Chapter 16 Spinal cord injury
INTRODUCTION
The prognosis for the patient sustaining spinal cord injury was poor until the latter part of the 20th century. An unknown Egyptian physician of 2500 BC describing spinal cord injury in the Edwin Smith Papyrus wrote:
This view continued until the work of Guttmann and others encouraged development of special centres throughout the world and saw the problems associated with spinal cord injury at last being addressed, although mortality from tetraplegia until the 1960s remained at 35% (Grundy & Swain 1996). Improvements in administering care at the time of the accident, technological advances in diagnosis and management have contributed to a continuing fall in mortality and morbidity rates over recent years. Kemp & Krause (1999) state that there has been a 2000% increase in life expectancy following spinal cord injury in the past 50 years compared with a 30% increase for the non-disabled population. Mortality is significantly higher during the first year after injury, especially for those with higher-level injuries. Life expectancy tables and other facts and figures are available online from the National Spinal Cord Injury Statistical Center at the University of Alabama, USA (see Further reading).
In a 50-year study in the United Kingdom, Frankel et al (1998) found that 92.3% of spinal cord injuries survived, the primary cause of death being attributed to respiratory complications. Another study has shown a projected mean life expectancy of 84% of normal for paraplegia and 70% for tetraplegia (Yeo et al 1998).
The respiratory care of patients with spinal cord injury is discussed in this chapter. It should, however, be remembered that effective management of patients requires a holistic, multidisciplinary approach, preferably in a spinal cord injuries unit, to ensure optimal rehabilitation.
MECHANICS OF RESPIRATION AND THE EFFECT OF SPINAL CORD INJURY
Normal respiration
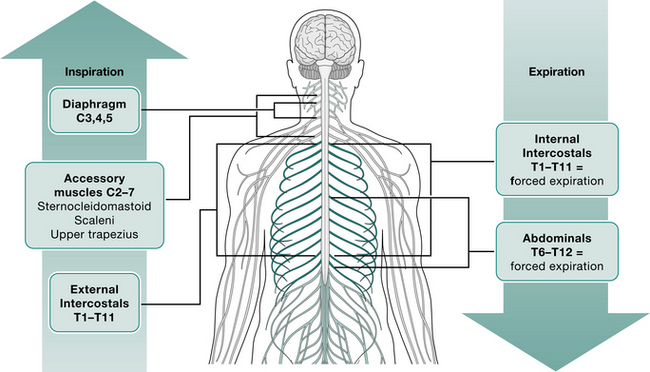
An understanding of normal respiratory mechanics is needed to appreciate the effect of a spinal cord injury. Figure 16.1 lists the muscles of respiration and their level of innervation.
The contraction and downward movement of the diaphragm and contraction of the intercostal muscles cause normal inspiration by generation of negative intrapleural and subsequent negative intrathoracic pressure (Lucke 1998). The intercostal muscles also work to stabilize the rib cage against the tendency for paradoxical inward movement caused by the negative intrathoracic pressure during inspiration.
Expiration is normally a passive process except during a forceful manoeuvre such as coughing or sneezing. This force is generated mainly by the abdominal muscles, assisted by the intercostals at large lung volumes (De Troyer & Heilporn 1980).
Respiratory complications are a major cause of death in the early stages of spinal injury (Lanig & Peterson 2000, Van Buren et al 1994), and those at the highest risk are:
Effects of spinal cord injury
Following a spinal cord injury the muscles below and, not uncommonly, at the level of the injury are paralysed. The higher the level of injury, the greater the effect on respiration (Linn et al 2000). To gain a more precise picture of the muscles affected, the physiotherapist should refer to the level of innervation of the respiratory muscles and relate this to the neurological level of injury.
Tetraplegic patients (i.e. those with an injury affecting T1 or above) will have lost the use of their intercostal muscles, which has a profound effect on respiratory function. Depending on the level of their injury, only part of the diaphragm may be spared, yet it has to provide them with all of their respiratory effort (Cohn 1993).
Paradoxical breathing
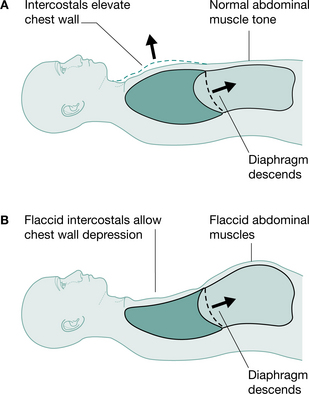
Tetraplegic patients injured at or below C4 will have partial or total innervation of the diaphragm and some accessory muscles of respiration and can be totally independent of mechanical ventilation. However, in the initial spinal shock stage, tetraplegic patients will have paralysis of their intercostal muscles, which, when flaccid, causes disruption to the mechanics of respiration. The usual splinting function of the intercostal muscles is lost and the negative intrathoracic pressure during inspiration causes paradoxical inward depression of the ribs (Lucke 1998, Menter et al 1997) (Fig. 16.2). This may lead to microatelectasis and an increase in the work of breathing (Fishburn et al 1990). With time the tendons, ligaments and joints of the rib cage stiffen owing to decreased active movement. This, together with spasticity of the intercostals, will provide some compensation for the loss of active control of these muscles and stabilize the rib cage, so that paradoxical breathing lessens (Axen et al 1985, Mansel & Norman 1990).
Cough
The ability to produce an effective cough is severely impaired in patients with cervical or high thoracic spinal cord injury (Roth et al 1997, Wang et al 1997). This is most marked when the intercostals are flaccid and the rib cage is at its most mobile. Patients who have loss of innervation to the abdominal muscles and the internal intercostals lose the ability to produce a forced expiration (Gouden 1997). De Troyer & Estenne (1991) have shown that patients with injuries at C5-8 can utilize the clavicular portion of pectoralis major to generate an expulsive force, although the extent to which this is functional is not clear. Linn et al (2000) found that in a group of patients with high tetraplegia (above C5) loss of peak expiratory flow rate was greater than 50% predicted. An effective cough for these patients requires external compression to produce the necessary large intrathoracic pressures; assisted coughing is discussed later.
The effect of position
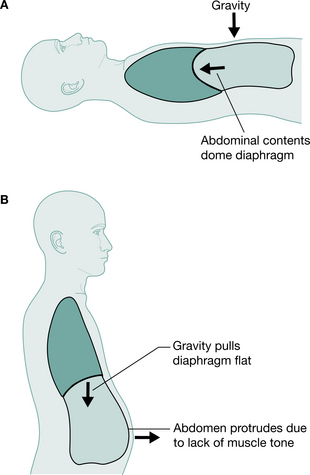
In the normal subject, mechanisms exist to ensure that adequate ventilation is maintained in all positions. In the supine position, contraction of the diaphragm displaces the abdominal contents without significantly expanding the rib cage, as the abdomen is more compliant than the rib cage. In standing, abdominal tone increases to support the abdominal contents, thereby decreasing abdominal wall compliance. Contraction of the diaphragm, intercostal and accessory muscles causes greater rib cage expansion, resulting in an increase in vital capacity in standing of about 5% (Chen et al 1990).
Positional changes will, however, affect the respiratory function of the tetraplegic patient. In supine, the weight of the abdominal contents forces the diaphragm to a higher resting level so that contraction produces greater excursion of the diaphragm. In sitting or standing, the weight of the unsupported abdominal contents increases the demand on the diaphragm, which now rests in a lower and flatter position (Chen et al 1990, Lucke 1998), decreasing effectiveness and restricting available excursion for creating negative intrapleural pressure (Fig. 16.3). Chen et al (1990) recorded a 14% drop in predicted vital capacity in the tetraplegic patient on changing position from supine to sitting or standing. Conversely, vital capacity of a tetraplegic patient rises by 6% when the bed is tipped 15° head down from supine (Bromley 1998). Linn et al (2000) showed a statistically significant decrease in forced vital capacity (FVC) in the erect position as compared with supine. It is therefore important with these patients not to assume that their respiratory ability will be sufficient in all positions.
Abdominal binders
Elasticated abdominal binders have been used on patients with high spinal cord injury for many years, both to minimize the effect of postural hypotension and aid respiration (Goldman et al 1986, McCool et al 1986, Scott et al 1993). Their effect is achieved by providing support to the abdominal contents, decreasing the compliance of the abdominal wall and thereby allowing the diaphragm to assume a more normal resting position in the upright posture (Alvarez et al 1981). Goldman et al (1986) investigated the effect of abdominal binders on breathing in tetraplegic patients and concluded that in the supine position there was no change, but when sitting there was a trend for improvement in lung volumes. This may help the patient considerably during the early stages of mobilization.
Incomplete injuries and position
If a spinal cord lesion is incomplete, i.e. there is sensation and/or movement below the level of injury, there are specific circumstances where the effect of position as described above can be reversed. A high-level C3–C5 incomplete lesion of the spinal cord can result in diaphragm paralysis but sparing of the intercostal muscles. Paradoxical breathing will not be observed. The patient will experience respiratory compromise in supine, which may be relieved by the sitting position, enabling the intercostal muscles to work more effectively. This scenario further emphasizes the need for accurate assessment of vital capacity in all positions where possible (see Respiratory assessment, below).
Sleep-disordered breathing and sleep apnoea syndrome after spinal cord injury
Sleep apnoea has been widely reported in patients with spinal cord injury and is known to be twice as prevalent as compared with the general population (Cahan et al 1993, McEvoy et al 1995, Young et al 1993). Stockhammer and colleagues (2002) defined two factors of sleep-disordered breathing and sleep apnoea. If a patient had more than 5 apnoeas per hour of sleep – the apnoea index (AI) and a respiratory disturbance index (RDI) of 15 or more, they were defined as having sleep apnoea syndrome (SAS). If their RDI was 15 or more but AI was less than 5 then they were defined as having sleep disordered breathing (SDB). In this study of tetraplegic patients 62% had SDB. Forty-eight per cent (55% of the men studied and 20% the women) had SAS. This is comparable with other studies that have found the prevalence of SAS to be 40% (Burns 2000) and 45% (Short et al 1992). Stockhammer et al (2002) found no correlation of level of lesion or spirometry values with the presence of RDI. They did, however, find a significant correlation of age, time since injury, body mass index (BMI) and neck circumference with RDI. They concluded that SAS incidence is high in tetraplegia, especially in older men with long-standing spinal cord injury. They also noted that patients with a high RDI did not complain of daytime drowsiness and were not obese, and therefore these individuals may not be investigated for SAS. Graham et al (2004) reported on two paraplegic men who had sleep apnoea who were aged 54 and 55 and were 37 and 25 years, respectively, post injury.
Berlowitz et al (2005) studied patients during the first year after cervical spinal cord injury. They found a high prevalence of SDB, which was first apparent 2 weeks following injury (60%) and peaked at 13 weeks or 3 months (83%). This then improved but remained significantly elevated (62%) at 1-year post injury. It should be noted that these outcomes cannot be compared directly with the studies quoted above, because this study reported on SDB whereas the others reported on SAS. Treatment for SAS with non-invasive ventilation is discussed later.
RESPIRATORY ASSESSMENT
Accurate assessment and regular review of respiratory status are vital. Initial assessment must be carried out as soon as possible to establish a baseline against which future deterioration or improvement can be monitored. Assessment is discussed in Chapter 1; however, in patients with spinal cord injury, the following details should also be considered:
PHYSIOTHERAPY TREATMENT
Respiratory management of the patient with spinal cord injury requires the application of the same principles as other respiratory problems; the skills used are discussed in Chapters 4 and 5. The goals of treatment include:
Treatment may be prophylactic or aimed at specific problems.
PROPHYLACTIC TREATMENT
This should include breathing exercises, modified postural drainage by regular turning and assisted coughing.
Breathing exercises
Breathing exercises to encourage maximal inspiration must be established at an early stage, but the therapist should be aware of the implications of lack of sensation of the chest wall. Exercises are directed to improve lateral basal and apical chest wall expansion and diaphragmatic excursion, but care must be taken to avoid tiring the diaphragm. Patients with intercostal paralysis are usually unable to perform localized breathing exercises.
Respiratory muscle training
Many authors have reported the use of respiratory muscle training for tetraplegia but there is a lack of randomized controlled trials, which makes the findings of the (often small) studies difficult to apply to the general patient group. Neither Stiller & Huff (1999) nor Brooks & O’Brien (2005) could recommend routine use of respiratory muscle training for tetraplegic patients (Chapter 5).
There have been some favourable findings. Liaw et al (2000) found that resistive inspiratory muscle training in tetraplegic patients, who were between 30 days and 6 months of injury, improved ventilatory function but noted that the patients needed to be highly motivated to gain benefit. Uijl et al (1999) reported on nine tetraplegic patients who underwent target flow endurance training and showed enhanced endurance capacity and an increase in aerobic exercise performance. Wang et al 2002 conducted a home programme of resistive inspiratory muscle training (RIMT) with a group of 14 tetraplegic patients who had sleep-disordered breathing and found that it had a positive effect on respiratory muscle strength and endurance. They suggested that this may reduce the patients’ sleep-disordered breathing. As with any training, the effect is soon lost when training ceases so this must be a lifelong commitment for long-term benefit to be maintained.
Incentive spirometry
Incentive spirometry enables respiratory training with immediate visual feedback to reinforce success. However, caution is needed in providing acutely injured tetraplegic patients with such a device, as a balance is needed between maintaining and improving lung volumes and respiratory muscle fatigue in the early days and weeks following injury.
Glossopharyngeal breathing
Glossopharyngeal breathing is another technique that can be used to increase lung volumes and assist secretion clearance (Chapter 5) in the high tetraplegic. Vital capacity may be increased by as much as 1000 ml (Alvarez et al 1981). Bach refers to the technique for augmenting inspired volume for patients with neuromuscular disorders to the extent where they can achieve a vital capacity of up to 1.7 litres (Bach 1993, Bach & McDermott 1990, Bach et al 1993). Pryor (1999) suggests that glossopharyngeal breathing (GPB) is a useful technique for increasing cough effectiveness in tetraplegia. In the high tetraplegic patient dependent on mechanical ventilation, other important benefits of GPB are to provide security in case of ventilator failure and independence from the ventilator for periods of time (Chapter 5).
TREATMENT OF THE PATIENT WITH RESPIRATORY PROBLEMS
In the presence of respiratory problems such as retained secretions or lung collapse, sputum clearance is of paramount importance and vigorous, aggressive treatment is often needed. Physiotherapy treatment plans will be determined by ongoing monitoring and assessment. Unless contraindicated by other complications, postural drainage either with an electric turning bed or manual turn into supported side lying, should be used as appropriate. Great care must be taken to maintain spinal alignment and cervical traction throughout treatment. The effect of positioning on lung ventilation and perfusion must be considered (Chapter 4). Patients should never be left unsupervised during postural drainage in case of sudden sputum mobilization, which could cause the patient to choke unless secretions are cleared by assisted coughing.
Treatment may consist of the active cycle of breathing techniques (Pryor 1999), vibration, shaking and chest clapping as necessary, followed by assisted coughing. ‘Little and often’ is the general rule as acutely injured patients will tire quickly, but treatment must be effective, using two physiotherapists if necessary. Where possible, treatment should link in with planned turn times to allow some rest between various procedures. Some authors recommend the use of the mechanical in-exsufflator (or CoughAssistTM machine), which has been used to great effect for many years (Bach 1993, Bach et al 1993, Tzeng & Bach 2000) (see Mechanical aids for assisted coughing, below).
Assisted coughing
Assisted coughing is a vital inclusion in any respiratory programme. Patients may be able to clear sputum from small to large airways, but will need assistance to produce an effective cough for expectoration. Assistance is provided by the application of a compressive force directed inwards and upwards against the thorax to create a push against the diaphragm, thus replacing the work of the abdominal and internal intercostal muscles. The sound of the resultant cough is the best indicator of the force required, but care must be taken to avoid movement of any fracture. Pressure directed down through the abdomen must be avoided, especially in the acute patient, owing to the possibility of associated abdominal injury or paralytic ileus. Care should also be taken in the presence of rib fractures or other chest injuries and therapists should position their hands away from the problem area to perform an assisted cough.
Bromley (1998) describes various methods of achieving assisted cough. Assisted coughing remains one of the most important techniques for airway clearance in the patient with an acute spinal cord injury. The technique needs to be relatively forceful and for this reason it is advisable for the therapist to lower the bed to gain the most advantageous position from which to perform the technique. However, great care must be taken not to allow any weights used for cervical traction to touch the floor.
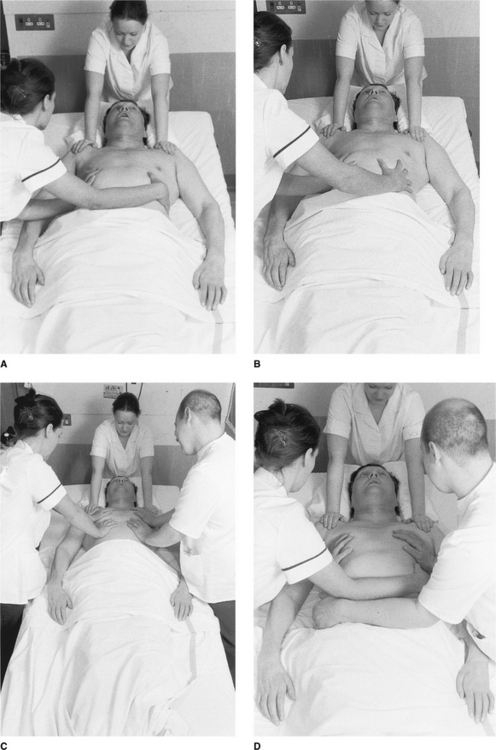
The spinal stability of the patient must be carefully considered and for the patient with an unstable cervical spine, a shoulder hold should be used to counter any movement of the fracture site. Figure 16.4 shows the methods that may be used in the supine patient requiring a shoulder hold.
If one person is assisting with the cough, hands should be placed so that one rests on the near side of the thorax and the other on the opposite side of the thorax, with the forearm resting across the lower ribs (Fig. 16.4A). As the patient attempts to cough the physiotherapist pushes inwards and upwards with the forearm and stabilizes the thorax with the hands. Alternatively, the hands are positioned bilaterally over the lower thorax (Fig. 16.4B) and, with elbows extended, the physiotherapist pushes inwards and upwards evenly through both arms. In the case of the patient with a large thorax or having particularly tenacious sputum, two people may be required to produce an effective cough (Fig. 16.4C & D).
Care must be taken to synchronize the applied compressive force with the expiratory effort of the patient. Once the cough is completed, pressure must be lifted momentarily from the lower ribs, thus enabling the patient to use their diaphragm to initiate the next breath. In the presence of paralytic ileus or internal injury, extreme care must be taken during assisted coughing to avoid the application of pressure over the abdomen. Patients should be encouraged to cough 3–4 times per day, with nursing staff involved in this process. If possible, patients should be taught self-assisted coughing when in a wheelchair and relatives should learn how to assist the patient to cough in both lying and sitting.
Mechanical aids for assisted coughing
The use of a mechanical in-exsufflation device to assist coughing has been documented in the literature as being effective with patients with neuromuscular disorders and respiratory muscle weakness (Chatwin et al 2003, Sancho et al 2004, Vianello et al 2005, Whitney et al 2002). Many of these studies used a device called the CoughAssistTM (JH Emerson Co, Cambridge, MA, USA). Whitney et al suggest using pressures in the range of 25 cmH2O positive pressure and −30 cmH2O negative pressure, while Chatwin & Simonds (2002) have reported effective coughs at pressures of +10 to +30 cmH2O and −10 to −30 cmH2O negative pressures.
There is an increasing body of literature which describes using electrical stimulation of the abdominal muscles to assist coughing (Langbein et al 2001, Lin et al 1998, Linder 1993, Stanic et al 2000, Taylor et al 2002, Zupan et al 1997). This technique, although often effective, is not widely available owing to the lack of standardized equipment available.
Airway suction
Nasopharyngeal suction may be used as a last resort if clearance by assisted cough alone is insufficient, but great care must be taken as pharyngeal suction can cause stimulation of the parasympathetic nervous system via the vagus nerve, resulting in bradycardia and even cardiac arrest. Hyperoxygenation of the patient with 100% oxygen before treatment will help to minimize this possibility (Wicks & Menter 1986). Atropine or an equivalent drug should be available for intravenous administration should profound bradycardia occur, defined as a heart rate of less than 50 with a continuing downward trend. Occasionally fibre-optic bronchoscopy may be necessary to treat unresolving lung or lobar collapse.
Intermittent positive pressure breathing and non-invasive ventilation
Intermittent positive pressure breathing (IPPB) and non-invasive ventilation (NIV) may be used in conjunction with other methods of treatment, particularly assisted coughing. Work by Rose et al (1987) concluded that increasing lung volumes had no major effect on lung function in stable tetraplegics. IPPB may be useful to aid the clearance of secretions by increasing inspiratory volume in patients with sputum retention and lung collapse (Chapter 5). The introduction of many new ventilators for NIV has overcome some of the limitations of IPPB machines such as lack of choice of interface and requirement for pressurized gas (Bott et al 1992).
It is useful to initiate NIV at an early stage, before the onset of fatigue, to enable the patient to become familiar with the machine (Tromans et al 1998) and if the patient does tire it can be used to provide assistance for up to 24 hours if required. This has been shown to be an effective way of preventing intubation and ventilation in acute spinal cord injury (Tromans et al 1998).
Unfortunately, in the case of acute spinal cord injury the psychological impact is devastating. Acceptance of a face or nose mask by the patient is often poor when they are also dealing with profound sensory deprivation from immobility and sensory loss. Kannan (1999) acknowledges that patient comfort and mental status can be reasons for failure of NIV and this is supported by clinical experience.
Non-invasive ventilation treatment for sleep apnoea syndrome
NIV can also be used to treat sleep apnoea syndrome (SAS) in spinal cord injury. Stockhammer et al 2002 reported that out of 31 patients recommended for BiPAP treatment only 16 opted to receive it and of those five subsequently discontinued treatment. They did comment that there was a strong correlation between symptoms of daytime drowsiness and acceptance of treatment. The group who experienced improvement of their drowsiness symptoms using the BiPAP demonstrated good adherence with treatment. Patients with SAS, who did not complain of drowsiness during the day, were the least likely to both accept treatment in the first instance and to continue with it. Burns et al (2000) also reported acceptance problems, with only 25% of patients with SAS undertaking treatment.
Readmissions to hospital with a chest infection
As survival following spinal cord injury increases, the cohort rises. This has led to an increase in hospital readmission for chest infection (Burns et al 2004, Capoor and Stein 2005). Physiotherapists in general hospitals are increasingly seeing patients with chronic spinal cord injury. This is partly due to the pressure on beds in the spinal units for treatment of acutely injured patients.
Patients with a vital capacity of less than 2 litres are at greatest risk of developing late-onset respiratory failure (Peterson & Kirshblum 2002). Some patients with long-standing spinal cord injury develop an altered response to carbon dioxide levels in the blood, in a similar way to patients with chronic obstructive pulmonary disease. Characteristically these patients are those who chronically hypoventilate and develop a raised partial pressure of carbon dioxide in the blood which leads to decreased chemoreceptor sensitivity to the presence of carbon dioxide (Stockhammer et al 2002). Consequently these patients have only a hypoxic element to their respiratory drive and administration of high inspiratory concentrations of oxygen may result in apnoea and respiratory arrest. If a readmitted patient requires ventilation, the weaning should be similar to that described later in Ventilation and weaning considerations as weaning should generally be slow in order to be successful.
Care of the ventilated patient
Around 20% of acute cervical spinal cord injuries will require some form of respiratory support (DiMarco 2005). Mechanical ventilation may be necessary in the following circumstances:
 Deterioration in respiratory function as a result of oedema or bleeding within the spinal canal causing the neurological level to rise, so affecting the diaphragm. Patients are most at risk during the first 72 hours
Deterioration in respiratory function as a result of oedema or bleeding within the spinal canal causing the neurological level to rise, so affecting the diaphragm. Patients are most at risk during the first 72 hours Respiratory muscle fatigue. The use of non-invasive ventilation, e.g. a bilevel positive pressure device, may be helpful in providing ventilatory assistance without the need for full ventilation (Tromans et al 1998) (see Intermittent positive pressure breathing and non-invasive ventilation above, and Chapter 10). Tracheostomy may be beneficial in reducing dead space by up to 50% (Bromley 1998)
Respiratory muscle fatigue. The use of non-invasive ventilation, e.g. a bilevel positive pressure device, may be helpful in providing ventilatory assistance without the need for full ventilation (Tromans et al 1998) (see Intermittent positive pressure breathing and non-invasive ventilation above, and Chapter 10). Tracheostomy may be beneficial in reducing dead space by up to 50% (Bromley 1998)Insertion of a minitracheostomy may be considered for patients with problems of retained secretions (Gupta et al 1989).
Physiotherapy goals for treatment of the ventilated patient are the same as those for the non-ventilated patient. Treatment will include modified postural drainage, vibration and shaking with manual hyperinflation, followed by suction to remove secretions. As previously stated, hyperoxygenation may be needed to prevent overstimulation of the vagus nerve resulting in bradycardia. Other methods for minimizing this are to use a suction catheter that is no more than half the diameter of the tracheostomy or endotracheal tube and to be as gentle and brief as possible (Carroll 1994, Dean 1997, Glass & Grap 1995). Frequency of treatment will be determined by assessment of the respiratory condition but should not exceed 15–20 minutes (Bromley 1998). Patients requiring ventilation due to complications from spinal cord injury are often not sedated and a system of communication must be established before physiotherapy is started.
Ventilation and weaning considerations
Early intervention with a non-invasive ventilatory technique may avoid progression of respiratory failure and the need for sedation, intubation and full mechanical ventilation (Tromans et al 1998). Where intubation and ventilation are necessary, weaning will typically progress from IPPV to pressure support and then, in the neurologically intact patient, continuous positive airway pressure (CPAP) is often used. However, in patients with hypercapnic respiratory failure, as seen in spinal cord injury, CPAP is not indicated as it cannot influence tidal volume or respiratory rate and therefore will not lower arterial carbon dioxide (Keilty & Bott 1992). Bilevel positive pressure may be of use during the weaning period, as it assists both expiration and inspiration (Ashworth 1990). Many machines now have a very sensitive flow trigger, which decreases the work of breathing.
Weaning from the ventilator should start as soon as the patient’s condition permits and is best performed with the patient supine, allowing the most effective diaphragm function (Chen et al 1990, Mansel & Norman 1990). Weaning must take into account the possibility that the patient’s respiratory muscles will have atrophied if ventilation has been prolonged. Cohn (1993) suggests that weaning should be thought of as a conditioning process for the diaphragm and also warns that fatigue of the muscle should be avoided. The goal must therefore be to achieve spontaneous breathing for short periods, several times a day, to avoid fatigue. A vital capacity of at least 15 ml per kilogram of bodyweight is generally used as a goal for the weaning patient and conversely as a threshold for ventilation for a self-ventilating patient (Chevrolet & Deleamont 1991, Mahanes & Lewis 2004).
A study by Peterson et al (1994) compared weaning onto a T-piece for progressive periods of time with the synchronized intermittent mandatory ventilation (SIMV) mode of ventilation, the latter being used frequently in the neurologically intact patient (Cull & Inwood 1999). Compared with SIMV, the T-piece group was almost twice as likely to wean successfully from the ventilator. Tromans et al (1998) reported on the use of BiPAP for weaning 15 patients from ventilation, with success in 13 out of 15 patients who weaned in an average of 32 days using a gradual decrease in pressure. Menter et al (1997) quote average weaning times for 74 spinal cord injured patients to be 36 days using progressive T-piece weaning. Peterson et al (1999) reported on the use of high tidal volumes to ventilate tetraplegic patients in association with T-piece weaning and concluded that tidal volumes of more than 20 ml per kilogram of bodyweight were most successful. McKinley (1996) reported one case of an initially ventilator dependent tetraplegic patient with a C3-4 lesion who was successfully weaned from a ventilator after 5 years. Other authors support the view that weaning can be prolonged and can take months, if not years (Fromm et al 1999, Oo et al 1999). Gutierrez et al (2003) used resistance and endurance respiratory muscle training to wean tetraplegic patients from ventilation. Although the numbers were small they reported success in fully weaning five patients with C4-C7 who had been ventilated for between 4 months and 2 years. They also achieved improvement in ‘off ventilator’ breathing time for two C2 tetraplegic patients using their weaning protocol.
Irrespective of technique used, regular recording of vital capacity, oxygen saturation and respiratory rate are minimal requirements to effectively monitor the patient’s progress (Menter et al 1997). If end-tidal carbon dioxide can be monitored, it is very useful and less traumatic than arterial blood gas sampling in the absence of arterial access (Cull & Inwood 1999). However, arterial blood gases are the gold standard and if the patient is unable to sustain his vital capacity, arterial carbon dioxide is likely to be rising and weaning should not be progressed until it has stabilized. The presence of other associated injuries and/or complications may have an effect on the success of the weaning process.
Long-term ventilation
For the patient on long-term ventilation, a battery-driven ventilator is attached to the wheelchair to enable mobility (Fig. 16.5). With increasing numbers surviving the initial injury owing to greater public awareness of resuscitation skills, home ventilation is now becoming more common, enabling these patients to undergo rehabilitation and to go home (Alderson 1999, Carter 1993). Planning must start at the earliest opportunity in conjunction with the patient’s local and community services in order to achieve timely discharge to an appropriate location. Comprehensive training is needed for the family and care team, for each patient, in order to minimize the risks of being fully ventilator dependent within the community.
The ethical dilemmas surrounding the ventilation of the high tetraplegic patient have challenged and continue to challenge medical practice (Gupta et al 1989, Maynard & Muth 1987). Only the ventilated tetraplegic knows what it is like to be a ventilated tetraplegic and only his carer knows what it is like to care for him. In a review of 21 patients who had required artificial ventilation (Gupta et al 1989), 18 stated that they would prefer a further period of continuous ventilation to being allowed to die. Sixteen of the 21 nearest caring relatives indicated that they were glad that their relative had been kept alive by ventilation. The study concluded that patients with spinal cord injury should be ventilated, provided that total emotional, educational and physical support could be given and maintained to all involved. This would seem to be most important.
In a case study, Maynard & Muth (1987) reveal how one individual’s request to cease life-supporting ventilation was met. They suggest that:
‘If rehabilitation is defined as achieving optimal quality of life for people with severe disability then quality must be defined by the disabled individual’
An individual’s perception of what constitutes acceptable quality of life will change over time (Purtilo 1986) and this poses the question of the feasibility of involvement of the newly injured patient and relatives in the decision regarding ventilation, as they are unable to appreciate the global implications of tetraplegia in the acute stage. However, ‘Right to Die’ issues have been in the media repeatedly in recent years, as individuals have challenged the established medical models of care (see Further reading). Physiotherapists must take great care to obtain consent and not to treat patients against their will.
Electrical and magnetic stimulation of breathing
A paralysed diaphragm can be electronically or magnetically stimulated if the phrenic nerve is intact and the cell bodies of C3, C4, C5 at the spinal cord are viable. The technique was first developed by Dr Glenn in the 1960s (Carter 1993). It should be noted, however, that many ventilator-dependent patients have sustained damage to the lower motor neurons and therefore are not suitable for this technique. Full preoperative assessment is required to ascertain a patient’s suitability for stimulation (DiMarco 2005). Electrodes may be placed to stimulate the phrenic nerve in either the neck, thorax or intramuscularly directly into the diaphragm (DiMarco 2005). Stimulation is achieved by means of a radio transmitter placed over the receiver. Extensive postoperative training is necessary to increase diaphragmatic endurance and to teach the patient, his family and carers the necessary skills and understanding of the device. For some patients phrenic nerve pacing will provide a full-time alternative to the ventilator and the tracheostomy will no longer be required, but for others the ventilator and tracheostomy will remain an emergency back-up.
Benefits include greater wheelchair mobility, elimination of the fear of accidental disconnection from the ventilator, loss of embarrassment and social stigma associated with being attached to a ventilator, improved speech, no noise from the ventilator, reduced need for carer input and improved well-being and overall health – particularly when decannulation of the tracheostomy has occurred (DiMarco 1999, 2005). Disadvantages include the need for major surgery involving thoracotomy, a prolonged hospital stay for the postoperative training, risk of surgical damage to the phrenic nerve and failure of the implanted device (DiMarco 2005).
CONCLUSION
Greater understanding of the problems of the spinal cord-injured patient has led to continuing improvements in morbidity and mortality rates. Respiratory complications can be managed more effectively with increased understanding of the problems facing these patients. Physiotherapists have, and will continue to have, much to offer in the respiratory care of the patient with spinal cord injury.
Alderson JD. Spinal cord injuries. Care of the Critically Ill. 1999;15(2):48-52.
Alvarez S, Peterson M, Lunsford B. Respiratory treatment of the adult patient with spinal cord injury. Physical Therapy. 1981;61(12):1737-1745.
Ashworth LJ. Pressure support ventilation. Critical Care Nurse. 1990;10(7):20-25.
Axen K, Pineda H, Shunfenthal I, Haas F. Diaphragmatic function following cervical cord injury: neurally mediated improvement. Archives of Physical Medicine and Rehabilitation. 1985;66:219-222.
Bach JR. Mechanical insufflation-exsufflation: comparison of peak expiratory flows with manually assisted and unassisted coughing techniques. Chest. 1993;104:1553-1562.
Bach JR, McDermott IG. Strapless oral-nasal interface for positive-pressure ventilation. Archives of Physical Medicine and Rehabilitation. 1990;71(11):910-913.
Bach JR, Smith WH, Michaels J, et al. Airway secretion clearance by mechanical exsufflation for poliomyelitis ventilator-assisted individuals. Archives of Physical Medicine and Rehabilitation. 1993;74(2):170-177.
Berlowitz DJ, Brown DJ, Campbell DA, Pierce RJ. A longitudinal evaluation of sleep in the first year after cervical spinal cord injury. Archives of Physical Medicine and Rehabilitation. 2005;86(6):1193-1199.
Bott J, Keilty SJ, Noone L. Intermittent positive pressure breathing - a dying art? Physiotherapy. 1992;78(9):656-660.
Bromley I. Tetraplegia and paraplegia: a guide for physiotherapists, 5th edn., Edinburgh: Churchill Livingstone, 1998.
Brooks D, O’Brien K. Is inspiratory muscle training effective for individuals with cervical spinal cord injury? A qualitative systematic review. Clinical Rehabilitation. 2005;19:237-246.
Burns SP, Little JW, Hussey JD, et al. Sleep apnea syndrome in chronic spinal cord injury: associated factors and treatment. Archives of Physical Medicine and Rehabilitation. 2000;81:1334-1339.
Burns SP, Weaver FM, Parada JP, et al. Management of community-acquired pneumonia in persons with spinal cord injury. Spinal Cord. 2004;42:450-458.
Cahan C, Gothe B, Decker MJ, et al. Arterial oxygen saturation over time and sleep studies in quadriplegic patients. Paraplegia. 1993;31:172-179.
Capoor J, Stein AB. Aging with spinal cord injury. Physical Medicine and Rehabilitation Clinics of North America. 2005;16:109-128.
Carroll P. Safe suctioning. Registered Nurse. 1994;57(5):32-36.
Carter RE. Experience with ventilator dependent patients. Paraplegia. 1993;31:150-153.
Chatwin M, Ross E, Hart N, et al. Cough augmentation with mechanical insufflation-exsufflation in patients with neuromuscular weakness. European Respiratory Journal. 2003;21:502-508.
Chatwin M, Simonds A. Mechanical technique for assisted cough (Correspondence). Physiotherapy. 2002;88(6):381-382.
Chen C, Lien I, Wu M. Respiratory function in patients with spinal cord injuries: effects of posture. Paraplegia. 1990;28:81-86.
Chevrolet J, Deleamont P. Repeated vital capacity measurements as predictive parameters for mechanical ventilation need and weaning success in Guillain-Barré syndrome. American Review of Respiratory Disease. 1991;144:814-818.
Cohn JR. Pulmonary management of the patient with spinal cord injury. Trauma Quarterly. 1993;9(2):65-71.
Cull C, Inwood H. Weaning patients from mechanical ventilation. Professional Nurse. 1999;14(8):535-538.
De Troyer A, Estenne M. Review article: the expiratory muscles in tetraplegia. Paraplegia. 1991;29:359-363.
De Troyer A, Heilporn A. Respiratory mechanics in quadriplegia. The respiratory function of the intercostal muscles. American Review of Respiratory Disease. 1980;122:591-600.
Dean B. Evidence-based suction management in accident and emergency: a vital component of airway care. Accident and Emergency Nursing. 1997;5:92-97.
DiMarco AF. Diaphragm pacing in patients with spinal cord injury. Topics in Spinal Cord Injury Rehabilitation. 1999;5(1):6-20.
DiMarco AF. Restoration of respiratory muscle function following spinal cord injury: review of electrical and magnetic stimulation techniques. Respiratory Physiology and Neurobiology. 2005;147:273-287.
Fishburn MJ, Marino RJ, Ditunno JF. Atelectasis and pneumonia in acute spinal cord injury. Archives of Physical Medicine and Rehabilitation. 1990;71:197-200.
Frankel HL, Coll JR, Charlifue SW, et al. Long-term survival in spinal cord injury: a fifty year investigation. Spinal Cord. 1998;36(12):868-869.
Fromm B, Hundt G, Gerner HJ, et al. Management of respiratory problems unique to high tetraplegia. Spinal Cord. 1999;37:239-244.
Glass CA, Grap MJ. Ten tips for safer suctioning. American Journal of Nursing. 1995;5:51-53.
Goldman J, Rose L, Williams S, et al. Effect of abdominal binders on breathing in tetraplegic patients. Thorax. 1986;41:940-945.
Gouden P. Static respiratory pressures in patients with post-traumatic tetraplegia. Spinal Cord. 1997;35:43-47.
Graham LE, Maguire SM, Gledhill IC. Two case reports of sleep apnoea in patients with paraplegia. Spinal Cord. 2004;42:603-605.
Grundy D, Swain A. ABC of spinal cord injury, 3rd edn., London: BMJ Books, 1996.
Gupta A, McClelland M, Evans A, El Masri W. Minitracheostomy in the early respiratory management of patients with spinal cord injury. Paraplegia. 1989;27:269-277.
Gutierrez CJ, Harrow J, Haines F. Using an evidence-based protocol to guide rehabilitation and weaning of ventilator-dependent cervical spinal cord injury patients. Journal of Rehabilitation Research and Development. 2003;40(5 Suppl 2):99-110.
Kannan S. Practical issues in non-invasive positive pressure ventilation. Care of the Critically Ill. 1999;15(3):76-79.
Keilty SEJ, Bott J. Continuous positive airways pressure. Physiotherapy. 1992;78(2):90-92.
Kemp BJ, Krause JS. Depression and life satisfaction among people aging with post-polio and spinal cord injury. Disability Rehabilitation. 1999;21:241-249.
Langbein WE, Maloney C, Kandare F, et al. Pulmonary function testing in spinal cord injury: effects of abdominal muscle stimulation. Journal of Rehabilitation Research and Development. 2001;38(5):591-598.
Lanig IS, Peterson WP. The respiratory system in spinal cord injury. Physical Medicine and Rehabilitation Clinics of North America. 2000;11(1):29-43.
Ledsome J, Sharp J. Pulmonary function in acute cervical cord injury. American Review of Respiratory Disease. 1981;124:41-44.
Liaw MY, Lin MC, Cheng PT, Wong MKA, Tang FT. Resistive inspiratory muscle training: its effectiveness in patients with acute complete cervical cord injury. Archives of Physical Medicine and Rehabilitation. 2000;81:752-756.
Lin VWH, Singh H, Chitkara RK, Perkash I. Functional magnetic stimulation for restoring cough in patients with tetraplegia. Archives of Physical Medicine and Rehabilitation. 1998;79:517-522.
Linder SH. Functional electrical stimulation to enhance cough in quadriplegia. Chest. 1993;103:166-169.
Linn WM, Adkins RH, Gong H, Waters RL. Pulmonary function in chronic spinal cord injury: a cross-sectional survey of 222 Southern California adult outpatients. Archives of Physical Medicine and Rehabilitation. 2000;81:757-763.
Lucke KT. Pulmonary management following acute SCI. Journal of Neuroscience Nursing. 1998;30(2):91-103.
Mahanes D, Lewis R. Weaning of the neurologically impaired patient. Critical Care Nursing Clinics of North America. 2004;16:387-393.
Mansel J, Norman J. Respiratory complications and management of spinal cord injuries. Chest. 1990;97(6):1446-1452.
Maynard F, Muth A. The choice to end life as a ventilator-dependent quadriplegic. Archives of Physical and Medical Rehabilitation. 1987;68:862-864.
McCool FD, Pichurko BM, Slutsky AS, et al. Changes in lung volume and rib configuration with abdominal binding in quadriplegia. Journal of Applied Physiology. 1986;60(4):1198-1202.
McEvoy DR, Mykytyn I, Sajkov D, et al. Sleep apnoea in patients with quadriplegia. Thorax. 1995;50:613-619.
McKinley WO. Late return of diaphragm function in a ventilator-dependent patient with a high tetraplegia: case report, and interactive review. Spinal Cord. 1996;34:626-629.
Menter RR, Bach JR, Brown DJ, et al. A review of the respiratory management of a patient with high level tetraplegia. Spinal Cord. 1997;35:805-808.
Oo T, Watt J, Soni BM, Sett PK. Delayed diaphragm recovery in 12 patients after high cervical spinal cord injury. A retrospective review of the diaphragm status of 107 patients ventilated after acute spinal cord injury. Spinal Cord. 1999;37:117-122.
Peterson P, Kirshblum S. Pulmonary management of spinal cord injury. In: Kirshblum S, Campagnolo D, DeLisa J, editors. Spinal cord medicine. Philadelphia, PA: Lippincott, Williams & Wilkins; 2002:136-155.
Peterson W, Barbalata L, Brooks CA, et al. The effect of tidal volumes on the time to wean persons with high tetraplegia from ventilators. Spinal Cord. 1999;37:284-288.
Peterson W, Charlifue MA, Gerhart A, Whiteneck G. Two methods of weaning persons with quadriplegia from mechanical ventilators. Paraplegia. 1994;32:98-103.
Pryor JA. Physiotherapy for airway clearance in adults. European Respiratory Journal. 1999;14(6):1418-1424.
Purtilo R. Ethical issues in the treatment of chronic ventilator dependent patients. Archives of Physical and Medical Rehabilitation. 1986;67:718-721.
Rose L, Geary M, Jackson J, Morgan M. The effect of lung volume expansion in tetraplegia. Physiotherapy Practice. 1987;3:163-167.
Roth EJ, Lu A, Primack S, et al. Ventilatory function in cervical and high thoracic spinal cord injury. American Journal of Physical Medicine and Rehabilitation. 1997;76(4):262-267.
Sancho J, Servera E, Diaz J, et al. Efficacy of mechanical insufflation-exsufflation in medically stable patients with amytrophic lateral sclerosis. Chest. 2004;125:1400-1405.
Scott MD, Frost F, Supinski G. The effect of body position and abdominal binders in chronic tetraplegic subjects more than 15 years post-injury. Journal of the American Paraplegia Society. 1993;16(2):117.
Short DJ, Stradling JR, Williams SJ. Prevalence of sleep apnoea in patients over 40 years of age with spinal cord lesions. Journal of Neurology, Neurosurgery, and Psychiatry. 1992;55:1032-1036.
Stanic U, Kandare F, Jaeger R, Sorli J. Functional electrical stimulation of abdominal muscles to augment tidal volume in spinal cord injury. IEEE Transactions on Rehabilitation Engineering. 2000;8(1):30-34.
Stockhammer E, Tobon A, Michel F, et al. Characteristics of sleep apnea syndrome in tetraplegic patients. Spinal Cord. 2002;40:286-294.
Stiller K, Huff N. Respiratory muscle training for tetraplegic patients: a literature review. Australian Journal of Physiotherapy. 1999;45:291-299.
Taylor PN, Tromans AM, Harris KR, Swain ID. Electrical stimulation of abdominal muscles for control of blood pressure and augmentation of cough in a C3/4 level tetraplegic. Spinal Cord. 2002;40:34-36.
Thomas E, Paulson SS. Protocol for weaning the SCI patient. SCI Nursing. 1994;11(2):42-45.
Tromans AM, Mecci M, Barrett FH, et al. The use of the BiPAP biphasic positive airway pressure system in acute spinal cord injury. Spinal Cord. 1998;36:481-484.
Tzeng AC, Bach JR. Prevention of pulmonary morbidity for patients with neuromuscular disease. Chest. 2000;118(5):1390-1396.
Uijl SG, Houtman S, Folgering HTM, Hopman MTE. Training of the respiratory muscles in individuals with tetraplegia. Spinal Cord. 1999;37:575-579.
Van Buren R, Lemons MD, Franklin C, Wagner MDJr. Respiratory complications after cervical spinal cord injury. Spine. 1994;19(20):2315-2320.
Vianello A, Corrado A, Arcaro G, et al. Mechanical insufflation-exsufflation improves outcomes for neuromuscular disease patients with respiratory tract infections. American Journal of Physical Medicine and Rehabilitation. 2005;84(2):83-88.
Wang AY, Jaeger RJ, Yarkony GM, Turba RM. Cough in spinal cord injured patients: the relationship between motor level and peak expiratory flow. Spinal Cord. 1997;35:299-302.
Wang TG, Wang YH, Tang FT, et al. Resistive inspiratory muscle training in sleep-disordered breathing of traumatic tetraplegia. Archives of Physical Medicine and Rehabilitation. 2002;83(4):491-496.
Whitney J, Harden B, Keilty S. Assisted cough: a new technique. Physiotherapy. 2002;88(4):201-207.
Wicks A, Menter R. Long-term outlook in quadriplegic patients with initial ventilator dependency. Chest. 1986;3:406-410.
Yeo JD, Walsh J, Rutkowski S, et al. Mortality following spinal cord injury. Spinal Cord. 1998;36:329-336.
Young T, Palta M, Dempsey J, et al. The occurrence of sleep-disordered breathing among middle-aged adults. New England Journal of Medicine. 1993;328:1230-1235.
Zupan A, Savrin R, Erjavec T, et al. Effects of respiratory muscle training and electrical stimulation of abdominal muscles on respiratory capabilities in tetraplegic patients. Spinal Cord. 1997;35:540-545.
Facts and figures at a glance. University of Alabama, Spinal Cord Injury Information Network. Online. (www.spinalcord.uab.edu/show.asp?durki=21446)(updated June 2006, accessed 18 July 2007)
Woman welcomes ‘right to die’ ruling. news.bbc.co.uk/1/hi/health/1887281.stm (accessed 18 July 2007)