CHAPTER 10 The shin
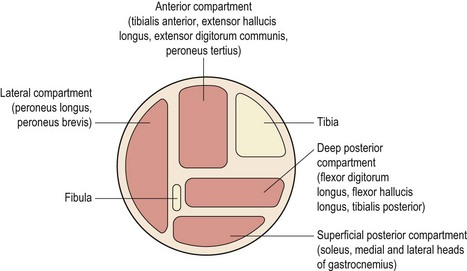
The term ‘shin splints’ is often used as a blanket description of any persistent pain occurring between the knee and ankle in an athlete. Originally called ‘fresher’s leg’, a more accurate description of this type of exercise induced leg pain comes with the various ‘compartment syndromes’ which identify the anatomical structures affected. The anterior compartment contains the tibialis anterior, extensor hallucis longus, extensor digitorum longus, and the anterior tibial artery and vein. The lateral compartment contains the peronei and the superficial peroneal nerve. The superficial posterior compartment contains the gastrocnemius and soleus and the deep posterior compartment contains the tibialis posterior, flexor digitorum longus, flexor hallucis longus, the peroneal artery and vein, and the posterior tibial artery and vein (Fig. 10.1). The deep and superficial posterior compartments are separated by the deep transverse fascia of the leg.
Compartment syndrome is an exercise induced leg pain which can affect several structures (Table 10.1). Direct bone pain may occur with a stress fracture or stress reaction, and indirect bone pain can come from traction through muscle fascia causing inflammation of the periosteum. Periosteal inflammation (periosteal contusion) may also occur through direct trauma such as a kick to the shin. Muscular pain may be acute immediately following exertion, or chronic building over time. Chronic pain may eventually become noticeable even at rest. Neural compression may be local or pain referred from nerve root compression in the lumbar sacral spine. Finally, vascular signs may exist in cases of popliteal artery entrapment.
Table 10.1 Structures affected in compartment syndrome
MTSS – medial tibial stress syndrome.
After Padhiar (2009).
Anterior compartment syndrome
Anterior compartment syndrome involves pain in the anterior lower leg (Fig. 10.2A), which is increased in resisted dorsiflexion. There is usually a history of a sudden increase in training intensity, frequently involving jumping or running on a hard surface. The anterior compartment muscles swell, and in some cases hypertrophy occurs. The fascia covering the muscles may be too tight and inflexible to accommodate the increase in size. As a consequence, when the muscles relax, their intramuscular pressure remains high and fresh blood is unable to perfuse the tissues freely. This decrease in blood flow leads to ischaemia with associated pain and impairment of muscle function.
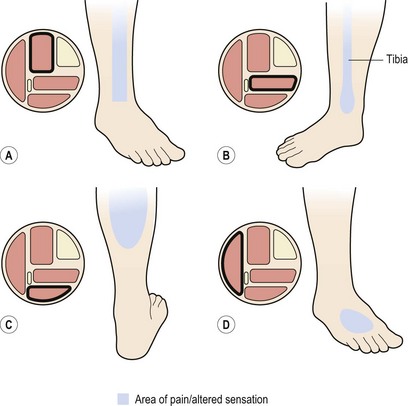
Figure 10.2 (A) Anterior compartment syndrome. Pain on the anterolateral aspect of the lower leg. (B) Deep posterior compartment syndrome (medial tibial stress syndrome). Pain over distal third of tibia (see also Fig. 10.3). (C) Superficial posterior compartment syndrome. Pain within the calf bulk. (D) Lateral compartment syndrome. Loss of sensation over the dorsum of the foot.
Usually, when a muscle contracts, its blood flow is temporarily stopped. Arterial inflow occurs once more between the muscle contractions as the intramuscular pressure falls. Normal resting pressure within the tibialis anterior in the supine subject is about 5–10 mmHg, increasing to as much as 150–250 mmHg with muscle contraction. Muscle relaxation pressure, that which occurs between repeated contractions, is between 15 and 25 mmHg in the normal subject, but in athletes with anterior compartment syndrome pressures may rise to 30–35 mmHg and take up to 15 minutes to return to normal values (Styf, 1989).
The intracompartment pressure (ICP) may be measured using a catheter introduced into the painful compartment via a cutting needle (medicut or similar). Saline and heparin are flushed into the area and the catheter is linked to a pressure transducer. A non-invasive technique has been developed which uses a 5 mm diameter indenter to measure quantitative hardness of the shin compartment to objectively measure the tissue tension which clinicians routinely measure subjectively. The use of this non-invasive compartment syndrome evaluator (NCSE) correlates well (Pearson correlation coefficient of 0.84) with direct measurement using a catheter (Steinberg, 2005).
Keypoint
In anterior compartment syndrome the resting pressure within the tibialis anterior can be 50% greater than normal.
The exact cause of the condition is not known. Hypertrophy may be one factor, as the condition occurs frequently when training intensity is increased. However, bodybuilders rarely suffer from the condition, so the rate rather than the amount of muscle hypertrophy may be important. Microtrauma and excessive stress to the blood capillaries and lymphatics may give rise to inflammation and in some cases myositis in the area (Styf, 1989).
Lateral compartment and superficial peroneal nerve
This is an unusual cause of shin pain and occurs when the peroneal muscles are affected, usually by hyperpronation (Brody, 1980). The condition may have existed for some time but is brought to the fore when running begins. Again, there is ischaemia and pain, but in addition the superficial peroneal nerve may be compressed as it emerges from the lateral compartment.
The nerve lies deep to the peroneus longus and then passes forwards and downwards between the peronei and the extensor digitorum longus. It pierces the fascia in the distal third of the leg where it divides into medial and lateral branches to enter the foot. Entrapment may occur if muscle herniation or fascial defect exist. In addition, ankle sprain, fasciotomy and an anomalous course of the nerve have been suggested as contributory factors (Styf, 1989). Clinically, the patient presents with loss of sensation over the dorsum of the foot, especially the second to fourth toes (Fig. 10.2D). Certain resting positions may compress the nerve and bring on the symptoms. To test the nerve, it is compressed over the anterior intermuscular septum 8–15 cm proximal to the lateral malleolus while the patient actively dorsiflexes and everts the foot. Tinel’s sign, involving local percussion over the compression site, may be positive.
Posterior compartment
The superficial compartment contains the soleus and gastrocnemius. These muscles are usually affected by trauma rather than ischaemia, and are dealt with separately below. Pain occurs within the calf bulk (Fig. 10.2C) and is increased with resisted plantarflexion.
The deep posterior compartment contains tibialis posterior, flexor digitorum longus and flexor hallucis longus, and is the most common site for shin pain. Pain in this region is usually experienced over the distal third of the medial tibia (Fig. 10.2B) and represents medial tibial stress syndrome (MTSS; Mubarak et al., 1982). The exact site of pain will vary depending on the specific structures affected, and Detmer (1986) described a system of classification involving three types of chronic condition (Fig. 10.3).
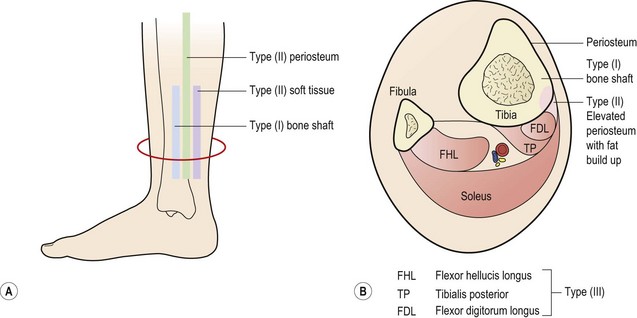
Figure 10.3 Medial tibial stress syndrome. (A) Painful areas of palpation. (B) Cross-section of structures affected.
After Detmer, D.E. (1986) Chronic shin splints: classification and management of medial tibial stress syndrome. Sports Medicine, 3, 436–444. With permission.
Definition
Medial tibial stress syndrome is pain over the distal third of the inner aspect of the tibia. The disorder may affect the bone, periosteum (bone membrane), fascia or muscles.
Type I involves microfractures or stress fracture of the bone itself. The patient is usually a runner who has recently increased his or her mileage. The stress imposed by the sport exceeds the ability of the bone to adapt and remodel. The condition may present as a stress fracture showing a concentrated positive uptake in a single area on bone scan and point tenderness to palpation (type IA), or as a diffuse area along the medial edge of the tibia giving more generalized pain (type IB). In chronic conditions which have existed for some time, the tibial edge may be uneven due to new bone formation.
Type II medial tibial stress syndrome involves the junction of the periosteum and fascia, and occurs particularly in sprinters and those involved in jumping activities. Pain is maximal just posterior to the bone, and has often persisted for a number of years. Initially, pain occurs only with activity, but as the condition progresses discomfort is felt with walking and even at rest. In this condition compartment pressures may not be elevated, and the periosteum is unchanged. One explanation is that the periosteum is traumatically avulsed from the bone by the action of soleus through its attachment to the fascia. During the chronic stage of this condition, adipose tissue has been found, during surgery, between the periosteum and underlying bone (Detmer, 1986). In the early stages of the condition the periosteum may heal back with rest, but when the condition becomes chronic, it is unable to heal, and continues to cause pain when stressed by activity.
The type III condition involves ischaemia of the distal deep posterior compartment and presents as a dull aching over the posterior soft tissues brought on by exercise. Intramuscular pressures are elevated as with anterior compartment syndrome (see above), and remain elevated after exercise.
Traction from muscle attachment is the most likely cause of the condition, the attachment of tibialis posterior normally being associated with the condition. However, both the soleus and flexor digitorum longus may also be implicated. The medial fibres of soleus and the attached fascia extend over the deep compartment forming a region known as the soleus bridge, an area implicated in MTSS. Excessive pronation is thought to be a significant factor in the development of the condition. It is argued that the muscles attaching to the posterior border of the tibia become overactive in an attempt to slow the rate of pronation and limit its degree. This deceleration results in eccentric overload and the consequent traction stress.
Management
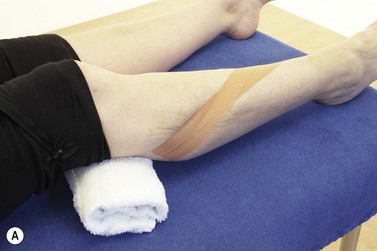
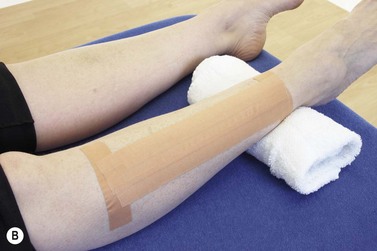
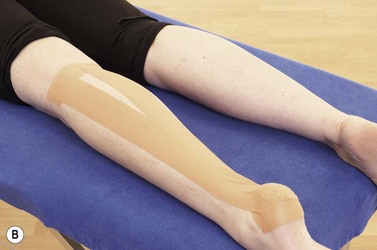
Initial management of shin pain involves a reduction of the stresses which caused the condition in the first place. This involves accurately identifying the structures affected and taking a thorough history of causal factors, particularly stresses imposed during training. Temporary pain relief may be achieved using deload taping to limit the fascial traction thought to be part of the pathology. Taping is applied in a spiral beginning on the lateral aspect of the ankle just above the lateral malleolus. The tape is wound around the shin going behind the calf and emerging once more onto the front of the tibia. As the tape is applied it is pulled proximally to unload the fascia (Fig. 10.4A). Another approach is to place anchors just below the knee and just above the ankle, and connect the anchors with 2 or 3 strips of zinc oxide taping gathering the skin up to take tension away (Fig. 10.4B). Both tapes alter the fascial loading and give temporary relief only.
Biomechanical assessment of the lower limb is mandatory and prescription of orthotics should be made where necessary. Initially, rest and anti-inflammatory modalities are used to allow the acute inflammation to settle, but external compression and elevation of the limb may exacerbate the problem (Di Manna and Buck, 1990). If training stresses can be modified, and the condition has been identified early enough, this may be all that is required.
In chronic conditions in which conservative management has failed, decompression by fasciotomy or fasciectomy may be called for. With fasciotomy the fascia of the affected compartment is surgically split along its length. The procedure is often performed on an out-patient basis, with two incisions being made, at the junction of the proximal and middle third of the leg and the middle and distal third. Athletes mobilize early and are often able to resume running after 3 weeks. Where fasciotomy fails, fasciectomy may be performed. Here, a longer open incision is made and a ribbon of fascia is removed. Because a longer incision is made, there is an increased risk of infection and dehiscence (incision bursting open). A longer rehabilitation period may be required, depending on incision healing time. Full return to sport may be expected in 8−12 weeks.
Definition
Fasciotomy is surgical splitting (cutting) of the fascia to reduce pressure within the fascial compartment. Fascietomy is the surgical removal of a ribbon of fascia.
Treatment note 10.1 Manual therapy for shin pain
Trigger point massage
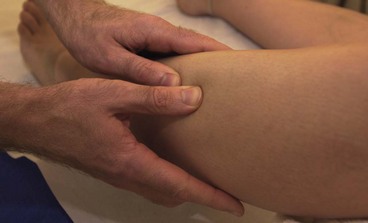
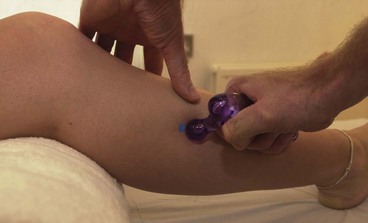
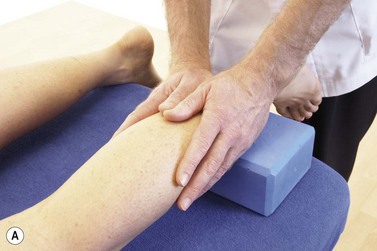
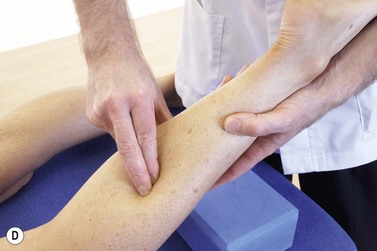
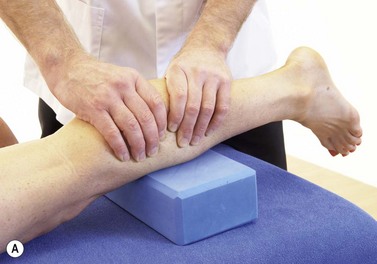
Trigger point (TrP) massage for anterior compartment syndrome focuses on the tibialis anterior and the extensor digitorum longus. The TrP for the tibialis anterior is located approximately one-third of the way distally from the knee and to the lateral side of the tibia. A muscle stripping technique can be used starting from half-way down the tibia progressing up towards the knee in a slow movement gradually progressing in depth (Fig. 10.5). The thickness of the muscle means that both thumbs must be used simultaneously or a pressure tool where ischaemic compression is used. The elbow is the tool of choice, direct compression is given and maintained for 3–10 seconds or until pain subsides.
Ice massage may be used over the same point, beginning from the knee and extending down the length of the tibia; a progressive stretch of plantarflexion and inversion is then used.
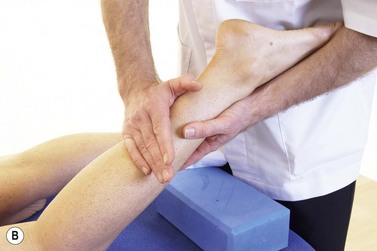
The long toe extensors (extensor digitorum longus (EDL) and extensor hallucis longus (EHL)) may be similarly treated. The EDL TrP is located approximately 8 cm distal and slightly anterior to the head of the fibula. The EHL is located at the junction of the middle of the distal thirds of the lower leg (Fig. 10.6). Home stretching may again be used, this time forcing the foot into plantarflexion and the distal toes into flexion.
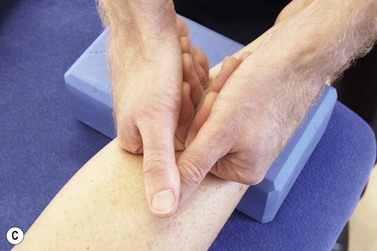
For lateral compartment syndrome, the peroneus longus and peroneus brevis muscles are targeted. The TrP for peroneus longus is approximately 2–4 cm distal to the fibular head, close to the shaft of the fibular itself, while that of the peroneus brevis is located at the junction of the middle and distal third of the lower leg.
Ischaemic compression may be used with the thumbs and ice stretch may again be given, this time stretching the foot into plantarflexion and inversion (Fig. 10.7).
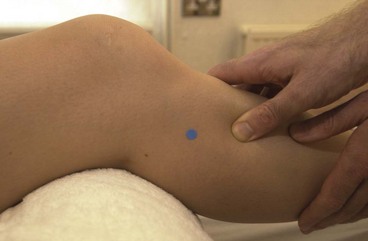
Posterior compartment syndrome and medial tibial stress syndrome require treatment of the flexor digitorum longus (FDL) and tibialis posterior along the lower third of the posterior edge of the tibia. Where the muscle itself is stressed, the TrP is targeted on the medial border of the upper tibia. The patient lies in crook side lying and the palpation point is between the medial edge of the tibia and the gastrocnemius muscles (Fig. 10.8). The gastrocnemius is pushed posteriorly and the pressure is then applied downward and then laterally. For the flexor hallucis longus (FHL), the patient may be treated in prone and this time the thumb is positioned lateral to the mid-line, pressing on the edge of the soleus at the junction between the middle and lower thirds of the lower leg.
Self-help trigger point methods
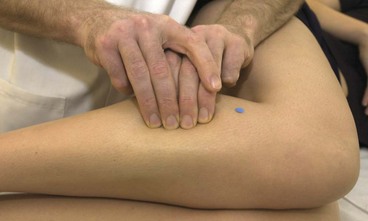
The athlete may be taught to use trigger point therapy as a self-treatment to relieve pain as it occurs. For the tibialis anterior, the easiest method is to use the heel of the opposite foot (Fig. 10.9). The movement is on the lateral edge of the tibia and begins at mid-shin level, the pressure being in a continuous sweep towards the lateral edge of the knee.
For the peroneal muscles, pressure may be given by the index finger, supported by the middle finger or by a single flexed knuckle (Fig. 10.10). For the flexor digitorum longus and tibialis posterior, one or two fingers may suffice. Alternatively, hands may wrap around the shin and both thumbs may be used to apply to the peroneus longus.
Where pressure on the trigger points is too tiring for the hands, apparatus such as the ‘backnobber’ (Physiomed, Manchester, UK) can be used to create more force (Fig. 10.11).
Dry needling techniques for trigger point therapy
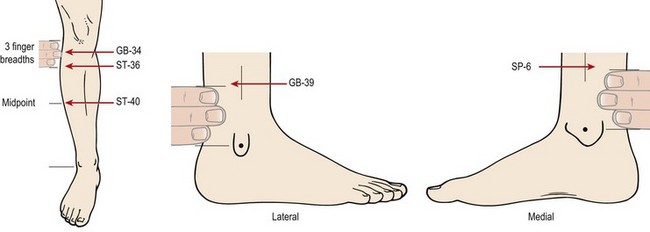
For anterior tibial syndrome, traditional acupuncture points on the stomach meridian may be used. Point ST. 36 located three finger breadths below the lateral aspect of the knee and point ST. 40 located midway between the lateral maleolus and the knee joint line may be used. Lateral compartment syndrome responds to needling along the gall-bladder meridian. GB 34 located distal and lateral to the head of the fibula and GB 39 located three finger breadths above the apex of the lateral maleolus on the edge of the tibia may be used.
For medial tibial stress syndrome, the point Sp. 6 may be used; located 3 finger breadths above the apex of the medial maleolus on the edge of the tibia, this point is directly opposite to GB 39 (Fig. 10.12). See Norris (2001) for specific details.
Stress fractures
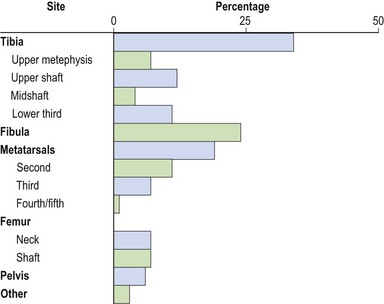
Over 50% of all stress fractures occur to the tibia and fibula (McBryde, 1985) with the remaining sites being mostly to the lower limb (Fig. 10.13). Stress fractures are usually the end point in a sequence of overuse. A number of causal factors usually coexist to begin the development of the condition. Training errors may account for 60–75% of such injuries in runners (McBryde, 1985). Common faults include high intensity work carried out for too long with an inadequate recovery, for example a distance runner who suddenly increases mileage. Faulty footwear which fails to attenuate shock and exercising on unforgiving surfaces will also contribute to lower limb pathology. These factors, coupled with an underlying malalignment problem of the lower limb (or biomechanical faults in technique of upper limb actions, see Chapter 5), will exacerbate the problem. With novice runners, an additional factor is muscular weakness in the lower extremity leading to a reduction in shock-absorbing capacity of the soft tissues. In each case, the overload on the tissues exceeds the elastic limit, causing a plastic deformation of bone.
Very often, the stress fracture is a direct result of a change of some type – in the athletes themselves, in the environment in which they train, or in the activity (Garrick and Webb, 1990). In terms of the athlete, stress fractures often emerge following the onset of a growth spurt or at the menopause as a result of the large body adaptations occurring at these times. Similarly, following illness, the body must be allowed time to readapt to training demands. Environmental changes, such as new clothing (shoes) or a new playing surface, will also require time to allow tissue adaptation, and failure to allow for this may lead to tissue breakdown, of which stress fractures are one type. Finally, alterations in the quality or quantity of a training programme itself will require a period of adaptation.
Far from being inert, bone is a dynamic tissue which is continually remodelling in response to mechanical stress. A balance usually exists between bone proliferation and reabsorption, which maintains the bone integrity. The result of athletic activity is normally that bone strengthens, but if unbalanced stresses cause bone reabsorption to exceed proliferation, the bone weakens.
Keypoint
Exercise (loading) normally causes microscopic damage within bone, and the body responds by producing stronger bone material. If the amount of damage caused by loading is greater than the ability of the bone to restrengthen, stress fracture occurs.
Examining excessive running and jumping in rabbits, Li et al. (1985) demonstrated that osteoblastic activity occurred from 7 to 9 days later than osteoclastic activity. Remodelling began on day 2, with the haversian blood vessels dilating, and by day 7 osteoclastic activity was noted in the bone cortex. New bone formation began in the periosteum by the 14th day of excessive stress. The adaptation to this excessive stress was reabsorption which occurred for some time before the formation of new cortical bone, thus weakening the bone structure. Abnormal x-rays were not found until day 21 after the stress was imposed.
Angular stresses, in particular, may cause failure of the bone with a resultant stress fracture. Two theories are generally accepted for the mechanisms by which stress affects bone. The first (fatigue) proposes that training which is too intense causes the muscles to fatigue so that they are no longer able to support the skeleton and absorb shock. The strain passes to the bone, causing the fracture. The second theory (overload) suggests that when certain muscles contract, they cause the bones to bend slightly. Training which is too intense will exceed the capacity of the bone to recover from this stress.
Signs and symptoms
The main symptom of a stress fracture is pain. This has been categorized into four types, depending on its characteristic (Puffer and Zachazewski, 1988). With type I pain, the athlete only feels discomfort after activity. With type II and III pain, discomfort is felt while training, but with type II this does not restrict activity. Type IV pain is chronic and unremitting in nature. Further symptoms include warmth and tenderness over the injured area, made worse by sporting activity and better with rest. Swelling may be evident in the later stages of the condition if the bony surface is superficial. Accuracy of palpation when assessing bone pain is vital. The tenderness of a stress fracture is usually well localized, whereas that of compartment syndrome is more diffuse. Initially radiographs are usually negative, it taking at least 2 weeks for x-ray changes to be apparent (Li et al., 1985; Rzonca and Baylis, 1988). Local periosteal reaction and new bone growth may be seen after 6 weeks in long bone (Markey, 1987) and compressive stress fractures in cancellous bone are sometimes visible after 24 hours (Puddu et al., 1994). Taunton and Clement (1981) found radiographs to be positive in only 47.2% of their cases, while bone scan was accurate in 95.8%. Bone scan is normally revealing at the onset of symptoms and is generally more reliable. Phosphate labelled with technetium-99 m is incorporated into osteoblasts and a hot spot appears over the active area, 6–72 hours after the onset of pain (Puddu et al., 1994).
Pain may be produced over the superficial fracture site by vibration. This may be produced from a tuning fork or ultrasound unit (Lowden, 1986), and is generally of more use in low risk areas, such as the foot and shin. Where there is a risk of complication through displacement (such as in the neck of the femur) bone scan with possible surgical intervention may be more appropriate (Fig. 10.14).
Treatment
Treatment of a stress fracture is primarily that of rest. As a general guide, with type I or II pain the workload should be reduced by 25% and 50%, respectively. Total rest is called for where type III pain is experienced, and type IV pain requires immediate medical investigation (Table 10.2). In the more severe conditions, rest should be total, because even allowing an athlete to train the upper body will often result in ‘just trying the leg out’ in the gym. Training should not resume until the athlete has been totally pain free for 10 days. It should be emphasized to the athlete that this means at the end of each day he or she should go to bed having felt no pain over the injured area during that day.
Table 10.2 Pain classification
| Classification | Characteristics | Action |
|---|---|---|
| Type I | Pain only after activity | ↓ workload by 25% |
| Type II | Pain during activity but not restricting performance | ↓ workload by 50% |
| Type III | Pain during activity restricting performance | Total rest |
| Type IV | Chronic unremitting pain at rest | Splint/cast Medical investigation |
After Puffer and Zachazewshi (1988).
The timescale of healing will vary depending on the site of injury. With reduced activity, fibular stress fractures will normally heal in 4–6 weeks, tibial stress fractures in 8–10 weeks and femoral neck stress fractures in 12–16 weeks (Reid, 1992).
When activity is resumed, the athlete should be closely monitored, and activities stopped if any pain occurs. Return to sport should be progressive and varied. Different speed, running surfaces and activities should all be used to spread the emphasis of training. Alternative training should be used, to reduce the weight bearing on the limb. Swimming and cycling may be utilized to restore cardiopulmonary fitness, for example.
Tennis leg
Of the sural muscles, it is the gastrocnemius which is usually injured. The soleus is infrequently affected by trauma being a single-joint muscle, but is more usually the victim of temporary ischaemia, giving rise to superficial posterior compartment syndrome. Both conditions must be differentiated from neural and vascular conditions. Pain in the calf may be referred from the lumbar spine or entrapment of the sciatic nerve further down, the S2 dermatome referring into the back of the thigh and calf. An inability to raise onto the toes of one foot in standing is a test for S1-2 nerve root entrapment (tibial nerve) in a case of low back history in the absence of calf symptoms. Neural involvement can be differentiated using the straight leg raise variations and slump test (see Chapter 14). Vascular symptoms include claudication usually from popliteal artery entrapment (see below) although in older patients calf and thigh claudication may occur in the presence of atherosclerotic disease.
True tennis leg is a strain of the gastrocnemius muscle itself, normally involving the medial head at its musculotendinous junction with the Achilles tendon. The history of injury is usually a sudden propulsive action, such as a lunge or jump as the athlete pushes off from the mark. Women are more commonly affected, and the athlete is typically over 30 years of age. The condition is often described as a rupture of the plantaris, more by tradition than anything else, because this muscle is rarely the cause of symptoms. A gastrocnemius strain usually occurs between the middle and proximal third of the muscle, with the medial belly more often affected. Palpation reveals a painful area, and with injuries affecting larger areas of the muscle, palpable scarring may be felt as tissue repair progresses.
Keypoint
Calf muscle strain usually affects the gastrocnemius muscle rather than the soleus. The medial head of the muscle is more commonly affected, at the junction of the middle and proximal third.
The athlete often feels something ‘go’ in the back of the leg, as though he or she were hit from behind. Pain and spasm occur rapidly, preventing the athlete from putting the heel to the ground. Later, swelling and bruising develop distal to the injury site, peaking at 48 hours after injury. There is local tenderness over the medial head of gastrocnemius at the junction between the middle and proximal third of the calf. Pain to passive stretch is worse with the knee straight than with it flexed.
Initial treatment is to immobilize the calf to prevent further tissue damage. Some authorities recommend a plantarflexed position with a 2–4 cm heel lift (Cyriax, 1982), while others claim that a 90° neutral ankle position gives a better result by preventing contraction and tightening the fascia to limit the spread of bruising (Garrick and Webb, 1990). Pain and the amount of tissue damage is usually the deciding factor. Partial ruptures are best immobilized non-weight bearing in plantarflexion to approximate the tissue, whereas less serious injuries can be prevented from shortening by adopting a neutral resting position. Taping has been shown to reduce gastrocnemius activity (Alexander et al., 2008). Applying undertaping and taping reduced the muscle activation measured by the Hoffman reflex (H-reflex) by 19% for the medial gastrocnemius and 13% for the lateral gastrocnemius.
Compression from an elastic bandage limits the formation of swelling, and strength and flexibility is maintained by starting rehabilitation early. Massage involving calf kneading and transverse frictions ensures adequate broadening of the muscle fibres. Stretching to the gastrocnemius begins in the long sitting position. A towel or band is placed over the foot and the athlete gently pulls the foot into dorsiflexion. Stretching with the toes on a block in a partial, and later full weight-bearing position is the exercise progression. Strength is regained by utilizing a comparable long sitting position but substituting an elastic band for the strap. The foot is pressed into plantarflexion against the resistance of the band. Heel raises are performed in a standing position, initially from the floor and later with the heel on a 2–3 cm block.
Gait re-education is used to encourage equal stride length and the adoption of a normal heel-toe rhythm avoiding external rotation of the leg. When the calf is pain free, strength activities give way to power movements, to build up the fast twitch nature of the muscle (Ng and Richardson, 1990). Gentle jogging, jumping and skipping are used and progressed to plyometrics. The athlete should be instructed to jump and land correctly. The toes should be the last point to leave the floor in a jump and the first to contact the floor on landing. Toe contact is followed by progressive lowering through the foot with the knee and hip flexing as the heal touches the floor to minimize shock. Flat foot landing and remaining on the toes must be avoided (Fig. 10.15). Slow squats raising up onto the toes at the top point give way to toe springing actions with and without weight resistance (dumb-bells or a weighted jacket). Leg drives from a mark are useful, as are ‘side hops’ and ‘hop and twist’ actions over a bar.
Popliteal artery entrapment
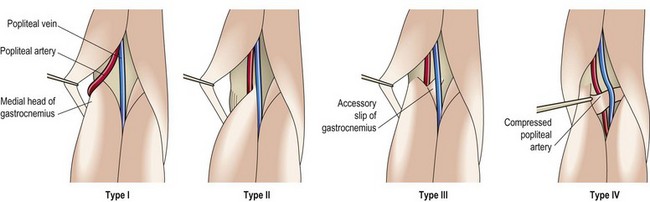
Popliteal artery entrapment syndrome (PAES) may be either structural or functional in nature (Stager and Clement, 1999). Structural changes (Fig. 10.16) include abnormal attachment of the medial head of gastrocnemius and additional tendinous remnants derived from the medial head. In long standing cases arterial wall degeneration may occur. Functional entrapment occurs when the popliteal structures appear normal, but compression occurs during activity. Differentiation of PAES during exercise induced calf pain must be made from compartment syndrome. Pain occurs during exercise in both conditions, but with PAES pain stops immediately on cessation of exercise, whereas compartment syndrome pain typically dies down slowly, taking 30−60 minutes to ease after exercise. Functional PAES is more normal with younger strength trained athletes and it is thought that plantarflexion compresses the artery between the underlying bone and muscle in the presence of hypertrophy of the gastrocnemius muscle. Prevalence of functional PAES has been estimated to be as high as 50% in the normal population (Pillai, 2008).
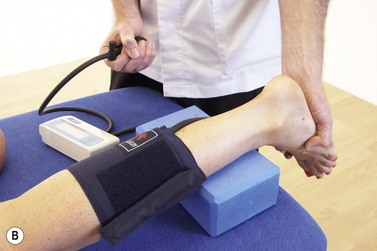
Although pain is the prevalent symptom, signs of ischaemia such as temperature changes, paraesthesia and skin discolouration may also occur. Initially evaluation is through the absence of distal pulses after exertion. Further information may be gained using the ankle to brachial pressure index (ABPI), the ratio of the blood pressure (BP) in the lower legs to that of the arms. The patient rests for 20 minutes (supine lying) and then an automatic blood pressure monitor (sphygmomanometer) is used to test systolic brachial pressure (arm) and systolic ankle pressure. The presence of the pulse is registered using Doppler ultrasound, and the BP at the point when the pulse is first detected is used. The ABPI index is calculated by dividing the ankle systolic BP by the higher of the two brachial systolic BPs. Normal index values are 0.90–1.0, with lower values representing moderate (0.75−0.90) or severe (0.75−0.50) disease. Values less than 0.50 are said to represent limb threatening vascular disease (Taylor and Kerry, 2002). Full examination is by angiography, but the condition may be missed if these tests are not carried out in resisted plantarflexion. Training modification is the treatment of choice initially, with surgical division of fascial bands in extreme cases.
Treatment note 10-2 Treatment of gastrocnemius tear
Taping
Anchors are placed over the heel and the back of the knee (Fig. 10.17A). Zinc oxide tape is then applied pre-stretched between the heel and posterior knee (Fig. 10.17B).
Massage
The patient lies prone with the foot over the couch end and the lower shin supported on a rolled towel or block to unlock the knee. Effleurage is applied using the flat palm (Fig. 10.18A) or webspace (Fig. 10.18B). Deep pressure may be applied using the supported fingers (Fig. 10.18C) and fascial stretch using the forearm placed across the muscle (Fig. 10.18D).
Exercise therapy
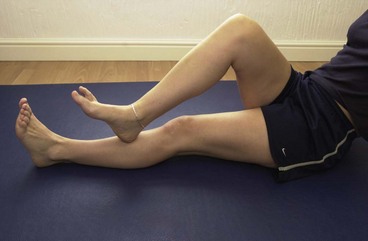
Stretching for the gastrocnemis is performed with the knee straight, taking the bodyweight against a wall (Fig. 10.19A). The stretch is increased by placing the toes on a slim book or block (Fig. 10.19B). Dynamic stretching is performed in a ‘sprint start’ position alternately plantarflexing and dorsiflexing the ankle (Fig. 10.19C). Heel raise activities using a dumbbell (Fig. 10.19D) may be used to strengthen the muscle and light jogging progresses to running, sprinting, jumping and bounding.
Achilles tendon
Achilles tendon pain (achillodynia) is a common condition in sport, with the incidence in runners being as high as 9% (Lysholm and Wiklander, 1987). Interestingly, the condition was relatively uncommon until the 1950s and is still very rarely seen in China (40 Achilles tendon ruptures among 2.5 million people over a 5-year period) (Jozsa, Kvist and Balint, 1989). The increased incidence of Achilles tendon rupture may be a result of the increasingly sedentary lifestyles seen in the Western industrialized countries (Kvist, 1994).
The Achilles tendon is the largest tendon in the body, being some 15 cm long and about 2 cm thick. It is able to sustain loads of up to 17 times bodyweight while utilizing only 13% of the oxygen supply of a muscle (Khan and Maffulli 1998). The tendon consists of connective tissue containing fibroblasts (tenoblasts) in a ground substance. The main extracellular component (80% of the dry weight of the tendon) is collagen, predominently type 1, with a small amount of type III. The ground substance consists of proteoglycans and glycosaminoglycan (GAG) chains. In normal tendon the amount of ground substance is minimal and not visible using light microscopy (Cook, Khan and Purdam, 2002).
Definition
Type I collagen is white and glistening and consists of large diameter fibres. It is found predominantly in skin and tendon. Type III collagen forms a delicate supporting network and is more common in young and repairing tissue. It is found commonly in skin and blood vessel walls.
Analysis of normal and ruptured Achilles tendon has shown that ruptured tendon has a greater percentage of type III collagen, making the tendon less resistant to tensile forces and increasing the risk of rupture (Maffulli, Ewen and Waterston, 2000).
The tendon originates from the musculotendinous junction of the calf muscles, the soleus inserting lower down on the deep surface of the tendon. The Achilles tendon gradually becomes more rounded as it travels distally, and flares out to insert into the posterior aspect of the calcaneum. The tendon is separated from the calcaneum by the retrocalcaneal bursa and from the skin by the subcutaneous calcaneal bursa. The tendon blood supply enters via the deep surface, making injection of any vessel abnormality with sclerosant more difficult than in other regions. Several structures are within the same body part and so should be considered in the differential diagnosis of Achilles tendon pain (Table 10.3).
Table 10.3 Structures to consider in differential diagnosis of Achilles tendon pain
Keypoint
The retrocalcaneal bursa separates the Achilles tendon from the calcaneum (heel bone). The subcutaneous calcaneal bursa separates the tendon from the skin.
The force from the Achilles tendon is delivered not through its insertion into the calcaneum, but via the point of contact on the posterior aspect of the calcaneum over the retrocalcaneal bursa (Reid, 1992). With increasing plantarflexion, the tendon ‘unrolls’ in such a way that this contact point moves lower down. In this way, the lever arm is maintained throughout the range of motion (Fig. 10.20).
The whole tendon rotates through 90° as it descends, so that the medial fibres become posterior by the time the tendon attaches to the calcaneum. This rotation is thought in part to account for the elastic properties of the tendon, giving it an elastic recoil when stretched. In the running action, as the lower limb moves from heel strike to mid-stance, the Achilles tendon is stretched, storing elastic energy. At toe off, the tendon recoils, releasing its stored energy and reducing the work required from the calf muscles to propel the body forwards.
The Achilles tendon is surrounded by a soft membranous paratenon (peritendon) which is continuous proximally with the muscle fascia and distally with the calcaneal periosteum. The paratenon consists of the epitenon or inner layer which lies directly over the achilles tendon itself, and the paratenon proper which comprises the outer layer. The paratenon does not have a synovial layer in contrast to the tendon sheaths of the hand, for example.
On the medial and lateral aspects of the dorsal surface there are thin spaces between the tendon and skin. These spaces are filled with thin gliding membranes covered in lubricating mucopolysaccharides. The membranes move freely over each other and greatly reduce friction. On the ventral side of the tendon there is fatty areolar tissue and connective tissue containing blood vessels.
The blood supply to the tendon is from either end and from the paratenon itself, but a relatively avascular zone exists between 2 and 6 cm proximal to the tendon insertion. Under normal circumstances, blood vessels do not travel from the paratenon to the tendon substance, and removal of the paratenon does not seem to compromise the blood supply (Williams, 1986). Tendon tissue in general has a low metabolic rate, and this, coupled with the poor blood supply to the tendon, means that the structure has a slow rate of healing. Although anatomically the blood flow to the tendon seems poor, investigation using laser Doppler has found that the blood supply is actually of a similar volume throughout its length (Astrom and Westlin, 1994). Changes in tendon blood flow are however important to tendon pathology (Malliaras et al., 2008).
Injuries to the Achilles tendon fall broadly into one of two categories. First, those which affect the tendon substance (partial or complete rupture), and, second, injury to the surface of the tendon and its covering (tendinopathy or peritendinopathy).
Tendinopathy
The term tendonitis (Cyriax, 1982) implies an inflammation within the tendon substance. The pathology of chronic tendon injury is that there is no triphasic inflammatory response. As such the term tendinopathy is now used to describe local pain and swelling around but not within the tendon. Tendinosus is a term used to refer to histological changes which have been confirmed using biopsy or scanning.
Definition
Tendinopathy refers to a combination of tendon pain and local swelling. Tendinosis is present when these changes are verified on tendon biopsy or through ultrasound scanning.
Tendinopathy presents as pain and thickening of the Achilles tendon of gradual onset. Histological changes show a failed healing reponse which leaves the tendon open to further injury. There is an increase in the amount of ground substance with a greater proportion of proteoglycans. Collagen bundles are often disrupted and the relative amount of type I collagen reduces while that of type III actually increases. This latter type is thought to be produced as a response to injury (Cook, Khan and Purdam, 2002), but its structure is thinner and weaker than type I, hence the tendon has reduced load tolerance and an increased risk of further disruption. Fibroblast cells migrate from the peritenon into the tendon substance and the total number of tenocytes (tendon cells) increases. The cells become more rounded in appearance and are metabolically active having an increased number of protein synthesizing organelles.
One of the most significant changes is an increase in vascularity, most easily seen on colour Doppler ultrasound. The new vessels have a small lumen and are twisted in appearance.
In peritendinitis the paratenon is inflamed and often thickened, and there is local oedema and crepitus. The condition can exist in isolation to tendinopathy and is normally due to repeated actions especially with tendon compression. Kager’s triangle (the space between the inner surface of the Achilles tendon, the deep flexors and the calcaneus) is obliterated. Hard scarred bands appear within the paratenon and adhesions develop between the Achilles tendon and surrounding tissue. Kvist et al. (1988) showed that the fatty tissue surrounding the tendon was still thickened and swollen, after nearly 2 years in subjects with paratenonitis. Connective tissue had increased, and local blood vessels had degenerated or been obliterated, suggesting the presence of immature scar tissue.
Tendon examination
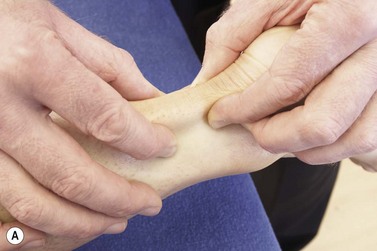
Specific examination of the Achilles tendon is carried out with the patients in prone lying with the foot over the couch end (shin supported on a rolled towel) and in standing and/or the position which reproduces pain (sprint start, for example). Tendon rupture is discounted from the history (sudden onset/trauma for rupture, gradual onset/overuse for tendinopathy) and using the calf squeeze test (see below). The tendon sheath can be differentiated from the tendon proper using the ‘painful arc’ sign (Williams, 1986). Using a pincer grip (Fig. 10.21A) the painful area of the tendon is palpated while the foot is actively plantarflexed and dorsiflexed. As the tendon moves through the sheath, the sheath will stay still as the foot is moved, but the tendon will move away from the palpating fingers. The painful arc test is positive (confirming tendinopathy) if the pain moves away from the fingers (Fig. 10.21B). For the posterior impingement test the pain in midrange plantarflexion is compared to that of full range plantarflexion. The therapist places one hand beneath the patient’s forefoot to resist plantarflexion and load the tendon. Pain which occurs in a midrange position is compared to that of full plantarflexion which compresses the posterior structures. The impingement test is positive if Achilles pain is increased with resisted plantarflexion.
Management
Initial management of tendinopathy or peritendinitis aims at reducing local oedema. Rest and ice are used in conjunction with electrotherapy modalities aimed at reducing pain and resolving inflammation. Ice must be used with caution due to the risk of producing an ice burn over the tendon or malleoli. Wet ice enclosed in a moist cloth is preferable to an ice pack. Commercial ice packs should never be placed directly onto the skin.
Several forms of manual therapy may be useful in addressing these conditions. Transverse friction massage has been used extensively (Cyriax and Cyriax, 1983), for both the teno-osseous (T/O) junction and tendon itself. The T/O junction may be frictioned with the patient prone and the foot plantarflexed (Fig. 10.22A). The therapist uses the side of his or her flexed forefingers to impart the friction, pulling the hands distally against the curved insertion of the Achilles tendon into the calcaneum. The musculotendinous junction is frictioned with the foot dorsiflexed, and the tendon gripped from above between the finger and thumb. The movement is perpendicular to the tendon fibres (Fig. 10.22B). The tendon sheath may be treated by placing the length of the finger alongside the tendon and pressing inwards. The forearm is pronated/supinated to impart the friction (Fig. 10.22C).
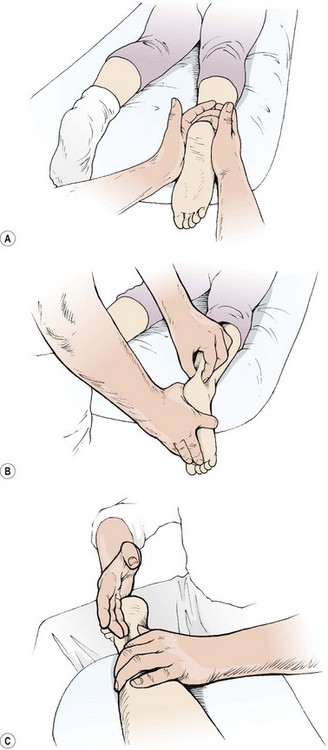
Figure 10.22 Transverse frictional massage for the Achilles tendon. (A) Teno-osseous junction. (B) Musculotendinous junction. (C) Tendon sheath/underside.
General massage may be used focussing on effleurage to relax the calf muscle and circular frictions along the tendon itself. Specific wringing actions (see Figs 10.18A,B and C and Fig. 10.24A) are useful both to test the tendon compliance as part of tissue assessment and for treatment itself. Specific soft tissue mobilization (SSTM) has been described (see Treatment note 2.3), and its use shown to be effective in the treatment of Achilles tendinopathy. The direction of the soft tissue mobilization was determined by reproduction of symptoms. A medial glide was used with the Achilles on stretch. This was progressed to gliding during isometric loading, and then gliding during dynamic through range loading against resistance (Christenson, 2007). Figure 10.23 shows SSTM of the Achilles.
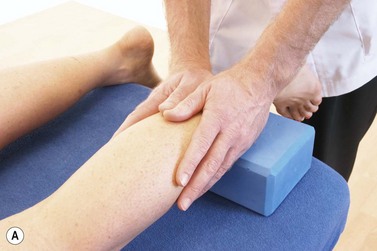

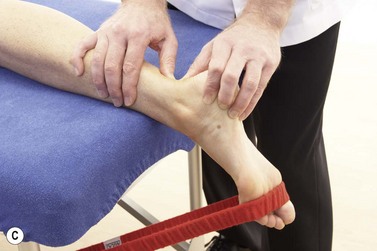
Figure 10.23 SSTM of the Achilles. (A) Calf massage, (B) Gastrocnemius and achilles stretch, (C) SSTM during resisted plantar flexion.
A heel raise is used to reduce the stretch on the tendon, and the calf may be strapped in plantarflexion. If pain is intense the patient should initially be non-weight bearing. A heel raise (built into an orthotic) may also be useful as the athlete returns to sport. Raising the heel can reduce the rate of eccentric loading during gait as well as the range of motion and it has been suggested that this may affect the strain response of the tendon (Hunter, 2000). In addition a medial heel post (wedge) used to control excessive subtalar pronation may reduce the shear stress on the tendon. In a study comparing lower limb kinematics of those with chronic Achilles tendon pain to controls, Donoghue et al. (2008a) found greater eversion and ankle dorsiflexion and less leg abduction in the symptomatic group. In a separate study of rear foot control for patients with Achilles tendon injury, Donoghue et al. (2008b) found a 92% reduction in symptoms when using a rear foot orthotic designed to reduce pronation. Determination of biomechanical faults in the lower limb and the subsequent use of orthotics in patients with chronic Achilles pain would seem mandatory.
Eccentric exercise in the management of tendinopathy
The use of eccentric loading in the treatment of Achilles tendinopathy was popularized in the late 1980s. The aim is to increase the tensile strength of the tendon by subjecting it to active lengthening and high tensile forces. Eccentric exercise also prepares the tendon for rapid unloading which has been associated with injury. The sudden release of force has been suggested to break interfibrillar connections within the tendon (Curwin, 1994). MRI has been used to assess Achilles tendon following eccentric loading. Shalabi, Kristoffersen-Wilberg, and Svensson. (2007) tested 25 subjects (mean age 51 years) using a 3-month eccentric calf muscle stretching training programme. Following the programme there was a 14% decrease in tendon volume with T1-weighted images reducing from 6.6 cm3 to 5.8 cm3. Collagen fibre synthesis has also been shown to change. Langberg et al. (2007) used a 12-week programme of heavy eccentric training in elite soccer players. Markers of type I collagen were measured using microdialysis, and increased from 3.9 µg/L prior to training to 19.7 µg/L following the 12-week programme. Sagittal thickness of the Achilles has been shown to reduce as a result of eccentric loading, and eccentric loading has been shown to induce a 4-fold greater thickness reduction than concentric training (Grigg, Wearing, and Smeathers, 2009). In this study average sagittal thickness reduced by 20% (4.4 mm to 3.5 mm) following eccentric training, and by 5% (4.5 mm to 4.3 mm) following concentric training. Tendon thickness did not return to post-treatment values until 24 hours after treatment. Training, which involves cyclic loading of this type, has been shown to exude water from the tendon core to the peritendinous space through straightening of crimped collagen fibres and reducing the interfibrillar space (Helmer et al., 2006; Screen: Cheng &, 2007).
Keypoint
Eccentric exercise causes the Achilles to exude water from the tendon core to the periphery, reducing cross-sectional area for up to 24 hours.
In a study of 200 Achilles tendinopathy patients treated by eccentric loading, 44% had complete pain relief and 43% showed a marked decrease in their symptoms (Stanish, Robinovich and Curwin, 1986). Comparing concentric to eccentric protocols, Niesen-Vertommen et al. (1992) showed better subjective pain scale scores for the eccentric group. Heavy eccentric loading has also been shown to be superior to surgery. The eccentric group achieved full function in 12 weeks compared to the surgery group timescale of 24 weeks, and pain response and strength deficit were better in the eccentric group (Alfredson et al., 1988).
Eccentric training may be given by performing a heel raise, rising on the uninjured leg and lowering on the injured side, both with the leg straight (gastrocnemius) and then bent (soleus). This type of programme has been shown to give relief in 90% of patients with mid tendon pain and pathology (Roos et al., 2004). Forty-four patients (mean age 45 years) were given a 12-week eccentric training programme and the results were measured using the Foot and Ankle Outcome Score. Significant pain reduction occurred which lasted for 1 year follow-up. As pain eases functional progression may be made using speed and power actions.
Eccentric training for the Achilles is performed using a step. The athlete lifts his or her bodyweight using both legs by plantarflexing the ankle. Weight is transferred to the painful side and the non-injured knee is flexed. Bodyweight is then lowered using the painful leg alone. Ideally athletes should perform 3 × 15 repetitions, twice daily on each day of the week (90 repetitions for each of the two exercises, giving 180 in total). Exacerbation of pain is to be expected as tissue repair is stimulated within the tendon. If pain does not occur, loading is likely to be suboptimal and more resistance should be applied.
Tendon substance
Focal degeneration (tendonosis) may occur in the tendon with athletes in their early 30s. The onset is usually gradual, giving highly localized pain. Microscopically there is proliferation of capillary cells and lacunae along the tendon fibres giving a loss of the normal wavy alignment of collagen. As we have seen this is a non-inflammatory condition resulting in haphazard collagen fibre orientation and relative absence of tenocytes, increased interfibrillar glycosaminoglycans (GAG) and scattered vascular in-growth. Collagen fibres fray and thin, losing their orientation and the quantity of type III collagen increases. There may be a reduction in fibre cross-linkage, reducing the tensile properties of the tendon (Movin et al., 1997; Hunter, 2000).
Tendon rupture generally occurs later in an athlete’s life, being more common from the late 30s to mid 40s. With partial rupture, a few fibres or nearly all of the tendon may be affected. The characteristic history is one of sudden onset. There is swelling, pain on resisted movement and the patient is unable to support his or her weight through the toes of the affected foot.
In complete rupture there is usually a single incident where the patient feels a sudden sharp ‘give’ in the tendon as though he or she had been struck from behind. Immediately afterwards there may be little pain but gross weakness. The patient is unable to take the weight through the toes, and the foot will hang straight downwards rather than being pulled into relaxed plantarflexion by calf muscle tone. Active plantarflexion is minimal, being accomplished by the peronei and posterior tibial muscles only. Three tests have been shown to be accurate diagnostically (Cleland, 2005): the calf squeeze test, the gap test and the Copeland test.
Without surgery, the tendon will heal, but very slowly. Granulation tissue forms poorly, partly due to repeated use, and partly as a result of the anticoagulant effect of synovial fluid contained by the unruptured paratenon (Williams, 1986).
Lesions of the tendon may be differentiated from those of the paratenon by using the ‘painful arc’ sign described above. When examining the patient it must be remembered that the Achilles tendon is painful to palpation normally, so the intensity of discomfort must always be compared with that of the contralateral limb.
Surgical management
Surgical management of Achilles tendon injuries carries a high rate of complication. Williams (1986) described complications in 64 out of 461 surgical procedures (13.9%), and Paavola, Orava and Leppilahti (2000) reported a similar percentage of 46 out of 432 patients (11%). These included mainly wound dehiscence (bursting), scar hypertrophy, wound infection and nerve irritation. Surgery may be called for where conservative management has failed and symptoms have persisted for more than 3 months.
Achilles tendon repair is the subject of considerable debate. Many authors claim that conservative treatment does not allow direct realignment of the torn tendon fibres and so the repair is less secure. In addition, they claim that re-rupture of surgically treated injuries is less likely (Turco and Spinella, 1987). Others argue that the risk of surgical complications is too great, and that the sophistication of rehabilitation is as important to the eventual outcome of the condition as whether the surgical or non-surgical route was chosen (Garrick and Webb, 1990). A meta-analysis of randomized controlled trials of treatment of Achilles tendon rupture concluded that open operative repair reduced the risk of re-rupture by 27% (statistically significant) compared to non-operative management. However, surgery was associated with a higher risk of complications. Use of a functional brace allowing early mobilization reduced the complication rate (Khan et al., 2005).
A number of repair methods have been described, including direct suture, reinforcement with plantaris or peroneus brevis tendon, and reconstruction using carbon fibre or polypropylene mesh.
Whatever the surgical method used, following immobilization the main problem is initially lack of flexibility to the Achilles tendon, ankle and subtaloid joint in particular. Intense rehabilitation of the foot, ankle and leg is needed to regain full function.
Rehabilitation following surgery
Early mobility following Achilles tendon repair is a vital factor to the success of the procedure. A typical rehabilitation period normally includes an equinus plaster for 8 weeks followed by a heel raise for a further 6 weeks (Apley and Solomon, 1993). However, Saw et al. (1993) showed improved mobility and strength and a reduction in complications using total immobilization in an equinus plaster for 1 week only. Following this, the plaster was removed and active dorsiflexion was practised in sitting. The plaster was replaced between therapy sessions. Sutures were removed after 2 weeks and the foot placed in a lower leg walker. Solveborn and Moberg (1994) showed similar results using a patellar tendon bearing cast with a metal frame attached for 6 weeks. The cast allowed pain-free motion of the foot while taking full bodyweight for walking. No complications were seen, with a reduction of skin/tendon adhesions and a rapid return to normal strength values.
On removal of the cast, stretching procedures are begun non-weight bearing, hooking a towel over the forefoot and gently pulling into dorsiflexion being sufficient. Partial weight-bearing activities in sitting are used to begin the closed kinetic chain exercises, with the athlete gently pressing into dorsiflexion using varying foot angles. The same starting positions are chosen for early static and later dynamic strength training using straps and elastic bands. Isokinetics is used for the calf specifically and the lower limb in general.
Walking re-education using parallel bars or sticks introduces functional movements and introduces skill work. When full weight-bearing activities are begun, the flexibility and strength work is progressed and augmented with skill training involving balance and coordination exercises (see Ankle rehabilitation, Chapter 11) and further walking re-education.
Both static and dynamic flexibility training is used from the earliest stages, and eccentric actions are begun as soon as possible (often in the pool to limit weight bearing) to mimic the true function of the Achilles tendon. When function has improved sufficiently, walking pace is increased and speed walking introduced, followed by on-the-spot and, later, small-step jogging. Straight running, zig-zags and circle running are all used. Thick-heeled training shoes are used, and gradually the heel is lowered. Toe walking progresses to jogging on the toes.
General fitness is maintained throughout the programme by cardiopulmonary activities in the pool (assuming good healing of the scar) and, later, static cycle riding, until the intensity of rehabilitation itself is sufficient to challenge aerobic fitness. Pain is the rate limiting factor for both exercise intensity and progression.
Assuming the surgery has been totally successful, the final stages of training must include the use of plyometric exercise to develop elastic strength. Poor results can be expected if the rehabilitation programme is stopped at the stage of heavy isotonic exercise and not progressed in terms of speed. Hopping and jumping actions are used on forgiving surfaces with progression to bounding.
Aetiology
A number of factors may combine to cause Achilles tendon pain, including training alterations, footwear and flexibility. Alterations in training type or intensity, such as a sudden increase in the distance run over a holiday period, or more intensive training in the lead up to a competition, can both produce problems. Only in recreational athletes is trauma the predominant cause. In the competitive athlete, as many as 90% of Achilles tendon injuries result from overuse in the form of microtrauma established over months, or, in some cases, many years (Kvist, 1994).
Footwear is an important factor to the development of this condition. Inadequate shock-absorbing properties stress the Achilles tendon during eccentric loading, a situation aggravated when an athlete has a rigid cavus foot. Good shock absorption of the shoe heel is important for athletes who exercise on a hard surface. In addition to reducing the impact at heel strike, the height of the heel will also reduce the stretch on the Achilles tendon. A rigid sole lengthens the ankle to forefoot lever arm, affecting Achilles tendon mechanics, but may be necessary to protect the first toe (see below). If the heel counter of a sports shoe is hard, it can abrade the Achilles tendon as the foot plantarflexes at toe off. A negative heel counter, or a soft tab, is to be encouraged.
Lack of flexibility in the calf and Achilles tendon may contribute greatly to injury. When flexibility is limited, the tendon loses some of its elastic recoil, making it more susceptible to damage through rapid eccentric loading of the calf in mid-stance. Flexibility exercises should be used for prevention and management of the subacute injury. Stretching may be performed with the forefoot on an angled block. The heel is kept on the ground and the hip is pressed forward, bringing the knee over the forefoot and increasing dorsiflexion. The exercise is first performed with the knee locked to stretch the gastrocnemius, and then with the knee flexed to 20° to stress the Achilles tendon and soleus. A static stretch is used and held for 20–30 seconds. The range of motion is generally greater when the tendon is warm, after a hot shower or bath for example.
Malalignment of the lower limb is probably a contributory factor to Achilles tendon pain, although, as such faults have generally been present for some time before the Achilles tendon condition started, it is unlikely to be the sole cause. Increased pronation of the foot stresses the medial aspect of the Achilles tendon. A 30° increase in rearfoot position has been shown to lengthen these fibres by 10% (Nichols, 1989). In addition, excessive tibial rotation caused by hyperpronation will twist the Achilles tendon fibres together, ‘wringing them out’. This will close any blood vessels between the tendon fibres, temporarily reducing an already poor blood supply.
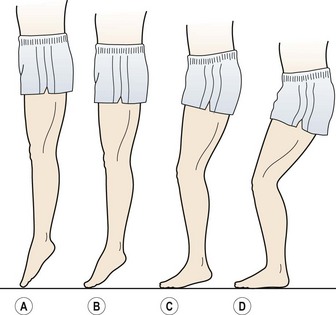
Viewing the athlete’s foot and lower limb from behind can be revealing. In an optimal alignment the Achilles tendon should appear vertical and the medial longitudinal arch be present. The gap between the floor and the navicular bone should be approximately one finger breadth. Excessive pronation (Fig. 10.25B) causes the Achilles to curve and deviate outwards with the medial longitudinal arch flatter (pes planus). Using a medial calcaneal wedge to control pronation at the subtalar joint (Fig. 10.25C) realigns the Achilles.
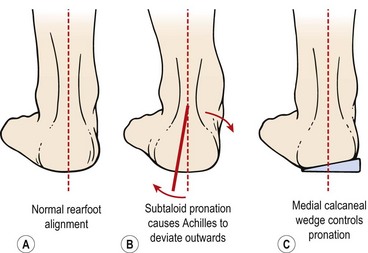
Figure 10.25 (A) Normal rearfoot alignment and the Achilles. (B) Subtaloid pronation causes Achilles to deviate outwards. (C) Medial calcaneal wedge controls pronation.
In an interesting survey of 109 runners with Achilles tendon problems, Clement, Taunton and Smart (1984) found 75% showed training errors, 56% demonstrated hyperpronation, 39% suffered inflexibility of the calf and 10% wore improper footwear.
Corticosteroid injections may affect ligament failure. Inhibition of collagen synthesis, reduction in the speed of tendon repair, lower elastic limit and fatty degeneration have all been described (Nichols, 1989). Various studies have shown an increased likelihood of Achilles tendon rupture after injection (Chechick et al., 1982; Kleinman and Gross, 1983; Urban, 1989). Cyriax (1982) argued that Achilles tendon rupture only occurred when an incorrect injection technique was used, the corticosteroid being introduced into the tendon substance rather than along its surface. However, Williams (1986) claimed that both techniques had been shown to precipitate rupture, and further argued that the benefits of such injections were relatively short-lived. In addition, he contested that in acute peritendinitis, similar results could be obtained using a local anaesthetic and hyaluronidase.
Accessory soleus muscle
Occasionally, athletes may present with an extra soleus muscle (accessory or supernumerary soleus) on one or both sides of the body. The condition occurs in 6% of the population and is more common in males than females with a ratio of 2 : 1 (Leswick et al., 2003). It is thought that the accessory muscle originates as a splitting of the embryonic cell cluster (anlage) in early development. The muscle is contained within its own fascial sheath with its origin typically on the distal posterior aspect of the tibia (Gordon and Matheson, 1973). Pain and swelling occur with increased tissue tension within the Kager triangle. Typically symptoms begin with an increase in training volume and are thought to represent a closed compartment ischaemia. Rest, ice and local massage may be used with advice on training modification. Persistent cases may respond to fasciotomy and debulking (Leswick et al., 2003). Ultrasound scanning reveals tissue of the same echogenicity as normal muscle with the appearance of muscle bundles often identified. Differential diagnosis must be made from Achilles tendinopathy, ganglion lipoma, sarcoma and haematoma.
Retrocalcaneal pain
Sever’s disease
Calcaneal apophysitis (Sever’s disease) may occur in adolescents during the rapid growth spurt, sometimes in association with Achilles tendon pain itself. The condition is more common in boys than girls, and occurs between the ages of 11 and 15 years. The posterior aspect of the calcaneum develops independently of the main body and is separated by an epiphyseal plate. The plate lies vertically and is therefore subjected to shearing stress from the pull of the Achilles tendon and through jarring stress in jumping. Pain frequently occurs during deceleration from running as well as take off and landing from a jump. Sever’s is inappropriately named as it is not a true disease entity, but rather a traction injury similar in many respects to Osgood–Schlatter’s disease in the adolescent knee.
On examination, there is tenderness to medial and lateral heel compression, but no noticeable skin changes. Radiographically, sclerosis and irregularity of the calcaneal apophysis is seen as a result of avascular necrosis (Caspi, Ezra and Horoszowski, 1989). Associated Achilles tendon tightness has been described, with the affected side showing 4–5° less passive dorsiflexion than the unaffected side (Mitcheli and Ireland, 1987). In addition, excessive foot pronation is also common in this patient group.
Initially, total rest is called for to allow the condition to settle. Later, any sporting activity which exacerbates the pain is avoided, and shock-absorbing heel pads are used in all shoes. Achilles tendon stretching is taught to restore dorsiflexion range, and foot position is corrected.
Haglund’s syndrome
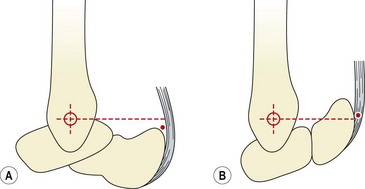
Haglund’s syndrome is present when an exostosis is found over the posterior lateral aspect of the calcaneum. Radiographically, a spur of bone is seen coming from the calcaneal insertion of the Achilles tendon, showing as a prominence of the posterior superior calcaneal angle (Wooten and Chandler, 1990). Assessment of radiographs may be made by comparing ‘parallel pitch lines’ (Pavlov et al., 1982). The first line joins the anterior and posterior calcaneal tubercles. A second line is drawn parallel to the first, but this time starting from the posterior lip of the talar articulation (Fig. 10.26). If the posterior superior calcaneal angle stands prominent to this second line, the diagnosis is positive.
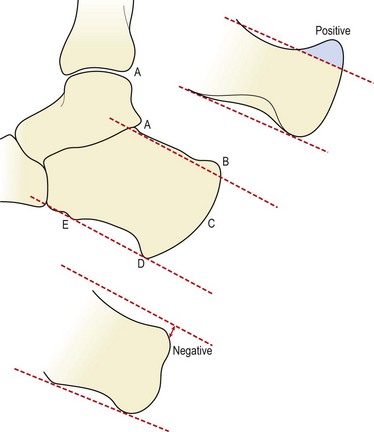
Figure 10.26 Parallel pitch lines to determine the position of the posterior superior calcaneal angle. A, Posterior lip of talar articulation. B, Posterior superior calcaneal angle. C, Posterior calcaneal tuberosity. D, Medial calcaneal tuberosity. E, Anterior calcaneal tuberosity.
From Rossi, F. et al. (1987) The Haglund syndrome: clinical and radiological features and sports medicine aspects. Journal of Sports Medicine, 27, 258–265. With permission.
In addition to the bony changes, a painful soft tissue swelling (pump bump) is noted at the insertion of the Achilles tendon. This swelling can vary in size and shape depending on the stage of the condition. Haglund’s syndrome commonly exists in association with retrocalcaneal bursitis, the inflamed bursa becoming visible on x-ray when low penetrating radiation is used (Rossi et al., 1987).
Contributing factors to the formation of Haglund’s syndrome include a hard edge to the posterior aspect of the shoe (heel tab) and a rearfoot varus. As the foot is plantarflexed, the hard heel tab will abrade the posterior aspect of the calcaneum. If a rearfoot varus exists, compensatory pronation will cause the heel to twist in the shoe and soft tissue pressure occurs over the posterior calcaneum. Although soft tissue changes could be expected from abrasion of this type, bony changes are less likely. The bony change itself could be the result of a secondary ossification centre in the calcaneus (Horn and Subotnick, 1989).
Management of retrocalcaneal pain is by pain relief until the condition settles. A horseshoe pad made from non-compressible rubber is placed over the painful calcaneal area, and held in place by an ankle sock. A heel raise may be used to reduce stress through the tendon onto the calcaneus, and gentle tendon stretching is taught. Electrotherapy modalities are used to alleviate pain and reduce inflammation.
Surgery includes bursectomy and resection of the superoposterior portion of the calcaneus, as far as the insertion of the Achilles tendon. Return to sport is relatively protracted, with athletes taking 3–5 months to begin full competition (Ghiggio, Nobile and Bronzo, 1993). With restoration of sport, athletes should still be advised to wear soft-back shoes to avoid local abrasion.
Alexander C., McMullan M., Harrison P. What is the effect of taping along or across a muscle on motoneurone excitability? A study using Triceps Surae. Manual Therapy. 2008;13(1):57-62.
Alfredson H., Pietila T., Johnsson P., Lorentzon R. Heavy load eccentric calf muscle training for the treatment of chronic Achilles tendonitis. American Journal of Sports Medicine. 1988;26(3):360-366.
Apley A.G., Solomon L. Apley’s System of Orthopaedics and Fractures, seventh ed. Oxford: Butterworth-Heinemann; 1993.
Astrom M., Westlin N. Blood flow in the normal Achilles tendon assessed by laser Doppler flowmetry. Journal of Orthopaedic Research. 1994;12(2):246-252.
Brody, D.M., 1980. Running injuries. Clinical Symposia 32 (4), Ciba Pharmaceutical Company, New Jersey.
Caspi I., Ezra E., Horoszowski H. Partial apophysectomy in Sever’s disease. Journal of Orthopaedic and Sports Physical Therapy. 1989;10(9):370-373.
Chechick A., Amit Y., Israli A., Horoszowki H. Recurrent rupture of the Achilles tendon induced by corticosteroid injection. British Journal of Sports Medicine. 1982;16:89-90.
Cheng V., Screen H. The micro-structural strain response of tendon. Journal of Material Science. 2007;42:8957-8965.
Christenson R.E. Effectiveness of specific soft tissue mobilizations for the management of Achilles Tendinosis: Single case study – Experimental design. Manual Therapy. 2007;12:63-71.
Cleland J. Orthopaedic clinical examination: An evidence based approach for physical therapists. New Jersey, USA: Icon learning systems; 2005.
Clement D.B., Taunton J.E., Smart G.W. Achilles tendinitis and peritendinitis: aetiology and treatment. American Journal of Sports Medicine. 1984;12(3):179-184.
Cook J.L., Khan K.M., Purdam C. Achilles tendinopathy. Manual Therapy. 2002;7(3):121-130.
Curwin S.L. The aetiology and treatment of tendinitis. In: Harries M., Williams C., Stanish W.D., Micheli L.J., editors. Oxford Textbook of Sports Medicine. Oxford: Oxford University Press, 1994.
Cyriax J. Textbook of Orthopaedic Medicine vol. 1. eighth ed London: Baillière Tindall, 1982.
Cyriax J.H., Cyriax P.J. Illustrated Manual of Orthopaedic Medicine. London: Butterworth; 1983.
Detmer D.E. Chronic shin splints: classification and management of medial tibial stress syndrome. Sports Medicine. 1986;3:436-446.
Di Manna D.L., Buck P.G. Chronic compartment syndrome in athletes: recognition and treatment. Athletic Training. 1990;25:1.
Donoghue O.A., Harrison A.J., Laxton P., Jones R.K. Lower limb kinematics of subjects with chronic Achilles tendon injury during running. Research in Sports Medicine. 2008;16(1):23-38.
Donoghue O.A., Harrison A.J., Laxton P., Jones R.K. Orthotic control of rear foot and lower limb motion during running in participants with chronic Achilles tendon injury. Sports Biomechanics. 2008;7(2):194-205.
Garrick J.G., Webb D.R. Sports Injuries: Diagnosis and Management. Philadelphia: W.B. Saunders; 1990.
Ghiggio P., Nobile G., Bronzo P. Haglund’s disease in athletes. Journal of Sports Traumatology. 1993;15:89-96.
Gordon S.L., Matheson D.W. The accessory soleus. Clinical Orthopaedics. 1973;97:129-132.
Grigg N., Wearing S., Smeathers J. Eccentric calf muscle exercise produces a greater acute reduction in Achilles tendon thickness than concentric exercise. British Journal of Sports Medicine. 2009;43:280-283.
Helmer K.G., Nair G., Cannella M., et al. Water movement in tendon in response to a repeated static tensile load using one-dimensional magnetic resonance imaging. Journal of Biomechanical Engineering. 2006;128:733-741.
Horn L.M., Subotnick S.I. Surgical intervention. In: Subotnick S.I., editor. Sports Medicine of the Lower Extremity. Edinburgh: Churchill Livingstone; 1989:461-566.
Hunter G. The conservative management of Achilles tendinopathy. Physical Therapy in Sport. 2000;1(1):6-14.
Jozsa L., Kvist M., Balint B.J. The role of recreational sports activity in Achilles tendon rupture: a clinical, pathoanatomical, and sociological study of 292 cases. American Journal of Sports Medicine. 1989;17:338-343.
Khan K.M., Maffulli N. Tendinopathy: an Achilles heel for athletes and clinicians. Clinical Journal of Sports Medicine. 1998;8(3):151-154.
Khan R.J., Fick D., Keogh A., Crawford J., Brammar T., Parker M. Treatment of acute Achilles tendon ruptures. A meta-analysis of randomized controlled trials. Journal of Bone Joint Surgery. 2005;87(10):2202-2210.
Kleinman M., Gross A.E. Achilles tendon rupture following steroid injection: report of three cases. Journal of Bone and Joint Surgery. 1983;65A:1345-1346.
Kvist M. Achilles tendon injuries in athletes. Sports Medicine. 1994;18:173-201.
Kvist M.H., Lehto M.U.K., Jozsa L., et al. Chronic Achilles paratenonitis. American Journal of Sports Medicine. 1988;16(6):616-622.
Langberg H., Ellingsgaard H., Madsen T., Jansson J. Eccentric rehabilitation exercise increases peritendinous type I collagen synthesis in humans with Achilles Tendinosis. Scandinavian Journal of Medicine Science and Sports. 2007;17(1):61-66.
Leswick D.A., Chow V., Stoneham G.W. Answer to case of the month 94: Accessory soleus muscle. Canadian Association of Radiologists Journal. 2003;54(5):313-315.
Li G., Zhang S., Chen G., et al. Radiographic and histological analysis of stress fracture in rabbit tibias. American Journal of Sports Medicine. 1985;13:285-294.
Lowden A. Application of ultrasound to assess stress fractures. Physiotherapy. 1986;72(3):160-161.
Lysholm J., Wiklander J. Injuries in runners. American Journal of Sports Medicine. 1987;15:168-171.
Maffulli N., Ewen S.W., Waterston S. Tenocytes from ruptured and tendinopathic Achilles tendons produce greater quantities of type III collagen than tenocytes from normal Achilles tendons: an in vitro model of human tendon healing. American Journal of Sports Medicine. 2000;28:499-505.
Malliaras P., Richards P.J., Garau G., Maffulli N. Achilles tendon Doppler flow may be associated with mechanical loading among active athletes. American Journal of Sports Medicine. 2008;36(11):2210-2215.
Markey K.L. Stress fractures. Clinics in Sports Medicine. 1987;6:405-425.
McBryde A.M. Stress fractures in runners. Clinics in Sports Medicine. 1985;4(4):737-752. October
Mitcheli L.J., Ireland M.L. Prevention and management of calcaneal apophysitis in children: an overuse syndrome. Journal of Paediatric Orthopaedics. 1987;7:34-38.
Movin T., Gad A., Reinholt F.P., Rolf C. Tendon pathology in long standing achillodynia: biopsy findings in 40 patients. Acta Orthopaedica Scandinavica. 1997;68(2):170-175.
Mubarak S.J., Gould R.N., Lee Y.F., Schmidt D.A. The medial tibial stress syndrome. American Journal of Sports Medicine. 1982;10:201-205.
Ng G., Richardson C.A. The effects of training triceps surae using progressive speed loading. Physiotherapy Practice. 1990;6:77-84.
Nichols A.W. Achilles tendinitis in running athletes. Journal of the American Board of Family Practice. 1989;2(3):196-203.
Niesen-Vertommen S.L., Taunton J.E., Clement D.B., Mosher R.E. The effect of eccentric versus concentric exercise in the management of Achilles tendonitis. Clinical Journal of Sports Medicine. 1992;2(2):109-113.
Norris C.M. Acupuncture: Treatment of Musculoskeletal Conditions. Oxford: Butterworth-Heinemann; 2001.
Paavola M., Orava S., Leppilahti J. Chronic Achilles tendon overuse injuries: complications after surgical treatment. American Journal of Sports Medicine. 2000;28(3):77-82.
Pavlov H., Henegan M., Hersch A., et al. The Haglund syndrome: initial and differential diagnosis. Radiology. 1982;144:83-88.
Pillai J. A current interpretation of popliteal vascular entrapment. Journal of Vascular Surgery. 2008:61S-64S.
Puddu G., Cerullo G., Selvanetti A., De Paulis F. Stress fractures. In: Harries M., Williams C., Stanish W.D., Micheli L.J., editors. Oxford Textbook of Sports Medicine. Oxford: Oxford University Press, 1994.
Puffer J.C., Zachazewski J.E. Management of overuse injuries. American Family Physician. 1988;38(3):225-232.
Read M.T. A Practical Guide to Sports Injuries. Oxford: Butterworth-Heinemann; 2000.
Reid D.C. Sports Injury Assessment and Rehabilitation. London: Churchill Livingstone; 1992.
Roos E.M., Engstrom M., Lagerquist A., Soderberg B. Clinical improvement after 6 weeks of eccentric exercise in patients with mid-portion Achilles tendinopathy – a randomized trial with 1-year follow-up. Scandinavian Journal of Medicine Science and Sports. 2004;14(5):286-295.
Rossi F., La Cava F., Amato F., Pincelli G. The Haglund syndrome: clinical and radiological features and sports medicine aspects. Journal of Sports Medicine. 1987;27:258-265.
Rzonca E.C., Baylis W.J. Common sports injuries to the foot and leg. Clinics in Podiatric Medicine and Surgery. 1988;5(3):591-612. July
Saw Y., Baltzopoulos V., Lim A., et al. Early mobilisation after operative repair of ruptured Achilles tendon. Injury. 1993;24:479-484.
Shalabi A., Kristoffersen-Wilberg M., Svensson L., et al. Eccentric training of the gastrocnemis-soleus complex in chronic Achilles Tendinopathy results in decreased tendon volume and intratendinous signal as evaluated by MRI. Scandinavian Journal of Medicine Science and Sports. 2007;17(3):298-299.
Solveborn S.-A., Moberg A. Immediate free ankle motion after repair of acute Achilles tendon ruptures. American Journal of Sports Medicine. 1994;22:607-610.
Stager A., Clement D. Popliteal artery entrapment syndrome. Sports Medicine. 1999;28(1):61-70.
Stanish D.W., Robinovich R.M., Curwin S. Eccentric exercise in chronic tendinitis. Clinical Orthopedics and Related Research. 1986;208(7):65-68.
Steinberg B.D. Evaluation of limb compartments with increased interstitial pressure. An improved noninvasive method for determining quantitative hardness. Journal of Biomechanics. 2005;38:1629-1635.
Styf J. Chronic exercise-induced pain in the anterior aspect of the lower leg. Sports Medicine. 1989;7:331-339.
Taunton J.E., Clement D.B. Lower extremity stress fractures in athletes. Physician and Sports Medicine. 1981;9:77-86.
Taylor A., Kerry R. Altered haemodynamics. Vascular issues in sport. Sportex medicine. 2002;12:9-13.
Turco V.J., Spinella A.J. Achilles tendon ruptures: peroneus brevis transfer. Foot and Ankle. 1987;7:253-259.
Urban K. Partial rupture of the Achilles tendon in athletes. Acta Chirurgiae Orthopaedicae et Traumatologiae Cechoslovaca. 1989;56(1):30-38.
Williams J.G.P. Achilles tendon lesions in sport. Sports Medicine. 1986;3:114-135.
Wooten B., Chandler J. Use of an orthotic device in the treatment of posterior heel pain. Journal of Orthopaedic and Sports Physical Therapy. 1990;11(9):410-413.