5 PERIODONTAL CONSIDERATIONS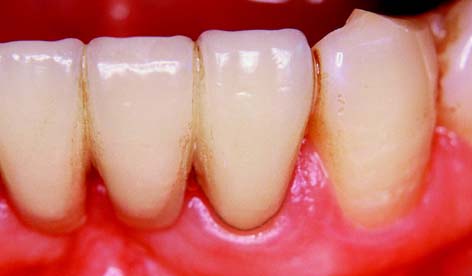
In the fabrication of any fixed prosthesis, the practitioner must determine the periodontal status of the involved abutment teeth. This allows a reliable and accurate prognosis for the restoration. Because periodontal disease is a major cause of tooth loss in adults, the dentist must be aware of the basic concepts and clinical modes of therapy available in periodontics to be able to develop an appropriate diagnosis and treatment plan.
This chapter reviews those concepts and treatment modalities and gives the practitioner a better understanding of periodontics and how it relates to restorative dentistry.
ANATOMY
The lining of the oral cavity consists of three types of mucosa, each with a different function1:
Gingiva
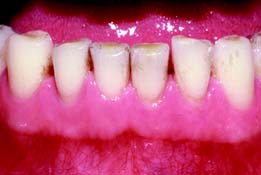
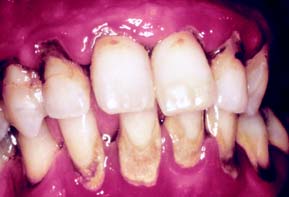
Normal gingiva (Fig. 5-1)—exhibiting no fluid exudate or inflammation caused by bacterial plaque—is pink and stippled. It varies in width from 1 to 9 mm and extends from the free margin of the gingiva to the alveolar mucosa. The gingiva and alveolar mucosa are separated by a demarcation called the mucogingival junction (MGJ), which marks the differentiation between stippled keratinized tissue and smooth, shiny mucosa; the latter contains more elastic fibers in its connective tissue. Apical to the MGJ, the alveolar mucosa then forms the vestibule and attaches to the muscles and fascia of the lips and cheeks.
The gingiva (Fig. 5-2) consists of three parts:
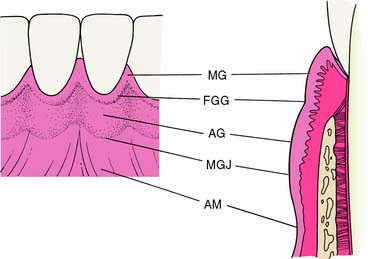
Fig. 5-2 Normal gingival structure and anatomic landmarks. MG, Marginal gingiva; FGG, free gingival groove; AG, attached gingiva; MGJ, mucogingival junction; AM, alveolar mucosa.
(Redrawn from Schluger S, et al: Periodontal Disease: Basic Phenomena, Clinical Management, and Occlusal and Restorative Interrelationships, 2nd ed. Philadelphia, Lea & Febiger, 1990.)
A V-shaped depression on the labial or buccal surface of the gingiva at or somewhat apical to the level of the epithelial attachment to the tooth is called the free gingival groove. It is not always readily apparent clinically but can be seen histologically and may serve as a reference point for dividing the free gingiva from the labially or buccally attached gingiva.2,3
The gingiva consists of dense collagen fibers, sometimes referred to as the gingivodental ligament, which can be divided into alveologingival, dentogingival, circular, dentoperiosteal, and transseptal groups. These fibers firmly bind the gingiva to the teeth and are continuous with the underlying alveolar periosteum. A more detailed description can be found in standard periodontal texts.4-8
Periodontium
The periodontium is a connective tissue structure attached to the periosteum of both the mandible and the maxilla that anchors the teeth in the mandibular and maxillary alveolar processes. It provides attachment and support, nutrition, synthesis and resorption, and mechanoreception. The main element of the periodontium is the periodontal ligament (PDL), which consists of collagenous fibers embedded in bone and cementum, giving support to the tooth in function (Fig. 5-3). These fibers, also known as Sharpey’s fibers, follow a wavy course and terminate in either cementum or bone. There are five principal fiber groups in the PDL that traverse the space between the tooth root and alveolar bone, providing attachment and support4:
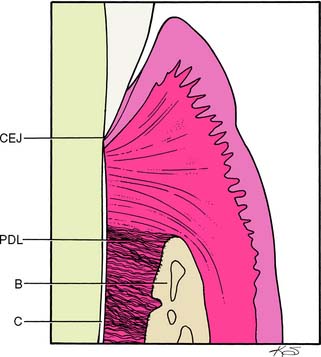
Fig. 5-3 Normal tooth-gingival interface and coronal periodontium. CEJ, cementoenamel junction; PDL, periodontal ligament. B, bone; C, cementum.
There are also smaller, irregularly arranged collagen fibers interspersed between the principal fiber groups. In addition, the PDL contains elastic fibers9 as well as oxytalan fibers.10
Cellular elements found in the PDL include fibroblasts (the main synthetic cells, which produce collagen and other proteoglycans), cementoblasts and cementoclasts, osteoblasts and osteoclasts (which maintain the viability of their respective tissues), and mast cells and epithelial rests (which play a role in pathologic conditions of the periodontium).1
Dentogingival Junction
At the base of the gingival sulcus (crevice) is the epithelium-tooth interface, also known as the dentogingival junction (DGJ). This structural relationship between hard and soft tissues is unique in the body. At the ultrastructural level, it is made up of hemidesmosomes and a basal lamina, which anchor the epithelial cells to the enamel and cemental surfaces.4,11
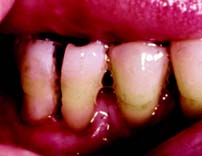
The depth of the sulcus varies in healthy individuals, averaging 1.8 mm.12 In general, the shallower it is, the more likely the gingiva is to be in a state of health. Sulcular depths up to 3 mm are considered maintainable. The continued maintenance of the gingiva in a state of health depends on tight, shallow sulci, which in turn depend on optimal plaque control and ensure the success of periodontal therapy, as well as affording a good prognosis for subsequent restorative treatment.
DISEASES OF THE PERIODONTIUM
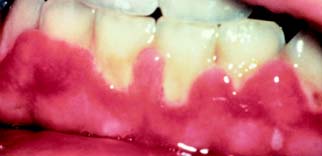
The general term periodontal disease is used to describe any condition of the periodontium other than normal. It covers such pathologic states as gingival hyperplasia, juvenile periodontitis (also known as periodontosis), and acute necrotizing ulcerative gingivitis—all distinct clinical entities that warrant specific treatment. Information concerning these disease states is available in any of the standard periodontal texts.4-8 Periodontal disease must be recognized and treated before fixed prosthodontics so that the gingival tissue levels can be determined for proper margin placement, esthetics, and gingival displacement (with an AlCl3-impregnated or plain cord; see Chapter 14). Only when the gingiva and periodontium are in an optimal state of health can these determinations be made with ease or predictability.
This discussion is limited to the etiology and progression of the inflammatory gingivitis-periodontitis lesion, which affects the majority of adults13 and constitutes the bulk of pathologic disorders necessitating treatment before restorative dentistry.
Etiology
Most gingival and periodontal diseases result from microbial plaque, which causes inflammation and its subsequent pathologic processes. Other contributors to inflammation include calculus, acquired pellicle, materia alba, and food debris.5,14
Terminology
Microbial plaque
Microbial plaque (Fig. 5-4) is a sticky substance composed of bacteria and their byproducts in an extracellular matrix; it also contains substances from the saliva, diet, and serum. It is basically a product of the growth of bacterial colonies and is the initiating factor in gingival and periodontal disease. If left undisturbed, it gradually covers an entire tooth surface and can be removed only by mechanical means.
Calculus
Dental calculus is a chalky or dark deposit attached to the tooth structure. It is essentially microbial plaque that has undergone mineralization over time. Calculus can be found on tooth structure in a supragingival and/or a subgingival location.
Structure of the dental plaque
Dental plaque consists mainly of microorganisms, scattered leukocytes, enzymes, food debris, epithelial cells, and macrophages in an intracellular matrix. Bacteria make up 70% of the solid portion of the mass. The remainder is an intracellular matrix consisting of carbohydrates, proteins, and calcium and phosphate ions.15-17
As the plaque mass increases and matures, the flora progresses apically from a supragingival position, facilitated by the presence of gingival crevicular fluid. The flora also change from a predominantly gram-positive, aerobic, and facultatively anaerobic population of coccoid morphology to a mix relatively high in gram-negative, anaerobic, and rodlike or filamentous organisms, along with increasing numbers of spirochetes.18,19 There is evidence5 that an increase in gram-negative organisms leads to an increase in disease activity within the periodontium and causes both direct and indirect tissue damage.
As the plaque colony matures and increases its mineral content, calculus forms within the plaque mass. Although gingival inflammation is often most severe in areas where calculus is present, the calculus itself is not the most significant source of inflammation; rather, it provides a nidus for plaque accumulation and retains the plaque in proximity to the gingiva. Dental plaque is the etiologic agent of the inflammation.20
Pathogenesis
The pathogenesis or sequence of events in the development of a gingivitis-periodontitis lesion is very complex. It involves not only local phenomena in the gingiva, PDL, tooth surface, and alveolar bone but also a number of complex host response mechanisms modified by the bacterial infection and behavioral factors.21 Implicated in the pathogenic mechanism are phagocytic cells, the lymphoid system, antibodies and immune complexes, complement and clotting cascades, immune reactions, and the microcirculation. Detailed descriptions of host response in the gingivitis-periodontitis lesion are available in standard periodontal texts and reviews.5-7
The chronic plaque-induced lesion has been investigated5,22 in great detail clinically, histopathologically, and ultrastructurally, and the model of disease activity has remained consistent over time. From these analyses, an indistinct division into initial, early, established, and advanced stages has been put forth. The salient features and approximate time frame for each stage are presented here.
Initial lesion
The initial lesion (Fig. 5-5) is localized in the region of the gingival sulcus and is evident after approximately 2 to 4 days of undisturbed plaque accumulation from a baseline of gingival health. The vessels of the gingiva become enlarged, and vasculitis occurs, allowing a fluid exudate of polymorphonuclear leukocytes to form in the sulcus. Collagen is lost perivascularly, and the resultant space is filled with proteins and inflammatory cells. The most coronal portion of the junctional epithelium becomes altered.
Early lesion
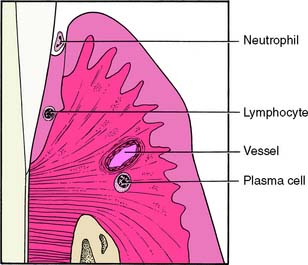
Although there is no distinct division between the stages of lesion formation, the early lesion (Fig. 5-6) generally appears within 4 to 7 days of plaque accumulation. This stage of development exhibits further loss of collagen from the marginal gingiva. In addition, an increase in gingival sulcular fluid flow occurs with increased inflammatory cells and the accumulation of lymphoid cells subjacent to the junctional epithelium. The basal cells of the junctional epithelium begin to proliferate, and significant alterations are seen in the connective tissue fibroblasts.
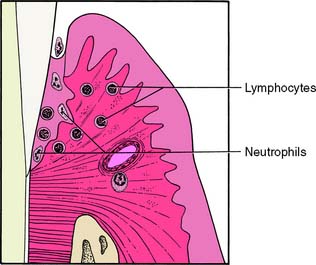
Fig. 5-6 Early lesion of gingivitis-periodontitis. The predominant inflammatory cells are lymphocytes subjacent to the junctional epithelium. The epithelium is beginning to proliferate into rete ridges.
(Redrawn from Schluger S, et al: Periodontal Disease: Basic Phenomena, Clinical Management, and Occlusal and Restorative Interrelationships, 2nd ed. Philadelphia, Lea & Febiger, 1990.)
Established lesion
Within 7 to 21 days, the lesion enters the established stage (Fig. 5-7). It is still located at the apical portion of the gingival sulcus, and the inflammation is centered in a relatively small area. There is continuing loss of connective tissue, with persistence of the features of the early lesion. This stage exhibits a predominance of plasma cells, the presence of immunoglobulins in the connective tissue, and a proliferation of the junctional epithelium (Fig. 5-8). Pocket formation, however, does not necessarily occur.
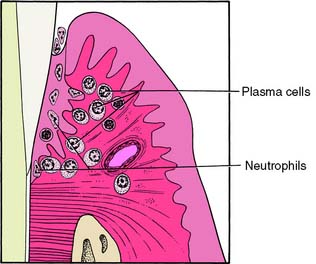
Fig. 5-7 Established lesion of gingivitis-periodontitis. The junctional epithelium is converted into pocket epithelium. Pocket formation may begin. The predominant inflammatory cells are plasma cells.
(Redrawn from Schluger S, et al: Periodontal Disease: Basic Phenomena, Clinical Management, and Occlusal and Restorative Interrelationships, 2nd ed. Philadelphia, Lea & Febiger, 1990.)
Advanced lesion
It is difficult to pinpoint the time at which the established lesion of gingivitis results in a loss of connective tissue attachment to the tooth structure and becomes an advanced lesion or overt periodontitis (Fig. 5-9). Upon conversion to the advanced stage, the features of an established lesion persist. The connective tissue continues to lose collagen content, and fibroblasts are further altered. Periodontal pockets are formed, with increased probing depths, and the lesion extends into alveolar bone. The bone marrow converts to fibrous connective tissue, with a significant loss of connective tissue attachment to the root of the tooth. This is accompanied by the manifestations of immunopathologic tissue reactions and inflammatory responses in the gingiva.
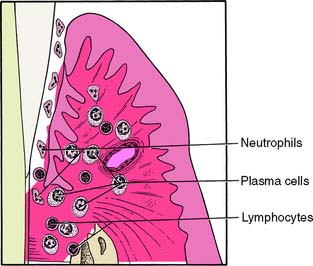
Fig. 5-9 Advanced lesion of gingivitis-periodontitis. Pocket formation has begun, with a loss of connective tissue attachment apical to the cementoenamel junction. Bone is converted into fibrous connective tissue and is subsequently lost. The predominant inflammatory cells are plasma cells, and there are scattered lymphocytes present.
Periodontitis
When a loss of connective tissue attachment occurs, the lesion transforms from gingivitis into periodontitis (Fig. 5-10), a disease that may be characterized by alternating periods of quiescence and exacerbation. The extent to which the lesion progresses before it is treated will determine the amount of bone and connective tissue attachment loss that occurs. It subsequently affects the prognosis of the tooth with regard to restorative demands.
EXAMINATION, DIAGNOSIS, AND TREATMENT PLANNING
Before treatment is rendered, all facts and findings related to the patient’s disease state should be recorded.23,24 These data can then be used to formulate a precise working blueprint for the proposed treatment. The diagnosis and treatment planning stages should be completed before therapy is initiated. In general practice, the data collection, diagnosis, and treatment planning for a patient’s restorative needs are accomplished at approximately the same time. Although laboratory assessments of gingival microflora and gingival crevicular fluid,24 as well as genetic tests for susceptibility to periodontal diseases, are available,25 traditional clinical assessments are most often employed to arrive at a periodontal diagnosis. These include such factors as probing depths, bleeding upon probing, clinical attachment levels, radiographic evidence of bone loss, and the presence or absence of the patient’s signs and symptoms.26,27 Once the diagnosis has been determined, a treatment plan is presented to the patient.
The treatment plan should be concise, logical, and rational—a realistic approach to therapy. It should not be a rigid or inflexible sequence of events, because often it must be emended as new information or changing circumstances dictate. The timing and sequencing of treatment are important in correcting the patient’s dental problems as efficiently as possible.
The following is a viable working model for periodontal treatment:
Initial Therapy
Initial therapy consists of all treatment carried out before evaluation for the surgical phases of periodontal therapy. A number of procedures in each patient’s treatment regimen may be accomplished before more definitive or invasive approaches are undertaken.
Control of microbial plaque
The most crucial aspect of periodontal therapy is the control of microbial flora in the sulcular area. If the patient does not maintain excellent oral hygiene and thereby the optimum condition of soft and hard tissues, the success of subsequent periodontal and restorative treatments will be jeopardized.
Bacterial plaque occurs on all surfaces of the teeth but is especially prevalent on the gingival third.28 It is strongly adherent to the tooth structure, which means that it is not removed by the chewing of fibrous foods.29 The prevention of plaque accumulation, by either mechanical or chemical means, is crucial in the prevention of hard and soft tissue pathosis. Although there are chemical means for removing plaque accumulation, only mechanical methods are considered in this text. Excellent reviews of the subject of chemical plaque removal are contained in standard periodontal texts.5,7
Toothbrushing
Plaque removal is accomplished with a toothbrush and other orophysiotherapy aids. Many types of toothbrushes can be used and are classified according to their size, shape, length, bristle arrangement, and whether they are manually or electrically powered. The many types of brushes and alternate techniques are reviewed in standard periodontal textbooks.4-7 The soft-bristle brush is particularly effective for cleaning in the gingival sulci and at buccal and lingual surfaces of interproximal areas30,31 without causing gingival damage and tooth abrasion, which can result from a hard-bristle brush.32
Technique
In toothbrushing, effective placement of the bristles is more important than the amount of energy expended. The Bass sulcular method of brushing is preferred for most fixed prosthodontics patients because it cleans the sulci, where the margins of restorations are often placed.
The bristles are placed in the sulci at an angle of approximately 45 degrees to the tooth surface, directed gingivally, and moved back and forth with short scrubbing motions under light pressure. The brush is applied in a similar manner throughout the mouth on all buccal and lingual or palatal surfaces of the teeth. In the anterior area, where interproximal spaces are small and where it may seem impossible to place the brush horizontally against the gingiva, the brush can be turned vertically for better access. After the sulcular areas have been cleansed, the occlusal surfaces are brushed, as is the dorsal surface of the tongue. Excellent descriptions and illustrations of toothbrush placement appear in standard periodontal texts.5-7
Flossing
Interproximal plaque can be controlled with dental floss.33,34 Both waxed and unwaxed types clean proximal surfaces, but the unwaxed floss has several advantages35:
Other Aids
Plaque may also be controlled effectively by orophysiotherapy aids such as dental tape, yarn, rubber and wooden tips, toothpicks, interdental stimulators, interproximal brushes, and electric toothbrushes.
When plaque is removed around a fixed dental prosthesis or a restoration involving splinted teeth, a floss threader may be needed. Alternatively, special lengths of floss with stiffened ends are available and have been shown to be quite effective.
Disclosing agents may be used to provide better visualization of areas where plaque control is difficult or deficient. Erythrosin dye in tablet or liquid form stains plaque, rendering it readily observable. Ultraviolet light has been used in combination with fluorescein dye to reveal plaque deposits, precluding the undesirable red stain that remains after erythrosin use.
All the previously mentioned items are useful in removing and controlling inflammation-inducing microbial plaque. However, the most important aspect of plaque control is patient motivation. Without motivation, all orophysiotherapy aids and the knowledge to apply them are useless.
Scaling and polishing
Removal of supragingival calculus (scaling) and polishing of the coronal portion of the tooth are the first definitive steps in débridement of the teeth. Scaling consists of the removal of deposits and accretions from the crowns of teeth and from tooth surfaces slightly subgingival. This is accomplished with the use of sharp scalers or curettes. The gingiva responds to this removal of supragingival and slightly subgingival calculus with a decrease in inflammation and bleeding. Thus, the patient is able to observe the first signs of therapeutic gain, especially when part or half of the mouth is instrumented at one appointment and the remainder is done after a short time has elapsed.
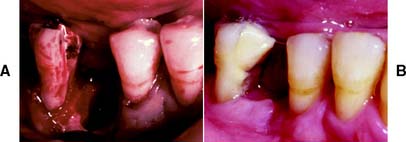
Correction of defective and/or overhanging restorations
Overhanging restorations, open interproximal contacts, and areas of food impaction contribute to local irritation of the gingiva and, of greater importance, impede proper plaque control. These deficiencies (Fig. 5-11) should be corrected during the initial therapy phase of treatment by either replacement or reshaping and/or removal of the overhang (Fig. 5-12). Close cooperation and communication between the periodontist and the restorative dentist are essential during this treatment phase.
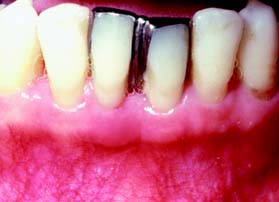
Fig. 5-11 Overhanging splinted restoration connecting the mandibular right and left central incisors, with obliteration of the interproximal space by the castings. The patient’s inability to clean this area properly has resulted in iatrogenic loss of attachment.
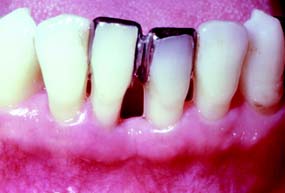
Fig. 5-12 Recontouring of the interproximal space of the castings seen in Figure 5-11 allows the patient to clean the area. Note the excellent gingival health between the central incisors as a result of good oral hygiene techniques.
Root planing
Root planing (Fig. 5-13) is the process of débriding the root surface with a curette. It is a more deliberate and more delicately executed procedure than scaling and necessitates the administration of a local anesthetic for most patients. At present, it constitutes the primary mode of initial therapy in periodontics, and there is evidence that tooth loss is substantially reduced over time when root surfaces are débrided on a continuing basis.36 Other work has also demonstrated that disease progression continues without root planing, even with effective oral hygiene.37
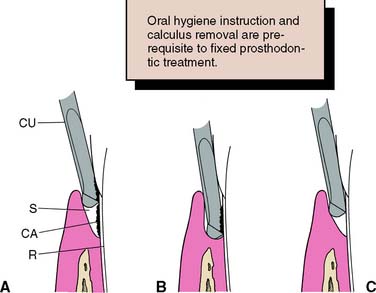
Fig. 5-13 Root planing. A, Curette placed in the sulcus to address calculus. B, The curette, initially placed apical to the calculus, moves coronally to dislodge the calculus. C, Accretions removed and the root planed to a smooth finish. CA, calculus; CU, Curette; R, root surface; S, sulcus.
(Redrawn from Carranza FA Jr: Glickman’s Clinical Periodontology, 7th ed. Philadelphia, WB Saunders, 1990.)
The curette is a spoon-shaped instrument well suited to cleaning and smoothing root surfaces. It is applied apically on the root with regard to the accretion and is moved coronally to lift deposits off the root surface and to plane it to a glasslike smoothness. As the patient’s plaque-control techniques improve, the changes observed when root planing is completed may necessitate changing or modifying the treatment plan, and further therapy may not be indicated.
Root planing and the incidental curettage of soft tissue that accompanies it may be an end point of active periodontal therapy. In many cases, the combination of root planing and improved oral hygiene on the part of the patient leads to manageable probing depths, and no further treatment is necessary. For this reason, the initial therapy requires careful evaluation.
Strategic tooth removal
An important part of treatment sequencing is the elimination of teeth that are hopelessly involved periodontally or are nonrestorable. Although no hard-and-fast rules exist regarding the timing of such extractions, removing teeth early in therapy is often more advantageous, when the patient has recently been informed of the prognosis and is prepared for treatment.
Extractions can be accomplished during initial therapy when the quadrant being instrumented is anesthetized. The operator can make an excellent determination of questionable teeth at this time by “sounding” the periodontium and can inform the patient of the verdict immediately. The patient is thus prepared psychologically (and also pharmacologically) for the removal. Teeth can also be removed during periodontal surgery, when the same conditions exist.
Early extraction of teeth and/or roots allows the socket areas to heal and can provide better access for plaque control of adjacent tooth surfaces. A transitional or provisional partial removable dental prosthesis or fixed dental prosthesis can also be fabricated; this stabilizes the arch and potentially maintains or improves occlusion, function, and esthetics.5,7
Stabilization of mobile teeth
Tooth mobility occurs when a tooth is subjected to excessive forces, especially when bony support is lacking. It is not necessarily a sign of disease, because it may be a normal response to abnormal forces, and it does not always need corrective treatment. However, it is sometimes a source of discomfort to the patient, and in these cases it should be treated by reduction of the abnormal forces after occlusal evaluation. Depending on the patient’s need, the teeth may also be treated by splinting with provisional restorations (see Chapter 15) or an acid-etch resin technique (see Chapter 26) in conjunction with occlusal adjustment (see Chapter 6). Such restorations should be carefully designed so they do not impede plaque control or future periodontal treatment. Close communication between the periodontist and the restorative dentist is crucial in this phase of treatment.
Minor tooth movement
Orthodontics can be of major benefit to periodontal therapy. Malposed teeth may be realigned to make them more receptive to periodontal treatment and to improve the efficacy of plaque-control measures. As described in Chapter 6, restorative procedures can also be aided by minor tooth movement. Thus, for the best treatment of a patient with complex dental problems, good communication among consulting dentists is essential.
Evaluation of Initial Therapy
The periodontium recovering from active disease should be regularly reexamined and reevaluated to determine the efficacy of treatment. Soft tissue responses to the initial therapy are observed along with the patient’s motivation and ability to maintain a relatively inflammation-free state. Probing depths should be recorded again, and the location of the MGJ noted in relation to the teeth. Changes must be assessed in regard to the necessity of further periodontal treatment.
Reevaluation gives the practitioner a firmer grasp on the progress of treatment, and if necessary, it allows revision of the initial treatment plan. At this time, the gingiva is healthier, probing depths may have decreased because of better plaque control and root planing, and the working knowledge of the patient’s abilities and desires should have improved. The combination of these factors facilitates decisions regarding further treatment of the periodontium and allows a more informed prognosis.
Surgical Therapy
There are a number of surgical procedures for the improvement of plaque removal aimed primarily at reducing or eliminating probing depths. Accurately diagnosing and choosing the most appropriate surgical regimen is crucial for maximum results.
Soft tissue procedures
Gingivectomy
Gingivectomy is the removal of diseased or hypertrophied gingiva. Introduced by G. V. Black,38,39 it was the first periodontal surgical approach to gain widespread acceptance. Gingivectomy is essentially the resection of keratinized gingiva only, and it may be applied to the treatment of suprabony pockets40 and to fibrous or enlarged gingiva, particularly when they result from diphenylhydantoin (Dilantin) therapy41 (see Fig. 1-4). However, it is unsuitable for the treatment of infrabony defects.
Technique
The surgical technique consists of establishing bleeding points (Fig. 5-14A) at the base of the gingival sulcus with a pocket marker or periodontal probe to serve as a guide for the gingival excision. The initial incision (Fig. 5-14B) is made at these points in a beveled manner with firm, continuous strokes from the gingivectomy knife. The interproximal tissue is freed by sharp excision and is removed from the site. The resulting ledge of tissue at the buccal and lingual or palatal terminations of the incision (Fig. 5-14C) is then smoothed with the knife or a rotary instrument to a margin continuous with the remaining tissue.
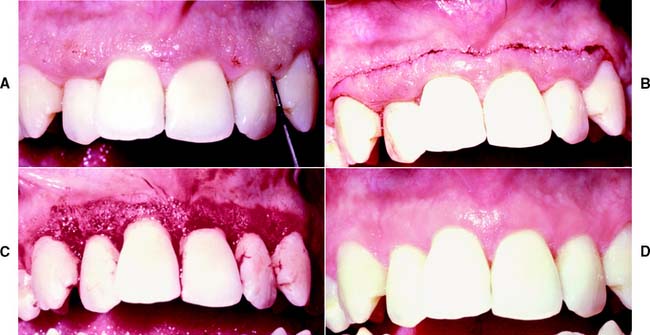
Fig. 5-14 A, Demarcation of pocket depth before the initial incision of a gingivectomy. B, Initial incision for the gingivectomy. C, Final gingival contours after removal of the coronal tissue and beveling of the incised area. D, Result of the gingivectomy, 6 months after surgery. Note the excellent gingival health and contours.
After vigorous débridement of the newly accessible tooth surfaces, a surgical dressing is applied for protection and hemostasis; it remains in place for 7 to 10 days. When it is removed, oral hygiene procedures are immediately resumed (Fig. 5-14D).
Contraindications
The major contraindication to gingivectomy-gingivoplasty is the absence of attached keratinized tissue. The procedure should be confined to areas of keratinized tissue to prevent leaving gingival margins that consist of alveolar mucosa (which is ill suited to resisting the trauma of restorative procedures and mastication).
Open débridement (modified Widman procedure)
Open débridement or curettage is a surgical procedure designed to gain better access to root surfaces for complete débridement and root planing. The modified Widman approach42 has been advocated because it allows good soft tissue flap control, minimum surgical trauma, and good postoperative integrity without excessive loss of osseous tissue or connective tissue attachment.
Technique
A sulcular or minimal internal bevel incision (Fig. 5-15) is made on the buccal or the lingual surfaces of the mandibular teeth. Next, a scalloped internal bevel incision is made on the palatal surfaces of maxillary teeth. The palatal flap is then thinned and the underlying connective tissue removed. The resulting flaps are reflected minimally yet sufficiently to allow access for complete débridement of the root surfaces and degranulation of any osseous lesions in the field. No osseous resection is accomplished, except where necessary for proper flap placement. The flaps are then carefully coapted and sutured to promote healing by primary intention (see Fig. 5-16).
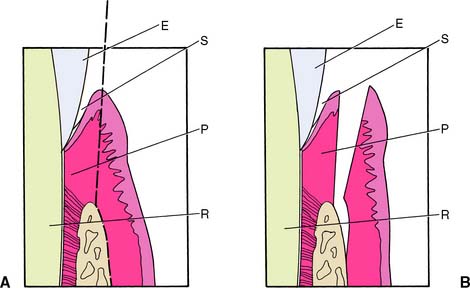
Fig. 5-15 Internal bevel incision. A, Ending on the bone, to allow reflection of the flap. B, Flap reflected. The supracrestal connective tissue and epithelium are to be removed. E, Enamel; P, supracrestal periodontium; R, root; S, sulcus.
(Redrawn from Carranza FA Jr: Glickman’s Clinical Periodontology, 7th ed. Philadelphia, WB Saunders, 1990.)
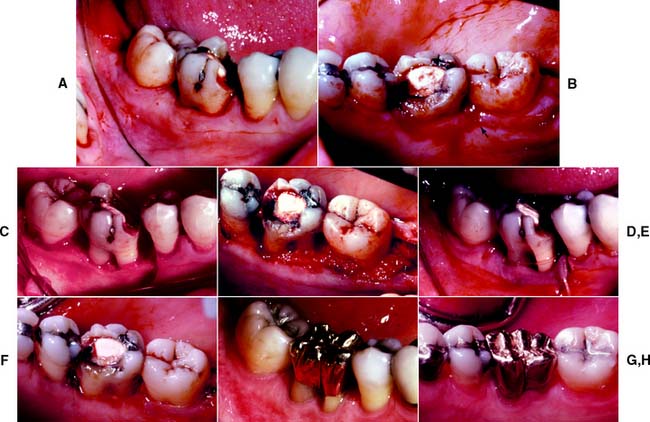
Fig. 5-16 Open débridement (modified Widman procedure). A, Initial thinning incision on the buccal surface for open débridement. B, Lingual flap thinned. C, Roots planed to remove subgingival accretions. D, Roots débrided and planed. E and F, Flaps coapted and sutured. G and H, The completed restoration, with a healthy periodontium.
Mucosal repair
Mucosal reparative surgery is used to increase the width of the band of keratinized gingiva. It is particularly useful when complete-coverage restorations are planned (see Chapter 6 for a more detailed discussion).
Hard tissue procedures
Hard tissue therapy is aimed at modifying the topography of areas where plaque control is difficult or impossible. Two examples are obvious:
Bone induction
Intrabony lesions (Fig. 5-17) are categorized as one-walled, two-walled, or three-walled, depending on the remaining osseous topography. The three-walled defect responds best to inductive or degranulation procedures, with resulting new attachment and resolution of all or part of the lesion. The one-walled and two-walled (crater) defects respond better to pocket elimination procedures.43
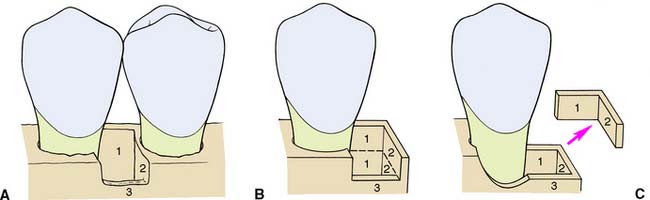
Fig. 5-17 Osseous defects. A, Three walls of bone present: at the lingual (1), distal (2), and buccal (3). B, Two walls of bone (1 and 2) in the coronal portion of the defect and three walls (1, 2, and 3) in the apical portion. C, The two coronal walls have been removed and the buccal surface of the bone recontoured, leaving the apical three-walled defect to fill with bone after degranulation.
(Redrawn from Carranza FA Jr: Glickman’s Clinical Periodontology, 7th ed. Philadelphia, WB Saunders, 1990.)
Many materials have been used to fill osseous defects: ceramic,44 sclera,45 cartilage,46 bone chips,47 cementum and dentin,48 osseous coagulum,49 freeze-dried bone,50 iliac crest marrow,51,52 hydroxylapatite,53 tricalcium phosphate,54 and bioactive glass materials.55,56 Laboratory and clinical studies with enamel matrix protein derivative have shown promising results in regenerating lost periodontium.57,58 Overall, results with these materials have been mixed, and no currently available alloplastic grafting material is clearly superior to any other in the regeneration of periodontal defects.
Technique
After the flaps have been reflected and the lesion has been thoroughly degranulated, the grafting material is packed firmly into the lesion until it is slightly overfilled. The flaps are then coapted, and interrupted sutures are placed (Fig. 5-18). A surgical dressing is applied and removed after 7 to 10 days.
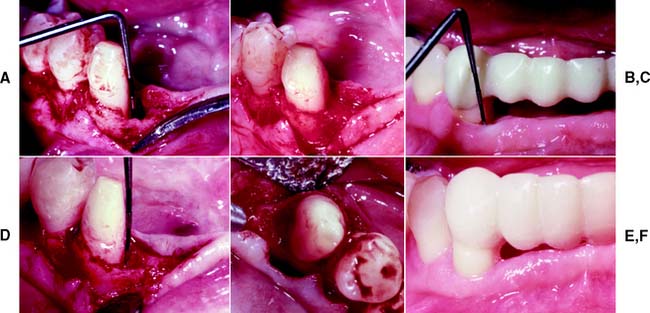
Fig. 5-18 Bone induction. A, Degranulation of a mesial defect on the mandibular right canine. This is a three-walled defect, with approximately 9 mm of intrabony lysis. B, The defect has been filled (slightly overfilled) with autogenous iliac crest marrow coagulum. C, Four months after surgery, the sulcular depth is approximately 3 mm. D, Osseous fill at reentry 1 year after surgery. Note the rim of bone at the margin of a previously existing defect (arrow). E, One year after surgery, there is a near-total fill of the defect. The rim of bone demarcates the margin of the previous intrabony lesion. F, Result of osseous grafting at the mesial of the canine 15 months after surgery. The gingival health and contours are excellent. Note the acrylic resin provisional restoration in place before the final restoration.
Osseous resection with apically positioned flaps
Chronic inflammatory periodontitis results in the loss of osseous tissue, destruction of osseous architecture, and creation of an intrabony lesion. The osseous tissue has no predictable or simple pattern of loss; the resorption may take the form of craters, hemiseptal defects, or well-like (troughlike) shapes. Craters in the interproximal areas (Fig. 5-19) are the most common type of lesion.4
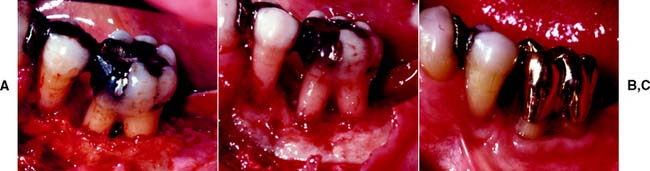
Fig. 5-19 Osseous resection. A, Osseous ledge and a crater defect. B, Osseous recontouring. C, Final restoration 3 months after apical positioning of the flap.
The objective of osseous resection is to shape the bone to form even contours. This is accomplished by leveling interproximal lesions, reducing osseous recontour lesions that are too wide and/or shallow for predictable repair or bony fill, thinning bony ledges, and eliminating or ramping crater defects. The result is intended to be a sound osseous base for gingival attachment and the elimination of pockets and excessive sulcular depth. Long-term studies59-61 have shown that although osseous resection surgery results in attachment loss and gingival recession, it is the most effective therapy for decreasing pocket depth, which can subsequently be maintained by the patient.
Technique
Before reflection of the flaps, the osseous topography of the lesion is assessed. After the area to be treated has been anesthetized, a periodontal probe is inserted into the pocket and forced through the epithelial attachment and connective tissue to the osseous crest. Multiple probings are made, and the surface morphology is observed. This “sounding” of the bone provides a reasonable representation of the width and depth of the lesion and is helpful in designing the incision.
Inverse bevel incisions are made on the buccal and lingual or palatal surfaces, and full-thickness mucoperiosteal flaps are reflected to expose the osseous tissue. After the flaps are thinned and the lesions are thoroughly degranulated, the roots of the teeth are planed vigorously. Osseous resection is then accomplished by the combination of rotary instrumentation with carbide and/or diamond burs, chisels, and bone files. When osteoplasty of the interproximal sluiceways, furcation areas, and buccal and lingual bone is completed, the flaps are positioned at the crest of the bone in an apical position on the tooth. Surgical dressings are applied, and in 7 to 10 days, the patient is seen again for suture removal and dressing removal or change.
Postsurgical healing
After surgery, the healing of the periodontium must be considered before any restorative procedures are performed. Initial connective tissue and epithelial healing is complete at 4 to 6 weeks. Final tissue maturation and sulcus reformation, however, may not be complete until 6 months to 1 year after surgery.
If the margins of the restorations are to be placed intrasulcularly (subgingivally) or at the gingival crest, or if gingival displacement procedures are to be used in making the impression, waiting as long as possible after surgery before attempting these procedures is recommended. If the restorative margins are to be placed at a suprasulcular (supragingival) position (which may not necessitate the use of a gingival displacement cord), these restorations may be started when the gingiva exhibits initial reepithelialization and a return to clinical health (approximately 4 to 6 weeks).
Treatment of furcation involvement
Diagnosis and treatment of furcation involvement of multiroot teeth is one of the more difficult problems encountered in the periodontal-restorative dentistry continuum. Familiarity with the furcation’s anatomic and morphologic variations is essential for formulating a treatment plan and prognosis for multiroot teeth.
Classification of involvements
Furcation involvements can be classified as Class (or Grade) I, II, III, and IV. Because these classifications are arbitrary, however, the reader should refer to periodontal textbooks and other readings5-7,62 for further detail and clarification.
The normal position of the osseous crest (Fig. 5-20) is approximately 1.5 mm apical to the cementoenamel junction (CEJ) in a young, healthy adult. Vertical loss of periodontal support that is less than 3 mm apical to the CEJ is considered Class I involvement (Fig. 5-21A). There is no gross or radiographic evidence of bone loss. Clinically, the furca can be probed up to 1 mm horizontally. Vertical loss greater than 3 mm but without involvement of the total horizontal width of the furcation is considered Class II involvement (Fig. 5-21B). A portion of the bone and periodontium remains intact, but osseous loss is evident on radiographs. The furca is penetrable more than 1 mm horizontally but does not extend through and through.
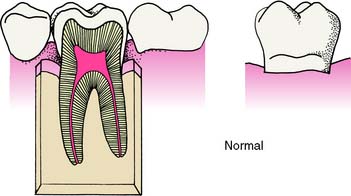
Fig. 5-20 Normal relationship of the cementoenamel junction and the osseous crest.
(From Baima RF: Considerations for furcation treatment. I. Diagnosis and treatment planning. J Prosthet Dent 56:138, 1986.)
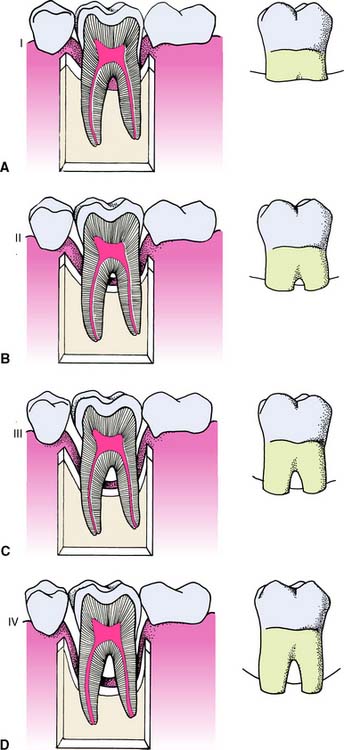
Fig. 5-21 Furcation involvements. A, Class I. B, Class II. C, Class III. D, Class IV.
(From Baima RF: Considerations for furcation treatment. I. Diagnosis and treatment planning. J Prosthet Dent 56:138, 1986.)
A horizontal through-and-through lesion that is occluded by gingiva but allows passage of an instrument from the buccal, lingual, or palatal surface is defined as a Class III involvement (Fig. 5-21C). The degree of osseous loss is grossly evident on radiographs. A horizontal through-and-through lesion that is not occluded by gingiva is defined as a Class IV involvement (Fig. 5-21D).
Review of root anatomy
The discussion of root anatomy is logically divided into subjects of maxillary and mandibular teeth.
Most maxillary molars have three roots—mesiobuccal, distobuccal, and palatal—although there may be variations, such as fused roots or fewer roots, particularly with second and third molars. The mesiobuccal root of most maxillary molars, especially the first molar, is usually biconcave and curves to the distal. The distobuccal root also is biconcave and somewhat less curved. The palatal root is wide buccolingually and mesiodistally, and it diverges palatally from the crown of the tooth. This configuration is unique to human dentition and may pose special problems in preparing, restoring, and designing restorations. The distobuccal and palatal roots tend to be in the same plane distally, and the distal furcation is more apical on the tooth than the is mesial furcation. In spite of this anatomy, the distal furca is more often involved in periodontal lesions than the mesial furca. From the apical perspective, a groove tends to unite the buccal and mesiopalatal openings of the trifurcation and can be probed when there is furcal involvement.
Most mandibular molars have two roots—mesial and distal—although, as with maxillary molars, there may be variations. The mesial root is flattened buccolingually, with concave surfaces on each proximal side. It curves distally, especially in first molars. The distal root is wider buccolingually than the mesial root and is concave on its mesial side. Its apex is often curved distally with a flat or convex distal aspect. Both root surfaces of mandibular molars facing the furca are concave, which results in an osseous chamber that is wider mesiodistally than either the buccal or the lingual furcation opening. The roof of the furcation is difficult to maintain because of mesiodistal bifurcation ridges.
Of importance is that maxillary and mandibular second and third molars often have more apically placed furcas than do first molars and often exhibit fused roots with little or no furcation.63-65
Maxillary premolars, particularly first premolars, and (at times) mandibular premolars also have furcations. However, because they are rarely amenable to treatment by odontoplasty-osteoplasty or root amputation procedures,5 they are not discussed here. Students should refer to oral anatomy and morphology textbooks66,67 for further clarification and study of molar root anatomy.
Odontoplasty-osteoplasty
Lesser degrees of furcation involvement can often be controlled by root planing and scaling, adequate oral hygiene, and/or gingivectomy-gingivoplasty. However, when the involvement is more extensive, recontouring of the tooth or bone may be necessary.
Class I and incipient Class II lesions (Fig. 5-22) can be treated by reflecting the soft tissue in the furcation area and recontouring both the tooth structure and the supporting bone to improve access for cleaning.4,68 Pocket elimination in this manner provides the best results and the fairest prognosis. A minimal amount of tooth structure and bone is lost, and the patient can easily maintain it.
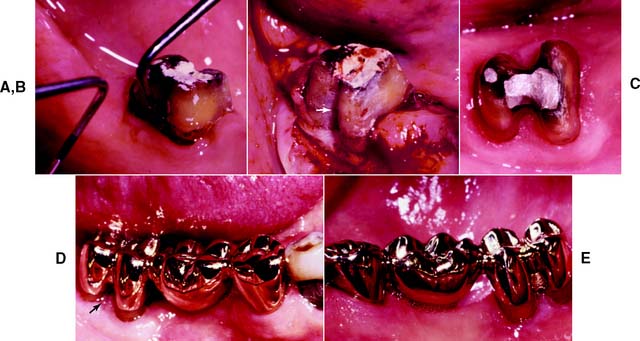
Fig. 5-22 Treatment of a Class II furcation lesion. A, The periodontal probe discloses approximately 3 mm of horizontal involvement. B, The lesion is reduced to Class I by odontoplasty-osteoplasty. Note the contours of the tooth at the coronal portion of the buccal furcation (arrow). C, Preparations for a fixed dental prosthesis to be placed in the right mandibular quadrant. Note the figure-8 shape of the molar preparation. D, Final restoration of the molar. There is excellent gingival health in the furcation area (arrow). E, Restoration of the quadrant. Note the slight contact of pontic on ridge and the open embrasures for access by oral hygiene instruments.
(Courtesy of Dr. H. J. Gulbransen.)
Class II and Class III involvements can be treated by a procedure known as tunneling.6,7 The osseous structure is completely removed in the furcation, which converts the lesion to a through-and-through defect. Teeth suitable for tunneling must have long, divergent roots, which will facilitate penetration by an oral hygiene aid (e.g., a proximal brush or a pipe cleaner). Patient selection is particularly important, because oral hygiene and patient motivation are crucial. Failure to maintain the furcation in a relatively plaque-free state may lead to caries, which are often impossible to correct. The common location of accessory canals in the roof of the furca can also be a problem. Because of irreversible pulp damage, endodontic treatment may be needed at a later date.63,64,68-70
Root amputation
In many patients, Class II and Class III furcation lesions are most effectively treated by root amputation (Fig. 5-23), which eliminates the furcation completely. The indications are as follows5,7,68,71-73:
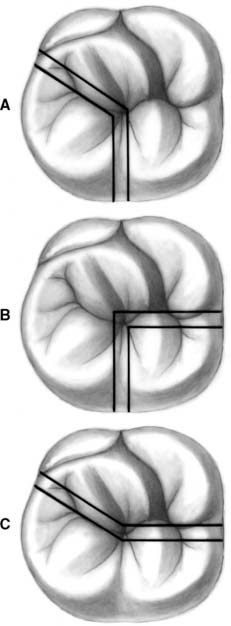
Fig. 5-23 Types of root amputation. A, Mesiobuccal. B, Distobuccal. C, Palatal or mesiobuccal and distobuccal.
Certain roots are not suitable for amputation. Individual considerations include the extent of furcation involvement, the anatomy and topography of the supporting bone, the anatomy of the root canal, and the periapical health of the tooth. The major contraindications to root resection are teeth exhibiting any of the following5,69,73:
Before the gingiva is reflected, the furca is probed with a curved furcation instrument so that the precise location of the bur cut can be determined (Fig. 5-24A). The cut is then made over the center of the furca but slightly toward the root to be removed. This will protect the residual root and/or tooth body. Whenever possible, the cut should be made before the flap is reflected so that the field is cleaner when the osseous tissue is exposed. When the cut is made into the root to be removed, the operator is able to inspect the residual root and remaining furcation area. A lip is often created in the furcation area, however (Fig. 5-24B), and after the root to be extracted has been delivered, the furcation lip is removed, and the tooth is finally contoured and finished. Removing the lip from the root of the furca is crucial to the treatment’s success. If this is not done, the osseous tissue will not be recontoured properly (Figs. 5-24C and D), plaque control will be impaired, and, in effect, the furca will still be present.4,73,74
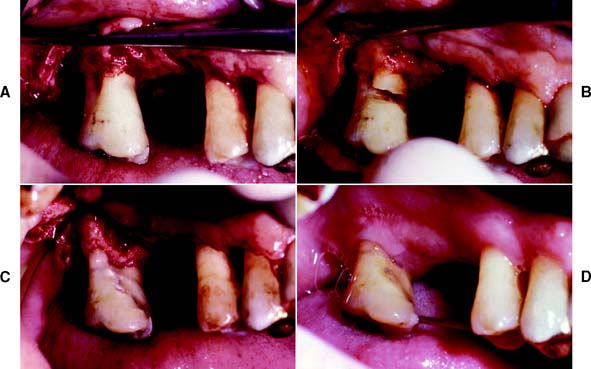
Fig. 5-24 Mesiobuccal root amputation. A, A full-thickness flap has been reflected to reveal Class I buccal furcation involvement and a Class II lesion in the mesiopalatal furca. B, The mesiobuccal root is sectioned at approximately 45 degrees to the tooth trunk. The section has been made into the root that is to be removed, and the result is a lip at the buccal furca. C, Final osseous contours after removal of the mesiobuccal root and osteoplasty-ostectomy. The furcation lip has also been removed. D, Mesiobuccal root amputation, 2 months after surgery. The remaining tooth structure is stabilized with a wire-and-acrylic resin provisional splint.
There are few surgical problems with root resection. The ones most frequently encountered are fracture of the root75 and loss of a root tip in the maxillary sinus.76,77 Osseous anatomic features such as a flat mandibular shelf and a flat palatal area can make access to the surgical site difficult and may complicate flap placement. Root proximity may complicate flap placement. Root proximity can pose a problem for separation and removal of the sectioned fragment from the surgical site. Mucogingival anatomy must be considered, because any flap procedure is contraindicated if there is a lack of keratinized attached gingiva.
Hemisection4,71-74
Hemisection means cutting a tooth in half. In the case of mandibular molars, hemisection is followed by removal and subsequent restoration of one root or restoration of each half of the tooth. The latter procedure is sometimes called premolarization or bicuspidization.74
The technical procedures of hemisection and root amputation are similar (Fig. 5-25). If one hemisected root is to be extracted, osteoplasty-osteoectomy and removal of the furcation lip are performed as previously described (Fig. 5-26). If the roots are to be maintained and restored separately, the furcation lips must be removed from each root. The individual roots may then be separated orthodontically, if necessary, to gain new interseptal osseous area.68,78
Provisionalization
Provisional stabilization is indicated in many cases of root resection to allow proper healing of the surgical site before definitive restorations are placed and to stabilize the remaining tooth structure against masticatory forces63,79 (Fig. 5-27).
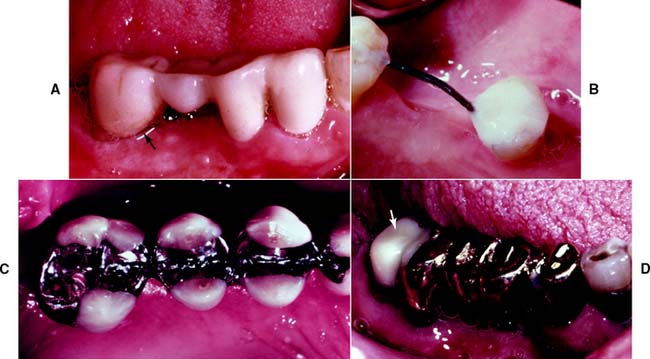
Fig. 5-27 Provisional restorations. A, Acrylic resin with an overcontoured area corresponding to the mesial root of the mandibular right second molar. Ideal contouring of such a provisional would remove excess resin where the root had been amputated (arrow). B, Wire-and-acrylic resin splint stabilizing the mandibular right quadrant. C, Wire-and-amalgam splint. D, Existing restoration lined with acrylic resin (arrow). This can serve adequately as a provisional restoration.
(A, Courtesy of Dr. S. B. Ross; C, courtesy of Dr. K. G. Palcanis; D, courtesy of Dr. H. J. Gulbransen.)
Normally, an acrylic resin provisional restoration (Fig. 5-27A) is provided (as described in Chapter 15 ), although on occasion an existing restoration can be successfully modified as a provisional (see Fig. 5-27D). Acid-etch retained composite resin or amalgam with orthodontic wire (see Fig. 5-27B and C) can also be used on an interim basis to maintain space and stabilize remaining tooth structure.
Restoration
Teeth with a resected root or roots may be restored in a variety of ways.60,69,76-78 They may be involved in a treatment plan as single units, as fixed or removable dental prosthesis abutments, or as vertical stops for an overdenture.
The most common types of restorations for teeth with resected roots involve the following:
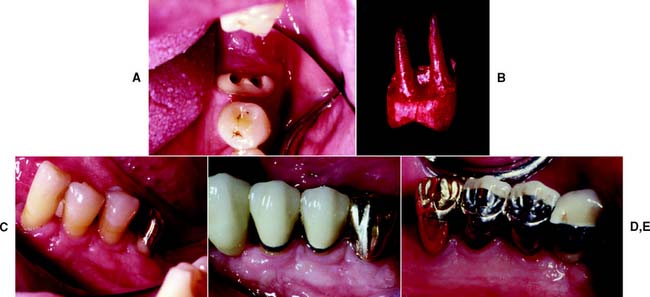
Fig. 5-28 Mesial root of a mandibular left first molar prepared for a single crown restoration. A, Canals have been made parallel for a dowel and core. B, Casting with parallel dowels. C, Dowel and core restoration have been cemented, and the root has been prepared for a single crown. D and E, Single nonsplinted restoration of the mesial root of a mandibular left first molar.
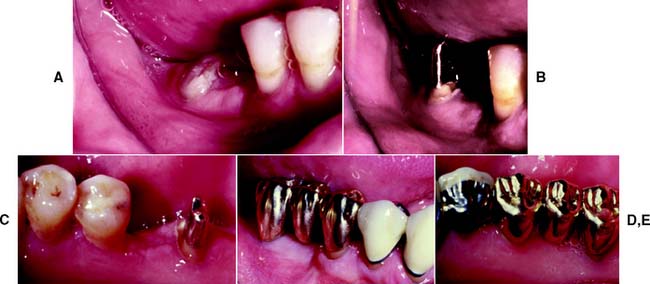
Fig. 5-29 Distal root of a mandibular right first molar prepared for a dowel and core restoration. A, The root will be used as an abutment for a fixed dental prosthesis. B and C, Dowel and core restoration of the root. D, Final restoration, with the root used as the distal abutment for a fixed dental prosthesis. Note the excellent gingival health and contours. E, Final restoration of the mandibular right quadrant, lingual view. The point contact of the totally convex pontic and the wide embrasure spaces allow optimum oral hygiene and excellent gingival health.
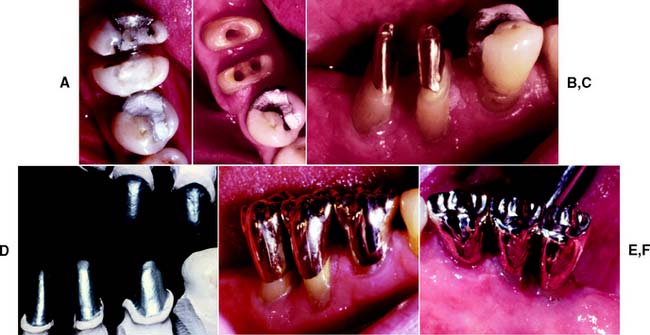
Fig. 5-30 Premolarization. Mesial and distal roots of a mandibular right first molar after hemisection. A, A wire-and-acrylic resin interim prosthesis is in place. B, The mesial and distal roots have been prepared for a dowel and core. Each will be restored as an individual premolar. Note that the distal root has been moved (orthodontically) 4 mm distally before the restoration was fabricated, to provide room in the newly created interproximal area for the dowel and core and crown restorations. C, The dowel and cores in place. Note the space between the roots created by the orthodontic movement. D, Dies with die relief placed and mounted on a suitable articulator for fabrication of the final restoration. E and F, Final result. The open interproximal areas and flat emergence profiles from the gingival area allow optimum oral hygiene and assist in the preservation of gingival health.
Evaluation of Surgical Therapy
The prognosis for a tooth whose root or roots have been resected and/or amputated depends on many factors. The manner in which the tooth is to be used in the restorative plan—as an abutment for a partial dental prosthesis or as a single crown—has a bearing on prognosis.7,71 The amount of residual osseous structure to support the remaining tooth also influences the outlook. Most important, however, are the motivation and oral hygiene of the patient. Long-term studies accounting for all of these factors have yielded results ranging from 4% to 38% loss of residual roots with up to 53 years of postsurgical service.68,84,85 With careful diagnosis, treatment planning, and good surgical technique, the tooth with resected roots may have a favorable prognosis. Plaque control is critical. For this reason, the patient makes the final determination about whether the tooth will ultimately be lost or remain as a healthy functioning unit in the dentition.
Guided Tissue Regeneration (Hard and Soft Tissue Procedures)
It has long been a goal of periodontal therapists to replace lost connective tissue attachment and bone. As previously described, many materials have been used in the quest for reattachment to diseased root surfaces. In the recent past, regaining lost attachment with cells from the host has been successful. Through the use of physical barriers that prevent cells from the gingival connective tissue and apically migrating oral epithelium from contacting the root surface, space is created over the root surface, which allows selective repopulation of this space by cells from the residual PDL. These cells become the regenerated PDL.86,87
Several types of barriers, both resorbable and nonresorbable,88-92 as well as native periosteum,93 have been used to regenerate the periodontium about root surfaces,94 in furcations,95-98 and with dental implants.99-101 The most significant evidence has been attained by the use of a nonresorbable, polytetrafluoroethylene (PTFE) barrier (Gore-Tex Periodontal Material). Although long-term follow-up results are not conclusive, coronal movement of the connective tissue attachment has been impressive in many clinical and laboratory investigations. Although guided tissue regeneration is a technique-sensitive mode of therapy and has yet to be viewed as widely successful, it may prove to be the most promising approach to regeneration.
Technique
After diagnosis of the lesion and any initial therapy deemed appropriate, full-thickness flaps are reflected in an attempt to maintain the maximum amount of tissue for coverage of the barrier (Figs. 5-31 and 5-32). The lesion is completely débrided of granulation tissue, and the roots are planed thoroughly.
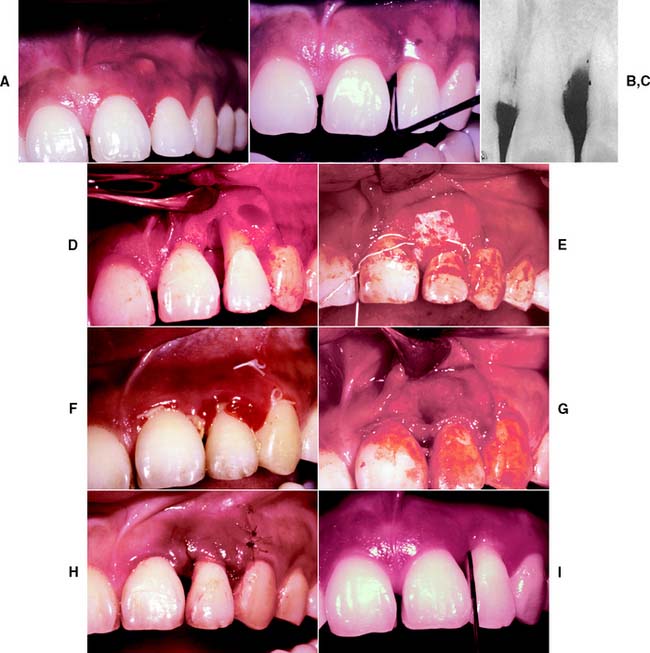
Fig. 5-31 Guided tissue regeneration about an anterior tooth. A, Abscess at the mesial of the left lateral incisor. B, After initial débridement and 3 days of antibiotic therapy. Probing depth is 8 mm. C, Radiograph taken at the time of barrier placement. D, The mesial surface after degranulation. Note the degree of bone loss. E, The polytetrafluoroethylene (PTFE) barrier placed at the cementoenamel junction (CEJ) completely covers the defect. F, Healing at 5 weeks. Note the new connective tissue coronal to the barrier and the CEJ. G, When the barrier is removed, the new connective tissue can be seen at the mesial and buccal surfaces. H, Healing 10 days after barrier removal. I, Healing at 9 months. Note the minimal sulcular depth with excellent tissue health. There is slight recession of the CEJ.
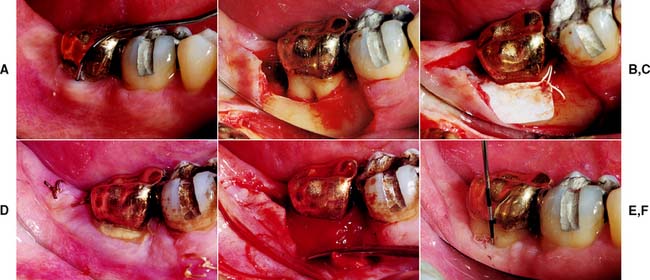
Fig. 5-32 Guided tissue regeneration in a furcation defect. A, Buccal aspect of a mandibular second molar showing the defect. A curved furcation probe reveals 6 mm of vertical bone loss and 3 mm of horizontal loss. B, With reflection of the flap, the Class II defect can be seen. C, Polytetrafluoroethylene (PTFE) barrier in place at the CEJ. D, Healing at 6 weeks. Note the recession of the flap at the coronal surface of the barrier. E, After barrier removal. Note the new connective tissue apical to the margin of the gold crown. F, Healing at 10 weeks. Despite minimum pocket depth, some loss of connective tissue is apparent.
The barrier is placed at the CEJ and secured with sutures placed in a suspensory (sling)–type manner, maintaining a position covering the entire root surface. The full-thickness flap is mobilized to cover the entire surface of the barrier in both an apicocoronal and a mesiodistal direction. Antibiotic coverage and an antibacterial mouth rinse may be prescribed for the postoperative interval. Weekly monitoring for possible infection is recommended.
After a healing period of 4 to 6 weeks, a full-thickness flap is again reflected and is teased away from the external portion of the barrier. The barrier is then carefully removed to reveal a glossy and very vascular surface of new connective tissue. After the internal surface of the flap is stripped of epithelium by either sharp or rotary excision, the flap is placed to cover the entire surface of the new connective tissue. A periodontal dressing and systemic antibiotics or antibacterial mouth rinse may be used at the operator’s discretion.
Clinical trials in humans102-104 have favorably demonstrated the use of calcium sulfate (plaster of Paris) as a resorbable barrier. In addition to a significantly reduced cost in comparison with a PTFE barrier, the main advantage of this type of barrier is that the desired guided tissue regeneration may be accomplished without the need for a second surgical procedure. The techniques of flap reflection, degranulation of the defect or defects, and wound closure are similar to those used in other barriers (Fig. 5-33), with primary wound closure over the barrier being the main objective of surgery.
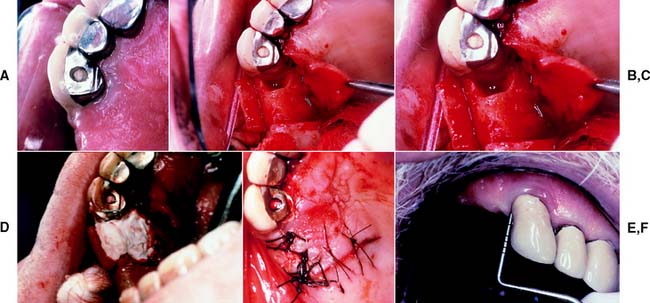
Fig. 5-33 Guided tissue regeneration with a resorbable calcium sulfate barrier. A, Preoperative view of the maxillary right canine to be restored with a new crown. B, Flaps reflected and granulomatous tissue removed from the defect at the distal surface. The three-walled intrabony defect prepared for barrier placement. C, Defect is filled with dense calcium sulfate graft. D, Calcium sulfate slurry placed to act as a barrier and facilitate guided tissue regeneration. E, Flap sutured to place. F, Minimal probing depths before final restoration. Favorable tissue contours after the elimination of the intrabony defect.
(Courtesy of Dr. V. Ng.)
Restoration
After the completion of guided tissue regeneration procedures, a period of healing that depends on the restorative needs of the patient is necessary. As a general guideline, 6 to 8 weeks should be allowed before displacement cord is used in the sulcus; this allows tissue maturation. The sub-sequent restorative procedures are accomplished as described earlier.
Maintenance
Continued reexamination and evaluation of periodontal status is an extension of active periodontal therapy performed at selective intervals to assist the periodontal patient in maintaining oral health. Of particular importance is the identification of areas where oral hygiene measures are partially effective or ineffective. The patient and the dentist must work together to preserve the health of the soft and hard tissues and prevent further periodontal breakdown or the recurrence of active disease. In this manner, recurrence and progression of disease and incidence of tooth loss may be minimized, and treatment of any recurrent disease may be offered in a timely manner.105
There is no standard maintenance schedule for patients requiring periodontal therapy. Some should be recalled only at 5- to 6-month intervals; others should be seen by the dentist (or periodontist) and the hygienist every 2 or 3 months. The maintenance regimen varies greatly among individuals, requires close coordination between the patient and the involved professionals, and should be tailored to the needs of each patient.106
Prognosis
The progress, course, and outcome of gingival and periodontal disease are crucially dependent on the patient. Without the ability and desire of the patient to maintain his or her teeth and periodontium, any treatment will ultimately fail. Determining a prognosis for the teeth and periodontium debilitated from moderate disease is therefore quite difficult. Unfortunately, failure is often the best teacher for patients.
There are many factors involved when the dentist attempts to arrive at a prognosis for a tooth or an arch. With optimal intentions and the best technique, a favorable result can be expected (even in the absence of good host resistance). Without them, treatment is doomed to ultimate failure. The age of the patient may help in predicting the success or failure of the treatment. In general, the prognosis is better for an older patient with a given amount of lost bone or tissue attachment than for a younger one. Many older individuals are more resistant to disease, and the disease has less effect. The amount of residual alveolar bone, the number of remaining teeth and their overall condition, any tooth mobility, and the patient’s general occlusion and systemic integrity all can influence the outcome of therapy. There is substantial evidence to suggest that smoking has a debilitating effect on the periodontium.107-109 Therapy for cessation of smoking may be incorporated into the comprehensive treatment plan for the patient in order to enhance the long-term prognosis of the dentition.
Also important to the long-term stability and function of the dentition are the condition of the arches to be restored and the ability of the restorative dentist to execute complex treatment plans without iatrogenically disturbing the gingiva and periodontium. This is a delicate undertaking and adversely affects a periodontally unstable arch if not skillfully performed.
SUMMARY
The periodontium is the most important anatomic structure of the oral cavity in fixed prosthodontics. Its main component, the PDL, anchors the teeth in the alveolar processes and provides attachment, nutrition, tissue synthesis and resorption, and mechanoreception. The practitioner embarking on a restorative program must therefore first make an accurate periodontal diagnosis and then institute effective treatment of any periodontal disease—whose main etiologic factor is neglected accumulations of plaque. Allowing the proper time for healing after periodontal surgery is also very important. The healing time required depends on the design of the restoration and is crucial to the correct placement of restorations near the free gingival margin.
 GLOSSARY*
GLOSSARY*
al·ve·o·lar \ăl-vē′a-ler\ adj (1799): that part of the jaws where the teeth arise
alveolar bone \ăl-vē′a-ler bōn\: the bony portion of the mandible or maxillae in which the roots of the teeth are held by fibers of the periodontal ligament—called also dental alveolus
alveolar mucosa \ăl-vē′a-ler myōō-kō′sa\: the fixed mucosal covering of the alveolar process, loosely attached to the bone
attached gingival \a-tăchd′ j n′ja-va, j
n′ja-va, j n′jī-\: the portion of the gingiva that is firm, dense, stippled, and tightly bound to the underlying periosteum, bone, and tooth
n′jī-\: the portion of the gingiva that is firm, dense, stippled, and tightly bound to the underlying periosteum, bone, and tooth
attachment apparatus \a-tăch′ment ăp′a-rā′tus, -răt′us\: in periodontics, a general term used to designate the cementum, periodontal ligament and alveolar bone
bi·fur·ca·tion \bī′fur-kā′shu\ n (1615): 1: division into two branches 2: the site where a single structure divides into two parts, as in two roots of a tooth
biologic width \bī′a-l j′
j′ -kal\ (1998): the combined width of connective tissue and junctional epithelial attachment formed adjacent to a tooth and superior to the crestal bone
-kal\ (1998): the combined width of connective tissue and junctional epithelial attachment formed adjacent to a tooth and superior to the crestal bone
Gargiulo, AW et al J Periodontology 1961; 32:261–267
Cohen, DW. Lecture at Walter Reed Army Medical Center, June 3, 1962
buccal vestibule \bŭk′al v s′ti-byōōl\: the portion of the oral cavity that is bounded on one side by the teeth, gingiva, and alveolar ridge (in the edentulous mouth, the residual ridge) and on the lateral side by the cheek posterior to the buccal frenula
s′ti-byōōl\: the portion of the oral cavity that is bounded on one side by the teeth, gingiva, and alveolar ridge (in the edentulous mouth, the residual ridge) and on the lateral side by the cheek posterior to the buccal frenula
calcium sulfate \kăl′sē-um sŭl′fāt′\: a product obtained by calcination of gypsum under steam pressure. The alpha form is composed of regularly shaped grains, with low porosity, and requiring little water for a satisfactory mix. Forms include alpha, alpha-modified and beta.
cemento-enamel junction \s ′m
′m n-tō
n-tō  -năm′al jŭngk′ shun\: that area where the enamel and cementum meet at the cervical region of a tooth
-năm′al jŭngk′ shun\: that area where the enamel and cementum meet at the cervical region of a tooth
ce·men·tum \s -m
-m n′tum\ n (1842): the thin calcified tissue of ectomesenchymal origin that covers the root of a tooth
n′tum\ n (1842): the thin calcified tissue of ectomesenchymal origin that covers the root of a tooth
connective tissue \ka-n k′t
k′t v t
v t sh′ōō\: a tissue of mesodermal origin rich in interlacing processes that supports or binds together other tissues
sh′ōō\: a tissue of mesodermal origin rich in interlacing processes that supports or binds together other tissues
crevicular epithelium \kr ′v
′v -kyōō′lar
-kyōō′lar  p′a-thē′lē-um\: the non-keratinized epithelium of the gingival crevice
p′a-thē′lē-um\: the non-keratinized epithelium of the gingival crevice
crown-root ratio \kroun-rōōt rā′shō, rā′shē-ō′\: the physical relationship between the portion of the tooth within alveolar bone compared with the portion not within the alveolar bone, as determined by radiograph
de·bride·ment \dā′brēd-mäN′, d -brēd′ment\ n (ca. 1842): the removal of inflamed, devitalized, contaminated tissue or foreign material from or adjacent to a lesion
-brēd′ment\ n (ca. 1842): the removal of inflamed, devitalized, contaminated tissue or foreign material from or adjacent to a lesion
donor site \dō′ner sīt\: an area of the body from which a graft is taken
eccentric \ k-s
k-s n′tr
n′tr k\ adj (14c): 1: not having the same center 2: deviating from a circular path 3: located elsewhere than at the geometric center 4: any position of the mandible other than that which is its normal position
k\ adj (14c): 1: not having the same center 2: deviating from a circular path 3: located elsewhere than at the geometric center 4: any position of the mandible other than that which is its normal position
etiologic factors \ē′tē-ō-l j′
j′ k făk′tors\: the elements or influences that can be assigned as the cause or reason for a disease or lesion—see LOCAL E.F., SYSTEMIC E.F.
k făk′tors\: the elements or influences that can be assigned as the cause or reason for a disease or lesion—see LOCAL E.F., SYSTEMIC E.F.
free gingival \frē j n′ja-va\: 1: the part of the gingiva that surrounds the tooth and is not directly attached to the tooth surface
n′ja-va\: 1: the part of the gingiva that surrounds the tooth and is not directly attached to the tooth surface
free gingival margin \frē j n′ja-val mär′j
n′ja-val mär′j n\: the unattached gingiva surrounding the teeth in a collar-like fashion and demarcated from the attached gingiva by a shallow linear depression, termed the free gingival groove
n\: the unattached gingiva surrounding the teeth in a collar-like fashion and demarcated from the attached gingiva by a shallow linear depression, termed the free gingival groove
frem·i·tus \fr m′
m′ -tus\ n (1879): a vibration perceptible on palpation; in dentistry, a vibration palpable when the teeth come into contact
-tus\ n (1879): a vibration perceptible on palpation; in dentistry, a vibration palpable when the teeth come into contact
frenectomy: surgical excision of a frenum
fur·ca·tion \fûr′kā′shun\ n: the anatomic region of a multirooted tooth where the roots diverge
gin·gi·va \j n′ja-va, j
n′ja-va, j n-j
n-j -\ n, pl –vae \-vē′\: the fibrous investing tissue, covered by epithelium, which immediately surrounds a tooth and is contiguous with its periodontal membrane and with the mucosal tissues of the mouth—see ATTACHED G., FREE G., MARGINAL G.
-\ n, pl –vae \-vē′\: the fibrous investing tissue, covered by epithelium, which immediately surrounds a tooth and is contiguous with its periodontal membrane and with the mucosal tissues of the mouth—see ATTACHED G., FREE G., MARGINAL G.
gingival crevice \j n′ja-val kr
n′ja-val kr v′
v′ s\: a shallow fissure between the marginal gingiva and the enamel or cementum. It is bounded by the tooth surface on one side, the crevicular epithelium on the other, and the coronal end of the junctional epithelium at its most apical point—called also gingival sulcus
s\: a shallow fissure between the marginal gingiva and the enamel or cementum. It is bounded by the tooth surface on one side, the crevicular epithelium on the other, and the coronal end of the junctional epithelium at its most apical point—called also gingival sulcus
guided tissue regeneration \gī′d d t
d t sh′ōō r
sh′ōō r -j
-j n′a-rā′shun\: any procedure that attempts to regenerate lost periodontal structures or alveolar process through differential tissue responses. Barrier techniques, using synthetic materials that may or may not resorb, to exclude epithelial ingrowth (periodontal regeneration) or connective tissue ingrowth (alveolar process regeneration) that is believed to interfere with regeneration
n′a-rā′shun\: any procedure that attempts to regenerate lost periodontal structures or alveolar process through differential tissue responses. Barrier techniques, using synthetic materials that may or may not resorb, to exclude epithelial ingrowth (periodontal regeneration) or connective tissue ingrowth (alveolar process regeneration) that is believed to interfere with regeneration
hemi·sec·tion \h m′
m′ -s
-s k′shun\ n: the surgical separation of a multirooted tooth, especially a mandibular molar, through the furcation in such a way that a root and the associated portion of the crown may be removed
k′shun\ n: the surgical separation of a multirooted tooth, especially a mandibular molar, through the furcation in such a way that a root and the associated portion of the crown may be removed
interdental papilla \ n′ter-d
n′ter-d n′tl pa-p
n′tl pa-p l′a\: a projection of the gingiva filling the space between the proximal surfaces of two adjacent teeth
l′a\: a projection of the gingiva filling the space between the proximal surfaces of two adjacent teeth
junctional epithelium \jŭngk′shun-al  p′a-thē′lē-um\: a single or multi-layer of non-keratinizing cells adhering to the tooth surface at the base of the gingival crevice—called also epithelial attachment
p′a-thē′lē-um\: a single or multi-layer of non-keratinizing cells adhering to the tooth surface at the base of the gingival crevice—called also epithelial attachment
ker·a·tin·i·za·tion \k r′a-t
r′a-t n-
n- -zā-shun\ n: the process of maturation of keratinocytes. The formation of a protein layer (keratin) on the surface of some epithelia
-zā-shun\ n: the process of maturation of keratinocytes. The formation of a protein layer (keratin) on the surface of some epithelia
keratinized gingival \k r′a-t
r′a-t -nīzd j
-nīzd j n′ja-va\: the oral surface of the gingiva extending from the mucogingival junction to the gingival margin. In gingival health, the coronal portion of the sulcular epithelium may also be keratinized. The pattern of keratinization may be ortho-or para-
n′ja-va\: the oral surface of the gingiva extending from the mucogingival junction to the gingival margin. In gingival health, the coronal portion of the sulcular epithelium may also be keratinized. The pattern of keratinization may be ortho-or para-
lengthening of the clinical crown \l ngk′tha-n
ngk′tha-n ng ŭv tha kl
ng ŭv tha kl n′
n′ -kal kroun\: a surgical procedure designed to increase the extent of supragingival tooth structure for restorative or esthetic purposes by apically positioning the gingival margin, removing supporting bone, or both
-kal kroun\: a surgical procedure designed to increase the extent of supragingival tooth structure for restorative or esthetic purposes by apically positioning the gingival margin, removing supporting bone, or both
marginal gingiva \mär′ja-nal j n′ja-va\: the most coronal portion of the gingiva; often used to refer to the free gingiva that forms the wall of the gingival crevice in health
n′ja-va\: the most coronal portion of the gingiva; often used to refer to the free gingiva that forms the wall of the gingival crevice in health
mo·bile \mō′bal, -bēl, -bīl\ adj (15c): capable of moving or being moved; movable
mucogingival junction \myōō′kō-j n′ja-val, j
n′ja-val, j n′jī-\: the junction of gingiva and alveolar mucosa
n′jī-\: the junction of gingiva and alveolar mucosa
mu·co·peri·os·te·um \myōō′kō-p r′ē-
r′ē- s′tē-am\ n: a term synonymous with a full-thickness flap implying the inclusion of both mucosa and periosteum during flap elevation
s′tē-am\ n: a term synonymous with a full-thickness flap implying the inclusion of both mucosa and periosteum during flap elevation
mu·co·sa \myōō-kō′sa\ n (1880): a mucous membrane comprised of epithelium, basement membrane, and lamina propria—see ALVEOLAR M., ORAL M.
o·don·to·plasty \ō-d n′ta-plăs-tē\: the reshaping of a portion of a tooth
n′ta-plăs-tē\: the reshaping of a portion of a tooth
oral mucosa \ôr′al, ōr′- myōō-kō′sa\: the lining of the oral cavity
per·i·o·don·tal \p r′ē-ō-d
r′ē-ō-d n′tl\ adj: pertaining to or occurring around a tooth
n′tl\ adj: pertaining to or occurring around a tooth
primary occlusal trauma \prī′m r′ē a-klōō′al trou′ma\: the effects induced by abnormal or excessive occlusal forces acting on teeth with normal periodontal support
r′ē a-klōō′al trou′ma\: the effects induced by abnormal or excessive occlusal forces acting on teeth with normal periodontal support
quadrant \kw d′rant\ n (15c): 1: any of the four quarters into which something is divided by two real or imaginary lines that intersect each other at right angles 2: in dentistry, one of the four sections of the dental arches, divided at the midline—see also SEXTANT, OCTANT
d′rant\ n (15c): 1: any of the four quarters into which something is divided by two real or imaginary lines that intersect each other at right angles 2: in dentistry, one of the four sections of the dental arches, divided at the midline—see also SEXTANT, OCTANT
re·at·tach·ment \rē-a-tăch′mant\ n: in periodontics, the reunion of epithelial and connective tissues with root surfaces and bone such as occurs after incision or injury
re·flec·tion \r -fl
-fl k′shun\ (14c) n: 1: the elevation and folding back of all or part of the mucosa to expose underlying structures 2: the return of light or sound waves from a surface
k′shun\ (14c) n: 1: the elevation and folding back of all or part of the mucosa to expose underlying structures 2: the return of light or sound waves from a surface
repositioning splint \rē-pa-z sh′a-n
sh′a-n ng spl
ng spl nt\ n: an intraoral maxillofacial prosthesis constructed to temporarily or permanently alter the relative position of the mandible to the maxillae
nt\ n: an intraoral maxillofacial prosthesis constructed to temporarily or permanently alter the relative position of the mandible to the maxillae
secondary occlusal trauma \s k′an-d
k′an-d r-ē a-klōō′zal\: the effects induced by occlusal force (normal or abnormal) acting on teeth with decreased periodontal support
r-ē a-klōō′zal\: the effects induced by occlusal force (normal or abnormal) acting on teeth with decreased periodontal support
split-thickness graft \spl t-th
t-th k′n
k′n s grăft\: a transplant of skin or mucous membrane consisting of epithelium and a portion of the dermis
s grăft\: a transplant of skin or mucous membrane consisting of epithelium and a portion of the dermis
stip·ple \st p′al\ vt stip·pled \st
p′al\ vt stip·pled \st ′puld\ stip·pling \st
′puld\ stip·pling \st p′l
p′l ng\ (1760): 1: to engrave by means of dots or/and flicks 2: to make small short touches that together produce an even or softly graded shadow 3: to speckle or fleck—stip·pler \st
ng\ (1760): 1: to engrave by means of dots or/and flicks 2: to make small short touches that together produce an even or softly graded shadow 3: to speckle or fleck—stip·pler \st p′lar\ n
p′lar\ n
STUDY QUESTIONS
1 Bhaskar SN. Orban’s Oral Histology and Embryology, 11th ed. St. Louis: Mosby, 1991.
2 Bowers GM. A study of the width of the attached gingiva. J Periodontol. 1963;34:210.
3 Ainamo J, Loe H. Anatomic characteristics of gingiva: a clinical and microscopic study of the free and attached gingiva. J Periodontol. 1966;37:5.
4 Newman MG, Takei H, Carranza FAJr. Clinical Periodontology, 9th ed. Philadelphia: WB Saunders, 2000.
5 Lindhe J. Textbook of Clinical Periodontology. Copenhagen: Munksgaard, 1989.
6 Schluger S, et al. Periodontal Disease: Basic Phenomena, Clinical Management, and Occlusal and Restorative Interrelationships, 2nd ed. Philadelphia: Lea & Febiger, 1990.
7 American Academy of Periodontology. The pathogenesis of periodontal diseases. J Periodontol. 1999;70:457.
8 Shafer WG, et al. A Textbook of Oral Pathology, 4th ed. Philadelphia: WB Saunders, 1983.
9 Thomas NG. Elastic fibers in periodontal membrane and pulp. J Dent Res. 1965;7:325.
10 Fullmer HM. A critique of normal connective tissues of the periodontium and some alterations with periodontal disease. J Dent Res. 1962;41(suppl 1):223.
11 Schroeder HE, Listgarten MA. Fine structure of the developing epithelial attachment of human teeth. Monogr Dev Biol. 1971;2:1.
12 Orban B, Kohler J. The physiologic gingival sulcus. Z Stomatol. 1924;22:353.
13 National Institute for Dental Research. Oral Health of United States Adults: The National Survey of Oral Health in U.S. Employed Adults and Seniors: 1985-1986 (NIH Publication No. 87-2868). Bethesda, Md: U.S. Department of Health and Human Services, Public Health Services, 1987.
14 Schwartz RS, Massler M. Tooth accumulated materials: a review and classification. J Periodontol. 1969;40:407.
15 Mandel ID. Dental plaque: nature, formation, and effects. J Periodontol. 1966;37:357.
16 Loe HE, et al. Experimental gingivitis in man. J Periodontol. 1965;36:177.
17 Newman HN. Calcium, matrix polymers, and plaque formation. J Periodontol. 1982;53:101.
18 Ritz HL. Microbial population shifts in developing human dental plaque. Arch Oral Biol. 1967;12:1561.
19 Slots J, et al. Microbiota of gingivitis in man. Scand J Dent Res. 1978;86:174.
20 Allen D, Kerr D. Tissue response in the guinea pig to sterile and non-sterile calculus. J Periodontol. 1965;36:121.
21 Wolff L, et al. Bacteria as risk markers for periodontitis. J Periodontol. 1994;65:498.
22 Page RC, Schroeder HE. Pathogenesis of inflammatory periodontal disease: a summary of current work. Lab Invest. 1976;34:235.
23 American Academy of Periodontology. Parameter on comprehensive periodontal examination. J Periodontol. 2000;71:847.
24 American Academy of Periodontology. Diagnosis of periodontal diseases. J Periodontol. 2003;74:1237.
25 Armitage GC. Clinical evaluation of periodontal diseases. Periodontol 2000. 1996;7:39.
26 Lang NP, et al. Monitoring disease supportive periodontal treatment by bleeding on probing. Periodontol 2000. 1996;12:44.
27 Greenstein G. Contemporary interpretation of probing depth assessments: diagnostic and therapeutic indications. J Periodontol. 1997;68:1194.
28 Turesky S, et al. Histologic and histochemical observations regarding early calculus formation in children and adults. J Periodontol. 1961;32:7.
29 Fine DH, Baumhammers A. Effect of water pressure irrigation on stainable material on the teeth. J Periodontol. 1970;41:468.
30 Bass CC. The optimum characteristics of tooth brushes for personal oral hygiene. Dent Items Interest. 1948;70:696.
31 Bass CC. The necessary personal oral hygiene for prevention of caries and periodontoclasia. J Louisiana Med Soc. 1948;101:52.
32 O’Leary TJ, et al. The Incidence of Recession in Young Males: Relationship to Gingival and Plaque Scores (Publication No. SAM-TR-67-97:1). U.S. Air Force School of Aerospace Medicine, July 1967.
33 Gjermo P, Flotra L. The plaque removing effect of dental floss and toothpicks: a group comparison study. J Periodont Res. 1969;4:170.
34 Graves R, et al. Comparative effectiveness of flossing and brushing in reducing interproximal bleeding. J Periodontol. 1989;60:243.
35 Arnim SS. The use of disclosing agents for measuring tooth cleanliness. J Periodontol. 1963;34:277.
36 Hujoel PP, et al. Non-surgical periodontal therapy and tooth loss. A cohort study. J Periodontol. 2000;71:736.
37 Westfelt E, et al. The effect of supragingival plaque control on the progression of advanced periodontal disease. J Clin Periodontol. 1998;25:536.
38 Black GV. A Work on Special Dental Pathology Devoted to the Diseases and Treatment of the Investing Tissues of the Teeth and Dental Pulp. Chicago: Medico-Dental Publishing, 1915.
39 Black AD. Treatment of chronic suppurative pericementitis. Natl Dent Assoc J. 1920;7:134.
40 Benjamin EM. The quantitative comparison of subgingival curettage and gingivectomy in the treatment of periodontitis simplex. J Periodontol. 1956;27:144.
41 Hassell TM. Epilepsy and the oral manifestations of phenytoin therapy. Monogr Oral Sci. 1981;9:1.
42 Ramfjord SP, Nissle RR. The modified Widman flap. J Periodontol. 1974;45:601.
43 Prichard J. Gingivoplasty, gingivectomy, and osseous surgery. J Periodontol. 1961;32:275.
44 Levin MP, et al. Healing of periodontal defects with ceramic implants. J Clin Periodontol. 1974;1:197.
45 Klingsberg J. Periodontal scleral grafts and combined grafts of sclera and bone: two year appraisal. J Periodontol. 1974;45:262.
46 Boyne PJ, Cooksey DE. Use of cartilage and bone implants in the restoration of edentulous ridges. J Am Dent Assoc. 1965;71:1426.
47 Forsberg H. Transplantation of os purum and bone chips in the surgical treatment of periodontal disease. Acta Odontol Scand. 1956;13:235.
48 Schaffer EM. Cementum and dentine implants in a dog and a rhesus monkey. J Periodontol. 1957;28:125.
49 Robinson RE. The osseous coagulum for bone induction technique: a review. J Calif Dent Assoc. 1970;46:18.
50 Mellonig JT, et al. Clinical evaluation of freeze-dried bone allografts in periodontal osseous defects. J Periodontol. 1976;47:125.
51 Dragoo MK, Sullivan HC. A clinical and histological evaluation of autogenous iliac bone grafts in humans. I. Wound healing 2 to 8 months. J Periodontol. 1973;44:599.
52 Schallhorn RG. Present status of osseous grafting procedures. J Periodontol. 1977;48:570.
53 Kenney EB, et al. Bone formation within porous hydroxylapatite implants in human periodontal defects. J Periodontol. 1986;57:76.
54 Stahl SS, Froum S. Histological evaluation of human intraosseous healing responses to the placement of tricalcium phosphate ceramic implants. J Periodontol. 1986;57:211.
55 Schepers EJG, Ducheyne P. The application of bioactive glass particle of narrow size range as a filler material for bone lesions. Bioceramics. 1993;6:401.
56 Ong MMA, et al. Evaluation of a bioactive glass alloplast in treating periodontal intrabony defects. J Periodontol. 1998;69:1346.
57 Haase HR, Bartold PM. Enamel matrix derivative induces matrix synthesis by cultured human periodontal fibroblast cells. J Periodontol. 2001;72:28.
58 Yukna RA, Mellonig JT. Histologic evaluation of periodontal healing in humans following regenerative therapy with enamel matrix derivative. A 10-case series. J Periodontol. 2000;71:752.
59 Olsen C, et al. A longitudinal study comparing apically positioned flaps, with and without osseous surgery. Int J Periodontics Restorative Dent. 1985;5:11.
60 Becker W, et al. A longitudinal study comparing scaling, osseous surgery and modified Widman procedures: results after one year. J Periodontol. 1988;59:351.
61 Kaldahl WB, et al. Evaluation of four modalities of periodontal therapy. Mean probing depth, probing attachment level and recession changes. J Periodontol. 1988;59:783.
62 Baima RF. Considerations for furcation treatment. I. Diagnosis and treatment planning. J Prosthet Dent. 1986;56:138.
63 Abrams L, Trachtenberg DI. Hemisection—technique and restoration. Dent Clin North Am. 1974;18:415.
64 Highfield JE. Periodontal treatment of multirooted teeth. Aust Dent J. 1978;23:91.
65 Gher ME, Vernino AR. Root anatomy: a local factor in inflammatory periodontal disease. Int J Periodontics Restorative Dent. 1981;1(5):52.
66 Cohen S, Hargreaves KM. Pathways of the Pulp, 9th ed. St. Louis: Mosby, 2006.
67 Ash MM, Nelson SJ. Wheeler’s Dental Anatomy, Physiology, and Occlusion, 8th ed. Philadelphia: WB Saunders, 2003.
68 Hamp SE, et al. Periodontal treatment of multirooted teeth. Results after 5 years. J Clin Periodontol. 1975;2:126.
69 Ross IF, Thompson RH. A long-term study of root retention in the treatment of maxillary molars with furcation involvement. J Periodontol. 1978;49:238.
70 Hellden LB, et al. The prognosis of tunnel preparations in treatment of class III furcations. A follow-up study. J Periodontol. 1989;60:182.
71 Amen CR. Hemisection and root amputations. Periodontics. 1966;4:197.
72 Newell DH, et al. Fixed prosthodontics with periodontally compromised dentitions. In Malone WF, Koth DL, editors: Tylman’s Theory and Practice of Fixed Prosthodontics, 8th ed., Tokyo: Ishiyaku-EuroAmerica, 1989.
73 Baima RF. Considerations for furcation treatment. II. Periodontal therapy. J Prosthet Dent. 1987;57:400.
74 Bergenholtz A. Radiectomy of multirooted teeth. J Am Dent Assoc. 1972;85:870.
75 Haskell EW, Stanley HR. A review of vital root resection. Int J Periodontics Restorative Dent. 1982;2(6):28.
76 Lee FMS. The displaced root in the maxillary sinus. Oral Surg. 1970;29:491.
77 Waldrep ACJr. Management of fractured root fragments. Dent Clin North Am. 1973;17:549.
78 Langer B, et al. An evaluation of root resections. A ten-year study. J Periodontol. 1981;52:719.
79 Basaraba N. Root amputation and tooth hemisection. Dent Clin North Am. 1969;13:121.
80 Polson AM. Periodontal considerations for functional utilization of a retained root after furcation management. J Clin Periodontol. 1977;4:223.
81 Baima RF. Considerations for furcation treatment. III. Restorative therapy. J Prosthet Dent. 1987;58:145.
82 Caplan CM. Fixed bridge placement following endodontic therapy and root hemisection. Dent Surv. 1978;54(6):28.
83 Haskell EW, Stanley HR. Resection of two vital roots. J Endodont. 1975;1:36.
84 Carnevale G, et al. Management of furcation involvement. Periodontol 2000. 1995;9:69.
85 Carnevale G, et al. Long-term effects of root-resective therapy in furcation-involved molars: a 10-year longitudinal study. J Clin Periodontol. 1998;25:209.
86 Melcher AH. On the repair potential of periodontal tissues. J Periodont Res. 1976;47:256.
87 Aukhil I, et al. Periodontal wound healing in the absence of periodontal ligament cells. J Periodontol. 1987;58:71.
88 Nyman S, et al. The regenerative potential of the periodontal ligament. An experimental study in the monkey. J Clin Periodontol. 1982;9:257.
89 Magnusson I, et al. New attachment formation following controlled tissue regeneration using biodegradable membranes. J Periodontol. 1988;59:1.
90 Pitaru S, et al. Collagen membranes prevent the apical migration of epithelium during periodontal wound healing. J Periodont Res. 1988;22:331.
91 Cortellini P, et al. Guided tissue regeneration with different materials. Int J Periodontics Restorative Dent. 1990;10:136.
92 Eickholz P, et al. Guided tissue regeneration with bioabsorbable barriers. II. Long-term results in infrabony defects. J Periodontol. 2004;75:957.
93 Kwan SK, et al. The use of autogenous periosteal grafts as barriers for the treatment of intrabony defects in humans. J Periodont. 1998;69:1203.
94 Tonetti MS, et al. Generalizability of the added benefits of guided tissue regeneration in the treatment of deep intrabony defects. Evaluation in a multi-center randomized controlled clinical trial. J Periodontol. 1998;69:1183.
95 Pontoriero R, et al. Guided tissue regeneration in the treatment of furcation defects in man. J Clin Periodontol. 1987;14:618.
96 Caffesse RG, et al. Class II furcations treated by guided tissue regeneration in humans: case reports. J Periodontol. 1990;61:510.
97 Vernino AR, et al. Use of biodegradable poly-lactic acid barrier materials in the treatment of grade II periodontal furcation defects in humans—Part I: a multicenter investigative clinical study. Int J Periodontics Restorative Dent. 1998;18:572.
98 De Leonardis D, et al. Clinical evaluation of the treatment of class II furcation involvements with bioabsorbable barriers alone or associated with demineralized freeze-dried bone allografts. J Periodontol. 1999;70:8.
99 Becker W, et al. Bone formation at dehisced dental implant sites treated with implant augmentation material: a pilot study in dogs. Int J Periodontics Restorative Dent. 1990;10:92.
100 Dahlin C, et al. Membrane-induced bone augmentation at titanium implants. A report on ten fixtures followed from 1 to 3 years after loading. Int J Periodontics Restorative Dent. 1991;11:273.
101 Becker W, Becker BE. Guided tissue regeneration for implants placed into extraction sockets and for implant dehiscences: surgical techniques and case reports. Int J Periodontics Restorative Dent. 1990;10:376.
102 Sottosanti J. Calcium sulfate: a biodegradable and biocompatible barrier for guided tissue regeneration. Compend Contin Educ Dent. 1992;13:226.
103 Payne JM, et al. Migration of human gingival fibroblasts over guided tissue regeneration barrier materials. J Periodontol. 1996;67:236.
104 Kim C-K, et al. Periodontal repair with intrabony defects treated with a calcium sulfate implant and calcium sulfate barrier. J Periodontol. 1998;69:1317.
105 American Academy of Periodontology. Parameter on periodontal maintenance. J Periodontol. 2000;71:849.
106 American Academy of Periodontology. Periodontal maintenance. J Periodontol. 2003;74:1395.
107 Bergstrom J, et al. A 10-year prospective study of tobacco smoking and periodontal health. J Periodontol. 2000;71:1338.
108 Calsina G, et al. Effects of smoking on periodontal tissues. J Clin Periodontol. 2002;29:771.
109 Baljoon M, et al. The association of smoking with vertical periodontal bone loss. J Periodontol. 2004;75:844.