Chapter 11 Streptococci, staphylococci and micrococci
Streptococci comprise a diverse group of Gram-positive cocci, which continuously undergo taxonomic revision. They are distributed widely in humans and animals, mostly forming part of their normal flora. A few species cause significant human morbidity. The oral streptococci, which include the cariogenic mutans group, are important members of the genus. Another common group of cocci, the staphylococci, live on the skin but are infrequently isolated from the oral cavity and are significant agents of many pyogenic (pus-producing) human infections.
Streptococci
General properties
Characteristics
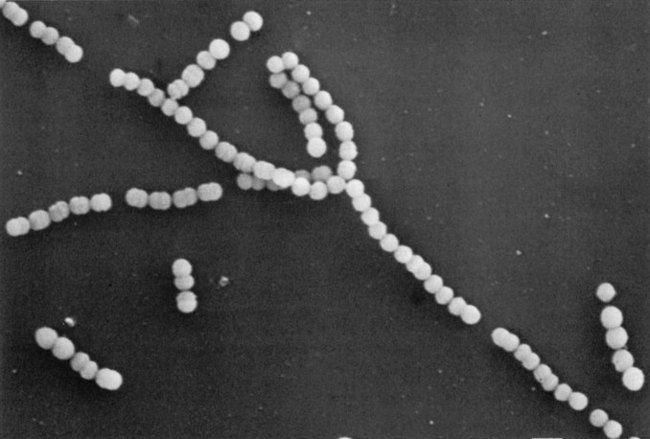
They are catalase-negative, Gram-positive spherical or oval cocci in pairs and chains; 0.7–0.9 µm in diameter. Chain formation is best seen in liquid cultures or pus.
Culture
These cocci grow well on blood agar, although enrichment of media with glucose and serum may be necessary. Typical haemolytic reactions are produced on blood agar (Fig. 11.1):
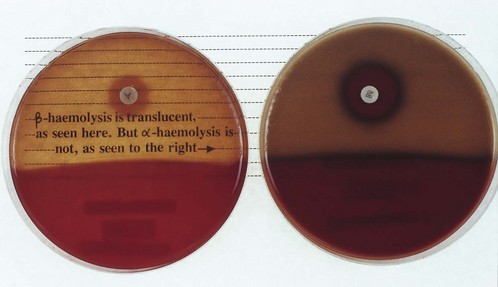
Fig. 11.1 α- and β-haemolysis: β-haemolytic colonies (e.g. Streptococcus pyogenes) produce complete translucence of blood agar, whereas α-haemolytic colonies (e.g. Streptococcus pneumoniae) do not. Note also the sensitivity of S. pneumoniae to a disc impregnated with optochin.
Serology
The carbohydrate antigens found on the cell walls of the organisms are related to their virulence. Hence, serogrouping, termed Lancefield grouping, is useful in the identification of the more virulent β-haemolytic species. Currently, 20 Lancefield groups are recognized (A–H and K–V) but not all are equally important as human pathogens. The following are worthy of note:
Streptococcus pyogenes (group A)
Habitat and transmission
The normal habitat of this species is the human upper respiratory tract and skin; it may survive in dust for some time. Spread is by airborne droplets and by contact.
Characteristics
It is found as a commensal in the nasopharynx of a minority of healthy adults, but more commonly (about 10%) in children. It grows well on blood agar, with a characteristic halo of β-haemolysis. Some strains produce mucoid colonies as a result of having a hyaluronic acid capsule. This may contribute to virulence by offering resistance to phagocytosis.
Exotoxins and enzymes
Produces a large number of biologically active substances, such as:
(Note: not all these products are produced by every strain; the combined action of enzymes and toxins contributes to the pathogenicity.)
Culture and identification
Culture on blood agar yields characteristic β-haemolytic colonies (lysis of blood due to streptolysins O and S). A Gram-stained smear may show characteristic cocci in chains (Fig. 11.2); these are more developed in liquid than in solid media. The isolate can be presumptively identified as Streptococcus pyogenes if it is sensitive to bacitracin.
If rheumatic fever is suspected, then testing the patient’s antistreptolysin O (ASO) antibody titre will demonstrate previous exposure to Streptococcus pyogenes.
Pathogenicity
Streptococcus pyogenes causes a number of infections; the most notable are:
Complications
After an episode of infection, some patients develop complications, such as rheumatic fever, glomerulonephritis and erythema nodosum, which may have long-lasting effects. Note that:
Streptococcus agalactiae (group B)
This species is increasingly recognized as a human pathogen, especially as a cause of neonatal meningitis and sepsis.
Habitat and transmission
Found in the human vagina; sometimes anorectal carriage occurs. Babies acquire infection from the colonized mother during delivery or during nursing.
Culture and identification
Gram-stained smear and culture yielding β-haemolytic colonies on blood agar; colonies on blood agar are generally larger than Streptococcus pyogenes. Lancefield group is determined by antiserum against cell wall polysaccharide.
Oral streptococci
Oral streptococci, which live principally in the oropharynx, are a mixed group of organisms with variable characteristics. New typing techniques, particularly those based on molecular biology, have revealed the complex nature of the origin and the taxonomy of this group. Hence, the nomenclature of oral streptococci is in a constant state of flux. They typically show α-haemolysis on blood agar, but this is not a constant feature as some strains are non-haemolytic and others β-haemolytic. Oral streptococci can be divided into four main species groups as follows:
Each of these groups comprises a number of species (Table 11.1).
Table 11.1 Some recognized species of oral streptococci
| Group | Species |
|---|---|
| mutans group | S. mutans, serotypes c, e, f |
| S. sobrinus, serotypes d, g | |
| S. cricetus, serotype a | |
| S. rattus, serotype b and others | |
| salivarius group | S. salivarius |
| S. vestibularis | |
| anginosus group | S. constellatus |
| S. intermedius | |
| S. anginosus | |
| mitis group | S. sanguinis |
| S. gordonii | |
| S. parasanguinis | |
| S. oralis and others |
Habitat and transmission
Streptococci make up a large proportion of the resident oral flora. It is known that roughly one-quarter of the total cultivable flora from supragingival and gingival plaque and half of the isolates from the tongue and saliva are streptococci. They are vertically transmitted from mother to child. Infective endocarditis caused by these organisms (loosely termed viridans streptococci) is generally a result of their entry into the blood stream during intraoral surgical procedures (e.g. tooth extraction), and sometimes even during tooth-brushing.
Culture and identification
Gram-positive cocci in chains; α-haemolytic; catalase-negative. Growth not inhibited by bile or optochin (ethylhydrocupreine hydrochloride), in contrast to pneumococci. Commercially available kits are highly useful in laboratory identification of these organisms.
Pathogenicity
The mutans group of streptococci are the major agents of dental caries (but in the absence of predisposing factors, such as sucrose, they cannot cause caries). They have a characteristic ability to produce voluminous amounts of sticky, extracellular polysaccharides in the presence of dietary carbohydrates (Fig. 11.3); these help tenacious binding of the organisms to enamel and to each other.
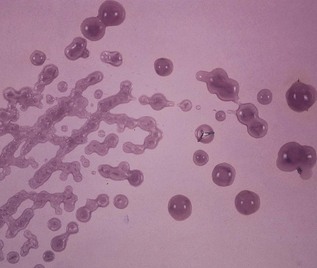
Fig. 11.3 Gelatinous colonies of mutans streptococci mainly comprising extracellular polysaccharides.
They are also important agents of infective endocarditis, and some 60% of cases are due to this organism. Usually, bacteria released during dental procedures settle on damaged heart valves, causing infective endocarditis (see Chapter 24).
Treatment and prevention
In patients at risk of infective endocarditis (e.g. those with damaged or prosthetic heart valves), prophylactic antibiotic cover should always be given before dental procedures (see Chapter 24).
See Chapter 32 for caries.
Streptococcus mutans
Streptococcus mutans gained notoriety in the 1960s when it was demonstrated that caries could be experimentally induced and transmitted in animals by oral inoculation with the organism. The name ‘mutans’ results from its frequent transition from coccal phase to coccobacillary phase. Currently, seven distinct species of human and animal mutans streptococci and eight serotypes (a–h) are recognized, based on the antigenic specificity of cell wall carbohydrates. The term Streptococcus mutans is limited to human isolates belonging to three serotypes (c, e and f).
Other oral streptococci
A group of oral organisms previously classified as nutritionally variant streptococci (Streptococcus adjacens, Streptococcus defectivus) and isolated under appropriate environmental conditions has been assigned to a new genus called Abiotrophia. Their role in oral disease is not well characterized.
Streptococcus pneumoniae (pneumococcus)
This organism causes a number of common diseases, such as pneumonia and meningitis in adults and otitis media and sinusitis in children.
Habitat and transmission
A normal commensal in the human upper respiratory tract; up to 4% of the population carry this bacteria in small numbers. Transmission is via respiratory droplets.
Characteristics
Gram-positive ‘lancet-shaped’ cocci in pairs (diplococci) or short chains; cells are often capsulate; α-haemolytic on blood agar; catalase-negative; facultative anaerobe (i.e. grows under both aerobic and anaerobic conditions).
Culture and identification
Forms α-haemolytic colonies. After incubation for 2 days, the colonies appear typically as ‘draughtsmen’ because of their central indentation (a result of spontaneous autolysis of older bacteria in the centre of the colony). The species is differentiated from other α-haemolytic streptococci by its sensitivity to optochin and solubility in bile (Fig. 11.1). Observation for the capsular swelling with type-specific antiserum (quellung reaction) confirms the identity and is the standard reference method. The latex agglutination test (see Fig. 6.7) for capsular antigen in spinal fluid can be diagnostic.
Pathogenicity
Although no exotoxins are known, this organism induces an inflammatory response. The substantive polysaccharide capsule retards phagocytosis. Vaccination with antipolysaccharide vaccine helps provide type-specific immunity. Viral respiratory infection predisposes to pneumococcal pneumonia by damaging the mucociliary lining of the upper respiratory tract (the mucociliary escalator). Other common diseases caused by pneumococci include lobar pneumonia, acute exacerbation of chronic bronchitis, otitis media, sinusitis, conjunctivitis, meningitis and, in splenectomized patients, septicaemia.
Gram-positive anaerobic cocci
Gram-positive anaerobic cocci (GPAC) all belonged to the genus Peptostreptococcus until recently. However, they now comprise three genera, namely Peptostreptococcus, Micromonas and Finegoldia. The representative species are Peptostreptococcus anaerobius, Finegoldia magnus (previously Peptostreptococcus magnus) and Micromonas micros (previously Peptostreptococcus micros).
These GPAC can often be isolated from dental plaque and the female genital tract. They are also found in carious dentine, subgingival plaque, dentoalveolar abscesses and in advanced periodontal disease, usually in mixed culture. Their pathogenic role is still unclear.
Staphylococci
Staphylococci are also Gram-positive cocci, but, unlike the chains of streptococci, they are arranged in characteristic grape-like clusters. The Staphylococcus genus contains more than 15 different species, of which the following are of medical importance: Staphylococcus aureus, Staphylococcus epidermidis and Staphylococcus saprophyticus.
Staphylococci cause a variety of both common and uncommon infections, such as abscesses of many organs, endocarditis, gastroenteritis (food-poisoning) and toxic shock syndrome. They are not infrequent isolates from the oral cavity. Higher proportions of Staphylococcus aureus are found in the saliva of healthy subjects older than 70 years.
Staphylococcus aureus
Habitat and transmission
The habitat is the human skin, especially the anterior nares and the perineum. Domesticated animals also carry staphylococci. Higher carriage rates are seen in hospital patients and staff. These bacteria are disseminated through air and dust and are always present in the hospital environment. The usual transmission route is via the hands and fingertips.
Characteristics
Gram-positive cocci in clusters (cluster formation is due to their ability to divide in many planes); non-sporing, non-motile; some strains are capsulate.
Culture and identification
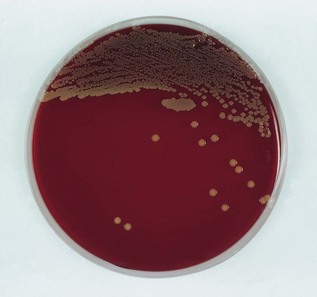
Grows aerobically as yellow or gold colonies on blood agar (Fig. 11.4); catalase-positive (this differentiates them from the catalase-negative streptococci).
Other tests used to differentiate the more virulent Staphylococcus aureus from the less pathogenic Staphylococcus epidermidis include the following.
Coagulase test
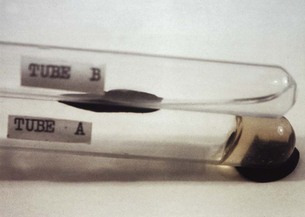
Staphylococcus aureus coagulates dilute human serum or rabbit plasma (i.e. it is coagulase-positive), whereas Staphylococcus epidermidis does not (coagulase-negative). This test could be done either in a test tube (the tube test), which requires overnight incubation (Fig. 11.5), or on a slide (the slide test), which is a rapid test.
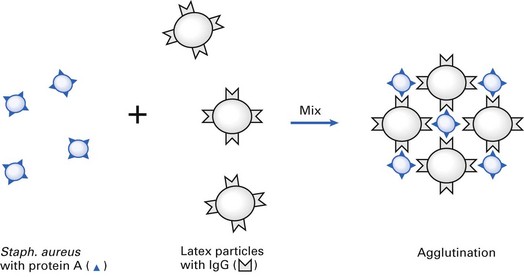
Protein A – latex agglutination test
Protein A, synthesized by almost all strains of Staphylococcus aureus, has a special affinity to the Fc fragment of immunoglobulin G (IgG). Hence, when latex particles coated with IgG (and fibrinogen) are mixed with an emulsified suspension of Staphylococcus aureus on a glass slide, visible agglutination of the latex particles occurs; no such reaction is seen with Staphylococcus epidermidis (Fig. 11.6).
Typing of Staphylococcus aureus
Typing is important to determine the source of an outbreak of infection. This was commonly done by the pattern of susceptibility to a set of more than 20 bacteriophages – phage-typing and serotyping. These methods are currently supplanted by molecular typing techniques. Antibiotic susceptibility patterns are also helpful in tracing the source of outbreaks.
Pathogenicity
A variety of enzymes and toxins are produced by Staphylococcus aureus, although no one strain produces the whole range listed in Table 11.2. The two most important are coagulase and enterotoxin. Coagulase is the best correlate of pathogenicity. Some of the diseases caused by Staphylococcus aureus are:
Table 11.2 Toxins and enzymes produced by Staphylococcus aureus
| Toxin/enzyme | Activity |
|---|---|
| Toxins | |
| Cytotoxins (α, β, γ, δ) | Cell lysis |
| Leukocidin | Kills leukocytes |
| Epidermolytic toxin | Exfoliation and splitting of epidermis |
| Toxic shock syndrome toxin | Shock, rash, desquamation |
| Enterotoxin (A–E) | Induces vomiting and diarrhoea |
| Enzymes | |
| Coagulase | Clots plasma |
| Catalase | Affects bactericidal activity of polymorphs |
| Hyaluronidase | Connective tissue breakdown |
| DNAase (nuclease) | DNA hydrolysis |
| Lipase | Breaks lipids of cell membranes |
| Penicillinase | Breaks down β-lactam drugs |
| Protein A | Antiphagocytic |
Predisposing factors for infection are minor and major breaks in the skin, foreign bodies such as sutures, low neutrophil levels and injecting drug abuse.
Treatment and prevention
The vast majority (>80%) of strains are resistant to β-lactam drugs, and some to a number of antibiotics. The latter phenomenon (multiresistance) is common, particularly in strains isolated from hospitals; these cause hospital (nosocomial) infection. Penicillin resistance is due to the production of β-lactamase encoded by plasmids. The enzyme destroys the efficacy of antibiotics with a β-lactam ring (i.e. the penicillin group drugs).
Antibiotics active against Staphylococcus aureus include penicillin for sensitive isolates, flucloxacillin (stable against β-lactamase), erythromycin, fusidic acid (useful for skin infections), cephalosporins and vancomycin.
Cleanliness, hand-washing and aseptic management of lesions reduce the spread of staphylococci.
Antibiotic resistance in staphylococci
This is a global problem of much concern and falls into several classes.
Staphylococcus epidermidis
Culture and identification
Grows as white colonies on blood agar, hence the earlier name Staphylococcus albus; catalase-positive; coagulase-negative; biochemically characterized by commercially available kits (e.g. APIStaph).
Staphylococcus saprophyticus
This organism causes urinary tract infections in women, an infection especially associated with intercourse. It has the ability to colonize the periurethral skin and the mucosa. The organism can be differentiated from Staphylococcus epidermidis (both grow as white colonies on blood agar) by the mannitol fermentation reaction and other biochemical tests.
Micrococci
Micrococci are catalase-positive organisms similar to staphylococci. They are coagulase-negative and usually grow as white colonies on blood agar, although some species are brightly pigmented – pink, orange or yellow.
Stomatococcus mucilagenosus, formerly classified in the genus Micrococcus, is found in abundance on the lingual surface. This species has the ability to produce an extracellular slime, which correlates with its predilection for the lingual surface. Its role in disease, if any, is unknown.
Key facts
Beighton D., Hardie J., Whiley R.A. A scheme for the identification of viridans streptococci. Journal of Medical Microbiology. 1991;35:367-372.
Greenwood D., Slack R., Peutherer J., editors. Medical microbiology, 16th ed, Edinburgh: Churchill Livingstone, 2003. Chs 15–17
Jone D., Board R.G., Sussman M. Staphylococci, Society for Applied Microbiology Symposium Series No. 19. Oxford: Blackwell Scientific. 1990.
Murdoch D.A. Gram-positive anaerobic cocci. Clinical Microbiology Reviews. 1993;11:81-120.
Review questions (answers on p. 352)
Please indicate which answers are true, and which are false.