45 Mechanisms of Viral Pathogenesis
Viruses cause disease after they break through the natural protective barriers of the body, evade immune control, and either kill cells of an important tissue (e.g., brain) or trigger a destructive immune and inflammatory response. The outcome of a viral infection is determined by the nature of the virus-host interaction and the host’s response to the infection (Box 45-1). The immune response is the best treatment, but it often contributes to the pathogenesis of a viral infection. The tissue targeted by the virus defines the nature of the disease and its symptoms. Viral and host factors govern the severity of the disease; they include the strain of virus, the inoculum size, and the general health of the infected person. The ability of the infected person’s immune response to control the infection determines the severity and duration of the disease. A particular disease may be caused by several viruses that have a common tissue tropism (preference), such as hepatitis—liver, common cold—upper respiratory tract, encephalitis—central nervous system. On the other hand, a particular virus may cause several different diseases or no observable symptoms. For example, herpes simplex virus type 1 (HSV-1) can cause gingivostomatitis, pharyngitis, herpes labialis (cold sores), genital herpes, encephalitis, or keratoconjunctivitis, depending on the affected tissue, or it can cause no apparent disease at all. Although normally benign, this virus can be life threatening in a newborn or an immunocompromised person.
Box 45-1
Determinants of Viral Disease
Viruses encode activities (virulence factors) that promote the efficiency of viral replication, viral transmission, the access and binding of the virus to target tissue, or the escape of the virus from host defenses and immune resolution (see Chapter 10). These activities may not be essential for viral growth in tissue culture but are necessary for the pathogenicity or survival of the virus in the host. Loss of these virulence factors results in attenuation of the virus. Many live-virus vaccines are attenuated virus strains.
The discussion in this chapter focuses on viral disease at the cellular level (cytopathogenesis), the host level (mechanisms of disease), and the population level (epidemiology and control). The antiviral immune response is discussed here and in Chapter 10.
Basic Steps in Viral Disease
Viral disease in the body progresses through defined steps, just like viral replication in the cell (Figure 45-1A). These steps are noted in Box 45-2.
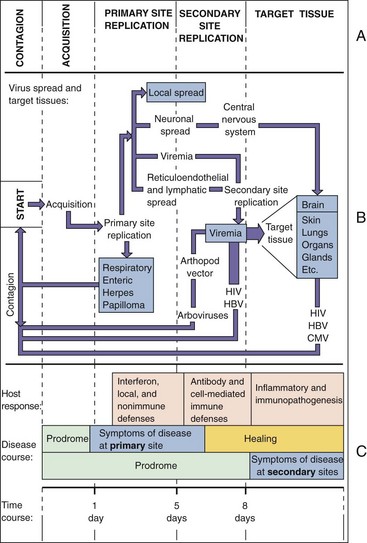
Figure 45-1 A, The stages of viral infection. The virus is released from one person, is acquired by another, replicates, and initiates a primary infection at the site of acquisition. Depending on the virus, it may then spread to other body sites and finally to a target tissue characteristic of the disease. B, The cycle starts with acquisition, as indicated, and proceeds until the release of new virus. The thickness of the arrow denotes the degree to which the original virus inoculum is amplified on replication. The boxes indicate a site or cause of symptoms. C, Time course of viral infection. The time course of symptoms and the immune response correlate with the stage of viral infection and depend on whether the virus causes symptoms at the primary site or only after dissemination to another (secondary) site. CMV, Cytomegalovirus; HBV, hepatitis B virus; HIV, human immunodeficiency virus.
Box 45-2
Progression of Viral Disease
1. Acquisition (entry into the body)
2. Initiation of infection at a primary site
3. Activation of innate protections
4. An incubation period, when the virus is amplified and may spread to a secondary site
5. Replication in the target tissue, which causes the characteristic disease signs
6. Host responses that limit and contribute (immunopathogenesis) to the disease
7. Virus production in a tissue that releases the virus to other people for contagion
The incubation period may proceed without symptoms (asymptomatic) or may produce nonspecific early symptoms, such as fever, head or body ache, or chills, termed the prodrome. Often, the virus infection is resolved by innate host protections without symptoms. The symptoms of the disease are caused by tissue damage and systemic effects caused by the virus and the immune system. These symptoms may continue through convalescence while the body repairs the damage. The individual usually develops a memory immune response for future protection against a similar challenge with this virus.
Infection of the Target Tissue
The virus gains entry into the body through breaks in the skin (cuts, bites, injections) or across the mucoepithelial membranes that line the orifices of the body (eyes, respiratory tract, mouth, genitalia, and gastrointestinal tract). The skin is an excellent barrier to infection. Tears, mucus, ciliated epithelium, stomach acid, bile, and immunoglobulin A protect the orifices. Inhalation is probably the most common route of viral infection.
On entry into the body, the virus replicates in cells that express viral receptors and have the appropriate biosynthetic machinery. Many viruses initiate infection in the oral mucosa or upper respiratory tract. Disease signs may accompany viral replication at the primary site. The virus may replicate and remain at the primary site, may disseminate to other tissues via the bloodstream or within mononuclear phagocytes and lymphocytes, or may disseminate through neurons (Figure 45-1B).
The bloodstream and the lymphatic system are the predominant means of viral transfer in the body. The virus may gain access to them after tissue damage, upon uptake by macrophages, or on transport past the mucoepithelial cells of the oropharynx, gastrointestinal tract, vagina, or anus. Several enteric viruses (picornaviruses and reoviruses) bind to receptors on M cells, which translocate the virus to the underlying Peyer patches of the lymphatic system.
The transport of virus in the blood is termed viremia. The virus may either be free in the plasma or be cell associated in lymphocytes or macrophages. Viruses taken up by phagocytic macrophages may be inactivated, may replicate, or may be delivered to other tissues. Replication of a virus in macrophages, the endothelial lining of blood vessels, or the liver can cause the infection to be amplified and initiate the development of a secondary viremia. In many cases, a secondary viremia precedes delivery of the virus to the target tissue (e.g., liver, brain, skin) and the manifestation of characteristic symptoms.
Viruses can gain access to the central nervous system or brain (1) from the bloodstream (e.g., arboencephalitis viruses), (2) from infected meninges or cerebrospinal fluid, (3) by means of the migration of infected macrophages, or (4) the infection of peripheral and sensory (olfactory) neurons. The meninges are accessible to many of the viruses spread by viremia, which may also provide access to neurons. Herpes simplex, varicella-zoster, and rabies viruses initially infect mucoepithelium, skin, or muscle, and then the peripheral innervating neuron, which transports the virus to the central nervous system or brain.
Viral Pathogenesis
Cytopathogenesis
The four potential outcomes of a viral infection of a cell are as follows (Box 45-3 and Table 45-1):
1. Failed infection (abortive infection)
2. Cell death (lytic infection)
3. Replication without cell death (persistent infection)
4. Presence of virus without virus production but with potential for reactivation (latent-recurrent infection)
Box 45-3
Determinants of Viral Pathogenesis
Interaction of Virus with Target Tissue
Cytopathologic Activity of the Virus
Table 45-1 Types of Viral Infections at the Cellular Level
| Type | Virus Production | Fate of Cell |
|---|---|---|
| Abortive | − | No effect |
| Cytolytic | + | Death |
| Persistent | ||
| Productive | + | Senescence |
| Latent | − | No effect |
| Transforming | ||
| DNA viruses | − | Immortalization |
| RNA viruses | + | Immortalization |
Viral mutants, which cause abortive infections, do not multiply and therefore disappear. Persistent infections may be (1) chronic (nonlytic, productive), (2) latent (limited viral macromolecular but no virus synthesis), (3) recurrent (periods of latency then virus production), or (4) transforming (immortalizing).
The nature of the infection is determined by the characteristics of the virus and the target cell. A nonpermissive cell may lack a receptor, important enzyme pathway, transcriptional activator, or express an antiviral mechanism that will not allow replication of a particular type or strain of virus. For example, neurons and nongrowing cells lack the machinery and substrates for replication of a DNA virus. These cells can also limit the amount of protein synthesis within the cells by phosphorylating elongation initiation factor-2α (eIF-2α) to prevent the assembly of ribosomes on 5′ capped mRNA, which shuts down most protein synthesis. This protection can be triggered by the large amount of protein synthesis required for virus production or the activation of the interferon-α (IFN-α) or interferon-β (IFN-β) induced antiviral state. Herpesviruses and some other viruses prevent this by inhibiting the phosphorylating enzyme (protein kinase R) or by activating a cellular protein phosphatase to remove the phosphate on eIF-2α. Another example is APOBEC3, an enzyme that causes hypermutation inactivation of the cDNA of retroviruses. The viral infectivity factor (Vif) protein of human immunodeficiency virus (HIV) overcomes this block by promoting the degradation of APOBEC3.
A permissive cell provides the biosynthetic machinery to support the complete replicative cycle of the virus. Replication of the virus in a semipermissive cell may be very inefficient, or the cell may support some but not all the steps in viral replication.
Replication of the virus can initiate changes in cells that lead to cytolysis or to alterations in the cell’s appearance, functional properties, or antigenicity. The effects on the cell may result from viral takeover of macromolecular synthesis, the accumulation of viral proteins or particles, modification or disruption of cellular structures, or manipulation of cellular functions (Table 45-2).
Table 45-2 Mechanisms of Viral Cytopathogenesis
| Mechanism | Examples |
|---|---|
| Inhibition of cellular protein synthesis | Polioviruses, herpes simplex virus, togaviruses, poxviruses |
| Inhibition and degradation of cellular DNA | Herpesviruses |
| Alteration of cell membrane structure | Enveloped viruses |
| Glycoprotein insertion | All enveloped viruses |
| Syncytia formation | Herpes simplex virus, varicella-zoster virus, paramyxoviruses, human immunodeficiency virus |
| Disruption of cytoskeleton | Nonenveloped viruses (accumulation), herpes simplex virus |
| Permeability | Togaviruses, herpesviruses |
| Toxicity of virion components | Adenovirus fibers, reovirus NSP4 protein |
| Inclusion Bodies | Examples |
|---|---|
| Negri bodies (intracytoplasmic) | Rabies |
| Intronuclear basophilic (Owl’s eye) | Cytomegalovirus (enlarged cells), adenoviruses |
| Cowdry type A (intranuclear) | Herpes simplex virus, subacute sclerosing panencephalitis (measles) virus |
| Intracytoplasmic acidophilic | Poxviruses |
| Perinuclear cytoplasmic acidophilic | Reoviruses |
Lytic Infections
Lytic infection results when virus replication kills the target cell. Some viruses damage the cell and prevent repair by inhibiting the synthesis of cellular macromolecules or by producing degradative enzymes and toxic proteins. For example, HSV and other viruses produce proteins that inhibit the synthesis of cellular DNA and mRNA and synthesize other proteins that degrade host DNA to provide substrates for viral genome replication. Cellular protein synthesis may be actively blocked (e.g., poliovirus inhibits translation of 5′-capped cellular mRNA) or passively blocked (e.g., through the production of much viral mRNA that successfully competes for ribosomes) (see Chapter 44).
Replication of the virus and the accumulation of viral components and progeny within the cell can disrupt the structure and function of the cell or disrupt lysosomes, causing cell death. The expression of viral antigens on the cell surface and disruption of the cytoskeleton can change cell-to-cell interactions and the cell’s appearance, making the cell a target for immune cytolysis.
Virus infection or cytolytic immune responses may induce apoptosis in the infected cell. Apoptosis is a preset cascade of events that, when triggered, leads to cellular suicide. This process may facilitate release of the virus from the cell, but it also limits the amount of virus that is produced by destroying the viral “factory.” As a result, many viruses (e.g., herpesviruses, adenoviruses, hepatitis C virus) encode methods for inhibiting apoptosis.
Cell surface expression of the glycoproteins of some paramyxoviruses, herpesviruses, and retroviruses triggers the fusion of neighboring cells into multinucleated giant cells called syncytia. Cell-to-cell fusion may occur in the absence of new protein synthesis (fusion from without), as occurs in infections with paramyxoviruses, or it may require new protein synthesis (fusion from within), as occurs in infection with HSV. Syncytia formation allows the virus infection to spread from cell to cell and escape antibody detection. Syncytia may be fragile and susceptible to lysis. The syncytia that occurs in infection with HIV also causes death of the cells.
Some viral infections cause characteristic changes in the appearance and properties of the target cells. For example, chromosomal aberrations and degradation may occur and can be detected with histologic staining (e.g., marginated chromatin ringing the nuclear membrane in HSV-infected and adenovirus-infected cells). In addition, new stainable structures called inclusion bodies may appear within the nucleus or cytoplasm. These structures may result from virus-induced changes in the membrane or chromosomal structure or may represent the sites of viral replication or accumulations of viral capsids. Because the nature and location of these inclusion bodies are characteristic of particular viral infections, the presence of such bodies facilitates laboratory diagnosis (see Table 45-2). Viral infection may also cause vacuolization, rounding of the cells, and other nonspecific histologic changes that are characteristics of sick cells.
Nonlytic Infections
A persistent infection occurs in an infected cell that is not killed by the virus. Some viruses cause a persistent productive infection because the virus is released gently from the cell through exocytosis or through budding (many enveloped viruses) from the plasma membrane.
A latent infection may result from DNA virus infection of a cell that restricts or lacks the machinery for transcribing all the viral genes. The specific transcription factors required by such a virus may be expressed only in specific tissues, in growing but not resting cells, or after hormone or cytokine induction. For example, HSV establishes a latent infection in neurons that don’t express the nuclear factors required to transcribe the immediate early viral genes, but stress and other stimuli can activate the cells to allow viral replication.
Oncogenic Viruses
Some DNA viruses and retroviruses establish persistent infections that can also stimulate uncontrolled cell growth, causing the transformation or immortalization of the cell (Figure 45-2). Characteristics of transformed cells include continued growth without senescence, alterations in cell morphology and metabolism, increased cell growth rate and sugar transport, loss of cell-contact inhibition of growth, and ability to grow in a suspension or pileup into foci when grown in a semisolid agar.
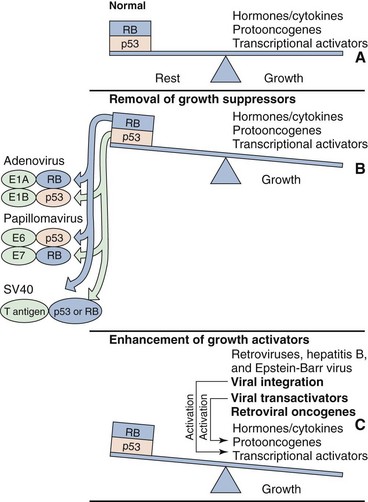
Figure 45-2 Mechanisms of viral transformation and immortalization. Cell growth is controlled (A) by the maintenance of a balance in the external and internal growth activators (accelerators) and by growth suppressors, such as p53 and the retinoblastoma (RB) gene product (brakes). Oncogenic viruses alter the balance by removing the brakes (B) or by enhancing the effects of the accelerators (C).
Different oncogenic viruses have different mechanisms for immortalizing cells. Viruses immortalize cells by (1) activating or providing growth-stimulating genes, (2) removing the inherent braking mechanisms that limit DNA synthesis and cell growth, or (3) preventing apoptosis. Immortalization by DNA viruses occurs in semipermissive cells, which express only select viral genes but do not produce virus. The synthesis of viral DNA, late mRNA, late proteins, or virus leads to cell death, which precludes immortalization. Several oncogenic DNA viruses integrate into the host cell chromosome. Papillomavirus, SV40 virus, and adenovirus encode proteins that bind and inactivate cell growth–regulatory proteins, such as p53 and the retinoblastoma gene product, thus releasing the brakes on cell growth. Loss of p53 also makes the cell more susceptible to mutation. Epstein-Barr virus immortalizes B cells by stimulating cell growth (as a B-cell mitogen) and by preventing programmed cell death (apoptosis).
Retroviruses (RNA viruses) use two approaches to oncogenesis. Some oncoviruses encode oncogene proteins (e.g., SIS, RAS, SRC, MOS, MYC, JUN, FOS), which are almost identical to the cellular proteins involved in cellular growth control (e.g., components of a growth-factor signal cascade [receptors, G proteins, protein kinases], or growth-regulating transcription factors). The overproduction or altered function of these oncogene products stimulates cell growth. These oncogenic viruses rapidly cause tumors to form. However, no human retrovirus of this type has been identified.
Human T-cell lymphotropic virus 1, the only human oncogenic retrovirus identified, uses more subtle mechanisms of leukemogenesis. It encodes a protein (TAX) that transactivates gene expression, including genes for growth-stimulating cytokines (e.g., interleukin-2 [IL-2]). This constitutes the second approach to oncogenesis. The integration of the DNA copy of HTLV-1 near a cellular growth-stimulating gene can also cause the gene to be activated by the strong viral enhancer and promoter sequences encoded at each end of the viral genome (long terminal repeat [LTR] sequences). HTLV-1–associated leukemias develop slowly, occurring 20 to 30 years after infection. Retroviruses continue to produce virus in immortalized or transformed cells.
Some viruses may initiate tumor formation indirectly. Hepatitis B virus (HBV) and hepatitis C virus (HCV) may have mechanisms for direct oncogenesis; however, both viruses establish persistent infections that cause inflammation and require significant tissue repair. Inflammation and continuous stimulation of liver cell growth and repair may promote mutations that lead to tumor formation. Human herpesvirus 8 (HHV-8) promotes the development of Kaposi sarcoma by means of growth-promoting cytokines encoded by the virus; this disease occurs most often in immunosuppressed patients, such as those with acquired immunodeficiency syndrome (AIDS).
Viral transformation is the first step but is generally not sufficient to cause oncogenesis and tumor formation. Instead, over time, immortalized cells are more likely than normal cells to accumulate other mutations or chromosomal rearrangements that promote development of tumor cells. Immortalized cells may also be more susceptible to cofactors and tumor promoters (e.g., phorbol esters, butyrate) that enhance tumor formation. Approximately 15% of human cancers can be related to oncogenic viruses such as HTLV-1, HBV, HCV, papillomaviruses 16 and 18, HHV-8, and Epstein-Barr virus. HSV-2 may be a cofactor for human cervical cancer.
Host Defenses against Viral Infection
The ultimate goals of the host antiviral innate and immune responses are to prevent entry, prevent spread, and to eliminate the virus and the cells harboring or replicating the virus (resolution). The immune response is the best and in most cases the only means of controlling a viral infection. Innate, humoral and cellular immune responses are important for antiviral immunity. The longer the virus replicates in the body, the greater the dissemination of the infection, the more rigorous the immune response necessary to control the infection, and the potential for immunopathogenesis. Interferon and cytotoxic T-cell responses may have evolved primarily as antiviral defense mechanisms. A detailed description of the antiviral immune response is presented in Chapter 10.
The skin is the best barrier to infection. The orifices of the body (e.g., mouth, eyes, nose, ears, and anus) are protected by mucous, ciliated epithelium, tears, and the gastric acid and bile of the gastrointestinal tract. After the virus penetrates these natural barriers, it activates the antigen-nonspecific (innate) host defenses (e.g., fever, interferon, macrophages, dendritic cells, natural killer [NK] cells), which attempt to limit and control local viral replication and spread. Viral molecules, including double-stranded RNA (which is the replicative intermediate of RNA viruses), certain forms of DNA and single-stranded RNA, and some viral glycoproteins, activate type I interferon production and innate cellular responses through interaction with cytoplasmic receptors or the Toll-like receptors (TLRs) in endosomes and on cell surfaces. Innate responses prevent most viral infections from causing disease.
Antigen-specific immune responses take several days to be activated and become effective. The goal of these protective responses is to resolve the infection by eliminating all infectious virus and virus-infected cells from the body. Antibody is effective against extracellular virus and may be sufficient to control cytolytic viruses because viral replication will eliminate the virion factory within the infected cell. Antibody is essential to control virus spread to target tissues by viremia. Cell-mediated immunity is required for lysis of cells infected with a noncytolytic virus (e.g., hepatitis A virus) and infections caused by enveloped viruses.
Prior immunity delivers antigen specific immunity much sooner and more effectively than during a primary infection. It may not prevent the initial stages of infection but, in most cases, does prevent disease progression. On rechallenge, cell-mediated responses are more effective at limiting the local spread of virus, and serum antibody can prevent viremic spread of the virus. Memory immune responses can be generated by prior infection or vaccination.
Many viruses, especially the larger viruses, have the means to escape one or more aspects of immune control (see Chapter 10, Table 10-4). These mechanisms include preventing interferon action, changing virus antigens, spreading by cell-to-cell transmission to escape antibody, and suppressing antigen presentation and lymphocyte function. By preventing the consequences of the antiviral state induced by IFN-α and IFN-β, herpes simplex viral protein synthesis and replication can continue. Inhibition of major histocompatibility complex (MHC) I expression by cytomegalovirus and adenoviruses prevents T-cell killing of the infected cell. Antigenic variation over the course of several years (antigenic shift and drift) by influenza or during the lifetime of the infected individual by HIV limits the antiviral efficacy of antibody. Failure to resolve the infection may lead to persistent infection, chronic disease, or death of the patient.
Immunopathology
The hypersensitivity and inflammatory reactions initiated by antiviral immunity can be the major cause of the pathologic manifestations and symptoms of viral disease (Table 45-3). Early responses to the virus and viral infection, such as interferon and cytokines, can initiate local inflammatory and systemic responses. For example, interferon and cytokines stimulate the flulike systemic symptoms (e.g., fever, malaise, headache) that are usually associated with respiratory viral infections and viremias (e.g., arboencephalitis viruses). These symptoms often precede (prodrome) the characteristic symptoms of the viral infection during the viremic stage. Some virus infections induce a large cytokine response, a cytokine storm, and this can dysregulate immune responses and may trigger autoimmune diseases in genetically predisposed individuals. Later, immune complexes and complement activation (classic pathway), CD4 T-cell–induced delayed-type hypersensitivity, and CD8 cytolytic T-cell action may induce tissue damage. These actions often promote neutrophil infiltration and more cell damage.
Table 45-3 Viral Immunopathogenesis
| Immunopathogenesis | Immune Mediators | Examples |
|---|---|---|
| Flulike symptoms | Interferon, cytokines | Respiratory viruses, arboviruses (viremia-inducing viruses) |
| Delayed-type hypersensitivity and inflammation | T cells, macrophages, and polymorphonuclear leukocytes | Enveloped viruses |
| Immune complex disease | Antibody, complement | Hepatitis B virus, rubella |
| Hemorrhagic disease | T cell, antibody, complement | Yellow fever, dengue, Lassa fever, Ebola viruses |
| Postinfection cytolysis | T cells | Enveloped viruses (e.g., postmeasles encephalitis) |
| Cytokine storm | – | Dendritic cells, T cells enveloped and other viruses |
| Immunosuppression | – | Human immunodeficiency virus, cytomegalovirus, measles virus, influenza virus |
The inflammatory response initiated by cell-mediated immunity is difficult to control and damages tissue. Infections by enveloped viruses, in particular, induce cell-mediated immune responses that usually produce more extensive immunopathologic conditions. For example, the classic symptoms of measles and mumps result from the T-cell–induced inflammatory and hypersensitivity responses rather than from cytopathologic effects of the virus. The presence of large amounts of antigen and antibody in blood during viremias or chronic infections (e.g., HBV infection) can initiate the classic type III immune complex hypersensitivity reactions. These immune complexes can activate the complement system, triggering inflammatory responses and tissue destruction. These immune complexes often accumulate in the kidney and cause glomerulonephritis.
In the case of dengue and measles viruses, partial immunity to a related or inactivated virus can result in a more severe host response and disease on subsequent challenge with a related or virulent virus. This is because antigen-specific T-cell and antibody responses are enhanced and induce significant inflammatory and hypersensitivity damage to infected endothelial cells (dengue hemorrhagic fever) or skin and the lung (atypical measles). In addition, a non-neutralizing antibody can facilitate the uptake of dengue and yellow fever viruses into macrophages through Fc receptors, where they can replicate.
Children generally have a less active cell-mediated immune response (e.g., NK or natural killer T [NKT] cells) than adults and therefore usually have milder symptoms during infections by some viruses (e.g., measles, mumps, Epstein-Barr, and varicella-zoster viruses). However, in the case of HBV, mild or no symptoms correlate with an inability to resolve the infection, resulting in chronic disease.
Viral Disease
The relative susceptibility of a person and the severity of the disease depend on the following factors:
1. The mechanism of exposure and site of infection
Once the host is infected, however, the host’s immune status and competence are probably the major factors that determine whether a viral infection causes a life-threatening disease, a benign lesion, or no symptoms at all.
The stages of viral disease are shown in Figure 45-1C. During the incubation period, the virus is replicating but has not reached the target tissue or induced sufficient damage to cause the disease. The incubation period is relatively short if the primary site of infection is the target tissue and produces the characteristic symptoms of the disease. Longer incubation periods occur when the virus must spread to other sites and be amplified before reaching the target tissue, or the symptoms are caused by immunopathology. Nonspecific or flulike symptoms may precede the characteristic symptoms during the prodrome. The incubation periods for many common viral infections are listed in Table 45-4. Specific viral diseases are discussed in subsequent chapters and reviewed in Chapter 46.
Table 45-4 Incubation Periods of Common Viral Infections
| Disease | Incubation Period (Days)* |
|---|---|
| Influenza | 1-2 |
| Common cold | 1-3 |
| Herpes simplex | 2-8 |
| Bronchiolitis, croup | 3-5 |
| Acute respiratory disease (adenoviruses) | 5-7 |
| Dengue | 5-8 |
| Enteroviruses | 6-12 |
| Poliomyelitis | 5-20 |
| Measles | 9-12 |
| Smallpox | 12-14 |
| Chickenpox | 13-17 |
| Mumps | 16-20 |
| Rubella | 17-20 |
| Mononucleosis | 30-50 |
| Hepatitis A | 15-40 |
| Hepatitis B | 50-150 |
| Rabies | 30-100+ |
| Papilloma (warts) | 50-150 |
| Human immunodeficiency virus | 1-15 years |
| AIDS | 1-10 years |
* Until first appearance of prodromal symptoms. Diagnostic signs (e.g., rash, paralysis) may not appear until 2 to 4 days later.
Modified from White DO, Fenner F: Medical Virology, ed 3, New York, 1986, Academic.
The nature and severity of the symptoms of a viral disease are related to the function of the infected target tissue (e.g., liver—hepatitis, brain—encephalitis) and the extent of the immunopathologic responses triggered by the infection. Inapparent infections result if (1) the infected tissue is undamaged, (2) the infection is controlled before the virus reaches its target tissue, (3) the target tissue is expendable, (4) the damaged tissue is rapidly repaired, or (5) the extent of damage is below a functional threshold for that particular tissue. For example, many infections of the brain are inapparent or are below the threshold of severe loss of function, but encephalitis results if the loss of function becomes significant. Despite the lack of symptoms, virus-specific antibody will be produced. For example, although 97% of adults have antibody (seropositive) to varicella-zoster virus, less than half remember having had chickenpox. Inapparent or asymptomatic infections are major sources of contagion.
Viral infections may cause acute or chronic disease (persistent infection). The ability and speed with which a person’s immune system controls and resolves a viral infection usually determine whether acute or chronic disease ensues, as well as the severity of the symptoms (Figure 45-3). The acute episode of a persistent infection may be asymptomatic (JC polyomavirus) or may later in life cause symptoms similar to (varicella and zoster) or different from (HIV) those of the acute disease. Slow viruses and prions have long incubation periods, during which sufficient virus or tissue destruction accumulates before a rapid progression of symptoms.
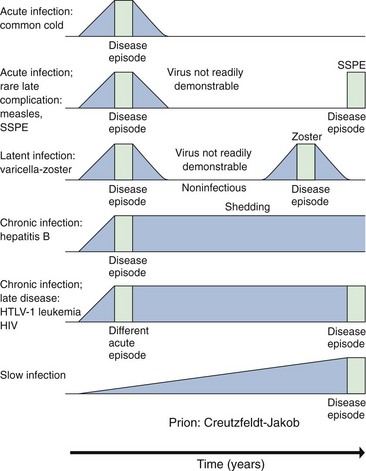
Figure 45-3 Acute infection and various types of persistent infection, as illustrated by the diseases indicated in the column at the left. Blue represents presence of virus; green indicates episode of disease. HIV, Human immunodeficiency virus; HTLV-1, human T-cell lymphotropic virus 1; SSPE, subacute sclerosing panencephalitis.
(Modified from White DO, Fenner FJ: Medical virology, ed 3, New York, 1986, Academic, 1986.)
Epidemiology
Epidemiology studies the spread of disease through a population. Infection of a population is similar to infection of a person in that the virus must spread through the population and is controlled by immunization of the population (Box 45-4). To endure, viruses must continue to infect new, immunologically naïve, susceptible hosts.
Box 45-4
Viral Epidemiology*
Mechanisms of Viral Transmission†
Disease and Viral Factors That Promote Transmission
* Infection of a population instead of a person.
† See also Table 45-5.
Exposure
People are exposed to viruses throughout their lives. However, some situations, vocations, lifestyles, and living arrangements increase the likelihood that a person will come in contact with certain viruses. In contrast, many viruses are ubiquitous. Exposure to HSV-1, HHV-6, varicella-zoster virus, parvovirus B19, Epstein-Barr virus, and many respiratory and enteric viruses can be detected in most young children or by early adulthood by the presence of antibodies to the virus.
Poor hygiene and crowded living, school, and job conditions promote exposure to respiratory and enteric viruses. Day-care centers are consistent sources of viral infections, especially viruses spread by the respiratory and fecal-oral routes. Travel, summer camp, and vocations that bring people in contact with a virus vector (e.g., mosquitoes) put them at particular risk for infection by arboviruses and other zoonoses. Sexual promiscuity also promotes the spread and acquisition of several viruses. Health care workers, such as physicians, dentists, nurses, and technicians, are frequently exposed to respiratory and other viruses but are uniquely at risk for acquiring viruses from contaminated blood (HBV, HIV) or vesicle fluid (HSV).
Transmission of Viruses
Viruses are transmitted by direct contact (including sexual contact), injection with contaminated fluids or blood, the transplantation of organs, and the respiratory and fecal-oral routes (Table 45-5). The route of transmission depends on the source of the virus (the tissue site of viral replication and secretion) and the ability of the virus to endure the hazards and barriers of the environment and the body en route to the target tissue. For example, viruses that replicate in the respiratory tract (e.g., influenza A virus) are released in aerosol droplets, whereas enteric viruses (e.g., picornaviruses and reoviruses) are passed by the fecal-oral route. Cytomegalovirus is transmitted in most bodily secretions because it infects mucoepithelial, secretory, and other cells found in the skin, secretory glands, lungs, liver, and other organs.
| Mode | Examples |
|---|---|
| Respiratory transmission | Paramyxoviruses, influenza viruses, picornaviruses, rhinoviruses, varicella-zoster virus, B19 virus |
| Fecal-oral transmission | Picornaviruses, rotavirus, reovirus, noroviruses, adenovirus |
| Contact (lesions, fomites) | Herpes simplex virus, rhinoviruses, poxviruses, adenovirus |
| Zoonoses (animals, insects) | Togaviruses (alpha), flaviviruses, bunyaviruses, orbiviruses, arenaviruses, hantaviruses, rabies virus, influenza A virus, orf (pox) |
| Transmission via blood | Human immunodeficiency virus, HTLV-1, hepatitis B virus, hepatitis C virus, hepatitis delta virus, cytomegalovirus |
| Sexual contact | Blood-borne viruses, herpes simplex virus, human papillomavirus, molluscum contagiosum |
| Maternal-neonatal transmission | Rubella virus, cytomegalovirus, B19 virus, echovirus, herpes simplex virus, varicella-zoster virus, HIV |
| Genetic | Prions, retroviruses |
HTLV-1, Human T-cell lymphotropic virus 1.
The presence or absence of an envelope is the major structural determinant of the mode of viral transmission. Nonenveloped viruses (naked capsid viruses) can withstand drying, the effects of detergents, and extremes of pH and temperature, whereas enveloped viruses generally cannot (see Chapter 44, Box 44-4). Specifically, most nonenveloped viruses can withstand the acidic environment of the stomach and the detergent-like bile of the intestines and mild disinfection and insufficient sewage treatment. These viruses are generally transmitted by the respiratory and fecal-oral routes and can often be acquired from contaminated objects, termed fomites. For example, hepatitis A virus, a picornavirus, is a nonenveloped virus that is transmitted by the fecal-oral route and acquired from contaminated water, shellfish, and food. Adenoviruses and many other nonenveloped viruses can be spread by contact with fomites, such as handkerchiefs and toys.
Unlike the sturdy nonenveloped viruses, most enveloped viruses are comparatively fragile (see Chapter 44, Box 44-5). They require an intact envelope for infectivity. These viruses must remain wet and are spread (1) in respiratory droplets, blood, mucus, saliva, and semen; (2) by injection; or (3) in organ transplants. Most enveloped viruses are also labile to treatment with acid and detergents, a feature that precludes their being transmitted by the fecal-oral route. Exceptions are HBV and coronaviruses.
Animals can also act as vectors that spread viral disease to other animals and humans and even to other locales. They can also be reservoirs for the virus, which maintain and amplify the virus in the environment. Viral diseases that are shared by animals or insects and humans are called zoonoses. For example, raccoons, foxes, bats, dogs, and cats are reservoirs and vectors for the rabies virus. Arthropods, including mosquitoes, ticks, and sandflies, can act as vectors for togaviruses, flaviviruses, bunyaviruses, or reoviruses. These viruses are often referred to as arboviruses because they are arthropod borne. A more detailed discussion of arboviruses is presented in Chapter 60. Most arboviruses have a very broad host range, capable of replicating in specific insects, birds, amphibians, and mammals, in addition to humans. Also, the arboviruses must establish a viremia in the animal reservoir so that the insect can acquire the virus during its blood meal.
Other factors that can promote the transmission of viruses are the potential for asymptomatic infection, crowded living conditions, certain occupations, certain lifestyles, day care centers, and travel. Virus transmission during an asymptomatic infection (e.g., HIV, varicella-zoster virus) occurs unknowingly and is difficult to restrict. This is an important characteristic of sexually transmitted diseases. Viruses that cause persistent productive infections (e.g., cytomegalovirus, HIV) are a particular problem because the infected person is a continual source of virus that can be spread to immunologically naïve people. Viruses with many different serotypes (rhinoviruses), or viruses capable of changing their antigenicity (influenza and HIV), also readily find immunologically naïve populations.
Maintenance of a Virus in the Population
The persistence of a virus in a community depends on the availability of a critical number of immunologically naïve (seronegative), susceptible people. The efficiency of virus transmission determines the size of the susceptible population necessary for maintenance of the virus in the population. Immunization, produced by natural means or by vaccination, is the best way of reducing the number of such susceptible people.
Age
A person’s age is an important factor in determining his or her susceptibility to viral infections. Infants, children, adults, and elderly persons are susceptible to different viruses and have different symptomatic responses to the infection. These differences may result from variations in body size, recuperative abilities, and most important, immune status in people in these age groups. Differences in lifestyles, habits, school environments, and job settings at different ages also determine when people are exposed to viruses.
Infants and children acquire a series of respiratory and exanthematous viral diseases at first exposure because they are immunologically naïve. Infants are especially prone to more serious presentations of paramyxovirus respiratory infections and viral gastroenteritis because of their small size and physiologic requirements (e.g., nutrients, water, electrolytes). However, children generally do not mount as severe an immunopathologic response as adults, and some diseases (herpesviruses) are more benign in children.
Elderly persons are especially susceptible to new viral infections and the reactivation of latent viruses. Because they are less able to initiate a new immune response, repair damaged tissue, and recover, elderly persons are therefore more susceptible to complications after infection and outbreaks of the new strains of the influenza A and B viruses. Elderly persons are also more prone to zoster (shingles), a recurrence of varicella-zoster virus, as a result of a decline in this specific immune response with age.
Immune Status
The competence of a person’s immune response and immune history determine how quickly and efficiently the infection is resolved and can also determine the severity of the symptoms. The rechallenge of a person with prior immunity usually results in asymptomatic or mild disease without transmission. People who are in an immunosuppressed state as a result of AIDS, cancer, or immunosuppressive therapy are at greater risk of suffering more serious disease on primary infection (measles, vaccinia) and are more prone to suffer recurrences of infections with latent viruses (e.g., herpesviruses, papovaviruses).
Other Host Factors
General health plays an important role in determining the competence and nature of the immune response and ability to repair diseased tissue. Poor nutrition can compromise a person’s immune system and decrease his or her tissue regenerative capacity. Immunosuppressive diseases and therapies may allow viral replication or recurrence to proceed unchecked. Genetic makeup also plays an important role in determining the response of the immune system to viral infection. Specifically, genetic differences in immune response genes, genes for viral receptors, and other genetic loci affect susceptibility to a viral infection and the severity of disease.
Geographic and Seasonal Considerations
The geographic distribution of a virus is usually determined by whether the requisite cofactors or vectors are present or whether there is an immunologically naïve, susceptible population. For example, many of the arboviruses are limited to the ecologic niche of their arthropod vectors. Extensive global transportation is eliminating many of the geographically determined restrictions to virus distribution.
Seasonal differences in the occurrence of viral disease correspond with behaviors that promote the spread of the virus. For example, respiratory viruses are more prevalent in the winter, because crowding facilitates the spread of such viruses, and the temperature and humid conditions stabilize them. Enteric viruses, on the other hand, are more prevalent during the summer, possibly because hygiene is more lax during this season. The seasonal differences in arboviral diseases reflect the life cycle of the arthropod vector or its reservoir (e.g., birds).
Outbreaks, Epidemics, and Pandemics
Outbreaks of a viral infection often result from the introduction of a virus (e.g., hepatitis A) into a new location. The outbreak originates from a common source (e.g., food preparation) and often can be stopped once the source is identified. Epidemics occur over a larger geographic area and generally result from the introduction of a new strain of virus into an immunologically naïve population. Pandemics are worldwide epidemics, usually resulting from the introduction of a new virus (e.g., HIV). Pandemics of influenza A used to occur approximately every 10 years as the result of the introduction of new strains of the virus.
Control of Viral Spread
The spread of a virus can be controlled by quarantine, good hygiene, changes in lifestyle, elimination of the vector, or immunization of the population. Quarantine was once the only means of limiting epidemics of viral infections and is most effective for limiting the spread of viruses that always cause symptomatic disease (e.g., smallpox). It is now used in hospitals to limit the nosocomial spread of viruses, especially to high-risk patients (e.g., immunosuppressed people). The proper sanitation of contaminated items and disinfection of the water supply are means of limiting the spread of enteric viruses. Education and resultant changes in lifestyle have made a difference in the spread of sexually transmitted viruses, such as HIV, HBV, and HSV. Elimination of an arthropod or its ecologic niche (e.g., drainage of the swamps it inhabits) has proved effective for controlling arboviruses.
The best way to limit viral spread, however, is to immunize the population. Immunization, whether produced by natural infection or by vaccination, protects individuals and reduces the size of the immunologically naïve, susceptible population necessary to promote the spread and maintenance of the virus.
1. What are the routes by which viruses gain entry into the body? For each route, list the barriers to infection and a virus that infects by it.
2. Describe or draw the disease path of a virus that is transmitted by an aerosol and causes lesions on the skin (similar to varicella).
3. Identify the structures that elicit a protective antibody response to adenovirus, influenza A virus, poliovirus, and rabies virus.
4. Describe the major roles of each of the following in promoting resolution of a viral infection: interferon, macrophage, NK cells, CD4 T cells, CD8 T cells, and antibody.
5. Why are IFN-α and IFN-β produced before IFN-γ?
6. How does the nucleoprotein of influenza virus become an antigen for cytolytic CD8 T cells?
7. What events occur during the prodromal periods of a respiratory virus disease (e.g., parainfluenza virus) and encephalitis (e.g., St. Louis encephalitis virus)?
8. List the viral characteristics (structure, replication, target tissue) that would promote transmission by the fecal-oral route, by arthropods, by fomites, by mother’s milk, and by sexual activity.
9. What are the different mechanisms by which oncogenic viruses immortalize cells? Describe them.
| Route | Barriers | Examples of Viruses |
| Oral | Saliva, IgA, mucous | Polio |
| Respiratory | IgA, mucous | Influenza |
| Contact | Skin, mucous | HSV, human papillomavirus |
| Sexual | Female: IgA, IgG, mucous; male: skin | HSV, HIV, human papillomavirus |
| Injection, blood products, transplant | Antibody, T cells | Cytomegalovirus, HIV, HTLV, hepatitis B, C, D |
| Insect or animal bite (zoonose) | Antibody, interferon | Eastern equine encephalitis virus, yellow fever virus, rabies |
| Maternal-neonatal | Maternal antibody | Rubella, cytomegalovirus |
2. Varicella is inhaled; initiates replication in the lung; activates interferon and local inflammatory responses; initiates viremia and spreads to T cells and lymphatics, liver, and spleen; initiates viremia; spreads to skin; and causes lesions on skin.
3. Protective antibody is generated against the viral attachment protein or structure as follows:
Influenza A virus: hemagglutinin
Poliovirus: capsid structure forming a valley
4. IFN-α and IFN-β: promote the expression of proteins for the antiviral state (e.g., PKR, 2′5′A polymerase) that are activated by virus infection; activate NK cells
IFN-γ: activate macrophage to become a killer cell and producer of IL-12, an inducer of T helper 1 (TH1) responses
Macrophage: antigen-presenting cell and upon activation by IFN-γ; will promote inflammatory killing of internalized microbes
NK cells: MHC-independent killing of infected cells; antibody-dependent cellular cytotoxicity killing of infected cells
CD4 T cells: helper T cells that promote the antiviral response by producing cytokines. Promote apoptosis of infected cell through Fas-FasL (ligand) interaction
CD8 T cells: MHC I–restricted killing of infected cells
Antibody: neutralization of virus; opsonization of virus
5. IFN-α and IFN-β are produced by the infected cell and activate antiviral response in surrounding cells, activate NK cells, and also enhance immune response. IFN-γ is produced by NK or T cells as part of the cellular innate or immune responses.
6. The nucleoprotein of influenza virus is the predominant viral protein that is degraded by the proteosome of dendritic cells and converted into small peptides to pass through the transporters associated with antigen processing (TAP) into the endoplasmic reticulum to fill the antigenic peptide cleft of the MHC I molecule. Binding of antigen is required for the movement of the MHC I molecule to the cell surface to present antigen as part of a CD8 T-cell response.
7. During the prodrome of a respiratory virus infection, the virus replicates in the lung, IFN-α and -β are produced, which produce flulike symptoms and tiredness. The virus replicates and spreads to other sites in the lung. Tissue damage is repaired after the virus has been controlled by innate and immune responses.
| Method of Transmission | Viral Characteristics That Promote Transmission |
| Fecal-oral | Capsid structure that is impervious to acid and bile of the gastrointestinal tract, replication in oral, intestinal cells or released into the gastrointestinal tract (e.g., hepatitis A virus) |
| Arthropods | Establishment of sufficient viremia to allow arthropod to acquire virus during a blood meal |
| Fomites | Stability to drying and heat, as for a naked capsule |
| Mother’s milk | Secretion by epithelial cells into milk |
| Sexual activity | Long asymptomatic period of virus shedding to allow transmission before knowledge of infection |
9. Fast oncogenesis: incorporation of the oncogene from the virus into the host chromosome to stimulate cell growth (no human viruses act in this manner); example: Rous sarcoma virus of chickens
Slow oncogenesis: integration near a growth-promoting gene to allow the promoters in the long terminal repeat of the virus to induce overexpression of these genes and stimulate growth; example: HTLV-1
Transactivation of growth-promoting genes; example: IL-2 and IL-2 receptor by HTLV-1