Chapter 7 Antimicrobial chemotherapy
Antimicrobial compounds include antibacterial, antiviral, antifungal and antiprotozoal agents. All of these, apart from the last group, are prescribed in dentistry.
All antimicrobials demonstrate selective toxicity; i.e. the drug can be administered to humans with reasonable safety while having a marked lethal or toxic effect on specific microbes. The corollary of this is that all antimicrobials have adverse effects on humans and should therefore be used rationally and only when required.
Antimicrobial therapy aims to treat infection with a drug to which the causative organism is sensitive. Antimicrobials can be administered on a ‘best-guess’ basis, with a sound knowledge of the:
This is called empirical antibiotic therapy and contrasts with rational antibiotic therapy in which antibiotics are administered after the sensitivity of the pathogen has been established by culture and in vitro testing in the laboratory. In general, empirical therapy is undertaken in the majority of situations encountered in dentistry.
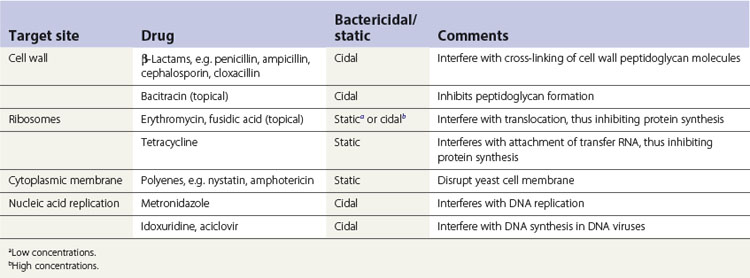
Bacteriostatic and bactericidal antimicrobial agents
Antimicrobial agents are classically divisible into two major groups: bactericidal agents, which kill bacteria; and bacteriostatic agents, which inhibit multiplication without actually killing the pathogen. However, the distinction is rather hazy and is dependent on factors such as the concentration of the drug (e.g. erythromycin is bacteriostatic at low concentrations and bactericidal at high concentrations), the pathogen in question and the severity of infection. Further, host defence mechanisms play a major role in the eradication of pathogens from the body, and it is not essential to use bactericidal drugs to treat most infections. A bacteriostatic drug that arrests the multiplication of pathogens and so tips the balance in favour of the host defence mechanisms is satisfactory in many situations.
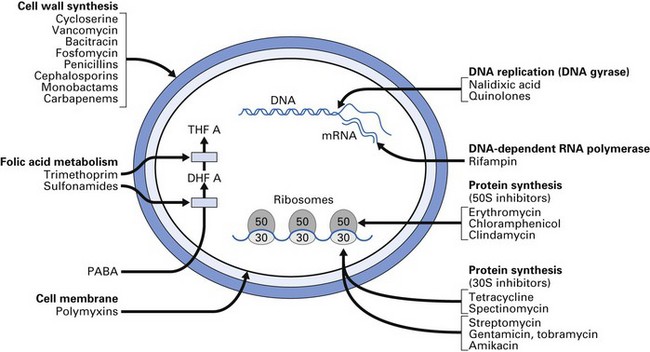
Mode of action of antimicrobials
Antimicrobial agents inhibit the growth of or kill microorganisms by a variety of mechanisms. In general, however, one or more of the following target sites are involved:
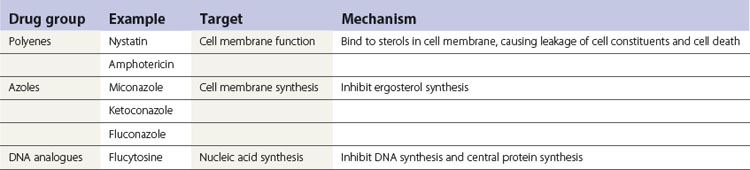
A summary of the mode of action of commonly used antimicrobials is given in Table 7.1 and Figure 7.1.
Principles of antimicrobial therapy
Antimicrobial agents should be prescribed on a rational clinical and microbiological basis. In general, therapy should be considered for patients when one or more of the following conditions are present:
(Note: this is not an exhaustive list.)
Choice of drug
The choice of drug is strictly dependent upon the nature of the infecting organisms and their sensitivity patterns. However, in a clinical emergency such as septicaemia or Ludwig’s angina, antimicrobial agents must be prescribed empirically until laboratory tests are completed. In general, another antimicrobial drug should be prescribed if the patient has had penicillin within the previous month because of the possible presence of penicillin-resistant bacterial populations previously exposed to the drug.
Spectrum of activity of antimicrobial agents
Antimicrobial agents can be categorized as broad-spectrum and narrow-spectrum antibiotics, depending on their activity against a range of Gram-positive and Gram-negative bacteria. For example, penicillin is a narrow-spectrum antibiotic with activity mainly against the Gram-positive bacteria, as is metronidazole, which acts almost entirely against strict anaerobes and some protozoa.
Broad-spectrum antimicrobials (e.g. tetracyclines, ampicillins) are active against many Gram-positive and Gram-negative bacteria, and they are often used for empirical or ‘blind’ treatment of infections when the likely causative pathogen is unknown. This unfortunately leads to ‘abuse’ of broad-spectrum agents, with the consequent emergence of resistance in organisms that were originally sensitive to the drug. The spectrum of activity of some broad-spectrum and narrow-spectrum antimicrobial agents is shown in Table 7.2.
Table 7.2 Spectrum of activity of some commonly used antimicrobial agents
| Drug | Spectrum |
|---|---|
| Phenoxymethylpenicillin (penicillin V) | 1. Aerobic Gram-positives (e.g. streptococci, pneumococci, β-lactamase-negative) |
| 2. Anaerobic Gram-positives (e.g. anaerobic streptococci) | |
| 3. Anaerobic Gram-negatives (e.g. most Bacteroides, fusobacteria, Veillonella) | |
| Penicillinase-resistant penicillins (e.g. flucloxacillin) | All the above, including β-lactamase-producing staphylococci |
| Ampicillin | As for penicillin, also includes Haemophilus spp. |
| Cephalosporins | As for penicillin, also includes some coliforms |
| Erythromycin | Gram-positives mainly but some anaerobes not susceptible at levels obtained by oral administration |
| Tetracycline | Broad-spectrum. Many Gram-positives and -negatives |
| Metronidazole | All strict anaerobes are sensitive, including some protozoa. Of questionable value for facultative anaerobes |
Combination therapy
Whenever possible, a single antimicrobial agent should be used to reduce the:
However, there are certain clinical situations where a combination of drugs is valuable: for example, to achieve a high bactericidal level when treating patients with infective endocarditis; the use of gentamicin and metronidazole in the empirical treatment of a patient with serious abdominal sepsis; and combination therapy in the management of tuberculosis. In dentistry, combination therapy should be avoided as far as possible.
Antimicrobial prophylaxis
Antimicrobial prophylaxis is the use of a drug to prevent colonization or multiplication of microorganisms in a susceptible host. The value of prophylaxis depends upon a balance between:
When used appropriately, prophylaxis can reduce morbidity and the cost of medical care. Irrational prophylaxis leads to a false sense of security, increased treatment cost and the possible emergence of resistant flora.
Aims
The aims of antimicrobial prophylaxis are:
In dentistry, antibiotics are used as prophylactic agents before dental or surgical treatment of patients who:
However, the advantages and disadvantages of prophylactic antimicrobial therapy in the latter three groups should be carefully weighed as some consider this superfluous.
Prescribing an antimicrobial agent
The following should be considered before any antimicrobial agent is prescribed.
Is there an infective aetiology?
When there is no good clinical evidence of infection, antimicrobial therapy is unnecessary, except in prophylaxis (discussed above).
Have relevant specimens been taken before treatment?
Appropriate specimens should be collected before drug therapy is begun as the population of pathogens may be reduced, and therefore less easily isolated, if specimens are collected after antimicrobial agents have been taken. Further, the earlier the specimens are taken, the more likely it is that the results will be useful for patient management.
When should the treatment be started?
In patients with life-threatening infections, e.g. Ludwig’s angina, intravenous therapy should generally be instituted immediately after specimen collection. Antimicrobial therapy may be withheld in chronic infections until laboratory results are available (e.g. actinomycosis).
Which antimicrobial agent?
Consider the pharmacodynamic effects, including toxicity, when choosing a drug from a number of similar antimicrobial agents that are available to treat many infections (see below). An adequate medical history, especially in relation to past allergies and toxic effects, should be taken before deciding on therapy.
Pharmacodynamics of antimicrobials
Dosage
Antimicrobial agents should be given in therapeutic doses sufficient to produce a tissue concentration greater than that required to kill or inhibit the growth of the causative microorganism(s).
Duration of treatment
Ideally, treatment should continue for long enough to eliminate all or nearly all of the pathogens, as the remainder will, in most instances, be destroyed by the host defences. Conventionally, this cannot be precisely timed, and standard regimens last for some 3–5 days, depending on the drug. However, a short-course, high-dose therapy of certain antibiotics such as amoxicillin is as effective as a conventional 5-day course. The other advantages of short courses of antimicrobial agents are good patient compliance and minimal disturbance to commensal flora, leading to an associated reduction in side effects such as diarrhoea.
Route of administration
In seriously ill patients, drugs should be given by the parenteral route to overcome problems of absorption from the intestinal tract. All antimicrobial agents given by mouth must be acid-stable.
Distribution
The drug must reach adequate concentrations at the infective focus. Some antibiotics, such as clindamycin, that penetrate well into bone are preferred in chronic bone infections; in meningitis, a drug that penetrates the cerebrospinal fluid should be given.
Excretion
The pathway of excretion of an antimicrobial agent should be noted. For example, drugs metabolized in the liver, such as erythromycin estolate, should not be given to patients with a history of liver disease because they may cause hepatotoxicity, leading to jaundice.
Toxicity
Most antimicrobials have side effects and the clinician should be aware of these (for examples, see the following section on antibacterial agents).
Drug interactions
Drug interactions are becoming increasingly common owing to the extensive use of a variety of drugs. For instance, antibiotics such as penicillin and erythromycin can significantly reduce the efficacy of some oral contraceptives, and antacids can interfere with the action of tetracyclines. All clinicians should therefore be aware of the drug interactions of any antimicrobial they prescribe. The major drug interactions of antimicrobials commonly used in dentistry are given in Table 7.3.
Table 7.3 Some drug interactions of antimicrobials commonly used in dentistry
| Drug affected | Drug interacting | Effect |
|---|---|---|
| Penicillins | Probenecid, neomycin | May potentiate the effect of penicillin. Reduced absorption |
| Erythromycin | Theophylline | Increase theophylline levels, leading to potential toxicity |
| Cephalosporins | Gentamicin | Additive effect leading to nephrotoxicity |
| Furosemide (Lasix) | Possible increase in nephrotoxicity | |
| Tetracycline | Antacids, dairy products, oral iron, zinc sulphate | Reduced absorption |
| Metronidazole | Alcohol | ‘Antabuse’ effect |
| Disulfiram, phenobarbital, phenytoin | Reduced effect |
Failure of antimicrobial therapy
Consideration should be given to the following potential problems if an infection does not respond to drugs within 48 h:
Antibiotic resistance in bacteria
Emergence of drug resistance in bacteria is a major problem in antibiotic therapy and depends on the organism and the antibiotic concerned. Whereas some bacteria rapidly acquire resistance (e.g. Staphylococcus aureus), others rarely do so (e.g. Streptococcus pyogenes). Resistance to some antibiotics is virtually unknown (e.g. metronidazole), but strains resistant to others (e.g. penicillin) readily emerge.
Antibiotic resistance develops when progeny of resistant bacteria emerge. As they will be at a selective advantage over their sensitive counterparts, and as long as the original antibiotic is prescribed, the resistant strains can multiply uninhibitedly (e.g. hospital staphylococci with almost universal resistance to penicillin). Such antibiotic resistance can be divided into:
Mechanisms of antibiotic resistance (Table 7.4)
Inactivation of the drug
This is very common, e.g. production of β-lactamase by staphylococci. The enzyme, which is plasmid coded, destroys the β-lactam ring responsible for the antibacterial activity of penicillins.
Table 7.4 Plasmid-mediated antibiotic resistance
| Antibiotic | Mechanism of resistance |
|---|---|
| β-Lactams | β-Lactamase breaks down the β-lactam ring to an inactive form |
| Aminoglycosides | Modifying enzymes cause acetylation, adenylation, phosphorylation |
| Chloramphenicol | Acetylation of the antibiotic to an inactive form |
| Erythromycin, clindamycin | Methylation of ribosomal RNA prevents antibiotic binding to ribosomes |
| Sulphonamides, tetracycline | Alteration of cell membrane decreases permeability to the antibiotic |
Altered uptake
The amount of drug that reaches the target is either reduced or completely inhibited (e.g. tetracycline resistance in Pseudomonas aeruginosa). This can be either due to altered permeability of the cell wall or to pumping of the drug out of the cell (efflux mechanism).
Emergence of drug-resistant bacteria and the role of the clinician
Emergence of antibiotic-resistant organisms is now a worldwide concern. This is accentuated by the extremely slow discovery of new antimicrobial agents due to the associated massive research and developmental costs, difficulties in conducting extensive clinical trials and a litigious society. Hence, all who prescribe antibiotics should be aware of general principles of minimizing the emergence of drug resistance, which include:
Antimicrobials commonly used in dentistry
Although a large array of antimicrobial agents have been described and are available to medical practitioners, only a limited number of these are widely prescribed by dental practitioners. The following therefore is an outline of the major antimicrobials (antibacterials, antifungals and antivirals) used in dentistry.
Antibacterial agents
Penicillins
Penicillins are the most useful and widely used antimicrobial agents in dentistry. A wide array of penicillins have been synthesized by incorporating various side chains into the β-lactam ring (Table 7.5). The spectrum of activity and indications for the use of these penicillins vary widely. The more commonly used penicillins such as phenoxymethylpenicillin (penicillin V) are described below in some detail. Others, such as the carboxypenicillins (carbenicillin and ticarcillin) and ureidopenicillins (azlocillin and piperacillin), which are active against Gram-negative organisms, are rarely used in dentistry, except for amoxicillin.
| Group | Type of penicillin |
|---|---|
| Narrow-spectrum | Benzylpenicillin |
| Phenoxymethylpenicillin | |
| Procaine penicillin | |
| Benzathine penicillin | |
| Broad-spectrum | Ampicillin |
| Amoxicillin | |
| Esters of ampicillin | |
| Penicillinase-resistant | Methicillin |
| Flucloxacillin | |
| Antipseudomonal | Piperacillin |
| Mezlocillin |
The commonly used penicillins are remarkably non-toxic but all share the problem of allergy. Minor reactions such as rashes are common, while severe reactions, especially anaphylaxis, although rare, can be fatal. Allergy to one penicillin is shared by all the penicillins and, in general, the drug should not be given to a patient who has had a reaction to any member of this group. Some 10% of patients sensitive to penicillin show cross-reactivity to cephalosporins.
Phenoxymethylpenicillin (penicillin V)
Mode of action
Bactericidal; inhibits cell wall synthesis by inactivating the enzyme transpeptidase, which is responsible for cross-linking the peptidoglycan cross walls of bacteria; an intact β-lactam ring is crucial for its activity.
Spectrum of activity
Effective against a majority of α-haemolytic streptococci and penicillinase-negative staphylococci. Aerobic Gram-positive organisms, including Actinomyces, Eubacterium, Bifidobacterium and Peptostreptococcus spp. are sensitive, together with anaerobic Gram-negative organisms such as Bacteroides, Prevotella, Porphyromonas, Fusobacterium and Veillonella species. The majority of S. aureus strains, particularly those from hospitals, are penicillinase producers and hence resistant to penicillin. (A small minority of α-haemolytic streptococci, and some Aggregatibacter actinomycetemcomitans strains implicated in aggressive periodontitis, are resistant.)
Resistance
Very common, owing to the β-lactamase produced by bacteria, which inactivates the drug by acting on the β-lactam ring.
Indications
As this drug can be administered orally, it is commonly used by dental practitioners in the treatment of acute purulent infections, post-extraction infection, pericoronitis and salivary gland infections.
Pharmacodynamics
Phenoxymethylpenicillin is less active than parenteral benzylpenicillin (penicillin G) because of its erratic absorption from the gastrointestinal tract. Therefore, in serious infections, phenoxymethylpenicillin could be used for continuing treatment after one or more loading doses of benzylpenicillin, when clinical response has begun.
Benzylpenicillin (penicillin G)
Broad-spectrum penicillins susceptible to staphylococcal penicillinase: ampicillin and amoxicillin
Spectrum of activity
Similar to penicillin but effective against a broader spectrum of organisms, including Gram-negative organisms such as Haemophilus and Proteus spp. Amoxicillin and ampicillin have similar antibacterial spectra.
Resistance
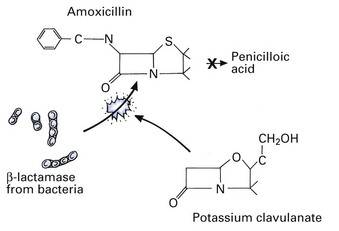
One drawback of amoxicillin is its susceptibility to β-lactamase, but if potassium clavulanate is incorporated with amoxicillin, the combination (co-amoxiclav) is resistant to the activity of β-lactamase (Fig. 7.2).
Indications
Ampicillin is sometimes used in the empirical treatment of dentoalveolar infections when the antibiotic sensitivity patterns of the causative organisms are unknown. In dentistry, amoxicillin is the drug of choice for prophylaxis of infective endocarditis in a restricted group of patients undergoing surgical procedures and scaling (see Chapter 24). A short course of high-dose amoxicillin (oral) has been shown to be of value in the treatment of dentoalveolar infections.
Toxicity
Associated with a higher incidence of drug rashes than penicillin, and hence should not be administered to patients with infectious mononucleosis (glandular fever) or lymphocytic leukaemia (because of the probability of a drug rash). Nausea and diarrhoea are frequent, particularly on prolonged administration; superinfection and colonization with ampicillin-resistant bacteria, such as coliforms and fungi, may also occur. The incidence of diarrhoea is less with amoxicillin.
Isoxazolyl penicillins: methicillin, cloxacillin and flucloxacillin
Spectrum of activity
Narrow-spectrum antistaphylococcal penicillins relatively resistant to β-lactamase produced by S. aureus.
Cephalosporins, cephamycins and other β-lactams
This group of drugs now includes more than 30 different agents and newer agents are being manufactured each year. All cephalosporins are β-lactams similar to penicillin but are relatively stable to staphylococcal penicillinase; the degree of stability varies with different cephalosporins. The group includes cephalosporins (cefotaxime, cefuroxime, cephalexin and cephradine), cephamycins (cefoxitin), monobactams (aztreonam) and carbapenems (imipenem and meropenem).
Administration
Cephradine and cephalexin, which can be given by mouth, and cephaloridine belong to the first generation of cephalosporins and are used in dentistry. The vast majority of cephalosporins are given parenterally; hence, they are virtually restricted to hospital use.
Spectrum of activity
Broad-spectrum; active against both Gram-positive and Gram-negative bacteria, although individual agents have differing activity against certain organisms.
Indications
Few absolute indications. In dentistry, cephalosporins should be resorted to as a second line of defence, depending on culture and antibiotic sensitivity test results.
Toxicity
Some 10% of penicillin-sensitive patients demonstrate cross-sensitivity; allergic reactions, including urticaria and rashes; possibly nephrotoxicity. Another disadvantage is that oral bacteria, including streptococci, may develop cross-resistance to both penicillins and cephalosporins. Hence, cephalosporins are not suitable alternatives for a patient who has recently had penicillin.
Erythromycin
The most popular member of the macrolide group of antibiotics.
Spectrum of activity
Similar, though not identical, to that of penicillin and thus the first choice in dentistry for treating penicillin-allergic patients. In addition, Haemophilus influenzae and Bacteroides, Prevotella and Porphyromonas spp. are sensitive. Erythromycin has the added advantage of being active against β-lactamase-producing bacteria. Not usually used as a first-line drug in oral and dental infections because obligate anaerobes are not particularly sensitive.
Clindamycin
Spectrum of activity
Similar to that of erythromycin (with which there is partial cross-resistance) and benzylpenicillin; in addition, it is active against Bacteroides spp.
Indications
Mainly reserved, as a single dose, for prophylaxis of infective endocarditis in patients allergic to penicillin; particularly effective in penetrating poorly vascularized bone and connective tissue.
Toxicity
Mild diarrhoea is common. Although rare, the most serious side effect of clindamycin, which can sometimes be fatal, is pseudomembranous (antibiotic-associated) colitis, especially in the elderly and in combination with other drugs. The colitis is due to a toxin produced by Clostridium difficile, an anaerobe resistant to clindamycin. Allergy to these drugs is extremely rare, and hypersensitivity to penicillin is not shared by them.
Tetracyclines
Formerly one of the most widely used antibiotic groups owing to their very broad spectrum of activity and infrequent side effects. Their usefulness has decreased as a result of increasing bacterial resistance. They remain, however, the treatment of choice for infections caused by intracellular organisms such as chlamydiae, rickettsiae and mycoplasmas, as they penetrate macrophages well. A range of tetracyclines is available, although tetracycline itself remains the most useful for dental purposes.
Spectrum of activity
Have a wide spectrum of activity against oral flora, including Actinomyces, Bacteroides, Propionibacterium, Aggregatibacter, Eubacterium and Peptococcus spp.
Indications
In dentistry, tetracyclines are used with some success as adjunctive treatment in localized aggressive periodontitis (formerly localized juvenile periodontitis); they are effective against many organisms associated with these diseases (see Chapter 33). They are also useful as mouthwashes to alleviate secondary bacterial infection associated with extensive oral ulceration, especially in compromised patients.
Pharmacokinetics
Widely distributed in body tissues, and incorporated in bone and developing teeth (Fig. 7.3); particularly concentrated in gingival fluid. Absorption of oral tetracycline is decreased by antacids, calcium, iron and magnesium salts.
Toxicity
Because of the deposition of tetracycline within developing teeth, its use should be avoided in children up to 8 years of age and in pregnant or lactating women; otherwise, unsightly tooth staining may occur. Diarrhoea and nausea may occur after oral administration, as a result of disturbance to bowel flora. However, when reduced dosages are used, even for prolonged periods (e.g. for acne), few side effects are apparent. Serious hepatotoxicity may occur with excessive intravenous dosage.
Metronidazole
The exquisite anaerobic activity of this drug, which was first introduced to treat protozoal infections, makes it exceedingly effective against strict anaerobes and some protozoa.
Mode of action
Bactericidal; it is converted by anaerobic bacteria into a reduced, active metabolite, which inhibits DNA synthesis.
Spectrum of activity
Active against almost all strict anaerobes, including Bacteroides spp., fusobacteria, eubacteria, peptostreptococci and clostridia.
Indications
The drug of choice in the treatment of acute necrotizing ulcerative gingivitis; also used, either alone or in combination with penicillin, in the management of dentoalveolar infections.
Pharmacokinetics
Well absorbed after oral (or rectal) administration; widely distributed and passes readily into most tissues, including abscesses, and crosses the blood–brain barrier into cerebrospinal fluid. The drug is metabolized in the liver.
Toxicity
Minor side effects of metronidazole include gastrointestinal upset, transient rashes and metallic taste in the mouth. Metronidazole interferes with alcohol metabolism and, if taken with alcohol, may cause severe nausea, flushing and palpitations (disulfiram-type effect). It potentiates the effect of anticoagulants and, if used for more than a week, peripheral neuropathy may develop, notably in patients with liver disease; allergenicity is very low.
Sulphonamides and trimethoprim
These drugs interfere with successive steps in the synthesis of folic acid (an essential ingredient for DNA and RNA synthesis). They are widely used in combination because of in vitro evidence of synergism.
Fusidic acid
A narrow-spectrum antibiotic with main activity against Gram-positive bacteria, particularly S. aureus. Angular cheilitis associated with S. aureus is a specific indication for the use of fusidic acid in the form of a topical cream. A small percentage of S. aureus strains show resistance to fusidic acid.
Antifungal agents
In contrast to the wide range of antibacterial agents, the number of effective antifungals is limited. This is because selective toxicity is much more difficult to achieve in eukaryotic fungal cells, which share similar features with human eukaryotic cells. Polyenes and the azoles are the most commonly used antifungals in dentistry. Nystatin and amphotericin are polyene derivatives; miconazole and fluconazole are two examples of a variety of azole antifungals currently available (Table 7.6).
Polyenes
Nystatin
Administration
Too toxic for systemic use; not absorbed from the alimentary canal and hence used to prevent or treat mucosal candidiasis; it is available in the form of pastilles, ready-mixed suspensions, ointments and powder.
Mode of action
Polyene binds to the cytoplasmic membrane of fungi, altering cell wall permeability, with resultant leakage of cell contents and death; in very low doses, it is fungistatic.
Amphotericin
Amphotericin is the other polyene group antifungal. It is used essentially in the same way as nystatin; lozenges, ointment and oral suspensions are available. As with nystatin, its absorption from the gut is minimal on topical administration. Amphotericin is the drug of choice for the treatment of systemic candidiases and other exotic mycoses (e.g. histoplasmosis, coccidioidomycosis).
Azoles
Miconazole
Mode of action
This drug, like other imidazoles, acts by interfering with the synthesis of chemicals needed to form the plasma membrane of fungi, resulting in leakage of cell contents and death.
Fluconazole
Fluconazole is a triazole drug that is highly popular because of its wide spectrum of activity on yeasts and other fungi. Specifically used to prevent Candida infection in HIV-infected individuals as intermittent or continuous therapy.
Administration
Oral; because of its long half-life, it is administered once a day, so patient compliance is good.
Itraconazole
Another azole with properties similar to fluconazole; useful for candidiasis in HIV infection.
New antifungal agents
Echinocandins
A new class of antifungals that disrupts cell wall integrity by inhibiting cell wall polysaccharide. The intravenous agent, capsofungin, available commercially, belongs to this group and is effective against systemic candidiasis and invasive aspergillosis; no specific role in dentistry.
Antiviral agents
Few antiviral drugs with proven clinical efficacy are available, in contrast to the great range of successful antibacterial agents. The shortage of antivirals is mainly due to the difficulty of interfering with the viral activity within the cell without damaging the host. Most antiviral agents achieve maximum benefit if given early in the disease. Immunocompromised patients with viral infections generally benefit from active antiviral therapy, as these infections may spread locally and systemically.
Other problems associated with the therapy of virus infections are:
Aciclovir is the major antiviral drug prescribed in dentistry.
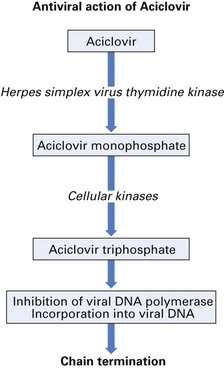
Aciclovir
Aciclovir is an efficient, highly selective antiviral agent useful in the treatment of primary as well as secondary herpetic stomatitis and herpes labialis.
Mode of action
Aciclovir blocks viral DNA production at a concentration of some thousand times less than that required to inhibit host cell DNA production (Fig. 7.4).
Indications
Topical aciclovir (5% cream) can be prescribed for recurrent herpetic ulcers; primary herpetic gingivostomatitis can be treated with either aciclovir cream or tablets. Treatment must be started in the prodromal phase (when there is a local tingling or burning sensation). Application at later stages of infection will reduce the length, discomfort and the viral shedding period correspondingly. Aciclovir tablets or oral suspension may be given for severe herpetic stomatitis or herpes zoster.
An alternative agent for herpetic ulcerations is penciclovir cream.
Key facts
Brook I., Lewis M.A.O., Sandor G.K.B., Jeffcoat M., Samaranayake L.P., Rojas V.R. Clindamycin in dentistry: More than just effective prophylaxis for endocarditis? Oral Surgery, Oral Medicine, Oral Pathology, Oral Radiology and Endodontics. 2005;100:550-558.
Ellepola A.N.B., Samaranayake L.P. Oral candidal infections and antimycotics. Critical Reviews in Oral Biology and Medicine. 2000;11:172-198.
Levy S.B. The antibiotic paradox. How miracle drugs are destroying the miracle. New York: Plenum; 1992.
Mims C., Playfair J., Roitt I., Wakelin D., Williams R. Antimicrobial agents and chemotherapy, Ch. 30. Medical microbiology, 2nd ed. London: Mosby. 1998.
O’Grady F., Lambert H.P., Finch R.G., Greenwood D. Antibiotics and chemotherapy: Their use in therapy, 7th ed. Edinburgh: Churchill Livingstone; 1997.
Samaranayake L.P., Johnson N. Guidelines for the use of antimicrobial agents to minimise the development of resistance. International Dental Journal. 1999;49:189-195.
Review questions (answers on p. 351)
Please indicate which answers are true, and which are false.