17 Overview of Solid Organ Transplantation
 Be sure to check out the supplementary content available at http://evolve.elsevier.com/Hazinski.
Be sure to check out the supplementary content available at http://evolve.elsevier.com/Hazinski.
History
History of Solid Organ Transplantation
Early attempts at solid organ transplantation were unsuccessful for many reasons. There was insufficient development of basic surgical techniques and methods for organ preservation, and also inadequate knowledge of the immune system and tissue rejection.
In the early 1950s steroids were used to suppress rejection, and this resulted in some successful kidney transplants. In 1954, Dr Joseph Murray21 performed the first truly successful kidney transplant between identical twins. Although human liver transplantation was attempted by Starzl in 1963,32 it was not successful until 1967. Also in 1967, the first successful human heart transplant was performed by Christiaan Barnard on a 55-year-old man who survived 18 days.
It wasn't until the mid-1970s that the development of more effective immunosuppression resulted in substantial improvement in transplantation survival. The first successful pediatric heart transplant was performed in 1984. The 4-year-old recipient received a second heart transplant in 1989 and is currently alive and well.
Evolution of Immunosuppression
In the late 1950s, kidney transplantation with nonidentical twins was attempted, with pretreatment of 10 recipients using sublethal total body irradiation. Nine of the ten recipients died within a month. Death was caused by the effects of radiation and not allograft failure.
In the 1960s, the development of tissue typing and drugs such as 6-mercaptopurine and azathioprine resulted in improved transplantation outcomes. Combinations of irradiation, splenectomy, thymectomy, high-dose corticosteroids, and azathioprine were used in attempts to overcome rejection. However, this combination therapy was associated with a remarkably high rate of opportunistic and often multiple bacterial, fungal, viral, and protozoal infections. Many of the infections were undetected and untreated before death.
In the late 1960s through the 1990s, as knowledge of the immune system evolved and more effective antibacterial and antiviral drugs were developed, results of transplantation again improved. Therapy targeting specific immunoregulatory sites became possible. The first polyclonal antilymphocyte globulin was used in 1967. Cyclosporin, a calcineurin inhibitor, became available in the 1980s. This drug, in combination with azathioprine and steroids, has been credited with a dramatic improvement in transplant graft survival.
In 1994, the next advance in transplantation came with the introduction of mycophenolate mofetil (MMF) and tacrolimus (another calcineurin inhibitor). Tacrolimus has gradually supplanted cyclosporine in many posttransplant protocols, because tacrolimus has been associated with lower rates of steroid-resistant acute rejection than cyclosporin.20 In contrast, MMF quickly and almost universally replaced azathioprine in posttransplant protocols.20,22
There are three stages of immunosuppressive therapies: induction, maintenance, and antirejection therapy. Induction immunosuppression therapy refers to all medications given in intensified doses immediately after transplantation for the purpose of preventing acute rejection. Although the drugs may be continued after discharge for the first 30 days after transplant, they are usually not used long term for immunosuppression maintenance.
Maintenance immunosuppression therapy includes all immunomodulation medications given before, during, or after the transplant with the intention of maintaining them long term. Antirejection immunosuppression therapy includes all immunosuppressive medication administered for the purpose of treating an acute rejection episode during the initial posttransplant period or during a specific follow-up period, usually as long as 30 days after the diagnosis of acute rejection.
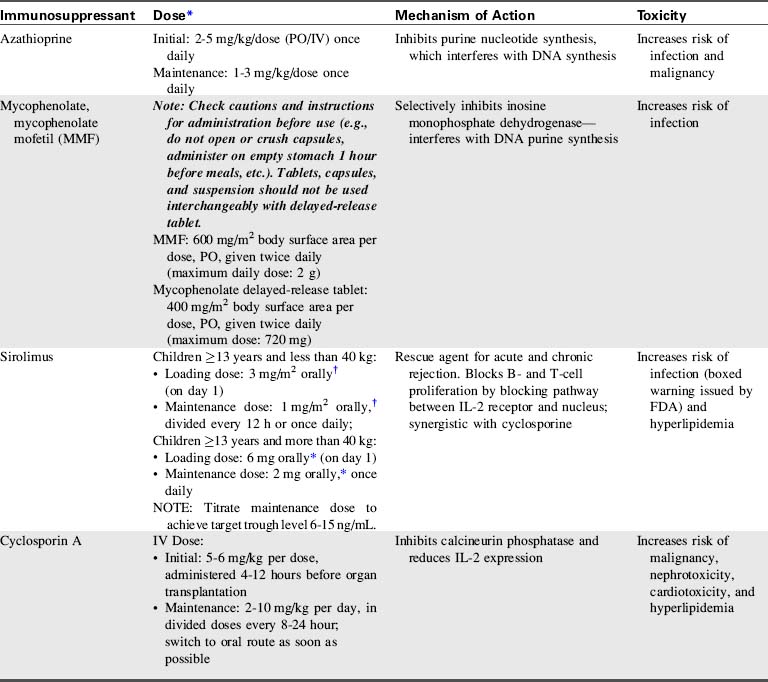
A combination of biologic agents (Table 17-1) and immunosuppressants (Table 17-2) are used to prevent and treat rejection. Because immunosuppressive regiments vary from institution to institution, providers should consult their institutional protocols and the institutional pharmacy to verify drugs and dosing regimens used.
| Biological Agent | Mechanism of Action | Toxicity |
| Antithymocyte globulin | Depletes lymphocytes | Increases risk of infection |
| Corticosteroid | Suppresses eicosanoid production: Increases TGF expression | Increases risk of infection and malignancy |
| IL-2 antibody | Selectively blocks IL-2 receptors on T helper cells | Increases risk of malignancy |
| Muromonab CD3 (OKT3) | Targets CD3 receptor complex on T cells Depletes T lymphocytes |
Increases risk of infection |
| Alemtuzumab | Targets CD52 on T cells, B cells, and NK cells causing depletion | Increases risk of infection |
| Rituximab | Binds CD20 and B cells | Increases risk of infection |
| Belatacept | Binds to T cells and prevents their activation, preventing CD28 signaling |
CD: Cluster of differentiation or cluster of designation (numerical nomenclature that identifies white blood cell surface molecules that affect cell signaling and other functions).
Pediatric heart transplantation
Overview
As of 2011, approximately 5437 pediatric heart transplants have been performed in the United States for recipients to the age of 17,24 and many more have been performed worldwide.7 Pediatric patients less than 17 years of age comprise approximately 10% of all heart transplant recipients.24
Heart transplantation is now an acceptable therapy for end-stage heart failure in infants, children, and adolescents (see, also, Chapter 8). One-year patient survival is 83% to 90%, with 5-year survival of 71% to 77%.24 The graft survival is somewhat lower than patient survival (82%-89% 1-year and 68%-75% 5-year graft survival), with some children requiring retransplantation.24 Unfortunately, about 20% of children and about 30% of infants less than 6 months of age who are waiting for a heart transplant die before receiving a heart.
Timing is crucial when listing a patient for a heart transplant, and the risks and benefits of transplantation must be evaluated in light of the limited donor pool. Bridge to transplant therapies such as extracorporeal membrane oxygenation (ECMO) and ventricular assist devices (VADs) are commonly needed for short- or long-term support (see Chapter 7). A recent review has demonstrated improved survival using the Berlin heart.16
Immediate Postoperative Care
After cardiac transplants, all patients are monitored in the critical care unit. Patients are usually intubated and receive mechanical ventilation for at least 24 hours. Antibiotics are usually continued until chest tubes and monitoring lines are removed. Daily chest radiographs are performed to verify endotracheal tube depth of insertion and monitor for evidence of pneumothorax, atelectasis, or pleural effusions, and to evaluate heart size and chest tube placement.
Close postoperative observation and cardiopulmonary support are needed to ensure adequate graft function and minimize risk of graft failure. Because hypoxia and metabolic acidosis can cause pulmonary vasoconstriction and worsen right heart failure, monitoring and therapy are planned to avoid both.
Pulse oximetry is monitored continuously to detect changes in arterial oxygenation and central venous oxygen saturation (SCVO2) is monitored frequently to evaluate the balance between oxygen delivery and utilization. Arterial blood gas analysis is performed on a scheduled basis and as needed to ensure that systemic arterial oxygenation, carbon dioxide tension, pH, and serum lactate are appropriate.
Hemodynamic status is also closely monitored, including continuous display and recording of heart rate and rhythm and intraarterial pressure, and at least hourly recording of central venous pressure. If a pulmonary artery catheter is in place, then pulmonary artery (PA) pressure, PA wedge pressure, and mixed venous oxygen saturation are monitored. Pulmonary artery pressure monitoring is helpful in determining the severity of pulmonary hypertension and response to therapy, particularly during administration and weaning of inotropes and vasodilators (including inhaled nitric oxide). The pulmonary artery wedge pressure will rise in the presence of left ventricular dysfunction, and the mixed venous oxygen saturation reflects the balance of systemic oxygen delivery with tissue oxygen consumption.
If a PA catheter is not in place, evaluation of the balance between oxygen delivery and consumption is accomplished through frequent evaluation of central venous oxygen saturation (SCVO2), typically drawn from a central venous catheter placed in the superior vena cava. The difference between arterial and central venous oxygen saturation is typically 30% if cardiac output is adequate (for further information, see Chapter 6, Clinical Recognition and Management of Shock, Use of Central Venous Oxygen Saturation). If arterial oxygen content is stable, a fall in SCVO2 indicates either a fall in cardiac output or an increase in tissue oxygen consumption. If a central venous catheter is not in place, a venous sample can be drawn simultaneous with an arterial blood gas sample to assess arterial and venous oxygen saturations and evaluate the balance of oxygen delivery and consumption.
Laboratory tests evaluated on a frequent basis include: complete blood count with differential, basic metabolic panel, ionized calcium, magnesium, coagulation panel, and liver function tests. Serum electrolytes are monitored frequently because electrolyte imbalances can cause cardiac arrhythmias in the postoperative period.
Viral surveillance is common before the transplant and weekly during the acute postoperative period. Bacterial cultures are also performed as needed when fever occurs beyond 48 hours postoperatively.
Both perioperative and immediate postoperative management focus on optimizing allograft function. Challenges to the function of a newly transplanted heart include: denervation, graft ischemia, and acclimation to the recipient's hemodynamics.7 The newly transplanted heart has a relatively fixed stroke volume; therefore, an increased heart rate is necessary to augment cardiac output. The heart rate can be increased through administration of inotropes or with atrial pacing.
Systolic function of the transplanted heart typically recovers rapidly, despite the ischemic insult that occurs between harvest and transplant. By comparison, the diastolic function may require weeks to recover and may respond to administration of low-dose inotropes in the early postoperative period. If the ischemic time was short and the recipient's pulmonary vascular resistance (PVR) is low, only short-term inotropic therapy is likely to be needed after the transplant.
The transplanted heart is no longer innervated by sympathetic nervous system fibers, and the vagal effects on the heart are blunted. Therefore, if sympathetic nervous system stimulation is needed, the child should receive exogenous catecholamines. Sympathomimetics that stimulate alpha-2 and beta-receptors are likely to be more effective than those that stimulate alpha-1 (innervated) receptors. For this reason, dobutamine is likely to produce more significant effects on heart rate and contractility than dopamine.
The most common postoperative complications include: pulmonary hypertension, acute allograft dysfunction, arrhythmias, vasodilatory heart failure, renal dysfunction, and hypertension. These complications are presented in more detail in the text that immediately follows.
Pulmonary Hypertension and Right Ventricular Failure
Most patients experience some degree of elevation in PVR before transplantation, and postoperative elevation in PVR can affect the transplanted heart function. Right heart failure often develops after 24 hours and typically lasts 3 to 5 days. Signs of right heart failure include a high right atrial or central venous pressure and hepatomegaly. Severe right heart failure may compromise cardiac output and systemic perfusion.
Several measures can be taken to reduce pulmonary vascular resistance and decrease right ventricular afterload, including the administration of milrinone, prostaglandin, prostacyclin, nitroprusside, and inhaled nitric oxide. Milrinone is often used because it has both vasodilatory properties (that can reduce pulmonary and systemic vascular resistance) and inotropic effects.1 Alveolar hypoxia and acidosis should be avoided, because these conditions can exacerbate pulmonary artery constriction, worsening right ventricular failure (for further information, see section, Pulmonary Hypertension in Chapter 8). Some patients with significant elevation in PVR benefit from administration of neuromuscular blockers with sedation (see Chapter 5) and controlled mechanical ventilation for 24 to 48 hours posttransplant.
Treatment of right ventricular failure is largely supportive and includes judicious fluid administration to maximize right ventricular preload, and inotropic support to improve contractility. In rare instances, right heart failure may be so severe that extracorporeal membrane oxygenation (ECMO) is required to support the circulation (see Chapter 7).1 Hemodynamic monitoring and serial echocardiograms will be used to monitor right ventricular function, particularly in response to therapy and during weaning of therapy.
Acute Allograft Dysfunction
Acute allograft dysfunction poses a significant clinical problem that accounts for approximately 30% of early posttransplant deaths. The causes include ischemia-reperfusion injury, problems with preservation, prolonged ischemic time, and unrecognized or underappreciated donor heart dysfunction.7 Allograft dysfunction may be detected even during the operative procedure with intraoperative transesophageal echocardiography (TEE).
A 12-lead electrocardiogram (ECG) is performed shortly after arrival from the operating room and daily after transplantation, because ECG changes may signify early rejection or graft dysfunction. Dobutamine and milrinone infusions are usually adequate to support lesser degrees of allograft dysfunction. On rare occasions, the recipient requires use of a ventricular assist device or ECMO until heart function improves (see Chapter 7).
Arrhythmias
The incidence of tachyarrhythmias after pediatric heart transplantation is about 15%, similar to that reported after adult cardiac transplantation.5 Most tachyarrhythmias resolve after a relatively brief period of medical treatment (see Arrhythmias in Chapter 8), and recurrence is uncommon.19 Although ectopic atrial tachycardia is more common in children than adults, it appears to be well tolerated in the younger age group.5 Atrial flutter tends to be associated with cardiac rejection, and atrial fibrillation is associated with a poor clinical outcome.5
Bradycardia, although not common, can result from sinus node dysfunction and typically requires pacemaker therapy. Because vagal (cholinergic) effects on the transplanted heart are blunted, administration of atropine (an anticholinergic) may not produce a significant rise in heart rate. Adrenergic drugs are likely to be more effective than an anticholinergic in increasing heart rate.
Vasodilatory Hypotension
Vasodilatory hypotension can result from several factors after cardiac transplantation. Loss of vascular tone after heart transplant can be caused by the use of drugs, including angiotensin-converting-enzyme (ACE) inhibitors, amiodarone, and inodilators (e.g., milrinone). Vasodilation can also be caused by a systemic inflammatory response from inflammatory cytokine activation following cardiopulmonary bypass.7 In addition, vasodilation can result from a baroreflex-mediated depletion of arginine vasopressin (AVP). Treatment of vasodilatory hypotension requires the use of vasopressors (catecholamines).
Renal Dysfunction and Hypertension
Urine output, central venous pressure, and serum blood urea nitrogen (BUN) and creatinine are monitored closely after transplantation. Renal dysfunction can result from decreased renal perfusion associated with prolonged aortic cross-clamp time, as well as from thromboemboli or perioperative hypotension. The introduction of calcineurin inhibitors for immunosuppression (see Table 17-2) can suppress renal function further.
The corticosteroids given for posttransplant immunosuppression can cause fluid retention. Administration of diuretics is often useful to eliminate excess intravascular fluid that can be overloading the transplanted heart. In rare cases, temporary dialysis may be initiated until the return of renal function.
Hypertension is common during the early and late postoperative periods, and is often a side effect of medications. Pediatric patients typically require antihypertensive therapy after heart transplantation. Calcium channel antagonists, angiotensin-converting enzyme inhibitors, and diuretics are all acceptable first-line treatments in children with hypertension after cardiac transplantation.27 Individual patient blood pressure profiles and evaluation of drug tolerance and responsiveness are needed to determine optimal drug choice and dosing.
Posttransplant Immunosuppression
The survival of the transplanted heart relies heavily on immunosuppressive therapy. Ideally the goal of immunosuppression is to manipulate the immune response to promote tolerance to the foreign antigens, without rendering the recipient vulnerable to infection.1
Induction immunosuppression is started in the perioperative period. Many institutions begin administration of calcineurin inhibitors, such as cyclosporine or tacrolimus, before the start of the transplant surgery. High-dose corticosteroids are given intraoperatively and continued for 48 hours; they are then either discontinued or tapered to a low-dose maintenance regimen.1
Additional immunosuppressive medications may be given postoperatively in the form of polyclonal antibodies such as antithymocyte globulin (ATG). The maintenance immunosuppressive regimen uses the “triple therapy” approach: a calcineurin inhibitor (cyclosporine or tacrolimus), mycophenolate mofetil (MMF), and corticosteroids (see Tables 17-1 and 17-2).
Rejection
Most pediatric heart transplant recipients experience at least one rejection episode during their lives, with a majority of these occurring during the first 3 months after transplantation.7 Clinical evaluation of rejection is important but can be misleading, especially in the pediatric population, because the inflammatory response associated with an infection can mimic the presentation of rejection.1 Clinical signs of rejection can include vague signs such as fatigue, irritability, gastrointestinal problems (e.g., vomiting, diarrhea, or poor feeding), or the development of specific cardiovascular signs such as heart failure, low cardiac output, or arrhythmias.
Cellular rejection, often referred to as T-cell-mediated rejection, is most common and is characterized by an infiltration of the heart by lymphocytes. The treatment options include optimizing current immunosuppressive therapy plus the administration of high-dose corticosteroids for mild cases of rejection and antithymocyte globulin (ATG) or muromonab CD-3 (OKT3) for severe cases of rejection (see Table 17-1).
Humoral- or antibody-mediated rejection occurs when donor-specific antibodies attack the transplanted allograft. This type of rejection is B-cell mediated and is associated with a higher rate of graft loss, development of transplant vasculopathy, and decreased long-term survival.7 Treatment for humoral rejection includes the use of high-dose corticosteroids, ATG, cyclophosphamide, and plasmapheresis.
Although the most effective way to detect rejection is through an endomyocardial biopsy (see Fig. 8-72), studies are underway to develop less invasive methods for rejection surveillance. Biohumoral markers such as B-type natriuretic peptide (BNP) and vascular endothelial growth factors (VEGF) are currently being studied as markers for rejection. Elevated BNP levels at 1 year or more posttransplant are associated with worse graft survival in pediatric heart transplant patients.28
Summary
Pediatric critical care nurses play an important role in the assessment and management of infants and children after cardiac transplantation. As noted, very subtle changes in the patient clinical presentation can indicate significant problems, such as acute rejection. Nurses are able to detect subtle and early changes in patient status, and provide both physical care and the psychosocial support that are so crucial during the early posttransplantation phases. Discharge planning and patient and family education can have a substantial impact on patient and family compliance and long-term success of transplantation.
Although late complications and long-term effects of immunosuppressive therapy are ongoing challenges for clinicians caring for pediatric patients after heart transplants, enormous progress has been made during the past 20 years to improve survival and functional outcome of these patients. Pediatric heart transplant recipients are often able to lead relatively normal lifestyles, although they do require daily medications and chronic medical surveillance. Incremental changes and refinements in therapy continue as researchers and clinicians strive to improve longevity and quality of life in the pediatric heart recipient.
Pediatric liver transplantation
The first human liver transplant was performed by Starzl in 1963.32 Since that time, graft and recipient survival and functional outcomes have steadily improved as the result of advances in immunosuppression and improved surgical techniques. There are now approximately 100 liver transplant centers in the United States, and about 250 pediatric liver transplantations are performed each year.24 Pediatric patients comprise approximately 9% of all liver transplant recipients.24
For the interested clinician, additional information about pediatric liver transplantation is contained in the Chapter 17 Supplement on the Evolve Website. The US Department of Health and Human Services maintains an Organ Procurement website with valuable information, including a numerical scale used for allocation of livers for transplantation (for a link to this website, see the Chapter 17 Supplement on the Evolve Website). Additional recommended readings are also provided (see Recommended Reading: Liver Transplantation in the Chapter 17 Supplement on the Evolve Website).
Immediate Postoperative Care
After liver transplant surgery, patients are cared for in a pediatric critical care unit. Because the transplant procedure and associated anesthesia are long, support with mechanical ventilation is typically planned for 24 to 48 hours. The length of stay in the pediatric critical care unit varies from several days to many weeks.
Immediately following liver transplantation, patients typically return to the critical care unit with multiple catheters and tubes in place (central venous and arterial catheters, urinary catheters, and nasogastric tubes). Careful monitoring of vital signs and urine output are always required, and administration of blood products may be needed. Laboratory studies, including sampling for arterial blood gas analysis, hematocrit, coagulation studies, liver function tests, and analysis of serum electrolytes, are performed frequently.
Graft function is confirmed biochemically if there is evidence of hepatic synthetic and metabolic function (e.g., correcting prothrombin time, reversal of acidosis). Alternatively, the lack of graft functional recovery can be evident in the first hours after transplantation, with findings such as a high serum lactate or prolonged prothrombin or activated partial thromboplastin time (aPTT), or if the patient fails to awaken despite suspension of sedation.
Failure of graft functional recovery is an extremely serious complication that must be treated aggressively and immediately by infusing prostaglandin E1, adopting the necessary measures to prevent brain edema (e.g., mannitol, oxygenation and ventilation), and addressing the effects of the liver failure by infusing plasma and glucose.31 Failure of a graft in the absence of vascular compromise (primary nonfunction) requires retransplantation in almost all cases, with the outcome best in patients with the shortest duration of liver failure before retransplantation. The incidence of primary nonfunction in pediatric patients is 5% to 10%.8,31
Posttransplant Immunosuppression
Currently, most liver transplantation centers use a tacrolimus-based regimen, combined with corticosteroid therapy with or without adjunctive agents (see Tables 17-1 and 17-2).26 Cyclosporine and tacrolimus share certain acute and long-term side effects while having some that are unique to each agent. The most important side effect of these immunosuppressives is nephrotoxicity, which acutely results from vasoconstriction of the afferent renal arterioles; this nephrotoxicity is usually reversible. These drugs can also produce a more chronic nephrotoxicity marked by tubular atrophy, interstitial fibrosis, and glomerulosclerosis. The chronic nephrotoxicity is variably reversible, depending on the degree of disease. To minimize acute toxicity and to allow lower tacrolimus levels, especially in patients with pretransplant renal insufficiency, a purine antimetabolite mycophenolate mofetil (MMF) can be used as an adjunctive agent.20,22
Treatment of acute rejection uses a high-dose methylprednisolone bolus, but unresponsive cases may require use of antibody therapy (e.g., OKT-3).8 Acute rejection accounts for less than 3% of overall patient and graft loss.20 However, treatment of acute rejection is an important risk factor for the development of cytomegalovirus and Epstein-Barr virus (EBV) infections in children. The EBV, in turn, is a risk factor for the development of posttransplant lymphoproliferative disorder.13Therefore, it is necessary to achieve a balance between adequate immunosuppression to prevent acute rejection and avoiding excessive immunosuppression to reduce the risks of toxicity and other complications.
Chronic rejection is a common cause of late graft loss in children, whereas disease recurrence is uncommon. Chronic rejection may result from a number of factors that share a final common pathway of graft injury. The hallmark of chronic rejection is the intrahepatic loss of bile ducts, which has been termed vanishing bile duct syndrome, based on the histologic appearance noted on biopsy. Rejection is suspected by the presence of progressive jaundice and a rising serum alkaline phosphatase level. Currently, there is no prophylactic or therapeutic agent available to treat chronic rejection. The only accepted treatment when decompensated graft failure occurs is retransplantation.
Long-term immunosuppression protocols focus on sustaining graft acceptance while limiting morbidity. Transplant centers have reported successful withdrawal of immunosuppression in patients who still achieved prolonged graft survival. Long-term clinical experience with steroid use has documented a host of adverse effects including hypertension, malignancy, and infections.31 In addition to age and growth deficit at the time of transplantation, two other major factors affecting growth after transplantation include allograft function and steroid therapy.20
Complications of Liver Transplantation
Complications of liver transplantation include the following:
These complications are addressed separately in the following.
Hepatic Artery and Portal Vein Thrombosis
Major vascular complications include hepatic artery thrombosis, portal vein thrombosis, and vena caval thrombosis or stenosis. Intravenous low-dose unfractionated heparin with or without low-molecular-weight dextran is routinely used for prophylaxis to prevent vascular thromboses. Early vascular complications are usually technical in nature; by comparison, immunologic and infectious (e.g., cytomegalovirus) causes have been ascribed to those occurring months after transplantation.
Hepatic artery thrombosis is the most common vascular complication, with an incidence that varies from 5% to 18% depending on patient age and type of graft.8,23,31 Hepatic artery thrombosis occurring in the first week after liver transplantation is commonly associated with graft nonfunction and biliary necrosis or leak. Graft nonfunction produces acute liver failure, rising bilirubin, and mental status deterioration. Biliary leaks can develop when ischemia from the arterial thrombotic event produces disruption of the anastomosis. Duplex ultrasonography and computed tomography or conventional angiography are accepted means of diagnosis.
Hepatic artery thromboses occurring later after the transplant may not affect graft function immediately but can produce biliary complications. These include intrahepatic biliary abscesses, biliary anastomotic stricture, and sclerosing cholangitis with sepsis, all of which lead to significant morbidity. If diagnosed sufficiently soon after the development of the thrombosis, some patients can be managed by thrombectomy and surgical revision. Others require urgent retransplantation. Late hepatic artery thrombosis with preserved graft function can be managed by radiologic interventional techniques, with later retransplantation (i.e., remote from the time of the initial transplant procedure).
Thrombosis of the portal vein occurs in 2% to 4% of pediatric liver transplant procedures and is often associated with loss of the graft.23 If interventional radiology techniques are unsuccessful, prompt retransplantation is required for patient salvage.
Patients with late portal vein thrombosis usually present with recurrent variceal bleeding or ascites and can be managed medically, endoscopically, or surgically with either shunting or retransplantation. Vena caval or hepatic vein thrombosis or stenosis occurs in 3% to 6% of pediatric liver transplant patients and is usually best managed with balloon dilation in an interventional radiology unit.23
Biliary Complications
Biliary complications are the most frequent technical complication after liver transplantation. Bile leaks and anastomotic strictures account for most notable biliary problems encountered in the postoperative period. Biliary complications may result from hepatic artery thrombosis; in some series, 70% of patients with hepatic artery thrombosis have concurrent biliary complications.18 Biliary complications may also be associated with prolonged preservation time, ischemia, or surgical technique.
Early biliary leaks are usually best handled by reoperation. Strictures (either anastomotic or intrahepatic) can frequently be managed nonoperatively with tube drainage, stent placement, or both.
Infection
Infections after liver transplantation follow a rather consistent time course. In the early postoperative period, bacterial infections are most common. After approximately 2 weeks, fungal infections are the increasing concern. Beginning about 6 weeks after the transplant and for the remainder of the patient's life, viral infections dominate the infectious disease concerns.4,25
Given the known time course of causes of likely infection, most patients are given prophylactic antibiotics, antifungals, and antiviral medications from the time of the transplant. The antibiotics and antifungals are usually stopped within the first days to weeks after transplant, if no signs of bacterial or fungal infections are present. Antiviral medications are usually given for the first several months after transplantation.
The most frequent viral infections in liver transplant recipients are cytomegalovirus (CMV), Epstein-Barr virus (EBV), adenovirus, and respiratory syncytial virus (RSV). Epstein-Barr virus is an especially important virus to identify in this population, because increased levels of EBV in the transplant recipient can lead to posttransplant lymphoproliferative disease (PTLD).4,13,25 Lymphoproliferative disorders are summarized in a section that follows (see section, Lymphoproliferative Disorders).
A major challenge in posttransplant management is preventing viral illness secondary to immunosuppression. Antiviral prophylaxis with ganciclovir has been proved to be valuable against CMV infections. Cytomegalovirus hyperimmune globulin may also be somewhat protective against EBV. Detection is paramount in ameliorating the negative effects of EBV- and CMV-associated illness. The use of polymerase chain reaction (PCR) for this purpose has been particularly useful.25,34
Nephrotoxicity
Although calcineurin inhibitors (e.g., tacrolimus and cyclosporine) are integral to posttransplant care, these drugs have a negative effect on kidney function. This phenomenon is dose related.
Histologic examination from kidney specimens of patients receiving calcineurin inhibitors shows chronic progressive interstitial fibrosis. Importantly, creatinine levels have not been shown to be a good indicator of renal function. A rise in serum creatinine concentration occurs only after a 50% reduction of glomerular filtration rate (GFR). Thus, ongoing monitoring of urine output, fluid balance, and renal function is essential.
Central Nervous System Toxicity
The use of tacrolimus as an immunosuppressant has been shown to induce seizures. This CNS toxicity has been attributed directly to elevated serum concentration of tacrolimus.2 As a result, when tacrolimus is initially administered, serum concentration is closely monitored.
Lymphoproliferative Disorders
Immunosuppression has led to the development of cancers in transplant recipients. For some tumor types, the incidence is 100 times more frequent among transplant recipients than in the general population. Skin cancers account for most of the newly diagnosed cases.
An important tumor recognized to result from immunosuppression is posttransplant lymphoproliferative disease (PTLD). Posttransplant lymphoproliferative disease is most common during the first 2 years following transplantation. This disease process has been attributed to EBV secondary to drug regimens that inhibit immune surveillance.13 Epstein-Barr virus-related PTLD plays a major role in morbidity and mortality among pediatric transplant recipients.
Posttransplant lymphoproliferative disease affects 6% to 20% of posttransplant patients.8 Treatments for PTLD are largely centered on reducing the level of immunosuppression. Novel therapies involving the use of rituximab, a monoclonal antibody against CD20, may also prove beneficial.2,8,13
Psychosocial Stress
A unique set of psychosocial risk factors may be present among pediatric liver transplant recipients, leading to poor adherence to medication regimens and subsequent graft dysfunction.20 Risk factors for poor compliance include inflicted trauma (intentional injury), single-parent households, substance abuse, and the patient dropping out of school. A formal assessment of these risk factors must be performed to identify them and assess and modify their impact on patient compliance with postsurgical treatment.10,17
Pediatric kidney transplantation
History
In 1954, Dr. Joseph Murray performed a successful kidney transplant between identical twins, thus skirting the problems of immune compatibility.9 Several transplants between twins followed. However, the possibility of kidney transplantation for patients with renal failure who did not have a twin donor remained unrealized.15,21 In the early 1960s, Calne demonstrated that a derivative of 6-mercaptopurine (azathioprine) increased the success of experimental kidney transplantation in dogs.3 Human use of azathioprine followed, and long-term graft survival from nonidentical donor kidneys became a possibility.
The success of kidney transplantation increased significantly when Goodwin and Starzl added prednisolone to azathioprine immunosuppression.33 Encouraged by this success, transplant centers began performing nonidentical living donor kidney transplantation.
Terasaki reported a marked decrease in early allograft failure from hyperacute rejection when a crossmatch between donor lymphocytes and recipient serum was performed.35 A negative crossmatch (no reaction against donor lymphocytes when incubated with recipient serum) indicated that no antibody directed against the donor's organ was present in the recipient.
Kidney transplantation has now become the primary method of treating end-stage renal disease (ESRD) in the pediatric population (see Chapter 13). During the past decade, survival following kidney transplantation has improved in all pediatric age groups. This improvement is particularly noteworthy in children younger than 2 years of age who previously had the worst outcomes; these children now have outcomes that equal the outcomes of any age group.9,29 Recent reports demonstrate that the youngest recipients now have the longest transplant half-lives of all recipients; this is especially true if the pediatric recipient receives an adult kidney that functions immediately.12 These improvements likely reflect better donor selection, improvement in surgical techniques, better immunosuppression agents, and a better understanding of immunosuppression management in children.
Although the current short-term success of kidney transplantation in the pediatric population is encouraging, it is also important to realize that long-term graft survival in the adolescent group (ages 11-17 years) is relatively poor.12 The reasons for this significant rate of graft loss are speculative, but noncompliance likely plays a significant role.20,29 Regardless of the cause, improving the long-term outcomes in this patient population represents one of the most significant challenges in pediatric transplantation.
Immediate Postoperative Care
To monitor and replace urine output on an hourly basis, patients are often admitted to the critical care unit for 1 to 2 days after renal transplantation. For some smaller children, mechanical ventilation is planned for 24 hours postoperatively; these patients will all be admitted to the critical care unit. If careful monitoring of fluid and electrolyte balance can be accomplished on a surgical acute care unit, larger children can be admitted to an area specializing in the care of renal transplant patients.
Careful attention to detail in the postoperative period is essential to the care of the child after renal transplant. Special care must be directed to monitoring and maintaining the child's fluid and electrolyte balance.
Many children are polyuric before transplant, and this obligate urine loss will continue in the immediate postoperative period. Intravenous fluids are administered, with administration rate adjusted in light of urine output as well as insensible losses. The composition of intravenous solutions is adjusted as needed, based on measurement of serum electrolytes at regular intervals. Serum sodium, potassium, and calcium levels are monitored closely and replaced as necessary.
Heart rate, blood pressure, and central venous pressure are carefully monitored. No single parameter alone is entirely reliable in assessing intravascular volume, and some variables may not reflect the true status of the patient.
Urine output is a critical indicator of graft function. However, urine volume must be evaluated in light of the patient's pretransplant renal function in combination with evaluation of the posttransplant graft function and the patient's volume status. For patients who were oliguric or anephric (i.e., underwent nephrectomy) before transplantation, if the graft functions immediately, improved urine output posttransplant will be an excellent indicator of graft function. If the patient had high urine output renal failure before transplant, a high posttransplant urine volume may mask delayed graft function.
Oliguria should be carefully evaluated; the urinary catheter may require flushing with small amounts of sterile saline. The volume status of the patient should also be carefully assessed. A fluid bolus followed by evaluation of urine output is usually warranted, both as a diagnostic test and as therapeutic intervention. An ultrasound with Doppler study may be helpful to confirm adequate arterial flow to the graft and adequate venous outflow. In addition, the ultrasound will reveal any evidence of fluid or blood around the kidney, and will allow assessment for possible ureteral obstruction.
In patients with oliguria who appear to have adequate intravascular volume and hemodynamic stability, a dose of diuretic may be given to monitor response. It is important to administer diuretics carefully, because sudden massive urine output can cause significant intravascular volume depletion, with resulting decrease in renal perfusion. If the patient is massively volume overloaded or has significant electrolyte abnormalities, dialysis or continuous venovenous hemofiltration (CVVH) may be indicated (see Chapter 13).
Hypertension can be problematic after renal transplantation. The volume loading associated with the renal transplant procedure, as well as the use of calcineurin inhibitors for immunosuppression, can result in significant hypertension. The hypertension can be severe and require aggressive therapy to prevent seizures and other sequelae.20
Enteral feedings are usually begun at a slow rate shortly after surgery if an extraperitoneal approach is used for the transplantation. Most children can leave the hospital within 5 to 7 days after transplantation, assuming graft function is adequate, the child is able to eat, and the family is familiar with the immunosuppression regimen.
Posttransplant Immunosuppression
Over the past several decades, significant advances have been made in our understanding of the immune response, and several new immunosuppressive agents have been introduced into clinical care. There are now several posttransplant immunosuppressive regimens available, but all require a balance between prevention of rejection and prevention of unwanted side effects associated with immunosuppression.12,29
Some transplant centers use a standard protocol for all renal transplant recipients, whereas other centers individualize the regimen for each patient. Immunosuppressive agents are used for induction, maintenance, and treatment of rejection episodes (see Tables 17-1 and 17-2).
Complications of Kidney Transplantation
Surgical Complications
Lymphocele
A lymphocele is an accumulation of lymphatic fluid around the kidney. Lymphoceles occur in 1% to 10% of pediatric renal transplant recipients.20 Because this complication is known to occur, some transplant surgeons prefer to place transplanted kidneys within the peritoneal cavity to allow lymph fluid to be reabsorbed by the peritoneum. Lymphoceles can produce fullness over the allograft, pain, or decreasing renal function. Large lymphoceles can compress the pelvis and ureter of the transplanted kidney and cause urinary obstruction. They can also cause venous obstruction.
Ultrasonography is the optimal means of imaging a lymphocele. A lymphocele appears as a fluid collection adjacent to the kidney transplant. If the diagnosis is in question, the fluid collection can be aspirated under sterile conditions using ultrasonographic guidance. Analyze fluid thus removed for creatinine level (high in urine leak, low in lymphocele), lipids (high in lymphocele, low in urine leak), and cell count (high lymphocyte count in lymphocele, low count in urine leak) to establish the diagnosis.
Treatment options include laparoscopic drainage with creation of a peritoneal window (communicating tract between the perinephric fluid collection and the peritoneum) and open creation of a peritoneal window. Laparoscopic surgery is currently the preferred method of treatment.
Wound Infection
With the use of lower doses and more rapid tapering of steroid therapy in kidney transplantation, the risk of wound infection is decreasing. Children with augmented bladders or complete diversion of the urine (e.g., ileal conduit) are at increased risk of wound infection after transplantation.14 Signs of wound infection include swelling, erythema, or purulent drainage from the incision, usually within days of transplantation.
Imaging studies are usually not needed to make the diagnosis. However, a febrile patient with wound tenderness or erythema should undergo ultrasonography or CT scan of the pelvis or abdomen to detect potentially infected perinephric fluid. Prompt surgical drainage and administration of parenteral antibiotics are required.
Thrombosis
One of the most devastating complications of transplantation, thrombosis (either of the renal artery or the renal vein) occurs in 0.5% to 3% of kidney transplants.6 Thrombosis should be suspected in any patient who demonstrates a sudden decrease in urine production after transplantation. Thrombosis may also produce a persistently elevated serum creatinine despite an adequate urine volume.
Thrombosis is readily diagnosed with the assistance of color Doppler ultrasonography. If a prompt diagnosis is made, emergency exploration will be performed to remove the thrombus, flush the kidney, and reconstruct the affected vascular anastomosis. However, once the clotting cascade is initiated in a transplanted kidney, the probability of salvage by surgery is low.
Renal Artery Stenosis
Obstruction of arterial inflow to a transplanted kidney occurs in 1% to 23% of kidney transplants.6 Renal artery stenosis should be suspected in patients who develop hypertension that is difficult to control, and appropriate evaluation should be performed. Renal artery stenosis can result from the transplantation surgery, causing constriction at the point of anastomosis, from kinking of the renal artery, or segmental hypertrophy of the intima of the renal artery or a branch thereof.
Cyclosporine causes an increase in tone of the smooth muscles of the efferent arteriole of the kidney. This commonly results in hypertension after transplantation. However, when multiple antihypertensive medications are required to control blood pressure or when controlling hypertension is impossible, the patient should be evaluated for renal artery stenosis.
Although ultrasonography may demonstrate a narrowing in the renal artery or decreased velocity of arterial flow, it is not sufficiently sensitive to confirm the diagnosis of renal artery stenosis. Digital subtraction angiography is the most sensitive diagnostic test. Three-dimensional CT scanning has also been used in making the diagnosis. Treatment options include balloon angioplasty (highest success rate with the lowest risk of complications) or open surgical revascularization.
Urologic Complications
Urologic problems are the most common surgical complications after pediatric kidney transplantation. These complications occur in approximately 7% to 13% of all kidney transplant patients.30
Obstruction
Obstruction of urine drainage may be caused by faulty surgical technique or ischemia of the distal ureter. It most often occurs at the point where the ureter is anastomosed to the bladder. Kidneys from young donors (<6 years of age) are at increased risk of ureteral ischemia because of the more tenuous blood supply present in these young children. Suspect obstruction in a recipient with hydronephrosis on posttransplantation ultrasonography, decreasing urine output, or if the expected drop in serum creatinine fails to occur.
Ultrasonography is the best method of diagnosing obstruction. When the diagnosis is in question, percutaneous antegrade pyelography and a pressure-flow study (Whitaker test) is indicated.36 Treatment options include balloon dilation or open surgical revision of the ureterovesical anastomosis. When obstruction is found in the recipient of a kidney from a pediatric donor, it may be preferable to proceed with open surgical revision. Occasionally, in such cases, complete loss of the ureter may be observed. When this occurs, anastomosis of the native ureter to the transplant kidney pelvis may be possible.
Urine Leak
A breakdown in the anastomosis of the ureter to the bladder results in leak of urine into the perinephric space. This can result from a technical failure or from ischemia of the distal ureter. Ischemic necrosis is more common in recipients of kidneys from pediatric (<6 years of age) donors. A urine leak may manifest as a leak of fluid from the incision, a perinephric fluid collection observed on ultrasonography, or a failure of the serum creatinine to decrease as expected after transplantation.
When significant drainage from the incision follows kidney transplantation, send a sample of the fluid for creatinine measurement. A high creatinine level (two times the serum creatinine) differentiates urine leak from other causes of perinephric fluid collections such as lymphocele or hematoma. If the volume of the leak is small, temporary diversion of urine with a nephrostomy tube and Foley catheter may be successful. A high-volume leak and persistent leak are treated with open surgical repair.
Use of cadaver allografts from infants is associated with an increased risk of ureteral complications (stenosis and leakage) because of the tenuous blood supply of infant ureters. Flechner et al. used a novel adaptation to the technique of en bloc infant kidney transplantation, harvesting the trigone and adjacent tissues intact and transplanting this entire segment, as a patch, onto the recipient bladder.11
Summary
Optimal outcome after solid organ transplantation in children requires careful patient selection, surgical technique, early postoperative care, and long-term follow-up and immunosuppression. Postoperative care of these patients requires careful assessment of patient cardiorespiratory and graft function, and immediate detection and treatment of surgical complications, including graft failure, infection and rejection.
The use of immunosuppressive therapy requires a careful balance to maintain adequate immunosuppression and maximize graft survival while minimizing short- and long-term complications. The pediatric transplant recipient and family require skilled physical, immunologic, and psychosocial support.
1 Boucek M.M., et al. Pediatric heart transplantation. In Allen J.D., Driscoll D.J., Shaddy R.E., Feltes T.F., editors: Moss & Adam's heart disease in infants, children, & adolescents, 7th ed, Philadelphia: Lippincott Williams & Wilkins, 2008.
2 Bucuvalas J.C., Ryckman F.C. Long-term outcome after liver transplantation in children. Pediatr Transplant. 2002;6(1):30-36. [Medline]
3 Calne R.Y., Alexandre G.P., Murray J.E. A study of the effects of drugs in prolonging survival of homologous renal transplants in dogs. Ann NY Acad Sci. 1962;99:743-761. [Medline]
4 Chang F.Y., et al. Fever in liver transplant recipients: changing spectrum of etiologic agents. Clin Infect Dis. 1998;26(1):59-65. [Medline]
5 Collins K.K., et al. Atrial tachyarrhythmias and permanent pacing after pediatric heart transplantation. J Heart Lung Transplant. 2003;22(10):1126-1133.
6 Dimitroulis D., et al. Vascular complications in renal transplantation: a single-center experience in 1367 renal transplantations and review of the literature. Transplant Proc. 2009;41(5):1609-1614.
7 Edwards N.M., Chen J.M., Mazzeo P.A. Cardiac transplantation. Totowa, NJ: Humana Press; 2004.
8 Evrard V., et al. Impact of surgical and immunological parameters in pediatric liver transplantation: a multivariate analysis in 500 consecutive recipients of primary grafts. Ann Surg. 2004;239(2):272-280. [Medline]
9 Filler G., Huang S.H. Progress in pediatric kidney transplantation. Ther Drug Monit. 2010;32(3):250-252.
10 Fine R.N., et al. Pediatric transplantation of the kidney, liver and heart: summary report. Pediatr Transplant. 2004;8(1):75-86. [Medline]
11 Flechner S.M., et al. Use of the donor bladder trigone to facilitate pediatric en bloc kidney transplantation. Pediatr Transplant. 2011;15(1):53-57. [Medline]
12 Gulati A., Sarwal M.M. Pediatric renal transplantation: an overview and update. Curr Opin Pediatr. 2010;22(2):189-196.
13 Guthery S.L., et al. Determination of risk factors for Epstein-Barr virus-associated posttransplant lymphoproliferative disorder in pediatric liver transplant recipients using objective case ascertainment. Transplantation. 2003;75(7):987-993. [Medline]
14 Hatch D.A., et al. Kidney transplantation in children with urinary diversion or bladder augmentation. J Urol. 2001;165(6 Pt 2):2265-2268. [Medline]
15 Hume D.M., et al. Experiences with renal homotransplantation in the human: report of nine cases. J Clin Invest. 1955;34(2):327-382. [Medline]
16 Imamura M., et al. Bridge to cardiac transplant in children: berlin heart versus extracorporeal membrane oxygenation. Ann Thorac Surg. 2009;87:1894-1901.
17 Kamath B.M., Olthoff K.M. Liver transplantation in children: update 2010. Pediatr Clin North Am. 2010;57(2):401-414.
18 Kling K., Lau H., Colombani P. Biliary complications of living related pediatric liver transplant patients. Pediatr Transplant. 2004;8(2):178-184. [Medline]
19 LaPage M.J., Rhee E.K., Canter C.E. Tachyarrhythmias after pediatric heart transplantation. J Heart Lung Transplant. 2010;29(3):273-277. Epub Sept 26, 2009
20 La Rosa C., Jorge Valuarte H., Meyers K.E.C. Outcomes in pediatric solid-organ transplantation. Pediatr Transplant. 2011;15:128-141.
21 Murray J.E., Merrill J.P., Harrison J.H. Kidney transplantation between seven pairs of identical twins. Ann Surg. 1958;148(3):343-359. [Medline]
22 Nobili V., et al. Mycophenolate mofetil in pediatric liver transplant patients with renal dysfunction: preliminary data. Pediatr Transplant. 2003;7(6):454-457. [Medline]
23 Ooi C.Y., et al. Thrombotic events after pediatric liver transplantation. Pediatr Transplant. 2010;14(4):476-482. Epub October 22, 2009
24 Organ Procurement and Transplantation Network: National Data Reports. OPTN. Available at http://optn.transplant.hrsa.gov/latestData/viewDataReports.asp Accessed 09.05.11
25 Paya C.V., et al. Risk factors for cytomegalovirus and severe bacterial infections following liver transplantation: a prospective multivariate time-dependent analysis. J Hepatol. 1993;18(2):185-195. [Medline]
26 Reding R. Tacrolimus in pediatric liver transplantation. Pediatr Transplant. 2002;6(6):447-451. [Medline]
27 Roche S.L., O'sullivan J.J., Kantor P.F. Hypertension after pediatric cardiac transplantation: detection, etiology, implications and management. Pediatric Transplantation. 2010;14(2):159-168. Epub July 16, 2009
28 Rossano J.W., et al. B-type natriuretic peptide levels late after transplant predict graft survival in pediatric heart transplant patients. J Heart Lung Transplant. 2010;29(3):385-386. Epub September 26, 2009
29 Shapiro R., Sarwal M. Pediatric kidney transplantation. Pediatr Clin North Am. 2010;57(2):393-400.
30 Shoskas D.A., et al. Urologic complications in 1,000 consecutive renal transplant recipients. J Urol. 1995;153:18-21.
31 Spada M., et al. Pediatric liver transplantation. World J Gastroenterol. 2009;15(6):648-674.
32 Starzl T.E., et al. Homotransplantation of the liver in humans. Surg Gynecol Obstet. 1963;117:659-676. [Medline]
33 Starzl T.E., Marchioro T.L., Waddell W.R. The reversal of rejection in human renal homografts with subsequent development of homograft tolerance. Surg Gynecol Obstet. 1963;117:385-395. [Medline]
34 Takada Y., Tanaka K. Living related liver transplantation. Transplant Proc. 2004;36(2 Suppl.):271S-273S. [Medline]
35 Terasaki P.I., et al. Serotyping for homotransplantation. V. Evaluation of a matching scheme. Transplantation. 1966;4(6):688-699. [Medline]
36 Whitaker R.H. Clinical assessment of pelvic and ureteral function. Urology. 1978;12(2):146-150. [Medline]