The gastrointestinal examination
Examination of the gastrointestinal system includes a complete examination of the abdomen. It is also important to search for the peripheral signs of gastrointestinal and liver disease. Some signs are more useful than others.1
Examination anatomy
An understanding of the structure and function of the gastrointestinal tract and abdominal organs is critical for the diagnosis of gastrointestinal disease (see Figure 14.1). The mouth is the gateway to the gastrointestinal tract. It and the anus and rectum are readily accessible to the examiner, and both must be examined carefully in any patient with suspected abdominal disease. The position of the abdominal organs can be quite variable, but there are important surface markings that should be kept in mind during the examination.
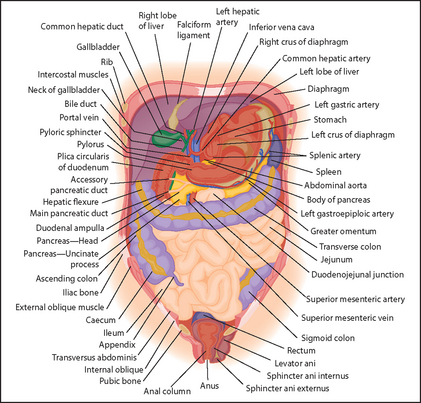
Figure 14.1 Anatomy of gut (Talley NJ, O’Connor S. Pocket clinical examination, 3rd edn. Sydney: Churchill Livingstone, 2009.) Churchill Livingstone
The liver is the largest organ in the abdomen; it comprises a large right lobe and smaller left lobe divided into eight segments, including the caudate lobe (segment I) squeezed in between. The lower border of the liver extends from the tip of the right tenth rib to just below the left nipple. Normally the liver is not palpable, but it may just be possible to feel the lower edge in healthy people.
The spleen is a lymphoid organ that underlies the ninth, tenth and eleventh ribs posteriorly on the left. It is usually not palpable in health (see Chapter 21).
The kidneys lie anteriorly four finger-breadths from the midline and posteriorly under the twelfth rib. Normally, the right kidney extends 2.5 centimetres lower than the left. The lower pole of the right kidney may be palpable in thin healthy people.
The gallbladder is a pear-shaped organ and the fundus (top) is at the tip of the right ninth costal cartilage; it cannot be felt in health. The pancreas is situated in the retroperitoneum (behind the peritoneum), with the head tucked into the C-shaped duodenum and the tail snuggling into the spleen. A huge pancreatic mass may rarely be large enough to be palpable.
The aorta lies in the midline and terminates just to the left of the midline at the level of the iliac crest. A pulsatile mass in the middle of the abdomen is likely to be arising from the aorta and may indicate an aneurysm.
The stomach is usually J-shaped and lies in the left upper part of the abdomen over the spleen and pancreas; it connects with the duodenum. The small intestine ranges from 3 to 10 metres in length and comprises the upper half (duodenum and jejunum) and the lower half (ileum). The small intestine lies over the middle section of the abdomen but is usually impalpable.
The colon is approximately 1.5 metres in length, and from right to left consists of the caecum, ascending colon, hepatic flexure, transverse colon, splenic flexure, descending colon, sigmoid colon, rectum and anal canal (anorectum). The appendix usually lies in the right lower abdominal area, arising posterior-medially from the caecum. The caecum and ascending colon lie on the right side of the abdomen, the transverse colon runs across the upper abdomen from right to left, and then the descending colon, sigmoid and rectum lie on the left side of the abdomen. Rarely, masses arising from the colon will be felt in the abdomen.
Other important anatomical areas include the inguinal canal and the anorectum, which are described later in this chapter in relation to examination of hernias and the rectal examination.
Positioning the patient
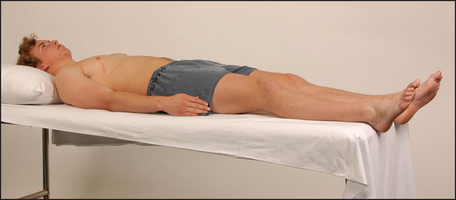
For proper examination of the abdomen it is important that the patient lies flat with the head resting on a single pillow (see Figure 14.2). This relaxes the abdominal muscles and facilitates abdominal palpation. Helping the patient into this position affords the opportunity to make a general inspection.
General appearance
JAUNDICE
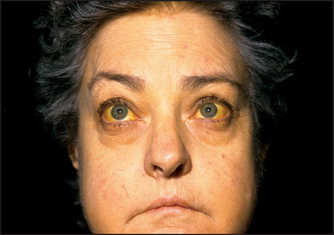
The yellow discolouration of the sclerae (conjunctivae) and the skin that results from hyperbilirubinaemia is best observed in natural daylight (page 30). Whatever the underlying cause, the depth of jaundice can be quite variable.
WEIGHT AND WASTING
The patient’s weight must be recorded. Failure of the gastrointestinal tract to absorb food normally may lead to loss of weight and cachexia. This may also be the result of gastrointestinal malignancy or alcoholic cirrhosis. Folds of loose skin may be visible hanging from the abdomen and limbs; these suggest recent weight loss. Obesity can cause fatty infiltration of the liver (non-alcoholic steatohepatitis) and result in abnormal liver function tests. Anabolic steroid use can induce increase in muscle bulk (sometimes considered desirable) and various liver tumours, including adenomas or hepatocellular carcinomas.
SKIN
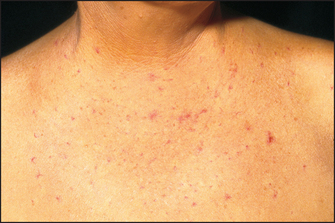
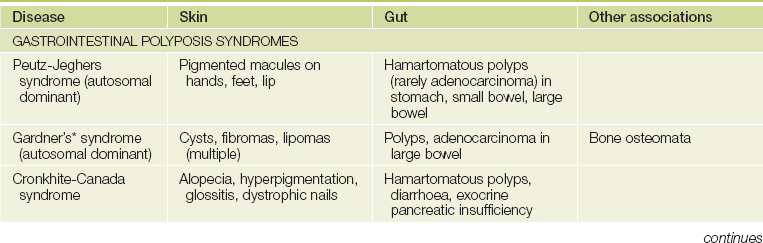
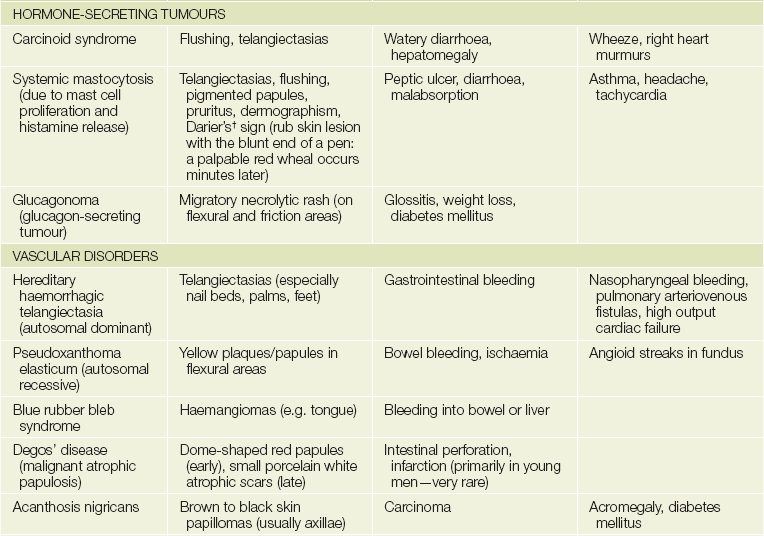
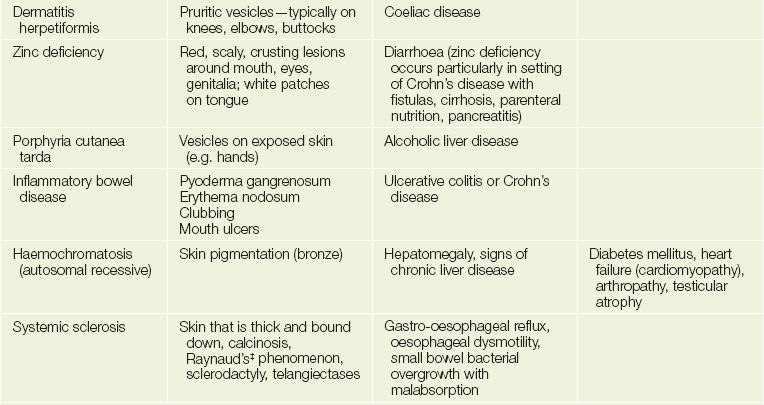
The gastrointestinal tract and the skin have a common origin from the embryoblast. A number of diseases can present with both skin and gut involvement (see Figures 14.3–14.8 and Table 14.1).2
TABLE 14.1
*Eldon John Gardner (1909–89), American geneticist.
†Ferdinand Jean Darier (1856–1938), Paris dermatologist.
‡Maurice Raynaud (1834–1881), Paris physician.
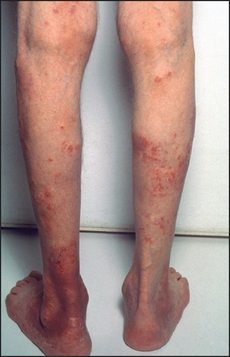
Figure 14.3 Dermatitis herpetiformis in coeliac disease (From McDonald FS, ed. Mayo Clinic images in internal medicine, with permission. © Mayo Clinic Scientific Press and CRC Press.)
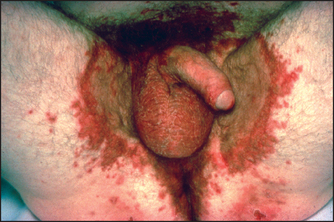
Figure 14.4 Glucagonoma: migratory rash involving the groin (very rare) (From McDonald FS, ed. Mayo Clinic images in internal medicine, with permission. © Mayo Clinic Scientific Press and CRC Press.)
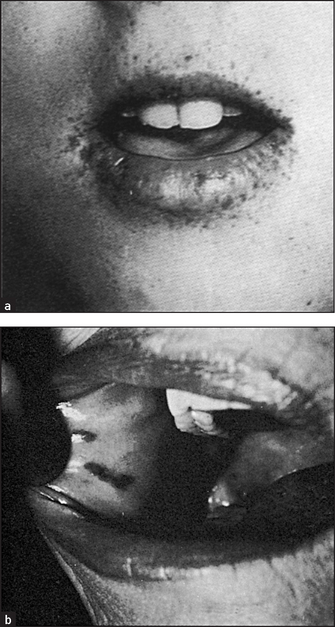
Figure 14.5 Peutz-Jeghers syndrome, with discrete brown-black lesions of the lips (Figure (a) from Jones DV et al, in Feldman M et al. Sleisenger & Fordtran’s gastrointestinal disease, 6th edn. Philadelphia: WB Saunders, 1998, with permission. Figure (b) from McDonald FS, ed. Mayo Clinic images in internal medicine, with permission. © Mayo Clinic Scientific Press and CRC Press.)
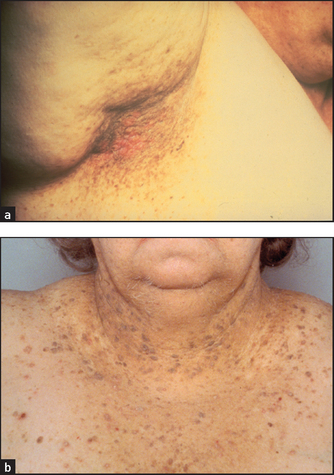
Figure 14.6 Acanthosis nigricans: (a) axilla; (b) chest wall (From McDonald FS, ed. Mayo Clinic images in internal medicine, with permission. © Mayo Clinic Scientific Press and CRC Press.)
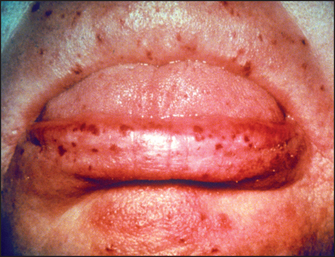
Figure 14.7 Hereditary haemorrhagic telangiectasia involving the lips (From McDonald FS, ed. Mayo Clinic images in internal medicine, with permission. © Mayo Clinic Scientific Press and CRC Press.)
Pigmentation
Generalised skin pigmentation can result from chronic liver disease, especially in haemochromatosis (due to haemosiderin stimulating melanocytes to produce melanin). Malabsorption may result in Addisonian-type pigmentation (‘sunkissed’ pigmentation) of the nipples, palmar creases, pressure areas and mouth.
Peutz-Jeghersa syndrome
Freckle-like spots (discrete, brown-black lesions) around the mouth and on the buccal mucosa (see Figure 14.5) and on the fingers and toes are associated with hamartomas of the small bowel (50%) and colon (30%), which can present with bleeding or intussusception. In this autosomal dominant condition the incidence of gastrointestinal adenocarcinoma is increased.
Acanthosis nigricans
These are brown-to-black velvety elevations of the epidermis due to confluent papillomas and are usually found in the axillae and nape of the neck (see Figure 14.6). Acanthosis nigricans is associated rarely with gastrointestinal carcinoma (particularly stomach) and lymphoma, as well as with acromegaly, diabetes mellitus and other endocrinopathies.
Hereditary haemorrhagic telangiectasia (Rendu-Osler-Weber syndromeb)
Multiple small telangiectasias occur in this disease. They are often present on the lips and tongue (see Figure 14.7), but may be found anywhere on the skin. When they are present in the gastrointestinal tract they can cause chronic blood loss or even, occasionally, torrential bleeding. Associated arteriovenous malformations may occur. This is an autosomal dominant condition and is uncommon.
Porphyria cutanea tarda
Fragile vesicles appear on exposed areas of the skin and heal with scarring (see Figure 14.8). The urine is dark in this chronic disorder of porphyrin metabolism associated with alcoholism and hepatitis C.
Systemic sclerosis
Tense tethering of the skin in systemic sclerosis is often associated with gastro-oesophageal reflux and gastrointestinal motility disorders (page 333).
MENTAL STATE
Assess orientation (page 401). The syndrome of hepatic encephalopathy, due to decompensated advanced cirrhosis (chronic liver failure) or fulminant hepatitis (acute liver failure), is an organic neurological disturbance. The features depend on the aetiology and the precipitating factors. Patients eventually become stuporous and then comatose. The combination of hepatocellular damage and porto-systemic shunting due to disturbed hepatic structure (both extrahepatic and intrahepatic) causes this syndrome. It is probably related to the liver’s failure to remove toxic metabolites from the portal blood. These toxic metabolites may include ammonia, mercaptans, short-chain fatty acids and amines.
Hands
Even experienced gastroenterologists must restrain their excitement and begin the examination of the gastrointestinal tract with the hands. The signs that may be elicited here give a clue to the presence of chronic liver disease. Whatever its aetiology, permanent diffuse liver damage results in similar peripheral signs. However, none of these signs alone is specific for chronic liver disease.
NAILS
Leuconychia
When chronic liver or other disease results in hypoalbuminaemia, the nail beds opacify (the abnormality is of the nail bed and not of the nail), often leaving only a rim of pink nail bed at the top of the nail (Terry’s nailsc; see Figure 14.9). The thumb and index nails are most often involved. The underlying mechanism may be compression of capillary flow by extracellular fluid.
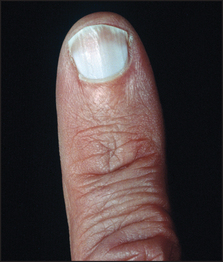
Figure 14.9 Leuconychia—Terry’s nails (From McDonald FS, ed. Mayo Clinic images in internal medicine, with permission. © Mayo Clinic Scientific Press and CRC Press.)
Muehrcke’s lines (transverse white lines) can also occur in hypoalbuminaemic states, including cirrhosis. Blue lunulae may be seen in patients with Wilson’s disease (hepatolenticular degeneration).
Clubbing
Up to one-third of patients with cirrhosis may have finger clubbing. In at least some cases, this may be related to arteriovenous (AV) shunting in the lungs, resulting in arterial oxygen desaturation. Cyanosis may be associated with severe longstanding chronic liver disease. The cause of this pulmonary AV shunting is unknown. Conditions such as inflammatory bowel disease and coeliac disease, which cause longstanding nutritional depletion, can also cause clubbing.
PALMS
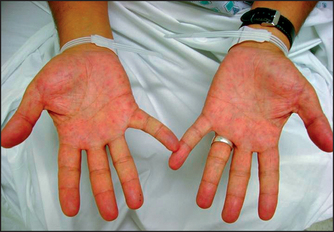
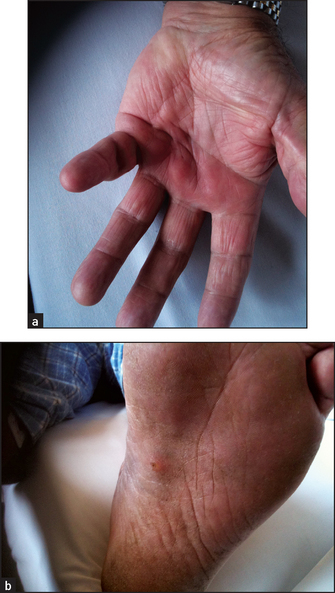
Palmar erythema (‘liver palms’)
Palmar erythema is reddening of the palms of the hands affecting the thenar and hypothenar eminences (see Figure 14.10). Often the soles of the feet are also affected. This can be a feature of chronic liver disease. While the finding has been attributed to raised oestrogen levels, it has not been shown to be related to plasma oestradiol levels, so the aetiology remains uncertain. Palmar erythema can also occur with thyrotoxicosis, rheumatoid arthritis, polycythaemia and, rarely, with chronic febrile diseases or chronic leukaemia. It may also be a normal finding, especially in women, and like spider naevi can occur in pregnancy.
Anaemia
Inspect the palmar creases for pallor suggesting anaemia, which may result from gastrointestinal blood loss, malabsorption (folate, vitamin B12), haemolysis (e.g. hypersplenism) or chronic disease.
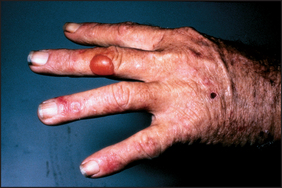
Dupuytren’s contracture
Dupuytren’s contractured is a visible and palpable thickening and contraction of the palmar fascia causing permanent flexion, most often of the ring finger (see Figure 14.11). It is often bilateral and occasionally affects the feet. It is associated with alcoholism (not liver disease), but is also found in some manual workers; it is often familial. The palmar fascia of these patients contains abnormally large amounts of xanthine, and this may be related to the pathogenesis.
HEPATIC FLAP (ASTERIXISe)
Before leaving the hands ask the patient to stretch out his or her arms in front, separate the fingers and extend the wrists for 15 seconds. Jerky, irregular flexion–extension movement at the wrist and metacarpophalangeal joints, often accompanied by lateral movements of the fingers, constitute the flapping of hepatic encephalopathy. It is thought to be due to interference with the inflow of joint position sense information to the reticular formation in the brainstem. This results in rhythmical lapses of postural muscle tone. Occasionally the arms, neck, tongue, jaws and eyelids can also be involved. It can sometimes be demonstrated if the patient is asked to close the eyes forcefully or to protrude the tongue. The flap is usually bilateral, tends to be absent at rest and is brought on by sustained posture. The rhythmic movements are not synchronous on each side and the flap is absent when coma supervenes.
Although this flap is a characteristic and early sign of liver failure, it is not diagnostic: it can also occur in cardiac, respiratory and renal failure, as well as in hypoglycaemia, hypokalaemia, hypomagnesaemia or barbiturate intoxication.
An apparent tremor (really a form of choreoathetosis) may occur in Wilson’s disease. A fine resting tremor is common in alcoholism.
Arms
Inspect the upper limbs for bruising. Large bruises (ecchymoses) may be due to clotting abnormalities. Hepatocellular damage can interfere with protein synthesis and therefore the production of all the clotting factors (except factor VIII, which is made elsewhere in the reticuloendothelial system). Obstructive jaundice results in a shortage of bile acids in the intestine and therefore may reduce absorption of vitamin K (a fat-soluble vitamin), which is essential for the production of clotting factors II (prothrombin), VII, IX and X.
Petechiae (pinhead-sized bruises) may also be present (see Figure 14.12). Chronic excessive alcohol consumption can sometimes result in bone marrow depression, causing thrombocytopenia, which may be responsible for petechiae. In addition, splenomegaly secondary to portal hypertension can cause hypersplenism, with resultant excessive destruction of platelets in the spleen; in severe liver disease (especially acute hepatic necrosis), diffuse intravascular coagulation can occur.
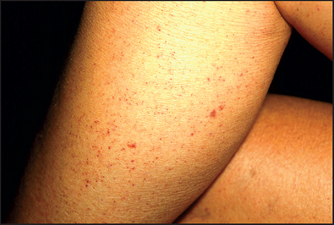
Figure 14.12 Petechiae (From Marks JG, Miller JJ. Lookingbill & Marks’ principles of dermatology, 4th edn. Saunders, 2006.)
Look for muscle wasting, which is often a late manifestation of malnutrition in alcoholic patients (see Text box 14.1). Alcohol can also cause a proximal myopathy (page 470).
Scratch marks due to severe itch (pruritus) are often prominent in patients with obstructive or cholestatic jaundice. This is commonly the presenting feature of primary biliary cirrhosisf before other signs are apparent. The mechanism of pruritus is thought to be retention of an unknown substance normally excreted in the bile, rather than bile salt deposition in the skin as was earlier thought.
Spider naevig (see Figure 14.13) consist of a central arteriole from which radiate numerous small vessels that look like spiders’ legs. They range in size from just visible to half a centimetre in diameter. Their usual distribution is in the area drained by the superior vena cava, so they are found on the arms, neck and chest wall. They can occasionally bleed profusely. Pressure applied with a pointed object to the central arteriole causes blanching of the whole lesion. Rapid refilling from the centre to the legs occurs on release of the pressure.
The finding of more than two spider naevi anywhere on the body is likely to be abnormal. Spider naevi can be caused by cirrhosis, most frequently due to alcohol. In patients with cirrhosis the number of spider naevi may increase or decrease as the patient’s condition changes, as does the prominence of palmar erythema. Spiders may occur transiently in patients with viral hepatitis. During the second to fifth months of pregnancy, spider naevi frequently appear, only to disappear again within days of delivery. It is not known why they occur only in the upper part of the body, but it may be related to the fact that this is the part of the body where flushing usually occurs. Like palmar erythema they are traditionally attributed to oestrogen excess. Part of the normal hepatic function is the inactivation of oestrogens, which is impaired in chronic liver disease. Oestrogens are known to have a dilating effect on the spiral arterioles of the endometrium, and this has been used to explain the presence of spider naevi, but changes in plasma oestradiol levels have not been found to correlate with the appearance and disappearance of spider naevi.
The differential diagnosis of spider naevi includes Campbell de Morganh spots, venous stars and hereditary haemorrhagic telangiectasia. Campbell de Morgan spots are elevated red circular lesions that occur on the abdomen or the front of the chest. They do not blanch on pressure and are very common and benign. Venous stars are 2- to 3-centimetre lesions that can occur on the dorsum of the feet, legs, back and lower chest. They are due to elevated venous pressure and are found overlying the main tributary to a large vein. They are not obliterated by pressure. The blood flow is from the periphery to the centre of the lesion, which is the opposite of the flow in the spider naevus. Lesions of hereditary haemorrhagic telangiectasia (page 187) occasionally resemble spider naevi.
Palpate the axillae for lymphadenopathy (page 273). Look in the axillae for acanthosis nigricans.
Face
EYES
Look first at the sclerae for signs of jaundice (see Figure 14.14) or anaemia (pale conjunctiva). Bitot’si spots are yellow keratinised areas on the sclera (see Figure 14.15). They are the result of severe vitamin A deficiency due to malabsorption or malnutrition. Retinal damage and blindness may occur as a later development. Kayser-Fleischer ringsj (see Figure 14.16) are brownish-green rings occurring at the periphery of the cornea, affecting the upper pole more than the lower. They are due to deposits of excess copper in Descemet’s membranek of the cornea. Slit-lamp examination is often necessary to show them. They are typically found in Wilson’s diseasel, a copper storage disease that causes cirrhosis and neurological disturbances. Kayser-Fleischer rings are usually present by the time neurological signs have appeared. Patients with other cholestatic liver diseases, however, can also have these rings. Iritis may be seen in inflammatory bowel disease (page 223).
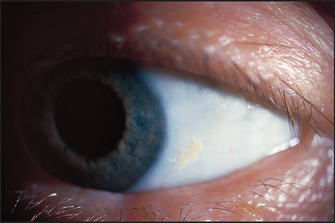
Figure 14.15 Bitot spot: focal area of conjunctival xerosis with a foamy appearance (From Mir MA. Atlas of clinical diagnosis, 2nd edn. Edinburgh: Saunders, 2003, with permission.)
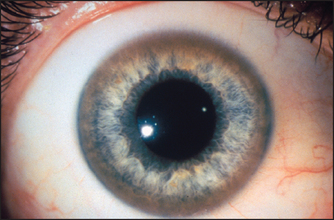
Figure 14.16 Kayser-Fleischer rings in Wilson’s disease (From McDonald FS, ed. Mayo Clinic images in internal medicine, with permission. © Mayo Clinic Scientific Press and CRC Press.)
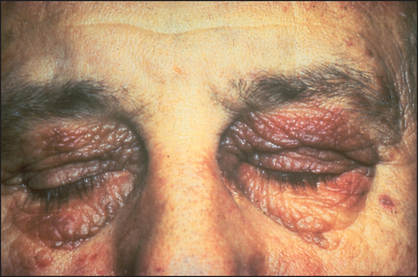
Xanthelasmata are yellowish plaques in the subcutaneous tissues in the periorbital region and are due to deposits of lipids (see Figure 5.21, page 70). They may indicate protracted elevation of the serum cholesterol. In patients with cholestasis, an abnormal lipoprotein (lipoprotein X) is found in the plasma and is associated with elevation of the serum cholesterol. Xanthelasmata are common in patients with primary biliary cirrhosis.
Periorbital purpura following proctosigmoidoscopy (‘black eye syndrome’) is a characteristic sign of amyloidosis (perhaps related to factor X deficiency) but is exceedingly rare (see Figure 14.17).
SALIVARY GLANDS
Next inspect and palpate the cheeks over the parotid area for parotid enlargement (see List 14.1). Ask the patient to clench his or her teeth so that the masseter muscle is palpable; the normal parotid gland is impalpable but the enlarged gland is best felt behind the masseter muscle and in front of the ear. Parotidomegaly that is bilateral is associated with alcoholism rather than liver disease per se. It is due to fatty infiltration, perhaps secondary to alcohol toxicity with or without malnutrition. A tender, warm, swollen parotid suggests the diagnosis of parotiditis following an acute illness or surgery. A mixed parotid tumour (a pleomorphic adenoma) is the most common cause of a lump. Parotid carcinoma may cause a facial nerve palsy (page 417). Feel in the mouth for a parotid calculus, which may be present at the parotid duct orifice (opposite the upper second molar). Mumps also causes acute parotid enlargement, which is usually bilateral.
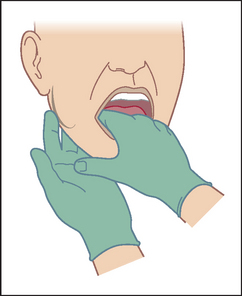
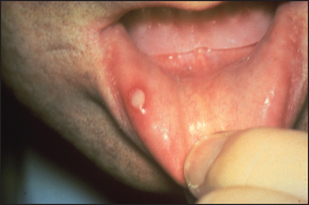
Submandibular gland enlargement is most often due to a calculus. This may be palpable bimanually (see Figure 14.18). Place your gloved index finger on the floor of the patient’s mouth beside the tongue, feeling between it and your fingers placed behind the body of the mandible. It may also be enlarged in chronic liver disease.
MOUTH
Teeth and breath
The very beginning of the gastrointestinal tract is, like the very end of the tract, accessible to inspection without elaborate equipment.3 Look first briefly at the state of the teeth and note whether they are real or false. False teeth will have to be removed for a complete examination of the mouth. Note whether there is gum hypertrophy (see List 14.2) or pigmentation (see List 14.3). Loose-fitting false teeth may be responsible for ulcers and decayed teeth may be responsible for fetorm (bad breath).
Other causes of fetor are listed in List 14.4. These must be distinguished from fetor hepaticus, which is a rather sweet smell of the breath. It is an indication of severe hepatocellular disease and may be due to methylmercaptans. These substances are known to be exhaled in the breath and may be derived from methionine when this amino acid is not demethylated by a diseased liver. Severe fetor hepaticus that fills the patient’s room is a bad sign and indicates a precomatose condition in many cases. The presence of fetor hepaticus in a patient with a coma of unknown cause may be a helpful clue to the diagnosis.
Unless the smell is obvious, ask the patient to exhale through the mouth while you sniff a little of the exhaled air.
Tongue
Thickened epithelium with bacterial debris and food particles commonly causes a coating over the tongue, especially in smokers. It is rarely a sign of disease and is more marked on the posterior part of the tongue where there is less mobility and the papillae desquamate more slowly. It occurs frequently in respiratory tract infections, but is in no way related to constipation or any serious abdominal disorder.
Lingua nigra (black tongue) is due to elongation of papillae over the posterior part of the tongue, which appears dark brown because of the accumulation of keratin. Apart from its aesthetic problems it is symptomless. Bismuth compounds may also cause a black discolouration of the tongue.
Geographical tongue is a term used to describe slowly changing red rings and lines that occur on the surface of the tongue. It is not painful, and the condition tends to come and go. It is not usually of any significance, but can be a sign of riboflavin (vitamin B2) deficiency.
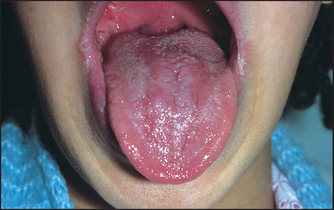
Leucoplakia (see Figure 14.19) is white-coloured thickening of the mucosa of the tongue and mouth; the condition is premalignant. Most of the causes of leucoplakia begin with ‘S’: sore teeth (poor dental hygiene), smoking, spirits, sepsis or syphilis, but often no cause is apparent. Leucoplakia may also occur on the larynx, anus and vulva.
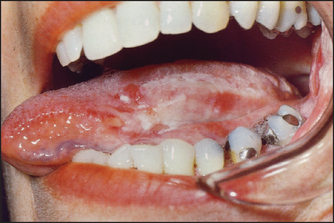
Figure 14.19 Leukoplakia (From Weidner N et al. Modern surgical pathology, 2nd edn. Saunders, 2009.)
The term glossitis (see Figure 14.20) is generally used to describe a smooth appearance of the tongue, which may also be erythematous. The appearance is due to atrophy of the papillae, and in later stages there may be shallow ulceration. These changes occur in the tongue often as a result of nutritional deficiencies to which the tongue is sensitive because of the rapid turnover of mucosal cells. Deficiencies of iron, folate and the vitamin B group, especially vitamin B12, are common causes. Glossitis is common in alcoholics and can also occur in the rare carcinoid syndrome. However, many cases, especially those in elderly people, are impossible to explain.
Enlargement of the tongue (macroglossia) may occur in congenital conditions such as Down syndrome or in endocrine disease, including acromegaly. Tumour infiltration (e.g. haemangioma or lymphangioma) or infiltration of the tongue with amyloid material in amyloidosis can also be responsible for macroglossia.
Mouth ulcers
This is an important topic because a number of systemic diseases can present with ulcers in the mouth (see List 14.5). Aphthous ulceration is the most common type seen (see Figure 14.21). This begins as a small painful vesicle on the tongue or mucosal surface of the mouth, which may break down to form a painful, shallow ulcer. These ulcers heal without scarring. The cause is completely unknown. They usually do not indicate any serious underlying systemic disease, but may occur in Crohn’sn disease or coeliac disease. HIV infection may be associated with a number of mouth lesions (page 556). Angular stomatitis (see Figure 14.22) refers to cracks at the corners of the mouth; causes include deficiencies in vitamin B6, vitamin B12, folate and iron.
Candidiasis (moniliasis)
Fungal infection with Candida albicans (thrush) causes creamy white curd-like patches in the mouth that are removed only with difficulty and leave a bleeding surface (see Figure 40.26 on page 546). The infection may spread to involve the oesophagus, causing dysphagia or odynophagia. Moniliasis is associated with immunosuppression (steroids, tumour chemotherapy, alcoholism or an underlying immunological abnormality such as HIV infection, or haematological malignancy), where it is due to decreased host resistance. Broad-spectrum antibiotics, which inhibit the normal oral flora, are also a common cause, because fungal overgrowth is permitted. Faulty oral hygiene, iron deficiency and diabetes mellitus can also be responsible. Chronic mucocutaneous candidiasis is a distinct syndrome comprising recurrent or persistent oral thrush, fingernail or toenail bed infection and skin involvement. It is usually the result of T-cell immunodeficiency. In about half of these patients, endocrine diseases such as hypoparathyroidism, hypothyroidism or Addison’s disease are associated.
Neck and chest
Palpate the cervical lymph nodes. It is particularly important to feel for the supraclavicular nodes, especially on the left side. The most common cause in many countries is tuberculosis. These may also be enlarged as a result of advanced gastric or other gastrointestinal malignancy, or with lung cancer. The presence of a large left supraclavicular node (Virchow’s node) in combination with carcinoma of the stomach is called Troisier’so sign. Look for spider naevi.
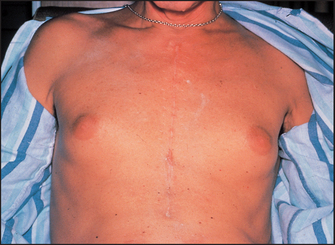
In males, gynaecomastia (enlargement of the male breasts) may be a sign of chronic liver disease. Gynaecomastia may be unilateral or bilateral and the breasts may be tender (see Figure 14.23). This may be a sign of cirrhosis, particularly alcoholic cirrhosis, or of chronic autoimmune hepatitis. In chronic liver disease, changes in the oestradiol-to-testosterone ratio may be responsible. In cirrhotic patients, spironolactone, used to treat ascites, is also a common cause. In addition, gynaecomastia may occur in alcoholics without liver disease because of damage to the Leydigp cells of the testis from alcohol. A number of other drugs may rarely cause gynaecomastia (e.g. digoxin, cimetidine).
Abdomen
Self-restraint is no longer required and it is now time to examine the abdomen itself.
INSPECTION
The patient should lie flat, with one pillow under the head and the abdomen exposed from the nipples to the pubic symphysis (see Figure 14.2). It may be preferable to expose this area in stages to preserve the patient’s dignity.
Does the patient appear unwell? The patient with an acute abdomen may be lying very still and have shallow breathing (page 219).
Inspection begins with a careful look for abdominal scars, which may indicate previous surgery or trauma (see Figure 14.24). Look in the area around the umbilicus for laparoscopic surgical scars. Older scars are white and recent scars are pink because the tissue remains vascular. Note the presence of stomata (end-colostomy, loop colostomy, ileostomy or ileal conduit) or fistulae. There may be visible abdominal striae following weight loss.
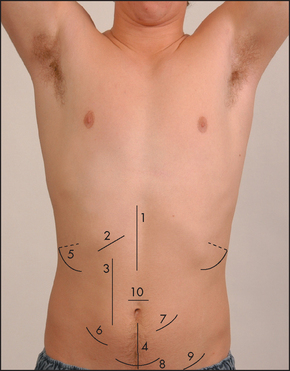
Figure 14.24 Abdominal scars
Note: Laparoscopic surgical scars are now common. Most of these procedures include a port about 2 cm in length, just above the umbilicus.
(Courtesy of Glenn McCulloch)
Generalised abdominal distension (see Figure 14.25) may be present. All the causes of this sound as if they begin with the letter ‘F’: fat (gross obesity), fluid (ascites), fetus, flatus (gaseous distension due to bowel obstruction), faeces, ‘filthy’ big tumour (e.g. ovarian tumour or hydatid cyst) or ‘phantom’ pregnancy. Look at the shape of the umbilicus, which may give a clue to the underlying cause. An umbilicus buried in fat suggests that the patient eats too much. However, when the peritoneal cavity is filled with large volumes of fluid (ascites) from whatever cause, the abdominal flanks and wall appear tense and the umbilicus is shallow or everted and points downwards. In pregnancy the umbilicus is pushed upwards by the uterus enlarging from the pelvis. This appearance may also result from a huge ovarian cyst.
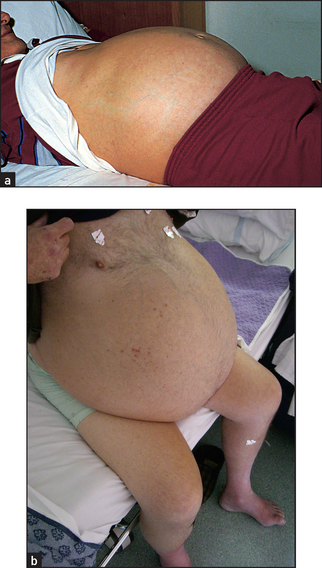
Figure 14.25 (a) Abdomen distended with ascites (patient supine): umbilicus points downwards, unlike cases of distension due to a pelvic mass; (b) Gross ascites (patient sitting) (Courtesy of Dr A Watson, Infectious diseases department, The Canberra Hospital)
Local swellings may indicate enlargement of one of the abdominal or pelvic organs. A hernia is a protrusion of an intra-abdominal structure through an abnormal opening; this may occur because of weakening of the abdominal wall by previous surgery (incisional hernia), a congenital abdominal wall defect or chronically increased intra-abdominal pressure.
Prominent veins may be obvious on the abdominal wall. If these are present, the direction of venous flow should be elicited at this stage. Use one finger to occlude the vein and then empty blood from the vein below the occluding finger with a second finger. Remove the second finger; if the vein refills, flow is occurring towards the occluding finger (see Figure 14.26). Flow should be tested separately in veins above and below the umbilicus.
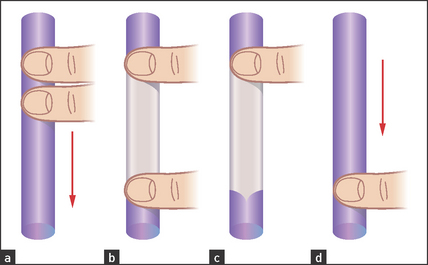
Figure 14.26 Detecting the direction of flow of a vein (a) Two fingers are placed firmly on the vein. (b) The second finger is moved along the vein to empty it of blood and keep it occluded. (c) The second finger is removed but the vein does not refill. (d) At repeat testing and removing the first finger, filling occurs, indicating the direction of flow. (Adapted from Swash M, ed. Hutchison’s clinical methods, 20th edn. Philadelphia: Baillière Tindall, 1995, with permission.)
In patients with severe portal hypertension, portal to systemic flow occurs through the umbilical veins, which may become engorged and distended (see Figure 14.27). The direction of flow then is away from the umbilicus. Because of their engorged appearance they have been likened to the mythical Medusa’s hair after Minerva had turned it into snakes; this sign is called a caput Medusae (head of Medusaa) but is very rare (see Figure 14.28). Usually only one or two veins (often epigastric) are visible. Engorgement can also occur because of inferior vena caval obstruction, usually due to a tumour or thrombosis but sometimes because of tense ascites. In this case the abdominal veins enlarge to provide collateral blood flow from the legs, avoiding the blocked inferior vena cava. The direction of flow is then upwards towards the heart. Therefore, to distinguish caput Medusae from inferior vena caval obstruction, determine the direction of flow below the umbilicus; it will be towards the legs in the former and towards the head in the latter. Prominent superficial veins can occasionally be congenital.
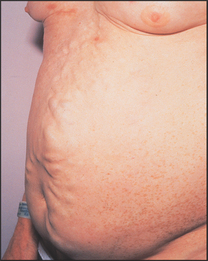
Figure 14.27 Distended abdominal veins in a patient with portal hypertension (From Mir MA. Atlas of clinical diagnosis, 2nd edn. Edinburgh: Saunders, 2003, with permission.)
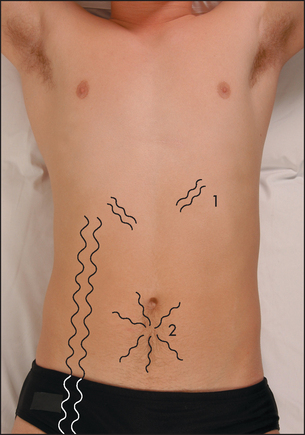
Figure 14.28 Prominent veins on the abdominal wall 1 = thin veins over the costal margin—not of clinical relevance; 2 = caput Medusae; 3 = inferior vena caval obstruction. (Courtesy of Glenn McCulloch. Based on Swash M, ed. Hutchison’s clinical methods, 20th edn. Philadelphia: Baillière Tindall, 1995.)
Pulsations may be visible. An expanding central pulsation in the epigastrium suggests an abdominal aortic aneurysm. However, the abdominal aorta can often be seen to pulsate in normal thin people.
Visible peristalsis may occur occasionally in very thin normal people; however, it usually suggests intestinal obstruction. Pyloric obstruction due to peptic ulceration or tumour may cause visible peristalsis, seen as a slow wave of movement passing across the upper abdomen from left to right. Obstruction of the distal small bowel can cause similar movements in a ladder pattern in the centre of the abdomen.
Skin lesions should also be noted on the abdominal wall. These include the vesicles of herpes zoster, which occur in a radicular pattern (they are localised to only one side of the abdomen in the distribution of a single nerve root). Herpes zoster may be responsible for severe abdominal pain that is of mysterious origin until the rash appears. The Sister Josepha nodule is a metastatic tumour deposit in the umbilicus, the anatomical region where the peritoneum is closest to the skin. Discolouration of the umbilicus where a faintly bluish hue is present is found rarely, in cases of extensive haemoperitoneum and acute pancreatitis (Cullen’s signa—the umbilical ‘black eye’; see Figure 14.29). Skin discolouration may also rarely occur in the flanks in severe cases of acute pancreatitis (Grey-Turner’s signa).
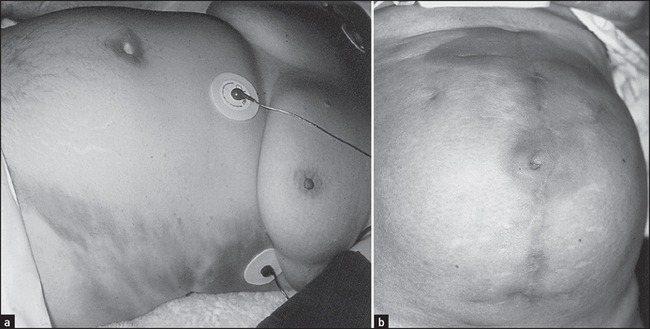
Figure 14.29 (a) Grey–Turner’s sign and (b) Cullen’s sign (umbilicus) in severe acute pancreatitis (From Jarnagin, Blumgart. Blumgart’s surgery of the liver, pancreas and biliary tract, 5th edn. Saunders, 2012.)
Stretching of the abdominal wall severe enough to cause rupture of the elastic fibres in the skin produces pink linear marks with a wrinkled appearance, which are called striae. When these are wide and purple-coloured, Cushing’s syndrome may be the cause (page 366). Ascites, pregnancy or recent weight gain are much more common causes of striae.
Next, squat down beside the bed so that the patient’s abdomen is at eye level. Ask him or her to take slow deep breaths through the mouth and watch for evidence of asymmetrical movement, indicating the presence of a mass. In particular a large liver may be seen to move below the right costal margin or a large spleen below the left costal margin.
PALPATION
This part of the examination often reveals the most information. Successful palpation is only possible if the patient’s abdominal muscles are relaxed. To this end, reassure the patient that the examination will not be painful and use hands that are gentle and warm. Ask the patient if any particular area is tender and examine this area last. Encourage the patient to breathe gently through the mouth. If necessary, ask him or her to bend the knees to relax the abdominal wall muscles.
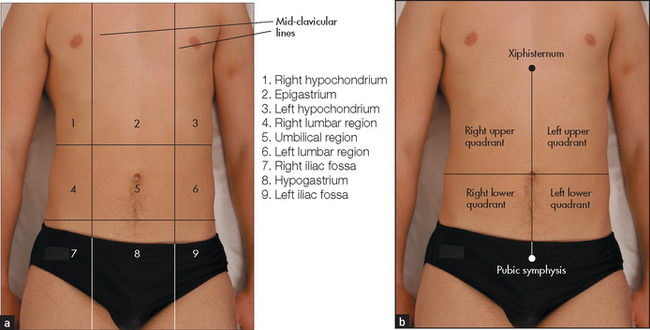
For descriptive purposes the abdomen has been divided into nine areas or regions (see Figure 14.30). Palpation in each region is performed with the palmar surface of the fingers acting together. For palpation of the edges of organs or masses, the lateral surface of the forefinger is the most sensitive part of the hand.
Palpation should begin with light pressure in each region. All the movements of the hand should occur at the metacarpophalangeal joints and the hand should be moulded to the shape of the abdominal wall. Note the presence of any tenderness or lumps in each region. As you move your hand over each region, consider the anatomical structures that underlie it. Deep palpation of the abdomen is performed next, though care should be taken to avoid the tender areas until the end of the examination. Deep palpation is used to detect deeper masses and to define those already discovered. Any mass must be carefully characterised and described (see List 14.6).
Guarding of the abdomen (when resistance to palpation occurs due to contraction of the abdominal muscles) may result from tenderness or anxiety, and is voluntary. It may be overcome by reassurance and gentleness. Rigidity is a constant involuntary reflex contraction of the abdominal muscles always associated with tenderness and indicates peritoneal irritation or inflammation (peritonitis).
Rebound tenderness is said to be present when the abdominal wall, having been compressed slowly, is released rapidly and a sudden stab of pain results. This may make the patient wince, so watch his or her face while this manoeuvre is performed. It strongly suggests the presence of peritonitis and should be performed if there is doubt about the presence of localised or generalised peritonitis. The patient with a confirmed acute abdomen should not be subjected to repeated testing of rebound tenderness because of the distress this can cause. Be careful not to surprise the patient by a sudden jabbing and release movement: rebound tenderness should be elicited slowly. If you suspect the patient may be feigning a tender abdomen, test for rebound with your stethoscope after telling the patient to lie still and quiet so that you can hear.
Liver
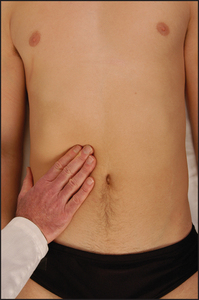
Feel for hepatomegaly (see Figure 14.31).4 With your examining hand aligned parallel to the right costal margin, and beginning in the right iliac fossa, ask the patient to breathe in and out slowly through the mouth. With each expiration advance your hand by 1 or 2 cm closer to the right costal margin. During inspiration the hand is kept still and the lateral margin of the forefinger waits expectantly for the liver edge to strike it.
The edge of the liver and the surface itself may be hard or soft, tender or non-tender, regular or irregular and pulsatile or non-pulsatile. The normal liver edge may be just palpable below the right costal margin on deep inspiration, especially in thin people. The edge is then felt to be soft and regular with a fairly sharply defined border and the surface of the liver itself is smooth. Sometimes only the left lobe of the liver may be palpable (to the left of the midline) in patients with cirrhosis.
If the liver edge is palpable the total liver span can be measured. Remember that the liver span varies with height and is greater in men than women, and that inter-observer error is quite large for this measurement. The normal upper border of the liver is level with the sixth intercostal space in the midclavicular line. At this point the percussion note over the chest changes from resonant to dull (see Figure 14.32(a)). To estimate the liver span (see Figure 14.32(b)), percuss down along the right midclavicular line until the liver dullness is encountered and measure from here to the palpable liver edge. Careful assessment of the position of the midclavicular line will improve the accuracy of this measurement. The normal span is less than 13 centimetres. Note that the clinical estimate of the liver span usually underestimates its actual size by 2 to 5 centimetres.
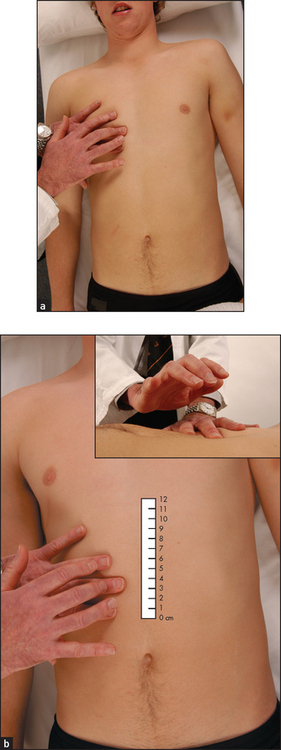
Figure 14.32 Percussing the liver span: (a) upper border; (b) lower border (Courtesy of Glenn McCulloch)
Other causes of a normal but palpable liver include ptosis due to emphysema, asthma or a subdiaphragmatic collection, or a Riedel’s lobe.a The Riedel’s lobe is a tongue-like projection of the liver from the right lobe’s inferior surface; it can be quite large and rarely extends as far as the right iliac fossa. It can be confused with an enlarged gallbladder or right kidney.
Many diseases cause hepatic enlargement and these are listed in List 14.7. Detecting the liver edge below the costal margin clinically is highly specific (100%) but insensitive (48% for enlargement)—positive LR 2.5, negative LR 0.5.4,5 Remember, the diseased liver is not always enlarged; a small liver is common in advanced cirrhosis, and the liver shrinks rapidly with acute hepatic necrosis (due to liver cell death and collapse of the reticulin framework).
Gallbladder
The gallbladder is occasionally palpable below the right costal margin where this crosses the lateral border of the rectus muscles. If biliary obstruction or acute cholecystitis is suspected, the examining hand should be oriented perpendicular to the costal margin, feeling from medial to lateral. Unlike the liver edge, the gallbladder, if palpable, will be a bulbous, focal, rounded mass that moves downwards on inspiration. The causes of an enlarged gallbladder are listed in List 14.8.
Murphy’sa sign should be sought if cholecystitis is suspected. On taking a deep breath, the patient catches his or her breath when an inflamed gallbladder presses on the examiner’s hand, which is lying at the costal margin. Other signs are less helpful.
When examining for an enlarged gallbladder always be mindful of Courvoisier’sa law, which states that, if the gallbladder is enlarged and the patient is jaundiced, the cause is unlikely to be gallstones. Rather, carcinoma of the pancreas or lower biliary tree resulting in obstructive jaundice is likely to be present. This is because the gallbladder with stones is usually chronically fibrosed and therefore incapable of enlargement. Note that if the gallbladder is not palpable, and the patient is jaundiced, some cause other than gallstones is still possible, as at least 50% of dilated gallbladders are impalpable (see Good signs guide 14.1).
Spleen
The spleen enlarges inferiorly and medially (see Figure 14.33). Its edge should be sought below the umbilicus in the midline initially. A two-handed technique is recommended. Place your left hand posterolaterally over the patient’s left lower ribs and your right hand on the abdomen below the umbilicus, parallel to the left costal margin (see Figure 14.34(a)). Do not start palpation too near the costal margin or a large spleen will be missed. As you advance your right hand closer to the left costal margin, compress your left hand firmly over the rib cage so as to produce a loose fold of skin (see Figure 14.34(b)); this removes tension from the abdominal wall and enables a slightly enlarged soft spleen to be felt as it moves down towards the right iliac fossa at the end of inspiration (see Figure 14.34(c)).
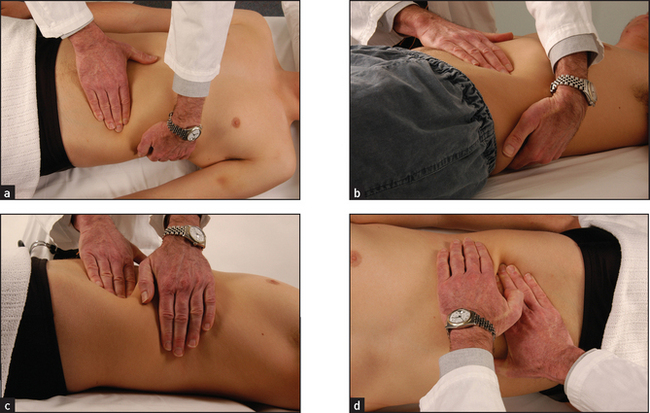
Figure 14.34 Palpation of the spleen
(a) Palpation begins in the lower mid-abdomen and finishes up under the left costal margin.
(b) The examiner’s hand supports the patient’s side …
(c) … and then rests over the lower costal margin to reduce skin resistance.
(d) If the spleen is not palpable when the patient is flat, he or she should be rolled towards the examiner. (Courtesy of Glenn McCulloch)
If the spleen is not palpable, roll the patient onto the right side towards you (the right lateral decubitusa position) and repeat palpation. Begin close to the left costal margin (see Figure 14.34(d)). As a general rule, splenomegaly becomes just detectable if the spleen is one-and-a-half to two times enlarged. Palpation for splenomegaly is only moderately sensitive but highly specific. The positive LR of splenomegaly when the spleen is palpable is 9.6 and the negative LR of splenomegaly if the spleen is not palpable is 0.6.5 Examination of the spleen is discussed further in Chapter 21. The causes of splenomegaly are listed in Table 21.5 (page 276). The causes of hepatosplenomegaly are listed in List 14.9.
Kidneys
The first important differential diagnosis to consider, if a right or left subcostal mass is palpable, must be a kidney. An attempt to palpate the kidney should be a routine part of the examination. The bimanual method is the best. The patient lies flat on his or her back. To palpate the right kidney, slide your left hand underneath the patient’s back to rest with the heel of the hand under the right loin. The fingers remain free to flex at the metacarpophalangeal joints in the area of the renal angle. The flexing fingers can push the contents of the abdomen anteriorly. Place your right hand over the right upper quadrant.
First an attempt should be made to capture the kidney between both hands. It is more often possible to feel a kidney by bimanual palpation (this is traditionally called ballotting, although this term should probably be reserved for palpation of an organ or a mass in a fluid medium). In this case the renal angle is pressed sharply by the flexing fingers of the posterior hand. The kidney can be felt to float upwards and strike the anterior hand. The opposite hands are used to palpate the left kidney.
When palpable, the kidney feels like a swelling with a rounded lower pole and a medial dent (the hilum). However, it is unusual for a normal kidney to be felt as clearly as this. The lower pole of the right kidney may be palpable in thin, healthy people. Both kidneys move downwards with inspiration.
It is particularly common to confuse a large left kidney with splenomegaly. The major distinguishing features are: (1) the spleen has no palpable upper border—the space between the spleen and the costal margin, which is present in renal enlargement, cannot be felt; (2) the spleen, unlike the kidney, has a notch that may be palpable; (3) the spleen moves inferomedially on inspiration while the kidney moves inferiorly; (4) the spleen is not usually ballottable unless gross ascites is present, but the kidney is, again because of its retroperitoneal position; (5) the percussion note is dull over the spleen but is usually resonant over the kidney, as the latter lies posterior to loops of gas-filled bowel; and (6) a friction rub may occasionally be heard over the spleen, but never over the kidney because it is too posterior.
Other abdominal masses
The causes of a mass in the abdomen, excluding the liver, spleen and kidneys, are summarised in List 14.10.
Stomach and duodenum
Although many clinicians palpate the epigastrium to elicit tenderness in patients with suspected peptic ulcer, the presence or absence of tenderness is not helpful in making this diagnosis.
With gastric outlet obstruction due to a peptic ulcer or gastric carcinoma, the ‘succussion splash’ (the sign of Hippocrates) may occasionally be present. In a case of suspected gastric outlet obstruction, after warning the patient what is to come, grasp one iliac crest with each hand, place your stethoscope close to the epigastrium and shake the patient vigorously from side to side. The listening ears eagerly await a splashing noise due to excessive fluid retained in an obstructed stomach. The test is not useful if the patient has just drunk a large amount of milk or other fluid for his or her ulcer; the clinician must then return 4 hours later, having forbidden the patient to drink anything further.
Pancreas
A pancreatic pseudocyst following acute pancreatitis may occasionally, if large, be palpable as a rounded swelling above the umbilicus. It is characteristically tense, does not descend with inspiration and feels fixed. Occasionally a pancreatic carcinoma may be palpable in thin patients.
Aorta
Arterial pulsation from the abdominal aorta may be present, usually in the epigastrium, in thin healthy people. The problem is to determine whether such a pulsation represents an aortic aneurysm (usually due to atherosclerosis) or not. Measure the width of the pulsation gently with two fingers by aligning these parallel to the aorta and placing them at the outermost palpable margins. With an aortic aneurysm, the pulsation is expansile (i.e. it enlarges appreciably with systole; see Figure 14.35). If an abdominal aortic aneurysm is larger than 5 centimetres in diameter, it usually merits repair. The sensitivity of examination for finding an aneurysm of 5 centimetres or larger is 82%.6,7 The sensitivity of the examination for detecting an aneurysm increases pari passu with the size of the aneurysm. The overall LR for a significant aneurysm when one is suspected on palpation is 2.7, with a negative LR of 0.43 if the examination is normal.5
Bowel
Particularly in severely constipated patients with soft abdominal walls and retained faeces, the sigmoid colon is often palpable. Unlike other masses, faeces can usually be indented by the examiner’s finger. Rarely, carcinoma of the bowel may be palpable, particularly in the caecum where masses can grow to a large size before they cause obstruction. Such a mass does not move on respiration. In the examination of children or adults with chronic constipation and a megarectum, the enlarged rectum containing impacted stool may be felt above the symphysis pubis, filling a variable part of the pelvis in the midline.
Bladder
An empty bladder is impalpable. If there is urinary retention, the full bladder may be palpable above the pubic symphysis. It forms part of the differential diagnosis of any swelling arising out of the pelvis. It is characteristically impossible to feel the bladder’s lower border. The swelling is typically regular, smooth, firm and oval-shaped. The bladder may sometimes reach as high as the umbilicus. It is unwise to make a definite diagnosis concerning a swelling coming out of the pelvis until you are sure the bladder is empty. This may require the insertion of a urinary catheter.
Testes
Palpation of the testes should be considered if indicated during the abdominal examination (page 252). Testicular atrophy occurs in chronic liver disease (e.g. alcoholic liver disease, haemochromatosis); its mechanism is believed to be similar to that responsible for gynaecomastia.
Anterior abdominal wall
The skin and muscles of the anterior abdominal wall are prone to the same sorts of lumps that occur anywhere on the surface of the body (see List 14.11). So to avoid embarrassment it is important not to confuse these with intra-abdominal lumps. To determine whether a mass is in the abdominal wall, ask the patient to fold the arms across the upper chest and sit halfway up. An intra-abdominal mass disappears or decreases in size, but one within the layers of the abdominal wall will remain unchanged. A divarication of the rectus sheath will be obvious when a patient sits up (see Figure 14.36). This weakness in the abdominal wall aponeurosis is very common and causes bulging of the central part of the abdomen when intra-abdominal pressure increases.
Pain can arise from the abdominal wall; this can cause confusion with intra-abdominal causes of pain. To test for abdominal wall pain, feel for an area of localised tenderness that reproduces the pain while the patient is supine. If this is found, ask the patient to fold the arms across the upper chest and sit halfway up, then palpate againa. If the tenderness disappears, this suggests that the pain is in the abdominal cavity (as tensed abdominal muscles are protecting the viscera), but if the tenderness persists or is greater, this suggests that the pain is arising from the abdominal wall (e.g. muscle strain, nerve entrapment, myositis).8–10 However, Carnett’s test may occasionally be positive when there is visceral disease with involvement of the parietal peritoneum producing inflammation of the overlying muscle (e.g. appendicitis).
PERCUSSION
Percussion is used to define the size and nature of organs and masses, but is most useful for detecting fluid in the peritoneal cavity, and for eliciting tenderness in patients with peritonitis.
Liver
The liver borders should be percussed routinely to determine the liver span. If the liver edge is not palpable and there is no ascites, the right side of the abdomen should be percussed in the midclavicular line up to the right costal margin until dullness is encountered. This defines the liver’s lower border even when it is not palpable. The upper border of the liver must always be defined by percussing down the midclavicular line. Loss of normal liver dullness may occur in massive hepatic necrosis, or with free gas in the peritoneal cavity (e.g. perforated bowel).
Spleen
Percussion over the left costal margin is more sensitive than palpation for detection of enlargement of the spleen. Percuss over the lowest intercostal space in the left anterior axillary line in both inspiration and expiration with the patient supine (see Figure 14.37). Splenomegaly should be suspected if the percussion note is dull or becomes dull on complete inspiration. If the percussion note is dull, palpation should be repeated.
ASCITES
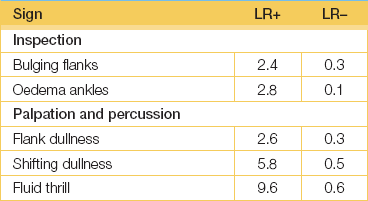
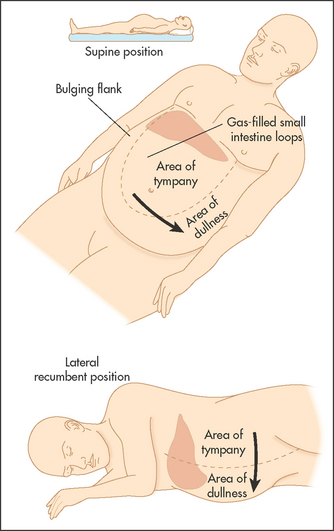
The percussion note over most of the abdomen is resonant, due to air in the intestines.11 The resonance is detectable out to the flanks. When peritoneal fluid (ascites) collects, the influence of gravity causes this to accumulate first in the flanks in a supine patient. Thus, a relatively early sign of ascites (when at least 2 litres of fluid have accumulated) is a dull percussion note in the flanks (see Good signs guide 14.2). When ascites is gross, the abdomen distends, the flanks bulge, umbilical eversion occurs (see Figure 14.25) and dullness is detectable closer to the midline. However, an area of central resonance will always persist. Routine abdominal examination should include percussion starting in the midline with the finger pointing towards the feet; the percussion note is tested out towards the flanks on each side.
If (and only if) dullness is detected in the flanks, the sign of shifting dullness (see Figure 14.38) should be sought.11 To detect this sign, while standing on the right side of the bed percuss out to the left flank until dullness is reached (see Figure 14.39(a)). This point should be marked (usually by leaving a finger over the spot) and the patient rolled towards you. Ideally 30 seconds to 1 minute should then pass so that fluid can move inside the abdominal cavity and then percussion is repeated over the marked point (see Figure 14.39(b)).
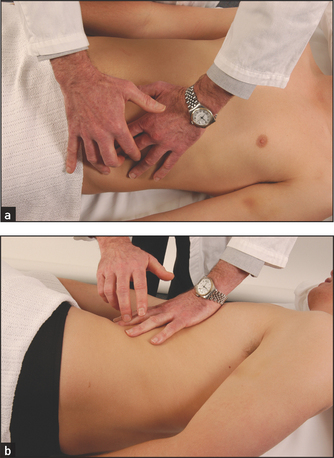
Figure 14.39 Shifting dullness (a) Percuss out to the left flank until the percussion note becomes dull. Mark this spot with your finger. (b) Roll the patient towards you, wait 30 seconds. Shifting dullness is present if the left lateral dull area is now resonant. (Courtesy of Glenn McCulloch)
Shifting dullness is present if the area of dullness has changed to become resonant. This is because peritoneal fluid moves under the influence of gravity to the right side of the abdomen when this is the lowermost point. Very occasionally, fluid and air in dilated small bowel in small intestinal obstruction, or a massive ovarian cyst filling the whole abdomen, can cause confusion.
To detect a fluid thrill (or wave), ask an assistant (or the patient) to place the medial edges of both palms firmly on the centre of the abdomen with the fingers pointing towards each other. Flick the side of the abdominal wall; a pulsation (thrill) is felt by the hand placed on the other abdominal wall. A fluid thrill is of more value when there is massive ascites. Interestingly, it may also occur when there is a massive ovarian cyst or a pregnancy with hydramnios.
The presence of bulging in the flanks has good sensitivity and specificity for the detection of ascites. Shifting dullness has both good sensitivity and specificity. The presence of ankle oedema increases the likelihood of ascites. (See Good signs guide 14.2.)
The causes of ascites are outlined in List 14.12.
When significant ascites is present, abdominal masses may be difficult to feel by direct palpation. Here is the opportunity to practise dipping. Using the hand placed flat on the abdomen, the fingers are flexed at the metacarpophalangeal joints rapidly so as to displace the underlying fluid. This enables the fingers to reach a mass covered in ascitic fluid. In particular, this should be attempted to palpate an enlarged liver or spleen. The liver and spleen may become ballottable when gross ascites is present.
AUSCULTATION
While some cardiologists believe that the sounds produced in the abdominal cavity are not as varied or as interesting as those one hears in the chest, they have some value.
Bowel sounds
Place the diaphragm of the stethoscope just below the umbilicus (see Figure 14.40). Bowel sounds can be heard over all parts of the abdomen in healthy people. They are poorly localised and there is little point in listening for them in more than one place. Most bowel sounds originate in the stomach, some from the large bowel and the rest from the small bowel. They have a soft gurgling character and occur only intermittently. Bowel sounds should be described as either present or absent; the terms ‘decreased’ or ‘increased’ are meaningless because the sounds vary, depending on when a meal was last eaten.
Figure 14.40 Auscultation of the abdomen (From Swartz MH. Textbook of physical diagnosis, 6th edn. Saunders 2009.)
Complete absence of any bowel sounds over a 4-minute period indicates paralytic ileus (this is complete absence of peristalsis in a paralysed bowel). As only liquid is present in the gut, the heart sounds may be audible over the abdomen, transmitted by the dilated bowel.
The bowel that is obstructed produces a louder and higher-pitched sound with a tinkling quality due to the presence of air and liquid (‘obstructed bowel sounds’). The presence of normal bowel sounds makes obstruction unlikely. Intestinal hurry or rush, which occurs in diarrhoeal states, causes loud gurgling sounds, often audible without the stethoscope. These bowel sounds are called borborygmi.
Friction rubs
These indicate an abnormality of the parietal and visceral peritoneum due to inflammation, but are very rare and nonspecific.
They may be audible over the liver or spleen. A rough creaking or grating noise is heard as the patient breathes. Hepatic causes include a tumour within the liver (hepatocellular cancer or metastases), a liver abscess, a recent liver biopsy, a liver infarct, or gonococcal or chlamydial perihepatitis due to inflammation of the liver capsule (Fitz-Hugh Curtis syndromea). A splenic rub indicates a splenic infarct.
Venous hums
A venous hum is a continuous, low-pitched, soft murmur that may become louder with inspiration and diminish when more pressure is applied to the stethoscope. Typically it is heard between the xiphisternum and the umbilicus in cases of portal hypertension, but is rare. It may radiate to the chest or over to the liver. Large volumes of blood flowing in the umbilical or para-umbilical veins in the falciform ligament are responsible. These channel blood from the left portal vein to the epigastric or internal mammary veins in the abdominal wall. A venous hum may occasionally be heard over the large vessels such as the inferior mesenteric vein or after portacaval shunting. Sometimes a thrill is detectable over the site of maximum intensity of the hum. The Cruveilhier-Baumgarten syndromeaa is the association of a venous hum at the umbilicus and dilated abdominal wall veins. It is almost always due to cirrhosis of the liver. It occurs when patients have a patent umbilical vein, which allows portal-to-systemic shunting at this site. The presence of a venous hum or of prominent central abdominal veins suggests that the site of portal obstruction is intrahepatic rather than in the portal vein itself.
Bruits
Uncommonly, an arterial systolic bruit can be heard over the liver. This sound is higher pitched than a venous hum, is not continuous and is well localised. This is usually due to a hepatocellular cancer but may occur in acute alcoholic hepatitis, with an arteriovenous malformation or transiently after a liver biopsy. Auscultation for renal bruits on either side of the midline above the umbilicus is indicated if renal artery stenosis is suspected. A bruit in the epigastrium may be heard in patients with chronic intestinal ischaemia from mesenteric arterial stenosis, but may also occur in the absence of pathology. A bruit may occasionally be audible over the spleen when there is a tumour of the body of the pancreas or a splenic arteriovenous fistula.
Scratch test
The scratch test can help identify the liver border if the abdomen is very tender, tense or distended. Place the stethoscope below the xiphoid and lightly but briskly stroke the skin in a direction at right angles to the expected liver edge, starting at the right lower quadrant and working slowly up to the right costal margin along the midclavicular line. When the liver edge is reached, the sound of the scratch is transmitted to the stethoscope. The point of initial (not maximal) sound transmission is most helpful in identifying the liver edge. This test has been controversial but is reliable with moderate accuracy (versus abdominal ultrasound).12
Hernias
Hernias are of surgical importance and should not be missed during an abdominal examination. They are very frequently the focus of student assessment examinations.
EXAMINATION ANATOMY
You need to know the anatomy of the inguinal and femoral canal. A key anatomical landmark is the pubic tubercle (or pubic spine), a palpable round nodule (tubercle) on the upper border of the medial portion of the superior ramus of the pubis (a midline cartilaginous joint uniting the superior rami of the left and right pubic bones). The inguinal ligament attaches to the pubic tubercle (see Figure 14.41). Find the femoral artery pulsation and go medially to identify the tubercle. The pubic tubercle can usually easily be felt lateral to the symphysis pubis (2 to 3 centimetres from the midline). In the obese individual, it may be difficult to locate the pubic tubercle; in such situations, if the thigh is flexed and abducted, the adductor longus muscle can be traced proximally, leading you to the pubic tubercle.
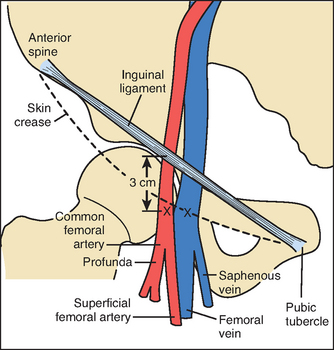
Figure 14.41 Anatomy of the pubic tubercle and inguinal ligament (From Kaplan J et al. Kaplan’s cardiac anesthesia, 6th edn. Saunders 2011.)
From the pubic tubercle to the anterior superior iliac spine lies the inguinal canal. At the mid-inguinal point (midway between the pubic symphysis and the anterior superior iliac spine) is an internal ring. At the pubic tubercle lies an external ring, in men the gateway to the scrotum. Remember that the femoral canal is situated lateral to the pubic tubercle, below the inguinal ligament.
HERNIAS IN THE GROIN
The principal sign of a hernia is a lump in the groin. Naturally, not all lumps in the region are hernias (see List 14.13).
A groin lump that is present on standing or during manoeuvres that raise intra-abdominal pressure (such as coughing or straining), and that disappears on recumbency (or reduces), is easily identified as a hernia.
Some hernias are irreducible. Another term used for irreducible is incarcerated, but this term is best avoided. Some irreducible hernias contain bowel, which may become obstructed, giving rise to symptoms of small bowel obstruction in addition to the irreducible lump. Sometimes the bowel contents’ blood supply becomes jeopardised, and these are known as strangulated hernias; they are usually painful, red, tense and tender.
Inguinal hernias typically bulge above the crease of the groin. The lump is found medial to the pubic tubercle, above the inguinal ligament (remember, inguinal = medial and above). Inguinal hernias may be indirect or direct. Indirect inguinal hernias are by far the most common groin hernia. The bowel follows the inguinal canal, bulging through the internal ring and sometimes out through the external ring. Direct inguinal hernias are usually the result of a muscular defect of the inguinal canal (an area known as Hesselbach’s triangle,bb bounded inferiorly by the inguinal ligament, laterally by the inferior epigastric vessels and medially by the lateral border of the rectus sheath). Usually the hernia does not reach the scrotum. However, differentiating a direct from an indirect hernia is difficult based on clinical signs alone.
A femoral hernia bulges through the femoral ring into the femoral canal. Femoral hernias bulge into the groin crease at its medial end. The lump is found lateral to the public tubercle, below the inguinal ligament (remember, femoral = lateral and below).
Examination technique
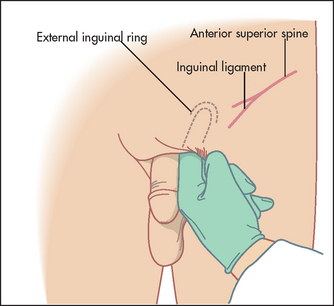
1. Wash your hands and glove up.
2. Position the patient: A thorough examination for a hernia should be commenced with the patient standing if possible. The patient should be asked to stand with full exposure from the thigh to the upper abdomen.
3. Point sign: Ask the patient to point where the lump has been seen or felt.
4. Inspect: Pay careful attention to scars from previous surgery, which may be difficult to see. Look for obvious lumps and swellings on both sides.
5. Cough impulse: Before palpation, ask the patient to turn his or her head away from you and cough. Fix your eyes in the region of the pubic tubercle and note the presence of a visible cough impulse. Ask the patient to cough again while you inspect the opposite side.
6. Palpation: Place your fingers over the region of the pubic tubercle. Once again the patient is asked to cough and a palpable expansile impulse is sought. If a hernia is present, attempts at reduction should not be performed while the patient is erect, as it is more difficult and painful than when the patient is placed supine.
7. Lay the patient supine: Ask the patient to lie supine on the examination couch. Perform the procedure of inspection and palpation in the same manner again. The exact position of any hernia is usually easier to define with the patient lying supine.
8. Identify the hernia type: If a lump is present, it must be determined whether this is a hernia and, if so, what sort of hernia. Identify the pubic tubercle. Remember, one cannot get above a hernia, but one can get above a hydrocele in the inguinal canal. Try to determine whether the hernia is inguinal (see Figure 14.42) or femoral based on the position in relation to the pubic tubercle and inguinal ligament (see above).
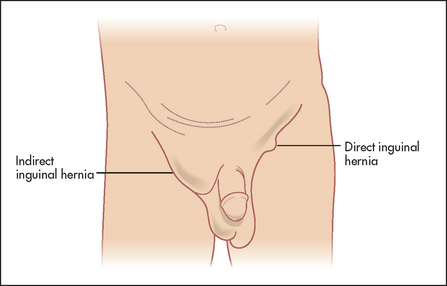
Figure 14.42 Note the elliptical swelling of an indirect inguinal hernia descending into the scrotum on the right side. Also note the globular swelling of a direct inguinal hernia on the left side
Remember, femoral hernias are more dangerous: they are usually smaller and firmer than inguinal hernias and commonly do not exhibit a cough impulse. Because they are frequently irreducible they are commonly mistaken for an enlarged inguinal lymph node. A cough impulse is rare from a femoral hernia and needs to be distinguished from the thrill produced by a saphena varix when a patient coughs.
9. In a male, examine the testes and scrotum. A large inguinal hernia may descend through the external ring immediately above the pubic tubercle into the scrotum. Gentle invagination of the scrotum with the tip of the gloved finger in the external ring may be performed to confirm an indirect hernia in men, but this can be difficult to interpret without substantial experience (see Figure 14.43). A maldescended testis can be confused with an inguinal hernia; always confirm that there is a testis in each scrotum. A large inguinal hernia may present as a lump in the scrotum. It is important to ascertain whether one can get above the lump. If you can get above the lump, the lump is of primary intrascrotal pathology and is not a hernia.
EPIGASTRIC HERNIA
A hernia in the epigastric region is common in older patients. This can be identified by asking the patient to do half a sit up from the supine position and looking for an obvious bulge. Identify scars that may explain the abdominal wall weakness. Ask the patient if the lump is painful and palpate it. Feel for a cough impulse. Asymptomatic epigastric hernias are usually best left alone!
INCISIONAL HERNIAS
Any abdominal scar may be the site of a hernia because of abdominal wall weakness. Assess this by asking the patient to cough while you look for abnormal bulges. Next have the patient lift the head and shoulders off the bed while you rest your hand on the patient’s forehead and resist this movement. If a bulge is seen, use your other hand to palpate for a fascial layer defect in the muscle, and test the cough impulse.
Rectal examination
The examining physician often hesitates to make the necessary examination because it involves soiling the finger.
The abdominal examination is not complete without the performance of a rectal examination (with the aid of gloves; see Figure 14.44).13,14 It should be considered in all patients with bowel symptoms admitted to hospital and who are aged 40 or older, unless the examiner has no fingers, the patient no anus or acute illness such as myocardial infarction presents a temporary contraindication.
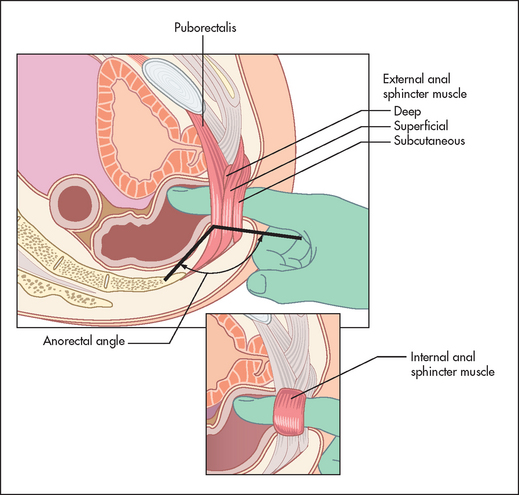
Figure 14.44 The rectal examination: regional anatomy (From Talley NJ. American Journal of Gastroenterology 2008; 108:802–803, with permission.)
The patient’s permission must be obtained and, if indicated, a chaperone introduced to the patient. Privacy must be ensured for the patient throughout the examination. Following an explanation as to what is to happen and why, the patient lies on his or her left side with the knees drawn up and back to the examiner. This is called the left lateral position. The examination can be performed with the patient standing and in the bent-over position; this may help provide good information about the prostate, but makes assessment of rectal function more difficult.
Don a pair of gloves and begin the inspection of the anus and perianal area by separating the buttocks. The following must be looked for:
1. Thrombosed external haemorrhoids (piles). Small (less than 1 centimetre), tense bluish swellings may be seen on one side of the anal margin. They are painful and are due to rupture of a vein in the external haemorrhoidal plexus. They are also called perianal haematomas.
2. Skin tags. These look like tags elsewhere on the body and can be an incidental finding or occur with haemorrhoids or Crohn’s disease.
3. Rectal prolapse. Circumferential folds of red mucosa are visible protruding from the anus. These may become apparent only when the patient is asked to strain as if to pass stool. If there is rectal prolapse, straining may cause a dark red mass to appear at the anal verge; mucosal prolapse causes the appearance of radial folds, while concentric folds are a sign of complete prolapse. This mass is continuous with the perianal skin and is usually painless. In cases of mucosal rectal prolapse, the prolapsed mucosa can be felt between the examiner’s thumb and forefinger. A gaping anus suggests loss of internal and external sphincter tone. This may coexist with prolapse.
4. Anal fissure (fissure-in-ano). This is a crack in the anal wall that may be painful enough to prevent rectal examination with the finger. Fissures-in-ano usually occur directly posteriorly and in the midline. A tag of skin may be present at the base: this is called a sentinel pile and indicates that the fissure is chronic. It may be necessary to get the patient to bear down for a fissure to become visible. Multiple or broad-based fissures may be present in patients with inflammatory bowel disease, malignancy or sexually transmitted disease.
5. Fistula-in-ano. The entrance of this tract may be visible, usually within 4 centimetres of the anus. The mouth has a red pouting appearance caused by granulation tissue. This may occur with Crohn’s disease or perianal abscess (page 224).
6. Condylomata acuminata (anal warts). Condylomata (see Figure 14.45) may be confused with skin tags, but are in fact pedunculated papillomas with a white surface and red base. They may surround the anus.
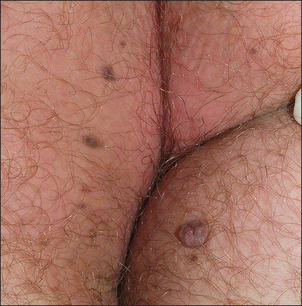
Figure 14.45 Condylomata (anal warts) (From Venkatesan A. Pigmented lesions of the vulva. Dermatologic Clinics 2010; 28(4):795–805.)
7. Carcinoma of the anus. This may be visible as a fungating mass at the anal verge.
8. Pruritus ani. The appearance of this irritating anal condition varies from weeping red dermatitis to a thickened white skin. It is usually caused by faecal soiling.
Next ask the patient to strain and watch the perineum: look for incontinence and leakage of faeces or mucus, abnormal descent of the perineum (>4 centimetres) or the presence of a patulous anus. The presence of a gaping anus often correlates with lower resting pressures on anorectal manometry. Internal haemorrhoids can prolapse in the right anterior, right posterior and left lateral positions.
Now test the anal wink. Stroke a cotton pad in all four quadrants around the anus. Usually you will see a brisk anal contraction that indicates the sacral nerve pathways are intact. Sometimes the response is weak in healthy people. However, the complete absence of an anal wink, particularly in the setting of faecal incontinence, suggests that there is a spinal cord problem and indicates the need to perform a more detailed neurological examination and consider further investigations accordingly.
Now the time for action has come. Lubricate the tip of your gloved right index finger and placed it over the patient’s anus. Ask the patient to breathe in and out quietly through the mouth, as a distraction and to aid relaxation.
If the patient feels excruciating pain at the start of the examination, this strongly suggests that there is an anal fissure and the remainder of the rectal examination should be abandoned. Often the fissure can be seen on inspection. An anal fissure can precipitate constipation but may be secondary to constipation itself. By liberally lubricating the rectum with lignocaine jelly, it may still be possible to complete the rest of the examination, but usually it is better to perform anoscopy under appropriate sedation for these patients. Other causes of significant anal pain during palpation include recently thrombosed external haemorrhoids, an ischiorectal abscess, active proctitis or anal ulceration from another cause.
Unless the patient feels pain, slowly increase the pressure applied with the pulp of your finger until the sphincter is felt to relax slightly. At this stage advance your finger into the rectum slowly. During entry, sphincter tone should be assessed as normal or reduced. The accuracy of this assessment has been questioned in the past, but more recently it has been shown to correlate well with anorectal manometry measurements.15 This resting tone is predominantly (70%–80%) attributable to the internal anal sphincter muscle. Reduced sphincter tone may indicate a sphincter tear. A high anal resting tone may contribute to difficulties with evacuation.
Palpation of the anterior wall of the rectum for the prostate gland in the male and for the cervix in the female is performed first. The normal prostate is a firm, rubbery bilobed mass with a central furrow. It becomes firmer with age. With prostatic enlargement, the sulcus becomes obliterated and the gland is often asymmetrical. A very hard nodule is apparent when a carcinoma of the prostate is present. The prostate is boggy and tender in prostatitis. A mass above the prostate or cervix may indicate a metastatic deposit on Blumer’s shelf.cc
Rotate your finger clockwise so that the left lateral wall, posterior wall and right lateral wall of the rectum can be palpated in turn. Then advance your finger as high as possible into the rectum and slowly withdraw along the rectal wall. A soft lesion, such as a small rectal carcinoma or polyp, is more likely to be felt in this waydd (see List 14.14).
THE PELVIC FLOOR—SPECIAL TESTS FOR PELVIC FLOOR DYSFUNCTION
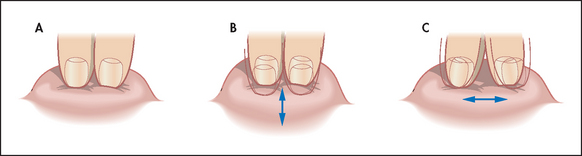
The first test is simple: ask the patient to strain and try to push out your finger. Normally, the anal sphincter and puborectalis should relax and the perineum should descend by 1–3.5 centimetres. If the muscles seem to tighten, particularly when there is no perineal descent, this suggests paradoxical external anal sphincter and puborectalis contraction, which in fact are blocking normal defecation (called pelvic floor dyssynergia or anismus). Ask the patient to strain again when your finger is rotated anteriorly. In this position a rectocele (a defect in the anterior wall of the rectum) may be palpable.
Second, press on the posterior rectal wall and ask if this causes pain; this suggests puborectalis muscle tenderness, which can also occur in pelvic floor dyssynergia.
Third, assess whether the anal sphincter and the puborectalis contract when you ask the patient to contract or squeeze the pelvic floor muscles. Puborectalis contraction is perceived as a ‘lift’; that is, the muscle lifts the examining finger towards the umbilicus. Many patients with faecal incontinence cannot augment anal pressure when asked to squeeze.
Finally, place your other hand on the anterior abdominal wall while asking the patient to strain again. This provides some information on whether the patient is excessively contracting the abdominal wall (e.g. by performing an inappropriate Valsalva manoeuvre) and perhaps also the pelvic floor muscles while attempting to defecate, which may impede evacuation. However, the exact value of this test is unclear.
Constipation that is due to pelvic floor dysfunction responds to biofeedback in about 70% of cases, and this treatment can result in a laxative-free existence for patients with troubling outlet constipation; the diagnosis should be entertained in all patients with chronic constipation, and a good rectal examination can help guide you as to whether anorectal manometry testing is warranted.
ENDING THE RECTAL EXAM
After your finger has been withdrawn, inspect the glove for bright blood or melaena, mucus or pus, and note the colour of the faeces. Haemorrhoids are not palpable unless thrombosed. Persistent gaping of the anal canal after withdrawal of the examining finger may indicate external anal sphincter denervation.
Proctosigmoidoscopy
The examination of the rectum with a sigmoidoscope is an extension of the physical examination for patients with symptoms referable to the anorectal region or large bowel. The principal indications include rectal bleeding, chronic diarrhoea, constipation or a change in bowel habit. It may usefully be performed in patients with abdominal pain, before treatment is begun for any anorectal condition and before a barium enema is ordered for any reason.
The examination can be performed without anaesthesia, except in patients who have a very painful anal condition.
PROCEDURE
Begin by inspecting the anal area as outlined earlier. Then perform a digital examination of the rectum.
Explain to the patient what is about to happen, warning that there will be a feeling of fullness and the desire to defecate, and possibly cramps in the rectal region. Then place the patient in the left lateral position and ask him or her to relax and breathe quietly through the mouth.
If a rigid sigmoidoscope is to be used, it is warmed slightly and, with the obturator in place, is inserted into the rectum in the direction of the umbilicus until the rectal ampulla is reached (4–5 centimetres). This is the only part of the examination that is performed blind. The obturator is then removed. The tip of the sigmoidoscope is gently swung posteriorly under direct vision to follow the curve of the sacrum. The important landmarks to note during sigmoidoscopy are the anal verge, the dentate line, the anorectal junction, the lowest and middle rectal valves and finally the rectosigmoid junction. Small amounts of air may be insufflated to assist with this. At about 12–15 centimetres, smooth rectal mucosa gives way to the concentric rugae of the distal sigmoid. It is possible to advance the rigid instrument into the distal sigmoid in the majority of men and in many women. The flexible sigmoidoscope can usually examine the entire left colon in skilled hands. The instrument must never be advanced unless the lumen is clearly visible and the patient is not experiencing pain.
Once the sigmoidoscope has been advanced as far as possible, it should be withdrawn gradually while the circumference of the mucosa is inspected carefully. Look behind for the valves of Houston.ee It is possible to sample faeces from areas away from the anal margin, which can be tested for occult blood and subjected to microbiological examination. Mucosal lesions can also be biopsied.
COMMON ABNORMALITIES SEEN ON SIGMOIDOSCOPY
1. Blood, seen to be arising from above the highest level examined, indicates that a total examination of the colon by colonoscopy is essential
2. Erythematous and ulcerated areas indicating inflammation, which may be local or diffuse
3. Mucosal oedema, where there is loss of the normal vascular pattern of the colon, may be seen in mild inflammatory bowel disease
4. Polyps, which may be sessile or pedunculated, solitary or multiple
6. Strictures, which may be due to carcinoma, Crohn’s disease, trauma, ischaemia, radiation or (very rarely) tuberculosis
If an abnormality of the anal canal is suspected, this is best seen using anoscopy, which can be carried out after sigmoidoscopy. Lesions to look for in the anal canal include swellings, masses, fissures, the internal openings of fistulae, squamous metaplasia and haemorrhoids. Haemorrhoids appear as swellings at the site of the normal anal cushions at 3, 7 and 11 o’clock, and they descend on straining. Remember that haemorrhoids are common and may coexist with more sinister bowel disease.
TESTING OF THE STOOLS FOR BLOOD
Testing of the stools for blood may be considered in the assessment of anaemia, iron deficiency, gastrointestinal bleeding or symptoms suggesting colonic cancer. In the guaiac test, stool is placed on a guaiac-impregnated paper; blood results in phenolytic oxidation, causing a blue colour. Newer tests can quantify the amount of blood in the stool.
Unfortunately, both false-positive and false-negative results occur with the occult blood tests. Peroxidase and catalase, present in various foods (e.g. fresh fruit, uncooked vegetables), and haem in red meat can cause false-positive results, as can aspirin, anticoagulants and oral iron. Vitamin C can reduce the sensitivity of guaiac results and should not be taken before testing. False-negative results are not uncommon with colorectal neoplasms because they bleed intermittently. Hence testing for faecal occult blood from the glove after a rectal examination is of little value,16 and more sensitive and specific testing (e.g. colonoscopy) is required, depending on the clinical setting.
Other
Examine the legs for bruising or oedema, which may be the result of liver disease. Neurological signs of alcoholism (e.g. a coarse tremor) or evidence of thiamine deficiency (peripheral neuropathy or memory loss) may also be present.
Examination of the cardiovascular system may be helpful in patients with hepatomegaly. Cardiac failure is a common cause of liver enlargement and can even cause cirrhosis. Measurement of the patient’s temperature is important, especially in an acute abdominal case or if there is any suggestion of infection.
Examine with particular care all lymph node groups, the breasts and chest if there is any evidence of malignant disease such as firm, irregular hepatomegaly.
Examination of the gastrointestinal contents
FAECES
Never miss an opportunity to inspect a patient’s faeces, because considerable information about the gastrointestinal tract can be obtained in this way.
Melaena
Melaena stools are poorly formed, black and have a tarry appearance. They have a very characteristic and offensive smell. The cause is the presence of blood digested by gastric acid and colonic bacteria. Melaena usually indicates bleeding from the oesophagus, stomach or duodenum. The most common cause is acute or chronic peptic ulceration. Less often, right-sided colonic bleeding and (rarely) small-bowel bleeding can cause melaena. The differential diagnosis of dark stools includes ingestion of iron tablets, bismuth, liquorice or charcoal. However, these tend to result in small well-formed non-tarry stools and the offensive smell is absent.
Bright-red blood (haematochezia)
This appearance usually results from haemorrhage from the rectum or left colon. Beetroot ingestion can sometimes cause confusion. Blood loss may result from a carcinoma or polyp, an arteriovenous malformation, inflammatory bowel disease or diverticular disease. It can occasionally occur with massive upper gastrointestinal bleeding. The blood is usually mixed in with the bowel motion if it comes from above the anorectum, but if blood appears on the surface of the stool or only on the toilet paper this suggests, but does not guarantee, that bleeding is from a local rectal cause, such as internal haemorrhoids or a fissure. Dark-red jelly-like stools may be seen with ischaemic bowel.
Steatorrhoea
The stools are usually very pale, offensive, smelly and bulky. They float and are difficult to flush away. However, the most common cause of floating stools is gas and water rather than fat.
Steatorrhoea results from malabsorption of fat. In severe pancreatic disease, oil (triglycerides) may be passed per rectum and this is virtually pathognomonic of pancreatic steatorrhoea (lipase deficiency). Oil may also be seen when the patient has used a weight-loss drug that induces fat malabsorption (orlistat).
VOMITUS
The clinician who is fortunate enough to have vomitus available for inspection (ill-informed staff may throw out this valuable substance) should not lose the opportunity of a detailed examination. There are a number of interesting types of vomitus.
‘Coffee grounds’
An old blood clot in vomitus has the appearance of the dregs of a good cup of espresso coffee. Unfortunately, darker vomitus is often described as having this appearance. This emphasises the need for personal inspection. Iron tablets and red wine, not to mention coffee ingestion, can have the same effect on the vomitus.
Bright-red blood (haematemesis)
Look for the presence of fresh clot. It usually indicates fresh bleeding from the upper gastrointestinal tract.
Yellow-green vomitus
This results from the vomiting of bile and upper small bowel contents, often when there is obstruction.
Faeculent vomiting
Here brown offensive material from the small bowel is vomited. It is a late sign of small intestinal obstruction. Recently ingested tea can have the same appearance but lacks the smell.
Brownish-black fluid in large volumes may be vomited in cases of acute dilation of the stomach. A succussion splash will usually be present. Acute dilation may occur in association with diabetic ketoacidosis or following abdominal surgery. It represents a medical emergency because of the risk of aspiration; there is a need for urgent placement of a nasogastric tube.
Urinalysis
Note that testing of the urine can be very helpful in diagnosing liver disease.
Strip colour tests can detect the presence of bilirubin and urobilinogen in the urine. False-positive or false-negative results can occur with vitamin C or even exposure to sunlight. An understanding of the reasons for the presence of bilirubin or urobilinogen in the urine necessitates an explanation of the metabolism of these substances (see Figure 14.46).
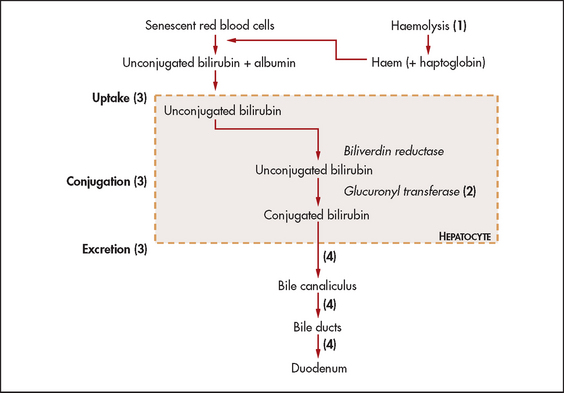
Figure 14.46 Schematic representation of the bilirubin pathway Increased haemolysis (1) overwhelms the hepatocytes’ ability to conjugate bilirubin and excrete the conjugated form, leading to increased serum levels of unconjugated bilirubin. Low levels of glucuronyl transferase (2) (e.g. Gilbert’s disease) cause decreased conjugation. Hepatocellular dysfunction (3) causes decreased uptake, conjugation and excretion with increases of unconjugated bilirubin and conjugated bilirubin. Post-hepatic obstruction (4) from stones or tumour prevents passage of bilirubin through the bile ducts into the bowel, leading to increased serum levels of conjugated bilirubin.
Red blood cells are broken down by the reticuloendothelial system, causing the release of haem, which is converted to biliverdin and then unconjugated bilirubin, a water-insoluble compound. For this reason, unconjugated bilirubin released with haemolytic anaemia will not appear in the urine (termed acholuric jaundice).
Unconjugated bilirubin is transported in the blood bound largely to albumin but also to other plasma proteins. Unconjugated bilirubin is then taken up by the liver cells and transported to the endoplasmic reticulum, where glucuronyl transferases conjugate bilirubin with glucuronide. This results in the formation of conjugated bilirubin, which is water-soluble. Conjugated bilirubin is then concentrated and excreted by the liver cells into the canaliculus.
Conjugated bilirubin is virtually all excreted into the small bowel; it is converted in the terminal ileum and colon to urobilinogen, and then to stercobilin. Stercobilin is responsible for the normal colour of the stools with other non-bilirubinoid dietary pigments. Up to 20% of urobilinogen is reabsorbed by the bowel, and small amounts are excreted in the urine as urinary urobilinogen. This can often be normally detected by reagent strips.
Total biliary obstruction, from whatever cause, results in absence of urinary urobilinogen, as no conjugated bilirubin reaches the bowel, resulting in pale stools (absence of stercobilin). The conjugated bilirubin, unable to be excreted (the rate-limiting step), leaks from the hepatocytes into the blood and from there is excreted into the urine (normally there is no bilirubin detected in urine). This results in dark urine (excess conjugated bilirubin). Acute liver damage, as in viral hepatitis, may sometimes initially result in excessive urinary urobilinogen, because the liver is unable to re-excrete the urobilinogen reabsorbed from the bowel. These changes are summarised inTable 14.2.
TABLE 14.2
Changes in urine and faeces with jaundice
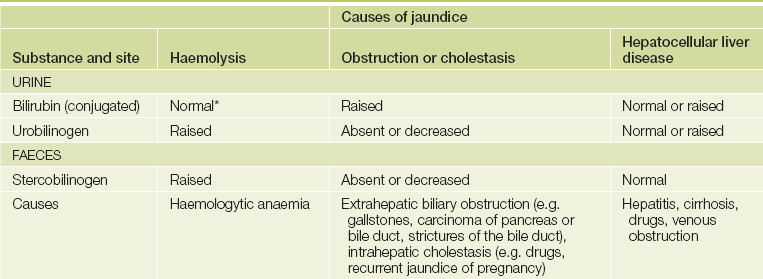
*Unconjugated bilirubin levels are elevated in the serum.
References
1. Sibarte, V, Shanahan, F. Clinical examination of the gastrointestinal system in the 21st century—is the emphasis right. Am J Gastroenterol. 2004; 99:1874–1875.
2. Walton, S, Bennett, JR. Skin and gullet. Gut. 1991; 32:694–697. [An excellent review of skin signs in gastroenterology.].
3. Beitman, RG, Rost, SS, Roth, JL. Oral manifestations of gastrointestinal disease. Dig Dis Sci. 1981; 26:741–747. [A detailed review.].
4. Naylor, CD. Physical examination of the liver. (The rational clinical examination. JAMA. 1994; 271:1859–1865. [A valuable review of technique. Palpation is probably superior to percussion (in part because the midclavicular line is a ‘wandering landmark’). Auscultation is usually of minimal value.].
5. McGee, S. Evidence-based physical diagnosis, 3rd edn. St Louis: Saunders, 2012.
6. Fink, HA, Lederle, FA, et al. The accuracy of physical examination to detect abdominal aortic aneurysm. Arch Intern Med. 2000; 160:833–836.
7. Chervu, A, Clagett, GP, Valentine, RJ, Myers, SI, Rossi, PJ. Role of physical examination in detection of abdominal aortic aneurysms. Surgery. 1995; 117:454–457.
8. Thomson, WH, Francis, DM. Abdominal wall tenderness: a useful sign in the acute abdomen. Lancet. 1977; 2:1053–1054. [While abdominal wall pain can be recognised when the patient tenses the abdominal wall muscles, intraperitoneal inflammation can cause a false-positive sign.].
9. Srinivasan, R, Greenbaum, DS. Chronic abdominal wall pain: a frequently overlooked problem. Practical approach to diagnosis and management. Am J Gastroenterol. 2002; 97:824–830.
10. Editorial. Abdominal wall tenderness test: could Carnett cut costs. Lancet. 1991; 337:1134.
11. Williams, JW, Jr., Simel, DL. Does this patient have ascites? How to divine fluid in the abdomen. (The rational clinical examination. JAMA. 1992; 267:2645–2648. [This review provides information on the discriminant value of signs. Bulging flanks, shifting dullness and a fluid wave are each reasonably sensitive and specific. The presence of ascites is more easily predicted than its absence.].
12. Gupta, K, Dhawan, A, Abel, C, Talley, N, Attia, J. A re-evaluation of the scratch test for locating the liver edge. BMC Gastroenterol. 2013; 13:35.
13. Muris, JW, Starmans, R, Wolfs, GG, Pop, P, Knottnerus, JA. The diagnostic value of rectal examination. Family Practice. 1993; 10:34–37. [A useful procedure in patients with rectal or prostatic symptoms. Based on a literature review but studies in general practice are lacking.].
14. Talley, NJ. How to do and interpret a rectal examination in gastroenterology. Am J Gastroenterol. 2008; 108:802–803.
15. Dobben, AC, Terra, MP, Deutekom, M, et al. Anal inspection and digital rectal examination compared to anorectal physiology tests and endoanal ultrasonography in evaluating faecal incontinence. Int J Colorectal Dis. 2007; 22:783–790.
16. Longstreth, GF. Checking for ‘the occult’ with a finger: a procedure of little value. J Clin Gastroenterol. 1988; 10:133–134. [A faecal occult blood test from the glove after rectal examination has too many false-positive and false-negative results to be of value.].
aJohn Peutz (1886–1957), physician at St John’s Hospital, The Hague, The Netherlands, first described this condition in 1921. Harold Jeghers (1904–90), professor of medicine, Boston City Hospital, USA, described it in 1949.
bHenri Rendu (1844–1902), French physician. Frederick Weber (1863–1962), English physician. The condition was described in 1907.
cThese changes were first described by Dr Richard Terry in 1954 in association with cirrhosis. They are also found in patients with cardiac failure and become more common in normal people with age. In a patient under the age of 50, their presence indicates cirrhosis, heart failure or diabetes, with a likelihood ratio of 5.3.
dBaron Guillaume Dupuytren (1777–1835), Surgeon-in-Chief at the Hotel-Dieu in Paris. A cold, rude, ambitious and arrogant man, he was called ‘the Napoleon of surgery’. He saw 10,000 private patients a year.
eFrom Greek a, ‘not’ and sterixis, ‘fixed position’.
fPrimary biliary cirrhosis (PBC) is an uncommon chronic nonsuppurative destructive cholangitis of unknown aetiology; 90% of affected patients are female.
gSpider naevi were first described in 1867 by Sir Erasmus Wilson (1809–84), an English surgeon and dermatologist.
hCampbell de Morgan (1811–76), London surgeon. He was one of the 300 original Fellows of the Royal College of Surgeons. He described his spots in 1872 and believed them to be a sign of cancer (which they are not).
iPierre Bitot (1822–88) described this in 1863.
jBernhard Kayser (1869–1954), German ophthalmologist, described these rings in 1902. Bruno Fleischer (1848–1904), German ophthalmologist, described them in 1903.
kJean Descemet (1732–1810), professor of surgery and anatomy, Paris. He described the membrane in 1785.
lSamuel Alexander Wilson (1878–1937), London neurologist at Queen Square. His colleagues there included Gowers and Hughlings Jackson. He described his disease in 1912 in his MD thesis. He also described the glabellar tap sign in Parkinson’s disease, which is sometimes called Wilson’s sign. He did not, however, describe the Kayser-Fleischer rings.
mThe Latin word means a stench.
nBurrill Bernard Crohn (1884–1983), American gastroenterologist at Mount Sinai Hospital, New York, described this disease in 1932. It had previously been described by Giovanni Battista Morgagni (1682–1771) in 1769.
oCharles Èmile Troisier (1844–1919), professor of pathology in Paris, described this sign in 1886.
pFranz von Leydig (1821–1908), Bonn anatomist and zoologist, Germany.
aIn Greek mythology Medusa was the only mortal among the three Gorgons. She had live snakes for hair and anyone who met her gaze was turned to stone (unless they looked at her in a mirror or polished shield).
aSister Joseph of St Mary’s Hospital, Rochester, Minnesota, described this sign to Dr William Mayo (1861–1939) of the Mayo Clinic.
aThomas S Cullen (1869–1953), professor of gynaecology at Johns Hopkins University, Baltimore, originally described this sign as an indication of a ruptured ectopic pregnancy.
aGeorge Grey-Turner (1877–1951), surgeon, Royal Victoria Infirmary, Newcastle-on-Tyne, England.
aBernhard Riedel (1846–1916), German surgeon, described this in 1888.
aJohn Murphy (1857–1916), American surgeon, professor of surgery at Rush Medical College, Chicago, described this in 1912.
aLudwig Courvoisier (1843–1918), professor of surgery, Switzerland, described this principle in 1890. He was a keen natural historian and wrote 21 papers on entomology.
aFrom the Latin accubitus—reclining at table. Remember how the Romans lay on their sides to eat their dinners.
aCarnett’s test: described by JB Carnett in 1926.
aAH Curtis described hepatic adhesions associated with pelvic inflammatory disease in 1930, while T Fitz-Hugh described right upper abdominal acute gonococcal peritonitis in 1934. However, this syndrome was actually first described by C Stajano in 1920.
aaJean Cruveilhier (1791–1874), professor of pathological anatomy, Paris, who had been Dupuytren’s registrar, and Paul von Baumgarten (1848–1928), German pathologist.
bbFranz Hesselbach (1759–1816), professor of surgery, Würzburg, described this triangle bounded by the inguinal ligament, the inferior epigastric artery and the rectus abdominis.
cccc George Blumer (1858–1940), professor of medicine at Yale, in 1909 described cancer in the pouch of Douglas forming a shelf-like structure.
dddd It can be useful to perform the rectal examination with the patient supine and the head of the bed elevated; this allows the intra-abdominal contents to descend and a bimanual examination (using the opposite hand to compress the lower abdomen) is possible.
eeee The middle one of three transverse folds of mucous membrane in the rectum, described in 1830 by John Houston (1802–45), Irish surgeon.