Pain Assessment
The Fifth Vital Sign
Outline
http://evolve.elsevier.com/Jarvis/
Structure and Function
Neuroanatomic Processing of Pain
Currently, we understand pain to develop by two main processes: nociceptive and/or neuropathic processing. It is important to understand how these two types of pain develop because patients will present with distinguishing sensations and respond differently to analgesics. When we are better able to assess the type(s) of pain, clinicians can more accurately select effective pharmacologic and nonpharmacologic strategies to interrupt the pain processing along multiple points within the pain messaging system and ultimately provide improved pain relief.38
Neuroanatomic Pathway
Pain is a highly complex and subjective experience that originates from the central nervous system (CNS) or peripheral nervous system (PNS), or both. Specialized nerve endings called nociceptors are designed to detect painful sensations from the periphery and transmit them to the CNS. Nociceptors are located within the skin, connective tissue, muscle, and the thoracic, abdominal, and pelvic viscera. These nociceptors can be stimulated directly by trauma or injury or secondarily by chemical mediators that are released from the site of tissue damage.
Nociceptors carry the pain signal to the central nervous system by two primary sensory (or afferent) fibers: Aδ and C fibers (Fig. 10-1). Aδ fibers are myelinated and larger in diameter, so they transmit the pain signal rapidly to the CNS. The sensation is very localized, short-term, and sharp in nature because of the Aδ fiber stimulation. In contrast, C fibers are unmyelinated and smaller and they transmit the signal more slowly. The “secondary” sensations are diffuse and aching, and they last longer after the initial injury.
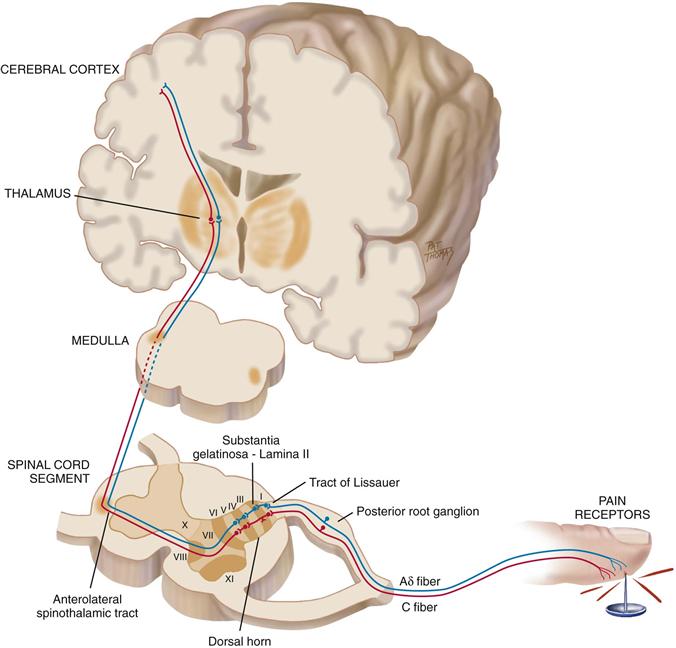
10-1
Peripheral sensory Aδ and C fibers enter the spinal cord by posterior nerve roots within the dorsal horn by the tract of Lissauer. The fibers synapse with interneurons located within a specified area of the cord called the substantia gelatinosa. A cross section shows that the gray matter of the spinal cord is divided into a series of consecutively numbered laminae (layers of nerve cells) (see Fig. 10-1). The substantia gelatinosa is lamina II, which receives sensory input from various areas of the body. The pain signals then cross over to the other side of the spinal cord and ascend to the brain by the anterolateral spinothalamic tract. Pain researchers are demonstrating that when pain is poorly controlled over an extended period, cells within the dorsal horn become altered in size and function and this damage ultimately turns future pain signals into more exaggerated or hypersensitive processing.6,8
Nociceptive Pain
Nociceptive pain develops when nerve fibers in the periphery and in the central nervous system are functioning and intact. Nociceptive pain starts outside of the nervous system from actual or potential tissue damage. Nociception can be divided into four phases: (1) transduction, (2) transmission, (3) perception, and (4) modulation (Fig. 10-2).
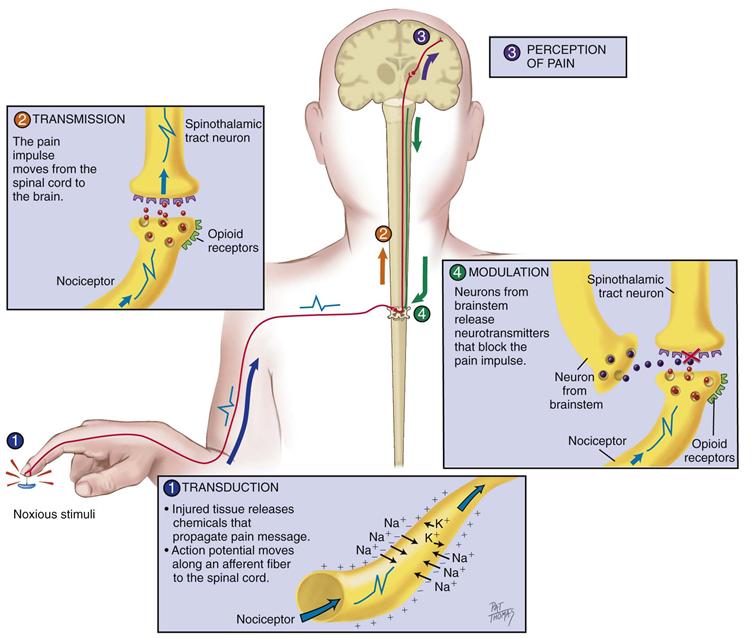
10-2
Initially, the first phase of transduction occurs when a noxious stimulus in the form of traumatic or chemical injury, burn, incision, or tumor takes place in the periphery. The periphery includes the skin, as well as somatic and visceral structures. These injured tissues then release a variety of chemicals, including substance P, histamine, prostaglandins, serotonin, and bradykinin. These chemicals are neurotransmitters that propagate a pain message, or action potential, along sensory afferent nerve fibers to the spinal cord. These nerve fibers terminate in the dorsal horn of the spinal cord. Because the initial afferent fibers stop in the dorsal horn, a second set of neurotransmitters carries the pain impulse across the synaptic cleft to the dorsal horn neurons. These neurotransmitters include substance P, glutamate, and adenosine triphosphate (ATP).
In the second phase, known as transmission, the pain impulse moves from the level of the spinal cord to the brain. Within the spinal cord, at the site of the synaptic cleft, are opioid receptors that can block this pain signaling with our own endogenous opioids or with exogenous opioids if they are administered. However, if not stopped, the pain impulse moves to the brain via various ascending fibers within the spinothalamic tract to the thalamus. Once the pain impulse moves through the thalamus, the message is dispersed to higher cortical areas via mechanisms that are not clearly understood at this time.
The third phase, perception, indicates the conscious awareness of a painful sensation. Cortical structures such as the limbic system account for the emotional response to pain, and somatosensory areas can characterize the sensation. Only when the noxious stimuli are interpreted in these higher cortical structures can this sensation be identified as “pain.”
Last, the pain message is inhibited through the phase of modulation. Fortunately, our bodies have a built-in system that will eventually slow down and stop the processing of a painful stimulus. If not, we would continue to experience pain from childhood injuries and beyond. Descending pathways from the brainstem to the spinal cord produce a third set of neurotransmitters that slow down or impede the pain impulse, producing an analgesic effect. These neurotransmitters include serotonin, norepinephrine, neurotensin, γ-aminobutyric acid (GABA), and our own endogenous opioids—β-endorphins, enkephalins, and dynorphins.
This type of nociceptive processing is protective.10 It is a warning signal that injury is about to or has taken place. We quickly learn to move our hand away from a burning flame. Other examples of nociceptive pain include a skinned knee, kidney stones, menstrual cramps, muscle strain, venipuncture, or arthritic joint pain. Nociceptive pain is typically predictable and time limited based on the extent of the injury.
Neuropathic Pain
Neuropathic pain is pain that does not adhere to the typical and rather predictable phases in nociceptive pain. Neuropathic pain implies an abnormal processing of the pain message from an injury to the nerve fibers. It is this type of pain that is most difficult to assess and treat. Pain is often perceived long after the site of injury heals and can start 2 to 3 years after an initial injury.
We are learning that the nociceptive pattern can change into a neuropathic pattern over time when pain has been poorly controlled. Because of the constant irritation and inflammation caused by a pain stimulus, the form of the nerve cells alters, making them more sensitive to any stimulus. The constant irritation also decreases the number of opioid receptors.
Conditions that may cause neuropathy include diabetes mellitus, herpes zoster (shingles), HIV/AIDS, sciatica, trigeminal neuralgia, phantom limb pain, and chemotherapy. Further examples include CNS lesions such as stroke, multiple sclerosis, and tumor. Pain is sustained on a neurochemical level that cannot be identified by x-ray, computerized axial tomography (CAT) scan, or magnetic resonance imaging (MRI); electromyography and nerve-conduction studies are needed.6,39
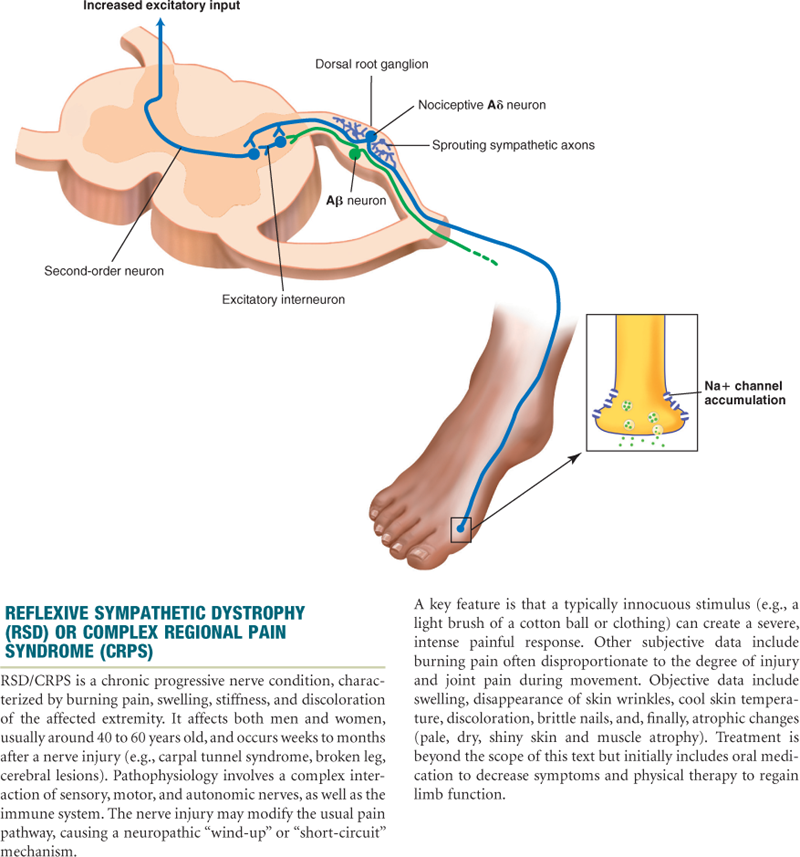
The abnormal processing of the neuropathic pain impulse can be continued by the peripheral or central nervous system. Exact mechanisms are unclear to date. A proposed mechanism is that injury to peripheral neurons can result in spontaneous and repetitive firing of nerve fibers, almost seizurelike in activity (Fig. 10-3). Neuropathic pain may be sustained centrally in a phenomenon known as neuronal “wind-up.” Within the dorsal horn of the spinal cord, neurons are thought to be transformed into a hyperexcitable state and a minimal stimulus can ultimately spiral into a much larger painful effect.
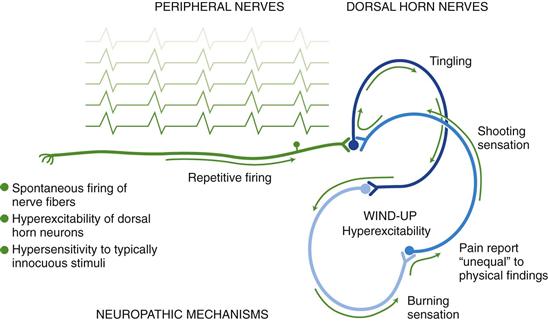
10-3
Sources of Pain
Pain sources are based on their origin. Visceral pain originates from the larger interior organs (i.e., kidney, stomach, intestine, gallbladder, pancreas). The pain can stem from direct injury to the organ or from stretching of the organ from tumor, ischemia, distention, or severe contraction. Examples of visceral pain include ureteral colic, acute appendicitis, ulcer pain, and cholecystitis. The pain impulse is transmitted by ascending nerve fibers along with nerve fibers of the autonomic nervous system. That is why visceral pain often presents along with autonomic responses such as vomiting, nausea, pallor, and diaphoresis.
Deep somatic pain comes from sources such as the blood vessels, joints, tendons, muscles, and bone. Injury may result from pressure, trauma, or ischemia. Cutaneous pain is derived from skin surface and subcutaneous tissues. The injury is superficial, with a sharp, burning sensation.
In past literature, you may have come across the term psychogenic pain. This term was attributed to pain with no known physical cause and assumed to have a psychiatric or emotional cause. It often was used as a derogatory label. Psychogenic is an obsolete term and is not accepted by the International Association for the Study of Pain. Linking pain to a mental disorder negates the person’s pain report. A clinician’s lack of awareness and understanding of neuropathic pain may contribute to this mislabeling.
Pain that is felt at a particular site but originates from another location is known as referred pain. Both sites are innervated by the same spinal nerve, and it is difficult for the brain to differentiate the point of origin. Referred pain may originate from visceral or somatic structures. Various structures maintain their same embryonic innervation. For example, an inflamed appendix in the right lower quadrant of the abdomen may have referred pain in the periumbilical area. It is useful to have knowledge of areas of referred pain for diagnostic purposes (see Table 21-2, Common Sites of Referred Abdominal Pain, on p. 559).
Types of Pain
Pain can be classified by its duration into acute or chronic categories (chronic is now more commonly called persistent because it carries a less negative, malingering connotation). The duration provides information on possible underlying mechanisms and treatment decisions.
Acute pain is short-term and self-limiting, often follows a predictable trajectory, and dissipates after an injury heals. Examples of acute pain include surgery, trauma, and kidney stones. Acute pain serves a self-protective purpose; acute pain warns the individual of actual or potential tissue damage. Incident pain is an acute type that happens predictably when certain movements take place. Examples include pain in the lower back upon standing or shoulder pain when arms are raised.
In contrast, persistent (or chronic) pain is diagnosed when the pain continues for 6 months or longer. It can last 5, 15, or 20 years and beyond. Persistent pain can be further divided into malignant (cancer-related) and nonmalignant. Malignant pain often parallels the pathology created by the tumor cells. The pain is induced by tissue necrosis or stretching of an organ by the growing tumor. The pain fluctuates within the course of the disease. Chronic nonmalignant pain is often associated with musculoskeletal conditions, such as arthritis, low back pain, or fibromyalgia.
Chronic pain does not stop when the injury heals. It persists after the predicted trajectory. Chronic pain outlasts its protective purpose, and the level of pain intensity does not correspond with the physical findings. Unfortunately, many patients with chronic pain are not believed and often are labeled as malingers, attention seekers, drug seekers, and so forth. Chronic pain originates from abnormal processing of pain fibers from peripheral or central sites. Because the pain is transmitted on a cellular level, our current technology cannot reliably detect this process. Therefore the most important and reliable indicator for pain is the patient’s self-report.
Finally, breakthrough pain is pain that starts again or escalates before the next scheduled analgesic dose. Pain breaks through when it is expected to be controlled by pain medications.21,26
 Developmental Competence
Developmental Competence
Infants
Infants have the same capacity for pain as adults. By 20 weeks’ gestation, ascending fibers, neurotransmitters, and the cerebral cortex are developed and functioning to the extent that the fetus is capable of feeling pain.29,30 However, inhibitory neurotransmitters are in insufficient supply until birth at full term. Therefore the preterm infant is rendered more sensitive to painful stimuli.
Preverbal infants are at high risk for undertreatment of pain because of persistent myths and beliefs that infants do not remember pain. In fact, new research indicates that repetitive and poorly controlled pain in infants (e.g., daily heel sticks, venipunctures) can result in lifelong adverse consequences such as neurodevelopmental problems, poor weight gain, learning disabilities, psychiatric disorders, and alcoholism.28
The Aging Adult
No evidence exists to suggest that older individuals perceive pain to a lesser degree or that sensitivity is diminished. Although pain is a common experience among individuals 65 years of age and older, it is not a normal process of aging. Pain indicates pathology or injury. Pain should never be considered something to tolerate or accept in one’s later years.
Unfortunately, many clinicians and older adults wrongfully assume that pain should be expected in aging, which leads to less aggressive treatment. Older adults have additional fears about becoming dependent, undergoing invasive procedures, taking pain medications, and having a financial burden. The most common pain-producing conditions for aging adults include pathologies such as arthritis, osteoarthritis, osteoporosis, peripheral vascular disease, cancer, peripheral neuropathies, angina, and chronic constipation.
People with dementia do feel pain. The somatosensory cortex is generally unaffected by dementia of the Alzheimer type. Sensory discrimination is preserved in cognitively intact and impaired adults.1 Because the limbic system is affected by Alzheimer disease, current research focuses on how the person interprets and reports these pain messages.24 See further discussion on pain assessment with dementia on p. 170.
Gender Differences
Gender differences are influenced by societal expectations, hormones, and genetic makeup. Traditionally, men have been raised to be more stoic about pain and more affective or emotional displays of pain are accepted for women. Hormonal changes are found to have strong influences on pain sensitivity for women. Women are two to three times more likely to experience migraines during childbearing years, are more sensitive to pain during the premenstrual period, and are six times more likely to have fibromyalgia.18 With recent findings from the Human Genome Project, genetic differences between both genders may account for the differences in pain perception.19 A pain gene exists, which helps explain why some people feel more/less pain even with the same stimulus. Efforts are being made to tailor pharmacologic agents to improve pain treatment based on genetic sequencing.
 Culture and genetics
Culture and genetics
Please review the cultural variations in Chapter 2. As clinicians, adopt the habit of asking each patient how he or she typically behaves when in pain.
Most of the research conducted on racial differences and pain has focused on the disparity in management of pain for various racial groups—comparing pain treatment for individuals of color (e.g., Blacks, Hispanics) with the standard treatment for all individuals with similar injuries or diseases. Various studies describe how Black and Hispanic patients are often prescribed and administered less analgesic therapy than whites, although the majority of these differences is quite small.16
Subjective Data
Pain is defined as an “unpleasant sensory and emotional experience associated with actual or potential tissue damage or described in terms of such damage. Pain is always subjective.”2
The subjective report is the most reliable indicator of pain. Because pain occurs on a neurochemical level, the diagnosis of pain cannot be made exclusively on physical examination findings, although these findings can lend support.

Pain Assessment Tools
Pain is multidimensional in scope, encompassing physical, affective, and functional domains. Various tools have been developed to capture unidimensional aspects (i.e., intensity) or multidimensional components. Select the pain assessment tool based on its purpose, time involved in administration, and the patient’s ability to comprehend and complete the tool. First, teach patients how to use each tool, with practice sessions to strengthen the validity and reliability of the response. Enlarge the print when appropriate for individuals with impaired vision. The printed language should be translated to the native language of the patient. Ask the patient to rate and evaluate all of the pain sites. Some forms allow for only one number, so be sure to add to your documentation. Finally, use the pain tool consistently before and after treatment to see if the treatment has been effective.
Standardized overall pain assessment tools are more useful for chronic pain conditions or particularly problematic acute pain problems. A few examples include the Initial Pain Assessment, the Brief Pain Inventory, and the McGill Pain Questionnaire.
In the Initial Pain Assessment,31 the clinician asks the patient to answer eight questions concerning location, duration, quality, intensity, and aggravating/relieving factors. Further, the clinician adds questions about the manner of expressing pain and the effects of pain that impair one’s quality of life (Fig. 10-4).
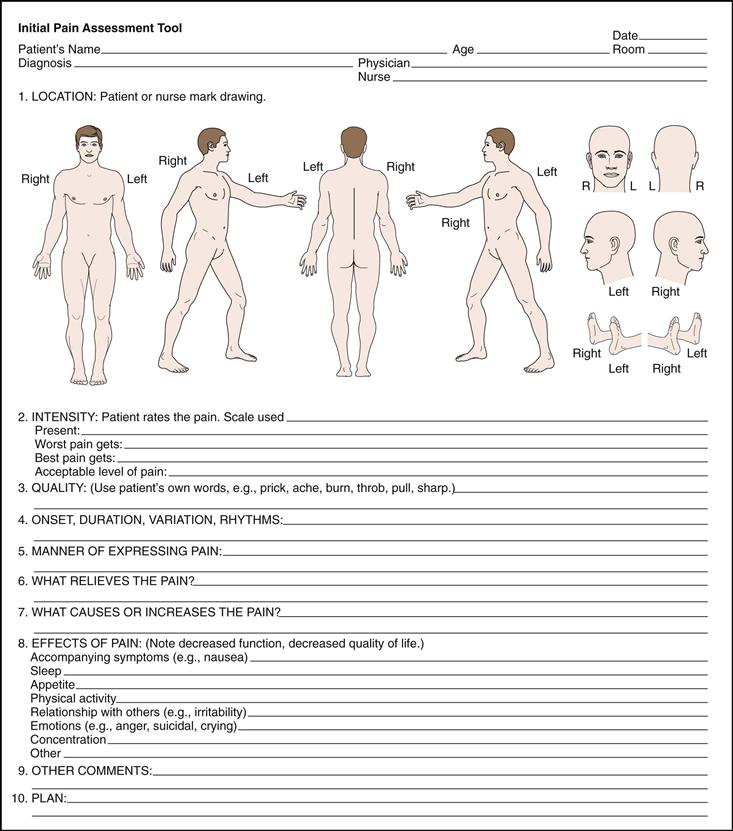
10-4
The Brief Pain Inventory15 asks the patient to rate the pain within the past 24 hours using graduated scales (0-10) with respect to its impact on areas such as mood, walking ability, and sleep (Fig. 10-5). The short-form McGill Pain Questionnaire33 (not illustrated) asks the patient to rank a list of descriptors in terms of their intensity and to give an overall intensity rating to his or her pain.
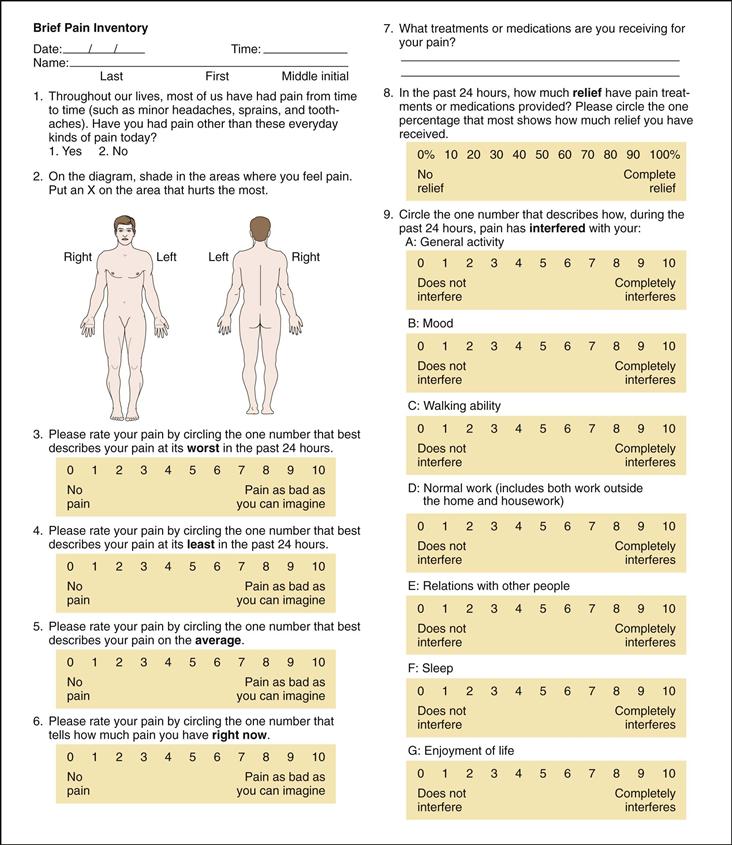
10-5
Pain rating scales are unidimensional and are intended to reflect pain intensity. They come in various forms. Pain rating scales can indicate a baseline intensity, track changes, and give some degree of evaluation to a treatment modality. Numeric rating scales ask the patient to choose a number that rates the level of pain for each painful site, with 0 being no pain and the highest anchor 10 indicating the worst pain ever experienced. It can be administered verbally or visually along a vertical or horizontal line (Fig. 10-6).
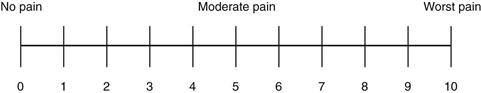
10-6
In general, older adults find the numeric rating scale abstract and have difficulty responding, especially with a fluctuating chronic pain experience. An alternative is the simple descriptor scale that lists words that describe different levels of pain intensity, such as no pain, mild pain, moderate pain, and severe pain. Older adults will often respond to scales in which words are selected. Again, it is essential to teach the person how to use the scale to enhance accuracy.
Infants and Children
Because infants are preverbal and incapable of self-report, pain assessment depends on behavioral and physiologic cues. Refer to the Objective Data section. It is important to underscore the understanding that infants do feel pain.
Children 2 years of age can report pain and point to its location. They cannot rate pain intensity at this developmental level. It is helpful to ask the parent or caregiver what words the child uses to report pain (e.g., boo–boo, owie). Be aware that some children will try to be “grown up and brave” and often deny having pain in the presence of a stranger or if they are fearful of receiving a “shot.”
Rating scales can be introduced at 4 to 5 years of age. The Faces Pain Scale–Revised (FPS-R) has six drawings of faces that show pain intensity, from “no pain” on the left (score of 0) to “very much pain” on the right (score of 10) (Fig. 10-7). Numbers are not shown to children, but the number scoring makes this tool compatible with the widely used 0-to-10 metric for numeric pain scales.23 This revised drawing has more realistic facial expressions with a furrowed brow and horizontal mouth stretch to rate pain. It avoids smiles or tears, so that children will not confuse pain intensity with happiness or sadness.

10-7
Similarly, the Oucher Scale7 has six photographs of young boys’ faces with different expressions of pain, ranked on a 0-to-5 scale of increasing intensity. The child is asked to point at the face that best matches his or her hurt/pain. You may use Oucher Scale variations for girls and diverse ethnic groups.
Objective Data
| PREPARATION | EQUIPMENT NEEDED |
| The physical examination process can help you understand the nature of the pain. Consider whether this is an acute or a chronic condition. Recall that physical findings may not always support the patient’s pain complaints, particularly for chronic pain syndromes. Pain should not be discounted when objective physical evidence is not found. Based on the patient’s pain report, make every effort to reduce or eliminate the pain with appropriate analgesic and nonpharmacologic intervention. According to the American Pain Society2: In cases in which the cause of acute pain is uncertain, establishing a diagnosis is a priority, but symptomatic treatment of pain should be given while the investigation is proceeding. With occasional exceptions (e.g., the initial examination of the patient with an acute condition of the abdomen), it is rarely justified to defer analgesia until a diagnosis is made. In fact, a comfortable patient is better able to cooperate with diagnostic procedures. (p. 3) |
Tape measure to measure circumference of swollen joints or extremities Tongue blade |
| Normal Range of Findings | Abnormal Findings |
| THE JOINTS | |
| Note the size and contour of the joint. Measure the circumference of the involved joint for comparison with baseline. Check active or passive range of motion (see discussion of complete technique beginning on p. 578in Chapter 22). Joint motion normally causes no tenderness, pain, or crepitation. | Swelling, inflammation, injury, deformity, diminished range of motion, increased pain on palpation. Crepitation is an audible and palpable crunching that accompanies movement. |
| THE MUSCLES AND SKIN | |
| Inspect the skin and tissues for color, swelling, and any masses or deformity. | Bruising, lesions, open wounds, tissue damage, atrophy, bulging, change in hair distribution. |
| To assess for changes in sensation, ask the person to close his or her eyes. Test the person’s ability to perceive sensation by breaking a tongue blade in two lengthwise. Lightly press the sharp and blunted ends on the skin in a random fashion and ask to identify it as sharp or dull (see Fig. 23-23). This test will help you identify location and extent of altered sensation. | Absent pain sensation (analgesia); increased pain sensation (hyperalgesia); or if a severe pain sensation is evoked with a stimulus that does not normally induce pain (e.g., the blunt end of the tongue blade; cotton ball; clothing) (allodynia). |
| THE ABDOMEN | |
| Observe for contour and symmetry. Palpate for muscle guarding and organ size (see discussion of complete technique beginning on p. 545in Chapter 21). Note any areas of referred pain (see Table 21-2). | Swelling, bulging, herniation, inflammation, organ enlargement. |
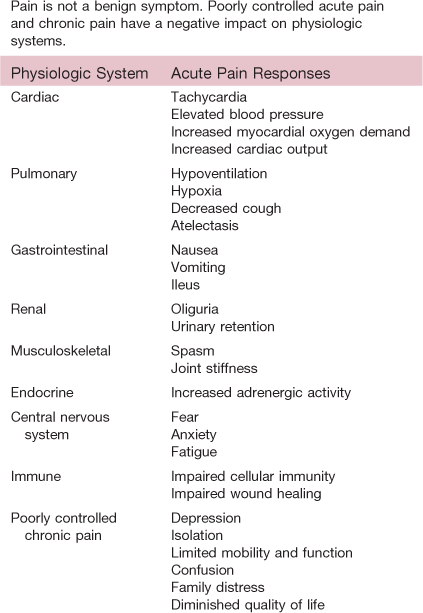
Table 10-1 lists physiologic changes resulting from poorly controlled pain.
Nonverbal Behaviors of Pain
When the individual cannot verbally communicate the pain, you can (to a limited extent) identify pain using behavioral cues. Recall that individuals react to painful stimuli with a wide variety of behaviors. Behaviors are influenced by a wide variety of factors, including the nature of the pain (acute vs. chronic), age, and cultural and gender expectations.
Acute Pain Behaviors
Because acute pain involves autonomic responses and has a protective purpose, individuals experiencing moderate to intense levels of pain may exhibit the following behaviors: guarding, grimacing, vocalizations such as moaning, agitation, restlessness, stillness, diaphoresis, or change in vital signs. This list of behaviors is not exhaustive because they should not be used exclusively to deny or confirm the presence of pain. For example, in a postoperative patient, pulse and blood pressure can be altered by fluid volume, medications, and blood loss.
Persistent (Chronic) Pain Behaviors
Persons with persistent pain often live with the experience for months and years. One cannot function physiologically and go on with life in a repetitive state of behaviors such as grimacing, diaphoresis, and guarding. The person adapts over time, and clinicians cannot look for or anticipate the same acute pain behaviors to exist in order to confirm a pain diagnosis.
Chronic pain behaviors have even more variability than acute pain behaviors. Persons with chronic pain typically try to give little indication that they are in pain and therefore are at higher risk for underdetection (Fig. 10-8). Behaviors that have been associated with chronic pain include bracing, rubbing, diminished activity, sighing, and change in appetite. Whenever possible, it is best to ask the person how he or she acts or behaves when in pain. Chronic pain behaviors, such as being with other people, movement, exercise, prayer, sleeping, or inactivity, underscore the more subtle, less anticipated ways in which persons behave when they are experiencing chronic pain. Sleeping is one way persons behave in response to chronic pain in order to self-distract. Unfortunately, clinical staff may inadvertently interpret this behavior as “comfort” and do not follow up with an appropriate pharmacologic intervention.

10-8
 Developmental Competence
Developmental Competence
Infants
Most pain research on infants has focused on acute procedural pain. We have a limited understanding of how to assess chronic pain in the infant. At this time, no one assessment tool adequately identifies pain in the infant. Using a multidimensional approach for the whole infant is encouraged. Changes in facial activity and body movements may help assess pain. Much effort and time is spent on decoding facial expressions (e.g., taut tongue, bulging brow, closing of eye fissures), which may be difficult for the general practitioner to carry out in a busy clinical setting.
One tool that has been developed for postoperative pain in preterm and term neonates is the CRIES score developed by Krechel and Bildner.27 It measures physiologic and behavioral indicators on a 3-point scale (Fig. 10-9).
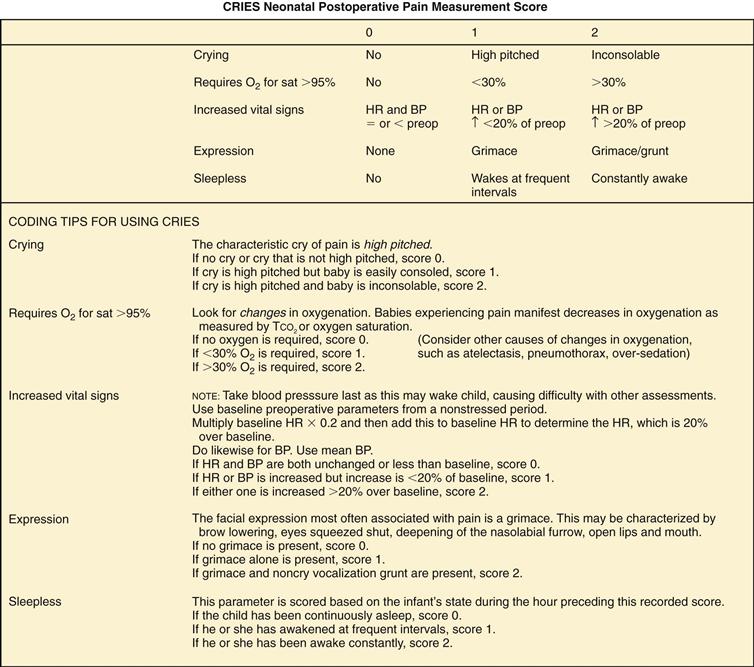
10-9
Because the sympathetic nervous system is engaged particularly in acute episodes of pain, physiologic changes take place that may indicate the presence of pain. These include sweating, increases in blood pressure and heart rate, vomiting, nausea, and changes in oxygen saturation. However, like the adult, these physiologic changes cannot be used exclusively to confirm or deny pain because of other factors such as stress, medications, and fluid changes.
Note that these measures target acute pain. No biological markers have been identified for long-term chronic pain in infants or children. Therefore evaluate the whole individual. Look for changes in temperament, expression, and activity. If a procedure or disease process is known to induce pain in adults (e.g., circumcision, surgery, sickle cell disease, cancer), it will induce pain in the infant or child.
The Aging Adult
Although pain should not be considered a “normal” part of aging, it is prevalent. When the older adult reports a history of conditions such as osteoarthritis, peripheral vascular disease, cancer, osteoporosis, angina, or chronic constipation, be alert and anticipate a pain problem. Older adults will often deny having pain for fear of dependency, further testing or invasive procedures, cost, and fear of taking pain killers or becoming a drug addict. During the interview, you must establish an empathic and caring rapport to gain trust.
When you look for behavioral cues, look at changes in functional status. Observe for changes in dressing, walking, toileting, or involvement in activities. A slowness and rigidity may develop, and fatigue may occur. Look for a sudden onset of acute confusion, which may indicate poorly controlled pain. However, you will need to rule out other competing explanations such as infection or adverse reaction from medications.
People with dementia become less able to identify and describe pain over time although pain is still present and destructive. The way people with dementia communicate pain is through their behavior. Agitation, pacing, and repetitive yelling may indicate pain and not a worsening of the dementia. People who are comfortable do not yell, cry, moan, hit, or kick. So when these behaviors occur, consider pain as a primary explanation.
When asked if they are having pain, people with dementia may say “No” when in fact they are very uncomfortable. Words have lost their meaning. Use the PAINAD scale (Fig. 10-10), which evaluates five common behaviors of breathing, vocalization, facial expression, body language, and consolability. Specific behaviors in these categories are quantified from 0 to 2, with a total score ranging from 0 to 10. This is consistent with the commonly used 0-to-10 metric on other pain tool scores. For the PAINAD, a score of 4 or more indicates a need for pain management.
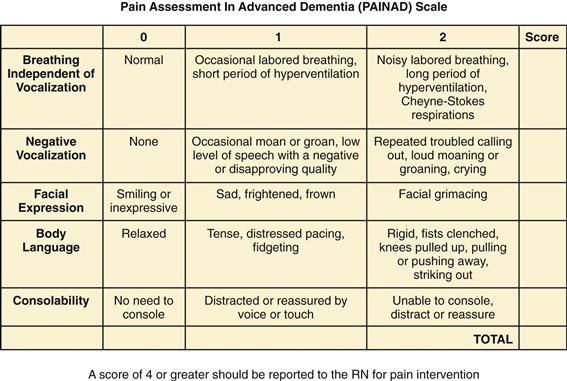
10-10
 Documentation and Critical Thinking
Documentation and Critical Thinking
Sample Charting
Subjective
Starting within the past 2 weeks, states having severe epigastric pain within a half-hour of eating greasy, fatty foods. Pain is stabbing and squeezing in nature with radiation to right shoulder blade. Rates pain as a 10 on a 0-to-10 scale. Nausea accompanies pain. Takes antacids with minimal relief. Pain diminishes after bringing knees to chest and “not moving” for a 1-hour period.
Objective
Patient diaphoretic, grimacing, and having difficulty concentrating. Breathless during history. Arms guarding upper abdominal area. Abdomen distended. Severe tenderness noted upon light left upper quadrant and epigastric palpation. Bowel sounds hyperactive in all four quadrants.
Assessment
Acute episodic pain
Focused Assessment: Clinical Case Study 1
R.M. is a 20-year-old African-American male diagnosed with sickle cell crisis. Admitted to the emergency department.
Subjective
Within the past 48 hours, R.M. reports increasing pain in upper and lower extremity joints and swelling of right knee. States having “stomach flu” 1 week before with periods of vomiting and diarrhea. Pain is aching and constant in nature. Rates pain as +10 on a 0-to-10 scale. Reports difficulty walking and climbing stairs. Taking ibuprofen, two tablets every 4 hours, and using ice packs with no relief.
Objective
Requiring assistance to sit on exam table. Unable to bear weight on right leg. Affect flat, clenches jaw during position changes. Tenderness localized in elbow, wrist, finger, and knee joints. Diminished range of motion in wrists and knees (right knee 36 cm, left knee 30 cm diameter). Right knee warm and boggy to touch.
Focused Assessment: Clinical Case Study 2
A.G. is an 85-year-old Irish-American female with a 20-year history of osteoarthritis.
Subjective
A.G. reports increased pain and stiffness in her neck, arms, and lower back for the past month. Denies radiation of pain. Denies tingling or numbness in upper or lower extremities.
Having difficulty getting in and out of bathtub and dressing herself. Describes pain as aching, with good and bad days. Becomes frustrated when asked to rate her pain intensity. Replies, “I don’t know what number to give; it hurts a lot, on and off.” Takes acetaminophen, extra strength, two tablets, when the pain “really gets the best of me,” with some degree of relief. Does not take part in “field trips” offered by assisted living facility because she “hurts too much.”
Objective
Localized tenderness noted upon palpation to C3 and C4; unable to flex neck to chest. Crepitus noted in both shoulder joints. No swelling noted. Muscle strength 1+ and equal for upper extremities. Lumbar area tender to moderate palpation. Rubs lower back frequently; limited flexion at the waist. Gait slow and unsteady. Facial expression stoic.
Assessment
Chronic pain
Abnormal Findings
TABLE 10-2
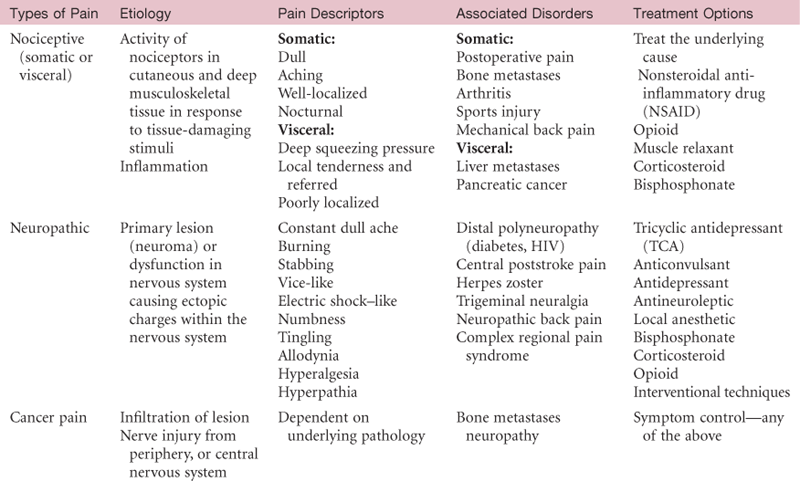
Data from Miller-Saultz, D. (2008). Identifying chronic pain: Awareness important. Nurse Practitioner 33(9):7.
Bibliography
1. American Geriatrics Society. Pharmacological management of persistent pain in older persons. Journal of the American Geriatrics Society. 2009;57(8):1331–1346.
2. American Pain Society (APS). Principles of analgesic use in the treatment of acute and cancer pain. 3rd ed. Glenview, IL: Author; 1992.
3. Anand KJS. The applied physiology of pain. In: Anand KJS, McGrath RJ, eds. Pain in neonates. Amsterdam: Elsevier; 1993.
4. Anand KJS. Effects of perinatal pain and stress. Progress in Brain Research. 2000;122:117–119.
5. Anderson KO, Green CR, Payne R. Racial and ethnic disparities in pain: causes and consequences of unequal care. The Journal of Pain. 2009;10(12):1187–1204.
6. Balasubramanyan S, Smith P. Cellular changes in the superficial dorsal horn in nerve-injury models of neuropathic pain. Journal of Neuropathic Pain & Symptom Palliation. 2007;2(2):9–42.
7. Beyer JE. The Oucher: a user’s manual and technical report. Evanston, IL: Judson; 1983.
8. Bird G, Han J, Fu Y, et al. Pain-related synaptic plasticity in spinal dorsal horn neurons: role of CGRP. Molecular Pain. 2006;2:31 Retrieved February 2010 from www.molecularpain.com/content/2/1/31; 2006.
9. Buffum MD, Miaskowski C, Sands L, Brod M. A pilot study of the relationship between discomfort and agitation in patients with dementia. Geriatric Nursing. 2001;22(2):80–85.
10. Cervero F. Pain: friend or foe? A neurobiologic perspective—the 2008 Bonica award lecture. Regional Anesthesia and Pain Medicine. 2009;34(6):569–574.
11. Chen Y, Kelly J. Reflex sympathetic dystrophy: a case of total body pain. The Nurse Practitioner. 2007;32(9):8–10.
12. Cruccu G, Truini A. Tools for assessing neuropathic pain. PLoS Medicine. 2009;6(4):e1000045 doi:10.1371/journal/pmed.1000045.
13. D’Arcy Y. Field guide to pain Part 1 Screening for pain in primary care. The Nurse Practitioner. 2005;30(9):46–48.
14. D’Arcy Y. Pain in the older adult. The Nurse Practitioner. 2008;33(3):18–24.
15. Daut RL, Cleeland CS. The prevalence and severity of pain in cancer. Cancer. 1982;50:1913–1918.
16. Ezenwa MO, Ameringer S, Ward SE, et al. Racial and ethnic disparities in pain management in the United States. Journal of Nursing Scholarship. 2006;38(3):225–233.
17. Fillingim RB. Sex, gender and pain. Seattle: IASP Press; 2000.
18. Fillingim RB, Wallace MR, Herbstaman DM, et al. Genetic contributions to pain: a review of findings in humans. Oral Diseases. 2008;14(8):673–682.
19. Fillingim RB, King CD, Ribeiro-Dasilva MC, et al. Sex, gender, and pain: a review of recent clinical and experimental findings. The Journal of Pain. 2009;10(5):447–485.
20. Flaherty E. Using pain-rating scales with older adults. The American Journal of Nursing. 2008;108(6):40–48.
21. Helms JE, Barone CP. Physiology and treatment of pain. Critical Care Nurse. 2008;28(6):38–50.
22. Herr K, Coyne PJ, Tonya K, et al. Pain assessment in the nonverbal patient: position statement with clinical practice recommendations. Pain Management Nursing. 2006;7(2):44–52.
23. Hicks CL, von Baeyer CL, Spafford P, et al. The Faces pain scale—revised: toward a common metric in pediatric pain measurement. Pain. 2001;93:173–183.
24. Horgas A, Elliott A, Marsiske M. Pain assessment in persons with dementia: relationship between self-report and behavioral observation. Journal of the American Geriatrics Society. 2009;57(1):126–132.
25. Horgas A, Miller L. Pain assessment in people with dementia. The American Journal of Nursing. 2008;108(7):62–71.
26. Karp JF, Shega JW, Morone NE, Weiner DK. Advances in understanding the mechanisms and management of persistent pain in older adults. British Journal of Anaesthesia. 2008;101(1):111–120.
27. Krechel SW, Bildner J. CRIES: a new neonatal postoperative pain measurement score—initial testing of validity and reliability. Paediatric Anaesthesia. 1995;5(1):53–61.
28. LaPrairie J, Murphy A. Neonatal injury alters adult pain sensitivity by increasing opioid tone in the periaqueductal gray. Frontiers in Behavioral Neuroscience. 2009;3:31.
29. Loizzo A, Loizzo S, Capasso A. Neurobiology of pain in children: an overview. The Open Biochemistry Journal. 2009;3:18–25.
30. Lowery CL, Hardman MP, Manning N, et al. Neurodevelopmental changes of fetal pain. Seminars in Perinatology. 2007;31:275–282.
31. McCaffery M, Pasero C. Pain: clinical manual. 2nd ed. St. Louis: Mosby; 1999.
32. McCarberg B, D’Arcy Y. Target pain with topical peripheral analgesics. The Nurse Practitioner. 2007;32(7):44–49.
33. Melzack R. The short-form McGill Pain Questionnaire. Pain. 1987;30:191–197.
34. Mendell JR, Sahenk Z. Painful sensory neuropathy. The New England Journal of Medicine. 2003;348(13):1243–1255.
35. Miller-Saultz D. Identifying chronic pain: awareness important. The Nurse Practitioner. 2008;33(9):6–9.
36. Mogil JS. Pain genetics: pre- and post-genomic findings. International Association for the Study of Pain Technical Corner Newsletter. 2002;2:3–6.
37. Polomano RC, Farrar JT. Pain and neuropathy in cancer survivors. The American Journal of Nursing. 2006;106(3):39–47.
38. Rosenblum A, Marsch L, Herman J, Portenoy R. Opioids and the treatment of chronic pain: controversies, current status, and future directions. Experimental and Clinical Psychopharmacology. 2008;16(5):405–416.
39. Schestatsky P, Nascimento OJ. What do general neurologists need to know about neuropathic pain? Arquivos de Neuro-psiquiatria. 2009;67(3-A):741–749.
40. Solano JP, Gomes B, Higginson IJ. A comparison of symptom prevalence in far advanced cancer, AIDS, heart disease, chronic obstructive pulmonary disease and renal disease. Journal of Pain and Symptom Management. 2006;31(1):58–69.
41. Spagrud LJ, Piira T, von Baeyer CL. Children’s self-report of pain intensity. The American Journal of Nursing. 2003;103(12):62–64.
42. Turk DC, Melzack R, eds. Handbook of pain assessment. New York: Guilford Press; 1992.
43. Vreeling FW, Houx PJ, Jolles J, Verhey FR. Primitive reflexes in Alzheimer’s disease and vascular dementia. Journal of Geriatric Psychiatry and Neurology. 1995;8(2):111–117.
Website of Interest
1. International Association for the Study of Pain (IASP). In: www.IASP.org.