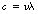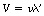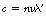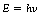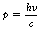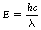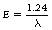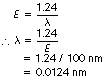Chapter 17 Electromagnetic radiation
Chapter contents
17.1 Aim
This chapter discusses the properties of electromagnetic radiations. The wave–particle duality of such radiations will be further discussed. Having identified the contents of the electromagnetic spectrum, we will finally consider some parts of this spectrum in more detail as these are of particular relevance to radiography.
17.2 Introduction
As can be seen from Figure 17.3 (p. 114), the electromagnetic spectrum encompasses a wide range of radiation types and we are only sensitive to a small section of these radiations. We can see the world around us because the retinas of our eyes are sensitive to a section of the electromagnetic spectrum which we know as light. Similarly, we can feel heat from the sun because our skin responds to the infrared part of the electromagnetic spectrum. We can also accidentally walk through a beam of X-rays or handle an isotope which is producing gamma radiation because none of our sense organs are able to detect them. It is important to remember, when we consider radiation protection, that although our sense organs are not able to detect the presence of ionizing radiations (X or gamma), they may still be damaged by them.
17.3 Properties of electromagnetic radiations
All electromagnetic radiations exhibit a set of general properties which are listed below:
• The waves are composed of transverse vibrations of electric and magnetic fields. (Transverse vibrations are ones which are oscillating at right angles to the direction of travel.)
• The vibrations have a wide range of wavelengths and frequencies.
• All electromagnetic radiations travel through a vacuum with the same velocity: 3×108 m.s−1.
• All electromagnetic radiations travel in straight lines.
• The radiations are unaffected by electric or magnetic fields.
• The radiations may be polarized so that they vibrate in one plane only.
• The radiations are able to produce constructive or destructive interference.
• All the radiations obey the duality principle (see Sect. 16.6) and so can either be considered as waves or as quanta with energy and momentum.
From the above list it is obvious that all electromagnetic radiations have a lot in common, so how can they be used for such widely differing purposes? The answer to this lies in the differences which they exhibit in their interactions with matter. These are outlined in Table 17.1.
Table 17.1 Interactions of different electromagnetic radiations with matter
| INTERACTION | NOTES |
|---|---|
| Emission | All bodies will emit electromagnetic radiation in certain circumstances but the most efficient emission is from a ‘black body’ (see Sect. 5.4.3.1) |
| Reflection | Reflection of electromagnetic radiation is not possible for the higher energy radiations (X and gamma radiation) |
| Refraction | Refraction of electromagnetic radiation is not possible for the higher energy radiations (X and gamma radiation) |
| Transmission | Different materials are transparent to different wavelengths or photon energies |
| Attenuation | Different attenuation processes are possible depending on the photon energy of the radiation but, if the photons all have the same energy, the attenuation is always exponential. Photoelectric absorption can occur from ultraviolet to gamma radiations. Compton scattering is produced by X and gamma radiations. Pair production is an attenuation process which is possible if the photon energies are higher than 1.02 MeV |
| Luminescence – fluorescence and phosphorescence | Electron transitions within the material being irradiated cause the emission of photons that have less energy than the incident radiation. A single photon of incident radiation can produce many fluorescent photons |
As discussed in Chapter 16, there is a wave–particle duality which exists when electromagnetic radiation interacts with matter. This will be discussed further in the following sections.
17.3.1 Wave-like properties
As the term electromagnetic suggests, electromagnetic radiation consists of both electric and magnetic fields. These fields are at right angles to each other and to the direction of propagation and are shown diagrammatically in Figure 17.1. As shown in E and B of the figure, both the electric and the magnetic vectors (see Sect. 4.3) vibrate transversely to the direction of propagation of the wave. In addition, the vectors vary in a sinusoidal manner, as shown in the figure. Thus, if we draw the variations of the electric vector (for example), a sine wave results, as shown in Figure 17.2. These periodic variations of the vectors are the reason why electromagnetic radiations are often referred to as electromagnetic waves and this was the sole method of explaining the behaviour of such radiations adopted by classical physics (see Sect. 16.6). The same parameters can be used to describe this type of wave formation as can be used for any sinusoidal waves. These are:
• the cycle– one complete waveform (this can start from any point on the wave and end at the corresponding point on another wave)
• the wavelength– the distance travelled in completing one cycle (λ)
• the frequency– the number of cycles per second (Hz)
• the amplitude– the magnitude of the peak of the wave above the x-axis (A).
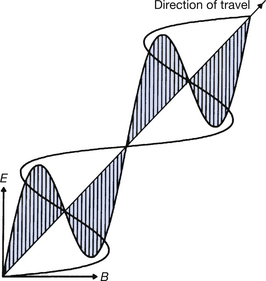
Figure 17.1 Electromagnetic radiation depicted as a wave consisting of alternating electrical and magnetic vectors vibrating at right angles to each other and to the direction of motion of the wave.
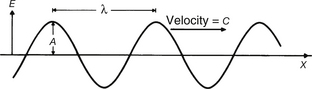
Figure 17.2 An electromagnetic sine wave produced by plotting energy (E) against distance (X). The wave has amplitude of A and a wavelength λ and travels at the velocity of light, c (3×108 m.s−1).
If we consider Figure 17.2, it should be apparent that we can calculate the distance travelled by the radiation in 1 second by multiplying the wavelength (λ) by the frequency (v). However, the distance travelled in 1 second is simply the velocity of the radiation (c in a vacuum) and so we have:
in a vacuum.
As mentioned earlier, one of the features of electromagnetic radiations is that they all travel with the same velocity (3×108 m.s−1) in a vacuum. One ray is distinguished from another by the difference in wavelength and frequency –blue light has a wavelength of about 400 nm and a frequency of 7.5×1014 Hz while red light has a wavelength of about 800 nm and a frequency of 3.75×1014 Hz. From this, it can be seen that the greater the frequency, the smaller the wavelength and vice versa.
In a transparent medium, the waves travel at a velocity V given by the equation c=nV, where n is a constant for a given medium and a given incident wavelength and is known as the refractive index. The frequency of the radiation is unaltered as it passes through a transparent medium but the wavelength is reduced due to the reduction in the velocity of the wave. If the wavelength in the medium is λ′, then Equation 17.1 becomes:
Substituting the value of V for c=nV, we obtain:
in a medium of refractive index, n.
Chapters 9 and 10 showed that it is possible to produce both a magnetic field from moving electrical charges (electromagnetism) and an electric field from a changing magnetic flux (electromagnetic induction). It can be shown mathematically that a changing electric field can sustain a changing magnetic field, and vice versa, if they both travel at the velocity of light. This is the basis both for the linear propagation of electromagnetic radiations and for the fact that the wave does not gradually diminish in amplitude with time. Electromagnetic radiation can therefore be described as a self-sustaining interaction of electric and magnetic fields travelling with the velocity of light.
Two further properties of electromagnetic radiation which are a consequence of its wave-like nature are polarization and interference.
17.3.1.1 Polarization
A beam of light, from a bulb for instance, consists of many millions of waves whose electric vectors are pointing in random directions with respect to each other. If this light beam is passed through a polarizing lens (similar to the one found in polarizing sunglasses), then there is optimum transmission of the waves whose electric vectors are pointing in one direction and complete absorption of those at right angles to this direction (this is the mechanism by which polarizing sunglasses limit the glare due to light reflection from water). The emergent light is said to be plane-polarized – all its vibrations are in the one plane.
17.3.1.2 Interference
Further evidence of the wave-like properties of electromagnetic radiations comes from the phenomenon of interference where the amplitudes of two coherent beams (beams which are in phase with each other) can be added together. If the peaks of the waves coincide, we get constructive interference and this produces bright areas. If the peak of the wave from one source coincides with the trough of the wave from the other source, we get destructive interference and this produces a dark area. Such interference patterns are used in radiographic science to produce holograms, but further discussion on this topic is well outside the scope of this text.
17.3.2 Particle-like properties
In Chapter 16 we discussed how consideration of electromagnetic radiations as quanta or photons having energy and momentum enabled us to explain a number of properties which are not explained by the wave theory. The energy of such a quantum, E, is proportional to the frequency of the associated wave such that:
where h is a constant known as Planck’s constant and v is the frequency of vibration of the associated wave. Thus, because there is a direct relationship between frequency and the energy of the quantum, as the frequency increases so does the energy. The momentum, p, of the quantum is also proportional to the frequency, and is given by:
where c is the velocity of electromagnetic radiation in a vacuum.
The photon energy and its wavelength can now be related. If we take Equation 17.3 and substitute v=c/λ from Equation 17.1, we get:
In this equation, h is Planck’s constant (6.62×10−34 J.s−1), c is the velocity of electromagnetic radiation in a vacuum (3×108 m.s−1), λ is the wavelength measured in meters and E is the photon energy measured in joules. For practical purposes, in radiography it is more convenient to measure the photon energy in keV (1 keV=1.16×10−16 J) and the wavelength in nanometers (1 nm=10−9 m). Because h and c are constants, Equation 17.5 can now be rewritten:
This equation gives us an easy link between the energy of the photon in keV and the wavelength of the radiation in nanometres.
If the energy of the X-ray photon is 100 keV, what will be its wavelength in nanometres?
Using Equation 17.6:
Lengths as small as 0.0124 nm or 1.24×10−11 m are difficult to imagine. This is less than the diameter of an atom of body tissue (about 10−10 m) but is greater than the diameter of the atomic nucleus (about 10−14 m).
17.4 The electromagnetic spectrum
The previous sections of this chapter have shown that electromagnetic radiation may have a very large range of wavelengths, i.e. a spectrum of wavelengths (and frequencies). It is convenient to split up such an electromagnetic spectrum into bands which are broadly categorized by their interaction with matter and hence the use to which the bands of radiation may be put. This is illustrated in Figure 17.3, which also shows the wavelengths, frequencies and energies corresponding to the approximate boundaries between the various bands. As we have previously discussed, the table of the electromagnetic spectrum shows that the smaller the wavelength of the radiation, the higher the frequency and the energy.
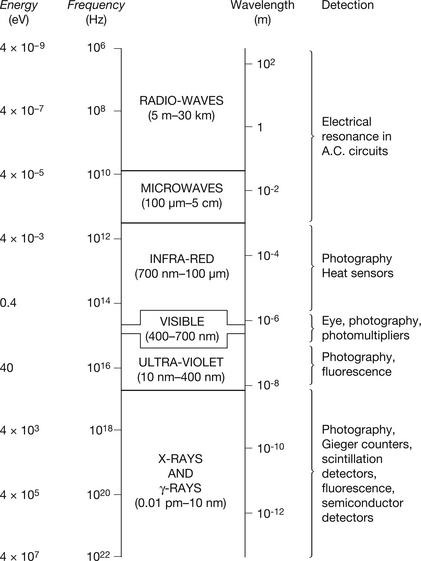
Figure 17.3 The electromagnetic spectrum (not drawn to scale). Typical values for wavelengths, frequencies and photon energies for different bands of the spectrum are shown, together with some of the methods used for their detection. (Note: 1 Hz=1 cycle per second.)
The common factor which links the interactions between different types of electromagnetic radiations is that the value of the wavelength determines the size of object with which the radiation will directly interact. This is illustrated in Table 17.2.
Table 17.2 Common electromagnetic radiations and the type of body with which they will directly interact
| TYPE OF RADIATION | BODY WITH WHICH RADIATION DIRECTLY INTERACTS |
|---|---|
| Radio waves | Transmitted and received by large metallic conductors – aerials |
| Infrared radiation | Interacts with whole molecules or atoms giving them an increase in their kinetic energy in the form of heat |
| Visible light and ultraviolet radiation | Interacts with the outer electrons (more loosely bound) of the atom |
| X-rays and γ-rays | Interact with the inner electron shells or, if they have very high energy, with the nucleus of the atom |
At the beginning of this chapter (see Table 17.1) are the types of interactions which are possible between electromagnetic radiations and matter, with brief comments. From this table you will note that refraction is associated with electromagnetic radiations which have wavelengths larger than X-rays. Refraction is a phenomenon associated with an interaction between a photon and the outer electron orbitals of atoms. It is therefore associated with wavelengths which are able to interact with these electrons – ultraviolet and longer wavelengths. Smaller wavelengths become progressively less affected by the outer electron orbitals, and so it is just possible to demonstrate refraction with very large wavelength X-rays but it is not possible to refract X-rays which have shorter wavelengths.
The interactions between X-rays and matter, which are of great relevance to radiographic science, are discussed in detail in Chapter 23 of this book.
17.5 Light amplification by stimulated emission of radiation (Laser)
In the last few years, devices containing a laser source have increasingly become a part of our everyday life and are also increasingly used in radiographic imaging. For this reason, a short section is included here describing the basic physics of laser production and also a section indicating the hazards of lasers if used carelessly.
17.5.1 Basic physics of laser production
Laser is an acronym for light amplification by stimulated emission of radiation. The theory which was first proposed by Albert Einstein in 1917 was further developed in the 1950s and the first laser was produced in 1960. We already know from Section 7.3 that in an atom electrons can exist at various energy levels depending on their position relative to the nucleus. We will also see in Chapters 18 and 24 that electrons can be temporarily raised to a higher energy level by the absorption of photons of energy. In many cases, the electron remains in this excited state for only a few milliseconds and subsequently decays to its lower energy level by the emission of a photon of light (or other energy) – this process is known as fluorescence (see Ch. 24 for a more detailed description). If the light emitted from an atom is incident on another atom which is in an excited state, then the first photon can stimulate the emission of a second photon from this atom. The two photons will be identical in wavelength, phase and direction. This is the basis of laser.
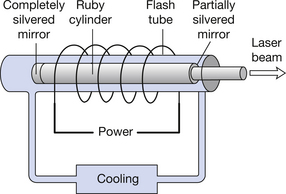
The basic components of a simple laser are shown in Figure 17.4. The excitation of electrons to a higher energy level is achieved by illuminating the material with light of a frequency higher than that which the laser will emit. This light is produced from the flash lamp and is known as optical pumping. The two ends of the laser rod are polished flat and parallel. One end is coated with a completely silvered mirror and the other is coated with a semisilvered mirror. Pulsed light is flashed into the laser at high intensity from the flash lamp and this is reflected within the rods using the two mirrors, thus causing coherent photon emission from the atoms of the rod material. Eventually the high-intensity laser radiation will leave the rod through the semisilvered mirror. The wavelength of the light emitted depends on the design of the laser so the beam may not be visible to the eye.
17.5.2 Potential hazards of lasers
Laser light has several properties which are important when we consider its safe use:
• The light is monochromatic, with its wavelength determined by the design of the laser.
• The light is in the form of a tightly collimated parallel beam, often less than 1 mm in diameter.
• Because the light is in the form of a small, highly collimated area, even lasers of small power can deposit significant amounts of energy on a small area.
Assume that a 10-W laser beam is directed at a material. This does not sound much in terms of power in that many night lights are rated at 10 W. However, the laser beam is a parallel, very narrow beam. If this beam is 1 mm in diameter then the power deposited on the material is greater than 1000 W.cm−2– this can ignite paper or cause significant skin burns. If the laser beam enters the eye, the problem is further complicated by the fact that the lens of the eye focuses the beam onto a small area of the retina. Thus, the power deposited on this small area of retina can be increased by a factor of up to 105 because of lens focusing. If we consider the original 10-W laser, this will mean a power deposited on a small area of the retina of 108 W.cm−2– compare this to power to the retina of about 10 W.cm−2 if we stare at the midday sun, and it is easy to appreciate how damaging this can be to the eye.
As mentioned earlier, the wavelength of the light from the laser is determined by the laser design. The retina of the eye can detect from about 400 nm to just over 700 nm. Thus, if a laser is operating at, say, 900 nm, we will not be able to see the beam from this laser. However, the beam can still penetrate the lens system of the eye and cause damage to the retina of the eye. For this reason it is important to wear eye protection whenever lasers are in use, e.g. in an operating theatre, even if the laser beam is not visible.
17.6 Electromagnetic radiations and radiography
The process of taking a radiograph (using a digital imaging system) results in the emission of radiations whose wavelengths and energies are from different parts of the electromagnetic spectrum, described in the previous section. These are summarized in Figure 17.5. When the anode is bombarded by electrons, it emits both heat and light as well as X-rays. The X-ray spectrum is composed of a continuous or Bremsstrahlung spectrum upon which is superimposed a characteristic or line spectrum from the tungsten target. The absorption processes within the aluminium filter and the lead collimators also produce characteristic radiation from these elements, although these are of fairly low intensity. When the radiation beam passes through the patient, some of the photons are absorbed (A in the diagram) and some are scattered (S in the diagram). The above processes produce a tiny amount of heat and other characteristic radiations from the elements which make up the body tissues. Some of the scattered radiation is absorbed by the grid. When the transmitted X-ray beam (T) strikes the digital imaging system, it produces changes in electron energies which can then be utilized to produce the digital image – this will be discussed in more detail in Chapter 34. Further examples of electromagnetic radiation relevant to radiography include:
• the emission of light from thin field transistors (TFTs) to produce images on flat-screened imaging devices
• the emission of light from cathode ray tubes and television monitors when the phosphors are bombarded by electrons
• the emission of light from fluorescent tubes to allow us to view hard copies of images
• the emission of gamma-rays from certain radioisotopes which allow imaging or treatment of organs
• the emission of light from sodium iodide crystals (in gamma cameras and computed tomography (CT) scanners) when bombarded with gamma-rays or X-rays
• the emission of light from photostimulable plates when scanned using a laser beam
• the use of a laser beam to ‘write’ information onto the film in a laser imager
• the use of low-intensity lasers to aid positioning of the radiation beam in radiotherapy.
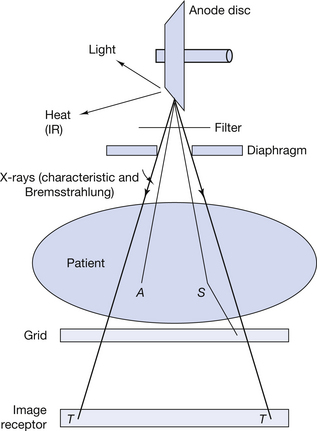
Figure 17.5 The production of radiation from different parts of the electromagnetic spectrum utilized when a digital radiographic image is produced.
In this chapter, you should have learnt the following:
• The properties which are common to all electromagnetic radiations (see Sect. 17.3).
• The different ways in which electromagnetic radiations can react with matter (see Sect. 17.3).
• How electromagnetic radiation can be described as having wave-like properties and how these properties can be used to explain polarization and interference (see Sect. 17.3.1).
• How electromagnetic radiation can be described as having particle-like properties and the equation which links the photon energy and the wavelength of the radiation (see Sect. 17.3.2).
• The various components of the electromagnetic spectrum and the principal interactions of the radiations with matter (see Sect. 17.4).
• A brief description of the physics of laser production and a note of its potential dangers (see Sect. 17.5).
• The electromagnetic radiations which are of importance to radiography (see Sect. 17.6).
Further reading
You will find further information on the methods of X-ray production in Chapter 21 and more information on the mechanisms by which X-rays interact with matter in Chapter 23 of this text. In addition, you may find the chapters of the following text useful:
Ball J.L., Moore A.D., Turner S. Ball and Moore’s Essential Physics for Radiographers, fourth ed. London: Blackwell Scientific, 2008. (Chapters 14 and 15)