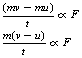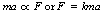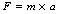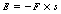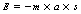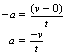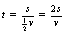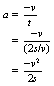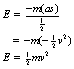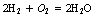Chapter 3 Laws of classical physics
Chapter contents
3.1 Aim
The chapter introduces the reader to some of the laws of classical physics. An understanding of these laws will aid in understanding some of the later chapters of the book. (Some of the physics in the later chapters of this book (e.g. pair production as an absorption mechanism) cannot be explained in terms of the laws of classical physics.) Before these concepts are considered (Ch. 20 onwards), the laws of modern physics will be considered (in Ch. 16).
3.2 Law of conservation of matter (Mass)
3.3 Law of conservation of energy
3.3.1 Statement of the law
Energy can neither be created nor destroyed but can be changed from one form to another. The amount of energy in a system is constant.
This law tells us that energy is never used up but changes from one form to another.
When an electron is released from the filament of an X-ray tube, it has potential energy. As it is accelerated across the tube, this potential energy is converted to kinetic energy. When it makes contact with the target of the tube, this kinetic energy is converted to heat and X-ray energy. At any time, the sum of all the energies remains constant.
Some of the common forms of energy are:
These two laws of conservation of mass and energy are combined into one law by modern physics (see Ch. 16).
3.4 Law of conservation of momentum
3.4.1 Statement of the law
This law is important when we consider collisions between two bodies.
There are two types of collision that occur, elastic and inelastic. An elastic collision is one where all kinetic energy is conserved, as in the case of a ‘perfect’ billiard ball colliding with a similar but stationary billiard ball; the moving ball stops, while the ball which was previously stationary moves with the same velocity as the first ball had before collision. In an inelastic collision, the total kinetic energy is not conserved, as in the case of two billiard balls colliding with a glancing blow, so that both continue to move after the collision. In both cases, the momentum is conserved, although the velocities of the bodies in each case will be different.
In radiographic science, conservation of momentum is mainly concerned with the interactions of X-rays with matter; these will be dealt with in Chapter 23.
3.5 Newton’s laws of motion
Newton’s laws of motion can be derived from the above laws, but they are so useful that they merit a separate section. They are defined as follows:
3.5.3 Law 3
The action of one body on a second body is always accompanied by an equal and opposite action of the second body on the first.
Note: The terms velocity and momentum in the first two laws imply direction, as both are vector quantities (see Ch. 4).
A body of mass m and velocity u has a force F applied to it. After a time t, its velocity has changed to v. Then the second law of motion can be stated as:
Now (v − u)/t is the rate of change of velocity or the acceleration, a, of the body (see Ch. 4). So, we can say that:
where k is the constant of proportionality (see Appendix A).
If we choose suitable units, it can be arranged that k is equal to 1 and we finally have:
In the International System of Units (SI; see Ch. 4), F is measured in newtons, m is measured in kilograms and a is measured in m.s−2. This makes Equation 3.1 the familiar mathematical statement of Newton’s second law and can also be used as the basis for the definition of the newton (1 N=1 kg × 1 m.s−2).
As an illustration of the use of this law, we are now in a position to calculate the kinetic energy of a body of mass, m, travelling with a velocity, v. If we apply a steady force, F, in the opposite direction to that of v, the body will slow down and eventually come to rest. The work done in bringing the body to rest must be equal to its kinetic energy. We can state this mathematically as:
where s is the distance taken for the body to come to rest. (The force, F, and its associated acceleration, a, are regarded as negative as they are applied in the opposite direction to v.)
From Equation 3.1 we can now change Equation 3.2 as follows:
The acceleration, a, is the change in velocity per unit time. Stated mathematically, this is:
As the action of the force is consistent throughout the deceleration, the time taken for the body to stop can be calculated by dividing the distance travelled by the average velocity (v/2). So:
If we now consider Equation 3.3, this can be rewritten:
Newton’s third law is usually paraphrased as: ‘To every action there is an equal and opposite reaction’. There are many examples of this in everyday life, such as a hammer hitting a nail, but it is important to realize that there need not necessarily be physical contact between the two bodies for one to act on the other. If we take two charged bodies and bring these close together (but not actually touching), the forces between the two bodies will be equal and opposite. The significance of this in the design of the cathode of the X-ray tube will be considered in Chapter 30.
3.6 Avogadro’s hypothesis, the mole and avogadro’s number
As we will see in Chapter 18, all substances consist of atoms or molecules. These may react chemically with the atoms or molecules of other substances. These reactions occur with fixed proportions in order to produce a given chemical compound and it is possible to predict the number of molecules of the compound from knowledge of the number of molecules of the original elements or compounds, for example:
In the case of gases, Avogadro’s hypothesis postulated that equal volumes of gases at the same temperature and pressure contain equal numbers of molecules. This hypothesis was first postulated in the early nineteenth century and has been verified by a number of experiments since then.
This is taken one stage further within the SI system in the more general statement that the number of molecules per mole is the same for any substance. The mole is the SI unit of the amount of substance and is defined as:
The mole is the amount of substance which contains as many elementary particles as there are atoms in 0.012 kg of carbon-12.
Carbon-12 is used as the standard for technical experimental reasons. From this, we can predict the number of atoms or molecules in a substance by knowing its atomic mass number and comparing this with carbon-12. For example, if we consider cobalt-60, then there will be the same number of atoms in 0.6 kg of cobalt-60 as there will be in 0.012 kg of carbon-12, as each constitutes one mole of substance. The number of molecules in a mole is given by Avogadro’s number (or constant) and is 6 × 1023 molecules.
If we consider X-ray photons being attenuated by matter, this is a reaction between the photons and the electrons of atoms of the material through which they pass. If we know the number of atoms per mole of the material, we can calculate the number of atoms per unit mass. By knowing the atomic number of the material, it is then possible to establish the electron density and so predict the likelihood of an X-ray photon interacting with an electron. This will be discussed further in Chapter 23.
Further reading
Further reading on the laws of classical physics can be found in most textbooks which are used in schools for study of AS level or A2 level physics. In addition, the following may prove useful:
Allan E., Harris J. New Higher Chemistry. London: Hodder Gibson, 1999. (Chapter 3)
Ball J.L., Moore A.D., Turner S. Ball and Moore’s Essential Physics for Radiographers, fourth edn. London: Blackwell Scientific, 2008. (Chapter 1)