare cases the EDH may be isodense with the brain tissue and IV contrast must be used to identify the hemorrhage.50
The patient with an acute SDH may demonstrate bilateral hematomas and evidence of diffuse neurologic injury. Because acute subdural hematomas are associated with underlying parenchymal injury, they are generally considered more lethal than EDHs. Approximately two thirds of all children with SDHs lose consciousness immediately after the cranial trauma. Frequently, the child demonstrates focal signs of injury, such as unilateral pupil dilation, focal seizures, or hemiparesis. Because most of these patients sustain additional cerebral injuries, including cerebral lacerations, contusions, and intracerebral hematomas, intracranial hypertension can develop rapidly and progress to severe levels.
Diagnosis of an acute or chronic SDH can be confirmed with a CT scan or angiography. The acute SDH is a crescent-shaped mass, and edema is usually present. The SDH is usually more diffuse than an EDH, the borders are less sharp, and the EDH may be less isodense because the blood mixes with CSF. Radiographic studies are extremely important because they can help determine the need for surgery.50
The patient with a chronic SDH may develop a minor headache, confusion, and a progressive decrease in level of consciousness weeks or months after a relatively minor head injury. Because the hematoma is present for a long period of time, papilledema may be present. The ipsilateral pupil will be large with a sluggish response to light. Hemiparesis and focal seizures can develop.62 Treatment of chronic subdural hematomas includes seizure control (anticonvulsants), correction of coagulopathies, and surgical evacuation.50
The patient with an SAH may experience a sudden onset of a severe headache (the most common symptom and present in nearly all cases), emesis, syncope, meningismus, and photophobia. These symptoms are associated with a rapid rise in ICP. Left untreated, the child will progress to coma and possibly death. Although spontaneous SAHs can arise from a variety of sources, including aneurysms, coagulation disorders, and small artery ruptures, trauma remains the leading cause of an SAH in children. CT scans performed within 48 hours of a hemorrhage will detect the vast majority of subarachnoid hemorrhages. The blood will appear as a white, hyperdense area in the subarachnoid spaces (see Fig. 11–12, B and C).
Complications of SAHs include rebleeding, hydrocephalus, hyponatremia, hypovolemia, arrhythmias, embolism, and seizures. Approximately half of patients with acute hydrocephalus have spontaneous resolution, whereas the remainder if symptomatic may require a ventriculostomy or ventricular shunt. If a ventriculostomy is placed, the recommended range of ICP for drainage is 15 to 25 mm Hg (20-34 cm H2O). Drainage should be gradual to avoid rapid decreases in the ICP that may increase the risk of rebleeding.50
Cerebral Edema
Signs of cerebral edema are those associated with increased ICP (see the Increased Intracranial Pressure section earlier in this chapter).
The “Talk and Die” Phenomenon
A small number of children who sustain closed head injury initially are awake and lucid, but then suddenly deteriorate and may develop cerebral herniation and death. This clinical picture may develop in the absence of any mass lesion (such as an EDH) or any initial symptoms of severe injury. This phenomenon has been referred to as the talk and die injury or the pediatric concussion syndrome.
Most children who “talk and die” sustain a potentially significant head injury; they usually are involved in a motor vehicle crash or a serious fall. Initially the child is alert, with a high GCS score (9 or more). Within 48 hours of the injury there is a sudden deterioration in the child's neurologic status. The child is restless, irritable, or difficult to arouse, with pupil dilation with decreased response to light. Seizures also may be associated with the deterioration.52
Postmortem examination reveals the presence of cerebral hyperemia, often associated with multiple cerebral contusions. The ultimate causes of death are cerebral herniation and ischemia. This phenomenon is thought to be related to the rapid development of cerebral swelling following significant head injury. It is important to note that the severity of the head injury may not be discernable from clinical examination, but usually will be apparent on CT scan.
Management
All children with moderate or severe head injury require critical care. Children with mild head injury usually are admitted to the critical or intermediate care unit for skilled continuous nursing observation. Because a small number of patients with apparently minor head injuries may deteriorate acutely as the result of rapid brain swelling and increased ICP, a CT scan is indicated when the child has a history or mechanism of injury consistent with a serious head trauma (e.g., a fall from a significant height, unrestrained occupant in a severe motor vehicle crash). This scan should be performed even if the child initially appears alert and oriented.50
All children with serious head trauma are presumed to have a spinal cord injury until it has been ruled out. The cervical spine should be immobilized, and the child should be log rolled whenever turning is necessary. Until definitive radiographic studies and clinical examination can be performed, the nurse must frequently and carefully evaluate the patient's movement and sensation in all four extremities. The development of a progressive neurologic deficit in the patient with a spinal cord injury is a neurosurgical emergency that will require urgent intervention (see Spinal Cord Injury). When assessing movement and sensation, it is important to verify that movement is voluntary and intentional; the reflex withdrawal of an extremity may occur in response to stimulation of a spinal reflex arc despite complete spinal cord transection.
Support of Cardiopulmonary Function
When the child with head trauma is admitted to the critical care unit, the first priority is establishing and maintaining adequate airway, oxygenation, ventilation, and systemic perfusion. Because increased ICP can cause decreased airway protective mechanisms, and because apnea and hypoventilation can cause hypoxia and hypercapnia that will contribute to increased CBF and increased ICP, establishing an effective airway, oxygenation, and ventilation are essential. If there is any doubt about the child's ability to maintain a patent airway or breathe spontaneously, intubation is performed and mechanical ventilation is provided.
If a cervical spine injury has been ruled out, the child's should be placed in a “sniffing” position to prevent upper airway obstruction. If the child is severely injured, intubation is performed and mechanical ventilation provided. Rapid sequence intubation is typically performed to prevent cough and gag and to reduce the risk of vomiting during the intubation (see Chapter 9). A nasogastric tube is inserted to decompress the stomach and prevent vomiting; if a basilar skull fracture or facial fractures are suspected the gastric tube is placed orally.
The entire healthcare team will carefully assess the child's systemic perfusion. Extremities should be warm with pink nail beds and brisk capillary refill. Peripheral pulses should be strong, urine output should average 1-2 mL/kg per hour, and the child's blood pressure should be appropriate for age. (The systolic blood pressure should average 90 mm Hg + twice the child's age for children 1-10 years old.) Hypotension (a systolic blood pressure less than 70 mm Hg + twice the child's age in years) is rarely caused by head injury; if it is present, hypovolemic shock is the likely cause. Tachycardia may indicate the presence of hemorrhage or the development of increased ICP; it also may indicate that the child is frightened, agitated, or in pain.
Hypovolemic shock is treated immediately (see Chapter 6). As soon as possible, at least one large-bore central venous catheter is inserted to allow measurement of the central venous pressure and administration of blood products, colloids, medications, or IV fluids.
Because head trauma frequently is associated with the injury of other major organs, the team will assess for signs of abdominal trauma, hemothorax, flail chest, and pneumothorax (see Chapter 19). Cerebrovascular injury alone usually will not account for a significant blood loss unless a massive intracranial hemorrhage and increased ICP develop. Severe cerebral injury will result in the release of tissue thromboplastin that may produce disseminated intravascular coagulation,61 which is treated with blood products (see Chapter 15).
The healthcare team will estimate the child's circulating blood volume (approximately 75 mL/kg in infants and children and 70 mL/kg in adolescents), and evaluate blood losses in light of that total estimate. Acute symptomatic traumatic blood loss is replaced immediately. Nurses will total all blood lost or obtained for laboratory analysis, and replacement is considered when blood loss totals 5% to 7% of the child's total circulating blood volume.
Assessment of Neurologic Function
Once adequate cardiopulmonary function has been established, the nurse should perform a careful neurologic assessment. This assessment includes an evaluation of the systemic perfusion and vital signs, including blood pressure, respiratory function, and level of consciousness (including the ability to follow commands, pupil size and response to light) and a careful assessment of motor activity and reflexes (see Box 11-2).
The healthy child will be awake, alert, and frightened in the hospital. The comatose child demonstrates no verbal response to external stimuli. To evaluate neurologic function and responsiveness, a standard rating scale, such as the GCS, should be applied in a consistent fashion by everyone who examines the child with head injury (see Table 11-6).
The child's pupil size and constrictive response to light should be evaluated frequently. The pupils normally are equal in size and constrict briskly and equally to light. Consensual pupil constriction also should be present (i.e., the right pupil constricts when light is shined into the left eye). High doses of morphine sulfate can cause pupil constriction, and high doses of atropine and atrovent can produce pupil dilation. The presence of fixed and dilated pupils during the initial evaluation of the child with a head injury often is regarded as a poor prognostic sign; however, children who have fixed pupils after a head injury have a better rate of recovery than do adults.26
Although the classic Cushing's triad (bradycardia, elevation in systolic blood pressure with widening of pulse pressure, and irregular respirations, including apnea) indicates the development of a severe increase in ICP, children rarely demonstrate such classic findings unless cerebral herniation is occurring. Often, the child exhibits tachycardia and hypertension. The widening of the pulse pressure and irregular respirations are typically late signs of deterioration in neurologic status.
Report the presence of any seizures to an on-call provider, and protect the child from injury during any seizure activity. A significant risk (30%-40%) of posttraumatic seizures is associated with the following conditions: severe head injury, diffuse cerebral edema, acute subdural hematoma, or an open depressed skull fracture with parenchymal damage.50 The routine use of prophylactic anticonvulsants has not been shown to prevent the development of seizures.
Status epilepticus should be reported and treated immediately, because it compromises CBF.58 Any abnormal posturing, such as decorticate posturing or decerebrate rigidity (see Fig. 11-9), should also be reported to an on-call provider.
As soon as the patient's condition is stable, the nurse should attempt to evaluate the child's cranial nerve function (see Table 11-2 and Box 11-2), and notify the on-call provider if any cranial nerve function is absent.
Key reflexes evaluated during critical care are the cough and gag reflex and the corneal reflex. The child should demonstrate a cough during suctioning of the airway, and a gag is typically observed during insertion of an orogastric or nasogastric tube. If the corneal reflex is intact, stroking of the eyelashes or the outer edge of the child's eye with a sterile cotton applicator will cause a blink. If this blink is absent, the brain stem is probably injured, and the child will require the regular application of ophthalmic ointment and eye patching to prevent corneal drying and lacerations.
When the child's condition is stable, CT scan, MRI and skull radiography may be performed to aid in the evaluation of the extent of the head injury (see Diagnostic Tests, later in this chapter). A nurse should always accompany the child to the CT scan to monitor the child's level of consciousness and to monitor and support cardiorespiratory function. Appropriate resuscitation equipment and medications should accompany the patient. If the child is extremely unstable with an elevated ICP, a provider who can perform intubation and direct management of increased ICP should accompany the nurse and the patient.
Increased ICP following head injury is treated with support of oxygenation and normocarbia, sedatives, analgesics, paralytics, and hyperosmolar therapy (e.g., hypertonic saline, mannitol); if refractory elevation in ICP is present, the child may be placed in a barbiturate coma. Diuretic therapy and therapeutic hypothermia currently are controversial, because clear benefit has not yet been documented from these therapies in children with TBI. (See Management in the Increased Intracranial Pressure section of this chapter.)
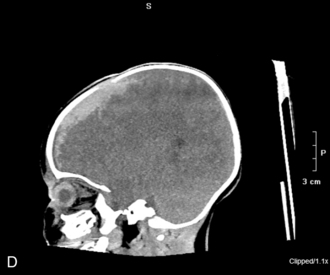
Throughout the child's care, the nurse must be alert for signs of increased ICP (see Increased Intracranial Pressure). The sudden appearance of irritability, confusion, lethargy, and pupil dilation must be reported to a physician or other on-call provider immediately. Emergency acute management of sudden increases in ICP requires immediate intervention with support of the airway, oxygenation, and ventilation and administration of mannitol (0.5-1 gm/kg IV) or 3% saline (3-5 mL/kg IV bolus), or both. A CT scan is typically performed to detect any mass lesion requiring surgical intervention; the CT scan also will enable the evaluation of cerebral edema (see Fig. 11-12).20
Poor prognostic findings following pediatric head injury include the following (assuming normothermia is present): cardiovascular instability despite adequate shock resuscitation, absence of spontaneous respirations, fixed pupils, flaccid extremities with no response to painful stimuli, the presence of diabetes insipidus on admission, severe disseminated intravascular coagulation and elevation of fibrinogen on admission, a GCS score of 4 or less, and persistent elevation in ICP (>20 mm Hg).61
Children who have an initial GCS score >8 usually have good long term outcomes, and those children with initial GCS score of 3-4 have significantly higher morbidity and mortality. Overall, children with head injuries have better outcomes than the adults with similar injuries, and a child's neurocognitive function may continue to improve for years after the initial injury.61
Temperature Control
In recent years, there has been increasing interest in the use of therapeutic hypothermia for children with TBI. In a large multicenter trial conducted by Hutchison and others, hypothermia initiated within 8 hours of injury and continued for 24 hours did not improve either survival or neurologic outcome in the pediatric patient.63 There were significantly more hypotensive episodes and lower average CPP in the hypothermic group, necessitating the use of inotropic drugs and fluid boluses. An earlier study of hypothermia in adult patients with TBI also failed to show improvement in survival or neurologic outcomes,33 although results of another study are anticipated.34 Additional pediatric hypothermia trials are in progress. Current recommendations for children are to maintain normothermia, avoid or rapidly treat hyperthermia, and consider therapeutic hypothermia for refractory intracranial hypertension.11,96
Supportive Care
The healthcare team will assess all major organ systems in the child with TBI (see Chapter 19) and assess the child's airway, oxygenation, ventilation, and perfusion at regular intervals and whenever the child's neurologic condition changes. Once the child's condition is stable, more complete examinations are performed to look for fractures or major lacerations that may require sutures. The examinations include inspection of the skin and body for signs of edema, contusions, petechiae, or hematomas and palpation of the scalp for evidence of depressed or compound skull fractures.
The child's skin and rectal temperature must be monitored closely; a high rectal temperature and low skin temperature can indicate poor systemic perfusion, and fever can indicate EDH or infection. Fever is treated with antipyretics and a cooling blanket if needed. Hypothermia may develop in children with severe head injury; however, hyperthermia blankets or warming lights should be used only to restore normothermia (or a temperature slightly below normal) while avoiding hyperthermia.
A urinary catheter is inserted whenever multisystem trauma or shock is present, unless blood is present at the urinary meatus, which suggests a urethral tear (see Chapter 19). Report any difficulty inserting the catheter or the presence of bloody urine to an on-call provider immediately, because these findings can indicate the presence of genitourinary trauma. The child's urine output normally averages 1 mL/kg per hour if fluid intake is adequate. Inadequate urine output can be caused by prerenal failure (e.g., inadequate systemic perfusion), renal failure (e.g., renal ischemic tubular injury or renal contusion), or postrenal failure (e.g., urethral obstruction). Oliguria requires immediate investigation and prompt treatment of the cause (see Chapter 13). As noted previously, hypertonic saline or hyperosmotic agents may be administered if increased ICP develops.
During the initial assessment and treatment of the child with head trauma, a nurse, social services member, chaplain or other resource typically remains with the parents to answer questions at the bedside. Although it can be difficult for the nurse to arrange the time to speak with the parents, brief updates are extremely helpful. Most parents understand the need for the nurse to focus attention on the physical care of the child. The parents often are reassured to see the careful treatment their child receives, seeing that every effort is being undertaken to stabilize and care for their child.
Injury-Specific Management
Once the child is stable, treatment of the child's specific injury is undertaken. The following information includes the specific management of the most common forms of pediatric TBI.
Concussion
Concussions are not associated with abnormalities on a CT scan, and they usually require no treatment. However, because the history of loss of consciousness followed by recovery and responsiveness also can be consistent with that of the development of an EDH, the child with concussion is often admitted to the hospital for observation. As noted earlier, children occasionally complain of headache, dizziness, malaise, and fatigue for days or weeks following a concussion. It is important that the parents be aware of this postconcussion syndrome so that the child will not be suspected of malingering. However, the healthcare team should not suggest to the child that symptoms are an expected part of the child's behavior.
Contusion
The appropriate treatment of a cerebral contusion is determined by the extent of the primary cerebral injury and the severity of secondary injuries, such as hemorrhage or cerebral edema. Treatment of skull fractures, EDHs, SDHs, and SAHs is reviewed in the sections immediately following. Approximately 10% of children with a cerebral contusion will develop posttraumatic seizures beginning hours, months, or years after the head injury.78
Skull Fractures
Most children with simple or linear skull fractures require no treatment. However, children should be observed carefully for signs of the development of an EDH or SDH.50
Depressed skull fractures are elevated surgically if the skull fragment is 5 mm or more (or a distance greater than the thickness of the skull) below the contour of the skull or if serious underlying cerebral injury or hemorrhage is present. Depressed skull fractures also can be elevated surgically for cosmetic reasons. Before any surgery is performed, the child's cardiorespiratory status is assessed thoroughly and shock resuscitation is provided, if needed. If the depressed skull fracture is located near the sagittal or lateral sinus, this venous channel might tear, causing profuse external or intracranial bleeding. In such a case, treatment of hypovolemic shock will be required (see Shock in Chapter 6). Immediate surgical control of the bleeding site is also necessary. The surgeon will elevate the depressed bone fragment and debride the wound in the operating suite.
When a compound (open, depressed) skull fracture is present, surgical elevation and repair is necessary. Because portions of the scalp or other foreign material can enter the wound and the intracranial space, the wound is carefully debrided. In addition, the surgeon will repair any dural tears.
Children with basilar skull fractures are hospitalized for observation. Because CSF drainage from the nose or ear indicates communication between the subarachnoid space and the nasal passages or external ear, such children are at risk for developing meningitis or intracranial abscess formation. Antibiotic therapy in CSF leaks is reserved for patients who show symptoms of CNS infection. The head of the patient's bed is typically kept level, unless the ICP is elevated, in an effort to decrease pressure and allow the CSF leak to spontaneously seal. In some cases, a lumbar drain can be placed in an effort to decrease pressure and allow the CSF leak to seal. Most CSF leaks will seal spontaneously within a few weeks, but occasionally children develop a chronic CSF leak as the result of entrapment of the dura between skull fragments during healing. This leak increases risk of meningitis and eventually will require surgical repair.
If a basilar skull fracture or head injury with facial bone fractures is present, the nurse should not attempt to insert a nasogastric tube, because as the tube may be directed intracerebrally. An orally inserted gastric tube is appropriate in this situation (see Chapter 19). Prophylactic antibiotics are frequently given if pneumocephalus is detected on the CT scan. Facial fractures involving the sinus cavities or producing pneumocephalus indicate a dural breach and are associated with increased risk of meningitis or abscess formation. Vaccines are given for pneumococcal and meningococcal meningitis prophylaxis.74
Intracranial hematoma and other forms of significant cerebral injury frequently are associated with a skull fracture.50,78 Any child admitted with a skull fracture must be closely monitored for seizures, evidence of hemorrhage, or increased ICP.
Vascular Injuries
When the child with an EDH demonstrates a sudden decreased level of consciousness, immediate surgical decompression of the hematoma is required. A CT scan will assist in the diagnosis of an epidural hematoma before deterioration in the child's clinical status.50,78 Before surgery the child requires treatment of increased ICP. After surgery many patients will demonstrate elevation in ICP, requiring aggressive medical management.50 The perioperative mortality is proportional to the degree of neurologic deterioration that develops preoperatively; if surgical decompression is performed before significant herniation or pupil dilation occurs, perioperative mortality is typically less than 10%.50 If, however, significant elevation in ICP, brain herniation, and ischemia develop before surgical relief is provided, perioperative morbidity and mortality will be high.50
The patient with an acute SDH requires careful assessment for and prevention of increased ICP. Generally, small subdural hematomas do not require surgical intervention; however, surgery may be considered if a large, acute SDH is producing significant signs of increased ICP (i.e., the hematoma is creating a significant mass effect exceeding that produced by cerebral edema or if a midline shift is present). Immediate surgical evacuation of a symptomatic hematoma is warranted if it is >5 mm at the thickest point. A delay in surgical intervention increases mortality to approximately 90%.50
Often, children with an SDH also have diffuse hyperemia and vasogenic cerebral edema that can be aggravated by surgery and will continue to progress even after removal of the hematoma. These children will require aggressive management of increased ICP. Because extremely high ICP has been recorded in both surgically treated and untreated patients, initiation of a barbiturate coma may be indicated.83,108
Mortality following an SDH is significant, and both morbidity and mortality seem to be closely related to the patient's level of consciousness at the time of surgery (if operated) and the degree and duration of intracranial hypertension.
The child with an SAH can develop a rapid increase in ICP requiring aggressive medical management. The presence of blood in the subarachnoid space can interfere with CSF reabsorption. As a result, children with an SAH are at risk for developing communicating hydrocephalus days or weeks after the injury.
Intracerebral hemorrhage can be the most damaging form of cerebral vascular injury, because it results in a rapid increase in ICP and in direct damage to surrounding brain tissue. When the diagnosis of an intracerebral hematoma (clot) with a mass effect is confirmed by CT scan or arteriography, surgery is performed immediately. The bleeding site is controlled, the clot is evacuated, and the area is debrided. Cerebral contusions are rarely removed, but they can contribute to the development of increased ICP. Postoperative management often is complicated by increased ICP, seizures, and motor or sensory deficits.62,96,110
Cerebral Edema
Children with head injury usually develop hyperemic cerebral edema for 24 to 48 hours after the injury. In addition, direct cerebral injury may produce vasogenic cerebral edema, and cytotoxic cerebral edema can result from hypoxia secondary to hypovolemic shock.83 Therefore, children with TBI are at risk for increased ICP and should be transferred to or cared for at a trauma center with experience in pediatric ICP monitoring and the management of intracranial hypertension. Medical and nursing management of this complication has been discussed previously.
Psychosocial Support
Children with head injury can be extremely agitated as the result of fear, pain, or increased ICP. It is important that the nurse be able to provide calm, efficient care, including assessment and physical and emotional support. The nurse is also the best person to recognize changes in the child's level of consciousness and separate these signs from those produced by fear, pain, or sleep deprivation.
The parents may feel extremely guilty if the child's injury occurred during a motor vehicle crash while they were driving the car, or if the child was injured from a preventable fall or sports activity. Parents often are inclined to think that they could or should have prevented the injury. If the child's injury is inflicted, the parents may feel distraught and guilty. If inflicted injury is suspected, the hospital child abuse team must be notified immediately so that the child is protected throughout recovery, documentation is accurate and complete, family support is provided and appropriate legal action is taken.
Children with TBI require close follow up care and appropriate referrals for supportive care after discharge. Many children demonstrate functional morbidity following TBI and may have difficulty returning to school, behavioral problems, sleep disturbances, and discipline problems.
Poor outcomes are thought to be closely related to periods of uncontrolled intracranial hypertension and inadequate cerebral perfusion. As a result, it is imperative that the nurse notify an on-call provider of sharp or prolonged increases in ICP or hypotensive episodes that lead to a prolonged decrease in calculated CPP.123
If the child's neurologic status deteriorates and the child dies, the parents require compassionate and consistent preparation and support (see Brain Death and Organ Donation in this chapter and in Chapter 3, and Withholding and Withdrawing Therapy in Chapter 24).
Spinal Cord Injury
Etiology
Any trauma victim can sustain spinal cord injury (SCI). Traumatic SCI can be primary or can result from a secondary insult. The mechanisms most commonly associated with pediatric SCI include falls and pedestrian-related motor vehicle crashes. In adolescents, motor vehicle crashes, sports and diving injuries and gunshot wounds are common causes of SCI. Intentional injury may also cause SCI.
Pathophysiology
The young child's head is large and heavy in proportion to the rest of the body. The child ejected from or struck by a car often is propelled head-first into the ground or another object, and resulting hyperextension or flexion of the neck is likely to produce cervical spine injury. Flexion-rotation or hyperflexion injuries can cause dislocation or locking of the facets (articulation surfaces) of two contiguous vertebrae with resulting spinal cord compression.
The cervical spine is relatively unstable and is still developing in young children. As a result, SCI patterns in children differ from those in adults. The ligaments along the child's cervical vertebrae are relatively lax, and the paraspinous muscles are incompletely developed. The child's vertebral bodies are wedge-shaped and not completely ossified. In addition, the facet joints of the cervical vertebrae are relatively flat. For these reasons, the vertebrae can shift several centimeters during injury or the application of force to the spine, resulting in spinal cord injury without evidence of injury to the vertebrae. Although the pediatric spine is relatively more elastic than the adult spine, it will be injured if significant cervical subluxation occurs (Fig. 11-19).
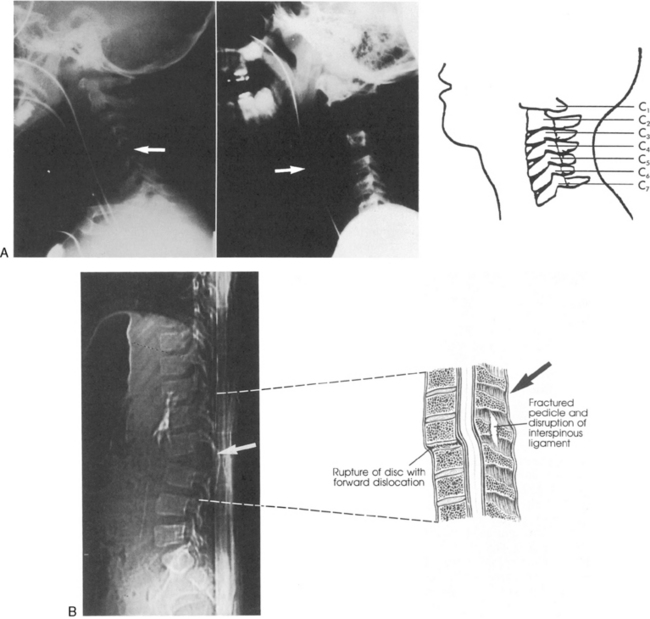
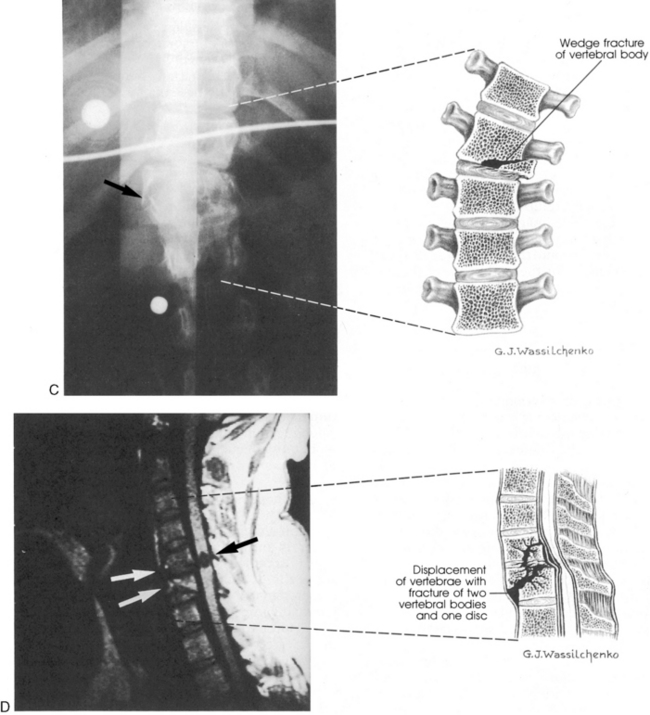
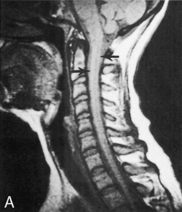
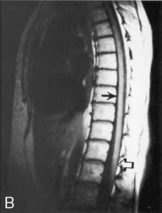
Fig. 11-19 Spinal cord injury. Many injuries resulting in spinal cord damage produce visible radiographic changes, although a significant number (20%-60%) are not associated with any skeletal fracture or dislocation. A, Lateral cervical spine radiographs demonstrating skeletal abnormalities associated with cervical spine injury. The first radiograph is from a 4 year old who was restrained in a car seat that was not anchored in the car. The separation between the fifth and sixth cervical vertebrae is subtle but detectable (arrow), especially when compared with the line drawing of normal anatomy (far right). Radiopaque orogastric and nasogastric tubes are visible; they are slightly displaced anteriorly, indicating a small amount of edema surrounding the spinal cord injury. The location of the injury is unusual for this age. The second radiograph shows a 5-year-old pedestrian struck by an automobile and demonstrates significant separation between the first and second cervical vertebrae. This is a more common site of cervical spine injury in young children. Note the anterior displacement of the nasogastric tube (arrow) produced by edema surrounding the injury. An endotracheal tube is present but not visible. The line drawing depicts normal cervical spinal anatomy in a 3- to 4-year-old child. B, This scan film performed before a computed tomography scan demonstrates lumbar vertebral and spinal cord trauma associated with a lap belt injury. Separation of the lumbar vertebrae can be seen (arrow) and resulted in paraplegia. This injury resulted from flexion of the lumbar spine (see drawing). C, Flexion injury of the lower thoracic vertebrae and spine is visible on this anteroposterior chest radiograph. This radiograph shows an unrestrained 16-year-old driver who was thrown from the car. The lateral flexion resulted in compression of the spinal cord and fracture of the thoracic vertebrae (see arrow and corresponding illustration). The rod placed during surgery is visible. D, This magnetic resonance imagery scan shows in detail the skeletal and spinal damage resulting from a flexion-rotation injury. This 16-year-old motorcycle driver sustained displacement of two vertebrae (white arrows) and fracture of two vertebrae and one disc. Resulting compression of the cervical spine produced a complete spinal cord injury. A contusion is visible in the spinal cord (black arrow). The line drawing depicts the injury.
(A, courtesy Carol Gilbert and John Feldenzer, Roanoke, Va. Drawing reproduced from Riviello JJ, et al: Delayed cervical central cord syndrome after trivial trauma, Pediatr Emerg Care 6:116, 1990. B, courtesy Bennett Blumenkopf, Vanderbilt University Medical Center, Nashville, Tenn. Line drawing reproduced from Rudy EB: Advanced neurological and neurosurgical nursing, St Louis, 1984, Mosby. C, courtesy Noel Tulipan, Vanderbilt University Medical Center, Nashville, Tenn. Illustration reproduced from Rudy EB: Advanced neurological and neurosurgical nursing, St Louis, 1984, Mosby. D, courtesy Bennett Blumenkopf, Vanderbilt University, Nashville, Tenn. Line drawing reproduced from Rudy EB: Advanced neurological and neurosurgical nursing, St Louis, 1984, Mosby.)
The most common areas of SCI in children younger than 9 years include the atlas, axis, and upper cervical spine. Generally, ligamentous injuries are more common than bone injuries. In patients older than 9 years, injury patterns resemble those of the adult, with less cervical involvement.123
Pediatric spinal cord injuries occur when vertebral bodies are fractured, or when vertebral subluxation (partial dislocation) occurs. Subluxation results in anteroposterior misalignment of contiguous vertebrae, with narrowing of the spinal canal and spinal cord compression. Young children are likely to sustain subluxation injuries without associated fractures. The severity of the neurologic deficit is related to the location and severity of the subluxation.123
The neurologic dysfunction associated with SCI can result from the injury itself or from secondary compromise of spinal cord perfusion (ischemia or infarction), edema and necrosis. In laboratory experiments, SCI produces altered permeability of neuron membranes, electrolyte flux across the membranes, and the release of catecholamines and endorphins. Vasospasm and thrombosis in spinal cord vessels can contribute to ischemia, infarction, and dysfunction.91
Severe traumatic cervical spine injury (especially to the upper cervical spine) usually produces respiratory arrest at the scene of the injury, and the patient dies unless immediate resuscitation is provided. Occasionally, children with a cervical spinal injury sustain injury to the cervical vertebrae, rendering them unstable. These children can move all four extremities and breathe at the scene of the accident; however, if the cervical spine is not immobilized (particularly during intubation), spinal cord injury, respiratory arrest, and tetraplegia can result.
If a spinal cord injury is mild or moderate, complete recovery of neurologic function is possible despite the presence of complete neurologic deficit on admission.123 However, if the initial insult is severe (particularly if severe subluxation occurred) or is associated with the development of edema, permanent loss of function and sensation below the level of injury can occur.
Traumatic atlantooccipital dislocation is the disruption of the supporting ligaments between the skull and the vertebral column, occurring in a transverse or vertical direction and resulting in complete transection of the spinal cord. Although this type of SCI is rare in children and adolescents, traumatic atlantooccipital dislocation is seen in infants and toddlers. Most children do not survive this initial insult, but as prehospital care has improved over recent years there are reports of survival when immediate CPR is provided at the scene or emergency responders arrive within a few minutes of the injury.123
Clinical Signs and Symptoms
Physical Examination
Approximately half of all children with SCIs demonstrate signs of neurologic deficit on initial examination.123 The most consistent sign of SCI is the loss of some or all movement and sensation below the level of injury. The degree of deficit is used to assess the severity of injury. Complete injury is present if the child demonstrates complete absence of all movement and sensation below the level of injury. Rectal sphincter tone is also lost. Partial injury is present if the child demonstrates transient weakness or paresthesias.123 Careful repeated clinical evaluation of movement and sensation in all extremities must be performed until definitive studies are completed, because many children demonstrate a delayed onset of symptoms.123
The clinical signs of cervical SCI include the development of respiratory depression or apnea and weakness in the upper arms. Complete cervical spine injury is associated with flaccid paralysis (usually tetraplegia) and anesthesia below the level of the injury. Paradoxic respirations may be observed if an injury occurs at the level of the third to fifth cervical vertebrae. Patients with these injuries typically require chronic ventilatory support.
Diagnostic Studies
Good quality cervical and thoracic lateral and anteroposterior radiographs will enable the detection of many spinal cord injuries (see Fig. 11-19).123 A lateral radiograph is obtained that includes the first cervical vertebra to the first thoracic vertebra.6 Occasionally the lateral displacement of vertebrae will be apparent on the anteroposterior radiograph and may be noted on the chest radiograph. If the radiographs are equivocal, or if nonvisualized SCI is suspected, flexion-extension radiographs are obtained after the child is alert and responsive.50
A CT scan will allow definitive visualization of the upper cervical spinal column, soft tissue, and vertebrae.50 This examination should be part of the CT scan performed for every child with a severe closed head injury.
Approximately 20% to 60% of children with SCIs will have no radiographic evidence of vertebral or spinal injury; this condition is known as spinal cord injury without radiographic abnormality (SCIWORA). The development of SCIWORA usually indicates the presence of severe subluxation and trauma. Children with SCIWORA are typically younger than 9 years and exhibit a wide range of neurologic findings from paresthesias to weakness or paralysis. The conscious child may complain of pain radiating down the neck or entire spine during movement. Full recovery is usually made in patients with normal cord signals, whereas children with transection of the cord or hemorrhages usually have poor outcomes.123
MRI will provide beautifully detailed images of the spinal cord and all surrounding structures and tissues. Although these scans are not practical for the evaluation of unstable patients, they can be used during follow-up care (see Common Diagnostic Studies).
Management
Management of the child with SCI is designed to minimize the potential for further injury while supporting maximal recovery of spinal cord function. In general, the spinal cord is immobilized until the child's condition is stable, and then definitive therapy is provided.47,50 The neck and spine are maintained in a neutral position, using the jaw-thrust maneuver when opening the airway and ensuring that manual stabilization is performed during intubation.
Steroid administration is advocated within the first 8 hours after injury to prevent secondary spinal cord edema and inflammation. A Cochrane meta-analysis of three studies documented that large dose methylprednisolone administration (a bolus dose of 30 mg/kg, followed by continuous infusion of 5.4 mg/kg per hour for 23 hours) significantly increased functional recovery of adult patients with SCI.28 Although this steroid administration has now become standard,50 controlled trials of this therapy have not been performed in children. There are no class I data to support the use of steroids in children younger than 18 years with SCI, and it should be avoided in children with brain injury.13,123
Children with SCI require close observation for signs of neurogenic shock. These children experience a loss of vasomotor tone leading to venous pooling, decreased venous return, and ultimately decreased cardiac output. Bradycardia results from loss of sympathetic tone with resulting parasympathetic dominance. Management includes volume resuscitation and vasopressor support. SCI will also produce hypothermia, because the vasodilation leads to heat loss through the skin.
Urgent surgical intervention is rarely needed during the acute management of SCI, particularly when complete SCI is present. Urgent surgical intervention is indicated for incomplete SCI with compromise or for incomplete SCI with progressive neurologic dysfunction, because such progression often indicates the presence of an emerging but reversible problem, such as an epidural hematoma, or an unstable spinal injury.50
If a cervical spine injury is unstable or subluxation is present, immobilization and alignment of the spine will be necessary through the use of Gardner-Wells tongs or halo traction. If the vertebral bodies are not reduced (i.e., they are misaligned), weight is added to the traction device, and clinical and radiographic examinations are repeated until the vertebrae are realigned or reduced. The alert patient will require analgesia during this procedure, but should not be sedated because the child must be responsive during clinical examination.50,123
Once the vertebrae are aligned, the patient is maintained in halo traction for several weeks. In children, the use of a halo vest will facilitate ambulation.50 If the area of injury continues to be unstable, spinal fusion occasionally is required, possibly several weeks later. Surgical stabilization is most likely to be required for children with SCI who are younger than 3 years.
Rehabilitation services should begin as soon as the pediatric patient with a SCI is stable. A physical medicine and rehabilitation consult should be obtained to provide guidance about the child's long term care needs. Physical and occupational therapy will be beneficial in maintaining range of motion, mobilization and integration into activities of daily living. Speech therapists will evaluate tolerance of oral intake and, if needed, can assist in providing alternative methods of communication. A bowel and bladder regimen should be initiated early and re-evaluated on a routine basis.
Intracranial Tumors
Etiology
Primary brain tumors are one of the most common forms of cancer in children, and medulloblastoma is the most common malignant brain tumor. Survival from brain tumors has now risen from approximately 60% in the 1980s to 80% to 85% in recent years.98 This improved survival can be attributed to earlier and more complete tumor resection (particularly for medulloblastomas) as well as more aggressive and targeted chemotherapy and radiation therapy. The involvement of the disease at presentation, the amount of tumor resection possible, and tumor grades are part of the prognostic factors involved in the outcomes of most brain tumors (e.g., ependymoma).21
Tumors are abnormal masses that can arise from any tissue in the body. Their cause is unknown, although the role of hereditary factors and environmental carcinogens continues to be explored. Although few tumors are present from birth, many tumors of childhood arise from the inappropriate development of primitive neuroepithelial cells. Astrocytomas are the most common primary intraaxial brain tumor, and pilocytic astrocytomas typically manifest in the second decade of life.50
Pathophysiology
Intracranial tumors in children produce an increase in intracranial volume; unless the skull can expand commensurately, the child will develop increased ICP. In addition, the tumor causes compression of the surrounding brain tissue, compromising important cerebral functions.
Tumors are classified according to their location, degree of malignancy, and histologic features. Classification by location is used here because it enables more straightforward prediction of the clinical consequences of tumor expansion and the possibility and risks of surgical tumor excision (Box 11-11).
Box 11-11 Pediatric Central Nervous System Tumors Classified By Location
3. Spinal cord tumors: ependymoma, astrocytoma
4. Generalized disease with brain tumor components: von Recklinghausen disease, tuberous sclerosis, Sturge-Weber disease, von Hippel-Lindau disease, ataxia telangiectasia, nevoid basal cell carcinoma syndrome
From Van Eys J: Malignant tumors of the central nervous system. In Sutow WW, Fernbach DJ, and Vietti TJ, editors: Clinical pediatric oncology, ed 3, St Louis, 1984, Mosby.
Supratentorial tumors involve the cerebral hemispheres and all structures located above the tentorium cerebelli. Infratentorial tumors are those that involve the brain stem and cerebral structures located below the tentorium cerebelli.
Classification of tumors by cell type allows some predictions about the speed of tumor growth and spread and about recurrence risks. It is important to note that intracranial tumors in children may be malignant by position as well as by cell type. This means that the tissue itself is not malignant, but tumor growth can compress or erode vital brain tissue, resulting in serious neurologic compromise or death.
In the following section, the most common intracranial tumors in children are described. This description includes the clinical consequences of tumor growth.
Supratentorial Tumors
The two most common supratentorial tumors in children are the astrocytoma and the craniopharyngioma. The astrocytoma is the most common of all supratentorial tumors accounting for approximately 12,000 new cases per year.50 This tumor arises from abnormal proliferation of the cerebral astrocytes. Astrocytomas can develop in the frontal, temporal, and central parietal areas of the cerebral hemispheres, and tumor growth can extend across the corpus callosum from one parietal lobe to the other. These tumors also can invade the brainstem or third ventricle and may cause hydrocephalus.50
Astrocytomas can grow slowly or rapidly. Tumor specimens are typically graded on a scale of one to four according to the degree of cell differentiation present in the tumor.50 When an astrocytoma is located above the tentorium, it is usually diffuse and expands into surrounding tissue or along long nerve fiber tracts. Expansion through metastases (transfer to other organs) is rare.
The craniopharyngioma is responsible for approximately 4% of all intracranial tumors in children and generally occurs between the ages of 5 and 10 years.50 It results from the growth of displaced neuroepithelial cells. The tumor consists of a solid mass or cyst that contains fluid, cellular debris, and calcified material. It develops within or just above the sella turcica (the skull pouch containing the pituitary) or within the third ventricle. As the craniopharyngioma grows, it can obstruct the foramen of Monro, producing hydrocephalus, or it can compress the optic chiasm, the pituitary, or the hypothalamus, producing visual disturbances, fever, hypoglycemia, diabetes insipidus (DI), or occasional hypotension.50
Infratentorial Tumors
Infratentorial tumors account for nearly two thirds of all pediatric brain tumors and for nearly half of all tumors in children.50 These tumors are typically detected early in their development because they can rapidly produce changes in vital body functions. The most common forms of infratentorial tumors in children are medulloblastomas, astrocytomas, ependymomas, and brain stem gliomas.
The medulloblastoma accounts for 15% to 20% of all intracranial tumors and one fourth of primary intracranial tumors in children.50 It is the most malignant of the posterior fossa tumors because it grows rapidly and tends to recur after surgical excision. The tumor rises from neuroepithelial cells located in the roof of the fourth ventricle, so it may cause early development of hydrocephalus.50 The tumor is usually a soft, gray mass that extends from the medulla along CSF pathways into the fourth ventricle, subarachnoid space, third ventricle, or spinal column. Symptoms include stiff neck or neck pain, increased ICP, obstructive hydrocephalus, ataxia, and fatigue. Hypotension or hypertension can result from compression of the medulla, and backache, limb weakness, or loss of bladder control will indicate spinal cord involvement. Medulloblastomas occur most commonly in children in the first decade of life. Five-year survival rates vary widely in published reports (21%-70%), but late relapses do occur.50
Astrocytomas also can grow in the brain stem, although they usually are confined to the pons. They produce sequential and multiple cranial nerve palsies, ataxia, and pyramidal (voluntary movement) dysfunction; headache and diplopia also occur frequently. The mean age at diagnosis of brainstem astrocytoma is 7 years. Five-year survival is approximately 40% despite aggressive chemotherapy and radiation therapy.50
Ependymomas account for approximately 5% to 6% of all intracranial tumors in children.50 This tumor rises from neuroepithelial cells, and it forms a fleshy gray mass that most frequently obstructs the fourth ventricle, producing hydrocephalus and cranial nerve palsies. These tumors can occur anywhere along the neuraxis.50
Brain stem gliomas are cysts that compress the cranial nerves, the pons, and medulla. If the glioma expands into the cerebellum, relatively large tumor growth can be accommodated without symptoms of cerebellar compression. The first symptoms of the brain stem glioma are usually those of cranial nerve dysfunction. Initially, compression of the abducens nerve (sixth cranial nerve) will cause nystagmus, facial nerve (seventh cranial nerve) compression will cause a facial palsy, and oculovestibular nerve (eighth cranial nerve) compression will result in hearing loss. As the glossopharyngeal and vagus nerves (ninth and tenth cranial nerves) become involved, the child will develop hoarseness and experience difficulty in swallowing. Increased ICP develops during the terminal stages of tumor expansion, producing headache, vomiting, and other signs of intracranial hypertension. The prognosis of this tumor is extremely poor, and most children with malignant brainstem gliomas die within 6 to 12 months of diagnosis. Treatment is usually not surgical, and a biopsy should not be performed if there is diffuse infiltrating brainstem lesion.50
Spinal cord tumors account for approximately 15% of all primary CNS tumors. They may be primary or secondary. Spinal tumors can be classified into three categories: extradural, intradural/extramedullary, and intramedullary. Extradural tumors typically arise within the vertebral bodies and are often metastatic. Intradural/extramedullary tumors arise inside the dura but outside of the spinal cord itself. Meningiomas and spinal cord sheath tumors are examples of intradural/extramedullary tumors. Intramedullary tumors arise within the spinal cord tissue. Typical tumor types are ependymomas and astrocytomas. These tumors may be metastatic.
Spinal cord tumors typically compress rather than invade the cord. Pain is the most common presenting symptom, and motor disturbances (typically weakness) are the second most common complaint. High-grade astrocytomas are typically treated with surgical excision and postoperative chemotherapy. Surgical removal is the only recommended treatment for ependymomas.
Prognosis is determined by the aggressiveness and resectability of the initial tumor. Astrocytomas have poorer outcomes than ependymomas and have a 50% recurrence rate within 4 to 5 years.50
Clinical Signs and Symptoms
Intracranial tumors in children can grow to a large size without producing significant symptoms until they invade vital brain tissue or cause increased ICP. In children up to 12 years of age, the skull can expand to accommodate a gradual increase in intracranial volume. Tumors may not be diagnosed in the young child with nonspecific signs of neurologic compromise, because testing of cognitive functions, fine motor skills, and sensation is very difficult in very young patients.
Signs and symptoms of any neoplasm in the child include a change in the child's appearance or growth patterns, swelling, lumps, masses, vague pains, or persistent irritability. The child may also change feeding patterns or bowel or bladder function or may develop unexplained clumsiness or stumbling or unexplained or persistent bleeding. General signs of an intracranial tumor during childhood include signs of increased ICP, headache, emesis, anorexia, ataxia, cranial nerve palsies, nystagmus, paresis, seizure activity, and hydrocephalus.
Specific signs of increased ICP caused by an intracranial tumor include papilledema, altered level of consciousness, visual disturbances (diplopia and blurring of vision), headache, and emesis. The headache is characteristically intermittent but progressive. It tends to be present after awakening, and it often is associated with vomiting. The child usually does not feel nauseated before vomiting. If vomiting or headaches are persistent, anorexia may develop.
As the tumor grows, an infant will develop a bulging fontanelle, and torticollis may result from the asymmetric compression of neck muscles by the tumor. Nuchal rigidity may be noted.35
If the tumor compresses the sixth cranial nerve or if uncal (temporal-lateral) herniation develops from increased ICP, the child may develop strabismus, diplopia, or blurring of vision. Ataxia or nystagmus will develop if the tumor compresses or erodes the cerebellum.50 Paresis will develop if the tumor compresses the brainstem or pyramidal tract. Seizures are rarely an early sign of an intracranial tumor, although they can develop late in the clinical course. If hydrocephalus is present, the tumor is obstructing CSF pathways.
The best means of diagnosing an intracranial tumor is with a thorough neurologic examination and a CT scan with and without contrast. MRI is also an essential tool in the diagnosis of brain tumors and is the preferred method of scanning for some brain stem tumors. (See Common Diagnostic Tests later in this chapter.) A plain skull radiograph may demonstrate characteristic changes associated with some tumors (e.g., calcification near the sella turcica that occurs with craniopharyngioma), but often the radiographs are not helpful. Arteriography can be performed to better locate and define the tumor.
Management
Care of the child with an intracranial tumor requires treatment of intracranial hypertension (see Increased Intracranial Pressure in the Common Clinical Conditions section of this chapter), surgical resection if possible, and initiation of chemotherapy or radiation therapy. Laser, stealth, and gamma knife surgeries are surgical techniques that have improved the efficacy of surgical intervention for intracranial tumors. Stealth or stereotactic guidance is especially useful in transsphenoidal tumor resection. Over the past decade, the development of frameless stereotaxy in combination with fluoroscopy assists the neurosurgeon in staying midline. It is considered safe and accurate and adds little cost and time to conventional surgical approaches.44
Radiation therapy is typically prescribed for intracranial tumors, and chemotherapy recently has been found to be helpful in the treatment of some intracranial tumors in children.50 Steroid therapy such as dexamethasone may be used for treatment of localized edema surrounding the tumor. Although the mechanism of action is unclear, the working theory is that the drug decreases the blood-tumor barrier permeability or it may increase parenchymal resistance to fluid transport.50
The child with a brain tumor is typically admitted to the critical care unit following neurosurgery. The child may also require critical care for management of sepsis or infections secondary to chemotherapy-induced immunosuppression (see Chapters 6 and 16).
The child and family will require long-term physical and emotional support. If the tumor initially produced vague clinical signs and symptoms, and the parents ignored the child's initial complaints, they may feel guilty and frustrated. Unless deterioration is rapid, the child will require surgery, radiation, and possible chemotherapy with frequent hospitalizations. The child may, in fact, have a chronic neurologic disease and require prolonged treatment. The child and family will require long-term follow-up and ongoing support.
Meningitis
Etiology
Meningitis is an acute inflammation of the meninges and CSF. It occurs far more commonly in children than in adults, and it is seen most frequently in children between 1 month and 5 years of age. Meningitis most commonly is produced by bacteria (called purulent meningitis) or viruses (usually called aseptic meningitis), although it can also result from fungi, parasites, or mycobacteria.
Over the past decade there have been advances in testing technology. The addition of pneumococcal and meningococcal vaccines into the routine immunization schedule and the adjunctive use of steroids have changed the management of meningitis. However, meningitis remains a leading cause of infection in children, with significant morbidity and mortality.
The three major organisms responsible for meningitis worldwide are Haemophilus influenzae, Neisseria meningitidis, and Streptococcus pneumoniae, but the causative organisms vary depending on the geographic location and age of the child. In the United States, meningococcus and pneumococcus account for approximately 95% of acute bacterial meningitis in children. Group B streptococcus remains a leading cause of meningitis in the neonatal population. These forms of meningitis usually result from the extension of a localized infection, with transient bacteremia and CNS spread of the organism. Staphylococcal meningitis occurs most commonly after neurosurgery or after a skull fracture with a dural tear.81,114
Pathophysiology
Most pathogens responsible for bacterial meningitis are introduced via the respiratory system. Colonization of the organism in the mucous membranes of the nasal passages causes injury to the epithelial cells, allowing access into bloodstream. After this hematogenous spread of bacteria (i.e., it is spread by blood), the bacteria then penetrate the blood-brain barrier, infecting the meninges. Once the CNS has been breached, the bacteria will multiply freely and rapidly spread throughout the entire surface of the spinal cord and brain. Inflammatory mediators and cytokines are then released causing an inflammatory response. Cerebral edema, inflammation, and ensuing increased ICP are frequently seen after bacterial invasion of the brain parenchyma. Cerebral vasculitis, thrombosis, and infarction are the consequence of endothelial damage at the vascular level. Edema or scarring of the outlet of the third ventricle produces stenosis of the Sylvian aqueduct and results in obstruction to CSF flow and hydrocephalus.50
Clinical Signs and Symptoms
Clinical presentation depends on the age of the child, the time lapse between initial symptoms and medical treatment, the immunity of the child, and the infecting organism. Onset can be acute or slow. In the neonate or infant of younger than 6 months, the signs of meningitis are often nonspecific. The infant may be extremely irritable or lethargic with a history of poor feeding, vomiting, and fever. Seizures may develop. A high-pitched cry may be observed in infants with increased ICP.
Extremely malnourished or very small infants with meningitis may be afebrile.114 If the ICP is high, the anterior fontanelle will be full and may be tense. Although the presence of nuchal rigidity (stiff neck) provides an index of suspicion, it is often not present in the young infant. The diagnosis is only confirmed by the results of the spinal tap.
Older children with meningitis usually complain of headache and exhibit nausea, vomiting, anorexia, photophobia, acute onset of fever, decreased level of consciousness or seizures.114 Nuchal rigidity, neck pain, and sensitivity to touch are also present. Kernig's sign (pain with extension of the legs) and Brudzinski's sign (flexion of the neck stimulates flexion at the knees and hips) also may be present. Often there is a history of an antecedent upper respiratory or gastrointestinal infection.114
When meningitis is suspected, blood samples are obtained for a complete blood cell count with white blood cell differential, glucose, electrolytes, and blood cultures. The results will help detect evidence of a localized infection or sepsis. Additional urine, serum, or wound cultures are obtained as indicated.
A lumbar puncture is the definitive diagnostic test for meningitis. During the lumbar puncture, CSF samples are taken. From these samples, a culture, Gram staining, and cell count will be performed, and protein and sugar levels will be measured. The general appearance of the fluid and the opening and closing CSF pressures should be noted in the nursing record.
When bacterial meningitis is present, the glucose concentration of the CSF is low, but the protein content is high. In addition, there will be a large number of cells present in the fluid, predominantly neutrophils (Table 11-11). The culture and Gram stain will be positive.114
When aseptic (viral or fungal) meningitis is present, the CSF glucose concentration is usually normal, and the protein content is only slightly elevated. In aseptic meningitis, there may be a moderate or large number of cells, predominantly polymorphonuclear leukocytes early in the course, and lymphocytes later in the course. The Gram stain is usually negative, and, with viral meningitis, the serologic culture is usually positive for virus.114
Untreated meningitis can cause rapid deterioration. The child may demonstrate mild irritability and fever and quickly progress to high fever, seizures, a decreased level of consciousness, and coma. Thus, the effectiveness of treatment can be related directly to the speed of diagnosis and the early initiation of appropriate treatment.
Management
Bacterial Meningitis
If the child is critically ill, support of airway, ventilation, and perfusion will be needed. However, the treatment of bacterial meningitis requires the prompt initiation (in less than 1 hour of first medical contact) and uninterrupted administration of appropriate IV antimicrobial agents. Vascular access must be established immediately and carefully maintained throughout therapy. Broad spectrum antibiotics are administered even before the results of the CSF cultures and sensitivities have been obtained. The infant or child is given nothing by mouth until systemic perfusion and neurologic function are acceptable.
Accurate recording of fluid intake and output and serum electrolyte concentrations is important, because many children will develop SIADH during or after the meningitis (see Chapter 12). The infant or child is typically given nothing by mouth, and IV maintenance fluids are administered. Fluid boluses may be required because these children usually are somewhat dehydrated related to fever and poor oral intake.
The infant's head circumference should be measured on admission and at least every 8 hours, because subdural effusions and obstructive hydrocephalus can develop after meningitis and can be detected by an increase in head circumference. The infant or child with H. influenzae or N. meningitidis meningitis is placed in respiratory isolation until antibiotic therapy has been administered for 24 hours.101
Treatment of bacterial meningitis should begin as soon as there is suspicion of infection. It is ideal to collect all cultures before beginning antibiotic therapy, but antibiotic administration should not be delayed if samples are difficult to obtain. Antibiotics should be given immediately, and the cultures can be obtained at a later time. Initially, broad spectrum antibiotics should be administered that will cover likely causative organisms for age (Table 11-12). The Gram stain is a guide for initial therapy, but coverage should not be narrowed based on this single test result. The CSF culture remains the gold standard for diagnosis of bacterial meningitis. The antimicrobial therapy can be modified once culture results (including organism and sensitivities) have been completed.
Table 11-12 Recommendations for Antimicrobial Therapy in Bacterial Meningitis Based on Predisposing Conditions or on Isolated Pathogen and Susceptibility Testing
| Predisposing Factor | Common Bacterial Pathogens | Antimicrobial Therapy |
| Age | ||
| <1 month | Streptococcus agalactiae, Escherichia coli, Listeria monocytogenes, Klebsiella species | Ampicillin plus cefotaxime or ampicillin plus an aminoglycoside |
| 1-23 months | Streptococcus pneumoniae, Neisseria meningitidis, S. agalactiae, Haemophilus influenzae, E. coli | Vancomycin plus a third-generation cephalosporin*,† |
| 2-50 years | N. meningitidis, S. pneumoniae | Vancomycin plus a third-generation cephalosporin*,† |
| >50 years | S. pneumoniae, N. meningitidis, L. monocytogenes, aerobic gram-negative bacilli | Vancomycin plus ampicillin plus a third-generation cephalosporin*,† |
| Head trauma | ||
| Basilar skull fracture | S. pneumoniae, H. influenzae, group A β-hemolytic streptococci | Vancomycin plus a third-generation cephalosporin* |
| Penetrating trauma | Staphylococcus aureus, coagulase-negative staphylococci (especially Staphylococcus epidermidis), aerobic gram-negative bacilli (including Pseudomonas aeruginosa) | Vancomycin plus cefepime, vancomycin plus ceftazidime, or vancomycin plus meropenem |
| Postneurosurgery | Aerobic gram-negative bacilli (including P. aeruginosa), S. aureus, coagulase-negative staphylococci (especially S. epidermidis) | Vancomycin plus cefepime, vancomycin plus ceftazidime, or vancomycin plus meropenem |
| CSF shunt | Coagulase-negative staphylococci (especially S. epidermidis), S. aureus, aerobic gram-negative bacilli (including P. aeruginosa), Propionibacterium acnes | Vancomycin plus cefepime,‡ vancomycin plus ceftazidime,‡ or vancomycin plus meropenem‡ |
| Microorganism, Susceptibility | Standard Therapy | Alternative Therapies |
| Streptococcus pneumoniae | ||
| Penicillin MIC | ||
| <0.1 mcg/mL | Penicillin G or ampicillin | Third-generation cephalosporin,* chloramphenicol |
| 0.1-1.0 mcg/mL§ | Third-generation cephalosporin* | Cefepime (B-II), meropenem (B-II) |
| ≥2.0 mcg/mL | Vancomycin plus a third-generation cephalosporin*, |
Fluoroquinolone¶ (B-II) |
| Cefotaxime or ceftriaxone MIC ≥1.0 mcg/mL | Vancomycin plus a third-generation cephalosporin*, |
Fluoroquinolone¶ (B-II) |
| Neisseria meningitidis | ||
| Penicillin MIC | ||
| <0.1 mcg/mL | Penicillin G or ampicillin | Third-generation cephalosporin,* chloramphenicol |
| 0.1-1.0 mcg/mL | Third-generation cephalosporin* | Chloramphenicol, fluoroquinolone, meropenem |
| Listeria monocytogenes | Ampicillin or penicillin G# | Trimethoprim-sulfamethoxazole, meropenem (B-III) |
| Streptococcus agalactiae | Ampicillin or penicillin G# | Third-generation cephalosporin* (B-III) |
| Escherichia coli and other Enterobacteriaceae †† | Third-generation cephalosporin (A-II) | Aztreonam, fluoroquinolone, meropenem, trimethoprim-sulfamethoxazole, ampicillin |
| Pseudomonas aeruginosa†† | Cefepime# or ceftazidime# (A-II) | Aztreonam,# ciprofloxacin,# meropenem# |
| Haemophilus influenzae | ||
| β-Lactamase negative | Ampicillin | Third-generation cephalosporin,* cefepime, chloramphenicol, fluoroquinolone |
| β-Lactamase positive | Third-generation cephalosporin (A-I) | Cefepime (A-I), chloramphenicol, fluoroquinolone |
| Staphylococcus aureus | ||
| Methicillin susceptible | Nafcillin or oxacillin | Vancomycin, meropenem (B-III) |
| Methicillin resistant | Vancomycin** | Trimethoprim-sulfamethoxazole, linezolid (B-III) |
| Staphylococcus epidermidis | Vancomycin** | Linezolid (B-III) |
| Enterococcus species | ||
| Ampicillin susceptible | Ampicillin plus gentamicin | |
| Ampicillin resistant | Vancomycin plus gentamicin | |
| Ampicillin and vancomycin resistant | Linezolid (B-III) | |
Note: All recommendations are A-III, unless otherwise indicated.
† Some experts would add rifampin if dexamethasone is also given.
‡ In infants and children, vancomycin alone is reasonable unless Gram stains reveal the presence of gram-negative bacilli.
§ Ceftriaxone/cefotaxime-susceptible isolates.
 Consider addition of rifampin if the MIC of ceftriaxone is 12 mg/mL.
Consider addition of rifampin if the MIC of ceftriaxone is 12 mg/mL.
¶ Gatifloxacin or moxifloxacin.
# Addition of an aminoglycoside should be considered.
** Consider addition of rifampin.
†† Choice of a specific antimicrobial agent must be guided by in vitro susceptibility test results.
Adapted from Tunkel, AR, Hartman BJ, Kaplan SJ, et al: Practice guidelines for the management of bacterial meningitis. Clin Infect Dis39:1267-1284, 2004.
The duration of antibiotic therapy is determined by the specific pathogen, but typically CNS infections are treated for 10 to 14 days. Infants infected with herpesvirus are generally treated for 28 days. In the neonate younger than 6 weeks, acyclovir is added because herpesvirus is a concern for CNS infection in the population. Vancomycin is added if the Gram stain is suggestive of pneumococcus. In the infant older than 2 months, vancomycin is added for empiric coverage because pneumococcal resistance to third-generation cephalosporins has emerged.81,114
Additional days of therapy are indicated if the patient fails to demonstrate clinical improvement or if additional CSF findings indicate partially treated meningitis. Throughout therapy the nurse must monitor for side effects of the antibiotics.
The use of steroids (dexamethasone is 0.6 mg/kg per day divided into four doses for 4 days) in bacterial meningitis has been shown to reduce the incidence of hearing loss and decrease CNS inflammation. However, the first dose must be administered before or in conjunction with the first dose of antibiotics. The targeted bacteria include H. influenzae and pneumococcus. The use of steroids in the neonatal population is yet to be established. Although recommended by the American Academy of Pediatrics, widespread use has not been adopted. Often the infant has already received the first dose of antibiotics before the diagnosis of meningitis is made, so this may partially account for the lack of steroid use in this population. Steroids are not recommended in cases of viral meningitis.
Gastrointestinal prophylaxis with ranitidine or a proton pump inhibitor should be administered when steroids are used.81,114 The nurse should be alert for signs of gastrointestinal hemorrhage or secondary infection that could complicate steroid administration.
A repeat lumbar puncture is not typically recommended at the end of therapy if the child's condition has improved and the child is afebrile. However, if the child is receiving appropriate therapy and does not show clinical improvement within 3 to 4 days of treatment, is experiencing new symptoms, culture results are positive for an unusual or resistant pathogen, or there is no improvement (i.e., in neurologic signs) or child remains febrile on specific therapy after 24 to 48 hours, then the lumbar tap should be repeated. In the neonate, Gram-negative meningitis is an indication for repeat lumbar puncture.81,114
Prophylactic antibiotic administration is recommended for household, daycare, and nursery-school contacts of the patient with Neisseria spp. meningitis. These contacts should begin taking antibiotics within 24 hours of the patient's diagnosis. Prophylaxis also is recommended for anyone having intimate contact with the patient, including babysitters who may have kissed the child. Ceftriaxone, ciprofloxacin, and rifampin are used for prophylaxis in adults, and rifampin is the drug of choice for prophylaxis in most children.101
Viral Meningitis
Supportive care is provided for the child with viral meningitis. Antibiotic administration is not indicated unless a concurrent bacterial infection is present.
Fungal Meningitis
Although rare in the pediatric population, fungal meningitis does occur. It most often occurs in immunosuppressed patients in the setting of HIV, lupus, diabetes, transplant, or cancer. It may also be seen in the premature neonatal population, typically caused by Candida albicans. Fungal meningitis can also complicate invasive neurosurgical procedures.81
Supportive Care
Throughout the first days of therapy, the infant or child should be monitored closely for signs of increased ICP, continued fever, or neurologic deterioration. These findings should be reported to a provider immediately. If the child does develop signs of increased ICP, then ICP monitoring and mechanical ventilation may be required (see Increased Intracranial Pressure in the Common Clinical Conditions section of this chapter).
Because SIADH is a known risk factor in meningitis, the child's serum sodium concentration and osmolality are closely monitored throughout therapy (see Chapter 12). Antipyretics are administered to reduce fever and decrease the risk of febrile seizures. If seizures develop and progress to status epilepticus, prompt anticonvulsant therapy is essential (see Status Epilepticus earlier in chapter). If bacteremia is present, septic shock and disseminated intravascular coagulation may develop (see Septic Shock, Chapter 6 and Immunology and Infectious Diseases, Chapter 16).
Prognosis varies in every patient. Mortality rates are as high as 20% and survivors, especially those with pneumococcal meningitis, often have neurologic deficits. These deficits can include decreased cognitive and motor skills, hydrocephalus, spasticity, hearing loss, and blindness. Hearing screens should be obtained on all children treated for bacterial meningitis, because risk of hearing loss is significant.81,114
Brain Abscess
Etiology
A brain abscess is an isolated intracranial collection of purulent fluid, typically located in the cerebral hemispheres or the cerebellum. The abscess usually develops after bacteremia, but it also can result from chronic sinusitis or following a head injury with a skull fracture. Children with cyanotic congenital heart disease (especially if untreated and older than 2 years) or those with bacterial endocarditis are at increased risk for developing brain abscesses.
Pathophysiology
The pathogen enters the cerebral circulation at the site of intracranial surgery or compound skull fracture or as the result of a systemic or blood-borne infection. Abscesses also can spread into adjacent cerebral tissue from middle ear or mastoid infections.
The infected tissue initially is localized and is invaded quickly by white blood cells. Over a period of several weeks, necrotic tissue within the abscess liquefies, and the abscess becomes encapsulated by fibroblasts.62 As the abscess grows, it can produce signs of increased ICP. If it ruptures, it can produce diffuse meningoencephalitis.
Clinical Signs and Symptoms
The child with a brain abscess may be asymptomatic during the initial period or may demonstrate nonspecific signs and symptoms including headache, fever, malaise, vomiting, confusion, seizures, motor deficits, sensory deficits, speech deficits, and leukocytosis. As the brain abscess enlarges, it produces signs of increased ICP, including a progressive headache, decreased level of consciousness, pupil dilation with sluggish or absent response to light (especially if uncal herniation develops), papilledema, cranial nerve deficits, and seizures. With enlargement of the abscess, progressive signs of increased ICP develop (see Increased Intracranial Pressure section in this chapter).
A brain abscess also can produce localizing symptoms. The child with a frontal lobe abscess may have contralateral hemiparesis, frontal headache, aphasia, or seizures. Temporal lobe abscesses can produce a temporal lobe headache and contralateral facial weakness. A cerebellar abscess often produces a postoccipital headache, nystagmus, ipsilateral ataxia, and limb weakness.62,114
A lumbar puncture will reveal an extremely high CSF pressure, a normal or increased cell count (with polymorphonuclear lymphocytes), an increased protein concentration, and a normal glucose concentration (see Table 11-4). The brain abscess can be localized with a CT scan or MRI.
Management
Prompt initiation and continued administration of IV antibiotics is the key treatment for a brain abscess. Treatment of increased ICP also may be required (see Management in the Increased Intracranial Pressure section of this chapter).114
The most common pathogens causing brain abscesses include anaerobic bacteria, Gram-negative organisms, streptococci, and staphylococci. The underlying cause of the abscess usually determines the causative organism. In children with heart disease, α-hemolytic streptococcus is the most common organism, whereas streptococcus and Staphylococcus aureus are common organisms associated with subacute bacterial endocarditis. S. aureus is commonly seen in abscesses associated with trauma, whereas streptococci, pseudomonas, or H. influenzae are the typical pathogens isolated in patients with otitis or sinusitis (see the Table 11-12 for specific antibiotic choices).
If the abscess appears to be well encapsulated, surgical excision may be attempted. If complete excision is not possible, serial aspiration and irrigation of the abscess may be required.
Throughout the child's care, it is important for the nurse to monitor for signs of neurologic deterioration, because increased ICP can develop and progress rapidly. Even with aggressive medical and surgical treatment of brain abscess, mortality is significant, and survivors may have neurologic deficits and seizures.62,114
Encephalitis
Etiology
Encephalitis is defined as an inflammation of the brain parenchyma. It can be associated with other CNS infections such as meningitis, or it can be related to viral illness such as rabies or herpes simplex. In the neonatal population, enterovirus and adenovirus are responsible pathogens. In children, arboviruses and enteroviruses are common causes of encephalitis. Adenovirus, Epstein-Barr virus, West Nile virus, measles, mumps and varicella are responsible for a small portion of pediatric encephalitis cases.
Encephalitis may appear during the course of an acute viral illness, or it may follow an infection. The term encephalitis is used to indicate an infective or inflammatory cerebral disorder. The term encephalopathy is used to refer to any neurologic disorder of unknown or noninfectious cause associated with a change in level of consciousness, irritability, seizures, and motor or sensory deficit.114
Pathophysiology
Encephalitis probably results from a toxic or infectious agent that enters the brain. The inflammatory mediators released or triggered by the agent produce an inflammatory response that results in cerebral edema, cellular damage, neuronal destruction, and transient neurologic dysfunction.
In the temporal region an acute inflammatory demyelination may be seen in association with viral infections or post immunization. In this circumstance if there is no direct viral involvement of the CNS it is called postinfectious encephalitis. In cases where the spinal cord is involved, it is referred to as acute disseminated encephalomyelitis (ADEM).114
Clinical Signs and Symptoms
Children with encephalitis demonstrate symptoms during or immediately after an acute viral illness or following exposure to a toxic or inflammatory agent. Affected children usually complain of a headache and may demonstrate irritability, lethargy, a change in level of consciousness, nuchal rigidity, visual, auditory and speech disturbances, seizures, or loss of consciousness. High fever is usually not present.
Herpes simplex virus encephalitis generally presents in children 6 months to 18 years of age and is equally distributed between males and females. Herpes simplex virus usually produces an acute, hemorrhagic, necrotizing encephalitis with associated cerebral edema.50,62
A lumbar puncture usually is performed to rule out a bacterial cause of the neurologic symptoms. It reveals normal CSF pressure, normal or increased cell count (lymphocytes may be elevated), normal or slightly increased protein concentration, and normal glucose concentration. The CSF culture and Gram stain will yield no bacterial growth, but serologic tests can aid in the diagnosis of a viral agent (see Table 11-4).
An EEG will reveal diffuse cortical inflammation with high-voltage discharges, usually from the temporal lobes. CT scans typically fail to demonstrate localized areas of infection, but they will show defined areas of edema, predominantly in the temporal lobes. Hemorrhagic lesions are seen on approximately 12% of scans.50,62 MRIs are more sensitive than CT scans, revealing edema as high signals on T2 weighted images, primarily in the temporal lobes.50
Management
Treatment of encephalitis is largely supportive, but acute encephalitis is considered an emergency. The child should be admitted to the pediatric critical care unit for close neurologic monitoring. These children can develop rapid neurologic deterioration, including coma. As always, support of airway, oxygenation, ventilation, and perfusion are priorities. Antibiotic administration is not indicated if the disease is viral or toxic in origin. If the toxic agent can be identified (e.g., a drug) and an antidote is available, it should be administered. Antiviral agents such as amantadine and rimantadine can be used in cases influenza infection.
Steroid use has not been shown to change outcomes in cases of acute viral encephalitis; however, they may be used in an effort to decrease cerebral and neuronal inflammation.114 In cases of acute disseminated encephalomyelitis, steroid administration is the initial treatment of choice.67
If seizures are present, they may be refractory to typical anticonvulsant therapy. Refractory status epilepticus in the child with encephalitis may require a induction of a barbiturate coma using either pentobarbital or thiopental (see Barbiturate Coma in the Management section under Status Epilepticus).
Plasmapheresis to remove circulating cytokines and other inflammatory mediators is an accepted mode of therapy in children with acute disseminated encephalomyelitis who are refractory to steroid therapy.67,89,105 Before initiating this mode of therapy, a large-bore double-lumen central venous catheter (e.g., a hemodialysis catheter) is placed. Intravenous immune globulin (IVIG) may also be used in children with encephalitis who are unable to produce an effective immune response.32,114
Children with encephalitis should be monitored closely for signs of neurologic deterioration that may indicate greater inflammation or the development of increased ICP. Analgesics that do not produce respiratory depression (e.g., codeine) can be prescribed to relieve a persistent or severe headache. If the child complains of sensitivity to light or noise, a private room or isolated bed space is usually necessary to enable reduction of room light and noise and to minimize stimulation.
Children with encephalitis may demonstrate mild symptoms and a rapid recovery or may develop progressive and fatal neurologic deterioration. The prognosis is determined by the causative agent and the general health of the patient.
Metabolic Encephalopathies
Etiology
Metabolic encephalopathies can be divided into three subgroups: endogenous intoxication related to accumulation of neurotoxic metabolites, energy failure related to a lack of metabolites needed for brain function, and acute water-electrolyte-endocrine disturbances.
Pathophysiology
In several disease processes, including liver failure and inborn errors of metabolism, the products of amino acid catabolism are not cleared or detoxified by either the liver or kidneys. These waste products (e.g., ammonia and urea) accumulate and affect organ function, often causing neurologic problems such as cerebral edema. Examples of these disorders include maple syrup urine disease, methylmalonic aciduria, and propionic aciduria.
Energy failure encephalopathies include hypoglycemia, thiamine deficiency, and mitochondrial energy metabolism defects. Water and electrolyte encephalopathies include diabetic ketoacidosis, nonketotic hyperosmolar coma, hyponatremia, and hypernatremia (see Chapter 12). Cerebral edema again is the cause.66,117
Clinical Signs and Symptoms
Children with metabolic encephalopathy can exhibit progressive confusion, motor and sensory deficits, hallucinations, and seizures. Respiratory failure develops as the level of consciousness deteriorates. Signs and symptoms of cerebral edema and increased ICP may also be present. (See Increased Intracranial Pressure in the Common Clinical Conditions section of this chapter.)
Management
Treatment is determined by the etiology of the encephalopathy. Patients usually are admitted to the critical or intermediate care unit for skilled continuous nursing observation. Initial priorities of care include establishing and maintaining an adequate airway, ventilation, oxygenation, and systemic perfusion. Careful monitoring for signs of increased ICP is imperative. Hypoventilation and hypercapnia will further exacerbate the already increased ICP. If the child is unable to maintain a patent airway or breathe spontaneously, elective intubation and mechanical ventilation are indicated before the development of airway obstruction, hypoxia, and hypercarbia (see Chapter 9).
Ischemic and Hemorrhagic Stroke
Etiology
Stroke is an acute loss of blood flow to a region of the brain that results in a loss of neurologic function. More than 3200 new strokes are diagnosed annually in children younger than 18 years.38,107 Approximately 30 neonates per 100,000 births are affected; in the pediatric population stroke incidence is approximately 2 to 13 per 100,000 children per year.38 The remainder of this section addresses stroke in infants beyond the neonatal period and in children.
Ischemic strokes usually occur within the first year of life, and subarachnoid hemorrhages are more common in the teenage population.39 Those at high risk are males and African-Americans.38,39 Prognosis in children is much better than in adults.
Cerebral vascular accidents and strokes are rare occurrences in children, but carry significant morbidity and mortality. Stroke is among the top 10 causes of childhood deaths, and more than 50% of survivors have cognitive and motor dysfunction. Increasing prevalence is likely related to improved imaging and recognition in addition to advances that allow children with predisposing conditions such as cancer, sickle cell disease, heart disease and neurologic disease to survive longer.38,39,65,99
Pathophysiology
Many factors and disease processes predispose children to strokes. A preexisting condition that increases the child's risk of stroke is identified in only about half of pediatric stroke victims. Common preexisting conditions include congenital heart disease and sickle cell anemia. Other known causes are leukemia, brain tumors, Down syndrome, trauma, recent infection, vasculitis, and bleeding disorders. Strokes can be classified as either ischemic or hemorrhagic.
Ischemic strokes are the result of either a thrombus or embolism and are responsible for approximately 85% of all strokes. Emboli generally originate in the heart, but extracranial arteries are another source. Thrombi are occlusive, and the most common sites are in the cerebral artery branches. Turbulent blood flow can also increase the risk for thrombus formation. Other causes include sickle cell disease, protein C disorders, polycythemia, hypoperfusion, anemia, and prolonged vasoconstriction.
Hemorrhagic strokes are intracerebral hemorrhages affecting the brain parenchyma. These hemorrhages can occur in small arteries damaged from hypertension, or they result from rupture of aneurysms, arteriovenous malformations, bleeding disorders, or cocaine abuse.99
Clinical Signs and Symptoms
Acute stroke in children can produce signs and symptoms such as nausea, emesis, headache, altered mental status, abrupt onset of hemiparesis, visual disturbances, ataxia, and aphasia. Symptoms may be single or occur in combination. It is imperative to establish the precise time of the onset of symptoms, because it can affect the potential use of thrombolytics for ischemic stroke.
Management
All children suspected of having an acute stroke require blood sampling on admission for initial serum chemistries (electrolytes, glucose, calcium, and BUN), complete blood count, and coagulation studies, including activated partial thromboplastin time (aPTT). A comprehensive urine toxicology screen is performed to rule out any toxins or drugs that can produce signs similar to a stroke. Additional testing may include lupus anticoagulant; antiphospholipid antibody; dilute Russell viper venom time; anticardiolipin immunoglobulin (Ig) G, IgM, IgA; antiphosphotidyl serine IgG, IgM, IgA; anti-β2 glycoprotein IgG, IgM, IgA; fasting lipid profile; cholesterol; lipoprotein A; plasma homocysteine; varicella titers; organic and amino acids; factor V Leiden; antinuclear antibody, erythrocyte sedimentation rate; and C-reactive protein. The child is evaluated for mitochondrial myopathy, encephalomyopathy, lactic acidosis, and stroke—a family of inherited mitochondrial cyopathies.
Noncontrast CT, CT angiography, and MRI are the mainstays of imaging for stroke diagnosis. The noncontrast CT is completed and read soon after the child's arrival to quickly differentiate ischemic from hemorrhagic insults. In adult acute stroke guidelines, the recommended goal for completion of the CT image is 25 minutes or less from the time of hospital arrival, with CT image interpretation within 45 minutes of hospital arrival.4 Although the sensitivity and specificity of CT has not been shown in children, expert consensus supports its use.107 However, it is often necessary to stabilize the child's condition before the CT scan.
CT angiography can identify filling defects and localize specific portions of the vessel that may be the cause of the stroke. MRI is sensitive in detecting both acute ischemic and hemorrhagic strokes. A cardiac echocardiogram should be obtained to rule out cardiac embolism as a cause of the stroke. Children with suspected strokes are admitted to the critical care unit for skilled continuous nursing observation. Obviously, establishing and maintaining an adequate airway, ventilation, and systemic perfusion are priorities.
Thrombolysis
A neurologist should be consulted to assist with both acute and long-term management of the pediatric stroke patient. Treatment with tissue plasminogen activator (tPA) has become a standard of care for adult ischemic stroke treated within hours of the event. No randomized studies have evaluated the use of tPA for stroke in children <18 years of age, so the FDA has not yet approved tPA for this use. Although small case series have documented tPA clot dissolution in children with systemic thromboses, the complication rate was high.107
If given within 3 hours of stroke symptom onset, tPA may be considered for some patients with occlusive ischemic stroke. In carefully selected adult patients, IV tPA may be given up to 4.5 hours after symptom onset, and intraarterial tPA may be administered up to 6 hours after symptom onset in carefully selected adult patients with anterior circulation strokes.107
Heparin therapy is the mainstay for anticoagulation in the pediatric stroke patient. The use of heparin can prevent repeat cardiac embolic events and inhibit further development of cerebrovascular thrombi. After imaging has ruled out hemorrhage, the child will be given a bolus dose of heparin (75 units/kg; maximum, 10,000 units) and given a heparin infusion of 15 units/kg per hour. Blood should be obtained for an aPTT 1 hour after the heparin infusion. The infusion will be adjusted with the goal maintaining the aPTT in the range of 60 to 85 seconds. Hematologists should be consulted to assist with transition to low molecular weight heparin or warfarin.
Children with hemorrhagic stroke require immediate transfer to the pediatric critical care unit for close neurologic and hemodynamic monitoring. An emergency craniectomy may be required if the lesion has a mass effect on the brain. Failure to intervene quickly can lead to increased ICP and subsequent brain death (see Increased Intracranial Pressure earlier in this chapter).99
Drowning
Etiology
The term drowning is used to describe a submersion event producing primary respiratory impairment.87 The victim typically requires some form of resuscitation (stimulation, rescue breathing, or compressions and ventilations). After a drowning event, the child may survive with or without significant neurologic impairment.
There are three categories of children at risk for drowning injuries: infants, children 1 to 4 years of age (toddlers and preschoolers), and adolescents. Infants usually drown in bathtubs and children 1 to 4 years of age are typically found submerged in shallow wading pools, home swimming pools, or spas.22,33 Adolescents often drown in natural bodies of water, and alcohol or diving injuries are often involved.
Pathophysiology
The pulmonary complications of submersion are summarized in Chapter 9. The following paragraphs address only the potential neurologic complications of submersion. Within 3 minutes of submersion, most patients will develop sufficient hypoxia and cerebral ischemia to produce loss of consciousness. If submersion continues, CNS dysfunction develops; further ischemia and hypoxia can produce brain death.
The hypoxic-ischemic insult during the submersion may not be immediately fatal. Cardiopulmonary resuscitation can produce the return of spontaneous circulation. However, profound cerebral cellular damage, cerebral edema, and reperfusion injury may develop. This edema may produce signs of increased ICP as late as 48 to 72 hours or longer after the submersion episode.
The severity of neurologic sequelae following submersion is related to the severity of the primary hypoxic insult and any secondary insults that occur. The severity of the primary insult is affected by the duration of immersion, the temperature of the submersion water, and the time that elapsed before effective cardiopulmonary resuscitation was provided. Secondary neurologic insults (e.g., hypotension, further hypoxic episodes, hyperthermia, decreased CBF) may occur after initial return of spontaneous circulation.87 The time of submersion as reported by bystanders is notoriously unreliable, so it is often impossible to determine the duration of cardiopulmonary arrest.
Submersion can stimulate the diving reflex. This reflex results in initial apnea, loss of consciousness, bradycardia, hypertension, and shunting of blood to vital organs and away from the skin and splanchnic vascular beds. When very small children are submerged in very cold water (<5° C), the diving reflex can slow metabolic rate and redistribute blood flow sufficiently to prevent profound neurologic injury. However, such protection cannot be assured, and intact survival following prolonged submersion is rare.22
Clinical Signs and Symptoms
Unless the submersion is extremely brief, most children are apneic and flaccid when pulled from the water. If high-quality cardiopulmonary resuscitation is immediately initiated, many of these children will demonstrate a perfusing cardiac rhythm and spontaneous respirations on arrival in the emergency department; these children are likely to recover completely from the episode. The presence of any spontaneous movement or posturing on arrival in the emergency department and even within the first 24 hours after submersion is consistent with neurologic recovery.87
No predictive factors evaluated during resuscitation can determine the outcome of drowning victims, so aggressive resuscitation and post-resuscitation care are generally indicated for the first hours following the submersion.87
If skilled resuscitation was performed at the scene and during transport, and the normothermic child is asystolic on arrival in the emergency department, reported outcome is poor.22 Additional poor prognostic indicators include absence of purposeful movement at 24 hours after admission.87
Serial neurologic examinations are required for children who remain comatose or severely impaired after return of spontaneous circulation and admission to the critical care unit.87 If these children receive aggressive hemodynamic support, it may be possible to restore effective systemic perfusion, but the child with severe hypoxic brain injury may never recover significant neurologic function. Therefore, the indication for aggressive or prolonged resuscitation beyond the first 24 hours should be considered carefully, and the parents should be included in these discussions.
Management
If the drowning victim is responsive after resuscitation, further neurologic support is typically not required. The child should be monitored closely, and aggressive respiratory support may be needed to treat pulmonary complications (see Chapter 9).
If the child with severe neurologic injury is supported vigorously during the first hours after the drowning episode, the child may demonstrate some gasps within 12 to 24 hours. However agonal gasps indicate brain stem function and do not indicate neurologic recovery.
Signs of increased ICP can develop 48 to 72 hours after submersion. There is currently no evidence that aggressive therapy to treat increased ICP and limit secondary neurologic injury is beneficial for drowning victims with increased ICP. 22,87 At this time, immediate resuscitation and therapeutic hypothermia are the only therapies that have been shown to improve neurologic outcome following hypoxic-ischemic brain injury, and evidence comes largely from the neonatal population. More data are needed regarding effectiveness of therapeutic hypothermia in children following resuscitation.
The parents of the drowning victim will need a great deal of compassionate support. They should be included in discussions and decisions to limit care or to pursue aggressive resuscitation and postresuscitation support (see Withholding and Withdrawing Care in Chapter 24).
If the child develops brain death, the parents should be offered the option of organ donation (see the Brain Death and Organ Donation section of this chapter). Support of the parents is reviewed in Chapter 3.