Anticonvulsants
1 Define epilepsy and briefly summarize the various types of seizures.
2 List and describe general adverse reactions to anticonvulsants.
3 Summarize the pharmacologic effects, adverse reactions, and drug interactions of the main anticonvulsants—carbamazepine, valproate, phenobarbital, and phenytoin.
4 Name two miscellaneous anticonvulsants and describe the workings of each.
5 Provide several examples of new types of anticonvulsants, including the mechanism of action, indications, and adverse reactions of each.
EPILEPSY
Epilepsy comprises a group of disorders that involve a chronic stereotyped recurrent attack of involuntary behavior or experience or changes in neurologic function caused by electrical activity in the brain that can be recorded via an electroencephalogram (EEG). This activity can be localized or generalized. Each episode is termed a seizure. The seizure may be accompanied by motor activity, such as convulsions, or by other neurologic changes (e.g., sensory or emotional).
Because seizure disorders are estimated to affect approximately 1% of the population, the dental team is likely to encounter a patient with epilepsy. Because these anticonvulsant agents are used chronically, one must consider potential adverse reactions that might alter dental treatment.
There are many etiologies for epilepsy, including infection, trauma, toxicity to exogenous agents, genetic or birth influences, circulatory disturbances, metabolic or nutritional alterations, neoplasms, hereditary factors, fevers, and degenerative diseases. The majority of epileptic patients have idiopathic epilepsy; this term is used when the cause is unknown.
Epilepsy has been classified based on causes, symptoms, duration, precipitating factors, postictal state (postseizure), and aura. Currently, The International Classification of Epileptic Seizures (Figure 16-1) divides seizures into two major groups and a miscellaneous group. The two major groups are partial and generalized seizures. Partial seizures are divided into simple and complex attacks. The most common generalized seizures are (1) tonic-clonic and (2) absence seizures (Box 16-1).
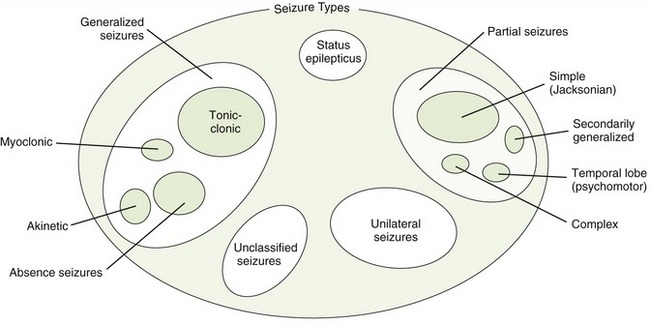
FIGURE 16-1 The International Classification of Epileptic Seizures: status epilepticus, elementary motor (Jacksonian) autonomic seizures, temporal lobe (psychomotor) seizures, secondarily generalized seizures, partial seizures, unilateral seizures, generalized seizures, tonic-clonic seizures, myoclonic seizures, akinetic seizures, and unclassified seizures. Seizure type patterns: same pattern indicates that same group of drugs are usually effective.
Generalized Seizures
Generalized seizures are divided into two large groups: (1) absence and (2) tonic-clonic types. Consciousness is lost in both types. Whereas little movement occurs in absence seizures, in tonic-clonic seizures, major movement of large muscle groups occurs. Management of seizures is discussed at the end of this chapter. Often, the patient may experience an aura (a brief period of heightened sensory activity) before the onset of the seizure. It may be characterized by numbness, nausea, or unusual sensitivity to light, odor, or sound.
 ABSENCE SEIZURES (PETIT MAL)
ABSENCE SEIZURES (PETIT MAL)
The symptoms of absence (petit mal) seizures include a brief (few seconds) loss of consciousness with characteristic EEG waves and little movement. Absence seizures usually begin during childhood and disappear in middle age. The patient is usually unaware that these seizures are occurring and body tone is not lost. There is no aura or postictal state, and the patient quickly returns to normal activity. The drug of choice in the treatment of typical absence seizures is either ethosuximide, valproate, or lamotrigine (Table 16-1).
TABLE 16-1
ANTICONVULSANT DRUGS OF CHOICE
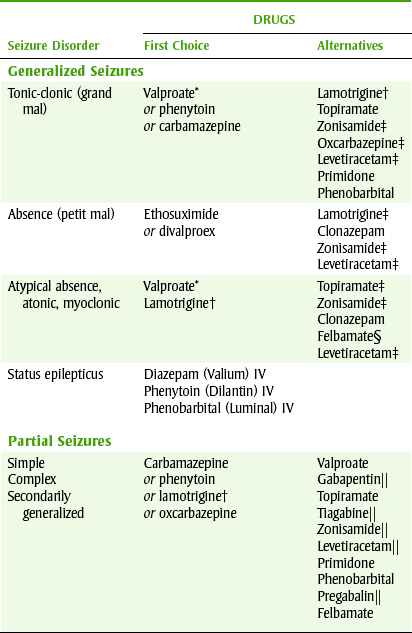
*Not Food and Drug Administration (FDA) approved unless absence is involved.
†FDA approved for adjunctive therapy in adults and children older than 2 years with partial seizures or with Lennox-Gastaut syndrome, and as monotherapy in adults with partial seizures as a substitute for carbamazepine, phenytoin, phenobarbital, or valproate as the single antiepileptic drug.
‡Not FDA approved for this indication.
§FDA approved as adjunctive therapy for patients with Lennox-Gastaut syndrome.
¶Only FDA approved for adjunctive therapy.
Management of absence seizures poses no problems for the dental team. The team’s main concern when treating patients with absence seizures is the adverse reactions that can occur from long-term administration of the drugs used to treat the disease.
 TONIC-CLONIC SEIZURES
TONIC-CLONIC SEIZURES
The generalized tonic-clonic (grand mal) seizures include longer periods of loss of consciousness and major motor activity of the large muscles of the body. The seizure begins by the body becoming rigid and the patient falling to the floor. Urination, apnea, and a cry may be present. Tonic rigidity is followed by clonic jerking of the face, limbs, and body. The patient may bite the cheek or tongue. Finally, the patient becomes limp and comatose. Consciousness returns gradually, with postictal confusion, headache, and drowsiness. Some patients experience prodromal periods of varying durations, but a true aura does not occur. Because this seizure type involves the violent movement of major muscle groups, it is more likely to result in serious injury to the patient. Valproic acid, phenytoin, and carbamazepine are used to treat tonic-clonic seizures.
 STATUS EPILEPTICUS
STATUS EPILEPTICUS
Status epilepticus seizures are continuous tonic-clonic seizures that last longer than 30 minutes or recur before the end of the postictal period of the previous seizure. This is an emergency situation, and rapid therapy is required, especially if the seizure activity has produced hypoxia. Parenteral benzodiazepines, such as diazepam (Valium), are the drugs of choice to control this seizure type (see Chapter 11).
Partial (Focal) Epilepsies
Partial epilepsies involve activation of only part of the brain, and the location of the activity determines the clinical manifestation. When consciousness is not impaired, the attack is called an elementary (simple) partial attack. When consciousness is impaired, the attack is termed a complex partial attack. Complex seizures are also called psychomotor or temporal-lobe seizures. In contrast to absence seizures that last a few seconds, these complex partial seizures last several minutes. Some patients with complex partial seizures have an aura, and full consciousness is slow to return. For the partial epilepsies, carbamazepine, phenytoin, phenobarbital, and primidone are used.
DRUG THERAPY OF PATIENTS WITH EPILEPSY
Drug therapy of the patient with epilepsy has variable efficacy, from complete control of all seizures to reducing the frequency of seizures. Anticonvulsant agents may be used singly or in combination. The goal is to control seizures and minimize potential adverse reactions. Some newer anticonvulsants are able to treat previously untreatable seizures, but more serious side effects can accompany them. General principles on the management of the dental patient taking any anticonvulsant agents are listed in Box 16-2.
Anticonvulsant agents are central nervous system (CNS) depressants that attempt to prevent epileptic seizures without causing excessive drowsiness. Although their exact mechanisms of action are unknown, these agents prevent the spread of abnormal electric discharges in the brain (Figure 16-2).
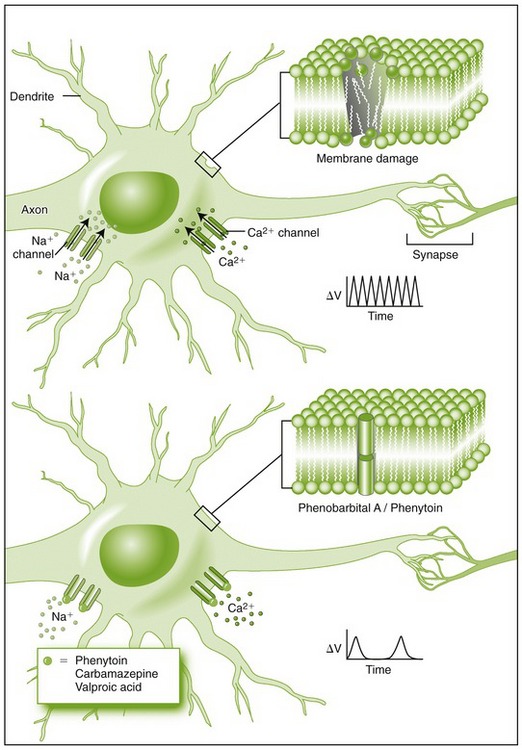
FIGURE 16-2 Effects of anticonvulsants on neurons. Upper, Damaged neuronal membranes, as shown in the section on the upper right of the figure, result in sodium and calcium influxes via their respective channels that cause repeated firing shown in the voltage-time curve to the right of the figure. Lower, Anticonvulsants, such as phenytoin (Dilantin), carbamazepine (Tegretol), and valproic acid (Depakote), represented by the small circles, block sodium and calcium channels, resulting in a substantially diminished rate of firing as shown in the voltage-time curve to the right of the figure. Both phenytoin and phenobarbital, shown as cylinders, are also thought to stabilize the damaged neuronal membrane as shown in the membrane section schematized in the upper right drawing. (From McPherson RA, Pincus MR: Henry’s clinical diagnosis and management by laboratory methods, ed 21, Philadelphia, 2007, Saunders.)
The anticonvulsant drug used to treat a specific patient depends on the type of seizures the patient has. Because these agents are usually taken for life, their chronic toxicity becomes an important consideration in choosing a particular anticonvulsant agent and determining the drug’s dental implications.
General Adverse Reactions to Anticonvulsant Agents
Several factors make dosing with anticonvulsants more difficult than with other drugs. First, the anticonvulsants have a narrow therapeutic index, so the dose must be carefully titrated to obtain the desired blood levels. It cannot be too low or too high. Second, most anticonvulsants stimulate liver microsomal enzymes that metabolize both themselves, other anticonvulsants, and other drugs that the patient may be taking. When a second anticonvulsant is added to the first, it changes the metabolism of both anticonvulsants. The third effect, which may be even more important than the others, is that the metabolism of the anticonvulsants can saturate the liver microsomal enzymes. With low doses, the metabolism is first order and the drug is removed from the body. At some point, when the enzymes become saturated, the metabolism converts to zero-order kinetics and the drug level can increase abruptly. At the point of saturation, a small change in the dose can lead to a large increase in the blood level of the drug. The dental team must be aware of the side effects of the anticonvulsant agents that might influence dental treatment.
The anticonvulsant drugs possess a unique set of adverse reactions. Adverse reactions that the anticonvulsants have in common are discussed first.
 CENTRAL NERVOUS SYSTEM DEPRESSION
CENTRAL NERVOUS SYSTEM DEPRESSION
Depressed CNS function is a common side effect of the anticonvulsant agents. Tolerance often develops to these sedative effects while the anticonvulsant effect persists. Impaired learning and cognitive abilities occur in some patients. Behavior alterations reported include both hyperactivity and sedation. Another CNS side effect is exacerbation of a seizure type that is not being treated. This CNS depression is additive with other CNS depressants such as the opioids. If another CNS drug is given to the patient, the dose should be reduced.
 GASTROINTESTINAL DISTRESS
GASTROINTESTINAL DISTRESS
Gastrointestinal distress, including anorexia, nausea, and vomiting, can occur with most anticonvulsants. These effects can be minimized by taking the drug with food. Agents with adverse reactions related to the gastrointestinal tract, for example, nonsteroidal antiinflammatory drugs (NSAIDs) or opioids, should be prescribed cautiously.
 DRUG INTERACTIONS
DRUG INTERACTIONS
Many drug interactions can occur with the anticonvulsants. They may interact with themselves, with each other, or with other drugs. The mechanisms of drug interactions include altering absorption or renal excretion and inducing or inhibiting metabolism. The outcome may alter the levels of the inducing drug itself, another concomitant anticonvulsant, or some other drug that is extensively metabolized by the liver microsomal enzymes.
The most important drug interaction of the anticonvulsants involves stimulation of the hepatic microsomal enzymes. Inducing these enzymes results in a reduction in the blood level of the affected drugs (those metabolized by the liver enzymes). Figure 2-13, A, shows the normal, unaffected enzyme situations. When the enzymes are stimulated (Figure 2-13, B), the level of the affected drug (D) is reduced because it is being metabolized more quickly, producing its metabolite (DM).
Drug interactions with the anticonvulsants are more significant than with other drug groups because of their narrow therapeutic indexes. If the level of an anticonvulsant is altered sufficiently by a drug interaction, either toxicity (level too high) or loss of seizure control (level too low) can result. Before any changes or additions are made to a patient’s therapy, the possibility of drug interactions should be considered.
Idiosyncratic Reactions: A wide range of idiosyncratic reactions occurs with the anticonvulsants. Dermatologic side effects include rash, Stevens-Johnson syndrome, exfoliative dermatitis, and erythema multiforme. Drug-induced systemic lupus erythematosus and hematologic effects have also been reported with most of these agents.
Teratogenicity/Growth: Reports have associated the anticonvulsant agents with alteration in growth, with profound effects on fetal development and children receiving anticonvulsant medications during growth and development. The teratogenic potential of the anticonvulsants has been documented. Several have been implicated in the production of fetal anomalies. However, antiseizure therapy may be necessary. In some instances the mother’s seizures may be more damaging to fetal development than the drug itself. In this case, the risk:benefit factor must be considered.
Withdrawal: Abrupt withdrawal of any anticonvulsant medication can precipitate seizures. Although many patients require medication for life, certain seizure types tend to disappear as the patient grows older. In these patients, gradual withdrawal of their seizure medication under controlled conditions can be undertaken after an appropriate interval of drug use.
Carbamazepine
Structurally related to the tricyclic antidepressants, carbamazepine (kar-ba-MAZ-e-peen) (Tegretol) is used to treat convulsions. It is of special interest in dentistry because of its use in the treatment of trigeminal neuralgia (tic douloureux). It is also indicated in the treatment of bipolar depression. In fact, several anticonvulsants are used to manage chronic pain syndromes.
 PHARMACOLOGIC EFFECTS
PHARMACOLOGIC EFFECTS
Carbamazepine has the following properties: anticonvulsant, anticholinergic, antidepressant, sedative, and muscle relaxant. It also has antiarrhythmic, antidiuretic, and neuromuscular transmission-inhibitory actions. Its mechanism of action involves blocking sodium channels, which block the propagation of nerve impulses. Other effects of carbamazepine include inhibition of high-frequency repetitive firing in neurons and decrease in synaptic transmission presynaptically.
 ADVERSE REACTIONS
ADVERSE REACTIONS
Carbamazepine can have many types of adverse reactions; some are serious, but most patients seem to tolerate the medication well. CNS depression and gastrointestinal tract problems are most common.
Central Nervous System Effects: Carbamazepine can produce dizziness, vertigo, drowsiness, fatigue, ataxia, confusion, headache, nystagmus, and visual (diplopia) and speech disturbances. Activation of a latent psychosis, abnormal involuntary movements, depression, and peripheral neuritis occur rarely.
Gastrointestinal Effects: Gastrointestinal side effects include nausea, vomiting, and gastric distress. Abdominal pain, diarrhea, constipation, and anorexia have also been noted. Taking carbamazepine with food can reduce its chance of producing nausea and vomiting.
Hematologic Effects: Fatal blood dyscrasias, including aplastic anemia and agranulocytosis, have been reported related to carbamazepine therapy. These effects usually occur within 4 months and have been reported in elderly patients taking carbamazepine for trigeminal neuralgia (may be caused by the higher doses used). Thrombocytopenia and leukopenia have also been reported. Because of the hematologic adverse effects, it is necessary to perform laboratory tests to follow these patients. Patients should be made aware of the symptoms of blood dyscrasias and warned to stop the drug and report any of the symptoms immediately. The dental team should observe the oral cavity of patients taking carbamazepine with these side effects in mind (look for petechiae or signs of infection).
Dermatologic Effects: Rashes, urticaria, photosensitivity reactions, and altered skin pigmentation can occur. Erythema multiforme, erythema nodosum, and aggravation of systemic lupus erythematosus have been reported. Alopecia can also occur.
Oral Effects: Dry mouth, glossitis, and stomatitis can sometimes be seen in patients taking carbamazepine. A child who is taking chewable carbamazepine, often four times daily, will be exposed to a sugar for an extended period of time (sticks to teeth). The pediatric dose form of carbamazepine contains 63% sugar in its chewable tablet. The parents should be questioned about the child’s medication use and the oral hygiene methods being used.
Other Effects: Cardiovascular side effects include congestive heart failure and alterations in blood pressure. Abnormal liver function tests and cholestatic jaundice have been reported. Urinary frequency and retention, oliguria, and impotence have been reported with carbamazepine use. Elevated blood urea nitrogen levels, albuminuria, and glycosuria have been seen. Lymphadenopathy, aching joints, and punctate lens opacities have occurred rarely.
 DRUG INTERACTIONS
DRUG INTERACTIONS
Carbamazepine has many drug interactions. Like many of the other anticonvulsants, it can induce liver microsomal enzymes, altering the metabolism of many drugs, including carbamazepine itself. Carbamazepine can decrease the effect of doxycycline, warfarin, theophylline, and oral contraceptives. Carbamazepine’s effects may be increased by erythromycin, isoniazid, propoxyphene, and calcium channel blockers. The dental management of patients taking carbamazepine is discussed in Box 16-3.
Valproate
A group of anticonvulsant agents that are not structurally related to any other anticonvulsants are the valproates, which include valproic (val-PRO-ik) acid, valproate (val-PRO-ate) sodium, and divalproex (dye-VAL-pro-ex) sodium. The term valproate is used here to refer to all of these agents. Divalproex sodium is a 1 : 1 ratio of valproic acid and valproate sodium. The mechanism of action of valproate may be its effect on sodium or potassium channels, a reduction in aspartate levels, or an increase in the inhibitory neurotransmitter γ-aminobutyric acid (GABA) (evidence is mounting against this as the only mechanism).
 ADVERSE REACTIONS
ADVERSE REACTIONS
Gastrointestinal Effects: Indigestion, nausea, and vomiting are the most frequent adverse effects associated with valproate. These can be minimized by giving the drug with meals or increasing the dose very gradually. Divalproex sodium may have fewer adverse gastrointestinal effects than its components. Other gastrointestinal side effects include hypersalivation, anorexia, increased appetite, cramping, diarrhea, and constipation.
Central Nervous System Effects: Sedation and drowsiness have been reported with valproate. Rarely, ataxia, headache, and nystagmus have been noted. Some children exhibit hyperactivity, aggression, and other behavioral disturbances. Weight gain and an increase in appetite have been reported.
Hepatotoxicity: The idiosyncratic toxicity of valproate is hepatotoxicity. Dose-related changes in liver enzymes often occur in these patients. Deaths caused by hepatic failure have also been reported. Because valproic acid can produce serious hepatotoxicity, hepatic function tests should be performed. Signs of hepatotoxicity include nausea, vomiting, abdominal pain, loss of appetite, and diarrhea.
 DRUG INTERACTIONS
DRUG INTERACTIONS
Other drugs that are CNS depressants can have an additive CNS depressant effect when used with valproate. Valproate inhibits the metabolism of phenobarbital, producing excessive sedation. Valproate has also been associated with drug interactions with phenytoin resulting in decreased action of valproate and increased phenytoin action. Because valproate can affect bleeding, other drugs that affect bleeding should be used cautiously. Box 16-4 summarizes the management of dental patients taking valproic acid.
Phenobarbital
The most common barbiturate used in the treatment of epilepsy is phenobarbital (fee-noe-BAR-bi-tal). The barbiturates are discussed in detail in Chapter 11. Primidone (PRYE-mih-done) (Mysoline) differs from phenobarbital by one functional group, and mephobarbital (me-foe-BAR-bi-tal) (Mebaral) is metabolized to phenobarbital in the body, so both have actions similar to phenobarbital. Because these agents are similar in their action, phenobarbital is discussed as the prototype for this group.
Phenobarbital is used alone and in combination with other anticonvulsants such as phenytoin. It is used to treat tonic-clonic and partial seizure types (see Table 16-1). Other anticonvulsants are often used first.
The most common side effect associated with phenobarbital is sedation. With continued use, tolerance to the drowsiness but not the anticonvulsant effect often develops. In children, excitement and hyperactivity are often produced. The elderly sometimes exhibit confusion, excitement, or depression.
Skin reactions occur in 1% to 3% of patients. Rarely, exfoliative dermatitis, erythema multiforme, or Stevens-Johnson syndrome has been reported. Stomatitis may herald the onset of cutaneous reactions, some of which have been fatal. The barbiturates should be discontinued if any skin reactions occur.
Phenytoin
Because phenytoin (FEN-i-toyn) (Dilantin), formerly called diphenylhydantoin, is the most commonly used hydantoin, it is discussed as the prototype for the hydantoin group. Because phenytoin is associated with gingival enlargement, the dental team plays an integral role in the management of these patients.
Phenytoin is used to treat both tonic-clonic and partial seizures with complex symptomatology. It is not useful in the treatment of pure absence seizures but may be used in combination with other agents indicated for absence seizures to control combined seizure types. It has also been used to treat trigeminal neuralgia. In addition to its anticonvulsant properties, phenytoin has quinidine-like antiarrhythmic properties.
 ADVERSE REACTIONS
ADVERSE REACTIONS
The adverse reactions associated with phenytoin are frequent, affect many body systems, and may be serious (rare). Because of phenytoin’s narrow therapeutic index, adverse reactions associated with elevated blood levels can occur. The chance for toxicity is also increased because phenytoin’s metabolism is a saturable process. Phenytoin has a propensity for drug interactions because of its enzyme-stimulating property.
Gastrointestinal Effects: Gastrointestinal adverse reactions are not uncommon. Taking the medication with food can reduce these side effects. Other drugs with the potential for adverse gastrointestinal tract effects, such as NSAIDs or opioids, should be used carefully.
Central Nervous System Effects: The CNS effects that can occur with phenytoin include mental confusion, nystagmus, ataxia, slurred speech, blurred vision, diplopia, amblyopia, dizziness, and insomnia. Because these effects are dose related, they can often be controlled by reducing the dose of phenytoin.
Dermatologic Effects: Skin reactions to phenytoin range from rash to (rarely) exfoliative dermatitis, lupus erythematosus, or Stevens-Johnson syndrome. Some patients experience irreversible hypertrichosis or hirsutism (excessive hairiness) on the trunk and face. This is one reason why alternative drugs are often selected, especially in the young female patient.
Vitamin Deficiency: Deficiency produced by phenytoin may involve vitamin D and folate. Osteomalacia may result from phenytoin’s interference with vitamin D metabolism. The first symptoms of folate deficiency may be oral mucosal changes such as ulcerations or glossitis. Treatment involves administering folic acid.
Teratogenicity/Growth: Fetal hydantoin syndrome is the term given to the congenital abnormality associated with maternal ingestion of phenytoin (Figure 16-3). It includes craniofacial anomalies, microcephaly, nail/digit hypoplasia, limb defects, growth deficiency, and mental retardation. Thickening of facial structures and coarsening of facial features have been noted.
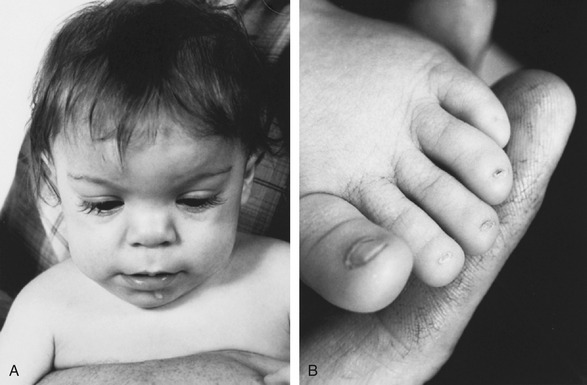
FIGURE 16-3 Infant with fetal hydantoin syndrome. A, Typical facial appearance with broad, depressed nasal bridge and widely spaced eyes. B, Hypoplasia of nails and distal phalanges. (From Bradley WG, Daroff RB, Fenichel GM, et al: Neurology in clinical practice, ed 5, Philadelphia, 2008, Butterworth-Heinemann.)
Another adverse reaction to phenytoin, gingival enlargement (previously referred to as gingival hyperplasia), occurs in approximately 50% of all chronic users (see Color Plates 16 and 17). The name change is the result of an increased understanding of the nature of the enlargement. In approximately 30% of affected patients, gingival enlargement is severe enough to require surgical intervention.
Symptoms: The clinical symptoms that occur with gingival enlargement may appear as little as a few weeks or as long as a few years after initial drug therapy. It often begins as a painless enlargement of the gingival margin. The gingiva is pink and does not bleed easily unless other factors are present. With time, the interproximal papillae become involved and finally coalesce to cover even the occlusal surfaces of the teeth. The hyperplasia is more commonly located in the anterior rather than the posterior surfaces, and the buccal rather than the lingual surfaces. The affected areas of the mouth in order of severity are the maxillary anterior facial, mandibular anterior facial, maxillary posterior facial, and mandibular posterior facial areas. In the affected patient, both normal and abnormal tissue may be found. Edentulous areas are rarely involved.
The better the patient’s oral hygiene, the less likely the lesions are to occur or the less severe they will be if they do occur. Younger patients are more likely to experience this adverse reaction. Controversy exists on the contribution of dose and duration of therapy to the risk for the development of gingival enlargement.
Etiology: The cause of phenytoin gingival enlargement is unknown. Many causes have been investigated, including alteration in the function of the adrenal gland, hypersensitivity or allergic reaction, immunologic reaction, and vitamin C or folate deficiency. Because it is known that phenytoin may be found in the saliva, some investigators suggest a local etiology.
Management: The management of phenytoin-induced gingival enlargement requires consultation between dental personnel and the patient’s physician. Some possible alternatives are as follows:
• Choose another antiepileptic drug. Choosing another effective antiepileptic drug is one method of handling the gingival enlargement produced by phenytoin.
• Discontinue phenytoin. Patients who have discontinued phenytoin will experience a decrease in gingival enlargement over a 1-year period. Surgical intervention should wait until at least 18 months after cessation of therapy because some patients experience additional reduction in the enlargement after the 1-year period.
• Improve oral hygiene. Scrupulous oral hygiene may delay the onset or reduce the rate of formation of enlargement. Avoiding irritating restorations may also reduce enlargement. Even with ideal oral hygiene, enlargement is not always totally preventable, and once it has formed is not easily reversed.
• Consider gingivectomy. When gingival enlargement interferes with plaque control, esthetics, or mastication and when oral hygiene has not been successful in controlling enlargement, surgical elimination is indicated. It is not a permanent solution because if the patient continues on phenytoin, enlargement quickly returns in most cases and can progress to the presurgical level in a short period.
• Consider other drugs. Although many types of drugs, such as diuretics, corticosteroids, mouthwashes, vitamin C, folic acid, and antihistamines, have been tried in the treatment of this condition, none has been shown to be effective in controlled trials.
Box 16-5 summarizes the management of dental patients taking phenytoin.
Miscellaneous Anticonvulsant Agents
The drug of choice for the treatment of absence seizures (see Table 16-1) is ethosuximide (eth-oh-SUX-i-mide) (Zarontin). Its mechanism of action may involve inhibiting the T-type calcium channels. It is ineffective in partial seizures with complex symptoms or in tonic-clonic seizures. In the treatment of mixed seizures, agents effective against tonic-clonic seizures must be used in addition to ethosuximide.
Gastrointestinal adverse effects include anorexia, gastric upset, cramps, pain, diarrhea, and nausea and vomiting. CNS adverse effects include drowsiness, hyperactivity, headache, and hiccups. Ethosuximide has been associated with blood dyscrasias, a positive direct Coombs’ test, systemic lupus erythematosus, Stevens-Johnson syndrome, and hirsutism. Oral effects reported with ethosuximide include gingival enlargement and swelling of the tongue.
 BENZODIAZEPINES
BENZODIAZEPINES
Benzodiazepines, such as clonazepam (kloe-NA-ze-pam) (Klonopin) and clorazepate (klor-AZ-e-pate) (Tranxene), are used orally as anticonvulsant adjuvants. Diazepam (Valium), lorazepam (Ativan), and midazolam (Versed) are used parenterally to treat recurrent tonic-clonic seizures or status epilepticus.
Clonazepam is used as an adjunct to treat absence seizures not responsive to ethosuximide. Drowsiness and ataxia occur often. Behavioral disturbances and adverse neurologic effects can occur. Other side effects reported relate to the gastrointestinal tract and to the dermatologic and hematologic systems. Oral manifestations include increased salivation, coated tongue, dry mouth, and sore gums. It is also used as an adjunct in the treatment of certain mental illnesses.
New Anticonvulsant Agents
The newer anticonvulsants are listed in Table 16-2. Because gabapentin is gaining in popularity, it is discussed in more depth.
TABLE 16-2
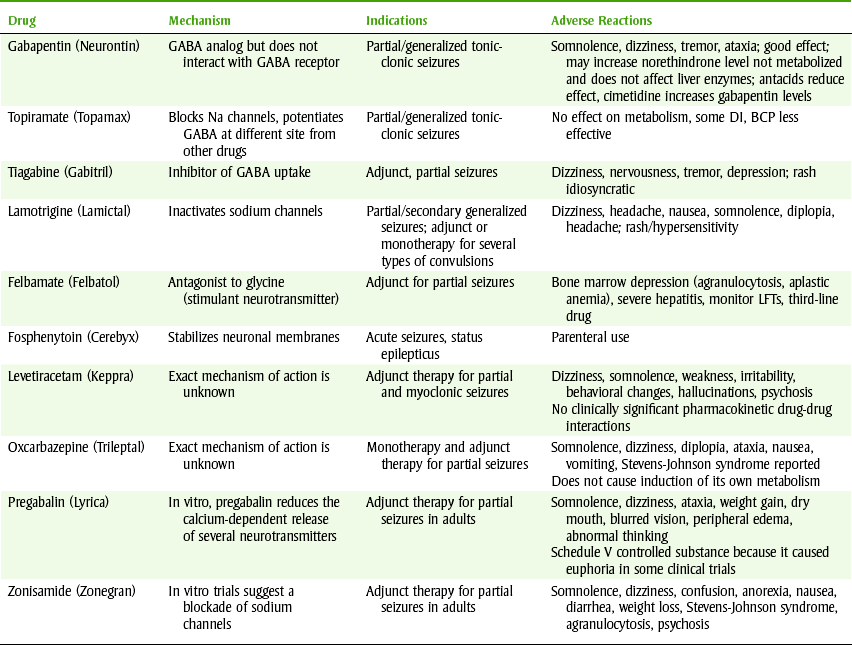
BCP, Birth control pill; DI, drug interaction; GABA, γ-aminobutyric acid; GE, gingival enlargement; LFTs, liver function tests; Na, sodium.
 FELBAMATE
FELBAMATE
Felbamate (fel-BA-mate) (Felbatol) is a newer anticonvulsant that has been associated with serious toxicities. Aplastic anemia and acute hepatic failure have been reported. The risk of death as a result of aplastic anemia depends on the severity and etiology of the anemia, with an estimated fatality rate of 20% to 30%. This agent should be reserved for use only if the seizures are refractory to other anticonvulsant agents. Prescribers are aware of these risks and patients must sign a patient information/consent form before they can receive the medication.
 GABAPENTIN
GABAPENTIN
Gabapentin (GA-ba-pen-tin) (Neurontin), an analog of GABA, is effective as an adjunct against partial and generalized tonic-clonic seizures. Its mechanism of action is unknown, but it is not a GABA agonist. Like other anticonvulsants, it can cause CNS effects such as somnolence, dizziness, tremor, and ataxia. Gastric complaints include dyspepsia, nausea, and vomiting. It can increase the blood pressure and produce edema. There is an increase in rhinitis, pharyngitis, and cough. Ophthalmic adverse reactions include diplopia (6%) and amblyopia (4%). Myalgia and back pain have been reported. Hypersensitivity reactions have included skin rash and pruritus. Oral manifestations of gabapentin include mucositis, hiccups, and nasal obstruction.
One major advantage of gabapentin over the other anticonvulsant agents is that it is not metabolized. Because it does not affect the hepatic microsomal enzymes, it lacks significant drug interactions, which gives it a distinct advantage over the other anticonvulsants.
DENTAL TREATMENT OF THE PATIENT WITH EPILEPSY
The dental team should not treat a patient who has a history of seizure disorders without reviewing the management of the patient with epilepsy, including the procedures for handling a patient experiencing tonic-clonic seizures. Preventive measures include a detailed seizure history, treatment planning to avoid excessive stress and missed medications, and education of the entire dental office staff. The management of the patient experiencing tonic-clonic seizures should include moving the patient to the floor if possible, tilting the patient’s head to one side to prevent aspiration, and removing objects from the patient’s mouth before the seizure to prevent fractured teeth. The use of tongue blades is not recommended because the blades may split and produce additional trauma in the oral cavity.
NONSEIZURE USES OF ANTICONVULSANTS
Several anticonvulsants are used to manage chronic pain syndromes. An example is carbamazepine, which is used to treat trigeminal neuralgia and atypical facial pain. Phenytoin has also been used to treat neurologic pain. Valproic acid has been used for migraine headache prophylaxis.
Psychiatric Use
Carbamazepine, valproic acid, clonazepam, and gabapentin have been used in the treatment of certain mental disorders (see Chapter 17). They are sometimes called mood stabilizers in this context. They can be used to “level out,” or stabilize, the mood in patients with bipolar disorder. Thus a patient taking an anticonvulsant drug may or may not have a seizure disorder.
CLINICAL SKILLS ASSESSMENT
1. What is gingival hyperplasia and how can it be treated?
2. Discuss methods to minimize gingival hyperplasia.
3. What is the mechanism of action of phenytoin and what are its clinical uses?
4. What is hirsutism? Why would this be a concern among young women?
5. What are some of the gastrointestinal adverse reactions associated with phenytoin?
6. What should a patient be told about the use of an NSAID and phenytoin?
7. What are the dental concerns associated with the CNS adverse reactions of phenytoin?
8. What is carbamazepine and what is its role in treating seizure disorders?
9. What are the major classes of adverse reactions associated with carbamazepine therapy?
10. Can any of the CNS adverse reactions affect oral health care?
11. Why should a parent be concerned about the pediatric dose form of carbamazepine?
12. What would you tell a parent about the pediatric dose form of carbamazepine?
13. What are the dental concerns associated with carbamazepine and what should a parent be told about them?
![]() Please visit http://evolve.elsevier.com/Haveles/pharmacology for review questions and additional practice and reference materials.
Please visit http://evolve.elsevier.com/Haveles/pharmacology for review questions and additional practice and reference materials.