Single functioning ventricle
Overview of Single Functioning Ventricle
Pearls
• Optimal circulation in patients with palliated single ventricle is associated with signs of adequate systemic perfusion and a systemic arterial oxygen saturation of 75% to 85%.
 Systemic arterial oxygen saturation greater than 85% with signs of decreased systemic perfusion suggest pulmonary overcirculation (and inadequate systemic blood flow). Ongoing assessment is critical to verify adequate oxygen delivery and stable systemic perfusion.
Systemic arterial oxygen saturation greater than 85% with signs of decreased systemic perfusion suggest pulmonary overcirculation (and inadequate systemic blood flow). Ongoing assessment is critical to verify adequate oxygen delivery and stable systemic perfusion. Systemic arterial oxygen saturation less than 75% requires further evaluation and can lead to inadequate oxygen delivery. Interventions or therapies are needed to promote adequate pulmonary blood flow and pulmonary vasodilation and prevent pulmonary vasoconstriction.
Systemic arterial oxygen saturation less than 75% requires further evaluation and can lead to inadequate oxygen delivery. Interventions or therapies are needed to promote adequate pulmonary blood flow and pulmonary vasodilation and prevent pulmonary vasoconstriction.• A hematocrit greater than 40% is necessary to optimize oxygen carrying capacity in the child with single ventricle physiology.
• All patients undergoing Glenn, Fontan, or Kawashima procedures must have normal pulmonary vascular resistance.
• Caution is required when the patient with single ventricle patient receives nothing by mouth (i.e., is NPO) or has inadequate fluid intake, because the patient can rapidly become dehydrated with resulting inadequate systemic perfusion and decreased oxygen saturation. Hemoconcentration can increase risk of thromboembolic events.
Overview of Etiology
Single ventricle refers to any congenital cardiac malformation where one ventricle is hypoplastic or absent. The heart may have significant hypoplasia of an AV valve or the apical portion of either ventricle.591 The etiology of each defect producing single ventricle physiology is described separately in the following sections.
Overview of Pathophysiology
The physiology and hemodynamics resulting from a univentricular heart are determined by individual variations such as obstruction to flow within the heart, status of flow across the atrial septum, the volume and mixing of systemic and pulmonary venous return, the pulmonary vascular resistance and the status of the AV valve regurgitation.468 In all defects with single ventricle physiology, the systemic and pulmonary venous return will mix in the single ventricle with resulting systemic arterial oxygen desaturation.591
The volume and ratio of systemic and pulmonary blood flows are determined by the vascular resistances in the pulmonary and systemic outflow tracts and pulmonary and systemic circulations.468 The presence or absence of obstruction to pulmonary blood flow helps to determine the presenting clinical signs, including congestive heart failure and cyanosis.342 If no obstruction to pulmonary (artery) blood flow exists, pulmonary hypertension will be present early and, if untreated, can progress to severe pulmonary vascular obstructive disease by the age of 2 years.342 Obstruction to pulmonary venous return can also lead to pulmonary hypertension.
The patient with single ventricle physiology may have varying degrees of obstruction to systemic outflow (see Hypoplastic Left Heart Syndrome [HLHS] and also Aortic stenosis elsewhere in this section of the chapter) and heterotaxy. Such obstruction can contribute to pulmonary overcirculation and, once the ductus begins to close, to poor systemic perfusion.
The goal for single ventricle patients is a Fontan circulation where all systemic venous return is directed passively into the pulmonary arteries, without the assist of a ventricular pumping chamber.591 Patients with single ventricle lesions requiring Glenn- and Fontan-type palliations require the lowest possible pulmonary vascular resistance for survival.512 These procedures create nonpulsatile pulmonary blood flow that is passive so flow is dependent on a gradient between the systemic venous (right atrial) pressure and left atrial pressure.589
If a single ventricle is present without significant obstruction to pulmonary blood flow, hypoxemia will be present from birth because pulmonary and systemic venous blood will mix in the ventricle. If there is good mixing of pulmonary and systemic venous blood, hypoxemia may be mild. Once pulmonary vascular resistance falls, pulmonary blood flow increases and signs of congestive heart failure develop.
If the single ventricle is associated with significant obstruction to pulmonary blood flow, hypoxemia will be present and may be severe from birth. If pulmonary atresia is present, pulmonary blood flow is dependent on the ductus arteriosus, and severe hypoxemia will develop when the ductus begins to close.
Unrepaired patients with univentricular anatomy have a poor prognosis. About 70% of those with left ventricular anatomy die by age 16 years. If the right ventricle is the systemic ventricle, about 50% die within 4 years after diagnosis.624 Patients with double inlet left ventricle have survived to the seventh decade of life with unoperated, well-balanced circulations.22 Survival is very poor (23% at 5 years) if the patient has associated total anomalous pulmonary venous return.307 All patients with single ventricle have very complex lesions and many potential complications, so require continuous, lifelong cardiac care by experts in the care of patients with congenital heart disease.927
Overview of Clinical Signs and Symptoms
If there is no significant obstruction to pulmonary blood flow, cyanosis may be mild during the neonatal period. Once pulmonary vascular resistance falls in the first 2 to 6 weeks of life, pulmonary blood flow increases significantly, and signs of congestive heart failure develop. If signs of congestive heart failure develop earlier (during the first 2 weeks of life), associated lesions can be present, including AV valve abnormalities, coarctation or other lesions. The signs of congestive heart failure include tachypnea, tachycardia, hepatomegaly, failure to thrive, and diaphoresis.342 The risk of pulmonary hypertension is significant. Arterial oxygen saturation may near 90%, reflecting excessive pulmonary blood flow.
A grade 3-4 over 6 systolic murmur is heard over the left sternal border. A diastolic murmur of pulmonary regurgitation may be heard at the upper left sternal border.686 The chest radiograph will reveal increased heart size and pulmonary vascular markings.686
The ECG can be highly variable because the anatomy of the univentricular heart may vary widely.468 The electrocardiogram is suggestive of a single ventricle if Q waves are abnormal and Q waves may be absent in right precordial leads or noted only with right precordial leads. First or second degree heart block may be present.686 The ECG in patients with a common-inlet atrioventricular (AV) connection and a common AV valve typically demonstrates moderate to severe left axis deviation.342 The risk of complete heart block is high when the single ventricle is associated with congenitally corrected transposition (CCTGA or levo-transposition of the great arteries [L-TGA]).81
The two-dimensional echocardiogram is the most useful noninvasive tool for the diagnosis, and typically reveals two atrioventricular valves opening into the single ventricle. Echocardiography enables classification of the single ventricle, identification of any rudimentary chamber, and other anatomic details. Magnetic resonance imaging (MRI) is extremely valuable in detailing anatomy, extracardiac abnormalities, volumes, and function.342
Cardiac catheterization will demonstrate specific details of cardiac anatomy and function, AV valve function, and the status of the pulmonary vascular bed.342 Initial palliative treatment may involve interventional cardiac catheterization.
Overview of Management (Including Surgical Procedures)
The management of the child with a single ventricle is individualized based on the child's symptomatology and anatomy. Nonsurgical management is determined by the magnitude of pulmonary blood flow and the severity of hypoxemia. The ultimate goal for a single functioning ventricle is a staged Fontan circulation. The management from birth is directed toward making each patient an optimal candidate for the Fontan procedure.
The Fontan circulation is typically achieved in two stages, beginning at approximately 4 to 6 months of age with surgery to create a superior vena cava (SVC) to pulmonary artery shunt (bidirectional Glenn). Timing for the Fontan surgery is individualized for each patient,589 but is typically performed at about 1 to 2 years of age.591 The Fontan redirects the IVC blood flow into the pulmonary arteries, resulting in total separation of systemic and pulmonary venous blood.591 Until the single ventricle is corrected it is imperative that no air be allowed to enter any intravenous system, because this air may be shunted into the systemic arterial circulation, producing a cerebral air embolus (stroke).
Children with single ventricle will require ongoing assessment of oxygen delivery and systemic perfusion to ensure that both are adequate. Careful assessment is required even when the arterial oxygen saturation is within the expected ranges for condition.
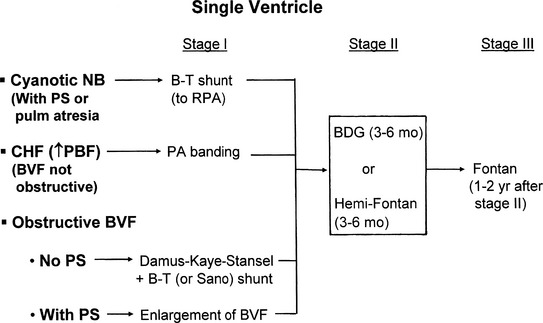
Beginning in the newborn period, most patients require palliative procedures to relieve cyanosis or prevent congestive heart failure (Box 8-39).589 The corrective procedures must also consider the presence of any associated defects; thus the particular surgical approach will be individualized. Cardiac transplantation may also be performed.
Box 8-39 Surgical Approach to Single Functioning Ventricle
From Park MK. Pediatr cardiol for practitioners, ed 5, Philadelphia, 2008, Elsevier (Fig. 14-63).BDG, Bidirectional Glenn; B-T Blalock-Taussig; BVF, bulboventricular foramen; CHF, congestive heart failure; NB, newborn; PBF, pulmonary blood flow; PS, pulmonic stenosis; RPA, right pulmonary artery.
If significant obstruction to pulmonary blood flow is present, prostaglandin E1 administration is required during the first days of life to maintain ductal patency; a palliative procedure is then performed to stabilize pulmonary blood flow. A surgical pulmonary-to-systemic shunt can be created (see Fig. 8-44), and the ductus can then be ligated (Additional palliative shunts are illustrated in Evolve Fig. 8-5, and are described in Evolve Table 8-2 in the Chapter 8 Supplement on the Evolve Website). Depending on the interventions required for associated conditions (e.g., pulmonary artery arterioplasty), cardiopulmonary bypass may be required for the surgery. In selected cases the patient may instead undergo interventional cardiac catheterization as the initial palliation, for placement of a stent in the ductus arteriosus to maintain ductal patency and adequate pulmonary blood flow.18,93,614
If significant obstruction to systemic blood flow is present, such as variants of hypoplastic left heart syndrome, management during the first days of life will include administration of prostaglandin E1 to maintain ductal patency and systemic flow. A palliative Norwood, Damus-Kaye-Stansel procedure or hybrid stage I palliation is then performed (see Hypoplastic Left Heart Syndrome later in this section of the chapter).
General Nursing Care
General nursing assessment and anticipatory interventions for single ventricle circulation involve support of adequate tissue oxygen delivery. In true single ventricle defects, the systemic arterial oxygen saturation is almost directly related to the volume of pulmonary blood flow. The greater the volume of pulmonary blood flow, the higher the volume of oxygenated pulmonary venous blood returning to the left atrium to mix with the (desaturated) systemic venous return, and the higher will be the resulting (mixed) systemic arterial oxygen saturation.514 An arterial oxygen saturation of 80% indicates a pulmonary to systemic blood flow ratio (QP/QS ratio) of nearly 1:1, assuming 95% to 100% pulmonary venous saturation and a 65% mixed venous saturation.935
Nursing care practices and therapies must promote a balance between systemic and pulmonary blood flow so both are optimized. Even when the arterial oxygen saturation is adequate (i.e., near 80%), typical clinical signs used to evaluate systemic perfusion do not reliably indicate actual tissue oxygen delivery.384 The arterial blood gas is, however, an excellent indicator of hemodynamic stability in the patient with single ventricle physiology.935 Adequate peripheral perfusion is indicated by a normal pH and an arterial oxygen saturation near 80% with a Qp:Qs ratio near 1:1.935 The arterial oxygen tension (PaO2) must remain greater than 30 mm Hg. Even when the arterial oxygen saturation is as expected, children with single ventricle physiology require ongoing assessment to verify adequate oxygen delivery and stable systemic perfusion. The arterial oxygen saturation may be within the acceptable or targeted range despite profound circulatory derangements.881
Care is generally provided in room air, because oxygen is a potent pulmonary vasodilator that will increase pulmonary blood flow. A high inspired oxygen concentration can lead to excessive pulmonary blood flow (indicated by an arterial oxygen saturation greater than 85% to 88%) and resultant systemic hypoperfusion and development of lactic acidosis.935 The inspired oxygen concentration and ventilation support is tailored to maintain the PaO2 near 40 mm Hg, the PaCO2 near 40 mm Hg, and the pH near 7.40. Hypocarbia and creation of alkalosis result in pulmonary vasodilation, and so are avoided unless there is a need to increase pulmonary blood flow.
The hematocrit is maintained at least at 40% to optimize oxygen carrying capacity in these patients with cyanosis and chronic arterial oxygen desaturation. Efforts are made to minimize oxygen consumption by maintaining normothermia (and a neutral thermal environment), with provision of appropriate sedation and analgesia (see section, Hypoplastic Left Heart and Chapter 5, Analgesia, Sedation and Neuromuscular Blockade), especially during periods of instability.
Indicators of potentially inadequate cardiac output and systemic perfusion include development of tachycardia, tachypnea, mottling, cool extremities, prolonged capillary refill, decreased peripheral pulses, decreased urine output, metabolic acidosis, or a rise in serum lactate (typically greater than 2.2 mmol/L—verify normal ranges used by the laboratory).
Indicators of adequate cardiac output and systemic perfusion are a heart rate and respiratory rate that are appropriate for age and clinical condition (typically slightly elevated as the result of hypoxemia), consistent skin color with pink nail beds and mucous membranes, warm extremities, brisk capillary refill, strong peripheral pulses, appropriate urine output for fluid intake, normal pH, and normal serum lactate (less than 2.2 mmol/L—verify normal ranges of the laboratory). The target mixed venous oxygen saturation for the patient with single ventricle circulation is near 50% when measured in the SVC of patients after the Norwood procedure.881 The normal target arteriovenous oxygen saturation difference (systemic arterial saturation [SaO2] minus mixed venous oxygen saturation, or 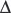 Sa-vO2) is near 25%.
Sa-vO2) is near 25%.
General care for patients with unrepaired single ventricle circulation will avoid factors that can lead to inadequate systemic perfusion. Hyperventilation with excessive use of oxygen is avoided, as are hypocarbia and alkalosis because they promote pulmonary vasodilation. Such pulmonary vasodilation can result in pulmonary overcirculation, with subsequent systemic hypoperfusion and development or worsening of congestive heart failure and myocardial dysfunction. This combination will produce acidosis despite an arterial oxygen saturation in excess of 90%,935 and requires rapid recognition and intervention.
Sinus tachycardia remains the most efficient way to increase cardiac output in newborns and infants. When any arrhythmias develop it is important to assess the impact of the arrhythmia on systemic perfusion; the greater the effect of the arrhythmia on perfusion, the more urgent the treatment needed. Excessive tachycardia, loss of AV synchrony and bradycardia are not well tolerated and may produce a fall in coronary as well as systemic perfusion, leading to rapid patient deterioration.
Systolic hypertension and high systemic vascular resistance may contribute to excessive pulmonary blood flow, increased myocardial work and increased oxygen consumption,520,602 requiring initiation of afterload reduction (vasodilator) therapy. No air can be allowed in any IV system because it may flow into the systemic arterial circulation causing a cerebral embolic event (e.g., stroke).
An arterial saturation less than 70% or PaO2 of less than 30 mm Hg is undesirable, as it indicates inadequate pulmonary blood flow, and eventually leads to the development of tissue hypoxia and acidosis. Management involves assessment and support of adequate airway and ventilation first. Pulmonary venous desaturation may be caused by hypoventilation, endotracheal tube obstruction or displacement, atelectasis, pleural effusion, infection and pneumothorax. Supplementary oxygen can be administered and titrated to achieve an arterial oxygen saturation above 75% but less than 85%. Preoperatively, if prostaglandin E1 is being administered and hypoxemia worsens, it is important to verify actual delivery of the infusion and assess for evidence of patency of the ductus arteriosus.
Presence of low mixed venous oxygen saturation (SvO2) can result from low cardiac output and inadequate systemic perfusion that may require support of both respiratory and hemodynamic function. An arteriovenous oxygen saturation difference (ΔSa-vO2) exceeding 30% can indicate worsening perfusion, as does a rising serum lactate concentration greater than 2.2 mmol/L (verify normal values with the laboratory). The SvO2 and serum lactate decrease before the clinical signs and symptoms change. A mixed venous oxygen saturation (SvO2) less than 40% is a very early indicator of cardiopulmonary deterioration in the patient with single ventricle circulation.384 These changes require immediate attention.
A rising AVO2 difference may also indicate an increase in oxygen demand in the face of limited oxygen delivery. Treat fever, prevent shivering and treat pain and identify and treat other causes of increased oxygen demand and consider neuromuscular blockade with sedation and analgesia during mechanical ventilation to reduce work of breathing. Anemia with subsequent impaired oxygen delivery can decrease arterial saturation and oxygen delivery, and requires transfusion to raise the hematocrit greater than 40%.
Hypotension may lead to decreased pulmonary blood flow and decreased arterial oxygen saturation, requiring IV volume administration or vasoactive therapy, and adjustment of vasodilator therapy. A diastolic blood pressure of less than 30 mm Hg is avoided because it will reduce coronary artery perfusion pressure (aortic end-diastolic pressure—right atrial pressure).591
An echocardiogram may be required to identify potential causes of arterial oxygen desaturation. Evaluation includes assessment of any existing shunt, examination of the ductus arteriosus (if present) and assessment of the size of the pulmonary artery and pulmonary veins (to identify any obstruction to pulmonary arterial or pulmonary venous flow), and assessment of diaphragm and cardiac function.
Caution is required to avoid excessive diuresis and avoid, identify, and promptly treat any dehydration. Dehydration with decreased intravascular volume can lead to lower cardiac output and systemic arterial oxygen saturation and can also produce hemoconcentration with subsequent risk of thromboemboli.
Treatment for elevated arterial oxygen saturation (greater than 85%), with pulmonary overcirculation and poor systemic perfusion includes measures to increase pulmonary vascular resistance. Inspired oxygen concentration is decreased to room air, to avoid the vasodilatory effects of increased alveolar oxygenation. If the child is receiving mechanical ventilation, interventions to increase pulmonary vascular resistance include changes in support to produce an increase in PaCO2 with intentional hypercarbia to 45 mm Hg or above. If the patient is tachypneic on the ventilator, sedation and neuromuscular blockade may be initiated. An arterial PaO2 of at least 30 mm Hg is desired,514 with a pH of 7.35 to 7.40. Sedation can be used to suppress the infant's intrinsic respiratory drive, allowing the PaCO2 to rise. PEEP can contribute to increased pulmonary vascular resistance.881
Alveolar hypoxia can be created with use of inhaled nitrogen to reduce the inspired oxygen concentration to 14% to 20% to create pulmonary vasoconstriction and thus limit pulmonary blood flow.935 Inhaled carbon dioxide (2% to 5%) can cause pulmonary vasoconstriction.935 When the arterial oxygen saturation is elevated, it may be appropriate to decrease systemic vascular resistance using intravenous vasodilators (e.g., Primacor up to 1 mcg/kg per minute, Phenoxybenzamine, or Nipride) or oral vasodilators (e.g., Enalapril, Captopril).
Despite the presence of an elevated arterial oxygen saturation, some infants may be minimally symptomatic, tolerating oral feedings with signs of adequate perfusion. Ongoing surveillance is still needed to evaluate adequacy of oxygen delivery and systemic perfusion. Echocardiogram may be completed to assess potential causes for elevated saturation, including extra sources of pulmonary blood flow from aortopulmonary collaterals or the development of an aortic arch obstruction with subsequent excessive flow through a prosthetic shunt or stented patent ductus arteriosus.
Throughout the care of patients with single ventricle, optimizing nutrition is critical for growth. Interventions are required to maximize caloric intake, monitor weight gain and minimize excessive metabolic demand. Infants with hypoplastic left heart palliation are expected to consume 110 to 130 cal/kg per day with fortified breast milk or formula prepared to deliver 24 to 27 calories per ounce.881 It is also common for the patient with single ventricle to have difficulty with oral intake, GE reflux, and altered feeding (see section, Common Clinical Conditions, Altered Nutrition). Normal rate of weight gains in infants following Norwood Stage I surgery have been achieved using calorie enhanced formulas with home surveillance of nutrition and weight gains.891
Palliative Surgery for Univentricular Heart with Excessive Pulmonary Blood Flow: Pulmonary Artery Banding
Palliative surgery for univentricular heart with uncontrolled increase in pulmonary blood flow can include pulmonary artery banding during early infancy. Banding can be performed via a midsternal incision, without cardiopulmonary bypass. The pulmonary artery banding can help prevent development of pulmonary vascular obstructive disease342; it will decrease the volume and pressure of pulmonary blood flow and decrease symptoms of congestive heart failure (CHF). Postoperative care includes titration of vasoactive support and intravascular volume and all aspects of typical perioperative cardiovascular care. The sudden application of new afterload by the pulmonary artery band may not be well tolerated, so myocardial dysfunction may be present postoperatively.
The systemic oxygen saturation following pulmonary artery banding should remain 75% to 80%. Following banding signs of an excessively tight band include signs of decreased pulmonary blood flow with excessive cyanosis, an arterial oxygen saturation less than 70%, poor perfusion, and acidosis. If the band is too loose, signs of pulmonary overcirculation with high arterial oxygen saturation (greater than 80%) and congestive heart failure may persist, and continued treatment for CHF will be required.
The Society of Thoracic Surgery 4-year database (2006-2010) reports hospital mortality for pulmonary artery banding is high at 12.8% in neonates and 5.5% in infants (compared to an overall pediatric surgical hospital mortality of 3.2%).823 The use of percutaneously adjustable pulmonary artery bands have improved survival and decreased the need for reoperation.842
Palliative Surgery for Univentricular Heart with Systemic Outflow Tract Obstruction: The Damus-Kaye-Stansel Procedure
This procedure requires cardiopulmonary bypass with aortic cross-clamping. In this procedure the pulmonary artery is transected before the bifurcation (i.e., the main trunk is separated from the branches). The end of the main pulmonary trunk is sewn into the side of the ascending aorta. This enables ventricular outflow to enter the original pulmonary artery trunk (bypassing the subaortic stenosis) and then flow into the aorta591 (Fig. 8-56). A modified Blalock-Taussig shunt is created to provide pulmonary blood flow. A patch is placed to close the opening in the distal main pulmonary artery.
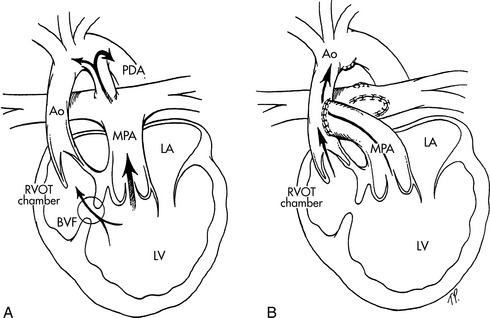
Fig. 8-56 The Damus-Kaye-Stansel procedure in a patient with double-inlet left ventricle and subaortic stenosis. A, Blood flow pathways before the surgical procedure. The aorta (AO) arises from a hypoplastic right ventricular outflow tract (RVOT) chamber that receives blood flow from the left ventricle through the bulboventricular foramen (BVF). The small outflow tract produces subaortic stenosis. The main pulmonary artery (MPA) arises from the left ventricle, and supplies blood flow to the pulmonary circulation, and, via the patent ductus arteriosus, to the aorta and systemic circulations. B, Blood flow pathways after the procedureThe MPA is transected and the trunk is sewn end-to-side to the ascending AO, providing unobstructed blood flow from the left ventricle into the aorta. A systemic shunt, such as a Blalock-Taussig shunt, is created (not shown) to provide pulmonary blood flow. LA, left atrium; LV, left ventricle; PDA, patent ductus arteriosus.
(From Nichols DG. Critical heart disease in infants and children, ed 2, Philadelphia, 1995, Elsevier, Fig. 38-3.)
Postoperative care for the Damus-Kaye-Stansel procedure is similar to that required after the Norwood procedure with a modified Blalock-Taussig shunt. Because the child has single ventricle hemodynamics, the desired arterial oxygen saturation is near 80%. Bleeding is a potential complication because the child has extensive aortic suture lines exposed to arterial pressure.591
Ongoing interstage mortality in patients with palliated single ventricle have led to establishment of specialized interstage home monitoring programs. These programs have improved survival of patients following the Norwood procedure and include monitoring of oxygen saturation and daily weight, and establishing parameters for additional repeat followup.318 National, multicenter quality improvement collaboratives by the Joint Council on Congenital Heart Disease are underway to reduce clinical practice variations and improve outcomes for children with HLHS.502 Risks for interstage death after the Norwood procedure and before the second stage Glenn procedure range from 5% to 15%881 (see Hypoplastic Left Heart Syndrome). Infants with palliative shunts are at risk for sudden interstage death.262
Family education and extensive preparation for optimal home management and follow-up care are required. Prophylaxis for risk of infective endocarditis is required because of the presence of prosthetic materials and presence of palliated cyanotic heart disease (see Bacterial Endocarditis later in this section of the chapter).
Palliative Surgery for Univentricular Heart with Inadequate Pulmonary Blood Flow: Systemic-to-Pulmonary Shunt
Pulmonary blood flow may be increased through the creation of a surgical systemic-to-pulmonary shunt. Alternatively, a stent may be placed in the ductus arteriosus to keep it open.
After surgery for a systemic to pulmonary artery shunt or placement of a stent within the ductus arteriosus to maintain ductal patency, titration of volume administration, inotropic support, and ventilatory management are required to balance systemic and pulmonary blood flow and prevent ventricular overload. Postoperatively, monitor for signs that the shunt is patent: an arterial oxygen saturation consistently greater than 70%, adequate systemic perfusion, low diastolic blood pressure with a wide pulse pressure (because the shunt creates a runoff of aortic blood flow into the pulmonary artery), and a continuous murmur. Clinical signs of a narrowing or closing shunt include a falling arterial oxygen saturation (a fall of greater than 10% from baseline or to less than 75%), rising diastolic blood pressure, a change in or disappearance of the shunt murmur, signs of poor perfusion, metabolic acidosis, or rising serum lactate greater than 2.2 mmol/L (verify threshold for lactic acidosis with the laboratory).
Shunt thrombosis is an emergency when the shunt is the only source of pulmonary blood flow. Immediate care can include administration of intravenous heparin, drugs to raise arterial blood pressure, and intravenous volume administration. Emergent echocardiogram is needed and urgent shunt revision may be needed.
The Society of Thoracic Surgery 4-year database (2006-2009) documents a relatively high mortality for prosthetic shunt surgery. Hospital mortality among neonates with single ventricle following creation of a modified Blalock-Taussig shunt is 6.9%, and 10% for a central shunt, while a 8.9% mortality is reported for a modified Blalock-Taussig shunt in infants without single ventricle physiology.823
Use of oral aspirin in children with prosthetic shunts appears to lower the risk of death and the occurrence of shunt thrombosis.533 Anticoagulation can initially include IV heparin (once the risk of postoperative bleeding is resolved), transitioning to oral aspirin therapy, but may include other anticoagulants such as Coumadin or Lovenox (a low molecular weight heparin) if the shunt or stent are judged to be at higher risk for thrombus occlusion.
Some patients continue to have antegrade pulmonary blood flow from the heart through the stenotic pulmonary outflow tract. When antegrade pulmonary blood flow from the heart is present, the arterial oxygen saturation is expected to trend higher than when a shunt alone provides all pulmonary blood flow.
Palliative Surgery for Univentricular Heart: The Bidirectional Glenn and Hemi-Fontan Procedure (Cavopulmonary Anastomosis)
The bidirectional Glenn and hemi-Fontan procedures may be performed as second-stage palliative procedures for hypoplastic left heart syndrome or as palliative procedures for other single functioning ventricle defects, such as tricuspid atresia. Both involve creation of a connection between the superior vena cava and the main pulmonary artery to divert all SVC blood to the pulmonary circulation. SVC flow is equal to about half of the total systemic venous return, and is slightly more than half of the systemic venous return in the infant.935
The bidirectional Glenn and the hemi-Fontan improve effective pulmonary blood flow and increase systemic arterial oxygen saturation by reducing the volume of systemic venous blood that enters the heart to mix with pulmonary venous blood. Both procedures also decompress the single ventricle because they reduce the total venous volume that the ventricle must eject, reducing the risk of ventricular dysfunction and atrioventricular valve insufficiency.935 As a result, both procedures improve survival and outcome from the later corrective procedure (the Fontan or variant of Fontan procedure).
The Glenn anastomosis can be performed with or without cardiopulmonary bypass. The hemi-Fontan is more extensive and requires cardiopulmonary bypass but facilitates later completion of the corrective Fontan (or modified Fontan) procedure.589,881 Typically, when either procedure is performed, previous palliative procedures (e.g., a systemic to pulmonary artery shunt or ductal stent) are taken down.
The bidirectional Glenn or hemi-Fontan and ultimate Fontan procedures require the presence of low pulmonary vascular resistance and low pulmonary venous pressures (which are related to the compliance of the single ventricle),298 and both require central pulmonary arteries that are undistorted and of adequate size.589 If pulmonary vascular resistance is elevated, these palliative procedures and ultimate correction cannot be performed. Therefore close collaboration between the cardiologist and cardiovascular surgeon is required to determine the ideal procedure and optimal time for surgical intervention.
Preoperative evaluation before second-stage palliation with the bidirectional Glenn or hemi-Fontan procedure includes echocardiogram with possible cardiac catheterization or cardiac MRI. The cardiac catheterization evaluates anatomy, hemodynamics, and suitability for the procedure. Interventional procedures during the preoperative catheterization may also include closure of aortopulmonary or venovenous collateral vessels and angioplasty dilation of the pulmonary arteries with possible stenting of any narrowed structures.
Cardiac MRI may be performed after first-stage palliations to evaluate for potential complications or sequelae. These can include aortic arch obstruction, pulmonary artery branch deformities and pulmonary vein compression or stenosis. The shunt status is also evaluated.314
The MRI before118 and after the Glenn procedure provides extensive information and in an increasing number of cases may substitute for diagnostic catheterization.314 Successful bidirectional Glenn procedures are achievable with calculated pulmonary vascular resistance up to 3-4 wood units (indexed to body surface area) or pulmonary artery mean pressure up to 20 mm Hg.589 For further information see Common Clinical Conditions, Pulmonary Hypertension.
The Hemi-Fontan Procedure
The hemi-Fontan procedure is so named because it diverts roughly half of systemic venous blood flow into the pulmonary circulation. The procedure requires cardiopulmonary bypass. It involves creation of an anastomosis between the side of the SVC and the main pulmonary artery (Fig. 8-57, A). The SVC is not disconnected from the right atrium, but a patch is placed at the SVC inlet to the right atrium to prevent SVC blood flow from entering the heart. The connection may alternatively include arteriotomies in the superior and inferior surfaces of the right pulmonary artery to allow for the connection to the superior vena cava (SVC).589
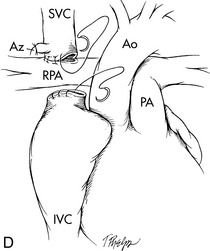
Fig. 8-57 The hemi-Fontan/Bi-Directional Glenn operations. A, The hemi-Fontan operation from the surgeon's point of view (this view is rotated 90 degrees counterclockwise from other figures of congenital heart defects and surgical repairs included in this chapter). A right atrial spiral incision is made, extending from the superior vena cava (SVC) to the surface of the right atrium (RA). B, After incision in the right pulmonary artery (RPA), a vascular confluence is created incorporating the cardiac end of the SVC, the cephalad portion of the RA (the portion nearest the head), and the RPA. C, The confluence is roofed with a patch of polytetrafluoroethylene (PTFE). A second PTFE baffle forms the floor of this confluence and separates the inferior vena cava (IVC) and coronary sinus (CS) blood from the SVC blood, thus diverting IVC and coronary sinus venous return through the atrial septal defect (ASD) to the left atrium. AO, aorta. D, The bidirectional Glenn operation. After transection of the superior vena cava (SVC), the cephalad end (i.e., bringing venous return from the head and upper extremities) is anastomosed to the right pulmonary artery (RPA). The cardiac end of the SVC is oversewn. The azygous (Az) vein is ligated (tied off). Blood flow from the inferior vena cava (IVC) passes through the atrial septal defect (ASD) to the left atrium, and thus contributes to left ventricular preload. AO, aorta.
(From Nichols DG. Critical heart disease in infants and children, ed 2, Philadelphia, 1995, Elsevier, Figs. 39-8 and 39-9.)
The Glenn Anastomosis
The Glenn anastomosis is performed with or without cardiopulmonary bypass. It involves division of the superior vena cava above the heart. The top portion (bringing venous blood from the head and upper extremities) is sewn directly into the top of the main pulmonary artery (on the patient's right side) and the cut end of the SVC that leads to the right atrium is oversewn. If a right and left SVC are present, a bilateral bidirectional Glenn is performed to join each SVC to the corresponding pulmonary artery.935 Occasionally, bilateral SVC's are present and the much smaller SVC can be ligated.935
The Glenn results in the diversion of all superior vena caval venous blood away from the heart and directly into the pulmonary arteries. The bidirectional Glenn immediately relieves overload of the functioning single ventricle, increases ventricular function, and improves the function of the AV valves.935 By directing a significant portion of desaturated systemic venous blood directly into the pulmonary circulation, effective pulmonary blood flow and systemic oxygenation is improved, particularly in young children.
In most centers, the Glenn procedure is performed as a stage 2 palliation technique for hypoplastic left heart syndrome. It follows an initial Norwood palliation, and is performed when the infant is about 4 to 6 months of age. This delayed timing for the second stage has followed reports that earlier performance of cavopulmonary anastomoses (i.e., at less than 4 months of age) resulted in lower arterial oxygen saturation in the early postoperative period and longer length of hospital stay.410
Postoperative Care After the Glenn or Hemi-Fontan
Potential early postoperative complications of the Glenn and the hemi-Fontan procedures include bleeding, chylothorax, pleural effusion, and superior vena caval obstruction. Arterial oxygen saturation should be near 80% to 85% for infants in room air.589,935 In the older patient, the arterial oxygen saturation may be lower after the Glenn because the volume of systemic venous return from the upper body that is diverted to the lungs is a smaller portion of total systemic venous return in older than in younger patients.935 Repair of additional lesions, such as pulmonary artery or aortic reconstruction, can add to the complexity of the postoperative course.
Oxygen therapy can be used without restriction following the Glenn procedure. During the immediate postoperative period, the head of the bed should be elevated with the head in a midline position, to enhance blood flow from the superior vena cava into the pulmonary arteries. The face and upper extremities are often edematous and plethoric in appearance during the immediate postoperative period (parents should be informed about this potential change in appearance in advance), but the child's appearance should return to normal within several days.
Small increases in pulmonary vascular resistance can lead to systemic venous hypertension with low cardiac output despite a technically successful procedure.476 Pulmonary hypertension can be present and pulmonary vascular resistance can be elevated after cardiopulmonary bypass.935,942 Treatment for elevated pulmonary vascular resistance can include the use of oxygen, vasodilators, and milrinone. Inhaled nitric oxide has been shown to be beneficial8,298 in the early postoperative period after Fontan and Glenn procedures.
Patients with elevated pulmonary vascular resistance (greater than 3.5 Wood units) or elevated mean pulmonary artery pressures (greater than 18 mm Hg) have been found to have higher risk for postoperative superior vena caval syndrome, low oxygen saturation (less than 70% to 75%), and death.174 High SVC pressure with high arterial oxygen saturation may indicate the presence of a large aortopulmonary collateral vessel producing an effective left-to-right shunt.591 Marked elevations in the SVC pressure result in decreased systemic perfusion.8
Initial postoperative SVC pressure may exceed 15 mm Hg, but should fall to 12 mm Hg or less within 2 days.591 For flow to occur through the Glenn, pulmonary vascular resistance must be low, so anything that causes pulmonary vasoconstriction will reduce blood flow through the Glenn and increase SVC pressure.
A superior vena caval syndrome may develop when SVC pressure is elevated; this syndrome produces upper body edema and plethora.935 Possible causes of superior vena caval syndrome include obstruction at the anastomosis, elevated pulmonary resistance or distortions of the pulmonary arteries with obstruction to flow.935 The elevated SVC pressure can be associated with an elevated transpulmonary gradient (SVC pressure minus intraatrial pressure).591 Identification of the cause for persistent SVC pressure elevation despite treatment requires a search for anatomic causes8 that can generally be treated during cardiac catheterization.
The Glenn anastomosis places cerebral blood flow and cardiopulmonary blood flow in a series, and autoregulatory mechanisms in these two tissue beds respond to changes in acid-base status and carbon dioxide tension in opposite ways.393,935 Increased alveolar oxygenation will produce pulmonary vasodilation. Factors known to produce pulmonary vasoconstriction (e.g., hypoventilation, acidosis, and reducing alveolar oxygenation) will cause cerebral vasodilation. Recent studies have shown that hypercarbia (maintaining the PaCO2 approximately 55 mm Hg) after a bidirectional Glenn will increase cardiac output by increasing cerebral blood flow, pulmonary blood flow and systemic arterial oxygenation, without significant elevation in pulmonary vascular resistance.98,99,393,935
Elevated atrial pressures may result from ventricular dysfunction, presence of arrhythmias with loss of AV synchrony, and atrioventricular valve regurgitation. The elevated atrial pressures can cause elevation in the SVC pressure.591 Heart rhythm disturbances may include sinus node dysfunction, treated with chronotropic medication therapy or temporary pacing.935
Pulmonary management includes weaning positive pressure ventilation to allow spontaneous ventilation as soon as possible. Early extubation is optimal to promote pulmonary blood flow. The arterial PaO2 should remain greater than 30 mm Hg. Hypercapnia with the PaCO2 approximately 55 mm Hg (range, 45-55 mm Hg) can increase cardiac output, systemic oxygenation, and pulmonary and cerebral blood flow.
After the bidirectional Glenn procedure the patient does not usually improve in response to inhaled nitric oxide,5 but cases of impaired pulmonary circulation after bidirectional Glenn have been treated successfully with inhaled nitric oxide298 and sildenafil646 to promote pulmonary vasodilation. Improved pulmonary blood flow results in improved systemic arterial oxygenation and systemic blood flow and decreases the transpulmonary gradient. If there is difficulty in weaning mechanical ventilation, rule out a diaphragm paralysis.
Systemic arterial hypertension is often present postoperatively. Causes can include pain, intracranial hypertension, or catecholamine secretion.935 Treatment may include administration of vasodilators. Caution is required to avoid aggressive lowering of the blood pressure, because it may have a negative affect on cerebral perfusion pressure.935 Irritability is a common postoperative finding and requires provision of comfort measures, analgesics and sedation (see Chapter 5, Sedatives, Analgesics, and Neuromuscular Blockade).
Severe hypoxemia and cyanosis, with low arterial oxygen saturation (less than 70% to 75%), or an arterial PaO2 of less than 30 mm Hg requires prompt identification and intervention. Such hypoxemia can be caused by pulmonary venous desaturation resulting from pulmonary problems such as atelectasis, elevated diaphragm, pleural effusion, tube obstruction, or pulmonary edema. Increased pulmonary vascular resistance and resulting decrease in pulmonary blood flow can also cause lower arterial oxygen saturation, because less pulmonary venous blood returns to the heart to mix with the systemic venous return from the inferior vena cava.
Persistent low cardiac output with low mixed venous oxygen saturation can lead to worsening hypoxemia and cyanosis, so treatment must focus on improving cardiac output. A hematocrit of at least 40% is maintained to optimize oxygen carrying capacity. A cardiac catheterization procedure may be required for complete assessment, definitive diagnosis, and intervention to treat the cause of the severe hypoxemia and cyanosis. Interventions may include sealing decompressing collateral venous vessels, angioplasty, and possible stenting of a stenotic anastomosis or pulmonary vessel.
Deterioration in arterial oxygen saturation in the weeks to months postoperatively can be caused by opening of veno-venous collaterals (from the superior vena cava into the right atrium or inferior vena cava). Such collaterals will decompress (reduce) the elevated SVC pressure,686 but will increase cyanosis. The venous collaterals can be identified by echocardiography with a “bubble study,” injecting agitated saline into an upper extremity and watching for the movement of the saline into the heart. An echocardiogram may also reveal a previously undiagnosed left superior vena cava or a baffle leak (in the intracardiac SVC patch for the hemi-Fontan).935
Persistent elevation of SVC pressure can lead to persistent pleural effusions and interstitial infiltrates if the pulmonary lymphatic vessels cannot drain into the hypertensive central veins.591 Pleural effusions are managed with diuresis and control of intravascular volume. Drainage is monitored for the development of chylous effusion, a whitish, opaque fluid. The chyle volume is increased, particularly with oral feedings containing fats (see Postoperative Care after Fontan Procedure, Pleural and Pericardial Effusions). Untreated pleural effusions further decrease pulmonary blood flow and arterial oxygen saturation, leading to worsening cyanosis.591
The bidirectional Glenn for infants has a hospital mortality of 1.8% in the 4-year (2006-2010) report from the Society of Thoracic Surgeons (STS) database, and 2.5% in infants undergoing bilateral bidirectional Glenn. Reported STS mortality for the hemi-Fontan procedure is 3.2%. Survival at 1 year was 96%, and at 5 years was 89% in a recent 10-year analysis,777 with preoperative atrioventricular valve regurgitation identified as a risk factor for death or transplant.
Patients undergoing the Glenn procedure in the past may have undergone the original Glenn connection (first reported in 1958), joining the SVC exclusively to the right pulmonary artery. This method is no longer used because it was associated with a high rate of complications, including the development of pulmonary arteriovenous fistulae, pulmonary artery or superior vena caval distortion, loss of continuity between right and left pulmonary arteries, and failure of normal right pulmonary artery development.342 The bidirectional Glenn procedure is not associated with significant occurrence of pulmonary arteriovenous fistulae.726
When pulmonic stenosis or a pulmonary band is present the bidirectional Glenn may be created and an existing systemic-to-pulmonary artery shunt may be left intact to provide additional antegrade pulmonary blood flow. The bidirectional Glenn with additional antegrade pulmonary blood flow may produce a resting arterial oxygen saturation of approximately 90%. This combination may serve as the final surgery if the patient is found to be a suboptimal Fontan candidate.133
If pulmonary arteriovenous malformations develop, desaturated venous blood within the lungs bypasses the pulmonary capillary bed and returns into the pulmonary veins without gas exchange, so hypoxemia and cyanosis worsen.881 The possible cause for the formation of these vessels is the lack of a “hepatic factor.”489 Hepatic factor is a postulated (but not yet identified) substance in hepatic venous blood; if hepatic venous blood does not traverse the pulmonary circulation (i.e., if it bypasses the lungs), patients are more likely to develop pulmonary arteriovenous malformations or fistulae. Completion of the Fontan procedure restores the “hepatic factor” flow through the pulmonary circulation, with potential resolution of pulmonary arteriovenous malformations.881
With time and growth the relative quantity of systemic venous return from the upper body diminishes, causing the child to become increasingly hypoxemic, cyanotic, and polycythemic.342 As the child grows, oxygen consumption increases and the growth of the lower body results in an increase in the volume of desaturated systemic venous returning to the heart via the inferior vena cava.881 Once this growth in the lower body occurs, the Glenn anastomosis will not provide sufficient pulmonary blood flow,241 and an additional surgical procedure will be needed to improve pulmonary blood flow. Typically, a Fontan-type correction is anticipated. In selected cases, an additional systemic to pulmonary artery shunt will be placed with no progression to the Fontan.
Selected patients will be eligible to have a “one-and-a-half” ventricle repair, with surgical correction of blood flow at the ventricular level and maintenance of the bidirectional Glenn.522 Alternatively, a bi-ventricular repair can be accomplished with takedown of the previously completed Glenn and Fontan procedures.379 Selected patients with unmanageable, severely decreased ventricular function may be referred for cardiac transplantation.
Corrective Surgery: The Completion Fontan Procedure (and Variations of the Fontan)
The correction of a single functioning ventricle is a variant of the original procedure described by Fontan in 1971. The procedure diverts all systemic venous blood flow into the pulmonary circulation. When accomplished after a bidirectional Glenn or hemi-Fontan (procedures that divert superior vena caval blood flow into the pulmonary circulation), the procedure diverts the inferior vena caval flow under a baffle or through an extracardiac conduit into the pulmonary circulation591 (Fig. 8-58). The Fontan is typically completed with use of cardiopulmonary bypass,591 although extracardiac Fontan completion without cardiopulmonary bypass is reported.797 Following the Fontan procedure the coronary sinus venous return will drain into the heart, resulting in a small right to left shunt with a decrease in systemic arterial oxygen saturation by one to two percent (Fig. 8-58).591
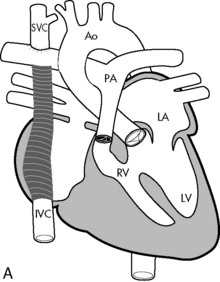
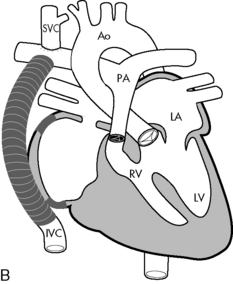
Fig. 8-58 Fontan correction for single ventricle. A, Lateral tunnel Fontan procedure for tricuspid atresia with normally related great arteries. Systemic venous return is channeled into the pulmonary circulation by direct anastomosis of the superior vena cava to the right pulmonary artery and insertion of an intraatrial baffle to divert the blood flow from inferior vena cava to the right pulmonary artery. If there is residual blood flow through the pulmonary valve, the main pulmonary artery is ligated (tied off) at the time of surgery. B, Extracardiac Fontan procedure for tricuspid atresia with normally related great arteries. This procedure is similar to the lateral tunnel Fontan procedure, except that the systemic venous return from the inferior vena cava is directed to the right pulmonary artery through an extraatrial conduit. AO, aorta; IVC, inferior vena cava; LA, left atrium; LV, left ventricle; PA, pulmonary artery; RV, right ventricle; SVC, superior vena cava.
(From Sidebotham D, McKee A, Gillham M, et al: Cardiothoracic critical care. Philadelphia, 2007, Butterworth Heinemann/Elsevier, Figs. 15-6 and 15-7.)
Preoperative Evaluation
Before the Fontan procedure, evaluation includes echocardiogram and cardiac catheterization to assess hemodynamics and suitability for Fontan procedure. Interventional procedures during the catheterization may include closure of aortopulmonary collateral vessels and dilation and possible stenting of narrowed structures including the pulmonary arteries or aorta.
“Ideal” hemodynamic criteria for a Fontan procedure include: low pulmonary vascular resistance (less than 2 Wood units, indexed to body surface area), pulmonary artery mean pressure less than 15 mm Hg, central pulmonary arteries that are large and without distortion, good ventricular function and low pulmonary venous atrial pressure (less than 5 mm Hg).589 Mild elevation in pulmonary vascular resistance (greater than 3 Wood units) can prevent a successful Fontan procedure.342 A 24-hour Holter monitor can detect loss of AV synchrony or bradycardia. After cardiac catheterization, patients may have transient complete heart block related to catheter manipulation.452
Fenestrated Fontan
The Fontan procedure produces passive flow of systemic venous blood through the pulmonary circulation. Because no ventricle ejects the flow forward into the lungs,942 blood flow requires that pulmonary venous and left atrial pressures and pulmonary vascular resistance remain low. Patients with increased risk factors, including elevated pulmonary pressure or elevated pulmonary vascular resistance or less than optimal ventricular function, may have restricted forward flow through a conventional Fontan, with decreased pulmonary venous return and low cardiac output. These patients may undergo a fenestrated Fontan, first described in 1990.108 In the fenestrated Fontan, there is a hole (fenestration) placed in the Fontan baffle to allow some inferior vena caval blood flow to enter the pulmonary venous atrium. This creates a right-to-left shunt whenever pressure in the Fontan baffle is elevated. The fenestration allows maintenance of cardiac output despite elevated pulmonary vascular resistance, although the systemic oxygen saturation will be lower than normal (as the result of the right-to-left shunt through the fenestration).
Use of the fenestrated Fontan and use of modified ultrafiltration to control intravascular volume have been shown to decrease the duration and severity of pleural effusions after the Fontan.306 Children with a fenestrated Fontan have been shown to have fewer complications, with less chest tube drainage and shorter length of hospital stay, and they are less likely to need additional postoperative procedures.523 With a right-to-left shunt, risk for systemic embolization exists. No air can be allowed to enter any intravenous device (see section, Common Clinical Conditions, Hypoxemia) as long as the fenestration is present.
Fenestrations are expected to cause systemic oxygen desaturation, so it is essential that the bedside nurse and receiving team know if such fenestrations were created, so the team will know to expect arterial oxygen desaturation. If the oxygen saturation rises or is higher than postoperative baseline values, the fenestration may have closed or become occluded; this must be promptly reported to the surgical team. Spontaneous fenestration closure may occur immediately after or days after surgery, causing acute deterioration with hypotension, renal failure, and excessive edema, in the presence of normally (fully) saturated blood.589 Echocardiographic evaluation for fenestration patency and prompt interventional venous cardiac catheterization may involve balloon dilation or placement of a stent55 or an ASD dilation device to reopen the fenestration. Cardiac catheter intervention perforation of the extracardiac conduit with implantation of a covered stent into the Fontan fenestration has been successful for the patient with a failing Fontan caused by closure of the fenestration.616
Outcomes have improved for both the standard and high-risk Fontan patient with fenestrated Fontan.621 By 1 year after surgery, 20% to 40% of fenestrations close spontaneously.686 Elective, future fenestration closure is completed with interventional cardiac catheterization and implantation of the Amplatzer device (see Atrial Septal Defect, and Evolve Fig. 8-2 in the Chapter 8 Supplement on the Evolve Website),389,621 or occlusion device. After fenestration closure, the arterial oxygen saturation is expected to rise.
If there is no fenestration in the Fontan circuit, all IVC blood flow will enter the pulmonary artery and systemic arterial blood should be near fully saturated (i.e., arterial oxygen saturation above 95% and near 100%) following surgery. Excellent outcomes with this procedure have been reported.758
Patients with heterotaxy variants may have complex venous anatomy including an interrupted IVC that does not enter the right atrium. Instead, the IVC joins to the azygous venous system and eventually enters the SVC; this anatomy is particularly likely in patients with polysplenia and bilateral left-sidedness.591 In these patients completion of a bilateral bidirectional Glenn is equivalent to near completion of a Fontan procedure because both the SVC and IVC blood flow will then enter the pulmonary arterial system. The only lower body venous flow not entering the pulmonary circulation is the hepatic blood.591 This bilateral cavopulmonary anastomosis procedure has been called the Kawashima procedure.
Postoperative Care After Fontan Procedure
Extubation with spontaneous ventilation should be allowed as soon as it is feasible after Fontan surgery because it may improve hemodynamics.935 Systemic venous return is facilitated by the subatmospheric pleural and intrathoracic pressures generated by spontaneous inspiration.942 If positive pressure ventilation must be provided, extremely high levels of positive end-expiratory pressure (PEEP) should be avoided because they can impede pulmonary blood flow and contribute to a fall in cardiac output.942 A PEEP of 3 to 5 cm H2O can be used without causing hemodynamic compromise,942 and can help improve ventilation-perfusion matching by reducing areas of microatelectasis.935 Positive pressure ventilation can decrease preload to the right and left atria and increase afterload to the pulmonary circulation.942 Ventilation goals, however, include extubation with spontaneous ventilation as soon as possible. Non-fenestrated Fontan patients should have arterial saturations above 95%.
Pulmonary pressure and pulmonary vascular resistance can be elevated following cardiopulmonary bypass.935,942 With even small increases in pulmonary resistance or any impediment to forward flow through the pulmonary vascular bed, the left heart filling will be compromised, leading to lower cardiac output.253 Treatment with oxygen and or nitric oxide has been successful in promoting pulmonary vasodilation and improving cardiac output.298,327
After the Fontan operation, low cardiac output is the most common and most severe complication. It is often caused by inadequate flow of blood into the pulmonary circulation that results from hypovolemia and inadequate systemic venous pressure (low right atrial [RA] and left atrial [LA] pressures), elevated pulmonary vascular resistance (low LA and high RA pressures), obstruction at the site of surgery, or pump failure.935 Additional causes include pulmonary artery distortion or hypoplasia, pulmonary venous obstruction, or residual left-to-right shunts (Table 8-34).342 The combination of low cardiac output with a high LA pressure is worrisome and may indicate potential left ventricular dysfunction, significant atrioventricular (AV) valve insufficiency or obstruction, loss of AV synchrony caused by arrhythmias, presence of obstruction to ventricular outflow, or cardiac tamponade.934,935 Any of these complications must be identified and treated.
Table 8-34 Differential Diagnosis of Low Cardiac Output After Fontan
| RA pressure | LA pressure | Cause(s) |
| Low | Low | Hypovolemia |
| High | Low | High pulmonary vascular resistance, baffle obstruction, pulmonary artery hypoplasia or stenosis |
| High | High | Ventricular dysfunction, atrioventricular valve stenosis or regurgitation, arrhythmia, outflow obstruction, tamponade |
Reproduced with permission from Wernovsky G, Bove EL: Early bidirectional cavopulmonary shunt in young infants. In Chang AC, Hanley F, Wernovsky G, Wessel DL, editors: Pediatric cardiac intensive care. Baltimore, 2008, Williams & Wilkins, p. 283.
Management of low cardiac output after Fontan is highlighted in Fig. 8-59. Inadequate intravascular volume and low central venous pressure can cause postoperative low cardiac output. Central venous pressure must equal or exceed the pulmonary artery pressure for flow into the pulmonary circulation to occur. A central venous pressure of 12 to 15 mm Hg may be needed to provide that pressure gradient and forward flow.750 Treatment of low cardiac output requires both judicious administration of intravenous fluid as well as administration of vasoactive medications (including inotropic agents and vasodilators). Therapy should be modified based on evaluation of the child's systemic perfusion, including assessment of mixed venous oxygen saturation, serum lactate and urine output. Avoid relative hypovolemia, with low central venous pressure and resultant low left atrial pressure.
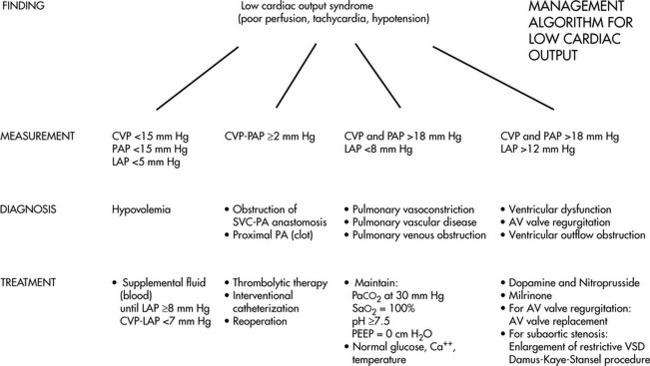
Fig. 8-59 Management algorithm for low cardiac output after Fontan-type correction of tricuspid atresia or single ventricle. AV, Atrioventricular; CVP, central venous pressure; LAP, left atrial pressure; PA, pulmonary artery; PAP, pulmonary artery pressure; PEEP, positive and expiratory pressure; SVC, superior vena cava; VSD, ventricular septal defect.
(Modified from Okanlami O, et al: Tricuspid atresia and the Fontan operation. In Nichols DG, et al, editors. Critical heart disease in infants and children. St Louis, 1995, Mosby.)
Diuretics, including Lasix and Aldactone (an aldosterone antagonist) are used to treat congestive heart failure,881 but aggressive diuresis is avoided. Afterload reduction is used to improve cardiac output and decrease single ventricle end-diastolic (filling) pressure,384 as well as decrease elevated systemic vascular resistance.589
Management of low cardiac output includes efforts to reduce pulmonary vascular resistance (see section, Common Clinical Conditions, Pulmonary Hypertension). These efforts include administration of supplementary oxygen even if arterial oxygen saturation is adequate.591 Avoid acidosis, hypoventilation, atelectasis (or other causes of alveolar hypoxia), hypothermia, and agitation. Anemia is treated to support adequate oxygen-carrying capacity and arterial oxygen content. Factors that increase oxygen consumption (fever, pain, agitation, and infection) should be avoided or promptly treated. When cardiac output is optimized and there is a good balance between oxygen delivery and oxygen demand, the mixed venous oxygen saturation will rise. In addition to monitoring and support of the arterial oxyhemoglobin saturation, the PaO2 must be monitored and maintained above 30 mm Hg (Table 8-35).
Table 8-35 Optimizing Pulmonary and Systemic Blood Flow in Patients with Single Ventricle
| Support Goals | Avoid and Correct |
| ARTERIAL oxygen saturation: 75%-85% | Arterial oxygen saturation >85%: May indicate excessive pulmonary blood flow, and cause CHF, respiratory distress, pulmonary edema |
| Arterial oxygen saturation <70%: Indicates decreased pulmonary blood flow, intrapulmonary shunt or reduced systemic venous oxygen saturation; will likely lead to development of acidosis. Verify patent airway and appropriate inspired oxygen and ventilation. | |
| Signs of adequate systemic perfusion | Inadequate systemic perfusion, metabolic acidosis |
| AIR in any IV system (risk of right-to-left shunt, embolus to systemic circulation) | |
| Mixed venous saturation near 50% (or within 10% of baseline) | Mixed venous saturation <40% indicates inadequate oxygen delivery-demand/consumption balance |
| Serum lactate <2.2 mmol/L | Serum lactate >2.2 mmol/L |
| OXYGEN: Carefully titrate use of oxygen to achieve desired oxygen saturation. Oxygen is a potent pulmonary vasodilator that typically increases pulmonary blood flow. | |
| ARTERIAL BLOOD GAS: Aim for “40-40-40” (PaO2: 40 mm Hg, PaCO2: 40 mm Hg, pH 7.40) | HYPERVENTILATION: Decreases cerebral blood flow and may contribute to reduced cardiac output and arterial oxygen saturation after cavopulmonary anastomosis. |
| ACIDOSIS increases pulmonary vascular resistance; metabolic acidosis indicates inadequate tissue oxygenation | |
| Keep Hematocrit at least 40% | Anemia |
A CVP greater than 15 to 18 mm Hg suggests difficulty with passive Fontan flow into the pulmonary capillary bed.591 Anatomic obstruction in the Fontan systemic venous pathway leads to elevated transpulmonary gradient (CVP minus LAP) with a value above 10 mm Hg suggesting difficulty with passive Fontan flow into the pulmonary circulation.591 Clinical signs include hepatomegaly, ascites, edema of the head and neck, anasarca, and signs of low cardiac output. The left atrial pressure is derived from the common atrial pressure with typical desired pressure slightly greater than 5 mm Hg (but not a lot higher).
Bleeding is most likely to occur if a coagulopathy existed preoperatively (related to chronic hypoxemia and polycythemia—see section, Common Clinical Conditions, Hypoxemia) or a significant amount of scar tissue (from previous palliative surgical procedures) was dissected. If synthetic polytetrafluoroethylene is used for the surgical correction, platelet adherence to the surface of this material will produce a fall in the child's platelet count immediately after surgery.
Continued cyanosis/hypoxemia may result from pulmonary venous desaturation related to atelectasis, elevated diaphragm, pleural effusions, pneumothorax, hypoventilation, or pulmonary edema (Table 8-36). Excessive cyanosis may be caused by a leak in the intracardiac baffle (or an intentional fenestration). If a fenestration is present within the Fontan circuit, right-to-left shunting with systemic desaturation is expected. Cyanosis may also result from decreased mixed venous oxygen saturation.
Table 8-36 Differential Diagnosis of Cyanosis After the Superior Cavopulmonary Anastomosis and the Fontan Completion
| Bidirectional Glenn/Hemi-Fontan | Fontan Completion | |
| Pulmonary venous desaturation | Ventilation/perfusion mismatch |
Ventilation/perfusion mismatch |
| Systemic venous desaturation | Decreased oxygen delivery |
Decreased oxygen delivery |
| Increased oxygen consumption | Increased oxygen consumption | |
| Decompressing vein | Decompressing vein | |
| Baffle leak (only for hemi-Fontan) | Baffle leak (only for lateral tunnel Fontan completion) | |
| Fenestration that is too large | ||
| Decreased pulmonary blood flow | Increased pulmonary vascular resistance | Increased pulmonary vascular resistance |
| Pulmonary venous hypertension | Pulmonary venous hypertension | |
| Restrictive atrial communication | Restrictive atrial communication | |
| Decompressing vein | Decompressing vein | |
| Baffle leak | Baffle leak | |
| Pulmonary artery obstruction |
From Marino BS, Spray TL, Greeley WJ. Separating the circulations: cavopulmonary connections (Bidirectional Glenn, hemi-Fontan) and the modified Fontan operation. In Nicholls DG, editor in chief. Critical heart disease in infants, children and adolescents, ed 2, Philadelphia, 2006, Saunders. Table 41-2.
Veno-venous collaterals may develop (from the SVC to the pulmonary venous atrium), but typically take weeks or months to develop after bidirectional Glenn or Fontan procedures. These collaterals increase cyanosis and require assessment and device closure during cardiac catheterization. It is uncommon for current bidirectional Glenn patients to develop pulmonary arteriovenous malformation (AVM) fistulae, which shunt blood flow away from the pulmonary capillaries and produce cyanosis. If these are present preoperatively in the Fontan patients, they will cause persistent cyanosis in the postoperative period. Pulmonary AVMs cannot be closed with coils or devices during cardiac catheterization, and must resolve over time. The cause for development of pulmonary AVMs is suspected to be lack of flow of a “hepatic factor” into the pulmonary circulation; when hepatic venous blood flows through the pulmonary circulation following the Fontan procedure, the problem resolves. Cyanosis and inability to wean from mechanical ventilation can also be related to diaphragm paralysis, with resultant risk for hypoventilation, atelectasis and increased pulmonary resistance.
Chronically cyanotic patients often develop collateral vessels from the systemic to the pulmonary arteries, resulting in increased pulmonary blood flow.434 The collateral vessels can cause ventricular volume overload,434 requiring transcatheter occlusion either before or after the Fontan procedure. Echocardiography with agitated saline contrast can be used to diagnose a right-to-left intrapulmonary or intracardiac shunt in the patient with unexplained cyanosis following a Glenn or Fontan operation.162,474 The agitated saline can help to detect location of a Fontan leak.474
Pleural and pericardial effusions are common postoperative problems requiring prolonged hospitalization.935 The causes are multifactorial.577 Elevated systemic venous pressure impedes lymphatic return into the venous circulation.591 Treatment by evacuation is needed because large fluid collections compress the lung, raising pulmonary vascular resistance,589 and significant pericardial effusions can cause tamponade.
An effusion is a chylothorax when milky appearing lymph fluid drains into the chest. Insertion of chest tubes may be necessary if pleural fluid accumulation compromises ventilation and oxygenation. Chylothorax can require initiation of and long-term management with a diet low in long-chain triglycerides,138 with Portagen as the infant formula. Because this fluid represents fluid loss from the body the total amount of fluid drained must be considered when evaluating the child's fluid balance. In addition, fat-soluble vitamins may be lost in the lymph fluid so replacement of vitamins A, D, K, and E is often required.138 Some children lose large proteins, including coagulation factors and fibrinogen, in this fluid; monitoring of the coagulation panel is advisable. Replacement of intravascular volume may be required with protein, electrolytes, and immunoglobulins.935 Octreotide IV infusion has been used with improvement.161 Thoracic duct ligation may be required for persistent drainage.
Recent factors associated with decreased incidence of effusions include use of fenestrations in the Fontan circuit, use of modified ultrafiltration, and the use of an “adjustable atrial septal defect.”935 Systemic venous congestion can lead to additional complications, including pericardial effusion, ascites, and liver congestion, which may produce liver dysfunction.
After the Fontan procedure, anticoagulation is typically initiated with aspirin, dipyridamole, or Coumadin (once intracardiac catheters and temporary pacing wires are removed). Slower venous blood flow occurs through the Fontan circuit, with passage through prosthetic materials, making patients susceptible to thrombus formation.591,881 Choice of therapy remains a matter of debate571 The persistent right-to-left fenestration shunt may also increase risk for thrombus formation and systemic embolization.
Rare complications include the development of plastic bronchitis with the formation of thick, tenacious protein casts within the bronchus.325,949 These casts cause life threatening obstruction of the airway and can result in pulmonary failure. Management is complex and involves repeated bronchoscopies.709
Arrhythmias can be problematic postoperatively. The presence of a normal sinus rhythm is not required for successful function of the Fontan, although the presence of chronic atrial fibrillation is worrisome because it often is associated with severe right atrial dilation and may indicate the presence of a severely restrictive atrial septal defect or left ventricular failure. Temporary or permanent cardiac pacing wires can be used postoperatively to control the cardiac rhythm.
Heart block or junctional tachyarrhythmias (loss of AV synchrony) raise intracardiac pressures, causing increased resistance to atrial filling and resulting in lower ventricular filling volume, additional pulmonary venous congestion, and low cardiac output as effective atrial contractility is lost or the atria contract against a closed AV valve.695 Loss of sinus rhythm, with escape rhythms such as junctional ectopic tachycardia, are poorly tolerated.589,935
Potential arrhythmias, including atrial flutter or fibrillation, primary atrial tachycardia, or accelerated junctional tachycardia, may develop postoperatively.342 Sinus node dysfunction is common after Fontan completion,591 possibly related to sinus node injury or interruption of sinus node blood supply.591 Sinus node dysfunction may result from an atrial septal defect; if an atrial septal defect is restrictive preoperatively, it leads to right atrial dilation.342 The atrial arrhythmias may reappear if significant right atrial hypertension develops postoperatively.
Preoperative Holter monitoring to establish baseline rhythm alterations can indicate the need for placement of permanent epicardial wires and a pacemaker generator during the Fontan procedure. Treatment can include temporary or permanent pacing and use of antiarrhythmic drugs. Atrial pacing wires allow for accurate rhythm diagnosis (see section, Common Clinical Conditions, Arrhythmias).589
Early perioperative Fontan failure may be related to multiple factors, including myocardial injury or elevated pulmonary vascular resistance.394,935 If elevated systemic venous pressure and low cardiac output persist despite maximal support, patients are brought to the cardiac catheterization laboratory for evaluation of their hemodynamics and anatomy. Transcatheter interventions have been completed safely in the early postoperative period after the Fontan procedure, including balloon angioplasty, occlusion of residual antegrade pulmonary flow or insertion of stents in narrowed Fontan or arterial structures.83 During interventional cardiac catheterization, an emergent fenestration may be created in the extracardiac Fontan circuit to improve cardiac output.616 A Fontan “takedown” to a bidirectional cavopulmonary shunt is uncommon, but may be a life-saving measure in the face of severe low cardiac output.589
Neurologic and developmental monitoring is required in patients with a single ventricle because risk for unfavorable developmental sequelae is high.648 Poor neurologic outcome is particularly likely in children with hypoplastic left heart syndrome who can have cognitive, motor, and neurologic deficits.554,767 Developmental evaluations are essential, with initiation of an early intervention programs, ongoing assessment and other interventions as needed.
Multiple factors create higher risks for developmental delay, including prolonged hypoxemia, unstable hemodynamics, multiple cardiac catheterizations, congestive heart failure, and a series of surgical procedures.648 An increased incidence of preoperative and postoperative periventricular leukomalacia, a nonspecific sign of cerebral white matter injury, has been found among patients who have undergone neonatal open-heart surgery.557
Physical activity level is reduced after Fontan procedures.596 Single ventricle patients with a Fontan procedure have been reported to have reduced exercise capacity.682,966 Obesity has the potential to increase pulmonary vascular resistance, as well as lead to other morbidities, and should be prevented in patients after the Fontan procedure.596 Ongoing care requires attention to promoting physical activity and healthy heart living.
After the Fontan procedure, most children demonstrate significant symptomatic improvement, although most also demonstrate abnormalities in exercise tolerance. In general the child demonstrates a high heart rate, high ventilation for oxygen consumption, and oxygen desaturation with exercise. Most children have an increase in physiologic dead space and a ventilation/perfusion mismatch, whether or not a Glenn anastomosis was performed before the Fontan procedure.934 Left ventricular ejection fraction often is reduced after the Fontan procedure, and the capacity to increase cardiac output in response to exercise varies from patient to patient. If left ventricular function is extremely poor, cardiac transplantation ultimately may be performed.934
Development of protein losing enteropathy (PLE) after a Fontan is associated with a poor clinical course.311 The incidence is 4% to 13%.756 Elevated systemic venous pressures are thought to contribute to protein-losing enteropathy, a loss of proteins throughout the GI tract,217 but the precise cause unknown.756 Symptoms include diarrhea, ascites, fatigue, abdominal pain, pleural effusions, shortness of breath, emesis, and peripheral edema.756 Fecal alpha1-antitrypsin is increased (greater than 200 mg/dL) and serum albumin is chronically low (less than 3.9 g/dL).756
Interventions include heparin administration, a high protein/low-fat diet, diuretic therapy, medications to improve cardiovascular function (afterload reduction, inotropic support), albumin infusions,612,756 prednisone,612,862 octreotide, budesonide,849 sildenafil730 and creation of atrial fenestration.612 After the onset of PLE, survival is 46% to 59%,756 with 50% mortality in 5 years.612
Monitoring of calcium, serum albumin, and total protein is required for patients with PLE, with intermittent infusions of these substances if needed. Subcutaneous heparin therapy has been shown to provide subjective symptomatic improvement in most patients, but it does not increase the clinical remission or decrease the need for albumin administration.756 No cure is known.217
The outcomes of the Fontan procedure with the evolving modalities of management require ongoing evaluation. Transcatheter fenestration with creation of an interatrial communication for the Fontan circuit has not prevented protein-losing enteropathy.914
Postoperative mortality associated with the Fontan procedure reported by centers in the Society of Thoracic Surgeons (2006-2010)823 database is 1.3% for the extracardiac fenestrated Fontan and 1.2% for the nonfenestrated procedure. The STS database reports postoperative hospital mortality of 1.0% for the fenestrated lateral tunnel.823 Adults (greater than 18 years old) presenting for Fontan revision/conversion have 8% mortality prior to discharge. Mortality for children with Down syndrome is higher following both the Glenn and Fontan surgery, and the procedures are rarely performed in these children.339
Results of the Fontan palliation continue to improve. Survival at 20 years after Fontan procedure ranges from 82.6%466 to 87%.672 The leading causes of death after Fontan are related to thromboembolism, sudden death, and heart failure.466 For double inlet left ventricle patients, 78% are reported to be alive at 12 years.468 Ten percent of the patients with single left ventricle develop complete heart block.452 A 15-year followup of extracardiac Fontan patients had 85% survival at 15 years (including operative deaths), with 92% of long-term survivors free from late heart failure. Three percent of the patients developed extracardiac conduit obstruction, and 3% experienced ventricular failure.319 Extracardiac conduit total cavopulmonary connection 10-year survival is reported at 93.6% with low morbidity, no restenosis, and 115/126 in normal sinus rhythm.642
Risk factors for adverse early and late outcomes include preoperative impaired ventricular function and elevated pulmonary vascular resistance.392 Late complications of the Fontan procedure include the development of conduit obstruction; this may cause sudden death if a Glenn anastomosis is not in place. Late arrhythmias, including atrial arrhythmias and complete heart block, also have been reported, requiring antiarrhythmic therapy and potential pacemaker insertion. Although many patients are able to resume normal daily activities without difficulty, others demonstrate persistent signs of systemic venous congestion and low cardiac output.
Late Fontan failure symptoms include those of congestive heart failure, as well as the development of protein losing enteropathy.394 Maintenance of low pulmonary artery pressure and vascular resistance with adequate unobstructed pulmonary blood flow and pulmonary venous return are important for optimal long-term Fontan hemodynamics.676 Cardiac catheter intervention dilation of Fontan pathway stenosis can improve hemodynamics, as well as improve or relieve chronic ascites.676 For those patients who develop intractable atrial arrhythmias requiring Fontan conversion, the procedure is completed with low mortality and is effective.582
Orthotopic cardiac transplantation remains an option for the patients with single functioning ventricule and may be an option for patients with severe ventricular dysfunction. The long-term morbidity and mortality related to transplant have prevented it from becoming a primary option for surgical management.342
Transcatheter Fontan Procedure
A transcatheter Fontan procedure has been developed as an interventional cardiac catheterization procedure using a covered stent.207,292,294,761 During the bidirectional Glenn, the patient undergoes placement of an aperture at the cardiac end of the SVC to pulmonary artery anastomosis. This aperture is perforated at the time of the transcatheter Fontan with placement of a stent. This procedure has resulted in improved outcomes compared with surgical Fontans, including shorter length of stay, postprocedural extubation, no arrhythmias, and no plural effusions. Successful Fontan stent dilation to accommodate for growth has been reported.761
Future Challenges and Advanced Concepts
Future issues for the young adult and adult with single ventricle palliations are numerous and reviewed by Khairy et al.468 Late medical, adult issues can include chronic low cardiac output, hypoxemia, protein-losing enteropathy, arrhythmias, ventricular dysfunction, thromboembolism, and Fontan pathway obstruction. Development of venous collaterals to the left atrium or development of pulmonary arteriovenous malformation may cause increased cyanosis.
Regular evaluation of the hepatic function is mandatory because late cirrhosis and hepatic dysfunction may develop and has been correlated with the duration of the Fontan circulation.48 Employment may be hindered because many patients have documented limitations in exercise capacity, as well as potential neurodevelopmental limitations. Lifelong care in an expert adult CHD clinic is critical to provide comprehensive, continuous, lifelong anticipatory management927 (see section, Common Clinical Conditions, Adult Congenital Heart Disease).
For advanced concepts in the care of the child with single functioning ventricle, see Box 8-40.
Box 8-40 Advanced Concepts: Single Functioning Ventricle
• Oxygen is a potent pulmonary vasodilator, and use of supplementary oxygen in patients with single ventricle is unnecessary if systemic arterial oxygen saturation is above 85% or the PaO2 is greater than 40 mm Hg.
• A fenestrated Fontan baffle will be created in patients with single ventricle who have less than optimal preoperative hemodynamics. The fenestration allows for right-to-left atrial shunting in the presence of elevated Fontan circuit pressures. This relieves venous congestion and helps to maintain cardiac output and oxygen delivery.
• Patients with a fenestrated Fontan are expected to have variable arterial oxygen saturation, with oxygen saturation inversely related to the magnitude of right-to-left shunting across the fenestration. Increasing saturation to near normal levels (>90%) may be a sign of a closing fenestration and must be evaluated immediately.
• New or increasing hepatomegaly or ascites is worrisome in patients after Fontan-type procedures, and suggests elevated Fontan circuit pressures.
Tricuspid Atresia
Pearls
• Type IB is the most common form of tricuspid atresia, and includes a ventricular septal defect and pulmonic stenosis. This defect presents with cyanosis in the first days of life.
• The infant with tricuspid atresia with large VSD and no pulmonic stenosis will develop heart failure in the first weeks of life as the result of excessive pulmonary blood flow.
• Single ventricle physiology care is required for all patients with tricuspid atresia before second-stage bidirectional Glenn (or hemi-Fontan).
• Staged palliations are required, concluding with Fontan-type hemodynamics.
Etiology
Tricuspid atresia results from a complete lack of formation of the tricuspid valve during fetal cardiac development. There is no blood flow between the right atrium and right ventricle. Tricuspid atresia is associated with a hypoplastic (very small) right ventricle (Fig. 8-60).
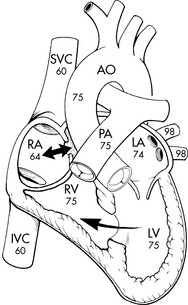
Fig. 8-60 Tricuspid atresia with normally related great arteries, ventricular septal defect and pulmonary stenosis (Type IB). Typical oxygen saturations within the cardiac chambers and great vessels are depicted. Tricuspid atresia may also be associated with transposition of the great arteries and without pulmonary stenosis.
(From Striepe V. Tricuspid atresia. In Kambam J, editor. Cardiac anesthesia for infants and children. St Louis, 1994, Mosby.)
In all forms of tricuspid atresia the systemic venous blood returns to the right atrium and must pass through an atrial septal defect or widely patent foramen ovale into the left atrium.253 The mitral valve (the only atrioventricular valve) connects the left atrium to the dominant left ventricle. A ventricular septal defect is often present.865
There are several forms of tricuspid atresia (Table 8-37). Normally related great arteries (Type I) are present in approximately 70% to 80%,253 but tricuspid atresia may also be associated with transposition of the great arteries (Type II) or other complex anomalies (Type III). Subcategories of tricuspid atresia have been named according to the degree of obstruction to pulmonary blood flow.406
Table 8-37 Classification of Tricuspid Atresia and Frequency of Occurrence
| Type I | Tricuspid atresia: normally related great arteries | 70%-80% |
| Ia | No VSD and pulmonary atresia | |
| Ib | Small VSD and pulmonary stenosis | |
| Ic | Large VSD without pulmonary stenosis |
| Type II | Tricuspid atresia: D-transposition of the great arteries | 12%-25% |
| IIa | VSD with pulmonary atresia | |
| IIb | VSD with pulmonary stenosis | |
| IIc | VSD without pulmonary stenosis |
| Type III | Tricuspid atresia: with L-transposition of the great arteries | 3%-6% |
From Epstein ML. Tricuspid atresia, stenosis and regurgitation. In Allen HD, Driscoll DJ, Feltes TF, Shaddy RE, editors. Moss and Adams' heart disease in infants, children, and adolescents, including the fetus and young adult, ed 7, Philadelphia, 2008, Lippincott Williams & Wilkins.
Tricuspid atresia was found in less than 3% of patients with congenital heart disease from the New England Regional Infant Cardiac Registry.330,724 Multiple cardiac anomalies are reported in less than 20% of cases.253 Coarctation of the aorta occurs most often, in about one third of patients with tricuspid atresia and transposition of the great arteries.253,454 Aortic arch obstruction was found in 25% of newborns with fetal diagnosis of tricuspid atresia.916 The 1-year survival rate for patients with tricuspid atresia without surgical intervention is as low as 10%, and is affected by the anatomy and associated defects.236 Survival into the sixth decade of life with no surgery has been reported in the presence of a balanced circulation.865 Continuous, lifelong care by congenital heart disease experts is required.
Pathophysiology
Tricuspid Atresia with Normally Related Great Arteries
The only outlet for the right atrium is an atrial septal defect, so systemic venous blood enters the right atrium and passes through the interatrial communication to the left atrium. Systemic and pulmonary venous blood completely mix in the left atrium and left ventricle, with the resulting saturation dependent on the relative volume of systemic and pulmonary venous blood. The left atrium is dilated but usually morphologically normal.406 The mixed venous blood passes through the mitral valve, into a large, well-developed left ventricle. From the left ventricle, some of the mixed venous blood can pass into the hypoplastic right ventricle though the ventricular septal defect (VSD), then into the pulmonary outflow tract and pulmonary artery. The volume of pulmonary blood flow depends on the size of the VSD and the degree of any pulmonary stenosis.253
The most common form of tricuspid atresia, present in about half of all patients with tricuspid atresia, is Type IB, tricuspid atresia with a small VSD and pulmonary stenosis.236,454 With this combination of defects, blood flow to the pulmonary circulation (and oxygenated pulmonary venous return to the left atrium) will be limited, so cyanosis will be present at birth. Pulmonary overcirculation will not develop when the pulmonary vascular resistance drops in the first weeks of life.253 In fact, pulmonary blood flow is decreased, and cyanosis can be significant, but no heart failure is present. The patients are at risk for the development of systemic consequences of chronic hypoxemia and resultant polycythemia (see section, Common Clinical Conditions, Hypoxemia). The infundibular stenosis can create a dynamic obstruction that can become severe.
The atrial septal defect is often a widely stretched patent foramen ovale or an ostium secundum atrial septal defect.253 If the interatrial communication is restrictive (too small), right atrial hypertension and systemic venous congestion with hepatomegaly will be present. In addition, signs of decreased cardiac output and hypoxemia are observed.253
As the infant with Type IB tricuspid atresia grows, the ventricular septal defect often becomes relatively smaller. In addition, the pulmonary infundibular stenosis may increase. For these reasons, pulmonary blood flow is typically further reduced, and the infant becomes progressively more cyanotic. Regular medical examinations are necessary to detect changes in the clinical presentation, and avoid the development of severe hypoxemia.
If the tricuspid atresia is present with no VSD and normally related great vessels (Type IA), there is no shunt from left to right at the ventricular level, so no intracardiac path for pulmonary blood flow, and the neonate usually demonstrates profound cyanosis from birth. This cyanosis worsens when the ductus arteriosus begins to close, because the ductus provides the only source of pulmonary blood flow. The right ventricle may be absent and the pulmonary valve atretic.253
Tricuspid Atresia with Transposition of the Great Arteries
If transposition of the great vessels is present, the pulmonary artery arises from the left ventricle and the aorta arises from the right ventricle. If no pulmonic stenosis is present (Type IIC), pulmonary blood flow will be increased once pulmonary vascular resistance falls at approximately 4 to 12 weeks of age. The excessive pulmonary blood flow under high pressure results in symptoms of congestive heart failure.253 The greater the volume of pulmonary blood flow, the greater the proportion of oxygenated blood returning to the left ventricle and the higher the systemic arterial oxygen saturation. As a result cyanosis usually decreases as pulmonary blood flow increases. However, the price of this increase in pulmonary blood flow is severe CHF, pulmonary hypertension, and the risk of pulmonary vascular disease. Increased pulmonary venous return contributes to the development of left ventricular volume overload and left ventricular dysfunction.
In tricuspid atresia with transposition of the great arteries, the right ventricle forms a subaortic chamber from which the aorta arises. Subaortic stenosis can result from a restrictive VSD, which minimizes shunting of blood from the left ventricle into the small right ventricle (the systemic outflow chamber),454 obstructing systemic blood flow. Subaortic stenosis can also result from a narrow infundibular area.253 Severe narrowing will decrease systemic circulation, which can produce shock, hypotension, or metabolic acidosis.253 When this combination of defects is present, systemic perfusion will deteriorate substantially as the ductus arteriosus begins to constrict after birth.
Coarctation of the aorta may be present with tricuspid atresia. The clinical findings of aortic coarctation will also be found, producing upper extremity hypertension with decreased blood pressure and pulses to the lower extremities, gastrointestinal tract, and kidneys.253 A bicuspid aortic valve may also be present.
The risk of bacterial endocarditis is significant in children with tricuspid atresia before and after palliative surgery. As a result, it is extremely important that antibiotic prophylaxis be administered during periods of increased risk of bacteremia preoperatively and after palliative surgery with prosthetic shunts and corrective surgery (see Bacterial Endocarditis later in this section of the chapter). Patients with tricuspid atresia and increased pulmonary blood flow who survive infancy are at risk for the development of pulmonary vascular disease.
Clinical Signs and Symptoms
All newborns with tricuspid atresia have systemic arterial desaturation resulting from the complete mixing of systemic and pulmonary venous return at the atrial level. By 1 week of age, most patients are noted to be cyanotic. The timing of onset and degree of cyanosis are determined by the source and volume of pulmonary blood flow.253 In an analysis of 225 patients presenting with tricuspid atresia, the most common presenting symptom was cyanosis (88%), followed by congestive heart failure (25%) and circulatory collapse/acidosis (6.2%).813
When normally related great vessels are present with a restrictive VSD and pulmonary stenosis, severe cyanosis will be present from birth. The cyanosis usually becomes profound when the ductus arteriosus begins to close.
The reported median arterial oxygen saturation in room air for newborns with prenatal diagnosis of tricuspid atresia was 79%, with a range of 40% to 91%.916 Polycythemia develops during the first months of life, and clubbing is observed beyond approximately 4 months of age. These infants also may develop hypercyanotic episodes (paroxysmal hypoxic spells) as a result of a decrease in the relative size of the VSD or an increase in the narrowing at the infundibulum.253 These episodes are similar to those seen in patients with tetralogy of Fallot, and may be precipitated by exertion, vigorous cry, feeding, or defecation. The spells result in profound cyanosis, irritability, and diaphoresis, and may result in loss of consciousness (see Box 8-34 for treatment). The development of these spells indicates the need for urgent therapeutic intervention.
When tricuspid atresia is present with a large VSD (with or without transposition) and no significant pulmonic stenosis cyanosis may be mild or may not be present at birth.253 However, signs of congestive heart failure and pulmonary overcirculation develop once pulmonary vascular resistance falls at approximately 4 to 12 weeks of age.253 Symptoms can include tachypnea, minimal cyanosis and hepatomegaly (see section, Common Clinical Conditions, Congestive Heart Failure).454
Murmurs are almost always present (described by Epstein253). If the pulmonary valve is patent a systolic pulmonary murmur may be heard at the second intercostal space along the left sternal border. The first heart sound is single because it is produced by mitral valve closure alone (no tricuspid valve closure). The second heart sound is usually also single. Flow through the VSD can produce a holosystolic murmur, and obstruction through the right ventricular outflow tract can produce an ejection murmur. A third heart sound and mid-diastolic rumble may be heard at the apex, caused by increased pulmonary blood flow. A restrictive VSD can produce a palpable thrill. The precordium is usually quiet. A left ventricular impulse is more likely to be appreciated than a right ventricular impulse.
Hepatomegaly may be present, particularly if the atrial septum is obstructing the right-to-left atrial shunt or CHF is present. The pulses are easily palpable in all extremities unless a coarctation is present and produces decreased lower extremity pulses.
ECG characteristics have been described by Epstein.253 Left axis deviation is present in 85% of patients with Type I tricuspid atresia. Right atrial enlargement often develops in older infants and children, but may not be seen at birth. When increased pulmonary blood flow is present, combined atrial enlargement may be noted. The precordial leads show reduced right ventricular forces. The P-R interval is normal in most patients. A sinus rhythm is typical, but older patients may have atrial tachycardias, including atrial flutter or fibrillation, particularly in the presence of a restrictive atrial septum or a dilated, hypertensive right atrium.
On chest radiograph, the heart size in tricuspid atresia is directly related to the volume of the pulmonary blood flow.454 The cardiac silhouette is often globular in appearance, with a normal heart size.253 The right heart border may be prominent when the right atrium is dilated.253 If pulmonary blood flow is increased (Types IC and IIC), cardiomegaly (caused by increased pulmonary venous return and left ventricular volume load) and increased pulmonary vascular markings develop once pulmonary vascular resistance falls and pulmonary blood flow increases.253 If pulmonary blood flow is obstructed, heart size will be normal and pulmonary vascular markings will be normal or decreased.253
The echocardiogram will confirm the presence of tricuspid atresia and is the diagnostic procedure of choice.253 The tricuspid valve will not be visible in the expected location; instead an imperforate linear echo density is present. Anatomic details, shunting, estimated pulmonary artery pressure, and ventricular function are identified by echocardiography. Associated defects, including coarctation of the aorta are identified.253 The degree of obstruction at the VSD, the size and location of the interatrial communication, and the size of the right ventricular outflow tract are quantified by the Doppler echocardiogram.454 Diagnosis of tricuspid atresia is well established by fetal echocardiography.916
Ongoing serial echocardiograms are used to monitor patients who undergo initial first-stage palliation, to monitor for the development of potential deterioration caused by problems such as pulmonary artery stenosis, ventricular dysfunction, evolution of atrial septal obstruction, or mitral valve regurgitation. If atrial fibrillation develops, the echocardiogram is performed to check for the development of atrial thrombi.253
Cardiac catheterization is often not required for diagnostic purposes because echocardiography provides detailed information, and advances in CT and MRI imaging yield clear and reliable images. Cardiac catheterization is used to measure the pulmonary vascular resistance, which is a critical factor for successful management, and must be determined before planning the next palliative surgery. Diagnostic studies may be needed to define the source(s) of pulmonary arterial flow, the impact of VSD obstruction, or evaluate other associated lesions, such as truncus arteriosus. Cardiac catheterization will also be required for interventional procedures, such as in selected cases requiring balloon atrial septostomy relief of a restrictive atrial septum, dilation of narrowed pulmonary arteries, or closure of collateral vessels.388
Angiocardiograms can evaluate the presence, location, and severity of any pulmonary stenosis or distortion. Initial surgical palliation with a systemic to pulmonary artery shunt or placement of a pulmonary artery band can lead to distortion of the pulmonary arteries. Angiography can also define any associated cardiac lesions. The precise size and location of collateral arteries (from the descending aorta to the lungs) or collateral venous vessels (creating a right-to-left shunt) are demonstrated by angiography.
Management
Medical Management
Greater than half of all newborns with tricuspid atresia demonstrate cyanosis in the first days of life. The cyanosis often progresses when the ductus arteriosus closes, the VSD becomes relatively smaller or closes, or there is progression in severity of the pulmonary stenosis. When significant cyanosis is present at birth, a substantial portion of pulmonary blood flow is probably dependent on the ductus arteriosus. If cyanosis is severe at birth, prostaglandin E1 (0.05 to 0.1 mcg/kg per minute initial infusion, titrated to 0.01 to 0.05 mcg/kg per minute infusion as tolerated) is administered to maintain ductal patency during the diagnostic studies and possibly until surgery can be performed (see section, Common Clinical Conditions, Hypoxemia, and Box 8-33).
Patients with tricuspid atresia typically undergo a series of procedures that eventually result in diversion of systemic venous return directly into the pulmonary arterial circulation. This eliminates the right-to-left atrial level shunt, and results in normal systemic arterial oxygenation and decompression of the left ventricle.253
Initial interventions are therapies to promote a balanced circulation, with sufficient pulmonary blood flow and adequate systemic oxygen delivery, while promoting growth and minimizing congestive heart failure and risk of pulmonary vascular disease. The goals of intervention for the newborn include: (1) establishing/maintaining a sufficient source of pulmonary blood flow to avoid extreme hypoxemia; (2) preventing excessive pulmonary blood flow, pulmonary hypertension, and the risk of pulmonary vascular disease; (3) maintaining a stable pulmonary artery anatomy for future surgical procedures.253 A balanced systemic and pulmonary circulation is indicated by signs of adequate systemic perfusion, normal pH and systemic arterial oxygen saturation near 80%.935 Activities to maintain effective pulmonary and systemic flow are listed in Table 8-35 (located earlier in the chapter).
If the echocardiogram identifies potential obstruction at the interatrial septum, a cardiac catheterization for a Rashkind balloon atrial septostomy (use of a balloon to tear a hole in the atrial septum) is performed. This septostomy usually is not required, however, because the foramen ovale typically is dilated.
If congestive heart failure is caused by increased pulmonary blood flow under high pressure (as occurs when a large ventricular septal defect is present without pulmonary stenosis), judicious medical management with diuretics and digoxin is indicated (see section, Common Clinical Conditions, Congestive Heart Failure). It is important to avoid aggressive diuresis, because hemoconcentration increases the risk of thromboembolic events, including cerebrovascular accident. The infant should be kept well hydrated to prevent hemoconcentration, although aggressive fluid administration should be avoided because it may precipitate or worsen congestive heart failure. Pulmonary artery banding may be performed if congestive heart failure is refractory to medical management.
Hypercyanotic spells occur in approximately 16% to 45% of infants with tricuspid atresia. If hypercyanotic episodes are observed, place the infant in the knee-chest position and administer 100% oxygen by nonrebreather face mask. Morphine sulfate (0.1 mg/kg IV or IM) provides sedation. The deeply cyanotic patient may require IV fluids and a vasopressor drug to increase the systemic vascular resistance (e.g., phenylephrine).935 This medication should be kept at the bedside of any infant known to have a history of such spells (for further information, refer to Box 8-34). Anything producing a fall in systemic vascular resistance should be avoided, because it will contribute to further reduction in pulmonary blood flow (i.e., blood is diverted to the systemic circulation) and worsening cyanosis.935 No air can be allowed to enter any IV system (may cause systemic/cerebral embolus).
Children with single ventricle physiology often are delayed in growth and may demonstrate difficulty feeding. Promoting adequate nutrition and growth is a critical component of care. Typically a concentrated formula (providing at least 24 calories per ounce) is provided with a goal of delivering more than 120 cal/kg per day. In the patient with single ventricle, when the arterial oxygen saturation is elevated (indicating excessive pulmonary blood flow that is likely associated with decreased systemic arterial flow) and during any low cardiac output state, mesenteric hypoperfusion is likely to be present. It is important to monitor for signs of necrotizing enterocolitis in the newborn including gastric distension, vomiting, and heme-positive stools. When enteral feedings are provided, monitor for increased volume of gastric residuals (see section, Common Clinical Conditions, Altered Nutrition).
Palliative Surgery
All surgical approaches are staged and palliative for tricuspid atresia; no biventricular repair is available. All staged procedures target a Fontan-type final procedure, so ultimately all systemic venous blood is directed into the pulmonary circulation. Palliative procedures are designed to divert some systemic venous (i.e., superior vena caval) blood to the pulmonary circulation and provide sufficient pulmonary blood flow to ensure adequate oxygenation while protecting the pulmonary vascular bed from developing elevated resistance. A low pulmonary vascular resistance is critical for a successful Fontan-type procedure.
The newborn with tricuspid atresia typically requires surgical intervention. If subpulmonic stenosis is present, a prostaglandin E1 infusion is provided and surgical or interventional catheterization is needed to increase pulmonary blood flow. A subclavian-to-pulmonary artery shunt, such as a Blalock-Taussig or modified prosthetic shunt (see Fig. 8-44; and Evolve Fig. 8-5 and Evolve Table 8-2 in the Chapter 8 Supplement on the Evolve Website) is often performed to provide adequate pulmonary blood flow under controlled pressure and stimulate growth of the pulmonary arteries. When the shunt is created, the prostaglandin E1 infusion will be discontinued. Some forms of palliative surgery are performed without cardiopulmonary bypass, while others require bypass. If the main pulmonary artery is small, a central prosthetic graft may be inserted between the aorta and the main pulmonary artery. These shunt procedures and perioperative care are discussed in Tetralogy of Fallot, Management, earlier in this chapter.
In selected cases, an alternative palliative procedure to increase pulmonary blood flow in the child with ductal-dependent circulation involves placement of a stent within the ductus arterious during cardiac catheterization.18,149 This avoids the need to create a surgical shunt in selected newborns. A balloon atrial septostomy is performed if a restrictive ASD is present.
In the presence of excessive pulmonary blood flow, pulmonary artery banding may be performed. Pulmonary artery banding can decrease the pulmonary blood flow and alleviate symptoms of congestive heart failure, but may not prevent development of pulmonary vascular disease. If the banding is too tight, the patient may develop severe hypoxemia, with systemic arterial oxygen saturation less than 75%, leading to metabolic acidosis. If the band is too loose, the symptoms of congestive heart failure will continue. Care after pulmonary artery banding is briefly reviewed in the Single Functioning Ventricle, Overview of Management section.
Alternatively, the main pulmonary artery may be detached from the heart and an aortopulmonary shunt can be created, to provide pulmonary blood flow through a path that has a controlled diameter to restrict the volume and the pressure of the pulmonary blood flow. A surgical atrial septectomy is also completed if the interatrial septum is restrictive or has the potential to be restrictive.
If D-transposition of the great arteries is present with obstruction to systemic flow through the VSD into the transposed aorta, surgery will be performed to provide unobstructed systemic flow. The Damus-Kaye-Stansel procedure may be performed: the pulmonary artery is transected before the bifurcation (i.e., the main trunk is separated from the branches). The end of the main pulmonary trunk is then sewn into the side of the ascending aorta. This procedure enables ventricular outflow to enter the original pulmonary artery trunk (bypassing the subaortic stenosis) and then flow into the aorta (see section, Single Functioning Ventricle, Overview of Management). An additional aortopulmonary shunt is created to provide pulmonary blood flow. A target arterial oxygen saturation for these patients with single ventricle hemodynamics is near 80%. Bleeding is a potential concern because of the extensive suture lines under systolic pressure591 (see section, Single Ventricle, Overview of Management). Following the surgery, flow into the aorta arises through both the pulmonary valve (from the left ventricle) and through the restrictive VSD and into the right ventricle and the aorta.253
Cavopulmonary Anastomosis
Either the bidirectional Glenn or the hemi-Fontan procedure is typically performed as the second stage surgery to improve effective pulmonary blood flow and arterial oxygen saturation and to decompress the left ventricle. These procedures and postoperative care are presented in detail in Single Functioning Ventricle, Overview of Management (see Box 8-39).
Completion Fontan-Type Procedure
The completion surgery for tricuspid atresia leads to total separation of the systemic and pulmonary venous blood by redirection of all systemic venous blood directly into the pulmonary arteries. The procedure is typically performed at 2 to 4 years of age.253,454 The bidirectional Glenn and Fontan procedures require low pulmonary artery pressure and vascular resistance.253 A cardiac catheterization is done before the surgery to determine the ideal procedure and the optimal time for surgical correction for each child.
This final stage Fontan-type procedure redirects inferior vena caval blood flow to the pulmonary arteries by means of an extracardiac conduit or with an intracardiac baffle.591 These procedures are illustrated and described in detail in Single Functioning Ventricle, Overview of Management, in previous pages. Pulmonary blood flow is provided without a ventricular pumping chamber.311 Cardiopulmonary bypass is required for most surgical Fontan-type procedures, but occasional cases have been reported of Fontan completion without use of bypass.123
The arterial oxygen saturation following the Fontan is typically in the high 80% to low 90% because the coronary sinus venous blood continues to drain into the right atrium, so it mixes with the pulmonary venous return, and then enters the systemic arterial circulation.591 This continued mixing of coronary sinus venous blood creates, in effect, a small right-to-left shunt. Arterial oxygen desaturation will also be present if a fenestrated Fontan is created to allow for right-to-left shunt flow and relief of potentially high pressure within the Fontan circuit. For details regarding postoperative care and complications, please see Single Functioning Ventricle, Overview of Management.
Several forms of cardiac catheterization based, nonsurgical Fontan completion have been successful and are under continued refinement.207,292,761
Variations of the Fontan procedure have been completed since 1971. The current mortality reported after a fenestrated Fontan procedure is 2.4%, and a bidirectional Glenn 1.4%.823 Interstage deaths caused by shunt thrombosis and unexplained deaths can also occur.813
Patients with tricuspid atresia who are not eligible for Fontan-type surgery may be eligible for cardiac transplantation. Factors that may prevent eligibility for the Fontan in tricuspid atresia include severe pulmonary artery distortion, pulmonary hypertension or elevated pulmonary vascular resistance, ventricular dysfunction, and progressive subaortic stenosis.
The survival of patients continues to improve with the wide use of Fontan-type interventions. Before use of the Fontan modifications, expected survival to young adulthood was about 50%.253 The outcomes vary widely with reported 10-year survival at 82% for tricuspid atresia patients with a Fontan procedure, whether diagnosis occurred during fetal life or after birth.813,916 The current goal is to achieve a Fontan circulation whenever possible.
Long-term complications are common and can include development of ventricular failure, atrial arrhythmias (particularly if the right atrium is included as part of the Fontan circuit), protein-losing enteropathy, Fontan pathway obstruction, increasing hypoxemia, collateral vessel malformations (venous and arterial), thromboembolism, recurrent pulmonary effusions, diminished exercise capacity, cirrhosis of the liver, and endocarditis.253,865 These potential complications and required care are detailed by Thorne,865 and are summarized in the Postoperative Care section of Single Functioning Ventricle, Overview of Management.
Advanced Concepts
Care of the patient with tricuspid atresia requires excellence in cardiopulmonary support. Continuous lifelong care by experts in congenital heart disease is required. Details regarding care after palliation are provided in the Overview of Single Functioning Ventricle, Overview of Management section and advanced concepts are listed in Box 8-41.
Box 8-41 Advanced Concepts: Tricuspid Atresia
• Approximately one third of patients with tricuspid atresia and transposition of the great arteries (Type II) have coarctation of the aorta.
• Patients with tricuspid atresia often have profound cyanosis at birth, and most require intervention during the neonatal period.
• In tricuspid atresia with a large VSD (with or without transposition) and no significant pulmonic stenosis, cyanosis may be mild or may not be present at birth. Signs of congestive heart failure and pulmonary overcirculation develop once pulmonary vascular resistance falls at approximately 4-12 weeks of age.
• The child with tricuspid atresia requires careful balance of systemic and pulmonary blood flow to maintain adequate systemic oxygenation and perfusion while minimizing risk of pulmonary vascular disease and ventricular dysfunction.
Pulmonary Atresia with Intact Ventricular Septum (IVS)
Pearls
• The newborn with pulmonary atresia and intact ventricular septum (IVS) is dependent on the presence of an atrial septal defect for right-to-left shunt, to allow exit of systemic venous blood flow from the right heart, and is also dependent on a patent ductus arteriosus to provide pulmonary blood flow.
• Right ventricular to coronary artery connections (sinusoids) develop with extreme RV hypertension; they develop most commonly in patients with small tricuspid valves and small, hypertrophied right ventricles. These sinusoids contribute to ischemia of both the right and left ventricles.
• Single ventricle physiology care is required.
• Staged palliations can lead to biventricular repair.
• After pulmonary valve perforation and dilation the newborn may not achieve sufficient arterial oxygen saturation for days or weeks and often requires continued observation and continuation of prostaglandin E1 therapy until that time.
Etiology
In pulmonary atresia there is complete obstruction to right ventricular outflow. When pulmonary atresia is present without a ventricular septal defect, this defect is called pulmonary atresia with intact ventricular septum (IVS). It results from failure of appropriate septation of the truncus arteriosus into both a pulmonary artery and aorta, with failure of pulmonary valve development. This form of the defect may also be called “hypoplastic right heart syndrome.”936
Right heart underdevelopment associated with pulmonary atresia can range from mild to severe, so the right ventricle size ranges from near normal to markedly hypertrophied with an underdeveloped chamber. The tricuspid valve, which is often dysplastic and stenotic, can be severely regurgitant with features of Ebstein anomaly.662 Typically the pulmonary valve is atretic, with fused valve leaflets,936 and there is a well-developed main pulmonary artery, with normally developed right and left pulmonary arteries. An atrial septal defect is present with either a patent foramen ovale or a secundum atrial septal defect; this allows the obligatory right-to-left atrial shunt (Fig. 8-61, A).662

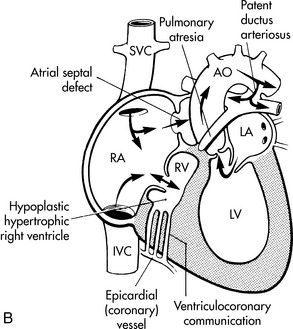
Fig. 8-61 Pulmonary atresia with intact ventricular septum. A, There is no outflow from the right ventricle, and the ventricle is small and extremely hypertensive and hypertrophied. There must be a source of pulmonary blood flow, such as the patent ductus arteriosus (PDA) depicted here. B, Right ventricular sinusoids may develop between the coronary arteries and the hypertensive right ventricular cavity. Desaturated systemic venous blood flows from the right ventricle through the sinusoids into the coronary arteries (particularly the left anterior descending branch). This results in perfusion of the myocardium with desaturated blood.
(From Striepe V. Pulmonary atresia with intact ventricular septum. In Kambam J, editor. Cardiac anesthesia for infants and children. St Louis, 1994, Mosby.)
Coronary artery anomalies are common, including the development of coronary sinusoids. These sinusoids are fistulae between the right ventricular cavity and the coronary arteries, producing retrograde flow of systemic venous blood from the right ventricular chamber through the coronary circulation.
Fetal studies suggest pulmonary atresia with intact ventricular septum is related to acquired progressive, altered hemodynamics in utero leading to lack of right heart ejection with subsequent fusion of the pulmonary valve leaflets.454 It is a rare lesion with an estimated incidence of 0.083 per 1000 live births.264 The New England Regional Infant Cardiac Program data reports that this defect represents 3.1% of all congenital heart defects. Extracardiac anomalies are rare.454 All patients with pulmonary atresia and intact ventricular septum have extremely complex congenital heart disease requiring lifelong, continuous care by a cardiologist with expertise in management of adult patients with congenital heart disease.927
Pathophysiology
The hemodynamics of pulmonary atresia with intact ventricular septum are similar to those resulting from other forms of single ventricle (including tricuspid atresia), with pulmonary blood flow dependent on patency of the ductus arteriosus (see Overview of Single Ventricle earlier in this section).936 When there is lack of anatomic continuity between the right ventricle and pulmonary artery, blood must enter the pulmonary circulation through another shunt or the newborn will develop profound and progressive hypoxemia when the ductus arteriosus begins to close; without intervention, this hypoxemia will lead to death.
Because there is no outflow from the right ventricle, an obligatory right-to-left shunt at the atrial level leads to complete mixing of the systemic and pulmonary venous return in the left heart. The right atrium is always dilated, and an interatrial communication is always present; this communication usually occurs through a patent foramen ovale or secundum atrial septal defect. The interatrial communication is rarely restrictive in size.662The pulmonary valve is atretic with fused leaflets.450
Systemic venous blood enters the right heart and quickly fills the right ventricle, but has no outflow path. Right ventricular end-diastolic and right atrial pressures rise, and tricuspid insufficiency often results. The increase in right atrial pressure opens the foramen ovale, so that systemic venous blood flows from the right to the left atrium and mixes with pulmonary venous blood. The mixed venous blood enters the left ventricle and is ejected into the aorta. A patent ductus arteriosus or some other form of systemic-to-pulmonary artery shunt must be present to provide flow from the systemic circulation into the pulmonary arterial circulation. The systemic arterial oxygen saturation varies directly with the volume of pulmonary blood flow; the greater the volume of pulmonary blood flow, the higher will be the systemic arterial oxygen saturation.662
The lack of a right ventricular outflow tract results in constant and extraordinarily high right ventricular afterload, severe right ventricular hypertrophy and extreme right ventricular hypertension; the RV systolic pressure may rise to 200 mm Hg.450 The hypertrophy and hypertension are responsible for many of the pathophysiologic changes that are associated with pulmonary atresia and intact ventricular septum.
The size of the right ventricle varies widely from a hypertrophied, tiny, hypertensive cavity to a hugely dilated, thin-walled cavity with low pressure, and can even include a normal-sized ventricle.219 The tricuspid valve is rarely normal, the annulus size is variable, and the valve is often hypoplastic, dysplastic, stenotic, or regurgitant.662 In some cases the tricuspid valve has features of Ebstein anomaly (see Ebstein Anomaly).662 The tricuspid valve diameter is presented as a “Z score” (the number of standard deviations by which the patient's tricuspid valve measurement deviates from the population mean).219 The size of the right ventricular cavity correlates well with the size of the tricuspid valve.349 In most (90%) patients with pulmonary atresia and IVS, the RV cavity size is small; it is severely reduced in 54% of the cases.349 Typically, the cavity size is severely compromised by massive right ventricular hypertrophy. Endocardial fibroelastosis can be present.662
Coronary artery abnormalities are common, and are found in as many as 70% of patients with this defect.770 Anomalies include coronary artery atresia at the origin, obstruction or occlusion of the coronary arteries, and sinusoids/fistulae between the right ventricle and coronary arteries.454 Newborns can demonstrate evidence of coronary artery abnormalities at birth, including stenoses and interruptions that can produce myocardial ischemia.662
The most common coronary abnormalities are the fistulae between the right ventricle and coronary artery. When these fistulae are present, endothelium-lined channels within the RV muscle mass, known as sinusoids, connect the RV cavity to the coronary artery tree.620 These sinusoids allow systemic venous blood from the hypertensive right ventricle to flow retrograde into the left anterior descending artery and/or the right coronary artery (see Fig. 8-61, B). This retrograde flow of desaturated blood into the coronary circulation results in ischemia of both the right and left ventricles. The involved coronary arteries may be dilated and tortuous, stenotic, or interrupted, with thick walls and small lumens. The coronary artery sinusoids are most likely to develop in patients with the smallest, most hypertensive right ventricles, the lowest “Z score” for the tricuspid valve (i.e., the most negative “Z score” value, indicating that the size of the tricuspid valve is much smaller than present in the population).662 Patients with a normal or near-normal right ventricle are unlikely to develop coronary sinusoids.770
The coronary sinusoids are thought to develop during fetal life because there is no outflow from the right ventricle and extreme RV hypertension is present. After birth, a variety of factors compromise coronary perfusion pressure and flow (coronary perfusion pressure = aortic end-diastolic pressure − right atrial pressure). Tachycardia, use of prostaglandin E1, creation of systemic to pulmonary shunts (by surgery or via stent within the ductus arteriosus) and use of vasodilator therapy can all decrease aortic end-diastolic pressure, and right atrial pressure is consistently elevated. These factors can reduce coronary artery perfusion pressure, enhance blood flow through the coronary sinusoids and compromise myocardial perfusion, causing myocardial ischemia and possible infarction.662
Coronary stenosis or atresia can develop proximal to the coronary fistulae, making the myocardium dependent on retrograde perfusion with desaturated blood from the RV through the fistulae into the coronary arteries. This combination is referred to as RV dependent coronary circulation (RVDCC).338 In these cases the RV pressure will need to remain high for any type of coronary perfusion to occur, because the aortic end-diastolic pressure may not be sufficient to provide antegrade coronary blood flow through the stenosed or atretic coronary vessels.662
The sinusoids can be minor or extensive and their development correlates directly with the degree of RV hypoplasia and resulting RV hypertension.936 When RVDCC is present, decompression of the RV (by a pulmonary valvotomy or an RV transannular patch), or other therapeutic interventions, which reduce RV hypertension can lead to coronary hypoperfusion, with resulting myocardial ischemia, infarction, ventricular dysfunction, malignant ventricular arrhythmias, and even death.662,936 RVDCC is reviewed by Guleserian et al338 and is reported in 3% to 34% of patients with pulmonary atresia and IVS.
The main pulmonary artery trunk is typically somewhat smaller than normal.450 The normal size of the pulmonary artery with this defect is in sharp contrast to the diminutive size of the pulmonary arteries observed in patients with pulmonary atresia with a ventricular septal defect (see Tetralogy of Fallot).450 Occasionally, hypoplasia of either the right or the left pulmonary artery is present.
The pulmonary venous return occurs through pulmonary veins that are typically joined normally to the left atrium. The left ventricular has variable degrees of hypertrophy.662 The left ventricle receives both systemic and pulmonary venous return, so left ventricular dilation and dysfunction may develop. This dysfunction is exacerbated by the development of right ventricular-coronary artery sinusoids and consequent compromise in oxygen delivery to the myocardium.
Successful fetal percutaneous pulmonary balloon valvuloplasty intervention has been initiated to alter the development of pulmonary atresia with intact ventricular septum in utero to enable ultimate biventricular management.33,877,883 Echocardiographic criteria have been established to identify potential candidates for fetal pulmonary valve intervention with the goal of preventing progressive RV hypoplasia.739,762 The risk of pulmonary hypertension is low.
Clinical Signs and Symptoms
Significant cyanosis and hypoxemia are present at birth. Profound cyanosis usually is observed once the ductus arteriosus begins to close, and will progress rapidly if untreated, causing metabolic acidosis and hemodynamic collapse. The hypoxemia is not responsive to oxygen administration.662
Once the ductus is reopened with prostaglandin E1 therapy, the desired arterial oxygen saturation is near 80%, with a normal pH. An arterial oxygen saturation greater than 90% suggests pulmonary overcirculation and will likely be associated with systemic hypoperfusion.935
A pansystolic murmur caused by tricuspid insufficiency can be heard along the left sternal border.662 A patent ductus arteriosus murmur can be heard at the second or third intercostal space, particularly once prostaglandin E1 therapy has begun.662 There may be no murmur.450
The pulse pressure is wide with a low diastolic pressure caused by the runoff of aortic blood flow into the ductus arteriosus, then into the pulmonary vascular circulation.620 The aortic diastolic pressure in a patient with a patent ductus arteriosus or a systemic to pulmonary shunt may be very low, leading to decreased coronary artery perfusion.620 The pulses are often normal, unless a large patent ductus produces bounding pulses or low cardiac output produces diminished pulses.662
The liver may become enlarged if the atrial septum is severely restrictive or the tricuspid valve is severely regurgitant.662 Tachycardia, and other signs of congestive heart failure also may be noted, and a left ventricular heave may be palpated.
Classic ECG findings include sinus rhythm, with left ventricular hypertrophy or dominance, decreased right ventricular forces, and right atrial enlargement.662 ECG evidence of left ventricular hypertrophy is in contrast to the typical dominant right ventricular forces normally observed in the neonate.620 Malignant ventricular arrhythmias may be observed. ST-segment abnormalities (depression or elevation) are frequently seen, indicating subendocardial ischemia662 (depression when the QRS is positive) or infarction (indicated by ST-segment elevation when the QRS is positive).
The cardiothoracic ratio on the chest radiograph ranges from mildly to substantially enlarged.662 Massive cardiomegaly will be seen in pulmonary atresia with Ebstein anomaly,662 or with significant tricuspid regurgitation.454 Pulmonary vascular markings will be diminished. The upper left heart border will be concave because the normal main pulmonary artery shadow is absent.
The echocardiogram is the primary mode of diagnosis and demonstrates absence of the right ventricular outflow tract. The echocardiogram enables evaluation of RV cavity size and function, tricuspid valve annulus size (including Z score) and function, the size and location of the atrial defect, the volume of the atrial shunting, the patency of the ductus arteriosus, and pulmonary artery branch anatomy. A tricuspid valve Z score of less than or equal to 2.5 has been shown to predict significant coronary artery abnormalities and the presence of right ventricular-dependent coronary circulation.770
Cardiac catheterization is often required for complete evaluation and to allow potential interventional therapies. Cardiac catheterization will conclusively demonstrate the anatomy needed to establish a treatment plan, including the distribution of the coronary arteries; identification of any atresia, stenosis, or interruption in the coronary arteries; the identification of right ventricle to coronary artery fistulae; and the status of right ventricular-dependent coronary circulation.662,935 Cardiac catheterization documents the absence of anatomic continuity between the pulmonary artery and aorta, and it the source, magnitude, and distribution of pulmonary arterial blood flow. In addition, the size of the common pulmonary artery or right and left pulmonary artery branches is visualized during angiography in order to determine the appropriate surgical intervention.
As part of the treatment during cardiac catheterization, a balloon atrial septostomy may be performed to treat a restrictive atrial septum. In selected cases, the plan will include perforation of the pulmonary valve or insertion of a PDA stent (see Palliative Procedures in the Management section, below).
Management
Medical Management
Initial management for pulmonary atresia with intact atrial septum involves establishment of vascular access and immediate administration of PGE1 (initial infusion: 0.05-0.1 mcg/kg per minute, then titrated to 0.01-0.05 mcg/kg per minute) to maintain ductal patency (see Box 8-33). Metabolic acidosis will require treatment. The infant is at risk for the development of complications of hypoxemia and polycythemia. No air can be allowed to enter any IV system because it can enter the systemic arterial circulation, producing an air embolus.
Single ventricle guidelines for care are followed, with goals including an arterial oxygen saturation of 75% to 85%, and signs of adequate oxygen delivery and systemic perfusion (see Overview of Single Functioning Ventricle, Overview of Management earlier in this section). A high arterial oxygen saturation (90% or above) with signs of poor perfusion can indicate excessive pulmonary blood flow with subsequent systemic hypoperfusion; this development requires intervention to improve the systemic blood flow, including changes in mechanical ventilation support, reduction in inspired oxygen concentration or inhaled nitrogen (to reduce inspired oxygen concentration and alveolar oxygenation). Excessively high systemic vascular resistance or very low pulmonary vascular resistance can result in increased pulmonary blood flow and higher arterial oxygen saturation, but may result in inadequate systemic and coronary artery perfusion and decreased tissue oxygen delivery with acidosis, myocardial ischemia, and compromised splanchnic and renal flow.
Inotropic support may be necessary if signs of inadequate perfusion are present. CHF requires careful management of intravascular volume while avoiding hemoconcentration. Severe tachycardia (which shortens diastolic filling time and coronary artery perfusion time) or further lowering of the aortic diastolic pressure must be avoided in patients with right ventricular-dependent coronary circulation because either condition will exacerbate coronary ischemia.662 Antibiotic prophylaxis is required before selected invasive procedures (see Bacterial Endocarditis).
The anatomy of pulmonary atresia with intact ventricular septum can be complex with wide variability in tricuspid, right ventricular, and coronary artery anatomy. Each patient requires an individualized approach in selection of surgical or interventional cardiac catheterization therapy, or a combination of these (“hybrid” procedure). No single procedure is appropriate for all patients.
Palliative Procedures
The initial palliative procedure provides some form of pulmonary blood flow.454 Interventions include creation of a systemic to pulmonary shunt, or right ventricular (RV) decompression (with a valvotomy or transannular patch), or a combination of RV decompression and a shunt.620 The goal of any palliative treatment is to establish adequate systemic arterial oxygenation with balanced pulmonary and systemic blood flow. The long-term goal is to maximize RV growth to enable a biventricular repair whenever possible, with separation of the systemic and pulmonary venous circulations, and RV to pulmonary artery continuity.219
Ultimately the degree of RV development will determine the strategy for biventricular repair, intermediate repair, or univentricular repair.620 A univentricular palliation treatment path is used when most or all of the coronary circulation is dependent on the RV, or when extensive RV-coronary fistulae are present.662 Options for management of the patient with pulmonary atresia with intact ventricular septum are summarized in Table 8-38.
Table 8-38 Newborn Interventions for Pulmonary Atresia with Intact Ventricular Septum by Anatomic Considerations315,620,662,936
| Anatomy Type | Intervention |
| RV and tricuspid valve—adequate size, no sinusoids | Surgery: Neonatal complete surgical repair with RV outflow reconstruction936 |
| Biventricular repair | |
| Mild RV hypoplasia, without RVDCC; tricuspid valve Z score of: 0 to −2620 | Surgery: Transannular patch along the right ventricle outflow tract |
| Cardiac catheterization: Pulmonary valvotomy620 | |
| Goal: Biventricular repair | |
| Moderate RV hypoplasia, tricuspid valve Z score is: −2 to −3; the coronary artery perfusion is not dependent on the RV620 | Surgery: Pulmonary outflow tract procedure with a Blalock-Taussig shunt620 |
| Goal: Biventricular repair | |
| Severe RV hypoplasia, the tricuspid valve Z value is less than or equal to: −3620 Likely to have sinusoids620 | Surgery: Blalock-Taussig shunt |
| Cardiac catheterization: Stent in PDA. | |
| Univentricular repair | |
| RV dependent coronary artery circulation, sinusoids662 | Surgery: Blalock-Taussig shunt |
| Cardiac catheterization: Stent in PDA | |
| Univentricular repair662 | |
| Severe tricuspid regurgitation | Surgery: Blalock-Taussig shunt662 with potential creation of tricuspid atresia662 |
| Consider cardiac transplantation662 | |
| Univentricular repair |
PDA, Patent ductus arteriosus; RV, right ventricle; RVDCC, right ventricular-dependent coronary circulation.
Interventional Catheterization
Perforation of the pulmonary valve during cardiac catheterization allows some blood flow across the right ventricular outflow tract. Since the 1990s radiofrequency energy has been used in the cardiac catheterization lab to perforate the pulmonary valve and establish antegrade pulmonary blood flow, with anticipation that the flow will stimulate RV growth and allow acute RV decompression. Laser-guided techniques also can be used. After pulmonary valve perforation, balloon dilation is performed. Perforation can be accomplished in 80% of patients with pulmonary atresia and intact ventricular septum.662 Complications occur in nearly 15% of patients, however, and can include perforation and tamponade. Mortality is approximately 8%.389
Another intervention that can be performed during cardiac catheterization to improve pulmonary blood flow is placement of a stent within the ductus arteriosus to maintain ductal patency.783 Future potential coarctation (i.e., stenosis or narrowing) of the left pulmonary artery associated with the site of ductus arteriosus stent insertion requires ongoing assessment.18
The atrial right-to-left shunt often persists despite relief of the RV outflow obstruction because of the combined effects of tricuspid stenosis, annular hypoplasia, and a small, noncompliant RV. The prostaglandin E1 infusion may be continued for days to weeks to ensure sufficient pulmonary blood flow, arterial oxygen saturation, and oxygen delivery. Postprocedure care includes the guidelines for single ventricle care presented in the section, Single Functioning Ventricle, Overview of Management earlier in this section of the chapter, and Cardiac Catheterization in the last section of this chapter.
If hypoxemia persists, an additional source of pulmonary blood flow can be created with a surgical Blalock-Taussig shunt or a stent placed within the ductus during a cardiac catheterization. A source of additional pulmonary blood flow is required in 33% to 70% of patients.350,662 Throughout the hospitalization, IV tubing must be kept free of air, because any air entering the systemic venous circulation may ultimately be shunted into the systemic arterial circulation, and may produce a cerebral air embolus.
When successful, these interventional therapies avoid the use of cardiopulmonary bypass in the newborn, and can facilitate later biventricular repair or “one-and-a-half ventricle” repair.350 The long-term results of this approach continue to be analyzed.662
Surgical Palliation to Increase Pulmonary Outflow and Blood Flow
Improved pulmonary blood flow can be achieved by a surgical pulmonary valvotomy. A closed transventricular pulmonary valvotomy does not require use of cardiopulmonary bypass. A curved blade is inserted through a small stab wound in the right ventricular outflow tract; the stab wound is surrounded by purse-string sutures (to prevent bleeding). A similar procedure may be performed using inflow occlusion and a small incision in the pulmonary artery. The valvotomy improves pulmonary blood flow and decompresses the right ventricle. In addition, it enables right ventricular ejection of blood, and so may stimulate growth of the hypoplastic right ventricle.620 Postoperatively, management as for single ventricle physiology is needed (as detailed in the Single Functioning Ventricle, Overview of Management).
Open-heart procedures performed in the newborn include an open pulmonary valvotomy followed by insertion of a patch in the pulmonary outflow tract. The ductus arteriosus can be ligated if systemic arterial saturation is adequate.620 If pulmonary blood flow (and systemic arterial saturation) remains inadequate after the valvotomy, a systemic-to-pulmonary artery shunt is also created. Postoperative complications include progressive right ventricular dysfunction, low cardiac output, and malignant ventricular arrhythmias. Congestive heart failure also may be present, with signs of systemic venous congestion. After any palliative procedure, absolutely no air can be allowed to enter any intravenous system because systemic venous blood continues to be shunted into the systemic arterial circulation.
Patients who have RV outflow reconstruction with creation of a Blalock-Taussig shunt can present with signs of low cardiac output if previously undiagnosed RV coronary sinusoids produce myocardial ischemia. Such low cardiac output is particularly likely if the right ventricular outflow reconstruction produced decompression of the right ventricle in patients with right ventricular-dependent coronary circulation. Signs of myocardial ischemia include ventricular arrhythmias and ECG changes (ST segment depression or elevation and T wave changes) immediately after surgery. An echocardiogram will confirm areas of myocardial dyskinesis or akinesis.936
If pulmonary insufficiency and tricuspid insufficiency are both present postoperatively, an ineffective “circular shunt” can develop. This shunt can develop after pulmonary valvotomy or RV outflow reconstruction that produces pulmonary insufficiency and creation of a systemic to pulmonary shunt. The systemic to pulmonary shunt delivers blood flow from the aorta to the pulmonary circulation; because pulmonary insufficiency is present, some of the shunted blood flows retrograde into the RV. If tricuspid valve regurgitation is also present, some of the blood from the RV is ejected back into the right atrium and then shunts right-to-left into the left atrium, ultimately flowing into the aorta and systemic circulation. This blood can then flow back through the systemic to pulmonary shunt, into the pulmonary artery, and back into the RV to the right atrium, left atrium, and aorta. The net result of this circular shunt is inadequate systemic blood flow. Signs of poor perfusion, including oliguria, metabolic acidosis, and systemic hypotension may develop several days after surgery.662 Therapies to decrease this postoperative physiology include increasing pulmonary vascular resistance and lowering systemic vascular resistance. The patient may require narrowing of the shunt or repair of the tricuspid valve.936
Surgical Correction
Following the initial procedure for pulmonary atresia with intact ventricular septum, the patient must be monitored to determine the timing and procedure appropriate for the second intervention. In children with mild RV hypoplasia who have successful RV outflow decompression (by balloon valvotomy, surgical valvotomy, or transannular patch), the RV hypertrophy can regress, RV compliance can improve, and the RV can grow, increasing the volume of pulmonary blood flow.
A lung perfusion scan may be performed to determine blood flow distribution and identify branch pulmonary artery narrowing. An echocardiogram and cardiac catheterization are typically performed when the infant is 3 to 6 months of age to determine if the RV will be capable of supporting normal pulmonary blood flow. If the RV has grown sufficiently, the existing aorta to pulmonary shunt will be closed in the cardiac catheterization laboratory with devices454 or via surgery. Some patients may only need an additional pulmonary balloon valvuloplasty and device closure of the atrial septal defect as the final intervention to complete biventricular repair.
More commonly an additional surgical procedure is needed to relieve RV outflow obstruction, including infundibular resection, pulmonary valvotomy, or placement of a transannular patch. The atrial septal defect may be closed, or the surgeon may choose to leave an atrial septal defect to allow a postoperative right-to-left shunt in case right ventricular function is borderline. The atrial defect can also be closed by cardiac catheterization device at a time in the future, when RV compliance and function improve.620
“One-and-a-half Ventricle” Repair
If RV development continues to be borderline, and the tricuspid valve is small, a “one-and-a-half ventricle” repair can be completed.620 This consists of a bidirectional Glenn (SVC to right pulmonary artery shunt) anastamosis936 to direct SVC flow directly into the pulmonary circulation. The IVC flow continues to enter the right atrium, and flows through the tricuspid valve, and is ejected by the right ventricle into the pulmonary circulation. A right ventricular outflow enlargement is performed to relieve residual pulmonary stenosis. When the atrial septal defect is closed, total separation of the systemic and pulmonary venous blood and separation of the systemic and pulmonary circulations is achieved.620 If atrial septal defect closure is not tolerated, a restrictive atrial opening may be left with plans for future closure via an interventional cardiac catheterization procedure. Immediately after surgery the atrial defect allows for right-to-left atrial shunting, and systemic arterial desaturation is present (see Single Functioning Ventricle, Overview of Management, Palliative Surgery for Univentricular Heart: The Bidirectional Glenn and Hemi-Fontan Procedure [Cavopulmonary Anastomosis] in this section, and the section, Postoperative Care and Anticoagulation).
Patients treated with an RV outflow tract enlargement procedure combined with a systemic to pulmonary artery shunt for mild to moderate RV hypoplasia require ongoing echocardiographic assessment of RV and tricuspid valve growth. If adequate growth is observed, a cardiac catheterization is performed and the shunt is temporarily occluded to evaluate adequacy of systemic arterial saturation (a sign of adequate pulmonary blood flow and adequate RV output). A temporary balloon may also be inserted to occlude the atrial septal defect to assess the ability of the RV to maintain adequate cardiac output. If the arterial oxygen saturation is maintained during catheterization testing, the shunt may be permanently closed. The catheterization may show need for additional surgery to enlarge the RV outflow tract as the next procedure.620
Biventricular Repair
When the right ventricle and both pulmonary arteries are of adequate size, total correction of the pulmonary atresia can be performed using a median sternotomy incision, cardiopulmonary bypass, and hypothermia in the newborn or after successful staged palliation during infancy. A transannular patch (with or without a cusp) or a conduit is inserted between the right ventricle and pulmonary artery; the use of antibiotic-treated cryopreserved valved pulmonary or aortic homografts currently is preferred over the use of prosthetic conduits. After biventricular repair, the atrial septal defect may be closed, or an atrial defect may be allowed to remain open temporarily to decompress the right heart in the immediate postoperative period; this will produce varying degrees of right-to-left shunting of blood and varying degrees of systemic arterial oxygen desaturation and cyanosis. Later, when flow patterns are established, the ASD may be closed during cardiac catheterization or additional surgery.
Postoperative complications after biventricular repair or “one-and-a-half ventricle” repair include low cardiac output, congestive heart failure, arrhythmias, bleeding, and neurologic complications. Severe congestive heart failure and right ventricular dysfunction (with signs of severe systemic venous congestion, including tachycardia, hepatomegaly, ascites, and pleural effusions) may develop after right ventricular outflow reconstruction. As noted, a small atrial defect may be left in place after any of these procedures, to allow for a right-to-left shunt when residual RV dysfunction is present. It is important to know if a residual ASD is present, because this will cause systemic arterial oxygen desaturation. In addition, if a residual ASD and right-to-left shunt is present, no air can be allowed to enter any intravenous catheter, because it may be shunted into the systemic arterial circulation and may produce a cerebral air embolus.
Ventricular dysfunction and signs of myocardial ischemia, ventricular arrhythmias, or ventricular infarction can develop in patients with RV sinusoids. It is important to monitor for ST-segment changes postoperatively.
Univentricular Repair
If the tricuspid valve and RV continue to be severely hypoplastic, they will be too small to support adequate pulmonary blood flow, and biventricular repair will not be possible. A staged univentricular pathway is planned and a bidirectional cavopulmonary anastomosis is performed with closure of the systemic to pulmonary artery shunt as the second stage. Postoperative evaluation will monitor RV and tricuspid valve status to determine future suitability for RV outflow tract enlargement with a “one-and-a-half- ventricle” repair, or completion of a Fontan procedure (see Single Functioning Ventricle, Overview of Management, Corrective Surgery: The Completion Fontan Procedure).
If the patient has an initial systemic to pulmonary artery palliation (shunt or stenting of the ductus arteriosus) and a univentricular heart pathway is anticipated, cardiac catheterization is performed at 3 to 6 months of age in preparation for the second-stage bidirectional Glenn with takedown of the systemic to pulmonary artery shunt. A total cavopulmonary connection (Fontan procedure) will then be completed.
For children with RV-dependent coronary circulation the Fontan pathway is planned to maintain elevated RV pressure, directing oxygenated (pulmonary venous) blood into the RV then into the coronary arteries via retrograde flow. This may include a lateral tunnel or extracardiac Fontan. The surgical procedure is planned to avoid even temporary RV decompression during cardiopulmonary bypass, because RV decompression can lead to lethal myocardial ischemia.620 The Fontan procedure (with or without a fenestration) joins the inferior vena caval flow to the pulmonary artery, so that systemic venous blood is diverted into the pulmonary arteries; oxygenated pulmonary venous blood must be able to flow through the tricuspid valve and into the RV and then into the coronary circulation (see Single Functioning Ventricle, Overview of Management, Corrective Surgery: The Completion Fontan Procedure).
Perioperative mortality varies widely and by specific procedure. The Society of Thoracic Surgeons, reporting 2006-2010 data, identify 7.1% (modified shunt) to 11.9% (central shunt) overall hospital mortality in neonates undergoing systemic to pulmonary artery shunts.823 Biventricular repair management strategies and outcomes have been reported, but broad application is limited due to the variability in intracardiac and great vessel anatomy.183 The Congenital Heart Surgeon Society database for 408 neonates with pulmonary atresia and intact ventricular septum reports 77% survival at 1 month, 60% survival at 5 years, and 58% survival at 15 years of age. A biventricular repair was reported in 33%, a Fontan in 20%, and a “one-and-a-half ventricle” repair in 5%. Outcomes continue to improve.34
The survivors of pulmonary atresia with intact ventricular septum are now beginning to reach adulthood and they will require lifelong care. Long-term results are affected by the patient's unique hemodynamics and the subsequent interventions. Late survivors can develop arrhythmias, residual right ventricular outflow obstruction, right atrial dilation, tricuspid regurgitation, and pulmonary regurgitation. Some may continue to have cyanosis from residual atrial septal defects or other shunts. The specific potential problems are varied because of the unique forms of palliation performed, possible progression of coronary artery abnormalities, and—for those with the univentricular interventions—all of the potential complications related to the Fontan procedure. These late complications, assessments, and late management options are detailed by Daubeney.219 Lifelong, continuous care is required into adulthood by experts in the management of adults with congenital heart disease.927
Some advanced concepts in the care of the child with pulmonary atresia and intact ventricular septum are listed in Box 8-42.
Box 8-42 Advanced Concepts: Pulmonary Atresia with Intact Ventricular Septum (IVS)
• A “circular shunt” can develop after creation of a systemic to pulmonary artery shunt if severe pulmonary and tricuspid insufficiency develop.
 Pulmonary insufficiency can result from RV outflow tract reconstruction with a transannular patch or from a percutaneous pulmonary valvotomy. Tricuspid insufficiency can result from right ventricular dysfunction and dilation.
Pulmonary insufficiency can result from RV outflow tract reconstruction with a transannular patch or from a percutaneous pulmonary valvotomy. Tricuspid insufficiency can result from right ventricular dysfunction and dilation. In the presence of both pulmonary and tricuspid regurgitation, blood from the aorta flows through the systemic to pulmonary artery shunt into the pulmonary artery, then flows retrograde (through the insufficient pulmonary valve) into the right ventricle. The blood can then flow retrograde (through the insufficient tricuspid valve) into the right atrium. Blood from the right atrium can flow through the atrial septal defect to the left atrium and into the ventricle and can be ejected into the aorta, to again flow through the systemic to pulmonary shunt.
In the presence of both pulmonary and tricuspid regurgitation, blood from the aorta flows through the systemic to pulmonary artery shunt into the pulmonary artery, then flows retrograde (through the insufficient pulmonary valve) into the right ventricle. The blood can then flow retrograde (through the insufficient tricuspid valve) into the right atrium. Blood from the right atrium can flow through the atrial septal defect to the left atrium and into the ventricle and can be ejected into the aorta, to again flow through the systemic to pulmonary shunt.• Decreased aortic diastolic pressure, tachycardia, and hypovolemia in the presence of RV sinusoids or RV-dependent coronary circulation will reduce myocardial oxygen delivery and produce myocardial ischemia. Such ischemia can produce ventricular arrhythmias and low cardiac output.
 Reduced aortic diastolic pressure decreases coronary perfusion pressure in general. In patients with RV sinusoids it will also increase retrograde flow of systemic venous blood from the right ventricle through the sinusoids.
Reduced aortic diastolic pressure decreases coronary perfusion pressure in general. In patients with RV sinusoids it will also increase retrograde flow of systemic venous blood from the right ventricle through the sinusoids. Hypovolemia reduces right ventricular end-diastolic pressure and decreases retrograde perfusion through the sinusoids. Patients with RV-dependent coronary circulation have limited antegrade flow from the coronary artery, so anything that reduces RV pressure will reduce the major source of blood flow to the coronary circulation. Decreased aortic diastolic pressure will also reduce what little antegrade flow is present through the coronary circulation
Hypovolemia reduces right ventricular end-diastolic pressure and decreases retrograde perfusion through the sinusoids. Patients with RV-dependent coronary circulation have limited antegrade flow from the coronary artery, so anything that reduces RV pressure will reduce the major source of blood flow to the coronary circulation. Decreased aortic diastolic pressure will also reduce what little antegrade flow is present through the coronary circulation• Signs of myocardial ischemia include ST-segment depression (in leads with upright QRS complex) and T-wave abnormalities. Myocardial infarction can produce ST-segment elevation (in leads with upright QRS complex). These changes require immediate recognition and therapy.
Hypoplastic Left Heart Syndrome (HLHS)
Pearls
• As the PDA closes in the newborn with HLHS, inadequate systemic perfusion will result in shock.
• The optimal circulation in patients with single ventricle physiology produces an arterial oxygen saturation of 75% to 85% with signs of adequate systemic perfusion.
• In the patient with single ventricle physiology:
 An arterial oxygen saturation greater than 85% with signs of decreased perfusion suggests pulmonary overcirculation (excessive pulmonary blood flow) that can lead to systemic hypoperfusion and inadequate systemic oxygen delivery.
An arterial oxygen saturation greater than 85% with signs of decreased perfusion suggests pulmonary overcirculation (excessive pulmonary blood flow) that can lead to systemic hypoperfusion and inadequate systemic oxygen delivery. An arterial oxygen saturation less than 70% indicates inadequate pulmonary blood flow or low cardiac output that can result in tissue hypoxia and lactic (metabolic) acidosis.
An arterial oxygen saturation less than 70% indicates inadequate pulmonary blood flow or low cardiac output that can result in tissue hypoxia and lactic (metabolic) acidosis.• A hematocrit greater than 40% is necessary to optimize oxygen carrying capacity when single ventricle physiology is present.
• It is important to monitor upper versus lower extremity pulses and blood pressures after the Norwood procedure. A pressure differential of greater than 10 mm Hg with elevation in upper extremity blood pressure may indicate recoarctation of the aorta after the Norwood procedure.
Etiology
Hypoplastic left heart syndrome (HLHS) consists of a small, often tiny left ventricle, aortic and/or mitral valve stenosis or atresia, normally related great vessels, and an intact ventricular septum (Fig. 8-62). The ascending aorta supplies the coronary circulation with retrograde flow (from the patent ductus into the aorta), but the aorta is hypoplastic; approximately half of patients with HLHS have an aortic diameter less than 2 mm.879 The left atrium is small because it receives limited blood flow during fetal development and the atrial septum is thick, with a foramen ovale that can be small or closed.514 Eighty percent of patients have localized coarctation of the aorta.358 The most serious form of the defect includes aortic atresia. The fetus with severe aortic valve disease has decreased or reversed blood flow through the patent foramen ovale in utero; the decreased flow results in less stimulus for development of left heart structures.561,881
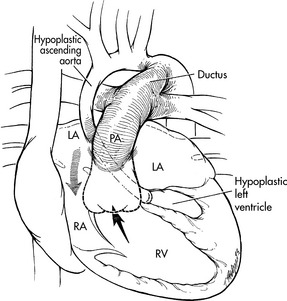
Fig. 8-62 Hypoplastic left heart syndrome: native anatomy. The syndrome includes a hypoplastic left ventricle, aortic valve atresia, and a diminutive ascending aorta. Systemic blood flow is generated by the right ventricle. Systemic venous blood flows from the right ventricle into the pulmonary artery (PA), through a patent ductus arteriosus (ductus) into the aorta. Pulmonary venous return enters the left atrium and flows through the foramen ovale or an atrial septal defect to the right ventricle. The amount of right ventricular output that flows into the pulmonary versus systemic circulations is determined by the relative resistances in the pulmonary and systemic circulations. LA, Left atrium; RA, right atrium; RV, right ventricle.
(From Nichols SC, Steven JM, Jobes AB. Hypoplastic left heart syndrome. In Nichols GD, et al, editors. Critical heart disease in infants and children, St Louis, 1995, Mosby.)
Hypoplastic left heart syndrome is responsible for between 1.4% and 3.8% of all congenital heart defects.881 Patients with HLHS can have extracardiac anomalies, including congenital diaphragmatic hernia, malrotation, and biliary atresia.881 A genetic basis for HLHS has been identified, with sibling recurrence risk at 8% and the presence of HLHS-associated cardiovascular malformation in one fifth of siblings.375 Before 1979 there was no intervention for HLHS, so mortality was 100%.514 Rapidly evolving treatment strategies for HLHS have dramatically improved survival in the last decade and include fetal echocardiographic diagnosis, prenatal transcatheter cardiac interventions, newborn staged surgical reconstruction, heart transplantation, and collaborative hybrid (cardiac catheter interventional and surgical) newborn palliation procedures. However, HLHS remains a leading cause of infant mortality and childhood morbidity among children with congenital heart disease.375
Pathophysiology
In HLHS systemic and pulmonary venous blood mix at the atrial level. Because there is a small left ventricular chamber with or without aortic or mitral valve obstruction, there is resistance to flow into the aorta and there is inadequate systemic perfusion. Survival after birth is dependent on the flow of blood from the right ventricle into the pulmonary artery and then through a patent ductus arteriosus to supply the systemic and coronary circulations. This flow from the ductus arteriosus supplies the descending aorta with antegrade flow; the ascending aorta (including the brain) and coronary arteries are perfused with retrograde flow. Thus, the right ventricle must supply the pulmonary, systemic, and coronary circulations. The left heart is too small to support the systemic and coronary circulations.
In HLHS, a parallel circulation exists, so the total ventricular output is divided between the pulmonary and systemic circulations and the volume of flow to each circulation is determined by the relative vascular resistance in each circuit.514,935 When the ductus arteriosus is widely patent and pulmonary vascular resistance is high at birth, there may be balanced flow to the systemic and pulmonary circulations with adequate arterial oxygen saturation and systemic perfusion.
An optimal balance of blood flow to the pulmonary and systemic circulations produces adequate systemic oxygen delivery without excessive volume load to the single, systemic right ventricle. This corresponds to a pulmonary to systemic flow ratio (Qp/Qs) of 1:1, an arterial oxygen saturation of near 80%, a pulmonary venous saturation about 95%, a mixed venous oxygen saturation about 65%, and cardiac output approximately normal.935 The systemic arterial oxygen saturation is almost directly related to the amount of pulmonary blood flow; the greater the amount of pulmonary blood flow, the higher is the amount of oxygenated pulmonary venous blood returning to the left atrium to mix with the (desaturated) systemic venous return, and the higher is the oxygen saturation ejected by the right ventricle into the pulmonary, coronary, and systemic circulations.514
Pulmonary vascular resistance decreases immediately after birth, leading to increased pulmonary blood flow.935 When the pulmonary vascular resistance is less than the systemic vascular resistance, blood flows preferentially into the pulmonary vascular bed, and the arterial oxygen saturation rises. The normal postnatal rise in systemic vascular resistance also enhances blood flow into the pulmonary vascular bed. Signs of congestive heart failure typically develop. Pulmonary congestion is likely to be significant because the increased volume of pulmonary venous return to the left atrium cannot flow freely into the left heart; obstruction to flow through the left heart increases pulmonary venous pressure with resultant pulmonary edema and work of breathing. The increased pulmonary blood flow may potentially cause systemic hypoperfusion because blood is diverted into the pulmonary circulation and away from the systemic circulation.
When the arterial oxygen saturation approaches 90%, pulmonary blood flow is very high and systemic flow is significantly compromised. If the oxygen tension in the alveoli is high, pulmonary vasodilation occurs, resulting in further increase in pulmonary blood flow. The single right ventricle has excessive volume load, so dysfunction will rapidly develop with dilation, elevated end-diastolic pressure, and progressive tricuspid regurgitation. Signs of heart failure include tachycardia, poor perfusion, and metabolic acidosis.935 The right ventricle exceeds maximal performance volume when the volume overload is four to five times normal, or the pulmonary to systemic flow ratio (Qp/QS) is 4:1.514
Progressive deterioration of systemic perfusion is associated with compromised coronary artery perfusion. Factors associated with progressive right ventricular dysfunction include excessive volume load (resulting from increased pulmonary blood flow and increased pulmonary venous return), increased right ventricular afterload (caused by the constricting ductus arteriosus and any pulmonary vasoconstriction that results from pulmonary edema and alveolar hypoxia), and poor coronary perfusion. As right ventricular dysfunction worsens, the ventricle will dilate and tricuspid regurgitation will develop. This leads to an increase in right and left atrial pressures and worsening of pulmonary venous hypertension and pulmonary edema.
An important determinant of symptoms from HLHS is the size of the interatrial communication. When severe mitral or aortic obstruction is present, blood will shunt from the left to the right atrium through a stretched foramen ovale. A nonrestrictive atrial septal defect allows an unrestricted left-to-right atrial shunt.514 If the interatrial communication is restrictive, the left atrial pressure will rise, increasing pulmonary venous pressure and ultimately increasing total pulmonary vascular resistance.514 This can eventually help to limit pulmonary blood flow.514 However, the left atrial hypertension will impair gas exchange.514 Pulmonary edema, respiratory distress, and progressive severe hypoxemia/cyanosis develop if a severely restrictive atrial septal defect or an intact atrial septum is present. Elevated pulmonary vascular resistance or a drop in systemic vascular resistance reduce pulmonary blood flow and increase cyanosis.
Clinical Signs and Symptoms
At birth the neonate may appear to be normal, but symptoms generally develop within 48 hours.2 There is no specific murmur associated with this defect.881 A third heart sound may be heard if ventricular dysfunction is present, and tricuspid regurgitation may produce a loud S1.881 If an unrestricted atrial septal defect is present, systemic perfusion and the systemic arterial oxygen saturation may initially be adequate while the ductus arteriosus is patent. As pulmonary vascular resistance begins to fall, pulmonary blood flow is increased, producing elevation in systemic arterial oxygen saturation and the development of signs of congestive heart failure, including tachycardia, tachypnea, and dyspnea. Because the infant is unable to breathe and suck at the same time, severe tachypnea is associated with poor feeding.
As the ductus arteriosus begins to spontaneously close, pulmonary blood flow increases and aortic blood flow and systemic perfusion decrease generally independent of the pulmonary vascular resistance.935 Cyanosis or pallor and signs of poor systemic perfusion develop rapidly, with signs including tachycardia, weak peripheral pulses, a narrowed pulse pressure, and peripheral vasoconstriction with possible lethargy.514 Continued ductal closure combined with the normal postnatal fall in pulmonary vascular resistance leads to progressive deterioration in systemic blood flow. Decreased coronary perfusion can lead to further myocardial dysfunction. The newborn may suddenly develop symptoms of circulatory collapse, including absence of palpable peripheral pulses, ashen color, gasping respirations, and severe cyanosis. Profound metabolic acidosis develops despite a high PaO2 (70 to 100 mm Hg).935
The development of pulmonary edema, respiratory distress, and progressive, severe cyanosis will be seen if a severely restrictive atrial septal defect or intact atrial septum is present.686 Respiratory support with elective intubation and assisted ventilation will be required. The patients with severely restrictive atrial septal defect or an intact septum are often unresponsive to conventional medical intervention and require urgent interventional catheterization.881
Newborns who are discharged home before the development of obvious symptoms may present at 2 to 3 days of age, with the parents reporting difficulty feeding or “funny breathing.” Gasping respirations with profound low cardiac output, acidosis, and renal failure may be seen on presentation to an emergency department.
Upper and lower extremity pulses are palpable and symmetric initially, but they become diminished in intensity once systemic perfusion is compromised. Right ventricular hypertrophy is present, and it may produce a sternal lift. The ECG reveals right ventricular hypertrophy, with diminished left ventricular forces, but this feature is typical of many normal newborns and therefore not specific for HLHS.514
The chest radiograph has few specific findings. Pulmonary edema with a normal heart size is seen when the atrial septal defect is severely restrictive; cardiomegaly with pulmonary edema may be present if HLHS is associated with a nonrestrictive atrial septal defect.881 Hepatomegaly is frequently observed, especially if presentation is delayed.881
Echocardiography confirms the presence of an extremely small left ventricle and any associated aortic or mitral atresia. Fetal echocardiographic diagnosis of HLHS is increasingly common.514 A cardiac catheterization is rarely required except in newborns with a severely restrictive atrial septal defect or intact atrial septum; catheterization allows interventional atrial septostomy for these newborns. These infants typically demonstrate severe pulmonary hypertension, pulmonary edema, low cardiac output, and cyanosis.
Management
Medical Management
Newborns with hypoplastic left heart syndrome can die at any time before institution of therapy. A prostaglandin E1 infusion must be provided immediately (initial infusion, 0.05 to 0.1 mcg/kg per minute) to keep the ductus arteriosus open until the diagnosis is confirmed (see Box 8-33). IV Prostaglandin E1 almost invariably reopens the ductus arteriosus, but may produce apnea, so providers should be prepared to perform intubation and provide mechanical ventilation.514
The PGE1 dose can be decreased to the lowest effective dose (0.01-0.05 mcg/kg per minute) once ductal patency is confirmed.881 Because all circulations are perfused by the right ventricle, until complete correction and separation of systemic and pulmonary venous blood is accomplished surgically, no air can be allowed to enter any IV system because it may be shunted into the cerebral arterial circulation, causing a cerebral air embolus (stroke). The patient is at risk for all complications associated with hypoxemia and polycythemia until that time (see section, Common Clinical Conditions, Hypoxemia).
Support of adequate systemic perfusion and oxygen delivery are critical throughout care in the patient with single ventricle circulation. The arterial blood gas is an excellent indicator of hemodynamic stability in these patients.935 Adequate peripheral perfusion is indicated by a normal pH and an arterial oxygen saturation near 80%; these findings are associated with a pulmonary to systemic flow ratio (Qp:Qs) near 1:1.935 Inadequate systemic perfusion and oxygen delivery, excessive pulmonary blood flow, and likely myocardial dysfunction are indicated by metabolic acidosis with arterial oxygen saturation in excess of 90%.935 Early detection and treatment of altered oxygen delivery is important to minimize end organ ischemia and improve survival. Note that arterial oxygen saturation may be within the acceptable range despite profound circulatory derangements, so continuous monitoring and careful assessment are needed.881
Development of signs of systemic hypoperfusion must be reported immediately to the healthcare provider responsible for managing the infant's care. Signs of systemic hypoperfusion include worsening tachycardia, diminished quality of peripheral pulses, decreased urine output, and pulmonary congestion. Serial serum lactate measurements help evaluate adequacy of oxygen delivery; a rising or persistently high lactate indicates acidosis that is not responding to therapy.
Near infrared spectroscopy (NIRS) sensors over the forehead and kidney area on the lower back have been used to identify real-time trends in oxygenation of the brain and somatic tissue, and to monitor response to interventions.315 After palliation for HLHS, cerebral saturations greater than 50% and somatic values greater than 60% have predicted better outcomes.590
After the reopening of the ductus, therapies to support right ventricular function may include inotropic support. If severe signs of low cardiac output develop, the right ventricle must be supported to recover from insults and stressors, including probable ischemia caused by poor coronary perfusion, volume overload, increased afterload, and acidosis.514 Hypoglycemia and hypocalcemia require correction.
Profoundly low cardiac output can lead to gastrointestinal dysfunction, hepatic dysfunction or failure, renal failure, fluid overload, and hyperkalemia; these complications require supportive therapies. Vasoactive therapy must be carefully titrated to prevent excessive vasoconstriction and further reduction in organ blood flow. Doses of all drugs should be evaluated and adjusted as needed if new renal or hepatic dysfunction develops.514
High doses of inotropic support are undesirable because they cause excessive vasoconstriction that will contribute to a further increase in pulmonary blood flow and will divert even more blood flow from the systemic to the pulmonary circulation. As a result, arterial oxygen saturation and mean arterial blood pressure require careful monitoring.935
Oxygen administration is necessary if the arterial oxygen saturation is less than 70%. The arterial blood gas must be closely monitored for development of acidosis or an oxygen tension below 30 mm Hg; these findings must be immediately reported to the on-call provider because they may be associated with inadequate oxygen delivery.
If the atrial septal defect is severely restrictive or the atrial septum is intact, obstruction to pulmonary blood venous return to the left atrium can contribute to pulmonary edema, and produce poor oxygenation and profound hypoxemia, with PaO2 of 20 mm Hg. When the atrial septal defect is restrictive, medical therapy to increase pulmonary blood flow, including oxygen administration and mechanical ventilation will not increase the PaO2.514 If the atrial septum is intact or the atrial septal defect is restrictive, urgent interventional cardiac catheterization or surgery is indicated to open the atrial septal defect and decompress the left atrium. This condition remains highly lethal despite prenatal diagnosis,323 but has shown improving outcomes with fetal atrial septostomy intervention.908
Cardiac catheterization intervention should improve systemic arterial oxygenation and relieve left atrial and pulmonary hypertension and pulmonary edema.329 Room air should be used to avoid the pulmonary vasodilation resulting from high alveolar oxygen tension that is produced by oxygen therapy. If high arterial oxygen saturation is associated with poor perfusion, metabolic acidosis, or other signs of inadequate tissue oxygen delivery, interventions are needed to increase pulmonary vascular resistance and reduce excessive pulmonary blood flow; this will improve systemic perfusion. Such measures include decreasing the inspired oxygen concentration, elective intubation, mechanical ventilation support, sedation, and possible neuromuscular blockade (see Chapter 5). Controlled hypoventilation is used to increase arterial carbon dioxide levels and create a mild respiratory acidosis. Alkalosis creates pulmonary vasodilation, and is therefore avoided. As a result, mild alveolar hypoxia and mild hypercarbia are maintained preoperatively and can improve systemic circulation.935
Sedation can be used to suppress the infant's intrinsic respiratory drive, because a rapid spontaneous respiratory rate will likely further decrease the PaCO2. Positive and-expiratory pressure (PEEP) can result in increased pulmonary resistance.881 Inspired gases can be manipulated to reduce excessive pulmonary blood flow and improve systemic hypoperfusion.935 Alveolar hypoxia can be created with addition of inhaled nitrogen to reduce the inspired oxygen concentration to 14% to 20%; such interventions will produce pulmonary vasoconstriction and thus limit pulmonary blood flow.935 Inhaled carbon dioxide (2% to 5%) can be used to promote pulmonary vasoconstriction.935
Afterload reduction can further decrease the systemic vascular resistance,881,935 but the diastolic blood pressure should not fall below 30 mm Hg to avoid compromising coronary blood flow.590 As pulmonary blood flow is controlled, the arterial oxygen saturation should decline to the acceptable range with signs of improved systemic perfusion. The right ventricular function will typically improve when the pulmonary to systemic flow ratio (QP/QS) falls from 4:1 to 2:1 or lower because the decrease in pulmonary venous return will reduce right ventricular volume overload.514
Inotropic support with milrinone can aid right ventricular contractility and reduce systemic vascular resistance.514 Hypertension and a rise in systemic vascular resistance are avoided because they will again increase pulmonary blood flow, with the potential return of poor systemic perfusion.
If at all possible the neonate should be allowed to continue to breathe spontaneously in room air. If mechanical ventilation is required, room air is often used to prevent excessive pulmonary vasodilation. Supplementary oxygen therapy may be required for severe hypoxia, lung pathology, or a restrictive atrial septal defect.881
Surgical Intervention
Since the late 1970s the Norwood procedure (Fig. 8-63, A) has become increasingly successful when performed as a first-stage palliation for hypoplastic left heart syndrome.315,659,881 Staged surgical intervention, completed by a Fontan-like procedure, ultimately results in separation of single ventricle circulations. Cardiac transplantation during the neonatal period may also be offered, particularly if right ventricular or tricuspid valve dysfunction remains severe despite medical management.
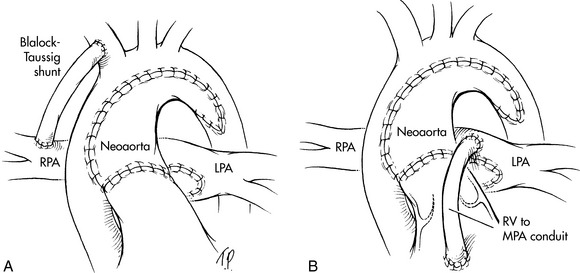
Fig. 8-63 Stage I Norwood procedure for hypoplastic left heart syndrome. A, Stage I Norwood procedure with pulmonary blood flow provided by a modified right Blalock-Taussig shunt. The main pulmonary artery is transected right before it branches into the right pulmonary artery (RPA) and left pulmonary artery (LPA). The new aorta (neoaorta) is created by joining the main pulmonary artery trunk to the original aorta. Pulmonary blood flow is provided by a Blalock-Taussig (subclavian to pulmonary artery) prosthetic shunt. B, In the Sano modification, a 5-mm conduit is inserted between the right ventricle (RV) and the main pulmonary artery (MPA) to provide pulmonary blood flow.
(From Nichols GD. Critical heart disease in infants and children. St Louis, 1995, Mosby, Fig. 40-7, A and B.)
A modification of the Norwood procedure, the Sano operation (see Fig. 8-63, B), was developed in the 1990s. Most recently a staged “hybrid” intervention combining interventional cardiac catheterization and surgery without cardiopulmonary bypass has become available.293 (See Hybrid Procedures in Cardiac Catheterization and Interventions.)
It is important to note that some parents may decline surgical intervention for the newborn with HLHS. In selected cases when unrecoverable decompensation occurs at presentation or multiple congenital anomalies are present, compassionate care is offered to the family. In these cases, comfort measures only are provided, with no surgical intervention, and the parents are supported until the infant dies (see Chapter 3).
The goal of each first stage intervention for HLHS is to provide unobstructed systemic blood flow from the right ventricle into the aorta. A source of controlled pulmonary blood flow is needed with care taken to protect the pulmonary artery architecture from development of distortions, and the pulmonary vascular bed from development of hypertension and vascular disease. Obstruction to pulmonary venous return is eliminated by establishing an adequate interatrial communication to ensure an unrestricted flow from the left to the right atrium. The final staged procedure for HLHS uses a modification of the Fontan procedure to separate pulmonary and systemic blood flow within the heart.
The Norwood Procedure
This operation establishes permanent, unobstructed systemic blood flow from the right ventricle into a reconstructed neo-aorta. This procedure requires cardiopulmonary bypass, altered perfusion (circulatory arrest or regional perfusion), and deep hypothermia.881 Pulmonary blood flow is provided by a prosthetic shunt from an aortic branch to a pulmonary artery. To eliminate obstruction to pulmonary venous return, the interatrial septum is resected.
The distal main pulmonary artery is transected. The aorta is incised longitudinally from the left subclavian artery, across the arch to the ascending aorta. An anastomosis is created between the trunk of the proximal main pulmonary artery and the ascending aorta and aortic arch. A portion of pulmonary homograft can be used to complete aortic reconstruction and prevent development of stenosis. Pulmonary blood flow is created by insertion of a 3- to 4-mm Gore-Tex systemic-to-pulmonary artery shunt between the innominate artery (or the aorta) and the pulmonary artery. The existing atrial septal defect or patent foramen ovale is enlarged (see Fig. 8-63, A and B).
After the completion of the stage 1 procedure, parallel circulations exist. The right ventricle receives systemic and pulmonary venous blood and ejects that mixed venous blood into the new aorta that was fashioned from the aorta and original pulmonary artery trunk. Pulmonary blood flow occurs through the systemic-to-pulmonary artery shunt. Pulmonary venous return enters the left atrium and flows chiefly into the right atrium, and is mixed with systemic venous blood in the right ventricle.
Several postoperative complications can develop after the Norwood procedure. The same single ventricle circulation guidelines for care are to be followed as during the preoperative period, with targeted arterial oxygen saturation near 80%.
Optimizing systemic oxygen delivery remains crucial. Low cardiac output syndrome is common during the first 24 to 48 postoperative hours,935 and inotropic medications are required. There are many causes of low cardiac output syndrome after intervention for complex neonatal congenital heart disease using cardiopulmonary bypass. Causes of low cardiac output after the Norwood procedure can include residual anatomic lesions such as residual aortic arch obstruction.935 In addition, metabolic demands are increased postoperatively. It is important to note that neonates can develop shock despite the presence of an adequate arterial blood pressure, so close observation is needed to detect signs of poor perfusion (see Shock in Chapter 6).568
Inotropic medications are titrated carefully to support cardiac output and systemic perfusion while avoiding a significant increase in systemic vascular resistance that can lead to increased pulmonary blood flow with systemic hypoperfusion and the potential for decreased systemic oxygen delivery. Cardiovascular surgical centers have reported improved survival and neurologic outcome with the routine initiation of mechanical circulatory assistance in the operating room, at the conclusion of the Norwood procedure (see Chapter 7). This practice enables support of cardiovascular function to meet the increased postoperative oxygen requirements.887
Continuous close monitoring for adequacy of oxygen delivery versus consumption (demand) is needed and includes the typical monitoring for the complex postoperative patient with additional measurements. Oxygen delivery status can be continually or intermittently evaluated with the use of a catheter in the superior vena cava to measure central venous oxygen saturation (ScvO2). Goal directed therapy targeting an ScvO2 of 75% as an indicator of adequate systemic oxygen delivery versus demand has resulted in improved survival after HLHS palliation.590,880 Continuous ScvO2 monitoring has also decreased the incidence of sudden circulatory collapse in the postoperative period.882 An ScvO2 saturation below 45% may be associated with ischemic brain injury.590 The difference between the arterial and central venous oxygen saturation should also be assessed. A rising arterial venous oxygen saturation difference is a strong indicator of inadequate oxygen delivery, and a difference of 40% to 50% or more indicates severely low cardiac output.384,935 A rising serum lactate also indicates inadequate systemic oxygen delivery.
Near infrared spectroscopy (NIRS) sensors over the forehead and kidney area on the lower back have been used to identify real-time trends in oxygenation of the brain and somatic tissue. After palliation for HLHS, cerebral saturations greater than 50% and somatic values greater than 60% have predicted better outcomes.590 Trends can be continually monitored and response to therapies assessed with good correlation between ScvO2 saturations and NIRS saturations590 for additional details of postoperative care.
Postoperatively, excessive pulmonary blood flow is present if the arterial oxygen saturation is greater than 85% to 88%.935 Although both pulmonary and systemic vascular resistance should be evaluated and optimized, the primary determinant of the pulmonary to systemic flow ratio is the systemic vascular resistance. Successful treatment methods to improve systemic perfusion involve therapies to decrease systemic vascular resistance.
Afterload reduction enhances systemic blood flow and improves systemic oxygen delivery as more blood flows to the systemic circulation (and less to the pulmonary circulation). Milrinone is often used as both a vasodilator and positive inotropic agent (it is an inodilator).
Phenoxybenzamine has been used successfully for prolonged afterload reduction through alpha-adrenergic blockade both post cardiopulmonary bypass and in the postoperative period. Decreasing the systemic vascular resistance can lead to a more balanced single ventricle circulation with a more stable postoperative course and higher systemic oxygen delivery.382 However, use of phenoxybenzamine can lead to sustained, excessive vasodilation and systemic hypoperfusion, requiring the initiation of catecholamines, including norepinephrine to optimize the systemic vascular resistance.591 When inotropic support is required during the use of phenoxybenzamine with low mixed venous saturation and elevated systemic vascular resistance, epinephrine may be useful.591 The arterial diastolic pressure should be maintained at greater than 30 mm Hg590 to maintain adequate coronary perfusion pressure.
Elevated systemic resistance and arterial blood pressure can result from a residual aortic arch obstruction. Clinical exam and echocardiography can be used to evaluate the reconstruction and detect any obstruction.
Monitor for bleeding. Bleeding may occur at any of the extensive suture lines along the aorta, because these suture lines are exposed to systemic pressure. Post-bypass coagulopathy can also contribute to bleeding. Anemia must be avoided and the hematocrit maintained at least 40% to support adequate oxygen-carrying capacity.
Titrate preload (with volume therapy) to maintain right atrial and right ventricular end-diastolic pressure, and support optimal cardiac output. Typically an initial CVP of 12 to 14 mm Hg is maintained.881
Monitor for arrhythmias. Sinus tachycardia is the newborn's most efficient mechanism to raise cardiac output, and it is a nonspecific sign of distress. As a result, development of tachycardia should prompt careful assessment of the newborn's airway, oxygenation, ventilation, and perfusion. Malignant arrhythmias including bradycardia, loss of sinus rhythm, or supraventricular tachycardia are not well tolerated and can lead to severe low cardiac output.
Monitor for neurologic complications, including seizures. Closely monitor the neonate's serum ionized calcium and provide supplementation as needed. Absolutely no air can be allowed to enter any intravenous system because it may be shunted into the cerebral circulation, producing a cerebral air embolus (stroke).
Additional support. Elective delayed sternal closure may be used. Methods to minimize oxygen consumption include providing narcotics and sedation and maintenance of normothermia and a neutral thermal environment. For further information about management, see Single Functioning Ventricle, Overview of Management.
Monitor for signs of deterioration. A decrease in pulmonary blood flow may result from hypotension or low systemic vascular resistance. A narrowed shunt can produce increasing cyanosis and development of metabolic acidosis at any time postoperatively. The shunt murmur may change and the diastolic blood pressure may increase. If shunt occlusion is suspected, immediate therapy is indicated, including heparinization to minimize clot progression, elevation of the arterial blood pressure with epinephrine (to enhance shunt flow), and rapid echocardiogram. Elective heparinization to decrease the possibility of prosthetic shunt occlusion may begin after the risk of postoperative bleeding has passed, with eventual transition to anticoagulation with aspirin or low molecular weight heparin. Hypoxemia unresponsive to therapy requires initiation of cardiopulmonary support or ECMO.
The atrial septal defect may become obstructive, leading to pulmonary venous hypertension; this is particularly likely in those patients undergoing the hybrid stage 1 intervention. Signs may develop gradually and include increasing tachypnea, decreasing arterial oxygen saturation, cyanosis, oxygen dependence, poor feeding, and signs of pulmonary venous congestion on chest radiograph. The shunt murmur intensity may decrease.514 The diagnosis can be made by echocardiography and treatment can include interventional cardiac catheterization to dilate the atrial septum, with possible stent implantation.
Right ventricular dysfunction and tricuspid regurgitation can develop requiring additional inotropic support and initiation of afterload reduction to reduce systemic vascular resistance (ventricular afterload). If treatment does not improve ventricular function, the infant may require an early second-stage bidirectional Glenn procedure, with tricuspid valve plication. Some infants are referred directly for cardiac transplantation.
Nutrition is initially provided parenterally. As the risk for systemic hypoperfusion subsides, enteral feeding begins, increasing gradually to 110 to 130 kcal/kg per day using fortified formula or breast milk.881 Optimal nutrition is critical for growth after palliation, but may be complicated by the development of poor oral feeding, impaired circulation with mesenteric hypoperfusion, necrotizing enterocolitis, or presence of postoperative recurrent laryngeal nerve injury.591,935 Necrotizing enterocolitis increases the risk for death.412 Malnutrition is common,461 with formula intolerance and prolonged feedings associated with impaired somatic growth until after the second stage surgical procedure.645,825
Nasogastric or gastrostomy tube feedings may be required. Normal rate of weight gain in infants following Norwood Stage I surgery have been achieved using calorie enhanced formulas with home surveillance monitoring of nutrition and weight gains.891 Management of these potential challenges is reviewed in the section, Common Clinical Conditions, Altered Nutrition and Potential Gastrointestinal Complications.
Re-coarctations in the aorta are reported in 11% to 37%170 of patients, and reintervention is most commonly required in the first 6 months after the Norwood procedure.35 Clinical symptoms include decreased femoral pulses and hypertension in the upper extremities. A gradient of 10 mm Hg or more between the blood pressures in the upper and lower extremities (with the higher pressure in the upper extremities) requires assessment by echocardiography. Re-coarctation also leads to high arterial saturation because the obstruction in the aorta increases systemic vascular resistance, so it increases pulmonary blood flow.935 The increased pulmonary blood flow can lead to even further pressure and volume load for the right ventricle, resulting in poor ventricular function and signs of heart failure. Re-coarctation is treated successfully by balloon dilation during interventional cardiac catheterization and possible aortic stent implantation or re-operation.170
Late complications include progressive congestive heart failure, pulmonary artery distortion, pulmonary vein stenosis and sudden death. The systemic to pulmonary artery shunt (Blalock-Taussig or other) or RV to PA conduit provide the only source for pulmonary blood flow and can progressively narrow at any time, causing increased cyanosis and hypoxemia.
Approximately 5% to 16%502,812 of immediate survivors of stage 1 Norwood die in the interval between stage 1 and the stage 2 procedures. Multiple risk factors for interstage death include postoperative arrhythmias, decreased ventricular function at discharge,502,812 and residual or recurrent lesions.
The limited cardiac reserve and chronic hypoxemia and cyanosis in patients with palliated HLHS reduce their tolerance of dehydration, fever, increased metabolic demands, serious respiratory illness, or advancing heart failure. Use of home monitoring protocols have improved interstage survival for surgical316-318,502 and hybrid stage I293 palliated HLHS by providing extensive parental education and combinations of home oxygen saturation monitoring, daily weights with digital scales, surveillance for intercurrent illness, and monitoring of oral intake.318,881,891 National, multicenter quality improvement collaboratives are underway by the Joint Council on Congenital Heart Disease to reduce clinical practice variations and improve long-term outcomes for children with HLHS.502
The Sano Procedure
This first stage intervention for HLHS is identical to the Norwood procedure except that the source for pulmonary blood flow is a 5- to 6-mm graft sewn between the right ventricle and the main pulmonary artery.881 Insertion of this graft requires a small right ventriculotomy (Fig. 8-63, B).
The described potential advantage of the Sano procedure includes the fact that the right ventricle to pulmonary artery connection avoids the low diastolic pressure that results from the aortic runoff with a systemic to pulmonary artery shunt.514 The aortic diastolic blood pressure has been found to be higher after the Sano procedure, but there is more variability in pulmonary blood flow.208,316
Potential postoperative complications include the development of conduit stenosis that produces cyanosis and hypoxemia and requires intervention.496 Oxygen therapy should be used for these infants with cyanosis because their shunt to the pulmonary system only occurs during systole, rather than during systole and diastole as following a systemic to pulmonary artery shunt.
Reports have not documented any difference between the Norwood and the Sano procedure when freedom from cardiac transplantation after 12 months670 and survival are evaluated.53,54,316,317 It may take years to determine the effects of the ventriculotomy and right ventricular to pulmonary artery shunt on right ventricular function and the pulmonary vascular bed.53
Long-Term Survival Following Palliation
Survival for infants undergoing the initial palliation for HLHS has dramatically improved in the last decade, with survival to hospital discharge as high as 93%209,880 to 95%.315 A 39% 15-year survival rate was reported for patients undergoing a Norwood procedure from 1984 to 1999.556 Mortality remains high for the Norwood procedure, with the Society of Thoracic Surgeons data centers reporting 18.2% mortality prior to hospital discharge (compared to the 3.2% overall mortality in the Society for Thoracic Surgeons congenital heart disease surgical registry).823
The long-term results of this procedure are unknown. There is concern that the right ventricle may not function indefinitely as a systemic ventricle, and that progressive right ventricular dysfunction will develop in a significant number of patients. Life-long continuous followup by experts in congenital heart disease care is mandatory.
Neurodevelopmental disabilities have been reported,326,554,798,836 include delays in visual-motor integration, motor development, executive function, and behavioral abnormalities. All patients with hypoplastic left heart syndrome require close, ongoing developmental screening for potential complications requiring therapeutic intervention, with a focus on maximizing the neurocognitive outcomes.115 The risks for developmental delay are summarized by Tweddell et al.881
Exercise performance has been shown to decline with age in children undergoing staged interventions for complex congenital heart disease.413 HLHS is an extremely complex congenital heart defect with multiple variations. To minimize morbidity and maximize quality of life, every child with HLHS requires continual, lifelong care by experts in management of adult congenital heart disease. Guidelines for management of adults with congenital heart disease are published by Warnes et al.927 Case reports have documented successful pregnancies following the Nowood procedure and later Fontan procedures.674,910
Fetal Intervention
Fetal aortic valvuloplasty has been performed to treat aortic stenosis with a goal of preventing the progression to HLHS in the fetus.562 Growth of the aorta and left ventricle with biventricular circulation has been documented postnatally following the fetal surgery.562
Stage 2 and Stage 3 Procedures
A bidirectional Glenn or hemi-Fontan procedure (SVC to pulmonary artery) may be performed at 4 to 6 months of age.514 A Fontan-type procedure (IVC to the pulmonary artery) anastomosis is the final stage for correction of hypoplastic left heart syndrome and is typically completed near the age of 2 to 4 years.
The Fontan operation separates the pulmonary and systemic circulations. The desaturated systemic venous blood is diverted directly into the pulmonary circulation. Fully saturated pulmonary venous blood is ejected into the systemic circulation by the right ventricle (see section, Single Ventricle, Overview of Management [Including Surgical Procedures]). Before these staged procedures, catheter-based interventions may be completed to treat residual lesions. The goal is to prepare the myocardium and pulmonary vascular bed, avoiding pulmonary vessel distortion and maintaining normal pulmonary vascular resistance.
Advanced Concepts
Advanced concepts in the care of the patient with hypoplastic left heart syndrome are listed in Box 8-43.
Box 8-43 Advanced Concepts: Hypoplastic Left Heart Syndrome
• Following stage I palliation, targeted oxygen saturation is approximately 80%. An elevated arterial oxygen saturation (above 85% to 88%) can indicate pulmonary overcirculation that can compromise systemic blood flow and perfusion.
• An elevated arterial oxygen saturation following creation of a systemic to pulmonary artery shunt can be caused by development of aortic re-coarctation distal to the placement of the Blalock-Taussig (or systemic to pulmonary artery) shunt. Assess for difference between upper versus lower extremity pulses and blood pressures.
• A restrictive atrial septal defect may develop following stage I palliation, particularly after a hybrid stage I procedure. A restrictive atrial septal defect limits the left to right atrial shunt and produces elevated left atrial and pulmonary venous pressures, pulmonary edema, increasing hypoxemia, cyanosis, and respiratory distress.
• Oxygen is a potent pulmonary vasodilator and use in single ventricle patients is unnecessary if the systemic arterial oxygen saturation is above75% or the PaO2 is greater than 30 mm Hg.
• Interstage morbidity and mortality remain significant after palliation for HLHS. Changes in feeding, increased respiratory effort, tachycardia, lack of weight gain, and irritability may signal adverse hemodynamic changes requiring intervention.