Chapter 21 Viruses of relevance to dentistry
This chapter gives an outline of the viruses that are of special relevance to dentistry. The DNA viruses are described first, followed by the RNA viruses (see Table 4.1).
DNA viruses
Papovaviruses
These DNA viruses infect both humans and animals; however, human disease is infrequent.
Human papillomavirus
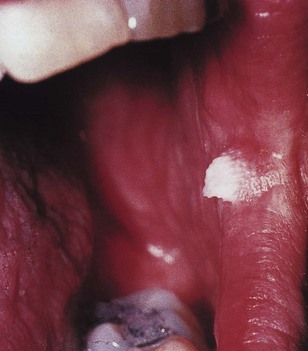
Human papillomavirus (HPV) mainly causes skin warts (verrucae); it is also associated with a number of lesions including oral papillomas, oral verrucous carcinomas and focal epithelial hyperplasia. There are more than 70 serological types of HPV, some of which are more closely associated with lesions (both benign and cancerous) than others.
Skin warts
Oral squamous papillomas and warts
Adenoviruses
These DNA viruses induce latent infections of the tonsils, adenoids and other lymphoid tissues of humans. However, most infections caused by adenoviruses are acute and self-limiting.
Adenoviral diseases
Acute respiratory disease is the most common adenovirus infection. It is an influenza-like illness seen commonly in military training camps. Clinically, the main symptoms are pharyngitis and conjunctivitis. Although self-limiting, acute respiratory disease may be complicated by pneumonia in some cases. Other infections caused by these viruses include pharyngoconjunctival fever (a disease of infants and children), epidemic keratoconjunctivitis, pneumonia and gastroenteritis.
Herpesviruses
There are a range of different human herpesviruses, currently numbered 1–8 (see Table 4.3). All of them are structurally similar (enveloped, icosahedral with double-stranded DNA) and infect both humans and animals. They are the most common causes of human viral infections. All have the important property of remaining latent, with the ability to reinfect the host a variable period after the primary infection. Important human pathogens include herpes simplex virus types 1 and 2 (HSV-1 and HSV-2), varicella-zoster virus (VZV), cytomegalovirus (CMV) and Epstein–Barr virus (EBV) (see Chapter 4). Students of dentistry should be thoroughly conversant with the herpes group of viruses as the majority of them either cause oral infection or are intimately associated with orofacial tissues and saliva.
HSV (human herpesviruses 1 and 2)
There are two types of HSV: HSV-1 and HSV-2. They can be differentiated by serotyping, by DNA homology and, to some extent, by clinical disease pattern.
Clinical disease
Disease due to HSV can be either a primary infection, due to first encounter with the virus, or a reactivation or recurrent infection, due to activation of the latent virus.
Primary infection
There is an incubation period of 2–20 days, depending upon the infected site and the infecting strain of virus. The lesions include:
Recurrent infections
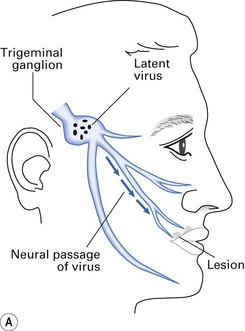
Recurrence or reactivation of HSV entails activation of the non-infectious form of the latent virus residing in the neurons of either the trigeminal ganglion (Fig. 21.3) or the sacral ganglia. Reactivation is provoked by menstruation, stress, sunlight (possibly ultraviolet rays), local trauma, etc.; the lesions tend to recur at the site of the primary lesion. HSV has been implicated in Bell’s palsy.
Epidemiology
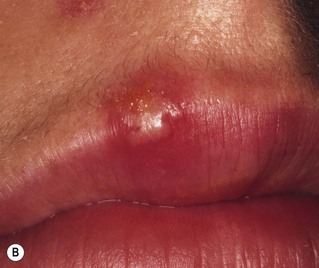
Humans are the only known reservoir for HSV-1 and HSV-2; experimental infection can be induced in animals and cell cultures. As the virus is highly labile, most primary infections are acquired through direct contact with a lesion or contaminated secretions. In general, HSV-1 causes orofacial lesions or lesions ‘above the belt’, while HSV-2 causes lesions ‘below the belt’, i.e. genital herpes (Fig. 21.4). However, because of sexual promiscuity or for other reasons, this may not be always true. HSV-1 is acquired early in life, while HSV-2 appears after the onset of sexual activity.
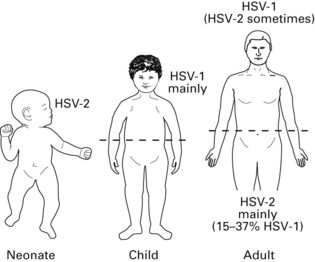
Fig. 21.4 Predominant distribution of infection with herpes simplex virus types 1 and 2 (HSV-1 and HSV-2), in different age groups.
As recurrent infection is common in the presence of high antibody titres, circulating antibodies appear to be unhelpful in controlling HSV infection. One reason for this may be the contiguous cell-to-cell spread of the virus, which cannot be prevented by antibody. Reactivation is not accompanied by a rise in herpes antibody titre.
Diagnosis
Diagnosis is usually achieved clinically; laboratory diagnosis is useful to confirm infection, especially in compromised patients. This entails:
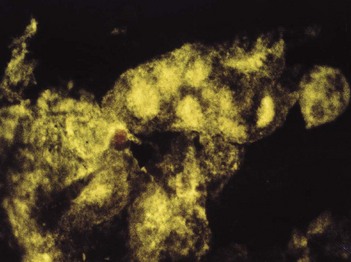
Fig. 21.5 Positive immunofluorescence of a smear taken from the lip lesion shown in Figure 21.3B (stained with anti-herpes antibody tagged to a fluorescing chemical), indicating that the patient has herpes labialis.
However, the foregoing traditional techniques are now supplanted by polymerase chain reaction-based rapid diagnostic methods.
Prevention
Control is difficult because of the high frequency of asymptomatic infection. It is important to avoid contact with acute herpetic lesions and contaminated body fluid (e.g. saliva) by routine wearing of gloves. No vaccine is available.
Treatment
The course of primary infection can be altered significantly with drugs that interfere with viral DNA synthesis, such as aciclovir and vidarabine, but these should be administered in the early prodromal phase of the disease for best results (see also Chapter 35).
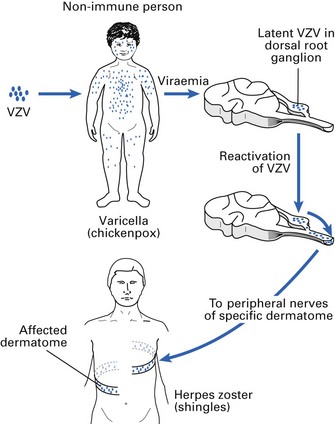
Varicella-zoster virus (human herpesvirus 3)
This organism causes both varicella (chickenpox) and herpes zoster (shingles) – two different diseases due to an identical organism. Chickenpox is the primary infection, and herpes zoster is the reactivation of illness.
Clinical disease
Varicella
A common childhood fever, varicella is mild and self-limiting. The disease is more severe if contracted in adulthood. After a 2-week incubation period, fever develops, followed by a papular rash of the skin and mucous membranes, including the oral mucosa. The papules rapidly become vesicular and itchy but painless (in contrast to the rash in zoster).
Zoster (shingles)
Occurs primarily as a reactivation of the virus in dorsal root or cranial nerve (usually trigeminal) ganglia (Fig. 21.6). The disease usually affects adults, and the virus is reactivated despite circulating antibodies. Zoster is triggered by trauma, drugs, neoplastic disease or immunosuppression.
The virus remains latent in ganglionic nerve cells and, after activation, travels back along the nerve fibre to the skin. Thoracic nerves supplying the chest wall are most often affected, and the lesion presents as a unilateral, painful vesicular rash, which extends in a horizontal strip from the middle of the back around the side of the chest wall (‘belt of roses from hell’). Fever and malaise accompany the lesion. The rash may last for 2–4 weeks, with pain (post-herpetic neuralgia) persisting for weeks or months.
The trigeminal nerve is affected in about 15% of cases, with involvement of the ophthalmic, maxillary and mandibular divisions (in that order of precedence). Severe localized oral pain precedes the rash and may be easily confused with toothache (see Chapter 35). Involvement of the ophthalmic nerve may lead to eye lesions and sometimes blindness.
Ramsay Hunt syndrome is a rare manifestation of zoster, with a vesicular rash on the tympanic membrane and the external auditory canal, together with unilateral facial nerve palsy.
Epidemiology
Shingles is primarily a disease of older adults and immunocompromised persons; it is rare in children. The incidence increases with advancing age and with decreasing degree of immunocompetence. It is a highly contagious infection in a host not previously exposed to the virus. Transmission occurs by direct contact with skin lesions or droplet infection from infectious saliva.
Diagnosis
The clinical picture is pathognomonic, as the lesion distribution overlaps and accurately maps the distribution of the sensory nerve (Fig. 21.7). Serology, if needed, entails detecting a fourfold rise in antibody titre in paired sera (compare herpes simplex reactivation, where antibody rise is not significant).
Treatment
Chickenpox is self-limiting and requires symptomatic treatment, if any. Disseminated zoster in immunocompromised patients requires antiviral drugs (e.g. aciclovir, vidarabine), which interfere with herpesvirus DNA replication. Varicella-zoster virus is less sensitive to aciclovir than is HSV, and hence, a higher dosage is required; therapy should start within 72 h of onset. Systemic aciclovir may reduce the duration of the early infective phase and the associated pain. In addition, it may reduce the prevalence of post-herpetic neuralgia.
EBV (human herpesvirus 4)
EBV is widespread in humans, and most adults have antibody to the virus. The virus persists in latent form within lymphocytes after primary infection (lymphotrophic, unlike HSV and varicella-zoster virus, which are neurotrophic). The genome resides in a latent form in B cells; latent EBV infection is common in the population. It is the aetiological agent of a number of diseases:
Infectious mononucleosis
An acute infection affecting lymphoid tissue throughout the body, infectious mononucleosis is commonly seen in teenagers, with a peak incidence at 15–20 years of age. The organism is present in saliva and is postulated to be transmitted during kissing – hence, it is called the ‘kissing disease’.
Signs and symptoms
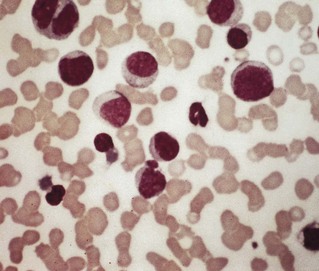
Low-grade fever with generalized lymphadenopathy and abnormal lymphocytes in the blood (note that a similar illness, glandular fever-like syndrome, develops during the first fortnight after infection with human immunodeficiency virus (HIV)). Fever, tonsillitis and fatigue are common, and many patients have splenomegaly. Lymphocytosis is a characteristic feature, hence the term ‘mononucleosis’; some 10% of the lymphocytes are atypical, with enlarged misshapen nuclei and increased cytoplasm (Fig. 21.8).
Chronic, persistent or reactivated EBV infection
This may take many clinical forms and is less common than acute mononucleosis, described above. The syndrome is characterized by persistent fatigue, with or without physical or laboratory findings.
Epidemiology
The virus is ubiquitous, and humans are its only known host. Spread of EBV is via respiratory secretions, primarily through oral contact. Those from lower socioeconomic classes are exposed to EBV at an early age and typically develop asymptomatic infections, while in higher socioeconomic classes, particularly in developed countries, primary infection is usually delayed to adolescence or young adulthood.
Diagnosis
As EBV cannot be easily propagated in culture, serological diagnosis is common:
Treatment
Infectious mononucleosis is generally mild and self-limiting; hence, therapy is usually symptomatic.
Burkitt’s lymphoma
Burkitt’s lymphoma is a highly malignant tumour that spreads rapidly, with widespread metastases; it is particularly common in African children. The disease is especially common in areas of Africa with endemic malaria. Hence, it is thought that the effect of the malarial parasite on the reticuloendothelial system could cause an abnormal response to infection with EBV. Under these conditions, the EBV may become frankly oncogenic, producing a malignant transformation in lymphoid tissue (lymphoma) instead of the benign proliferation seen in infectious mononucleosis.
Nasopharyngeal carcinoma
A tumour with a remarkable geographic and probably racial distribution, it is particularly common among the southern Chinese. EBV DNA is regularly present in the malignant epithelial cells of the tumour.
Hairy leukoplakia
The term ‘hairy leukoplakia’ is given to raised, white areas of thickening, particularly on the lateral border of the tongue (see Fig. 30.3). Although this new clinical entity was first described in HIV-infected patients, other immunosuppressed patients may develop the lesion, which is very closely associated with EBV. The DNA of EBV is present in the epithelial cells of hairy leukoplakia. The lesion is non-malignant but about a third of HIV-infected individuals who develop hairy leukoplakia may develop acquired immune deficiency syndrome (AIDS) in 3–5 years. Demonstration of EBV in biopsy tissue of hairy leukoplakia is essential for a definitive diagnosis.
CMV (human herpesvirus 5)
CMV rarely causes disease unless other precipitating factors, such as immunocompromising states, are present. However, it can infect the foetus during pregnancy.
Clinical disease
Symptomless infection
The majority of infants show no signs of infection, and diagnosis is made by serology. Although a large proportion of the infants are unharmed, a significant number of neonates with congenital infection show neurological sequelae, such as deafness and mental retardation, later in life. A minority develop a severe, often fatal illness associated with infection of the salivary glands, brain, kidneys, liver and lungs.
Human herpesvirus 6
A DNA virus closely related to CMV, human herpesvirus 6 (HHV-6) was originally isolated from peripheral blood cells of immunocompromised patients, such as those with AIDS. The virus shows affinity for T and B cells in particular. Infection with HHV-6 is common in childhood, and most primary infections are asymptomatic followed by latency. The pathogenicity of HHV-6 is as yet unclear. Recently, it has been found in active plaques in patients with multiple sclerosis.
Exanthem subitum (roseola infantum)
A common childhood disorder characterized by mild fever and a facial rash, and appears to be associated with HHV-6 infection.
Mononucleosis with cervical lymphadenopathy
This is a febrile illness in adults with bilateral cervical lymphadenopathy, somewhat like glandular fever; thought to be a primary infection with HHV-6.
HHV-6 and the oral cavity
The virus is present in the saliva of most healthy adults, and it can also be demonstrated in ductal and alveolar epithelium of major salivary glands. There are no specific oral lesions reported for HHV-6, though erythematous papules seen in soft palate and uvula (Nagayama’s spots) and in the pharynx are thought to be due to this organism. No occupational hazard from HHV-6 has been proved in dentistry, but the virus may well be transmitted in saliva.
Human herpesviruses 7 and 8
These viruses have been recently identified: human herpesvirus 7 (HHV-7) is a T-lymphotrophic virus and is implicated in rashes; human herpesvirus 8 (HHV-8) is the agent responsible for Kaposi’s sarcoma, a vascular endothelial tumour commonly seen in HIV disease (Chapter 30); it is also implicated in sarcoidosis.
The relatively recent discovery of these new herpesviruses in individuals with HIV disease suggests that there are more herpesviruses yet to be uncovered. These viruses appear to be evolving in humans and primates. HHV-6 and HHV-7 in particular are found in 70–90% of the population and hence considered universal herpesviruses.
RNA viruses
Orthomyxoviridae
These RNA viruses cause worldwide epidemics of influenza. They are subdivided into types A, B and C on the basis of the antigenic properties of their major nucleocapsid protein (NP) and viral envelope matrix protein (M protein). In addition to these antigenic differences, they are characterized by a unique mechanism of frequent immunological variations within the subtypes. These variations are due to structural changes in the surface spike glycoproteins: haemagglutinin (H antigen) and neuraminidase (N antigen). The nomenclature of influenza viruses is based on the H and N antigens. For instance, the first pandemic influenza virus was called H1N1 and the current avian flu virus is H5N1. Influenza epidemics are due to the emergence of a new virus strain containing a haemagglutinin (and sometimes a neuraminidase) that differs from that of previously circulating viruses, so that the population has no (herd) immunity to the new haemagglutinin. Antigenic variation may occur due to:
These antigenic shifts are critically important in the production of vaccines for influenza: the vaccine used in previous years may have little or no effect because of these phenomena.
Influenza
Clinical features
Symptoms are sudden and appear 1–2 days after exposure. Major symptoms are high fever, accompanied by myalgia, sore throat, headache, cough and nasal congestion. Pneumonia is the most common serious complication of influenza; it is caused by secondary bacterial infection of the respiratory tract with weakened defences.
Epidemiology
Epidemic illness is common in non-immune or partially immune populations. Transmission occurs by aerosolization and subsequent inhalation of virus-laden respiratory secretions during sneezing and coughing (droplet spread). Rapid spread of illness may occur in confined populations (e.g. nursing homes, classrooms).
Treatment and prevention
Only symptomatic treatment is indicated. Amantadine is helpful for relieving symptoms and enhancing the effectiveness of immunization. The low success rate (about 70%) of the vaccination is mainly due to the difficulty of predicting the proper antigenic profile of the influenza strain; this unfortunately cannot be determined until the onset of the particular disease cycle.
Avian influenza or bird flu (H5N1 virus)
The first known cases of avian influenza caused by H5N1 were discovered in Hong Kong in 1997, and since then, there have been sporadic outbreaks, mainly in South-East Asia. Usually, such infections are preceded by lethal outbreaks of H5N1 influenza in waterfowl, which are the natural hosts of these viruses and therefore normally have asymptomatic infection. The acquisition by the viruses of characteristics that enhance virulence in humans and their potential for wider distribution by infected migrating birds have caused renewed pandemic concern. The factors that account for the severe symptoms of viral influenza are still not well understood. However, it is believed that the cytopathic effects of the virus itself and the cytokines evoked by the infection account for both local and systemic effects that are life-threatening.
Clinical features
H5N1 infection and its replication in the respiratory tract have been shown to injure directly the nasal and tracheobronchial epithelium, possibly due to virus-induced cellular apoptosis and resulting loss of respiratory epithelial cells. These may account for symptoms such as cough, depressed tracheobronchial clearance and altered pulmonary function.
Incubation is thought to be 7–12 days after exposure. Major symptoms are high fever, sore throat, headache, chest pain and cough, bleeding nose and gums, and diarrhoea. These may lead to pneumonia, encephalitis and organ failure. The predicted death rate of avian flu ranges from 90% to 60%.
Epidemiology
At the time of writing, there have been sporadic cases (up to 70), mainly in South-East Asia, with 14 associated fatalities.
Treatment and prevention
Preventive measures include good personal hygiene, thorough adherence to routine hand-washing, respiratory precautions such as wearing masks and avoiding crowded places during an outbreak. The routine flu vaccine is ineffective against avian flu. Drugs such as Tamiflu reduce the severity of infection if taken within 2 days of initial symptoms. Patients may need quicker and larger doses. In dentistry, additional precautions or transmission-based precautions must be implemented during an outbreak situation (Chapter 36).
Paramyxoviridae
The paramyxoviruses are enveloped, RNA viruses with an unsegmented genome, which cause major diseases of infancy and childhood. There are four groups of paramyxoviruses:
Parainfluenza viruses
The parainfluenza viruses cause human respiratory infections, especially in autumn and winter.
Mumps virus
Mumps, measles, rubella and varicella (chickenpox) are the common childhood fevers. Mumps virus typically causes parotitis (mumps) of acute onset involving one or both parotid glands. The attenuated form of the mumps virus, incorporated in the combined measles–mumps–rubella (MMR) vaccine, leads to the development of antibody in 95% of vaccinees (see also Chapter 35).
Measles virus
Another agent of common childhood fever, measles virus causes one of the most highly infectious diseases known. Infection results in permanent immunity.
Clinical features
Measles is an acute febrile illness with a characteristic exanthematous rash. The virus enters through the respiratory tract and multiplies in the respiratory epithelium and regional lymphoid tissue for up to 12 days. In the next (viraemic) phase, the virus spreads throughout the lymphoid tissues and skin. This stage is accompanied by prodromal symptoms: conjunctivitis, nasal discharge, headache, low-grade fever, sore throat and Koplik’s spots. These are bluish-white, pinpoint spots surrounded by dark-red areolae, which appear on the buccal mucosa opposite the molar teeth and sometimes near the orifice of the parotid duct. The measles rash appears to result from the interaction between virus-infected cells and either sensitized lymphocytes or antibody–complement complexes. The rash consists of fine, sparse, discrete macules. As the rash develops, the Koplik’s spots disappear.
Complications
The complications of measles virus infection are serious and could be:
Epidemiology
Measles is readily transmissible, usually via respiratory secretions and urine, especially during the prodromal phase and when the rash appears.
Prevention
The measles component of the MMR vaccine is a live attenuated virus that induces immunity for up to 10 years. However, in developing countries such as West Africa, where universal vaccination in childhood is not feasible, measles remains a severe disease and a major cause of death in childhood.
Respiratory syncytial virus
A major agent of lower respiratory tract disease, RSV causes worldwide epidemics of respiratory tract infection in infants and young children. Adults, although infected, develop only mild or non-apparent symptoms. The virus can cause colds, bronchiolitis and pneumonia, especially during the first 6 months of life. Approximately one-third of infants develop antibodies in the first year of life.
Picornaviridae
Picornaviridae are non-enveloped, RNA viruses with an unsegmented genome. Four members of this family cause significant human disease: polioviruses, coxsackieviruses, echoviruses and rhinoviruses. The first three of these are collectively termed enteroviruses.
Polioviruses
Polioviruses are agents of paralytic poliomyelitis.
Clinical features
Poliovirus infection is initiated by ingestion of infectious virions, after which primary replication occurs in oropharyngeal and intestinal mucosa. The virus drains into the cervical and mesenteric lymph nodes and then into the systemic circulation. Subsequent replication continues in a number of non-neural sites, leading to a persistent viraemia and spread into the central nervous system.
Paralytic poliomyelitis is unusual and depends on host factors that may predispose to neural infection. The incidence and severity of paralytic disease increase with age (e.g. teenagers are more likely than younger children to develop crippling disease).
Epidemiology
Polioviruses have a wide geographic distribution and spread rapidly, especially in densely populated areas with poor sewage control, such as in developing countries. Infection occurs mainly in the hot season and is spread in the faeces. Transmission is primarily by person-to-person contact through pharyngeal secretions, although the disease may spread by infected water.
Coxsackieviruses
Coxsackieviruses are subdivided into two major groups, A and B, on the basis of the lesions they induce in suckling mice. Each group also has several serologically distinct subgroups. Most human coxsackievirus infections are mild and frequently asymptomatic. Serious infection, although rare, results in severe disease. Two diseases caused by group A coxsackieviruses are of particular dental interest: herpangina, and hand, foot and mouth disease.
Herpangina
Herpangina, caused by group A coxsackievirus, is common in children but may affect any age group.
Clinical features
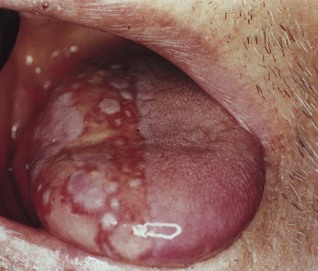
The disease is characterized by fever, headache, sore throat, dysphagia, anorexia and occasionally a stiff neck. These symptoms are accompanied by herpes-like oropharyngitis, where the ulceration is predominantly on the tonsil, soft palate and uvula. The small, papulovesicular lesions are about 1–2 mm in diameter, with a greyish-white surface surrounded by red areolae. The disease is self-limiting and lasts for 3–4 days (see also Chapter 35).
Hand, foot and mouth disease
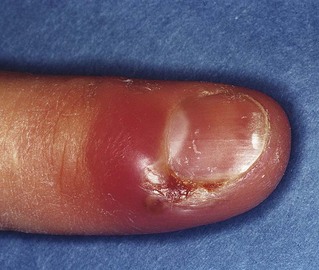
Hand, foot and mouth disease, also caused by group A coxsackievirus, is a relatively common infection in children. It is easily diagnosed because of its classic distribution in the hands, feet and mouth. The incubation period is about 3–5 days and resolution occurs within a week.
Clinical features
The disease may begin with facial pain, with tenderness along the course of the parotid duct and a few vesicles around the duct orifice. The onset of the oral and skin eruptions is accompanied by headache, malaise and sore throat, but in many, there is little systemic upset. The oral lesions are generally bright-red macules, which later form oval or grey vesicles with red areolae (see Chapter 35). The plantar surface of the feet and the palmar surface of the hands and sometimes the buttocks may be affected. These skin lesions are bright-red macules with pale centres, which develop into thin-walled bullae or small ulcers with surrounding erythema. The lesions in the mouth, and on the hands and feet, are not always seen.
All serotypes of coxsackievirus have a worldwide distribution. They are highly infectious within families and closed communities, and the greatest epidemic spread occurs in the summer and autumn. Viral transmission is by the faecal–oral route and from nasal and pharyngeal secretions. They enter through the mouth and nose, multiply locally and spread viraemically (compare polioviruses).
Rhinoviruses
The aetiological agents of the ‘common cold’ and a group of acute, afebrile upper respiratory diseases, rhinoviruses are readily inactivated at low pH conditions and require an incubation temperature of 33°C for maximal replication; hence, they multiply well in the upper respiratory tract where the incoming air provides low temperature conditions suited to the virus.
Antigenicity
There is a vast array (more than 100) of immunologically distinct groups of rhinoviruses based on a single type-specific antigen, hence the reason for recurrent colds, as the succeeding infective virus is likely to be antigenically different from the virus that caused the previous episode (i.e. immunity is only effective against homologous challenge).
Epidemiology
In a family unit, rhinovirus transmission is usually initiated when a child introduces the virus, which spreads rapidly via nasal secretions. The disease is most common in the autumn, winter and early spring. Note, however, that rhinoviruses are not the only agents of the common cold, although they are the major culprits.
Togaviridae
Rubella
The agent of rubella (German measles) is a togavirus. Rubella is a childhood fever resembling measles, except that it has a milder clinical course and shorter duration. If rubella is contracted in early pregnancy, the virus can cause severe congenital abnormalities and may cause the death of the foetus.
Epidemiology
Rubella is a highly contagious disease spread by nasal secretions. Because of its mild clinical symptoms, the infection is often non-apparent, and viral dissemination may be widespread before it is recognized. The disease may spread in the dental clinic environment. Females (especially of child-bearing age) should be immunized against the virus: the rubella component of the combined MMR vaccine contains a live attenuated virus, which confers adequate protection.
Other RNA viruses
Other RNA virus families that have not been discussed here include the Arenaviridae, Bunyaviridae, Coronaviridae, Reoviridae, Rhabdoviridae and Retroviridae. HIV, which is in the latter family, is discussed in detail in Chapter 30 because of its major relevance to dentistry.
Viruses and cancer
Viruses that have the ability to cause cancer are called oncogenic viruses. A number of DNA viruses are oncogenic, but only one RNA virus is known to have this potential. The virus groups and the cancers they cause are summarized below.
Papovaviruses
HPVs cause benign warts, malignant carcinomas and cervical cancers.
The polyomavirus and the simian virus 40 (SV40) are oncogenic in laboratory animals.
Hepadnaviruses
Hepatitis B virus is a well-known agent of human hepatocellular carcinoma (Chapter 29).
Retroviruses
Retroviruses include the human T cell leukaemia viruses (HTLVs):
Key facts
DNA viruses
RNA viruses
Bagg J. Virology and the mouth. Reviews in Medical Microbiology. 1994;5:209-216.
Cleator G.M., Klapper P.E. The Herpesviridae. In Zuckerman A.J., Banatvala J.E., Pattison J.R., editors: Principles and practice of clinical virology, 3rd ed., Chichester: John Wiley, 1994. Ch. 2A
Greenberg M.D. Herpesvirus infections. Dental Clinics of North America. 1996;40:359-368.
Scully C., Samaranayake L.P. Clinical virology in oral medicine and dentistry. Cambridge: Cambridge University Press; 1992.
Review questions (answers on p. 353)
Please indicate which answers are true, and which are false.