Chapter 4 Viruses and prions
Viruses are one of the smallest forms of microorganism and infect most other forms of life: animals, plants and bacteria. They can also cause severe acute oral and orofacial disease, produce oral signs of systemic infection, and be transmitted to patients and dental staff. The main features that characterize viruses are:
Structure
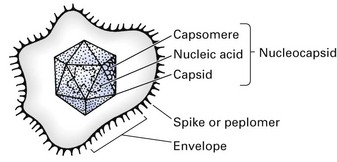
Viruses consist of a nucleic acid core containing the viral genome, surrounded by a protein shell called a capsid (Figs 4.1 and 4.2). The entire structure is referred to as the nucleocapsid. This may be ‘naked’, or it may be ‘enveloped’ within a lipoprotein sheath derived from the host cell membrane. In many viruses (e.g. orthomyxoviruses, paramyxoviruses), the ensheathment begins by a budding process at the plasma membrane of the host cell, while others, such as herpesviruses, ensheath at the membrane of the nucleus or endoplasmic reticulum.
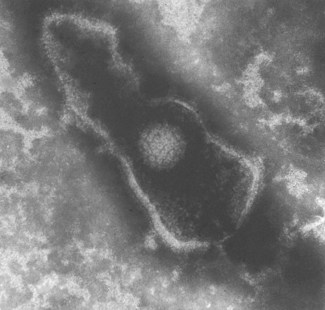
Fig. 4.2 Scanning electron micrograph of a herpesvirus. Note the extensive outer lipid envelope and the icosahedral nucleocapsid.
The protein shell or capsid consists of repeating units of one or more protein molecules; these protein units may go on to form structural units, which may be visualized by electron microscopy as morphological units called capsomeres (Fig. 4.1). Genetic economy dictates that the variety of viral proteins be kept to a minimum as viral genomes lack sufficient genetic information to code for a large array of different proteins. In enveloped viruses, the protein units, which comprise the envelopes and are visualized electron microscopically, are called peplomers (loosely referred to as ‘spikes’).
Viral nucleic acid
Viral nucleic acid may be either DNA or RNA. The RNA, in turn, may be ss or ds, and the genome may consist of one or several molecules of nucleic acid. If the genome consists of a single molecule, this may be linear or have a circular configuration. The DNA viruses all have genomes composed of a single molecule of nucleic acid, whereas the genomes of many RNA viruses consist of several different molecules or segments, which are probably loosely linked together in the virion.
Viral protein
In terms of volume, the major bulk of the virion is protein, which offers a protective sheath for the nucleic acid. The viral protein is made up of two or three different polypeptide chains, although in some only one kind of polypeptide chain may be present. Virion surface proteins may have a special affinity for receptors on the surface of susceptible cells and may bear antigenic determinants.
Although most viral proteins have a structural function, some have enzymatic activity. For instance, many viruses such as the human immunodeficiency virus (HIV) contain a reverse transcriptase, whereas several enzymes (e.g. neuraminidase, lysozyme) are found in larger, more complex viruses.
Viral lipid and carbohydrate
In general, lipids and carbohydrates of viruses are only found in their envelopes and are mostly derived from the host cells. About 50–60% of the lipids are phospholipids; most of the remainder is cholesterol.
Virus symmetry
The nucleocapsids of viruses are arranged in a highly symmetrical fashion (symmetry refers to the way in which the protein units are arranged). Three kinds of symmetry are recognized (Fig. 4.3):
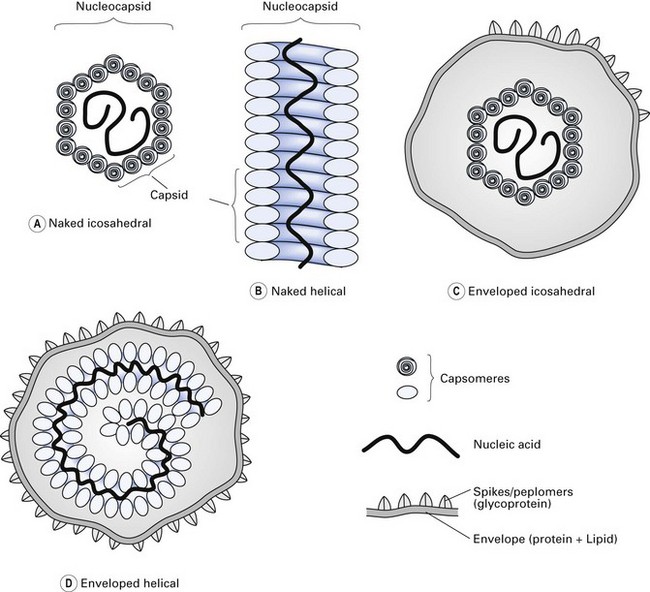
Fig. 4.3 Structural components and symmetry of different viruses. (A) Naked icosahedral; (B) naked helical; (C) enveloped icosahedral; (D) enveloped helical.
Taxonomy
Vertebrate viruses are classified into families, genera and species. The attributes used in classification are their symmetry, the presence or absence of an envelope, nucleic acid composition (DNA or RNA), the number of nucleic acid strands and their polarity. Classification of some of the recognized families of RNA and DNA viruses is given in Table 4.1. (Note: to memorize which viruses contain DNA, remember the acronym ‘PHAD’: P is for papova and pox, H for herpes and AD for adenoviruses. Most of the remainder are RNA viruses, including the self-evident picornaviruses.)
Table 4.1 Classification of some of the viruses causing human disease
| Morphology | Virus |
|---|---|
| DNA | |
| Enveloped, double-stranded nucleic acid | Herpesviruses |
| Herpes simplex virus | |
| Varicella-zoster virus | |
| Epstein–Barr virus | |
| Cytomegalovirus | |
| Human herpesvirus 6 | |
| Poxviruses | |
| Vaccinia | |
| Orf | |
| Enveloped, single-stranded | Parvoviruses |
| Non-enveloped, double-stranded | Adenoviruses |
| Papovaviruses | |
| Polyomaviruses | |
| Papillomaviruses | |
| Hepadnaviruses | |
| Hepatitis B virus | |
| RNA | |
| Enveloped, single-stranded | Orthomyxoviruses |
| Influenzavirus | |
| Paramyxoviruses | |
| Parainfluenza | |
| Respiratory syncytial | |
| Mumps | |
| Measles | |
| Togaviruses | |
| Rubella | |
| Retroviruses | |
| Human immunodeficiency viruses HTLV-I, -III | |
| Rhabdoviruses | |
| Rabies | |
| Non-enveloped, double-stranded | Reoviruses |
| Rotavirus | |
| Non-enveloped, single-stranded | Picornaviruses |
| Rhinovirus | |
| Enterovirus | |
| Coxsackievirus | |
| Echovirus | |
| Poliovirus |
HTLV-I, human T cell leukaemia virus type I.
The following is a concise description of the families of mammalian viruses.
DNA viruses
Papovaviruses
Papovaviruses are small, icosahedral DNA viruses with a capacity to produce tumours in vivo and to transform cultured cell lines. The name ‘papova’ is an acronym derived from the papillomavirus, polyomavirus and vacuolating agent simian virus 40 (SV40), which make up this family.
Papillomavirus
This genus contains human serotypes that cause benign skin tumours or warts and both oral and skin papillomas (e.g. hand and plantar warts). Although they were regarded as a cosmetic nuisance rather than a specific disease, it is now known that the papillomaviruses may be involved in genital and oral cancers.
Adenoviruses
Adenoviruses are icosahedral DNA viruses, commonly associated with respiratory and eye infections in humans. These viruses were so named because they were first isolated from cultured adenoid tissue eliciting cytopathic effects. Syndromes associated with adenoviruses include:
Herpesviruses
Herpesviruses are the predominant viral cause of oral infections in humans; often the infections are recurrent, and latent.
Structure
These enveloped, icosahedral viruses are 180–200 nm in diameter and contain a linear dsDNA molecule. The Herpesviridae family has over 100 members spread widely among vertebrates, and invertebrates and new species are continuously being added. Herpesviruses are unstable at room temperature and are rapidly inactivated by lipid solvents such as alcohol and other common disinfectants owing to the disruption of the outer lipid envelope.
During reproduction, maturation of the progeny begins in the nucleus of the host cell, which buds through the nuclear membrane and acquires the viral envelope. Typical and highly pathognomonic intranuclear inclusions are therefore found in cells that have undergone active virus replication. As many herpesviruses can fuse with the cells they infect, polykaryocytes or giant cells readily appear in tissue lesions. Such cells, e.g. Tzanck cells or nuclear inclusions (Lipschiitz bodies), are hallmarks of herpetic infections.
Different herpesviruses cause a variety of infectious diseases, some localized and some generalized, often with a vesicular rash. Herpesviruses establish latent infection, which can be readily reactivated by immunosuppression (Table 4.2).
Table 4.2 Latent viruses relevant to dentistry
| Virus | Site of latency |
|---|---|
| Herpes simplex virus | Trigeminal ganglion |
| Varicella-zoster virus | Sensory ganglia |
| Epstein–Barr virus | Epithelial cells |
| B lymphocytes | |
| Cytomegalovirus | Salivary gland cells |
| Papillomaviruses | Epithelial cells |
| Human immunodeficiency viruses | Lymphocytes and other CD4+ cells (see Chapter 30) |
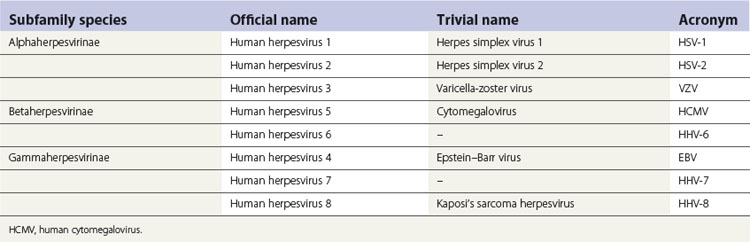
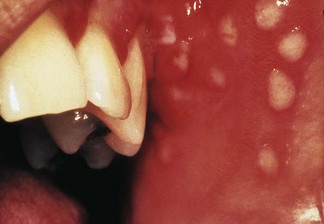
The nomenclature of herpesviruses is contentious; there is thus a historical or a traditional (trivial) nomenclature and an official name for each virus (Table 4.3). The herpesviruses that commonly infect humans can be distinguished by their antigenic and genomic profiles, although they cannot be differentiated by electron microscopy owing to identical capsid morphology. They also have a universal ability to establish latent infection in the host in which they reside, and manifest a number of common epidemiological features. Herpes simplex virus, herpes zoster virus, Epstein–Barr virus, human cytomegalovirus and herpesviruses 6 and 8 can all cause infections in oral and perioral tissues (Fig. 4.4); see Chapter 35 for details.
Poxviruses
The poxviruses are the largest viruses to infect humans or animals. Molluscum contagiosum in humans is caused by a poxvirus, as is smallpox, which is now a disease of only historical interest. Humans occasionally acquire infection by animal poxviruses, e.g. cowpox.
Parvoviruses
Parvoviruses are icosahedral viruses with ssDNA. Three serologically distinct types of autonomous parvoviruses are recognized in human disease. The first group is found in stool specimens, the second (the B19 virus) in the serum of asymptomatic blood donors, while the third has been recovered from synovial tissues of rheumatoid arthritis patients. The B19 virus is responsible for a febrile illness, particularly in children, manifesting as a maculopapular rash.
The exanthem is characterized by a fiery-red rash on the cheeks – the ‘slapped-cheek’ syndrome (also termed fifth disease).
Hepadnaviruses
Hepadnaviruses are small, spherical DNA viruses causing hepatitis, chronic liver infections and possibly liver cancer. They are of particular interest in dentistry because of their mode of transmission via blood and saliva (see Chapter 29).
RNA viruses
Picornaviruses
Picornaviruses are the smallest family of RNA viruses but incorporate a very large group of viruses, including the genus Enterovirus. Human enteroviruses have been further subdivided into three major subgroups:
The enteroviruses reside and multiply asymptomatically in the gut but may cause a spectrum of disease ranging from mild undifferentiated rashes, respiratory infections and pharyngitis (coxsackie A) to more serious diseases, including carditis (coxsackie B) which may be lethal in the newborn (see Chapters 21 and 35).
Orthomyxoviruses
Orthomyxoviruses are RNA viruses with a tubular nucleocapsid and a lipoprotein envelope. Influenza A viruses of birds, mammals and humans are in this category. Some of these viruses, for example, Asian influenza viruses, may cause severe and often fatal generalized infections. The nomenclature of these viruses is based on the first letter H and N of the spike glycoproteins haemagglutinin and neuraminidase, respectively. Thus, the earliest recognized virus was termed H1N1, followed by H2N2 and so on. The current bird-flu virus that causes sporadic human infections in Asia is termed H5N1. As the latter outbreaks indicate that H5N1 virus has crossed the species barrier from birds to humans, there is great concern that human-to-human transmission of this rather virulent virus may create a worldwide pandemic of avian flu. At the time of writing, there have been more than 60 human fatalities associated with avian flu transmitted directly from avian sources to humans, but no confirmed cases of human-to-human transmission (Chapter 23).
Paramyxoviruses
Paramyxoviruses are large, pleomorphic enveloped RNA viruses. The family contains four common and important human pathogens: measles, mumps, parainfluenza and respiratory syncytial viruses. Paramyxoviruses are a common cause of croup (laryngotracheobronchitis), while respiratory syncytial viruses cause regular winter epidemics of bronchiolitis/pneumonitis in infants.
Coronaviruses
These are enveloped RNA viruses with a helical nucleocapsid. They resemble orthomyxoviruses but have petal-shaped surface projections like a solar corona, hence the name. They infect both animals and humans. Most human infections lead to mild upper respiratory tract infections including the ‘common cold syndrome’. Human coronaviruses infect the respiratory tract by the airborne route, i.e. by inhalation or aerosols by coughs and sneezes of infected individuals. Additionally, inanimate reservoirs (i.e. fomites) are a secondary factor in transmission. Rhinoviruses together with coronaviruses are the major agents of the common cold. A coronavirus that crossed the ‘species barrier’ from civet cats in China to humans is the agent of severe acute respiratory syndrome (SARS). The latter infection – considered the first emerging infection of the new millennium – spread worldwide in 2003 causing many deaths particularly among health care workers (Chapter 23). Human coronaviruses are also implicated in gastroenteritis in infants.
Retroviruses
Retroviruses are large, spherical enveloped RNA tumour viruses characterized by a unique genome, a unique enzyme and a unique mode of replication. The viral genome RNA is first transcribed into DNA by a virus-specific enzyme, reverse transcriptase. This DNA can then serve as a template for messenger RNA (mRNA) synthesis. The RNA viruses infecting humans comprise a single taxonomic group with three subfamilies:
Viroids
As a result of advances in molecular biology, two new classes of infectious agents, prions and viroids, have been discovered. These are the smallest known agents of disease. Viroids cause diseases in plants and comprise naked, covalently linked, closed circles of ssRNA, less than 300–400 nucleotides in length. Despite their minute size, they replicate using host cell enzymes. Viroids are not associated with human disease, thus far. Prions are discussed at the end of this chapter.
Viral replication
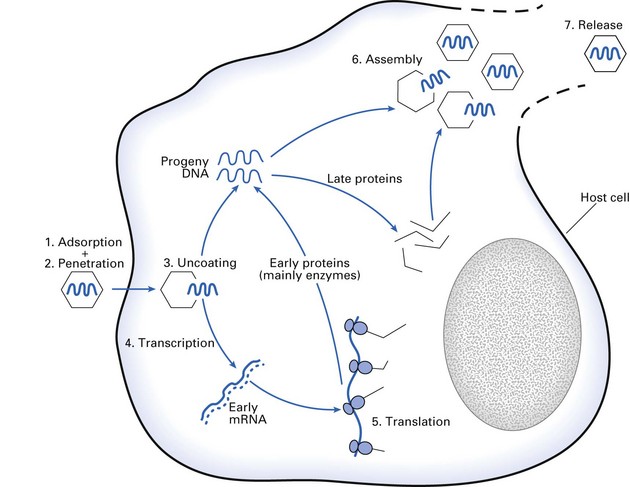
Viral replication (Fig. 4.5) is a highly complex process and only a brief summary is given here. There are a number of general steps in the replication cycle of all viruses: adsorption, penetration, uncoating and eclipse, transcription, synthesis of viral components, assembly and release of virions. In some viruses, however, these steps may not be clearly defined and may overlap, e.g. penetration and uncoating. It is noteworthy that in some families (e.g. Herpesviridae) many of the critical events occur in the cell nucleus, while others (e.g. Picornaviridae) multiply exclusively within the cytoplasm. The period between infection and the production of the new virion (eclipse or latent period) could be as short as 3 h (e.g. Orthomyxoviridae) or as long as several months or years (e.g. HIV).
Figure 4.5 depicts the steps in the replication of a DNA virus. However, this picture has to be somewhat modified when RNA viruses are considered, as the basic unit of information is now RNA instead of DNA. The strategies of viral replication become more complex when ds rather than ss viruses, and those with RNA of positive polarity and negative polarity are considered. The basic steps in replication are:
 endocytosis: most of the virions taken up by endocytosis appear to be degraded by lysosomal enzymes and therefore fail to initiate infection, but this is the normal route to successful infection by many viruses
endocytosis: most of the virions taken up by endocytosis appear to be degraded by lysosomal enzymes and therefore fail to initiate infection, but this is the normal route to successful infection by many virusesThe foregoing is a brief, composite picture of processes involved in viral multiplication. It should be noted that the replication cycle of each family of viruses has unique characteristics that differ from other viruses.
Cellular antiviral response
The antiviral response is mostly mediated immunologically and is described in Part 2.
Prions and prion diseases
Prions (proteinaceous infectious particles) are unique elements in nature, and they are the agents of a group of chronic diseases called prion diseases or transmissible spongiform encephalopathies. They infect essentially the nerve tissues of animals and humans, and manifest with long incubation periods lasting up to decades. The relevance of this chronic disease to dentistry relates to the realization (1) that the infectious agent is extremely difficult to destroy and (2) of the probability of infection transmission (variant Creutzfeldt–Jakob disease (vCJD)) in clinical settings.
The major features of prions are given below:
Prion-induced diseases or transmissible spongiform encephalopathies
Kuru
Kuru is the fatal neurological disease first described in societies in Papua New Guinea who consumed human brain. It is no longer prevalent owing to the cessation of this practice.
Creutzfeldt–Jakob disease
Creutzfeldt–Jakob disease (CJD) is a globally prevalent, rare, chronic encephalopathy; 10% of cases are familial and carry the mutated prion gene; the remainder are either acquired or sporadic. Onset is in middle to late life (40–60 years); the clinical course lasts for about 7–18 months.
Variant Creutzfeldt–Jakob disease
A variant form (vCJD) is localized to Europe, especially the UK; it almost always affects teenagers or young adults, with a mean age of onset of 24 years. Up to now more than 120 individuals have been affected in the UK alone. The disease is spread by consumption of prion-infected animal tissues.
Pathogenesis
Prions appear to replicate incessantly, first in lymphoid tissue, and then in brain cells where they produce intracellular vacuoles and deposition of altered host prion protein (PrPSc). These vacuoles give rise to the sponge-like appearance of the brain on microscopic examination. The disease is uniformly fatal.
Transmission
Kuru is transmitted in infected human brain by cannibalism.
The mode of transmission of CJD is mostly unknown. There are a few reports of iatrogenic transmission by medical and surgical procedures; hereditary acquisition occurs in familial cases; contaminated food (beef from cattle with ‘mad cow’ disease or bovine spongiform encephalopathy) is thought to cause acquired disease.
Prevention and dental implications
North American guidelines
For patients with highly suspected or confirmed CJD or vCJD:
 Either use disposable instruments or clean instruments thoroughly and sterilize for 18 min at 134°C in a vacuum autoclave (i.e. repeat the standard cycle six times).
Either use disposable instruments or clean instruments thoroughly and sterilize for 18 min at 134°C in a vacuum autoclave (i.e. repeat the standard cycle six times).British guidelines
Special precautions for patients with any form of CJD are not required, but strict adherence to standard precautions is required (Chapter 37).
Key facts
Collier L., Oxford J. Human virology: A text for students of medicine, dentistry and microbiology. Oxford: Oxford University Press; 1993.
. Epidemiologic concepts and methods, Ch. 1. Evans A.S., Kaslow R.A., editors. Viral infections of humans. Epidemiology and control, 4th ed., New York: Plenum, 1997.
Field D.N., Knipe D.M., Howlley P.M., editors. Virology, 3rd ed., Philadelphia: Lippincott-Raven, 1996.
Kohn W.G., Collins A.S., Cleveland J.L., Harte J.A., Eklund K.J., Malvitz D.M. Guidelines for infection control in dental health-care settings. Morbidity and Mortality Weekly Report. 2003;52(RR17):1-61. 19 December 2003
Porter S.R. Prion disease: Possible implications for oral health care. Journal of the American Dental Association. 2003;134:1486-1491.
Samaranayake L.P., Peiris J.S.M., Scully C. Ebola virus infection: An overview. British Dental Journal. 1996;180:264-266.
Scully C., Samaranayake L.P. Clinical virology in oral medicine and dentistry, Chs 1 and 2. Cambridge: Cambridge University Press. 1992.
Review questions (answers on p. 351)
Please indicate which answers are true, and which are false.