Vestibular Disorders
VESTIBULAR DISORDERS
Definition and Overview
The organs and nervous system circuitry make up the vestibular system, which detects both the forces created by gravity and the forces generated as we move. The ability to maintain clear vision during head motion and to determine head position or speed and direction of movement is provided by the vestibular system, which generates appropriate postural reflexes to maintain balance.34 Until the system fails, the activity of the vestibular system is noticed only in particular circumstances when it is stimulated beyond normal, such as during a long spin or an amusement ride. Vestibular disorders, with the exception of aggressive form of neoplasm, are not life threatening but can cause significant disability.
Because the vestibular system provides information about orientation in space, disorders of the vestibular system can cause a devastating abnormal sense of movement, visual instability, and loss of balance. The vestibular system works as part of the sensory triad for postural stability and must be integrated with somatosensory and visual input to determine appropriate postural strategies. The disorders of the vestibular input cannot always be assumed to be an actual loss of function, or even hypofunction, as many individuals complaining of dizziness and imbalance have normal tests of vestibular function but do not make appropriate use of the information within the environmental context.
Incidence and Etiologic and Risk Factors
The vestibular system can be involved in many conditions either directly or indirectly; therefore it is difficult to determine the incidence within the general population. Even the most common causes of lesions vary. Box 38-1 gives common causes of vestibular disorders. Although dizziness is common in all age groups, the frequency of dizziness increases with age. Aging has a significant effect on vestibular function but may not be the primary cause of imbalance. Hair cell loss occurs with aging, particularly in the ampulla. It has been estimated that neuronal loss in the vestibular nuclei occurs at a rate of approximately 3% per decade from the age of 40 years.51 Nearly 10% of people older than 45 years visit their physicians with this complaint. Most of this group is older than 75 years.16 Changes in the vestibular system happen during the normal aging process, but the problems reported to physicians are most likely related to a pathologic process. Dizziness is reported more frequently by women than by men.21
A dysfunction can affect the system at any level (Box 38-2). Disorders of the labyrinth, or inner ear, can affect the sensory end organs, resulting in abnormal input into other levels of the vestibular system. Damage to the peripheral vestibular nerve can be from a neurologic pathology, mechanical deformation from a nonneurologic pathologic condition, or trauma to the structures surrounding the nerve. A central deficit can be the result of damage to the area in the brainstem or cortex that processes vestibular information. Often the signs and symptoms help to indicate the location of the lesion or level of dysfunction.
At its most peripheral level, the end organ of the vestibular system is primarily a mechanical system designed to identify movement of the head. It consists of a fluid-filled labyrinth and is bordered laterally by the middle ear and medially by the petrous portion of the temporal bone. The pathologic condition at this level is related to the structures involved. Figs. 38-1 and 38-2 show the mechanism of the vestibular system and the relationship of the vestibular system to the nearby structures of the brain and bony structures.
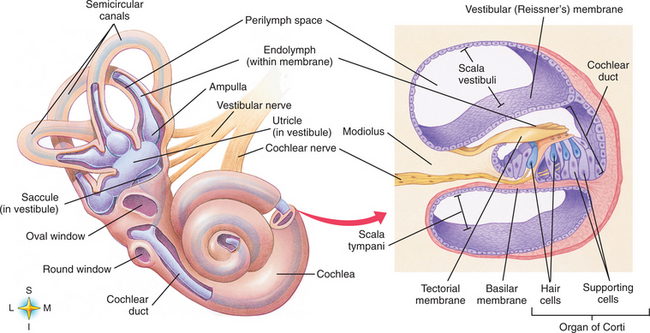
Figure 38-1 Components of the vestibular system and cochlea with distribution of neural connections. (From Thibodeau GA: Anatomy and physiology, ed 6, St Louis, 2006, Mosby.)
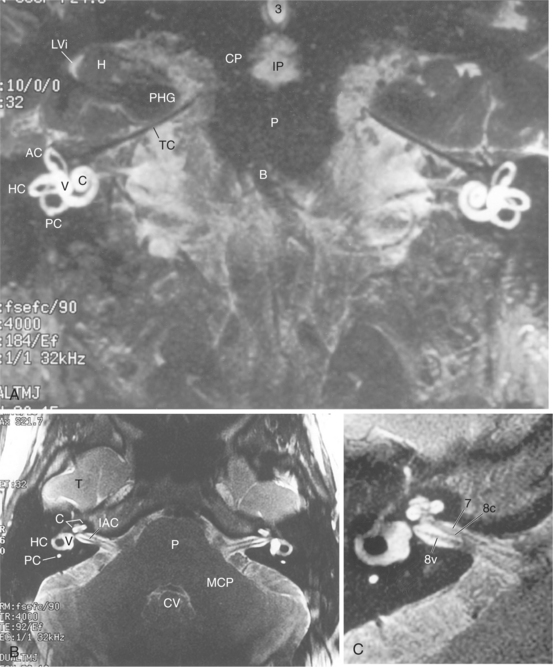
Figure 38-2 MRIs of the labyrinth. A, Coronal view. B, Axial view. C, Enlarged view. AC, Anterior semicircular canal; HC, horizontal canal; PC, posterior semicircular canal; C, cochlea; IAC, internal auditory canal; P, pons; 8v, eighth cranial nerve (vestibulocochlear nerve). (From Nolte J: The human brain: an introduction to its functional anatomy, ed 5, St Louis, 2002, Mosby.)
The semicircular canals are ring-shaped, fluid-filled structures oriented in three dimensions that provide sensory input about head velocity, or angular acceleration of the head. This is accomplished by movement of the fluid (endolymph) in the direction opposite to the head movement. Fig. 38-3 represents the relationship of the movement of the head and the resulting effect on the direction of the endolymph movement. The ampulla is deflected away from the direction of head movement by the movement of the endolymph. Figs. 38-4 and 38-5 show the relationship between the movement of the fluid within the canal, the orientation of the canal to the head, and the ampullar deflection. The speed and direction of the deflection of the hair cells of the ampulla determine the rate of firing of the vestibular nerve. The difference between the rate of firing of each vestibular nerve is interpreted by the brain as to the amount of angular acceleration of the head.42 Disorders at this level are most often related to fluid pressures, changes in the contents of the fluid, and inflammatory or infectious agents that affect the homeostasis of the system. Blows to the head or acceleration-related injuries can cause direct damage to the labyrinthine system. Ischemia in the surrounding vasculature can cause disrupted function.
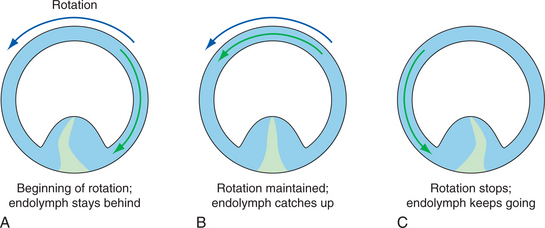
Figure 38-3 Relative movement of semicircular canals. A, Beginning of rotation. B, During rotation. C, End of rotation. (From Nolte J: The human brain: an introduction to its functional anatomy, ed 5, St Louis, 2002, Mosby.)
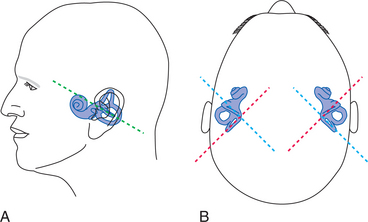
Figure 38-4 Orientation of the labyrinth. A, Side view. B, From the top. Labyrinth is enlarged relative to the head in this drawing for clarity. (From Nolte J: The human brain: an introduction to its functional anatomy, ed 5, St Louis, 2002, Mosby.)
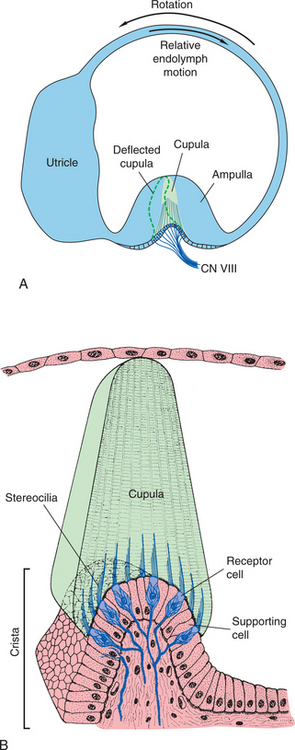
Figure 38-5 Two views of the ampulla. A, View of the semicircular canal and movement of the endolymph and resulting deflection of the cupula within the ampulla relative to head motion. B, Enlargement of the cupula showing its components. (From Nolte J: The human brain: an introduction to its functional anatomy, ed 5, St Louis, 2002, Mosby.)
Both ends of each fluid-filled semicircular duct open into the otolith, which contains the utricle and saccule. A portion of the floor of the utricle and the saccule is thickened and contains hair cells covered with a gelatinous membrane know as the otolithic membrane. Calcium carbonate crystals, or otoconia, adhere to this membrane. Fig. 38-6 shows how the otoconia sit above the hair cells. The weight of the otoconia produces a shear force on the hair cells with acceleration of the head. Information about linear acceleration, the tilt of the head, or the static position of the head with respect to gravity from the movement of the hair cells is transmitted to the vestibular nerve. Changes in the position of the hair cell respond to changes in head position in relation to gravity. Fig. 38-7 shows the relationship of the otolith to head positions changes. When this system is not working properly, there is loss of the ability to orient to gravity, and the person complains of bouncing or feels as if he or she were on a ship. It is difficult to orient to the environment when a person is not able to orient to gravity.5,23
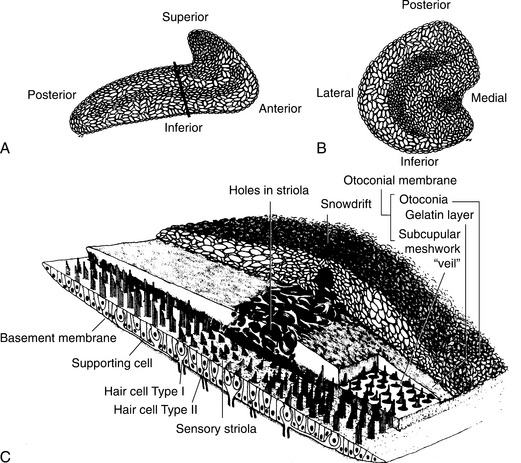
Figure 38-6 Arrangement of otoliths in the two maculae. A, Saccule. B, Utricle. C, Composition of the saccular otoconial membrane in a section taken at the level shown in A. (From Paparella MM, Shumrick DA, eds: Textbook of otolaryngology, vol 1, Philadelphia, 1980, WB Saunders.)
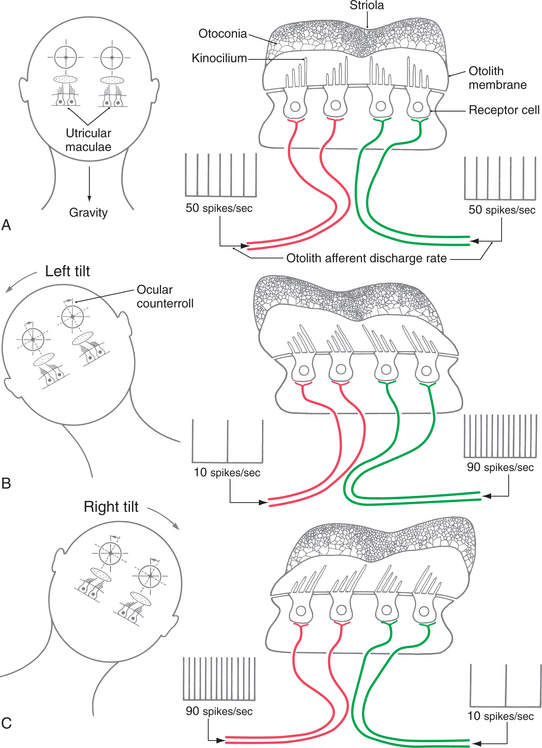
Figure 38-7 Patterns of excitation and inhibition for the left utricle and saccule when the head is tilted with the right ear 30 degrees down (A), upright (B), and tilted with the left ear 30 degrees down (C). The utricle is seen from above and the saccule from the left side. The background color represents baseline activity and black and white represent depolarization and hyperpolarization, respectively. (From Haines DE: Fundamental neuroscience for basic and clinical applications, ed 3, 2006, Churchill Livingstone.)
The hair cells, or nerve endings in both the semicircular canal and the otolith, work together to determine head position and rate of movement. There is a tonic resting firing rate of the hair cells that is carried through the vestibular nerve. The vestibular nerve transmits these afferent signals from the labyrinths, extending through the internal auditory canal and entering the brainstem at the pontomedullary junction. Somatosensory, visual, tactile, and auditory information is also processed in the vestibular nuclei.13,27 The brain uses the combined input in determining orientation within the environmental context.
When there is disruption of the vestibular nerve on one side causing loss of tonic firing, it results in abnormal information relayed to the brain about the position or movement of the head. The brain, as it compares the two sides, then interprets the abnormal input as movement when the head actually is at rest. This is the phenomenon of vertigo, or the illusion of turning or spinning. The individual will sense that the head is turning or the room is rotating.
The brain, however, has a remarkable ability to be able to adapt this system, and if the lesion is stable or nonfluctuating, the brain will learn to interpret movement based on the information that is available from the intact vestibular nerve on the opposite side. This adaptation depends on intact function of the central nervous system (CNS) and requires time and available visual information. The brain can then compare the vestibular information to the visual information in order to recalibrate. This adaptation takes about 2 weeks in an intact nervous system.5,10,17 when the CNS is unable to make the adaptation, or it is not adequate for the task at hand, the resulting sensation is one of spinning, especially when the eyes are closed or the visual environment is not stable.31
The vestibular system drives eye movement through its connections in the vestibular nucleus through the oculomotor nuclei to the extraocular muscles. This is known as the vestibulo-ocular reflex (VOR). The extraocular muscles are arranged in pairs and connected to the vestibular system so that a pair of canals is connected to a pair of extraocular muscles. This allows conjugate movement of the eyes as a result of head movement. Through the complex connections of the vestibular and oculomotor nuclei, information regarding direction and speed of head movement is directed to the eye muscles so that they react and move in the opposite direction as the head is moving. This keeps the visual environment in focus during rapid head movements. Fig. 38-8 shows the connection between stimulation of the semicircular canals and eye movement.
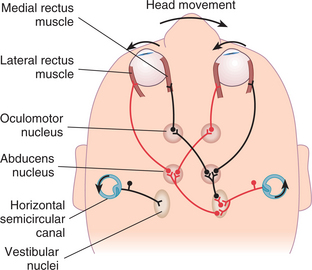
Figure 38-8 Neural connections in the direct pathway for the VOR from excitation of the left horizontal canal (left HC). As seen from above, a counterclockwise head rotation (head) produces relative endolymph flow in the left HC that is clockwise and toward the utricle. The cupular deflection excites the hair cells in the left HC ampulla, and the firing rate in the afferents increases (inset). Excitatory interneurons in the vestibular nuclei (vest. N.) connect to motor neurons for the medial rectus muscle in the ipsilateral third nucleus (III) and lateral rectus muscle in the contralateral sixth nucleus (VI). Firing rates for these motor neurons increase (bar graphs). The respective muscles contract and pull the eyes clockwise—opposite the head—during the slow phases of nystagmus. Inhibitory interneurons in the vestibular nuclei connect to motor neurons for the ipsilateral lateral rectus and contralateral medial rectus. Their firing rates decrease (bar graphs), and these antagonist muscles relax to augment the eye movement. (From Lundy-Ekman L: Neuroscience: fundamentals for rehabilitation, ed 3, Philadelphia, 2007, Saunders.)
When one side of the system is damaged or stimulated abnormally, the intricate coupling of the vestibular system and eye movements becomes disturbed.17,33 Because of the imbalance in the firing rate between the two sides of the vestibular system, the brain perceives movement of the head, although no movement may have occurred. Through the VOR mechanism, eye movements are triggered as if the head were moving. As the eyes move, the sensation created is one of the room spinning around.
The vestibular system is also responsible for motor output through the vestibulospinal reflexes (VSRs). The purpose of the VSR is to stabilize the body using the information provided by the vestibular system. Three pathways connect the vestibular nuclei to the anterior horn cells of the spinal cord. The lateral vestibulospinal tract, with its connection to the lateral vestibular nucleus, receives the majority of its input from the otoliths and the cerebellum. It is responsible for postural activity in the lower extremities in response to head position changes that occur with respect to gravity.32 The medial vestibulospinal tract from the medial, superior, and descending vestibular nuclei gets its input from the semicircular canals and triggers postural responses with regard to angular head motion. The medial vestibulospinal tract descends only through the cervical spinal cord. The reticulospinal tract gets its input from all the vestibular nuclei in addition to other systems concerned with maintaining balance. These reflexes together provide automatic control of the activity of the postural muscles in the trunk and limb. Because these tracts are the output for the vestibular system, damage to any part of the system can result in abnormal postural responses to movement.43
The vestibular nuclei are connected to each other in a complex manner across the brainstem. The descending nucleus is a relay between all the other nuclei and also to the cerebellum. The cerebellum receives output from the vestibular nucleus. The vestibular reflexes become uncalibrated and ineffective when the cerebellum is dysfunctional (see Chapter 28).
The brainstem can be affected in many ways. Pathologic processes can be direct, as in mechanical damage of the neurons in brain injury, bleeding disorders, and neoplasia. Hypoxic damage can occur in stroke or other conditions that cause decreased profusion of oxygen into the brainstem. Degenerative diseases such as multiple sclerosis (MS) and Alzheimer’s disease can cause abnormal function in the brainstem and its connections.
The vertebral-basilar artery supplies blood to the components of the vestibular system. The posterior and inferior cerebellar arteries feed the CNS. Because there is redundant blood supply via the circle of Willis, ischemia in this area is rare. The anterior inferior cerebellar arteries supply the peripheral mechanism via the labyrinthine, common cochlear, and anterior vestibular arteries.13 Disorders that affect the small vessels can cause direct damage to the peripheral vestibular system, or the ischemia can cause direct damage to the vestibular nuclei. Fig. 38-9 demonstrates the pathways associated with the end organ, vestibular nuclei, fibers of the cerebellum, and the extraocular muscles. This pathway involves inhibition in the process of modulating output to extraocular muscles.
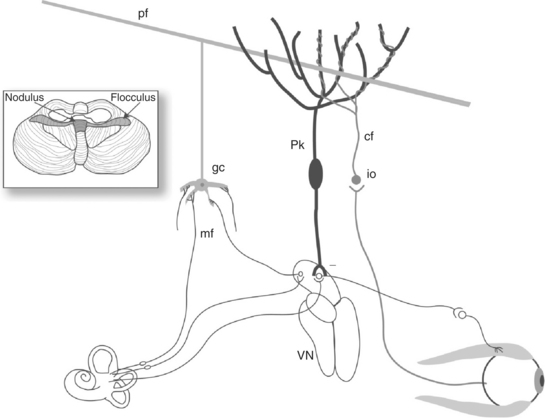
Figure 38-9 Circuitry of the cerebellum involved in modifying the VOR. Inputs from primary vestibular afferents and secondary neurons in the vestibular nuclei (VN) form mossy fiber (mf) inputs to cerebellar granule cells (gc). Parallel fibers (Pf) that originate from granule cells synapse weakly with Purkinje cells (Pk) and cause a high tonic inhibitory output of simple spikes from the Purkinje cells onto secondary vestibular neurons controlling the VOR. Climbing fiber (cf) input from the inferior olive (io) carries sensorimotor error information such as retinal slip. Climbing fibers make extensive and strong synapses onto Purkinje cells. Climbing fiber activity leads to complex spikes in the Purkinje cells, which can alter the efficacy of the parallel fibers’ synapses onto the Purkinje cells, a form of learning. (From Cummings CW, Haughey BH, Thomas R, et al: Cummings otolaryngology: head and neck surgery, ed 4, St Louis, 2004, Mosby.)
MEDICAL MANAGEMENT
History.
Dizziness is one of the most common clinical complaints when the vestibular system is involved in some way. Dizziness is a broad term for the sensation that results from the disruption of information from the vestibular, visual, and somatosensory systems. The description of dizziness may reflect the nature of the disorder. The history of the dizziness should give an indication of the cause of the symptoms and lead toward diagnosis. The symptoms as reported can reflect an abnormality in the vestibular system or may indicate some other general medical cause. Often the history is complex, and much insight is gained by listening carefully and allowing the client to provide his or her own thoughts on the probable cause. See Box 38-1 for a list of common vestibular causes of dizziness.
Static disturbances come about because of the tonic imbalance of vestibular tone when the head is still. Determining sensation when the head is stable, with the individual sitting unsupported, can give initial cues. A sensation of rotation may represent maladaptation of the input, reflecting a unilateral disorder. Floating, swimming, or leaning may represent abnormal otolith response.
When the complaint is of feeling lightheaded, several conditions should be considered. Presyncope is related to blood flow and may be related to orthostatic hypotension. Anxiety or panic can trigger sensation of lightheadedness, but usually there are other abnormal sensations such as paresthesia or shortness of breath. (See section on somatoform disorders later in this chapter.) When the complaint of lightheadedness is accompanied by a lack of feeling grounded, the integration between the vestibular and somatosensory system may be disrupted.
Head motion–provoked disturbances reflect abnormalities in the ability of the system to identify movement accurately, with abnormal gain of the system. This can be tested through responses to head rotation. The time it takes the brain to resettle after this motion can also reflect lack of CNS recalibration.
Nausea is a frequent complaint associated with dizziness and reflects the stimulation of the medullary centers. Direct damage to the medulla causes the most severe nausea, as in stroke, but it can also be reflected in traumatic brain injury. Sudden-onset conditions often create a high level of nausea that decreases as the system recalibrates. Canal stimulation can cause nausea; horizontal canal stimulation provokes higher levels of nausea than does the posterior or anterior canal. Headache pain can have a relationship to dizziness and reflects a concomitant disorder of the CNS. Migraine headache and traumatic brain injury may trigger dizziness as a part of the abnormal processing of input through the brainstem.
Measurement of the interaction of the visual and vestibular systems can be accomplished by a variety of tests, with each test providing information that can be compared with the results of other tests to help determine the diagnosis.
The visual-ocular system is measured when looking for a disorder of the vestibular system and, when abnormal, can provide some clues as to where the damage to the system is located. If the motor component of the VOR is damaged, visual and vestibular controlled eye movements are abnormal. If the sensory component is damaged, visually controlled eye movements are usually normal, but vestibular-dependent eye movements are abnormal. Nystagmus is involuntary, rhythmic oscillation of the eye. Nystagmus can be a part of another brain dysfunction in addition to vestibular dysfunction. The nystagmus related to vestibular disorders usually has a particular direction, intensity, and shape. The characteristics can often offer clues to the pathologic condition. Pathologic nystagmus can be spontaneous, gaze evoked, or positional. Box 38-3 describes three types of physiologic nystagmus. Examination for pathologic nystagmus must include observation of the effects of fixation, eye position in the orbit, and head position.62
Disorientation is often described as feeling out of sync with the environment and is usually associated with other deficits, such as short-term memory loss, lack of concentration, and irritability, reflecting the intricate integration with the limbic, hippocampal, and reticular systems through the brainstem.8
Disequilibrium is reported as feeling unsteady or clumsy or as if one were swaying, and a history of falls should be obtained. Fear of falling is also of importance, and the efforts made to avoid a fall can shed light on the degree of relative vestibular spinal responses.
The side effects associated with use of some medications, anemia, hypoperfusion of the brain from postural hypotension, cardiac arrhythmia, endocrine disorders, and hypoglycemia may mimic a vestibular disorder. If the cause is a medical condition or use of medications, the symptoms should decrease when the appropriate condition is treated or when the medication is stopped.30
Elderly patients tend to show reductions in the gain of the VOR and smooth pursuit and increases in saccade latencies. Such age-related alterations might interfere with appropriate responses to fast head movements. Initially, reduction of the inhibitory system mediated through the cerebellum can compensate for decreased sensitivity. Over time, the combination of reduced peripheral sensitivity and central inhibition decreases the range in which the system can respond.51 Most falls in the elderly population result from an accidental slip or trip, often in association with an unsteady gait. Fewer than 10% of falls in the elderly population are a result of an acute attack of vertigo or dizziness. It appears that over time there is selective nonuse of vestibular cues during balance activities. Age-related increases in sway have been found for conditions that involved sway referencing of visual or somatosensory cues.
Video Nystagmography or Electronystagmography.
Capturing eye movements related to vestibular dysfunction can be done through use of video goggles (video nystagmography [VNG]) or by surrounding the orbit with electrodes (electronystagmography [ENG]). Most testing now is done using VNG. The angular velocity, amplitude, and frequency of nystagmus can be quantified. The ability to move the eyes quickly from one target to another intended target can be tested for accuracy and is called saccadic eye movement. When the target is missed on the first movement, there is a catch-up saccade used to move the eye directly to the target. Catch-up saccades are normal in some cases, such as for jumps made more than 20 degrees, because there is a consistent undershooting. Overshooting rarely occurs in normal individuals. Slow saccadic eye movements can be related to lesions in the central pathways that control their movement. This may be an abnormal motor response of the VOR, or it can be a part of many other degenerative or static lesions that can affect the same part of the system, such as MS or parkinsonian disorders. Saccadic eye movements can be induced by using dots or lights at different amplitudes and asking the client to move the focus quickly from target to target. Accuracy can be recorded using ENG to demonstrate the undershooting and overshooting resulting from disorders affecting control of eye movement.
The ability to follow a target through a trajectory, or smooth pursuit, is another ocular skill that reflects an intact visual-ocular system and vestibulo-ocular output. An acute vestibular lesion will cause an impairment of smooth pursuit, but accuracy should naturally return. Abnormalities of the smooth pursuit system can reflect disorders throughout the CNS. Cerebellar lesions are often a cause of abnormal smooth pursuit. Table 38-1 describes different functions of eye movements.
Table 38-1
Different Functions of Eye Movements
| Class of Eye Movement | Main Function |
| Vestibular | Holds images steady on the retina during brief head rotations |
| Visual fixation | Holds the image of a stationary object on the fovea |
| Optokinetic | Holds images steady on the retina during sustained head rotation |
| Smooth pursuit | Holds the image of a small moving target on the fovea or holds the image of a small near target on the retina during linear self-motion; with optokinetic responses, aids gaze stabilization during sustained head rotation |
| Nystagmus quick phases | Reset the eyes during prolonged rotation and direct gaze toward an oncoming visual scene |
| Saccades | Bring objects of interest onto the fovea |
| Vergence | Moves the eyes in opposite directions so that images are placed or held simultaneously on both foveae |
Smooth pursuit, or tracking of the target, is observed by recording the eye movement as the client attempts to follow targets at varying velocities. Use of a pendulum or computer-generated movement of lighted targets allows a sinusoidal tracking that can be measured by electrooculographic recording. Two measures are usually applied to the recordings. Pursuit gain compares the velocity of the eye to the velocity of the target. Pursuit phase lag refers to the difference in time between waveforms of the target and the eye movement. A person with normal tracking will predict the target motion and make the precise eye movements necessary to stay on target.70
Optokinetic nystagmus is the eye movement elicited by tracking a field instead of a target. The purpose of optokinetic nystagmus is to stabilize visually an entire moving visual field. Optokinetic nystagmus is recorded by ENG when a striped drum surrounding the client is moved at a constant velocity at a slow speed of 30 degrees and a fast speed of 60 degrees. Abnormalities of slow components parallel those of smooth pursuits and are related to disorders of the cortex, brainstem, diencephalon, or cerebellum. Abnormalities of the fast components reflect damage, as do abnormal saccades. ENG is used to measure eye movements caused by activation of the VOR and records spontaneous eye movements caused by alteration of the vestibular discharge rate.
The limitations of smooth pursuit and optokinetic nystagmus illustrate the important concept that reflexive sensorimotor systems have optimal operating ranges. Smooth visual pursuit functions are best for lowfrequency and slow head movements. Autonomic gravity receptors function best for static and very-low-frequency conditions. These and other reflexes overlap with the vestibular system for part of its operating range, but nonvestibular systems largely break down during quick head movements. The VOR is essential for gaze stabilization during high-frequency, high-velocity, and high-acceleration head movements.
The direction of the nystagmus reflects possible origin. Purely vertical or torsional spontaneous nystagmus suggests a central origin. Nystagmus caused by a central lesion can be in any direction (vertical, oblique, horizontal, rotational), may change direction as gaze direction changes, and is not suppressed by fixation of gaze.47
Bithermal Caloric Test.
Manipulation of endolymphatic flow in the semicircular canals by creating a temperature gradient is known as bithermal caloric stimulation. When warm air is infused in the external auditory meatus, the skin of the horizontal canal is heated, resulting in a temperature change that is transmitted to the horizontal semicircular canal. The endolymph closest to the canal wall is heated, causing it to become relatively less dense than the surrounding endolymph. The fluid movement that results from the heating of the endolymph deflects the hair cells in the ampulla, simulating head movement; the result is a nystagmus with the fast component moving toward the canal that was stimulated. Cold air or water has created movement in the opposite direction. The mnemonic COWS (cold opposite, warm same) describes the movement of nystagmus related to the temperature of the stimulus used. The caloric examination allows the clinician to evaluate each horizontal semicircular canal separately.41 VNG recordings during this stimulation can indicate abnormalities in different locations of the vestibular system and brain. Paresis can indicate damage anywhere from the end organ to the entry of the nerve root in the brainstem. A central disorder that affects the nerve root, such as MS or brainstem strokes, can cause paresis recording on caloric testing. Lesions of the cerebellum evoke heightened caloric responses and a suppression fixation deficit. Abnormalities in the characteristics of the nystagmus, such as vertical or oblique responses, are associated with CNS disorders.47 Fig. 38-10 describes three ways to invoke nystagmus.
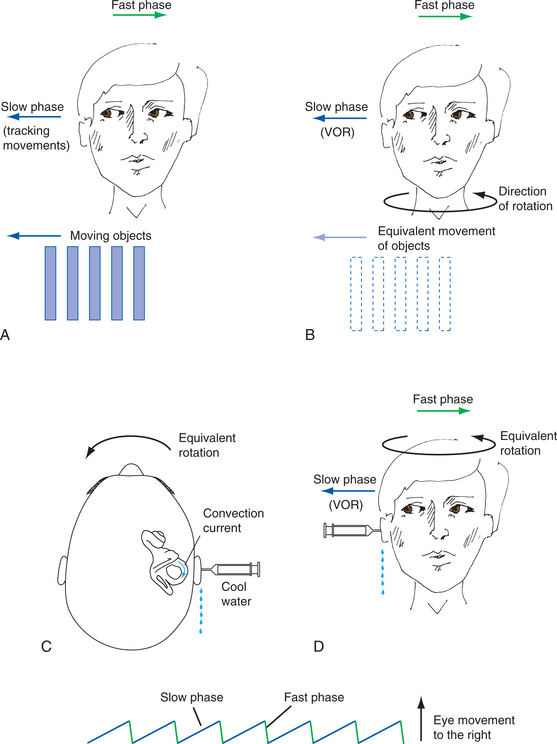
Figure 38-10 Three different ways to cause nystagmus. A, Movement of a series of objects to the individual’s right causes slow tracking eye movements to the right followed by rapid movements to the left. B, Rotation to the left is equivalent to the movement of objects to the right as perceived by the retina. C, Cool water or air placed near the horizontal canal via the external ear canal causes the same movement of the endolymph as head rotation in B. D, Electrical recording of horizontal nystagmus with its fast phase to the left. (From Nolte J: The human brain: an introduction to its functional anatomy, ed 5, St Louis, 2002, Mosby.)
Rotational Chair Testing.
Rotational testing of the horizontal semicircular canal is provided by use of a motorized rotational chair. Angular acceleration can be controlled and responses to angular acceleration measured. Persons who suddenly lose vestibular function on one side have asymmetric responses to rotational stimuli. Rotational stimuli are ideally suited for testing persons with bilateral peripheral vestibular lesions because both labyrinths are stimulated simultaneously and the degree of function is accurately quantified. As with lesions of the peripheral vestibular structures, lesions of the central VOR patterns can lead to changes in the gain of rotational-induced nystagmus. Cerebellar dysfunction will result in abnormal amplitudes and arrhythmia.
Subjective Visual Vertical or Horizontal.
The ability to recognize vertical and horizontal in a darkened room in relation to gravity indicates intact functioning of the utricle. This is known as subjective visual vertical or subjective visual horizontal. There is an ocular torsion that occurs after unilateral lesions of the vestibular system that is related to dysfunction of the otoliths. It is believed that this ocular rotation results in the inability, in the acute phase of vestibular damage, to accurately determine vertical or horizontal visually. This is tested in a darkened room using a light bar that is controlled by the client’s relation to vertical or horizontal. Clients are asked to determine when the bar is in either a horizontal or vertical position in relationship to gravity as they sit in a chair. The offset from the true position is often up to 15 degrees. With a compensated lesion, the accuracy improves to 4 degrees but does not ever appear to return to normal. This test will not detect bilateral utricular lesions.34,67
Vestibular-Evoked Myogenic Potentials.
Vestibular-evoked myogenic potentials (VEMPs) may be another way to test otolith function in the clinic. The VEMP is generated by synchronous discharges of groups of muscle cells innervated by a single motor unit, or myogenic potential; this response is found in the sternocleidomastoid as part of the vestibulocolic reflex. It appears that the VEMPs arise from stimulation of the saccule, and the saccule is very sensitive to sound. Click-sensitive neurons in the vestibular nerve are the same neurons that respond to tilts. VEMP testing involves generating a standard sound set and looking for inhibition of the sternocleidomastoid recorded via electromyography (EMG). When one side of the system is dysfunctional there will be a corresponding decrease in EMG activity in one of the sternocleidomastoid muscles. It appears at this time that the VEMP test can provide information in addition to the tests of the semicircular canals to isolate dysfunction in the otolith.
Posturography.
Testing of VSRs is performed by posturography, which evaluates somatosensory and visual influences on posture and equilibrium. The clinical applicability of computerized dynamic posturography (CDP) assessment varies with the chief complaints of the individual. CDP has the most clinical value in persons with symptoms of unsteadiness, disorientation, and vertigo in whom the history and physical examination do not suggest an obvious localized cause.44 In the motor control test the individual’s postural responses are recorded during displacement of the support surface using EMG recordings that reflect the activation of the segmental, spinal, and long-loop response pathways. Prolonged motor control test latencies are evidence for abnormality in any one or a combination of components making up the long-loop automatic system and therefore are strong indications for nonvestibular, spinal cord, brainstem, and subcortical involvement.14,54,55 The sensory organization test, the second component of the CDP, defines six different sensory conditions in which an individual’s postural sway is measured. These six conditions vary the amount and accuracy of the sensory information (somatosensation, vision, vestibular input) available to maintain balance.53 Vestibular dysfunction patterns are seen in virtually all persons with bilateral peripheral vestibular deficits. These persons are able to maintain balance when the vision or somatosensation is available, but they free fall repeatedly when conditions require dependence primarily on vestibular input.7 Similar vestibular dysfunction patterns are seen in persons with peripheral vestibular lesions and CNS lesions affecting central pathways of the vestibular system. Persons who compensate after a unilateral lesion will have a normal sensory organization test result 2 to 4 weeks after the initial insult.28 An abnormal preference for vision, either alone or in combination with vestibular dysfunction, is most frequently observed in persons with posttraumatic vertigo and unsteadiness. Fig. 38-11 shows sensory conditions during the sensory organization test.
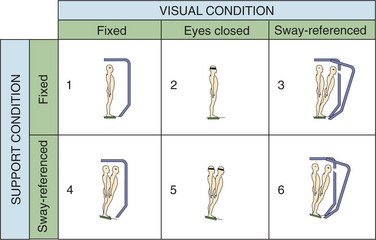
Figure 38-11 Diagram of the sensory conditions related to the sensory organization test, a component of posturography. (Courtesy NeuroCom International, Clackamas, OR.)
Measurement of movement strategies (ankle, hip, stepping) used to maintain balance is the third component of the CDP. Inappropriate use of ankle movements during large-amplitude sway might be an abnormal adaptation used to minimize head movements and associated stimulation of the vestibular system.53
Electrocochleography.
Electrocochleography is used to record elevation of endolymphatic fluid pressure. It is the recording of acoustically evoked electrical potentials arising from the cochlea and the eighth cranial nerve. An electrode is placed on the tympanic membrane or on the wall of the external ear canal near the tympanic membrane.
Imaging.
When a central cause is suspected based on clinical findings and vestibular testing, magnetic resonance imaging (MRI) with contrast is indicated. Central brainstem lesions, such as stroke, trauma, or MS, can be identified. Neoplasms, including schwannomas, meningiomas, and metastases, can also be identified. Vascular loop or sling can be identified on MRI. Inflammatory lesions can be identified with MRI, including labyrinthitis and inflammatory lesions of the eighth cranial nerve.
TREATMENT.
Treatment of vestibular disorders reflects the spectrum of etiologies. A multidisciplinary approach is critical in the management of symptoms to determine the role of medications, surgical interventions, and therapy. The need for lifestyle changes should be carefully evaluated related to the effect of other interventions.
When symptoms are due to a peripheral vestibular lesion, functional recovery will begin within 2 days to 4 weeks through the adaptive mechanism of the brain.70 If the symptoms are severe, sedatives may be given, ideally for the first 24 hours only. Vestibular rehabilitation should begin within the first 3 days if possible. Treatment of recurrent dizziness depends on the nature of the underlying disorder. The goal is to eliminate the frequency and duration of the abnormal sensation of motion and the symptoms of nausea, vomiting, and anxiety.4
If the central adaptation process is inadequate, vestibular suppression can be helpful but must be used judiciously. Benzodiazepines, antihistamines, or anticholinergic agents are used frequently. Serotonin 5-HT receptor antagonists have been reported to be of some use.
Surgical intervention is considered when the symptoms are unrelenting and the underlying condition is determined but is unresponsive to other medical measures. Local application of gentamicin to selectively destroy the end organ is often used rather than ablative surgery. Surgery is indicated with neoplasia (see Chapter 30) perilymph fistula, persistent benign paroxysmal positional vertigo, hydrops, and vascular loop. These conditions are covered later in this chapter.46
PROGNOSIS.
When there is a unilateral lesion in the peripheral vestibular system and the CNS is intact, recovery of function is possible. The recovery related to the static imbalance, the difference in the tonic firing rate within the vestibular nuclei, is spontaneous. The spontaneous nystagmus at rest resolves, and there is no longer a sensation of movement at rest. The recovery of dynamic disturbances, reflecting the relationship of the coupling of the two sides of the system during movement, requires visual experience. The visual experience allows the brain to recognize the error of the system through the retinal slip, causing oscillopsia that occurs immediately after the lesion. It is this sensory mismatch provided by the visual system that allows the CNS to recognize the need for adaptation and drives the readjustment of motor reflexes.44 There is a limitation to this recovery, and the system is able to recover function related to slow to moderate movements only. The VOR should return to near normal within 2 to 6 weeks for slower movements, so that motion of the head during typical activities no longer disrupts vision or causes nausea. However, there will continue to be an abnormal reaction to rapid head turns. Recovery is minimized when the response of the individual experiencing the symptoms is to avoid the provoking motions and the CNS is never given the opportunity to adapt to the asymmetric firing patterns of the peripheral vestibular system.
Complete bilateral loss is relatively rare; more often there is decreased function in both sides, often to a different degree on each side. Rehabilitation can provide adaptation to whatever degree possible and compensation for the remaining loss of function.45 In the instance of complete bilateral dysfunction, substitution of the other intact sensory inputs for balance is required. The use of visual and somatosensory inputs to substitute for loss of vestibular function is possible when the environment provides adequate cues, as in well-lighted environments and firm, level surfaces. Substitution of small saccades in the direction opposite to head rotation can augment inadequate gain. Smooth pursuit accuracy can improve, and predictive strategies can improve gaze stability. More accurate use of spatial localization is reflected in the ability to imagine the location of stationary targets.70
The VSRs are slower to return, and the individual may continue to experience instability when turning quickly or walking on uneven surfaces or in the dark for weeks or months after the insult.
Recovery rate in a central vestibular system disorder causing dizziness or disequilibrium depends on the nature of the lesion and the concomitant neurologic dysfunction. If vertigo is part of a progressive disease, the prognosis is less optimistic than if it is part of a transient disorder, such as a minor vascular incident. A disease such as MS can cause episodes of symptoms and progressive dysfunction with poor adaptation and compensation because of the other damage within the CNS.8 In a disorder of the central as well as the peripheral vestibular system, as in head injury or multisensory disorders, the recovery is significantly reduced.58 Recognizing and treating the individual components will successfully enhance the recovery.
Benign Paroxysmal Positional Vertigo
Episodic, intense vertigo related to head position is most often a benign disorder called benign paroxysmal positional vertigo (BPPV), also known simply as benign positional vertigo (BPV). It is considered a benign condition, since it is not the sign of a disease process but a mechanical disorder of the labyrinths.
Incidence and Risk Factors.: BPPV is the most common cause of vertigo seen by otolaryngologists, representing 20% to 40% of patients with peripheral vestibular disease. The incidence is difficult to estimate given the benign, typically self-limited course of the disease. It is thought to vary from 10 to 64 cases per 100,000 persons. Affected women outnumber men by a ratio of 1.6: 1. Involvement in more than one canal is found in 20% of cases. Spontaneous remissions are common, but the disorder can recur in up to 40% of the cases. The condition may trouble the individual intermittently for years, but in this condition a close examination of potential causes will often identify an underlying disorder, and recurrences decline when the disorder is managed. Increased fluid pressure within the labyrinth may dislodge otoconia (see discussion of endolymphatic hydrops below). Migraine-induced ischemia may be responsible for the release of otoconia. Head trauma or infectious or inflammatory disorders may precede the onset by months or even years. Adverse life events are reported often prior to the onset of BPPV. Aging, perhaps because of the increase of dehydration, can increase incidence.18
Clinical Manifestations.: Typically, a person with BPPV will complain of brief episodes of vertigo precipitated by rapid head movement in a specific direction. The vertigo follows getting into bed and lying down. Often the first report is that of waking up suddenly in the night with the room spinning. Bending, looking up to take an object off a shelf, tilting the head back to shave, getting a haircut, or turning the head rapidly while backing up a car can trigger symptoms. These single bouts of vertigo are frequently clustered in time and separated by remissions lasting months or more. Lightheadedness, worsened by head movement, is common and balance problems develop over time when the crystals are not removed from the canals. Symptoms occur suddenly and typically last 20 seconds, but not more than a minute. The subjective impression of attack duration reported is frequently longer than the actual period of dizziness.25,36 Box 38-4 gives typical complaints related to BPPV.
Pathogenesis.: The otoconia in the otolith can become loose, clump together, and form densities known as canaliths. Canaliths move through the fluid and become problematic when they float into the semicircular canals. There is potential for them to go into any of the three canals, but most often they will move into the posterior canal because of the relationship of the posterior canal to the otolith. The posterior canal is placed in the vertical position when the body is supine and the head is extended beyond neutral and rotated 45 degrees to the same side. The canaliths drift out of the otolith through the opening to the posterior canal and begin moving through the endolymph in the semicircular canal, causing drag on the endolymph and creating cupular deflection, or move- ment of the ampulla. This movement of the cupula is interpreted in the brain as movement of the head. There is an intense sensation of rotation. As the canaliths reach the bottom of the semicircular canal, they slow down and stop moving, stimulation of the vestibular nerve ceases, and the sensation of movement stops. This typically takes about 15 to 60 seconds. Canalithiasis of the posterior semicircular is the most frequent cause of BPPV. Box 38-5 describes the phenomenon of canalithiasis.
A less common variant of BPPV is cupulolithiasis. It is believed that the otoconia can become adherent to the ampulla. In this instance there is an inappropriate deflection of the hair cells with movement, causing vertigo and nausea. The symptoms will begin immediately with movement of the head in the provoking position and will persist as long as the head is held in the position. The symptoms may decrease slightly because of adaptation of the CNS and therefore may mimic the fatigue of canalithiasis.38
The horizontal or semicircular canal has been identified as the affected structure in about 17% of cases. Cupulolithiasis, either occurring independently or in combination with canalithiasis, is more likely to be involved in the etiology of lateral canal BPPV than is the case for posterior canal BPPV.
MEDICAL MANAGEMENT
The diagnosis of BPPV is made based on a suggestive history and report of current clinical manifestations as described above. The classic eye movements are found when performing the Dix-Hallpike maneuver. The maneuver is repeated with the head in the opposite direction.
Dix-Hallpike Maneuver.
Diagnostic criteria for BPPV include whirling vertigo induced by a specific head position or movement. There is a burst of positioning nystagmus that is rotary with latency of symptoms on placing the head in a provoking position; the nystagmus is fatigable, that is, it diminishes with repeated positioning and lasts less than 60 seconds.
The Dix-Hallpike maneuver, or passive movement of the head from the upright position to one with the head hanging extended and rotated to 45 degrees, is the standard test performed to establish the diagnosis of BPPV. When the head is in this position, the clinician observes the eyes for evidence of nystagmus. It may take at least 20 seconds for the nystagmus to occur, and the nystagmus should fatigue within 60 seconds. The nystagmus occurs when the affected ear is placed down. Fig. 38-12 shows the testing positions. The pattern of response is torsional nystagmus that is identified by rotation of the superior poles (the center, vertical component of the iris) toward the involved ear. This is accompanied by an upbeat nature to the nystagmus. There is a latency of onset of nystagmus. Duration of nystagmus is less than 1 minute. Vertiginous symptoms are reported as a sensation that the room is spinning, or the individual is spinning or falling out of control. Symptoms often recur, and the nystagmus beats in the opposite direction on return of the head to the upright position. There are also reports of BPPV affecting the anterior semicircular canal, but they are found infrequently. The nystagmus expected in cases of anterior canal BPV would be torsional, such that the superior poles would beat to the right but would have a downward nature.18 Although a positive test is pathognomonic for BPV, a negative test indicates only that BPV is not active at that movement.
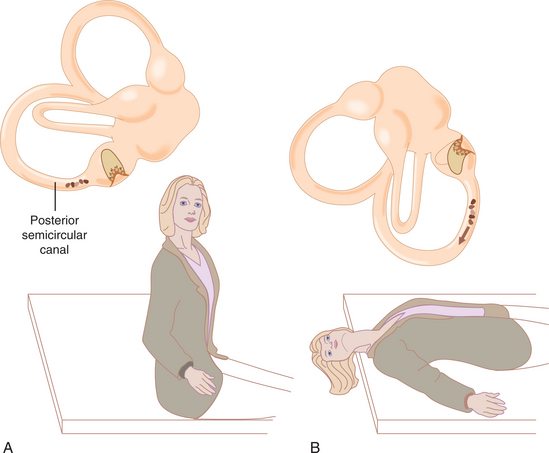
Figure 38-12 The Dix-Hallpike maneuver. A, Starting position with head rotated toward the side to be tested. B, Lowering the patient’s head backward and to the side allows debris in the posterior canal to fall to its lowest position, activating the canal and causing eye movements and vertigo. (From Lundy-Ekman L: Neuroscience: fundamentals for rehabilitation, ed 3, Philadelphia, 2007, Saunders.)
To test for BPPV in the horizontal canal, the head is first brought to the supine position resting on the examining table or is slightly flexed. The head is then turned rapidly to the right so that the patient’s right ear rests on the table. The nystagmus with lateral canal BPV is horizontal. It often begins with a shorter latency, increases in magnitude while maintaining the test position, and is less susceptible to fatigue. Dizziness is likely to occur with head rotation in both directions, but the direction that causes the most intense horizontal nystagmus or is the most symptomatic is considered the side involved. If a brief period of dizziness is experienced during the Dix-Hallpike maneuver as the head is approaching the full supine position, and there is no evidence of posterior or anterior canal BPPV, there is a possibility of horizontal canal involvement, which should be further investigated. Horizontal canalithiasis can also occur, producing symptoms provoked by rolling in the supine position. Horizontal nystagmus may occur in either direction but will be greatest when rolling the head to the involved side.38,57
Another form of positional dizziness that must be ruled out in making the diagnosis of BPPV is central positional vertigo. Nystagmus that is sustained and not suppressed by visual fixation reflects a central lesion. These findings can be indicative of ischemia of the pontomedullary brainstem or another part of the central vestibular pathway. Cerebellar involvement will cause dizziness in positions of supine, head rotated left with extension, and head rotated right with extension. Pure downbeating nystagmus in either the sitting or supine position may indicate an infratentorial disorder. This can be related to vascular insults such as Arnold-Chiari malformation, stroke, or subdural hematoma. MS lesions are common in this area and are associated with the progressive forms. Unilateral lesions, perilymphatic fistula, superior semicircular canal dehiscence, or middle ear problems can cause positional dizziness and are discussed later in this chapter. Intermittent dizziness that is made worse with position changes could also be part of the aura of migraine.
It should also be noted that it is typical to experience dizziness with the head pitched backward, as it puts the otoliths at an angle that is not a part of daily activity. Orthostatic hypotension can also create a brief episode of dizziness when going from supine to standing.
TREATMENT.
Several approaches have been developed to treat persons with BPPV, including maneuvers or positions that attempt to dislodge the provoking substances and move them through the canal system back to the otolith, known as canalith repositioning procedures or the libratory maneuver. Individuals can also be instructed in exercises (e.g., Brandt’s) to try to habituate to the stimulus. The use of gravity to move canalith debris out of the affected semicircular canal and into the vestibule is a safe and reliable method. Even patients who have been symptomatic for years can be cured. The posterior and superior canal maneuver begins with placement of the head into the Dix-Hallpike position that evokes vertigo. Lateral or horizontal canal BPPV responds to positional procedures that involve head rolling with the horizontal canal in the plane and allow gravity to pull the otoconia through the horizontal canal. The maneuvers are repeated until no nystagmus is elicited. In some cases this may take several repositioning sessions. In the case of bilateral BPPV, the canals cannot be treated on the same day, as the maneuvers move the crystals in the opposite direction. Oscillation to the mastoid process can be used in conjunction with the repositioning maneuvers but should be avoided in patients who have had retinal detachment or who might be susceptible to such a detachment because of high myopia. A low dose of meclizine or a benzodiazepine can be given 1 hour earlier if the patient is unusually anxious or susceptible to nausea and vomiting with vestibular stimulation. After undergoing one of the canalith repositioning maneuvers, patients are instructed to avoid bending over and are told to sleep with the head elevated at least 30 degrees for the next 1 to 2 days. Because BPPV can be related to conditions that also are responsible for abnormal vestibular function, exercises stimulating the vestibular system should follow treatment of BPPV once it has been cleared.38
The canalith-repositioning maneuvers are highly effective in the treatment of BPPV in the vast majority of patients. Chairs that are specially designed for precise positioning or repositioning are in use in some centers. Surgical procedures have been described, although their current use is rare. These procedures include singular neurectomy, which is rarely used due to the potential for sensorineural hearing loss, and posterior semicircular canal occlusion. Posterior semicircular canal occlusion has resulted in the abolition of symptoms but is also rarely done.
Infection
Acute unilateral vestibulopathy, or vestibular neuritis, is the second most common cause of vertigo. Viral infection is common and usually affects the vestibular nerve unilaterally. (See section above on unilateral lesions.) Vestibular neuritis can be a partial unilateral vestibular lesion, and this partial lesion can affect the superior division of the vestibular nerve, which includes the afferents from the horizontal and anterior semicircular canals.24
Incidence and Etiology.: Incidence is 3.5 in 100,000. Viral infections are common between the ages of 30 and 60 years, with a peak for women in the fourth decade and men in the sixth decade.25 The etiology of the disease is multifactorial. Viral pathogens include mumps, rubella, herpes simplex virus type 1, cytomegalovirus, and Epstein-Barr virus. Enteroviruses are among the other rare viral causes. Onset is often preceded by a systemic viral illness, such as an upper respiratory tract infection or gastritis. The illness may precede the vestibular dysfunction by up to 2 weeks but can happen within the course of a bout of cold or flu. When illness such as measles, mumps, or infectious mononucleosis is the source, hearing loss may accompany the vestibular symptoms.
The use of antibiotics in general has decreased the incidence of bacterial infections affecting the vestibular system. However, infections still do arise and can be introduced into the vestibular apparatus through the various fluid systems involved or through breakdown of the bony labyrinth.
Pathogenesis.: Histopathologic studies have suggested involvement of the superior vestibular nerve and vestibular ganglion, often with little or no involvement of the actual end organ. This is true of vestibular neuritis, in which there is no cochlear involvement. However, many viruses do damage throughout the labyrinth and cochlea. Typically, in vestibular neuritis, the end organ is filled with lymphocytes. Intracytoplasmic particles have been found in the vestibular ganglia. These particles are thought to be dormant forms of a virus that may produce infection, with resultant inner ear disease.5
Persons with bacterial meningitis develop labyrinthitis when bacteria enter the perilymphatic space from the cerebrospinal fluid by way of the cochlear aqueduct or the internal auditory canal. Some bacterial infections result in biochemical irritation of the membranes through a toxic reaction. Both congenital and acquired syphilitic infections produce labyrinthitis as a latent manifestation.
Clinical Manifestations.: Unilateral vestibular neuronitis causes sudden onset of rotatory vertigo, spontaneous horizontal nystagmus, nausea, and vomiting. Immediately after the onset of unilateral vestibular hypofunction there is intense disequilibrium. There are profound disturbances of position and motion perception. There is a false sense of angular motion (i.e., rotation). With the eyes closed, there is an illusion of spinning, or sense of motion of the body turning on its long axis toward the involved side. When the eyes are open, the illusion is of spinning of the environment in the opposite direction. There is a tonic ocular tilt reaction consisting of head tilt, conjugate eye torsion, skew deviation, and lateropulsion, seen as an abnormal weight shift toward the side of the lesion. This is due to the otolithic hypofunction on the side with the lesion.53,54 This sensation of movement at rest should resolve within 2 days.
MEDICAL MANAGEMENT
The VOR becomes abnormal with unilateral hypofunction. There is loss of gaze stabilization with head movement. Movement of the head then causes blurring of vision, resulting in dizziness and loss of ability to use visual cues to maintain balance. EMG testing shows a unilateral hypofunction.
A spontaneous mixed torsional and horizontal nystagmus is often present immediately after unilateral hypofunction. The slow phases are directed toward the side of the lesion, and the quick phases are to the intact side. As the eyes are observed, it is the quick phases that are apparent, and therefore the nystagmus appears to be moving away from the side with the lesion. The nystagmus is suppressed by gaze fixation, or looking at a static object.54 Peripheral vestibular nystagmus is usually mixed torsional and horizontal in a fixed direction opposite the side of the lesion.
In severe unilateral vestibular hypofunction, passive head shaking for about 20 seconds will elicit a quick burst of nystagmus directed toward the intact end organ. The nystagmus can be suppressed with visual fixation.
TREATMENT.
The main groups of drugs used for symptoms of acute vertigo include antihistamines, anticholinergic agents, antidopaminergic agents, steroids, and antivirals such as acyclovir.
Polyneuritis of the seventh and eighth cranial nerves, known as Ramsay Hunt syndrome, can be the result of herpes zoster, causing perivascular, perineural, and intraneural infiltration. Other viruses are suspected but have yet to be positively identified. The cortical vestibular projection fibers can be affected by herpes zoster encephalitis, which has a predilection for the temporal lobe.
Recovery of function and resolution of dizziness is accomplished through a program to facilitate the vestibular system. Recalibration, or adaptation of the system, comes by facilitating the integration of somatosensory input and recovery of normal postural responses. See Special Implications for the Therapist: Vestibular Dysfunction in this chapter.
PROGNOSIS.
The symptoms slowly resolve over 6 weeks to 3 months, but there can be persistent complaints of imbalance, motion intolerance, and headache related to decrease in the natural motion of the head. Over time, decreased functional use of the vestibular mechanism develops and is related to excess caution on uneven surfaces, dizziness that is more easily provoked, an overall decrease in activity level, or avoidance of activity that may cause dizziness.
Endolymphatic Hydrops and Menière’s Syndrome
Definition and Overview.: Endolymphatic hydrops is a disorder relating to the membranous inner ear as a consequence of the overaccumulation of endolymph compromising the perilymphatic space. There is lack of absorption of endolymph in the endolymphatic duct and sac and fluid backs up into the system. Menière’s syndrome is the most common form of endolymphatic hydrops and is characterized by episodic vertigo; fluctuating, sensorineural hearing loss; sensation of fullness in the ears; and tinnitus. Tinnitus is an abnormal sound in the ear usually described as a ringing, buzzing, clicking, or crackling sound. It is often associated with other abnormal sensations, such as fullness of the ear. Vertiginous attacks are the most debilitating symptom, with intervals of hours to days. Acute attacks can be superimposed on a gradual deterioration in sensorineural hearing in the involved ear, typically in the low frequencies initially. Over time, a reduction in responsiveness of the involved peripheral vestibular system can occur.5 Fig. 38-13 shows the fluid mechanism of the inner ear, including the endolymphatic sac.
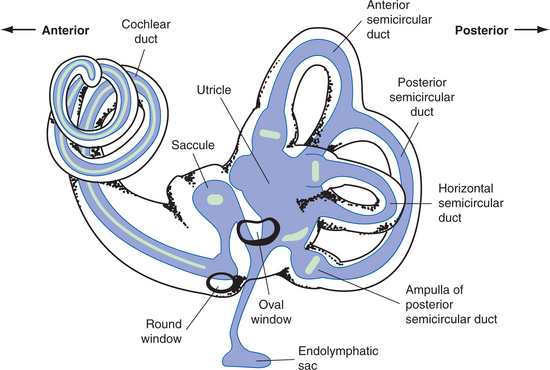
Figure 38-13 Membranous labyrinth as seen through an outline of the bony labyrinth. The endolymphatic sac provides the absorption of endolymph. This is the probable site of dysfunction in hydrops, causing a backup of fluid within the membranous labyrinth. (From Nolte J: The human brain: an introduction to its functional anatomy, ed 5, St Louis, 2002, Mosby.)
Incidence and Etiologic and Risk Factors.: Female Caucasians are most prone to the disorder. However, diagnostic criteria have varied across epidemiologic studies. These vary from 157 per 100,000 persons in England to 46 per 100,000 in Sweden and 7.5 per 100,000 in France. The peak incidence is in the 40-to 60-year-old age group, with a nearly equal female to male ratio (1.3: 1). Estimates of symptoms arising in the opposite ear vary from 2% to 50%. Whether the variability in prevalence rates is caused by differences in environment, genetics, or diagnostic criteria is unclear.63
Familial occurrence of Menière’s syndrome has been reported in 10% to 20% of cases. Genetic inheritance plays a role. The mode of transmission appears variable; however, an autosomal-dominant mode of inheritance with increased penetrance has been documented.
The incidence of Menière’s syndrome is greater in individuals with certain genetically acquired major histocompatibility complexes. Specifically, human leukocyte antigens B8/DR3 and Cw7 have been associated with Menière’s syndrome. The etiology for disease in these individuals may be autoimmune.
The cause of endolymphatic hydrops is multifactorial and may be related to fibrosis, atrophy of the sac, obstruction of the endolymphatic duct, infection, or the vascularity in the region of the inner ear. It can also be caused by otosyphilis, or involvement of the inner ear in collagen vascular diseases. Immune responses are likely within the complex, including the endolymphatic sac, related to allergic reactions and histamine. There may be a predisposing viral infection that may cause the inner ear to be more susceptible to changes in thyroid, sodium, or hormone dysfunction. The deficit may also be related to overproduction of endolymph by the stria vasularis.19 Posttraumatic endolymphatic hydrops can be observed following a blow to the head, a fall, or flexion or extension injury sustained in an automobile accident.
Pathogenesis.: Although the pathophysiologic mechanism involves a disruption of homeostasis of inner ear fluid, the mechanism and the pathophysiology of the symptoms are uncertain. Endolymphatic hydrops may be an epiphenomenon rather than directly responsible for the symptoms. Deficits are related to the volume and pressure changes within closed fluid systems. The increase in the volume of endolymph causes the membranous labyrinth to progressively dilate until the wall makes contact with the stapes footplate and the cochlear duct fills the entire scala vestibuli, causing both vestibular and cochlear dysfunction. Fig. 38-14 shows the cross view of the canal and the changes that can occur with increased pressure. Distension of the otoliths can put pressure on the ampulla, creating the sensation of spinning that is characteristic of acute unilateral dysfunction. The mem- brane that separates the endolymph from the perilymph may rupture, and there is a leakage of potassium into the endolymph that may result in palsy of the vestibular nerve fibers.
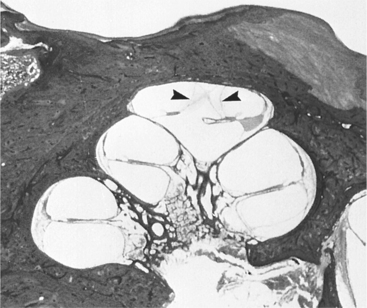
Figure 38-14 Cross-section of human cochlea demonstrating endolymphatic hydrops in a patient with Menière’s syndrome. Note the distension of Reissner’s membrane into the scala tympani in the apical turn of the cochlea (arrowhead). (From Cummings CW, Haughey BH, Thomas R, et al: Cummings otolaryngology: head and neck surgery, ed 4, St Louis, 2004, Mosby.)
Pathologic studies of the human sac in the hydrops patient have recorded ischemia and fibrosis around the endolymphatic sac. Alterations in the size of the endolymphatic duct and sac along with reductions in the lining of these structures have also been noted in both the diseased and nondiseased inner ears. This supports the theory that an abnormal endolymph drainage system may predispose individuals to the future development of Menière’s syndrome. Alterations in calcium and chloride metabolism may alter the osmotic gradients in the endolymph, causing accumulation of endolymph as well as the loss of endocochlear potentials necessary for hair cell function. These ionic disturbances may be caused by ischemia resulting from changes in local vasculature. A common vascular mechanism for migraine headaches and Menière’s disease has also been proposed. A hydropic state may leave those persons sensitive to stresses to the inner ear.63
Clinical Manifestations.: The typical attack of hydrops related to Menière’s syndrome is experienced as an initial sensation of fullness of the ear, a reduction in hearing, and tinnitus. This is usually followed by a rotational vertigo, postural imbalance, nystagmus, and nausea. The vertigo may last from 30 minutes to 24 hours. The symptoms abate over time, and the individual regains the ability to maintain balance. However, there may still be some sense of disequilibrium. Hearing slowly returns, but over time there may be a permanent loss of hearing. Tinnitus is common in hydrops and is commonly described as a low-pitched roaring or similar to a seashell noise. Box 38-6 gives some of the characteristics consistent with the diagnosis of Menière’s syndrome.
MEDICAL MANAGEMENT
The complaints of vertigo, hearing loss, and tinnitus do not automatically confirm a diagnosis of endolymphatic hydrops or Menière’s syndrome. The definitive vertiginous attack is sudden in onset with nausea and vomiting, lasts 20 minutes, but abates by 24 hours. Typically, any movement during an attack aggravates the vertigo. The presence of neurologic signs or symptoms such as syncope, visual aura, and motor weakness suggest another diagnosis. Disorders that can present with similar symptoms include migraine, acoustic neuroma, perilymphatic fistula, dehiscence of the superior semicircular canal, labyrinthitis, autoimmune inner ear disorder, and MS.
An audiogram, or test of hearing, typically demonstrates low-frequency hearing loss on one side. Perceived hearing loss can be difficult to verify without audiometry, especially during an exacerbation of tinnitus or aural pressure. Improvement in audiograms can reflect better control of fluid, and decreases in hearing are suggestive of progressive disease.63
Electrocochleography may provide objective evidence of the presence of endolymphatic hydrops in the presence of large summating potentials. However, the diagnostic utility of the test is limited by the variability of the ratio both in individuals with hydrops and in normal individuals. The results should be used in the face of other subjective history.
TREATMENT.
Diuretics can control vertigo and stabilize hearing in more than half of the individuals with reported hydrops. Restricting salt, caffeine, alcohol, and nicotine is routinely suggested. The goal of salt restriction and diuretics is to reduce endolymph volume by fluid removal and reduced production.
Histamine is used in the belief that hydrops are a result of strial ischemia or immune modulated disorders. Other agents used include the papaverine analog eupavarine, nicotinic acid, adenosine triphosphate, and dipyridamole. Antivertiginous medications, antiemetics, sedatives, antidepressants, and psychiatric treatment have been reported to be beneficial in reducing the severity of the vertigo and vegetative symptoms.
Corticosteroid infusion of the middle ear via a transtympanic route is under investigation as hearing and vestibular conservation therapy for endolymphatic hydrops. The presumed site of action is in the inner ear. The rationale for the treatment is based on the apparent role of autoimmune and inflammatory injury in the etiology of hydrops.18
Intratympanic gentamicin is used in the treatment of chronic, unrelenting unilateral hydrops. Gentamicin typically results in a reduction of vestibular function in the treated ear. Surgical treatment to restore normal endolymph volume includes endolymphatic decompression procedures. Other procedures designed to ablate vestibular function in the affected ear without damage to the cochlea include vestibular neurectomy, intratympanic gentamicin, cryosurgery, ultrasound, and cochlear dialysis.18
PROGNOSIS.
The natural history of endolymphatic hydrops and Menière’s syndrome is highly variable. Clusters of attacks may be separated by periods of long remission. Balance function between attacks can be normal, although a sense of disequilibrium often persists later in the course of the disorder. Menière’s syndrome initially affects only one ear. The attacks increase in frequency in the first years and then decrease. The frequency of bilateral disease ranges from 2% to 78%, with an average incidence of 45%. If bilateral involvement has not occurred within 5 years of onset of disease in the first ear, then there is less likelihood of developing bilateral involvement.
The hearing loss in Menière’s syndrome is a fluctuating, low-frequency sensorineural loss early in the clinical course. Eventually, the loss becomes irreversible, often progressing in severity with involvement of higher frequencies and loss of speech discrimination.
An estimated 2% to 6% of patients with Menière’s syndrome of long duration can experience “drop attacks” known as otolithic crisis of Tumarkin, characterized by being abruptly thrown to the ground without loss of consciousness and with little or no vertigo.29
Perilymph Fistula
Definition and Overview.: Perilymph fistula, an abnormal communication of the inner and middle ear spaces, can cause vertigo. Fistulas commonly occur at the round and oval windows of the middle ear. Attempts to identify the prevalence and characterize auditory and vestibular symptoms have been inconclusive. Some studies report vestibular symptoms as the major presenting complaint, whereas others indicate hearing loss equal to or more common than balance-related symptoms. It is believed that most fistulas result from congenital malformations or prior ear surgery. Damage can also result from pressure applied via the external ear, via the eustachian tube, or by an increase in the pressure of the cerebrospinal fluid.9
Clinical Manifestations.: Characteristics of perilymph fistula include easing of symptoms at rest and increases with activity, including the Valsalva maneuver. Barotrauma, violent exercise, heavy lifting, or even sneezing may cause a fistula. Other mechanisms include head trauma, explosive blast, or barotrauma. Sensorineural hearing losses vary from an isolated high-frequency loss to a low-frequency or flat one. Speech discrimination test results are not characteristic. Both the pure-tone threshold and speech discrimination scores have been noted to fluctuate. Isolated mild conductive losses have been noted. Vestibular symptoms are also variable and include episodic incapacitating vertigo, equivalent to a Menière’s attack, positional vertigo, motion intolerance, or occasional disequilibrium. Disequilibrium after increases in CSF pressure (e.g., nose blowing, lifting), called Hennebert’s sign, has been noted, as has vertigo after exposure to loud noises, which is known as Tullio’s phenomenon.
MEDICAL MANAGEMENT
The inability to reliably predict the presence of a fistula before surgical exploration, as well as the lack of standard criteria for recognizing a fistula intraoperatively, have resulted in confusion and even doubt as to the existence of symptomatic fistulas. Because fistulas have been identified intraoperatively and their repair has resulted in symptomatic and objective improvement, this diagnosis must be kept in mind in the evaluation of the vertiginous patient.
Audiologic tests considered to be helpful in the diagnosis include electrocochleography. This demonstrates a larger summating potential due to endolymph/perilymph disequilibrium. However, the test is not sensitive or specific for perilymph fistula.
Results of vestibular testing are nondiagnostic. The most consistent abnormality seen is a unilateral reduced caloric response in the affected ear.
A fistula test is done by introducing positive pressure into the suspected ear, either by rapid pressure on the tragus, compression of the external canal, or use of a pneumatic otoscope, while observing the eyes. A positive fistula sign consists of conjugate contralateral slow deviation of the eyes followed by three to four beats of nystagmus. Vertigo is usually elicited at the same time. Measuring body sway during pressure on the tympanic eardrum can help make the diagnosis.
TREATMENT.
Recommendations with suspected inner ear fistula include head elevation during bed rest, laxatives to reduce the risk of increased intracranial pressure, and monitoring of both hearing and vestibular function. In those instances in which hearing loss worsens or vestibular symptoms persist, surgical exploration is warranted.
Intraoperative identification of a fistula, regardless of criteria used, is reported in about 50% of individuals explored. At the time of surgery, the oval and round windows are patched with tissue, such as blood clot, fat, fascia, or absorbable gelatin sponge.18
PROGNOSIS.
The outcome of surgical repair is variable. An appropriate surgical candidate is probably the most significant factor in outcome. Reduction in vestibular-related complaints has been reported in more than 50% of surgeries. Hearing is improved about 25% of the time.
Superior Semicircular Canal Dehiscence Syndrome
Definition and Overview.: Superior semicircular canal dehiscence syndrome is a syndrome of vertigo and oscillopsia induced by loud noises or by stimuli that change middle ear or intracranial pressure in patients with a dehiscence of bone overlying the superior semicircular canal. Tullio’s phenomenon (eye movements induced by loud noises) or Hennebert’s sign (eye movements induced by pressure in the external auditory canal) develop and often there is chronic disequilibrium.
Pathogenesis and Clinical Manifestations.: The dehiscence creates a “third mobile window” into the inner ear, thereby allowing the superior canal to respond to sound and pressure stimuli. The evoked eye movements in this syndrome typically align with the affected superior canal. Loud sounds, positive pressure in the external auditory canal, and the Valsalva maneuver can cause characteristic eye movements. A larger length of dehiscence overlying the superior canal (5 mm or greater) can lead to dysfunction in the affected canal when evaluated by responses to rapid head movements in the plane of the superior canal. Visual fixation can suppress the evoked eye movements.20 Fig. 38-15, A, shows the schematic representation of superior semicircular canal dehiscence; Fig. 38-15, B, shows the condition as seen on MRI.
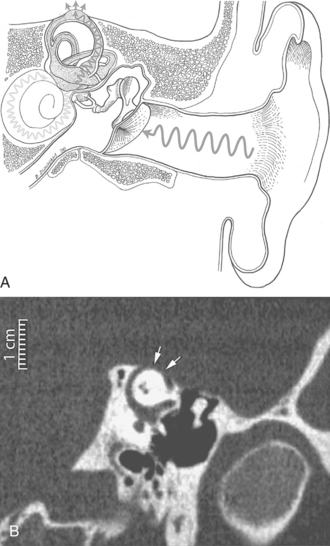
Figure 38-15 A, In superior semicircular canal dehiscence syndrome, sound waves can excite the superior canal because the “third mobile window” created by the dehiscence allows some sound pressure to be dissipated along a route through the superior canal in addition to the conventional route through the cochlea. B, CT scan demonstrating dehiscence (arrows) of the superior canal. (From Cummings CW, Haughey BH, Thomas R, et al: Cummings otolaryngology: head and neck surgery, ed 4, St Louis, 2004, Mosby.)
Ototoxicity
Overview.: The aminoglycoside antibiotics can be ototoxic, with auditory toxicity estimated at 20% and vestibular toxicity affecting 15% of the individuals receiving the drug. It appears that there is a possible genetic vulnerability, which is under study. Streptomycin and gentamicin specifically target the vestibular end organ. Other members of this group of drugs include kanamycin, tobramycin, amikacin, netilmicin, and sisomicin. Ototoxicity is usually seen in individuals who are given multiple doses over time or one large dose, usually aimed at managing a threatening infection. Damage to the hair cells in the inner ear can result in complete loss of vestibular function within 2 to 4 weeks after these drugs are given.32
Etiologic and Risk Factors.: Approximately 3% of an orally administered aminoglycoside is absorbed from the gastrointestinal tract. They are normally injected for severe systemic infections. Penetration of the blood-brain barrier is generally poor, so that aminoglycosides are injected intrathecally to treat meningitis. Aminoglycosides are excreted primarily by the kidney by glomerular filtration, and therefore high concentrations of drug in the urine may be achieved. Impaired renal function reduces the rate of excretion. Therefore renal failure is a risk factor for ototoxicity, and dosing of aminoglycosides must be modified to compensate for delayed renal excretion. Measurement of peak and trough serum levels of aminoglycosides provides rough guidelines for therapeutic efficacy but is not an absolute guarantee for prevention of ototoxicity, particularly vestibular ototoxicity.18
Pathogenesis.: The aminoglycoside reacts with inner ear tissues to form an active, ototoxic metabolite. The drug in its inactive form combines with iron to form an ototoxic complex. This complex reacts with oxygen to produce reactive oxygen species. These species can then react with various cell components—including the phospholipids in the cell membrane, proteins, and DNA—to disrupt the function, primarily in the outer hair cell. This process can then trigger programmed cell death, resulting in apoptosis. Histopathologic studies demonstrate that the cochlear and/or vestibular hair cells serve as primary targets for injury. Inner hair cells seem to be more resistant to injury than the outer hair cells. This could be a result of the higher concentration of the natural antioxidant glutathione in the inner hair cells. In some cases, spiral ganglion cells may be damaged directly by aminoglycosides without injury to outer hair cells. The stria vascularis may become thinner as a result of cell death. The damage is primarily bilateral, although there may be a difference in severity of loss between the two inner ear systems.
When there is bilateral dysfunction of the vestibular system the ability to determine head position in space can also be lost or decreased. Gaze stability with head movement is severely affected as the vestibular ocular reflex is disabled. With loss of the vestibular system function the individual must rely on somatosensory and visual cues for perception of stability and movement.
Clinical Manifestations and Treatment.: One of the most debilitating early symptoms is oscillopsia. Oscillating vision is the illusion of environmental movement caused by excessive motion of images of stationary objects on the retina, known as retinal slip. Oscillopsia of vestibular origin is brought on or accentuated by head movement. It is due to insufficient VOR. When the vestibular system is unable to keep an image stationary by controlling the movement of the eyes, this slip occurs. Severe or complete bilateral loss of peripheral vestibular system function will result in inability to stabilize vision during head movement and produce oscillopsia.30
MEDICAL MANAGEMENT
It is generally considered that the damage is permanent and that the recovery of function of the vestibular mechanism is limited. Initially there is severe disability related to sensation of dizziness, oscillopsia, and imbalance. Compensatory strategies using visual references and somatosensory input can improve mobility, taking approximately 6 weeks to become effective. There are some circumstances that will always be problematic such as walking in the dark, especially on uneven surfaces, or swimming. Night driving or driving in inclement weather should be avoided as vision is unavailable to provide stability.
Mal de Debarquement
Overview and Definition.: Mal de debarquement (MDDS) is a syndrome that is named essentially for the symptoms related to “getting off the boat.” It is usually triggered after a long time spent on a ship, such as during a cruise, or by an extended train ride. The complaints occasionally occur after international or extended air travel, especially if there is turbulence. The symptoms of dizziness and disequilibrium that usually subside within hours become persistent and can last for weeks, months, and even years. It is also reported as rocking vertigo, since the sensation is usually one of rocking back and forth and is experienced to a greater degree at rest than during movement.12 The individual with MDDS would prefer to be moving in a car rather than standing still.
Pathogenesis, Clinical Manifestations, and Treatment.: The basis for MDDS is uncertain but seems to be related to abnormal adaptation from one sensory context to another. During passive motion there may be a mismatch between the vestibular inputs that encode head motion and other sensory and motor cues. Central adaptation may minimize the symptoms provoked by this mismatch, but when the movement ends, the brain must readapt to the stationary environment. It has been suggested that patients with MDDS undergo a physiologic adaptive process during passive motion, but for unknown reasons they do not readily adapt back to the stable environment. This impaired return to baseline results in a perception of motion when the patient is stationary.49 Somatosensory input is no longer able to override vestibular input for stability. This sensation of constant motion at rest can be debilitating in that the person has difficulty managing the symptoms and tends to limit activity or overmedicate to dampen the sensation.43,56 Providing cues outside the vestibular system is helpful, and using somatosensory weighting or providing cues about position in space using an external level appear to help the system to recalibrate.
Autoimmune Ear Disease
Overview and Definition.: Autoimmune ear disease (AIED) is rapidly progressive bilateral sensorineural hearing loss that responds to the administration of immunosuppressive therapy. The disease is typically characterized by symptoms of pressure and tinnitus in the ears with or without dizziness. Autoimmune disease can affect the inner ear with resultant vertigo, sensorineural hearing loss, aural fullness, and tinnitus.
Clinical Manifestations.: Symptoms usually progress over weeks or months, and there is often a known systemic immune disease, such as rheumatoid arthritis. The otologic symptoms may occur as a direct assault by the immune system or as the deposition of antibody-antigen complex in the capillaries or basement membranes of inner ear structures. AIED mimics Menière’s disease with fluctuating hearing loss and vestibular dysfunction. Approximately 50% of patients testing positively for AIED have positive serum antibody tests. The Western blot is currently the most widely reported diagnostic tool used in the diagnosis of AIED with autoantibodies directed against the 68 kilodalton (kd) protein.11
Treatment.: Typical patients with AIED are managed medically with corticosteroids at the lowest dose that prevents fluctuating hearing. Most patients achieve mild recovery of sensorineural hearing loss on corticosteroids. In more severe cases there is report of medical management with methotrexate.35
Neoplasia
Overview.: Primary carcinoma can directly involve the end organ, the middle ear, or the mastoid. Glomus tumors are the most common tumor of the middle ear, arising from the chemoreceptor system of the ninth through twelfth cranial nerves and producing focal symptoms. See Chapter 30 for more information on the type of tumor presented here and information regarding treatment.
Schwann cell tumors arise from the nerve sheath of the vestibular nerve. The term acoustic neuroma is commonly used to describe this tumor, especially with regard to surgery. The tumor usually arises from the vestibular component of the nerve rather than the cochlea. Schwannomas are usually small, firm, encapsulated tumors that grow very slowly. They form in the internal auditory canal or cerebellopontine angle and produce symptoms by compressing the nerve. Initial symptoms are usually related to hearing loss. Eventually mild symptoms of dizziness or balance disorders can develop as the tumor increases in size and the ability to adapt to the loss of function is lost. Fig. 38-16 shows the resulting findings on audiogram and imaged with MRI.
Figure 38-16 A, Normal audiogram. B, Abnormal audiogram in an individual with bilateral hearing loss. C, Abnormal audiogram in left ear with loss of both air and bone conduction. D, Vestibular schwannoma pressing against the left side of the brainstem and into the internal auditory canal. (A, B, and C redrawn from and D from Nolte J: The human brain: an introduction to its functional anatomy, ed 5, St Louis, 2002, Mosby.)
MRI with gadolinium contrast is used to diagnose these tumors. Surgical removal of schwannoma can be achieved by performing middle fossa craniotomy. The translabyrinthine approach is appropriate for tumors up to 3.0 cm. Radiosurgery (a single treatment of high-dose irradiation stereotactically administered) has been used successfully with fewer complications than surgery. There is a chance of progressive loss of hearing in the years following this procedure.
Meningiomas arise from the arachnoid layer in the area of the petrosal and sigmoid sinuses. The tumor is encapsulated and therefore does not invade the neural tissue. Displacement of the cranial nerves, brainstem, and cerebellum is common, causing complaints consistent with compressive damage.
Gliomas arising from the brainstem grow slowly and progressively disrupt the brainstem centers, invading the vestibular and auditory systems in 50% of cases.16 Gliomas arising in the cerebellum are relatively silent until there is compression of the brainstem or obstruction of cerebrospinal fluid. Medulloblastomas, which occur primarily in children and adolescents, are rapidly growing tumors of the vermis and hemispheres of the cerebellum.
Metastatic neoplasms involve the vestibular and auditory functions primarily through involvement of the temporal bone. The internal auditory canal is a frequent site of metastatic tumor growth. Medical interventions to control growth can cause disruption of the neural tissue and trigger edema in the area of the vestibular system. This can cause acute symptoms; there is usually a good response to vestibular rehabilitation in that case.
Traumatic Brain Injury
Risk Factors.: Complaints of dizziness and imbalance are common after traumatic brain injury (TBI) and may affect as many as 50% of the individuals who experience TBI. There may be direct injury to the vestibular mechanism as a result of temporal bone fracture resulting in tearing of the tympanic membrane or the membranous labyrinth. The vestibular and cochlear nerve can be damaged in the trauma, producing peripheral vestibular disorders. Labyrinthine concussion can trigger BPPV.51 Posttraumatic endolymphatic hydrops can cause intermittent dizziness but responds to management as described above. Perilymphatic fistula is more common after head injury and can be difficult to assess given the fluctuating nature of brain injury and difficulty with maintaining consistent performance of home programs.
Pathogenesis.: Direct damage of the vestibular nuclei or cerebellar connections can cause persistent and disabling positional dizziness.3,48 Brainstem or midbrain damage involving the vestibular nuclei can create inability to integrate somatosensation, visual, or vestibular input. In this case the individual will often choose to decrease the use of the vestibular system by limiting head motion and using predominantly visual or somatosensory cues for balance. The ability of the vestibular system to perform is further decreased by general lack of challenge. Then when the vestibular system is stimulated during daily activity, there is a sensation of dizziness and loss of postural stability. The individual with TBI may have more difficulty providing a clear history and may be plagued by other issues such as photophobia, hyperacusis, and visual motion sensitivity. Because of the damage to the central mechanisms that are necessary for the adaptation of vestibular impairment, recovery can take up to three times as long as would otherwise be expected. Flexion-extension injury is common and may limit the ability to perform therapy involving head motion. There is often a cervicogenic component to the dizziness that can respond well to intervention. See Chapter 33 for further discussion of the brainstem disorders.
Psychologic Disorders and Somatoform Dizziness
Overview and Definition.: Forty percent of dizzy individuals have psychologic disorders and, in general, individuals with psychiatric disorders report more disability with dizziness than do individuals without psychiatric involvement. Determining the correct etiology is critical in determining the treatment. Interest in this area is growing, and methods to more accurately identify the cause of the abnormalities are being developed. Connections between the locus coeruleus and lateral vestibular nucleus and serotonergic effects have implications for both processes. In a somatoform disorder the cause of the dizziness symptoms cannot be traced to any objective organic pathologic findings and are experienced by patients as symptoms resulting from the dizziness and not the cause of it. Dizziness is usually the chief symptom and remains dominant after several years. There appears to be greater impairment in work and daily activities when the condition has a somatoform nature than for individuals who have dizziness with a known cause.
Anxiety and phobic disorders are the most frequent psychologic disorders underlying somatoform dizziness. Primary somatoform dizziness may develop without a prior fundamental organic disease, with individuals exhibiting dizziness sensations of varying quality and associated with impairment of daily activities as the chief symptom. Examination reveals typical symptom complexes for each of the underlying psychiatric disorders, such as anxiety, phobia, depression, or dissociation. Box 38-7 provides guidelines regarding phobic dizziness.
Secondary, reactive, or comorbid psychiatric disorders can emerge as a consequence of organic dizziness. Vestibular neuritis and BPPV are two common causes of dizziness that respond well to intervention. Often these individuals are given the standard intervention and acute symptoms subside, but recovery is illusive because of the underlying psychiatric disorder. Patients who have predisposed personality traits or previously latent anxiety disorders interpret the physical sensations catastrophically. The physical sensations function as stimuli triggering panic attacks. As a consequence, panic attacks can be triggered by other stimuli through conditioning, thereby persisting despite adequate central vestibular compensation.22
Vestibular disorders have an influence on autonomic regulation. In somatoform disorder there is often a higher frequency of other autonomic symptoms such as heart palpitations, fainting spells, and chronic fatigue. As central neurologic links are found to exist between the vestibular and autonomic systems, it is hypothesized that dizziness and panic symptoms may have a common neuroanatomic basis. Vertiginous migraineurs have a higher frequency of comorbid anxiety disorders.
Clinical Manifestations.: Dizziness attacks can be an expression of a panic attack lasting from a few minutes to half an hour or up to 2 hours and can be accompanied by vegetative symptoms, feelings of depersonalization, feelings of anxiety and panic, and hypochondriacal anxieties. In the case of panic disorder, recurrent dizziness attacks (i.e., several attacks over the course of 1 month) justify the diagnosis. In severe cases, the attacks may occur several times a day. Initially, the dizziness attacks cannot be related to any specific trigger situation. Nevertheless, the result is that patients frequently tend to avoid those places where they experienced their first dizziness attack. Not until later, during the course of psychotherapy, does it become clear what unconscious conflicts are underlying.
Nighttime dizziness attacks combined with sleep disorders also can occur. The patients subjectively believe that they have been awakened by the dizziness and afterwards often are unable to sleep for long periods. After the attacks, patients frequently feel exhausted. It is not uncommon for these patients to contact emergency medical services. Many patients develop hypochondriac anxieties and insist on repeated diagnostic measures. Episodes of hyperventilation also may be associated with panic disorder, yet patients subjectively may not perceive them as such. The hyperventilation leads to subjective feelings and sensations, such as dizziness, emptiness, lightheadedness, fear of fainting, and paresthesias, which in turn intensify the fear in a vicious circle because patients interpret these sensations as an expression of a severe physical illness.
In generalized anxiety disorder patients have dizziness attacks and permanent dizziness with fluctuating intensity. In addition to the symptoms, patients complain of constant inner tension and nervousness, a persistent generalized diffuse anxiety accompanied by psychomotor agitation and anxiety about the future. Establishing a diagnosis of somatoform dizziness as an expression of a panic disorder or a generalized anxiety disorder is simple after careful clinical examination. Anxiety disorders can be associated with depression. Often depressive disorders are not distinguished easily from each another.22
Agoraphobia and social phobia are the two phobic disorders underlying somatoform dizziness most frequently. On closer exploration, it becomes clear that the dizziness frequently occurs in specific situations (e.g., agoraphobia when shopping in the supermarket, waiting in line, or driving a car). A classic phobia involves the fear of failing in such situations, becoming panicked, and falling into a state of helplessness and powerlessness, ultimately leading to embarrassment and shame. Avoidance behavior is not perceived subjectively as such; rather, it is a logical consequence of the dizziness. They often conceal their psychologic symptoms of anxiety and phobic avoidance and they are able to continue with their activities of daily life and work.
Somatoform dizziness as an expression of a depressive disorder usually takes the form of permanent diffuse dizziness. As with the anxiety disorders and phobic disorders, all further symptoms are interpreted as resulting from the dizziness. The symptoms can be manifold: depressive moods; rapid changes in mood; loss of drive and concentration; sleep disorders; or typical somatic symptoms, such as loss of appetite, loss of weight, and libidinal disorders. In severe cases, social withdrawal, fear of the future, nihilistic thoughts and, rarely, suicidal indications may occur. Older patients often are misdiagnosed as suffering from vascular diseases or dementia. An organic lesion can act as a trigger and can make these persons feel completely unsure of themselves, leading in some instances to withdrawal from social life and isolation. Older people generally are poorly cared for in terms of psychotherapeutic and psychiatric therapy. In particular, older patients benefit most from appropriate treatment to improve their quality of life when it is hampered considerably by somatoform dizziness.
Somatoform dizziness can be an expression of a dissociative disorder (conversion disorder). In these cases, dizziness has the psychodynamic meaning of a conversion symptom. Careful clinical diagnostics commonly reveal a close temporal connection between past conflicts or stress situations and the onset of dizziness, a relationship of which patients initially are not aware. In contrast to patients who have anxiety or phobic or depressive disorders, these patients initially do not show such pronounced suffering and are less limited in their daily life and work activities. They do tend to demonstrate clear signs, however, of reaping secondary gains from the disorder, which primarily is connected with acute relief from unsolved, usually subconscious, internal conflicts. During the course of illness, however, secondary conditioning processes can start. Then the dizziness eventually occurs independently of the triggering conflict situation whenever low-level tension or mental irritation arises. In these cases, diagnosis can be difficult because the original conflict that triggered the dizziness is hidden. Therefore it is important to identify the exact time of onset of dizziness and the associated living circumstances at that time.
Psychogenic hearing disorders nearly always are dissociative disorders (conversion disorders). Initially, patients are not aware of the underlying conflicts. They willingly tell about their hearing disorder, which they often attribute to an earlier illness or previously occurring event, such as loud music and noises, an upper respiratory tract infection, or an ear infection. Frequently, it is only during thorough psychiatric-psychodynamic diagnostic procedures that conflicts come to light that are connected with the hearing disorder. Depending on the underlying conflict, long-term psychotherapy may be required in some cases.
Pathogenesis.: The parabrachial nucleus holds particular importance as it is tied to the central core of the amygdala and the infralimbic cortex through reciprocal connections. A special significance is attributed to these brain structures during the development of fear and panic sensations and the conditioning of avoidance. States of hyperventilation can alter balance of the vestibular system, induce nystagmus, and provoke dizziness sensations in patients who have subclinical vestibular disorders.
Patients who suffer from a depersonalization or derealization syndrome often describe themselves as having diffuse dizziness or feeling dazed. Frequently, the depersonalization disorder is overlooked, but closer examination reveals that patients are not suffering from dizziness per se. The patients describe a strange feeling in their head, feeling dazed, like being wrapped in cotton, feeling unreal or foreign, as though they are outside their body. They often barely can express these symptoms in words, as they find the symptoms highly disturbing and embarrassing. Isolated depersonalization syndromes are rare; a depersonalization disorder usually is accompanied by anxiety and depressive disorders. In the presence of a depressive disorder or an anxiety disorder, the existence of a depersonalization disorder often is underestimated.
MEDICAL MANAGEMENT
Somatoform dizziness usually is included late in the differential diagnosis or not at all, which is why the diagnosis is delayed in most cases—often after several months or years of the disorder—or is not made at all.
Individuals often consult first with an ear, nose, and throat physician, neurologist, or internist. To physicians who are less familiar with somatoform dizziness, these symptoms do not seem suspicious, and they usually are not recognized because the patients present with dizziness as the chief symptom. Initially it seems logical that, for example, a specific avoidance behavior or the reported vegetative symptoms result exclusively from organically caused dizziness.
This circumstance often increases the tendency of somatoform dizziness to become chronic, frequently leading to severe impairment of the quality of life, even early retirement, and high health care costs. Patients who have somatoform dizziness lasting more than 12 weeks and involving marked impairment should undergo interdisciplinary diagnostics routinely, including medical and ophthalmologic diagnostics, in addition to differentiated neurologic or neuro-otologic and psychosomatic diagnostics. Over long periods misdiagnoses are made or novel diagnoses invented, such as cervical dizziness or Menière’s disease.
When complex persistent dizziness syndromes are present, careful organic diagnostics always must be performed followed by a differentiated psychiatric-psychodynamic examination performed by an experienced clinician; this practice makes possible the diagnosis of somatoform dizziness in approximately two thirds of patients.
Attempts at symptomatic treatment such as long-term use of antivertiginous drugs are based on the assumption of residual signs and symptoms after a previous organic vertigo syndrome. These treatments typically lead to a short-term placebo effect, if they are successful at all, and contribute to perpetuating the dizziness symptoms by encouraging patients to believe their dizziness to be organic in origin. The consequence is that other symptoms, such as reactive depression and motivational disorders, crop up in addition to the primary underlying psychiatric disorder because all attempts at treatment are unsuccessful.
Clinical interactions have an important influence on the course of somatoform dizziness. When somatoform dizziness develops as a consequence of a vestibular disorder, explaining the psychosomatic connections in detail can be the first step in recovery. Patients may insist on new diagnostic measures because of hypochondriac anxieties. If their health care providers do not give in to patients’ insistence, instead reassuring them that their symptoms are taken seriously and repeatedly pointing out the necessity of psychosomatic diagnostics and therapy, they may help prevent the development of chronic symptoms.
TREATMENT.
Treatment is determined by clinical presentation. Psychotherapy should begin immediately. Psychodynamic techniques and behavioral therapy are effective. The indication is determined by the clinical findings and the underlying conflict and stress situation. If the disorder is of short duration and pronounced only mildly, focused outpatient treatment can be successful. Depending on the underlying conflict, a long-term psychodynamic therapy should be the treatment of choice. If the illness is severe, pharmacotherapy to help manage symptoms may be necessary.
PROGNOSIS.
Follow-up studies of the course of complex somatoform dizziness show an urgent need for early differentiated interdisciplinary diagnostics. The follow-up period in individual studies ranges from 4 months to 2 years. Depression, anxiety, and compulsion are influenced more heavily by how the dizziness is experienced subjectively than by the objective balance disorder. Subjective experiences depend on the underlying or associated psychologic disorder. The extent of the handicap and form of emotional distress interact with each other; depression and anxiety impede the ability to cope, thus exacerbating the dysfunction.
Tinnitus can be a trigger for depressive disorders. Sleep disorders, concentration difficulties, irritability, and a low threshold for stress can cause depression and lead to social withdrawal. The subjectively perceived intensity of the tinnitus often depends on the level of stress. Phobic reactions may develop, particularly when the tinnitus is accompanied by hyperacusis.
Comorbid Disorders with Vestibular Consequences
Congenital Vestibular Loss.: Events before birth can cause loss of vestibular function and are related to genetics or intrauterine infection, intoxication, or anoxia. Rubella and cytomegalovirus are responsible for most cases. Thalidomide, no longer commonly used, can cause loss of vestibular function.25
Vascular Disease.: Vertebrobasilar insufficiency is a common cause of vertigo in persons older than 50 years. This is often due to atherosclerosis of the vertebral and basilar arteries. Ischemia confined to the labyrinthine artery distribution results in infarction of the vestibular labyrinth and cochlea. Ischemia can also cause vertigo without hearing loss. A vascular loop compressing the eighth cranial nerve can cause vertigo, tinnitus, and hearing loss. Spontaneous hemorrhage into the inner ear, resulting in vertigo and hearing loss, mainly occurs in persons with underlying bleeding disorders. Leukemia is the most common disorder associated with labyrinthine hemorrhage.
Cerebellar infarction simulating vestibular neuritis is more common than previously thought. Early recognition of the pseudo-vestibular neuritis of vascular cause may allow specific management. Infarcts located dorsolateral to the fourth ventricle can cause interruption of the vestibular nuclei-archicerebellar loop seems to be responsible for central paroxysmal positional vertigo.
Vestibular dysfunction is a common component with brainstem stroke and may respond to intervention. See Chapter 32 for a more complete description of these stroke syndromes.3
Vestibular Migraine.: A type of migraine headache that results in symptoms of dizziness is associated with vascular changes in the area of the vestibular apparatus and is common in both children and adults. (See Chapter 37 for the full description of vestibular migraine.) Approximately 30% of the population with migraine report episodic vertigo, in some cases during the headache-free period.
Metabolic Disorders.: When vascular and nerve changes associated with diabetes mellitus occur in the area of the peripheral or central vestibular system, symptoms may develop. Loss of proprioceptive input and visual degeneration will further exacerbate the sense of disequilibrium. Metabolic disorders affecting the resorption of bone, such as otosclerosis and Paget’s disease, may develop to the point of causing degeneration of the labyrinths or nerves, resulting in vertigo and dizziness.
Allergies.: Adverse reactions to foods and chemicals have been recognized as important etiologic agents in allergy. Otolaryngologic allergists address the clinical aspects of food sensitivity and dizziness after exposure to specific chemicals. There is clinical evidence of the relationship of dizziness with food allergies. The pathophysiology, however, remains unclear.47,56
Aural Cholesteatomas.: Aural cholesteatomas are cysts of the middle ear or mastoid arising from squamous epithelial lining, containing keratin. Cholesteatomas erode the temporal bone and may be congenital or acquired. The cholesteatoma can cause resorption of the adjacent bone by a process of pressure erosion. This process can cause damage to the labyrinths and result in vestibular dysfunction. Annual incidence in children is 3 per 100,000 and 12.6 per 100,000 in adults. The diagnosis of aural cholesteatoma is made on otoscopic examination, including endoscopic and microscopic evaluation or surgical exploration. Special imaging procedures, such as high-resolution CT scanning and MRI, may suggest the presence of cholesteatoma within the temporal bone and may be used to complement the clinical examination. High-resolution CT scanning is useful for operative planning and is recommended for all revision mastoid operations. The symptoms of cholesteatoma vary; some cholesteatomas are asymptomatic, whereas others become infected and rapidly cause bone destruction.
Malignant External Otitis.: Malignant external otitis, an infection affecting older people with diabetes or immunosuppression, begins in the external auditory canal and spreads to the temporal bone, putting pressure on the facial nerve or the surrounding nerves.
The most common intracranial complication of otitic infections is extradural abscess, a collection of purulent fluid between the dura mater and bone of the middle or posterior fossa. Spread of the infection across the dura from the epidural space may result in thrombophlebitis of the lateral venous sinus, subdural abscess, meningitis, and brain abscess.
In some cases the damage is temporary, as in serous labyrinthitis, and vestibular and auditory function return to varying degrees. Permanent damage is possible when the infection causes damage to the structures of the labyrinth or the eighth cranial nerve. When the membranous labyrinth becomes permanently damaged, endolymphatic hydrops may result (see the section on Menière’s disease in this chapter) and the symptoms can become episodic.
Malignant external otitis in the mastoid bone is a major clinical problem. It often occurs in persons with a chronic illness, such as diabetes, or a malignancy who are receiving broad-spectrum antibiotics. The infection enters the middle and inner ear via the nasal sinuses. The infection can spread into the intracranial cavity and create thrombosis of the cerebral arteries. The eighth cranial nerve can be affected along with surrounding cranial nerves in the presence of basilar meningitis.5
References
1. Agrup, C. Systemic autoantibodies against discrete inner ear compartments in bilateral vestibular loss. Neurology. 2005;65(1):167.
2. Allison, L, Fuller, K. Balance and vestibular function. In: Umphred DA, ed. Neurological rehabilitation. ed 5. St Louis: Mosby Elsevier; 2007:732–774.
3. Arai, M, Terakawa, I. Central paroxysmal positional vertigo. Neurology. 2005;64(7):1284.
4. Arenberg, IK. Menière’s disease: diagnosis and management of vertigo and endolymphatic hydrops. In: Arenberg IK, ed. Dizziness and balance disorders. New York: Kugler; 1993:503–510.
5. Baloh, RW, Honrubia, V. Clinical neurophysiology of the vestibular system, ed 2. Philadelphia: FA Davis, 1990.
6. Barrs, DM. Intratympanic corticosteroids for Menière’s disease and vertigo. Otolaryngol Clin North Am. 2004;37(5):955–972.
7. Black, FO, et al. Vestibular reflex changes following aminoglycoside induced ototoxicity. Laryngoscope. 1987;97:582–587.
8. Brandt, T, Dietrich, M. Assessment and management of disorders affecting central vestibular pathways. In: Herdman SJ, ed. Vestibular rehabilitation. ed 3. Philadelphia: FA Davis; 2007:409–432.
9. Britton, BH. Common problems in otology. St Louis: Mosby Year Book, 1991.
10. Brookes, GB, et al. Sensing and controlling rotational orientation in normal subjects and patients with loss of labyrinthine function. Am J Otol. 1993;14:349–361.
11. Boulassel, MR, Deggouj, N, Tomasi, JP, et al. Inner ear autoantibodies and their targets in patients with autoimmune inner ear disease. Acta Otolaryngol. 2001;121:28–34.
12. Brown, JJ, Baloh, RW. Persistent mal de debarquement syndrome: a motion induced subjective disorder. Am J Otolaryngol. 1987;8:219.
13. Burt, AM. Textbook of neuroanatomy. Philadelphia: WB Saunders, 1993;272.
14. Cevette, MJ, et al. A physiologic performance on dynamic posturography. Otolaryngol Head Neck Surg. 1995;112:676–679.
15. Clendaniel, RA, Helminski, JO. Rehabilitation strategies for patients with vestibular deficits. In: Arenberg IK, ed. Dizziness and balance disorders. New York: Kugler; 1993:663–676.
16. Collard, M, Chevaliar, Y. Vertigo. Curr Opin Neurol. 1994;7:88–92.
17. Curthoys, IS, Halmagyi, GM. Vestibular compensation: a review of the oculomotor, neural and clinical consequences of unilateral vestibular loss. J Vestib Res. 1995;5:67–107.
18. Cummings, CW, Haughey, BH, Thomas, R, et al. Cummings otolaryngology: head and neck surgery, ed 4. St Louis: Mosby, 2004.
19. Derebery, MJ. Allergic and immunologic aspects of Menière’s disease. Otolaryngol Head Neck Surg. 1996;114(3):360–365.
20. Deutschländer, A, Strupp, M, Jahn, K, et al. Vertical oscillopsia in bilateral superior canal dehiscence syndrome. Neurology. 2004;62:784–787.
21. Draznin, E, Theriot, KO. Multidisciplinary clinic’s model: a comprehensive approach for management of dizziness and balance disorders. In: Arenberg IK, ed. Dizziness and balance disorders. New York: Kugler; 1993:649–654.
22. Eckhardt-Henn, A, Dieterich, M. Psychiatric disorders in otoneurology patients. Neurol Clin. 2005;3(23):731–749.
23. Epley, JM. Aberrant coupling of otolithic receptors: manifestations and assessment. In: Arenberg IK, ed. Dizziness and balance disorders. New York: Kugler; 1993:183–202.
24. Fetter, M, Dichgans, J. Vestibular neuritis spares the inferior division of the vestibular nerve. Brain. 1996;119(Pt 3):755–763.
25. Fetter, M. Vestibular system disorders. In: Herdman SJ, ed. Vestibular rehabilitation. ed 3. Philadelphia: FA Davis; 2007:98–106.
26. Foster, CA. Vestibular rehabilitation. Baillieres Clin Neurol. 1994;3:577–591.
27. Fukushima, K. Corticovestibular interactions: anatomy, electrophysiology, and functional considerations. Exp Brain Res. 1997;117:1–16.
28. Furman, JM. Role of posturography in the management of vestibular patients. Otolaryngol Head Neck Surg. 1995;112:8–12.
29. Glasscock, ME, Cueva, RA, Thedinger, BA. Handbook of vertigo. New York: Raven Press, 1990.
30. Goebel, JA. Understanding eye movements in balance disorders. In: Arenberg IK, ed. Dizziness and balance disorders. New York: Kugler; 1993:49–56.
31. Hain, TC, Helminski, JO. Anatomy and physiology of the normal vestibular system. In: Herdman SJ, ed. Vestibular rehabilitation. ed 3. Philadelphia: FA Davis; 2007:2–20.
32. Halmagyi, GM, et al. Gentamicin vestibulotoxicity. Otolargyngol Head Neck Surg. 1994;111:571–577.
33. Halmagyi, GM, Curthoys, IS. Vestibular compensation: clinical changes in vestibular function with time after unilateral vestibular loss. In: Herdman SJ, ed. Vestibular rehabilitation. ed 3. Philadelphia: FA Davis; 2007:76–97.
34. Halmagyi, GM, Curthoys, IS. Otolith function tests. In: Herdman SJ, ed. Vestibular rehabilitation. ed 3. Philadelphia: FA Davis; 2007:144–161.
35. Harris, JP, et al. Treatment of corticosteroid-responsive autoimmune inner ear disease with methotrexate. JAMA. 2003;290(14):1875–1883.
36. Herdman, SJ. Advances in the treatment of vestibular disorders. Phys Ther. 1997;77:602–609.
37. Herdman, SJ, et al. Vestibular adaptation exercises and recovery: acute stage after acoustic neuroma resection. Otolaryngol Head Neck Surg. 1995;113:77.
38. Herdman, SJ, Tusa, RJ. Physical therapy management of benign positional vertigo. In: Herdman SJ, ed. Vestibular rehabilitation. ed 3. Philadelphia: FA Davis; 2007:233–260.
39. Horak, FB, et al. Effects of vestibular rehabilitation on dizziness and imbalance. Otolaryngol Head Neck Surg. 1992;106:175.
40. Horak, FB, Shupert, CL. Role of the vestibular system in postural control. In: Herdman SJ, ed. Vestibular rehabilitation. ed 3. Philadelphia: FA Davis; 2007:32–53.
41. Jacobson, GP, Newman, CW. Background and technique of caloric testing. In: Jacobson GP, Newman CW, Kartush JM, eds. Handbook of balance function testing. St Louis: Mosby Year Book; 1993:156–192.
42. Kelly, JP. Vestibular system. In: Kandel ER, Schwartz JH, eds. Principles of neural science. ed 2. New York: Elsevier; 1985:584–596.
43. Keshner, EA. Postural abnormalities in vestibular disorders. In: Herdman SJ, ed. Vestibular rehabilitation. ed 3. Philadelphia: FA Davis; 2007:54–75.
44. Khater, TT, et al. Dynamics of adaptive change in human vestibulo-ocular reflex direction. J Vestib Res. 1990;1:23–29.
45. Krebs, DE, et al. Double blind, placebo-controlled trial of rehabilitation for bilateral vestibular hypofunction: preliminary report. Otolaryngol Head Neck Surg. 1993;109:735–741.
46. Larouere, MJ, Seidman, MD, Kartush, JM. Medical and surgical treatment of vertigo. In: Jacobson GP, Newman CW, Kartush JM, eds. Handbook of balance function testing. St Louis: Mosby Year Book; 1993:337–358.
47. Leigh, RJ, Zee, DS. The neurology of eye movements. Philadelphia: FA Davis, 1999.
48. Lee, H, Sohn, SI, Cho, YW, et al. Cerebellar infarction presenting isolated vertigo: Frequency and vascular topographical patterns. Neurology. 2006;67(7):1178–1183.
49. Lewis, RF. Frequency-specific mal de debarquement. Neurology. 2004;63(10):1983–1984.
50. Maki, BE, McIlroy, WE. The role of limb movements in maintaining upright stance: “the change of support strategy”. Phys Ther. 1997;77:488.
51. Matsumura, BA. Balance in the elderly. Clin Geriatr Med. 2006;22(2):395–412.
52. Murphy, TP. Mal de debarquement syndrome: a forgotten entity? Otolaryngol Head. Neck Surg. 1993;109:10–11.
53. Nashner, LM. Computerized dynamic posturography. In: Jacobson GP, Newman CW, Kartush JM, eds. Handbook of balance function testing. St Louis: Mosby Year Book; 1993:280–307.
54. Nashner, LM. Computerized dynamic posturography: clinical applications. In: Jacobson GP, Newman CW, Kartush JM, eds. Handbook of balance function testing. St Louis: Mosby Year Book; 1993:308–334.
55. Nashner, LM. Practical biomechanics and physiology of balance. In: Jacobson GP, Newman CW, Kartush JM, eds. Handbook of balance function testing. St Louis: Mosby Year Book; 1993:261–279.
56. Nonas, NG. Inner ear allergy diathesis and environmental factors: evaluation and management. In: Arenberg IK, ed. Dizziness and balance disorders. New York: Kugler; 1993:555–562.
57. Nuti, D, et al. Benign paroxysmal positional vertigo of the horizontal canal: a form of canalithiasis with variable clinical features, J. Vestib Res. 1996;6:173–177.
58. Shepard, NT, Telian, SA. Programmatic vestibular rehabilitation. Otolaryngol Head Neck Surg. 1995;112:173–177.
59. Shepard, NT, Telian, SA, Smith-Wheelock, M. Habituation and balance retraining therapy: a retrospective view. In: Arenberg IK, ed. Dizziness and balance disorders. New York: Kugler, 1993.
60. Shumway-Cook, A. Assessment and management of the patient with traumatic brain injury and vestibular dysfunction. In: Herdman SJ, ed. Vestibular rehabilitation. ed 3. Philadelphia: FA Davis; 2007:444–457.
61. Shumway-Cook, A, Woollacott, MH. Motor control: theory and practical applications. Baltimore: Lippincott, Williams & Wilkins, 2001.
62. Sung, KB, Lee, TK, Furman, JM. Abnormal eye movements in dizzy patients. Neurol Clin. 2005;23(3):675–703.
63. Venecia, RD, Nadol, JB. Menière’s disease. In Rakel RE, Bope ET, eds.: Conn’s current therapy, ed 58, Philadelphia: Saunders, 2006.
64. Szturm, T, Ireland, DJ, Lessing-Turner, M. Comparison of different exercise programs in the rehabilitation of patients with chronic vestibular dysfunction. , J Vestib Res. 1994;4:461.
65. Tusa, RJ, et al. The contribution of the vertical semicircular canals to high-velocity horizontal vestibulo-ocular reflex (VOR) in normal subjects and patients with unilateral vestibular nerve section. Acta Otolaryngol. 1996;116:507–512.
66. Tusa, RJ. Bedside assessment of the dizzy patient. Neurol Clin. 2005;23(3):655–673.
67. Wade, S, Curthoys, IS. The effect of ocular torsional position on perception of the roll-tilt of visual stimuli. Vision Res. 1997;37:1071–1077.
68. Wrisley, DM, Pavlou, M. Physical therapy for balance disorders. Neurol Clin. 2005;23(3):855–874.
69. Yardley, L, et al. A randomized controlled trial of exercise therapy for dizziness and vertigo in primary care. Br J Gen Pract. 1998;48:1136–1140.
70. Zee, DS. The management of patients with vestibular disorders. In: Barber HO, Sharpe JA, eds. Vestibular disorders. Chicago: Year Book; 1988:254–274.
71. Zee, DS. Vestibular adaptation. In: Herdman SJ, ed. Vestibular rehabilitation. ed 3. Philadelphia: FA Davis; 2007:19–31.