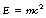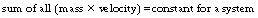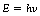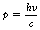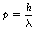Chapter 16 The laws of modern physics
Chapter contents
16.1 Aim
The aim of this chapter is to introduce the reader to the laws of modern physics. In many cases these are refinements or extensions to the laws of classical physics which were discussed in Chapter 3, and in this chapter reference will be made to the laws outlined in Chapter 3. In other areas, there is disagreement between the classical and the modern laws and these will be identified and discussed.
16.2 Introduction
The laws of classical physics were discussed in Chapter 3 and these have been sufficient to explain the phenomena outlined in previous chapters of this text. In the following chapters on atomic and radiation physics, some important aspects of modern physics must be introduced in order to explain many of the phenomena discussed. The purpose of this chapter is to describe the laws of modern physics which are relevant to our need in the rest of the text.
16.3 Classical versus modern laws
Modern physics started at the turn of the twentieth century with Planck’s quantum hypothesis in 1900 in which he conjectured that radiation energy could only be absorbed or emitted by a body at discrete values of energy. Other dates of interest to the rest of this text include the mass–energy relationship postulated by Einstein in 1905, the Bohr model of the atom which was suggested in 1913 and the de Broglie wavelength of particles which was introduced in 1924. Since these dates, there have been major technical and theoretical strides, but these form part of the firm experimental foundation upon which modern physics is built.
One of the essential differences between classical and modern physics is the way in which matter is regarded. In classical physics, matter and energy are completely separate entities and so we have the law of conservation of matter (see Sect. 3.2) and the law of conservation of energy (see Sect. 3.3) with no interconnection being produced between the two laws. In classical physics, matter is supposed to behave in one way – like matter! – and waves are supposed to behave like waves, and one cannot behave like the other; classical physics does not allow for the existence of a particle with a wavelength. There are no such rigid boundaries in modern physics. In particular, the work of Einstein showed that matter can be thought of as being interchangeable with energy if the conditions are right. This principle is known as mass–energy equivalence and will be discussed in more detail later in this chapter (see Sect. 16.4.1). In addition, it is found in modern physics that particles of matter do behave like waves and vice versa and this is known as the wave–particle duality principle(see Sect. 16.6). In this way, an X-ray beam may be considered as photons (particles) which have a specific energy and so can liberate electrons from atoms of the materials they pass through.
With these concepts in mind, we will now consider the laws of modern physics in more detail in the remainder of this chapter.
16.4 Law of conservation of energy
This law now states that the amount of energy in a system is constant. In the context of modern physics, this can be thought of as the sum of all the energies (rest energies + kinetic energies + potential energies) being a constant for any given system. The phrase ‘a system’ can be used to define either a very small or a very large area, provided the influence of other bodies outside the system is negligibly small. In the context of modern physics, the use of the term energy in the above law embraces the contribution of matter to the total energy of a system under consideration. This concept of mass–energy equivalence will now be discussed.
16.4.1 Mass–energy equivalence
Einstein showed that the mass of a body, m, and its energy, E (excluding potential energy), are related by the formula:
where c is the velocity of electromagnetic radiation (often referred to as the velocity of light, as light is probably the best known form of electromagnetic radiation). Since c is a constant, the energy of a body is proportional to its mass (and vice versa). If we consider a stationary body with a rest mass, m0, then the rest energy of this body is given by E0=m0c2. If we now consider this body travelling with a velocity, V, then its energy is now EV and Einstein’s equation is EV=mVc2. Since the energy of the body when moving, EV, is greater than the energy of the body when at rest, E0, and since c is a constant, then mV must be greater than m0– a body increases in mass as its velocity increases. The above statement seems to contradict our common experiences (because of the small values of velocity which we can normally produce) but if we take particles and accelerate them until they travel with a velocity close to the velocity of light (3×108 m.s−1) then there is a measurable increase in the mass of the particles. The mass which they then possess is known as the relativistic mass of the particles. Thus, if we take a car which is travelling at 40 kilometres per hour, we see no measurable increase in its mass, but if we take electrons travelling at high speeds in a linear accelerator then there is a measurable increase in their mass.
The law of conservation of energy, stated earlier, is sometimes referred to as the law of conservation of mass–energy because of the concept of equivalence between mass and energy. It is also not uncommon to quote the rest mass of subatomic particles, either in units of mass or in units of energy – the rest mass of the electron can be stated as 9.1×10−31 kg (mass) or 0.511 MeV (energy).
As we can see from the above discussion, energy and mass can be considered as two manifestations of the same thing, and may be changed from one form to the other in appropriate circumstances, as the following examples show:
• The forces which hold the atomic nucleus together are obtained because some of the mass of the nuclear particles is converted into energy. Because of this, the mass of the nucleus is less than the sum of the masses of the individual nuclear particles.
• If a gamma-ray has an energy greater than 1.02 MeV and passes close to the nucleus of an atom, the ray may spontaneously disappear and create two particles of matter – an electron and a positron. This process is known as pair production and will be described in more detail in Chapter 23. The positron created in this interaction will interact with an electron and their mass will be converted into two photons of radiation, each photon having an energy of 0.51 MeV. The positron and the electron will now cease to exist and the radiation is referred to as annihilation radiation. The positron can thus be regarded as the antiparticle of the electron. This interaction shows that energy can be converted into mass and that mass can be converted into energy.
16.5 Law of conservation of momentum
This law may be stated as the total linear momentum in a system is constant. The word ‘system’ is used in the same context as for the law of conservation of energy. As we discussed in the section dealing with the laws of classical physics, the momentum of a body is the product of its mass and its velocity. Thus we can say:
Here, the mass referred to in the equation is the relativistic mass of the body, i.e. its mass when it is moving with the velocity, V. An example of the law of conservation of momentum being applied to modern physics is in Compton scattering (see Ch. 23) when some of the momentum of the incoming photon is given to an electron – the combined momentum of the scattered photon and the ejected electron is the same as the momentum of the incident photon.
16.6 Wave–particle duality
Duality simply means two different features of the same thing. As we have just seen, modern physics regards mass and energy as being two manifestations of the same phenomenon. Similarly, modern physics blurs the distinction which exists in classical physics between a particle and a wave.
16.6.1 Waves as particles
Classical physics was very successful in explaining many of the phenomena associated with electromagnetic radiation (e.g. diffraction and interference) by assuming that such radiation was made up of waves travelling at the velocity of light, c. In this case c=vλ, where v is the frequency of vibration of the radiation and λ is its wavelength. However, phenomena like the Compton effect and photoelectric absorption (both will be discussed in detail in Ch. 23) are not easy to explain using the wave theory. These effects are explained by considering that sometimes electromagnetic radiation behaves as ‘packets’ of energy which have an associated momentum. Such a packet of energy is called a photon or a quantum and the quantum theory predicts that:
where h is a constant known as Planck’s constant and v is the frequency of vibration of the associated wave. We will frequently use this formula in the chapters which follow on atomic physics!
The electromagnetic wave may also behave like a particle, possessing energy and momentum.
16.6.2 Particles as waves
Moving particles of matter, whether these are large or very small, have both kinetic energy and momentum. Are there then occasions when they behave as waves? Perhaps the most dramatic example of particles behaving like waves is in the operation of the electron microscope. Here high-energy electrons are passed through or are scattered by a sample. A very highly magnified image of the sample is obtained so that individual large molecules may be seen in materials. The reason for the high degree of magnification is due to the very small wavelength of the electrons. Whether we consider an optical microscope or an electron microscope, the smaller the wavelength of the radiation used, the finer the detail it is possible to see.
De Broglie proposed that the following relationship exists between the momentum, p, of the particle and its associated wavelength, λ:
In such cases, λ is called the de Broglie wavelength and the existence of particles behaving like waves can be verified by a number of experiments.
Note the inverse relationship between the momentum of the particle and its associated wavelength. The wavelength decreases as the momentum increases and vice versa. For example, the de Broglie wavelength associated with an electron moving at half the velocity of light is about 4×10−12 m which is less than the diameter of the hydrogen atom (100×10−12 m). When the velocity of the electron is one-hundredth of that of light then the de Broglie wavelength is at the larger value of 240×10−12 m, which is now in the X-ray range of wavelengths (see Ch. 17) and so such electrons can be used for X-ray crystallography.
Within the context of radiographic science, it is more common to consider waves behaving as quanta rather than quanta behaving as waves. This is particularly true when we consider the production of X-rays (see Ch. 21) and the interactions of X-rays with matter (see Ch. 23).
16.7 Heisenberg’s uncertainty principle
In classical physics it is possible in principle to measure exactly a number of quantities concerning the state of a body, e.g. the body’s energy, position, and momentum. Furthermore, if it were possible to build measuring apparatus which was infinitely precise, it would be possible to make simultaneous exact measurements of several of these quantities.
According to modern physics, it is not possible to treat quantities like mass and energy or matter and waves as being totally independent of each other. Heisenberg’s uncertainty principle is an extension of the principle of wave–particle duality and concerns the maximum possible precision which may be obtained in ideal circumstances when measuring two quantities simultaneously. The central point of the principle is that measuring one quantity affects another quantity so that it is never possible to measure both quantities simultaneously with complete accuracy – if we try to measure the momentum of a particle, this will automatically affect the position of the particle so that it is never possible to measure momentum and position simultaneously with complete accuracy. Effects due to this principle are too small to be observed in everyday life and concern atomic and nuclear systems. This principle is yet another difference between modern and classical physics – in classical physics, it is assumed that perfect instruments produce perfect results.
If we now apply Heisenberg’s uncertainty principle to the duality theory, we can say that, although we can demonstrate that waves can behave like particles and that particles can behave like waves, it is not possible to set up a situation where both properties are demonstrated simultaneously.
In this chapter you should have learnt the following:
• The differences which exist between the laws of classical physics and the laws of modern physics (see Sect. 16.3).
• The law of conservation of energy as applied to modern physics (see Sect. 16.4).
• The concept of mass–energy equivalence (see Sect. 16.4.1).
• The law of conservation of momentum as applied to modern physics (see Sect. 16.5).
• The concept of wave–particle duality (see Sect. 16.6).
• The concept that waves may behave as particles and that particles may behave as waves, with examples of each situation (see Sects 16.6.1 and 16.6.2).
• A brief outline of Heisenberg’s uncertainty principle (see Sect. 16.7).
Further reading
You may find Chapter 17 of this text and chapters from the following text provide useful further reading.
Ball J.L., Moore A.D., Turner S. Ball and Moore’s Essential Physics for Radiographers, fourth ed. London: Blackwell Scientific, 2008. (Chapters 14 and 15)