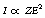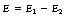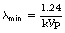Chapter 21 Production of X-rays
Chapter contents
21.1 Aim 151
21.2 Interactions of electrons with matter 151
21.3 Interactions between electrons from the filament and the outer electrons of the target atoms in the X-ray tube 151
21.4 Interactions between electrons from the filament and the nuclei of the target atoms in the X-ray tube 152
21.5 Inelastic collisions with the electrons of the target atoms – production of characteristic radiation 154
21.6 The X-ray spectrum 155
Further reading 157
21.1 Aim
The aim of this chapter is to consider the mechanisms by which X-rays are produced at the target of the X-ray tube. The concept of the X-ray spectrum will be introduced and this chapter will act as a foundation for Chapter 22 where the factors affecting the X-ray spectrum will be considered.
21.2 Interactions of electrons with matter
In Chapter 30 we will discuss the construction of the X-ray tube and the functions of its various components. In this chapter we will look in detail at the processes involved at subatomic level in the target of the X-ray tube to allow the production of X-rays.
Electrons are produced at the filament of the X-ray tube, accelerated towards the target of the tube and then made to collide with the target atoms. There are a number of ways in which high-energy electrons from the filament of the X-ray tube may lose their energy when they collide with the atoms of the target. These are shown in Figure 21.1:
• Electrons from the filament lose energy because of interactions between them and the outer electrons surrounding the atoms of the target material (see electron 1 in Fig. 21.1).
• Electrons from the filament lose energy because of interactions between them and the nuclei of the atoms of the target material (see electrons 2 and 3 in Fig. 21.1).
• Electrons from the filament lose energy because of interactions between them and individual inner electrons of the target atoms (see electron 4 in Fig. 21.1).
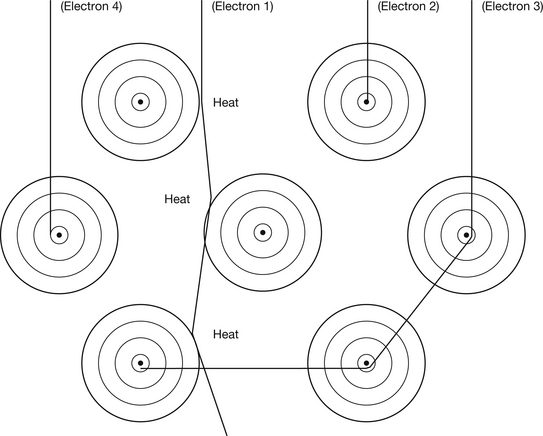
Figure 21.1 Diagrammatic representation of possible interactions between electrons from the filament and target atoms in an X-ray tube. An explanation of the interactions is given in the text.
The interactions between electrons and target atoms may be elastic where there is conservation of kinetic energy or inelastic collisions in which kinetic energy is lost (see Section 3.3). It is also important to realise that an electron from the filament may experience many interactions (typically about 1000) before it is brought to rest within 0.25–0.5 mm of the target surface.
21.3 Interactions between electrons from the filament and the outer electrons of the target atoms in the x-ray tube
As you will see in Chapter 30, the main material of the X-ray tube target is tungsten, which has an atomic number of 74. Thus each target atom is surrounded by 74 electrons in various orbitals. These electrons have the effect of deflecting the approaching electron from the filament from their original path because of the electrostatic repulsion between them and the filament electron. Such deflections from the electron’s original path are small and so the loss of kinetic energy is small. The kinetic energy lost by the electron is emitted as a photon of electromagnetic radiation. The energy of this photon is such that it falls into the infrared part of the spectrum and so heat is produced in the target material. If we consider the statistical probability of this process occurring rather than the other two processes identified in Section 21.2, we can see that it has a very high probability of occurring.
This is because the other two processes involve an interaction between either the incoming electron and the nucleus or the incoming electron and the inner orbital electrons. The area occupied by the nucleus and the inner electron orbitals is very small compared to the size of the whole tungsten atom and so an incoming electron has a much greater chance of interacting with the whole atom. For the above reasons, about 95–99% of the energy produced at the target of the X-ray tube is in the form of heat.
21.4 Interactions between electrons from the filament and the nuclei of the target atoms in the x-ray tube
As mentioned at the beginning of this chapter, the interactions of electrons from the filament with the nuclei of the target atoms can be elastic interactions or inelastic interactions. As each of these results in a different outcome, they will now be considered separately.
21.4.1 Elastic collisions with the nuclei
The electron has a relatively low mass and a negative charge while the tungsten nucleus has a larger mass (about 334 000 times that of the electron) and a positive charge (74 times that of the electron). Thus the electron is attracted to the more massive tungsten nucleus.
As shown in Figure 21.2, the closer the electron travels to the nucleus, the more it is deflected from its original path by the attraction of the positive nucleus. Because the mass of the electron is so tiny compared to that of the nucleus, the amount of energy it transfers to it is proportionately small.
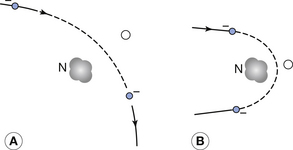
Figure 21.2 The deflection of an electron from the filament when it interacts with the nucleus of a target atom in the X-ray tube: (A) a small deflection; (B) a large deflection.
These events therefore serve only to produce very tortuous paths for the electrons within the tungsten, without transferring much energy to the tungsten. The number of such events compared to the number of inelastic collisions discussed in the next section increases with the atomic number of the target material and so they are very frequent in tungsten.
21.4.2 Inelastic collisions with the nuclei – production of Bremsstrahlung radiation
Classical physics concluded that when a charged particle changed its velocity, this always resulted in the emission of electromagnetic radiation. However, we have already seen that this is not the case, as the electrons in the Bohr model of the atom are continuously changing their velocity (see Sect. 18.4). We have also seen in the previous section that electrons can undergo elastic collisions with the nucleus which result in no electromagnetic radiation being produced. As shown in Figure 21.2, the electron must accelerate towards the nucleus during its deflection but, despite this acceleration, a quantum of electromagnetic radiation is emitted in only a small percentage of cases. When such a quantum is emitted, the kinetic energy of the electron is suddenly reduced by an amount equal to the energy of the quantum, and so the electron is suddenly slowed down or braked.
This is an example of an inelastic interaction since the total kinetic energy of the electron and of the nucleus is not conserved because some energy is removed by the emission of the quantum of radiation. The energy of the quantum may be in the X-ray part of the electromagnetic spectrum and the radiation is known as braking or Bremsstrahlung radiation. The exact energy of this quantum will vary and may be very small if the electron does not lose much energy in its interaction with the nucleus or it may be up to the total kinetic energy of the electron if it is involved in a direct collision with the nucleus (see electron 2 in Fig. 21.1).
Note that not every electron passing close to the nucleus will emit such a photon of radiation but only those which are able to undergo a sudden loss of energy by an inelastic interaction with the nucleus.
The Bremsstrahlung process is illustrated in Figure 21.3, where the X-ray photon is emitted at the point P and the electron continues with reduced energy on the pathway shown. The probability of Bremsstrahlung radiation being produced is very small (about 1–5% for diagnostic X-ray tubes). The intensity (I) of the radiation produced is related to the atomic number of the target material (Z) and the energy of the electron beam (E) by the equation:
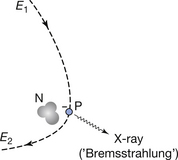
Figure 21.3 The production of Bremsstrahlung X-rays. An electron at point P suddenly loses energy by the emission of an X-ray photon. The electron continues with a reduced energy, E2.
In diagnostic X-ray tubes, tungsten is used as the target material as its high atomic number (74) leads to a higher intensity of X-ray production.
The effect on the X-ray spectrum of changing the energy of the electron beam will be discussed in more detail in Chapter 22, but it is worth noting at this stage that the higher electron energies used in linear accelerators mean that there is more efficient X-ray production and that a smaller percentage of the energy is converted into heat.
If the law of conservation of momentum (see Sect. 3.4) is applied, then, in order to conserve momentum, the greater the energy of the incident electrons, the more likely the Bremsstrahlung radiation is to be emitted in the same direction as the electrons. Thus, for low-electron-energy beams the X-rays are directed almost isotropically, in diagnostic X-ray tubes (medium-energy range) the central ray of the X-ray beam is at right angles to the electron beam, and in linear accelerators (high-energy) the useful X-ray beam is transmitted forward through the anode in the same direction as the electrons.
21.5 Inelastic collisions with the electrons of the target atoms – production of characteristic radiation
As can be seen from Figure 21.1 (see route of electron 4), it is also possible for an electron from the filament to be involved in an inelastic collision with one of the orbital electrons of the target atoms. Such interactions are shown in more detail in Figure 21.4. Depending on the energy transferred to the orbital electron, such collisions may result either in excitation of the atom (Fig. 21.4A) or ionization of the atom (Fig. 21.4B). For excitation to occur the incident electron must be given sufficient energy to raise it to its new orbital, whereas for ionization the incident electron must be given sufficient energy to overcome its binding energy and thus be liberated from the atom. In the case of ionization of the target atom, the electron is ejected from its orbital with a given kinetic energy and is often referred to as a delta-ray. This electron may have sufficient energy to excite or ionize other atoms in its path.
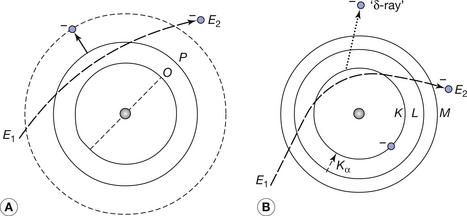
Figure 21.4 (A) Excitation and (B) ionization caused by the passage of an energetic electron through a tungsten atom. Both processes result in the production of heat and ionization produces characteristic radiation (Kα radiation is shown in the diagram). E2 is less than E1 in both diagrams. Note that, for clarity, only the electron orbitals which have a direct bearing on the interaction are shown.
The process of ionization causes a vacancy in the electron shell involved – the K-shell in the atom shown in Figure 21.4B. This vacancy is quickly filled by one of the outer electrons undergoing a quantum jump downwards to fill the vacancy and emitting a quantum of electromagnetic radiation in the process. The energy of the quantum emitted is given by:
where E1 is the energy of the electron before the jump and E2 is the energy of the electron after the jump. As this energy varies for the same transition in different materials, it is known as characteristic radiation as it is characteristic of the material emitting the radiation.
If we consider Figure 21.5 where a vacancy has been created in the K-shell of the atom, then the most probable jump is from the L-shell with the next probability being a jump from the M-shell followed by the probability of a jump from the N-shell. These are known as Kα′, Kβ′, Kγ transitions and the radiation known as Kα- characteristic radiation, etc. If we assume that the vacancy in the K-shell has been filled by a transition from the L-shell, there will now be a vacancy in this shell which can be filled from the M-shell or the N-shell, etc., with the emission of Lα or Lβ characteristic radiation. In this way, a cascade of electrons will occur with each one emitting a photon of energy given by Equation 21.2. In the diagnostic X-ray tube, only the K and L characteristic radiations are of any significance as the others are absorbed by the target material or the insert envelope.
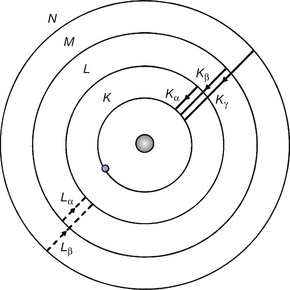
Figure 21.5 The production of characteristic radiation. The K-lines are formed by quantum jumps down to the K-shell. Also note that the energy of the Kα-line must be less than the binding energy of the K-shell. (The significance of this fact is discussed when photoelectric absorption edges are considered in Sect. 23.5.)
As well as the characteristic radiation produced, both excitation and ionization result in the production of heat within the target. In excitation, this is by the absorption of energy by the target as the electron falls back to its original orbital and in ionization by the increased kinetic energy received by the tungsten atoms as they slow down the electrons ejected by the collision.
21.6 The x-ray spectrum
So far in this chapter we have considered the production of X-ray quanta by two major mechanisms:
1. Energetic electrons from the filament interact with the nuclei of target atoms and are slowed down, thus giving off energy in the form of X-ray quanta – Bremsstrahlung radiation. As we mentioned earlier, the energy of the quantum may be very small if the interaction with the nucleus is small, or it may be up to the energy of the incoming electron if this electron is brought to rest by the nucleus. All energies between the two extremes are possible and so it can be seen that the Bremsstrahlung radiation will form a continuous spectrum (see Sect. 17.6).
2. Vacancies created in inner electron orbitals are filled by the electrons from orbitals further away from the nucleus making a quantum drop to fill the vacancy and giving off a photon of radiation in the process. This is the characteristic radiation from the atom. As the energy differences between specific orbitals is constant for an atom, this will take the form of a series of line spectra (see Sect. 17.6).
If the overall X-ray intensity is plotted against the energy of the radiation, a summation of the two effects known as the X-ray spectrum is produced. Such a spectrum produced at the tungsten target of an X-ray tube is shown in Figure 21.6.
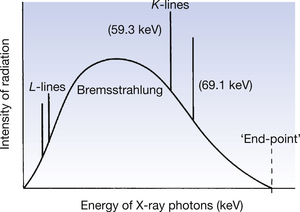
Figure 21.6 An X-ray spectrum as emitted at the anode of an X-ray tube with a tungsten target. For an explanation of this spectrum, see the text.
Certain important features of this spectrum are worthy of further discussion:
• The energy of the Bremsstrahlung radiation is expressed in keV and lies somewhere between zero and a maximum value. This maximum photon energy is achieved if an electron which has the maximum kinetic energy gives up all its energy to form a single photon. The value of this photon for a specific beam may be deduced as follows. Suppose the potential difference across the X-ray tube is 100 kVp. An electron accelerated across the tube when the peak voltage is applied would have achieved a kinetic energy of 100 keV at its point of contact with the anode. If it now gives up all this energy as a single photon, the energy of the photon will be 100 keV and this will be the maximum photon energy for a tube operating at this kVp. Similarly, for an X-ray tube operating at 50 kVp, the maximum photon energy will be 50 keV. Thus it can be seen that the maximum photon energy is dependent on the potential difference across the tube (kVp) but is independent of the material of the target.
• The Bremsstrahlung radiation is a continuous spectrum. The intensity of the low-energy photons within this spectrum is decreased because of absorption of these photons by the target material; as will be seen in Chapter 22, the intensity is further decreased by the filtration of the X-ray beam before it leaves the tube.
• The average energy of the X-ray beam is about one-third to one-half of its maximum energy – this is related to the quality of the X-ray beam (see Ch. 22).
• The total intensity of the beam – the quantity of the radiation – is given by the area under the curve.
• The energies of the K, L, M, etc. lines are always in the same position, although the energy of lines from M onwards is so small that it is likely to be totally absorbed in the X-ray tube. These discrete energies form a line spectrum – the exact energy of the line is determined by Equation 21.2.
• The line spectra will not be produced unless the energy of the electron beam from the filament exceeds the binding energy of the appropriate electron shell of the atom. To produce the K-characteristic lines at a tungsten target, electrons must have an energy ≥69 keV and to produce L-lines, electrons must have an energy ≥11 keV. In practice, this means that K-lines are not produced when the potential across the tube is less than 70 kVp.
In the spectrograph in Figure 21.6 we have considered the intensity of the radiation plotted against the photon energy. It is sometimes useful to calculate the wavelength of the radiation rather than know its photon energy. As you may remember from Section 17.3.2, the photon energy and its associated wavelength are related by Planck’s equation (see Equation 17.6). From this we can see that the maximum photon energy corresponds to the minimum wavelength. Since the numerical value of the maximum photon energy is the same as the kVp, we can calculate the minimum wavelength from the equation:
Although we have explained the maximum and minimum energies in the continuous spectrum, we have not explained the shape – the distribution of energies – of the spectrum. This is quite a complicated process and involves consideration of the chances of photons of various energies being produced, the direction of the radiation emitted and the chances of this radiation being absorbed or scattered within the target.
Consider an ultra-thin target, which is constructed in such a way that there are only a few layers of target atoms and so incoming electrons from the filament are only likely to have one interaction and there is negligible attenuation by the target of the radiation produced. In such circumstances, the number of photons of each energy produced will be related to the statistical chance of an electron losing that amount of energy in its Bremsstrahlung interaction with the nucleus. As the nucleus of the atom is relatively small compared to the whole atom, the chance of an electron from the filament colliding with it and so producing a maximum-energy photon is equally small. The chance of a filament electron getting close to the nucleus is statistically larger and so there should be more photons produced which have less than the maximum energy compared with the number which have the maximum energy. Thus, as we increase the diameter of the circle around the nucleus within which we consider electrons interacting to produce X-ray photons, so we increase the statistical possibility of producing photons with that energy – as these electrons are further from the nucleus of the atom, the energy of the photon produced would be smaller.
The production of lower-energy photons is further increased in a target with a finite thickness if we still ignore any radiation attenuation by the target. A target of finite thickness – normally a few millimetres – can really be considered as a series of ultra-thin targets one on top of the other. Because of this, the energy of the electrons reaching the second ultra-thin layer is less due to their energy loss in the first layer – this again favours the production of lower-energy X-ray photons. Thus, the deeper we go into the target, the lower the predicted energy of the photons produced.
Because of the two processes described, we would expect the number of photons produced to increase as the photon energy decreases. This is shown by the broken line in Figure 21.7.
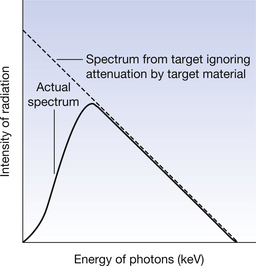
Figure 21.7 A comparison between the continuous spectrum produced if attenuation by the target material is ignored and the actual spectrum produced from a tube target.
In an X-ray tube, the target is just a few millimetres thick so we must consider X-rays being produced below the surface of the target. As these photons leave the target they are attenuated by the target atoms. Both scattering and absorption (see Ch. 23) favour attenuation of the lower-energy photons. Thus, lower-energy photons are heavily attenuated by the target atoms. The attenuation of the lower-energy photons is increased because they are more likely to be produced deeper in the target material and so have further to travel through the target. This attenuation of the photons produced by the target material produces a continuous spectrum similar to the solid line in Figure 21.7.The effect of various parameters on the spectrum of radiation emitted from the X-ray tube will form the material of the next chapter.
The effect of various parameters on the spectrum of radiation emitted from the X-ray tube will form the material of the next chapter.
In this chapter, you should have learnt the following:
• The three major mechanisms by which electrons from the filament of the X-ray tube may lose energy when they interact with the atoms of the target (see Sect. 21.2).
• The result of interactions between electrons from the filament of the X-ray tube and the outer electrons around atoms of the target material (see Sect. 21.3).
• The result of elastic interactions between electrons from the filament and the nuclei of atoms of the tube target (see Sect. 21.4.1).
• How inelastic collisions between electrons from the filament and the nuclei of atoms of the target of the X-ray tube result in the production of Bremsstrahlung radiation (see Sect. 21.4.2).
• How inelastic collisions between electrons from the filament and the orbital electrons of target atoms may result in the production of characteristic radiation (see Sect. 21.5).
• The graphical representation of the X-ray spectrum and the significant features of this graph (see Sect. 21.6).
Further reading
You will find that Chapter 22 of this text deals with further detail of the factors which affect the spectrum of X-rays from the X-ray tube. In addition, you may find that the chapters from the following texts provide useful further reading.
Ball J.L., Moore A.D., Turner S. Ball and Moore’s Essential Physics for Radiographers, fourth ed. London: Blackwell Scientific, 2008. (Chapter 16)
Dowsett D.J., Kenny P.A., Johnston R.E. The Physics of Diagnostic Imaging. London: Chapman & Hall Medical, 1998. (Chapter 3)
Webb S., editor. The Physics of Medical Imaging. Bristol: Institute of Physics, 2000. (Chapter 2)