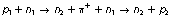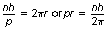Chapter 18 The elementary structure of the atom
Chapter contents
18.1 Aim
The aim of this chapter is to introduce the reader to the key components of an atom. The principal particles which form the nucleus will be identified, as will the factors which determine whether or not the nucleus is stable. The differing electron orbitals and the influence of the electron orbitals on the chemical properties of the material will be discussed. The consequences of transitions of electrons between orbitals will also be identified.
18.2 Introduction
Any attempt to understand the universe around us must start with the fundamental question: what is matter made of? The atom as the fundamental building block of matter has been the subject of a great deal of both theoretical debate and experimental study by physicists. Many of the modern theories concerning atomic and subatomic structures are extremely complex and are the subject of a number of textbooks in their own right. Most of the phenomena which we encounter in radiography can be explained using a relatively simple planetary model of the atom. In this model, there are solid electrons orbiting a solid nucleus – some phenomena can also be explained using the quantum physics model and where this is appropriate this model will be referred to in Insights.
The planetary model of the atom was first described by Rutherford in 1911. It describes an atom consisting of a small, positively charged central nucleus (containing protons and neutrons) around which negatively charged electrons move in defined orbitals. This model can be used to illustrate the carbon atom, as shown in Figure 18.1. As we can see from the diagram, the nucleus of this atom consists of 12 elementary particles – six protons and six neutrons. These particles are bound together in a small volume of extremely high density – about three thousand million, million times greater than the density of water. The protons in the nucleus carry a positive charge and the electrons carry an equal negative charge, so this atom is electrically neutral – the neutrons carry no charge. The electrons are arranged in orbitals or shells called K, L, M … starting from the orbital closest to the nucleus. The K-shell can only contain two electrons and, in the case of the carbon atom, the L-shell contains the remaining four. Different atoms contain different numbers of protons and neutrons in the nucleus and different numbers of electron configurations; this will be discussed later in this chapter. The particles which make up the atom are very tiny and the atom consists largely of empty space. For example, if an atom were to be enlarged until it was the size of a house, the size of the nucleus would be about the size of a pin-head, although it contains 99.95% of the total mass of the atom.
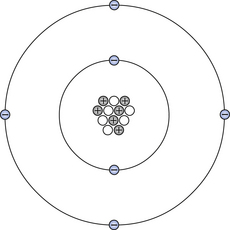
Figure 18.1 The basic structure of a carbon atom. At the centre of the atom is a nucleus which contains six protons (positively charged) and six neutrons (zero charge) – i.e. the nucleus contains 12 nucleons. Six electrons (negatively charged) orbit the nucleus in defined orbitals. As the atom contains equal numbers of positive and negative charges, the whole atom is electrically neutral.
The masses and charges of the subatomic particles which will be considered in this and later chapters are summarized in Table 18.1.
Table 18.1 Masses and charges of the main subatomic particles
| REST MASS | REST ENERGY | |||||
|---|---|---|---|---|---|---|
| PARTICLE | SYMBOL | kg | AMUa | MeV | CHARGEb | COMMENTS |
| Proton | p | 1.672×10−27 | 1.007 | 938 | +1 | Nucleon, i.e. present in the atomic nucleus |
| Neutron | n | 1.675×10−27 | 1.009 | 939 | 0 | Nucleon, i.e. present in the atomic nucleus |
| Alpha-particle | α | 6.645×10−27 | 4.003 | 3718 | +2 | Two protons and two neutrons |
| Ejected in α decay | ||||||
| Electron | e− or β− | 9.109×10−31 | 0.00055 or 1/1820 | 0.511 | −1 | Form stable discrete orbits around nuclei |
| Ejected from nucleus in β decay | ||||||
| Positron | e+ or β+ | 9.109×10−31 | 0.00055 or 1/1820 | 0.511 | +1 | Antiparticle of the electron – produces annihilation radiation when both meet |
| Pi meson | π+ | 2.480×10−28 | 0.150 | 139 | +1 | Keep the nucleus together (π0 and π− also exist) |
| Neutrino | ν | 0 | 0 | 0 | 0 | Emitted during β decay and electron capture |
| Very weak attenuation by matter | ||||||
| Photon or quantum | hv | – | – | – | 0 | Travels at 3×108 m.s−1 Forms part of the electromagnetic spectrum |
a 1 amu is 1 atomic mass unit which is one-twelfth of the mass of a neutral C126 atom.
b A charge of +1 is +1.602×10−19 coulomb.
Rutherford carried out some elegant experimental work connected with the scattering of alpha-particles by atoms. From this, he concluded that the only explanation for the wide scattering angles which he found experimentally was given by assuming that the atom consists of a very heavy positively charged nucleus with orbiting electrons. The alternative model, whereby all the subatomic particles were contained in a very small volume, was unacceptable because it would produce much smaller scattering angles for the alpha-particles.
Similar experiments have recently been carried out by physicists to try to establish whether protons and neutrons can be broken into smaller pieces. Unfortunately, if the energy of the projectile is large enough to break up the proton or neutron, then the energy is great enough to create new particles – remember E=mc2 – and so it is difficult to tell which are fragments of the proton and which have been created as a result of energy being converted into matter. More subtle experiments use high-energy electrons to bombard the protons and neutrons and measure the angle of deflection of these particles. These angles are again over a wide range, suggesting that there are small solid structures within the protons and neutrons. These may be the basic building blocks of the universe; they are known as quarks and 18 different types of quark have been identified. Further discussion regarding this search to identify whether there is a smaller structure within the quark is well outside the scope of this text.
18.3 The atomic nucleus
The number of protons and neutrons in the atomic nucleus determines both the mass and the charge of the nucleus and the configuration of electron orbitals of the atom. There are several important terms, which we will use in this and in following chapters of this text, that require definition at this stage. These terms, which will help us to understand atomic structure, are defined in Table 18.2.
Table 18.2 Terms used to describe a nucleus
| TERM | SYMBOL | DEFINITION |
|---|---|---|
| Nucleon | A proton or neutron within a nucleus | |
| Atomic number | Z | The number of protons in the nucleus |
| Atomic mass number | A | The total number of nucleons in the nucleus |
| Neutron number | N | The number of neutrons within the nucleus |
| Nuclide | A nucleus with a specific value of Z and A | |
| Element | E | A nucleus with a given value of Z |
| Isotope (of an element) | Any nucleus which contains the same number of protons as the given nucleus but has a different mass number | |
| Isobar | Any nucleus which has the same atomic mass number as another nucleus (i.e. has the same value of A) | |
| Radionuclide or radioisotope | Any nuclide or isotope which is radioactive |
We can now consider the use of some of the terms in the tables. The most common naturally occurring stable isotope of carbon has six protons and six neutrons, as shown previously in Figure 18.1. The atomic number (Z) of this isotope is six and the atomic mass number is 12. The whole atom can be written as 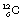 . Thus
. Thus 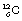 is an example of a nuclide – one which contains six protons and six neutrons. In general, an element E is written as
is an example of a nuclide – one which contains six protons and six neutrons. In general, an element E is written as 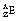 . An isotope of carbon, which is also naturally occurring but less abundant, has seven neutrons in its nucleus and may be written as
. An isotope of carbon, which is also naturally occurring but less abundant, has seven neutrons in its nucleus and may be written as 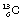 .
.
Note that it is not necessarily the case that isotopes of an element are radioactive, as shown by this example; 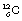 and
and 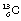 are both isotopes of carbon but neither is radioactive.
are both isotopes of carbon but neither is radioactive.
An isotope of carbon which is radioactive is 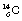 – the well-known carbon-14. This again contains the six protons which identify it as a carbon nucleus, but this time it contains eight neutrons.
– the well-known carbon-14. This again contains the six protons which identify it as a carbon nucleus, but this time it contains eight neutrons. 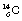 is an example of a radionuclide or a radioactive isotope. It decays, as we shall see in Chapter 19, by beta decay to form
is an example of a radionuclide or a radioactive isotope. It decays, as we shall see in Chapter 19, by beta decay to form 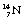 (nitrogen) as the daughter product.
(nitrogen) as the daughter product.
18.3.1 The stability of the nucleus
At first sight the atomic nucleus would appear to be inherently unstable as the neutrons are uncharged and the protons have a positive charge and so would electrostatically repel each other. This would suggest that the nucleus should fly apart because of the electrostatic forces between the protons.
In practice, some nuclei are so stable as to possess no measurable radioactivity (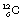 is an example) while others decay with a half-life (see Sect. 20.5) of less than one-millionth of a second. The nucleus must be visualised as a dynamic rather than a static structure where there are opposing forces acting – forces which tend to hold the nucleus together and forces which tend to disrupt the nucleus. A stable nucleus is one where the disruptive forces never win and an unstable nucleus is one where they do succeed. This nucleus is said to undergo radioactive decay. It is not possible to predict the exact moment when any particular nucleus will decay, as it is a matter of probability rather than certainty. However, if there are a large number of unstable nuclei in a sample, the law of radioactive decay (see Sect. 20.3) is obeyed.
is an example) while others decay with a half-life (see Sect. 20.5) of less than one-millionth of a second. The nucleus must be visualised as a dynamic rather than a static structure where there are opposing forces acting – forces which tend to hold the nucleus together and forces which tend to disrupt the nucleus. A stable nucleus is one where the disruptive forces never win and an unstable nucleus is one where they do succeed. This nucleus is said to undergo radioactive decay. It is not possible to predict the exact moment when any particular nucleus will decay, as it is a matter of probability rather than certainty. However, if there are a large number of unstable nuclei in a sample, the law of radioactive decay (see Sect. 20.3) is obeyed.
The forces which hold the nucleus together are quite unlike the other forces – e.g. gravity – with which we are familiar. They are known as short-range nuclear forces and act over distances of about 10−15 metres, over which range they are much more powerful than the electrostatic forces between the protons. These forces are shown diagrammatically in Figure 18.2. A strong force of attraction is evident below 10−15 m and this changes to a force of repulsion at about 10−16 m. The energy expended in keeping the nucleus together is known as the nuclear binding energy (NBE). If the NBE is divided by the number of nucleons within the nucleus, then a figure of about 8.4 MeV is obtained for most nuclei. This is known as the binding energy per nucleon. The binding energy between the nucleons is provided by the transformation of some of the nuclear mass into energy, as given by Einstein’s equation E=mc2. Each nucleon has a mass of approximately 931 MeV per atomic mass unit (amu), of which about 8.4 MeV is used for its NBE to nearby nucleons. Because of this, the mass of the nucleus is always less than the sum of the masses of the nucleons.
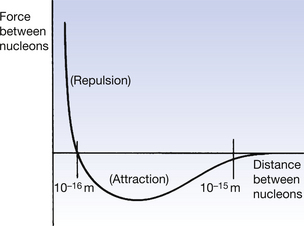
Figure 18.2 If the separation of the nucleons is between 10−15 and 10−16 metres, then a force of attraction exists between the nucleons which helps to hold the nucleus together. If the separation is greater or less than this distance, then the force is a repulsive force.
Considering the graph in Chapter 19 (Fig. 19.2) it can be seen that, for low atomic numbers, equal numbers of protons and neutrons produce the greatest stability. This leads to the concept of nuclear shells, which suggests that there is a layering within the nucleus, with maximum stability being produced when a shell is complete. Further discussion of this concept is beyond the scope of this text.
The π-meson is a particle with a mass between that of an electron and a nucleon and is thought to be responsible for the forces holding the nucleus together. The short-range forces are known as exchange forces and result in (for example) an adjacent proton and a neutron changing continually into a neutron and a proton and back again. This interchange may be written as:
The π+-meson has left the original proton p1 , leaving it as a neutron n2, and then forms a proton p2 by combining with the original neutron n1. The proton and the neutron are continuously exchanging their positions. Negative and neutral π-mesons also exist and are exchanged between nucleons.
18.4 Electron orbitals
Consider an atom of hydrogen, as shown in Figure 18.3. It is assumed in the planetary model of the atom that the solitary electron is on a circular path (path 1) around the nucleus. It may be shown that a body moving in a circle of radius r at a velocity V has an acceleration of V2/r towards the centre of the circle. According to classical physics, such acceleration would result in the emission of electromagnetic radiation from the electron so that the electron is continuously losing energy and would eventually collide with the nucleus (see path 2). Electrons do not behave in this manner, or atoms as we know them would not exist. The electrons orbit in stable paths (path 1) – discrete electron orbitals. Further, these orbitals are grouped in ‘shells’ where there is a particular number of electrons of approximately the same energy in each orbital. (An explanation of the apparent contradiction of the predictions of classical physics (path 2 and path 1) is given in the ‘Insight’ below.) The electrons fill up the inner shells first since the energies of the inner shells are less than the outer shells.
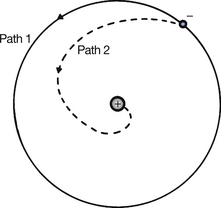
Figure 18.3 A stable, discrete electron orbital (path 1) compared to a ‘decaying electron’ (path 2), as predicted by the laws of classical physics.
The wave–particle duality of matter (see Sect. 16.6) may be used to explain the existence of discrete electron orbitals. Here it is assumed that an orbiting electron has a de Broglie wavelength which is able to fit around the circumference of an orbital an exact number of times (see Fig. 18.4). This fixes the size of each orbital. The condition necessary for this to occur is that nλ=2πr where n is a whole number and r is the mean radius of the orbital. Now λ is the de Broglie wavelength of the electron with momentum p and is given by λ=h/p:
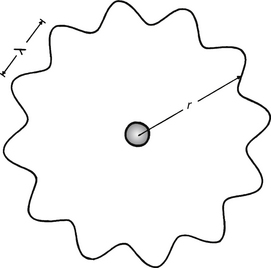
Figure 18.4 The use of the de Broglie wave concept to explain why electrons exist in discrete orbitals.
But pr is the angular momentum of the electron and this must be a multiple of h/2π. There is now no question of electromagnetic radiation occurring from an orbital electron. This is an example of a situation where modern physics can be used to describe atomic phenomena which are not explicable by classical physics.
Table 18.3 shows the maximum number of electrons in each shell from the inner K-shell to the N-shell. The shell number, n, starts from n=1 for the K-shell and is known as the principal quantum number. The chemical properties of an element are controlled by the electron configuration of its atoms. Atoms with filled outer electron shells are chemically inert – neon (Z=10) has full K- and L-shells, as illustrated in Figure 18.5, and so is a chemically inert gas. Fluorine (Z=9), where the K-shell contains two electrons and the L-shell contains seven, is a chemically active electron acceptor (the electron fills the vacancy in the L-shell). Sodium (Z=11), where the K-shell contains two electrons, the L-shell contains eight electrons and the M-shell contains one, is a chemically active electron donor (the electron in the M-shell is donated, resulting in a filled L-shell).
Table 18.3 Numbers of electrons in atomic shells
| PRINCIPAL QUANTUM NUMBER OR SHELL NUMBER (n) | SHELL LETTER | MAXIMUM NUMBER OF ELECTRONS | 2n2 |
|---|---|---|---|
| 1 | K | 2 | 2 |
| 2 | L | 8 | 8 |
| 3 | M | 18 | 18 |
| 4 | N | 32 | 32 |
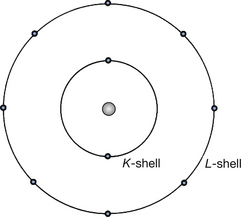
Figure 18.5 An atom of neon, showing both K- and L-shells containing their maximum number of permitted electrons. This means that the neon atom is chemically inert.
The outer electron shells may contain subshells within them. Argon (Z=18) has two electrons in its K-shell, eight electrons in its L-shell and eight electrons in the M-shell. The M-shell has a maximum complement of electrons of 18 and yet argon is chemically inert. An outer subshell of eight electrons is particularly chemically stable, a fact which is confirmed by the next inert gas, krypton, which has an electron configuration of 2, 8, 18, 8.
18.5 The periodic table of elements
If the elements are arranged in order of increasing atomic number, it may be shown that their chemical properties, such as valency, and their physical properties, such as specific heat, tend to occur in a periodic manner. Arranging these elements in these similar groups produces a periodic table as shown in Table E (See page 356). Chemical similarities of the elements in each group may be explained by reference to their electron structure as shown in Table F (See page 357), the electron configuration of the elements. It has already been noted that the number of electrons increases with atomic number and that each electron takes an orbital of the lowest possible energy. This means that the inner shells are filled to stable or substable levels before the outer shells accommodate electrons. There are two rules which determine the way in which the electron shells are gradually built up as the atomic number increases:
1. An electron shell, n, cannot contain more than 2n2 electrons where n is the shell number.
2. The outer shell cannot contain more than eight electrons.
These rules are known as the Bury–Bohr Rules after their co-discovers and ensure that the orbitals of minimum energy are filled first.
One additional constraint is required in order that electrons may fill orbitals in the correct manner. This is known as the Pauli exclusion principle and states that no two electrons may have precisely the same orbital. The two K-shell electrons of an atom, for example, are not at precisely the same energy level because they orbit the atom in opposing directions and hence have slightly different orbitals. These two electrons complete the K-shell so that the electrons must start to fill up the L-shell (see lithium in Table F) at a greater distance from the nucleus and so at a higher energy. These L-shell electrons all have slightly different energies from each other and the shell is complete when it has eight electrons – neon satisfies these conditions where n=2 and so 2n2=8.
The K- and L-shell are thus completed in sequence but the M-shell (which from the 2n2 formula can contain up to 18 electrons), when it reaches nine electrons (argon), then obeys the rule that the outer shell cannot contain more than eight electrons and the ninth electron is placed in the N-shell (see potassium in Table F). This process is repeated each time there are eight electrons in the outer orbital (see rubidium, caesium, fracium, etc.).
As mentioned above, the number of electrons in the outer orbital determines the chemical activity of the element. The ability of one atom to join another is called valency and the electron linkage between the atoms is called the valency bond. There are two types valency bond:
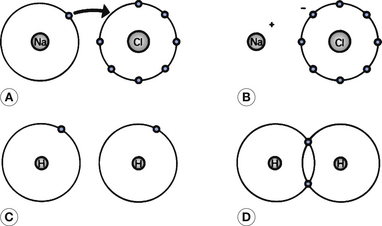
1. Ionic bonds (see Fig. 18.6A and B): this type of bond is created when one or more electrons are transferred from one atom to another forming charged atoms (or ions) which are attracted towards each other by electrostatic attraction forming the bond. After the electron exchange, the shells of each ion appear to be closed.
2. Covalent bonds (see Fig. 18.6C and D): formed by the apparent sharing of electrons such that each atom appears to increase its number of electrons forming an apparently closed shell.
18.6 Electron orbital changes
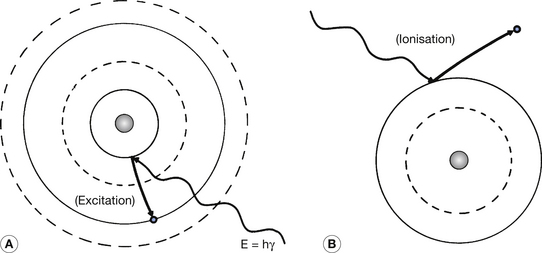
The previous sections of this chapter have shown that electrons may only take up fixed or discrete orbitals around an atomic nucleus. We have also discussed the fact that the inner orbitals are filled before the outer orbitals, since this constitutes the lowest energy state of the whole atom. An atom in this state is said to be in its ground state, since it cannot have an electron configuration which will produce a lower energy. However, this is not to say that any particular atom at a given moment of time will be at its ground state, since interatomic collisions or interactions with electromagnetic radiations may have raised the energy of one of its electrons so that it is able to take up an orbital of higher energy – the electron will move further away from the nucleus. This process is called excitation of the atom. The excited electron is able to return to its original orbital and releases a quantum of electromagnetic radiation in the process. The energy of this quantum is equal to the energy difference between the excited state and the ground state. Alternatively, an orbiting electron may receive sufficient energy to be able to escape from the atom completely – this might happen as a result of the interaction of a photon with the electron. This process is called ionization since the remaining atom will now form a positive ion. Both processes are shown diagrammatically in Figure 18.7.
18.7 Binding energy of the electron shells
Because the various electron shells are positioned at different distances from the atomic nucleus, they experience different forces of attraction from the nucleus. The K-shell is closest to the nucleus and so experiences the greatest force of attraction from the nucleus. It is therefore most difficult to remove an electron from the K-shell – the K-shell has the highest electron binding energy. The electron binding energy of a shell is the amount of work which must be done to remove an electron from that shell (it is normally stated in KeV or eV). The L-shell is further away from the nucleus and so experiences less force of attraction. It also experiences some repulsion from the electrons in the K-shell. For this reason, the binding energy of the electrons in the L-shell is less than the binding energy of the K-shell for a particular atom. Thus we can say that there is a reduction in the binding energy as we move from the K-shell to the L-shell to the M-shell, etc., within a particular atom.
The reason for the existence of the binding energy is the electrostatic attraction between the nucleus and the electrons in the shells. Thus we would expect to find an increase in the K-shell binding energy with an increase in the atomic number of the element. This is found to be the case in practice.
A knowledge of the binding energy of the various electron shells in different elements is important in radiation physics for the following reasons:
• A knowledge of the binding energies of the different shells allows us to predict the energies of the characteristic radiations which the atom might produce (see Ch. 21 for further details).
• A knowledge of the binding energies of different electron shells within an atom of an element will allow us to determine the likely position of absorption edges during photoelectric absorption (see Ch. 23 for further details).
• If we know the energy of the characteristic radiation from an element and we know the position of absorption edges when radiation is attenuated by this material, then this allows us to explain why elements are relatively transparent to their own characteristic radiation (see Ch. 23 for further details).
Although the binding energy of an electron orbital increases as we move closer to the nucleus, its potential energy reduces. Thus, if an electron moves from the L-shell to the K-shell, it must lose energy (normally in the form of a photon of characteristic radiation). This will be discussed in more detail in Chapter 23.
In this chapter, you should have learnt the following:
• The main subatomic particle which can be joined together to produce matter (see Sect. 18.2).
• A basic planetary model of the atom which consists of a nucleus containing protons and neutrons and has electrons in specific orbitals around this nucleus (see Sect. 18.2).
• The basic structure of the atomic nucleus and the reasons for its stability or otherwise (see Sects 18.3 and 18.3.1).
• The configuration of electron orbitals for differing atoms and the factors which determine the maximum number of electrons in an orbital (see Sects 18.4 and 18.5).
• The basic structure of the periodic table of elements and the electron configuration of elements (see Sect. 18.5).
• The meaning of the term ground state when applied to an atom and the consequences of raising the energy of the electrons above the ground state (see Sect. 18.6).
• An explanation of the term binding energy and a brief outline of areas where a knowledge of the binding energy of electron shells is of importance in radiation physics (see Sect. 18.7).