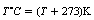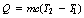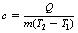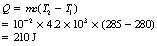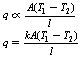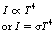Chapter 5 Heat
Chapter contents
5.1 Aim
This chapter considers heat as a transfer of energy. Links between heat and temperature are established. The mechanisms of transferring heat are also established.
5.2 Introduction
We are familiar with the feelings of hot and cold; indeed, we are crucially dependent on our bodily temperature staying within a very limited range in order to survive. Compared to the vast ranges of temperature which exist across the universe, our experience of ‘hot’ and ‘cold’ is very limited indeed.
In fact, we are better at noticing ‘hotter’ or ‘colder’ than we are at determining an absolute value of ‘hot’ or ‘cold’. This can easily be shown from a simple experiment for which you require a bowl of hot water, a bowl of cold water and two bowls of tepid water. Place the right hand in the bowl of hot water and the left hand in the bowl of cold water and leave them in position for about 2 minutes. Now transfer both hands to the bowls of tepid water. You will note that this water feels cold to the right hand and warm to the left hand. This shows that we are better at detecting changes in temperature than we are at detecting absolute values of temperature.
In order to investigate heat further, we must use objective measures of heat and cold, as described in the following sections.
5.3 Heat energy and temperature
When heat is given to a body, its atoms or molecules are given increased kinetic energy in the form of increased lattice vibration (this is the reason why substances expand when heated). A body whose atoms have a higher kinetic energy than those of another body is said to be hotter or at a higher temperature. If two bodies are placed in contact, then heat will be transferred from the hotter body to the cooler body by collisions between the molecules at the point of contact. The molecules of the cooler body receive a net increase in kinetic energy and so its temperature rises. The molecules of the hotter body have lost kinetic energy and so its temperature falls. This process continues until the two bodies are at the same temperature when no further exchange of energy takes place. The bodies are now in a state of thermal equilibrium. Notice that the thermal energy is always transferred from the body at the higher temperature to the body at the lower temperature, irrespective of the size of the bodies. Also note that the temperature existing at thermal equilibrium will always lie somewhere between the initial temperatures of the two bodies.
If a body is made to give away thermal energy, then the lattice (the bound framework of a solid or semisolid) vibrations of its atoms or molecules will decrease. It is logical to assume that there must be a temperature at which no lattice vibration exists. As all movement of molecules ceases to exist at this temperature, this is the lowest temperature that we can attain. This temperature is known as absolute zero and will be further discussed in the next section.
5.3.1 Temperature scales
There are two temperature scales used in modern physics: Celsius (also called centigrade) and kelvin. The Celsius scale is defined as 0°C at the temperature of melting ice, and 100°C at the temperature of boiling water at an atmospheric pressure of 1.01 × 105 newtons per square metre (76 mm of mercury). On this scale, the temperature of absolute zero (see previous Insight) is approximately −273.15°C. This temperature is zero on the kelvin scale (0 K) while the temperature of melting ice is 273.15 K. From this, we can see that the units of temperature are the same on both scales but the scales have different starting points, i.e. one unit on the Celsius scale is equivalent to one unit on the kelvin scale. Note that temperature on the kelvin scale does not have the degree symbol in front of the K. It is a convenient approximation to assume that 0°C=273 K, so that the simple conversion formula may be used:
where T is temperature.
5.3.2 Units of heat energy, specific heat capacity and thermal capacity
If heat is given to a body, then its molecules will have a higher kinetic energy and its temperature will rise. Hence, it is convenient to express a quantity of heat in terms of the temperature change it produces in a given body. Consider the situation where we wish to apply an amount of energy, Q, which will raise the temperature of a body by 1 kelvin unit (for simplicity, assume that this does not change the state of the body, i.e. it does not change from a solid to a liquid). This will be affected by the mass and the type of material of the body. It also seems logical to assume that we would need twice the amount of heat to raise the temperature of the body by 2 kelvin units. If we take all these factors together, we can write the equation:
where Q is the heat energy required to raise the temperature of a body of mass m from T1 to a temperature T2. The factor c is approximately constant for a particular material and is known as the specific heat capacity of the material. We can rearrange Equation 5.2 to get:
so that c, the specific heat capacity, is in units of c joules per kilogram per kelvin (J.kg−1.K−1). Specific heat capacity can be defined thus:
The specific heat capacity of a body is the energy in joules required to raise the temperature of 1 kilogram of the body by 1 kelvin unit.
The specific heat capacity of a substance is thus unique to that substance and it allows us to predict the behaviour of different masses of the same substance.
The specific heat capacity of water is about it 4.2 (J.kg−1.K−1). How much heat energy is required to raise a mass of 10 g of water from 280 K to 285 K? (Remember that the unit of energy is the joule and the unit of mass is the kilogram.)
Using Equation 5.2, we have:
In the situations considered so far, we have considered the amount of heat required to raise the temperature of 1 kg of the material. Another unit of heat energy, which is useful in practice, is the thermal capacity.
The thermal capacity of a body is the heat energy in joules which is required to raise the temperature of the body by 1 kelvin unit.
Note that this definition differs from the previous one for specific heat capacity in that no mention is made of unit mass. Thus, the thermal capacity refers to the whole of the body and not just 1 kg of it. It should also be noted that the thermal capacity of a body is the specific heat capacity of the body multiplied by the mass of the body. The units of thermal capacity are joules per kelvin.
The anode discs of two X-ray tubes are made of the same material but one has twice the mass of the other. If the same amount of heat is applied to each anode, which will have the higher temperature rise at the end of the exposure?
The thermal capacity is the product of the mass and the specific heat. Thus, the larger anode will have twice the thermal capacity of the smaller one. If the same amount of heat is applied to each, the smaller one will experience twice the temperature rise of the larger one. The importance of this will be considered when we consider the rating of the X-ray tube (see Ch. 31).
5.4 Transfer of heat
As mentioned at the beginning of this chapter, heat can be given to a body from some other structure with greater thermal energy than the body. Similarly, if a body can be isolated from its environment or is in a state of thermal equilibrium with its surroundings, there is no net gain or loss of heat from the body.
The mechanisms of heat transfer form an important part of the study of radiography because of the large amounts of heat energy produced at the target of the X-ray tube. The mechanical and thermal stresses associated with this make it possible to damage the X-ray tube unless adequate precautions are taken. The anodes of all X-ray tubes must therefore be designed to transfer heat away from the focal spot area as quickly as possible in order to minimize the temperature rise in this region. The practical application of this knowledge to the design of X-ray tubes will be considered in Chapter 31. However, we must first understand the different mechanisms of heat transfer: these are conduction, convection and radiation.
5.4.1 Conduction
Conduction is the transfer of heat between bodies by physical contact of those bodies and results in a transfer of kinetic energy by interatomic collision, thus forming the main process by which heat is transferred through a solid. If heat is applied to one end of a metal bar, the atoms at this end of the bar receive copious supplies of kinetic energy. These atoms, because of their increased vibrational energy, collide with neighbouring atoms and so kinetic energy is gradually transferred along the bar. This method of heat flow along the bar is known as conduction of heat.
We can find by experiment that the rate of flow of heat, q (joules per second), by conduction is controlled by a number of things:
• q∝A (the cross-sectional area of the rod)
• q∝ (Tl−T2) (the temperature difference between the ends of the rod; this is known as the temperature gradient)
Combining all these factors, we have:
Here k is the constant of proportionality and this is a constant for any given material. It is known as the thermal conductivity of the material. Materials are classed as ‘good’ or ‘bad’ conductors depending on the value of k. Typical values of k are shown in Table 5.1 (See page 30).
Table 5.1 Thermal conductivity of materials
| MATERIAL | THERMAL CONDUCTIVITY (W.m−1.K−1) | COMMENTS |
|---|---|---|
| Copper | 386 | Excellent conductor – used as the anode material in the stationary anode tube |
| Tungsten | 202 | Fairly good conductor – used as the target material in stationary X-ray tubes |
| Molybdenum | 147 | Relatively poor conductor – used as the anode stem in the rotating anode tube |
| Glass | 1.0 | Poor conductor – involved in the transfer of heat to the oil in the tube housing |
| Rubber | 0.05 | Very poor conductor |
| Air | 0.02 | Very poor conductor – removes heat from the housing of the X-ray tube by convection |
Note that the rate of flow of heat along a bar can be changed by altering the length, the cross-sectional area and the temperature gradient as well as by altering the material.
5.4.2 Convection
Convection is the main process by which heat is transferred in fluids (i.e. liquids and gases). Consider heat being applied to a liquid in a beaker (Fig. 5.1). The liquid near the source of heat has thermal energy transferred to it by conduction. This increase in thermal energy causes the liquid to expand and become less dense than the surrounding liquid. The heated liquid rises (because of hydrostatic pressure) and, as it rises, it transfers heat to the surrounding molecules by conduction. The temperature of the small section of heated liquid returns to the same temperature as the surrounding liquid and so sinks back into the beaker, to be heated a second time. Thus, convection currents are established, as shown in Figure 5.1. The overall effect is that the whole volume of fluid is heated and not just the region where the heat is applied.
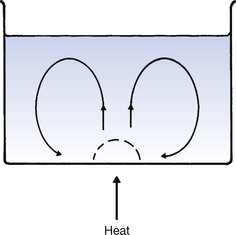
Figure 5.1 Principles of convection. Applied heat causes the broken-line region of fluid to expand, become less dense and rise, being replaced by cooler parts of the fluid. This sets up convection currents.
Some interesting points emerge from this:
• Convection can only occur where the molecules are free to move through the medium, i.e. in liquids and gases.
• It is not possible to have convection currents without some conduction.
• It is necessary to have a gravitational field in order for the convection currents to be initiated as the warm fluid moves in the opposite direction to the gravitational force, by hydrostatic pressure.
5.4.3 Radiation
In this process, heat is lost from a body with high thermal energy in the form of electromagnetic radiations. Radiation is the only heat transfer process that will take place through a vacuum. The most obvious and striking example is the heat we can feel from our sun where the radiation must pass through a near-perfect vacuum for 93 million miles (approximately 150 million km) to reach us. Light and heat also reach us from other stars but we are only able to discern the light, as we are not sufficiently sensitive to small amounts of heat (although the latter can be detected with infrared sensors).
We are able to feel heat by radiation because it imparts kinetic energy to molecules of our tissues, which we discern as an increase in temperature. Heat radiations occur in a band of energies just beyond the red part of the visible spectrum (see Ch. 17 for further detail of the electromagnetic spectrum). This band of energies is known as infrared radiations. All bodies radiate electromagnetic energies if their temperature is above absolute zero. This does not necessarily mean that we can discern the radiations, as we perceive only a narrow band of energies extending from violet light to infrared. Bodies of different colours and different surface compositions radiate somewhat differently and so it is conventional to consider black-body radiation. This we will do next.
5.4.3.1 Black-body radiation
A body looks black because very little of the light incident upon it is reflected or transmitted. A black body is defined as one that will absorb 100% of all radiations at all frequencies incident upon it. If such a black body is in a state of thermal equilibrium with its surroundings, equal amounts of radiation must be absorbed and emitted per second. From this it can be deduced that the black body must radiate more energy than any other type of body since no other body absorbs 100% of the energy incident upon it. The foregoing statements can be summarized thus:
• All bodies are capable of emitting radiation.
• A black body absorbs 100% of all radiations incident upon it.
• A black body is the most efficient emitter of radiation of any body.
We now need to know in more detail the spectrum of radiations emitted by a black body. Figure 5.2 shows such a spectrum for a black body at different temperatures. The following should be noted from the graph:
• The wavelength corresponding to the peak radiation progressively decreases as the temperature of the body increases.
• The height of the graph (intensity of radiation) is very sensitive to changes in temperature.
• The spectrum of the radiation is a smooth curve, of which we perceive only a small part (violet to infrared).
• The total intensity of the radiation emitted by a body at a given temperature is the sum of the intensities at each wavelength. This is related to the area under the curve in each case. Stefan’s law states that the total intensity of the emitted radiation is proportional to the fourth power of the kelvin temperature. Thus:
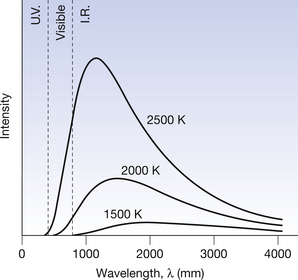
Figure 5.2 The spectrum of electromagnetic radiation emitted by a black body at different temperatures. The total intensity (I) is a function of the kelvin temperature (I∝T4). IR, infrared; UV, ultraviolet.
where σ is Stefan’s constant.
Doubling of the kelvin temperature results in the amount of heat radiation emitted being increased by a factor of 24= 16 times. As this shows, the intensity of the radiation emitted varies greatly with the temperature of the body. This has important consequences in the design of the rotating anode X-ray tube disc, as we shall see in Chapter 30.
A curve of emitted radiation at about 1000 K is equivalent to a metal rod which has been heated until it is glowing cherry-red. As the temperature increases, the colour changes through light red to white since an increasing amount of the violet end of the spectrum is emitted along with the red (see 2000 K and 2500 K lines in Fig. 5.2). At higher temperatures, objects emit white light. This is true of the filament of an electric light bulb or the anode disc of the rotating anode tube after a large exposure.
The mechanism that causes the emission of radiation is the acceleration and deceleration of the charged particles, which make up the atoms or molecules of the body involved. An example particularly relevant to radiography is the sudden deceleration of electrons when they strike the anode of the X-ray tube – the Bremsstrahlung or braking radiation. Here, some (or all) of the kinetic energy of the electron is transformed into an X-ray photon (see Ch. 21). Another example is the production of radio waves, where electrons are forced to oscillate at high frequencies in a radio-transmitting aerial.
The interactions of energetic atoms produce a broad band of emitted quanta wavelengths because of the range of interactions that are possible. All of these radiations form part of the electromagnetic spectrum. A narrow range of frequencies (in the infrared part) is capable of being absorbed by the whole atom (as opposed to the electron shells or the nucleus) and so these atoms gain kinetic energy. This increase in the kinetic energy of the atoms is in the form of heat. Higher frequencies of radiation interact with the electrons in the orbitals and even higher frequencies will interact with the atomic nuclei.
5.5 Thermal expansion
Most substances expand when heated, owing to the increased kinetic energy of the atoms. The linear expansivity isa measure of this thermal expansion, defined as the fractional change in unit length per unit change in kelvin temperature. The thermal expansion of different parts of the X-ray tube is an important design consideration as the tube is subject to large temperature variations during its working life. If materials of very different thermal expansivity are used, this can cause mechanical stress that could result in fracture of one of the components. A particularly weak point in this respect is the seal between the glass and the copper anode in a stationary anode tube. A special type of glass with a thermal expansivity very similar to copper is chosen. This reduces the mechanical stress and thus the chances of fracture of the glass envelope and loss of vacuum in the tube.
5.6 Evaporation and vaporization
We are all familiar with the evaporation of liquids that occurs when heat is applied to them, e.g. boiling kettles. Evaporation is caused by the loss of whole atoms from the surface of the liquid. Some of the atoms are given sufficient kinetic energy to escape from the forces of attraction of their neighbours near the surface of the liquid and produce a vapour in the free space above this surface.
Whole atoms may also be liberated from the surfaces of solid materials under the action of heat. There is a stronger force of attraction between the atoms in a solid compared to a liquid and so liberation of the atoms is more difficult to accomplish. This means that in a solid the atoms require higher kinetic energy for liberation, i.e. solids need to be subjected to a higher temperature. The tungsten at the target and in the filament of the X-ray tube is subjected to such high temperatures that a certain amount of vaporization takes place. The tungsten vapour can condense to form a thin layer of tungsten on the inside of the glass of the envelope. A major effect of this is to reduce the electrical insulation provided by the glass, as tungsten is a reasonable conductor of electricity. This can produce an effect called a ‘gassy tube’, which renders the X-ray tube inoperable. Fortunately, this does not occur readily as tungsten has a low vapour pressure so does not readily vaporize at its normal working temperatures.
In this chapter, you should have learnt:
• That heat is a form of energy produced by the kinetic energy of atoms in a body (see Sect. 5.3).
• The greater the kinetic energy of the atoms, the higher the temperature and vice versa (see Sect. 5.3).
• That bodies which are in thermal equilibrium with their surroundings lose heat as fast as they gain heat and so experience no change in temperature (see Sect. 5.3).
• Absolute zero temperature corresponds to zero atomic kinetic energy and has a temperature of 0 K (see Sect. 5.3.1).
• On the kelvin scale, the temperature at which ice melts is approximately 273 K (see Sect. 5.3.1).
• The link between heat and temperature provided by specific heat capacity and thermal capacity (see Sect. 5.3.2).
• Heat may be transferred from one point to another by conduction (mainly in solids), convection (in fluids) and radiation (may travel through a vacuum) (see Sects 5.4.1-5.4.3).
Further reading
Further information on X-ray tube design and rating is available in Chapters 30 and 31 of this text. The following may also prove useful: