The genitourinary examination
Examination anatomy
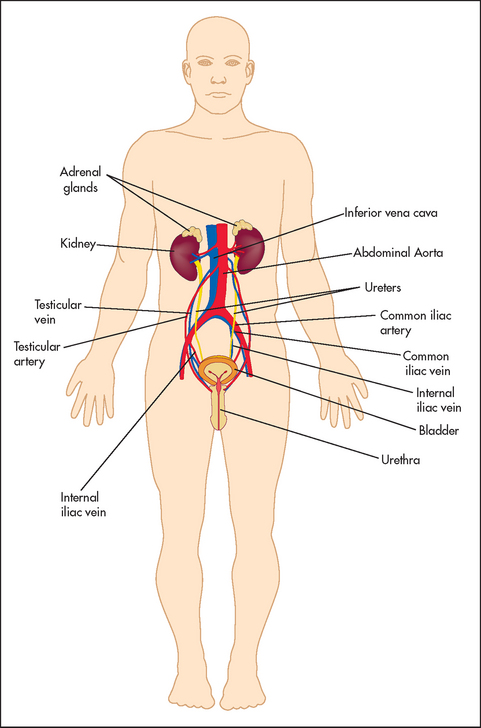
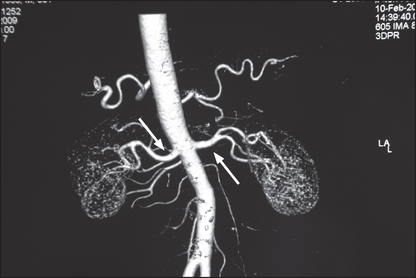
Figure 18.1 shows an outline of the anatomy of the urinary tract. Figure 18.2 shows the arterial supply of the kidneys as demonstrated on a CT renal angiogram and Figure 18.3 shows the outline of the renal collecting system. Problems with function can arise in any part, from the arterial blood supply of the kidneys, the renal parenchyma, the ureters and bladder (including their innervation) to the urethra.
The examination
A set examination of the genitourinary system is not routinely performed. However, if renal disease is suspected or known to be present, then certain signs must be sought. These are mostly the signs of chronic kidney disease (CKD) (uraemia) and its causes (see List 18.1). On the other hand, examination of the male genitalia or female pelvis is part of the routine general examination.
General appearance
The general inspection remains crucial. Look for hyperventilation, which may indicate an underlying metabolic acidosis. Hiccupping may be present and can be an ominous sign of advanced uraemia. There may be the ammoniacal fish breath (uraemic fetor) of kidney failure. This musty smell is not easy to describe but once detected is easily remembered. Patients with CKD commonly have a sallow complexion (a dirty brown appearance or uraemic tinge). This may be due to impaired excretion of urinary pigments (urochromes) combined with anaemia. The skin colour may be from slate grey to bronze, due to iron deposition in dialysis patients who have received multiple blood transfusions, but these signs are becoming less frequent with the use of exogenous erythropoietin. In terminal renal failure, patients become drowsy and finally sink into a coma due to nitrogen or toxin retention. Twitching due to myoclonic jerks, and tetany and epileptic seizures due to neuromuscular irritability or a low serum calcium level, occur late in renal failure. Over-vigorous correction of acidosis (e.g. with bicarbonate infusions) may also precipitate seizures and coma. There may be typical skin nodules related to calcium phosphate deposition.
It is essential to assess the state of fluid balance in all patients with renal disease. Severe fluid-volume depletion can be a cause of AKI and can cause precipitous decompensation in patients with CKD. Conversely, volume excess can result from intravenous infusions of fluid used in an attempt to correct AKI, resulting in pulmonary oedema. Patients should be weighed regularly as an objective measure of their fluid status.
The distinctive ketone-like smell of a urinary tract infection (UTI) may be apparent. There may be evidence of urinary incontinence on the patient’s clothing.
Hands
The patient’s nails should be inspected. Look for leuconychia. Muehrcke’s nailsa refer to paired white transverse lines near the end of the nails; these occur in hypoalbuminaemia (e.g. nephrotic syndrome).1 A single transverse white band (Mees’ linesb; see Figure 18.4) may occur in arsenic poisoning, as well as in renal failure. Half-and-half nails (distal nail brown or red, proximal nail pink or white) are also seen in CKD.
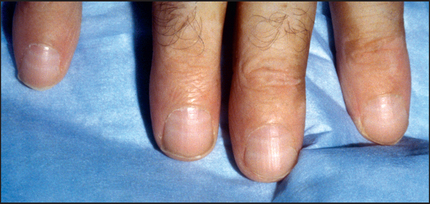
Figure 18.4 Mees’ lines (From McDonald FS, ed. Mayo Clinic images in internal medicine, with permission. © Mayo Clinic Scientific Press and CRC Press.)
Anaemia is common and causes palmar crease pallor. There are a number of causes of anaemia in patients with CKD, including poor nutrition (especially folate deficiency), blood loss, erythropoietin deficiency, haemolysis, bone marrow depression and the chronic disease state.
Asterixis (page 188) may be present in terminal CKD.
Arms
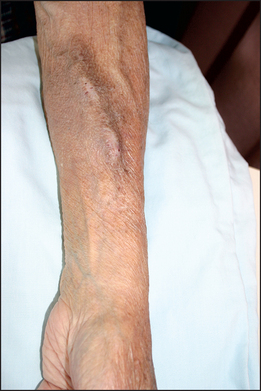
Inspect the patient’s wrists and forearms for scars and palpate for surgically created arteriovenous fistulae or shunts, used for haemodialysis access. There is a longitudinal swelling and a palpable continuous thrill—a characteristic buzzing feel—present over the fistula (see Figure 18.5). There may be scars from previous thrombosed shunts or carpal tunnel syndrome surgery present on either side. Look for signs of the carpal tunnel syndrome.
Bruising occurs because of nitrogen retention, which causes impaired prothrombin consumption, a defect in platelet factor III and abnormal platelet aggregation in CKD. Skin pigmentation is common, reflecting a failure to excrete urinary pigments. Scratch marks and excoriations, due to uraemic pruritus, often associated with hyperphosphataemia, may be present. This occurs commonly and can be extremely debilitating. Uraemic frost is a fine white powder present on the skin where very high concentrations of urea have precipitated out of the sweat in terminal CKD; it is very rare. Evidence of vasculitis, which can cause renal disease, should also be sought.
Look for signs of peripheral neuropathy (page 464) in the limbs. Sensory impairment is more marked than motor impairment initially. Myopathy and bone tenderness can also occur.
Face
The presence of a rash or skin tethering may indicate an underlying connective tissue disease such as systemic lupus erythematosus or systemic sclerosis.
The presence of hearing aids may be consistent with Alport’s syndrome (hereditary nephritis, often with sensorineural hearing loss and eye disease of the retina or cornea).
Inspect the patient’s eyes and look for signs of anaemia and, rarely, jaundice (retention of nitrogenous wastes can cause haemolysis). Band keratopathy is a calcium deposition beneath the corneal epithelium in line with the interpalpebral fissure—it is due to secondary or tertiary hyperparathyroidism, or excessive replacement of calcium in patients with CKD.
A uraemic fetor may be present in the patient’s mouth. This is an ammoniacal, musty odour that results from the breakdown of urea to ammonia in the saliva. Mucosal ulcers can occur as there is a decrease in saliva flow, and patients with CKD are prone to infection (e.g. thrush), due to decreased acute inflammatory responses as a result of nitrogen retention. Transplant patients treated with calcineurin inhibitors (cyclosporin and tacrolimus) frequently develop gingival hyperplasia (thickening of the gums).
Neck
Carefully check the jugular venous pressure to help assess the intravascular volume status. Auscultate for carotid artery bruits; these provide a (rather unreliable) clue that there may be generalised atherosclerotic disease (which can cause renal artery stenosis or complicate CKD). Look for signs of previous jugular vein puncture due to previous vascular access insertion (‘vascath’) for haemodialysis. Surgical scars from previous parathyroidectomy performed for management of tertiary hyperparathyroidism may be present.
Chest
Examine the heart and lungs. In CKD there may be congestive cardiac failure due to fluid retention, and hypertension as a result of sodium and water retention or excess vasoconstrictor activity, or both. Signs of pulmonary oedema may also be present due to uraemic lung disease (a type of non-cardiogenic pulmonary oedema associated with a typical ‘bat’s wing’ pattern on chest X-ray; see Figure 8.5, page 120), volume overload or uraemic cardiomyopathy.
Pericarditis, which can be fibrinous or haemorrhagic in CKD, is secondary to retained metabolic toxins and can cause a pericardial effusion; there may be a pericardial rub or signs of cardiac tamponade. Lung infection is also common due to the immunosuppression present from the CKD itself or as a result of treatment.
The abdominal examination
Abdominal examination is performed as described on page 194. However, particular attention must be paid to the following.
INSPECTION
The presence of a Tenckhoff catheter (peritoneal dialysis catheter) should be noted. It is important to look for nephrectomy scars (see Figure 14.24, page 195). These are often more posterior than one might expect. It may be necessary to roll the patient over and look in the region of the loins. Renal transplant scars are usually found in the right or left iliac fossae. A transplanted kidney may be visible as a bulge under the scar, as it is placed in a relatively superficial plane. Peritoneal dialysis results in small scars from catheter placement in the peritoneal cavity; these are situated on the lower abdomen, at or near the midline.
The abdomen may be distended because of large polycystic kidneys or ascites (as a result of the nephrotic syndrome, or peritoneal dialysis fluid).
PALPATION
Particular care is required here so that renal masses (see List 18.2) are not missed. Remember that an enlarged kidney usually bulges forwards, while perinephric abscesses or collections tend to bulge backwards. Transplanted kidneys in the right or left iliac fossa may be palpable as well. Tenderness over the transplant can be a sign of rejection. Patients with polycystic kidneys may also have a polycystic liver and there may be hepatomegaly as a result of hepatic cysts (see List 18.3). Feel for the presence of an enlarged bladder.2 Also palpate for an abdominal aortic aneurysm. In the patient with abdominal pain, renal colic should be suspected if there is renal tenderness (LR+ 3.6) or loin tenderness (LR+ 27.7).3
BALLOTTING
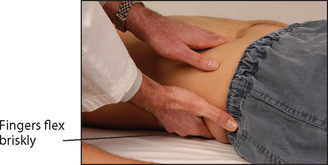
From the French word meaning to shake about, ballotting is an examination technique for palpating the kidney by attempting to flick it forwards. Place one hand under the renal angle and flick your fingers upwards while your other hand, placed anteriorly in the right or left upper quadrant, waits to feel the kidney move upwards and then float down again (see Figure 18.6).
PERCUSSION
This is necessary to confirm the presence of ascites by examining for shifting dullness. Also percuss for an enlarged bladder. Obesity and ascites make direct percussion of the bladder difficult. This is an opportunity to attempt auscultatory percussion.2 Place the diaphragm of the stethoscope just above the border of the symphysis pubis and perform direct percussion of the abdominal wall, starting at the subcostal margin in the middle line. There is a sudden increase in loudness when the upper border of the bladder is reached. It is even possible to estimate the volume of urine in the bladder by this method. An upper border less than 2 centimetres from the stethoscope suggests a fairly empty bladder, while an upper border more than 8 centimetres higher corresponds to a urine volume of between 750 mL and 1 L.
AUSCULTATION
The important sign here is the presence of a renal bruit. Renal bruits are best heard above the umbilicus, about 2 centimetres to the left or right of the midline. Listen with the diaphragm of the stethoscope over both these areas. Next ask the patient to sit up, and listen in both flanks. The presence of a systolic and diastolic bruit is important. A diastolic component makes the bruit more likely to be haemodynamically significant. Its presence suggests renal artery stenosis due to fibromuscular dysplasia or atherosclerosis. Approximately 50% of patients with renal artery stenosis will have a bruit. In a patient with hypertension that is difficult to control, the presence of a systolic/diastolic abdominal bruit has a positive LR for renal artery stenosis of over 40.3 On the other hand, if only a soft systolic bruit is audible, at least half these patients do not have any significant renal artery stenosis. In such cases the aorta or splenic artery may be the source of the sound. The absence of hypertension makes the diagnosis of renal artery stenosis less likely. The occurrence of unexplained pulmonary oedema of sudden onset (‘flash’ pulmonary oedema) in a patient with renal impairment and hypertension makes a diagnosis of renal artery stenosis more likely.
Rectal and pelvic examination
Here the presence of prostatomegaly4,5 in men and a frozen pelvis from cervical cancer in women is important, as this may be a cause of urinary tract obstruction and secondary renal failure.
The back
Strike the patient’s vertebral column gently with the base of your fist to elicit bony tenderness. This may be due to renal osteodystrophy from osteomalacia, secondary hyperparathyroidism or multiple myeloma. Back pain in the context of renal failure should always raise the possibility of an underlying paraproteinaemia.
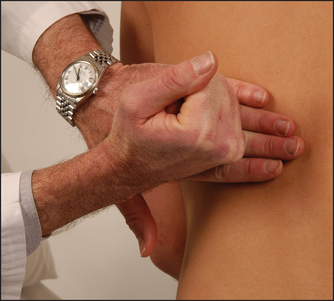
Gentle use of the clenched fist to strike the patient in the renal angle is known as Murphy’s kidney punch (see Figure 18.7) and is designed to elicit renal tenderness in patients with renal infection. Similar information may be gained from more gentle ballotting of the renal angle when the patient lies supine. Look also for sacral oedema in a patient confined to bed, particularly if the nephrotic syndrome or congestive cardiac failure is suspected. The presence of ulcerations of the toes suggests atheroembolic disease.
Legs
The important signs here are oedema, purpura (page 278), livedo reticularis (a red-blue reticularc pattern from vasculitis or atheroembolic disease), pigmentation, scratch marks and signs of peripheral vascular disease. Examination for peripheral neuropathy and myopathy is indicated, as in the arms. Gouty tophi or the presence of gouty arthropathy may very occasionally provide an explanation for the patient’s renal failure (although secondary uric acid retention is common with CKD, it rarely causes clinical gout).
Blood pressure
It is of the utmost importance to take the blood pressure in every patient with renal disease. This is because hypertension can be the cause of renal disease or one of its complications. Test for postural hypotension, as hypovolaemia may precipitate acute kidney injury (AKI).
Fundi
Examination of the fundi is important. Look especially for hypertensive changes and diabetic changes. Diabetes can be a cause of CKD.
Male genitalia
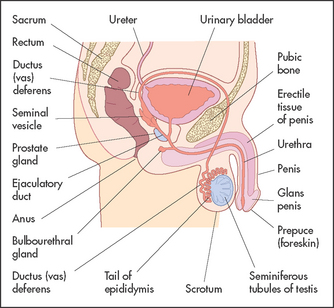
Inspect the genitals (see Figures 18.8 and 18.9) for evidence of mucosal ulceration. This can occur in a number of systemic diseases, including Reiter’s syndrome (reactive arthritis) and the rare Behçet’s syndrome. For aesthetic and protective reasons, it is essential to wear gloves for this examination. Retract the foreskin to expose the glans penis. This mucosal surface is prone to inflammation or ulceration in both infective and connective tissue diseases (see List 18.4). Look also for urethral discharge. If there is a history of discharge, attempt to express fluid by compressing or ‘milking’ the shaft. Any fluid obtained must be sent for microscopic examination and culture.
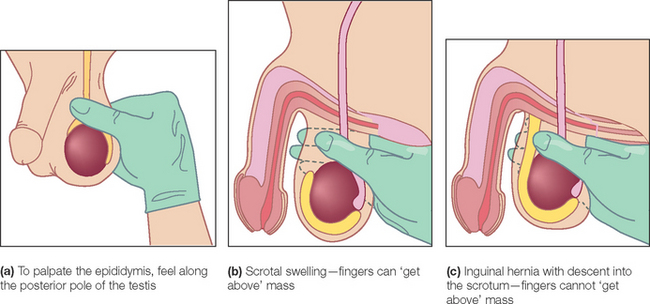
Figure 18.9 Examination of scrotum (From Douglas G, Nicol F, Robertson C. Macleod’s clinical examination, 12th edn. Edinburgh: Churchill Livingstone, 2009, with permission.) Churchill Livingstone
Inspect the scrotum with the patient standing. Usually the left testis hangs lower than the right. This is the only part of the body that consistently does not appear bilaterally symmetrical on inspection. Torsion of the testis may cause the involved testis to appear higher and to lie more transversely than normal. Inspect for oedema of the skin, sebaceous cysts, tinea cruris (an erythematous rash caused by a fungal infection of the moist skin of the groin) or scabies. Scrotal oedema is common in severe cardiac failure and may occur with the nephrotic syndrome and ascites.
Palpate each testis gently using the fingers and thumb of the right hand or cradle the testis between the middle and index fingers of the right hand and palpate it with the ipsilateral thumb.6 The testes are normally equal in size, smooth and relatively firm. Absence of one or both testes may be due to previous excision, failure of the testis to descend or to a retractile testis. In children the testes may retract as examination of the scrotum begins because of a marked cremasteric reflex. A maldescended testis (one that lies permanently in the inguinal canal or higher) has a high chance of developing malignancy. An exquisitely tender, indurated testis suggests orchitis.7 This is often due to mumps in postpubertal patients and occurs about 5 days after the parotitis. An undescended testis may be palpable in the inguinal canal, usually at or above the external inguinal ring. The presence of small firm testes suggests an endocrine disease (hypogonadism) or testicular atrophy due to alcohol or drug ingestion.
Feel posteriorly for the epididymis and then upwards for the vas deferens and the spermatic cord. It should be possible to differentiate the vas from the testis.
A varicocele feels like a bag of worms in the scrotum. The testis on the side of the varicocele often lies horizontally. It is unclear whether this is a cause or effect of the varicocele. A left varicocele is sometimes found when there is underlying left renal tumour or left renal vein thrombosis. The significance of the rarer right varicocele is disputed.8
DIFFERENTIAL DIAGNOSIS OF A SCROTAL MASS
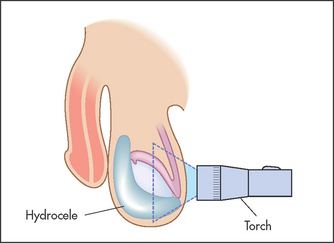
If a mass is palpable in the scrotum, decide first whether it is possible to get above it. Have the patient stand up. If no upper border is palpable, it must be descending down the inguinal canal from the abdomen and is therefore an inguino-scrotal hernia (see Figure 18.10). If it is possible to get above the mass, it is necessary to decide whether it is separate from or part of the testis, and to test for translucency. This is performed using a transilluminoscope (a torch; see Figure 18.11). With the patient in a darkened room, a small torch is applied to the side of the swelling by invaginating the scrotal wall. A cystic mass will light up, while a solid mass remains dark.
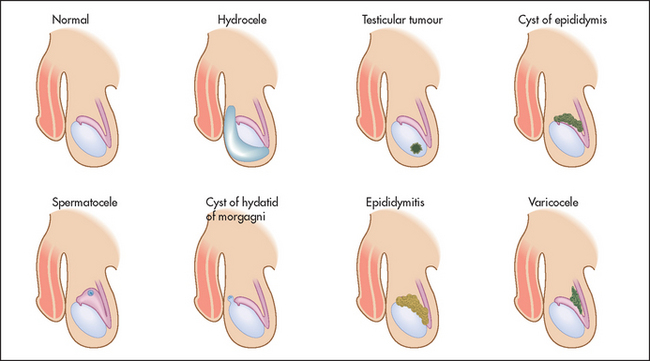
Figure 18.10 Differential diagnosis of a scrotal mass (Adapted from Dunphy JE, Botsford TW. Physical examination of the surgical patient. An introduction to clinical surgery, 4th edn. Philadelphia: WB Saunders, 1975.)
A mass that is part of the testis and that is solid (non-translucent) is likely to be a tumour or, rarely, a syphilitic gumma. The testes may be enlarged and hard in men with leukaemia. A mass that is cystic (translucent) with the testis within it is a hydrocele (a collection of fluid in the tunica vaginalis of the testis). A mass that appears separate from the testis and transilluminates is probably a cyst of the epididymis, while a similar mass that fails to transilluminate is probably the result of chronic epididymitis. By feeling along the testicular–epididymal groove it is usually possible to separate an epididymal mass from the testis itself.
Pelvic examination
The pelvic examination should be performed when indicated as the final part of any complete physical examination.9 It is essential to obtain informed consent and for male students and doctors to have a female chaperone (see List 18.5). The patient’s privacy must be promised and ensured. Gloves must be worn (see Figure 18.12).
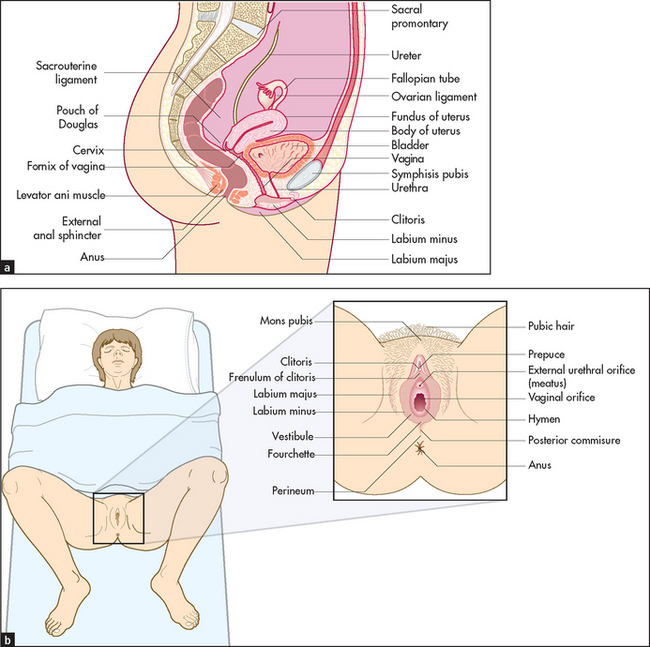
Figure 18.12 Female reproductive anatomy (a) Lateral view, showing the relationship of the genitals to the rectum and bladder. (b) Position for examination. (From Douglas G, Nicol F, Robertson C. Macleod’s clinical examination, 12th edn. Edinburgh: Churchill Livingstone, 2009, with permission.) Churchill Livingstone
The patient should have first emptied her bladder. She should be on her back with her legs apart, ankles together and her knees bent (the frog-legged position). The left lateral position is used when the woman cannot assume the lithotomy position or when a view of the anterior vaginal wall is required—for example, when a urinary fistula is suspected.
The perineum should be brightly illuminated by a lamp. Put a glove on each hand. Inspect first the external genitalia. Note any rash (e.g. sclerotic white areas of leucoplakia, or redness, swelling and excoriation from thrush or trichomoniasis), ulceration, warts, scars, sinus openings or other lesions. Separate the labia with the thumb and forefinger. A Bartholind cyst or abscess is palpated between the thumb and index finger in the posterior part of the labia major; the normal gland is impalpable. Note the size and shape of the clitoris, and the presence or absence of discharge from the urethral orifice and vaginal outlet. A bloody vaginal discharge suggests menstruation, a miscarriage, cancer or a cervical polyp or erosion. A purulent discharge suggests vaginitis, cervicitis or endometritis (e.g. gonorrhoea) or a retained tampon. Trichomonas vaginalis causes a frothy, watery, pale, yellow-white discharge, while thrush (Candida albicans) causes a thick cheesy discharge associated with excoriations and pruritus. Physiological discharge may be present; this is almost colourless.
Ask the patient to bear down; a cystocele (descent of the bladder through the anterior vaginal wall) or rectocele (descent of the rectum through the posterior vaginal wall) or uterine prolapse may become apparent. Then ask the patient to cough; this may demonstrate stress incontinence. Note the presence of vaginal atrophy in older women.
Speculum examination should be performed before digital examination to avoid any contamination with lubricant. A bivalve or Cusco’s speculum is most commonly used and enables a clear view of the cervix to be obtained.
Holding the lips of the labia minora open with the left hand, insert the speculum into the introitus with the blades of the speculum vertical. Then turn the speculum 90° and advance it into the vagina. When the speculum reaches the top of the vagina, gently open the blades and visualise the cervix. Make a note of the presence of any discharge or bleeding from the cervix and of any polyps or areas of ulceration. Remember that the appearance of the cervix is changed after childbirth, with the external os more irregular and slit-like. The most common finding is a so-called erosion or ectropion. This is an area of cervical epithelium around the cervical os that appears a darker red colour than the smooth pink of the rest of the cervix. It is not an erosion at all but normal columnar epithelium extending from the endocervical canal onto the ectocervix. If the clinical history suggests possible infection, take swabs from the vaginal fornices and cervical os and place in transport medium to look for Candida, Trichomonas and Neisseria; take a separate swab from the endocervix for Chlamydia.
Where vaginal wall prolapse is suspected, a Sims’ speculum should be used, as it provides a clearer view of the vaginal walls. Where the Sims’ speculum is used, it is preferable to examine the patient in the semiprone or Sims’ position.
Perform bimanual examination by introducing the middle finger of your examining hand into the vaginal introitus and applying pressure towards the rectum. As the introitus opens, introduce the index finger as well. Palpate the cervix; it has the consistency of the cartilage of the tip of the nose. It must be remembered that the abdominal hand is used to compress the pelvic organs onto the examining vaginal hand. The size, shape, consistency and position of the uterus must be noted. An enlarged uterus is described as being equivalent to a pregnant uterus (e.g. a 14-week sized uterus). The uterus is commonly pre-axial or anteverted, but will be postaxial or retroverted in some 10% of women. Provided the retroverted uterus is mobile, the position is rarely significant.
It is important to feel in the pouch of Douglas for the presence of thickening or nodules, and then to palpate laterally in both fornices for the presence of any ovarian or tubal masses. An attempt should be made to differentiate between adnexal and uterine masses, although this is often not possible. For example, a pedunculated fibroid may mimic an ovarian tumour, whereas a solid ovarian tumour, if adherent to the uterus, may be impossible to distinguish from a uterine fibroid. The ovaries may be palpable in the normal pelvis if the patient is thin, but the Fallopian tubes are palpable only if they are significantly enlarged.
In a child or woman with an intact hymen, it may be necessary to perform a single-finger vaginal examination or rectal examination. It should always be remembered that a rough or painful examination rarely produces any useful information and, in certain situations such as tubal ectopic pregnancy, may be dangerous.
TAKING A CERVICAL (PAP) SMEAR
This should be done at least 3 months after pregnancy and not during menstruation. Explain the purpose of the test and warn the patient that she may notice some spotting afterwards.
Record the patient’s name and hospital number on a suitable slide. After inserting a speculum as above, wipe away any discharge or blood. Note the appearance of the cervix. A 360° sweep should be taken with a suitable spatula or brush pressed firmly against the cervix at the junction of the columnar epithelium of the endocervical canal and the squamous epithelium of the ectocervix. Spread the specimen immediately onto a clear glass slide in a thin even layer and fix the slide with 95% alcohol solution. Fixation requires 30 minutes in solution. Finally, complete the cytology request form with details of previous smears, the last period and method of contraception.
References
1. Muehrcke, RC. The fingernails in chronic hypoalbuminaemia: a new physical sign. BMJ. 1956; 1:1327–1328. [The classic description of this sign.].
2. Guarino, JR. Auscultatory percussion of the urinary bladder. Arch Intern Med. 1985; 145:1823–1825. [This careful study makes a convincing case for the use of this technique, especially in obese patients or those with ascites.].
3. McGee, S. Evidence-based physical diagnosis, 3rd edn. St Louis: Saunders, 2012.
4. Guinan, P, Bush, I, Ray, V, et al. The accuracy of the rectal examination in the diagnosis of prostate carcinoma. N Engl J Med. 1980; 303:499–503. [This article suggests that rectal examination is an excellent technique for distinguishing benign prostatic hyperplasia and cancer, but this has subsequently been questioned.].
5. Schroder, FH. Detection of prostate cancer. BMJ. 1995; 310:140–141.
6. Zornow, DH, Landes, RR. Scrotal palpation. Am Fam Physician. 1981; 23:150–154. [This article describes standard examination techniques.].
7. Rabinowitz, R, Hulbert, WC, Jr. Acute scrotal swelling. Urol Clin Nth Am. 1995; 22:101–105.
8. Roy, CR, Wilson, T, Raife, M, Horne, D. Varicocele as the presenting sign of an abdominal mass. J Urol. 1989; 141:597–599. [A sign of late-stage renal cell carcinoma, due to testicular vein compression, but can be on the left or right side!].
9. Deneke, M, Wheeler, L, Wagner, G, Ling, FW, Buxton, BH. An approach to relearning the pelvic examination. J Fam Pract. 1982; 14:782–783. [This study provides useful hints.].
aRC Meuhrcke reported this sign in the British Medical Journal in 1956.
bRA Mees, Dutch physician, reported this sign in 1919. It had previously been reported (1901) in the Lancet by E Reynolds among drinkers of beer contaminated by arsenic in the north of England.
cReticulum is the Latin word for a ‘little net’.
dCaspar Bartholin Secundus (1655–1738), professor of philosophy at Copenhagen at the age of 19, then professor of medicine, anatomy and physics. He described the glands in 1677.