CHAPTER 1 Healing
The basic processes of soft tissue healing underlie all treatment techniques for sports injuries. We need to know what occurs in the body tissues at each successive stage of healing to be able to select the treatment technique which is most appropriate for that time. A technique aimed at reducing the formation of swelling, for example, would be inappropriate when swelling had stopped forming and adhesions were the problem. Similarly, a manual treatment designed to break up adhesions and mobilize soft tissue would not be helpful when inflammation is still forming and the tissues are highly irritable.
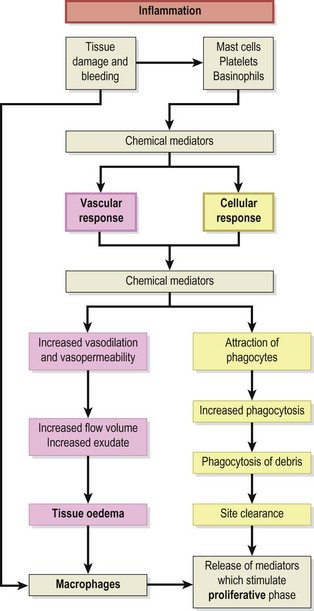
The stages of healing are, to a large extent, purely a convenience of description, since each stage runs into another. The term phasing rather than separate stages may be more suitable. Traditionally, the initial tissue response has been described as inflammation, but some authors see inflammation as a response separate to the processes occurring at the time of injury. Van der Meulen (1982) described both in terms of the ‘reaction phase’, arguing that the classical inflammatory period is preceded by a short (10 minute) period before the inflammatory mechanism is activated. Others (Hunter, 1998), looking at the changes in strength of the healing tissue, have termed the first stage the ‘lag phase’ because tissue strength does not change. In any traumatic injury the initial stage is bleeding which is the precursor for the inflammatory cascade seen as both a vascular and cellular response.
The second stage of healing has been variously called repair, proliferation and regeneration. The tertiary stage has been termed remodelling (Van der Meulen, 1982; Kellett, 1986; Dyson, 1987). The terms injury, inflammation, repair and remodelling will be used in this text.
When describing the stage of healing, the terms acute, subacute and chronic are helpful. The acute stage (up to 48 hours following injury) is the stage of inflammation. The subacute stage, occurring between 14 and 21 days after injury, is the stage of repair. The chronic stage (after 21 days) is the stage of remodelling. The term chronic is also sometimes used to describe self-perpetuating inflammation, where the inflammatory process has restarted due to disruption or persistent irritation of the healing tissues. The total healing process occurs over a continuum, shown in Fig. 1.1.
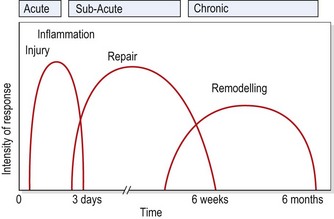
Figure 1.1 Timescale for healing.
From Oakes, B.W. (1992) The classification of injuries and mechanisms of injury, repair and healing. In Textbook of Science and Medicine in Sport (eds J. Bloomfield, P.A. Fricker and K.D. Fitch). Blackwell Scientific Publications, Melbourne. With permission.
Definition
Treatment must be adapted to the stages of healing, which are injury, inflammation, repair and remodelling.
Injury
This stage represents the tissue effects at the time of injury, before the inflammatory process is activated. With tissue damage, chemical and mechanical changes are seen. Local blood vessels are disrupted causing a cessation in oxygen to the cells they perfused. These cells die and their lysosome membranes disintegrate, releasing the hydrolysing enzymes the lysosomes contained. The release of these enzymes has a twofold effect. First they begin to break down the dead cells themselves, and second, they release histamines and kinins which have an effect on both the live cells nearby and the local blood capillary network.
The disruption of the blood vessels which caused cell death also causes local bleeding (extravasated blood). The red blood cells break down, leaving cellular debris and free haemoglobin. The blood platelets release the enzyme thrombin which changes fibrinogen into fibrin. The fibrin in turn is deposited as a meshwork around the area (a process known as walling off). The dead cells intertwine in the meshwork forming a blood clot. This network contains the damaged area.
The changes occurring at injury are affected by age (Lachman, 1988). Intramuscular bleeding, and therefore haemorrhage formation, are more profuse in individuals over 30 years of age. The amount of bleeding which occurs will be partially dependent on the vascularity of the injured tissues. A fitter individual is likely to have muscle tissue which is more highly vascularized, and therefore greater bleeding will occur with muscle injury. In addition, exercise itself will affect gross tissue responses. Muscle blood flow is greatly increased through dilatation of the capillary bed, and again bleeding subsequent to injury will be greater.
Inflammation
The next stage in the healing sequence is that of inflammation summarized in Fig. 1.2. This may last from 10 minutes to several days, depending on the amount of tissue damage which has occurred. The inflammatory response to injury is the same regardless of the nature of the injuring agent or the location of the injury itself (Hettinga, 1990). Various agents can give rise to injury, and Evans (1990a) listed physical, thermal, radiational, electrical and chemical causes.
Inflammation is not simply a feature of soft tissue injuries. It occurs when the body is infected, in immune reactions and when infarction stops blood flowing to an area. Some of the characteristics of the inflammatory response have even been described as excessive (Cyriax, 1982) and better suited to dealing with infection, by preventing bacterial spread, than healing injury (Evans, 1990a).
The cardinal signs of inflammation are heat (calor), redness (rubor), swelling (tumor) and pain (dolor). These in turn give rise to the so-called fifth sign of inflammation disturbance of function of the affected tissues (functio laesa).
Keypoint
Inflammation is often seen as undesirable. However, inflammation is the first stage of healing and so is a vital step on the road to recovery. The aim should be to prevent excessive inflammation and move the athlete on through the stages of healing towards eventual full function.
Heat and redness
Heat and redness take a number of hours to develop, and are due to the opening of local blood capillaries and the resultant increased blood flow. Chemical and mechanical changes, initiated by injury, are responsible for the changes in blood flow.
Chemically, a number of substances act as mediators in the inflammatory process. The amines, including histamine and 5-hydroxytryptamine (5-HT or serotonin) are released from mast cells, red blood cells and platelets in the damaged capillaries and cause vessel dilatation and increased permeability lasting 10–15 minutes (Lachman, 1988). Kinins (physiologically active polypeptides) cause an increase in vascular permeability and stimulate the contraction of smooth muscle. They are found normally in an inactive state as kininogens. These in turn are activated by the enzyme plasmin, and degraded by kininases.
The initial vasodilatation is maintained by prostaglandins. These are one of the arachidonic acid derivatives, formed from cell membrane phospholipids when cell damage occurs, and released when the kinin system is activated. The drugs aspirin and indometacin act to inhibit this change—hence their use as anti-inflammatory agents in sports injury treatment. The prostaglandins E1 and E2 are two of the substances responsible for pain production, and they will also promote vasodilatation, blood vessel permeability and lymph flow (Oakes, 1992).
Keypoint
Aspirin is one of a group of drugs called non-steroidal anti-inflammatory drugs (NSAIDs). These work by affecting prostaglandins, which are also involved in action of the stomach. For this reason NSAIDs may cause gastric irritation and should not be given to those with a history of peptic ulcers.
The complement system, consisting of a number of serum proteins circulating in an inactive form, is activated and has a direct effect on the cell membrane as well as helping to maintain vasodilatation. Various complement products are involved, and these are activated in sequence. Finally, polymorphs produce leukotrienes, which are themselves derived from arachidonic acid. These help the kinins maintain the vessel permeability.
Blood flow changes also occur through mechanical alterations initiated by injury. Normally, the blood flow in the venules, in particular, is axial. The large blood proteins stay in the centre of the vessel, and the plasmatic stream, which has a lower viscosity, is on the outside in contact with the vessel walls. This configuration reduces peripheral resistance and aids blood flow.
In a damaged capillary, however, fluid is lost and so the axial flow slows. Marginalization occurs as the slower flow rate allows white blood cells to move into the plasmatic zone and adhere to the vessel walls. This, in turn, reduces the lubricating effect of this layer and slows blood flow. The walls themselves become covered with a gelatinous layer (Wilkinson and Lackie, 1979), as endothelium changes occur (Walter and Israel, 1987).
Some 4 hours after injury (Evans, 1980) diapedesis occurs as the white cells pass through the vessel walls into the damaged tissue. The endothelial cells of the vessel contract (Hettinga, 1990), pulling away from each other and leaving gaps through which fluids and blood cells can escape (Fig. 1.3). Various substances, including histamine, kinins and complement factors, have been shown to produce this effect (Fox, Galey and Wayland, 1980; Walter and Israel, 1987).
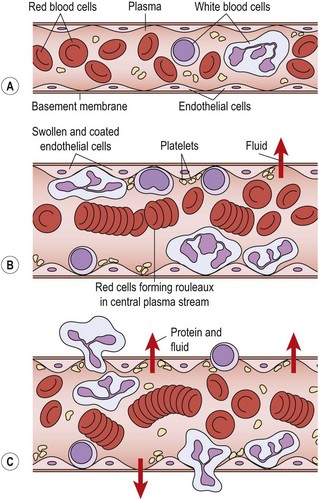
Figure 1.3 Vascular changes which occur in inflammation. (A) Blood vessel starts to dilate. (B) Dilated vessel showing marginalization. (C) White blood cells and fluid pass into tissue.
From Evans, D.M.D. (1990a) Inflammation and healing. In Cash’s Textbook of General Medical and Surgical Conditions for Physiotherapists (ed. P.A. Downie), 2nd edn. Faber and Faber, London. With permission.
Swelling
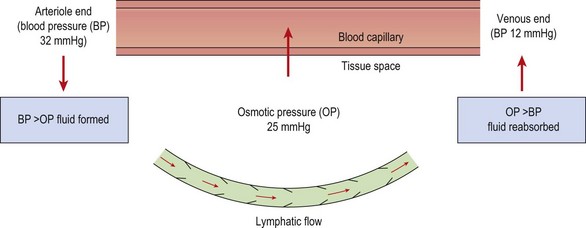
The normal pressure gradients inside and outside the capillary balance the flow of fluid leaving and entering the vessel (Fig. 1.4). The capillary membrane is permeable to water, and so water will be driven out into the interstitial fluid. However, because the tissue fluids usually contain a small amount of protein, and the blood contains a large amount, an osmotic pressure is created which tends to suck water back from the tissue fluid and into the capillary once more. The magnitude of this osmotic pressure is roughly 25 mmHg. At the arteriole end of the capillary the blood pressure (32 mmHg) exceeds the osmotic pressure and so tissue fluid is formed. At the venous end of the capillary the blood pressure has reduced (12 mmHg) and so, because the osmotic pressure now exceeds this value, tissue fluid is reabsorbed back into the capillary.
During inflammation the capillary bed opens and blood flow increases (heat and redness). The larger blood volume causes a parallel increase in blood pressure. Coupled with this, the tissue fluid now contains a large amount of protein, which has poured out from the more permeable blood vessels. This increased protein concentration causes a substantial rise in osmotic pressure, and this, together with the larger blood pressure in the capillary, forces fluid out into the interstitium, causing swelling.
Protein exudation in mild inflammation occurs from the venules only and is probably mediated by histamine (Evans, 1990a). More severe inflammation, as a result of trauma, results in protein exudation from damaged capillaries as well.
During inflammation, lymphatic vessels open up and assist in the removal of excess fluid and protein. The lymph vessels are blind-ending capillaries which have gaps in their endothelial walls enabling protein molecules to move through easily. The lymph vessels lie within the tissue spaces, and have valves preventing the backward movement of fluid. Muscular contraction causes a pumping action on the lymph vessels and the excess tissue fluid is removed to the subclavian veins in the neck.
Pain
Pain is the result of both sensory and emotional experiences, and is associated with tissue damage or the probability that damage will occur. It serves as a warning which may cause us to withdraw from a painful stimulus and so protect an injured body part. Unfortunately, pain often continues long after it has ceased to be a useful form of protection. Associated muscle spasm, atrophy, habitual postures, guarding and psychological factors all combine to make chronic pain almost a disease entity in itself.
Types of pain
Pain may be classified as somatogenic (acute or chronic), neurogenic or psychogenic. Chronic pain may be considered as pain which generally lasts for more than 6 weeks, while acute pain is pain of sudden onset which lasts for less than 6 weeks (Donley and Denegar, 1990).
Definition
Acute pain has a sudden onset and lasts for less than 6 weeks. Chronic pain lasts in excess of 6 weeks.
Musculoskeletal pain is not usually well localized—the surface site where the pain is felt rarely correlates directly to injured subcutaneous tissue. Generally, the closer an injured tissue is to the skin surface, the more accurate the athlete can be at localizing it.
Deep pain is normally an aching, ill-defined sensation. It usually radiates in a characteristic fashion, and may be associated with autonomic responses such as sweating, nausea, pallor and lowered blood pressure (Lynch and Kessler, 1990). Pain referral corresponds to segmental pathways, most often dermatomes. The extent of radiation largely depends on the intensity of the stimulus, with pain normally radiating distally, and rarely crossing the mid-line of the body (Cyriax, 1982).
Neurogenic pain is different again. Compression of a nerve root gives rise to ill-defined tingling, especially in the distal part of the dermatome supplied by the nerve. This is a pressure reaction, which quickly disappears when the nerve root is released. Greater pressure causes the tingling to give way to numbness. Compression or tension to the dural sleeve covering the nerve root gives severe pain, generally over the whole dermatome. In contrast, pressure on a nerve trunk usually causes little or no pain, but results in a shower of ‘pins and needles’ as the nerve compression is released. Pressure applied to a superficial nerve distally gives numbness and some tingling, with the edge of the affected region being well defined (Cyriax, 1982).
Irritability
Irritability may be defined as ‘the vigour of activity which causes pain’ (Maitland, 1991). It is determined by the degree of pain which the patient experiences, and the time this takes to subside, in relation to the intensity of activity that brought the pain on in the first place. The purpose of assessing irritability is to determine how much activity (joint mobilization, exercise, etc.) may be prescribed without exacerbating the patient’s symptoms.
An assessment of irritability may be made at the second treatment session. The amount of movement which the patient was subjected to in the previous session is known, as is the discomfort that he or she feels now. These subjective feelings are then used to determine the intensity of the second treatment session. Similarly, at the beginning of each subsequent treatment session the irritability is again assessed.
Keypoint
Irritability is a measure of the amount of pain a patient experiences as a result of movement (including that of treatment). Irritability should be used to guide the type and intensity of treatment to avoid excessive post-treatment soreness.
Treatment note 1.1 Pain description in examination
During both the subjective examination and the objective examination (see Treatment note 1.5, p. 24) the patient will usually describe pain. Both the type (nature) of pain and its behaviour are important factors in making an accurate clinical diagnosis, and a number of factors should be considered:
 When pain is decreasing the condition is generally resolving, while increasing pain suggests a worsening condition.
When pain is decreasing the condition is generally resolving, while increasing pain suggests a worsening condition. Constant pain which does not change with time, alteration of static posture or activities suggests a non-mechanical condition such as chemical irritation, tumours or visceral lesions (Magee, 2002).
Constant pain which does not change with time, alteration of static posture or activities suggests a non-mechanical condition such as chemical irritation, tumours or visceral lesions (Magee, 2002). Where pain changes (episodic pain), the therapist should try to determine what activities make the pain worse (exacerbation) and what make it better (remission).
Where pain changes (episodic pain), the therapist should try to determine what activities make the pain worse (exacerbation) and what make it better (remission). The therapist should try to determine if the pain is associated with particular events (e.g. movements, visceral function), or time of day.
The therapist should try to determine if the pain is associated with particular events (e.g. movements, visceral function), or time of day. Pain with sporting activity which reduces with rest in general suggests a mechanical problem, irritating pain sensitive structures.
Pain with sporting activity which reduces with rest in general suggests a mechanical problem, irritating pain sensitive structures. Morning pain which eases with movement indicates chronic inflammation which takes time to build up and reduces with movement.
Morning pain which eases with movement indicates chronic inflammation which takes time to build up and reduces with movement.The description of pain itself may indicate the structure causing it (see Table 1.1) and the behaviour of the pain on physical examination clarifies the picture.
Table 1.1 Pain descriptions and related structures
| Type of pain | |
|---|---|
| Cramping, dull, aching, worse with resisted movement | Muscle |
| Dull, aching, worse with passive movement | Ligament, joint capsule |
| Sharp, shooting | Nerve root |
| Sharp, lightning-like, travelling | Nerve |
| Burning, pressure-like, stinging, with skin changes | Sympathetic nerve |
| Deep, nagging, poorly localized | Bone |
| Sharp, severe, unable to take weight | Fracture |
| Throbbing, diffuse | Vasculature |
Source Magee (2002) and Petty and Moore (2001) with permission.
Recording pain
The intensity of pain may be recorded on a visual analogue scale (VAS). The patient is asked to indicate the pain description or number which best represents their pain. Where a 10 cm line is used the distance from the left of the scale to the point marked by the patient may be measured in millimetres and used as a numerical value (Fig. 1.5).
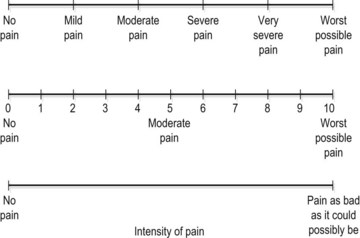
Figure 1.5 Visual analogue scales (VAS) used in pain description.
From Petty and Moore (2001) with permission.
Red flags
It is important for the therapist to appreciate when pain and other symptoms may suggest serious pathology which requires medical investigation—so called ‘red flags’ (Table 1.2). Where the patient has persistent pain and is generally unwell, the indication is that a pathology other than a musculoskeletal condition exists. In addition, changes in bladder and bowel habits, alteration in vision or gross changes in gait all require further investigation.
Table 1.2 Red flags in sport examination indicating medical investigation
| System/possible pathology | Pain behaviour |
|---|---|
| Cancer | Persistent night pain |
| Constant (25 hour) pain | |
| Unexplained weight loss (e.g. 4–6 kg in 10 days) | |
| Loss of appetite | |
| Unusual lumps or growths | |
| Sudden persistent fatigue | |
| Past history of carcinoma | |
| Cardiovascular | Shortness of breath |
| Dizziness | |
| Pain or feeling of heaviness in the chest | |
| Pulsating sensations in the body | |
| Discolouration in the feet | |
| Persistent swelling with no history of injury | |
| Gastrointestinal/genitourinary | Frequent or severe abdominal pain |
| Frequent heartburn or indigestion | |
| Frequent nausea or vomiting | |
| Change in bladder or bowel habits | |
| Unusual menstruation | |
| Neurological | Changes in hearing |
| Frequent or severe headache | |
| Problems in swallowing or changes in speech | |
| Gait disturbance, or problems with balance/ coordination | |
| Drop attacks (fainting) | |
| Sudden weakness |
Source Magee (2002) and Waddell, G., Feder, G. and Lewis, M. (1997) Systematic reviews of bed rest and advice to stay active for acute low back pain. British Journal of General Practice, 47, 647–652. With permission.
Pain production
Free or ‘bare’ nerve endings (type IV) respond to painful stimuli and are termed nociceptors. They are largely unresponsive to normal stimuli, but have a low threshold to mechanical and thermal injury, anoxia and irritation from inflammatory products. Tissues vary in the intensity of pain they will produce when stimulated. The joint capsule and periosteum are the most sensitive to noxious stimuli. Subchondral bone, tendons and ligaments are the next in line in terms of sensitivity, followed by muscle and cortical bone, the synovium and cartilage being largely insensitive.
The pain receptors are supplied by a variety of different nerve fibres. Skin receptors are supplied by thinly myelinated (A delta) fibres which carry ‘fast’ pain and respond to strong mechanical stimuli and heat above 45°C (Low and Reed, 1990). They give the initial sharp well-localized pain feeling (pinprick). The function of fast pain is to help the body avoid tissue damage and it often provokes a flexor withdrawal reflex.
Impulses from free nerve endings found in deeper body tissues are carried by non-myelinated C fibres. This is ‘slow’ pain, which tends to be aching and throbbing in nature, and poorly defined. Its onset is not immediate, and the sensation it produces persists after the pain stimulus has gone. The function of slow pain seems to be to enforce inactivity and allow healing to occur and it is therefore often associated with muscle spasm. The C fibres respond to many different types of stimuli and, as such, are said to be ‘polymodal’. However, they are most sensitive to chemicals released as a result of tissue damage. Histamine, kinins, prostaglandins E1 and E2, and 5-HT have all been implicated in this type of pain production during inflammation (Walter and Israel, 1987; Lachman 1988).
It can be seen that the pain experienced as a result of sporting injury will usually be either mechanical or chemical in nature. Mechanical pain is the result of forces which deform, or damage the nociceptive nerve endings, and so may be caused by stretching contracted tissue or by fluid pressure. This type of pain is influenced by movement. Chemical pain, on the other hand, results from irritation of the nerve endings, and is less affected by movement or joint position, but will respond to rest.
Articular neurology
In addition to pain receptors (type IV), three other joint receptors are important. Type I receptors are located in the superficial layers of the joint capsule. They are slow adapting, low-threshold mechanoreceptors, which respond to both static and dynamic stimulation. These receptors provide information about the static position of a joint, and contribute to the regulation of muscle tone and movement (kinaesthetic) sense. The type I receptors sense both the speed and direction of movement.
Type II receptors are found mainly in the deeper capsular layers and within fat pads. These are dynamic receptors with a high threshold, and they adapt quickly. They respond to rapid changes of direction of joint movement.
The type III fibres are found in the joint ligaments, and are again high threshold dynamic mechanoreceptors, but are slow adapting. These receptors monitor the direction of movement, and have a ‘braking’ effect on muscle tone if the joint is moving too quickly or through too great a range of motion. The type IV receptor is the nociceptor described above. Table 1.3 provides a synopsis of the various movement categories to which the receptors respond.
Table 1.3 Function of joint receptors
| Function | Receptor |
|---|---|
| Static position | Type I |
| Speed of movement | Type I |
| Change in speed | Type II |
| Direction of movement | Types I and II |
| Postural muscle tone | Type I |
| Tone at initiation of movement | Type II |
| Tone during movement | Type II |
| Tone during harmful movements | Type III |
Adapted from Hertling, D. and Kessler, R.M. (1990) Management of Common Musculoskeletal Disorders. JB Lippincott, Philadelphia.
Alteration in the feedback provided by joint receptors is of great importance following sports injury, and is dealt with in the section on proprioceptive training.
Pain pathways
Three categories or ‘orders’ of neurone make up the pain pathways. First order neurones travel from the pain receptors to the spinal cord, second order neurones travel within the cord to the brainstem and third order neurones travel from the brainstem to the higher centres of the cerebral cortex.
Keypoint
Type I joint receptors are found in the superficial layers of the joint capsule, type II in the deeper layers. Type III receptors are contained within the joint ligaments themselves.
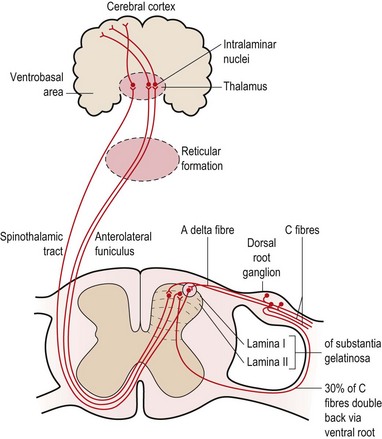
Seventy percent of the C fibres (slow pain) enter the spine via the dorsal root, while 30% of the fibres enter via the ventral root. The C fibres synapse with second order neurones in the substantia gelatinosa (SG) of the cord and these neurones ascend in the anterolateral funiculus on the opposite side of the cord (Fig. 1.6). From here they travel via the reticular formation to the intralaminar nuclei of the thalamus. The neurones synapse here once more and travel to the prefrontal region of the cerebral cortex. Some of the C fibres travel to the limbic system (cingulate gyrus) and generate emotional responses to pain (described as anxiety, fear and dread). C fibre pain is therefore poorly localized with a large emotional effect (White, 1999).
The A delta fibres (fast pain), on the other hand, synapse in the outer part of the posterior horn of the cord and cross to ascend in the spinothalamic tract to the ventrobasal nuclei in the thalamus, and then to the postcentral gyrus of the cortex.
Fast pain is registered in the parietal lobe and visceral pain in the insular cortex.
With more major injuries both fibres will produce a pain effect. The response to an ankle sprain, for example, will be an intense, well-defined stabbing sensation (A delta) followed by a dull ache accompanied by an emotional response (C fibre).
Pain relief mechanisms
Three concepts of pain control are generally used within physiotherapy:
The pain gate
The pain gate theory (Melzack and Wall, 1965) proposed that pain perception was regulated by a ‘gate’ which could be opened or closed. When stimulated, mechanoreceptors in the skin send impulses via A beta fibres to the posterior horn of the cord. Here, collateral branches are given off. These collaterals affect A delta and C pain fibres in the substantia gelatinosa (SG), reducing their excitability by presynaptic inhibition. Stimulation of A delta fibres by low intensity, high frequency transcutaneous electrical nerve stimulation (TENS) (100–200 Hz) will therefore reduce pain through this gating mechanism (Fig. 1.7).
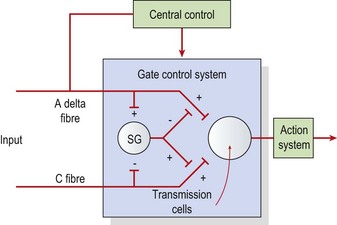
Figure 1.7 The pain gate mechanism. Central control—effects caused by higher centre (brain), action system—pain response. SG, substantia gelatinosa.
From Melzack, R. and Wall, P.D. (1965) Pain mechanisms: a new theory. Science, 150, 197. American Association for the Advancement of Science. With permission.
Endogenous opioids
Interneurones within the substantia gelatinosa are able to produce endogenous (made in the body) opioid peptides, which have similar effects to opioids such as morphine, heroin and codeine, which are some of the most powerful analgesics known. These opioids inhibit C fibre cells. Collateral branches of A delta fibres in the posterior horn connect to the interneurones and stimulate them to produce opioids.
The opioids, including enkephalins and endorphins, are produced in various areas of the central nervous system (CNS) including the limbic system and thalamus, pituitary gland, substantia gelatinosa and nerve terminals. These substances inhibit transmission in the A delta and C fibre pathways and so block pain before it reaches sensory levels. Stimulation of the A delta fibres with high intensity low frequency TENS (2–10 Hz) will damp down C fibre activity and reduce pain through this method.
Descending inhibition
Descending inhibition occurs when A delta fibres activate a chain of neurones which travel down the length of the spinal cord. Two separate systems are generally said to be involved (White, 1999), one involving serotonin, the other noradrenaline (norepinephrine). In the first, fibres from the periaqueductal grey matter (PAG) of the midbrain travel to the nucleus raphe magnus and then to the stalked cells in the dorsal horn of the spinal cord. In these cells, serotonin is released, which in turn causes the release of enkephalin to inhibit the cells of the SG. In the second system, the arcuate nucleus of the hypothalamus activates nuclei in the brainstem. Descending fibres in turn release noradrenaline (norepinephrine) into the dorsal horn of the cord to again inhibit the SG. Stimulation at frequencies above 50 Hz may affect this system (De Domenico, 1982).
Treatment note 1.2 Dry needling
Dry needling is an acupuncture technique which involves using a solid (no bore) atraumatic needle inserted into the body tissues. Pain relief has been described through both the pain gate mechanisms and diffuse noxious inhibitory control. In addition, according to Oriental medicine pain can be a result of a blockage and/or stagnation of acupuncture energy (qi) and acupuncture is designed to increase the flow of qi energy to stimulate healing and pain relief.
Both painful points (trigger points) and classic acupuncture points may be used. Classic acupuncture points are needled to specific depths described in acupuncture literature (see Norris, 2001). Trigger points may be either needled superficially to a depth of 0.5 cm (Baldry, 1998) or up to 8–10 cm (Gunn, 1996), depending on the mechanism being used. Superficial needling is said to activate A delta nerve fibres responsible for acupuncture pain relief, while deep needling is said to reduce pain but also induce healing through the production of platelet derived growth factor (PDGF) to stimulate new collagen formation (Gunn, 1996).
It should be noted that dry needling carries risks, and requires postgraduate training. Deep needling of the type used in intramuscular stimulation (IMS) in particular is a technique which involves highly specialist training (see Training section at the end of this box).
Technique
Acupuncture needles used for musculoskeletal conditions are of pre-sterilized disposable stainless steel construction and they vary from 0.22 mm to 0.45 mm in diameter and may be up to 4 inches in length. The patient’s skin should be cleaned prior to the treatment and the practitioner’s hands should be washed and sterilized with an alcohol-based sanitizer before treatment.
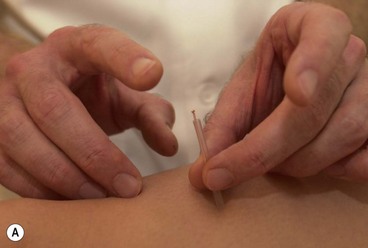
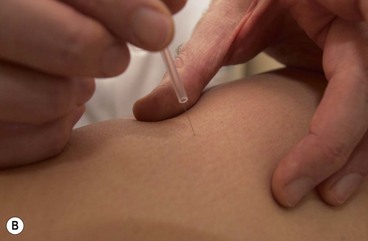
The needles are normally inserted using a plastic guide tube which is slightly shorter than the needle (Fig. 1.8). The guide tube is placed on the skin with the needle within it and the needle inserted through the skin surface with a small, brisk tap. Once the needle is inserted the guide tube is removed, and the needle pressed in further to the required depth (Fig. 1.9). Throughout this procedure, sterile practice must be maintained. To guard against infection, each needle is only used once and the shaft of the needle should not be touched.
The needle is often manipulated by rotation, scraping or flicking to increase the sensation felt by the patient—known as sensory propagation along channels or deqi in Oriental medicine. The needle may be inserted perpendicular to the skin or at oblique and transverse angles (Fig. 1.10). Once the needle is withdrawn the acupuncture point may be pressed with a cotton bud or probe to reduce the likelihood of bruising.
Needles are in general inserted for between 10 and 15 minutes, though shorter insertion techniques may be used to directly stimulate the muscle through intramuscular stimulation (IMS). In addition to muscular treatment, the periosteum of the bone itself may be used as a form of treatment, particularly for joint degeneration. The aim is to strike the periosteum, and the sensation felt is a dull ache rather than the immediate pain of a tissue needle.
Precautions and contraindications
Patients who are on anticoagulant therapy and those who have haemophilia should not be treated with dry needling. Diabetic patients should be closely monitored if they are to have needles inserted into hands and feet. Patients with prosthetic or damaged heart valves should be treated with caution. Epileptic patients should not be treated if they have had a fit during the last 3 months. Points on the lower abdomen and the lumbar sacral region should be used with caution during the first trimester of pregnancy. Following needling, it is common for patients to feel drowsy and they should be warned about this.
It is vital that the practitioner using dry needling has a thorough knowledge of underlying anatomy as trauma to body tissues, including the lungs and other organs, has been reported.
Management of inflammation
The effects of acute inflammation can be reduced by slowing the body’s response represented by the cardinal signs. Redness and heat are therefore treated by trying to reduce localized bleeding through the use of cold or ice and compression. Swelling is similarly managed by the use of compression to contain local oedema, and gentle movement to assist lymphatic drainage. By reducing the chemical and mechanical effects of the three inflammatory signs above, pain is also reduced.
The amount of rest prescribed during inflammation will depend on the stage of inflammation and the amount of tissue damage.
Chronic inflammation
Inflammation is the beginning phase in the healing process. Ordinarily tissues progress through the healing process sequentially to restore full function. However, in certain cases injuries remain in the inflammatory period, like a computer program that has become stuck in a loop. This is then termed chronic inflammation and occurs because macrophages have been unable to completely clear (debride) the area of foreign substances. This material may be dead cells, extracellular blood, or sand or dirt in some cases. Either way, the material is surrounded by collagen to isolate it from the body. This mass of encapsulating scar is called a granuloma.
Definition
A granuloma is a mass of collagen which occurs in chronic inflammation. It surrounds and isolates foreign material in a wound.
Chronic inflammation has been shown to have a low concentration of growth factors and a high concentration of protease (Hom, 1995). Adding growth factor to a chronic wound has been shown to improve healing in a number of soft tissues. Platelet derived growth factor has been used to treat ligaments and tendons in general (Evans, 1999) whilst insulin like growth factor (IGF-1) has been used with the achilles tendon (Kurtz, Loebig and Anderson, 1999) and articular cartilage (Nixon, Fortier and Williams, 1999).
Repair
Inflammation may continue for 5 days, but with minor trauma it is usually complete by the third day after injury (Evans, 1980). Following this, tissue repair can take place. Repair is by resolution, organization or regeneration, depending on the severity of the injury and the nature of the injured tissues. A minor injury will result in acute inflammation as described above, and the phagocytic cells will clear the area. If there is little tissue damage, the stage of resolution will result in a return to near normal (Lachman, 1988). True resolution rarely occurs with soft tissue injuries, but is more common with inflammatory tissue reactions such as pneumonia.
On the periphery of the injured area, macrophages and polymorphs are active because they can tolerate the low oxygen levels present in the damaged tissue. Cellular division by mitosis is seen in the surrounding capillaries about 12 hours after injury. During the next 3 days capillary buds form and grow towards the lower oxygen concentration of the injured area. These capillaries form loops and blood begins to flow through them. This new capillary rich material is known as granulation tissue. Plasma proteins, cells and fluid pour out of these highly permeable vessels. The gradually increasing oxygen supply to the previously deoxygenated area means phagocytosis can now begin.
New lymphatic vessels bud out from the existing lymphatics, linking to form a renewed lymphatic drainage system. As this process is occurring, fibroblast cells multiply and move towards the injured tissue. By the fifth day after injury they begin to lay down fibrils of collagen, a process requiring adequate amounts of vitamin C.
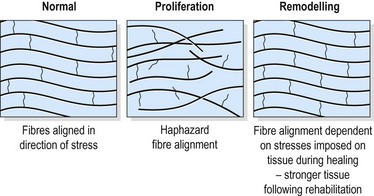
The individual fibrils form into parallel bundles lying in the direction of stress imposed on the tissue. If no movement occurs to stress the collagen bundles, they are laid down in a haphazard and weaker pattern (Cyriax, 1982). Controlled movement causes the fibrils to align lengthways along the line of stress of the injured structure (Burri, Helbing and Spier, 1973). Variation in longitudinal fibre alignment will determine the stress–strain response of the tissue to loading (Fig. 1.11). Where fibre alignment is parallel to the tissue body, the steep stress–strain curve (C) indicates that less deformation will occur for a given tissue loading. The tissue is therefore ‘stronger’.
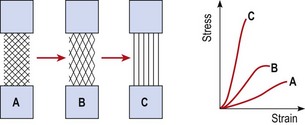
Figure 1.11 Relationship between fibre orientation and stress–strain response.
From Oakes, B.W. (1992). The classification of injuries and mechanisms of injury, repair and healing. In Textbook of Science and Medicine in Sport (eds J. Bloomfield, P.A. Fricker and K.D. Fitch). Blackwell Scientific Publications, Melbourne. With permission.
It becomes clear that external mechanical factors, and not the previous organization of the tissue, dictate the eventual pattern of fibril arrangement. Total rest during this stage of healing is therefore contraindicated in most cases.
Keypoint
External mechanical factors have a positive influence on tissue healing and can dictate the eventual strength of the healing tissues. Long periods of total rest are therefore rarely required when treating sports injuries.
In some tissue full regeneration occurs, damaged cells being replaced by functioning normal tissue. Fractured bone exhibits this property, as do torn ligaments and peripheral nerves providing conditions are suitable (Evans, 1990b).
Remodelling
The remodelling stage overlaps repair, and may last from 3 weeks to 12 months (Kellett, 1986). During this stage, collagen is modified to increase its functional capacity. Remodelling is characterized by a reduction in the wound size, an increase in scar strength, and an alteration in the direction of the collagen fibres (Van der Meulen, 1982).
Contraction of granulation tissue will occur for as long as the elasticity of the fibres will allow (Van der Meulen, 1982). Fibroblast cells transform into myofibroblasts which then form intercellular bonds. These contain contractile proteins (actomyosin) and behave much like smooth muscle fibres.
Three weeks after injury, the quantity of collagen has stabilized (Van der Meulen, 1982) but the strength of the fibres continues to increase. Strength increases are a result of an expansion in the number of cross-bonds between the cells, and the replacement collagen cells themselves. There is a continuous turnover of collagen, a process influenced by a number of factors, including the age of the patient, the type of tissue injured, the quantity of scar tissue present, the site and direction of the scar and external forces (Van der Meulen, 1982; Frank et al., 1983).
Final collagen fibre alignment should match the tissue function (Fig. 1.12). Fibres within a ligament will respond to a range of motion exercises which tense the ligament rhythmically. This may cause mild discomfort (VAS 2−3) but not pain (VAS 7−8). Pain is an indication of tissue damage indicating that the healing process has reverted back from the remodelling stage to the inflammatory stage. Fibres within muscle respond similarly, but to force transmission encountered by active and light resisted exercise during rehabilitation.
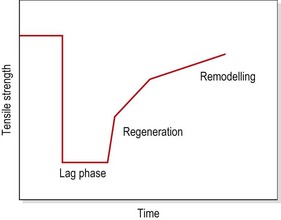
Matching treatment to the healing timescale
The tensile strength of injured soft tissue will reduce substantially after injury due to mechanical damage to the tissues. By the first postinjury day, tensile strength may have reduced by some 50%. Although healing begins immediately, collagen is not laid down until the fifth postinjury day (Garrett, 1990). The period between injury and the beginning of collagen synthesis has been described as the ‘lag phase’ (Fig. 1.13). Manual therapy techniques applied in this period should be aimed at pain resolution and oedema reduction. Only when collagen synthesis begins should therapy aim to prevent adhesions and align collagen fibres in the direction of stress.
Treatment note 1.3 Influencing the mechanical properties of healing tissue
Soft tissue manipulation
The use of deep transverse frictional massage (DTF) has been claimed to assist soft tissue healing. By applying shear and gliding movements to an injured tendon, ligament or muscle, tensile strength of the healing scar may be improved and adhesions reduced. Gentle DTF applied in the acute phase of healing could increase the rate of phagocytosis by inducing agitation of tissue fluid. In the chronic stage of healing the therapeutic movement produced by DTF is said to soften and mobilize adhesions (Kesson and Atkins, 1998).
Specific soft tissue mobilization
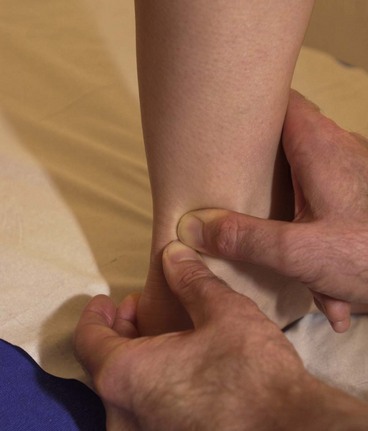
Specific soft tissue mobilization (SSTM) is a technique pioneered by Hunter (1998). The procedure involves tensioning the tissue using physiological joint movement and accessory joint movement and adding a dynamic soft tissue mobilization. In the acute phase of healing, SSTM is claimed to influence the mechanical properties of healing tissue by altering collagen and ground substance synthesis.
A sustained load of five repetitions of a 30-second hold is used in a treatment session and the patient performs home exercise by self-stretching for three repetitions of a 30-second hold every 3–4 hours. Using the Achilles tendon as an example (Fig. 1.14), the mobility of the tendon is assessed by subjecting it to shearing forces, beginning distally and moving proximally. The aim is to fix the proximal segment of the tendon with the fingers and then move the tendon in the opposite direction with the fingers of the other hand. The shearing motion is moved up the tendon progressively, assessing range and quality of movement.
This same shearing action is used with the tendon on stretch (passive loading) or loaded by mild muscle contraction (dynamic loading).
Eccentric loading
Eccentric loading has been used extensively for rehabilitation of the Achilles tendon. It is claimed that the controlled lengthening of the tendon during eccentric actions increases the tensile strength of the tendon (Stanish, Rubinovich and Curwin, 1986; Kannus, 1997) and allows for more storage of elastic energy in the stretch shorten cycle. In addition, eccentric loading may prepare the tendon for rapid unloading. The sudden release of force in this way produces shearing forces within the tendon which could conceivably break up adhesions within the Achilles itself (Curwin, 1994).
Individual tissue response to injury
In this section we will look at the responses of the individual tissues to injury, and the effects these have upon subsequent rehabilitation. Aspects of tissue structure relevant to sports injury are discussed.
Synovial membrane
The synovium consists of two layers, the intima, or synovial lining, and the subsynovial (subintimal) tissue. The intimal layer is made up of specialized cells known as synoviocytes, arranged in multiple layers. Two types of synoviocytes are present, type A cells, which are phagocytic, and type B cells, which synthesize the hyaluronoprotein of the synovial fluid. The two types are not distinct, however, and appear to be functional stages of the same basic cells (Hettinga, 1990).
The subsynovial tissue lies beneath the intima as a loose network of highly vascular connective tissue. Cells are interspaced with collagen fibres and fatty tissue. The subsynovial tissue itself merges with the periosteum of bone lying within the synovial membrane of the joint. Similar merging occurs with the joint cartilage through a transitional layer of fibrocartilage.
The blood vessels of the joint divide into three branches, one travelling to the epiphysis, the second to the joint capsule and the third to the synovial membrane (Paget and Bullough, 1981). From here the vessels of the subsynovium are of two types. The first is thin walled and adapted for fluid exchange, and the second thick walled and capable of gapping to allow particles, especially nutrients, to pass through.
Once free of the vessels, any material must pass through the synovial interstitium before entering the synovial fluid itself. The passage of this material is by diffusion on the whole, but by active transport for glucose molecules.
The synovium must adapt to movement with normal function of the joint. Rather than stretching, the synovium unfolds to facilitate flexion. The synovium is well lubricated by the same hyaluronate molecules found within the synovial fluid itself, and so the various layers slide over each other. Since the synovium must alter shape within the confines of the joint capsule, the process of synovial adaptation is at its best when the fluid volume of the joint is at a minimum.
Synovial fluid plays a significant role in joint stability. The negative atmospheric pressure within the joint creates a suction effect, which, aided by the surface tension of the synovial fluid, draws the bony surfaces of the joint together.
Response to injury
With minor trauma the synovium is not microscopically disturbed, but will instead suffer a vasomotor reaction (Hettinga, 1990). The synovium will dilate and fluid filtration increases. Protein leaks into the interstitium, changing the osmotic pressure and causing local oedema and joint exudation. This process constitutes a post-traumatic synovitis.
Keypoint
Post-traumatic synovitis initially involves a vasomotor reaction of the synovium rather than actual structural damage.
The slight hyperaemia gives way later to alterations of the intimal layer, the total number of layers increasing threefold. If the trauma does not continue, the protein molecules which were released are cleared by the lymphatics and the osmotic pressures return to normal. If mechanical irritation persists, the intimal layer will continue to thicken. The deep synovial cells now show increased activity and protein synthesis escalates.
Alterations occur in the number of type A and type B synoviocyte cells. The number of type A cells reduces as some of these move into the synovial fluid to become macrophages. The synovial lining becomes filled with fibroblasts, which in turn change into type B cells. Neutrophil cells die, releasing proteolytic enzymes which attack the near joint structures. This process can self-perpetuate the synovitis, even in the absence of further trauma, giving rise to a reactive synovitis.
Onset of symptoms following post-traumatic synovitis usually occurs between 12 and 24 hours after injury and can last for between 1 and 2 weeks. Patients complain mainly of joint tightness, with warmth, erythema and pain being encountered less often. The tightness is due to joint effusion, the increased fluid volume causing the normally negative intra-articular pressure to become positive.
The stability of the joint, no longer created by a negative intra-articular pressure, comes from joint distension instead. This places a traction force on the joint capsule and surrounding ligaments. Pressures are greatest in the effused joint in extremes of flexion and extension, and are reduced at about 30° flexion, this being the resting position taken up by the patient. Haemarthrosis is usually present if swelling occurs within 2 hours of injury, and pain is intense.
Keypoint
If synovitis continues beyond 72 hours the synovium thickens and proteolytic enzymes are released which attack the near joint structures. This process can continue in the absence of further trauma, and represents reactive synovitis.
Surgical removal of the synovial membrane (synovectomy) may be performed in a number of inflammatory joint disorders. With time the synovium will regrow, and the use of continuous passive motion (CPM) may be used to assist this process (Salter, 1990). Without mechanical stimulation synovial membrane will degenerate through the loss of proteoglycan, so movement is vital to its effective healing.
Synovial fluid
Synovial fluid is similar in many ways to blood plasma. The main differences being that synovia does not contain fibrinogen or prothrombin and so is unable to clot. The mucopolysaccharide hyaluronate (hyaluronic acid) secreted by the synoviocytes is contained within the fluid.
The amount of synovial fluid present in a joint is very little—about 0.5–4 ml within large joints such as the knee—and this is spread throughout the joint by structures such as the cartilage, menisci and fat pads.
Synovial fluid is a highly viscous fluid, which becomes more elastic as the rate of joint movement increases. As weight is taken by the joint, synovial fluid is squeezed out from between the opposing joint surfaces. This is resisted by the tenacity of the fluid itself.
As the joint moves, the synovial fluid is pulled in the direction of movement and so a layer of fluid is maintained between the joint surfaces. Any friction produced by movement will therefore occur within the synovial fluid rather than between the joint surfaces. When the joint is statically loaded, however, fluid flows away from the point of maximal load and the joint relies on the articular cartilage to provide lubrication (see below). The synovial fluid provides nutrition for about two-thirds of the articular cartilage bordering the joint space.
Following injury, fluid volume may increase as much as 10 or 20 times, with a decrease in hyaluronate and, with it, fluid viscosity. Pain due to the accumulation of synovial fluid is dependent not on the amount of fluid present, but on the speed with which it forms. Blau (1979) claimed that as much as 100 ml may be extracted from a joint which caused little pain because it took a long time to form, while 15 ml may be exquisitely painful if formed rapidly following trauma.
With injury in which bleeding is not present, the constituents of the synovial fluid remain basically the same. In reactive synovitis, the protein concentration is slightly elevated, and the number of white blood cells increases somewhat from normal values of 100/ml to as much as 300/ml. With post-traumatic synovitis, however, the white cell count is further increased, to as much as 2000/ml.
Haemarthrosis (blood within the joint) causes rapidly developing fluid, which contains fibrinogen. If the synovial membrane is torn, fat can enter the joint from the extrasynovial adipose tissue and will show up in the synovial fluid. The blood from haemarthrosis will mostly stay fluid, and is quickly absorbed by the phagocytic cells to ultimately disappear after several days.
Bone
Types
Bone is essentially a fibrous matrix impregnated with mineral salts (mainly calcium phosphate), and it therefore combines the properties of both elasticity and rigidity. It is a living tissue, which is continually remodelled, and subject to hormonal control. Bone injury within sport is assessed using x-ray, see Treatment note 1.4.
Treatment note 1.4 Diagnostic imaging
Diagnostic imaging is used to support a thorough examination and should not be relied upon as the only means of clinical diagnosis. In some cases physical examination will be superior to imaging, a situation recognized, for example, with ankle injury (see Ottawa ankle rules, Chapter 12).
Imaging types
There are several imaging types used within the management of sports injuries. Reflective imaging includes ultrasound (US) and magnetic resonance imaging (MRI).
With this type of imaging energy is passed into the tissues and reflected back to form an image. Ionizing radiation imaging includes standard x-rays (plain films) and computed tomography (CT). X-rays use radiation which passes through the tissues onto film to cause an image, whereas CT again uses radiation but converts the radiation into digital units to create slices (3 mm minimum) through the body. Emission imaging (bone scan) uses a radiopharmaceutical agent injected into the blood. This is taken up by areas of the body with increased metabolic activity and these show up as images.
Standard x-rays
X-rays pass through the tissues at different rates depending on tissue density. Air is the least dense (radiolucent) and metal the most dense (radio-opaque). X-rays pass easily through air creating the darkest image on x-ray film. The lungs, airways and colon therefore appear black. Fat is the next densest tissue and so appears as very dark grey and is found throughout the body. Fluid absorbs more of the x-rays than air or fat, giving a whiter image. Bone is the most dense naturally occurring tissue, appearing white on x-ray film. This may be either bone itself or where tissues have calcified, for example in myositis ossificans or calcific tendon pathology. Metal completely blocks x-rays and so metal implants are easily visualized on plain film as totally white.
Because x-rays pass through the body in a straight line, they create a planar image, that is one structure superimposed on another. Two images are therefore used lying at 90° to one another, most commonly anteroposterior (AP) and lateral (lat.). X-rays obey the laws of light, and so body positioning is important. The closer the body area to be imaged is to the x-ray film (further from the x-ray beam) the clearer the image will be.
X-ray analysis
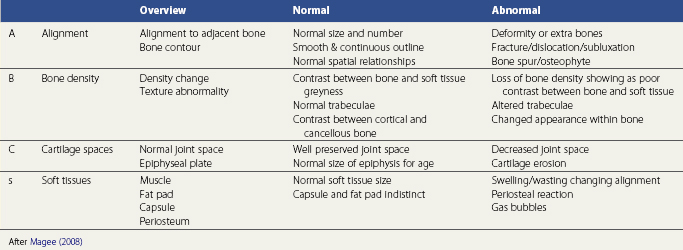
A systematic analysis of x-rays may be made using the ABCs mnemonic, standing for Alignment, Bone density, Cartilage and Soft tissue (Table 1.4).
 Alignment is assessed by comparing the contour of one bone to that of the adjacent bone. This is normally assisted by drawing a mental line along the edge of the bones. Changed alignment suggests altered stability through soft tissue changes and/or fracture.
Alignment is assessed by comparing the contour of one bone to that of the adjacent bone. This is normally assisted by drawing a mental line along the edge of the bones. Changed alignment suggests altered stability through soft tissue changes and/or fracture. Bone is assessed for changes in both density and dimension. In long bones the shaft should appear uniform with increased density towards the edge and less density within the medullary canal. The periosteum should not be lifted and the edge of the bone continuous. At the bone ends the trabeculae of the cancellous bone should be uniform. Bone dimension is compared to neighbouring segments, for example within the spine to the vertebrae above and below to identify wedging.
Bone is assessed for changes in both density and dimension. In long bones the shaft should appear uniform with increased density towards the edge and less density within the medullary canal. The periosteum should not be lifted and the edge of the bone continuous. At the bone ends the trabeculae of the cancellous bone should be uniform. Bone dimension is compared to neighbouring segments, for example within the spine to the vertebrae above and below to identify wedging.Two types of bone are generally described, cancellous or spongy bone and compact bone, and important differences exist in both their mechanical properties and methods of healing. Cancellous bone is found at a number of sites, including the bone ends, and is arranged in a system of trabeculae aligned to resist imposed stresses. The shaft of a long bone consists of a ring of compact bone surrounding a hollow cavity. This structure in turn is surrounded by a further layer of compact bone. The bone cavity contains marrow. In infants the bone marrow is red, but this is gradually replaced by yellow bone marrow until, by puberty, only the cancellous bone cavities at the ends of the long bones contain red marrow. With age the bone marrow in these cavities too is replaced, but red bone marrow may still be found in the vertebrae, sternum and ribs, as well as the proximal ends of the femur and humerus.
The bone is enclosed in a dense membrane called the periosteum, which is absent in the region of the articular cartilage. The periosteum is highly vascular and responsible for the nutrition of the bone cortex which underlies it. The deep layers of periosteum contain bone-forming cells (osteoblasts). These lay down successive layers of bone during growth, and so the periosteum is responsible for alterations in the bone width; in addition these cells play an important part in bone healing. In contrast, the bone cavity is lined by bone-destroying cells (osteoclasts) which erode the inner surface of the bone. A direct blow to the bone which produces bleeding beneath the periosteum will lift it, causing bone deposition by the osteoblasts. This is a common problem over the anterior tibia in footballers and hockey players.
Keypoint
A direct blow to unprotected bone (with a hard ball or boot, for example) may cause bleeding beneath the bone periosteum. Ultimately this may lead to calcification (bone deposits) within the bruised area.
Bone may be further classified into four major types. The long bones are found within the limbs, and consist of a shaft and two enlarged ends. Short bones have a block-like appearance such as those of the carpals, and are mainly cancellous bone. Flat bones are thinner, and consist of two layers of compact bone sandwiching a thin layer of cancellous bone; examples are the skull vaults and the ribs. Finally, irregular bones consist of a thin layer of compact bone surrounded by cancellous bone; examples are the vertebrae.
Development
Skeletal development begins with loosely arranged mesodermal cells which are mostly converted to hyaline cartilage. Between the seventh and twelfth intrauterine week a primary ossification centre appears within the shaft of the long bone, and spreads towards the bone ends. The centre of the shaft is hollowed out and filled with red bone marrow, and the whole shaft is called a diaphysis. At the end of the bone, secondary centres of ossification appear, usually after birth. Gradually the main part of the cartilage is replaced, leaving only the articular cartilage, and a cartilage plate (growth plate, or physis) between the shaft and end of the bone.
The growth plate is of great importance in paediatric sports medicine. This cartilage layer is responsible for the increase in bone length. As the cartilage grows it becomes thicker, and its upper and lower surfaces are converted to bone. Eventually the cartilage stops growing, but its ossification continues so that the cartilage becomes thinner, until it eventually disappears. At this point the diaphysis and epiphysis are united, and growth in length of the bone is no longer possible. The point at which this occurs may be influenced by a number of factors, including impact stresses (see below).
Intramembranous ossification occurs in the mandible, clavicle and certain bones of the skull (Palastanga, Field and Soames, 1989). Here, the intermediate stage of cartilage formation is omitted and the bone ossifies directly from connective tissue.
Epiphyseal injury
Two types of epiphysis are found. Pressure epiphyses are found at the end of long bones, and are interarticular. They are subjected to compression stress with weight bearing, and are responsible for changes in bone length. Traction epiphyses (apophyses) occur at the insertion of major muscles. They experience tension stress as the muscles contract, and alter bone shape.
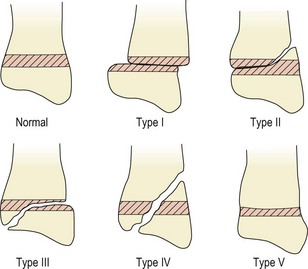
The growth plate itself forms a weak link in the immature skeleton, and shearing or avulsion stresses can cause it to be dislodged. Because the epiphyses are weaker than the major joint structures, injuries which in the adult would cause dislocation or tendon rupture may cause epiphyseal injury in a child.
Five types of injury have been described (Salter and Harris, 1963), as shown in Fig. 1.15. The type I lesion involves horizontal displacement of the growth plate while with the type II lesion the fracture line runs through to the adjacent metaphysis. In the type III and IV fractures the joint surface is involved, so the complication rate is higher (Watson, 1992). Gentle manipulation under anaesthetic is normally used to realign the epiphysis in the first fracture types. Crush injuries and those through the growth plate may cause more severe problems of growth disturbance. When a fracture crosses the growth plate, bone may fill the gap formed, giving unequal growth. Crush injuries may lead to premature closure of the plate with associated deformities of shortening and altered bony angulation.
Osteochondrosis
Osteochondrosis affects the pressure epiphyses during their growth period, and are the most common overuse injuries seen in children. There is an interference with the epiphyseal blood supply, causing an avascular necrosis to the secondary ossification centre. The bone within the epiphysis softens, dies and is absorbed (Fig. 1.16). Because the cap of articular cartilage surrounding the epiphysis receives nutrition from the synovial fluid, it remains largely unaffected. The combination of softening bone contained within an intact articular cap leads to flattening of the epiphysis. Gradually the dead bone is replaced by new bone by a process of ‘creeping substitution’. Trauma may be one factor which initiates the ischaemic process, either through direct injury or vascular occlusion as a result of traumatic synovitis (Gartland, 1987).
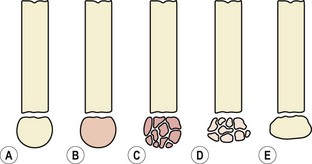
Figure 1.16 Stages of osteochondritis. (A) Normal epiphysis. (B) The bony nucleus undergoes necrosis, loses its normal texture and becomes granular. (C) The bony nucleus becomes fragmented during the process of removal of dead bone. (D) If subjected to pressure the softened epiphysis becomes flattened. (E) Re-ossification with restoration of normal bone texture, but deformity may persist. The whole process takes 2–3 years.
From Gartland (1987) with permission.
The osteochondroses may be categorized into four groups (Pappas, 1989):
Any pressure epiphysis may be affected by osteochondrosis, the condition taking the name of the person who first described it in that region. The most common conditions are shown in Table 1.5, and details of these are given in the relevant clinical chapters in Section II of this book.
| Classification | Name | Site(s) |
|---|---|---|
| Non-articular traction (pulling) | Osgood–Schlatter’s Sinding–Larsen—Johannsson’s disease Sever’s disease |
Tibial tubercle Interior pole of patella (quadriceps) Calcaneous (gastrocnemius) |
| Articular subchondral (crushing) | Perthes’ disease Kienböck’s disease Köhler’s disease Freiberg’s disease |
Femoral head Lunate (wrist) Navicular (mid-foot) Second metatarsal head |
| Articular chondral (splitting) | Osteochondritis dissecans | Medical femoral Condyle (knee) Capitellum (elbow) Talar dome (ankle) |
| Physeal | Scheuermann’s disease Blount’s disease |
Thoracic spine Tibia (proximal) |
Healing
Bone healing is governed by a number of factors, including the type of tissue which is damaged, the extent of the damage and position of the bony fragments, the amount of movement present at the fracture site as healing progresses and the blood supply. In a long bone, healing may be divided into five stages, as shown in Fig. 1.17 (Crawford-Adams, 1978; Apley and Solomon, 1988).
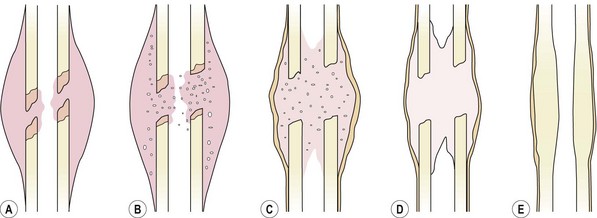
Figure 1.17 Stages of fracture healing. (A) Haematoma. Tissue damage and bleeding occur at the fracture site, the bone ends die back a few millimetres. (B) Inflammation. Inflammatory cells appear in the haematoma. (C) Callus. The cell population changes to osteoblasts and osteoclasts, dead bone is mopped up and woven bone appears in the fracture callus. (D) Consolidation. Woven bone is remodelled to resemble the normal structure (E).
From Apley and Solomon (1988) with permission.
The rate of healing is dependent on a number of factors. The type of bone is important, cancellous bone healing far more quickly than cortical bone. Also, the fracture type will dictate the speed of healing, with a spiral fracture healing more quickly than a transverse type. If the blood supply has been compromised at the time of injury, or if the fracture has occurred in an area with a poor blood supply, the healing rate will be slower. The age and health of the patient are other determining factors, with fractures in children healing almost twice as fast as those in adults. In general terms, callus may be visible radiographically within 2–3 weeks of injury, with firm fracture union taking about 4–6 weeks for upper limb fractures and 8–12 weeks for those in the lower limb. Full consolidation may take as much as 8 and 16 weeks for upper and lower limb fractures, respectively.
In contrast to long bone, cancellous bone remains fairly immobile when fractured, and heals by ‘direct repair’ with a minimum of callus formation. The main difference occurs because there is no medullary canal in cancellous bone, and the area of contact between the two injured bone fragments is much greater. Following haematoma formation, new blood vessels and osteogenic cells penetrate the area and meet similar tissue from the opposite bone fragment. The intercellular matrix which is laid down by osteoblasts is calcified into woven bone. This type of healing also occurs when internal fixation is used.
Mechanical properties
Bone responds to mechanical stress in similar ways to other connective tissue, but at a considerably slower rate. Its ability to adapt its structure, size and shape depends on the mechanical stresses placed upon it. When stress is reduced, by prolonged bed rest for example, mineral reabsorption occurs and the bone reduces in strength. Raising stress to an optimal level, by exercise, leads to an increase in bone strength.
In addition to changes in total mineral content, bone varies its strength according to the direction of the imposed stress. At bony attachment sites such as tubercles, alignment of collagen fibres is parallel to the direction of the imposed force. In the shaft of a long bone, fibre orientation is along the bone axis, indicating that this part of the bone is designed to resist tension and compression forces. In cancellous bone at the epiphysis, shear stresses are maximal and so fibre alignment is in the direction of the shearing forces.
Keypoint
As with other tissues in the body, bone responds to mechanical stress by adapting its structure. If stress is reduced (prolonged rest) bone reduces in strength. Raising stress to an optimal level (exercise) leads to an increase in bone strength.
A number of mechanisms have been proposed to explain how the bone remodels to imposed stress. Bassett (1965) argued that, in theory, mechanical stresses on bone create electrical charges (piezoelectric effect). Areas of compression were said to develop negative charges while those subjected to tensile stress are more positive. The suggestion is that bone deposition by osteoblasts occurs in negatively charged areas, while reabsorption by osteoclasts takes place in regions of positive excitation. Experiments with dogs (Bassett and Pawlick, 1964) have shown that new bone is laid down at an implanted cathode site, although other authors have described bone deposition at both negative and positive sites (Hert and Zalud, 1971) so the effects of polarity are still uncertain.
Electrical fields formed in bone as a result of imposed stresses have been shown to stimulate protein synthesis in frog osteogenic cells (Becker and Murray, 1970), and increased rate of bone formation has been shown as a result of direct current (d.c.) administered through implanted electrodes (Becker, Spadaro and Marino, 1977).
Bone trabeculae subjected to maximum stress will be strengthened by bone deposition, in the direction of the imposed force. Regions of high stress have denser trabeculae, aligned to minimize the bending effect on a bone. Where a bone is subjected to an unbalanced force, which would tend to bend it, remodelling occurs as a result of changes in surface strain (Reigger, 1990). On the convex surface of the bone, osteoclastic activity leads to bone reabsorption, while on the concave surface the reverse occurs and bone deposition is seen. Bone therefore moves towards the concavity which has been induced through loading of the shaft, to minimize the bending effect, a process known as flexural drift. The stress imposed on the bone causes an increase in the calcium concentration found within the interstitial fluid, as a result of changes in bone crystal solubility.
Osteoporosis
Osteoporosis (bone mineral loss) has now reached epidemic proportions in the Western world (Carbon, 1992). The condition involves a progressive decrease in bone mineral density (BMD), due to an imbalance between bone formation and bone reabsorption. Bone mass reaches its peak in the third and fourth decade, and loss occurs normally in both sexes with ageing, but this rate is increased markedly with osteoporosis. Normal bone loss of 3% per decade may increase to as much as 10% per decade for trabecular bone. This leads to a reduction per decade in ultimate stress of 5%, ultimate strain of 9% and energy absorption of 12% (Behiri and Vashishth, 2000).
Osteoporosis may be either primary, governed by age and sex, or secondary as a result of disease. The most common primary types are postmenopausal osteoporosis and senile osteoporosis.
Definition
Osteoporosis may be either primary, governed by sex (postmenopausal) or age (senile), or secondary resulting from disease.
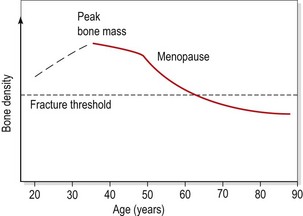
The loss of mass makes the bone susceptible to fracture, particularly as a result of microtrauma. Common fracture sites include the distal radius and vertebrae for postmenopausal osteoporosis, and the proximal femur for senile osteoporosis. Fracture of the distal radius presents as a Colles’ fracture, while vertebral wedging gives rise to an increased thoracic kyphosis or ‘dowager’s hump’.
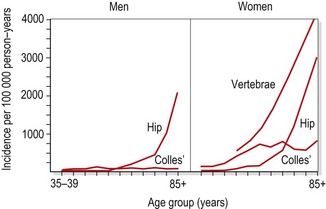
There is a marked increase in the risk of fracture once bone density reduces below a certain level (known as the fracture threshold, Fig. 1.18). The incidence of hip and vertebral fractures rises with age (Fig. 1.19), but the rise in hip fracture incidence in men occurs some 5 years later than that in women (Wolman and Reeve, 1994), an indication of the effect of postmenopausal osteoporosis.
Assessment of osteoporosis
Changes in BMD is the standard objective measure for osteoporosis, but low bone density in itself is asymptomatic. It is only when a fracture occurs or they develop pain or substantial postural changes that individuals seek help. Assessment of BMD may be made by direct examination of x-rays. Cortical index measures the thickness of the bone cortex in comparison to the bone shaft. The combined width of the two cortices should be 50% of the total width of the bone at mid-shaft (Apley and Solomon, 1993). Trabecular index is a method of assessing bone mass from the radiographic pattern of bone trabeculae in the proximal femur or calcaneus. A more accurate measure of BMD is obtained by dual energy x-ray absorpiometry (DXA). The amount of radiation used is relatively small, representing no more than 3% of the natural background radiation in a large city (Lewis, Blake and Fogelman, 1994; Huda and Morin, 1996). Measurement is taken yearly, and only changes greater than 2–3% are significant (Bennell, Khan and McKay, 2000). Results are expressed as BMD in g/cm2, as a Z score which compares the value to an age matched group, or as a T score which compares the obtained value to a young healthy population. The World Health Organization has classified degrees of BMD loss as shown in Table 1.6.
Table 1.6 Classification of osteoporosis
| Classification | DXA reading |
|---|---|
| Normal | >−1 |
| Osteopenia | −1 to −2.5 |
| Osteoporosis | <−2.5 |
| Severe osteoporosis | <−2.5 plus history of fragility fracture |
BMD expressed as standard deviations (SD) below the mean of a young adult (T score). World Health Organization (1994) Assessment of Fracture Risk and its Application to Screening for Osteoporosis. Report of WHO study group. World Health Organization, Geneva. With permission.
Subjective assessment includes elements such as family history, fracture status, history of falls, menstrual history, smoking and dietary habits, and exercise status. Objective examination is of posture, pain and functional limitation (CSP, 1999; Bennell, Khan and McKay, 2000).
Three main factors are important in the development of osteoporosis: diet, oestrogen level and physical activity.
Diet
Modern diet can fail to provide an adequate daily intake of calcium. Recommended requirements as high as 1500 mg of calcium and 400 IU of vitamin D have been made for postmenopausal women (MacKinnon, 1988), but many women may consume as little as 300 mg of calcium (McArdle, Katch and Katch, 1986), placing themselves in negative calcium balance. Excess salt or caffeine, and a large intake of meat promote calcium loss in the urine, and an excessive intake of fibre and alcohol can bind calcium in the gut, preventing its absorption (Deakin and Brotherhood, 1992).
Calcium deficiency in animals has been shown to produce osteoporosis (Martin and Houston, 1987), but effects in man are less clear. Studying early postmenopausal women, Nilas, Christiansen and Rodbro (1984) gave a 500 mg calcium supplement over a 2-year period and assessed bone density in the distal radius, while Ettinger, Genant and Cann (1987) gave a 1000 mg supplement and assessed the lumbar vertebrae. Both of these studies failed to show any significant differences between the treatment and non-treatment groups. However, in late postmenopausal women calcium supplementation may be more beneficial (MacKinnon, 1988). However, the effect of calcium supplementation on the incidence of fractures remains unproven (Evans, 1990b).
Adequate calcium is clearly necessary for health, and those individuals who show a deficiency in their calcium intake may need dietary supplementation. Others should receive advice on good diet to enable them to maintain an adequate intake of calcium and vitamin D, remembering that excessive vitamin D intake can be toxic.
Exercise
Weight-bearing exercise creates bone stress and acts as a stimulus for maintaining bone mass. Loss of bone mass has been reported both as a result of prolonged bed rest (Donaldson, Hulley and Vogel, 1970) and following weightlessness (Mazess and Whedon, 1983). Similarly, athletes have been shown to have greater bone density than non-athletes (Nilsson and Westlin, 1977), and tennis players have been shown to have a greater bone density in their dominant arm (Huddleston, Rockwell and Kuland, 1980).
A number of authors have demonstrated the beneficial effects of weight-bearing exercise in slowing bone loss. Smith, Smith and Ensign (1984) assessed the effects of exercise (45 minutes, 3 days per week) on bone loss in postmenopausal women and showed a 1.4% increase in bone mass during the second and third years of their study. Krolner, Toft and Nielsen (1983) studied the effects of exercise (1 hour, twice per week) on postmenopausal women. They showed a 3.5% increase in bone mineral content of the lumbar spine for the exercising group compared to 2.7% for their control.
High impact exercise which generates a ground reaction force greater than twice body weight is likely to create bone remineralization (Heinonen, Kannus and Sievanen, 1996). Exercise should be performed at least three times per week at intensities beginning with 40–60% maximal oxygen uptake (VO2 max) and progressing to as much as 80% VO2 max, even in the elderly (ACSM, 1998). Strength (weight) training at 70% 1 RM has been shown to maintain hip and spine BMD, but programmes must be given progressively, taking clinical history into account. A flowchart to guide exercise prescription is shown in Fig. 1.20.
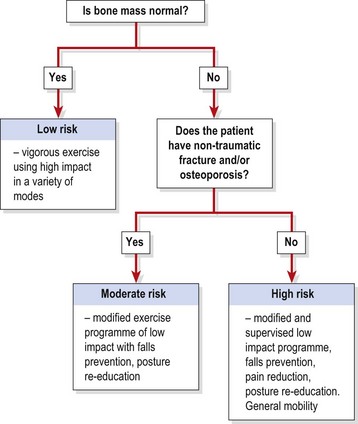
Figure 1.20 Exercise prescription for osteoporosis.
Adapted from Bennell, Khan and McKay (2000), with permission.
Clearly, regular weight-bearing exercise is important for the prevention and management of osteoporosis. However, the increased risk of fracture in this group means that caution must be shown. Repeated spinal rotation or flexion should be avoided, and high impact activities should be limited at the early stages of treatment, especially where osteoporosis already exists.
Hormone replacement therapy
The use of low dosage oestrogen in hormone replacement therapy (HRT) to slow or even halt bone loss in postmenopausal women is widely recommended. HRT is used to make up for the decline in oestrogen levels seen during the menopause. Oestrogen plays an important part in calcium absorption by the body. Secreted by the ovaries and corpus luteum, oestrogen (a steroid hormone) stimulates regeneration of the endometrium, increases mucus output from the cervical glands, and promotes epiphyseal closure and growth of the bones and skeletal muscle.
HRT has been linked with an increased risk of endometrial carcinoma, although the risk is reduced when oestrogen is combined with progesterone (Hedlund and Gallagher, 1988).
Transient osteoporosis
Transient osteoporosis most commonly affects the hip, where it is termed idiopathic transient osteoporosis of the hip (ITOH) (Harrington et al., 2000). It is a relatively rare condition which must be differentiated from both stress fracture and avascular necrosis. The condition was first described in women during late pregnancy (Curtiss and Kincaid, 1959). However, the main group to suffer from this condition are middle-aged (40–70 years) men, who represent over 70% of cases (Lakhanpal, Ginsburg and Luthra, 1987). ITOH is a self-limiting condition which demonstrates bone marrow oedema and slight bone cortex thinning. There is no evidence of the focal subchondral bone defect of the femoral head seen in avascular necrosis (Harrington et al., 2000). Joint fluid is sterile (non-infected) and only mild synovial thickening is present (McCarthy, 1998).
Subjective examination reveals progressive unilateral hip pain referred into the groin and anterior thigh. Pain is exacerbated with weight bearing and eased with rest (contrast with malignancy which gives pain at rest). Typically pain is worse 4–8 weeks after onset, and enforced rest may show disuse atrophy of the quadriceps and gluteal muscles. The joint and greater trochanter may be tender to palpation and the FABRE test positive.
Management is by protected weight-bearing (crutches) and non-weight-bearing or partial weight-bearing exercise to pain tolerance (pool/walking/jogging). Non-steroidal anti-inflammatory drugs (NSAIDs) may be of use, and protracted cases have responded to core decompression of the femur (Apel, Vince and Kingston, 1994).
Articular cartilage
Cartilage is essentially a connective tissue consisting of cells embedded in a matrix permeated by fibres. Two major types are recognized, hyaline cartilage and fibrocartilage. Hyaline cartilage is found over bone ends and is described in more detail below. Fibrocartilage may be either white or yellow. White fibrocartilage is found in areas such as the intervertebral discs and glenoid labrum. Yellow fibrocartilage is found in structures such as the ears and larynx.
Definition
Hyaline cartilage is found in synovial joints, white fibrocartilage in the vertebrae and yellow fibrocartilage in the ear and larynx.
Articular (hyaline) cartilage is made up of collagen fibres, a protein–polysaccharide complex, and water. Fibres of collagen are embedded in a ground substance of gel-like material. The water content of cartilage is high, between 70 and 80% of its total weight (Bullough, 1981).
Cartilage has no blood vessels, lymphatics or nerve fibres. Nutrition is supplied directly from the synovial fluid, and by diffusion of blood products from the subchondral bone. The tangled structure of cartilage causes it to behave in some ways like a microscopic sieve, filtering out large molecules such as plasma proteins. Fluid movement through the cartilage is by osmosis and diffusion. Movement of the joint increases the rate of diffusion, but in the mature joint there is no transfer across the bone–cartilage interface (Hettinga, 1990).
Intermittent loading creates a pumping effect, squeezing fluid from the cartilage and allowing fresh fluid to be taken up as the load is released. Prolonged loading will gradually press fluid out of the cartilage, without allowing new fluid to be taken up. As much as a 40% reduction in cartilage depth can occur by compression (Hettinga, 1990).
Keypoint
Because cartilage has no direct blood supply, regular movement is vital to supply it with nutrients from the synovial fluid.
The cartilage is arranged in four layers (Fig. 1.21). The collagen fibres of the calcified zone bind the cartilage to the subchondral bone, resisting shear stresses. Within the mid-zone the fibres are randomly oriented. With joint compression, these fibres stretch and will resist the tension forces created within them (Fig. 1.22). This property of elastic deformation (spring) occurs instantly. However, the fluid within the matrix of the cartilage will be compressed. As it does so, proteoglycans within the cartilage will tend to retain water and control its movement through the cartilage matrix. The cartilage will slowly flow away from the compression force, demonstrating the property of creep. When the load is removed, the fluid lost at the time of compression is reabsorbed. These two properties of instant spring and slower creep make cartilage a viscoelastic material.
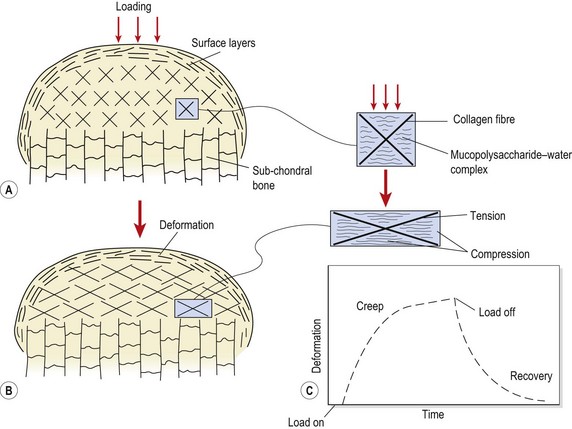
Figure 1.22 Effects of joint compression on articular cartilage. (A) Compressive loading of the cartilage results in (B) tension stress to the collagenous elements and compression stress to the mucopolysaccharide–water complex. (C) The total response is viscoelastic. The viscous creep with sustained loading is largely the result of a time-dependent squeezing out of fluid.
From Hertling, D. and Kessler, R.M. (1990) Management of Common Musculoskeletal Disorders. JB Lippincott, Philadelphia. With permission.
Cartilage assists joint lubrication by boundary lubrication, and weeping lubrication.
Injury
Articular cartilage is to a great extent protected from injury by the elasticity of other joint structures, and the neuromuscular reflexes. Reflex muscular contraction and soft tissue ‘give’ will largely dampen shock before it reaches the cartilage. However, unexpected loading (such as trauma) which occurs too rapidly to invoke reflex protection will cause injury, but this is more frequently bony fracture rather than damage to the cartilage itself.
If cartilage injury does occur, it is most likely due to slip or shear. Three injury stages have been described (Hettinga, 1990): first, splitting of the cartilage layer at the tidemark between the calcified and uncalcified tissue; second, cartilage depression into the subchondral bone; and third, fissuring of the cartilage and underlying bone.
Partial thickness injuries, limited to the articular cartilage alone, heal poorly. The healing which does take place is by proliferation and invasion from soft tissue, and the further away from this tissue the injured area is, the poorer the healing. Full thickness defects, extending through the subchondral bone, heal by superficial tissue bridging the break. Blood vessels grow into the uncalcified cartilage. Osteogenic cells and granulation tissue from the base of the break invade the area, resulting in the formation of fibrous tissue, bony trabeculae and fibrocartilage. There is obvious demarcation of the region into zones, a chondrin-free ring surrounding the uninjured tissue. The healed defect appears as a slightly discoloured roughened area of fibrous tissue.
Cartilage repair
Several surgical techniques may be used to aid repair of injury to articular cartilage (Table 1.7). The joint surface may be debrided (scraped) and the joint itself washed out (lavaged) to remove fragments of cartilage or subchondral bone. Damaging enzymes produced through cartilage degeneration are also removed. New cartilage cells may be introduced into the damaged area to stimulate healing. Osteochondral grafts of this type may be taken from the patient (autografts) or from cadaveric donors (allografts). A number of tissues have been used for autografts including rib perichondrial cells, periosteum and chondrocytes themselves. The technique involves two surgical procedures. First the cells must be removed, then allowed to grow in a laboratory, and finally the cells must be replanted into the damaged cartilage. Allograft techniques can result in high degrees of rejection as a complication (Wroble, 2000) but only involve a single surgical procedure.
Table 1.7 Management of articular cartilage degeneration
| Method | Technique |
|---|---|
| Debridement | Cartilage layer is removed down to subchondral bone. Results may be poor |
| Arthroscopic washing | Washing out loose fragments of cartilage/subchondral bone and joint fluid containing degradative enzymes |
| Drilling, abrasion and microfracture | Work by penetrating the subchondral bone and stimulating cell regrowth. Fibrocartilage rather than renewed hyaline cartilage is produced |
| Autologous implantation | Transplanting cells of various types into chondral defects. Cells used include rib perichondrial cells, periosteum and chondrocytes |
| Allograft | Implanting cadaveric cartilage. Rejection is possible |
Definition
Autografts involve implanting cells or tissue taken from the patient. Allografts are taken from a cadaver (dead person).
Drilling, abrasion and microfracture all work by penetrating the subchondral bone and stimulating cell regrowth. The aim is to allow a conduit for clot formation containing mesenchymal stem cells capable of forming repair tissue (Steadman, Rodkey and Singleton, 1997). Unfortunately fibrocartilage rather than renewed hyaline cartilage is produced by these procedures. Further details of cartilage repair and rehabilitation are given in Chapter 9.
Arthritis
The term arthritis tends to be used to describe any chronic inflammatory reaction affecting a joint. However, the term simply means ‘joint inflammation’, and as such must be qualified by a description of the cause of inflammation. Acute joint injury which causes intracapsular swelling may be termed ‘traumatic arthritis’. True osteoarthritis involves cartilage degeneration, initially with little inflammation, so the term osteoarthrosis would seem more appropriate. This condition must be differentiated from inflammatory states affecting multiple joints such as rheumatoid arthritis. The following description is concerned with osteoarthritis (OA) and its connection to sport and exercise.
The initial changes in OA are usually painless and show no gross joint swelling. The tissue affected first appears to be the joint cartilage. This shows an increased water content resulting from proteolysis of the cartilage macromolecules (Jones, 2007).
Mild fraying or flaking of superficial collagen fibres within the hyaline cartilage occurs. This happens first at the periphery of the joint in the non-weight-bearing region. Later, damage (fibrillation) is to the deeper cartilage layers in the weight-bearing areas of the joint, extending down to one-third of the cartilage thickness. Small cavities form (blistering) between the cartilage fibres which gradually extend to become vertical clefts. If cartilage fragments break off, they may float free in the joint fluid as loose bodies, giving rise to joint locking and sudden twinges of pain. The presence of a loose body and the by-products of cartilage destruction causes the synovium to inflame, and it is only now that many patients become aware that a problem exists.
Keypoint
Patients usually only become aware that they have arthritis when a joint becomes inflamed and painful. Treatment aimed at reducing pain/inflammation and restoring function will often alleviate the problem, but the structure of the bone remains the same.
Turnover of proteoglycan and collagen within the cartilage ground substance is increased, and the proteoglycan molecules near the fibrillated cartilage are smaller than normal. Mechanically, this altered cartilage is weaker to both compression and tension stresses, but it is still resistant to gliding, and its coefficient of friction remains low (Threlkeld and Currier, 1988). As the cartilage thins the joint space is reduced.
The subchondral bone beneath the fibrillated cartilage becomes shiny and smooth (eburnated). Below the eburnated region the area becomes osteoporotic and local avascular necrosis causes cyst formation where there is complete bone loss. Osteophytes covered with fibrocartilage form at the periphery of the joint, and may protrude into the joint space or more frequently into surrounding soft tissue.
Definition
Two classic signs of osteoarthritis are eburnation, where the joint cartilage becomes shiny and smooth, and osteophytes, which are bony spurs forming at the edge of the joint.
The synovial membrane becomes thickened and its vascularity increases in line with an inflammatory response (see above). The joint capsule demonstrates small tears filled with fibrous tissue, causing thickening. Contracture usually alters both physiological and accessory movements. For example, when OA affects the knee joint, flexion is often limited and mediolateral stability reduced. It is important to appreciate that many tissues are affected by arthritis, and bone itself is often not the most important tissue clinically. The synovial proliferation alters the consistency of the synovial fluid. There is an increased production of synovial fluid which has a lower viscosity. Injections of hyaluronic acid are designed to slow this process. Increased growth of blood vessels (angiogenesis or neovascularization) occurs in OA in bone, synovial membrane and joint capsule. In addition new vessels may also grow across the subchondral barrier dragging nerve fibres with them (Jones, 2007). Table 1.8 summarizes the common tissue changes seen in arthritis, and further details of arthritis with repect to the knee are given in Chapter 10.
Table 1.8 Tissue changes in knee arthritis
| Tissue | Effect |
|---|---|
| Cartilage | |
| Bone | |
| Blood vessels | |
| Fibrocartilage | |
| Synovium | |
| Tendons & ligaments | |
| Muscles & nerves |
Adapted From Jones, 2007.
The joint capsule
The joint capsule is in two parts. The outer part (stratum fibrosum) is fibrous and thickened in areas to form ligaments. The inner layer (stratum synoviale) is loose and highly vascular and blends with the synovial membrane. The capsule consists of parallel fascicles of collagen and some fibrocytes. Blood vessels enter the subchondral bone at the line of capsular attachment and small vessels are found between the individual cartilage fascicles. The nerve supply is very rich, with large fibres giving proprioceptive feedback and small fibres terminating in pain endings.
The capsular response to trauma is an increase in vascularity and eventually the development of fibrous tissue. Cross-linking of collagen fibres occurs, causing a palpable thickening of the capsule. Capsular shrinkage combined with fibrous adhesions will cause loss of movement and occurs particularly after immobilization.
Keypoint
The joint capsule will become thickened and less elastic following trauma and prolonged rest. Ultimately capsular shrinkage will significantly limit movement.
Accumulation of joint fluid through swelling will stretch the capsule and capsular ligaments. The nerve endings situated between the collagen fascicles of the capsule will be stretched, giving rise to mechanical pain. Should the fluid accumulation exceed the elastic limit of the joint capsule, rupture may result.
Joint effusion will stretch portions of the capsule which are normally lax, to facilitate movement. The patient will tend to rest the joint in a position where the joint cavity is of maximum volume; in addition, passive movements will be limited in characteristic ‘capsular patterns’ (Table 1.9).
Table 1.9 The capsular patterns
Ligaments
When a ligament is put under stress, it responds by getting progressively stiffer before later deforming in a regular manner (Amis, 1985). There are two reasons for this. First, the collagen fibres within the ligament are not in line, and so the initial tensile stress is used up by pulling the fibres straight. Secondly, the fibres are not attached to a single point but to an area of bone, and so are of slightly different lengths. When the fibres are stretched, there will be a progressive tightening of the ligament with some fibres becoming taut sooner than others.
Ligaments demonstrate viscoelastic properties. Rapid stretch has been shown to increase stiffness by as much as 20% (Woo, Gomez and Akeson, 1981), and sustained stretch to cause ligament tension to reduce significantly after 2 minutes (Viidik, 1966). The mode of failure also changes with speed, avulsion occurring at slower speeds and ligamentous rupture at higher speeds (Noyes, De Luca and Torvik, 1974).
Clinically, three grades of ligament injuries are recognized (Fowler, 1984). Grade I sprains involve minimal tissue damage with some local tenderness. Swelling is only slight, and function is almost normal. With the grade II sprain, more ligament fibres are injured or the ligament may become partially detached from its bony attachment. Local pain is more intense and movement more limited. Grade III injuries constitute a complete rupture. There is a rapid onset of effusion with considerable pain. The joint is unstable and loss of function is complete. Noyes et al. (1984) argued that this traditional classification, although useful, could be misleading. The grade II injury could refer equally to an injury where only a few ligament fibres are torn, or to a lesion where virtually the total ligament was affected. The grade III injury, they claimed, represented not total rupture, but total loss of function of the ligament, actual continuity of the ligament often being maintained (Table 1.10).
| Grade | Signs and symptoms |
|---|---|
| (I) Mild | Minimal loss of structural integrity |
| No abnormal motion | |
| Little or no swelling | |
| Localized tenderness | |
| Minimal or no bruising | |
| (II) Moderate | Significant structural weakening |
| Some abnormal motion | |
| Marked bruising and swelling | |
| Often associated with haemarthrosis and joint effusion | |
| (III) Severe | Loss of structural integrity |
| Marked abnormal motion | |
| Significant bruising which may track away from area | |
| Definite haemarthrosis if capsule remains intact |
Adapted from Reid (1992) with permission.
With grades I and II, pain is increased by placing the ligament on stretch. With a grade II injury, some instability may be present, but with a complete rupture instability is always present. However, clinically this may be difficult to assess in instances of severe pain and muscle spasm.
Ligamentous viscosity changes with age. The collagen fibres within the ligament enlarge, reducing the water content in the ground substance. Noyes and Grood (1976) showed marked reductions in tensile strength of ligament in the 48–83 year age group when compared with the 16–26 year age group. Noyes et al. (1984) showed threefold decreases in maximum stress, elastic modulus and strain energy between the ligaments from donors aged 50 years compared to those aged 20 years. The older ligaments failed by bony avulsion rather than ligamentous failure as occurred in the younger tissues. They claimed that ageing produced changes in the ligament/bone systems similar to those found through disuse.
Immobility also has marked effects on ligaments. Laros, Tipton and Cooper (1971) showed strength reductions of 39% after 9 weeks’ immobilization, full strength not being regained for 30 weeks. Exercise has been shown to have a beneficial effect on ligament tensile strength (Adams, 1966; Heikkinen and Vuori, 1972; Cabaud et al., 1980). Exercise seems to act as a ‘mechanical stimulant’, causing increased collagen turnover within the ligament (Weisman, Pope and Johnson, 1980).
Keypoint
Immobilization weakens ligaments, while exercise acts as a mechanical stimulant to cause increased collagen turnover and strengthen the ligament.
This has important implications for rehabilitation following ligament injury. Wherever possible, complete immobilization should be avoided. While an injured ligament should be protected against excessive external forces which could cause further damage, gentle exercise within the pain-free range should still be encouraged.
The effect of corticosteroids on ligament failure is important, since steroid usage in sport is still popular. Decreases in maximum load of 21% and 39% have been shown 6 and 15 weeks after large dosage cortisone injection (methylprednisolone acetate), with a load reduction remaining for 1 year. In addition, fibrocyte death was seen within the ligament, with delay in reappearance of new fibrocytes for 15 weeks (Noyes et al., 1984).
Muscle
The term ‘strain’ is generally used to imply a minor injury to any soft tissue structure. However, in this book ‘strain’ will be confined to injury of muscle while the term ‘sprain’ will be used for injured joint structures. Strains may be subdivided into acute and chronic types. Acute strains are usually the result of a single violent stretch to a contracting muscle (Fowler, 1984; Keene, 1990), while chronic strains develop over a period of time as a result of repetitive loading.
Definition
Injuries to muscles are termed strains while injuries to ligaments and other joint structures are called sprains.
Muscle injuries may be classified into a variety of types (Table 1.11). At one end of the spectrum is muscle pain, including ischaemia and delayed onset muscle soreness (DOMS) which occurs some time after intense muscle training. These two conditions are covered in Chapter 5. Muscles may be injured through direct trauma (extrinsic injury) or through their own power (intrinsic injury). Extrinsic injury includes contusions (bruising) and tissue damage through laceration such as may occur through an open impaling injury or compound fracture, where the fractured bone pierces muscle and skin.
| Grade | Signs and symptoms |
|---|---|
| Exercise induced muscle soreness | Muscle pain during training (ischaemia) or aching occurring 24–48 hours after intense unaccustomed activity (delayed onset muscle soreness—DOMS) |
| Contusion | Pain and marked bruising tracking distally. Failure to increase range of motion suggests blood clot is trapped (intramuscular haematoma) |
| (I) Mild strain | Small number of muscle fibres torn, giving minimal bleeding. Pain and spasm well localized. Muscle function normal, but endurance may be reduced |
| (II) Moderate strain | Larger number of muscle fibres injured, but muscle fascia remains intact. Bleeding contained causing an increase in intramuscular pressure. Palpation reveals definite mass and considerable pain. Contractility and extensibility of muscle reduced. |
| (III) Severe strain | Greater area of muscle affected, with muscle fascia partially torn. Bleeding more profuse, and blood may track distally. Function severely impaired |
| (IV) Rupture/avulsion |
Sources Fowler, J.A. (1984) Soft tissue injuries and sports injuries. In Cash’s Textbook of Orthopaedics and Rheumatology for Physiotherapists (ed. P.A. Downie). Faber and Faber, London. With permission. Keene (1990) and Reid (1992).
Contusions are common in contact sports such as rugby, where a quadriceps contusion or ‘dead leg’ is a common site. Bruising may be greater than for grade (I) or grade (II) injuries, and the amount of blood released into the tissues makes possible scarring or calcification a concern. Contusions, as with grade (III) injuries, may be classified as intermuscular, where the blood is released from the muscle and spreads downwards (tracking). Tracking disperses the blood, facilitating healing and minimizing the risk of scarring. The tense muscle resolves fairly quickly, allowing an earlier recovery of range of motion. Blood appears in distal parts of the limb. With an intramuscular haematoma, blood is trapped within the muscle septum, giving a palpable mass. The inflammatory response is generally greater (Reid, 1992) and the risk of scarring and myositis ossificans must be considered. Range of motion remains restricted, and this is one of the most important early clinical signs.
Keypoint
Following a muscle contusion, if range of motion remains restricted, intramuscular haematoma must be suspected.
Three grades are used to describe actual muscle strains, while a complete rupture is sometimes described as a grade (IV) sprain (Fowler, 1984).
Figure 1.23 An intermuscular haematoma affecting the rectus femoris.
From Fowler, J.A. (1984) Soft tissue injuries and sports injuries. In Cash’s Textbook of Orthopaedics and Rheumatology for Physiotherapists (ed. P.A. Downie). Faber and Faber, London. With permission.
Table 1.12 Common sites for avulsion injuries
| Site | Muscle |
|---|---|
| Anterior superior iliac spine (ASIS) | Sartorius |
| Anterior inferior iliac spine (AIIS) | Rectus femoris |
| Iliac crest | External oblique |
| Ischial tuberosity | Hamstrings |
| Olecranon process | Triceps |
| Patella | Quadriceps tendon |
| Lesser trochanter | Iliopsoas |
The site of the injury is especially important with regard to the treatment regimen chosen. Muscle−tendon injuries can be to the muscle belly itself, the musculotendinous (MT) junction or the teno-osseous (TO) junction. Garrett (1990) showed that both passive stretching and stretching a precontracted isolated rabbit muscle consistently caused failure at the MT junction, a small (0.1–1 mm) amount of muscle tissue being left attached to the tendon.
Garrett (1990) also showed that precontracted muscles absorbed 100% more energy than passively stretched muscle fibre, and used these results to emphasize the importance of muscles as energy absorbers. He argued that conditions which interfere with a muscle’s contractile ability, such as fatigue or muscle weakness, might diminish the energy absorbing ability of the muscle and possibly predispose to injury.
Cyriax (1982) claimed that muscle belly injuries would respond to therapeutic exercise. Muscle contraction causes broadening of the fibres and, he claimed, would reduce intramuscular scarring. Injury at the MT or TO junctions would not respond to exercise as no muscle broadening occurs at these points. Cyriax argued that passive therapy, such as transverse frictional massage or injection, was more appropriate to these sites.
Myositis ossificans traumatica
Myositis ossificans traumatica (MOT) is soft tissue ossification of muscle resulting in the formation of non-neoplasmic bone (Booth and Westers, 1989). It usually occurs in the proximal limb muscles, and is seen most commonly following contusion of the quadriceps femoris musculature (Jackson, 1975), although other regions include the elbow flexors and hip abductors.
Several aetiological theories have been suggested (Booth and Westers, 1989). Calcification of a muscle haematoma can occur following fibrosis, or intramuscular bone formation may result from periosteal detachment. Rupture of the periosteum may lead to the proliferation of osteoblasts and their escape into the surrounding muscle. Intramuscular connective tissue may undergo metaplasia into bone, and Urist et al. (1978) demonstrated that certain skeletal muscle cells, known as inducible osteogenic precursor cells (IOPC), have the capability of differentiating into osteoblasts. Certain individuals have been shown to have a predisposition to the condition (Rothwell, 1982).
Clinically, the patient presents with severe post-traumatic pain and limitation of movement. There is local swelling and erythema, with the area being warm and tender to palpation. Tenderness usually becomes more pronounced with time, and tissue signs do not respond to conservative management. The factor which should alert the practitioner to the possibility of MOT is a reduction in movement range over time. Normally, an increasing range would be expected as a condition resolves. Radiological evidence of ossification is usually seen within 2 months following injury, and other investigations, including computed tomography (Vas et al., 1981) and ultrasound scanning (Kramer et al., 1979) have been used.
Keypoint
Myositis is most common after quadriceps haematoma (dead leg). The classic sign is reduction in movement range over time.
Management of MOT is aimed at lessening any disability rather than affecting the bone mass. Initially, the soft tissue response to trauma, especially bleeding, is limited by the use of such regimens as rest, ice, compression and elevation (RICE). Rehabilitation is slower than with a normal muscle haematoma, isometric exercises being used 7–14 days following injury, but active exercises (within the pain-free range) are not used until 2–4 weeks after injury (Booth and Westers, 1989).
Tendons
Tendinopathy is pain and/or pathology of tendons. A variety of conditions are included within this term, the most common in sport being tenosynovitis, tendinitis and tenovaginitis. Tendinitis is an inflammation and scarring of the tendon which can occur in varying degrees. Scarring may interfere with tendon function and/or cause persistent local inflammation. Tenosynovitis is a lesion to the gliding surfaces of the outside of the tendon and the inside of its sheath, but not the sheath itself (Fowler, 1984). The lesion is usually as a result of overuse or compression (Williams and Sperryn, 1976). Pathological features are similar to those seen in rheumatoid arthritis (Williams and Sperryn, 1976), including an increase in the number of lining cells of the sheath, together with proliferation of local blood vessels, oedema and cellular changes. As the roughened surfaces of the sheath move against each other pain and crepitus occur. The same condition occurring to tendons which do not have a true sheath is termed peritendonitis. Here, the paratenon thickens, and shows fibrinoid degeneration and dense fibrous adhesions (Snook, 1972). Local oedema is evident, sometimes with palpable crepitus, and pain may disappear with activity (Williams and Sperryn, 1976).
Tenovaginitis occurs when the tendon sheath is chronically inflamed and thickens. It is the fibrous wall of the tendon sheath rather than the synovial lining which is affected. Common sites include the flexor sheaths of the fingers or thumb. When the sheaths of extensor pollicis brevis and abductor pollicis longus are affected, de Quervain’s syndrome is present.
Keypoint
Tendinitis is an inflammation and scarring of the tendon; tenosynovitis is a lesion of the gliding surfaces of the tendon and its sheath; tenovaginitis is a chronic inflammation and thickening of the tendon sheath.
Specific tendon pathology is covered in more detail in Chapters 10 (patellar tendon), 11 (Achilles tendon) and 18 (rotator cuff tendons).
Nerves
The nervous system can be affected by injury through both internal (intraneural) and external (extraneural) means. Intraneural pathology has an effect on the elasticity of the neural system (Slater, Butleter and Shacklock, 1994). When tension is taken up at one point in the nervous system, this is transferred further along, a little like pulling on a rope. Altered mechanics may be accompanied by changes in physiology. Altered microcirculation and axonal transport may occur through compression (Lundborg, 1988), with nerve swelling occurring both proximal and distal to a compression force (MacKinnon, Dellon and Hudson, 1984).
Definition
Axonal transport is active movement of chemical substances through the cell fluid (axoplasm) of a nerve.
The nervous system consumes 20% of the oxygen used by the body. This is supplied to the nerves through the vasa nervorum, a network of extraneural and intraneural vessels.
Nerve injury may occur along a number of sites (Fig. 1.24) where nerve movement is restricted. Typically, injury is either through trauma or through overuse conditions caused by friction, compression or stretch which cause adverse neural tension (ANT). If sustained, compression may lead to deterioration of the neurone.
Definition
Adverse neural tension (ANT) is an abnormal physiological or mechanical response from neural structures caused by alteration in their movement or stretch capability (Butler, 1991).
The two major issues affecting nerves in sport injury management are vascular factors and mechanical factors. An alteration in pressure to the blood vessels supplying the nerves of as little as 20 mmHg will affect nerves, and at pressures of 80 mmHg the nerve blood flow is cut off completely (Rydevik, Lundborg and Bagge, 1981). With increasing pressure, the nerve will initially become hypoxic, leading to local venous stasis. If maintained, oedema will form due to alteration in osmotic pressure. With time, the blood capillaries will deteriorate and fibrosis will set in. Eventually the condition becomes chronic, with scar tissue formation, and the nerve becomes a tough fibrous cord (Butler, 1991).
Mild pressure or friction can lead to oedema of the epineurium, and tearing of the epineurium itself is common in ankle injury (Nitz, Dobner and Kersey, 1985). In such injuries the intraneural and extraneural blood vessels may also rupture. Mechanical damage can again lead to fibrosis with increased stiffness and reduced elasticity occurring in the damaged nerve (Beel, Groswald and Luttges, 1984). The mechanical change in the nerve will alter tension in the whole nervous system as the structures are continuous. Further, the combination of mechanical change and vascular alteration will alter the firing pattern of the nerve and the transport system within the nerve itself (axonal transport). This type of alteration can cause changes further along the nerve creating a double crush syndrome. In these cases a condition such as carpal tunnel syndrome can give rise to altered neural mechanics and eventual pathology in the neck (Upton and McComas, 1973), or carpal tunnel syndrome on the other side of the body (Hurst, Weissberg and Carroll, 1985).
Skin
Skin wounds may be divided into two categories: open and closed. Closed wounds occur when there is no penetration of the epidermis, and open wounds are those where the epidermis has been pierced. Closed wounds encompass contusions, abrasions and friction burns, such as those caused by gravel. Open wounds in sport include cuts such as lacerations and puncture wounds.
A contusion or bruise involves a direct blow to the skin surface. Bleeding occurs from torn blood vessels into the skin and subcutaneous tissues, forming a bruise or ecchymosis. The size of the bruise is dependent on the vascularity of the affected tissue as well as the size of the blood vessels damaged and the laxity of the surrounding tissues. Normally, bruising requires conservative management, but conditions such as retrobulbar haemorrhage (behind the eyeball) and scrotal haematoma may require surgical evacuation (Ray and Soutar, 1993). Following contusion, released blood will clot and degrade, producing superficial colour changes.
An abrasion or graze occurs through a glancing injury or repeated microtrauma to the skin surface. The skin surface breaks through, but the damage is not full thickness. The condition may be complicated by ingrained tattooing where embedded particles remain beneath the surface. The material may become covered by epithelium, giving a visible and hypertrophic scar. An abrasion of this type should be treated as early as possible after the injury by vigorous scrubbing with a stiff nail brush and an antiseptic wash to remove the embedded dirt.
Clinical note
An abrasion from a fall onto a grit surface runs the risk of ingrained tattooing. The wound must be thoroughly scrubbed with an antiseptic wash to remove all dirt, before being dressed.
Extensive areas of skin may be affected during an abrasion, and intense pain usually occurs as raw nerve endings are exposed. Bleeding may be widespread, but healing is rapid unless infection ensues. Extensive debriding injuries involving large areas of skin loss (for example from a motor-cycle accident) may require grafting.
A laceration is a full thickness skin injury which exposes the subcutaneous tissue. A force sufficient to break the skin in this way will also give considerable associated deep tissue damage. In addition the edge of the skin break will be uneven and surrounded by a contusion. Clean lacerations may require stitching but heal well; jagged injuries involving skin tearing may leave more scarring.
Pressure or friction may cause an abrasion where the skin surface is removed, or a blister where the epidermal surface skin layer is detached from the underlying tissue. The gap between the two layers is filled with lymph, exposing nerve endings to fluid pressure, so causing pain. When pressure or friction is applied progressively, the epidermal skin layer may adapt to form a thickened callus (Williams, 1979).
Healing of an incised wound such as a cut, where the wound edges are in apposition, occurs by first intention. If the wound edges are separated and more major skin damage has occurred (or if infection has intervened), then healing is by second intention (Evans, 1990a).
With healing by first intention, slight haemorrhage occurs and the cut is filled with a blood clot on the first day. By the second and third day the clot has become organized and epithelial cells from the two sides of the cut have joined. This single layer of covering multiplies to form stratified squamous epithelium—normal epidermis. Fibroblasts lay down collagen in the granulation tissue and a band of scar tissue is formed by 2 weeks after the injury.
Healing by second intention takes longer as the spread of epithelial cells over the larger area takes time. The large amount of granulation tissue results in more extensive scarring and likely contracture.
Treatment note 1.5 Principles of physical examination
Physical examination is conducted after a full subjective examination (see Treatment note 1.1), and aims to determine the structure(s) which are responsible for producing the patient’s symptoms (Petty and Moore, 2001). The examination consists of a number of stages within the whole framework of clinical diagnosis (Table 1.13) and some or all of the elements will be used on each patient.
Table 1.13 Clinical examination
| Element | Detail |
|---|---|
| Observation | Face |
| Gait | |
| Posture | |
| History (subjective examination) | Age, occupation, activities |
| Site and spread | |
| Onset and duration | |
| Symptoms and behaviour | |
| Past medial history (PMH) | |
| Other joint involvement | |
| Drug history (medication) (DH) | |
| Inspection | Deformity (bony?) |
| Colour changes | |
| Muscle wasting | |
| Swelling | |
| Palpation | Temperature |
| Swelling | |
| Synovial thickening | |
| Active movements | Range/movement pattern |
| Pain/symptoms | |
| Power | |
| Willingness | |
| Resisted movements | Pain/symptoms |
| Power | |
| Passive movements | Pain/symptoms |
| Range and quality | |
| End-feel |
After Kesson and Atkins (1998).
Observation and inspection
Palpation
Movements
| Type | Example |
|---|---|
| Normal | |
| Hard | Bone to bone approximation—elbow extension |
| Soft | Soft tissue approximation—elbow flexion |
| Elastic | Tissue stretch—lateral rotation of the shoulder joint |
| Abnormal | |
| Hard | Bony degeneration—osteoarthritis (OA) |
| Springy | Mechanical joint displacement—loose body, meniscal tear |
| Spasm | Involuntary muscle spasm—irritable joint |
| Empty | Patient stops movement—anticipation of pain/instability |
From Cyriax (1982), Kesson and Atkins (1998) with permission.
Adams A. Effect of exercise upon ligament strength. Research Quarterly. 1966;37:163-176.
American College of Sports Medicine (ACSM). Position stand on exercise and physical activity for older adults. Medicine and Science for Sports and Exercise. 1998;30:992-1008.
Amis A.A. Biomechanics of ligaments. In: Jenkins D.H.R., editor. Ligament Injuries and their Treatment. London: Chapman and Hall, 1985.
Apel D.M., Vince K.G., Kingston S. Transient osteoporosis of the hip: a role for core decompression? Orthopedics. 1994;17(7):629-632.
Apley A.G., Solomon L. Concise System of Orthopaedics and Fractures. Oxford: Butterworth-Heinemann; 1988.
Apley A.G., Solomon L. Apley’s system of orthopaedics and fractures, seventh ed. Oxford: Butterworth Heinemann; 1993.
Baldry P.E. Trigger point acupuncture. In: Filshie J., White A., editors. Medical Acupuncture. Edinburgh: Churchill Livingstone, 1998.
Bassett C.A.L. Electrical effects in bone. Scientific American. 1965;213(4):18.
Bassett C.A.L., Pawlick R.J. Effects of electric currents on bone in vivo. Nature. 1964;204:652.
Becker R.O., Murray D.G. The electrical control system regulating fracture healing in amphibians. Clinical Orthopaedics and Related Research. 1970;73:169.
Becker R.O., Spadaro J.A., Marino A.A. Clinical experience with low intensity direct current stimulation of bone growth. Clinical Orthopaedics and Related Research. 1977;124:75-83.
Beel J.A., Groswald D.E., Luttges M.W. Alterations in the mechanical properties of peripheral nerve following crush injury. Journal of Biomechanics. 1984;17:185-193.
Behiri J., Vashishth D. Biomechanics of bone. In: Dvir Z., editor. Clinical Biomechanics. Edinburgh: Churchill Livingstone, 2000.
Bennell K., Khan K., McKay H. The role of physiotherapy in the prevention and treatment of osteoporosis. Manual Therapy. 2000;5(4):198-213.
Blau S.P. The synovial fluid. Orthopaedic Clinics of North America. 1979;10:21-35.
Booth D.W., Westers B.M. The management of athletes with myositis ossificans traumatica. Canadian Journal of Sports Science. 1989;14(1):10-16.
Bullough P.G. Cartilage. In: Owen R., Goodfellow J., Bullough P., editors. Scientific Foundations of Orthopaedics and Traumatology. Oxford: Heinemann Medical, 1981.
Burri C., Helbing G., Spier W. Rehabilitation of knee ligament injuries. In: The Knee. New York: Springer; 1973.
Butler D.S. Mobilisation of the Nervous System. Edinburgh: Churchill Livingstone; 1991.
Cabaud H.E., Chatty A., Gildengorin V., Feltman R.J. Exercise effects on the strength of the anterior cruciate ligament. American Journal of Sports Medicine. 1980;8:79-86.
Carbon R.J. The female athlete. In: Bloomfield J., Fricker P.A., Fitch K.D., editors. Textbook of Science and Medicine in Sport. Melbourne: Blackwell Scientific Publications, 1992.
Chartered Society of Physiotherapy (CSP). Physiotherapy Guidelines for the Management of Osteoporosis. London: Chartered Society of Physiotherapy; 1999.
Crawford-Adams J. Outline of Fractures. London: Churchill Livingstone; 1978.
Curtiss P.H., Kincaid W.E. Transitory demineralization of the hip in pregnancy: a report of three cases. Journal of Bone and Joint Surgery (Am). 1959;41:1327-1333.
Curwin S.L. The aetiology and treatment of tendinitis. In: Harries M, et al, editors. Oxford Textbook of Sports Medicine. Oxford: Oxford Medical Publications, 1994.
Cyriax J. Textbook of Orthopaedic Medicine vol. 1, eighth ed. London: Baillière Tindall, 1982.
De Domenico G. Pain relief with interferential therapy. Australian Journal of Physiotherapy. 1982;28(3):14-18.
Deakin V., Brotherhood J.R. Nutrition and energy sources. In: Bloomfield J., Fricker P.A., Fitch K.D., editors. Textbook of Science and Medicine in Sport. Melbourne: Blackwell Scientific Publications, 1992.
Donaldson C.L., Hulley S.B., Vogel J.M. Effects of prolonged bed rest on bone mineral. Metabolism. 1970;19:1071-1084.
Donley P.B., Denegar C. Pain and mechanisms of pain relief. In: Prentice W.E., editor. Therapeutic Modalities in Sports Medicine. Philadelphia: Times Mirror/Mosby College Publishing, 1990.
Dyson M. Mechanisms involved in therapeutic ultrasound. Physiotherapy. 1987;73(3):116-120.
Ettinger B., Genant H.K., Cann C.E. Postmenopausal bone loss is prevented by low dosage estrogen with calcium. Annals of Internal Medicine. 1987;106:40-45.
Evans P. The healing process at cellular level: a review. Physiotherapy. 1980;66(8):256-259.
Evans D.M.D. Inflammation and healing. In Downie P.A., editor: Cash’s Textbook of General Medical and Surgical Conditions for Physiotherapists, second ed, London: Faber and Faber, 1990.
Evans R.A. Calcium and osteoporosis. Medical Journal of America. 1990;152:431-433.
Evans C.H. Cytokines and the role they play in the healing of ligaments and tendons. Sports Medicine. 1999;28:71-76.
Fowler J.A. Soft tissue injuries and sports injuries. In: Downie P.A., editor. Cash’s Textbook of Orthopaedics and Rheumatology for Physiotherapists. London: Faber and Faber, 1984.
Fox J., Galey F., Wayland H. Action of histamine on the mesenteric microvasculature. Microvascular Research. 1980;19:108-126.
Frank G., Woo S.L., Amiel O., et al. Medial collateral ligament healing: a multidisciplinary assessment in rabbits. American Journal of Sports Medicine. 1983;11:379-389.
Garrett W.E. Muscle strain injuries: clinical and basic aspects. Medicine and Science in Sports and Exercise. 1990;22:436-443.
Gartland J.J. Fundamentals of Orthopaedics, fourth ed. Philadelphia: W.B. Saunders; 1987.
Gould J.A., editor. Orthopaedic and Sports Physical Therapy, second ed, St Louis: C.V. Mosby, 1990.
Gunn C.C. Treatment of Chronic Pain, second ed. Edinburgh: Churchill Livingstone; 1996.
Harrington S., Smith J., Thompson J., Laskowski E. Idiopathic transient osteoporosis. Physician and Sportsmedicine. 2000;28(4):1-9.
Hedlund L.R., Gallagher J.C. Estrogen therapy for postmenopausal osteoporosis: current considerations of the menopause. Annals of Clinical and Laboratory Science. 1988;15:219-228.
Heikkinen E., Vuori I. Effect of physical activity on the metabolism of collagen in aged mice. Acta Physiologica Scandinavica. 1972;84:543-549.
Heinonen A., Kannus P., Sievanen H. Randomised controlled trial of effect of high impact exercise on selected risk factors for osteoporotic fractures. Lancet. 1996;348:1343-1347.
Hert J., Zalud J. Reaction of bone to mechanical stimuli. (VI). Bioelectrical theory of functional adaptation of bone. Acta Chirurgicae Orthopaedicae et Traumatologicae Cechoslovaca. 1971;38:280.
Hettinga D.L. Inflammatory response of synovial joint structures. In: Gould J.A., editor. Orthopaedic and Sports Physical Therapy. second ed. St Louis: C.V. Mosby; 1990:87-117.
Hom D.B. Growth factors in wound healing. Otolaryngologic Clinics of North America. 1995;28:933-953.
Huda W., Morin R.L. Patient doses in bone mineral dosimetry. British Journal of Radiology. 1996;69:422-425.
Huddleston A.L., Rockwell D., Kuland D.N. Bone mass in lifetime tennis athletes. Journal of the American Medical Association. 1980;244:1107-1109.
Hunter G. Specific soft tissue mobilization in the management of soft tissue dysfunction. Manual Therapy. 1998;3(1):2-11.
Hurst L.C., Weissberg D., Carroll R.E. The relationship of double crush to carpal tunnel syndrome. Journal of Hand Surgery. 1985;10B:202-204.
Jackson D.W. Managing myositis ossificans traumatica. Physician and Sports Medicine. 1975;October:56-61.
Jones A. The osteoarthritic knee. In Touch Journal. 2007;119:16-21.
Kannus P. Tendon pathology. Sports Exercise and Injury. 1997;3:62-75.
Keene J.S. Ligament and muscle tendon unit injuries. In Gould J.A., editor: Orthopaedic and Sports Physical Therapy, second ed, St Louis: C.V. Mosby, 1990.
Kellett J. Acute soft tissue injuries: a review of the literature. Medicine and Science in Sports and Exercise. 1986;18(5):489-500.
Kesson M., Atkins E. Orthopaedic Medicine. Oxford: Butterworth-Heinemann; 1998.
Kramer F.L., Kurtz A.B., Rubin C., Goldberg B.B. Ultrasound appearance of myositis ossificans. Skeletal Radiology. 1979;4:19-20.
Krolner B., Toft B., Nielsen S. Physical exercise as a prophylaxis against involutional bone loss: a controlled trial. Clinical Science. 1983;64:541-546.
Kurtz C.A., Loebig T.G., Anderson D.D. Insulin like growth factor 1 accelerates functional recovery from achilles tendon injury in a rat model. American Journal of Sports Medicine. 1999;27:363-369.
Lachman S. Soft Tissue Injuries in Sport. Oxford: Blackwell; 1988.
Lakhanpal S., Ginsburg W.W., Luthra H.S. Transient regional osteoporosis: a study of 56 cases and review of the literature. Annals of Internal Medicine. 1987;106(3):444-450.
Laros G.S., Tipton C.M., Cooper R.R. Influence of physical activity on ligament insertions in the knees of dogs. Journal of Bone and Joint Surgery. 1971;53A:275-286.
Lewis M.K., Blake G.M., Fogelman I. Patient dose in dual X-ray absorptiometry. Osteoporosis International. 1994;4:11-15.
Low J., Reed A. Electrotherapy Explained: Principles and Practice. Oxford: Butterworth-Heinemann; 1990.
Lundborg G. Nerve Injury and Repair. Edinburgh: Churchill Livingstone; 1988.
Lynch M.K., Kessler R.M. Pain. In Hertling D., Kessler R.M., editors: Management of Common Musculoskeletal Disorders, second ed, Philadelphia: J.B. Lippincott, 1990.
McArdle W.D., Katch F.I., Katch V.L. Exercise Physiology: Energy, Nutrition, and Human Performance, second ed. Philadelphia: Lea and Febiger; 1986.
McCarthy E.F. The pathology of transient regional osteoporosis. Iowa Orthopedic Journal. 1998;18:35-42.
MacKinnon J.L. Osteoporosis: a review. Physical Therapy. 1988;68(10):1533-1540.
MacKinnon S.E., Dellon A.L., Hudson A.R. Chronic nerve compression: an experimental model in the rat. Annals of Plastic Surgery. 1984;13:112-120.
Magee D.J. Orthopedic Physical Assessment, fourth ed. Philadelphia: Saunders; 2002.
Magee D.J. Orthopedic Physical Assessment, fifth ed. Missouri: Saunders/Elsevier, St. Louis; 2008.
Maitland G.D. Peripheral Manipulation, third ed. Oxford: Butterworth-Heinemann; 1991.
Martin A.D., Houston C.S. Osteoporosis, calcium and physical activity. Canadian Medical Association Journal. 1987;136:587-593.
Mazess R.B., Whedon G.D. Immobilization and bone. Calcified Tissue International. 1983;35:265-267.
Melzack R., Wall P.D. Pain mechanisms: a new theory. Science. 1965;150:971.
Nilas L., Christiansen C., Rodbro P. Calcium supplementation and post-menopausal bone loss. British Medical Journal. 1984;289:1103-1106.
Nilsson B.E., Westlin N.E. Bone density in athletes. Clinical Orthopaedics and Related Research. 1977;77:179-182.
Nitz A.J., Dobner J.J., Kersey D. Nerve injury and grade II and III ankle sprains. American Journal of Sports Medicine. 1985;13:177-182.
Nixon A.J., Fortier L.A., Williams J. Enhanced repair of extensive articular defects by insulin like growth factor 1 laden fibrin composites. Journal of Orthopaedic Research. 1999;17:475-487.
Norris C.M. Acupuncture: Treatment of Musculoskeletal Conditions. Oxford: Butterworth-Heinemann; 2001.
Noyes F.R., De Luca J.L., Torvik P.J. Biomechanics of anterior cruciate ligament failure: an analysis of strain-rate sensitivity and mechanisms in primates. Journal of Bone and Joint Surgery. 1974;56A:236-253.
Noyes F.R., Grood M.J. The strength of the anterior cruciate ligament in humans and rhesus monkeys: age-related and species-related changes. Journal of Bone and Joint Surgery. 1976;58A:1074-1082.
Noyes F.R., Kelly C.S., Grood E.S., Butler D.L. Advances in the understanding of knee ligament injury, repair and rehabilitation. Medicine and Science in Sports and Exercise. 1984;16(5):427-443.
Oakes B.W. The classification of injuries and mechanisms of injury, repair and healing. In: Bloomfield J., Fricker P.A., Fitch K.D., editors. Textbook of Science and Medicine in Sport. Melbourne: Blackwell Scientific Publications, 1992.
Paget S., Bullough P.G. Synovium and synovial fluid. In: Owen R., Goodfellow J., Bullough P., editors. Scientific Foundations of Orthopaedics and Traumatology. Oxford: Heinemann Medical, 1981.
Palastanga N., Field D., Soames R. Anatomy and Human Movement. Oxford: Heinemann Medical; 1989.
Pappas A.M. Osteochondroses: diseases of growth centers. Physician and Sports Medicine. 1989;17:51-62.
Petty N.J., Moore A.P. Neuromusculoskeletal Examination and Assessment, second ed. Edinburgh: Churchill Livingstone; 2001.
Ray A.K., Soutar D.S. Skin injuries and wound healing. In: McLatchie G.R., Lennox C.M., editors. The Soft Tissues. Edinburgh: Churchill Livingstone, 1993.
Reid D.C. Sports Injury Assessment and Rehabilitation. Edinburgh: Churchill Livingstone; 1992.
Reigger C.L. Mechanical properties of bone. In: Gould J.A., editor. Orthopaedic and Sports Physical Therapy. second ed. St Louis: C.V. Mosby; 1990:3-47.
Rothwell A.G. Quadriceps haematoma: a prospective study. Clinical Orthopaedics and Related Research. 1982;171:97-103.
Rydevik B., Lundborg G., Bagge U. Effects of graded compression on intraneural blood flow. Journal of Hand Surgery. 1981;6:3-12.
Salter R.B. The biological concept of continuous passive motion of synovial joints. In: Wing J.W., editor. Articular Cartilage and Knee Joint Function. New York: Raven Press, 1990.
Salter R.B., Harris W.R. Injuries involving the epiphyseal plate. Journal of Bone and Joint Surgery. 1963;45A:587.
Slater H., Butleter D.S., Shacklock M.O. The dynamic central nervous system: examination and assessment using tension tests. In Boyling J., Palastanga N., editors: Grieve’s Modern Manual Therapy, second ed, Edinburgh: Churchill Livingstone, 1994.
Smith E.L., Smith P.E., Ensign C.J. Bone involutional decrease in exercising middle-aged women. Calcified Tissue International. 1984;36:129-138.
Snook G.A. Tenosynovitis in long distance runners. Medicine and Science in Sports and Exercise. 1972;4:166.
Stanish W.D., Rubinovich R.M., Curwin S. Eccentric exercise in chronic tendinitis. Clinical Orthopaedics. 1986;208:65-68.
Steadman J., Rodkey W., Singleton S. Microfracture technique for full-thickness chondral defects. Operative Techniques in Orthopaedics. 1997;7(4):300-304.
Threlkeld A.J., Currier D.P. Osteoarthritis: effects on synovial joint tissues. Physical Therapy. 1988;68(3):364-370.
Toomey M. The pelvis, hip and thigh. In: Zaluaga M., et al, editors. Sports Physiotherapy. Edinburgh: Churchill Livingstone, 1995.
Upton A.R., McComas A.J. The double crush in nerve entrapment syndromes. Lancet. 1973;2:359-362.
Urist M.R., Nakagawa M., Nakata N., Nogami H. Experimental myositis ossificans: cartilage and bone formation in muscle in response to a diffusible bone matrix derived morphogen. Archives of Pathology and Laboratory Medicine. 1978;102:312-316.
Van der Meulen J.C.H. Present state of knowledge on processes of healing in collagen structures. International Journal of Sports Medicine. 1982;3:4-8.
Vas W., Cockshott W.P., Martin R.F., Pai N.K., Walker I. Myositis ossificans in haemophilia. Skeletal Radiology. 1981;7:27-31.
Viidik A. Biomechanics and functional adaptation of tendons and joint ligament. In: Evans F.G., editor. Studies on the Anatomy and Function of Bone and Joints. Berlin: Springer, 1966.
Walter J.B., Israel M.S. General Pathology, sixth ed. London: Churchill Livingstone; 1987.
Watson A.S. Children in sport. In: Bloomfield J., Fricker P.A., Fitch K.D., editors. Textbook of Science and Medicine in Sport. Melbourne: Blackwell Scientific Publications, 1992.
Weisman G., Pope M.H., Johnson R.J. Cyclic loading in knee ligament injuries. American Journal of Sports Medicine. 1980;8:24-30.
White A. Neurophysiology of acupuncture analgesia. In: Ernst E., White A., editors. Acupuncture: A Scientific Appraisal. Oxford: Butterworth-Heinemann, 1999.
Wilkinson P.C., Lackie J.M. The adhesion, migration and chemotaxis of leucocytes in inflammation. Current Topics in Pathology. 1979;68:47-88.
Williams J.G.P. Injuries in Sport. England: Bayer (UK), Haywards Heath; 1979.
Williams J.G.P., Sperryn P.N. Sports Medicine. London: Edward Arnold; 1976.
Wolman R.L., Reeve J. Exercise and the skeleton. In: Harries M., et al, editors. Oxford Textbook of Sports Medicine. Oxford: Oxford University Press, 1994.
Woo S.L.Y., Gomez M.A., Akeson W.H. The time and history-dependent viscoelastic properties of the canine medial collateral ligament. Journal of Biomechanical Engineering. 1981;103:293-298.
Wroble R.R. Articular cartilage injury and autologous chondrocyte implantation—which patients might benefit? Physician and Sportsmedicine. 2000;28(11):43-49.