31 LUTING AGENTS AND CEMENTATION PROCEDURES
INTERIM CEMENTATION
On many occasions, cementing a restoration on an interim basis is advised so that the patient and dentist can assess its appearance and function over a time longer than a single visit. However, these trial cementations should be managed cautiously. On one hand, removing the restoration for definitive cementation may be difficult, even when temporary zinc oxide–eugenol (ZOE) cement is used. To avoid this problem, the interim cement can be mixed with a little petrolatum or silicone grease. The modified luting agent is applied only to the margins of the restoration to seal them and yet allow subsequent removal without difficulty. On the other hand, an interim cemented restoration may come loose during function. If a single unit is displaced, it can be embarrassing or uncomfortable for the patient. If one abutment of a partial fixed dental prosthesis (FDP) becomes loose, the consequences can be more severe. If the patient does not return promptly for recementation, caries may develop very rapidly. Interim cementation should not be undertaken unless the patient is given clear instructions about the objectives of the procedure, the intended duration of the trial cementation, and the importance of returning if an abutment loosens. If removing an interim cemented fixed prosthesis is difficult, the use of a crown-removal device such as the CORONAflex* (see Chapter 32) is recommended.
DEFINITIVE CEMENTATION
Conventional Cast Restorations
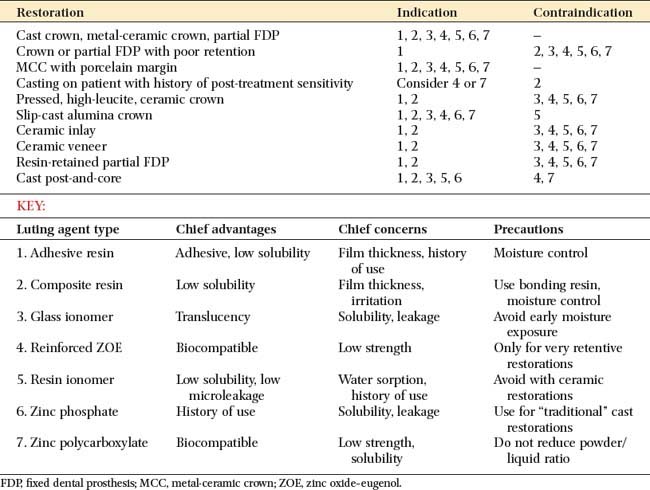
Definitive cementation often does not receive the same attention to detail as do other aspects of restorative dentistry. Careless luting agent selection can result in margin discrepancies and improper occlusion and may even necessitate cutting the restoration from the patient’s mouth and making a new one. The choice of cement depends first on whether a conventional casting or an adhesively bonded restoration, such as a ceramic inlay or resin-bonded partial FDP, is to be cemented. Traditional dental cements can be used for cast crowns and FDPs, but not where adhesion is needed. Adhesive resins are necessary for some restorations, but they can be difficult to use; in addition, there are no long-term data to justify more general use with conventional castings.
Dental Cements
Most luting agents traditionally used for cast restorations are dental cements (Fig. 31-1). These consist of an acid combined with a metal oxide base to form a salt and water. The setting mechanism results from the binding of unreacted powder particles by a matrix of salt to harden the mass. However, because they are ionic, these agents are susceptible to acid attack and are therefore somewhat soluble in oral fluids.1-4 Traditionally, the success of restorations cemented with these luting agents has been attributed to excellent adaptation between the casting and the prepared tooth. In vitro, however, cement dissolution is independent of the marginal width up to a certain critical value. After that, it increases only slightly, which is explained by Fick’s first law of diffusion.*5
This study and others identify dissolution (rather than physical disintegration) as the mechanism for cement erosion.6 They explain the success of cast restorations, despite the prevalence of relatively large subgingival marginal discrepancies, which are difficult to detect even at 0.1 mm.7
Zinc phosphate cement
This traditional luting agent continues to be popular for cast restorations. It has adequate strength, a film thickness (thickness of the layer) of about 25 μm (Fig. 31-2) (which is within the tolerance limits required for making cast restorations),8 and a reasonable working time. Excess material can be easily removed.
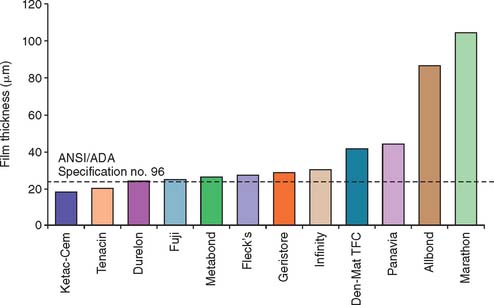
Fig. 31-2 The film thickness of a range of luting agents was tested according to American Dental Association (ADA) specification No. 8 for zinc phosphate cement (now American National Standards Institute [ANSI]/ADA specification No. 96) by White and Yu.64 Some of the adhesive materials possessed unacceptably high film thicknesses, which may translate into clinical problems for complete restoration seating.
(From Rosenstiel SF, et al: Dental luting agents: a review of the current literature. J Prosthet Dent 80:280, 1998.)
The toxic effects of zinc phosphate, or, more specifically, phosphoric acid, are well documented.9 However, the success of the use of this material over many years suggests that its effect on the dental pulp is clinically acceptable as long as normal precautions are taken and the preparation is not too close to the pulp.
Zinc polycarboxylate cement
One advantage of this luting agent is its relative biocompatibility,10 which may stem from the fact that the polyacrylic acid molecule is large and therefore does not penetrate into the dentinal tubule.
Zinc polycarboxylate cement also exhibits specific adhesion to tooth structure because it chelates the calcium (although it has no adhesion to gold castings). Because of its high viscosity, zinc polycarboxylate cement can be difficult to mix, but this problem can be overcome by using encapsulated products.*
In clinical trials, polycarboxylate performs as well as or slightly better than zinc phosphate.11,12 However, dentists have reported varying success rates, and claims of inferior long-term retention have been made. These problems may be related to the powder/liquid ratio. At manufacturers’ recommended powder/liquid ratios, mixed polycarboxylate cement is very viscous. Some dentists may prefer a more fluid working consistency for reliable seating during cementation. However, polycarboxylate cements have different rheologic or flow properties than zinc phosphate, exhibiting thinning with increased shear rate.13 This means that they are capable of forming low film thicknesses despite their viscous appearance. When the dentist unnecessarily reduces the powder/liquid ratio, the solubility (how susceptible something is to being dissolved) of the cement increases dramatically (as much as threefold).14 This may be the cause of increased clinical failures. By fabricating luting agents, including polycarboxylate in encapsulated form, manufacturers have reduced problems arising from manipulative variables.
The working time of polycarboxylate is much shorter than that of zinc phosphate (about 2.5 minutes, in comparison with 5 minutes). This may be a problem when multiple units are being cemented. Residual zinc polycarboxylate is more difficult to remove than zinc phosphate, and there is some evidence15,16 that it provides less crown retention than zinc phosphate (Fig. 31-3). Its selection therefore should probably be limited to restorations with good retention and resistance form where minimum pulp irritation is wanted. Its use as a base material and to block out minor undercuts in preparations on vital teeth may also be worth considering.
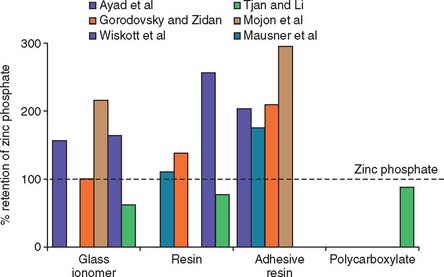
Fig. 31-3 Crown retention studies. Effect of luting agent. These six in vitro studies evaluated the effect of luting agent on crown retention. The data were normalized as a percentage of the retention value with zinc phosphate cement. Adhesive resins had consistently greater retention than zinc phosphate. Conventional resins and glass ionomers yielded less consistent results.
(From Rosenstiel SF, et al: Dental luting agents: a review of the current literature. J Prosthet Dent 80:280, 1998.)
Glass ionomer cement
This cement adheres to enamel and dentin and exhibits good biocompatibility. In addition, because it releases fluoride,17,18 it may have an anticariogenic effect, although this has not been documented clinically.19 The set cement is somewhat translucent, which is an advantage when it is used with the porcelain labial margin technique (see Chapter 24).
The mechanical properties of glass ionomer cement are generally superior compared with zinc phosphate or polycarboxylate cements (Fig. 31-4). A disadvantage is that during setting, glass ionomer appears particularly susceptible to moisture contamination20 and should be protected with a foil or resin coat or by leaving a band of cement undisturbed for 10 minutes.21 The water changes the setting reaction of the glass ionomer as cement-forming cations are flushed away and water is absorbed, which leads to erosion.22 Nevertheless, zinc phosphate has also demonstrated significant early erosion when exposed to moisture.18 Glass ionomers should not be allowed to desiccate during this critical initial setting period. The newer resin-modified glass ionomers are less susceptible to early moisture.23
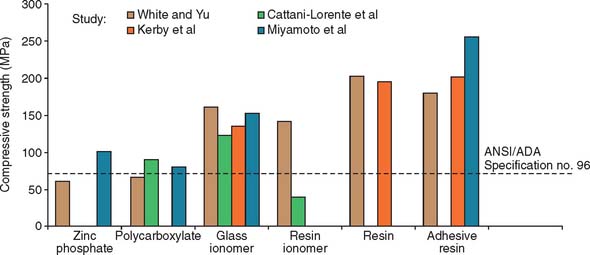
Fig. 31-4 Compressive strength of luting agents. Higher strength values were reported in these studies with the resin cements and glass ionomers than with zinc phosphate or polycarboxylate. Resin-modified glass ionomer exhibited greater variation than did other cements.
(From Rosenstiel SF, et al: Dental luting agents: a review of the current literature. J Prosthet Dent 80:280, 1998.)
Although glass ionomers have been reported to cause sensitivity,24 there appears to be little pulpal response at the histologic level,25 particularly if the remaining dentin thickness exceeds 1 mm.26 Side effects such as posttreatment sensitivity thought to result from a lack of biocompatibility may actually be a result of desiccation or bacterial contamination27 of the dentin rather than irritation by the cement. The anecdotal reports that glass ionomer causes more post-treatment sensitivity have not been replicated in clinical trials. Authors have reported little association between the choice of zinc phosphate or glass ionomer cement and increased pulpal sensitivity, provided that manufacturers’ recommendations were followed28-30 (Fig. 31-5). If postcementation sensitivity becomes a problem, dentists should carefully evaluate their technique, particularly avoiding desiccation of the prepared dentin surface.31 Resin-modified glass ionomer materials have been reported to provoke less posttreatment sensitivity. Again, this information is anecdotal and has not been confirmed by clinical research.32 A desensitizing agent may prevent sensitivity, although it may also reduce retention, at least with some luting cements.16,33 Some formulations of glass ionomer and resin cements are radiolucent (Fig. 31-6), which may prevent the practitioner from distinguishing between a cement line and recurrent caries, as well as detecting cement overhangs.34 The use of a glass ionomer luting agent in general practice has been favorable35; however, any reduction in caries activity that might be anticipated by the fluoride content has not been demonstrated by clinical research.36
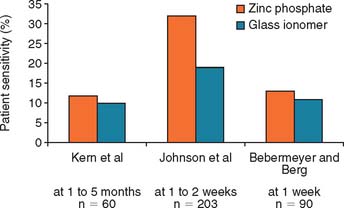
Fig. 31-5 Clinical trials28-30 that evaluated postcementation sensitivity of patients with crowns cemented with zinc phosphate or glass ionomer cement. Contrary to anecdotal evidence, those with glass ionomer cemented crowns did not exhibit increased postcementation sensitivity.
(From Rosenstiel SF, et al: Dental luting agents: a review of the current literature. J Prosthet Dent 80:280, 1998.)
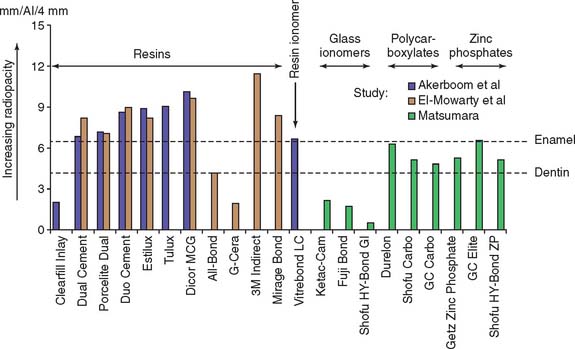
Fig. 31-6 Radiopacity of luting agents. In three in vitro studies, investigators compared the radiographic appearance of various luting agents to aluminum. The data were normalized to account for different specimen thicknesses used by the investigators. Excess luting agent is more difficult to detect if materials with lower values are chosen. In addition, margin gaps and recurrent caries are more difficult to diagnose.
(From Rosenstiel SF, et al: Dental luting agents: a review of the current literature. J Prosthet Dent 80:280, 1998.)
Zinc oxide–eugenol with and without ethoxybenzoic acid
Reinforced ZOE cement is extremely biocompatible and provides an excellent seal. However, its physical properties are generally inferior to those of other cements, which limits its use.37 In terms of compressive strength, solubility, and film thickness, another luting agent (e.g., zinc phosphate) should be used. The ethoxybenzoic acid (EBA) modifier replaces a portion of the eugenol in conventional ZOE cement, although the change improves compressive strength without affecting its resistance to deformation; the cement should be used only in restorations with good inherent retention form in which emphasis is on biocompatibility and pulpal protection. The EBA cement has a relatively short working time, and excess material is difficult to remove.
Resin-modified glass ionomer luting agents
Resin-modified glass ionomers* were introduced in the 1990s in an attempt to combine some of the desirable properties of glass ionomer (i.e., fluoride release and adhesion) with the higher strength and low solubility of resins. These materials are less susceptible to early moisture exposure38 than is glass ionomer and are currently among the most popular materials in general practice. The materials exhibit higher strength than the conventional cements; values are similar to the resin luting agents.39 Resin-modified glass ionomers should be avoided with all-ceramic restorations because some brands have been associated with fracture,40,41 which is probably caused by their water absorption and expansion.42
Resin luting agents
Unfilled resins have been used for cementation since the 1950s. Because of their high polymerization shrinkage and poor biocompatibility, these early products were unsuccessful, although they had very low solubility. Composite resin cements with greatly improved properties were developed for resin-bonded prostheses (see Chapter 26) and are used extensively for the bonded ceramic technique (see Chapter 25). Resin cements are available with adhesive properties (i.e., they are capable of bonding chemically to dentin).43 Bonding is usually achieved with organophosphonates, hydroxyethyl methacrylate (HEMA), or 4-methacryloxyethyl-trimellitic anhydride (4-META).44 These developments, and their lack of solubility, have rekindled an interest in the use of resin cements for crowns and conventional fixed prostheses (Fig. 31-7). Resin luting agents are less biocompatible than cements such as glass ionomer, especially if they are not fully polymerized. They also tend to have greater film thickness.45
Choice of Luting Agent
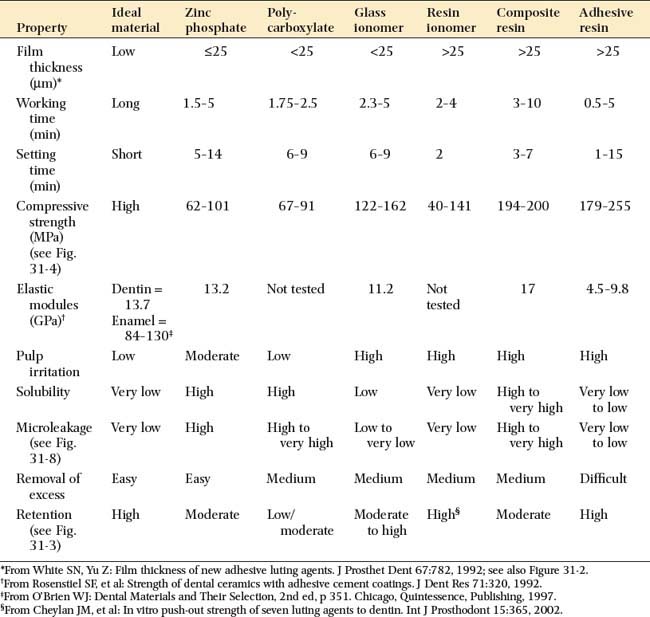
An ideal luting agent has a long working time, adheres well to both tooth structure and cast alloys, provides a good seal, is nontoxic to the pulp, has adequate strength properties, is compressible into thin layers, has low viscosity and solubility, and exhibits good working and setting characteristics. In addition, any excess can be easily removed. Unfortunately, no such product exists (Tables 31-1 and 31-2).
Zinc phosphate cement
Despite its limited biocompatibility in terms of pulp irritation, zinc phosphate has a long history, and its limitations are well documented. This factor is important for cast restorations, which should be designed for long-term service. Zinc phosphate cement is probably still the luting agent of choice for otherwise normal, conservatively prepared teeth. Cavity varnish can be used to protect against pulp irritation from phosphoric acid and appears to have little effect on the amount of retention of the cemented restorations.46 In addition, crowns cemented with zinc phosphate displayed increased resistance to dislodgment on preparations lacking resistance form.31
Zinc polycarboxylate cement
This agent is recommended on retentive preparations when minimal pulp irritation is important (e.g., in children with large pulp chambers).
Glass ionomer cement
This has become a popular cement for luting cast restorations. It has good working properties, and is more translucent than zinc phosphate.
Resin-modified glass ionomer luting agents
Currently among the most popular luting agents, resin-modified glass ionomer luting agents have low solubility, adhesion, and low microleakage* (Fig. 31-8). The popularity of these materials is derived mainly from the perceived benefit of reduced postcementation sensitivity, although this benefit has not been confirmed in clinical studies.47
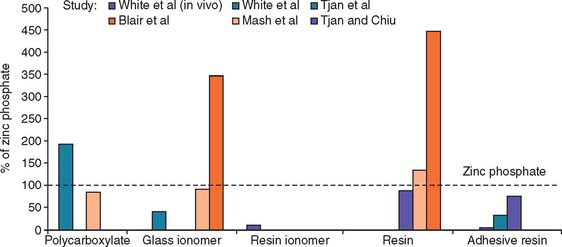
Fig. 31-8 Microleakage of luting agent. A comparison of data from one clinical study and five laboratory studies, expressed as a percentage of the value obtained for zinc phosphate cement. Considerable variation was reported, with adhesive resins and resin-modified glass ionomer exhibiting low microleakage values.
(From Rosenstiel SF, et al: Dental luting agents: a review of the current literature. J Prosthet Dent 80:280, 1998.)
Adhesive resins
Long-term evaluations of these materials are not yet available, and so they cannot be recommended for routine use; however, they are indicated for all-ceramic and laboratory-processed composite restorations. Laboratory testing yields high retention strength values,48 but there is concern that stresses caused by polymerization shrinkage, magnified in thin films,49 lead to marginal leakage. Adhesive resin may be indicated when a casting has become displaced through lack of retention, and is recommended for all-ceramic restorations.50
Preparation of the Restoration and Tooth Surface for Cementation
The performance of all luting agents is degraded if the material is contaminated with water, blood, or saliva. Therefore, the restoration and tooth must be carefully cleaned and dried after the evaluation procedure, although excessive drying of the tooth must be avoided to prevent damage to the odontoblasts (Fig. 31-9). The casting is best prepared by airborne particle abrading the fitting surface with 50-μm alumina. This should be done carefully to avoid abrading the polished surfaces or margins. Air abrasion has increased the in vitro retention of castings by 64%.51 Alternative cleaning methods include steam cleaning, the use of ultrasonics, and the use of organic solvents.
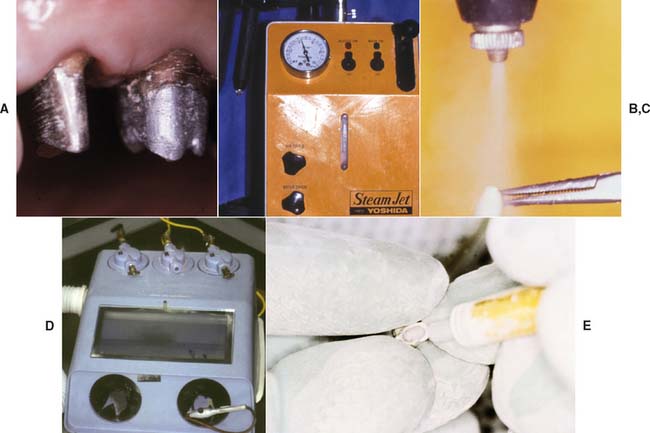
Fig. 31-9 Teeth and restorations must be carefully prepared immediately before cementation. A, These preparations need to be cleaned of interim luting agent and dried but not excessively desiccated. B and C, A steam cleaner is convenient for removing traces of polishing compound from the restorations. D and E, Airborne particle abrasion of internal restoration surface.
Before the initiation of cement mixing, isolating the area of cementation and cleaning and drying the tooth is mandatory. However, the tooth should never be excessively desiccated. Overdrying the prepared tooth leads to postoperative sensitivity. (The techniques for moisture control, essential to proper cementation, are described in Chapter 14.) If a nonadhesive cement (zinc phosphate) is to be used, the tooth should be cleaned,* gently dried, and coated with cavity varnish or dentin-bonding resin.
Armamentarium (Fig. 31-10)
Step-by-step procedure
Zinc phosphate cement is used to illustrate a typical procedure, but the steps may vary slightly, depending on the cement chosen (Fig. 31-11). The differences with glass ionomer are pointed out in the description.

Fig. 31-11 Cementation technique with zinc phosphate. A, Armamentarium. B, The powder is divided into small increments and mixed with liquid on a cool slab over a large surface area. C, The consistency of the mix is evaluated by pulling out a “thread” of cement. The thread should break at about 20 mm (¾ inch). D and E, The internal surface of the restoration is coated. F, The restoration is seated. G and H, Applying an orangewood stick with a rocking motion against the restoration ensures that all excess cement is expressed. I, Complete seating is immediately verified with an explorer. J, When set, the excess cement is removed from around the margins. A length of dental floss with a knot tied in it is useful for removing excess interproximally. K, Cemented restoration.
(A to C, Courtesy of Dr. J. H. Bailey; G, Reprinted from Campagni WV: The final touch in the delivery of a fixed prosthesis. CDA J 12(2):21, 1984.)
A paper mixing pad is generally used with glass ionomer or polycarboxylate cements.
Frozen slab technique
This technique is a practical way to increase the working time and reduce the setting time of zinc phosphate cement.52,53 The technique entails the use of a 50% increased powder/liquid ratio, and mixing is performed on a frozen glass slab. There is no adverse effect on compressive strength, solubility, or retention,54,55 and the pH rise in the cement may be accelerated.56 The frozen slab technique is reliable and is particularly effective when multiple castings are to be cemented. An incidental advantage of this technique is that the condensation that forms on the glass slab facilitates cleanup of excess cement. Reduced-temperature mixing has also been applied to glass ionomer cements as a way of increasing working time and increasing powder/liquid ratio. Film thickness measurements suggest that the procedure is beneficial.57
The consistency of properly mixed glass ionomer is noticeably more viscous than zinc phosphate, but the material thins out with seating pressure.
Resin luting agents
Resin luting agents are available in a wide range of formulations. These can be categorized on the basis of polymerization method (chemical-cure, light-cure, or dual-cure) and the presence of dentin-bonding mechanisms. Metal restorations require a chemically cured system, whereas a light- or dual-cure system is appropriate with ceramics. Resins formulated for cementing conventional castings must have lower film thickness than materials designed for ceramics or orthodontic brackets. However, this may be achieved at the expense of filler particle content and will adversely affect other properties such as polymerization shrinkage.
Manipulative techniques vary widely, depending on the brand of resin cement. For example, Panavia EX* sets very rapidly when air is excluded. The directions call for the material to be spatulated in a thin film. It sets rapidly if piled up on the mixing pad. Another material, C&B-MetaBond,† is mixed in a ceramic well that must be chilled to prevent premature setting. Mixing techniques for these materials are illustrated in Figures 31-12 and 31-13.

Fig. 31-12 Cementation with C&B-MetaBond resin cement. A, The brush-on separating film is applied to the prosthesis, the proximal teeth, in order to prevent the adhesive from bonding where it is not wanted. B, The recommended dentin conditioner is applied for 10 seconds and rinsed off, and the tooth is dried. C and D, Four drops of base and 1 drop of catalyst are mixed for each crown. E, After the preparation and interior of the crown are wetted with this mixed liquid, the powder is added (F). G, The casting is painted, and the crown is seated. H, Excess resin is removed after it has completely set. Cleanup is greatly facilitated when the separating film is used. It is important not to remove resin before it has fully set, because the rubbery material will pull away from the margins.
(Courtesy of Parkell Products, Farmingdale, New York.)
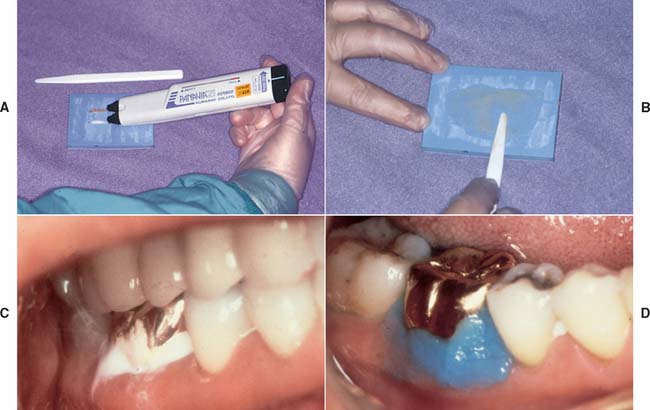
Fig. 31-13 A, Panavia resin cement. B, Measured powder and liquid are spatulated for 60 to 90 seconds. The mix becomes creamier as it is mixed. The cement sets if oxygen is excluded, and so it should not be piled up. Instead, it should be spread out over a large surface area. C, A thin coat of the cement is applied, the restoration is seated, and excess cement is removed. D, The cement is coated with oxygen-inhibiting gel to promote polymerization.
(Courtesy of J. Morita USA, Inc., Irvine, California.)
CEMENTATION PROCEDURES FOR CERAMIC VENEERS AND INLAYS
These restorations rely on resin bonding for retention and strength. The cementation steps are critical to the restoration’s success; careless handling of the resin luting agent may be a key factor in their prognosis. Bonding is achieved by performing the following steps:
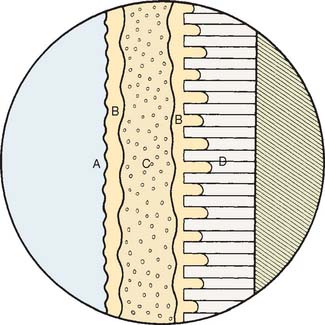
Fig. 31-14 Schematic of resin-bonding technique. A, Ceramic surface (etched and silanated); B, unfilled resin; C, resin luting agent; D, etched enamel.
The etching and silanating steps are presented in Chapter 25.
Selection of Resin Luting Agent
Composite resin luting agents are available in a range of formulations. For veneers, a light-cured material can be used. For inlays, a chemical-cure material is preferred, to ensure maximum polymerization of the resin in the less accessible proximal areas. In clinical testing, restorations luted with chemically cured materials have performed better than dual-cured luted restorations.61
The shade of veneers can be modified by the shade of the luting agent. To facilitate shade selection, color-matched try-in pastes are available from some manufacturers (e.g., Nexus*).
Bonding the Restoration
Armamentarium (Fig. 31-15)
Step-by-step procedure (Fig. 31-16)
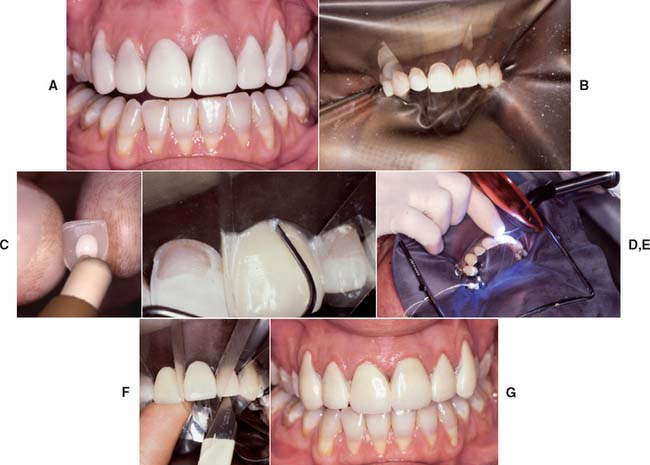
Fig. 31-16 Evaluation and bonding procedure. A, The veneers are evaluated very carefully. A drop of glycerin on the fitting surface aids in shade assessment and provides retention. If necessary, the shade can be modified slightly with colored luting agents. The luting agent must not polymerize during evaluation; in particular, the unit light must not shine directly on the restoration. The veneers are thoroughly cleaned in acetone and are silanated according to the manufacturer’s recommendations. B, The teeth are isolated, pumiced, and etched. Mylar strips are placed between adjacent teeth. C, The veneers are filled with resin luting agent and gently seated. D, Excess resin is removed with an explorer. E, The resin is polymerized. F, Gross excess resin is trimmed with a scalpel, and the margins are finished with fine-grit diamonds and diamond polishing paste. G, The completed restorations.
REVIEW OF TECHNIQUE
Figure 31-17 illustrates the cementation of six maxillary anterior metal-ceramic crowns.
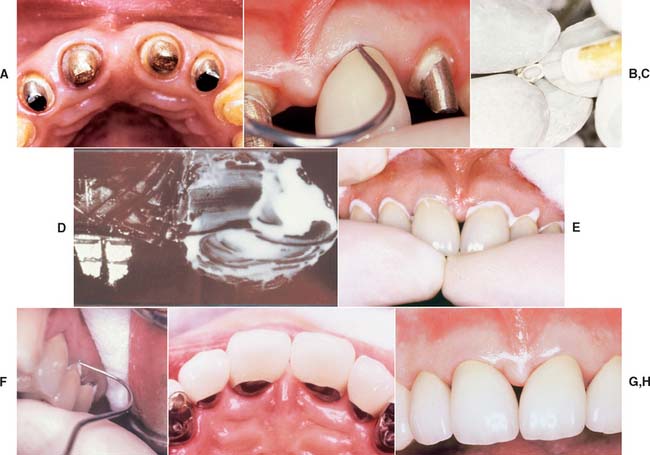
Fig. 31-17 Technique review. A, The preparations are thoroughly cleaned; all interim luting agent should be removed. B, The restorations are seated, and a readily accessible area of the margin is examined with an explorer. C, The restorations are thoroughly cleaned with airborne particle abrasion, steam cleaning, or ultrasonics. D, The luting agent is mixed according to the manufacturer’s recommendations. E, The restorations are seated to place with a firm rocking pressure. F, The accessible margin area is quickly reexamined to ensure complete seating. G and H, Once the luting agent has completely set, all excess is removed.
SUMMARY
Proper moisture control is essential for the cementation step. The restoration must be carefully prepared for cementation, including the removal of all polishing compounds. Airborne particle abrading the fitting surface is recommended. The luting agent of choice is mixed according to the manufacturer’s recommendations, and the restoration is seated, with the use of a rocking action. The luting agent must be protected from moisture during its initial set. Removal of excess luting agent from the gingival sulcus is crucial for continued periodontal health.
Additional steps are necessary for adhesively bonded restorations. These steps must be carefully sequenced according to the manufacturer’s directions.
 GLOSSARY*
GLOSSARY*
ac·cel·er·a·tor \ăk-s l′a-rāter\ n (1611) 1: a substance that speeds a chemical reaction 2: in physiology, a nerve, muscle, or substance that quickens movement or response
l′a-rāter\ n (1611) 1: a substance that speeds a chemical reaction 2: in physiology, a nerve, muscle, or substance that quickens movement or response
ad·her·ence \ăd-hîr′ens, -enz\ n (1531): the act, quality, or action of adhering; persistent attachment
ad·he·sion \ăd-hē′zhun\ n (1624) 1: the property of remaining in close proximity, as that resulting from the physical attraction of molecules to a substance or molecular attraction existing between the surfaces of bodies in contact 2: the stable joining of parts to each other, which may occur abnormally 3: a fibrous band or structure by which parts abnormally adhere—comp CAPSULAR FIBROSIS, FIBROUS A., INTRACAPSULAR A., MYOFIBROTIC CAPSULAR CONTRACTURE
1ce·ment \s -m
-m nt\ n (14c) 1: a binding element or agent used as a substance to make objects adhere to each other, or something serving to firmly unite 2: a material that, on hardening, will fill a space or bind adjacent objects- syn luting agent
nt\ n (14c) 1: a binding element or agent used as a substance to make objects adhere to each other, or something serving to firmly unite 2: a material that, on hardening, will fill a space or bind adjacent objects- syn luting agent
ce·men·ta·tion \s ′m
′m n-tāshun\: 1: the process of attaching parts by means of cement 2: attaching a restoration to natural teeth by means of a cement (GPT-4)
n-tāshun\: 1: the process of attaching parts by means of cement 2: attaching a restoration to natural teeth by means of a cement (GPT-4)
com·po·mer \k m′pō-m
m′pō-m r\ n (1998): a poly-acid modified composite resin, composed of glass filler, acid-modified dimethacrylate resin, and a photo initiator
r\ n (1998): a poly-acid modified composite resin, composed of glass filler, acid-modified dimethacrylate resin, and a photo initiator
hybrid ionomer \hī′br d ī′a-noōm
d ī′a-noōm r\: a conventional glass ionomer that has been modified to include methacrylate groups in the liquid component. It may contain photoinitiators. Setting is by an acid-base reaction with light and dual cure polymerization syn—RESIN-MODIFIED GLASS IONOMER
r\: a conventional glass ionomer that has been modified to include methacrylate groups in the liquid component. It may contain photoinitiators. Setting is by an acid-base reaction with light and dual cure polymerization syn—RESIN-MODIFIED GLASS IONOMER
1lute \lōōt\ n (15c): a substance, such as cement or clay, used for placing a joint or coating a porous surface to make it impervious to liquid or gas—see CEMENT
luting agent: any material used to attach or cement indirect restorations to prepared teeth
STUDY QUESTIONS
1 Swartz ML, et al. In vitro degradation of cements: a comparison of three test methods. J Prosthet Dent. 1989;62:17.
2 Stannard JG, Sornkul E. Demineralization resistance and tensile bond strength of four luting agents after acid attack. Int J Prosthodont. 1989;2:467.
3 Dewald JP, et al. Evaluation of the interactions between amalgam, cement and gold castings. J Dent. 1992;20:121.
4 Knibbs PJ, Walls AW. A laboratory and clinical evaluation of three dental luting cements. J Oral Rehabil. 1989;16:467.
5 Jacobs MS, Windeler AS. An investigation of dental luting cement solubility as a function of the marginal gap. J Prosthet Dent. 1991;65:436.
6 Dupuis V, et al. Solubility and disintegration of zinc phosphate cement. Biomaterials. 1992;13:467.
7 Dedmon HW. Ability to evaluate nonvisible margins with an explorer. Oper Dent. 1985;10:6.
8 Phillips RW. Skinner’s Science of Dental Materials, 9th ed., Philadelphia: WB Saunders; 1991:491.
9 Langeland K, Langeland LK. Pulp reactions to crown preparation, impression, temporary crown fixation, and permanent cementation. J Prosthet Dent. 1965;15:129.
10 Going RE, Mitchem JC. Cements for permanent luting: a summarizing review. J Am Dent Assoc. 1975;91:107.
11 Dahl BL, et al. Clinical study of two luting cements used on student-treated patients: final report. Dent Mater. 1986;2:269.
12 Black SM, Charlton G. Survival of crowns and bridges related to luting cements. Restorative Dent. 1990;6:26.
13 Phillips RW. Skinner’s Science of Dental Materials, 9th ed., Philadelphia: WB Saunders; 1991:490.
14 Osborne JW, Wolff MS. The effect of powder/liquid ratio on the in vivo solubility of polycarboxylate cement. J Prosthet Dent. 1991;66:49.
15 Øilo G, Jørgensen KD. The influence of surface roughness on the retentive ability of two dental luting cements. J Oral Rehabil. 1978;5:377.
16 Mausner IK, et al. Effect of two dentinal desensitizing agents on retention of complete cast coping using four cements. J Prosthet Dent. 1996;75:129.
17 Swartz ML, et al. Long term F release from glass ionomer cements. J Dent Res. 1984;63:158.
18 Muzynski BL, et al. Fluoride release from glass ionomers used as luting agents. J Prosthet Dent. 1988;60:41.
19 Rosenstiel SF, et al. Dental luting agents: a review of the current literature. J Prosthet Dent. 1998;80:280.
20 Um CM, Øilo G. The effect of early water contact on glass-ionomer cements. Quintessence Int. 1992;23:209.
21 Curtis SR, et al. Early erosion of glass-ionomer cement at crown margins. Int J Prosthodont. 1993;6:553.
22 McLean JW. Glass-ionomer cements. Br Dent J. 1988;164:293.
23 Cho E, et al. Moisture susceptibility of resin-modified glass-ionomer materials. Quintessence Int. 1995;26:351.
24 Council on Dental Materials, Instruments, and Equipment, American Dental Association. Reported sensitivity to glass ionomer luting cements. J Am Dent Assoc. 1984;109:476.
25 Heys RJ, et al. An evaluation of a glass ionomer luting agent: pulpal histological response. J Am Dent Assoc. 1987;114:607.
26 Pameijer CH, et al. Biocompatibility of a glass ionomer luting agent. II. Crown cementation. Am J Dent. 1991;4:134.
27 Torstenson B. Pulpal reaction to a dental adhesive in deep human cavities. Endod Dent Traumatol. 1995;11:172.
28 Johnson GH, et al. Evaluation and control of post-cementation pulpal sensitivity: zinc phosphate and glass ionomer luting cements. J Am Dent Assoc. 1993;124:38.
29 Bebermeyer RD, Berg JH. Comparison of patient-perceived postcementation sensitivity with glass-ionomer and zinc phosphate cements. Quintessence Int. 1994;25:209.
30 Kern M, et al. Clinical comparison of postoperative sensitivity for a glass ionomer and a zinc phosphate luting cement. J Prosthet Dent. 1996;75:159.
31 Rosenstiel SF, Rashid RG. Postcementation hypersensitivity: scientific data versus dentists’ perceptions. J Prosthodont. 2003;12:73.
32 Proussaefs P. Crowns cemented on crown preparations lacking geometric resistance form. Part II: effect of cement. J Prosthodont. 2004;13:36.
33 Pameijer CH, et al. Influence of low-viscosity liners on the retention of three luting materials. Int J Periodontics Restorative Dent. 1992;12:195.
34 Goshima T, Goshima Y. Radiographic detection of recurrent carious lesions associated with composite restorations. Oral Surg. 1990;70:236.
35 Brackett WW, Metz JE. Performance of a glass ionomer luting cement over 5 years in a general practice. J Prosthet Dent. 1992;67:59.
36 Moura JS, et al. Effect of luting cement on dental biofilm composition and secondary caries around metallic restorations in situ. Oper Dent. 2004;29:509.
37 Silvey RG, Myers GE. Clinical study of dental cements. VI. A study of zinc phosphate, EBA-reinforced zinc oxide eugenol and polyacrylic acid cements as luting agents in fixed prostheses. J Dent Res. 1977;56:1215.
38 Cho E, et al. Moisture susceptibility of resin-modified glass-ionomer materials. Quintessence Int. 1995;26:351.
39 Piwowarczyk A, et al. Laboratory strength of glass ionomer cement, compomers, and resin composites. J Prosthodont. 2002;11:86.
40 Letters to the editor. Quintessence Int. 1996;27:655.
41 Leevailoj C, et al. In vitro study of fracture incidence and compressive fracture load of all-ceramic crowns cemented with resin-modified glass ionomer and other luting agents. J Prosthet Dent. 1998;80:699.
42 Knobloch L, et al. Solubility and sorption of resin-based luting cements. Oper Dent. 2000;25:434.
43 Cheylan J-M, et al. In vitro push-out strength of seven luting agents to dentin. Int J Prosthodont. 2002;15:365.
44 Anusavice KJ. Phillips’ Science of Dental Materials, 11th ed., Philadelphia: WB Saunders; 2003:456.
45 Caughman WF, et al. Glass ionomer and composite resin cements: effects on oral cells. J Prosthet Dent. 1990;63:513.
46 Felton DA, et al. Effect of cavity varnish on retention of cemented cast crowns. J Prosthet Dent. 1987;57:411.
47 Hilton T, et al. A clinical comparison of two cements for levels of post-operative sensitivity in a practice-based setting. Oper Dent. 2004;29:241.
48 Tjan AHL, Tao L. Seating and retention of complete crowns with a new adhesive resin cement. J Prosthet Dent. 1992;67:478.
49 Feilzer AJ, et al. Setting stress in composite resin in relation to configuration of the restoration. J Dent Res. 1987;66:636.
50 Malament KA, Socransky SS. Survival of Dicor glass-ceramic dental restorations over 16 years. Part III: effect of luting agent and tooth or tooth-substitute core structure. J Prosthet Dent. 2001;86:511.
51 O’Connor RP, et al. Effect of internal microblasting on retention of cemented cast crowns. J Prosthet Dent. 1990;64:557.
52 Henschel CJ. The effect of mixing surface temperatures upon dental cementation. J Am Dent Assoc. 1943;30:1583.
53 Kendzior GM, Leinfelder KF. Characteristics of zinc phosphate cements mixed at sub-zero temperatures [Abstract no. 134]. J Dent Res. 1976;55(Special Issue B):B95.
54 Myers CL, et al. A comparison of properties for zinc phosphate cements mixed on room temperature and frozen slabs. J Prosthet Dent. 1978;40:409.
55 Rosenstiel SF, Gegauff AG. Mixing variables of zinc phosphate cement and their influence on the seating and retention of complete crowns. Int J Prosthodont. 1989;2:138.
56 Fakiha ZA, et al. Rapid mixing of zinc phosphate cement for fixed prosthodontic procedures. J Prosthet Dent. 1992;67:52.
57 Brackett WW, Vickery JM. The influence of mixing temperature and powder/liquid ratio on the film thickness of three glass-ionomer cements. Int J Prosthodont. 1994;7:13.
58 Rosenstiel SF, Gegauff AG. Improving the cementation of complete cast crowns: a comparison of static and dynamic seating methods. J Am Dent Assoc. 1988;117:845.
59 Irie M, et al. Marginal and flexural integrity of three classes of luting cement, with early finishing and water storage. Dent Mater. 2004;20:3.
60 Della Bona A, et al. Effect of ceramic surface treatment on tensile bond strength to a resin cemen. Int J Prosthodont. 2002;15:248.
61 Sjögren G, et al. A 10-year prospective evaluation of CAD/CAM-manufactured (Cerec) ceramic inlays cemented with a chemically cured or dual-cured resin composite. Int J Prosthodont. 2004;17:241.
62 Mojon P, et al. A comparison of two methods for removing zinc oxide–eugenol provisional cement. Int J Prosthodont. 1992;5:78.
63 Schwartz R, et al. Effect of a ZOE temporary cement on the bond strength of a resin luting cement. Am J Dent. 1990;3:28.
64 White SN, Yu Z. Physical properties of fixed prosthodontic, resin composite luting agents. Int J Prosthodont. 1993;6:384.