External preparations
 Skin structure and sites of action of drugs
Skin structure and sites of action of drugs
 The types and functions of solid, liquid and semi-solid skin preparations
The types and functions of solid, liquid and semi-solid skin preparations
 The ingredients used in skin preparations
The ingredients used in skin preparations
Introduction
Skin is the largest organ in the body and has three distinct regions. The hypodermis is the innermost and is often called subcutaneous fat. The dermis is the bulk of the thickness of the skin and contains blood vessels, nerve fibres, sweat glands and hair follicles. The outermost region is the epidermis, which is made up of several layers. One of these layers is the stratum basale, in which cells divide and as they move towards the surface, they change appearance and function. The outermost layer, the stratum corneum, acts as the skin barrier. It is made up of about 20 layers of dead keratinized cells. The hair follicles and sweat ducts pass through the stratum corneum to reach the surface. A simplified diagram showing the main skin structures is given in Figure 36.1.
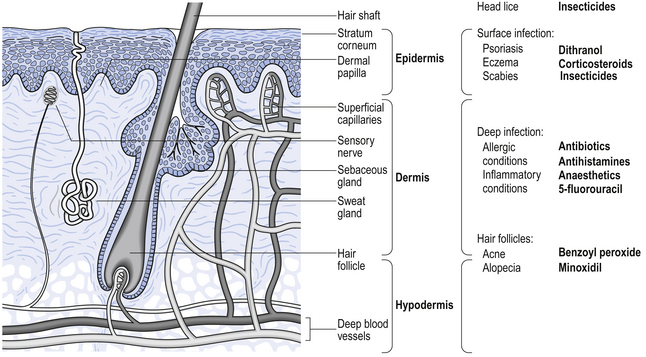
Figure 36.1 Diagrammatic representation of the skin showing the main structures, location of diseases and the sites of action of drugs.
There are a large number of diseases which may affect different regions of the skin. Any drug used will need to reach the site of the disease in order to act. Unless it is for a surface effect only, the drug must either pass through the stratum corneum or go through hair follicles or sweat ducts. Examples of drugs applied to the skin and their sites of action are shown in Figure 36.1. Once in the skin, a lipid-soluble drug will tend to accumulate in lipid regions, while more water-soluble drugs will tend to enter the blood capillaries and be removed from the skin. There are also many metabolic enzymes in the skin which can deactivate drugs.
Effective formulation makes it possible to achieve adequate and reproducible percutaneous absorption, which is close to zero order kinetics. As a consequence, absorption through the skin from toxic materials is possible and so gloves should always be worn when preparing external preparations.
There are an increasing number of drugs that are effective against skin diseases, but drugs are not the only way of treating skin conditions. Creating physiological changes in the skin can also be beneficial. The main change is to control the moisture content of the skin. Normal skin has 10–25% moisture in the stratum corneum. This level may be reduced in, for example, eczema, or increased, as in skin maceration between the toes. By using an occlusive product (that is, an oily product), water leaving the body through the skin will be trapped and the moisture content will increase. These products are called emollients. An excess of moisture may be removed using an astringent, a hygroscopic material or, to a lesser extent, a dusting powder. Where an oily vehicle is needed, but moisture must not increase, adding solid particles to the vehicle will allow water to escape. Lubrication of sensitive skin is achieved by using finely divided solids, applied either as a powder or, more efficiently, as a suspension. Cooling the skin relieves inflammation and eases discomfort. It is achieved by evaporating a solvent, usually water or a water and alcohol mixture. Volatile solvents sprayed on the skin give intense cooling.
Types of skin preparation
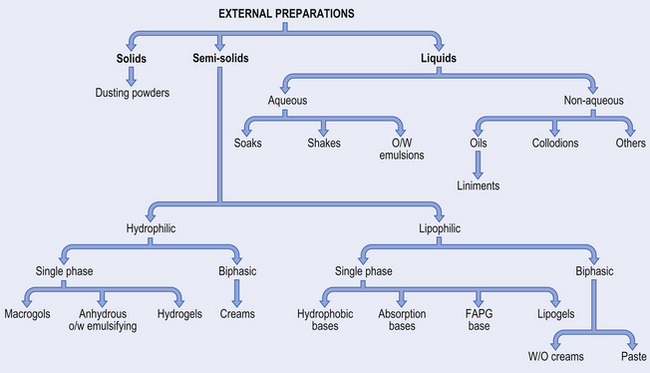
There are a large number of different types of external medicine, ranging from dry powders through semi-solids to liquids. The names are often traditional making classification difficult. Figure 36.2 illustrates the formulation of the main types of preparation used on the skin.
Solids
Dusting powders are applied to the skin for a surface effect such as drying or lubricating, or an antibacterial action. They are made of a fine-particle-size powder, which may be a drug alone or together with excipients.
Liquids
Soaks have an active ingredient dissolved in an aqueous solvent and are often used as astringents, for cooling or to leave a film of solid on the skin. Oily vehicles can be used in bath additives to leave an emollient film on the skin surface.
Lotions are aqueous solutions, suspensions (see Example 36.1) or emulsions that cool inflamed skin and deposit a protective layer of solid.
Liniments are alcoholic or oily solutions or emulsions (see Example 36.2) designed to be rubbed into the skin. The medicament is usually a rubefacient.
Applications are solutions or emulsions that frequently contain parasiticides (see Example 36.3).
Paints and tinctures are concentrated aqueous or alcoholic antimicrobial solutions.
Collodions are organic solvents containing a polymer and drug.
There are also many other liquid products including shampoos and foot washes.
Semi-solids
Ointments are usually oily vehicles that may contain a surfactant to allow them to be washed off easily (barrier creams). They are used as emollients, or for drug delivery either to the surface or for deeper penetration.
Creams are traditionally oil-in-water (o/w) emulsions while oily creams are water-in-oil (w/o) emulsions. However, there are also ‘creams’ that are not emulsions. Emulsified creams usually give cooling, are less greasy than ointments and can be used for drug delivery onto or into the skin. They require antimicrobial preservatives.
Pastes are vehicles (aqueous or oily) with a high concentration of added solid. This makes them thick so they do not spread and so localizes drug delivery (e.g. Dithranol in Lassar’s Paste, see Example 36.9). They can also be used for sun blocks.
Gels are usually aqueous gels used for lubrication or applying a drug to the skin. Oily gels are also available where occlusion is required.
Ingredients used in skin preparations
Water-miscible vehicles
These include water, alcohol and the macrogols. Alcohol, usually industrial denatured alcohol, is often added to water to increase the rate of evaporation and produce a more intense cooling effect. The macrogols (polyethylene glycols) are available with a range of molecular weights. As chain length increases, so the properties change from liquid, through semi-solid to waxy solid. They have good solvent properties for a wide range of drugs and can be blended to produce intermediate consistencies. They tend to dry the skin, inactivate some antimicrobials, interact with some plastics and can give poor release of drugs.
Oily vehicles
Oils used in external preparations come from one of three sources.
Mineral oils (paraffins) are the most widely used. They are complex mixtures of mainly saturated hydrocarbons, which are available in different fractions. Different names are used in different pharmacopoeias (Table 36.1).
Table 36.1
Paraffins used in external preparations: the names used are different in the UK, USA and European pharmacopoeias
| UK | USA | European |
| Light liquid paraffin | Light mineral oil | Paraffinium perliquidum |
| Liquid paraffin | Mineral oil | Paraffinium liquidum |
| Soft paraffin | Petrolatum | Paraffinium molle |
| Hard paraffin | Paraffin | Paraffinium durum |
Light liquid paraffin is not normally used in external medicines. Soft paraffin is the main ingredient in many products, with liquid or hard paraffin being used to thin or thicken them respectively. There are two forms of soft paraffin: yellow and white. The latter has been bleached, residues of which may remain. As a general rule, white is used with white or pale coloured ingredients, while yellow is used for darker ingredients. The paraffins are occlusive and chemically inert, but do not give good skin penetration.
Vegetable oils come from plant sources such as castor, olive, peanut and coconut. The latter two would be rarely used because of severe reactions in patients with nut allergies.
Synthetic oils, such as the silicone oils (Dimethicone BP), are used as water repellents and occlusives because they are very hydrophobic. The semi-synthetic isopropylmyristate is similar to vegetable oil in its properties and use.
Emulsifying agents
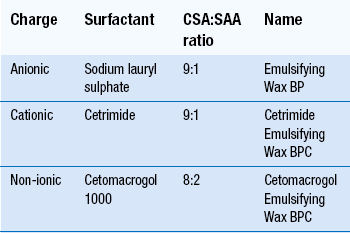
Liquid and semi-solid emulsions, both o/w and w/o, are used externally and require the addition of emulsifying agents. The latter may also be added to an oil without water as in Emulsifying Ointment BP. The presence of a surfactant usually increases the skin penetration of any drug. A wide range of materials can be used as surfactants, either alone or in combinations. Selection is made in view of the type of emulsion required (o/w or w/o) and the charge on the other ingredients (anionic, cationic or non-ionic).
Emulsifiers – w/o
Hypoallergenic commercial wool fat is a complex mixture of fatty acid esters of cholesterol and other sterols and alcohols. Wool alcohol, a solid, is richer in cholesterol and lanosterol and has fewer impurities. Both it and wool fat increase the ‘water-holding’ capacity of greasy bases. Hydrous wool fat is 7 parts wool fat and 3 parts water and is a softer material. Beeswax is a traditional w/o emulsifier which is occasionally used.
Emulsifiers – o/w
Emulsifying waxes each contain two ingredients: cetostearyl alcohol and a surface-active agent, as shown in Table 36.2. All three bases are waxy solids that mix with oily materials. Addition of water produces an o/w emulsion – a cream. Both the non-aqueous blends and the creams are easily washed off the skin. Varying the amount of bodying agent, usually cetostearyl alcohol, can control consistency. The ratio of oil to water will also alter the consistency of a cream.
Other emulsifiers
Calcium soaps and soft soap have traditionally been used to make o/w emulsions.
Synthetic surface-active agents are used particularly in commercial products. Low HLB (hydrophilic–lipophilic balance) materials will produce w/o emulsions, while higher HLB surfactants give o/w emulsions.
Suspending agents
These materials can be used for suspending solids in lotions, or to produce gels, depending on the concentration used. Examples are the clays, of which there are many forms, including bentonite, attapulgite, montmorillonite and Veegum® (aluminium magnesium silicate). They leave a lubricant layer of powder on the skin. They are unsuitable for use below pH 3.5 and their consistency may be affected by alcohol and electrolytes (see Example 34.5).
Gelling agents can be used to produce a wide range of consistency from slightly thickened (as in artificial tears), through lubricants and semi-solids for the delivery of drugs to very thick bases used to immobilize the skin. For aqueous gels the materials used include tragacanth, alginates, pectin, gelatin, methylcelluloses, carbomer, polyvinyl alcohol and clays. Oils may be thickened using cetostearyl alcohol, hard paraffin, beeswax, wool alcohols and polyvalent soaps such as magnesium stearate. The latter, when heated with an oil, produces a clear ‘lipogel’.
Other ingredients
Wetting agents are required for hydrophobic solids. Tincture of quillaia is the traditional material (see Example 36.1), but alcohol alone may be effective. Synthetic materials, such as Manoxol OT, can also be used.
Humectants are materials added to reduce the rate of water loss from creams and gels. They are all hygroscopic materials and include glycerol, propylene glycol, PEG 300 and sorbitol syrup, typically used at concentrations of 5–15%.
Solids may be added to semi-solid occlusive bases. They provide channels for the migration of water from the skin surface and so reduce the occlusiveness. Solids used include zinc oxide, talc, starch and Aerosil®. Some, such as talc, must be sterilized to kill bacterial spores.
Whenever there is a danger of microbial growth, antimicrobial preservation is required.
Preparation of external preparations
A wide range of dispensing techniques are used in compounding external medicines, some of which have been reviewed in other chapters (see Chs 30, 33, 34, 35, 38). In the section that follows, only those types of product which require different dispensing techniques are described in detail.
Dusting powders
A simple mixing in a mortar and pestle using ‘doubling-up’ is used (see Ch. 38). Sieving may be necessary to disperse aggregates of cohesive powders. A 180 μm sieve should be used. Powders such as starch, which contains a lot of moisture, may need drying to ensure optimum flow properties. With coloured materials, considerable working with the pestle is required before proceeding to ‘doubling-up’, otherwise a speckled product may result. A liquid may be added by pipette to a small quantity of the powder and be worked in before further mixing. A worked example of a dusting powder is given in Example 38.3.
Liquid preparations
These include solutions, suspensions and emulsion. Most liquid preparations are used unsterilized, but if they are intended for application to broken skin, eyes or body cavities, they should be sterilized. They should be labelled ‘For external use only’, and carry a ‘Shake the bottle’ label if they are emulsions or suspensions. Worked examples are given, of a lotion in Example 34.3 and of an oily lotion in Example 35.3.
Formulation notes. This an example of a ‘shake’ lotion, an aqueous suspension prepared without a suspending agent, but including a wetting agent for the hydrophobic sulfur.
Method of preparation. Sieve the precipitated sulfur. Weigh out 4 g and place in a glass mortar. Using a 1 mL pipette, add 0.5 mL quillaia tincture and work well into the sulfur using a pestle. Add 6 mL of industrial methylated spirits followed by 2 mL glycerol, working in after each addition (thus achieving maximum wetting before water is added). Add 20–30 mL calcium hydroxide solution to produce a pourable suspension. Transfer to a tared bottle. Rinse the mortar with calcium hydroxide solution, adding it to the bottle, before making up to volume.
Shelf-life and storage. There are no special requirements for storage. An expiry date of 4 weeks is suitable.
Formulation notes. The methyl salicylate requires to penetrate the skin. The vegetable oil, arachis oil, is used as the solvent to assist in the penetration of the skin by methyl salicylate. Other similar fixed oils can be used.
Method of preparation. Measure 25 mL of methyl salicylate in a 100 mL measure and add arachis oil to make up to volume. Transfer to a dry 100 mL amber ribbed bottle.
Shelf-life and storage. This liniment should be kept in a well-closed container in a cool place. An expiry date of 4 weeks is appropriate.
Formulation notes. Benzyl benzoate is water immiscible and is being emulsified using the anionic Emulsifying Wax BP. The application is an o/w emulsion.
Method of preparation. Weigh the emulsifying wax and place it in an evaporating basin on a water bath or hot plate to melt. Add the benzyl benzoate and mix and warm. Warm about 75 mL of the water to the same temperature. Add about half of this to the evaporating basin and mix very gently. Transfer the mixture, again very gently to avoid frothing, to a tared bottle. Add warmed water to volume. Close the bottle and shake vigorously. Care is required to avoid frothing when water is present, because it will be very difficult to make up to the tare mark when froth has formed. Shake frequently during cooling.
Shelf-life and storage. The application should be kept in a cool place, but not be allowed to freeze. An expiry date of 4 weeks is appropriate.
Semi-solid preparations
Mixing by fusion
The compounding of many semi-solid preparations includes the blending together of oily materials, some of which are solids at room temperature. The process called ‘mixing by fusion’ achieves this. As the name implies, it involves melting the ingredients together (see Example 36.4). The process is carried out in an evaporating basin on a water bath or hot plate. It should be noted that a high temperature is not required so 60–70°C is usually adequate. Waxy solids should be grated before weighing and should be added first, so that melting can start while other ingredients are being measured. When all the ingredients are melted, remove the basin from the water bath and gently stir until cold. Mixing, which should be gentle to avoid air bubbles, is necessary to avoid lumps forming. This could happen because the higher melting point ingredients in the eutectic system may precipitate out. Any medicament may be added at different stages of preparation depending on its properties. If soluble and stable, it can be added when the base is molten. If it is less stable, or insoluble but easy to disperse, it can be added during cooling. However, if it is unstable or if dispersion is difficult, it should be added when cold using mixing by trituration.
When evaporating basins are being used, recovery of all the product is not possible. Thus, in order to be able to pack the prescribed amount, it is necessary to make an excess of about 10%.
Formulation notes. This is a simple blend of solid and semi-solid oily ingredients made by fusion. Yellow or white soft paraffin is chosen according to the colour of the finished product. In this case, since there is nothing else to be added, white soft paraffin should be used; 60 g is made to allow 50 g to be dispensed.
Method of preparation. Grate the hard paraffin and cetostearyl alcohol. Weigh 3 g of each and place in an evaporating basin on a water bath or hot plate. Weigh the wool fat, using a piece of paper to allow full recovery of the material, and add it to the evaporating basin, followed by the soft paraffin (also weighed on paper). Stir gently until fully melted. Remove from the heat and continue to stir gently until cold. Weigh 50 g of base into a tared ointment jar. If an ointment jar is used, a greaseproof paper disc should be placed on the surface of the ointment to protect the liner of the lid from the greasiness.
Shelf-life and storage. Store in a cool place. An expiry date of 4 weeks is appropriate.
Mixing by trituration
Insoluble solids or liquids are incorporated into bases using the technique called ‘mixing by trituration’. Any powders should be passed through a 180 μm sieve before weighing to avoid grittiness in the finished product. Mixing by trituration is carried out on an ointment slab or tile, which may be made of glass or glazed porcelain. A flexible spatula is used to work the materials together. Powders are placed on the tile and incorporated into the base using ‘doubling-up’ as it is worked in. However, it is usually necessary to have two to three times the volume of base to powder, otherwise it will ‘crumble’. Liquids, if present, are usually present in small amounts. To incorporate a liquid, a portion of the base is placed on the slab and a recess made to hold the liquid which is then worked in gently. Larger quantities of liquid should be added a little at a time using the same method. In theory it is possible to recover all material from the slab, but it is normal to allow up to 10% excess for losses. These processes can be carried out in a mortar with a flat base using a pestle with a flat head. However, because recovery of the product is difficult, this is usually reserved for larger-scale batches.
Formulation notes. The BP directs that the simple ointment be prepared with white soft paraffin. If simple ointment is available, the trituration can be carried out on a slab and all the product recovered. However, if simple ointment is also being made, 50 g should be adequate to ensure that 45 g are available. Precipitated sulfur, while of smaller particle size than sublimed sulphur, can give a gritty feel unless it is passed through a 180 μm sieve.
Method of preparation. Sieve and then weigh out the precipitated sulfur and place it on the slab. Weigh out the simple ointment (using a piece of paper to prevent it sticking to the balance), and place it on a different part of the slab. Take a portion of the sulfur and a portion of the base of about three times the volume of the sulfur and work them together vigorously until there is no sign of any particles of sulfur. Spreading a thin layer on the slab helps check this. Gradually add the remaining sulfur and base. Collect the ointment together on the slab using the spatula and pack 50 g.
Shelf-life and storage. Store in a cool place. An expiry date of 4 weeks is appropriate.
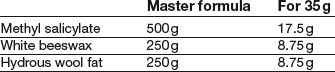
Formulation notes. Methyl salicylate is a liquid. With the high proportion present, the product would be runny without the addition of the beeswax as a thickening agent. The base ingredients require to be blended by fusion.
Method of preparation. Grate and weigh the beeswax. Melt it with the hydrous wool fat (weighed on a piece of paper) in an evaporating basin on a water bath or hot plate. Remove from the heat and stir until almost cold before adding the methyl salicylate (it is volatile). Continue stirring until cold. Pack 30 g in a glass ointment jar (plastic should be avoided with methyl salicylate).
Shelf-life and storage. Store in a cool place. An expiry date of 4 weeks is appropriate.
Creams
Creams are emulsified preparations containing water. They are susceptible to microorganisms which may cause spoilage of the cream or disease in the patient. While preservatives are included, they are usually inadequate to cope with a heavy microbial contamination and growth so the possibility of microbial contamination during preparation should be minimized. Ideally aseptic techniques should be used, but this is not normally possible in extemporaneous dispensing and so thorough cleanliness is employed. As a minimum, all apparatus and final containers should be thoroughly cleaned and rinsed with freshly boiled and cooled purified water, then dried just prior to use. Swabbing of working surfaces, spatulas and other equipment with ethanol will also reduce the possibility of microbial contamination.
The basic method of making an emulsified cream is to warm both the oily phase and aqueous phase separately to a temperature of about 60°C, mix the phases and stir until cold. It is important that the temperatures of the two phases are within a few degrees of each other and it is advisable to use a thermometer to check this. Rapid cooling will cause the separation of high melting point materials, and excessive aeration as a result of vigorous stirring will produce a granular appearance in the product. Medicaments may, if they are stable, be dissolved in the appropriate phase before emulsification, or can be added by trituration when cold.
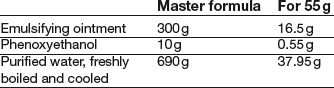
Formulation notes. This is an o/w cream made using an anionic emulsifying agent. To reduce the risk of microbial contamination, all equipment should be washed before use. Phenoxyethanol is present as an antimicrobial preservative. It is a liquid, so has to be weighed, or, if its density is obtained, it could be measured by pipette. If the emulsifying ointment has to be made, exactly 16.5 g can be made because the emulsification can be carried out in the same evaporating basin.
Method of preparation. The phenoxyethanol is dissolved in the water warmed to 60°C. Weigh the emulsifying ointment (using a piece of paper to prevent it sticking) and melt it in an evaporating basin on a water bath or hot plate. Ensure that both phases are close to 60°C, then add the aqueous phase to the melted ointment. Remove from the heat and stir continuously until cold, taking care not to incorporate too much air. Weigh 50 g and pack in an ointment jar.
Shelf-life and storage. The preparation should be stored in a cool place, but not allowed to freeze. A shelf-life of 2–3 weeks is appropriate because the preparation has not been made in the cleanest conditions.
Formulation notes. This is a w/o cream prepared using wool alcohols as the emulsifying agent. Phenoxyethanol is present as preservative, but all equipment should be washed before use. Phenoxyethanol is a liquid and so must be weighed, or, if its density is obtained, it can be measured by pipette. Quantities for 55 g produce amounts that cannot be weighed on a dispensing balance, so 60 g is made. If the wool alcohols ointment is also to be made, exactly 30 g is adequate, because it does not have to be removed from the evaporating basin.
Method of preparation. All equipment should be thoroughly cleaned before use. Dissolve the magnesium sulphate and phenoxyethanol in the water and warm to 60°C on a water bath or hot plate. Weigh the wool alcohols ointment, using a piece of paper, and melt it in an evaporating basin at 60°C. Check that the two temperatures are the same. Add the water, little by little, to the ointment, stirring constantly until a smooth creamy mixture is produced, while maintaining the temperature at 60°C. When all the water is added, remove from the heat and stir gently until the cream is at room temperature. Pack 50 g in an ointment jar.
Shelf-life and storage. Store in a cool place but do not allow to freeze. If liquid separates on storage, stirring may reincorporate it. An expiry date of 4 weeks is appropriate.
Dilution of creams
It is sometimes necessary to prepare a dilution of a commercially produced cream, although the practice is undesirable. Choice of diluent is crucial, since the diluent may impair the preservative system in the cream, may affect the bioavailability of the medicament, or be incompatible with other ingredients. The process of dilution also increases the risk of microbial contamination. Thus, dilutions should only be made with the diluent(s) specified in the manufacturer’s data sheet and heat must be avoided. All diluted creams should be freshly prepared and be given a 2-week shelf-life.
Pastes
Pastes are dispersions of high concentrations of solid in either an aqueous or oily vehicle. They can be used to treat infections by making use of their high osmotic pressure, or as very thick materials to prevent irritant drugs spreading over the skin surface. Incorporation of the solid is by mixing on an ointment slab.
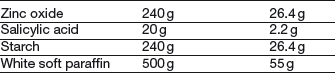
Formulation notes. The Lassar’s paste has to be made first before incorporating the dithranol. Dithranol is prone to oxidation, so contact with metal should be avoided. Gloves should be worn during preparation.
Method of preparation. Sieve the zinc oxide and salicylic acid through a 180 μm sieve before weighing. Weigh the soft paraffin (on a piece of paper) and melt in an evaporating basin on a water bath. Take some of the powder and stir into the melted base. Continue until all the powder is added, then stir gently until cold. Weigh out the Lassar’s paste (using paper to avoid sticking). Only when the Lassar’s paste has been completed, weigh out the dithranol. Care is required because it is very irritant to skin. Place it on a slab and incorporate it in a small portion of the paste using a plastic spatula, ensuring that a smooth, even product is produced. Dilute gradually with the remainder of the paste. Pack in a brown ointment jar, with a circle of greaseproof paper and a tight-fitting closure.
Shelf-life and storage. The product should be kept in a cool place, protected from light. An expiry date of 2 weeks is appropriate because of chemical instability.
Transdermal delivery systems
Transdermal drug delivery systems aim to provide continuous drug release over a period of time which can be from a few hours to 7 days.
The principle of this dosage form is that, by optimization of physicochemical factors, the drug is absorbed through the skin into the systemic circulation. Absorption through the skin is variable so the rate of release of the drug must be controlled to a slower rate than the skin can absorb it. This may be achieved either by using a matrix system or a rate-limiting membrane. These devices are commonly known as ‘patches’. Drugs available as transdermal therapeutic systems include Glyceryl trinitrate, Oestradiol, Nicotine, Hyoscine, Testosterone, Fentanyl, Rivastigmine. Transdermal therapeutic systems are always produced by pharmaceutical manufacturers because of the technology involved.
Advantages
 Continuous drug delivery, producing steady-state plasma levels
Continuous drug delivery, producing steady-state plasma levels
 No drug deactivation in the gastrointestinal tract
No drug deactivation in the gastrointestinal tract
 No first pass effect, as the liver is bypassed (although there is metabolism in the skin)
No first pass effect, as the liver is bypassed (although there is metabolism in the skin)
 Cessation of treatment by removing the patch. (This is not immediate because of a reservoir effect which will continue to deliver drug from the skin for several hours.)
Cessation of treatment by removing the patch. (This is not immediate because of a reservoir effect which will continue to deliver drug from the skin for several hours.)
Although these are benefits, various problems are associated with this type of dosage form. For these reasons, few drugs so far have been formulated in this way.
Disadvantages
 Only potent drugs, i.e. those with a small therapeutic dose, are suitable to be incorporated into a patch. Skin permeability is inadequate to allow larger doses from an acceptable size of patch
Only potent drugs, i.e. those with a small therapeutic dose, are suitable to be incorporated into a patch. Skin permeability is inadequate to allow larger doses from an acceptable size of patch
 Because the drug is being absorbed through the skin, lipid-soluble drugs are most likely to be effective
Because the drug is being absorbed through the skin, lipid-soluble drugs are most likely to be effective
 Drugs with long half-lives are not suitable for this type of formulation
Drugs with long half-lives are not suitable for this type of formulation
 There have been reports of local skin reactions due to irritancy by drugs To minimize possible skin reactions, new patches should be placed on fresh skin each time, the same site being not used for at least 7 days
There have been reports of local skin reactions due to irritancy by drugs To minimize possible skin reactions, new patches should be placed on fresh skin each time, the same site being not used for at least 7 days
 In some instances the steady-state blood levels have produced tolerance, e.g. glyceryl trinitrate. This has led to the practice of patients being given a ‘nitrate-free’ period which prevents tolerance occurring
In some instances the steady-state blood levels have produced tolerance, e.g. glyceryl trinitrate. This has led to the practice of patients being given a ‘nitrate-free’ period which prevents tolerance occurring
 Steady-state blood levels of nicotine have caused central nervous system disturbance; in particular, patients have reported suffering nightmares. Normally, nicotine levels in a smoker will fall during the hours of sleep as no cigarette smoking occurs. No such fall will occur when 24-h nicotine patches are used. For this reason, manufacturers have developed patches which are applied for 16 h then removed. A new one is applied 8 h later.
Steady-state blood levels of nicotine have caused central nervous system disturbance; in particular, patients have reported suffering nightmares. Normally, nicotine levels in a smoker will fall during the hours of sleep as no cigarette smoking occurs. No such fall will occur when 24-h nicotine patches are used. For this reason, manufacturers have developed patches which are applied for 16 h then removed. A new one is applied 8 h later.
Method of use
It is important that patients are informed how to use patches correctly. All patients who purchase or are prescribed patches should be given the following information about their use:
 To ensure adequate adhesion, the patch must be applied to a clean, dry area of skin
To ensure adequate adhesion, the patch must be applied to a clean, dry area of skin
 The old patch must always be removed before applying a new one
The old patch must always be removed before applying a new one
 When a patch is replaced with a new one it must be applied to a different area of skin. The area of skin from which a patch has just been removed will be soft and possibly moist. This alters the permeability of the skin. In order to maintain the same level of drug absorption, a different, intact area of skin must be used
When a patch is replaced with a new one it must be applied to a different area of skin. The area of skin from which a patch has just been removed will be soft and possibly moist. This alters the permeability of the skin. In order to maintain the same level of drug absorption, a different, intact area of skin must be used
 The patch must be disposed of carefully. It should be folded together to prevent it being stuck on to another person’s skin. Particular care should be taken to keep patches away from children.
The patch must be disposed of carefully. It should be folded together to prevent it being stuck on to another person’s skin. Particular care should be taken to keep patches away from children.
Key Points
 Drugs applied to the skin are usually for a local effect, although systemic action is possible
Drugs applied to the skin are usually for a local effect, although systemic action is possible
 Skin preparations may be solids, liquids or semi-solids
Skin preparations may be solids, liquids or semi-solids
 For liquids and semi-solids, the vehicles may be water based, water miscible, oily or emulsified
For liquids and semi-solids, the vehicles may be water based, water miscible, oily or emulsified
 A wide range of emulsifying agents may be used to produce either o/w or w/o emulsions
A wide range of emulsifying agents may be used to produce either o/w or w/o emulsions
 Suspending agents used on the skin are usually clays
Suspending agents used on the skin are usually clays
 Other ingredients include wetting agents, humectants and finely divided solids
Other ingredients include wetting agents, humectants and finely divided solids
 All skin preparations should carry the label ‘For external use only’
All skin preparations should carry the label ‘For external use only’
 Dusting powders are simple mixtures made by ‘doubling-up’
Dusting powders are simple mixtures made by ‘doubling-up’
 Lotions are aqueous solutions, suspensions or emulsions
Lotions are aqueous solutions, suspensions or emulsions
 Liniments are oily solutions or emulsions
Liniments are oily solutions or emulsions
 Mixing by fusion is the process of melting together the ingredients of ointment bases followed by stirring until cold
Mixing by fusion is the process of melting together the ingredients of ointment bases followed by stirring until cold
 Mixing by trituration is the incorporation of solids or liquids into semi-solid vehicles on an ointment slab
Mixing by trituration is the incorporation of solids or liquids into semi-solid vehicles on an ointment slab
 Cleanliness is essential when making creams to avoid excessive microbial contamination
Cleanliness is essential when making creams to avoid excessive microbial contamination
 Transdermal delivery systems (skin patches) are used to give prolonged constant plasma concentrations for a number of drugs
Transdermal delivery systems (skin patches) are used to give prolonged constant plasma concentrations for a number of drugs
 Patients must be carefully counselled on the use of skin patches
Patients must be carefully counselled on the use of skin patches