Traumatic Brain Injury
TRAUMATIC BRAIN INJURY
Traumatic brain injury (TBI) is the negative effect on brain function resulting from external physical force. TBI has great influence on outcome from complex trauma. Almost half of the people sustaining a closed head injury die on site, and TBI is often the main cause of persistent morbidity, driving the need for discharge to long-term rehabilitation. Appropriate management of the brain injury results in improved outcome from trauma.35
Typically there is, at least initially, a diminished or altered state of consciousness. Impairment of cognition and physical function are common and may be temporary or permanent. Changes are seen in behavior and emotional control. Functional disability or psychologic maladjustment can be persistent and can have a devastating impact on lifestyle. The long-term effects associated with closed head injury vary depending on the severity of the injury. Differences in recovery are seen in people who appear to have identical injuries.
One of the great challenges to the management of TBI is that the need to control cerebral perfusion or lower intracranial pressure (ICP) can collide with therapeutic goals focusing on problems with infection, ventilation, renal function, or even generalized perfusion. For this reason, integration of brain and systemic care is critical on all levels, from understanding the pathophysiologic principles to treatment through interdisciplinary communication.8
Incidence and Risk Factors
TBI accounts for a disproportionate share of morbidity and mortality in traumatized individuals. The past 2 decades have witnessed a significant decline, however, in overall TBI mortality from the mid-30% range in the 1970s to less than 20% in the 1990s. This improvement has paralleled an understanding of the secondary injury process and an appreciation that all neurologic damage does not occur at the moment of insult but evolves over the ensuing hours and days from various biochemical and molecular derangements.
TBI leads to 500,000 hospitalizations per year, resulting in over 175,000 deaths and significant disability. Although the statistics are somewhat variable, it is estimated that the incidence of TBI currently approaches 222 in 100,000 population per year.18
The incidence of head injury appears to be close to 2 million persons per year. Many are left with lifelong disabilities that keep them from returning to their preinjury lifestyles. It is believed that there are now more than 5 million persons living with the effects of TBI. Men have twice the chance of sustaining head injury than women based on activities. The average age at the time of injury is 29 years. The incidence if TBI increases with declining income and with rising population density, suggesting that those most at risk for brain trauma are low-income inner-city dwellers.
TBI peaks at three different age levels. The first peak occurs in early childhood at age 1 to 2 years and is related most often to child abuse. The second occurs in late adolescence and early adulthood between ages 15 and 24 years and may be related to risk-taking behaviors. One of the most widespread causes of head injury among young people is bicycling. Wearing an appropriate helmet reduces the risk of severe head injury by 88%.2
Approximately 300,000 sports-related concussions occur annually. Actual incidence may be higher because of potential underreporting of concussion symptoms by athletes. Concussion may occur in sports and situations not typically thought to put the athlete at risk and not necessarily during games. A single concussion does not necessarily lead to long-term neuropsychologic or cognitive complications, but multiple concussions can cause long-term neuropsychologic abnormalities, particularly in executive functioning and information-processing speed. Athletes who have had previous concussions are more likely to have future concussions with longer recovery time.14
The third peak in TBI occurs in the elderly population and is related most often to falls. This group is the most likely to be hospitalized, and approximately 7% will die while hospitalized. Although elderly individuals account for less than 15% of trauma admissions due to falls, they account for half of deaths due to falls. Stretching of the bridging vessels over the surface of the brain results in increased susceptibility to tearing under shear forces. In addition, there seems to be a significant, age-related decline in cerebrovascular autoregulation that may partially explain the worse outcomes seen in elderly individuals with TBI.1
Of the severely brain-injured, approximately 60% of adults and 92% of children are injured in a motor vehicle accident. Pedestrians injured by automobiles represent some of the most seriously injured individuals in trauma. The elderly are at particular risk for being struck as pedestrians and make up a significant percentage of pedestrians who have been struck by a motor vehicle. Slow ambulation; impaired reflexes; misjudgment; and visual, auditory, and gait impairment appear to be involved, as elderly individuals are frequently struck within marked crosswalks or walk directly into the path of an oncoming vehicle. In the elderly there are significantly increased mortality rates, with a majority of deaths occurring at the scene or at the emergency department.1 Motor vehicle accidents still account for the majority of injuries, but the increasing mandate for seatbelt use and availability of air bags appear to be reducing injuries. The incidence of penetrating TBI from gunshot wounds is increasing, and in some urban communities it is now the most common type of injury seen.18 Brain injury due to firearms is associated most often with attempted suicide.32
Alcohol use and abuse are frequently associated with brain trauma. Fifty percent of people admitted into hospitals with head trauma are intoxicated at the time. Brain injury may be two to four times higher in alcoholics than in the general population.23
Etiologic Factors
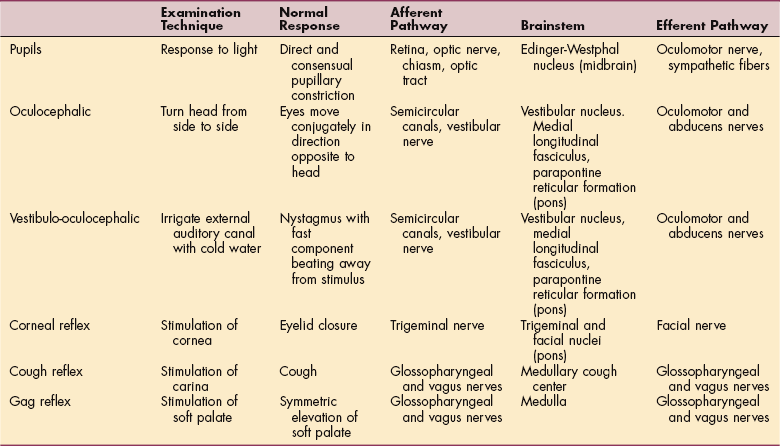
TBIs can come from open head injury or closed head injury. With an open head injury, the meninges have been breached, leaving the brain exposed. Penetrating missile injuries create localized, focal lesions that, when not fatal, cause limited damage to the brain. It is not the size of a missile but its velocity that generally determines the extent of damage. Penetrating injury also causes vascular injury, including disruption or the formation of aneurysms or pseudoaneurysms.35 Fig. 33-1 shows the kind of damage that can occur from a gunshot wound.
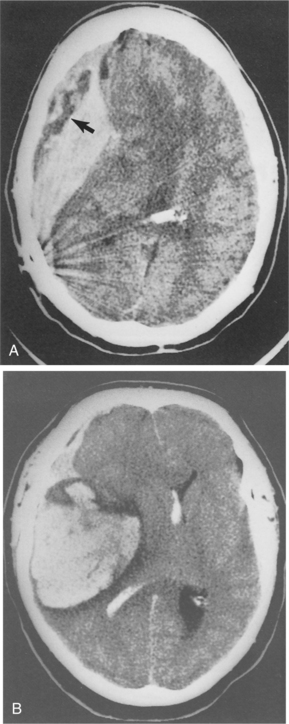
Figure 33-1 Gunshot wound resulting in both intracerebral and epidural hemorrhage. A, The bullet is shown on CT scan resting in a midline position with streaking effect of fragments also seen. The arrow points to area of decreased density thought to be epidural bleeding. B, Large intracerebral hemorrhage is noted with blood present in the ventricle. (Reprinted from Ramsey R: Neuroradiology, Philadelphia, 1994, WB Saunders, p 407.)
A closed head injury occurs when there is no skull fracture or laceration of the brain. A closed head injury occurs when the soft tissue of the brain is forced into contact with the hard, bony, outer covering of the brain, the skull. The initial blow occurs under the point of impact; then, as the brain decelerates against the contralateral skull, injury occurs to tissue on the opposite side. Such contrecoup injury is frequently worse than the injury underlying the impact.
Actual loss of consciousness does not always occur, although there is generally an altered state of consciousness. Mild closed head injuries can occur after a severe neck injury without the head actually striking any surface. The symptoms are worse when there is a rotational component to the head injury in addition to the back-and-forth jarring.17 Both diffuse injury and rupture of veins bridging from the brain to the venous sinuses producing subdural and subarachnoid hemorrhage can occur. Rotational forces are the most likely forces to cause diffuse axonal injury, including damage to brainstem structures, such as the reticular activating system.
Severe head injuries result from significant bruising and bleeding within the brain. Approximately 25% of people with a normal initial computed tomographic (CT) scan will develop late hemorrhages. Contusions are usually more severe in persons with skull fracture than in those without fracture. Although contusion is the hallmark of TBI, severe or even fatal damage to the brain can occur without contusion.4
Pathogenesis
Primary damage is the result of forces exerted on the brain at the time of injury. Secondary damage refers to changes compromising brain function that result from the brain’s reaction to trauma or other system failure. Causes of secondary damage include brain swelling and impaired cerebral perfusion (Fig. 33-2). Diffuse brain injury includes axonal injury, hypoxic damage, and edema. Multiple small hemorrhages may occur and are predictive of a poor outcome.7
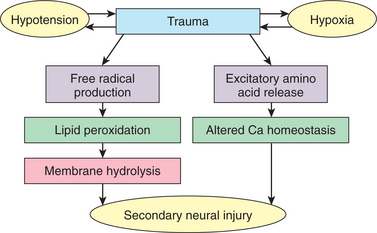
Figure 33-2 Biochemical and molecular substrates of the secondary injury cascade. (Reprinted from Salaman M: Current techniques in neurosurgery, ed 2, Philadelphia, 1993, Current Medicine.) Current Medicine
Vascular Changes.: Focal brain injuries usually result in cerebral contusions. Vascular damage is sustained at the moment of impact and leads to infarction within the cortical grey matter. Glial elements encapsulate the infarction, ultimately creating a residual cystic cavity.26 Typically, contusions occur at the poles and on the inferior surfaces of the frontal and temporal lobes. Occipital blows are more likely to produce contusions than are frontal or lateral blows. Areas where the cranial vault is irregular, such as on the anterior poles, undersurface of the temporal lobes, and undersurface of the frontal lobes are commonly injured. With fracture of the cranial vault, there may be damage to the superficial epidural vessels and, particularly in the case of falls, there can be rupture of the bridging vessels between hemispheres.42 Fig. 33-3 shows CT scan images of changes seen after TBI.
Figure 33-3 CT scans of two patients with closed head injury. A, This patient has a right temporal epidural hematoma (arrows). The mesencephalic cisterns are patent in the top left, indicating a lack of brainstem compression despite mass (arrowheads). B, This patient has suffered an acute left subdural hematoma (arrowheads) with midline shift (arrows). (Reprinted from Townsend CM: Sabiston textbook of surgery, ed 17, Philadelphia, 2004, Saunders.)
TBI can be associated with other forms of vascular change. Gliding contusions, or hemorrhagic lesions in the cortex, may be the result of movement of the cortical grey matter in relation to the underlying white matter, causing shear strains to damage the penetrating vessels found at the grey and white matter interface.57 Fig. 33-4 shows the effects of shearing injury as seen on CT scan. Subarachnoid hemorrhage is common due to the rupture of pial vessels within the subarachnoid space. This may trigger vasospasm that can lead to reduced regional blood flow. Injury to the vessels within the white matter can also cause significant neurologic consequences, especially if it is in the area of the basal ganglia.26
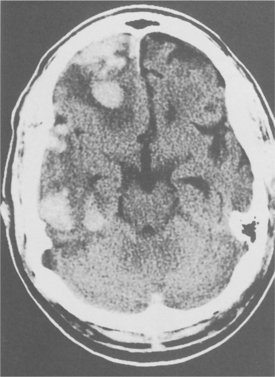
Figure 33-4 Contusion with shearing injury. CT scan shows multiple rounded areas of blood density with surrounding edema. Many of these areas are at the junction of grey and white matter consistent with shear injury. (Reprinted from Ramsey R: Neuroradiology, Philadelphia, 1994, WB Saunders, p 409.)
The increase in blood volume is considered to be the most important cause of increased ICP after head trauma. There can be bleeding into the epidural compartment, creating a mass effect that can displace the brain and increase ICP. The shear and tensile forces of traumatic injury can also create a subdural hematoma by disruption of the bridging veins. Acute hydrocephalus occurs when blood accumulates in the ventricular system, expanding the size of the ventricles and causing increased pressure on brain tissue being compressed between the skull and the fluid-filled ventricles.16 Vascular volume can increase if venous outflow is blocked or increased cerebral blood flow (CBF) increases passively because of loss of autoregulation. Cerebrospinal fluid (CSF) volume increases may be the result of blockage of outflow pathways or interference with reabsorption. When the volume of one compartment changes slowly, compensatory decreases in the volume of other compartments may prevent a rise in ICP. When the volume change is rapid or the compensatory mechanisms are exhausted or dysfunctional, the ICP goes up.8
The overall result of these vascular changes is the decreased ability of the cerebral vessels to maintain necessary homeostasis in the face of changing blood pressure or blood gas composition. Initially, within the first few hours after severe injury, there is decreased CBF both globally and at the impact site, which can induce ischemia. Within 24 hours the blood flow can be at normal or above-normal levels.26
The impairment of autoregulation of circulation has been described in the presence of moderate to severe head injury. This allows blood flow to the brain to become dependent upon the systemic arterial pressure. Elevated blood pressure can result in hyperemia and decreased blood pressure, causing hypoperfusion. Impaired vascular responsiveness of blood gas changes in head injury results in abnormal arteriolar vasoconstriction in the presence of carbon dioxide. Vascular volume can increase if venous outflow is blocked or increased CBF is recruited for metabolic reasons (e.g., seizures, pyrexia) or increases passively because of loss of autoregulation.
Posttraumatic aneurysms of the intracavernous internal carotid artery can be associated with delayed and sometimes lethal massive epistaxis. This can be a result of basal skull fractures in the region of the carotid canal or cavernous sinus and/or orbital fractures and compromise of the optical nerves. Knowledge of these risk factors and early diagnosis can minimize the high mortality risk. Fig. 33-5 demonstrates this relationship. It can take from days to years for the artery weakening to develop, with an average time of 3 weeks. Because of the close anatomic relationship of the intracavernous portion of the internal cerebral artery (ICA) to the oculomotor, optic, abducens, trochlear, and trigeminal nerves, these structures may also be damaged during the aneurysm development, resulting in effects such as blindness, facial numbness, and/or oculomotor palsy.19
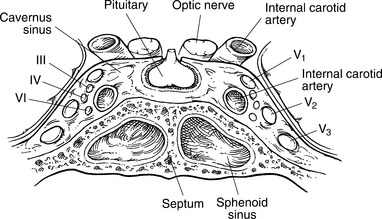
Figure 33-5 Diagram showing the close proximity of the intracavernous internal carotid artery and the sphenoid sinus. (Reprinted from Cummings CW, Haughey BH, Thomas R, et al: Cummings otolaryngology: head and neck surgery, ed 4, St Louis, 2004, Mosby.)
There appears to be a change in the endothelium, or walls, of the blood vessels following brain injury. In the normal brain, neurotransmitters such as acetylcholine induce dilation of the vessels through the release of endothelium-derived releasing factor, causing relaxation of the smooth muscle in the vessel wall. In the injured brain this reaction can be missing, resulting in abnormal vasoconstriction.15 Additional changes at the level of the endothelium result in a disturbed blood-brain barrier in the injured brain. This results in leakage of serum proteins and neurotransmitters into the parenchyma, causing edema. The effects of edema in the brain are described in Chapter 28.
Parenchymal Changes.: Axonal injury is a consistent feature of the traumatic event. Shear and tensile forces most likely disrupt the axolemma, which impairs transport of proteins from the cell body and causes swelling of the axon. The distal axon segment detaches and undergoes wallerian degeneration. These reactive axonal swellings, or retraction balls, full of axon material develop and can be detected in the injured brain within 12 hours of injury. The myelin sheath pulls away from the axon. The axonal changes are seen throughout the brain regardless of site of impact. The damage is different from that of stroke or tumor, which produces a more complete but local deafferentation. Typically, with diffuse axonal injury, there remain intact axons interspersed with the damaged axons. There is evidence of the potential for recovery of function based on the possible sprouting of undamaged axons to reoccupy the areas left vacant by degenerating axons. Secondary cell death by necrosis of the cellular membrane can be a result of edema. Apoptosis, or programmed destruction from within the cell through changes in the DNA, can result in cell death that occurs days, weeks, or months after injury.43
Study of excitotoxicity related to diffuse brain injury shows that the increase in extracellular neurotransmitters, resulting in increased potassium, causes a massive depolarization of the injured brain. There is a complex interaction of the various amino acids and neurotransmitters, which may affect the postsynaptic functions, resulting in secondary dysfunction of the neural mechanisms of the brain. The excitatory neurotransmitter glutamate appears to rise to abnormal amounts following brain injury. Glutamate is neurotoxic when concentrations increase. See Chapter 28 for information on the damage to the nervous system associated with glutamate.
Free radicals are generated by TBI. Extensive membrane depolarization, induced by trauma, allows for a nonselective opening of the voltage-sensitive calcium channels and an abnormal accumulation of calcium within neurons and glia. Such calcium shifts are associated with activation of lipolytic and proteolytic enzymes, protein kinases, protein phosphatases, dissolution of microtubules, and altered gene expression.18
Frank blood that moves into the parenchyma is possible and can cause extensive damage and infection of the tissue, especially with open wounds.
Compressive Damage.: Intracranial hypertension can produce herniation. During trauma, the brain may shift from its normal symmetric position. The most common herniation is the lateral tentorial membrane separating the cerebral hemispheres from the posterior fossa. This shift may cause compression of the brainstem, the pituitary, or other delicate brain structures. Since the brainstem controls the body’s major visceral functions, brainstem involvement may result in paralysis or death. In less severe situations, autonomic nervous system changes may include changes in pulse and respiratory rates and regularity, temperature elevations, blood pressure changes, excessive sweating, salivation, tearing, and sebum secretion. Because the brain is surrounded by the rigid skull, swelling of the brain, or pooling of blood, pushes tissue through openings in the base of the skull or through the other compartments of the brain, resulting in herniation through the foramen magnum. Fig. 33-6 shows the herniation possible with brain injury with epidural bleeding. Table 33-1 lists the possible signs of intracranial hypertension and associated herniation syndromes.
Table 33-1
Signs of Intracranial Hypertension and Associated Herniation Syndromes
| Sign | Mechanism | Type of Herniation |
| Coma | Compression of midbrain tegmentum | Uncal, central |
| Pupillary dilation | Compression of ipsilateral third nerve | Uncal |
| Miosis | Compression of the midbrain | Central |
| Lateral gaze palsy | Stretching of the sixth nerves | Central |
| Hemiparesis* | Compression of contralateral cerebral peduncle against tentorium | Uncal |
| Decerebrate posturing | Compression of the midbrain | Central, uncal |
| Hypertension, bradycardia | Compression of the medulla | Central, uncal, cerebellar (tonsillar) |
| Abnormal breathing patterns | Compression of the pons or medulla | Central, uncal, cerebellar (tonsillar) |
| Posterior cerebral artery infarction | Vascular compression | Uncal |
| Anterior cerebral artery infarction | Vascular compression | Subfalcine (cingulate) |
*Hemiparesis will occur ipsilateral to the hemispheric lesion (false localizing sign).
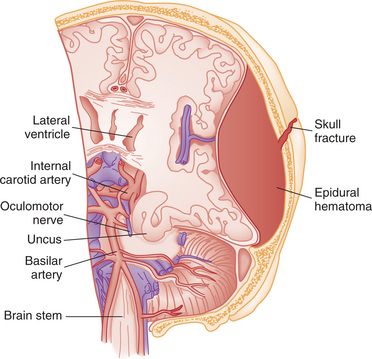
Figure 33-6 Anterior view of transtentorial herniation caused by large epidural hematoma. Skull fracture overlies hematoma. (Reprinted from Rockswold GL: Head injury. In Tintinalli JE et al, eds: Emergency medicine, New York, 1992, McGraw-Hill, p 915.) McGraw-Hill
Hypoxia.: Hypotension (systolic blood pressure less than 90 mm Hg) occurring between injury and resuscitation occurs in one third of severe TBI victims. It can be caused by blockages resulting in decreased blood in the brain or by decreased oxygen in the blood due to concomitant pulmonary insult. It is associated with doubling of mortality rate and a significant increase in morbidity. Early hypotension is also a strong predictor of poor outcome and can lead to intracranial hypertension in later stages.44
Hypertension.: Intracranial hypertension can interfere with perfusion by lowering the cerebral perfusion pressure (CPP). Under normal circumstances, cerebral pressure autoregulation maintains CBF constant over a CPP range of approximately 50 to 150 mm Hg. Following trauma, this relationship may be partially or totally dis- rupted; the brain can weather limited changes in CPP without notable alterations of CBF.
Clinical Manifestations
Signs and Symptoms.: Mild TBI is termed a concussion. Concussions are infrequently associated with structural brain injury and rarely lead to significant long-term sequelae. Moderate TBI may be associated with significant structural injury such as hemorrhage or contusion, but death is uncommon. Severe TBI generally results in some form of cognitive and/or physical disability or in death, especially with very low Glasgow Coma Scale (GCS) scores.18
Concussion.: In minor head injury, or concussion, the loss of consciousness lasts a relatively short time, or there may be no loss of consciousness. The postconcussion syndrome is a distinct entity. Symptoms usually associated with concussion are dizziness, disorientation, nausea, and headache. The client may be irritable or distractible and have difficulty with reading and memory. There may be complaints of headache, fatigue, personality changes, and decreased control of emotions.
The symptoms generally reflect both the focal and the diffuse nature of the damage. Changes to neurons, axons, neurotransmitter metabolism, neuroendocrine system (pituitary gland), CBF, and reticular activating system are common. The shearing effects and coup/countercoup can be responsible for dysfunction in frontal and temporal lobes. A right-sided cortical lesion could cause problems of visual-spatial processing, whereas a left-sided lesion could result in verbal processing deficits. Damage in the area of the amygdala may lead to heightened arousal, which enhances sensory information processing and is linked to emotional responses. The function of the amygdala is essential to the learning process and understanding of the consequences of action. Divided attention deficit, a reduction in information processing capacity, speed, or amount of information that can be processed, is associated with acceleration and deceleration head injury. This may be related to the diffuse white matter lesion, brainstem dysfunction, or a disruption in the frontal-limbic reticular activating system.13 Neuropsychologic testing has shown significant cognitive disability following a concussion. Neuropsychologic testing has documented cognitive impairments, including a reduction in information processing speed, attention, reaction time, and memory for new information.18
Concussions in athletes can lead to symptoms as described above. A single concussion does not correlate with long-term neuropsychologic or cognitive complications, but multiple concussions can result in long-term neuropsychologic abnormalities, particularly in executive functioning and information-processing speed.
Although cognitive impairment has been shown to resolve within about 7 days for most concussions, cognitive impairment has been shown to persist, particularly for athletes suffering multiple concussions. Epidural hematomas frequently result from skull fractures with subsequent laceration of the middle meningeal artery. Classically, epidural hematoma leads to an initial loss of consciousness, which is followed by recovery of consciousness and a lucid period. The athlete then progressively deteriorates neurologically, with development of headache, a decline in mental status, and focal neurologic findings such as contralateral weakness or numbness, pupillary reflex abnormalities, or facial asymmetry.
Migraine headaches with and without aura can develop in the hours to weeks after a mild concussion. Immediately after mild TBI in sports such as soccer, football, rugby, and boxing, children, adolescents, and young adults may have a first-time migraine with aura. This syndrome may be triggered multiple times after additional mild TBI and has been termed footballer’s migraine. Cluster headaches can develop after mild TBI. Subdural hematomas can result in headaches that are nonspecific and that can be mild to severe, paroxysmal or constant, and bilateral or unilateral.18
Nonspecific psychologic symptoms such as personality change, irritability, anxiety, and depression are reported by over one half of individuals within 3 months of mild TBI. Fatigue and disruption of sleep patterns are also often reported. Posttraumatic stress disorder, which has many symptoms similar to those of the postconcussion syndrome, may occur after mild TBI.
Levels of Consciousness.: Altered level of consciousness is a state that can occur with both diffuse and focal head injuries. This can be a result of diffuse bilateral cerebral hemispheric damage or a smaller lesion that affects the brainstem. In many cases, it is probably a combination. In moderate or severe head injury, unconsciousness can be prolonged or persistent. Arousal is associated with wakefulness and depends on an intact reticular formation and upper brainstem.
Coma is regarded as the lowest level of consciousness and is characterized as not obeying commands, not uttering words, not opening the eyes, or a state of unresponsiveness. This indicates advanced brain failure, with bilateral cerebral hemispheric or direct involvement of the brainstem. Coma rarely lasts longer than 4 weeks. The GCS is the most widely used instrument for determining level of consciousness; it is used to determine current status and potential for improvement (Box 33-1).
Some individuals may continue to exhibit a reduced level of consciousness, a condition referred to as persistent vegetative state, or postcomatose unawareness, characterized by a wakeful, reduced responsiveness with no evident cerebral cortical function. This includes eye opening with sleep-wake cycles and tracking of the eyes, controlled at a subcortical level. The vegetative state (VS) is notable for preserved arousal mechanisms associated with a complete lack of self or environmental awareness. Individuals in a VS will open their eyes spontaneously, without visual tracking or gaze fixation. There is no purposeful movement and the individual remains mute. The VS can result from diffuse cerebral hypoxia or from severe, diffuse white matter impact damage. The brainstem is usually relatively intact.3
The locked-in syndrome consists of quadriplegia in the setting of preserved awareness and arousal. It is caused by injury to the ventral pons.18 It spares vertical eye movements and can be seen with disordered breathing patterns associated with injury to brainstem respiratory centers. Cheyne-Stokes breathing is a rhythmic pattern of alternating rapid breathing and momentary stopping of breathing. It often presents in individuals with hemispheric lesions that are bilateral or can be the result of lesions in the diencephalon. Hyperventilation is seen in individuals with pontine or midbrain lesions. Apneustic breathing is characterized by a prolonged pause at the end of inspiration and indicates lesions of the mid-and caudal portions of the pons. Ataxic breathing, seen with damage to the medulla, is irregular in both rate and tidal volume.
Cognitive and Behavioral Impairments.: Cognitive impairments include problems with attention, memory, concentration, and executive functions. Residual cognitive and behavioral deficits often remain despite a return to full consciousness. Deficits, including disorders of learning, memory, and complex information processing and loss of abstract thinking and complex problem solving, reflect the frontal lobe pathology associated with TBI. Loss of executive functions is observed and there is often confusion and disorientation in addition to difficulty in problem solving, delayed processing, and lack of initiation. Mood disturbances include depression and anxiety. Symptoms are related to the area of the brain injured.
When the damage is in the orbitofrontal area, behavior may be excessive and disinhibited. Inappropriate social and interpersonal behaviors, including inappropriate sexual behavior, occur with lesions in this area. Septal area lesions result in irritability and rage. Pseudobulbar injuries can result in emotional lability, including euphoria, involuntary laughing, or crying that is not associated with negative emotions.
Cognitive deficits are not always directly observable, but the observable behavior provides information regarding the ability to integrate cognitive processes. The observable behavior of a brain-injured person is directly related to the integrity of cognitive function. The behaviors reflect the inability to adjust to the environment. Typical behaviors include erratic wandering; motor, sensory, and verbal perseveration; imitation of gestures; restlessness; refusal to cooperate; and striking out in response to stimulus or in random fashion. Often the individual will attempt to run away from the institution or home. Deficits in attention are also common. Clients show impulsiveness, hyperactivity, and difficulty sustaining attention. Behavioral changes can be present without cognitive or physical deficits.50 Box 33-2 describes some of the typical cognitive characteristics and the resulting behavioral disturbances seen in persons after TBI. A useful tool to assess behaviors as a function of cognitive recovery is the Rancho Los Amigos Scale (Table 33-2). Table 33-3 includes some of the behavioral disturbances and their manifestations in people with TBI.
Table 33-2
Rancho Los Amigos Scale for Levels of Cognitive Functioning
| Level | Behaviors Typically Demonstrated |
| I | No response: Client appears to be in a deep sleep and is completely unresponsive to any stimuli. |
| II | Generalized response: Client reacts inconsistently and nonpurposefully to stimuli in a nonspecific manner. Responses are limited and are often the same regardless of stimulus presented. Responses may be physiologic changes, gross body movements, or vocalization. |
| III | Localized response: Client reacts specifically but inconsistently to stimuli. Responses are directly related to the type of stimulus presented. May follow simple commands in an inconsistent, delayed manner, such as closing eyes or squeezing hand. |
| IV | Confused-agitated: Client is in heightened state of activity. Behavior is bizarre and nonpurposeful relative to immediate environment. Does not discriminate among persons or objects; is unable to cooperate directly with treatment efforts. Verbalizations frequently are incoherent or inappropriate to the environment; confabulation may be present. Gross attention to environment is very brief; selective attention is often nonexistent. Client lacks short-term and long-term recall. |
| V | Confused-inappropriate: Client is able to respond to simple commands fairly consistently. However, with increased complexity of commands or lack of any external structure, responses are nonpurposeful, random, or fragmented. Demonstrates gross attention to the environment, but is highly distractible and lacks ability to focus attention on a specific task. With structure, may be able to converse on a social-automatic level for short periods of time. Verbalization is often inappropriate and confabulatory. Memory is severely impaired, often shows inappropriate use of objects; may perform previously learned tasks with structure but is unable to learn new information. |
| VI | Confused-appropriate: Client shows goal-directed behavior but is dependent on external input for direction. Follows simple directions consistently and shows carryover for relearned tasks with little or no carryover for new tasks. Responses may be incorrect due to memory problems but appropriate to the situation; past memories show more depth and detail than recent memory. |
| VII | Automatic-appropriate: Client appears appropriate and oriented within hospital and home settings; goes through daily routine automatically, but frequently robot-like with minimal-to-absent confusion; has shallow recall of activities. Shows carryover for new learning, but at a decreased rate. With structure is able to initiate social or recreational activities; judgment remains impaired. |
| VIII | Purposeful-appropriate: Client is able to recall and integrate past and recent events and is aware of and responsive to environment. Shows carryover for new learning and needs no supervision once activities are learned. May continue to show a decreased ability relative to premorbid abilities, abstract reasoning, tolerance for stress, and judgment in emergencies or unusual circumstances. |
Modified from Hagen C, Malkmus D, Durham P: Levels of cognitive functioning. In Rehabilitation of the head injured adult: comprehensive physical management, Downey, CA, 1979, Professional Staff Association of Rancho Los Amigos Hospital, pp 87-88.
Table 33-3
Typology of Behavioral Disturbances after Traumatic Brain Injury
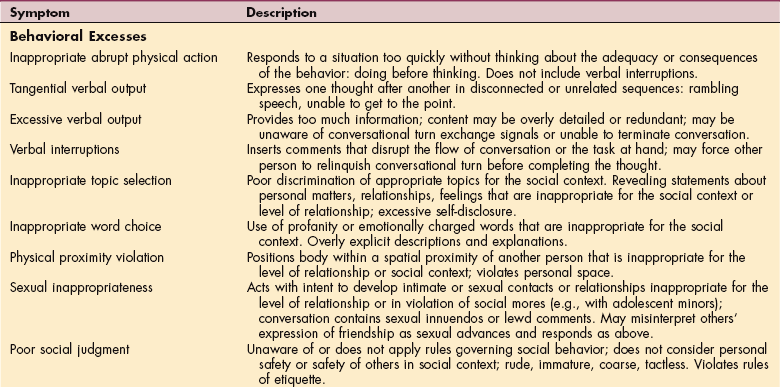

Modified from Vomoto JM: Neuropsychological assessment and rehabilitation after brain injury. In Berrol S, ed: Physical medicine and rehabilitation clinics of North America. Traumatic brain injury, Philadelphia, 1992, WB Saunders, p 307.
Impairment of memory is common with head injury. Retrograde amnesia is the partial or total loss of ability to recall events that have occurred during the period immediately preceding head injury. Posttraumatic amnesia is the time lapse between the injury and the point at which functional memory returns.5 During this time there may be improvement in automatic activities, but there is no carryover of tasks requiring memory or learning. The duration of posttraumatic amnesia is considered a clinical indicator of the severity of the injury.37
Anterograde memory is the ability to form new memory. Loss of anterograde memory is common and manifests as decreased attention or inaccurate perception. The capacity for anterograde memory is frequently the last function to return following recovery from loss of consciousness.
Memory disturbance is common with concussion and minor head injury. Memory function is disbursed throughout the brain (see Chapter 28), and there appears to be a lack of ability to use semantic organizational strategy to remember something by associating it with relevant cues. There is commonly difficulty with identifying nonverbal stimuli, reproducing visual stimuli, and recalling verbal material. Complaints of memory problems are associated with poor performance on tests of speed, reaction time, attention tasks, and complex perceptual-motor abilities. Language deficits are often seen as word-and name-finding problems. However, recovery of language function appears to surpass that of memory in individuals with minor head injury.13
TBI is associated with several neuropsychiatric disturbances that can range from subtle deficits to severe disturbances including cognitive deficits, mood and anxiety disorders, psychosis, and behavioral problems. More than 50% of individuals with TBI develop psychiatric sequelae.40
Pain.: Pain is a common complaint after brain injury with complex interaction on both physical and neuropsychologic function. Even with a mild concussion there is some level of trauma associated. Head and neck pain is common with whiplash, and there is an increased incidence of physical trauma associated with the severity of head injury. Pain can cause a persistent distraction that pulls the individual’s attention away from activity and can decrease the ability to concentrate. It can affect the ability to sleep, which leads to daytime lethargy, and it contributes to emotional reactions such as anxiety and depression.45
Neuropathic pain can result from the aberrant somatosensory processing in the peripheral or central nervous system, most common with damage in the area of the thalamus. Myofascial pain is common with trigger points, stiffness, and weakness. A deep, burning pain followed by persistent, involuntary, and irregular movements of the toes and feet, termed painful legs and moving toes, can be associated with minor foot and ankle injuries. Fibromyalgia can develop, as it is related to sleep disturbances, anxiety, and depression. Another component of pain is suffering, in which the intensity is dependent on the person’s mood, life experience, and level of social support. The result can often be that the cycle of pain and limitation of central processing can lead to a condition that mimics chronic pain syndrome. Chronic pain can have an impact on the neuropsychologic function as part of a vicious cycle. Managing this syndrome in the individual with head injury can be challenging and warrants good decisions regarding both the pharmacologic and neuropsychologic approaches.13
Cranial Nerve Damage.: Focal damage in the brainstem can be reflected in the loss of cranial nerve function. The following are signs of specific cranial nerve deficits.21,46 Examination of the eyes may yield valuable information about the level of brainstem disease causing coma, given the proximity of centers governing eye movement, pupillary function, and elements of the ARAS. Completely normal pupillary function and eye movements suggest that the lesion causing coma is rostral to the midbrain. Table 33-4 lists brainstem reflexes in the comatose individual.
The olfactory nerve is well protected in the cribriform plate, but shearing of the fibers to the extent of damage occurs in about 7% of head injuries. In about 50% of those cases, this is a temporary condition.
The optic nerve is not a true cranial nerve but rather a direct extension of the brain. The most vulnerable component of the optic nerve in people with head injury is the portion of the nerve located within the optic canal. Damage to this portion can result in monocular blindness, a dilated pupil with an absent direct pupillary response, and a brisk consensual response to light. Partial visual defects may take the form of scotomata, sector defects, and upper or lower hemianopia.
The oculomotor nerve works in conjunction with the trochlear and abducens nerves to move the eyeball in the orbit to maintain gaze stability and scanning. Damage is often due to direct insult to the musculature, but it can also be due to cerebral herniation. This nerve is damaged in less than 3% of people with head injury. In some cases there is development of misdirection of regeneration, resulting in constriction of the pupil when any one of the extraocular muscles supplied by the third nerve is activated. Also, because of misdirection of the growing axons, the levator muscle of the lid may receive fibers destined for other muscles. When the person affected attempts to look down, instead of the globe moving down, the lid becomes elevated.
It is important to understand the difference between peripheral damage to the oculomotor system and the abnormal movement that represents damage of a central nature that affects eye movements. Conjugate lateral deviation of the eyes is a sign either of an ipsilateral hemisphere lesion, a contralateral hemisphere seizure focus, or damage involving the contralateral pontine horizontal gaze center. Lateral gaze palsy may signal central herniation with compression of bilateral sixth nerves. Tonic downward deviation of gaze is suggestive of injury or compression involving the thalamus or dorsal midbrain, such as may occur with acute obstructive hydrocephalus or midline thalamic hemorrhage. Tonic upward gaze has been associated with bilateral hemispheric damage. Ocular bobbing, a rapid downward jerk followed by a slow return to mid-position, is indicative of pontine lesions. Rapid intermittent horizontal eye movements suggest seizure activity.18
The fourth cranial nerve is the least frequently injured oculomotor nerve. Damage is usually in the form of contusion or stretching. With severe frontal blows, there can be direct damage to the fourth nerve or hemorrhage of the tentorial incisura. There can be a vertical diplopia mimicking a third nerve palsy. The prognosis for recovery in fourth nerve palsy is poor because the nerve is so slender that it is often avulsed by the trauma.
The most common form of trigeminal nerve injury after head trauma involves the supraorbital and supratrochlear nerves as they emerge from the orbit. Damage results in anesthesia of a portion of the nose, eyebrow, and forehead. Facial trauma may extend the sensory deficits to the cheek, upper lip, gums, teeth, and hard palate. In deep coma, the eyelids can be opened easily, and the corneal reflex (indicating fifth nerve palsy) is often absent.
The abducens nerve is often injured when the head is crushed in an anteroposterior plane with resultant lateral expansion and distortion of the skull. It can also be damaged in fractures of the petrous bone. Vertical movement of the brainstem may severely stretch the sixth nerve as it leaves the pons. There can also be damage in relation to the third and fourth nerves in the orbital fissure. There is failure of the eye to abduct when the head is passively turned away from the side of the lesion. Abnormal wandering movements are present in midbrain lesions, and they usually disappear when the person regains consciousness.
Trauma to the facial nerve is common in head injury. With injury to the temporal bone or swelling of the nerve, external compression caused by hematoma symptoms of facial nerve palsy may occur. Loss of tear production, saliva secretion, and taste in the anterior two thirds of the tongue and loss of stapedius muscle function may be noted. Muscles controlling facial expressions become weak.
Hearing and vestibular dysfunction occur in head injuries. Transverse fractures of the temporal bone may cause disruption of the auditory and vestibular end organs or transient eighth nerve dysfunction. A blow to the head creates a pressure wave that is transmitted through the petrous bone to the cochlea, resulting in hair cell damage and degeneration of cochlear nerves. For further information on dizziness and vertigo, see Chapter 38.
The glossopharyngeal, vagus, spinal accessory, and hypoglossal cranial nerves pass through the jugular foramen at the base of the skull. The twelfth nerve passes through the hypoglossal foramen nearby. Injury is most often from a missile wound, but fractures of the occipital condyle can also produce lower cranial nerve palsies. Symptoms include cardiac irregularities, excessive salivation, loss of sensation and gag reflex of the palate, loss of taste on the posterior third of the tongue, hoarse voice, dysphagia, and deviation of the tongue to the side of the lesion.
Motor Deficits.: Abnormalities of movement include monoplegia, hemiplegia, and abnormal reflexes. Often there is flaccidity, the absence of motor responses, at the onset, which is gradually replaced by increased tone, spasticity, and rigidity. Decorticate posturing, or hyperactive flexor reflexes in the upper extremities and hyperactive extensor response in the lower extremities, is common initially and reflects the loss of cortical control. Decerebrate posturing, or hyperactive extensor reflexes in both the upper and lower extremities, reflects injury at the superior border of the pons resulting in the loss of inhibitory control of the cortex and basal ganglia.2,34
The specific manifestations of hemiparesis may include loss of selective motor control, abnormal balance reactions, and sensory loss. Cerebellar and basal ganglia dysfunction can result in ataxia, dysmetria, and tremor or bradykinesia. See Chapter 32 for further information regarding focal damage to the brain associated with the area of infarct.
Direct trauma to subcortical and substantia nigral neurons can result in movement disorders occurring shortly after an injury. Movement disorders occurring months following the injury have been hypothesized to be related to sprouting, remyelination, inflammatory changes, oxidative reactions, and central synaptic reorganization. Peripheral trauma that precedes the development of a movement disorder may alter sensory input, leading to central cortical and subcortical reorganization.
Postural and kinetic tremor can be due to direct traumatic lesions of the dentatothalamic circuit. Postural-kinetic tremors of the arms, legs, or head may occur within weeks of mild TBI even without loss of consciousness. Peripheral trauma can induce tremor, which can occur along with reflex sympathetic dystrophy, dystonia, and myoclonus. Myoclonus, dystonia, and athetosis may be present in individuals with posttraumatic tremors.
Contralateral dystonia can be due to a lesion in the striatum, particularly the putamen. The onset of dystonia may have a latency period from 1 month to 9 years. Spastic dystonia due to pyramidal and extrapyramidal injury and paroxysmal nocturnal dystonia are variants of posttraumatic dystonia. Often individuals develop posttraumatic dystonia as a delayed sequela of severe TBI, initially characterized by coma and quadriplegia. After the individual awakens and the plegia improves, severe action dystonia develops.18
Heterotopic Ossification.: Another complication associated with head injury is the formation of heterotopic ossification (HO), or abnormal bone growth around a joint. The cause and pathogenesis of HO is unknown, but bone scans show evidence of increased uptake, and there is also elevation of alkaline phosphatase.
The onset of HO is usually 4 to 12 weeks after the head injury, and it is first represented as a loss of range of motion. Local tenderness and a palpable mass can be detected, and there can be erythema, swelling, and pain with movement. HO in the hip area can mimic deep vein thrombosis. Peripheral nerve compression will sometimes develop, especially if the HO is in the elbow. HO can also result in vascular compression and possible lymphedema.53
Medical Complications.: Multiple medical complications can also occur after TBI. Cardiovascular effects of TBI include neurogenic hypertension and cardiac dysrhythmias. Respiratory complications such as neurogenic pulmonary edema, aspiration pneumonia, and pulmonary emboli usually caused by deep venous thrombosis are common. Other complications include disseminated intravascular coagulation, hyponatremia, diabetes insipidus, and stress gastritis. Iatrogenic infections are common.
MEDICAL MANAGEMENT
The diagnosis of brain injury starts at the level of concussion, and the American Academy of Neurology guidelines include three levels of concussion. The second impact syndrome should be addressed as a diagnostic entity, as the cumulative damage from multiple concussions can lead to long-term brain damage and disability. Every possible concussion should therefore be reported and maintained as part of an individual’s medical record. Athletic injury, falls, and minor auto accidents can result in concussions that are often not reported to health care providers. In general, people who have lost consciousness for 2 minutes or more following head injury should be observed medically from the time of the impact.
When the person sustains a severe head injury, the GCS is used to assess level of consciousness. Using this scale, three aspects of coma are observed independently: eye opening, best motor response, and verbal response. A score of 8 or less indicates coma. Reflex responses tested by applying a noxious stimulus, such as pressure on a nail bed, fall into three categories: appropriate, inappropriate, or absent.
There is some controversy over including motor responses to describe the depth of coma because neural structures regulating consciousness differ from and are more anatomically distant from those regulating motor function.41
Oculomotor and pupillary signs are valuable in assisting with the diagnosis, localizing brainstem damage, and determining the depth of coma. Pupillary examination should document size and reactivity to light. Greater than 1 mm difference in size or asymmetry should be considered abnormal. Once the baseline neurologic status has been determined, repeated evaluations are critical to monitor improvement, provide prognostic data, or detect deterioration, which should be addressed immediately. Symptoms of focal neurologic deficits, lethargy, or skull fractures should be monitored. A mental status examination is important in all head-injured individuals. Subtle abnormalities may be a guide to significant intracranial injury.
Higher GCS is associated with greater than normal cardiac index responses and better tissue oxygenation. Poor outcomes are related to low GCS, hypertension, mild tachycardia, normal pulmonary function, and reduced tissue oxygenation.39
Diagnostic imaging can provide significant information, which can guide the intervention and allow a more accurate prognosis. CT is the primary imaging modality for the initial diagnosis and management of the head-injured person. CT scanning of the head reveals the presence of hemorrhage, swelling, or infarction. In individuals with traumatic coma, patterns on CT that have been associated with worse neurologic outcome include lesions in the brainstem, encroachment of the basal cisterns, and diffuse axonal injury (Fig. 33-7).51 An initially normal CT scan, however, is no assurance that hemorrhagic lesions will not occur.
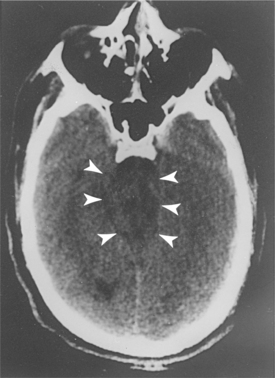
Figure 33-7 CT scan of the head in a patient with a closed head injury. Severe compression of mesencephalic cisterns is seen, indicating midbrain compression. (Reprinted from Townsend CM: Sabiston textbook of surgery, ed 17, Philadelphia, 2004, Saunders.)
Diffuse axonal injury (DAI) is a frequent CT and pathologic correlate of severe TBI, accounting for about 50% of primary brain injuries. DAI is usually associated with a poor outcome. DAI is readily identifiable on CT as multiple punctate hemorrhages, typically in the deep white matter and corpus callosum and occasionally in the brainstem. DAI may also occur as a result of mild TBI and may culminate in subtle types of cognitive deficits. Approximately 10% to 15% of individuals with clinically severe TBI have a normal CT scan. In such situations, the possibility of extracranial or intracranial vascular disruptions may exist, and angiography should be considered.18
Current imaging with CT or magnetic resonance imaging (MRI) does not permit quantification of neural injury that occurs after a traumatic brain concussion. Proton magnetic resonance spectroscopic imaging can be used to assess the neurochemical damage derived from a cerebral concussion by monitoring N-acetyl-L-aspartate (NAA) levels over time. Current research indicates that NAA diminution appears to be linked to a general mitochondrial dysfunction, and therefore NAA restoration can be considered a surrogate marker of metabolic recovery. The ability to identify postconcussive individuals who are in a vulnerable cellular state is important as there can be a catastrophic deterioration in individuals even with a simple head injury. The combination of metabolic data, physiologic data, and clinical observations satisfactorily addresses the complete recovery from concussion.36
CT and Doppler measurements could be combined to detect individuals at risk for secondary neurologic deterioration on admission to improve their initial disposition.30
Clinical examination is of no diagnostic value in predicting head CT scan findings and should not be used as a means of avoiding head CT scans in pediatric practice. If clinical examination alone is used to evaluate children with loss of consciousness or amnesia and minor TBI, intracranial injuries will be missed. Due to possible implications for learning, return to athletic activity, parent education, and potential medicolegal issues, in all children with observed loss of consciousness or amnesia a head CT scan should be considered as part of the evaluation. Warning signs that may portend need for urgent intervention include any vomiting, restlessness, any GCS score decrease, severe headache, confusion, and focal temporal blow.10 Vomiting may also represent an individual who is predisposed due to migraine familial characteristics, so this should also be considered.11
MRI is complementary to CT and is used in conjunction with, not as a replacement for, CT. The multiplanar capabilities of MRI are important to better demonstrate extra-axial hemorrhage located subfrontally, subtemporally, or along the tentorium. Lesions in the posterior fossa, as well as shear injury, are better demonstrated on MRI than on CT. MRI can also detect small hemorrhages in the corpus callosum, intraventricular hemorrhages, or effacement of basal cisternal structures in the absence of brain shift or mass lesions. This can lead to the diagnosis of increasing ICP, a significant risk in brain injury.25
MRI offers a sensitive window of detection for neuropathology from mild TBI. MRI is more sensitive than CT to DAI. Anatomic distribution of tissue damage and precise indications of the volume of lesions seen on MRI can predict recovery of the brain in the subacute phase. Prediction of outcome should not be based on CT scanning or MRI alone.22 Positron emission tomography (PET) can be used to identify both structural and functional consequences and is especially valuable for mild head injury.29
Electrophysiologic tests that have been used for predicting coma outcomes include somatosensory evoked potentials, transcranial motor evoked potentials, brainstem auditory evoked potentials, and event-related potentials. Visual, auditory, and somatosensory evoked potentials make it possible to observe changes in a lesion, and therefore may aid in prognosis, but are not routinely used in isolation.
Neuropsychologic evaluation is valuable in identifying the extent of the cognitive deficits. The evaluation consists of a series of cognitive challenges given to the individual, including assessment of sensorimotor status, attention span, memory, language, sequencing, problem solving, and verbal and spatial integration tasks. Comparisons of normal and brain-injured persons have been well documented. Previous tests of intellectual function, including IQ tests and achievement tests, can be helpful for comparison, especially in mildly brain-injured clients. Cognitive impairment is the primary contributor to disability with moderate to severe brain injury.28 Language and cognitive problems are examined by speech pathologists and can include neuropsychologic testing with naming tests, aphasia examinations, as well as tests of auditory comprehension and speed of comprehension.
Athletes should undergo formal neuropsychologic evaluations when injury is suspected because this may unmask subtle continued deficits when compared with baseline testing. Such deficits have been shown to correlate with duration of symptoms. This has become an increasingly important tool in concussion evaluation. Postural stability testing may also be undertaken for adjunctive data in determination of concussion severity.
Cognitive and behavioral dysfunctions caused by brain injury are similar to posttraumatic stress syndrome, conversion or hysterical reactions, malingering, depression, and anxiety. Therefore careful evaluation of each individual should be performed to determine cause of symptoms. It can be considered as well that the trauma occurring at the time of the injury may trigger posttraumatic syndrome or other psychoses in susceptible individuals with history of prior trauma.
Approximately 5% to 10% of individuals with severe TBI have an associated spine and/or spinal cord injury. Initial head injury evaluation and management thus require simultaneous evaluation and management for potential spinal injuries. The majority of individuals with severe TBI have multisystem injury. Possibility of other significant and potentially life-threatening injuries should be evaluated and the proper treatment priorities accordingly established. Fig. 33-8 demonstrates intubating an individual with acute trauma and an uncertain cervical spine.
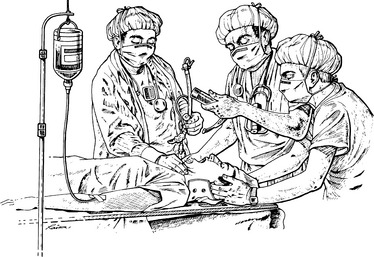
Figure 33-8 Intubating an acute trauma patient with an uncertain cervical spine. A hypnotic and a relaxant have been administered. One assistant maintains in-line axial stabilization with the occiput held firmly to the backboard; a second applies cricoid pressure. The posterior portion of the cervical collar remains in place to “discourage” atlanto-axial extension. (Reprinted from Stene JD: Anesthesia for the critically ill trauma patient. In Siegel JH, ed: Trauma: emergency surgery and critical care, Melbourne, Australia, 1987, Churchill Livingstone, p 843.)
TREATMENT
Treatment of TBI requires coordinated care and service from the onset of injury through the person’s lifetime. Fig. 33-9 represents the levels of care that are utilized in the course of treatment. Note that primary prevention is the first step, and it is only when prevention is not provided that the client must begin the acute medical phase.
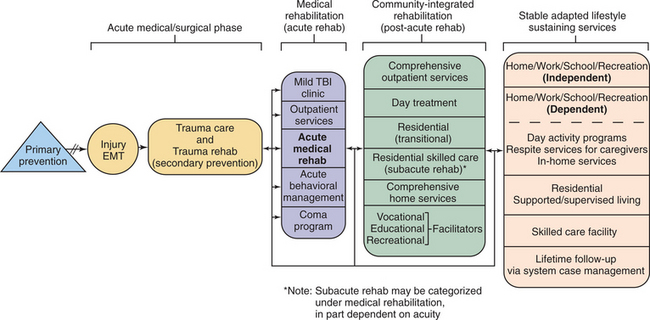
Figure 33-9 System of care for the person with TBI showing the components that should be considered in each phase. (Reprinted from Horn LJ: Systems of care for the person with traumatic brain injury. In Berrol S, ed: Physical medicine and rehabilitation clinics of North America. Traumatic brain injury, Philadelphia, 1992, WB Saunders, p 479.)
Prehospital management of the severely head-injured person includes rapid triage, resuscitation, and efficient transport. Survival and medical management with the goal of stabilization and prevention of secondary com- plications are the primary medical focus. Hypoxia is a frequent secondary insult; often the upper airway is obstructed, and clearing the airway is the first treatment administered. Intubation and ventilation are critical, with positive-pressure breathing techniques supplemented by 100% oxygen and early intervention.25 Hypotension (systolic blood pressure less than 90 mm Hg) and hypoxia (PaO2 less than 60 mm Hg) should be avoided if possible and corrected if present.
Emergency department treatment includes determination of head injury severity, identification of persons at risk of deterioration, and control of hypoxia and hypotension. Prevention of secondary brain damage caused by edema, increased ICP, or bleeding should be addressed. Treatment of the medical complications of the trauma are paramount but is not discussed here; the focus of this chapter is on the control and treatment of neurologic sequelae. Close clinical observation remains the best tool for neurologic monitoring in the early stages of head injury.
Surgical intervention is critical in the presence of hemorrhage to prevent neurologic compromise and can improve both short-and long-term outcomes. Uncal, transtentorial, or tonsillar herniations can occur with hematomas. In some cases, the individual may be lucid after the injury and then, in the presence of undetected hematoma, lapse into coma and die.
Injury to the dural sinus can occur with a depressed fracture over a major sinus and requires evacuation. Decompression of the skull, often using burr holes, is warranted in the presence of significant cerebral edema or subdural hematoma. Decompressive craniectomy for intractable brain swelling is an older treatment that has recently received renewed attention. A number of operations have been developed for or applied to decompression of the brain at risk for the sequelae of uncontrollable intracranial hypertension.
Individuals with a GCS score of less than 8, or individuals whose neurologic status cannot be assessed because of administration of sedative drugs or neuromuscular blocking agents, should be monitored for ICP increases. The institution of ICP monitoring remains somewhat controversial; however, ICP monitors provide the most reliable guide to treatment of the underlying brain injury. Elevation of ICP more than 20 mm Hg is a significant predictor of a poor outcome. Monitoring of ICP can be accomplished in a number of ways. The ventriculostomy catheter allows monitoring and drainage of CSF but it is the most invasive method and is associated with risk of infection. The epidural catheter, hollow subarachnoid bolt, and subarachnoid fiberoptic catheter are other options. All must be surgically placed. The noninvasive Doppler waveform can also provide information regarding ICP. If an ICP monitor is in place, the drainage of CSF may have significant therapeutic benefits.
CPP is determined by subtracting the ICP value from mean arterial blood pressure and represents the pressure driving CBF. Thus CPP and CBF can be positively affected by either lowering ICP or elevating systemic blood pressure. It is generally believed that active attempts to maintain CPP above 70 mm Hg improve outcome; however, there are no systematic scientific studies to validate this.18
Cerebral fluid volume can be reduced pharmacologically. Mannitol is used to control blood volume. Hyperventilation has been used as mechanism for controlling cerebral blood volume by increasing in blood pCO2, resulting in vasoconstriction of the central vessels and reduced CBF. This must be considered a short-term procedure to be used judiciously because the cerebral vasoconstriction induced may produce ischemia. There remains debate on its usefulness in the light of possibility for worse outcome.37 High-dose barbiturate therapy is generally reserved for those situations in which ICP changes becomes refractory to other available therapies.
Blood pressure control is important in brain-injured clients, and systolic blood pressure should be kept at a minimum of 90 mm Hg. If fluid management cannot keep the blood pressure at an adequate level, then vasopressor drugs are used. Phenylephrine is effective at maintaining stability. CSF can also play a role in ICP. This can be controlled by the use of hypertonic saline or mannitol. Removal of CSF can be accomplished by ventriculoscopy. In clinical studies, the use of mild hypothermia appears to reduce neuronal injury by decreasing the amount of glutamate released.
Glucocorticoids have been used to treat cerebral edema, but there appears to be little long-term effect.12 They are still used to reduce brain swelling and neuronal injury in select cases. They may be used counter the negative effects of TBI on the stress response to systemic trauma, then are rapidly withdrawn once a individual’s failure to improve neurologically clearly identifies him or her as a nonresponder. This usually becomes apparent in the first 48 hours.47
Management of secondary injury is as critical in TBI as it is in other brain disorders. Control of free radicals through use of vitamin E appears to show some effect. Study of neurotrophic agents has not yet proved to be effective in clinical studies, although they show effect in animals. The most active area of research in TBI today is in the field of molecular genetics. It has been noted that certain genes are upregulated, whereas others are downregulated, after both trauma and ischemia. Particular attention has been focused on the apolipoprotein E gene and its various alleles. Certain alleles have been associated with an increased susceptibility and severity of brain injury, and others have been linked to improved recoveries after TBI.
Because of the intense sympathetic stimulation seen with head injury, hypertension and tachycardia are prevalent. Cushing’s phenomenon, or a rise in blood pressure in the presence of an acute rise in ICP (most often caused by brainstem compression), may be present. Moderate increases in blood pressure can be tolerated, but extreme hypertension should be treated because it could lead to increased blood volume.25
For sports-related minor TBI, the American Academy of Neurology has defined three grades of concussion and recommended guidelines for return to play. For grades 1 and 2, there is transient confusion and no loss of consciousness. Resolution of concussion symptoms or mental status abnormalities occurs in less than 15 minutes in grade 1 and more than 15 minutes in grade 2. Grade 3 concussion is any loss of consciousness. Players with grade 1 concussion may return to play the same day if they have a normal sideline neurologic assessment, including a detailed mental status examination. Players with grade 2 and 3 concussions should not return to play the same day. Guidelines for return to play are necessary because of concern over the cumulative effects of even mild TBI. See Table 33-5 for current guidelines.14,18
Table 33-5
When to Return to Play after Removal from Contest
| Grade of Concussion | Time Until Return to Play* |
| Multiple grade 1 concussions | 1 week |
| Grade 2 concussion | 1 week |
| Multiple grade 2 concussions | 2 weeks |
| Grade 3: brief loss of consciousness (seconds) | 1 week |
| Grade 3: prolonged loss of consciousness (minutes) | 2 weeks |
| Multiple grade 3 concussions | 1 month or longer; based on clinical decision of evaluating physician |
*Only after being asymptomatic with normal neurologic assessment at rest and with exercise.
Reprinted from Practice parameter: the management of concussion in sports (summary statement). Report of the Quality Standards Subcommittee, Neurology 48:584, 1997.
Subacute.
In addition to attempting to maintain homeostasis in the brain, management of the other sequela of brain injury is important. Spasticity is controlled by the administration of baclofen, diazepam, or dantrolene. These medications must be used carefully because of their side effects, which include increased weakness, lethargy, and drowsiness. Intrathecal baclofen can be used selectively to decrease tone with a baclofen pump, and the overall side effects are decreased. Abnormal muscle tone can also be controlled by nerve and motor point blocks or by the administration of botulinum toxin directly into the muscle belly.25
Control of seizures is provided by the use of medication such as divalproex sodium (Depakote). If the thalamus is affected, there can be abnormal sensations or intractable pain. The use of antiseizure medications is effective but carries high side effects and is often not tolerated by the individual whose system is already compromised. Attempts to control aggressive behavior through use of carbamazepine (Tegretol) and propranolol (Inderal) have had limited success. The nontricyclic antidepressants seem to be the most effective when the person is depressed.20
Rehabilitation.
Rehabilitation of the head-injured person and return to optimal function are the goals once the medical status is stabilized. Highly skilled, specially trained interdisciplinary teams provide an organized approach to the complex deficits encountered after head injury. Rehabilitation management of the individual is dependent on the cognitive and behavioral level of function of the individual. The Rancho Los Amigos Scale (see Table 33-2), which assesses components of cerebral function, is widely used. Treatment protocols are established according to the level at which the individual is functioning.
Psychotherapy is critical in posttraumatic stress disorder and can be helpful to establish coping mechanisms to address the cognitive deficits and problem solving in relationship to daily activity.
Restoration of mobility, self-care, employment, and recreational activities depends on the level of sensorimotor impairment as well as cognitive status. See Special Implications for the Therapist: Traumatic Brain Injury, in this chapter.
Community Reentry.
Community reentry programs for the head-injured person enhance the transition from rehabilitation unit to independent living. Therapists play a significant role in such programs.
In order to return to a lifestyle that may include work and school, the person with TBI needs to learn how to cope with the multiple demands on his or her attention that are part of that lifestyle. The person with TBI will have difficulty with executive functions such as organizing time and information, self-monitoring, and selfcorrecting. Self-motivation is often lacking, and structuring is necessary to ensure follow-through on assigned activities. Extensive use of checklists and environmental cues is helpful when attempting to reintegrate the client into the community.
The therapist working with brain-injured individuals must understand the interaction between the deficits related to cognitive and social behaviors and the ability to learn to move.54 Cognitive rehabilitation and physical rehabilitation are closely related. Functional outcomes are limited by the cognitive status, and the understanding of the techniques that foster behavioral modification and learning in the head-injured should be used by the therapist while motor skill acquisition is being attempted.50 For further explanation regarding this motor control and for information specific to learning, see Chapter 28. For information regarding working on mobility in individuals with focal brain injury, see Special Implications for the Therapist: Traumatic Brain Injury, in this chapter.
Individuals with higher level physical skills and moderate-or low-level cognition skills are often the most difficult to reintegrate into the family and society. Therefore they may have higher levels of handicaps remaining after rehabilitation because family and coworker expectations are high based on motor function. Generally it is the cognitive functions that make one more successful in society. Aggressive counseling should start as soon as the behavioral and cognitive impairments are identified. Neuropsychologists and counselors can suggest interventions that help with cognitive functions, especially techniques to deal with memory loss, decreased attention span, and inappropriate behavior.
Significant deficits in motor skills but higher levels of cognitive skills generally lead to a higher quality of life. There are numerous upper and lower extremity adaptive devices to help perform activities of daily living. Electric wheelchairs controlled by the head, mouth, or hand and electric lifts for vans as well as hand controls for driving can assist with mobility. Computerized communications systems can improve interactions when speech is disrupted and cognition is still present.
The lack of motivation associated with TBI becomes a challenge for the therapist. Lack of internal initiation and decreased ability to learn may persist despite cues from the external environment. Determining goals that are meaningful to the client even though they may seem to be unrealistic is the first step in establishing motivation.
Alterations in attention span can be detrimental to progress in therapy. Reducing distracting stimuli can be helpful initially; distractions can be reintroduced as the ability to manage multiple input improves. In most cases the family of the survivor needs help understanding their family member’s social and behavioral changes.49
PROGNOSIS.
Because of the complex nature of the injury, predicting outcomes in brain injury is difficult at best. One major problem is with the consistency of the tests performed and interpretation of those tests. However, there are some indicators that can be considered broadly.52 The probability of having persistent symptoms and neuropsychologic deficits is the same whether a individual is only dazed or loses consciousness for less than 1 hour. The effects of repeated concussions are cumulative because the ability of the brain to accommodate the damage is compromised by previous trauma. Individuals with moderate TBI usually experience both cognitive and physical disabilities and typically require rehabilitation services after acute hospitalization. Nevertheless, the incidence of severe long-term disability is small.18
Injury severity is one of the main factors determining outcome. The depth of impaired responsiveness and the duration of altered consciousness have been related to outcome.31,55 The current gold standard for assessing those mental states in the head-injured person is the Glasgow Outcome Scale (Box 33-3). In addition, the duration of posttraumatic amnesia has been used as a predictor of severity of the head injury. Other aspects of neurologic functioning are predictive of outcome. Loss of pupillary light reflexes following head injury reflects significant damage to the brainstem and portends a poor prognosis. Oculomotor deficits often signal concomitant cerebral damage resulting in severe cognitive deficits. The degree of hypoxemia and hypotension encountered in the early stages can also have an effect on the long-term prognosis.48
CT has increased the ability to predict outcome in the head-injured person with a lesion of the brain parenchyma, intercranial hematoma, subdural hematoma, or massive hemispheric swelling. Acute hemispheric swelling with an extracerebral hematoma is associated with the worst prognosis. Unilateral brain contusion and DAI also carry a poor prognosis. A midline shift of brain structures, absent or compressed basal cisterns (indicating rising ICP), and subarachnoid hemorrhage will increase the risk of death or remaining in a VS.51
Epilepsy occurring within 7 days is often related to severe injury, depressed fracture, or intracranial hemor- rhage. Posttraumatic epilepsy may emerge months or years following brain trauma and is more common after severe brain injury. Late epilepsy occurs most often as grand mal seizures or temporal lobe seizures.23 For further information on seizures, see Chapter 36.
Dementia has been long recognized as a sequela of multiple head injuries in boxing, as evidenced by the term “punch drunk.” Neuropathologic studies of brains of boxers with dementia demonstrate β-amyloid protein-containing diffuse plaques and neurofibrillary tangles, which are pathologic features of Alzheimer’s disease.
Neuropsychologic dysfunction appears greater in people over the age of 30 years and those with less education. Social outcomes may be related to the premorbid status of the individual. History of substance abuse, low educational level, and psychiatric disorders can also limit success. Social and family problems are common and can cause isolation and poor quality of life.33
Cognitive deficits that affect motivation, attention, emotion, memory, or learning will slow progress. Lack of social skills has been reported to affect an individual’s ability to reintegrate into the community. Often it becomes difficult to sustain relationships that were stable before the injury. Working with professionals who recognize these deficits and are trained to treat them will improve the chances of increasing quality of life after head injury.
A compensation case or lawsuit is often filed in circumstances in which another party may be responsible for the TBI, such as a motor vehicle accident or on-the-job injury. In this circumstance, when individuals have persistent complaints, many physicians are appropriately concerned about compensation neurosis or malingering being the cause. Individuals with claims, however, have similar symptoms that improve with time and similar cognitive test results as those without claims. For many claimants, the end of litigation does not mean the end of symptoms or return to work. They are not cured by a verdict.18
TRAUMATIC BRAIN INJURY IN CHILDREN
TBI is one of the leading causes of death and disability in children of all ages.38 Nonaccidental injury is a common cause of head injury in infants and toddlers and is often the result of the battered child syndrome. The head injury is often caused by shaking or striking the child.
Although the pathology of the brain injury in the child reflects damage similar to that in the adult, there are differences. Infants typically have tears in the white matter of the temporal and orbitofrontal lobes. The infant will more often sustain a subdural or epidural hemorrhage than an older child but is less likely to have skull fracture because of the pliancy of the skull.
Drowning is the third leading cause of death in children aged 1 to 4 years. Peak incidences occur in 1-to 4-year-olds and in adolescent boys. Boys are three times more likely to be injured. Rapid resuscitation leads to better outcomes. As in adults, the motor activity return and pupillary light response are prognosticators of outcome (see Chapter 15).
A children’s coma scale has been developed for use in children under the age of 3 years (Box 33-4). The highest level of ocular response is eye tracking. The verbal response is rated highest by crying, then by spontaneous respirations, with the lowest score given for apneic breathing. The motor response has a highest score for flexing and extending the extremities.
Early management of the infant or child with TBI follows that of the adult, with some difference in the child’s ability to tolerate the medications used. Late seizures are less common with children than with adults, so the need to be maintained on seizure medication is less.
Rehabilitation goals for the child are similar to those of the adult, although play is used during therapy. Orthotic and assistive devices are used frequently but for a shorter time than for adults. Agitation is common and is often difficult for the parents and siblings to handle. Aggression, decreased attention span, hyperactivity, and socially inappropriate behavior are seen. These children often require a great deal of behavior modification.
Community reintegration can be as difficult for the child as it is for the adult. Schools are better prepared to handle cognitive delays than abnormal behaviors. Cognitive status may return in one area and remain defective in another. Attention and memory deficits may produce the greatest obstacles to learning.
References
1. Aschkenasy, MT. Trauma and falls in the elderly. Emerg Med Clin North Am. 2006;24(2):413–432.
2. Bernard, PG. Closed head injury: a clinical source book. Charlottesville, VA: Michie, 1994.
3. Berrol, S. Evaluation and the persistent vegetative state. Head Trauma Rehabil. 1986;1:7.
4. Bryan, VL. Management of residual physical deficits. In: Ashley MJ, Krych DK, eds. Traumatic brain injury rehabilitation. Salem, MA: CRC Press; 1995:319–365.
5. Cartlidge, N, Shaw, DA. Head injury. London: WB Saunders, 1981.
6. Cecchini, A. Functional assessment after traumatic brain injury. Neurol Report. 1998;22(4):136–143.
7. Chestnut, R, Marshall, L, Klauber, M. The role of secondary brain injury in determining outcome from severe head injury. J Trauma. 1993;34:216–222.
8. Chesnut, RM. Care of central nervous system injuries. Surg Clin North Am. 2007;87(1):119–156.
9. Chiu, ML. Wheelchair seating and positioning. In: Montgomery J, ed. Physical therapy for traumatic brain injury. New York: Churchill Livingstone; 1995:117–136.
10. Clement, CM. Clinical features of head injury individuals presenting with a Glasgow Coma Scale score of 15 and who require neurosurgical intervention. Ann Emerg Med. 2006;48(3):245–251.
11. Da Dalt, L. Characteristics of children with vomiting after minor head trauma: a case-control study. J Pediatr. 2007;150(3):274–278.
12. Dearden, N, Gibson, J, McDowall, D. Effects of high-dose dexamethasone on outcome from severe head injury. J Neurosurg. 1986;64:81–88.
13. Deiter, JN. Neuropsychology of minor head injury. In: Swerdlow B, ed. Whiplash and related headaches. Boca Raton, FL: CRC Press; 1999:879–916.
14. Dimberg, EL. Management of common neurologic conditions in sports. Clin Sports Med. 2005;24(3):637–662.
15. Domino, KB. Pathophysiology of head injury: secondary systemic effects. In: Lam AM, ed. Anesthetic management of acute head injury. New York: McGraw-Hill; 1995:25–59.
16. Evans, E, Randall, R. Predicting outcome following traumatic brain injury. Neurol Report. 1998;22(3):144–148.
17. Evans, RW. Mild traumatic brain injury. In: Berrol S, ed. Physical medicine and rehabilitation clinics of North America. Traumatic brain injury. Philadelphia: WB Saunders; 1992:427–437.
18. Evans, RW, Wilberger, JE. Traumatic disorders. In Goetz CG, ed.: Textbook of clinical neurology, ed 2, Philadelphia: Saunders, 2003.
19. Fontela, PS. Posttraumatic pseudoaneurysm of the intracavernous internal carotid artery presenting with massive epistaxis. Pediatr Crit Care Med. 2006;7(3):260–262.
20. Fulop, Z, Wright, D, Stein, D. Pharmacology of traumatic brain injury: experimental models and clinical implications. Neurol Report. 1998;22(3):100–109.
21. Gelber, DA. The neurologic examination of the traumatically brain-injured patient. In: Ashley MJ, Krych DK, eds. Traumatic brain injury rehabilitation. Salem, MA: CRC Press; 1995:23–42.
22. Gentry, R. Imaging of closed head injury. Radiology. 1994;191:1–17.
23. Giles, GM, Clark-Wilson, J. Brain injury rehabilitation: a neurofunctional approach. San Diego, CA: Singular Publishing Group, 1993.
24. Gill-Body, KM, Giorgetti, MM. Acute care and prognostic outcomes. In: Montgomery J, ed. Physical therapy for traumatic brain injury. New York: Churchill Livingstone; 1995:1–32.
25. Grady, MS, Lam, AM. Management of acute head injury: initial resuscitation. In: Lam AM, ed. Anesthetic management of acute head injury. New York: McGraw-Hill; 1995:87–100.
26. Grady, MS, Shapiro, X. Pathophysiology of head injury: primary central nervous system effects. In: Lam AM, ed. Anesthetic management of acute head injury. New York: McGraw-Hill; 1995:11–24.
27. Halley, M. Loss of consciousness: when to perform computed tomography? Pediatr Crit Care Med. 2004;5(3):230–233.
28. Heinman, A. Functional states and therapeutic intervention during rehabilitation. Am J Phys Med Rehabil. 1995;74:315–326.
29. Hughes, M, Cohen, WA. Radiographic evaluation. In: Cooper PR, ed. Head injury. ed 3. Baltimore: Williams & Wilkins; 1993:65–90.
30. Jaffres, P, Brun, J, Declety, P, et al. Transcranial Doppler to detect on admission patients at risk for neurological deterioration following mild and moderate brain trauma. Intens Care Med. 2005;31:785–790.
31. Kampfi, A, Schmutzhard, E, Franz, G, et al. Prediction of recovery from post-traumatic vegetative state with cerebral magnetic-resonance imaging. Lancet. 1998;351(9118):1763–1767.
32. Kraus, JF. Epidemiology of head injury. In: Cooper PR, ed. Head injury. ed 3. Baltimore: Williams & Wilkins; 1993:1–26.
33. Leahy, BJ, Lam, CS. Neuropsychological testing and functional outcome for individuals with traumatic brain injury. Brain Inj. 1998;12:1025–1035.
34. Leahy, P. Traumatic head injury. In: O’Sullivan SB, Schmitz TJ, eds. Physical rehabilitation. Philadelphia: FA Davis; 1994:491–508.
35. Langlois, JA, Rutland-Brown, W, Thomas, KE. Traumatic brain injury in the United States: emergency department visits, hospitalizations, and deaths. Atlanta, GA: US Department of Health and Human Services, 2006.
36. Le, TH, Gean, AD. Imaging of head trauma. Semin Roentgenol. 2006;41(3):177–189.
37. Muizelaar, J, Marmarou, A, Ward, JD. Adverse effects of prolonged hyperventilation in patients with severe head injury: a randomized clinical trial. J Neurosurg. 1991;75:731–739.
38. Nelson, VS. Pediatric head injury. In: Berrol S, ed. Physical medicine and rehabilitation clinics of North America. Traumatic brain injury. Philadelphia: WB Saunders; 1992:461–474.
39. Nicholls, TP. Survival, hemodynamics, and tissue oxygenation after head trauma. J Am Coll Surg. 2006;202(1):120–130.
40. O’Brien, RF. Medical conditions with psychiatric manifestations. Adolesc Med Clin. 2006;17(1):49–77.
41. Plum, F, Posner, JB. The diagnosis of stupor and coma, ed 3. Philadelphia: FA Davis, 1980.
42. Povlishock, JT, Christman, CW. The pathobiology of traumatic brain injury. In: Salsman SK, Faden AI, eds. The neurobiology of nervous system trauma. New York: Oxford University Press; 1994:109–130.
43. Povlishock, JT, Valadka, A. Pathobiology of traumatic brain injury. In: Finlayson MA, Garner SH, eds. Brain injury rehabilitation: clinical considerations. Baltimore: Williams & Wilkins, 1994.
44. Rangel-Castillo, L. Management of intracranial hypertension. Crit Care Clin. 2006;22(4):713–732.
45. Rosenthal, M, Christensen, BK, Ross, TP. Depression following traumatic brain injury: a review. Arch Phys Med Rehabil. 1998;79(1):90–103.
46. Rovit, RL, Murali, R. Injuries of the cranial nerves. In: Cooper PR, ed. Head injury. ed 3. Baltimore: Williams & Wilkins; 1993:183–202.
47. Salcman, M. An old debate joined again: should we give steroids to all head-injured patients? Crit Care Med. 2005;33(10):2440–2441.
48. Signorini, DF, Andrews, PJ, Miller, JD, et al. Adding insult to injury: the prognostic value of early secondary insults for survival after traumatic brain injury. J Neurol Neurosurg Psychiatry. 1999;66(1):26–31.
49. Springer, J, Farne, J, Bower, D. Common misconceptions about traumatic brain injury among family members of rehabilitation patients. J Head Trauma Rehabil. 1997;12(3):41–50.
50. Sullivan, K. Cognitive rehabilitation. In: Montgomery J, ed. Physical therapy for traumatic brain injury. New York: Churchill Livingstone; 1995:33–54.
51. Van der Naalt, J, Hew, JM, van Zorneren, AH, et al. Computed tomography and magnetic resonance imaging in mild to moderate head injury: early and late imaging related to outcome. Ann Neurol. 1999;46(1):70–78.
52. Van der Naalt, J, van Zorneren, AH, Minderhoud, JM. One year outcome in mild to moderate head injury: the predictive value of acute injury characteristics related to complaints and return to work. J Neurol Neurosurg Psychiatry. 1999;66(2):207–213.
53. Varghese, G. Heterotopic ossification. In: Berrol S, ed. Physical medicine and rehabilitation clinics of North America. Traumatic brain injury. Philadelphia: WB Saunders; 1992:407–416.
54. Winkler, PA. Head injury. In: Umphred DA, ed. Neurological rehabilitation. ed 5. St Louis: Mosby Elsevier; 2007:532–566.
55. Wedekind, C, Fischbach, R, Pakos, P. Comparative use of magnetic resonance imaging and electrophysiologic investigation for the prognosis of head injury, J Trauma. Inj Infect Crit Care. 1999;47(1):44–49.
56. Zablotny, C. Evaluation and management of swallowing dysfunction. In: Montgomery J, ed. Physical therapy for traumatic brain injury. New York: Churchill Livingstone; 1995:99–116.
57. Zimmerman, RA, Belaniuk, LT, Genneralli, T. Computed tomography of shearing injuries of the cerebral white matter. Radiology. 1978;127:393–395.