CHAPTER 12 Cerebellum
Basic anatomy
The cerebellum, often refered to as the ‘little brain’, makes up only 10% of the total brain volume but contains more than 50% of the total number of neurons. It is composed of an outer layered cortical region and an inner subcortical mass of cells which form the deep cerebellar nuclei (DCN). The cerebellum is topographically arranged and has ipsilateral control of movement, that is, the left hemisphere of the cerebellum controls the left side of the body. The cerebellum is divided by two deep transverse fissures into three lobes, anterior, posterior and flocculonodular lobe. On the upper surface there are three functional regions separated anatomically by two longtitudinal stripes. The central vermis (medial zone) and two paravermal regions or hemispheres. The paravermal regions are divided into the lateral and intermediate zones.
The deep cerebellar nuclei
There are four deep cerebellar nuclei (DCN):
All output from the cerebellum leaves via the DCN, with the exception of output to the vestibular nuclei which has a direct connection. Afferent pathways also send collaterals that synapse on neurons in the DCN.
The functional loops
Based on its function, the cerebellum can be broadly divided into four loops:
The cerebrocerebellum
This incorporates a loop between the cerebral cortex and the cerebellum. It is located in the lateral zone of the paravermal regions and is strongly connected with the dentate nucleus.
The vestibulocerebellum
This incorporates a loop between the vestibular system and the cerebellum. It roughly corresponds to the flocculonodular lobe and has no connection to the DCN.
Peduncles
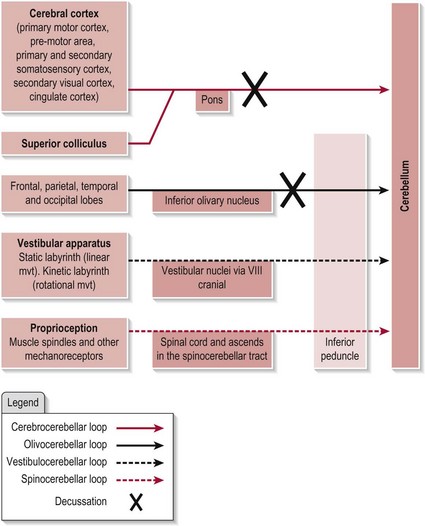
The cerebellum is connected to the rest of the central nervous system by three bundles of nerves or pathways termed ‘peduncles’. The superior peduncle carries almost entirely efferent information away from the cerebellum. The middle peduncles, of which there are two, carry afferent information into the cerebellum. The inferior peduncle carries both afferent and efferent information.
Afferent connections
The cerebellum receives information from many sources. Figure 12.1 shows the main afferent connections. Note that the vestibular and spinal inputs to the cerebellum remain ipsilateral as the information conveyed from these sources is also ipsilateral. However, the inputs from the cerebral cortex and the inferior olive (which contralateral) need to decussate (cross-over) before entering the cerebellum.
Efferent connections
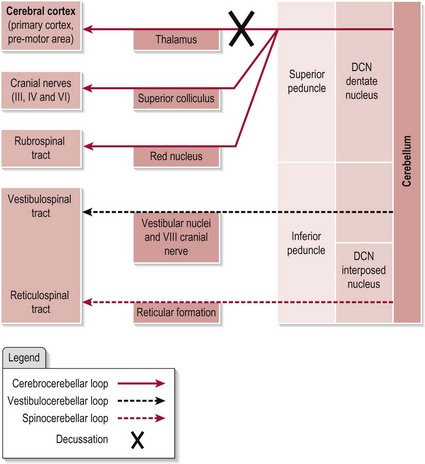
The efferent connections from the cerebellum are shown in Figure 12.2. Note that the pathway to the vestibular nuclei is a direct projection, however all other efferent pathways exit the cerebellum by one of the DCN.
The axons from the dentate nucleus must again decussate to ensure the cerebral cortex receives information related to the appropriate side of the body. Output is ultimately destined to influence the the descending tracts which will innervate the relevant muscles for movement.
The intrinsic circuitry within the cerebellum
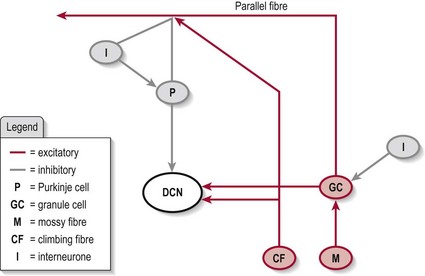
Somatosensory information remains topographically arranged within the cerebellum although less organized than that of the cerebral cortex. Histologically, the cerebellum is organized into five cell layers which form a modulatory circuit throughout the cerebellar cortex. This circuitry is highly complex and beyond the scope of this book. However, in brief, the cells involved are:
In brief, the cerebellar circuitry forms a loop between the input neurons (mossy fibres and granule cells/parallel fibres and climbing fibres) and the output neurons (Purkinje cells) (Fig. 12.3) – the final output being via the DCN. The Golgi, stellate and basket cells work as interneurons to control/modulate the flow of information through this loop with their inhibitory contacts on the Purkinje cell. Also, the whole circuit has various other connections which allow it to be self-modulating. The final output is decided by the balance of excitation and inhibition at the DCN (granule cell and climbing fibres excitatory and Purkinje cell inhibitory).
Function of the cerebellum
The three regions of the cerebellum have similar intrinsic circuitry (Fig. 12.3) but different functions which are a consequence of the origins and destinations of the input and output neurons, respectively. In brief, the cerebrocerebellum loop provides the cerebellum with knowledge about the intended plan for movement and the vestibulocerebellum and spinocerebellum loops monitor the actual position and motion of the body during the movement. Any mismatch/error is corrected and the movement is refined in terms of both timing and magnitude to meet the original goal. The correction is managed directly or indirectly via various descending tracts.
Cerebrocerebellum
This loop is specifically involved in the regulating, planning and timing of movement. Its connections with the superior colliculus also give it a role in visually guided coordination of voluntary movement. In addition, it has a further role in regulating distal limb movment and speech, with damage to the region presenting as limb ataxia (S3.18, 26) and dysarthria (S3.16), respectively.
Vestibulocerebellum
This loop is important in the regulation of posture and balance (S3.18, 32) via axial muscle control and vestibular reflexes. It also governs the eye movements associated with equilibrium (vestibulo-ocular reflex) (S2.10).
Spinocerebellum
This loop is primarily involved in the execution and control of axial and proximal muscle activity via the medial descending tracts. Therefore a role in postural control and balance is likely. Not surprisingly, damage to the area results in trunk ataxia (S3.26) but damage to the more lateral areas of the intermediate zone may also present with limb ataxia. The spinocerebellum also influences the level of muscle tone via the reticular system (S3.21). Recent studies are also challenging the view that this region participates only in the execution of movement and suggests that the intermediate zone also has a role in predictive control and adaptation.
The detail
The error correction system of the cerebellum is a consequence of modulation by the intrinsic circuitry. This forms the basis for regulation of movement as it occurs and over the longer term to initiate the changes that underly motor learning. This regulation occurs by altering the firing frequency of the Purkinje cells to ultimately influence the excitatory output at the DCN. The Purkinje cells and DCN are both tonically active at rest. During movement, there is a change in the rate of firing of these cells, the pattern of which exactly represents the movement being performed. The patterns of firing that are concurrently available to both cells are compared. Any error is corrected by altering the firing pattern, the Purkinje cells modulating (through inhibitory contacts) the excitatory output of DCN. A correct signal is then sent to the appropriate motor neurons to improve the accuracy of the movement. This provides an ongoing corrective mechanism while movement is occuring. Over time, the climbing fibres change the firing patterns via long-term depression (neuroplasticity) to reflect the corrected pattern of movement. In simple terms, the error has been corrected immediately during the movement and over time removed from the circuit; learning has occurred.
References and Further Reading
Ausim AS. And the olive said to the cerebellum: organization and functional significance of the olivo-cerebellar system. The Neuroscientist. 2007;13:616-626.
Bakker M, Allum JH, Visser JE, et al. Postural responses to multidirectional stance perturbations in cerebellar ataxia. Experimental Neurology. 2006;202:21-35.
Bastian AJ. Learning to predict the future: the cerebellum adapts feedforward movement control. Current Opinion in Neurobiology. 2006;16:645-649.
Ilg W, Giese MA, Gizewski ER, et al. The influence of focal cerebellar lesions on the control and adaptation of gait. Brain. 2008;131:2913-2927.
Kandel ER, Schwartz JH, Jessell TM. Principles of neural science, ed 4. New York: McGraw-Hill Health Professions Division; 2000.
Lo YL, Fook-Chong S, Chan LL, et al. Cerebellar control of motor activation and cancellation in humans: an electrophysiological study. The Cerebellum. 2009;8:302-311.
Ohki M, Kitazawa H, Hiramatsu T, et al. The role of primate cerebellar hemisphere in voluntary eye movement control revealed by lesion effects. Journal of Neurophysiology. 2009;101:934-947.
Purves D, Augustine GJ, Fitzpatrick D, et al. Neuroscience. Sunderland: Sinauer Associates; 2008.
Schoch B, Dimitrova A, Gizewski AB, et al. Functional localization in the human cerebellum based on voxelwise statistical analysis: a study of 90 patients. Neuroimage. 2006;30:36-51.
Shadmehr R, Krakauer JW. A computational neuroanatomy for motor control. Experimental Brain Research. 2008;185:359-381.
Timmann D, Konczak J, Ilg W, et al. Current advances in lesion-symptom mapping of the human cerebellum. Neuroscience. 2009;1:1-16.
Voogd J, Barmack NH. Oculomotor cerebellum. Progress in Brain Research. 2005;151:231-268.