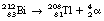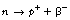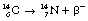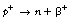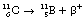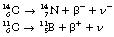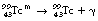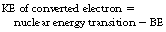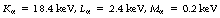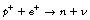Chapter 19 Radioactivity
Chapter contents
19.1 Aim 127
19.2 Introduction 127
19.3 Nuclide chart 128
19.4 Alpha decay or alpha-particle emission 129
19.5 Beta decay or beta-particle emission 129
19.6 Gamma decay or gamma-ray emission 132
19.7 Electron capture 134
19.8 Branching decay programmes 135
19.9 Fission 135
19.10 Summary of radioactive nuclear transformations 137
Further reading 137
19.1 Aim
The aim of this chapter is to discuss the various types of radioactive decay which can occur. Within the chapter the relevance of these processes to nuclear medicine will be considered.
19.2 Introduction
Chapter 18 referred to various terms which may be used to define nuclear structure. This chapter will examine the changes which may take place in the nucleus during radioactive decay.
The term radioactive is applied to nuclei which are unstable. In these nuclei, the forces disrupting the nucleus are stronger than the forces which hold the nucleus together. The instability of the nucleus is demonstrated by the fact that it changes its internal structure to a more stable form, often (but not always) ejecting a charged particle from the nucleus in the process. Each time a nucleus changes its structure it is called a radioactive disintegration or a nuclear transformation and may result in a change in the atomic number – number of protons in the nucleus – (and therefore element) or a change in the mass number – number of protons and neutrons in the nucleus – or a change of both the atomic number and the mass number.
A pictorial representation of the decay process (see Fig. 19.1) is called a decay scheme. In addition to this, it is possible for a nucleus to undergo more than one type of transformation (see Fig. 19.12 later in this chapter) and this type of decay is called a branching scheme.
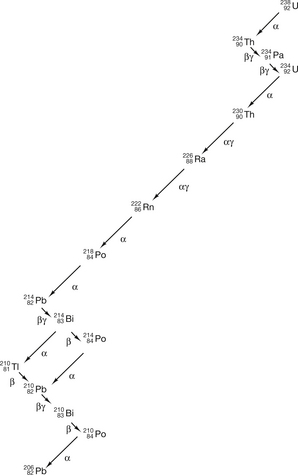
Figure 19.1 The uranium decay series. Note that arrows to the right indicate an increase in the atomic number while arrows to the left indicate a decrease. The downward direction of the arrows indicates that the nucleus loses energy with each transformation (downward direction not drawn to scale).
It is impossible to determine the exact time when a particular nucleus will transform, but the laws of probability may be used to determine the behaviour of a large number of nuclei (see Ch. 20 for a discussion of radioactive decay and the exponential law).
The unit of radioactivity is the becquerel (Bq) where 1 becquerel is 1 nuclear disintegration per second.
Before the introduction of International System of Units (SI) units, radioactivity was measured in curie (Ci). 1 curie was equal to 3.7×1010 nuclear disintegrations per second. Thus, an activity of 1 Ci is the same as an activity of 3.7−1010 Bq or 37 GBq. From this it can be seen that the curie was a much larger unit than the becquerel.
It is often useful to know the specific activity of a sample. This is the activity of radionuclide per unit mass of the sample and so is measured in Bq.kg−1 or submultiples thereof.
19.3 Nuclide chart
It is often useful to draw a graph, plotting the number of neutrons in a nucleus against the number of protons, as all nuclides may be included in this nuclide chart. Such a chart is shown in simplified form in Figure 19.2. Note that isotopes (lines of equal atomic number) are given by any vertical line on the figure. In such a graph, an angle of 45° to the x-axis represents a situation where the nucleus contains equal numbers of protons and neutrons (Z=N).
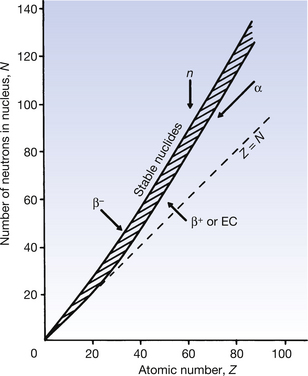
Figure 19.2 A graph of the neutron number (N) plotted against the atomic number (Z) showing the position of the stable nuclides as a shaded band. Note that, as the atomic number increases, a higher proportion of neutrons to protons is required to achieve stability. Also shown are the directions which a nucleus takes for various forms of radioactive decay (these movements are not to scale). EC, electron capture.
It is found on such a graph that there is a broad band of nuclides with low atomic numbers at about 45° to the x-axis and these are all stable or only weakly radioactive. Thus, for the lighter elements, nuclear stability can be produced with equal numbers of protons and neutrons. From the graph it can be seen that, as the atomic number increases, a proportionately larger number of neutrons is necessary to produce nuclear stability. Thus we can say that as the Coulomb repulsion (see Sect. 6.4) between the protons increases, then more neutrons are required to produce the short-range nuclear forces (see Sect. 18.3.1), thus producing the cohesive forces required for a stable nucleus. Nuclei which have atomic numbers greater than 83 (bismuth) are so large that it is impossible to produce a stable nuclear configuration.
Nuclides whose combination of neutrons and protons means that they land outside the band of stability shown in Figure 19.2 have nuclei possessing higher energies than those within the band. As a consequence, such a nucleus is unstable and tends, on decay, to produce a new nucleus of lower energy which is closer to, or within, the stable band. The energy difference between the nucleus before and after decay is emitted either as a charged particle or as a quantum (or quanta) of electromagnetic radiation. The decay scheme towards stability may be in the form of a single step (e.g. 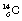 decays to stable
decays to stable 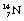 1 by the emission of a beta-particle (see Fig. 19.4) or it may be in the form of a multistage route involving many nuclear transformations (e.g. the decay of
1 by the emission of a beta-particle (see Fig. 19.4) or it may be in the form of a multistage route involving many nuclear transformations (e.g. the decay of 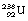 to
to 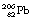 involves at least 14 steps, as shown in Figure 19.1). As a general rule, the greater the mass number of the nuclide, the more complicated the decay path to eventual stability.
involves at least 14 steps, as shown in Figure 19.1). As a general rule, the greater the mass number of the nuclide, the more complicated the decay path to eventual stability.
The effect of different decay modes is illustrated using arrows on Figure 19.2 The direction of the arrow shows the direction of change of position on the chart before and after the decay process. You may find it helpful to refer back to this chart while studying the decay processes in more detail in the following sections of this chapter.
19.4 Alpha decay or alpha-particle emission
For the spontaneous emission of an alpha-particle from a nucleus, the nuclide must have an atomic mass number greater than 150. The nucleus must also have too few neutrons for the number of protons – a higher neutron-to-proton ratio would be required to produce nuclear stability. The alpha-particle consists of two protons and two neutrons tightly bound together (a helium nucleus). It may be considered as a free particle having high kinetic energy which is trapped in the parent nucleus. Thus the daughter nucleus has two protons and two neutrons fewer than the parent nucleus (see Equation 19.1 below).
The mechanism of production of the alpha-particle is quite complex. As we have already identified (see Sect. 18.3.1), the nucleus depends on a balance of disruptive electrostatic (Coulomb) forces and attractive forces between the nucleons caused by the short-range nuclear forces. In very large nuclei there is a large amount of electrostatic repulsion between the protons which extends across the whole nucleus. This is balanced by the short-range nuclear force which exists between adjacent nucleons. Thus, if the nucleus becomes elongated, the electrostatic forces dominate and the nucleus becomes even more elongated. This process continues until the nucleus divides into two fragments, the daughter nuclide and the alpha-particle.
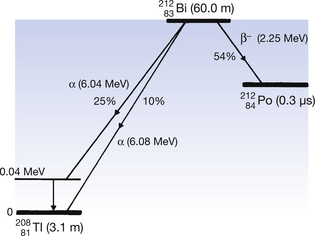
An example of alpha-decay is the decay of bismuth-212 to thallium-208 with the emission of alpha-particles. This process is shown in the following equation:
Note that there is a reduction of four in the atomic mass number and two in the atomic number between parent and daughter product.
This reaction can also be written as shown in Figure 19.3 (the decay of 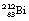 also includes the emission of a beta-particle and is discussed later as an example of a branching programme; for simplicity only the alpha-particle reaction is shown in Fig. 19.3 and the percentages shown refer only to the alpha-particles). Because the daughter product is to the left of the parent nuclide, this shows that there is a reduction in the atomic number. The reaction also shows that the difference in energy between the parent nuclide and the daughter nuclide is 6.08 MeV. If we consider only the alpha-particles emitted, we find that approximately 71% of these have an energy of 6.04 MeV and 29% have an energy of 6.08 MeV. In the case of the first group of alpha-particles, the nucleus is left in an excited state with excess energy of 0.04 MeV which it emits as a gamma-ray.
also includes the emission of a beta-particle and is discussed later as an example of a branching programme; for simplicity only the alpha-particle reaction is shown in Fig. 19.3 and the percentages shown refer only to the alpha-particles). Because the daughter product is to the left of the parent nuclide, this shows that there is a reduction in the atomic number. The reaction also shows that the difference in energy between the parent nuclide and the daughter nuclide is 6.08 MeV. If we consider only the alpha-particles emitted, we find that approximately 71% of these have an energy of 6.04 MeV and 29% have an energy of 6.08 MeV. In the case of the first group of alpha-particles, the nucleus is left in an excited state with excess energy of 0.04 MeV which it emits as a gamma-ray.
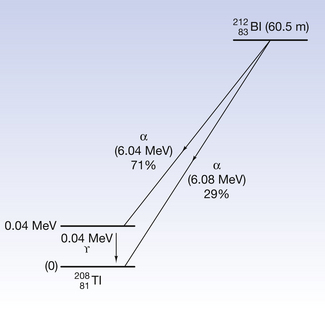
Figure 19.3 An example of alpha-particle emission. The nucleus decreases its atomic number and energy as shown.
The ejection of an alpha-particle means that, to preserve momentum, the nucleus must also recoil with an equal and opposite momentum to that of the alpha-particle. The energy of the recoiling nucleus is typically about 2% of that of the emitted alpha-particle. (Recoil also exists for beta-decay but is much smaller due to the tiny mass of the beta-particle.)
Alpha-particles are intensely ionizing but have a very short range in tissue so have little practical application in radiology.
19.5 Beta decay or beta-particle emission
In the process of beta decay, a particle, having a mass equal to that of an electron, is ejected from the nucleus. The ejected particles, however, may have either a positive or negative charge and so, although they are known collectively as beta-particles, negative beta-particles (β−) or negatrons and positive beta-particles (β+) or positrons both exist. Although the negatron is exactly the same as an electron, in this and subsequent chapters the term negatron will be used to describe the particle which exits from the nucleus of an atom while the term electron will be used to describe particles which orbit the nucleus of the atom. Because the processes are different for the production of the negatron and the positron, they will be dealt with under separate headings.
19.5.1 Negatron (β−) emission
As we can see from Figure 19.2, β−-particles are emitted from nuclei which have too many neutrons for nuclear stability. As we saw in Chapter 18, nucleons are being constantly changed from proton to neutron and back within the atomic nucleus. A neutron may be thought of as consisting of a proton and a negatron:
It appears that in nuclei which have too many neutrons, there is a finite possibility that a neutron becomes isolated within the nucleus and then decays to form a proton and a negatron. The proton rejoins the nucleus and the negatron is ejected. This transformation results in the atomic mass number remaining unchanged (the combined number of protons and neutrons is still the same) but the atomic number will increase by one as one extra proton has been added to the nucleus (this results in the formation of a different element with the consequent rearrangement of electron orbitals to suit the new element).
An example of β−-particle decay is shown below, where carbon-14 decays to form nitrogen with the emission of a β−-particle:
This can also be illustrated using the methods discussed for alpha-particle emission to show the transformation from parent to daughter product. Such a diagram is shown in Figure 19.4. Note that this time there is an increase in the atomic number so the line is down and to the right. This simple decay process is an example of pure beta emission since no other transformations are involved.
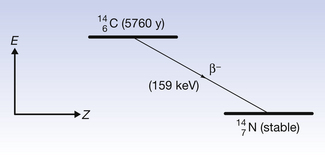
Figure 19.4 An example of pure beta-particle (negatron) emission. The nucleus increases in atomic number and loses energy as shown.
Figure 19.5 shows a more complex emission pattern where the parent and daughter nuclei are separated by an energy difference of 2.81 MeV. The β−-particle has an energy of 0.31 MeV and so the nucleus is left in an excited state – 2.50 MeV above its ground state. The nucleus emits this energy in the form of two gamma-rays, one of energy 1.17 MeV and the other of energy 1.33 MeV. (This will be discussed further when we consider gamma-ray emission in Sect. 19.6.)
19.5.2 Positron (β−) emission
As mentioned earlier, protons are continuously changing into neutrons and back. In these reactions we can consider a proton as consisting of a neutron and a positron.
If we consider Figure 19.2, we see that positron emission takes place from nuclei which have too many protons to achieve stability. In such cases, protons appear to become isolated within the nucleus and then to decay to form a neutron which rejoins the nucleus and a positron which is ejected from the nucleus. In this reaction, the atomic mass number will remain the same but the atomic number will decrease by one (the total number of protons and neutrons is unchanged but one proton has been converted into a neutron).
An example of such a reaction is the decay of carbon-11 to boron:
If we consider the energy changes which take place within the nucleus, as shown in Figure 19.6, we see that the situation regarding energy of the β+-particle is not as simple as it is for the β−-particle. The energy difference between the parent and daughter nucleus in this case is 1.99 MeV. An energy loss of 2mc2 is required to create the positron and allow it to escape from the nucleus – where m is the mass of the positron and c is the velocity of electromagnetic radiation. As the mass of the positron is the same as the mass of the electron, then 1.02 MeV is required to create the positron and eject it from the nucleus and any remaining energy (in this case 0.97 MeV) is given to the positron as kinetic energy.
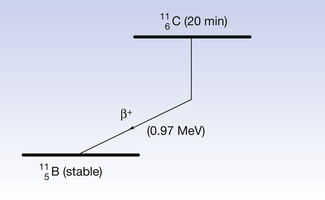
Figure 19.6 An example of positron decay. The nucleus experiences a decrease in the atomic number and loses energy, as shown.
A negatron or a positron is influenced by the electrostatic forces which exist between it and the nucleus until it is able to escape from the atom. As a result, the kinetic energy of the β−-particle is reduced by the force of attraction which exists between it and the nucleus, while the kinetic energy of the β+-particle is increased by electrostatic repulsion. Energy and momentum are conserved in this process.
19.5.3 The fate of the positron
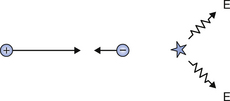
An energetic positron emitted by a nucleus will move through the surrounding atoms and will lose kinetic energy because of collisions with them. As its momentum decreases, it is more likely to interact with a free electron in the material. The positron is the antiparticle of the electron and when they meet they completely annihilate each other to form two gamma-rays of annihilation radiation. This process is shown diagrammatically in Figure 19.7, where the annihilation radiation consists of two photons, each of energy 0.511 MeV, ejected at 180° to each other. In this way, both energy and momentum are conserved. The mutual annihilation of the electron and the positron is an example of the principle of mass–energy equivalence (see Sect. 16.4.1) as given by Einstein’s equation E=mc2, and shows that each particle has a mass–energy of 0.511 MeV, in agreement with Table 18.1.
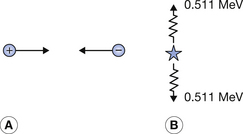
Figure 19.7 (A) A collision between a positron and an electron where both have a minimal kinetic energy. (B) The annihilation radiation where two photons, each of 0.511 MeV, are produced at 180° to each other.
The annihilation radiation produced by the reaction may now interact with neighbouring atoms by the processes of Compton scattering and photoelectric absorption (see Ch. 23) and may produce characteristic radiation and Auger electrons (see Sect. 19.6.3) by processes which will be discussed later in this chapter.
The positron and the electron may annihilate each other when the positron still has considerable kinetic energy. In this case, the two quanta are not emitted at 180° to each other and each has an energy higher than 0.511 MeV. The excess of energy of the positron is divided equally between each quantum. In each case, energy and momentum are conserved. A diagrammatic representation of the process is shown in the figure.
19.5.4 The neutrino
Consider the carbon-14 reaction which we discussed earlier. Here the carbon-14 decayed to nitrogen where the difference in energy between the parent and the daughter nucleus was 159 keV. As only beta decay takes place, we could confidently expect that all the β−-particles should have an energy of 159 keV. If we plot a graph of the energy of the beta-particles against the numbers emitted, we get a graph similar to the β− graph shown in Figure 19.8. The maximum or end-point energy of the β−-particles in this case is 159 keV. The graph means that, although only beta decay takes place and although the difference between the parent and the daughter nucleus is 159 keV, some of the β−-particles have energies less than 159 keV. Such a situation would appear to suggest that this does not obey the law of conservation of energy (see Sect. 16.4). This difficulty was overcome by Wolfgang Pauli in 1933 when he postulated that another particle – the neutrino – is always ejected with a beta-particle. He suggested that a neutrino (symbol ν) is ejected at the same time as a positron and that an antineutrino (symbol ν−) at the same time as a negatron. The total energy of the emitted negatron and antineutrino corresponds to the energy difference between the parent and daughter nucleus. How this energy is shared between the two particles will differ for each decay of the nucleus. In this way, a continuous distribution of energies is obtained both for the β−-particle and the antineutrino as shown in Figure 19.8.
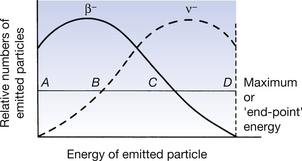
Figure 19.8 The sharing of energy between the beta-particle and the neutrino in beta decay – the case shown is for negatron emission.
Because of the presence of the neutrino and antineutrino, we now need to modify some of the equations we considered earlier:
Although Pauli postulated the existence of neutrinos and antineutrinos in 1933, the presence of these particles proved difficult to detect until much later. There is now conclusive proof of their existence because of experiments performed using nuclear reactors and particle accelerators. The neutrino and the antineutrino differ in their direction of spin relative to their direction of motion – the neutrino spins anticlockwise and the antineutrino spins clockwise. They are antiparticles and if they are made to collide will produce electromagnetic radiation in the form of annihilation radiation. The reason the particles are so difficult to detect can be seen by considering their three major properties:
Because of these properties, it is extremely difficult to detect the presence of neutrinos (or antineutrinos) – they have a half-value thickness (see Sect. 20.8) of many miles in lead!
19.6 Gamma decay or gamma-ray emission
Gamma-rays are part of the electromagnetic spectrum and are similar in many ways to X-rays. Gamma-rays are emitted from a nucleus which has excess energy whereas X-rays are from electrons as they lose energy (this will be further discussed in Ch. 21). It is possible for gamma-rays to have lower energies than X-rays and vice versa. However, the maximum energy possible from gamma-rays exceeds that of X-rays.
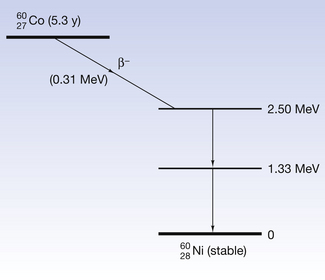
We discussed in this chapter (see Sect 19.5.1) the fact that cobalt-60 decayed with the emission of a β-particle but the daughter nucleus was left in an excited state. The situation for this daughter nucleus is depicted in Figure 19.9A. The figure illustrates the energy states of the daughter nucleus by means of horizontal lines above the thick line representing the ground state of the nucleus. In this case, the 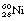 nucleus is left at an energy 2.5 MeV above its ground state. It immediately decays to its ground state in two jumps:
nucleus is left at an energy 2.5 MeV above its ground state. It immediately decays to its ground state in two jumps:
1. It decays to 1.33 MeV above its ground state by the emission of a 1.17 MeV gamma-ray.
2. It then drops to its ground state by the emission of a second gamma-ray of energy 1.33 MeV.
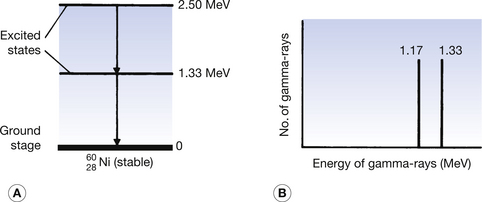
Figure 19.9 (A) The excited states of a nickel-60 nucleus which is left in an excited state as the result of the previous decay of a cobalt-60 nucleus. This decays to its ground state by the emission of two gamma photons, each representing the difference in energy between the excited states. (B) The line spectrum produced by the gamma radiation.
Figure 19.9B shows the line spectra of the gamma radiation emitted by the nucleus where each gamma-ray has a precise energy corresponding to the discrete energy transformations within the nucleus.
19.6.1 Metastable states and isomeric transitions
In the case of 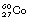 decaying to
decaying to 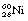 , the nucleus remains in an excited state before the emission of the gamma-rays for a time so short that it is incapable of accurate measurement. However, this is not always the case, and those excited states which last sufficiently long for their durations to be measured are known as metastable states. The transition from a metastable state to a more stable state is known as an isomeric transition. Such metastable radionuclides prove to be useful in nuclear medicine because of the low dose which they deliver to the patient – the patient gets no dose from β−-particles emitted before metastability. Technetium-99m (usually written
, the nucleus remains in an excited state before the emission of the gamma-rays for a time so short that it is incapable of accurate measurement. However, this is not always the case, and those excited states which last sufficiently long for their durations to be measured are known as metastable states. The transition from a metastable state to a more stable state is known as an isomeric transition. Such metastable radionuclides prove to be useful in nuclear medicine because of the low dose which they deliver to the patient – the patient gets no dose from β−-particles emitted before metastability. Technetium-99m (usually written 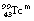 or, less commonly,
or, less commonly, 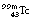 ) is a radionuclide which is widely used in nuclear medicine. A simplified decay scheme for it is shown in Figure 19.10 and it will be discussed in more detail Chapter 37 which deals with radionuclide imaging. The half-life of
) is a radionuclide which is widely used in nuclear medicine. A simplified decay scheme for it is shown in Figure 19.10 and it will be discussed in more detail Chapter 37 which deals with radionuclide imaging. The half-life of 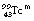 is 6 hours and it emits a gamma-ray of energy 140 keV when it decays to
is 6 hours and it emits a gamma-ray of energy 140 keV when it decays to 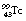 . The half-life of
. The half-life of 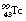 is so long (2.1×105 years) that it can be considered stable for all practical purposes.
is so long (2.1×105 years) that it can be considered stable for all practical purposes.
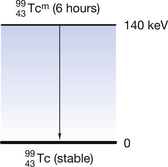
Figure 19.10 An example of the decay of technetium from a metastable to a stable state with the emission of a 140 keV gamma photon.
The reaction can be written as:
The next section of this chapter considers the possible effects of gamma emission from the nucleus on the whole atom rather than on the nucleus only.
19.6.2 Internal conversion
When a nuclide is decaying by gamma emission, there is a competing process within the atom called internal conversion. This process results in electrons with discrete energies (unlike the continuous spectrum of energies of negatrons) being emitted from the atom. The process is a result of a direct interaction between the nucleus and an orbiting electron such that the nucleus is able to drop to its ground state by giving all of its excess energy to the electron.
The innermost electrons of the atom have orbitals which pass close to or even through the nucleus. Thus it is a matter of statistical probability whether the excess energy of the nucleus will result in a gamma-ray emission from the nucleus or electron emission from the inner shells. The K-shell is situated closest to the nucleus so is most likely to participate in such a reaction followed by the L-, M-shells, etc. The converted electron escapes from the atom with an energy equal to the energy donated by the nucleus minus the binding energy of that particular electron. This can be stated:
where KE is kinetic energy and BE is binding energy of the electron.
The internal conversion coefficient, α, is defined as the ratio of the number of nuclear transformations which result in internal conversion to the number which result in gamma-ray emission. Thus, α may take a value between 0, resulting in no internal conversions, and infinity, corresponding to complete internal conversion. This can be illustrated by considering the figures for the decay of 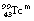 to
to 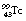 . Approximately 9% of the nuclear transitions result in internal conversions of electrons from the K-shell, 1.1% from the L-shell and 0.3% from the M-shell. This means that 10.4% of the nuclear transitions result in internal conversions and 89.6% result in gamma-ray emission. This gives an internal conversion coefficient (α) for
. Approximately 9% of the nuclear transitions result in internal conversions of electrons from the K-shell, 1.1% from the L-shell and 0.3% from the M-shell. This means that 10.4% of the nuclear transitions result in internal conversions and 89.6% result in gamma-ray emission. This gives an internal conversion coefficient (α) for 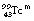 of 0.116.
of 0.116.
19.6.3 X-rays and Auger electrons
If a radioactive decay results in a vacancy occurring within one of the inner electron shells of the atom, then electrons from orbitals further away from the nucleus will perform quantum jumps inwards until there are no inner shell vacancies. Each such quantum jump will result in the emission of electromagnetic radiation from the atom equal to the energy difference between the two shells. For inner-shell transitions, the energy of this electromagnetic radiation may be in the X-ray part of the spectrum. Such radiation is known as fluorescent radiation and its energy is characteristic of the atom concerned. If we consider the internal conversion process which we have just described and take a situation where a K-shell electron is removed from the atom, the vacancy thus created in the K-shell may be filled from the L-shell (a Kα transition) or from the M-shell (a Kβ transition). Such a quantum jump would be accompanied by the emission of a photon of electromagnetic radiation equivalent to the energy difference between the two shells. If it was a Kα transition, there would now be a vacancy in the L-shell and this might be filled from the M-shell, etc. This would again be accompanied by the emission of a photon of electromagnetic radiation equal to the energy difference between the L- and M-shells. This process continues until the atom is able to capture a free electron to fill the vacancy – usually in one of its outermost shells. Until this occurs the atom contains more protons than electrons and so is regarded as a positive ion.
This already rather complex situation is further complicated by the fact that some of the photons emitted by such transitions may have sufficient energy to interact with other electrons in the atom and to remove them from their orbitals and eject them from the atom – this process is called the photoelectric effect and may occur if the photon energy is greater than the binding energy of the electron (see Ch. 23 for a more detailed description). The electrons thus ejected are called Auger electrons and have discrete energies equal to the photon energy minus the binding energy of the electron. The ejection of an Auger electron from a shell leaves a vacancy in that shell which will be filled by electrons jumping down from orbitals even further away from the nucleus, with the release of more fluorescent radiation and perhaps the release of even more Auger electrons.
From the above we can see that the ejection of an electron from one of the orbitals by internal conversion may result in a complicated sequence of orbital quantum jumps accompanied by the release of photons of electromagnetic radiation and the ejection of Auger electrons from the atom. The term fluorescent yield is used to describe the fraction of the electron transitions which result in the production of fluorescent radiation.
If we consider 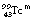 from the previous discussion, you may remember that 11.6% of the energy from the nucleus results in internal conversion (α=0.116). Of the resulting electron quantum jumps, 80% of the transitions result in fluorescent radiation and 20% result in the production of Auger electrons. Thus the fluorescent yield for technetium is 0.8. Some typical fluorescent radiation energies for
from the previous discussion, you may remember that 11.6% of the energy from the nucleus results in internal conversion (α=0.116). Of the resulting electron quantum jumps, 80% of the transitions result in fluorescent radiation and 20% result in the production of Auger electrons. Thus the fluorescent yield for technetium is 0.8. Some typical fluorescent radiation energies for 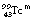 are:
are:
Note: The fact that the electrons emitted by this process carry discrete amounts of energy makes it easy to distinguish them from negatron emission where there is a continuous spectrum of energies.
For each decay, the total energies for all the fluorescent radiation and of all the electrons emitted from the shells of the atom equal the energy lost by the nucleus in the nuclear transition. This is because the binding energy of each ejected electron is recovered in the fluorescent radiation emitted in the subsequent cascade of orbital jumps. Thus the whole process obeys the law of conservation of energy (see Sect. 3.3).
19.7 Electron capture
If a nucleus of low mass number has too few neutrons for stability but has insufficient excess energy (<1.02 MeV) to eject a positron, then an alternative way by which the nucleus may undergo an isobaric transformation and lose energy is electron capture (shown in Fig. 19.11). In this process, the nucleus captures one of the orbiting electrons, the most likely being a capture of a K-shell electron. Sometimes the terms ‘K capture’ and ‘L capture’ are used to denote the shell from which the electron was captured. A situation where electron capture is the only process involved is shown in Figure 19.11A where 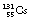 decays to
decays to 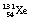 .
.
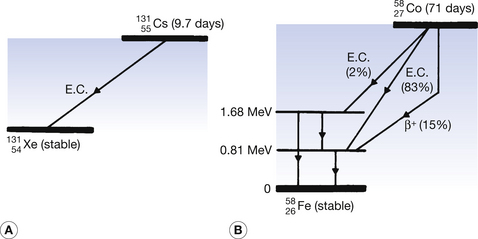
Figure 19.11 (A) Pure electron capture (EC) decay. (B) Electron capture and positron decay occurring in the same nuclide (85% EC and 15% positron decay). Also emitted are the X-rays from the iron-58 atoms and the 0.511 MeV annihilation radiations from the positron–electron annihilations.
In the diagram, the atomic number is reduced by one because the capture of an electron by the nucleus results in one of the protons in the nucleus changing into a neutron. Also note that during the process of electron capture, a neutrino is emitted by the nucleus. The processes involved in electron capture may be shown by Equation 19.9:
In situations where a low-mass nucleus has too few neutrons and an excess energy greater than 1.02 MeV, then the process of electron capture may compete with the process of positron emission. An example of such a competing process is shown in the decay of 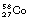 into stable
into stable 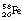 in Figure 19.11B.
in Figure 19.11B.
Note that both the process of electron capture and the process of positron emission involve the conversion of a proton to a neutron. Thus the atomic mass number remains unaltered but the atomic number is reduced by one.
As we mentioned earlier in this chapter (Sect. 19.6.2), creating a vacancy in an inner electron shell will result in the emission of characteristic fluorescent radiation from the atom. It is interesting to note that in the case of electron capture this is characteristic of the daughter product. This means that the electron orbitals of the daughter product are established before the consequent electron transitions occur.
19.8 Branching decay programmes
If the nucleus is very large, it is possible that it may disintegrate in more than one way. We have already encountered this – although it was not described in detail – in the uranium series in Figure 19.2. In this case, 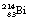 can either decay to
can either decay to 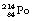 by β−-particle emission or it can decay to
by β−-particle emission or it can decay to 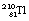 by alpha-particle emission.
by alpha-particle emission.
Another isotope of bismuth, 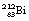 , decays in a similar branching programme. The decay process for this isotope involves the emission of alpha-particles of energies 6.04 and 6.08 MeV, gamma-rays of energy 0.04 MeV and β−-particles of maximum energy 2.25 MeV. The initial decay scheme for the
, decays in a similar branching programme. The decay process for this isotope involves the emission of alpha-particles of energies 6.04 and 6.08 MeV, gamma-rays of energy 0.04 MeV and β−-particles of maximum energy 2.25 MeV. The initial decay scheme for the 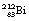 nuclide is shown in Figure 19.12. Note that, for simplicity, only the first disintegrations are shown. As can be seen from the diagram, both
nuclide is shown in Figure 19.12. Note that, for simplicity, only the first disintegrations are shown. As can be seen from the diagram, both 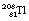 and
and 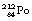 have short half-lives and so are also radioactive.
have short half-lives and so are also radioactive.
19.9 Fission
As we mentioned in our discussion of alpha decay (see Sect. 19.4), the short-range forces holding the nucleus together exist between adjacent nucleons, whereas the Coulomb forces act across the whole of the nucleus. This fact becomes increasingly important as the size of the nucleus increases. A very large nucleus may be pictured as being rather like a liquid drop in which the nucleons are moving about with very high energy and continuously deforming the shape of the nucleus. During this process, it is possible for the nucleus to become very elongated and then to break into two fragments – usually of fairly similar sizes. Such a phenomenon is known as spontaneous fission and can occur for very large nuclei, e.g. thorium-232 is capable of spontaneous fission. As well as the fission fragments from such a reaction, one or more neutrons are usually liberated and the whole fission process is accompanied by the release of large amounts of energy.
Neutron-activated fission is a more controllable process than spontaneous fission. This occurs as a result of a heavy nucleus capturing an incoming neutron and then breaking into large fragments in a similar way to spontaneous fission. An example of such a reaction is the disintegration of 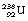 into two fission fragments of
into two fission fragments of 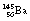 and
and 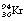 if the uranium-238 nucleus is made to absorb a neutron. The fission of the nucleus is normally accompanied by the release of gamma-rays and neutrons. Both of the fissile fragments are extremely rich in neutrons and so each will usually release a neutron. The neutrons from both of the above processes can now react with two
if the uranium-238 nucleus is made to absorb a neutron. The fission of the nucleus is normally accompanied by the release of gamma-rays and neutrons. Both of the fissile fragments are extremely rich in neutrons and so each will usually release a neutron. The neutrons from both of the above processes can now react with two 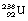 nuclei, resulting in the release of four fissile fragments and four neutrons, and so a chain reaction can be set up. Such a process is accompanied by the release of large amounts of energy (see Insight).
nuclei, resulting in the release of four fissile fragments and four neutrons, and so a chain reaction can be set up. Such a process is accompanied by the release of large amounts of energy (see Insight).
On average, a uranium-238 nucleus will release an energy of 200 MeV on fission. If we consider the complete fission of a 1-kg mass of uranium (this is about the size of a golf ball), we need to consider the energy released by all the uranium atoms in a 1-kg mass. By applying Avogadro’s equation (see Sect. 3.6) to this we can calculate that 1 kg of uranium-238 contains 2.5×1024 atoms. If each of these atoms releases 200 MeV of energy, the total energy released will be 5.1×1027 MeV. This is the same as 8.1×1013 joules of energy.
A pictorial representation of the neutron-activated fission process is shown in Figure 19.13. The incoming neutron is captured in Figure 19.13A and delivers sufficient energy to the nucleus (Fig. 19.13B) to elongate its shape into an ellipsoid. As mentioned earlier, the Coulomb forces across the nucleus are now at an advantage over the short-range nuclear forces and the nucleus further distorts to form a ‘peanut’ shape (Fig. 19.13C) and eventually breaks to form two fragments because of the electrostatic repulsion between the two main nuclear masses. The two fragments fly apart (their kinetic energy accounts for about 80% of the total disintegration energy) and several neutrons and gamma-ray photons are usually emitted (Fig. 19.13D). The neutrons emitted by this reaction may now interact with other atoms to cause fission and the release of further neutrons and so a chain reaction is set up, with the consequent liberation of large amounts of energy. The fissile fragments (Fig. 19.13E) are themselves rich in neutrons for their atomic numbers and so will undergo further disintegration to move towards more stable nuclei. This initial process results in the production of β−-particles (with associated gamma-rays) which produces a more stable proton/neutron configuration or, if the nucleus is very excited, the ejection of a neutron from the nucleus. These neutrons are known as delayed neutrons to distinguish them from the prompt neutrons which are emitted at the moment of fission.
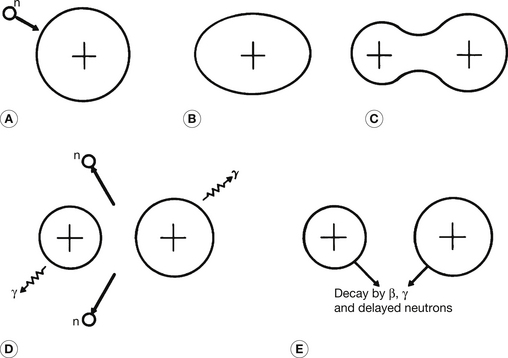
Figure 19.13 A diagrammatic representation of neutron-activated fission. For a fuller explanation of the process, see the text.
Fission products from any type of nucleus are not always the same and may have different relative sizes on each disintegration. Thus a great range of other decay chains is possible from other fissile fragments.
It is also possible to initiate fission in the nucleus by bombarding it with protons (or with alpha-particles) or by striking it with high-energy photons. Examples of these are:
• Copper (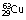 ), if bombarded with protons, can be made to undergo fission to produce sodium (
), if bombarded with protons, can be made to undergo fission to produce sodium (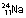 ) and potassium (
) and potassium (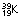 ). This process is accompanied by neutron emission.
). This process is accompanied by neutron emission.
• Uranium-238 (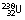 ), if it is bombarded by photons of energy equal to or greater than 5.1 MeV, will undergo fission. This latter process is known as photofission.
), if it is bombarded by photons of energy equal to or greater than 5.1 MeV, will undergo fission. This latter process is known as photofission.
19.10 Summary of radioactive nuclear transformations
Table 19.1 is a summary of the types of radioactive decay discussed so far in this chapter. You may find it useful to refer back to this table when considering the production of artificial radionuclides and also in considering the use of radionuclides in medicine in Chapter 37.
Table 19.1 Summary of the effects of radioactive decay
| EFFECT ON NUCLEUS | ||||||
|---|---|---|---|---|---|---|
| TYPE OF DECAY | SYMBOL | Z | N | A | EFFECT ON ATOM | COMMENTS |
| Alpha | α | Z − 2 | N − 2 | A − 4 | Electron orbits change to that of daughter nucleus | Occurs in elements with a mass number greater than 150. Daughter products may undergo further decay by a variety of the processes mentioned below |
| Negatron emission | β− | Z + 1 | N − 1 | – | Electron orbits change to that of daughter nucleus | Proton changes to neutron in nucleus. Negatron emitted from the nucleus with a spread of energies but the energy of the negatron plus the energy of the antineutrino is constant for a given nuclide. Daughter nucleus may further decay by prompt or delayed gamma-ray emission competing with internal conversion (IC) |
| Positron emission | β+ | Z − 1 | N + 1 | – | Electron orbits change to that of daughter nucleus | Neutron changes to proton in nucleus. The positron is emitted with a neutrino and the sum of their energies is constant for a given nuclide. The process only occurs if the energy loss by the parent nucleus can be greater than 1.02 MeV. Positron is annihilated by collision with electron and two photons of annihilation radiation, each of energy 0.511 MeV are produced. Daughter nucleus may decay by gamma radiation and/or IC |
| Gamma radiation | γ | – | – | – | No effect on the number of nucleons in the nucleus but the process may compete with IC | Produced by the quantum jump from excited state of the nucleus to a lower energy. Excited states of measurable half-life are defined as metastable and the transition is isomeric |
| Internal conversion | IC | – | – | – | Characteristic radiation and Auger electrons emitted | An inner orbital electron of the atom interacts with the nucleus and is ejected from the atom. The electron is given the excess nuclear energy. This creates a vacancy in the shell and results in the emission of characteristic radiation and Auger electrons |
| Electron capture | EC | Z − 1 | N + 1 | – | Characteristic radiation and Auger electrons of the daughter nucleus are emitted | An inner orbital electron is captured by the nucleus and changes a proton to a neutron. A neutrino is emitted, carrying energy changes of the nucleus. The nucleus may decay spontaneously by gamma-ray emission. The vacancy left in the electron orbit results in the emission of characteristic radiation and Auger electrons |
| Fission | f | Size and structure of the fragments may vary | Bonds may be broken or ionization caused by fragment | Fission may be spontaneous or neutron activated. The nucleus splits, producing two or more fragments, gamma-rays and neutrons. Controlled fission is used in nuclear reactors and the neutrons can be used to produce artificial radionuclides (see Ch. 37) | ||
In this chapter you should have learnt:
• The mechanism for alpha decay and alpha-particle emission (see Sect. 19.4)
• Beta decay leading to negatron emission (see Sect. 19.5.1)
• Beta decay leading to positron emission (see Sect. 19.5.2)
• The fate of the positron after emission (see Sect. 19.5.3)
• The fact that a neutrino or an antineutrino forms part of beta emission (see Sect. 19.5.4)
• The mechanism of gamma decay and gamma-ray emission (see Sect. 19.6)
• The meaning of metastable nuclei and isomeric transitions (see Sect. 19.6.1)
• The mechanism of the process of internal conversion (see Sect. 19.6.2)
• The mechanisms involved in the emission of X-rays and Auger electrons from the atoms of radionuclides (see Sect. 19.6.3)
• The mechanism of electron capture and the subsequent changes within the atom (see Sect. 19.7)
• Branching decay programmes (see Sect. 19.8)
• The processes involved in nuclear fission (see Sect. 19.9)
• Summary of radioactive nuclear transformations (See Sect 19.10)