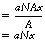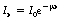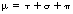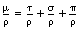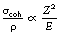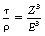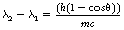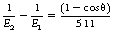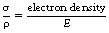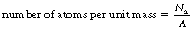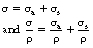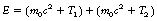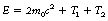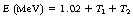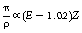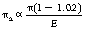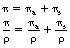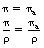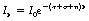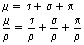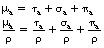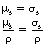Chapter 23 Interactions of X-rays with matter
Chapter contents
23.1 Aim 165
23.2 Outline of possible interactions 165
23.3 Attenuation and absorption 168
23.4 Elastic (coherent) scattering 168
23.5 Photoelectric absorption 169
23.6 Compton scattering 171
23.7 Pair production 173
23.8 Relative importance of the attenuation processes in radiography 174
23.9 Conclusion 175
Further reading 176
23.1 Aim
The aim of this chapter is to introduce the reader to the interaction processes which may occur when radiation photons interact with matter. The factors which influence such interaction processes will be discussed. An understanding of the processes is important both for consideration of the production of the radiograph and in the consideration of the biological effects of radiation on tissue.
23.2 Outline of possible interactions
When a beam of X-rays interacts with a medium, e.g. a volume of patient tissue, there are a number of possible interactions between the photons and the atoms of the medium. These are outlined in Figure 23.1 (see page 166). If we consider photon A, we can see that this interacts with an atom of the medium and is deflected from its path – this deflection may or may not be accompanied by a loss of photon energy. This process is known as scattering. Photon B, on the other hand, interacts with an atom of the medium and loses all of its energy to the atom. This process is known as absorption. The third photon, photon C, passes through the material without interacting with any of the atoms. If we measure the intensity of the radiation in a given area before it interacts with the medium and again in a similar area after the interactions, we find that there is a lower intensity of radiation after passing through the medium – the beam of radiation has been attenuated (see Sect. 20.7).
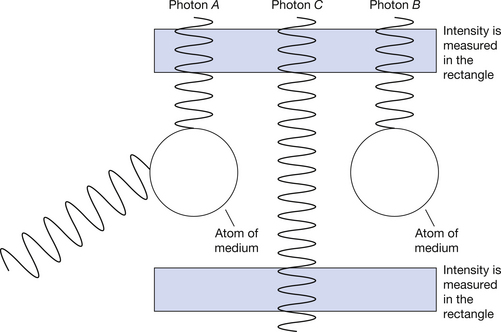
Figure 23.1 Mechanisms of interaction of X-ray photons with matter. Photon A is scattered, photon B is absorbed and photon C is transmitted. Note that the number of photons passing through the lower rectangle is less than the number passing through the upper rectangle and so attenuation has taken place.
When a beam of radiation passes through a medium, it is attenuated by the processes of absorption and scattering.
The reactions considered above involve an interaction between a photon and an orbiting electron rather than between a photon and the nucleus of the atom (this will be discussed in more detail in the rest of this chapter) and so, for an interaction to occur, a photon must pass close to the electron orbiting the atom with which it is capable of interacting. Thus we can consider the atoms in a medium as targets which, if hit, will attenuate a photon from the primary radiation beam.
Note: For the purposes of this discussion, we are considering a parallel beam of radiation so that reduction in intensity only occurs here because of the attenuation processes and no reduction in intensity occurs because of the inverse square law.
23.2.1 Probability and cross-sections
The probability of an X-ray photon interacting with a particular atom is low, but the very large number of atoms even in a small volume of a solid substance makes the probability of a photon interacting with some atom much greater. In Figure 23.2, a beam of X-rays of area A is incident upon a medium whose atoms appear to the beam to have an area of a. This area is called the cross-section of the atom to a particular type of radiation, a typical area being 1.5×10−28 m2. If an X-ray photon hits one of the atoms, it is either absorbed or scattered from the primary beam.
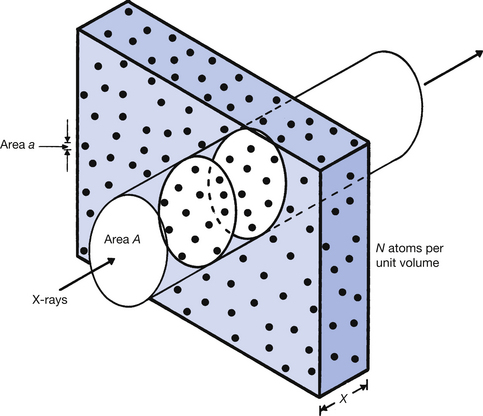
Figure 23.2 Consideration of the area of individual atoms as part of the total irradiated area. If any of the X-ray photons passing through area A hits an atom (area a) then the photon is removed from the beam. As fewer photons will exit from the medium than entered the same area, the beam is said to be attenuated. For further explanation, see the text.
Note that the area a is not the true size of the atom but is the apparent area of the atom likely to interact with the X-ray beam. The value of a depends on a number of things, including the atomic number of the material and the energy of the X-ray photon.
The probability of an interaction occurring can be predicted by dividing the total area of the atoms within the irradiated area by the size of this area. If we consider Figure 23.2 and take it that there are N atoms per unit volume of the material, then the number of atoms in the irradiated cylinder of cross-sectional area A is N×A×x. Thus the total area of the atoms in the irradiated area is a(N×Ax) or aNAx.
The probability of an interaction:
23.2.2 The total linear attenuation coefficient (μ)
As we discussed in Section 20.7, a parallel beam of monoenergetic radiation – radiation where all the photons have the same energy – will undergo exponential attenuation as it passes through a uniform medium. The intensity of the incident beam (I0) and the intensity after a thickness x are given by the equation:
where μ is the total linear attenuation coefficient. If we now consider Equations 23.1 and 23.2, it can be seen that as the probability of an interaction increases (aNx), the linear attenuation coefficient μ will also increase. The total linear coefficient may be defined:
23.2.3 The total mass attenuation coefficient (μ/ρ)
As we have seen from Equation 23.1, the probability of an interaction between an X-ray photon and a medium containing N atoms is proportional to N. If we consider the medium shown in Figure 23.2 and take a situation where it is heated until it has expanded to a thickness of 2x, then the number of atoms in the thickness x will be N/2 and the linear attenuation coefficient (μ) will also be halved. As the number of atoms per unit volume has halved, the density of the material (ρ) will also have halved. However, the ratio μ/ρ will be the same in both cases. This ratio μ/ρ is known as the total mass attenuation coefficient and is defined:
The total mass attenuation coefficient, μ/ρ, is the fraction of the X-rays removed from a beam of unit cross-sectional area by unit mass of the medium.
If we wish to calculate the amount of radiation which would pass through a given length of a material, then we would use the linear attenuation coefficient. In most other cases the mass attenuation coefficient would be used as this is unaffected by the state of the medium; it may be solid, liquid or gas as long as the changes in density are taken into account. Thus, if we wish to consider the effect of a change, for instance, of photon energy on the transmitted beam, this type of calculation would be undertaken using the mass attenuation coefficient.
The following sections of this chapter will consider the effect on the mass attenuation coefficient of the different types of X-ray interactions and of different attenuating materials. An understanding of these is fundamental to consideration of the formation of the radiograph (see Ch. 25).
Note the use of the term total in both of the above coefficients. This is because there is a mass attenuation coefficient due to each of the attenuation processes and so the total linear attenuation coefficient is the sum of the individual linear attenuation coefficients and the total mass attenuation coefficient is the sum of the individual mass attenuation coefficients. Thus:
where μ is the total linear attenuation coefficient, τ is the linear attenuation coefficient due to the photoelectric effect, σ is the linear attenuation coefficient due to Compton scattering and π is the linear attenuation coefficient due to pair production. Similarly:
where μ/ρ is the total mass attenuation coefficient, τ/ρ is the mass attenuation coefficient due to the photoelectric effect, σ/ρ is the mass attenuation coefficient due to Compton scattering and π/ρ is the mass attenuation coefficient due to pair production.
23.3 Attenuation and absorption
The four interaction processes described in the rest of this chapter involve attenuation since the intensity of the primary X-ray beam is reduced as a result of each process. In some of the processes, energy is transferred from the photons in the X-ray beam to the atoms of the medium and so absorption is said to have taken place. Thus, an absorption process must involve the transfer of energy from the photons to the atoms of the material.
In other processes, the photon is deflected from its original path and so is said to be scattered. This may involve no transfer of energy to the medium (see coherent or elastic scatter, Sect. 23.4) or it may involve deflection of the photon and a transfer of energy to the medium (see Compton scattering, Sect. 23.6).
As we have already noted, attenuation consists of both absorption and scattering and the contribution of each of the individual processes to the whole is shown in Table 23.1. As can be seen from the table, the total mass absorption coefficient will be composed of the mass absorption coefficients from each of the attenuation processes. This may be defined as follows:
Table 23.1 Contribution of each of the attenuation processes to absorption and scatter
| PROCESS | CONTRIBUTION TO ABSORPTION COEFFICIENT | CONTRIBUTION TO SCATTER |
|---|---|---|
| Elastic (coherent) scattering | No contribution to absorption as no energy is permanently absorbed by the medium | Makes only a very small contribution to the total scatter in medical radiography |
| Photoelectric absorption | Usually the energy of the photon is absorbed and so this process makes a contribution to the total absorption coefficient | No scattering of the photon occurs as part of the photoelectric process |
| Compton scattering | Some of the energy of the photon is transferred to the medium during the scattering process and so it contributes to the absorption coefficient | This is the major source of scatter in medical radiography |
| Pair production | Depending on the energy of the photon, this process produces partial or complete absorption | This process produces no scattering of the primary beam photons |
23.4 Elastic (coherent) scattering
When the energy of a photon is considerably less than the binding energies of orbiting electrons of the atoms of the attenuator, the photon may be deflected from its path with no loss of energy after it has interacted with one of the electrons. This process is known as elastic scattering or coherent scattering (sometimes also known as classical scattering or Rayleigh scattering).
In this process the incoming photon interacts with an electron and raises its energy but does not give it sufficient energy to become excited or ionized. The electron then returns to its previous energy level by the emission of a photon which is equal in energy to the incoming photon but is in a different direction; hence scattering has occurred. The photon is scattered predominantly in the forward direction, because elastic scattering cannot occur if the recoil experienced by the atom as a whole during the scattering process is sufficient to produce excitation or ionization. There is no absorption since no energy has been given permanently to the material and the attenuation, although present, is small since the majority of the photons are only scattered through a small angle. This is particularly so if the energy of the photon is higher than 100 eV and the atomic number of the attenuator is relatively low. The factors affecting the mass attenuation coefficient due to elastic scattering are shown in Equation 23.5:
where σcoh/ρ is the mass attenuation coefficient due to elastic scattering, Z is the atomic number of the attenuator and E is the photon energy.
In medical radiography, the effect of elastic scattering can be largely ignored since the average atomic number of tissue is low (approximately 7.4) and the photon energy is too high to allow significant elastic scatter.
Elastic scattering may occur as a result of a photon interacting with the nucleus of an atom of the attenuator but the effect is even less and so may safely be ignored in medical radiography.
In X-ray diffraction studies of crystals, it is the elastic scattering which is responsible for the coherent ‘reflections’ from the various atomic planes within the crystal lattice. Low-energy, monoenergetic X-rays are used for this purpose but only a small percentage are able to take part in the elastic scattering process.
23.5 Photoelectric absorption
The previous section has shown that elastic scattering has little significance for radiography except at extremely low photon energies. The process of photoelectric absorption, which we will now discuss, is important even at very low energies and continues to be important at the photon energies found in the typical X-ray spectrum for diagnostic radiography.
In photoelectric absorption, the X-ray photon is involved in an inelastic collision with an orbiting electron of an atom of the absorber. The photon gives up all its energy to the electron (and thus disappears) and the electron is ejected from the atom. As ejection of the electron from the atom is a necessary part of the process, photoelectric absorption can only take place if the photon energy is equal to, or greater than, the binding energy of the electron.
The process of photoelectric absorption is shown schematically in Figure 23.3, where it is assumed that the X-ray photon of energy hν ejects an electron from the K-shell of the atom. Some of the energy of the photon is used in overcoming the binding energy of the electron and the rest is given to the electron as kinetic energy. If we assume that the electron has a binding energy of B, then the kinetic energy after ejection (it is then referred to as a photoelectron) is (hv − B). The vacancy thus created in the K-shell will be filled by electrons in orbitals further from the nucleus performing a series of quantum jumps downwards, producing characteristic radiation (see Sect. 21.5) and the possible emission of Auger electrons (see Sect. 19.6.3) from orbitals further out from the nucleus. As you may remember from Section 21.5, the energy of the characteristic radiation is equal to the difference in energy of the electron before and after the quantum jump. In the case of X-ray photons interacting with atoms of body tissue, this energy difference is very small (normally between 1.2 × 10−2 eV and 1.8×10−2 eV) and so is in the infrared part of the electromagnetic spectrum.
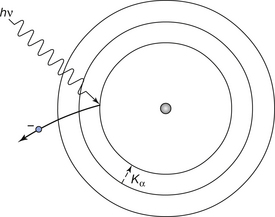
Figure 23.3 The process of photoelectric absorption and the subsequent emission of a quantum of characteristic radiation. The incoming photon (hv) is absorbed by the K-shell electron which is then displaced from that shell. The vacancy is filled from the L-shell with the emission of Kα characteristic radiation.
The probability of a photoelectric interaction occurring at a particular shell depends on the binding energy of the electrons in the shell and the energy of the incoming photon. The probability is zero when the energy of the photon is less than the binding energy of the electron; it is greatest when the photon energy is equal to the binding energy and thereafter decreases rapidly with increasing photon energy. A graph of the mass absorption coefficient for photoelectric absorption in lead is shown in Figure 23.4 to illustrate these points.
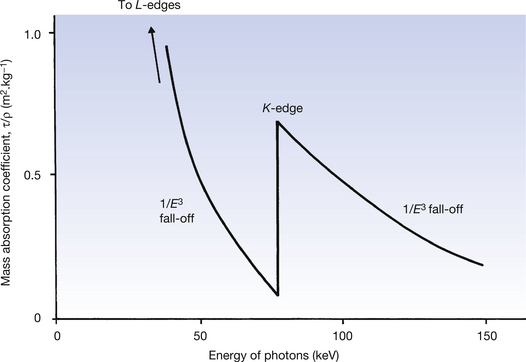
Figure 23.4 The mass absorption coefficient for the photoelectric effect in lead and its variation with photon energy.
The outer orbital shells of the atom are affected by the lower photon energies, only to have a reducing absorption as the photon energy increases. When the photon energy reaches the binding energy of the orbital then electrons in that orbital can take part in photoelectric absorption, thus producing a sudden increase in the amount of absorption. Such an increase is shown for the K-shell electrons of the lead atom in Figure 23.4. This sudden increase is known as an absorption edge and so Figure 23.4 shows the K absorption edge for the lead atom. Between the absorption edges, the mass attenuation coefficient due to the photoelectric effect (τ/ρ) is approximately proportional to 1/E3.
23.5.1 Photoelectric absorption, attenuation and absorption coefficients
The mass attenuation coefficient for the photoelectric effect (τ/ρ) is related to the atomic number (Z) of the absorber and the photon energy of the radiation (E). A very approximate guide to this relationship is given by the equation:
This equation applies to energies up to about 200 keV. At higher energies, the E3 term approximates to E2 and eventually to E.
In photoelectric absorption, the X-ray beam is both attenuated and absorbed since individual photons are removed from the beam (attenuation) and energy is imparted to the absorbing medium (absorption). The absorbed energy is composed of the following parts:
• Kinetic energy of the ejected photoelectron.
• Energy of recoil of the absorbing atom.
• Energy of the photons of characteristic radiation and Auger electrons
The ejected photoelectron is quickly brought to rest by the surrounding atoms and delivers its energy to them in the process. A similar thing happens with the Auger electrons. It is possible, however, for the characteristic radiation to escape from the absorber, especially if the energy of the characteristic radiation is high and it is produced near the surface of the absorber. In such cases, the mass absorption coefficient is slightly less than the mass attenuation coefficient since not all of the energy of the original photon has been absorbed by the medium. For this difference to be significant, the characteristic radiation must have high photon energy. The effect is thus only important in materials with high atomic numbers. As most of the atoms which make up body tissues have low atomic numbers, it may be considered that the mass absorption coefficient and the mass attenuation coefficient are equal for photoelectric absorption in practical radiography.
When we consider the absorption of X-ray photons to produce a radiographic image, we need to look at the linear attenuation coefficient. From Equation 23.6 we can see that, for the photoelectric effect at a given photon energy, this is proportional to the density of the absorber and its atomic number cubed. Bone is approximately twice as dense as soft tissue and its atomic number is also approximately twice that of soft tissue. For these reasons, the linear attenuation coefficient for photoelectric absorption for bone is approximately 16 times that of soft tissue. As the blackening on a radiograph is proportional to the radiation dose it receives, this explains why bones appear lighter than the same thickness of soft tissue. This process is the main one responsible for the contrast of the radiographic image.
23.6 Compton scattering
In the case of the photoelectric effect, the X-ray photon has an energy close to or just above the binding energy of the electron. If the energy of the X-ray photon is very much higher than the binding energy of the electron, the electron may be regarded as a free electron. The reaction between an X-ray photon and a free electron is known as Compton scattering and results in the partial absorption of the energy of an X-ray photon which undergoes such scatter. Because the interaction is between a photon and a free electron, the electron density of the material is important in determining the probability of Compton scatter occurring.
The Compton scattering process is shown diagrammatically in Figure 23.5. The incident X-ray photon has an energy E1(=hν1) and collides with the electron, which recoils, thus taking some of the photon energy. The energy remaining, E2(=hν2), is the energy of the deflected (or scattered) photon. Since E2 is less than E1, then the wavelength of the scattered photon must be greater than the wavelength of the incident photon, as shown in Figure 23.5 (see Sect. 17.3.2 for the relationship between photon energy and wavelength). Compton scattering interactions are termed inelastic in that the photon energy is not conserved, although the total energy of the interaction is conserved.
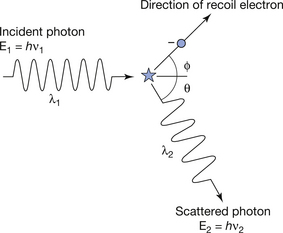
Figure 23.5 The process of Compton scattering. The scattered photon has less energy than the incident photon and may be scattered through any angle. The recoil electron is always scattered in a ‘forward’ direction.
After a Compton scattering process, the photon may travel in any direction but the electron can only travel in a forward direction relative to the incident photon. Thus, in Figure 23.5, θ may be any value, while Φ lies between 1°±90° relative to the direction of the incident photon. The division of the energy of the original photon between the electron and the scattered photon depends on the original photon energy (E1) and the angle through which it has been scattered. It may be shown that in order to preserve both energy and momentum, the following equation must be obeyed:
where the quantity λ2−λ1 is called the Compton wavelength shift, h is Planck’s constant, m is the mass of the electron and c is the velocity of electromagnetic radiation.
Two important conclusions may be drawn from Equation 23.7:
1. λ2−λ1 depends only on h, m, c and θ and so is not dependent on the wavelength of the incident photon or the composition of the attenuating material.
2. For a given value of scattering angle, θ, the value of λ2−λ1 is constant. Low-energy photons have longer wavelengths, so that the wavelength change represents a smaller fractional change in λ and hence only a slight reduction in the energy of the scattered photons. By applying the same argument, high-energy photons scattered through the same angle will experience a much larger fractional change in their wavelength and energy.
Equation 23.7 shows what happens to the photon wavelengths when scattered through an angle of θ. It does not give any information on the relative probability of a photon actually being scattered through that angle. Mathematical predictions based on quantum mechanics, and practical measurements, have shown that low-energy photons (up to about 100 eV) are scattered in all directions with almost equal probability. High-energy photons (greater than 1 MeV) are scattered predominantly in a forward direction, i.e. they have small scattering angles.
An alternative way of writing Equation 23.7 using the photon energy change instead of the wavelength change is:
where E1 and E2 are in keV.
23.6.1 Compton attenuation, absorption and scatter coefficients
The mass attenuation coefficient for Compton scattering (σ/ρ) is given by the equation:
As shown in the above equation, the probability of Compton scattering occurring in unit mass is inversely proportional to the energy of the photon – the amount of Compton scattering occurring in unit mass decreases as the photon energy increases.
It can also be seen from the above equation that the probability is also directly proportional to the electron density. As we saw in Section 3.6, it is possible to use Avogadro’s number to calculate the number of atoms per mole of an element (Na=6×1023 mol−1). We can therefore calculate the number of atoms per unit mass by simply dividing this by the mass number (A) for the element:
The number of electrons in a normal atom is the same as the number of protons and is given by Z – the atomic number (see Sect. 18.3). The electron density is the number of electrons per unit mass and is given by the equation:
If we assume that most elements in tissue have approximately equal numbers of protons and neutrons in the atomic nucleus, then the value of Z/A is 0.5. The exception to this rule is hydrogen which contains no neutron in its nucleus and so Z/A=1. Thus hydrogen contains 6×1023 electrons per gram (or 6×1026 electrons per kilogram) whereas all other substances contain approximately half this value (between 2.5 and 3.5×1023 electrons per gram). The lower values are for the heavier elements which have a larger neutron-to-proton ratio.
Thus we can see from Equation 23.9 that, at a given photon energy, the mass attenuation coefficient due to Compton scattering is very similar for all elements except hydrogen where it is twice as large.
The mass absorption coefficient for Compton scattering (σa/ρ) represents the average energy transferred to the electron (and hence the medium) as a fraction of the total energy in the beam. As shown in the previous section, the higher the energy of the photon, the higher the average energy lost by the photon as a result of Compton scattering. This means that the electron takes more energy in the recoil process as the photon energy increases. As a result of this, the values for the mass absorption coefficient (σa/ρ) and the mass attenuation coefficient (σ/ρ) are closer to the same value at higher photon energies than they are at lower photon energies. This situation is shown in Figure 23.6.
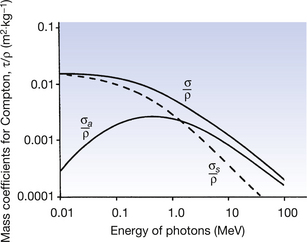
Figure 23.6 The mass attenuation, mass absorption and mass scattering coefficients for Compton scattering. As the energy increases, the absorbed energy (σa/ρ) becomes an increasing proportion of the total energy of the interaction (σ/ρ). See text for further details.
As we have already noted in Equation 23.9, the mass attenuation coefficient (σ/ρ) is inversely proportional to the photon energy and so we would expect σ/ρ to decrease as the photon energy increases. This is also shown in Figure 23.6.
Also shown in Figure 23.6 (represented by the broken line) is the mass scattering coefficient (σs/ρ). This represents the fraction of the total beam energy left to the photons after a Compton scattering event. At the point where the two curves cross (approximately 1.5 MeV), the electrons and the photons share equal amounts of energy. Below this energy, the scattered photon carries more energy than the electron, and above the energy, the reverse is true.
When considering the coefficients, it is important to remember that the mass attenuation coefficient is a measure of the total energy removed from the primary beam, whereas the absorption coefficient and the scattering coefficient are a measure of the proportion of energy removed from the beam by the electron and scattered photon respectively.
Since the sum of the energies of the electron and the scattered photon must equal the energy of the incident photon, then we can say:
In diagnostic radiography, many consider that the Compton process produces scatter which degrades the radiograph so makes no contribution to contrast. However, it should be remembered that linear absorption is also affected by the density of the medium and so the Compton process is responsible for the differences in absorption (and hence contrast) which we get between soft tissue and air or contrast agents and air in high kV radiography.
23.7 Pair production
Pair production resulting in the formation of two charged particles from a single high-energy photon can only occur at photon energies of 1.02 MeV or above. (Because of this high photon energy requirement, this is not a process encountered in diagnostic radiograph but is encountered in therapeutic radiography and in positron emission tomography (PET) scanning.) This is because this value represents the energy equivalent of the masses of two electrons. One of these, the negatron, has a negative charge, just like a normal electron, while the other particle, the positron, has an equal and opposite positive charge. The total charge present as a result of the interaction is therefore zero and equal to the uncharged photon creating the interaction. The process is illustrated in Figure 23.7.
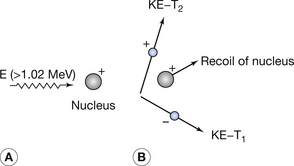
Figure 23.7 The mechanism of pair production. (A) A photon of energy (E) ≥1.02 MeV approaches the nucleus of an atom of the attenuating material. When the photon passes close to the nucleus, it produces a positron and a negatron, as shown in (B). KE, kinetic energy.
The interaction, which occurs as the photon interacts with the electrical field of the nucleus, is an example of mass–energy equivalence (see Sect. 16.4.1) as the energy from the photon is converted into mass. Any photon energy remaining after the interaction is passed to the particles as kinetic energy. This can be stated using the following equation:
where E is the energy of the photon, m0 is the rest mass of positron or electron, c is the velocity of electromagnetic radiation, T1 is the kinetic energy of the electron and T2 is the kinetic energy of the positron.
This equation can be further simplified to give:
If we consider the rest mass of the electron and positron pair then we can calculate that 2m0c2 is equal to 1.02 MeV. This explains why pair production will not take place if the photon energy is less than 1.02 MeV as this amount of energy is required to create the electron and positron pair. The equation may now be written in the form:
This shows that 1.02 MeV of the energy of the photon is used to create the electron and positron pair and the rest is given to the two particles in kinetic energy.
23.7.1 Attenuation, absorption and scatter coefficients for pair production
The linear attenuation for pair production is usually given the symbol π so that the mass attenuation coefficient is π/ρ. This is related both to the photon energy and the atomic number, as shown by Equation 23.14:
where E is the photon energy in MeV and Z is the atomic number of the attenuation material. (Note that this is the only process where the amount of attenuation increases with an increase in the photon energy.)
The kinetic energies of the electron and positron pair are absorbed by the medium as the particles slow down in it. Thus the energy absorbed by the medium is less than the energy of the original photon and is equal to (E − 1.02) MeV from Equation 23.14. The electron will eventually lose all its kinetic energy and come to rest. However, the positron will eventually collide with an electron and both will disappear with the emission of two photons of annihilation radiation where each photon has an energy of 0.51 MeV. This is the conversion of matter into energy, sometimes termed annihilation, and it is the reverse of pair production. If the two photons of annihilation radiation produced by this reaction are completely absorbed by the material, then the total energy absorbed is given by the relationship (E−1.02) + (2×0.51)=E, i.e. the whole of the energy of the original photon which caused the pair production.
There is, however, no certainty that this will happen. In such cases, the absorption coefficient (πa) is less than the attenuation coefficient (π) by a fraction: (E−1.02)/E. This can be rewritten as 1 − (1.02/E) and so we have:
By analogy with the absorption and scatter coefficient for Compton scatter (see Equation 23.12), in the case of pair production, we may write:
where πs is the fraction of the energy carried by the two annihilation photons each of energy 0.51 MeV, so πs=1.02/E.
However, in all but the most accurate work, it is usual to ignore the scattering coefficient, πs, since it is usually very small at the photon energies used in therapeutic radiography and so the more exact Equation 23.16 is replaced by more approximate relationships which assume that πs=0:
23.8 Relative importance of the attenuation processes in radiography
As we have already established in Equations 23.3 and 23.4, at a particular photon energy, some or all of the above processes may be competing to remove photons from the radiation beam. Thus the total linear attenuation coefficient, μ, is the sum of the linear attenuation coefficients due to photoelectric absorption, Compton scattering and pair production. Thus, the relationship Ix=I0e−μx can be rewritten as:
where τ is the linear attenuation coefficient due to photoelectric absorption, σ is the linear attenuation coefficient due to Compton scattering and π is the linear attenuation coefficient due to pair production. From the above equation we can also deduce the relationships given in Equations 23.3 and 23.4:
By applying the same argument to absorption, we can arrive at very similar equations:
Similarly, for scatter, we can write equations:
where the contribution to the scattering from pair production is ignored. It should also be remembered that photoelectric absorption contributes to energy absorption but not to scatter.
These points are illustrated in Figures 23.8 and 23.9 (see page 175) where the mass attenuation coefficients for photoelectric absorption (τ/ρ), Compton scattering (σ/ρ) and pair production (π/ρ) are shown for air and for lead. The total mass attenuation coefficients (μ/ρ) are shown as thick lines and the total mass absorption coefficients ((μa/ρ) are shown as broken lines.
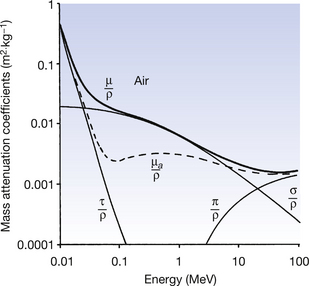
Figure 23.8 Mass attenuation coefficients for air – μ/ρ is the total mass attenuation coefficient, τ/ρ is the mass attenuation coefficient due to photoelectric absorption, σ/ρ is the mass attenuation coefficient due to Compton scattering and π/ρ is the mass attenuation coefficient due to pair production. Also shown (broken line) is the total mass absorption coefficient (μa/ρ).
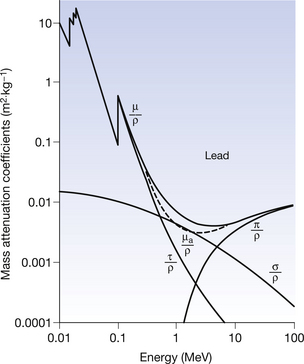
Figure 23.9 Mass attenuation coefficients for lead – μ/ρ is the total mass attenuation coefficient, τ/ρ is the mass attenuation coefficient due to photoelectric absorption, σ/ρ is the mass attenuation coefficient due to Compton scattering and π/ρ is the mass attenuation coefficient due to pair production. Also shown (broken line) is the total mass absorption coefficient (μa/ρ).
The following important points about the attenuation processes should be noted from the graphs:
• Photoelectric absorption dominates at low energies (up to 50–500 keV depending on the atomic number of the absorber).
• The absorption edges become more pronounced as the atomic number of the absorber increases.
• Compton scattering dominates over a wide range of energies (≈50 keV to 5 MeV) in all materials.
• The Compton region is almost identical in all attenuating materials (except hydrogen) because of the similarity of electron density of all materials. Thus the shape of the Compton curve is independent of the attenuator (the curve for σ/ρ is the same shape for both air and lead).
• Only photoelectric absorption and Compton scattering are important to diagnostic radiography.
• Pair production is only significant for high energies (≥1.02 MeV) and for attenuating materials of high atomic number.
• The significance of the contribution of pair production increases as the photon energy increases above 1.02 MeV. This is the opposite to the effect on the other processes of increasing the photon energy.
• Pair production is an important absorption mechanism in therapeutic radiography and in PET scanning.
23.9 Conclusion
As can be seen from the above, the attenuation of an X-ray beam as it passes through a patient’s tissues is a complicated affair depending on the energy of the photons, the tissue thickness and the atomic number of the tissue through which the beam passes. In both radiography and radiotherapy, we are able to alter the photon energy and this has important consequences for the absorption pattern and the intensity pattern of the beam emerging from the patient. These will be discussed in more detail in Chapter 25 when the formation of the radiographic image is considered.
The significance of the high absorption of materials with high atomic numbers (as shown by the graph for lead in Fig. 23.9) will also be considered in Chapter 27, which considers the topic of radiation protection.
A summary of the interaction processes, which may be useful in subsequent chapters of this text, is given in Table 23.2.
Table 23.2 Summary of the interaction processes
| PROCESS | DESCRIPTION OF INTERACTIONS | EFFECT OF Z, E | COMMENTS |
|---|---|---|---|
| Elastic scattering | Photon interacts with bound atomic electron. Photon energy is less than the electron binding energy. Photon is re-radiated from the material with no energy loss | σcoh/ρ ∝ Z2/E | No energy absorption in the medium. Photon scattered in the forward direction. Effect is negligible in biological tissues because of low Z |
| Photoelectric absorption | Photon of energy ≥ the binding energy of an electron interacts with bound electron and ejects it from its orbital. Photon disappears as all its energy is absorbed by the electron. Kinetic energy of the electron=E−binding energy. Atom recoils, conserving momentum | τ/ρ ∝ Z3/E3 | Ejected electron loses velocity to surrounding atoms. giving energy to them; i.e. absorption takes place. The electron vacancy created is filled by electrons making quantum jumps and so characteristic radiation is emitted |
| Compton scattering | Photon behaves like a particle and collides with a free electron. Energy of the incident photon is shared between the electron and the scattered photon | σ/ρ ∝ electron density/E | Energy of the displaced electron is absorbed by the medium, so Compton process produces attenuation and partial absorption. Electron densities of all materials except hydrogen are similar and so σ/ρ values are largely independent of the type of attenuator |
| Pair production | Photon of energy ≥1.02 MeV may spontaneously disappear in the vicinity of the nucleus of an attenuator atom, producing an electron and a positron. Atom recoils and preserves momentum. Positron eventually annihilated with an electron to form two photons of annihilation radiation, each with energy of 0.51 MeV | π/ρ ∝ (E − 1.02)Z | Probability of pair production increases with E above 1.02 MeV. Contribution of πs is very small compared to π and is often ignored |
In this chapter, you should have learnt the following:
• The meaning of the terms attenuation, absorption and scattering as applied to a beam of radiation passing through matter (see Sect. 23.2).
• The factors which control the probability of an interaction taking place between the X-ray photon and an atom of the attenuator (see Sect. 23.2.1).
• The meaning of the terms total linear attenuation coefficient and total mass attenuation coefficient and where it is appropriate to use each (see Sects 23.2.2 and 23.2.3).
• The interrelationships between attenuation and absorption (see Sect. 23.3).
• The mechanism of elastic (coherent) scattering and the factors affecting it (see Sect. 23.4).
• The mechanism of photoelectric absorption and the factors affecting it (see Sect. 23.5).
• The mechanism of Compton scattering and the factors affecting it (see Sect. 23.6).
• The mechanism of pair production and the factors affecting it (see Sect. 23.7).
• The relative importance of each of the attenuation processes in radiography (see Sect. 23.8).
Further reading
Ball J.L., Moore A.D., Turner S. Ball and Moore’s Essential Physics for Radiographers, fourth ed. London: Blackwell Scientific, 2008. (Chapter 17)
Curry T.S., Thomas S., Dowdey J.C., et al. Christensen’s Physics of Diagnostic Radiology. London: Lea & Febiger, 1990. (Chapters 4 and 5)
Webb S., editor. The Physics of Medical Imaging. Bristol: Institute of Physics, 2000. (Chapter 2)