Chapter 33 Microbiology of periodontal disease
Periodontal diseases can be defined as disorders of supporting structures of the teeth, including the gingivae, periodontal ligament and supporting alveolar bone. Everyone suffers from various degrees of periodontal disease at some point, and it is one of the major diseases afflicting humankind. However, in most people, the common chronic inflammatory diseases involving the periodontal tissues can be controlled, using mechanical cleansing techniques and good oral hygiene. A minority experience rapid progressive disease that requires assessment and management by periodontists.
Classification of periodontal disease
Periodontal disease can be broadly categorized into gingivitis and periodontitis. These are yet again subdivided into numerous categories; a recent classification of periodontal diseases is given in Table 33.1. It should be noted that there is no universally acknowledged classification of periodontal disease and the clinical descriptors used relate to:
Table 33.1 Classification of periodontal diseases
| Gingival diseases |
|
A Dental plaque-induced gingival diseases
|
| Periodontal diseases |
Periodontitis usually develops from a pre-existing gingivitis; however, not every case of gingivitis develops into periodontitis.
Ecology of the gingival crevice and the periodontal pocket
The healthy gingival crevice is a unique environment created by a mineralized structure, the tooth, that is part embedded in the connective tissue and part exposed to the oral environment. The gingival crevice is more anaerobic than most locales of the mouth and is constantly bathed by the gingival crevicular fluid (GCF) and its humoural and cellular defence factors, including polymorphs. Dramatic changes ensue during the transition of the crevice into a periodontal pocket. The oxygen tension or Eh falls further and becomes highly anaerobic and the flow of GCF increases. The mostly proteolytic bacteria living in the periodontal pocket raise the pH to alkaline levels (pH 7.4–7.8; compared with neutral values in health), which in turn promotes the growth of bacteria such as Porphyromonas gingivalis.
The exposed cemental surface of the tooth is first colonized mainly by pioneer dwellers, including streptococci and Actinomyces spp. Secondary colonizers such as Prevotella and Porphyromonas spp. can adhere to this layer of cells by coaggregation. Others, such as Peptostreptococcus micros, can adhere to the crevicular epithelium. Thus, the inhabitants and the ecology of a deep periodontal pocket are markedly different from that of the gingival crevice.
Aetiological factors
The main aetiological agent of periodontal disease is microflora inhabiting subgingival plaque biofilms.
However, the host tissues and its specific and non-specific host defence mechanisms play crucial modulating roles (i.e. modifying factors) in the disease process. The latter will be described first.
Host tissues
The periodontium comprises the gingivae, periodontal ligament, cementum and alveolar bone (Fig. 33.1). Although the dentogingival junction is perhaps the most vulnerable site for microbial attack, it is not breached as long as oral hygiene is satisfactory. However, when plaque accumulates close to the gingival margin, the host defences are overcome, and gingival inflammation (gingivitis) and subsequent periodontal inflammation with loss of attachment ensue (periodontitis).
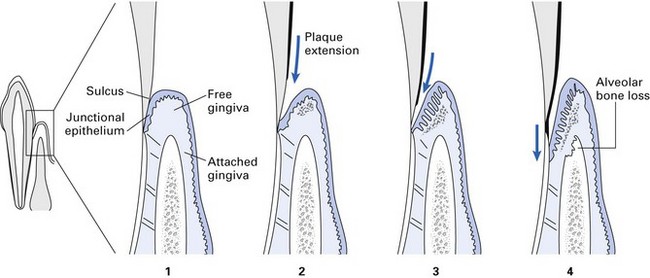
Fig. 33.1 The progression of a marginal periodontium from health to disease. (1) A healthy gingival sulcus with minimal supragingival plaque. (2) Established chronic gingivitis with minor inflammatory enlargement. (3) Long-standing chronic gingivitis with subgingival plaque extension into the pocket. (4) Chronic periodontitis with destruction of the periodontal membrane, alveolar bone loss and apical migration of the epithelial attachment.
Host defence factors
Both the specific and non-specific immune responses of the host to subgingival plaque are considered to play critical roles in the initiation, progression and recovery from periodontal diseases. One of the most important components of the host response is the GCF, which contains both specific and non-specific defence factors (Table 33.2).
Table 33.2 Specific and non-specific defence factors in gingival crevicular fluid
| Specific | Non-specific |
|---|---|
| B and T lymphocytes | Polymorphs |
| Macrophages | |
| Antibodies: IgG, IgA, IgM | Complement system |
| Proteases | |
| Lysozyme | |
| Lactoferrin |
IgG, immunoglobulin G.
Polymorphonuclear leukocytes
Clinically healthy gingiva contain small numbers of polymorphonuclear leukocytes (PMNLs). Their numbers increase markedly during the onset of gingivitis and periodontitis. The PMNLs migrate from venules and enter the gingival sulcus through the junctional epithelial cells. When PMNLs encounter bacteria, phagocytosis ensues, and the ingested organisms are then killed with a combination of proteolytic and hydrolytic enzymes, and other cell-derived killing agents such as hydrogen peroxide and lactic acid. Although phagocytosis can occur in the absence of antibody, the presence of immunoglobulins and complement enhances the process. The interaction between PMNLs and plaque bacteria may result in:
Thus, PMNLs may have both a protective and a damaging effect on host tissues. Phagocytosis, which may occur within the host tissues and possibly at the interface with subgingival plaque, is important in preventing the microbial ingress into the tissues.
Antibody
Locally derived specific antibodies (IgM, IgG and IgA) to subgingival plaque organisms are found in the GCF. An elevated titre of specific antibody to a periodontopathogen may be:
The presence of antibody implies that the T cell (helper and suppressor) and B cell interactions occur in periodontal tissues. Cells required for a wide range of immune reactions, present in gingival tissues of periodontitis patients, possess antigen specificity for plaque bacteria. When stimulated, either antibodies (from B lymphocytes) or lymphokines (from T lymphocytes) are produced.
Antibodies and complement present in the periodontal tissues interact to produce hypersensitivity reactions, which may damage host tissues and also contribute to periodontal disease. There is evidence that all four types of hypersensitivity may be involved in the pathogenesis of periodontal disease.
Microorganisms in subgingival plaque biofilm
That dental plaque biofilm is the essential aetiological agent of the common forms of chronic gingivitis and periodontitis is shown by the following:
Microbiological studies of periodontal plaque flora
As most of the periodontal plaque flora is anaerobic, special care must be taken to preserve the viability of these organisms during sampling, dispersion and cultivation of plaque samples. Ideally, the sample should be taken from the advancing front of the lesion at the base of the pocket, although in practice, this is difficult because of contaminants from the superficial plaque at the top of the pocket. The techniques involved in microbiological studies of pocket flora include:
Specific and non-specific plaque hypotheses
Although bacteria are definitive agents of periodontal diseases, there are conflicting views as to whether a single or a limited number of species are involved in the disease process – the specific plaque hypothesis – or disease is caused by any combination of a wider range of non-specific bacteria – the non-specific plaque hypothesis.
The specific plaque hypothesis
In certain disease states such as necrotizing ulcerative gingivitis, the key aetiological agents are fusobacteria and spirochaetes. Furthermore, this disease can be resolved by appropriate antibiotics active against anaerobes (e.g. metronidazole). Other studies have convincingly shown the direct involvement of Aggregatibacter actinomycetemcomitans in aggressive (juvenile) periodontitis, and disease resolution after therapy with tetracycline, which is active against this organism. These observations led to the theory of specific plaque hypothesis.
The non-specific plaque hypothesis
This hypothesis proposes that collective groups or consortia of different bacteria have the total complement of virulence factors required for periodontal tissue destruction and that some bacteria can substitute for others absent from the pathogenic consortium. This hypothesis implies that plaque will cause disease irrespective of its composition, and it is supported by the clinical findings of numerous bacterial species in diseased periodontal pockets.
It is likely that the two theories represent the extremes of a complex series of host–parasite interactions.
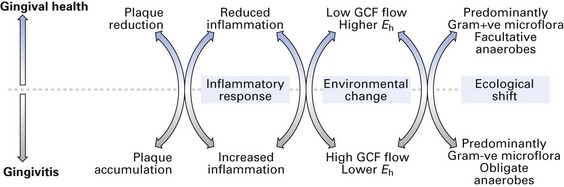
The ecological plaque hypothesis
The ecological plaque hypothesis has also been proposed for the aetiology of periodontal disease. This postulates the following causative process:
This simple yet elegant hypothesis implies that periodontal disease is an endogenous or an opportunistic infection, caused by an imbalance in the composition of the resident microflora at a site, owing to an alteration in the ecology of the local habitat (Fig. 33.2).
Clinical implications
The non-specific plaque hypothesis and the ecology hypothesis imply that periodontal disease may be treated by reducing the plaque to an acceptable level and the maintenance of healthy plaque, or by achievement of total plaque control. In contrast, the specific plaque hypothesis implies that therapy should be directed at elimination of specific pathogens, for instance, by appropriate antibiotic therapy.
Periodontal health and disease
Healthy gingival sulcus has a scant flora dominated by almost equal proportions of Gram-positive and facultative anaerobic organisms; spirochaetes and motile rods make up less than 5% of the organisms (Table 33.3). With increasing severity of disease, the proportions of strict anaerobic, Gram-negative and motile organisms increase significantly (Fig. 33.3).
Table 33.3 Microorganisms associated with various types of periodontal disease
| Condition | Predominant microorganisms | Comments |
|---|---|---|
| Health | Streptococcus sanguinis (previously Streptococcus sanguis) | Mainly Gram-positive cocci with few spirochaetes or motile rods |
| Streptococcus oralis | ||
| Actinomyces naeslundii | ||
| Actinomyces viscosus | ||
| Veillonella spp. | ||
| Chronic marginal gingivitis | Streptococcus sanguinis | About 55% of cells are Gram-positive with occasional spirochaetes and motile rods |
| Streptococcus milleri | ||
| Actinomyces israelii | ||
| Actinomyces naeslundii | ||
| Prevotella intermedia | ||
| Capnocytophaga spp. | ||
| Fusobacterium nucleatum | ||
| Veillonella spp. | ||
| Chronic periodontitis | Porphyromonas gingivalis | About 75% of cells are Gram-negative (90% being strict anaerobes). Motile rods and spirochaetes are prominent |
| Prevotella intermedia | ||
| Fusobacterium nucleatum | ||
| Tannerella forsythia (formerly Bacteroides forsythus) | ||
| Aggregatibacter actinomycetemcomitans | ||
| Selenomonas spp. | ||
| Capnocytophaga spp. | ||
| Spirochaetes | ||
| Aggressive periodontitis | Aggregatibacter actinomycetemcomitans | About 65–75% of bacteria are Gram-negative bacilli. Few spirochaetes or motile rods are present. These diseases may be associated with cellular immune or genetic defects |
| Capnocytophaga spp. | ||
| Porphyromonas gingivalis | ||
| Prevotella intermedia |
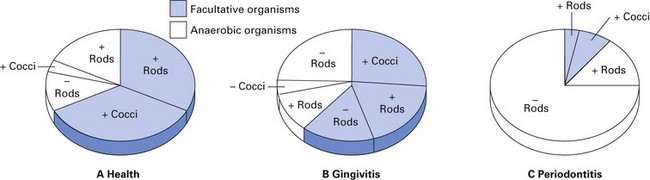
Fig. 33.3 Predominant plaque bacterial morphotypes in (A) health, (B) gingivitis and (C) periodontitis. +, Gram-positive; –, Gram-negative.
A wide range of microbial products potentially toxic to host tissues have been identified in plaque bacteria; these virulence determinants are shown in Table 33.4. If these toxic products are released into the periodontal tissues, then rapid destructive inflammatory disease could be expected. However, tissue destruction is usually slow, sporadic and episodic, suggesting the existence of powerful host defence mechanisms, of which little is known. However, the nature of the periodontal disease and its elusive progression or regression could be explicable by the following:
Table 33.4 Some microbial virulence determinants in periodontal disease
A note on the role of viruses in periodontal disease
There are a few who surmise that viral infections may play a role in periodontal disease. Such an association has been suggested for HIV and herpesviruses, especially in view of the aggravation of periodontal diseases in HIV disease (Chapter 30). The demonstration of viral DNA in gingival tissues, crevicular fluid and subgingival plaque in diseased sites has added some credence to this hypothesis. However, conclusive data are warranted to confirm an aetiological role for viruses in periodontal disease.
Relationship between chronic marginal gingivitis and periodontitis
Both chronic marginal gingivitis and periodontitis are inflammatory diseases: the lesions of the former are confined to the gingivae; the latter involves destruction of both the connective tissue attachment of the tooth and the alveolar bone. Gingivitis is common in both adults and children, although early periodontitis is rarely seen before late adolescence. It is considered that chronic periodontitis is preceded by chronic gingivitis; however, in some cases, gingivitis may exist for prolonged periods without progressing to periodontitis. The main stages in the development of chronic gingivitis and periodontitis are shown in Figure 33.1.
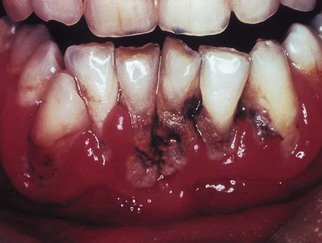
Chronic marginal gingivitis
Clinical presentation
The gingivae are red and swollen, with rounded edges; bleeding gums and halitosis are common. However, pain, discomfort and unpleasant taste are uncommon.
Pathogenesis
Plaque-associated gingivitis is divided into three separate but contiguous phases:
The initial lesion
Early histological examination shows an acute inflammatory reaction associated with vasculitis, perivascular collagen destruction, increase in crevicular fluid and polymorphonuclear leukocytosis in the junctional epithelium and crevice. At this stage, no clinical change is evident.
The early lesion
After about 7 days, clinically recognizable chronic gingivitis with gingival inflammation is seen. A dense infiltration of lymphocytes (75%) with macrophages and plasma cells can be observed, especially at the periphery of the lesion. The lymphocytic infiltrate occupies approximately 15% of the marginal connective tissue with areas of local collagen destruction. Polymorph infiltration of the gingival sulcus peaks 7–12 days following the onset of clinically detectable gingivitis.
The established lesion
This develops after a variable period of time when the above-mentioned changes in the gingival crevice support the growth of predominantly anaerobic flora. Histologically, a predominance of plasma cells and B lymphocytes are seen, together with a heavy neutrophil infiltrate in the junctional and the newly developed pocket epithelium. It is during this stage that periodontal pocket formation begins.
If oral hygiene is improved at this juncture without the removal of subgingival plaque, then the lesion may persist for years without extending into the deeper periodontal tissues.
Microbiology
Gingivitis is related to the prolonged exposure of host tissues to a non-specific mixture of gingival plaque organisms. The microbiological features of the gingival pocket necessarily change during the transition from the initial lesion to the established lesion. In the initial stage, Gram-positive and facultative organisms predominate, including streptococci (see Table 33.3). In the early lesion, Actinomyces spp. increase together with proportions of capnophilic species such as Capnocytophaga spp. and obligately anaerobic Gram-negative bacteria. For example, in one study, in the initial stage (of non-bleeding gingivitis), proportions of Actinomyces israelii and Actinomyces naeslundii almost doubled. When the disease progresses to the established lesion, where bleeding is seen, the flora further changes, and levels of black-pigmented anaerobes such as Porphyromonas gingivalis and Prevotella intermedia increase quantitatively (e.g. 0.1–0.2% of total plaque flora), together with spirochaetes.
The transition from gingivitis to periodontitis
Chronic marginal gingivitis may be present for up to 10 years in some individuals before progressing to periodontitis. This transition may be due to one or a combination of the following:
Chronic periodontitis (formerly adult periodontitis)
Periodontitis can be classified into various groups (Table 33.1), but chronic periodontitis is by far the most prevalent disease globally.
Morbidity
About 70–80% of all adults suffer from this universal disease, and chronic periodontitis comprises 95% of all periodontal diseases. Prevalence and severity increase with age.
Clinical presentation
All the features of the established lesion are present in addition to the following:
Pathogenesis
The main processes that produce loss of attachment and pocket formation are (Fig. 33.1):
Microbiology
Microorganisms implicated in chronic periodontitis are listed in Table 33.3. The depth of the periodontal pocket creates a highly anaerobic locale with a shift from neutral to alkaline pH (7.4–7.8). The protein-rich fluid in the pocket encourages the growth of anaerobes, which possess many proteolytic enzymes. The subgingival plaque has two distinct zones: a zone of Gram-positive cocci and bacilli close to the tooth surface, and a zone of Gram-negative organisms next to the gingival crevice. In active pockets, Porphyromonas gingivalis, A. actinomycetemcomitans, Prevotella intermedia and F. nucleatum may be present. Specific microbes are discussed below.
Spirochaetes
Significantly lower numbers of spirochaetes (Chapter 18) are present in healthy periodontal tissue, compared with diseased sites. Thus, it was thought that a high percentage of spirochaetes in a subgingival sample strongly suggested a site undergoing – or about to experience – active, destructive disease. However, it is now clear that the number of spirochaetes cannot predict active periodontitis, and therefore, the evidence for ‘spirochaete specificity’ is conflicting and confused. It is possible that one or more Treponema spp. are involved in the disease process.
Porphyromonas, Prevotella and Tannerella spp.
Although now divided into three species (Chapter 17), these organisms formerly belonged to a single group of organisms called ‘black-pigmented Bacteroides species’. These bacteria are often isolated from periodontal pockets in large numbers and are believed to be intimately associated with all forms of periodontitis.
The evidence for the specificity of Porphyromonas and Prevotella species depends mainly on the following:
Capnocytophaga and corroding bacteria
Capnocytophaga spp. (Chapter 14) are members of the commensal oral flora and were implicated as prime pathogens in periodontal infections at one time, especially in localized aggressive periodontitis (formerly localized juvenile periodontitis). Various corroding bacteria such as Wolinella spp. and Eikenella corrodens have been associated with a number of forms of periodontal disease. However, their precise role is uncertain.
Aggressive periodontitis
Periodontal diseases, previously classified as juvenile periodontitis (localized and generalized), rapidly progressive periodontitis, early-onset periodontitis and prepubertal periodontitis, are now categorized under this common heading.
Localized and generalized aggressive periodontitis (formerly localized/generalized juvenile periodontitis)
Morbidity
The condition is relatively rare – 0.1% in young whites – but is more common in West Africans and Asians. It appears around puberty and is relatively common in girls; case clusters are usually seen in families.
Initiation and course
Approximately around 13 years, with onset of puberty; rather rapid progress with active and quiescent periods.
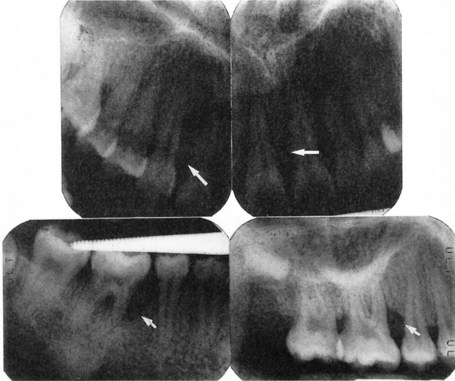
Clinical features
In the localized variant, the incisors and/or first permanent molars in both jaws are affected for unknown reasons. Later, other teeth may be involved, producing the appearance of generalized alveolar bone loss (Fig. 33.5). Alternatively, in the generalized variant of the disease, many areas may be involved in a similar manner. The disease is insidious in nature and lesions are discovered incidentally on radiographs. In some generalized cases, about 50% of the supporting alveolar bone is affected and teeth may be lost. The condition may or may not manifest with gingivitis, and patients can present with various levels of oral hygiene. In contrast to chronic periodontitis, little plaque or calculus is present in periodontal pockets. The disease may be inherited (autosomal recessive).
Microbiology and immunology
A majority of patients with aggressive periodontitis have peripheral blood lymphocytes with impaired ability to react to chemotactic stimuli. This deficiency may be associated with, or is a direct cause of, the presence of large numbers of A. actinomycetemcomitans, a Gram-negative coccobacillus. Other organisms, such as Capnocytophaga spp. and Porphyromonas gingivalis, may be synergistically associated with the disease. The evidence for the specific involvement of A. actinomycetemcomitans in aggressive periodontitis includes:
A. actinomycetemcomitans is a rare but recognized pathogen in medical microbiology and has been implicated in actinomycosis (Chapter 13), abdominal and brain abscesses, septicaemia and infective endocarditis.
Necrotizing ulcerative gingivitis
Necrotizing ulcerative gingivitis, also known as acute necrotizing ulcerative gingivitis (ANUG), is rare in the West but may be seen in developing countries; it is commonly associated with poor and neglected oral hygiene, malnutrition and possibly systemic diseases.
Clinical features
The condition is characterized by actually inflamed, red, shiny and bleeding gingivae with irregularly shaped ulcers, which initially appear on the tips of the interdental papillae. If untreated, the ulcers enlarge and spread to involve the marginal and, rarely, the attached gingivae (Fig. 33.6). The lesions are extremely painful and are covered by a pseudomembrane (or slough), which can be wiped from the surface. The slough consists of leukocytes, erythrocytes, fibrin, necrotic tissue debris and microorganisms. Characteristically, the patient’s breath is malodorous. The patient may complain of an unpleasant metallic taste. There is little or no systemic upset, and mild submandibular lymphadenitis; involvement of the cervical lymph nodes only occurs in severe cases. Generalized fever or malaise is very uncommon.
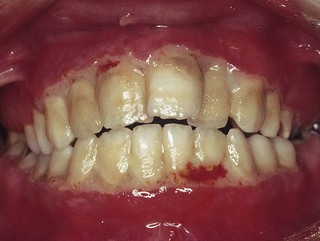
Fig. 33.6 Acute necrotizing ulcerative gingivitis. Note the loss of papillae, spontaneous bleeding and gross plaque accumulation.
If the disease is inadequately treated, tissue destruction slows down and the disease may enter a chronic phase with pronounced loss of supporting tissues (noma).
Microbiology
The disease is a specific, anaerobic, polymicrobial infection, mainly due to the combined activity of fusobacteria (F. nucleatum) and oral spirochaetes (Treponema spp.) – the so-called fusospirochaetal complex. The main evidence for the microbial specificity of ANUG is:
Cultural studies indicate that medium-sized spirochaetes account for a third, and fusobacteria less than 5%, of the total flora. The remaining organisms comprise Prevotella intermedia, Veillonella and streptococci.
Diagnosis
The clinical appearance together with the offensive smell is pathognomonic. Confirmatory evidence is obtained by microscopy of a Gram-stained, deep gingival smear of the ulcerated lesion. A predominance of three components – fusobacteria, spirochaetes and leukocytes – is essential for a confident diagnosis (see Fig. 18.2); some, but not all three, of these components may be observed in primary herpetic stomatitis, gonococcal gingivitis, benign mucous membrane pemphigoid, desquamative gingivitis and some forms of leukaemia.
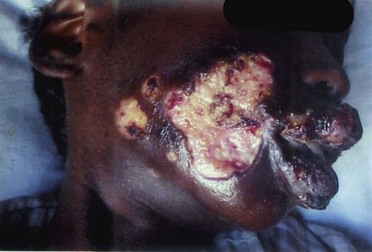
Noma or cancrum oris
In some developing countries (e.g. sub-Saharan Africa), an extremely severe form of ANUG called noma or cancrum oris, also called gangrenous stomatitis, is seen in children. Typically, the child is less than 10 years old, severely malnourished (especially with regard to protein) and has a recent history of viral infection, e.g. measles or other debilitating diseases such as tuberculosis. As a result, the specific immune system of the child may be compromised, and the initial necrotic lesion may spread locally from the gingivae into the cheek and sometimes to the face, causing extensive tissue loss and severe disfigurement (Fig. 33.7). Noma is extremely rare in developed countries.
Peri-implant microbiology
Peri-implant health
Plaque biofilms found in the healthy peri-implant sulci around dental implants are similar in composition to biofilms found in healthy gingival sulci around teeth. There are low levels of bacteria with a predominance of facultative Gram-positive coccoid bacteria.
Peri-implant disease
Peri-implant diseases are inflammatory conditions affecting the peri-implant tissues and include peri-implant mucositis and peri-implantitis. Peri-implant mucositis refers to inflammation of the peri-implant soft tissues without loss of supporting bone. Peri-implantitis is inflammation of the peri-implant soft tissues with loss of supporting bone.
Peri-implant diseases are mixed microbial infections and in most cases show a similar microflora to that found in chronic periodontitis, dominated by diverse Gram-negative anaerobic bacteria (Fusobacterium spp., Porphyromonas gingivalis, Prevotella spp., Aggregatibacter actinomycetemcomitans). However some studies show high numbers of other organisms such as Peptostreptococci, Staphylococci (S. aureus), enteric rods and yeasts.
Studies evaluating the dynamics of colonization around dental implants in partially dentate individuals indicate that colonization begins within the first 30 minutes after exposure of the implant to the oral cavity. With time, a complex microflora gradually develops which is similar in composition to biofilms found on neighbouring teeth. This underlines the importance of treating periodontitis and establishing a microflora conducive to periodontal health prior to placing dental implants.
Clinical implications of microbiological tests in periodontal disease
Microbiological tests are useful in the management of periodontal disease to identify sites of active tissue destruction and to monitor the effects of treatment and decide when recall is necessary. The presence of a specific putative pathogen associated with any of the periodontal diseases mentioned above could be detected by:
However, these tests are only useful if the identified organisms are definitively known to cause the disease, and if samples can be collected accurately from the site of disease (i.e. probably the base of the periodontal pocket). As this stage has not yet been reached, doubt exists as to the value of these tests in diagnosis. Sampling for the presence of A. actinomycetemcomitans in aggressive periodontitis is the only microbiological test that is likely to contribute to the treatment of chronic periodontal diseases at present. A positive test would suggest that systemic antibiotic therapy could be considered a useful adjunct after root instrumentation.
Key facts
Armitage G.C. Development of a classification system for periodontal disease. Annals of Periodontology. 1999;4:1-6.
Cappuyns I., Gugerli P., Mombelli A. Viruses in periodontal disease – A review. Oral Diseases. 2005;11:219-229.
Enwonwu C.O. Noma – The ulcer of extreme poverty. New England Journal of Medicine. 2006;354:221-224.
Marsh P.D., Marin M.V. Oral microbiology, 5th ed. London: Churchill Livingstone; 2009.
Meyer D.H., Fives-Taylor P.M. The role of Actinobacillus actinomycetemcomitans in the pathogenesis of periodontal diseases. Trends in Microbiology. 1997;5:224-228.
Mombelli A., Samaranayake L.P. Topical and systemic antibiotics in the management of periodontal disease. International Dental Journal. 2004;54:3-14.
Slots J. Microflora of the healthy gingival sulcus of man. Scandinavian Journal of Dental Research. 1997;85:247-254.
Socransky S.S., Haffajee A.D. Microbiology of periodontal disease. In Lindhe J., Karring T., Lang N.P., editors: Clinical periodontology and implant dentistry, 4th ed., Oxford: Blackwell, Munksgaard, 2003. Ch. 4
Review questions (answers on p. 355)
Please indicate which answers are true, and which are false.