19 FRAMEWORK DESIGN AND METAL SELECTION FOR METAL-CERAMIC RESTORATIONS
All patients want a pleasing smile, and so esthetics is an essential part of restorative practice, in which attention must be given to color, shape, surface texture, and proportion. Because anterior and maxillary posterior teeth are the most visible, they require the greatest attention to esthetic detail.
Tooth-colored restorative materials have evolved from the soluble silicate cements of the past to the composite resin materials and resin-modified glass ionomer cements of today. Currently, metal-ceramic prostheses are widely accepted and are the most commonly used extracoronal restoration.1,2 They combine the superior fit of a casting with the outstanding esthetics of dental porcelain. Because the ceramic veneer is chemically bonded to the metal substructure, this restoration is not subject to the discoloration problems associated with acrylic resin veneer crowns and, when appropriate clinical and laboratory protocols are followed, excellent longevity can be predicted.3 In addition, the material properties of dental porcelain are better able than resin to withstand wear under functional loading.
The concept of combining a brittle material with an elastic material to arrive at more desirable physical properties has many engineering applications. Dental porcelains (which are, chemically speaking, glasses) resist compressive loading but tend to succumb to tensile stress. Therefore, the metal substructure must be designed so that any tensile stresses in the porcelain are minimized.
To avoid fracture, the thickness of a ceramic veneer must not exceed 2 mm; however, a minimum thickness of 1 mm is needed for an esthetically pleasing restoration.
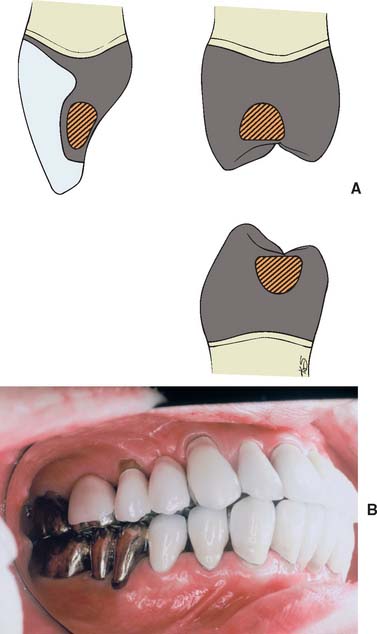
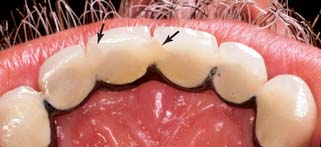
Restorations with porcelain occlusal surfaces must be planned carefully. Although they are esthetically very acceptable, these restorations have disadvantages, especially wear of the opposing enamel.4 Ideally, an esthetic restoration should wear at approximately the same rate as the enamel it replaces (about 10 μm per year5). In addition, the restoration should not increase the wear rate of an opposing enamel surface. Dental porcelain is more abrasive of enamel than of other restorative materials (e.g., gold or amalgam6-10) and has been implicated in severe occlusal wear, particularly when the porcelain is not glazed or highly polished (Fig. 19-1).11 This factor should be considered whenever a metal-ceramic restoration is being designed,12 and the practitioner should realize that although abrasiveness may be correlated with the composition of the ceramic material, the selection of a lower fusing ceramic (sometimes labeled by the manufacturer as “low-wear”) does not necessarily mean less enamel wear.13 Less wear on opposing teeth was cited as the most important need for improvement of posterior tooth-colored crowns.1 In addition, porcelain occlusal coverage leads to restorations with lower strength,14 and anatomically correct occlusal form with sharp cusps can be difficult to obtain in dental porcelain.
Fig. 19-1 A to D, Destructive enamel wear associated with metal-ceramic restorations.
(Courtesy of Dr. M. T. Padilla.)
Some technicians may attempt to fabricate a framework by dipping the die into molten wax, obtaining an even thickness. After the excess wax is trimmed away, a gingival collar is added, and the pattern is sprued, invested, and cast. When this is completed, the veneer is then applied. This technique almost always produces an uneven porcelain thickness, with an increased potential for material fracture as a result of the porcelain’s lack of proper support (Fig. 19-2). If porcelain thickness is not well controlled, appearance suffers as well, because the shade of the final crown depends on porcelain thickness.15 For predictable success, the framework must be carefully designed and shaped.
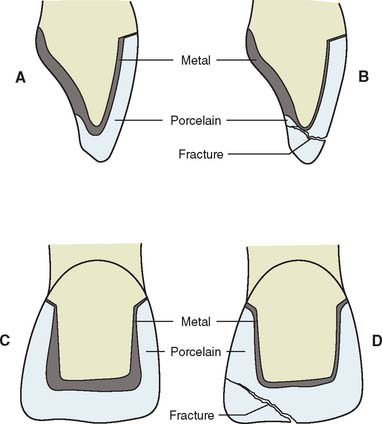
Fig. 19-2 A and C, Cross section through a metal-ceramic restoration. Ideal porcelain thickness is ensured by waxing to the full anatomic contour and cutting back. B and D, Incorrect framework design has insufficient support for the incisal porcelain. This can lead to fracture.
PREREQUISITES
The framework design for a fixed prosthesis should be considered during the treatment planning stage (see Chapter 3) and should be evaluated at the diagnostic tooth preparation and waxing stages, particularly in more complex treatments. A properly designed framework for a metal-ceramic crown or fixed dental prosthesis (FDP) can be achieved routinely only by waxing the restoration to complete anatomic contour first and then cutting back a consistent amount for the veneer. This allows an even thickness of porcelain, proper porcelain-metal interfaces, good connector design, and optimally placed occlusal contacts.
Waxing to Anatomic Contour
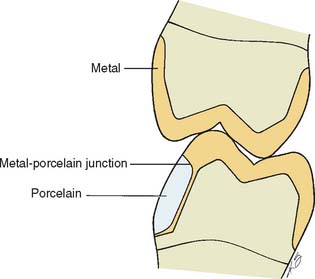
The main objective in waxing a framework is a substructure that supports a relatively even thickness of porcelain. Simultaneously, if the retainer is to serve as part of an FDP, it must allow for proper connector configuration and location. Furthermore, the restoration must conform to the normal anatomic configuration of the tooth that is being replaced. At the porcelain-metal interface, the ceramic material should be at least 0.5 mm thick. The framework should be shaped to allow for a distinct margin so that the porcelain is not overextended (Fig. 19-3). There should be no abrupt contour change between the metal and the adjacent porcelain.
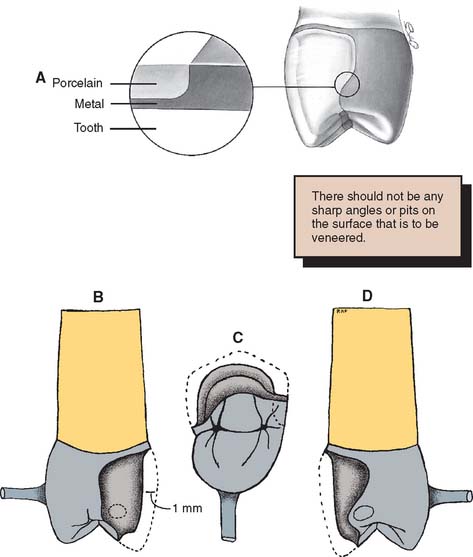
Fig. 19-3 A, The metal substructure should have a distinct margin for finishing the veneer. The location of the ceramic-metal interface varies, depending on the material chosen to contact adjacent and opposing teeth. B, Cutback for proximal contact in porcelain. C, Occlusal contact in metal. D, Proximal contact in metal.
(B to D, Courtesy of Dr. R. Froemling.)
The most effective way to consistently meet these criteria, with a minimum number of failures, is to develop the final contours of the proposed restoration in wax (Fig. 19-4). Once completed, the area to be veneered can be demarcated and an even thickness of wax removed. If this technique is not followed, one or more of the objectives is almost certainly missed, and the contours of the framework are not in harmony with the required ceramic configuration (Fig. 19-5).
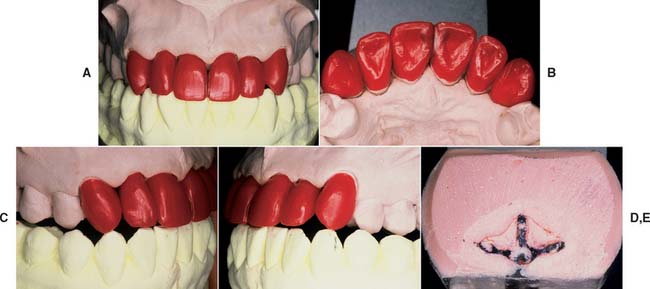
Fig. 19-4 A and B, Waxing anterior metal-ceramic restorations. C, Right lateral excursion. D, Left lateral excursion. E, The anterior guidance is determined with a custom table fabricated from the diagnostic waxing procedure.
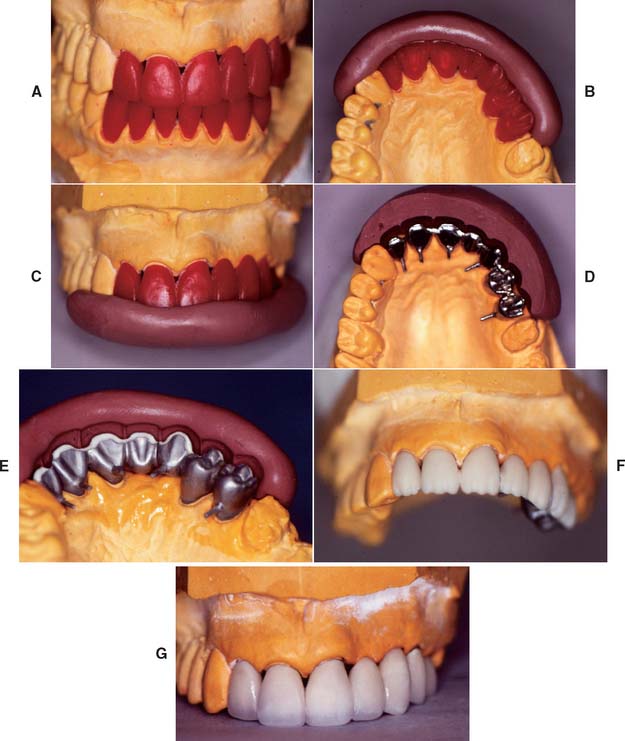
Fig. 19-5 Predictable esthetic result ensured by waxing to anatomic contour. A, Anatomic contour wax patterns. B and C, Incisal and labial indices were used to verify even cut-back. D, Cast substructures. E, The labial index is reused during porcelain application. F, The porcelain application. G, After contouring, the restorations are ready for clinical evaluation. The metal-ceramic restorations.
(Courtesy of Dr. M. Chen.)
Occlusal Analysis
The centric stops of any metal-ceramic restoration can be located on either porcelain or metal. However, they must be at least 1.5 mm away from the junction16 to prevent porcelain fracture from deformation of the metal (Fig. 19-6). Care is needed to minimize sliding contacts over the porcelain-metal interface. When this is not possible, the framework must be modified so that the porcelain is well supported in the area of functional contact.
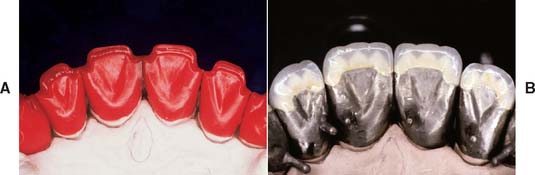
Fig. 19-6 A, The metal-ceramic junction must be carefully placed to avoid areas of high stress near occlusal contacts. B, Waxing to the anatomic contour ensures a smooth transition from porcelain to metal.
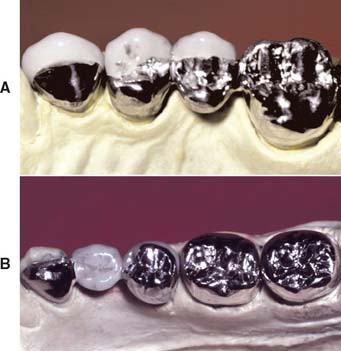
Existing restorations in the opposing arch can influence framework design. Because sliding contact of a porcelain restoration with a cast crown abrades the gold, the framework design must be modified as necessary. A complete cast crown in the mandibular arch presents little difficulty. It can be opposed by a maxillary restoration with only a metal occlusal surface and a facial ceramic veneer (Fig. 19-7). An existing metal crown on a maxillary molar, however, restricts the design of a mandibular metal-ceramic restoration if metal-to-porcelain contact is to be avoided (Fig. 19-8). Here the facial veneer can no longer be extended to include the buccal cusp tips and associated centric stops without contacting the opposing restoration. A complete cast crown is usually preferred because most patients do not show the facial surfaces of their mandibular posterior teeth. In other situations, particularly on mandibular first premolars, a facial veneer is esthetically essential, and the design of opposing restorations should allow for it (Fig. 19-9).
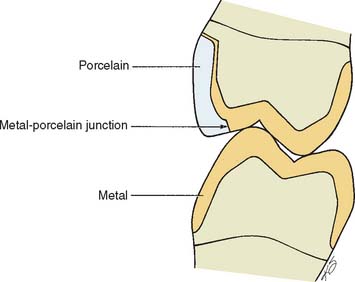
Fig. 19-7 The metal-ceramic restoration should be designed so that porcelain does not oppose an existing gold restoration. This presents few problems in the maxillary arch because the less visible lingual cusps are in cuspal contact.
CUTTING BACK
The criteria for waxing to anatomic contour are discussed in Chapter 18. This section deals with cutting back the veneering area.
Step-by-Step Procedure
Designing the cut-back
Esthetic and functional needs govern the design of the veneering surface. The ceramic veneer should extend far enough interproximally, particularly in the cervical half of the restoration, to avoid metal display. Wherever possible, the functional occlusal surfaces should be designed in metal, because an accurate occlusion is then easier to achieve (Fig. 19-10). However, esthetic demands may require extension of the porcelain veneer (e.g., on the mesial incline of a mandibular buccal cusp). The extent to which a restoration can be veneered is determined largely by the location of the centric stops.
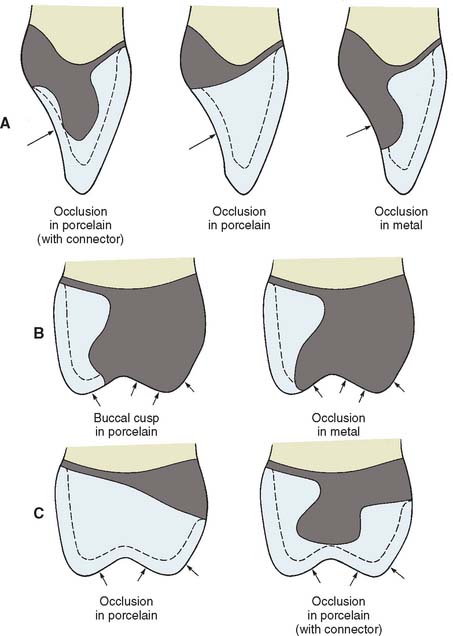
Fig. 19-10 Framework designs for a maxillary incisor (A) and a maxillary posterior tooth (B). The cut-back should be designed so the occlusal contacts are 1.5 mm away from the metal-porcelain junction. C, Framework designs for porcelain occlusal surfaces.
Troughing the pattern
Just as guiding grooves are used to mark the amount of substance to be removed in tooth preparation, depth cuts (troughing) can be used to standardize the amount of wax to be removed from the veneering area.
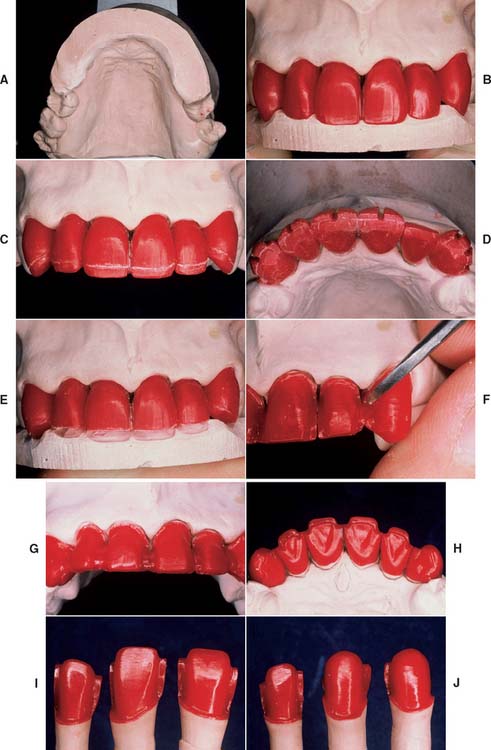
Fig. 19-12 Cut-back procedure. A and B, For extensive restorations, a matrix or index can be made to assist with the evaluation of the cut-back and subsequent porcelain application. C, It is important to follow the incisal contour carefully. D, Guiding troughs prepared in the area to be veneered. E, Wax is removed from between the troughs. F, The porcelain-metal interface is carved to a distinct butt joint. G, Note the correctly shaped proximal. These units will have soldered connectors. H, The finished cut-back. I and J, Patterns before reflowing of the margins.
Finishing
Connector design
Pontics
Evaluation
Immediately before the investing stage, the following criteria should have been met:
METAL SELECTION
Clinicians and dental laboratories face a potentially bewildering set of choices when selecting alloys for metal-ceramic restorations. Both noble metal and base metal casting alloys exist, and there are different alloy types for each of these two major groups. There are advantages and disadvantages for each alloy type, including significant differences in cost. Successful clinical practice depends on the selection of a compatible metal-porcelain combination that provides predictable results, depending on the particular patient’s case. Improper selection can cause catastrophic failure (Fig. 19-16). For a better understanding of the different properties provided on the packaging of casting alloys, the meanings and clinical relevance of these properties are discussed next.
Dental Connotations of Mechanical and Physical Properties for Ceramic Alloys
Mechanical properties of major clinical relevance are modulus of elasticity, yield strength (or proportional limit), hardness, and creep or distortion at elevated temperatures. Ultimate tensile strength (UTS), ductility, and toughness should also be reviewed, although these properties have less relevance for metal-ceramic restorations. Except for hardness (and elevated temperature creep or distortion), all these mechanical properties are determined by loading a cast specimen of the alloy to the point of failure in a tension test at room temperature. The physical property of thermal contraction is crucial in choosing an alloy that is compatible with the porcelain selected. From a practical standpoint, the density is important in both the economics of alloy selection and the dental laboratory procedure with the casting machine.
Modulus of elasticity
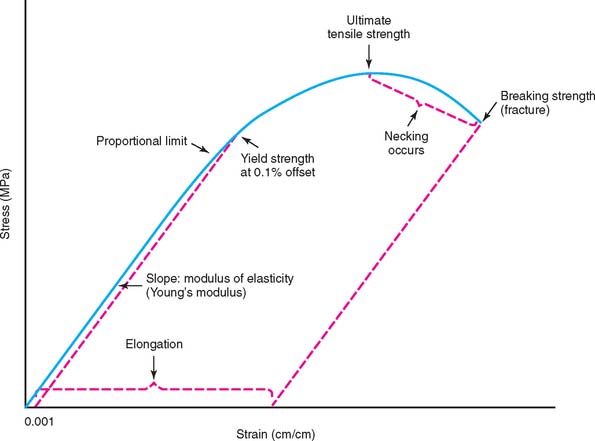
Figure 19-17 illustrates schematically the tensile stress-strain plot for a ductile casting alloy that undergoes substantial permanent deformation before fracture. This plot consists of two portions: (1) a linear or elastic region that ends at the proportional limit, where the stress is proportional to strain, and (2) a subsequent curved region corresponding to plastic or permanent deformation (which terminates when the test specimen fractures). The modulus of elasticity (also called Young’s modulus) is the slope of the stress-strain plot in the elastic region. The elastic modulus has the same value for tensile and compressive strains, which occur during bending of a prosthesis, in which regions on opposite sides of the neutral axis (centerline for a symmetric cross section) undergo opposite senses of deformation. An alloy with a higher modulus of elasticity has greater stiffness or rigidity for elastic deformation. For the fabrication of a long-span FDP, an alloy with a relatively high modulus of elasticity to reduce the amount of bending deflection under loading is preferred, because excessive flexure can cause fracture of the brittle porcelain (Fig. 19-18). The modulus of elasticity has units of stress/strain and is reported for dental alloys in units of gigapascals (1 GPa = 109 Pa = 145,000 psi [pounds per square inch]). The unit of 1 Pa = 1 N/m2 is much too small to be useful for the elastic modulus of materials.
Proportional limit and yield strength
In standard testing practice, the proportional limit of an alloy is determined by placing a straight edge on the stress-strain plot (or performing this operation with computer software) and noting the value at which the plot first deviates from a straight line. The proportional limit is often considered synonymous with the elastic limit, which corresponds to the value of stress at which permanent deformation occurs. However, the value of the elastic limit is highly dependent on the sensitivity of the strain-measuring apparatus. Moreover, precise location of the proportional limit on the stress-strain plot is somewhat problematic. Consequently, dental alloy manufacturers generally report the yield strength (sometimes called offset yield strength), which corresponds to the amount of stress for a very small designated amount of permanent deformation, at 0.1% or 0.2% (permanent strains of 0.001 or 0.002, respectively). The unit for yield strength is megapascal: 1 MPa = 106 Pa = 145 psi. As shown in Figure 19-17, the yield strength is obtained by constructing a line parallel to the initial straight-line portion of the stress-strain plot, starting with the specified value of offset on the horizontal strain axis and then noting the point of intersection with the curved portion of the plot. Because the 0.2% yield strength can be substantially higher than the 0.1% yield strength for a given alloy, depending on the rate of work hardening (slope of the curved portion of the stress-strain plot), manufacturers specify the offset value at which the yield strength was determined on the alloy packaging. The yield strength is often called the useful strength of a dental alloy, because stresses caused by biting forces should not exceed the yield strength and result in permanent deformation of the alloy. Although a sufficiently high value of yield strength is essential for a ceramic alloy, values that are too high create difficulties when the casting is adjusted in the dental laboratory or dental office.
Hardness
The Vickers hardness number (VHN) is generally measured for dental alloys by means of a symmetric diamond pyramidal indenter. The VHN is the quotient of the indenting load and the surface area of the indentation, for which the square of the mean diagonal length is multiplied by a constant related to the indenter geometry. The Knoop hardness number (KHN), obtained with a diamond indenter having long and short axes, is sometimes reported for dental alloys. For the KHN, only the length of the long diagonal of an indentation is measured, and the indenting load is divided by the unrecovered projected area of the indentation. Harder alloys, which have smaller indentations, have higher VHN and KHN values. Conversion scales available for the two different hardness tests should be used with caution, because such conversions are alloy dependent. Both the VHN and KHN are measures of the microhardness, in contrast to the older Brinell and Rockwell tests, in which much larger indenters are used to measure the macrohardness. When measuring the Vickers hardness of an alloy, an understanding of the microstructure is crucial. Use of the large indenting load of 1 kg stipulated in standards for dental alloys provides information about the overall hardness of the alloy microstructure, whereas light indenting loads (e.g., 10 g) can be used to obtain information about the hardness of individual grains, constituents, or phases. The hardness is an important practical property, inasmuch as very high values of hardness cause difficulty in the dental laboratory when the casting is ready to be finished. Alloys with VHN or KHN values exceeding that of enamel (approximately 350) cause abrasive wear of opposing teeth.
Elevated-temperature creep and distortion
Castings undergo elevated-temperature dimensional changes during the porcelain firing cycles. These changes have many causes, such as bulk creep of the alloy from several metallurgical mechanisms, distortion of the alloy as a result of the relief of residual stresses from the casting process, and alloy oxidation. The latter may be higher for high-palladium and other alloys that undergo internal (bulk and grain boundary) oxidation with the formation of oxide precipitate particles, in addition to the formation of an external oxide layer. Measuring the dimensional changes that occur in alloys during the porcelain bonding sequence is tedious, but concern has been expressed about the clinical fit for castings prepared from certain alloys. Nevertheless, in most cases, an experienced dental laboratory should be able to vary techniques and obtain successful results.
Ultimate tensile strength
The UTS (also called tensile strength or simply strength) is the maximum point on the stress-strain curve (see Fig. 19-17) and represents the greatest value of stress that can be developed in the alloy without fracture. The unit of measure for UTS is megapascal. Two types of stress-strain curves are observed for tensile testing of casting alloys. Alloys of high ductility undergo substantial necking between the UTS and the breaking strength, as shown in Figure 19-17. Other alloys of more limited ductility undergo much less necking, and the stress continues to increase after the yield strength until fracture occurs at the UTS. The UTS has minimal practical importance for a ceramic alloy, because the corresponding permanent strain does not occur under clinical conditions for a restoration. Nevertheless, this property is easy to measure, inasmuch as a strain-gauge extensometer does not need to be attached to the specimen, and manufacturers often quote the UTS.
Percentage elongation
For metals, the ductility—the capability of undergoing permanent tensile deformation—is measured in two ways when the test specimen is loaded to fracture: as percentage elongation or as reduction in area. For dental alloy castings, the ductility is measured as the percentage of permanent elongation of the starting gauge length, after the two portions of the fractured specimen are placed back together. This is done because the castings typically fracture on inclined planes whose locations are determined by porosity, and a well-defined area for the fracture surface is not available for measurement of the reduction in area. Obtaining precise registration of the two fractured portions and defining the location of the original gauge length are difficult, and therefore determining the percentage elongation to better than the nearest 1% is difficult to achieve, although values to the nearest 0.1% have been quoted. In principle, the percentage elongation can be obtained during the stress-strain test if a breakaway extensometer is attached to the specimen. However, such extensometers are rarely available in dental materials laboratories. Figure 19-17 exaggerates the more important elastic range of the stress-strain curve, inasmuch as the values of percentage elongation for current casting alloys used for porcelain veneering generally exceed 10% (Table 19-1). It is instructive to draw the stress-strain plots for some alloys listed in Table 19-1, because it is readily seen that the region of permanent deformation is much more extensive on the strain axis than the region of elastic deformation. When considering the ease of adjustment for cast restorations, the practitioner must remember that both yield strength and percentage elongation are involved.18 Alloys with high yield strength cannot be burnished by hand, even if they have high values of percentage elongation.
Toughness
Historically, toughness, which is the total area under the stress-strain curve, was considered an important property of casting alloys. However, with the focus on stresses that do not exceed the yield strength, this property no longer receives as much attention. The toughness represents the total energy per unit volume necessary to fracture the alloy and has units of stress × strain, or megapascals. For an alloy that does not work harden greatly and has substantial ductility, toughness is approximately equal to UTS × elongation. Determining toughness from stress-strain plots is laborious, and manufacturers do not report this property.
Thermal expansion/contraction
The linear coefficient of thermal expansion is a crucial property for an alloy that is to be bonded to dental porcelain. These coefficients should be closely matched to within about 0.5 × 10−6/° C below the glass transition temperature of the porcelain (approximately 500° to 600° C, depending on the cooling rate), at which the ceramic can no longer undergo viscous flow to relieve thermal incompatibility stresses. The thermal contraction coefficient (α), generally assumed to be the same as that for thermal expansion, should be slightly higher for the metal so that the ceramic is in a state of beneficial residual compressive stress at room temperature. Values of α typically range from 13.5 to 14.5 × 10−6/° C for metals and 13.0 to 14.0 × 10−6/° C for porcelains.
Density
Density is the ratio of mass to volume; specific gravity is the ratio of the density of a substance to the density of water. Densities for the important types of noble and base metal casting alloys are provided in Table 19-1. The high–gold content alloys have much higher densities than the low–gold content, palladium-based, and base metal casting alloys. This is because gold has a much higher density (19.3 g/cm3) than palladium (12.0 g/cm3), nickel (8.9 g/cm3), and cobalt (8.8 g/cm3). These differences in density have two consequences. First, for cast restorations of the same size and configuration, less mass of metal is required for the lower density alloy; the difference in the metal cost for a restoration can be substantial when both the unit metal cost and the density difference are considered. Second, additional winding of the spring on the centrifugal casting machine is necessary to achieve the needed casting pressure for the lower density alloys.
Available Alloy Systems
The nomenclature for dental casting alloys usually creates confusion. Classifying noble and base metal casting alloys according to the mechanism for corrosion resistance is the preferred method of categorization. The gold-based and palladium-based noble metal casting alloys achieve corrosion resistance because of the inherent nobility of the gold and palladium atoms, which do not form stable oxides at room temperature. In contrast, the conventional base metal casting alloys, in which nickel and cobalt are the principal elements and chromium is present to provide corrosion resistance, oxidize rapidly to form a passivating chromium oxide surface layer that blocks the diffusion of oxygen and prevents corrosion of the underlying metal.
Historically, terms such as precious, semiprecious, and nonprecious have been used to describe dental casting alloys. Such precious or semiprecious alloys usually contained a greater quantity of silver, along with more palladium and a reduced gold content. Silver, which is not a noble metal in the oral environment, assumes some noble metal character in the presence of palladium. The terms precious, semiprecious, and nonprecious, which refer to unit metal cost, are now less preferable than the terms noble and base metals, which refer to the electrochemical character of the alloys.
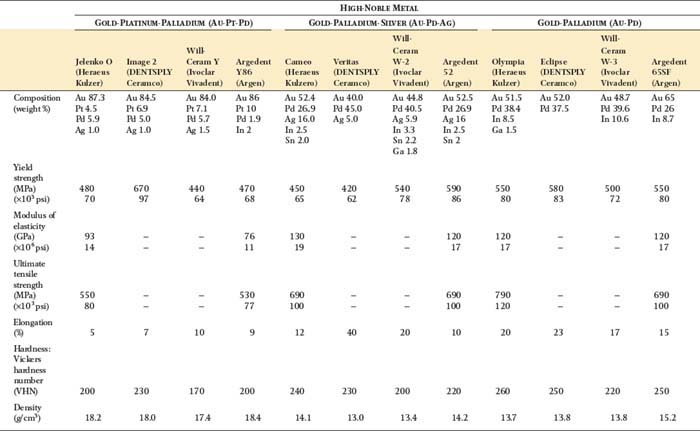
The major noble metals in dental alloys are gold, platinum, and palladium. (The other noble metals are iridium, ruthenium, rhodium, and osmium.) The total percentage of gold, platinum, and palladium in a dental alloy is referred to as the noble metal content. Iridium (much less than 1% by weight) and ruthenium (up to about 1%) are used as grain-refining elements in gold-based and palladium-based casting alloys, respectively. The original metal-ceramic alloy compositions (e.g., Jelenko “O,” described in Table 19-1) had approximately 98% noble metal content by weight. Rapid increases in the price of gold during the 1970s stimulated the development of lower gold content (from about 85% to 50% by weight) alloys and base metal alloys for fixed prosthodontics.19 During the 1980s, the high-palladium alloys were developed as economic alternatives to the gold-based alloys.20*
A classification system21 developed by the American Dental Association for casting alloys is presented in Table 19-2 and includes alloys for all-metal and metal-ceramic restorations. Because the classification is based solely on noble metal content and ignores other, often crucial alloying elements, general statements cannot be made about mechanical properties, clinical performance, and biocompatibility, even within each of the three groups in Table 19-2. Hundreds of dental alloys are commercially available, and appropriate testing is necessary to characterize the properties, safety, and efficacy of each. However, when each of these major groups is further subdivided into alloy types, some accurate generalizations are possible. They are discussed in the following sections.
Table 19-2 CLASSIFICATION FOR DENTAL CASTING ALLOYS
| Noble metal content: Au, Pt, Pd (minimum percentage by weight) | |
|---|---|
| High-noble metal | 60% (>40% gold) |
| Noble metal | 25% (no gold requirement) |
| Predominantly base metal | <25% (no gold requirement) |
Au, gold; Pd, palladium; Pt, platinum.
High-noble metal alloys
The high-noble metal content alloys contain a minimum of 60% by weight of noble elements; at least 40% is gold. There are three systems in this class: gold-platinum-palladium (Au-Pt-Pd), gold-palladium-silver (Au-Pd-Ag), and gold-palladium (Au-Pd), in the historical order of their development. Table 19-1 lists some mechanical properties and the density for representative alloys of each system.
Au-Pt-Pd
As previously noted, these were the first casting alloys formulated to bond with dental porcelain. Because of concern about adverse effects on the color of dental porcelain, copper, which was traditionally used for strengthening the high-gold casting alloys for all-metal restorations, could not be incorporated in the ceramic alloy compositions. Instead, these alloys were strengthened by precipitates of an iron-platinum (Fe-Pt) intermetallic compound.22 Porcelain adherence was achieved by incorporating tin and indium in the alloys, in addition to the contribution from iron. During the initial alloy oxidation step for the porcelain firing cycles, tin and indium (as well as some iron) diffused to the alloy surface and became oxidized. Subsequent chemical bonding was achieved between this oxide layer and the dental porcelain (see Chapter 24). Although these alloys have excellent corrosion resistance, they are susceptible to some dimensional changes during the porcelain firing cycles and are not recommended for multiple-unit FDP restorations.
Au-Pd-Ag
These were the first lower gold content alternative alloys to be widely used in the 1970s. Platinum was eliminated from the alloy compositions, and the gold content was reduced to about 50%, with corresponding increases in the amounts of palladium and silver.23,24 Some alloy strengthening was achieved by solid solution hardening from the dissimilar atomic sizes of the three major elements (gold, palladium, and silver), which form solid solutions with each other. Additional solid solution strengthening was hypothesized to be caused by tin or indium, which were again incorporated as oxidizable elements to provide porcelain bonding. Further alloy strengthening may be caused by precipitates formed by these elements. Although these alloys have excellent mechanical properties and porcelain adherence, green discoloration (resulting from diffusion of silver atoms into the porcelain) has been reported for some alloy-porcelain combinations.25 Possible reasons for this effect may be the high sodium concentration of the porcelain or the relative sizes of the metal ions in the porcelain. The discolored region can be ground away, but this involves an additional processing step. In addition, silver vapor generated in the porcelain furnace during processing can contaminate the muffle, and periodic purging of the furnace with a carbon block is required. Green discoloration has apparently been eliminated in some porcelain compositions by substituting potassium ions for sodium ions; the larger potassium ions impede the diffusion of silver into the porcelain.
Au-Pd
Gold-palladium alloys that are silver-free were developed during the late 1970s and have become very popular. Alloy strengthening is achieved with a combination of solid solution hardening and microstructural precipitates. The hardness (assumed to be related to strength) of these alloys is independent of heat-treatment temperature within the porcelain-firing range, unlike that of Au-Pd-Ag alloys.24 The Au-Pd alloys have excellent mechanical properties, elevated-temperature creep behavior,26 and porcelain adherence,27 without the green discoloration associated with Au-Pd-Ag alloys.
Discussion
The data in Table 19-1 show that the Au-Pd and Au-Pd-Ag alloys, in comparison with the Au-Pt-Pd alloys, generally have higher values of yield strength and modulus of elasticity, along with lower density. Consequently FDP restorations fabricated from alloys in the former two groups are more resistant to masticatory forces and undergo less bending deflection. They also have the economic advantage of providing more restorations per unit of alloy cost. Selection of the proper porcelain for Au-Pd-Ag alloys is essential if discoloration problems are to be avoided.
Noble metal alloys
The noble metal alloys have a minimum of 25% by weight of noble metal, with no requirement for gold percentage. There are three alloy systems in this class: palladium-silver (Pd-Ag), palladium-copper-gallium (Pd-Cu-Ga), and palladium-gallium (Pd-Ga), in the historical order of their development. Table 19-1 lists some mechanical properties and the density for representative alloys of each system.
Pd-Ag
These alloys, developed in the 1970s, continued the trend by manufacturers of reducing the gold content (to between 0% and 2% by weight), with corresponding increases in the palladium and silver contents.28 A small percentage of gold in these alloys and the high-palladium alloys has little effect on their properties but may facilitate third-party payments. As previously noted, in the presence of palladium, silver appears to assume noble metal character, which is beneficial for corrosion resistance. Because of their high silver content (approximately 30% to 35% by weight), these alloys have been called semiprecious, a term that should no longer be used. In comparison with the Au-Pd-Ag and Au-Pd alloys, the Pd-Ag alloys have similar values of yield strength and modulus of elasticity and much lower density values. Because of their high silver contents, porcelain greening and furnace contamination can result during fabrication of partial FDP restorations, unless the porcelain is carefully selected. Nevertheless, these alloys are frequently chosen as a compromise between the more expensive high-noble alloys and the relatively inexpensive base metal alloys.
Pd-Cu-Ga
The Pd-Cu-Ga alloys contain more than 70% by weight of palladium and were developed in the early 1980s as economical alternatives to the gold-based alloys.20 The melting point of palladium (1555° C) is much higher than that of gold (1064° C); gallium has a melting point of 30° C. The addition of gallium to palladium yields high-palladium alloys that can be fused and cast with the same dental laboratory technology developed for the gold-based casting alloys. Multiorifice torches are necessary to fuse the high-palladium alloys, and the use of ceramic crucibles dedicated to individual alloys is recommended.20 Carbon-containing investments should not be used, because the incorporation of very small amounts of carbon in these alloys degrades the bond strength with porcelain.29 The Pd-Cu-Ga alloys appear to have casting accuracy comparable with that of the high-noble metal alloys.30
Measurements31,32 of the mechanical properties of some Pd-Cu-Ga alloys have produced values of yield strength, modulus of elasticity, and percentage elongation that differ from values in Table 19-1. This suggests some technique sensitivity in the fabrication of cast specimens for the tension test. Although a near-surface eutectic structure was present in Pd-Cu-Ga alloy castings that simulated copings for maxillary incisors,20 this constituent was absent in the 3-mm diameter cast specimens for the tension test.31,32 Some Pd-Cu-Ga alloys have hardness values comparable with or exceeding that of tooth enamel, and castings from these alloys may be difficult to finish in the dental laboratory. In addition, chairside adjustments may be difficult for patients. However, substitution of indium for tin yields Pd-Cu-Ga alloys with much lower hardness (VHN ≍ 270).33 All these alloys achieve substantial hardening by solid-solution incorporation of other elements within the palladium crystal structure. The hardest Pd-Cu-Ga alloys (VHN > 300) contain a hard grain boundary phase whose composition is close to that of Pd5Ga2.33 Transmission electron microscopic studies indicate that representative high-palladium alloys have the same bulk ultrastructure.34 Analyses of x-ray diffraction patterns have revealed that oxidized Pd-Cu-Ga alloys have complex internal oxidation regions that can contain up to five different oxide phases.35 Oxides of copper, gallium, tin, indium, and even palladium that were formed under the conditions present in the porcelain furnace were subsequently detected in the oxidized alloys at room temperature. The results of creep experiments on the Pd-Cu-Ga alloys have been mixed.26 The creep rates associated with relatively high thermal incompatibility stresses near the glass transition temperature of dental porcelain were high for two Pd-Cu-Ga alloys, whereas these alloys had excellent creep resistance at high temperatures and low stresses simulating the deflection of a long-span FDP as a result of gravity during processing.
Pd-Ga
The copper-free Pd-Ga alloys were subsequently developed during the 1980s to provide compositions with lower hardness than that of the initial Pd-Cu-Ga formulations. The hard Pd5Ga2 phase is absent in these alloys, which are strengthened by solid solution hardening.33 The alloys have a complex fine precipitate structure at the grain boundaries,20,36 and their mechanical properties are generally more similar to those of Pd-Ag alloys than to the Pd-Cu-Ga alloys. In comparison with the Pd-Ga alloys, porcelain adherence is superior for the Pd-Cu-Ga alloys.27 A palladium-gallium-cobalt (Pd-Ga-Co) alloy37 has a particularly dark oxide that is more difficult to mask with dental porcelain, and this alloy did not achieve widespread clinical acceptance.
Discussion
In one study in which the dimensional changes at various stages of the simulated porcelain firing cycles were compared for copings for metal-ceramic single-unit restorations of selected high-palladium alloys, investigators found that most of the selected high-palladium alloys had acceptable high-temperature distortion.31,38 Because of the considerable price volatility for palladium since the mid-1990s, dentists and dental laboratories have tended to select gold-palladium, palladium-silver, and lower gold content alloys, rather than high-palladium alloys. Currently, the unit metal cost of gold is substantially higher than that of palladium, and high-palladium alloys again have a significant economic advantage over gold-palladium alloys. When the high-palladium alloys were introduced in the 1980s, the unit metal cost was between half and one third of the Au-Pd alloys.20 However, caution is needed with the Pd-Ag alloys to prevent porcelain from acquiring green discoloration. Some biocompatibility concerns have been raised about the high-palladium alloys, particularly in Germany with the Pd-Cu-Ga alloys. Review articles39,40 suggest that there are minimal health hazards associated with the high-palladium alloys, and this has been borne out by extensive research41 involving cell culture42,43 and animal implantation44 methods.
Predominantly base metal alloys
Table 19-2 defines these alloys (sometimes termed nonprecious) as having less than 25% by weight of noble metal with no requirement for gold. Most of these alloys used for fixed prosthodontics are nickel-chromium (Ni-Cr) alloys, but some cobalt-chromium (Co-Cr) alloys have also been formulated for porcelain application. The complex metallurgy of these alloys is described in a review article.45
Ni-Cr
Yield strength, hardness, and modulus of elasticity can be greatly affected by small differences in weight percentages of minor elemental components among the compositions of these alloys. Table 19-1 illustrates some of these variations. For example, values of yield strength for the representative alloys listed vary from 310 to 550 MPa, and the VHN varies from 180 to 240. Consequently, the selection of a specific brand of Ni-Cr alloy depends on the clinical application. If burnishing or extended finishing of a crown is anticipated, a brand with a relatively low yield strength and hardness should be used.
One benefit of these alloys is that their values of modulus of elasticity are much higher than those of the noble metal alloys. Therefore, long-span fixed prostheses fabricated from Ni-Cr alloys undergo much less flexure than do similar prostheses fabricated from noble metal alloys, with less likelihood of fracture of the brittle dental porcelain component. These base metal casting alloys are generally considered more technique sensitive and difficult to cast than the noble metal casting alloys. However, this assessment may reflect the lack of experience of some dental laboratories with the Ni-Cr alloys, because excellent results for castability of these alloys have been published.46 Therefore, the choice of dental laboratory is particularly important when these alloys are selected.
Beryllium
Many Ni-Cr alloy formulations contain up to 2% by weight of beryllium. The major reason for incorporating this element in the alloy is to lower the melting range and to decrease the viscosity of the molten alloy, thereby improving its castability. Beryllium also provides strengthening and affects the thickness of the oxide layer, when the alloy is oxidized for porcelain firing. The latter is an important consideration for base metal casting alloys, which can form much thicker oxide layers than noble metal casting alloys. Fracture through the oxide layer may occur and cause failure of the base metal-ceramic restoration.
The use of beryllium has created some doubt about the safety of some Ni-Cr alloys. Of importance: When the densities of nickel (8.9 g/cm3) and chromium (7.2 g/cm3) are compared with that of beryllium (1.8 g/cm3), 2% by weight of beryllium in the alloy composition can be equivalent to nearly 10% beryllium on an atomic basis. Consequently, the atomic proportion of beryllium atoms in these alloy compositions can be relatively large.
Nickel
The U.S. Federal Standard for exposure to metallic nickel and soluble nickel compounds (1 mg/m3 for an 8-hour time-weighted average concentration) is much higher than the National Institute for Occupational Safety and Health recommendation for such exposure (15 μg/m3 for a 10-hour time-weighted average workday). Occupational exposure of refinery workers to nickel has been associated with lung and nasal cancer. Acute effects of exposure to nickel include skin sensitization that can lead to chronic eczema. Therefore, as a health precaution, an operator should wear a mask and use efficient suction when grinding and finishing a dental nickel-base alloy.
It has been reported that 9% of the female population and 0.9% of the male population are sensitive to nickel.47 This prompts a question: Are such individuals likely to manifest an adverse reaction to dental Ni-Cr alloys? In a 20-patient clinical study48 to investigate this question, each of 10 controls (who had no known sensitivity to nickel) showed a negative dermal response and a negative intraoral response to a dental Ni-Cr alloy. Of 10 patients with a known sensitivity, 8 showed a positive dermal response to the alloy. When these patients wore an intraoral appliance containing the Ni-Cr alloy, 30% manifested an allergic response within 48 hours.
According to the American Dental Association’s labeling requirement for base metal alloys that contain nickel, such alloys should not be used in individuals with known nickel sensitivity. Another question now arises: Can patients who are not allergic to nickel become sensitive to it from fixed prostheses made with nickel-containing alloys?
In one investigation,49 researchers found that Ni-Cr alloys not containing beryllium were more resistant than beryllium-containing alloys to in vitro corrosion. The four alloys studied showed lower corrosion rates in cell culture solutions after cold solution sterilization. Although the corrosion products released from the alloys did not alter the cellular structure and viability of human gingival fibroblasts, reductions in cellular proliferation were observed. The authors concluded that there still exist biocompatibility concerns relating to the exposure of local and systemic tissues to elevated levels of corrosion products from the Ni-Cr alloys.
Co-Cr
The potential health problems associated with beryllium- and nickel-containing alloys have led to the development of another alternative base metal alloy system: cobalt-chromium.50,51 The representative Co-Cr alloys listed in Table 19-1 have higher hardness than the Ni-Cr alloys listed, which suggests that finishing restorations made with the former alloys may be more difficult.
Titanium
Titanium-based alloys have been studied since the late 1970s as potential casting alloys.52 Advantages of titanium and titanium alloys include excellent biocompatibility and corrosion resistance, which results from the presence of a thin, adherent, passivating surface layer of TiO2. The low density (4.5 g/cm3) of titanium, in comparison with that of gold or palladium, also results in lighter and potentially less expensive restorations.* Alloys studied have principally been commercially pure titanium, which contains an upper limit of 1% impurities for American Society for Testing and Materials (ASTM) Grade 4, and the highly popular engineering alloy titanium–6% aluminum–4% vanadium (Ti-6Al-4V).53 There has been substantial interest in the titanium–6% aluminum–7% niobium (Ti-6Al-7Nb) alloy, which was advocated because of potential concern with cytotoxicity of vanadium and the poor wear resistance of commercially pure titanium.54-56
The dental casting of titanium and titanium alloys poses special problems because of the high melting point of titanium (1668° C) and its strong tendency to oxidize and react with other materials.57 Special casting machines that provide either a vacuum environment or an argon atmosphere must be used. Both vacuum/argon pressure and centrifugal dental casting machines have been developed, and both argon-arc melting and induction melting have been used to fuse titanium and titanium alloys.58,59 Studies have also discussed the use of face coats on the wax pattern,57,60,61 casting into low-temperature (350° C) phosphate-bonded investments,62 effects of the pressure differential for the argon gas pressure,63 permeability of the investment,64 and mold venting.65 Good accuracy for titanium castings on their dies can be achieved with the proper choice of investment.62,66 A study has compared commercially pure titanium cast with helium, argon, krypton, and xenon atmospheres and found that while the microstructure is not affected, porosity and mechanical properties depend upon gas type and pressure.67 Selecting a dental laboratory experienced in fabricating these castings is essential, and such dental laboratories are not common in the United States. Further research is needed to optimize the metallurgical structure and casting technology for titanium alloys, which have a dendritic microstructure57 resulting from the lack of a suitable grain-refining element. A very hard near-surface region (termed α case) that can exceed 50 μm in thickness is also present on the castings as a result of reaction of the titanium alloy with the investment (and perhaps also with the residual atmosphere in the casting machine).57,60 One study61 has shown that use of a zirconia-based coating on the wax pattern substantially reduced the thickness of the complex reaction layer with the investment and yielded titanium castings with a high-quality surface.
REVIEW OF TECHNIQUE
Figure 19-19 summarizes the steps involved in producing wax patterns for metal-ceramic restorations.
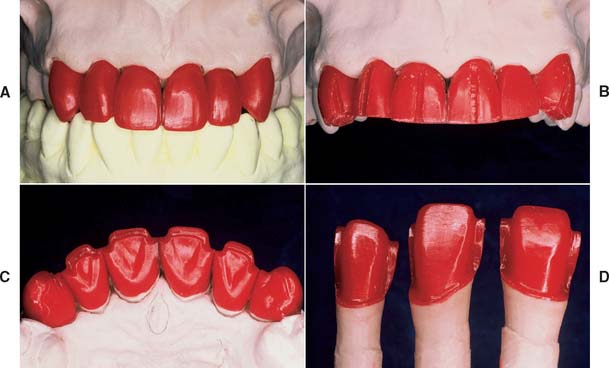
Fig. 19-19 Technique review. A, The restorations are waxed to anatomic contour. B, The patterns are troughed to obtain correct porcelain thickness in the completed restoration. C, The cut-back is completed. D, The margins are finalized before investing.
SUMMARY
Framework design for metal-ceramic restorations must be based on an understanding of fundamental material properties. Restorations should be waxed to anatomic contour and then cut back in the area that is to be veneered. This creates an even porcelain thickness, which provides superior mechanical properties in the completed restoration while simultaneously standardizing shade reproduction.
 GLOSSARY*
GLOSSARY*
airborne-particle abrasion \âr′bôrn′ pär′t -kal a-brā′zhun\: the process of altering the surface of a material through the use of abrasive particles propelled by compressed air or other gases
-kal a-brā′zhun\: the process of altering the surface of a material through the use of abrasive particles propelled by compressed air or other gases
al·loy \ăl′oi′, a-loi′\ n (14c): a mixture of two or more metals or metalloids that are mutually soluble in the molten state; distinguished as binary, ternary, quaternary, etc., depending on the number of metals within the mixture. Alloying elements are added to alter the hardness, strength, and toughness of a metallic element, thus obtaining properties not found in the pure metal. Alloys may also be classified on the basis of their behavior when solidified—usage: see BASE METAL, NOBLE METAL
alloying element \a-loi′ ng
ng  l′a-ment\ (1998): metallic or non-metallic elements added to or retained by a pure metal for the purpose of giving that metal special properties
l′a-ment\ (1998): metallic or non-metallic elements added to or retained by a pure metal for the purpose of giving that metal special properties
aluminum oxide \a-lōō′mi-num  k′sīd′\: 1: a metallic oxide constituent of dental porcelain that increases hardness and viscosity 2: a high strength ceramic crystal dispersed throughout a glassy phase to increase its strength as in aluminous dental porcelain used to fabricate aluminous porcelain crowns 3: a finely ground ceramic particle (frequently 50 um) often used in conjunction with air-borne particle abrasion of metal castings before the application of porcelain as with metal ceramic restorations
k′sīd′\: 1: a metallic oxide constituent of dental porcelain that increases hardness and viscosity 2: a high strength ceramic crystal dispersed throughout a glassy phase to increase its strength as in aluminous dental porcelain used to fabricate aluminous porcelain crowns 3: a finely ground ceramic particle (frequently 50 um) often used in conjunction with air-borne particle abrasion of metal castings before the application of porcelain as with metal ceramic restorations
an·neal \a-nēl′\ vt (1580): 1: to heat a material, such as metal or glass, followed by controlled cooling to remove internal stresses and create a desired degree of toughness, temper, or softness to a material 2: to heat a material, such as gold foil, to volatilize and drive off impurities from its surface, thus increasing its cohesive properties. This process is termed degassing 3: to homogenize an amalgam alloy by heating in an oven
base metal \bās m t′l\: any metallic element that does not resist tarnish and corrosion—see NOBLE METAL
t′l\: any metallic element that does not resist tarnish and corrosion—see NOBLE METAL
bio·ac·cept·ability \bī′ō-ăk-s p′ta-b
p′ta-b l′
l′ -tē′\ (1998): the quality of compatibility in a living environment in spite of adverse or unwanted side effects
-tē′\ (1998): the quality of compatibility in a living environment in spite of adverse or unwanted side effects
bio·com·pat·i·ble \bī′ō-kom-păt′a-bal\ adj: capable of existing in harmony with the surrounding biologic environment
bur·nish·ibil·i·ty \bûr′n sh-a-b
sh-a-b l′
l′ -tē\ n: the ease with which a material can be burnished
-tē\ n: the ease with which a material can be burnished
butt \bŭt\v (14c): to bring any two flat-ended surfaces into contact without overlapping, as in a butt joint
car·at \kăr′at\ n (15c): a standard of gold fineness. The percentage of gold in an alloy, stated in parts per 24. Pure gold is designated 24 carat
centric stop \s n′tr
n′tr k st
k st p\: opposing cuspal/fossae contacts that maintain the occlusal vertical dimension between the opposing arches
p\: opposing cuspal/fossae contacts that maintain the occlusal vertical dimension between the opposing arches
compressive stress \k m-pr
m-pr s′
s′ v str
v str s\: the internal induced force that opposes the shortening of a material in a direction parallel to the direction of the stresses; any induced force per unit area that resists deformation caused by a load that tends to compress or shorten a body
s\: the internal induced force that opposes the shortening of a material in a direction parallel to the direction of the stresses; any induced force per unit area that resists deformation caused by a load that tends to compress or shorten a body
1cop·ing \kō′p ng\ n: 1: a long, enveloping ecclesiastical vestment 2a: something resembling a cope (as by concealing or covering) 2b: coping
ng\ n: 1: a long, enveloping ecclesiastical vestment 2a: something resembling a cope (as by concealing or covering) 2b: coping
2cop·ing \kō′p ng\ n (ca. 1909): a thin covering or crown—usage: see C. IMPRESSION, TRANSFER C.
ng\ n (ca. 1909): a thin covering or crown—usage: see C. IMPRESSION, TRANSFER C.
cor·rode \ka-rōd\ vt (15c): 1: deterioration of a metal due to an electrochemical reaction within its environment 2: to eat away by degrees as if by gnawing 3: to wear away gradually usually by chemical action
cor·ro·sion \ka-rō′zhen\ n (15c): the action, process, or effect of corroding; a product of corroding; the loss of elemental constituents to the adjacent environment
2creep \krēp\ n (1818): the slow change in dimensions of an object due to prolonged exposure to high temperature or stress
de·gas \dē-găs′\ vt; de·gassed \dē-găsd′\ pt., pp; de·gassing \dē-găs′ing\ ppr (1920): 1: to remove gas from an object or substance 2: the name commonly used to denote the first heat cycle (oxidation cycle) in fabrication of a metal ceramic restoration that removes surface impurities from the metallic component and produces surface oxides prior to the application of opaque porcelain
de·pas·siv·a·tion \dē-păs′s -vā′shun\ n: loss of corrosion protection due to damage or removal of the protective oxide surface film on a passivated metal
-vā′shun\ n: loss of corrosion protection due to damage or removal of the protective oxide surface film on a passivated metal
duc·til·i·ty \dŭk-t l′
l′ -tē\ n (14c): the ability of a material to withstand permanent deformation under a tensile load without rupture; ability of a material to be plastically strained in tension. A material is brittle if it does not have appreciable plastic deformation in tension before rupture
-tē\ n (14c): the ability of a material to withstand permanent deformation under a tensile load without rupture; ability of a material to be plastically strained in tension. A material is brittle if it does not have appreciable plastic deformation in tension before rupture
dwt : abbr [denarius + weight] pennyweight; called also pennyweight; a measurement of weight in the troy system equal to 24 grains, or 0.05 ounce. Its metric equivalent is 1.555 grams
elastic limit \ -lăs′t
-lăs′t k l
k l m′it\: the greatest stress to which a material may be subjected and still be capable of returning to its original dimensions when such forces are released
m′it\: the greatest stress to which a material may be subjected and still be capable of returning to its original dimensions when such forces are released
elastic modulus \ -lăs′t
-lăs′t k m
k m j′a-lus\: the stiffness or flexibility of a material within the elastic range. Within the elastic range, the material deforms in direct proportion to the stress applied as represented by Hooke’s law
j′a-lus\: the stiffness or flexibility of a material within the elastic range. Within the elastic range, the material deforms in direct proportion to the stress applied as represented by Hooke’s law
e·las·tic·i·ty \ -lă-st
-lă-st s′
s′ -tē\ n (1664): the quality that allows a structure or material to return to its original form on removal of an external force—see MODULUS OF E.
-tē\ n (1664): the quality that allows a structure or material to return to its original form on removal of an external force—see MODULUS OF E.
e·lon·ga·tion \ -lông′gā′shun\ n (14c) 1: deformation as a result of tensile force application 2: the degree to which a material will stretch before breaking 3: the over eruption of a tooth
-lông′gā′shun\ n (14c) 1: deformation as a result of tensile force application 2: the degree to which a material will stretch before breaking 3: the over eruption of a tooth
fa·tigue \fa-tēg′\: the breaking or fracturing of a material caused by repeated cyclic or applied loads below the yield limit; usually viewed initially as minute cracks followed by tearing and rupture; also termed brittle failure or fracture <metal~>—see FAILURE
fracture strength \frăk′chur str ngkth\: strength at fracture based on the original dimensions of the specimen
ngkth\: strength at fracture based on the original dimensions of the specimen
frame·work \frām′wûrk\ n (1644): 1. an interior or imbedded, openwork, or structural frame used to support some other object or objects 2. the skeletal portion of prosthesis (usually metal, sometimes ceramic) around which and to which are attached the remaining portions of the prosthesis to produce a finished restoration—usage: for dental prostheses, the framework may be any metal or combination of metals, with various forms including designed slots, incorporated corrective angulation patterns etc. which provide ridgity to a prosthesis. Such a framework can be made in whole or made of component parts. Frequently used to anchor a prosthesis to natural teeth (by cementation) or dental implant abutments (by cementation, mechanical undercuts, screws) or both
Knoop hardness tests [Frederick Knoop, U.S. engineer, U.S. Department of Commerce]: eponym for a surface hardness test using a diamond stylus. It is used for harder materials and is characterized by the diamond or rhomboid shaped indentation. The indentation micro-hardness test uses a rhombic-based pyramidal diamond indenter. The long diagonal of the resulting indentation is measured to determine the hardness. This test is suitable for most classes of materials including brittle and elastomeric
Knoop F, Peters CG, Emerson WB. A sensitive pyramidal diamond tool for indentation measurements. J Res Nat Bur Stand 1939;12:39–45.
mal·le·a·ble \măl′ē-a-bal\ adj (14c): capable of being extended or shaped with a hammer or with the pressure of rollers
1met·al \m t′l\ n (13c): any strong and relatively ductile substance that provides electropositive ions to a corrosive environment and that can be polished to a high luster. Characterized by metallic atomic bonding
t′l\ n (13c): any strong and relatively ductile substance that provides electropositive ions to a corrosive environment and that can be polished to a high luster. Characterized by metallic atomic bonding
modulus of elasticity \m j′a-las ŭv
j′a-las ŭv  -lă-st
-lă-st s′
s′ -tē, ē-lă-\: in metallurgy, the coefficient found by dividing the unit stress, at any point up to the proportional limit, by its corresponding unit of elongation (tension) or strain. A ratio of stress to strain. As the modulus of elasticity rises, the material becomes more rigid
-tē, ē-lă-\: in metallurgy, the coefficient found by dividing the unit stress, at any point up to the proportional limit, by its corresponding unit of elongation (tension) or strain. A ratio of stress to strain. As the modulus of elasticity rises, the material becomes more rigid
modulus of resilience \m j′a-las ŭv r
j′a-las ŭv r -z
-z l-yans\: the work or energy required to stress a cubic inch of material (in one direction only) from zero up to the proportional limit of the material, measured by the ability of the material to withstand the momentary effect of an impact load while stresses remain within the proportional limit
l-yans\: the work or energy required to stress a cubic inch of material (in one direction only) from zero up to the proportional limit of the material, measured by the ability of the material to withstand the momentary effect of an impact load while stresses remain within the proportional limit
noble metal \nō′bal m t′l\: those metal elements that resist oxidation, tarnish, and corrosion during heating, casting, or soldering and when used intraorally; examples include gold and platinum—comp BASE METAL
t′l\: those metal elements that resist oxidation, tarnish, and corrosion during heating, casting, or soldering and when used intraorally; examples include gold and platinum—comp BASE METAL
nonprecious metal : see BASE METAL
pas·si·vate \păs′ -vāt\ vt -at·ed; -at·ing (1913): 1: to render inactive or less reactive 2: to protect against contamination by coating or surface treating
-vāt\ vt -at·ed; -at·ing (1913): 1: to render inactive or less reactive 2: to protect against contamination by coating or surface treating
precious metal \pr sh′as m
sh′as m t′l\: a metal containing primarily elements of the platinum group, gold, and silver
t′l\: a metal containing primarily elements of the platinum group, gold, and silver
precious metal alloy \pr sh′as m
sh′as m t′l ăl′oi′, a-loi′\: an alloy predominantly composed of elements considered precious, i.e., gold, the six metals of the platinum group (platinum, osmium, iridium, palladium, ruthenium, and rhodium), and silver
t′l ăl′oi′, a-loi′\: an alloy predominantly composed of elements considered precious, i.e., gold, the six metals of the platinum group (platinum, osmium, iridium, palladium, ruthenium, and rhodium), and silver
proportional limit \pra-pôr′sha-nal l m′
m′ t\: that unit of stresses beyond which deformation is no longer proportional to the applied load
t\: that unit of stresses beyond which deformation is no longer proportional to the applied load
shearing stress \shîr′ ng str
ng str s\: the internal induced force that opposes the sliding of one plane on an adjacent plane or the force that resists a twisting action
s\: the internal induced force that opposes the sliding of one plane on an adjacent plane or the force that resists a twisting action
static fatigue \stăt′ k fa-tēg\: the delayed failure of glass and ceramic materials resulting from stress enhanced chemical reactions aided by water vapor acting on surface cracks. Analogous to stress corrosion occurring in metals
k fa-tēg\: the delayed failure of glass and ceramic materials resulting from stress enhanced chemical reactions aided by water vapor acting on surface cracks. Analogous to stress corrosion occurring in metals
strain \strān\: change in length per unit length when stress is applied; the change in length/original length
stress \str s\ n (14c): force per unit area; a force exerted on one body that presses on, pulls on, pushes against, or tends to invest or compress another body; the deformation caused in a body by such a force; an internal force that resists an externally applied load or force. It is normally defined in terms of mechanical stress, which is the force divided by the perpendicular cross sectional area over which the force is applied—see COMPRESSIVE S., SHEARING S., TENSILE S.
s\ n (14c): force per unit area; a force exerted on one body that presses on, pulls on, pushes against, or tends to invest or compress another body; the deformation caused in a body by such a force; an internal force that resists an externally applied load or force. It is normally defined in terms of mechanical stress, which is the force divided by the perpendicular cross sectional area over which the force is applied—see COMPRESSIVE S., SHEARING S., TENSILE S.
tensile stress \t n′sal, -s
n′sal, -s l str
l str s\: the internal induced force that resists the elongation of a material in a direction parallel to the direction of the stresses
s\: the internal induced force that resists the elongation of a material in a direction parallel to the direction of the stresses
ten·sion \t n′shun\ n (1533): the state of being stretched, strained, or extended
n′shun\ n (1533): the state of being stretched, strained, or extended
tough·ness \tŭf′n s\ n: the ability of a material to withstand stresses and strains without breaking
s\ n: the ability of a material to withstand stresses and strains without breaking
tox·ic·i·ty \t k-s
k-s s′
s′ -tē\ n: the adverse reactions (dose-response-time relationships) of tissues to selected foreign substances resulting in unacceptable in-vivo interactions. The toxicity can be at the local or systemic level depending on the amount, rate of release, and specific type of substance available to the tissues
-tē\ n: the adverse reactions (dose-response-time relationships) of tissues to selected foreign substances resulting in unacceptable in-vivo interactions. The toxicity can be at the local or systemic level depending on the amount, rate of release, and specific type of substance available to the tissues
ultimate strength \ŭl′ta-m t\: the greatest stress that may be induced in a material at the point of rupture—called also ultimate tensile strength
t\: the greatest stress that may be induced in a material at the point of rupture—called also ultimate tensile strength
yield strength \yēld str ngkth\: the strength at which a small amount of permanent (plastic) strain occurs, usually 0.1% or 0.2%, and most frequently measured in MPa or psi
ngkth\: the strength at which a small amount of permanent (plastic) strain occurs, usually 0.1% or 0.2%, and most frequently measured in MPa or psi
STUDY QUESTIONS
1 Christensen GJ. The use of porcelain-fused-to-metal restorations in current dental practice: a survey. J Prosthet Dent. 1986;56:1.
2 Seymour KG, et al. Metal ceramic crowns—a review of tooth preparation. Eur J Prosthodont Restor Dent. 1999;7:79.
3 Walton TR. A 10-year longitudinal study of fixed prosthodontics: clinical characteristics and outcome of single-unit metal-ceramic crowns. Int J Prosthodont. 1999;12:519.
4 Oh W-S, et al. Factors affecting enamel and ceramic wear: a literature review. J Prosthet Dent. 2002;87:451.
5 Pintado MR, et al. Variation in tooth wear in young adults over a two-year period. J Prosthet Dent. 1997;77:313.
6 Monasky GE, Taylor DF. Studies on the wear of porcelain, enamel, and gold. J Prosthet Dent. 1971;25:299.
7 Ekfeldt A, Øilo G. Occlusal contact wear of prosthodontic materials. Acta Odontol Scand. 1988;46:159.
8 Kelly JR, et al. Ceramics in dentistry: historical roots and current perspectives. J Prosthet Dent. 1996;75:18.
9 Hacker CH, et al. An in vitro investigation of the wear of enamel on porcelain and gold in saliva. J Prosthet Dent. 1996;75:14.
10 Ramp MH, et al. Evaluation of wear: enamel opposing three ceramic materials and a gold alloy. J Prosthet Dent. 1997;77:523.
11 Al-Wahadni AM, Martin DM. An in vitro investigation into the wear effects of glazed, unglazed and refinished dental porcelain on an opposing material. J Oral Rehabil. 1999;26:538.
12 Magne P, et al. Wear of enamel and veneering ceramics after laboratory and chairside finishing procedures. J Prosthet Dent. 1999;82:669.
13 Clelland NL, et al. Relative wear of enamel opposing low-fusing dental porcelain. J Prosthodont. 2003;12:168.
14 Marker JC, et al. The compressive strength of nonprecious versus precious ceramometal restorations with various frame designs. J Prosthet Dent. 1986;55:560.
15 Terada Y, et al. The influence of different thicknesses of dentin porcelain on the color reflected from thin opaque porcelain fused to metal. Int J Prosthodont. 1989;2:352.
16 Craig RG, et al. Stress distribution in porcelain-fused-to-gold crowns and preparations constructed with photoelastic plastics. J Dent Res. 1971;50:1278.
17 Warpeha WS, Goodkind RJ. Design and technique variables affecting fracture resistance of metal-ceramic restorations. J Prosthet Dent. 1976;35:291.
18 Moon PC, Modjeski PJ. The burnishability of dental casting alloys. J Prosthet Dent. 1976;36:404.
19 Valega TM, editor. Alternatives to Gold Alloys in Dentistry [DHEW Publication No. (NIH) 77-1227]. Washington, DC: U.S. Department of Health, Education, and Welfare, 1977.
20 Carr AB, Brantley WA. New high-palladium casting alloys. I. Overview and initial studies. Int J Prosthodont. 1991;4:265.
21 American Dental Association Report. Classification system for cast alloys. J Am Dent Assoc. 1984;109:838.
22 Fuys RA, et al. Precipitation hardening in gold-platinum alloys containing small quantities of iron. J Biomed Mater Res. 1973;7:471.
23 Civjan S, et al. Further studies on gold alloys used in fabrication of porcelain-fused-to-metal restorations. J Am Dent Assoc. 1975;90:659.
24 Vermilyea SG, et al. Observations on gold-palladium-silver and gold-palladium alloys. J Prosthet Dent. 1980;44:294.
25 Moya F, et al. Experimental observation of silver and gold penetration into dental ceramic by means of a radiotracer technique. J Dent Res. 1987;66:1717.
26 Anusavice KJ, et al. Interactive effect of stress and temperature on creep of PFM alloys. J Dent Res. 1985;64:1094.
27 Papazoglou E, et al. Porcelain adherence to high-palladium alloys. J Prosthet Dent. 1993;70:386.
28 Goodacre CJ. Palladium-silver alloys: a review of the literature. J Prosthet Dent. 1989;62:34.
29 Herø H, Syverud M. Carbon impurities and properties of some palladium alloys for ceramic veneering. Dent Mater. 1985;1:106.
30 Byrne G, et al. Casting accuracy of high-palladium alloys. J Prosthet Dent. 1986;55:297.
31 Papazoglou E: On porcelain bonding, oxidation, mechanical properties and high-temperature distortion of high-palladium dental casting alloys. PhD dissertation, The Ohio State University, 1999.
32 Papazoglou E, et al. Comparison of mechanical properties for equiaxed fine-grained and dendritic high-palladium alloys. J Mater Sci Mater Med. 2000;11:601.
33 Wu Q, et al. Heat-treatment behavior of high-palladium dental alloys. Cells Mater. 1997;7:161.
34 Cai Z, et al. Transmission electron microscopic investigation of high-palladium dental casting alloys. Dent Mater. 1997;13:365.
35 Brantley WA, et al. X-ray diffraction studies of oxidized high-palladium alloys. Dent Mater. 1996;12:333.
36 Brantley WA, et al. X-ray diffraction studies of as-cast high-palladium alloys. Dent Mater. 1995;11:154.
37 Syverud M, et al. A new dental Pd-Co alloy for ceramic veneering. Dent Mater. 1987;3:102.
38 Papazoglou E, et al. Evaluation of high-temperature distortion of high-palladium metal-ceramic crowns. J Prosthet Dent. 2001;85:133.
39 Cai Z, et al. On the biocompatibility of high-palladium dental alloys. Cells Mater. 1995;5:357.
40 Wataha JC, Hanks CT. Biological effects of palladium and risk of using palladium in dental casting alloys. J Oral Rehabil. 1996;23:309.
41 Sun D: On the corrosion behavior and biocompatibility of palladium-based dental alloys. PhD dissertation, The Ohio State University, 2004.
42 Sun D, et al. Influence of palladium alloy elements on cell proliferation and viability [Abstract no. 2698]. J Dent Res. 84(Special Issue A on CD-ROM), 2005.
43 Sun D, et al. Elemental release from palladium casting alloys to cell culture media [Abstract no. 2699]. J Dent Res. 84(Special Issue A on CD-ROM), 2005.
44 Sun D, et al. Initial biocompatibility evaluation of two palladium-based alloys and a high-gold alloy from animal study [Abstract no. 131]. J Dent Res. 82(Special Issue A on CD-ROM), 2003.
45 Baran GR. The metallurgy of Ni-Cr alloys for fixed prosthodontics. J Prosthet Dent. 1983;50:639.
46 O’Connor RP, et al. Castability, opaque masking, and porcelain bonding of 17 porcelain-fused-to-metal alloys. J Prosthet Dent. 1996;75:367.
47 American Dental Association, Council on Dental Materials, Instruments, and Equipment. Biological effects of nickel-containing dental alloys. J Am Dent Assoc. 1982;104:501.
48 Moffa JP, et al. An evaluation of nonprecious alloys for use with porcelain veneers. II. Industrial safety and biocompatibility. J Prosthet Dent. 1973;30:432.
49 Bumgardner JD, Lucas LC. Corrosion and cell culture evaluations of nickel-chromium dental casting alloys. J Appl Biomater. 1994;5:203.
50 Vermilyea SG, et al. Observations on nickel-free, beryllium-free alloys for fixed prostheses. J Am Dent Assoc. 1983;106:36.
51 Barakat MM, Asgar K. Mechanical properties and soldering of some cobalt base metal alloys. Dent Mater. 1986;2:272.
52 Waterstrat RM. Comments on casting of Ti-13Cu-4.5Ni alloy. In: Valega TM, editor. Alternatives to Gold Alloys in Dentistry. Washington, DC: U.S. Department of Health, Education, and Welfare; 1977:224-233. [DHEW Publ. No. (NIH) 77-1227]
53 Donachie MJJr. Titanium: A Technical Guide. Metals Park, OH: ASM International, 1988;31. 444
54 Kobayashi E, et al. Mechanical properties and corrosion resistance of Ti-6Al-7Nb alloy dental castings. J Mater Sci Mater Med. 1998;9:567.
55 Wang TJ, et al. Castability of Ti-6Al-7Nb alloy for dental casting. J Med Dent Sci. 1999;46:13.
56 Iijima D, et al. Wear properties of Ti and Ti-6Al-7Nb castings for dental prostheses. Biomaterials. 2003;24:1519.
57 Taira M, et al. Studies of Ti alloys for dental castings. Dent Mater. 1989;5:45.
58 Takahashi J, et al. Effect of casting methods on castability of pure titanium. Dent Mater J. 1993;12:245.
59 Eliopoulos D, et al. Porosity of cpTi casting with four different casting machines. J Prosthet Dent. 2004;92:377.
60 Koike M, et al. Corrosion behavior of cast titanium with reduced surface reaction layer made by a face-coating method. Biomaterials. 2003;24:4541.
61 Luo XP, et al. Titanium casting into phosphate bonded investment with zirconite. Dent Mater. 2002;18:512.
62 Takahashi J, et al. Casting pure titanium into commercial phosphate-bonded SiO2 investment molds. J Dent Res. 1990;69:1800.
63 Watanabe I, et al. Effect of pressure difference on the quality of titanium casting. J Dent Res. 1997;76:773.
64 Syverud M, Herø H. Mold filling of Ti castings using investments with different gas permeability. Dent Mater. 1995;11:14.
65 Herø H, et al. Mold filling and porosity in casting of titanium. Dent Mater. 1993;9:15.
66 Hung CC, et al. Pure titanium casting into zirconia-modified magnesia-based investment molds. Dent Mater. 2004;20:846.
67 Zinelis S. Effect of pressure of helium, argon, krypton, and xenon on the porosity, microstructure, and mechanical properties of commercially pure titanium castings. J Prosthet Dent. 2000;84:575.
68 Knoop F, et al. A sensitive pyramidal diamond tool for indentation measurements. J Res Nat Bur Stand. 1939;12:39.