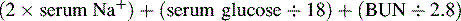Irreversible Condition
Requires observation over time (proposed length of observation time increases during infancy)
Absence of complicating factors
Adequate resuscitation provided
Absence of Brain Function
Flaccid paralysis (no posturing, no response to central pain stimulus)
Absence of brain stem and cranial nerve function:
No pupil response to light
No corneal (blink) reflex
No oculocephalic reflex (doll’s eyes)
No oculovestibular reflex (cold water calorics)
Apnea despite documented PaCO
2 >55-60 mm Hg or despite rise in PaCO
2 of >20 mm Hg
Possible Confirmatory Tests
Electroencephalogram (requires special electrode placement and amplitude, absence of sedative drug levels, normothermia)
Radionuclide angiogram
Carotid arteriogram
Magnetic resonance angiography
Reliability of additional tests in children, including cold xenon blood flow study, brain stem-evoked potentials, and Doppler cerebral blood flow studies are under investigation
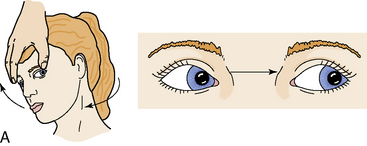
The oculocephalic reflex is tested (testing of “doll's eyes” reflex) by briskly rotating the head from midline to the side while holding the eyelids open to observe eye movement. If the oculocephalic reflex is intact, head rotation is detected by the semicircular canals (innervated by the vestibular branch of the acoustic nerve) and neck proprioceptors, and they will stimulate the third (oculomotor) and sixth (abducens) cranial nerves to rotate the eyes in the sockets in the direction opposite head rotation (i.e., if the head is turned from midline to the right, the eyes will rotate to the left). This normal eye rotation is designed to help focus on objects despite head rotation. When the oculocephalic reflex is absent, the eyes will remain fixed in their sockets, despite rotation of the head (Fig. 11-16).The nurse can imagine the eyes painted on the face of a doll—that is, they will not move in the sockets. The reflex will not be present when brain function ceases.
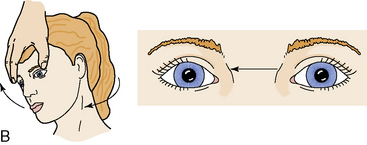
Testing of the oculovestibular reflex (the cold water calorics test) tests brainstem function by stimulating the vestibular branch of the eighth (acoustic) cranial nerve as well as the function the third (oculomotor) and sixth (abducens) cranial nerves. If the brain stem is intact, the cold water will have the same effect on the semicircular canals as head rotation, resulting in stimulation of eye movement. This test should be performed only if the tympanic membrane is intact and the patient is unconscious. If the reflex is tested/elicited in conscious patients, it will produce eye movement, but typically produces severe vertigo and nausea.
To perform the test, the head of the bed is elevated 30 degrees and ice water is quickly instilled by syringe (without a needle) deep into the ear canal. If the oculovestibular reflex is intact, bilateral conjugate eye movement will occur with slow horizontal nystagmus initially toward the stimulus, and then rapid nystagmus away from the stimulus. If the reflex is abnormal, disconjugate or asymmetrical eye movements will occur. If the reflex is absent, the eyes will remain fixed in midposition (Fig. 11-17).
Testing of the cough, gag, cornea, and pupil reflexes is relatively simple. A suction catheter inserted into the back of the pharynx normally will stimulate a cough or gag reflex; if brain stem function is absent, neither response is observed. When the brain stem is intact, an automatic blink will occur when any object approaches the eye; when brain stem function is absent, the cornea can be stroked lightly with a cotton-tipped applicator, and a blink will not be observed. When the brain stem is intact, the pupils will constrict in response to light; when brain stem function is absent, the pupils will be fixed and unresponsive to light. The pupils typically are fully dilated when brainstem function ceases, although they may be midsize.
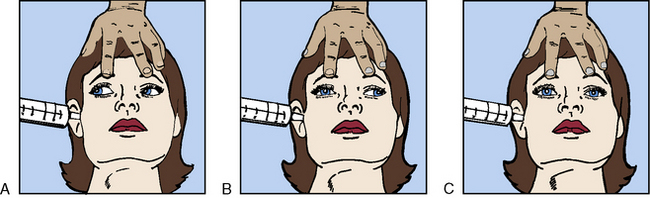
The presence of apnea must be carefully assessed and documented (Box 11-8). Apnea can be documented if the patient fails to demonstrate spontaneous ventilation despite an arterial carbon dioxide tension exceeding 55 to 60 mm Hg. The arterial carbon dioxide tension must be documented to be sufficiently high to stimulate ventilation. Before initiating the test, support of ventilation is adjusted to ensure normocarbia, and preoxygenation (using 100% oxygen) is provided to ensure adequate oxygenation. The apnea test should begin, if possible, with an arterial carbon dioxide tension of 35 to 40 mm Hg.
Box 11-8 Apnea Test to Document Absence of Spontaneous Respirations
1. Before study, adjust mechanical ventilation support (if needed) to ensure normal oxygenation and normocarbia (PaCO2 = 35-45 mm Hg). Obtain arterial blood gases before and at conclusion of study. Monitor pulse oximetry throughout study to ensure effective oxygenation. Monitor exhaled CO2 monitoring, if possible, to estimate PaCO2 during study.
2. Before study, preoxygenate patient for 5-10 min using 100% oxygen.
3. Remove patient from ventilator and provide passive oxygenation with 100% oxygen (6 L/min or twice the minute ventilation appropriate for child’s weight) delivered to endotracheal tube (using blow-by tubing or T-piece) or via suction catheter (joined to green oxygen tubing) inserted into endotracheal tube.
4. Closely observe patient during 5-10 min duration of study. No chest movements or respiratory effort will be present if brain function has ceased. Abort study if cardiovascular deterioration occurs.
5. During the study, titrate oxygen support as needed to ensure effective oxygenation (per pulse oximetry). When exhaled CO exceeds 60 mm Hg or the 5-10-min observation period has elapsed, draw arterial blood gas sample. The PaCO2 will rise approximately 4 to 5 mm Hg/min during apnea.
6. To confirm apnea, no respiratory effort can be noted, and the PaCO2 at end of study should exceed 55-60 mm Hg.
Oxygen must be provided during the apnea test to avoid hypoxemia; pulse oximetry should be used to monitor systemic oxygenation during the study. Supplementary oxygen can be delivered with a T-connector joined to the endotracheal tube. However, many providers prefer to administer the oxygen directly into the endotracheal tube. To accomplish this, a suction catheter is joined to standard green oxygen tubing, and the suction catheter is inserted into the endotracheal tube. Oxygen then flows through the suction catheter into the endotracheal tube. If the child becomes hypoxemic during the test, it may be necessary to interrupt the test and resume support of mechanical ventilation.
An arterial blood gas sample usually is obtained at the beginning of the study to document normocarbia. Correlation between the exhaled carbon dioxide (PETCO2) and the PaCO2 can be established with the initial blood gas, and then the PETCO2 can be monitored during the study. Based on the initial PaCO2, providers can predict the length of time needed to raise the PaCO2 to 60 to 65 mm Hg. During apnea, the PaCO2 will rise approximately 4 to 5 mm Hg for each minute of apnea97; therefore, it will require approximately 4 minutes of apnea to raise the PaCO2 from 40 mm Hg to 56 to 60 mm Hg. Remember that this is an estimate however, and formal documentation is necessary for the apnea test to be valid.
During the apnea test, the nurse must remain at the bedside, watching the child closely for any evidence of respiratory effort. In addition, the nurse must monitor the child's heart rate and systemic perfusion. At the end of a 5- or 10-minute observation period, an arterial blood gas sample is taken and mechanical ventilation is resumed.
Confirmatory and Adjunct Tests
Confirmatory tests are unnecessary in children older than 1 year when the cause of brain function cessation has been established and is irreversible, and there is no evidence of brain function on clinical examination. These patients may be examined twice (with the examinations separated by 12-24 hours) and pronounced brain dead at the time of the second examination.
The Task Force on Brain Death Determination in Children (TFBDDC) recommends electroencephalography as the confirmation test of choice for children younger than 1 year and for those with hypoxic ischemic cerebral insult.121 The EEG was favored at the time of the recommendations because clinical experience with the EEG during childhood was more extensive than the clinical experience with xenon perfusion scans or brainstem evoked potentials. However, a “technically satisfactory radionuclide angiogram” also was noted as being acceptable by the Task Force.
If the EEG is performed to confirm brain death, the technician must be informed that the study has been ordered to confirm brain death. For such studies, the electrodes are placed farther apart than for a normal EEG, and the voltage is increased. A long, uninterrupted recording is made at the end of the EEG brain death study to document 30 minutes of electrocerebral silence.
Slow EEG activity can persist despite brain death. EEG activity persisted in as many as 25% of adult donors following the pronouncement of brain death.51 EEG activity has also been shown to persist in children, despite the absence of CBF on angiography.15-17 A case study documented the return of cerebral activity following a flat EEG during the neonatal period.15 These reports of false positive and false negative EEGs raise doubts about the reliability of the EEG as a confirmatory test of brain death.
An added disadvantage to the use of the EEG as a confirmatory test is that serum concentrations of any sedative drugs and barbiturates present must be subtherapeutic, and electrolytes must be normal. The body temperature must be higher than 32° C. If a barbiturate coma has been induced, several (3-5) days may elapse before the serum barbiturate concentration is sufficiently low to allow the first EEG.
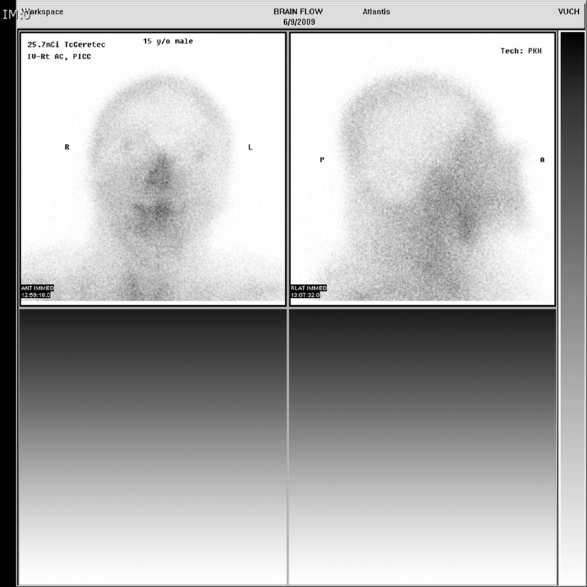
Many centers use cerebral angiography or radionuclide brain flow scans to determine the presence or absence of cerebral perfusion131 (Fig. 11-18) as an adjunct to the clinical determination of brain death. Although the reliability of these studies has not been reported with a large series of children, no significant issues of reliability have been raised for older infants and children. These blood flow studies are not influenced by the presence of electrolyte imbalance or barbiturates. Additional data suggest that brain stem-evoked potential, transcranial Doppler, and magnetic resonance angiography may be useful adjunctive tests.19
Observation Period
The Task Force on Brain Death Determination in Children recommended repeated clinical examinations, separated by an observation period, to ensure that irreversible cessation of brain function has occurred. The younger the child, the longer the suggested interval between examinations.125 Some institutions adhere strictly to these criteria, and others use cerebral perfusion studies to shorten the observation time or replace the EEG. It is important for the nurse to be familiar with the hospital protocol and ensure strict adherence to and documentation of the protocol.
Infants 7 Days to 2 Months of Age
The Task Force recommends two examinations separated by at least 48 hours. Additional confirmation is recommended using two EEGs separated by at least 48 hours.121
Infants 2 Months to 1 Year of Age
The Task Force recommends two examinations separated by 24 hours. One EEG consistent with brain death is recommended, and a cerebral radionuclide angiographic study can be substituted for the second EEG.121
Beyond 1 Year of Age
The Task Force recommends two examinations separated by at least 12 hours. A 24-hour observation period is suggested if the cause of death is a hypoxic-ischemic insult. Confirmatory tests or adjunct studies are not suggested.121
Brain Death in the Neonate
Standards are still evolving for the pronouncement of brain death in the neonate.18,19,121 After the Task Force on Brain Death Pronouncement in Children guidelines became available, additional studies suggested that brain death can be pronounced in the neonate and preterm infant older than 34 weeks' gestation.18 Diagnosis of brain death in this group can use the standard clinical examination and confirmatory testing, but the clinician should be knowledgeable about the challenges of declaring brain death in the neonate. A 48-hour observation period is recommended between examinations. The cause of brain death should be determined and the timing of cessation of brain function established, if possible. Repeated examinations are performed, and confirmatory or adjunct studies are almost always performed, although EEG activity may be present despite the absence of CBF.18
Contact with the Local Organ Procurement Organization
Federal law requires that the hospital notify the local organ procurement organization (OPO) about a potential organ donor. A coordinator from the OPO will determine whether there are any absolute or relative contraindications to organ donation. Such contraindications are few, but they should be ruled out so that the family does not become hopeful about organ donation that is not feasible (Box 11-9).
Box 11-9 Donor Contraindications to Solid Organ Donation
Untreated sepsis
Organ ischemia, inadequate resuscitation
Some systemic diseases (always consult with local federally funded organ procurement agency)
Significant organ dysfunction
The OPO coordinator will explain the donation process to the parents and answer their questions. The coordinator is skilled in dealing with parents during an extremely stressful time and can provide the parents with current, accurate information. The coordinator will write to the family months after the donation (if the family desires), to inform them in general terms about the recipient of their child's organs. Parents often find this information extremely comforting. Parents should be aware that there is no cost to them for the organ donation, and that the process will not delay or alter standard funeral arrangements.
If the child's parents consent to organ donation, a written consent is required and the OPO assumes all financial responsibility for donor care once the child is pronounced brain dead. If any diagnostic studies are obtained before the pronouncement of death that will contribute to the donation process, those costs also are assumed by the OPO. Once pronouncement of death has occurred, the donor coordinator participates in the management of systemic and organ perfusion until actual donation takes place. Some OPOs will reimburse the hospital for the cost of nursing care of the donor between brain death pronouncement and recovery of the organs.
Once the parents sign the consent for organ donation, the child's age, weight, blood type, and available organs are listed on a national website via the United Network for Organ Sharing. If the organs are compatible with several potential recipients, priority is given to the most severely ill local recipient. Organs are distributed at a regional level before they are available at a national level. It is extremely important that both donor and recipient families realize that these organs are distributed fairly according to strict criteria developed by the United Network for Organ Sharing.
Psychosocial Support of the Family
The death of a child is always tragic. Because brain death often is the end result of a tragic injury or illness, the parents are often physically and emotionally exhausted when death is pronounced. When brain function ceases, it can be extremely difficult for the parents to wait for confirmation that their child has, in fact, died. Often, brain death pronouncement is attempted to remove the burden of the decision to withdraw support and to enable donation of the child's organs. Therefore, it is important that the parents be approached in a sensitive and compassionate manner about this issue (see Chapters 3 and 24).
Organ donation can be life saving for the recipients who are dying of organ failure. Since the inception of the Health Resources Services Administration's Organ Donation and Transplantation Breakthrough Collaboratives in 2003, the number of organ donors has increased. Yet, hundreds of children awaiting organ transplantation will die each year before a donor is located.129 Organ donation does not benefit only the recipient, however. Parents have expressed gratitude that something positive could come out of the tragic death of their child. Parents have stated that they feel a part of their child is able to survive through the gift of organ donation. In a national survey of pediatric organ donation, approximately two-thirds of families (range, 63%-75%) of eligible pediatric donors consented to donation, with highest consent rates occurring when the potential donor was 12 years of age or older.129
The family's decision about organ donation can be influenced by religious or cultural beliefs.45 However, the family's interaction with healthcare providers and the manner in which the subject of organ donation is introduced can have a profound effect on the family's decision and memories of the child's death (see Chapter 3).
The nurse is typically the member of the healthcare team who is closest to the family of the dying child. Although it is necessary to offer hope to parents of dying children, the nurse is often the best member of the healthcare team to help the family prepare for their child's death. If open visitation is in place and parents are allowed to participate in their child's care, they can see that everything possible was done for their child. However, if parents are separated from the child for most of the child's final hours, it will be more difficult for the parents to be aware of the efforts to save the child's life.
Each healthcare team and OPO will determine the best person to approach the family about organ donation. Often, family members will introduce the subject of organ donation; if not, the nurse will be aware of the best time to begin to discuss potential organ donation with the family. This discussion must be compassionate and accurate. Parents should not be approached about organ donation if there is still hope that the child will survive. The bedside nurse should be aware of the hospital and unit protocol and be available to support the family through this difficult period.
Understandably, the family will be easily confused by inconsistent statements and terminology from members of the healthcare team. When a child is brain dead, it is possible to maintain oxygenation, ventilation, and perfusion, but the ventilator does not keep the child alive. The ventilator is merely moving air into and out of the child's lungs. Parents must be aware that the diagnosis of brain death is made according to established protocols, and there is no possibility of error when these protocols are followed strictly.
Parents should be allowed to remain with the child as much as possible during the time preceding the child's death, and they should be asked whether they would like to stay with the child while the brain death examinations are performed. Many parents express the wish to be alone with the child before the final examinations and studies are performed, to say goodbye before they are informed that the child has died.
Support of the Cadaveric Donor
Maintenance of the cadaveric donor requires expert critical care to support excellent perfusion to solid organs. This care requires close coordination among members of the healthcare team and with the organ donation coordinator. If medications are administered to the donor, the recipient transplant surgeons may be consulted regarding potential effects of the drugs on the organs to be donated.
Because several transplant teams may be coming to obtain organs from the same donor, the organ procurement coordinator and the bedside nurse are often responsible for integrating the orders of teams from several hospitals. This responsibility requires skill and flexibility. The nurse should be aware that the goals of management change after brain death is pronounced. The goal then becomes provision of adequate substrate and oxygen delivery to the organs to be donated, rather than maintenance of brain perfusion and function.
At the time of brain death pronouncement, most organ donors are relatively hypovolemic for several reasons. They usually have been treated for increased ICP with fluid limitation and osmotic diuretics, and the loss of brain stem regulation of vascular tone and blood pressure results in the vasodilation and expansion of the vascular space. Finally, diabetes insipidus (DI) can develop in as many as 90% of donors,75 and unreplaced fluid lost in the urine can result in the rapid development of hypovolemia and inadequate organ perfusion.75 Fluid administration with frequent bolus therapy of isotonic crystalloid (20 mL/kg) is generally required.68
Cardiovascular dysfunction is common in the pediatric donor. Arrhythmias and hypotension are encountered in more than half of donors. The most common arrhythmias include bradycardia and ventricular arrhythmias.68
Electrolyte imbalances also are encountered frequently, particularly hypokalemia, hypernatremia, and hyperglycemia. If DI is present, urine losses should be replaced with an equal volume of 0.2% or 0.45% sodium chloride in order to minimize hypernatremia and maintain euvolemia. If urine losses are excessive, exogenous antidiuretic hormone (ADH or vasopressin) often is administered. Although the administration of this hormone can result in reduced liver and renal perfusion, this condition is usually preferable to the fluid and electrolyte imbalances that can accompany urine volume loss and attempted replacement.
If possible, blood pressure and systemic perfusion are maintained through volume administration alone. If it is necessary to administer an inotropic or vasopressor medication, a low dose of dopamine (<10 mcg/kg per minute) has traditionally been used; however, more recent data support the use of vasopressin as the therapy of choice for both hemodynamic instability and DI.75 If large doses of vasoactive medications are required to maintain systemic perfusion, myocardial injury or dysfunction may be present. Thyroid hormone may be administered to help stabilize hemodynamic status and allow for weaning of inotropic and vasopressor medications.109,110
The temperature of the donor must be maintained. An overbed warmer or warming blanket usually is required because the hypothalamus is no longer functional. Hypothermia should be prevented because it may further depress myocardial function.68
Care of the Donor after Cardiac Death
The protocol for care of patients who become candidates for donation after cardiac death varies by institution. This donation often follows a devastating neurologic injury that does not result in brain death. In this population, the family is approached about potential organ donation only after a decision has been made by the family and healthcare team to withdraw support.
The subject of donation is usually introduced by a member of the OPO staff or others trained in this discussion. If the family agrees, arrangements are made to withdraw support as would normally occur. Once cardiac death is pronounced, the patient is then taken to the operating room for removal of organs, usually the kidneys and liver. The amount of time between pronouncement of death and retrieval of organs is usually approximately 5 minutes, making team coordination crucial.
Withdrawal of care can take place in the critical care unit, operating suite, or another location. The role of the nurse is to provide the same care normally provided for the patient, including support the grieving family and assistance in transporting the donor to the operating room, if necessary, after cardiac death occurs (see Chapter 3 for more information).
The concept of and procedures for donation after cardiac death are evolving. This issue remains one of ethical concern for many, and widespread acceptance and implementation remains a challenge for the future (see Chapter 24).
The Emotional Toll on the Nurse
It is extremely difficult to care for a dying child and to support the family. At the very time that the child requires the most attentive physical care, the family requires the most sensitive emotional support. The bedside nurse should ask for help at the bedside to be able to spend time with the family. Additional support from chaplain services or social work specialists can be utilized.
When a nurse is closely involved with the child and family, the nurse must have the opportunity to grieve about the child. If at all possible, before the nurse begins to care for the organ donor, the nurse should take a few minutes to think about the child and family and derive some comfort from the fact that they were given the best support possible.60
Care of the organ donor is extremely hectic, but it can also be rewarding. Many nurses have voiced satisfaction about participation in the donor process because they could see something wonderful come from a tragedy. For further information about the care of the dying child and family see Chapter 3.
Postoperative care of the pediatric neurosurgical patient
Care of the child after neurosurgery requires maintenance of vital functions, assessment of neurologic function, recognition and treatment of potential complications of neurosurgery, regulation of fluid and electrolyte balance, and emotional support of the child and family.
Postoperative assessment is facilitated if the nurse is able to perform a thorough preoperative examination of the child. This examination enables the nurse to quickly recognize changes in the child's condition or level of response. When a detailed preoperative assessment is not available, it is important to determine what the child's baseline status was before surgery in order to determine recovery goals. Thus, the following discussion includes the preoperative assessment.
Preoperative Assessment
The nurse must obtain as much information as possible about the child's preoperative neurologic function from the parents or primary caretaker. If the child is transported to the hospital by medical personnel, the nurse should receive a report from the medical team that cared for the child before transport and from the transport providers. The nurse will also obtain information from medical records from the transferring hospital, emergency medical services providers or transport team and from the parents or primary caretaker in person or by telephone.
It is important to know the child's normal motor activity, self-comforting measures, communication, cognitive and motor skills, sleep patterns, feeding preferences, and behavior when frightened or angry; all of this information will be helpful during the postoperative period. The names of family members, special friends, pets, and favorite activities should be noted in the nursing plan of care to enable the evaluation of level of consciousness through questions about familiar people or things.
The nurse also should note the presence and severity of any preoperative neurologic symptoms or deficits such as seizures, coma, blindness, cranial nerve palsies, delayed developmental milestones, abnormal posturing or motor activity, or abnormal or absent reflexes, to establish a baseline for evaluating postoperative progress or deterioration. The preoperative head circumference should be recorded in the care plan of every infant and young child.
The nurse will carefully evaluate and document the child's preoperative level of consciousness. Whenever possible, use descriptions rather than general classifications to avoid confusion. When the child is older and responsive, the nurse should ask the child specific questions about name, age, birthday, and normal activities. Alert, accurate answers are normal; confused answers and lack of response to painful stimuli are clearly abnormal. During infancy, evaluation of the level of consciousness will be made through observation of the infant's cry, response to auditory and tactile stimuli, feeding and sleeping behavior, and through evaluation of the infant's achievement of developmental milestones. A high-pitched, breathless cry is considered abnormal in an infant; extreme lethargy, poor feeding, and delay in achieving developmental milestones are all abnormal.
The nurse will evaluate and document the child's motor ability, including head control, grasp, strength and movement of extremities, and symmetrical withdrawal of all extremities following painful stimulus. The older child should be able to move all extremities, follow commands, and squeeze the observer's fingers upon request. In addition, hand use and strength should be symmetrical and equal bilaterally. Evaluation of antigravity muscles is important, because discrepancies can be appreciated most readily in these muscles. If the child is walking, abnormalities of gait and balance are important to note.
The child's reflexes will be evaluated preoperatively, including the child's pupil size and response to light. The presence of papilledema should also be noted (see the Increased Intracranial Pressure section of this chapter). The complexity and extent of the assessment will be determined by the child's general condition. If time permits, the nurse should observe or perform an assessment of the child's cranial nerve function (see Table 11-2). In general, it is too difficult to test the olfactory nerve in infants and young children. If tested, strongly pungent substances should be avoided, and familiar odors such as lemon, orange, or peppermint should be used.58 The evaluation of some reflexes, such as the oculomotor or oculovestibular reflexes, should not be performed in an alert, mobile, responsive child, but may be required if the child is comatose.
It is important to assess and document the child's preoperative cardiovascular function and systemic perfusion. Signs of adequate perfusion include warm skin with brisk capillary refill, consistent color tones, pink nail beds and mucous membranes, and strong peripheral pulses, with heart rate and blood pressure appropriate for age and clinical condition. If systemic perfusion is poor, cerebral perfusion can be compromised and the child's level of consciousness can deteriorate. The child's typical heart rate and blood pressure should be noted carefully for comparison with postoperative values (see pages inside front cover for normal pediatric heart and respiratory rate ranges and blood pressure ranges for age). The median systolic blood pressure for a child 1 to 10 years old is estimated by adding 90 mm Hg to twice the child's age in years; a systolic blood pressure lower than the sum of 70 mm Hg plus twice the child's age in years (for children 1-10 years of age) is definitely hypotensive.104 Signs of increased ICP are discussed in the Increased Intracranial Pressure section earlier in this chapter.
The child's respiratory status is assessed carefully preoperatively, because neurologic disease can produce characteristic respiratory patterns (such as Cheyne-Stokes respirations), apnea, or respiratory arrest (see Fig. 11-7). It is important to note the presence and strength of the child's gag and cough reflexes, because they will affect the child's ability to maintain a patent airway and handle respiratory secretions and oral or enteral feedings postoperatively. Increased ICP may produce central hypoventilation or apnea. Serious hypercapnia or hypoxemia must be corrected preoperatively, and the reports of the child's preoperative arterial blood gases must be readily available to serve as a basis for comparison during the postoperative period.
Evaluation of the child's preoperative fluid balance and general nutrition is important. The child who is admitted with SIADH will demonstrate hyponatremia and water intoxication (with potential cerebral edema). The child with diabetes insipidus (DI) may demonstrate massive intravascular volume depletion and hypernatremia. Careful evaluation of the child's preoperative fluid balance will aid in both perioperative and postoperative fluid administration. It is extremely helpful if the child's normal feeding behavior, food preferences, and sleep patterns are documented in the nursing plan of care so that attempts can be made to provide these postoperatively.
If neurosurgery is planned on an elective basis, the child (as age appropriate) can be prepared for the sights, sounds, and sensations encountered during postoperative care through a visit to the postoperative unit. A child life specialist, if available, can assist in the preoperative teaching process and address specific questions and concerns that the child or siblings may have. During these sessions the parents can clarify some of the child's concerns and reassure the child that they will be present throughout the child's postoperative care (see Chapter 2).
If the child is admitted to the critical care unit following trauma, intracranial hemorrhage, or acute illness, the nurse may want to spend a few moments with the parents and family before they enter the critical care unit for the first time. These moments may help prepare the parents for the sights and sounds of the critical care unit. At a later time it will be important to verify that the critical care unit policies and procedures have been discussed with the parents; however, such discussion should take place later, after the parents and family have visited the child. If the child has suffered major multisystem trauma, the family should be warned about the child's altered appearance. Too often the medical team welcomes the parents to the bedside of a recently stabilized child with the comment, “He looks good,” when the parents are confronted with the sight of a bruised or bloodied, puffy, unconscious child covered with tubes and bandages. The parents should be reassured that the child is not aware of his or her appearance, and that the phrase looks good refers to the child's neurologic status and vital functions. If at all possible, the nurses should clean the child as much as possible and partially cover the child with a colorful blanket so that tubes and incisions are not all immediately visible.
Preparation for Admission or Transfer to the Critical Care Unit
The nurse and respiratory therapists will assemble all equipment necessary for postoperative care at the bedside so it is ready for use before the child returns from neurosurgery. All IV administration equipment is ready for use at the bedside. Arterial and central venous pressure transducers, flush systems, and an ICP monitoring system should be prepared per unit policy if they will not arrive with the child.
Equipment to support airway and ventilation must be set up and ready for use at the bedside, including a pediatric mechanical ventilator, face mask or oxygen administration device, a functional bag and mask connected to an oxygen source, and pediatric intubation equipment. Resuscitation drugs and drugs used in the prevention or treatment of increased ICP (e.g., lidocaine, 3% saline and mannitol), those used to control status epilepticus (e.g., lorazepam, phenobarbital, and phosphenytoin), analgesics, sedatives and neuromuscular blockers (see Chapter 5) and steroids (i.e., dexamethasone) for control of local edema associated with brain tumors must be readily available.50,96,120 Most nurses prepare or obtain a computerized, precalculated emergency drug sheet based on the child's age and body weight. The pharmacy may provide such a sheet. The Broselow Resuscitation Tape is a quick reference that assists in determining proper equipment sizes and emergency drug doses (see Fig. 1-1).79
The neurosurgeon discusses the planned surgery and potential postoperative complications with the family to obtain informed consent before the procedure. It is extremely useful if the nurse who will care for the patient postoperatively is present for these discussions.
Postoperative Care
Initial Assessment
When the child returns from neurosurgery, the nurse receives a report from the surgical team while assessing the child's airway, oxygenation, and respiratory function or ventilation support, systemic perfusion, and evidence of neurologic function. First priority must be given to establishing airway, oxygenation, ventilation, and perfusion.
If the child is breathing spontaneously, the child is generally positioned to extend the neck, avoiding flexion or hyperextension. If the blood pressure is adequate and no spine injury is present, the head of the child's bed can be elevated approximately 30 degrees to maximize chest expansion and diaphragm excursion. Although this position will enhance cerebral venous return, its effect on actual cerebral perfusion varies so the position must be reevaluated based on the child's progress.
If ventilation is effective, the child's aeration will be equal and adequate bilaterally, and the respiratory rate will be appropriate for the child's age and clinical condition (see Chapter 9). A respiratory rate of 50/minute can be appropriate in a crying, vigorous infant, but this rate is fairly rapid for an adolescent. Conversely, a respiratory rate of 12/minute in a sleeping but arousable adolescent may be appropriate, but it is too slow for an infant.
Evidence of respiratory distress such as retractions, nasal flaring, grunting, stridor, apnea, gasping, or cyanosis should be reported immediately to an on-call provider. Inadequate ventilation can produce hypercapnia, hypoxemia, cerebral vasodilation, and increased cerebral blood flow (CBF) and increased cerebral blood volume (CBV), leading to increased ICP. If ventilation is inadequate, hand ventilation is provided with a bag and mask until intubation can be accomplished and mechanical ventilation can be provided. If the intubated child deteriorates suddenly, the nurse must check for tube displacement, tube obstruction, pneumothorax and equipment failure (recalled with the mnemonic “DOPE”).104
As soon as possible after admission of the unstable child, a blood sample is taken for blood gas analysis to confirm or rule out the presence of hypercapnia, hypoxemia, or acidosis. Based on the blood gas analysis results, oxygen administration and support of ventilation are initiated or adjusted.
The nurse should quickly assess the child's systemic perfusion. Signs of adequate perfusion include warm skin with brisk capillary refill, consistent color tones, pink nail beds and mucous membranes, and strong peripheral pulses, with heart rate and blood pressure appropriate for age and clinical condition. Hypotension may be the result of bleeding or hypovolemia and can compromise the child's CPP (CPP = MAP − ICP). Hypertension may be an early sign of increased ICP; if present, it should not be corrected unless an extremely high blood pressure raises concern about the development of hypertensive encephalopathy, because hypertension may be necessary to maintain the CPP.
If an ICP monitoring device is in place, the ICP should be monitored closely and the nurse should be alert for the development of clinical signs of increased ICP (see earlier sections of this chapter). If intraarterial pressure monitoring was not established during surgery and the child's condition is unstable, the nurse should request the insertion of an arterial catheter to enable continuous monitoring of the arterial blood pressure, calculation of CPP, and sampling of blood for laboratory analysis. If the child requires frequent venipunctures for blood sampling, vigorous crying can result in a rise in ICP every time blood samples are drawn.
The child's heart rate may vary if increased ICP develops, so close continuous observation of heart rate is required. Although bradycardia is one of the signs of a severe increase in ICP with imminent brainstem herniation, tachycardia may be an early sign of increased ICP. The heart rate should be appropriate for age and clinical condition; tachycardia is appropriate if the child is apprehensive, crying, febrile, or in pain.
A quick but thorough assessment of the child's neurologic status is needed as soon as possible after the child returns from surgery. Pupils should be equal and reactive to light; pupil inequality or sluggish response to light should be reported immediately to an on-call provider, because these signs can indicate the development of increased ICP. When pupil size and responsiveness are checked, the nurse should assess the corneal reflex and notify a physician immediately if it is absent.
The nurse should notify an on-call provider immediately of decorticate rigidity, decerebrate posturing, or absence of normal reflexes such as gag and cough reflexes assessed during suctioning of the child's airway. Normal withdrawal of an extremity is demonstrated by abduction (movement outward or away from the midline) when a painful stimulus is applied to the medial aspect of the extremity; lack of this withdrawal is abnormal and worrisome. The patient normally moves purposefully (grabs toward the nurse's hand) in response to a central painful stimulus (i.e., one applied to the trunk or by pinching the trapezius muscle).
The child is monitored at all times for seizure activity because seizures can develop following intracranial surgery or traumatic brain injury.83,96 If neuromuscular blocking agents are administered postoperatively, seizures may progress unrecognized because the neuromuscular blocking agents will prevent tonic-clonic movements. Whenever seizures are suspected in the paralyzed child or whenever unexplained fluctuations in heart rate or blood pressure, poor systemic perfusion, nystagmus, alternating dilation and constriction of pupils, or a disconjugate gaze develops, notify an on-call provider immediately; it may be necessary to obtain an EEG to confirm that seizures are present. Continuous EEG monitoring may be indicated to detect seizure activity or status epilepticus.
If an ICP monitoring system is in place, both the ICP and the calculated CPP are recorded. The nurse will need specific orders for actions to take if the ICP exceeds 20 mm Hg for more than 10 minutes or if the CPP falls significantly; a fall near or below 40 mm Hg is extremely worrisome and requires immediate treatment. (Additional management is discussed in the Increased Intracranial Pressure section earlier in this chapter.)5,13
Maintenance of Cardiorespiratory Function
As noted earlier, optimal cerebral perfusion requires adequate systemic oxygenation and ventilation (including normocarbia). If the child demonstrates increased ICP postoperatively, the child will remain intubated with mechanical ventilation. The child's PaCO2 is maintained between 35 and 40 mm Hg and the PaO2 at 100 mm Hg. Appropriate analgesia will be needed and sedation with or without neuromuscular blockade may be required (see Chapter 5).
Preoxygenation is needed before endotracheal suctioning, and the child may require premedication with lidocaine (1 mg/kg IV bolus) before suctioning or other noxious stimuli to prevent or limit increases in ICP. Suctioning should be brief with careful assessment of the child's color, arterial blood pressure, and ICP (if monitored). Generally, two nurses are needed to suction efficiently yet provide appropriate monitoring and minimize hypoxia and hypercarbia. If an inline suction system is used, careful airway suctioning may be accomplished with only one care provider. Suctioning of the airway should be interrupted if the child develops bradycardia, hypotension, hypertension, deterioration in systemic perfusion, or increased ICP.
If the child's condition is relatively stable and the child is breathing spontaneously upon arrival in the critical care unit, the goals of respiratory care are maintenance of adequate respiratory function and detection of early signs of respiratory insufficiency. Oxygenation is monitored by pulse oximetry and blood gas analysis. The child's airway is assessed and supported as needed, and ventilation is evaluated through clinical examination, monitoring of exhaled CO2 (PETCO2), and arterial blood gas analysis.
A nasogastric tube is inserted to decompress the stomach and prevent vomiting that may result in aspiration. Gastric decompression and drainage is especially important if the patient demonstrates inadequate cough and gag reflex.
Assessment of systemic perfusion, heart rate, and blood pressure will continue throughout the postoperative period. Extreme tachycardia, bradycardia, and systolic hypertension with a widened pulse pressure should be reported to an on-call provider immediately, because these signs can indicate the development of increased ICP.
After neurosurgery, most children will receive 80%-100% of maintenance fluid requirements consisting of 0.9% sodium chloride with appropriate dextrose added to maintain normoglycemia (Table 11-8). The child can develop hypovolemia if this fluid volume is inadequate to provide for maintenance requirements plus replace fluid lost via insensible losses, bleeding, urine output, and gastric suction. Hypotension and hypovolemia require prompt treatment, because poor systemic perfusion can significantly compromise cerebral perfusion. Occasionally, vasopressors may be required to increase the MAP and maintain the CPP.
Aggressive fluid administration is avoided unless it is needed to maintain systemic perfusion during treatment of shock. If shock resuscitation is required, bolus therapy with isotonic crystalloid or 3% saline is provided until systemic perfusion is adequate. Hypotonic fluids are not used because they are likely to produce a fall in the child's serum sodium concentration (see Chapter 12). Neurosurgery patients are likely to retain free water as the result of ADH secretion or SIADH; therefore, the child's fluid balance, urine output, and serum sodium concentration require close monitoring. If a central venous catheter is in place, the child's central venous pressure can provide helpful information about the child's intravascular volume status.
Neurologic Assessment
The nurse must be able to perform a rapid but thorough neurologic assessment to detect potential complications of neurosurgery, including acute neurologic deterioration. These potential complications include increased ICP, status epilepticus, SIADH, diabetes insipidus (DI), CSF otorrhea or rhinorrhea, and CNS infection. In most cases a thorough neurologic examination, ongoing assessment of vital signs and fluid intake and output, and evaluation of the child's complete blood count, serum electrolytes, and acid-base balance should alert the nurse to the development of any of these complications.
Because assessment, pathophysiology, and treatment of each of the major postoperative complications have been summarized in the Common Clinical Conditions section of this chapter, the following discussion is designed to highlight important aspects of the neurologic examination of the critically ill child and to provide a brief discussion of the major complications of neurosurgery. For more detailed information about these complications, please refer to the Common Clinical Conditions section earlier in this chapter.
Highlights of the Neurologic Examination
Evaluation of the level of consciousness is one of the most important aspects of the neurologic examination of a nonsedated infant or child. Because the infant is unable to communicate verbally, the nurse must evaluate the infant's alertness and response to the environment. The alert infant will awaken to auditory or tactile stimuli, visually track bright objects or lights, cry in response to painful stimuli, be comforted when held or fed, suck vigorously, and sleep when not disturbed, stimulated, or in pain.
The critically ill infant may be extremely irritable, reacting strongly to even mild stimulation. The irritable infant may cry often, will not be comforted when held or fed, will sleep or feed only for short periods, and may hold extremities rigidly. Lethargy is also a potential sign of neurologic dysfunction. When lethargy is present, the infant is difficult to arouse, seems uninterested in surroundings or feeding, will fail to maintain eye contact or respond to parents, and may demonstrate poor muscle tone and a weak suck.
Evaluation of the level of consciousness in the nonsedated child can rely heavily on the child's response to questions and ability to follow commands—ask the child to hold up two fingers, stick out his or her tongue, or wiggle toes. Sluggish or confused responses are usually signs of decreased level of consciousness. However, if the child is suffering from sleep deprivation, then drowsiness, confusion, or irritability can be appropriate. If questions used to evaluate the child's alertness include questions about familiar family members or activities (using information obtained from the parents or primary caretaker) the child may be more likely to respond, because such questions are less likely to be intimidating.
The intubated but ordinarily verbal child will be able to answer questions by nodding or shaking his or her head (or the child may hold up one finger for yes and two fingers for no). Rating of the child's level of consciousness should be performed using a standard rating scale, such as the GCS score (see Table 11-6).
The child's pupil size and response to light can be extremely sensitive indicators of intracranial events. With the development of increased ICP, one or both pupils dilate and begin to react sluggishly to light. If drugs are administered that can cause pupil dilation (e.g., large doses of atropine or atrovent) or the pupils have been medically dilated, then this should be noted prominently in the patient record and with a sign placed at the bedside. The pupils will dilate with pain, but they will remain reactive to light (see Fig. 11-8 for variations in pupil size that can result from neurologic disorders).
As noted previously, assessment of the child's vital signs is an essential part of the neurologic examination. Cushing's reflex causes a triad (often called Cushing's triad) of late clinical signs of increased ICP; these signs develop when brain stem herniation is imminent. The classic clinical triad includes bradycardia, systolic hypertension with widening pulse pressure, and irregular respirations. In children the change in heart rate can include tachycardia or bradycardia, and irregular respirations can include respiratory depression or apnea.
An important component of the neurologic examination and of the GCS score is assessment of motor function and reflexes (see Table 11-6); this assessment is particularly important if the child is unconscious (i.e., with no eye opening or verbal response). Decorticate rigidity and decerebrate posturing are abnormal (see Fig. 11-9). A positive Babinski's reflex is abnormal after the infant has begun walking (see Fig. 11-10).
The child should withdraw extremities in response to pain, and he or she should demonstrate corneal, gag, and cough reflexes (described under Clinical Signs and Symptoms for Increased Intracranial Pressure and for Coma). Any deterioration in responsiveness or motor function should be reported to an on-call provider immediately.
If the child has been awake and alert and is old enough to follow commands, the nurse can assess muscle strength by asking the child to move all extremities and to move each extremity against resistance. Testing strength of antigravity muscles may reveal unilateral muscle weakness (e.g., ask the child to extend both arms and close eyes; if the right arm falls, a left hemisphere lesion is suspected).
The nurse should ensure that the child moves all four extremities equally and appropriately and is able to sense light touch and pain. The nurse can evaluate motor coordination by asking the child to touch the nurse's index finger and then his or her own nose—even toddlers will perform this activity. Seizure activity should be reported to the provider and must be treated promptly.
Many of the cranial nerves can be evaluated while other nursing care is performed (see Table 11-2 and Box 11-2). For example, the glossopharyngeal (ninth cranial) nerve and vagus (tenth cranial) nerve are probably intact if the child coughs and gags during suctioning of the airway and if the child is able to swallow.
The child may complain of headaches, nausea, malaise, vomiting, blurred vision, diplopia, and poor feeding; these can be nonspecific symptoms of increased ICP and should be reported to the on-call provider. Unless a large head dressing is in place, the infant's head circumference is measured when the infant returns from surgery and at daily or other intervals per hospital protocol. An increase in head circumference can be caused by a gradual increase in intracranial volume, such as occurs with hydrocephalus or a subdural empyema.
Postoperative Complications
Increased ICP results from an uncompensated increase in the volume of blood, brain, or CSF within the skull. Hemorrhage, cerebral venous obstruction, or cerebral vasodilation can increase the child's CBV. The brain size can increase with cerebral edema, and the CSF volume can increase with obstruction to CSF flow or decreased CSF reabsorption. Although intracranial volume can initially increase without a rise in ICP, further small increases in intracranial volume will produce significant increases in ICP once a critical volume is reached (see Increased Intracranial Pressure).
Signs and symptoms of increased ICP are summarized in Box 11-3, and they include a decrease in the level of consciousness, pupil dilation and sluggish response to light, decreased motor function, hypertension, tachycardia or bradycardia, altered respiratory pattern, and ultimately apnea and cardiorespiratory arrest. If ICP monitoring is used, an on-call provider should be notified of rises in ICP or a compromise in calculated CPP.
The treatment of increased ICP includes control of CBV through maintenance of adequate oxygenation and normocarbia. If an extraventricular drainage (EVD) system is in place, CSF drainage (if ordered) can reduce CSF and intracranial volume. The use of hypertonic agents such as 3% saline or hyperosmolar agents such as mannitol can reduce cerebral edema. Analgesics, such as fentanyl, and sedatives, such as midazolam, can also help control increased ICP.
Acute hyperventilation is reserved for management of increased ICP when signs of impending herniation (i.e., Cushing triad of bradycardia, systolic hypertension with widening pulse pressure, and irregular respirations) develop.83 When all other methods of medical treatment fail, the child may be placed in a barbiturate coma to reduce cerebral metabolic requirements and ICP (see Management, Barbiturate Therapy, in the Increased Intracranial Pressure section of this chapter).
Status epilepticus is repetitive or continuous seizure activity for more than 30 minutes without a return to consciousness. Status epilepticus requires immediate treatment because it can produce cerebral vasodilation, an increase in cerebral metabolic requirements, and in an increase in ICP.
When the child receives neuromuscular blocking agents during the postoperative period, clinical evidence of seizure activity is masked. Therefore, the presence of seizures or status epilepticus should be considered if the child receiving neuromuscular blockade demonstrates wide fluctuations in blood pressure, sudden pupil dilation, or other unexplained deterioration in clinical status. Once herniation has been ruled out, an EEG will likely be necessary to identify seizures.
An individual seizure does not require treatment. Status epilepticus, however, must be treated immediately. In children, lorazepam (0.05-0.1 mg/kg IV), phenobarbital (10-20 mg/kg IV), fosphenytoin (15-20 mg (PE)/kg IV), are used most commonly120 (see Status Epilepticus, in the second part of this chapter). Monitoring and support of therapeutic serum drug levels is also needed.
SIADH frequently develops after neurosurgery. Clinical signs and symptoms of this syndrome include hyponatremia, persistently high urine sodium concentration, high urine osmolality, and low urine volume. Treatment of SIADH includes water restriction to approximately ½ to ⅔ of maintenance fluid requirements and administration of isotonic fluid, such as 0.9% sodium chloride. If the serum sodium concentration is dangerously low or if neurologic signs of water intoxication (e.g., lethargy, irritability) are present, 3% sodium chloride may be administered to reach a target sodium of 125 mEq/L or to elevate the child's serum sodium concentration approximately 0.5 to 1 mEq/L. The remaining sodium deficit is replaced over the next 12 to 24 hours. The following formula can be used to calculate sodium requirements:
(0.6* × Body weight [kg]) × (Desired-Actual [Na+]) = mEq Na+ required to correct
*Use 0.5 in females.
Diabetes insipidus most commonly results from decreased production of ADH, also called central or neurogenic DI. The child with DI has massive free water loss in the urine. If DI progresses unrecognized, fluid depletion can rapidly cause shock, hypernatremia, and neurologic deterioration. Clinical signs of DI include excretion of large quantities of dilute urine often in excess of 10 to 15 mL/kg per hour, hypernatremia, low urine sodium concentration, and low urine osmolality. Treatment of DI includes replacement of urinary fluid and electrolyte losses and administration of IV vasopressin (for further discussion of management of Diabetes Insipidus and SIADH, see Chapter 12).
General Supportive Care
Fluids and Nutrition
Serum electrolytes, serum osmolality, urine output, specific gravity, and urine osmolality must be closely monitored to determine ongoing fluid and electrolyte needs. The child's level of hydration should be monitored closely. Signs of adequate hydration include evidence of good systemic perfusion, moist mucous membranes, adequate urine output (1 mL/kg per hour) and tearing with cry. The infant's fontanelle will not be depressed if hydration is adequate.
The dehydrated infant will demonstrate dry mucous membranes, a sunken fontanelle, decreased urine output with increased specific gravity, poor skin turgor, and an elevation in blood urea nitrogen (BUN). With more severe levels of dehydration, hemoconcentration produces a rise in serum electrolyte concentrations and signs of circulatory compromise (see Chapters 6 and 12).
IV fluids containing 5% to 10% glucose will not provide maintenance caloric requirements. Plans should be made to provide parenteral nutrition if the child does not tolerate enteral feedings within 1 to 2 days after surgery.
Analgesia
Relief of pain during the postoperative period is mandatory for all neurosurgery patients; however, the degree of analgesia and sedation must be balanced with the need to evaluate the patient's neurologic status. If the child is tolerating an enteral feeding, codeine (0.5-1 mg/kg per dose, orally) is often the preferred analgesic because it does not produce the respiratory depression associated with morphine sulfate or fentanyl. If the child is intubated, and receiving mechanical ventilatory support, fentanyl (0.5-2 mcg/kg per dose as an IV bolus or a continuous infusion of 0.5-2 mcg/kg per hour), or morphine sulfate (0.05-0.1 mg/kg per dose IV) may be administered (see Chapter 5). These medications are titrated to clinical effect. If the child becomes agitated, it is important to verify that the child's airway, oxygenation, and ventilation are adequate, and identify and treat any increased ICP before assuming that analgesia or sedation are needed for simple agitation.
Sedation
Agitation during the immediate postoperative period can be treated with midazolam, (0.05-0.1 mg/kg per dose IV bolus [maximum dose, 2-5 mg] or by continuous infusion of 0.05-0.1 mg/kg per hour). Sedatives are especially helpful if it is anticipated that the child will be intubated with mechanical ventilation for any length of time. It is important to determine whether agitation is caused by hypoxia or increased ICP before sedation is administered.
Prevention of Infection
Routine postoperative administration of prophylactic antibiotics is no longer recommended for prevention of infection. However, some patients will receive intraoperative antibiotics.
The single most effective tool for prevention of hospital-acquired infection is good handwashing technique. Stellar wound care and judicious use of central and arterial access also minimizes the risk of postoperative infection (see Chapters 16 and 22).
Infection risk increases if the surgery was performed to repair a cerebral contusion or skull penetration by a foreign object or if the child develops a CSF leak from the nose or ears (see the Head Trauma section of this chapter). The child's temperature and white blood cell count are monitored to detect evidence of infection; appropriate blood, wound, and catheter cultures are taken (per order or protocol) if infection is suspected.
Treatment of Fever
Fever increases metabolic rate, so will increase cerebral oxygen consumption. Such an increase in oxygen consumption can be extremely detrimental in the face of limited oxygen delivery to the brain. As a result, it is especially important to prevent or promptly treat fever in the child with head injury. Fever should be treated with antipyretics such as acetaminophen (10-15 mg/kg per dose orally, nasogastric or rectally with a maximum of 650 mg every 6 hours) or ibuprofen (10 mg/kg per dose orally or via nasogastric tube every 4-6 hours) if bleeding is not a risk.
It is important to rule out sources of infection if high or persistent fever develops. If antipyretics are not effective, a hypothermia blanket may be used to reduce fever if it does not cause shivering. Shivering can increase cerebral metabolism and contribute to increased ICP.83,96
Prevention of the Complications of Immobility
The comatose or paralyzed child can quickly develop atelectasis, pulmonary infiltrates, contractures, deep vein thrombosis, and other complications of immobility unless the nurse provides good pulmonary toilet, passive range-of-motion exercises, and other preventive care (for further information see Management in the Coma section of this chapter).
Psychosocial Support
Neurosurgery is likely to be extremely frightening for the child and parents. In recent years, the general public has become more aware that a patient can survive a neurologic insult in a chronic vegetative state. Many parents have expressed the willingness to cope with their child's physical handicap as long as a chronic vegetative state does not develop. Thus, the prospect of neurosurgery and its possible complications can be extremely threatening. If the child requires surgery after head trauma, the parents may be overwhelmed by the acuity and severity of the injury and by guilt for not having prevented it.
If possible before the surgery, the nurse should determine the parents' and child's understanding of and response to the child's condition. If the parents have major misconceptions, the nurse can attempt to clarify them. If the child or parents have unanswered questions before surgery, the nurse should notify the neurosurgeon or on-call provider to ensure that these questions are answered. Providing informed consent for surgery requires that the parents be aware of the child's condition, potential postoperative complications, and alternatives to surgery.
During the surgery, it is helpful for the nurse to keep the parents informed about the progress of surgery. It is not necessary for the parents to be made aware of each aspect of the surgical technique or specifics of dissection, suturing, or debridement, but it is helpful for the parents to know of the general progress of the surgery. The nurse also can provide the parents with interim reports of the child's condition during surgery; however, these reports must be carefully worded and require the consent and input of the surgeon. Such interim reports can reduce the parents' anxiety during the waiting period and also provide the opportunity to prepare the family for bad news if the child's condition deteriorates. If the family's first hint of trouble comes when the surgeon arrives to inform them that the child suffered a catastrophic cerebral insult, the parents can be overwhelmed by grief or anger and be too shocked to respond or ask questions.
After surgery, the parents will require support when visiting their child for the first time. They should be given consistent reports of the child's condition and prognosis throughout postoperative care. The child will require gentle care and encouragement. When discussing a child's condition and prognosis with the child and the child's siblings, a child life specialist is often very helpful (see Chapters 2 and 24 for further information). Box 11-10 summarizes the important aspects of nursing care of the postoperative pediatric neurosurgery patient.
Box 11-10 Postoperative Care of the Pediatric Neurosurgery Patient and the Patient with Increased Intracranial Pressure
Postoperative care of the neurosurgery patient includes assessment for deterioration (particularly development or worsening of increased ICP) and prevention or detection and treatment of potential surgical complications of the surgical procedure. This box highlights nursing care priorities.
I Potential Respiratory Insufficiency, Related to:
Anesthesia
Airway obstruction
Hypoventilation, atelectasis
Increased intracranial pressure
Neurogenic pulmonary edema
Pneumonia
Expected Patient Outcomes
1. Patient will demonstrate appropriate respiratory rate for age and clinical condition.
2. Patient will demonstrate no evidence of airway obstruction.
3. Patient will demonstrate equal and adequate breath sounds and lung expansion bilaterally, with no evidence of increased respiratory effort or distress.
4. Oxygenation and ventilation as evaluated by arterial blood gases and arterial oxygen saturation (pulse oximetry) and exhaled CO2 will be appropriate for clinical condition. Hypoxemia and hypercarbia will be prevented or promptly treated (both increase cerebral blood flow and can contribute to potential rise in ICP).
5. Systemic oxygen delivery (arterial oxygen content × cardiac output) will be adequate and metabolic acidosis will be absent.
Nursing Interventions
1. Obtain report of patient condition and surgical procedure before the child’s arrival. Prepare all equipment and support needed in the postoperative care unit.
2. If child is intubated and receiving mechanical ventilation:
a.
Assess airway and effectiveness of ventilation. If positive pressure ventilation is adequate, chest expansion will be equal and adequate bilaterally. If the chest does not expand during positive pressure ventilation, ventilation is not adequate.
b.
Evaluate oxygenation through pulse oximetry, arterial blood gases, and ventilation (carbon dioxide elimination) through exhaled carbon dioxide.
c.
Monitor child’s heart rate, spontaneous respiratory effort, color, and general appearance; notify physician/provider of signs of distress.
d.
Adjust ventilation support as indicated and ordered to maintain oxygenation and normocarbia. Monitor for hypoxemia and hypercarbia and treat immediately.
e.
Once child’s condition is stable, wean mechanical ventilation as indicated (and per order or protocol); closely monitor neurologic function.
f.
Keep emergency hand ventilator (bag), oxygen source, mask, and reintubation equipment readily available
3. If child is breathing spontaneously, evaluate and support airway, oxygenation, and ventilation:
a.
Be alert for signs of airway obstruction, hypoventilation, or inadequate gas exchange.
b.
Monitor level of consciousness and responsiveness; if child’s response to painful stimulus is compromised, airway protective mechanisms (cough, gag) may also be depressed; consider elective intubation.
4. Monitor vital signs and report excessive tachycardia, bradycardia, systolic hypertension, or irregular breathing to physician; be prepared to treat increased ICP.
5. Auscultate breath sounds bilaterally, and report decreased intensity or change in pitch of breath sounds (may be associated with unilateral pulmonary pathology) to provider. Monitor for clinical evidence of pulmonary edema (see Chapter 9), pulmonary edema on chest radiograph (see Chapter 10), or hypoxemia (and report findings to on-call provider).
6. Evaluate presence of cough and gag; if these reflexes are depressed, withhold oral fluids and discuss potential intubation with on-call provider.
7. Monitor for signs of increased ICP: deterioration in level of consciousness, decreased responsiveness, pupil dilation with decreased response to light. Be prepared to treat increased ICP as needed and ordered.
8. Administer analgesics and sedatives as needed to maintain patient comfort and ensure effective mechanical ventilation (see Chapter 5). If agitation develops, assess for hypoxia, increased ICP and pain.
II Potential Compromise in Systemic Perfusion Related to:
Hemorrhage
Increased intracranial pressure
Inadequate fluid administration or excessive diuretic therapy
Fluid loss associated with diabetes insipidus
Electrolyte imbalance
Hypoventilation and hypoxemia
Expected Patient Outcomes
1. Patient will demonstrate a heart rate and blood pressure appropriate for age and clinical condition.
2. Systemic perfusion will remain adequate, as demonstrated by urine output approximately 1-2 mL/kg per hour (if fluid intake adequate), warm skin with brisk capillary refill, strong peripheral pulses, normal serum lactate (no lactic acidosis).
Nursing Interventions
1. Monitor systemic perfusion: heart rate, blood pressure, warmth of extremities and trunk, capillary refill, quality of peripheral pulses and urine output. Notify the provider immediately of any deterioration.
a.
Assess intravascular volume status; administer fluid or blood as needed.
b.
Assess neurologic function.
c.
Assess and support fluid and electrolyte balance.
d.
Ensure that oxygenation and ventilation are adequate; be prepared to provide supplementary oxygen and ventilation as needed (and ordered).
2. Administer isotonic (e.g., normal saline) intravenous fluids as ordered:
a.
Avoid hypotonic fluids (they may contribute to a fall in serum sodium with development of cerebral edema).
b.
If intravascular volume and systemic perfusion are adequate, typical fluid administration volume is less than maintenance fluids (see
Table 11-8).
c.
Plan to provide adequate nutrition (see
Chapter 14).
d.
Monitor for evidence of SIADH: fall in serum sodium concentration (with possible associated change in mental status) and serum osmolality, oliguria with high urine sodium concentration. Treatment: restrict water intake to approximately 50%-75% of calculated maintenance requirements (see
Chapter 12).
e.
Monitor for evidence of diabetes insipidus: extremely high urine volume with low urine specific gravity. Treatment: replace urine losses (milliliter for milliliter), monitor for and treat hypovolemia or electrolyte imbalance, administer ADH (see
Chapter 12).
3. Monitor fluid balance closely and be aware of potential sources of increased insensible water loss (including fever); notify provider of positive or negative fluid balance.
4. Monitor electrolyte balance; notify provider of imbalance.
5. Administer hypertonic saline and osmotic agents as ordered; monitor result and notify provider of effect on ICP and neurologic function.
6. Ensure that oxygen delivery (arterial oxygen content × cardiac output) is adequate (inadequate oxygen delivery will result in development of metabolic acidosis), and treat reversible causes of increased oxygen consumption (fever, pain, anxiety).
III Potential Cerebral Ischemia or Dysfunction Related to:
Increased intracranial volume (caused by increased blood volume, brain volume or CSF volume) and pressure and reduction in cerebral perfusion pressure
Status epilepticus
Cerebral oxygen consumption in excess of oxygen and substrate delivery
Expected Patient Outcomes
1. Patient will demonstrate no deterioration in neurologic function.
2. Complications of increased ICP, such as a compromise in cerebral perfusion or brain death, will not develop.
3. Seizure activity will be detected promptly and treated as needed.
Nursing Interventions
1. Closely monitor neurologic function including (see Box 11-2):
a.
Level of consciousness and responsiveness
b.
Pediatric Glasgow Coma Scale score (see
Table 11-6)
Note: Notify the provider immediately of deterioration in child’s responsiveness. Responsiveness should be assessed whenever clinical condition changes.
c.
Pupil size and response to light
d.
Evidence of cranial nerve function during nursing care activities
2. Monitor systemic perfusion; report compromise to provider immediately.
3. Monitor patient for signs of increased intracranial pressure, including: deterioration in level of consciousness, decreased responsiveness and ability to follow commands, pupil dilation with decreased response to light. Late signs of increased intracranial pressure (and impending herniation) include: bradycardia (note that tachycardia may be present early), systolic hypertension with widened pulse pressure, and irregular respirations with possible apnea. Notify on-call provider immediately if these signs are observed, and be prepared to provide mild hyperventilation if signs of impending herniation develop.
a.
Maintain normocarbia (arterial carbon dioxide tension approximately 35-40 mm Hg) with mechanical ventilation to control cerebral blood flow
b.
Prevent development of hypoxemia (maintain PaO
2 ≥100 mm Hg).
c.
Maintain serum sodium concentration and serum osmolality.
d.
Administer analgesics and sedatives as ordered (see
Chapter 5)
e.
If deterioration in clinical condition is observed, quickly assess neurologic function and report any change to provider immediately.
4. Maintain head in midline position (to enhance cerebral venous return). Avoid any condition that will impede cerebral venous return, such as extremely high levels of positive end-expiratory pressure. If systemic arterial pressure is adequate, elevate head of the bed to enhance cerebral venous return (per order or protocol).
5. Administer hypertonic saline or osmotic diuretics as ordered. Monitor serum sodium concentration and evaluate serum osmolality (via laboratory) and estimate according to the following formula:
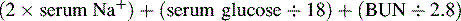
Normal serum osmolality is approximately 275-295 mOsm/L. As a rule, diuresis is performed to maintain serum osmolality at approximately 300-310 mOsm/L, with the serum sodium concentration maintained at approximately 145-150 mEq/L. Prevent an abrupt fall in serum sodium concentration or serum osmolality that can contribute to intracellular fluid shift and cerebral edema.
6. If intracranial pressure monitor is in place, verify calibration and accuracy of system.
7. Chart the ICP to reflect the peak (hourly) intracranial pressure, whether this peak occurred with stimulation, and the average ICP (see Fig. 11-15 and Box 11-5)
8. Calculate CPP; notify provider of rise in ICP (>20 mm Hg) or compromise in CPP. For further information regarding ICP monitoring, see below
9. If intracranial pressure monitoring is performed, discuss plan with the provider for response to rises in ICP (e.g., draining of CSF through EVD, administration of osmotic diuretics, administration of anesthetic agents). Report significant or spontaneous rises in intracranial pressure to on-call provider immediately; be prepared to report any associated changes in neurologic function.
10. Maximize oxygen delivery (arterial oxygen content × cardiac output) and eliminate treatable causes of increased oxygen consumption (e.g., fever, pain, seizures).
11. Monitor for evidence of seizure activity, including lateral eye deviation, a rise in ICP, or a sudden change in vital signs. Posttraumatic seizures are most likely to develop in patients with severe head injury, diffuse cerebral edema, acute subdural hematoma, or an open depressed skull fracture.
12. If ICP continues to rise despite provision of mild hyperventilation, and osmotic therapy, administration of anesthetic agents may be prescribed. This will require transfer to appropriate facility. See Increased Intracranial Pressure, Management)
IV Maintain ICP Monitoring System and Verify Function
Expected Patient Outcomes
1. Intracranial pressure measurements will be accurate and trends recorded on patient flow chart will reflect trends in patient ICP and cerebral perfusion pressure accurately.
2. Longevity of intracranial pressure monitoring device will be maximized.
3. Infection will be prevented.
Nursing Interventions
1. Assemble, zero, and calibrate ICP monitoring system according to manufacturer’s specifications.
a.
Fluid-filled monitoring systems use standard pressure transducers, which must be properly leveled (at the level of the intracranial ventricle), zeroed, and calibrated per the manufacture’s instruction and unit policy.
b.
If fiberoptic catheter is used, zero and calibrate per the manufacturer’s instruction before insertion. Calibrate waveform after insertion. Damage to the transducer or faulty signal will result in dampening of the waveform signal.
c.
Notify provider of any dysfunction.
2. Support insertion of ICP monitoring catheter under sterile conditions, apply dressing over the skin entrance site. Discuss removal of monitoring catheter as soon as possible, to reduce infection.
3. If a ventricular catheter is placed with EVD system, obtain specific information from the provider regarding the technique of CSF drainage, including:
a.
Continuous versus intermittent drainage (and pressure for intermittent drainage)
b.
Position of drainage drip chamber above lateral ventricles
c.
Placement of the head of the bed
Note: If the ventricular catheter is open to drainage at all times, CSF drainage should automatically occur when the ICP exceeds the pressure above the mm Hg (or cm H2O) corresponding to the height at which drip chamber set. For example, if the drainage drip chamber is set at the 27 cm mark above the lateral ventricles, the CSF should automatically drain when the ICP exceeds 27 cm H2O (or 20 mm Hg). If the system is patent, the ICP should never exceed that pressure.
4. Monitor volume of CSF drainage; change the drainage bag (using sterile technique) when it is half full (or per unit practice). If obstruction of CSF drainage is suspected, notify provider.
5. If the nurse is uncertain of the accuracy of ICP monitoring results, or if the ICP rises, the nurse should assess patient’s neurologic function and report changes to provider.
6. For further information, see Increased Intracranial Pressure in this chapter, and Intracranial Pressure Monitoring in Chapter 21.
V Potential Infection, Related to:
Breaks in skin barrier
Invasive monitoring catheters
Multiple trauma
Contamination of surgical wound
Expected Patient Outcomes
1. Patient will demonstrate no signs of infection such as fever, localized inflammation, leukocytosis or leukopenia, or positive wound or blood cultures.
2. Existing infection will resolve promptly.
Nursing Interventions
1. Ensure strict handwashing
2. Ensure that sterile and aseptic procedures are used as needed for insertion and maintenance of monitoring catheters and systems.
3. Change dressings, transducer tubing, and catheters according to unit policy. Maintain occlusive dressings over central venous catheter skin entrance sites.
4. Monitor for signs of infection, including localized signs of inflammation, fever, leukocytosis or leukopenia. Notify physician of cloudy urine, CSF drainage, or drainage from wounds or catheter insertion sites. Obtain cultures as ordered or per unit policy.
5. Administer antibiotics as ordered; monitor peak and trough levels as ordered.
6. Be alert for signs of ventriculitis following intraventricular pressure monitoring. Signs can include deterioration in level of consciousness, sluggish pupil response to light, fever, and headache. Notify a provider immediately if these are observed.
VI Fluid and Electrolyte Imbalance, Related to:
Alterations in ADH secretion, including SIADH
Diabetes insipidus (DI)
Limited fluid intake
Administration of diuretic agents
Expected Patient Outcomes
1. Fluid balance will be appropriate for patient clinical condition.
2. Electrolyte balance will be normal or appropriate for clinical condition (e.g., serum sodium will be maintained at approximately 145-150 mEq/L). Abrupt fall in serum sodium and osmolality will not develop.
Nursing Interventions
1. Monitor and accurately record all sources of fluid intake and output. Monitor effects of diuretic agents on urine volume and neurologic function. Monitor urine specific gravity and serum osmolality (estimate serum osmolality according to formula in III., 5., above). Notify provider of fall in the serum sodium concentration or osmolality.
2. Administer intravenous fluids as ordered (see Table 11-8). Avoid hypotonic fluids or excessive fluid administration.
3. Monitor for signs of SIADH, including a fall in the serum sodium concentration, oliguria, and high urine sodium concentration. Notify a provider if these signs develop. Treatment requires restriction of water intake. Treat symptomatic hyponatremia with 2-4 mL of 3% saline as ordered.
4. Monitor for signs of diabetes insipidus, including increased urine volume with a low urine specific gravity (<1.005). If urine volume increases and diabetes insipidus is suspected, replace fluid lost in the urine and administer ADH as ordered.
5. If ADH administered, monitor urine output before and after administration.
6. Monitor serum electrolyte concentration and notify provider of imbalance.
7. Assess systemic perfusion and notify physician of signs of dehydration, hypovolemia or hypervolemia.
8. When the patient begins oral fluid intake, restrict fluid intake as ordered and needed.
9. Monitor neurologic function closely and notify the provider of any deterioration.
Additional Nursing Diagnoses or Patient Problems
Patient or family anxiety or anger related to: severity of patient condition, potential sequelae of neurosurgery or underlying disease or injury, unknown aspects of treatment or prognosis
Knowledge deficit regarding: patient’s condition, long-term treatment and home care required
Impaired patient mobility related to: neurologic deficit, associated injuries, complications of the underlying disease or initial trauma
Potential pain related to: underlying disease or initial trauma, multiple invasive catheters, surgical incision
ADH, Antidiuretic hormone; BUN, blood urea nitrogen; CPP, cerebral perfusion pressure; CSF, cerebrospinal fluid; EVD, extraventricular drainage; ICP, intracranial pressure; SIADH, syndrome of inappropriate antidiuretic hormone.