The respiratory system
Respiratory examination
Anatomy
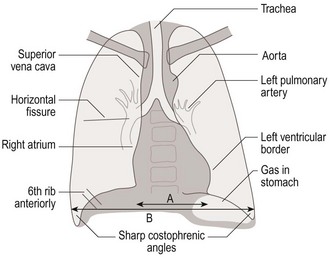
The respiratory system comprises the upper airway; the nose, mouth, oropharynx and larynx, and the lower airway; the trachea and lungs. The left lung only contains 45% of the total surface area available for gas exchange, because the heart lies principally within the left side of the chest. The right lung has three lobes (upper, middle and lower) and the left, two (upper and lower) (Fig. 7.1).
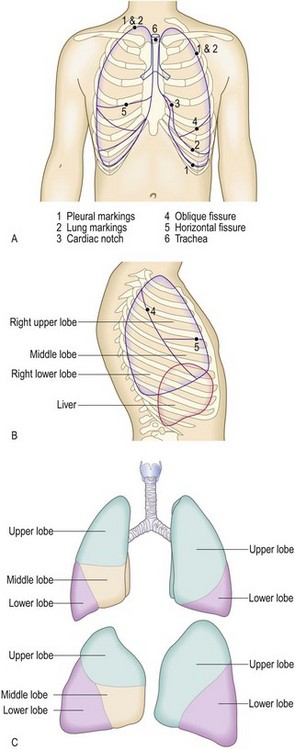
Fig. 7.1 Surface anatomy of the thorax.
(A) Surface markings of the lungs and pleura, trachea and bronchi. The trachea is normally central. The bifurcation of the trachea corresponds on the anterior chest wall with the sternal angle, the transverse bony ridge at the junction of the sternum and manubrium sternum. Count the ribs downwards from the second costal cartilage at the level of the sternal angle. (B) Surface markings of the right lung and underlying viscera. (C) Lobes of the lungs: anterior view (upper) and lateral view (lower).
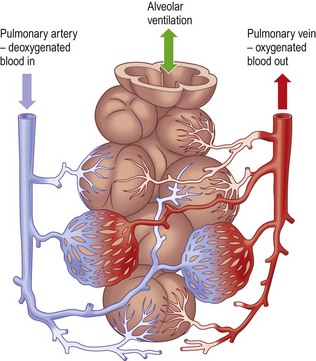
The airways (bronchi) transport air to the alveoli on inspiration and carry waste gases, e.g. carbon dioxide, away on expiration. The gas exchange unit of the lung is the acinus, with branching bronchioles leading to clusters of alveoli (Fig. 7.2). Alveoli are tiny air sacs lined by flattened epithelial cells (type I pneumocytes) and covered in capillaries where gas exchange occurs. The alveoli and capillaries have extremely thin walls and come into very close contact (the alveolar–capillary membrane); carbon dioxide and oxygen readily diffuse between them. There are approximately 300 million alveoli in each lung, with a total surface area for gas exchange of 40–80 m2.
The lung has two blood supplies: the bronchial arteries arise from the aorta and supply oxygenated blood to the bronchial walls. The pulmonary arteries circulate deoxygenated blood to the capillaries surrounding the alveoli.
Symptoms and definitions
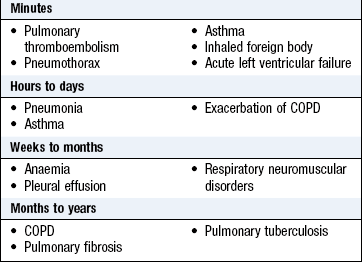
Cough is a characteristic sound caused by a forced expulsion against an initially closed glottis. Acute cough is one lasting less than 3 weeks; chronic cough lasts more than 8 weeks. The most common cause of acute cough is acute upper respiratory tract viral infection. Acute cough is usually self-limiting and benign, but may occur in more serious conditions (Box 7.1). Chronic cough in a non-smoker with a normal chest X-ray is usually caused by gastro-oesophageal reflux disease, chronic sinus disease with postnasal drip or angiotensin-converting enzyme inhibitors (Box 7.2).
Severe asthma or chronic obstructive pulmonary disease (COPD) causes prolonged wheezy cough and often a paroxysmal dry cough after a viral infection that lasts several months (bronchial hyperreactivity).
A feeble non-explosive ‘bovine’ cough with hoarseness suggests lung cancer invading the left recurrent laryngeal nerve causing left vocal cord paralysis, but may also occur with respiratory muscle weakness due to neuromuscular disorders.
Laryngeal inflammation, infection or tumour causes harsh, barking or painful coughs and associated hoarseness and the rasping or croaking inspiratory sound of stridor (p. 142). A moist cough suggests secretions in the upper and larger airways from bronchial infection and bronchiectasis. A persistent moist ‘smoker’s cough’ first thing in the morning is typical of chronic bronchitis. Smokers often consider this normal but any change in this cough may indicate lung cancer. Tracheitis and pneumonia cause dry, centrally painful and non-productive cough.
Chronic dry cough occurs in interstitial lung disease, e.g. idiopathic pulmonary fibrosis (formerly fibrosing alveolitis).
Timing and associated features: Nocturnal cough disrupting sleep is a feature of asthma. Occupational asthma and exposure to dusts and fumes cause a chronic cough which improves during weekends and holidays. Coughing during and after swallowing liquids suggests neuromuscular disease of the oropharynx.
Cough syncope may result from raised intrathoracic pressure impairing venous return to the heart, reducing cardiac output.
Dysphonia (hoarseness)
Dysphonia (hoarseness) is most commonly caused by laryngitis. Damage to the left recurrent laryngeal nerve by lung cancer at the left hilum causes hoarseness with a prolonged, low-pitched, ‘bovine’ cough as the left vocal cord cannot adduct to the midline (Ch. 13).
Wheeze
Wheeze is a high-pitched whistling sound produced by air passing through narrowed small airways. It occurs with expiration, but patients may call rattling sounds from secretions in the upper airways or larynx or the inspiratory sound of stridor wheeze. Wheeze on exercise is common in asthma and COPD. Night wakening with wheeze suggests asthma or paroxysmal nocturnal dyspnoea, but wheeze after wakening in the morning suggests COPD.
Stridor
Stridor is a high-pitched, often harsh noise produced by airflow turbulence through a partial obstruction of the upper airway. It occurs most commonly on inspiration but also on expiration or biphasically. Inspiratory stridor indicates narrowing at the vocal cords; biphasic stridor suggests tracheal obstruction, while stridor on expiration suggests tracheobronchial obstruction. Narrowing of smaller, peripheral airways produces wheeze (Fig. 13.22). Stridor always needs investigation. Common causes include infection/inflammation, e.g. acute epiglottitis in children and young adults, and tumours of the trachea and main bronchi or extrinsic compression by lymph nodes in older adults. Rarer causes include anaphylaxis and foreign body.
Stertor
Stertor, or muffled ‘hot potato’ speech, occurs with naso- or oropharyngeal blockage, e.g. quinsy.
Sputum
Sputum is mucus produced from the respiratory tract. The normal lung produces about 100 ml of clear sputum each day, which is transported to the oropharynx and swallowed. There are four main types of sputum (Box 7.3).
• Clear or ‘mucoid’ sputum is produced in chronic bronchitis and COPD with no active infection.
• Yellow sputum occurs in acute lower respiratory tract infection (live neutrophils) and in asthma (eosinophils).
• Green purulent sputum (dead neutrophils) indicates chronic infection, e.g.in COPD or bronchiectasis. Purulent sputum is green because lysed neutrophils release the green-pigmented enzyme, verdoperoxidase. The first sputum produced in the morning by a patient with COPD may be green because of nocturnal stagnation of neutrophils.
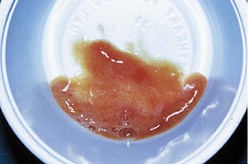
• Rusty red sputum can occur in early pneumococcal pneumonia, as pneumonic inflammation causes lysis of red cells (Fig. 7.3).
Amount: Bronchiectasis causes large volumes of purulent sputum, which varies with posture. Suddenly coughing up large amounts of purulent sputum on a single occasion suggests rupture of a lung abscess or empyema into the bronchial tree. Large volumes of watery sputum with a pink tinge in an acutely breathless patient suggest pulmonary oedema but, if occurring over weeks (bronchorrhoea), suggests alveolar cell cancer.
Taste or smell: Foul-tasting or smelling sputum suggests anaerobic bacterial infection, and occurs in bronchiectasis, lung abscess and empyema. In bronchiectasis a change of sputum taste may indicate an infective exacerbation.
Solid material: In asthma and allergic bronchopulmonary aspergillosis thick secretions can accumulate in airways and be coughed up as ‘worm-like’ structures that are casts of the bronchi (Fig. 7.4). Other solid matter sometimes coughed up includes necrotic tumour and inhaled foreign bodies, e.g. food, teeth and tablets.
Haemoptysis
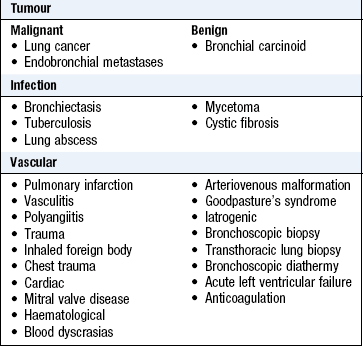
Haemoptysis is coughing up blood from the respiratory tract and always requires investigation (Box 7.4).
Amount and appearance: Blood-streaked clear sputum or clots in sputum for more than a week suggest lung cancer. Haemoptysis with purulent sputum suggests infection. Coughing up large amounts of pure blood is rare but potentially life-threatening; causes include lung cancer, bronchiectasis and tuberculosis and, less commonly, lung abscess, mycetoma, cystic fibrosis, aortobronchial fistula and granulomatosis with polyangiitis (formerly Wegener’s granulomatosis).
Duration and frequency: Bronchiectasis causes intermittent haemoptysis associated with copious purulent sputum over years. Daily haemoptysis for a week or more is a symptom of lung cancer; other causes include tuberculosis and lung abscess.
Single episodes of haemoptysis, if associated with symptoms, e.g. pleuritic chest pain and breathlessness, suggest pulmonary thromboembolism and infarction and need immediate investigation.
Dyspnoea
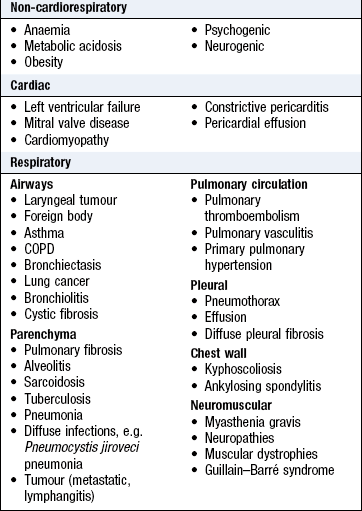
Dyspnoea (breathlessness) is undue awareness of breathing and is normal with strenuous physical exercise. Patients use terms such as ‘shortness of breath’, ‘difficulty getting enough air in’, or ‘tiredness’ (Box 7.5).
Mode of onset, duration and progression: Psychogenic breathlessness may occur suddenly at rest or while talking. Patients often say they cannot get enough air into their chest and need to take deep breaths. The resultant hypocapnia causes lightheadedness, dizziness, tingling in the fingers and round the mouth and chest tightness. These symptoms, in turn, aggravate anxiety and exacerbate the situation (Box 7.6 and Ch. 2).
Variability and aggravating/relieving factors: Breathlessness when lying flat (orthopnoea) is usually associated with left ventricular failure. It can also be a feature of respiratory muscle weakness, large pleural effusion, massive ascites, morbid obesity or any severe lung disease.
Breathlessness on sitting up (platypnoea) with relief on lying down is rare and due to right-to-left shunting through a patent foramen ovale, atrial septal defect or a large intrapulmonary shunt.
Breathlessness when lying on one side (trepopnoea) is due to unilateral lung disease (patient prefers the healthy lung down), dilated cardiomyopathy (patient prefers right side down) or tumours compressing central airways and major blood vessels.
Breathlessness that wakes the patient from sleep is typical of asthma and left ventricular failure (paroxysmal nocturnal dyspnoea). Patients with asthma typically wake between 3 and 5 a.m. and have associated wheezing. Breathlessness worse on waking is more typical of COPD and may improve after coughing up sputum.
Patients with exercise-induced asthma may notice that the breathlessness continues to worsen for 5–10 minutes after stopping activity. If you suspect asthma, ask about exposure to allergens, smoke, perfumes, fumes, cold air or drugs, e.g. aspirin, non-steroidal anti-inflammatory drugs. Common allergens are house dust mite (shaking bedding, hoovering), animals (cats, dogs, horses) and grass pollens (mowing the lawn, the ‘hayfever season’) and tree pollens. Breathlessness improving at weekends or holidays suggests occupational asthma. Ask about symptoms that accompany the breathlessness, e.g. chest pain, cough, wheeze (Box 7.11).
Severity: Breathlessness while walking on the flat, up gentle inclines or stairs indicates a significant condition. Severely breathless patients are dyspnoeic at rest, walking around the house, washing, dressing and even eating (Box 7.7).
COPD is characterised by airflow obstruction that is usually progressive and not fully reversible. It is defined as a reduced post-bronchodilator forced expiratory volume in 1 second (FEV1)/forced vital capacity (FVC) ratio of <70%.
Chest pain
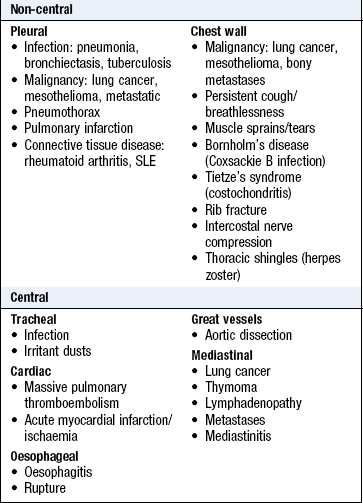
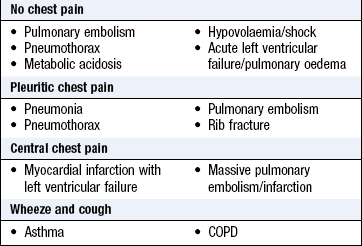
Chest pain can originate from the parietal pleura, the chest wall and mediastinal structures (Box 7.8). The lungs do not cause pain because their innervation is exclusively autonomic.
Pleural pain is sharp, stabbing and intensified by inspiration or coughing. Irritation of the parietal pleura of the upper six ribs causes localised pain. Irritation of the parietal pleura overlying the central diaphragm innervated by the phrenic nerve is referred to the neck or shoulder tip. The lower six intercostal nerves innervate the parietal pleura of the lower ribs and the outer diaphragm, and pain from these sites may be referred to the upper abdomen. Common causes of pleuritic chest pain are pulmonary embolism, pneumonia, pneumothorax and fractured ribs.
Chest wall pain which is sudden and localised after vigorous coughing or direct trauma is characteristic of rib fractures or intercostal muscle injury. Prevesicular herpes zoster and intercostal nerve root compression can cause chest pain in a thoracic dermatomal distribution. Chest wall pain due to direct invasion by lung cancer, mesothelioma or rib metastasis is typically dull, aching or gnawing, unrelated to respiration, progressively worsens and disrupts sleep. Pancoast’s tumour of the lung apex may involve the first rib and the brachial plexus, causing referred pain down the medial side of the ipsilateral arm.
Mediastinal pain is central, retrosternal and unrelated to respiration or cough. Irritant dusts or infection of the tracheobronchial tree produce a raw, burning retrosternal pain worse on coughing. A dull, aching retrosternal pain that disturbs sleep is a feature of cancer invading mediastinal lymph nodes or an enlarging thymoma. Massive pulmonary thromboembolism acutely increasing right ventricular pressure may produce central chest pain similar to myocardial ischaemia (Ch. 6).
Respiratory pattern
Respiratory rate: Tachypnoea is a respiratory rate >25 breaths/min. It is caused by increased ventilatory drive in fever, acute asthma and exacerbation of COPD, or reduced ventilatory capacity in pneumonia, pulmonary oedema and interstitial lung disease (Boxes 7.17 and Box 7.18). A slow respiratory rate of <10 breaths/min (bradypnoea) occurs in opioid toxicity, hypercapnia, hypothyroidism, raised intracranial pressure and hypothalamic lesions.
Breathing patterns: Periodic breathing (Cheyne–Stokes respiration) is cyclical with increasing rate and depth of breathing, followed by diminishing respiratory effort and rate, ending in a period of apnoea or hypopnoea. This relates to altered sensitivity of the respiratory centre to CO2 and delay in circulation time between the lung and chemoreceptors.
Hyperventilation is a common response to acute anxiety or emotional distress, and is often associated with respiratory alkalosis with hypocapnia. Breathing is deep, irregular and sighing, and patients feel unable to fill their lungs completely. When acute hyperventilation is sustained, tetany and occasionally grand mal seizure can occur. Hyperventilation with deep, sighing respirations (Kussmaul respiration) occurs in metabolic acidosis caused by diabetic ketoacidosis, acute renal failure, lactic acidosis, and salicylate and methanol poisoning. Although patients may not be aware of breathlessness, their respiratory rate increases and they appear to have ‘air hunger’.
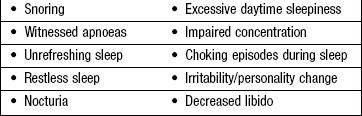
Apnoea is the absence of breathing; hypopnoea is a reduction in airflow or respiratory movements by >50% for 10 seconds or more. Obstructive sleep apnoea/hypopnoea syndrome (OSAHS) is the combination of excessive daytime sleepiness and recurrent upper airway obstruction with sleep fragmentation caused by upper airway obstruction from collapse of the retropharynx. A total of 2–4% of middle-aged men and 1–2% of middle-aged women (Box 7.10) have OSAHS with multiple apnoeas during sleep. They usually describe loud snoring, then a pause in breathing followed by a grunting noise and restoration of snoring. Simple (benign) snoring is more common than OSAHS, affecting 40% of middle-aged men and 20% of middle-aged women. Urgently investigate patients with daytime sleepiness and drowsiness while driving, especially if they have a heavy goods or public service vehicle licence.
The history
Cough: Ask about the duration of the cough and when, during the day, it is most severe. Cough on lying down in the evening may be due to gastro-oesophageal reflux; cough disrupting sleep is typical of asthma; cough on rising in the morning can be caused by rhinosinusitis and postnasal drip.
Wheeze: Ask about precipitating factors such as exercise and exposure to allergens (pets, pollens) and relationship to occupation.
Sputum: Ask patients specifically about sputum as they may find it difficult to discuss or may swallow it. Ask about the colour of any sputum produced and how many teaspoonfuls are coughed up daily.
Haemoptysis: Establish the volume and nature of the blood. Clarify whether the blood was coughed up from the respiratory tract, vomited (upper gastrointestinal tract) or suddenly appeared in the mouth without coughing (nasopharyngeal).
Breathlessness: Use the Medical Research Council grading scale to characterise breathlessness (Box 7.7). How far can the patient walk before stopping to rest? Get an estimate of the time taken to walk a given distance and the number of stops involved: say, from home to a local shop. Ask about the effect on work and hobbies such as golf, gardening, dancing, swimming or hill walking, Can the patient walk up hills or keep up with contemporaries? Box 7.9 outlines some combinations of symptoms in acutely breathless patients.
Chest pain: Characterise chest pain using SOCRATES (Box 2.10).
Respiratory pattern: Ask about any change in the rate or pattern of breathing. If the patient has daytime sleepiness, ask the patient’s bed partner about apnoea, loud snoring, nocturnal restlessness, irritability and personality change (Box 7.11).
Drug history
Detail the type of inhaler, dose in micrograms (not puffs) and frequency. Ask patients to demonstrate how they use the inhaler to check that they are doing this correctly. Note the effectiveness of previously prescribed medications, e.g. oral corticosteroids and β-agonist inhalers and current and previous medications, if you suspect drug-induced respiratory disease (Box 7.13).
Family history
Cystic fibrosis is the most common severe autosomal recessive disease in Europeans, with a carrier rate of 1 : 25 and an incidence of ~1 : 2500 live births. α1-antitrypsin deficiency (associated with emphysema and COPD) also has recessive inheritance.
A family history of asthma, eczema and hayfever increases the chance of a predisposition to form excess IgE in response to allergen (atopy). ‘Asthma’ in parents or grandparents who were smokers may have been misdiagnosed COPD. A family history of tuberculosis can represent significant past exposure that may reactivate later in life. Patients with asbestos-related disease and no obvious occupational exposure may have had significant exposure through asbestos-contaminated work clothes brought home for cleaning by a relative in a relevant occupation.
Social history
Smoking: Although cigarette smoking has declined in the UK, the current prevalence of COPD and incidence of lung cancer reflect historical smoking patterns. Establish when patients started and stopped smoking and their average tobacco consumption as cigarettes/day or ounces/grams of ‘roll-up’ tobacco/week. Calculate the ‘pack year’ consumption: smoking 1 pack of 20 cigarettes per day for 1 year equates to 1 pack year (Box 2.20). Patients with COPD usually have smoked >20 pack years. Stopping smoking at any age is crucial to improving health; beyond 40 years people lose, on average, 3 months of life expectancy for every further year they continue smoking.
Occupational history
Occupation is important in many respiratory disorders. Record all the occupations, full- and parttime, since the patient left school and the number of years spent in each job (Box 7.14). Find out exactly what the job entailed and the length of any exposure. Exposure to a recognised hazard helps diagnosis, has implications for current employment and may be the basis for compensation.
Asbestos was widely used in shipyards, building construction and plumbing until the 1970s, and asbestos-related lung disease, e.g. mesothelioma and asbestosis, is the greatest single cause of work-related death in the UK. Occupation is particularly important in adult-onset asthma, e.g. baker’s asthma due to flour dust, and interstitial lung disease, e.g. farmer’s lung. Some occupational diseases are worse at the beginning of the working week, e.g. byssinosis due to cotton dust and humidifier fever due to organism-contaminated water in air-conditioning systems. Occupational asthma gets better on holidays away from work.
The physical examination
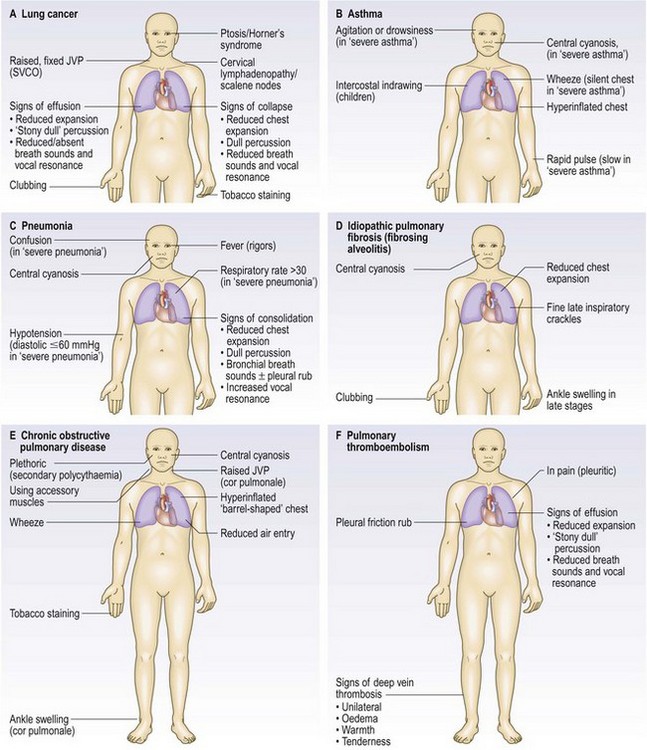
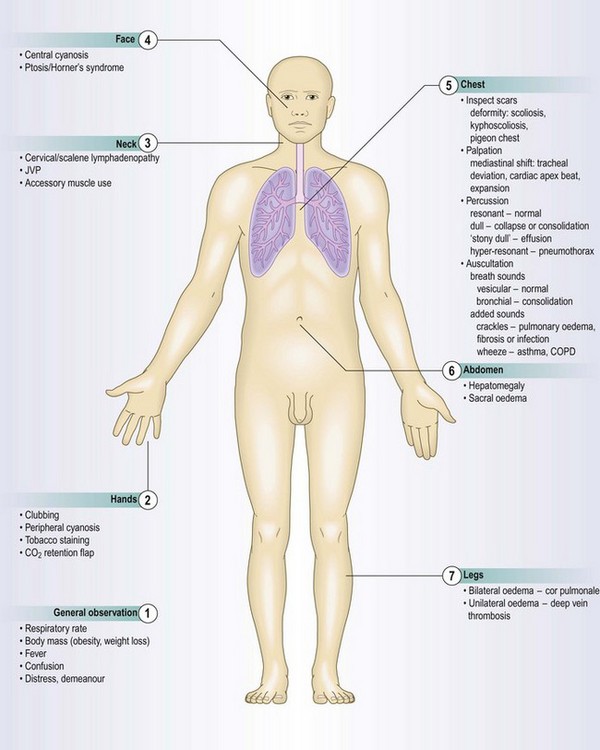
Observe patients as you first meet them. Look for breathlessness, weight loss, cyanosis and their mental state.
Abnormal findings: The sternocleidomastoid, platysma and trapezius muscles are accessory muscles of respiration and their use is an early sign of airways obstruction. If the patient sits forward with the hands/arms on the thighs or knees to ‘fix’ the shoulder girdle, he raises the clavicles and upper chest, increasing lung volume and negative intrathoracic pressure. Use of accessory muscles is characteristic in severe COPD and acute severe asthma.
In severe respiratory failure or, rarely, bilateral phrenic nerve lesions causing diaphragmatic palsy, the abdomen and chest move paradoxically; during inspiration the abdomen moves inwards as the chest wall moves out.
Some patients with severe COPD appear to breathe with ‘pursed lips’. This manoeuvre increases positive end-expiratory pressure, reducing small-airway collapse and improving ventilation.
Abnormal findings: Cyanosis is the blue colour caused by an absolute concentration of deoxygenated haemoglobin of >50 g/l.
Central cyanosis reflects arterial hypoxaemia. With a normal haemoglobin concentration, central cyanosis occurs when the arterial oxygen saturation falls below 90%, corresponding to a PaO2 of approximately 8 kPa (60 mmHg). In anaemic or hypovolaemic patients, you may not see cyanosis because severe hypoxia is required to produce the necessary concentration of deoxygenated haemoglobin. Patients with polycythaemia become cyanosed at higher arterial oxygen tensions. Rarely, central cyanosis is caused by methaemoglobinaemia due to intravascular haemolysis or drugs.
Peripheral cyanosis is only seen in the fingers and toes and is usually due to circulatory disorders or cold, but can also occur in patients with severe central cyanosis.
Abnormal findings: Diastolic pressure of <60 mmHg is associated with increased mortality in community-acquired pneumonia (Boxes 7.15 and 7.17). In pneumothorax, hypotension may indicate the development of ‘tension’ with reduction in venous return to the heart and risk of cardiac arrest. Pulsus paradoxus is an exaggeration of the normal variability of pulse volume with the respiratory cycle (Box 7.18). A fall in pulse volume and consequent fall in systolic blood pressure of >10 mmHg during inspiration is abnormal and can occur in cardiac tamponade (Ch. 6).
Hands
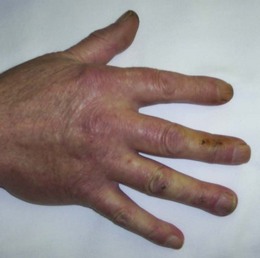
Clubbing: The majority of patients with finger clubbing have thoracic disease (lung cancer, bronchiectasis, interstitial lung disease) but it is also associated with gastrointestinal disorders and can be familial (Box 3.10). Rarely, clubbing develops relatively quickly over several weeks, in empyema.
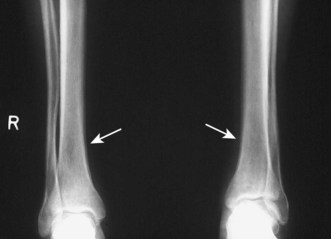
Hypertrophic pulmonary osteoarthropathy is rare and almost always associated with lung cancer, usually squamous cancer. Pronounced clubbing of fingers and toes occurs, with pain and swelling affecting the wrists and ankles. X-rays of the distal forearm and lower legs show subperiosteal new bone formation separate from the cortex of the long bones (Fig. 7.7).
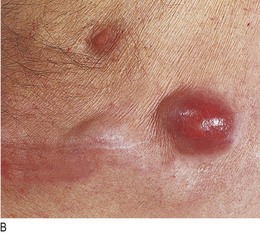
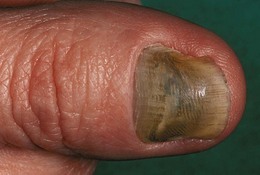
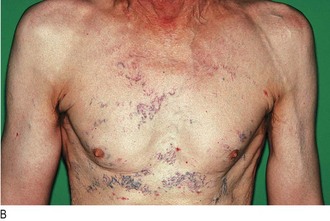
Discoloration of the fingers and nails: A brownish stain on the fingers and nails in cigarette smokers is caused by tar, not nicotine (Fig. 7.8). The rare ‘yellow nail syndrome’ is associated with lymphoedema and an exudative pleural effusion (Fig. 7.9).
A fine finger tremor is often caused by excessive use of β-agonist or theophylline bronchodilator drugs.
A coarse flapping tremor (asterixis) is seen with severe ventilatory failure and carbon dioxide retention. This is the result of intermittent failure of parietal mechanisms required to maintain posture. Unilateral asterixis is due to structural abnormality in the contralateral cerebral hemisphere.
Neck
Jugular venous pressure (JVP):
Abnormal findings: The JVP is raised in right-sided heart failure. Chronic hypoxia in COPD leads to pulmonary arterial vasoconstriction, pulmonary hypertension, right heart dilatation and peripheral oedema with elevation of the JVP. This is cor pulmonale. The JVP is high if the intrathoracic pressure is raised in tension pneumothorax or severe acute asthma. Massive pulmonary embolism may cause the JVP to be so high that the patient has to be sitting upright to see it.
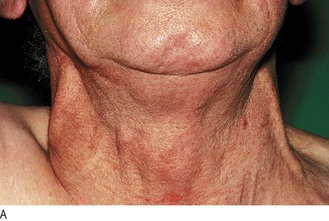
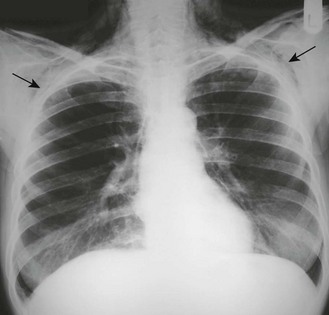
In superior vena caval obstruction (SVCO) (Fig. 7.11A) the JVP is raised and non-pulsatile, and the abdominojugular reflex is absent. Most cases are due to lung cancer compressing the superior vena cava. Other causes include lymphoma, thymoma and mediastinal fibrosis. Facial flushing, distension of neck veins and stridor can occur in SVCO when the arms are raised above the head.
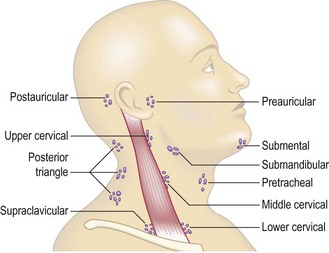
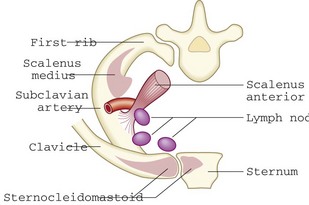
Abnormal findings: Scalene lymph node enlargement may be the first evidence of metastatic lung cancer and localised cervical lymphadenopathy is a common presenting feature of lymphoma. In Hodgkin’s disease, the lymph nodes are typically ‘rubbery’; in dental sepsis and tonsillitis they are usually tender; in tuberculosis and metastatic cancer they are often ‘matted’ together to form a mass; and calcified lymph nodes feel stony hard. Palpable lymph nodes, fixed to deep structures or skin, are usually malignant (Box 7.19).
Thorax
Ask the patient to sit over the edge of a bed or on a chair if possible with the chest and upper abdomen fully exposed. Fully examine the back of the chest first and then the front.
Normal findings: The chest should be symmetrical and elliptical in cross-section. The anteroposterior diameter should be less than the lateral diameter.
Abnormal findings: When the anteroposterior diameter is greater than the lateral diameter, the chest is ‘barrel-shaped’. This is associated with lung hyperinflation in patients with severe COPD (Fig. 7.14A), although the degree of deformity does not correlate with the severity of airways obstruction or lung function.
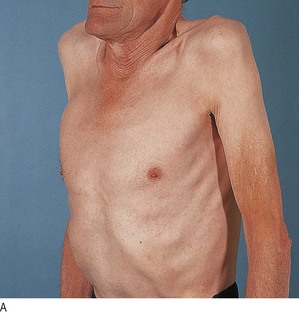
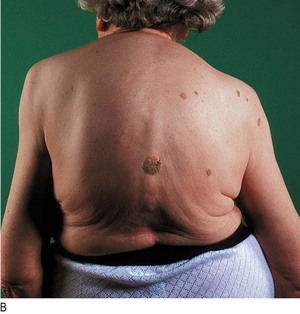
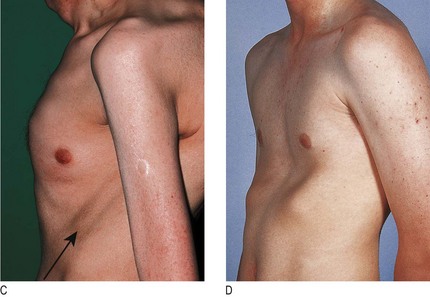
Fig. 7.14 Abnormalities in the shape of the chest.
(A) Hyperinflated chest with intercostal indrawing. (B) Kyphoscoliosis. (C) Pectus carinatum with prominent Harrison’s sulcus (arrow). (D) Pectus excavatum.
Kyphosis is an exaggerated anterior curvature of the spine and scoliosis is lateral curvature. Kyphoscoliosis, involving both deformities (Fig. 7.14B), may be idiopathic or secondary to childhood poliomyelitis or spinal tuberculosis, and may be grossly disfiguring and disabling. It may reduce ventilatory capacity and increase the work of breathing. These patients develop progressive ventilatory failure with carbon dioxide retention and cor pulmonale at an early age.
Pectus carinatum (pigeon chest) is a localised prominence of the sternum and adjacent costal cartilages, often accompanied by indrawing of the ribs to form symmetrical horizontal grooves (Harrison’s sulci) above the costal margin (Fig. 7.14C). These result from lung hyperinflation with repeated vigorous contractions of the diaphragm while the bony thorax is in a pliable prepubertal state. It is most often caused by severe and poorly controlled childhood asthma but can occur in osteomalacia and rickets.
Pectus excavatum (funnel chest) is a developmental deformity with a localised depression of the lower end of the sternum (Fig. 7.14D) or, less commonly, of the whole length of the sternum. Patients are usually asymptomatic but concerned about their appearance. In severe cases the heart is displaced to the left and the ventilatory capacity is reduced.
Skin: Subcutaneous lesions may be visible, including metastatic tumour nodules, neurofibromas and lipomas, as may vascular anomalies, e.g. the dilated venous vascular channels of SVCO (Fig. 7.11B).
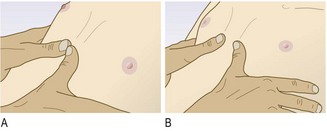
Palpation: Determine the position of the mediastinum by examining the trachea and cardiac apex beat (Box 7.20).
Abnormal findings: Shift of the upper mediastinum causes tracheal deviation. Displacement of the cardiac apex beat may indicate shift of the lower mediastinum (Ch. 6). Displacement of the cardiac impulse without tracheal deviation is usually due to left ventricular enlargement but can occur in scoliosis, kyphoscoliosis or severe pectus excavatum. The cardiac apex beat may be difficult to localise in obesity, pericardial effusion, poor left ventricular function or lung hyperinflation, as in COPD. The heave of right ventricular hypertrophy, found in severe pulmonary hypertension, is best felt at the left sternal edge (Fig. 6.22C).
A ‘tracheal tug’ is found in severe hyperinflation; resting on the patient’s trachea, your fingers move inferiorly with each inspiration.
Normal findings: Both sides of the thorax should expand equally during normal (tidal) breathing and maximal inspiration.
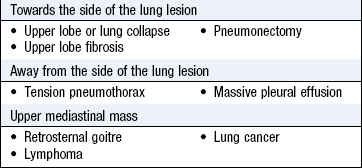
Abnormal findings: Reduced expansion on one side indicates abnormality on that side: for example, pleural effusion, lung or lobar collapse, pneumothorax and unilateral fibrosis. Bilateral reduction in chest wall movement is common in severe COPD and diffuse pulmonary fibrosis.
Paradoxical inward movement may indicate diaphragmatic paralysis or, more commonly, severe COPD. Double fracture of a series of ribs or of the sternum allows the chest wall between the fractures to become mobile or ‘flail’ (Fig. 19.6).
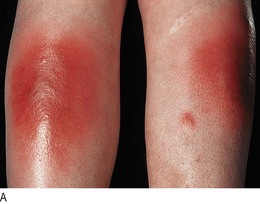
Subcutaneous emphysema produces a characteristic crackling sensation over gas-containing tissue (Fig. 7.17) and there may be diffuse swelling of the chest wall, neck and face. It may complicate severe acute asthma, spontaneous or traumatic pneumothorax, or rupture of the oesophagus, and is a complication of intercostal drainage. Mediastinal emphysema occurs if air tracks into the mediastinum and is associated with a characteristic systolic ‘crunching’ sound on auscultating the precordium (Hamman’s sign).
Tenderness over the costal cartilages is found in the costochondritis of Tietze’s syndrome. Localised rib tenderness can be found over areas of pulmonary infarction or fracture.
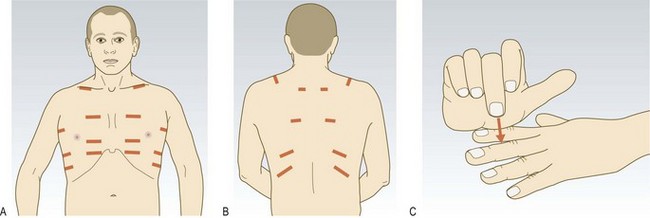
Percussion: Percussion allows you to hear the pitch and loudness of the percussed note and to feel for postpercussive vibrations. Percuss in sequence over corresponding areas on both sides of the chest (Fig. 7.18).
Normal findings: Normal lung produces a resonant note (Box 7.21).
Abnormal findings: A pneumothorax produces a hyperresonant note, whereas percussion over solid structures, e.g. liver, heart or a consolidated area of lung (pneumonia) produces a dull note. Percussion over fluid, e.g. pleural effusion (Box 7.21), produces an extremely dull (‘stony dull’) note. Find the upper level of liver dullness by percussing down the anterior wall of the right chest; in adults the upper level of liver dullness is the fifth rib in the mid-clavicular line. Resonance below this is a sign of hyperinflation (COPD or severe asthma). The area of cardiac dullness over the left anterior chest may be decreased when the lungs are hyperinflated. Basal dullness due to elevation of the diaphragm is easily confused with pleural fluid.
Auscultation: Most sounds reaching the chest wall are low-frequency and best heard with the stethoscope bell. The diaphragm locates higher-pitched sounds, such as pleural friction rubs. Stretching the skin and hairs under the diaphragm during deep breathing can produce anomalous noises like crackles, and in thin patients it may be difficult to apply the diaphragm fully to the chest wall skin.
Normal findings: Turbulent flow in large airways causes normal breath sounds heard at the chest wall. Through a stethoscope they have a rustling (vesicular) quality. The larynx makes little contribution in quiet breathing but may accentuate the noise in deep respiration. The intensity of breath sounds relates to airflow and the tissue through which the sound travels.
The pattern and intensity of breath sounds reflect regional ventilation. Sounds are decreased through normal lungs since the parenchyma transmits sounds poorly. In an upright patient breath sounds are normally loudest at the apex in early inspiration and at the bases in mid-inspiration. During expiration, normal breath sounds rapidly fade as airflow decreases.
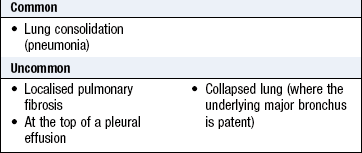
Abnormal findings: Diminished vesicular breathing occurs in obesity, pleural effusion, marked pleural thickening, pneumothorax, hyperinflation due to COPD and over an area of collapse where the underlying major bronchus is occluded (Box 7.22). If breath sounds appear reduced, ask the patient to cough. If the reduced breath sounds are due to bronchial obstruction by secretions, they are likely to become more audible after coughing.
Bronchial breathing is a high-pitched breath sound with a hollow or blowing quality similar to that heard over the trachea and larynx during tidal breathing. The breath sounds are of similar length and intensity in inspiration and expiration, with a characteristic pause between the two phases (Fig. 7.19). Bronchial breath sounds are found when normal lung tissue is replaced by uniformly conducting tissue and the underlying major bronchus is patent (Box 7.27), so it tends to exclude the possibility of an obstructing lung cancer. Bronchial breathing and whispering pectoriloquy are heard over pulmonary consolidation (pneumonia), at the top of a pleural effusion and over areas of dense fibrosis.
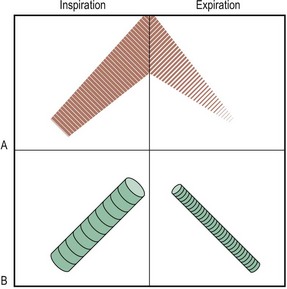
Fig. 7.19 Diagrammatic representation of breath sounds.
(A) Vesicular. (B) Bronchial. Note the gap between inspiration and expiration and change in pitch and the blowing, tubular quality of bronchial breath sounds.
Aegophony is a bleating or nasal sound heard over consolidated lung (pneumonia) or at the upper level of a pleural effusion. It is due to enhanced transmission of high-frequency noise across abnormal lung, with lower frequencies filtered out (Box 7.28).
Added sounds: Crackles are interrupted non-musical sounds and result from collapse of peripheral airways on expiration. On inspiration, air rapidly enters these distal airways, and the alveoli and small bronchi open abruptly, producing the crackling noise.
Note when crackles occur within the respiratory cycle (Box 7.25). Early inspiratory crackles suggest small airways disease and can occur in bronchiolitis. In pulmonary oedema crackles occur in mid-inspiration. Fine late inspiratory crackles, which sound similar to rubbing hair between your fingers, are characteristic of pulmonary fibrosis. Bronchiectasis can cause crackles throughout inspiration and expiration.
Crackles may be heard when air bubbles through secretions in major bronchi, dilated bronchi in bronchiectasis or in pulmonary cavities. These crackles sound coarse, have a gurgling quality and change if the secretions are dislodged by coughing.
Wheeze is caused by continuous oscillation of opposing airway walls and has a musical quality. Wheezes imply airway narrowing and are timed in relation to the respiratory cycle. They are louder on expiration because airways normally dilate during inspiration and narrow on expiration. Inspiratory wheeze implies severe airway narrowing. High-pitched wheeze arises from smaller airways and has a whistling quality. Low-pitched wheeze originates from larger bronchi. Distinguish wheeze from the harsh rasping sound of stridor (p. 143).
Wheeze is characteristic of asthma and COPD, but is a poor guide to severity of airflow obstruction. In severe airways obstruction wheeze may be absent because of reduced airflow, producing a ‘silent chest’. A fixed bronchial obstruction, most commonly due to lung cancer, may cause localised wheeze with a single musical note that does not clear on coughing.
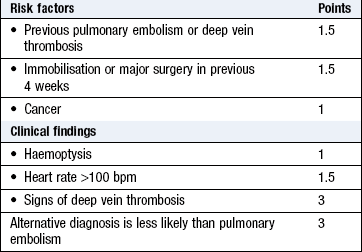
A pleural friction rub is a creaking sound similar to that produced by bending stiff leather or treading in fresh snow. It is produced when inflamed parietal and visceral pleurae move over one another and is best heard with the stethoscope diaphragm. It may be heard only on deep breathing at the end of inspiration and beginning of expiration. A pleural rub is usually associated with pleuritic pain and may be heard over areas of inflamed pleura in pulmonary infarction due to pulmonary embolism. Pulmonary embolism is obstruction of part of the pulmonary vascular tree by thrombus that has travelled from a distant site, e.g. deep veins in the legs or pelvis (Figs 7.18 and 7.19; Boxes 7.23 and 7.24). Pulmonary embolism is frequently unrecognised and fewer than one-third of patients have symptoms or signs of deep vein thrombosis (Box 7.26). A pleural friction rub may be heard in pneumonia or pulmonary vasculitis. If the pleura adjacent to the pericardium is involved, a pleuropericardial friction rub may also be heard. Pleural friction rubs disappear if an effusion separates the pleural surfaces.
A pneumothorax click is a rhythmical sound, synchronous with cardiac systole, and produced when there is air between the two layers of pleura overlying the heart.
A mid-expiratory ‘squeak’ is characteristic of obliterative bronchiolitis – a rare complication of rheumatoid arthritis – where small airways are narrowed or obliterated by chronic inflammation and fibrosis.
Investigations
See Box 7.29.
Chest X-ray
The standard chest X-ray is a posteroanterior (PA) view taken with the film in front of the anterior chest and the X-ray source 2 metres behind the patient (Fig. 7.22). In an anteroposterior (AP) film the X-ray source is in front of the patient, which tends to enlarge anterior structures such as the heart. Always compare an abnormal chest X-ray with previous films to see if abnormalities are resolving or longstanding.
Sputum examination
Inpatients with respiratory symptoms should have a sputum pot for inspection.
Gram stain helps rapid identification of the causative organism: for example, Gram-positive – pneumococcus or staphylococcus; Gram-negative – Haemophilus influenzae. If the patient’s symptoms and chest X-ray suggest tuberculosis send several sputum samples urgently for auramine staining (screening); if these are positive, obtain a Ziehl–Neelsen stain. Positive samples indicate a high degree of infectivity; the patient should be urgently isolated and treated, and the condition notified.
Pulse oximetry
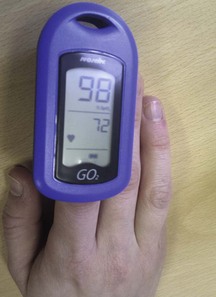
An oximeter is a spectrophotometric device that measures arterial oxygen saturation (SpO2) by determining the differential absorption of light by oxyhaemoglobin and deoxyhaemoglobin. Modern oximeters use a probe incorporating a light source and sensor attached to a patient’s ear or finger (Fig. 7.25).
Oximeters are easy to use, portable, non-invasive and inexpensive. They are widely used for the continuous measurement of SpO2 and to adjust oxygen therapy. In acutely ill patients with no risk of CO2 retention, SpO2 should be maintained at 94–98%. Movement artifact, poor tissue perfusion, hypothermia and nail varnish can lead to spuriously low SpO2 values. Dark skin pigmentation and raised levels of bilirubin or carboxyhaemoglobin can result in false increases in SpO2. Oximetry is less accurate with saturations <75%.
Arterial blood gas analysis
Arterial blood gas (PaO2, PaCO2) and acid–base (pH and HCO3–) status are obtained from heparinised samples of arterial blood from the radial, brachial or femoral artery (Box 7.30 and Fig. 7.26).
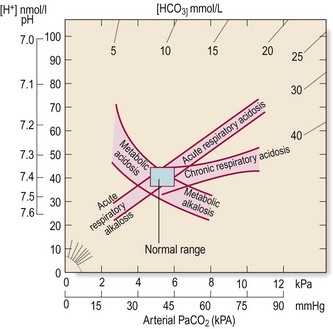
Fig. 7.26 Acid–base diagram.
Acid–base nomogram showing the relationships between pH (H+), PaCO2 and bicarbonate. The bands represent 95% confidence limits of acid–base disturbance.
Respiratory acidosis: An acute rise in PaCO2 caused by alveolar hypoventilation associated with a decrease in pH occurs in severe acute asthma, severe pneumonia, exacerbations of COPD and neuromuscular disorders. Elevation of PaCO2 for more than 2–3 days may occur in COPD, respiratory muscle weakness due to neuromuscular disorders and thoracic skeletal deformities, and leads to renal retention of HCO3– and normalisation of pH, i.e. compensated respiratory acidosis. In some patients with COPD low PaO2 levels drive respiratory effort. Removal of this stimulus by excessive oxygen therapy may result in alveolar hypoventilation with further increase in PaCO2, which can lead to deterioration and death.
Respiratory alkalosis: Hyperventilation occurs with respiratory conditions (asthma, pulmonary thromboembolism, pleurisy), high altitude and acute anxiety. Alveolar hyperventilation leads to decrease in PaCO2 and a consequent increase in pH. If hyperventilation persists, as occurs with stays at high altitude, increased renal excretion of HCO3– results in normalisation of pH, i.e. compensated respiratory alkalosis.
Spirometry
Dynamic lung volumes are measured by inhaling to total lung capacity and then exhaling into a spirometer with maximal effort to residual volume. The volume exhaled in the first second is the FEV1 and the total volume exhaled is the FVC. Normal predictive values for FEV1 and FVC are influenced by age, gender, height and race. In healthy young and middle-aged adults the FEV1/FVC ratio is usually >75%. In the elderly the ratio is usually 70–75%. Reduction in the FEV1/FVC ratio indicates airway obstruction. The severity of obstruction is represented by the absolute FEV1 expressed as a percentage of predicted. Airway obstruction that reverses with inhaled β2-agonist or oral steroid over 5 days or more (an absolute increase in FEV1 >200 ml that is >15% of baseline) favours a diagnosis of asthma over COPD (Box 7.31).
In interstitial lung disorders, e.g. idiopathic pulmonary fibrosis, pulmonary sarcoidosis or hypersensitivity pneumonitis, there is a decrease in FVC with preservation of FEV1/FVC ratio, a restrictive defect (Fig. 7.27).
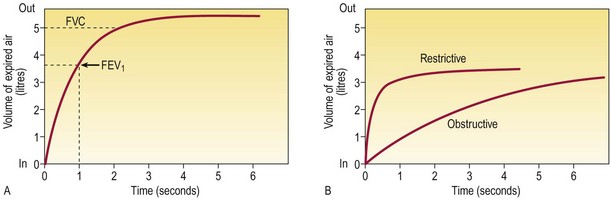
Fig. 7.27 Volume–time curves obtained using a wedge-bellows spirometer.
(A) The patient takes a full inspiration and exhales forcibly and fully. Maximal flow decelerates as forced expansion proceeds. (B) Obstructive and restrictive patterns. In obstruction, FEV1/FVC is low; in restriction, it is normal or high. FEV1, forced expiratory volume in 1 second; FVC, forced vital capacity.
Peak expiratory flow
Peak expiratory flow (PEF) is measured by inhaling to total lung capacity and exhaling into a peak flow meter with maximal effort. Measuring PEF is essential in the assessment of acute asthma and for the diagnosis of occupational asthma where falls in PEF occur during the working week but improve during weekends and holidays. Early-morning falls in PEF of >60 L/min (>20% maximal PEF) are very suggestive of asthma. A >60 L/min fall in PEF (>15% baseline) after exercise is diagnostic of asthma (Box 7.29).