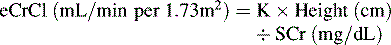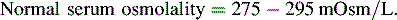Care of the child during dialysis, hemoperfusion, and hemofiltration
Dialysis in Children
Dialysis is indicated for the child with AKI when aggressive medical management has failed to control hypervolemia, hypertension, bleeding, hyperkalemia, hyperuricemia, or acidosis. Dialysis also is indicated when uremia produces cardiovascular or neurologic deterioration or when elimination of toxins or poisons is required (Box 13-9).
Box 13-9 Indications for Dialysis in Children
Hypervolemia with congestive heart failure, uncontrolled hypertension, or hypertensive encephalopathy
Deterioration in neurologic status
Bleeding that is unresponsive to blood component therapy
Biochemical alterations (these criteria are not absolute):
• Serum potassium concentration above 6.5-7 mEq/L, despite maximal medical therapy and administration of sodium polystyrene sulfonate exchange resin
• Persistent metabolic acidosis, particularly in the presence of hypervolemia or hyperkalemia
• Serum BUN greater than 125-150 mg/dL
Acute poisonings or drug toxicity, including ingestion of the following substances:
Both hemodialysis and peritoneal dialysis use osmotic and concentration gradients between the child's blood and the dialysate to reduce the child's intravascular volume and to alter intravascular electrolyte concentrations. The content of the dialysate, or dialysis solution, will determine the specific changes made in the child's volume and electrolyte status.
When peritoneal dialysis is used, a peritoneal catheter is inserted and the dialysate is infused into the peritoneal cavity, so that it comes into contact with the peritoneal membrane. The peritoneal membrane acts as the semipermeable membrane, allowing diffusion of electrolytes and water between the peritoneal capillaries and the dialysate.
In children, peritoneal dialysis removes water and electrolytes from the blood by virtue of the osmotic and electrolyte concentration gradients that exist between the dialysate and the patient's blood (across the peritoneal membrane). Manipulation of the osmolality and electrolyte concentration of the dialysate determines the quantity and speed of fluid movement. Peritoneal dialysis enables fluid removal at a rate slower than hemodialysis, so can avoid the complications created by rapid intravascular and extravascular fluid and electrolyte shifts.
Hemodialysis uses an artificial semipermeable membrane and dialysate located outside the patient's body (i.e., it is extracorporeal). Vascular access for acute hemodialysis can be achieved with single- or double-lumen catheters inserted in the vena cava or upper right arm, using the femoral, internal jugular, or subclavian approach. These catheters can be maintained in place for long periods of time. Chronic hemodialysis in the older child may require placement of an arteriovenous fistula or graft.
If good circulatory access is achieved, hemodialysis is much more efficient than peritoneal dialysis in the child and adolescent. Hemodialysis with hemoperfusion is especially effective for the removal of poisons after drug overdose. However, good circulatory access can be difficult to obtain in infants or young children. In addition, the volume of the hemodialysis circuit cannot exceed 10% of the child's circulating blood volume unless the circuit is primed with blood before each use. As a result, hemodialysis during infancy should be performed only at institutions experienced in the procedure.
Two additional techniques, hemoperfusion and hemofiltration, can be used to adjust serum water and electrolyte concentrations. These techniques will be discussed in two separate sections, Hemoperfusion and Hemofiltration, below.
Acute Peritoneal Dialysis
When the decision to begin peritoneal dialysis (PD) is made, informed consent is obtained from the parents by the physician. The results of serum chemistries obtained within the previous 8 hours should be available at the bedside, and the child's weight is obtained before dialysis. If the child is small, the predialysis weight should be obtained after the peritoneal catheter is in place and dressings are applied.
There are few contraindications to PD in children. Patient age and size do not constitute any contraindication, because PD has been performed in small neonates. However, neonates with omphalocele, diaphragmatic hernia, or gastroschisis cannot be treated with PD.
Recent abdominal surgery is not a contraindication to PD, provided the patient has no draining abdominal wounds. However, smaller infusion volumes will be required in these patients. Minor abdominal adhesions will not preclude successful PD, although extensive adhesions may prevent successful instillation and removal of the dialysate.
The presence of a vesicostomy or other urinary diversion, polycystic kidneys, colostomy, gastrostomy, or prune-belly syndrome does not preclude the use of PD. Acute renal failure associated with renal transplant rejection can be treated with peritoneal dialysis, provided the allograft has been placed in the extraperitoneal space.
Bedside (Percutaneous) Placement of Peritoneal Catheter
If peritoneal dialysis is expected to be required for a short time (<72 hours), the catheter can be placed percutaneously at the bedside (i.e., not surgically). When bedside placement is planned, all needed equipment is assembled and checked before the procedure (see Box 13-10). Because the incidence of catheter-related infection increases when percutaneously placed catheters remain in place beyond 72 hours, surgical catheter placement (i.e., in the operating suite) is indicated if peritoneal dialysis is expected to be required beyond 3 days.
Box 13-10 Equipment Typically Needed for Bedside Placement of Peritoneal Dialysis (PD) Catheter
1. Two pediatric PD catheters with trocars, Y tubing, and a PD tray, dialysate fluid with warmer.
2. Acute PD is accomplished with four or six 2-L bags of dialysate containing either 1.5% glucose or 4.25% glucose. The dialysate must be warmed to body temperature before infusion, to prevent hypothermia. Dialysate bags must be checked for punctures or leaks before use.
3. A patent urinary catheter must be in place. If the child's catheter has been in place for several days, it may be wise to replace it to ensure patency; this ensures the emptying of the bladder and reduces the risk of bladder perforation when the PD catheter is placed.
4. Laboratory results obtained within the previous 8 hours should include hemoglobin, hematocrit, BUN, electrolytes, glucose, phosphorus, uric acid (if appropriate, as in uric acid nephropathy associated with chemotherapy), a PT, aPTT, and platelet count, as well as a type and cross match for a unit of blood (or packed cells).
5. One thousand units of sodium heparin are added to each 2-L bag of dialysate (500 U/L) unless frank abdominal bleeding is present. Heparin crosses the peritoneal membrane poorly, and its presence in the dialysate will reduce fibrin formation and assist in maintaining peritoneal catheter patency.
6. Two 16-gauge polyethylene over-the-needle catheters and two short sets of extension tubing. Two sets of tubing are used to infuse a volume of solution into the peritoneum to distend the peritoneal space and reduce the risk of bowel perforation when the trocar is inserted.
7. Two small (1-mL) syringes and lidocaine (Xylocaine) without epinephrine.
9. Sterile gloves, masks, and gowns.
10. Sterile dressings, tape, surgical skin cleaner, and povidone-iodine solution.
11. Tubes for culture of the peritoneal fluid. The first outflow is cultured, and then cultures of fluid are obtained from every sixth pass
aPTT, Activated partial thromboplastin time; PD, periotoneal dialysis; PT, prothrombin time.
When the decision for dialysis is first considered, the preparation of the child must begin. The discussion should be appropriate for the child's age and comprehension, and it should involve the physician, family, and nurse. The nurse must attempt to understand the aspects of the procedure that are frightening or confusing to the child and address those points directly. It is important that the parents understand the procedure and support the child throughout the dialysis.
The parents and the nurse must be comfortable with the facts before attempting to discuss them with the child. Often a sedative will be prescribed for the child to reduce pain and anxiety during the procedure.
A surgical preparation of the abdomen is performed, using a surgical skin cleaner, followed by an appropriate scrub. Sterile procedure will be used throughout catheter placement, which requires that the surgeon and any assistants wear gown, gloves, and masks. Local anesthetic is infiltrated along the lower quadrant of the abdominal wall. Before perforation of the abdominal wall, the child's vital signs should be documented for comparison during the procedure.
The 16-gauge polyethylene over-the-needle catheter is joined to the primed dialysate tubing, and the needle and catheter are inserted into the abdomen at the midline. Using the symphysis pubis and the umbilicus as distance markers, the catheter will be inserted one third of the total distance down from the umbilicus. The catheter is joined to the tubing of a warmed and primed bag of dialysate and is advanced into the peritoneal cavity until the drip chamber of the inflow line demonstrates free flow of solution into the abdomen. The inflow should be interrupted temporarily while the inflow line is disconnected and the steel needle is removed from the catheter. The catheter is then advanced into the abdomen (to the hub), the tubing is connected to the catheter, and a volume of 30 mL/kg is infused into the abdomen to distend it. Occasionally, a volume of up to 50 mL/kg is required to elevate the anterior abdominal wall sufficiently. During fluid infusion, the child's ventilation and perfusion must be monitored closely; infusion should be interrupted if cardiorespiratory distress develops.
When the peritoneal space is judged to be full and the abdominal wall is elevated and tense, the catheter is withdrawn and a small stab wound is made at the site of catheter insertion (without entering the peritoneum). The catheter and trocar are then inserted using steady pressure aimed at the right or left lower quadrant. Once the abdominal wall is penetrated, the catheter will be advanced as the trocar is withdrawn. Easy inflow and outflow of fluid should occur through the catheter.101
The catheter will be trimmed to leave only 4 to 6 cm outside the abdominal wall. The catheter should be secured with a silk purse-string suture and water-resistant tape. The outflow tubing is then clamped, and the first warmed exchange dialysate of 20 to 30 mL/kg is infused.
The child's blood pressure, temperature, respiratory rate, and heart rate are assessed and documented every 15 minutes for 1 hour, then every hour once the child's condition is stable. Changes in the child's level of consciousness and activity level should be noted and reported to a physician, because these can indicate serious fluid or electrolyte disturbances.
The dialysate remains in the peritoneal space for 15 to 90 minutes (this may vary), and then the outflow connection is opened and the fluid is drained slowly. All subsequent weights are obtained at the end of the outflow cycle when the peritoneal cavity is empty.
If the dialysate fails to drain easily, the catheter is probably obstructed by omentum. If the problem continues, surgical replacement of the catheter may be required. If the dialysate returns cloudy or consistently bloody, or if diarrhea or polyuria are noted, a physician should be notified immediately; these signs may indicate perforation of the bowel or bladder. The catheter must be removed and replaced, and the patient should be observed closely for evidence of further symptoms.101
Surgical Placement of Dialysis Catheter
When the dialysis catheter is placed surgically, a cuffed catheter is used and is inserted at the level of the umbilicus through a small incision in the rectus muscle. The catheter is inserted to the level of the Dacron cuff and is held in place with a peritoneal purse-string suture. Once the catheter is inserted, a small volume of dialysate or normal saline is infused in the catheter to ensure that the site does not leak and that fluid flows easily into and out of the catheter. Finally, the catheter is tunneled under the skin and exits through the skin at a site separate from the catheter entrance into the peritoneal cavity.3
Dialysate Solution
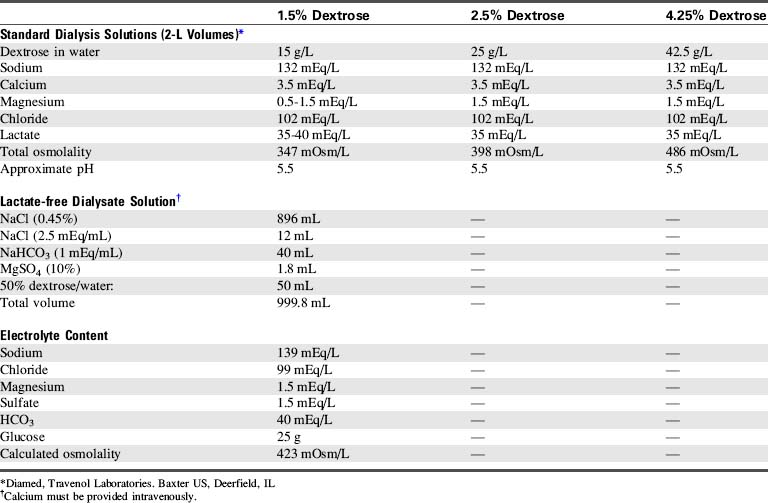
Commercially available dialysis solutions contain electrolytes in concentrations similar to that of normal plasma, except that potassium is absent and the concentration of glucose and osmolality vary. The absence of potassium ion creates a concentration gradient for potassium between the dialysate and the capillary vessels of the peritoneum so that potassium moves from the vascular space into the dialysate.
The dialysate is selected by glucose concentration and osmolality; the higher the glucose and osmolality of the dialysate, the greater will be the fluid shift from the vascular space to the dialysate (i.e., more fluid is withdrawn from the child). Commercially available solutions contain 1.5% (15 g glucose/L, osmolality of 347 mOsm/L), 2.5% (25 g glucose/L, osmolality of 398 mOsm/L), and 4.25% glucose (42.5 g glucose/L, osmolality of 486 mOsm/L). Because glucose can move from the dialysate into the vascular space according to a concentration gradient, the child's serum glucose must be monitored closely. In the patient without diabetes, endogenous insulin secretion should prevent hyperglycemia, but insulin should be added to the dialysate of diabetic patients (see “Fluid or Electrolyte Imbalance”).
Commercially available dialysates vary in electrolyte concentration (Table 13-6). Some critically ill infants are unable to tolerate lactate in the dialysate, because the lactate may worsen acidosis. Dialysate can be reformulated in the hospital pharmacy, using a sterile hood, to contain bicarbonate instead of lactate (see Table 13-6). When the dialysate contains bicarbonate, calcium must be administered intravenously and cannot be added to the dialysate; it will precipitate with the bicarbonate.
If the serum potassium concentration begins to fall after several dialysis cycles (usually 4-6 cycles are required), a small amount of potassium (typically to a maximum of 4 mEq/L) may be added to the dialysate with a physician or other on-call provider order.
Dialysis Exchange
The initial dialysate volume (inflow or exchange volume) is determined by the method of catheter placement. If the catheter is placed percutaneously at the bedside, initial exchange volumes of 20 to 30 mL/kg are used. If the catheter is placed surgically, smaller initial volumes (15-20 mL/kg) are used to reduce the likelihood of leak around the catheter.3 Small exchange volumes also may be necessary if respiratory distress is present and the child is breathing spontaneously.101 Heparin (500 units/L) usually is added to the dialysate for the first 24 hours of dialysis.
Exchange volumes are increased gradually as tolerated to 35 to 50 mL/kg/exchange. These volumes are ideal because they enable the correction of acidosis, electrolyte imbalance, and uremia.
Typically, 2.5% glucose dialysate is utilized for initial exchanges in the uremic child with acidosis and hyperkalemia. If fluid removal is not required, 1.5% glucose dialysate may be utilized. The 4.25% glucose dialysate is used for those patients with hypervolemia requiring fluid removal. The prolonged use of 4.25% glucose dialysate can be associated with hyperglycemia, hyponatremia, and hypovolemia.
For each exchange, dialysate is instilled over 5 minutes with a dwell time of 15 to 90 minutes (this may vary). The drain time varies with the size of the patient and the exchange volume; usually 5 to 10 minutes is sufficient, although drain times of 20 minutes may be required. Peritoneal dialysis is maximally effective during the first 15 to 90 minutes of dwell time. Therefore if maximum fluid removal and correction of hyperkalemia, acidosis, and uremia are required, frequent exchanges (often every 30 minutes) are performed.
Automated peritoneal dialysis cyclers are now available that provide exchange volumes as small as 50 to 100 mL. These cyclers incorporate a heater to warm the dialysate, and they automatically monitor the drain volume or outflow and can calculate the volume of fluid removed from the patient. Audible alarms indicate volume or infusion problems. Because the equipment manipulates the dialysate it can substantially reduce the nursing time needed for each exchange.
Manual peritoneal dialysis requires the use of buretrols or other graduated cylinders to monitor the precise volume of fluid infused and drained. When the exchange volume is small, buretrols can be used to measure both inflow and outflow (drain) volume. Graduated urine collection systems also can be utilized to measure outflow volume. Finally, the serial dialysate drainage bags used for continuous ambulatory peritoneal dialysis also can be utilized to measure the drain volume; clamps are used to direct the draining fluid into a separate bag following each exchange.
The large (2-L) dialysate bags should never be hung so they infuse directly into the child; if a clamp loosens or is inadvertently left open, the entire 2 L volume could be infused into the child's abdomen, causing respiratory distress and possible cardiovascular collapse. The dialysate fluid always should be warmed before use in infants and children to prevent heat loss and reduce discomfort. Room temperature dialysate may be used in adolescents unless it produces discomfort.
For dialysis in infants, exchange volumes will be small and tubing dead space should be minimized. Adult peritoneal dialysis Y tubing sets contain too much dead space for use in infants, so intravenous tubing (containing a Y or a stopcock) can be utilized to direct dialysate flow. Infant peritoneal dialysis circuits are commercially available.
Because dialysis removes fluid from the vascular space, the nurse should assess the child's volume status frequently. Signs of hypovolemia include tachycardia and signs of poor systemic perfusion (such as decreased intensity of pulses, pale mucous membranes, and cool extremities with weak pulses and sluggish capillary refill). The CVP will be low unless heart failure is present. The development of hypotension indicates critical hypovolemia and hypotensive shock.
If edema is present, peritoneal dialysis will not immediately abolish all fluid excess. However, as fluid is removed from the vascular compartment the intravascular proteins and sodium ions (osmolality) will draw water out of the edematous tissues into the vascular space, (where it can then be removed during dialysis).
Calculation of Fluid Balance
During peritoneal dialysis, two patient records of total fluid intake and output must be strictly maintained. One record documents the dialysate infused and the dialysate recovered (drain volume) at the end of each cycle (Fig. 13-9).

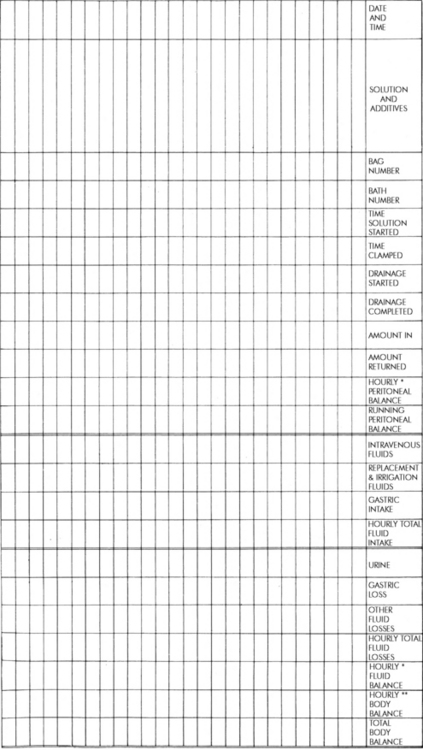

Fig. 13-9 Example of peritoneal dialysis flow chart template.
(Courtesy Children's Memorial Hospital, Chicago, IL.)
The amount of fluid recovered always should equal or exceed the amount infused; this produces a negative fluid balance. If less dialysate is recovered than was infused, the nurse should check for signs of catheter or tubing obstruction (see section, Catheter Dysfunction and Obstruction). If additional dialysate cannot be recovered, the difference between the amount infused and the amount recovered is recorded as a positive fluid balance, and a physician should be notified.
During the initial cycles of peritoneal dialysis, a larger volume may be recovered than was infused, indicating removal of intravascular fluid. When this occurs, the amount of fluid recovered in excess of the amount infused should be recorded as a negative number, because this represents fluid loss for the child. If significant fluid loss continues, a physician or on-call provider should be notified; it may be necessary to reduce the osmolality of the dialysate to prevent excessive fluid loss and dehydration.
The time and the duration of each infusion, dwell time, and drainage cycle should be recorded. Because maximum solute transfer occurs during the first 30 to 90 minutes the dialysate is in the peritoneal cavity, the dwell time is rarely longer than this. The temperature of the dialysate also should be measured and recorded; this temperature should be as close as possible to 37° C to improve the efficiency of dialysis and to minimize the child's heat loss and discomfort.
The second record of the child's fluid balance includes a total of all sources of fluid intake and output. The net dialysis balance is recorded as part of this total. It is extremely important that this record be carefully maintained, because it will aid in the evaluation of the child's progress and need for modifications in dialysis. When dialysis is begun, drug doses and administration schedules should be adjusted, because many drugs are removed by dialysis.
Potential Complications
Peritonitis
As many as one third of children who receive peritoneal dialysis develop peritonitis. The risk is directly proportional to the duration of the dialysis and is inversely proportional to the child's age. Critically ill patients are especially susceptible to the development of peritonitis. Clinical signs and symptoms of peritonitis during peritoneal dialysis include cloudy return, abdominal pain and tenderness, and leukocytosis. Fever is usually present in children, but young infants may become hypothermic. Paralytic ileus and constipation also may develop.
Because the risk of peritonitis is significant in critically ill patients, a sample of outflow often is obtained on a daily basis and should be obtained whenever peritonitis is suspected. The sample is centrifuged and a Gram stain, cell count, and culture and sensitivity tests are performed. If clinical evidence of peritonitis is present, antibiotics usually are administered as soon as the cultures from the outflow sample and blood are obtained. Cefazolin and gentamicin may be administered into the peritoneal cavity. If the patient is clinically unstable, a single dose of IV vancomycin may be given.19
The risk of fungal and bacterial infections is reduced by scrupulous attention to sterile technique during the catheter insertion process and aseptic technique during exchanges. When the outflow collection bag is examined, the bag must not be raised above the level of the bed, because elevation will produce reflux of outflow back into the peritoneal cavity.
Catheter Dysfunction and Obstruction
When dialysate will not flow either into or out of the peritoneal cavity, an external kink or an internal plug is likely to be present in the tubing. If external causes of flow obstruction are eliminated and flow does not resume, a catheter plug is presumed to be present.
A physician (or other on-call provider) can gently irrigate the catheter with normal saline or urokinase, using aseptic technique.101 Every time a break is made in the dialysis tubing system, the patient's risk of peritonitis increases. Before the dialysis tubing is separated, the connection between the tubing and the catheter must be scrubbed for 5 minutes with a povidone-iodine solution, and clamps are placed on both the catheter and the tubing. The separated ends of the tubing should be wrapped in dry sterile 4 × 4 gauze during separation. Aspiration should not be performed in an attempt to dislodge any plugs, because this may result in catheter occlusion with omentum.
A flat plate radiograph (x-ray film) of the abdomen can be made to confirm the presence of a catheter plug and rule out catheter migration. A solution consisting of three parts 1.5% dialysate and one part Renografin M60 can be infused by gravity or administered by syringe into the catheter at the time the abdominal films are made. This solution will opacify the lumen of the catheter and enable identification and location of plugs.
After catheter manipulation, the dialysis tubing can be reconnected if it is not contaminated during the manipulation. As noted above, the ends should be wrapped in sterile gauze during the manipulation. If the tubing is to be reused, ensure that no fluid leaks out of the system. If a solid column of fluid is maintained in the tubing, air does not enter the tubing and fluid does not drain out of the tubing during catheter manipulation.
With most forms of catheter obstruction, dialysate will flow freely into the peritoneal cavity, but fluid will not drain freely from the peritoneal cavity. Often the catheter floats above the level of the fluid in the peritoneal cavity or becomes wrapped in the omentum. Catheter obstruction also may be caused by constipation, which locks the catheter into a position that restricts drainage. Once the bowel is evacuated, dialysis can proceed. If the child is repositioned or turned from side to side, drainage often can be restored.
Sluggish outflow also can be caused by loops in the dialysis tubing that hang off the edge of the bed. The collection bag and tubing should be repositioned so that the tubing falls straight down from the bed to the collection bag. Any extra tubing should be coiled on the bed to facilitate drainage.
Pain
Almost all patients with new peritoneal catheters complain of pain during the initial dialysis infusions and outflows. The pain experienced upon inflow can be relieved by slowing the rate of infusion or by infusing smaller dialysate volumes.
Pain also can be caused by encasement of the catheter in a false passage; this causes the dialysate to fill only a small area of the peritoneal cavity instead of spreading throughout the peritoneal space. That small area can distend and become painful. If the catheter has been immobilized so that the dialysate flow is directed at the same point in the peritoneal cavity, it usually causes pain in the lateral or posterior peritoneal wall. It may be possible to float the catheter to another position when the abdomen is filled with dialysate. Occasionally, insertion of a new catheter may be necessary. Painful inflow also may be related to extremes of dialysate temperature.
Pain is usually present during dialysate inflow; patients rarely complain of pain only during outflow. Pain at the end of outflow will occur when the abdomen is emptied completely, and it can be abolished by stopping the outflow when a small volume of solution remains in the peritoneal cavity. The presence of this residual solution also diminishes the likelihood of catheter obstruction by omentum. Limiting the outflow time to 5-10 min also should alleviate this problem.
Miscellaneous Complications
Bloody return is a common observation during the initial 24 to 48 hours following catheter implantation. This condition is usually self limiting, and heparin still should be added to the dialysate to prevent the formation of fibrin plugs in the catheter. Heparin will not cross the peritoneal membrane, and it will not affect the patient's coagulation. If the amount of blood in the outflow seems excessive, serial hematocrits can be obtained from the outflow to quantify the amount of blood present. Transfusion may be required if excessive blood loss occurs.
Leaking around the catheter is common when the catheter is placed under urgent conditions at the bedside; it seldom occurs in surgically placed or chronic catheters. Whenever a leak occurs, the nurse should check for overfilling of the abdomen by feeling the tenseness of the abdomen at the end of inflow. The abdomen will feel full, but should not feel rigid. Reassess the catheter insertion wound and tubing to determine whether the catheter is migrating into or out of the peritoneal cavity.
If a leak occurs, weigh and then pack (using aseptic technique) sterile dressings around the catheter and change and weigh them when they become soaked, to measure the volume of the leak. Notify a physician or on-call provider of the leak volume; a smaller volume of dialysate inflow may be ordered.
Fluid leak into the abdominal subcutaneous tissues occasionally develops. The fluid is likely to accumulate in dependent perineal areas. The volume of the subcutaneous leak is usually small and is commonly reabsorbed. If a large volume leaks into closed tissue areas such as the penis or scrotum, it may be necessary to replace the catheter.
Pulmonary Complications
Because peritoneal dialysis results in abdominal fullness, it may compromise diaphragm excursion, resulting in shallow breathing and atelectasis, particularly if the patient is breathing spontaneously. Atelectasis especially in the lower lobes is more likely to develop when the child remains in the supine position.
Assess the child's breath sounds frequently, and constantly evaluate the effectiveness of ventilation. Provide chest physical therapy if areas of atelectasis are noted. If the child is alert and cooperative, encourage the child to cough and take deep breaths or perform inspiratory exercises (e.g., incentive spirometry) to prevent atelectasis. The infant may require other exercises to encourage deep breathing (see Chapter 9). The head of the child's bed should be elevated to facilitate maximal diaphragm excursion and chest expansion.
Fluid or Electrolyte Imbalance
Throughout the dialysis period, the child's electrolyte and acid-base balance should be monitored closely, and electrolyte or acid-base imbalances should be discussed immediately with an on-call provider.
Hypertonic dehydration and hemoconcentration can develop if peritoneal dialysis removes too much water too rapidly; this can result in hypernatremia and can exacerbate hyperkalemia. If dehydration is suspected the nurse should assess the child's level of hydration, heart rate, systemic perfusion, and blood pressure.
If the serum sodium concentration is elevated and the child is dehydrated or hypovolemic, free water may be administered orally, intravenously (with 5% dextrose), or intraperitoneally (using a less concentrated dialysate). The osmolality of the dialysate solution should be reduced for subsequent peritoneal dialysis.
Hypokalemia can develop if a hypokalemic dialysate is used after the serum potassium concentration normalizes. If hypokalemia develops, small doses of potassium can be added to the dialysate, or small doses of potassium chloride can be administered intravenously (0.5-1.0 mEq/kg over several hours). Hypokalemia also can develop if a 4.25% glucose dialysate is used for prolonged periods; the treatment of choice is to change the dialysate solution if the child's condition permits.
Hypoproteinemia can develop if peritoneal dialysis is required for several days, because 0.2 to 8.0 g of protein is lost per liter of outflow. Higher amounts of protein loss occur during episodes of peritonitis. As a result, the child's total protein and albumin should be monitored, and the nurse should assess the child for signs of peripheral edema. If hypoproteinemia develops, administration of amino acids may be needed.
Hyperglycemia can develop if concentrated glucose dialysate is required to eliminate large amounts of free water. The serum glucose concentration (and point of care evaluation of glucose, if available) should be monitored closely, and hyperglycemia or hypoglycemia should be reported to an on-call provider. It may be necessary to reduce the glucose concentration of the dialysate. If the patient has diabetes mellitus, exogenous insulin should be added to the dialysate.
Catheter Removal
The percutaneously placed PD catheter is removed using sterile technique. While one provider withdraws the catheter, a second provider maintains tension on the purse-string stitch that was placed during the catheter insertion. The suture is drawn tight as the catheter is withdrawn. The catheter tip can be sent to the laboratory for culture (per policy). Sterile dressings are placed over the catheterization site. If the dialysis catheter is placed surgically, it must be removed surgically.
Extended Peritoneal Dialysis: Continuous Ambulatory Peritoneal Dialysis and Continuous Cycling Peritoneal Dialysis
Some patients with AKI will require PD for a long period of time (i.e., longer than 5-10 days). In these patients a permanent cuffed peritoneal catheter can be placed surgically for ambulatory peritoneal dialysis.
Method
Continuous Ambulatory Peritoneal Dialysis
Continuous ambulatory peritoneal dialysis (CAPD) is a form of continuous dialysis that does not require bed rest or hospitalization. CAPD uses a surgically placed, cuffed PD catheter and disposable plastic bags of dialysate. Approximately four to five exchanges are performed daily, and each exchange volume totals approximately 30 to 50 mL/kg (or 0.5-2 L total). The exchange time is approximately 4 to 6 hours, which is much longer than the exchange time during acute peritoneal dialysis.101 However, during the exchanges the empty dialysate bag is clamped and strapped to the patient's abdomen, and the patient is free to be relatively active. At the end of each exchange period, the empty dialysate bag is placed at a level lower than the patient's abdomen, the drainage tubing is unclamped, and outflow from the patient's abdomen drains into the bag. The bag of outflow is then discarded, and a new disposable dialysate bag is obtained for use in the next exchange.
CAPD will not rapidly correct hypervolemia, acidosis, or hyperkalemia; therefore it is not the dialysis method of choice for acutely ill children. However, CAPD allows excellent regulation of fluid and serum electrolyte concentrations when it is used on a daily basis for the child in stable condition with chronic kidney disease. Children who receive CAPD generally require less frequent blood transfusions than children who receive chronic hemodialysis, and serum urea nitrogen and phosphorus concentrations may be better controlled than with hemodialysis. However, renal osteodystrophy and hyperphosphatemia do occur.36
Children receiving CAPD have few dietary restrictions, because their relatively continuous dialysis can remove excess fluid and allow constant regulation of electrolyte and acid-base balance. As a result, children receiving CAPD may be better nourished than those who require intermittent forms of dialysis. Control of hypertension is also excellent when children with renal failure receive CAPD.
Continuous Cycling Peritoneal Dialysis
Continuous peritoneal dialysis may be made less labor intensive with the addition of mechanical cycling to the dialysis process. Continuous cycling peritoneal dialysis (CCPD) provides mechanical (automatic) delivery of a prescribed volume of dialysate at prescribed intervals, with a set indwell time. Drainage of outflow is initiated mechanically, and audible alarms sound if flow problems occur.72 Current cycling machines are capable of infusing dialysate volumes of 50 to 100 mL.
CCPD is performed most commonly while the child sleeps. The dialysis cycling machine should not require any attention during the night unless an alarm sounds. A small dialysate volume is allowed to remain in the peritoneal cavity during the day, providing continuous dialysis. The child is able to resume school and other normal childhood activities. All exchange bags are hung at the same time when CCPD is begun at night; this can reduce the risk of contamination and peritonitis.72
Complications
The most frequent complications associated with CAPD and CCPD are mechanical problems and infection. The mechanical problems are related to cuff erosion and fluid leaks, and the infection problems are related to peritonitis.
The cuffed peritoneal catheter can erode the abdominal wall; this can cause a fluid leak and require catheter replacement. Hernias can develop from subcutaneous fluid leaks around the dialysis incision, and these hernias often require surgical repair.
The incidence of peritonitis among children receiving CAPD and CCPD varies widely in clinical reports, but averages one infection every 14.7 patient months.36 Many patients also develop local infections around the catheter site.
When selecting patients for CAPD, it is important that the child and the parents are reliable and able to follow the established protocol. Children or families must be taught the dialysis technique, and they should be instructed to contact the CAPD nurse whenever the patient experiences abdominal pain, inflow or outflow occlusion, inflammation of the catheter site, a feeling of weakness or dizziness when standing, hypotension, cloudy outflow, catheter disconnection or contamination, fever, excessive weight gain, edema, or other illness.
Because the dialysate dwells in the peritoneal cavity for a long time and the risk of peritonitis is relatively high in children receiving CAPD and CCPD, hospital protocols often try to minimize the number of personnel providing any in-hospital CAPD or CCPD that the child requires. This action minimizes the child's exposure to people and contaminants and may reduce the risk of peritonitis.
Hemodialysis
Method
Hemodialysis is one of the most efficient artificial methods of removing nitrogenous wastes from the body and of restoring fluid, electrolyte, and acid-base balance. However, pediatric hemodialysis requires the assembly of skilled personnel capable of establishing and maintaining vascular access, recognizing and responding to potential complications of dialysis, and supporting cardiorespiratory function in extremely unstable patients. If urgent dialysis is required and experienced personnel are not available, PD may be provided until the child's condition can be stabilized and the child transported to an appropriate facility.
Indications for pediatric hemodialysis are GFR <15 mL/min per 1.73 m2 or intractable complications of AKI, even at a higher GFR. GFR is roughly equivalent to the creatinine clearance estimated by the Schwartz formula (see Box 13-5).
Complications of hemodialysis include but are not limited to hypervolemia, hyperkalemia, metabolic acidosis, hyperphosphatemia, hypocalcemia, hypercalcemia, and neurologic dysfunction.52
Hemodialysis requires access in an artery, arterialized vessel, or large vein. If chronic hemodialysis is planned, an arteriovenous fistula may be created or a graft may be placed in a large artery. In the critical care unit, vascular access is often achieved with a single- or double-lumen catheter (at least size 6 French) placed through the subclavian vein into the right atrium, or from the femoral vein into the inferior vena cava.15
During hemodialysis, blood is withdrawn from the body and pumped at high flow rates to a blood compartment that makes contact with a semipermeable membrane. This blood compartment and semipermeable membrane are immersed in dialysate, which is pumped at rates that exceed blood flow rate by 50%. The dialysate flows in the direction opposite the blood flow.37
The dialysate contains a fairly standard concentration of electrolytes, but the potassium concentration usually is determined individually (based on the amount of potassium to be removed). Nitrogenous wastes pass from the blood into the dialysate as the result of a concentration gradient. Free water will move from the blood into the dialysate if the osmolality of the dialysate is greater than the serum osmolality. Free water movement is also enhanced by a positive transmembrane hydrostatic pressure from the blood compartment to the dialysate compartment. This positive pressure can be generated by the blood pump and by manipulation of resistance to blood outflow, although newer dialysis machines automatically generate the transmembrane hydrostatic pressure required to provide the volume of ultrafiltrate prescribed.37
The amount of fluid and solute removal from the blood is determined by the flow rates of the blood and dialysate, the surface area and permeability of the membrane, the concentration and osmolality gradients between the dialysate and blood, and the transmembrane pressure gradient. Electrolytes move across the semipermeable membrane as the result of concentration gradients. If the concentration of an electrolyte (e.g., potassium) or other small molecule (e.g., urea) is lower in the dialysate than in the blood, that electrolyte or molecule will move out of the blood and into the dialysate. If the concentration of an electrolyte or other molecule (e.g., glucose) is higher in the dialysate than in the blood, that electrolyte or molecule will move into the blood. Other substances can be removed from the blood as the result of ultrafiltration and solvent drag (the passive movement of solutes as the result of movement of large amounts of water).
Because hemodialysis requires that blood be drawn from and returned to the body, pumps must be present in the dialysis circuit. In addition, the filter and the tubing (the dialysis circuit) must be primed with fluid or blood before the dialysis begins. Because the circulating blood volume of the infant or child is low, extracorporeal movement of a large quantity of blood (e.g., to prime the dialysis circuit) is likely to produce hypovolemia. As a rule, the filling volume of the dialysis circuit should be no greater than the equivalent of 10% of the child's circulating blood volume. If the circuit requires a larger filling volume, the circuit should be primed with colloid (5% albumin or packed RBCs diluted with albumin to a hematocrit of 35%).37 Few centers have experience in the hemodialysis of infants, and it should not be attempted by inexperienced personnel. In adolescents, the dialysis circuit can be primed with normal saline or 5% albumin.
The dialysate contains glucose, sodium, calcium, and potassium, in concentrations that are specified by the physician. The dialysate usually contains little potassium (0-4 mEq/L) and no urea, so that high concentration gradients between the dialysate and the blood will hasten removal of these solutes from the blood. The presence of glucose in the dialysate at levels of 200 to 250 mg/dL creates a high osmolality in the dialysate, favoring the movement of water from the blood to the dialysate, provided the patient's serum glucose concentration and osmolality are lower than the glucose and osmolality of the dialysate.
The glucose concentration in patients with diabetes mellitus is often higher than 200 to 250 mg/dL. As a result, the osmotic forces will favor free water movement from the dialysate into the patient's blood, exacerbating hypervolemia. This inappropriate fluid movement can be prevented by increasing the pressure gradient across the dialysis filter (creating higher pressure within the blood compartment or generating negative pressure in the dialysate compartment) or by adding albumin to the dialysate.
The high glucose concentration in the dialysate produces a serum glucose of approximately 200 mg/dL during dialysis. This mild hyperglycemia is usually well tolerated in the patients without diabetes mellitus; however, patients with diabetes will require adjustment of insulin dose (particularly long-acting insulin) in anticipation of this period of relatively low serum glucose.
The blood and the dialysate are typically pumped through the circuit in opposite directions. This pumping maximizes the concentration and osmotic gradients between the dialysate and the blood so that dialysis can be accomplished within a short period of time (exchange time of approximately 3-4 hours).
As noted previously, the creation of either positive pressure in the blood compartment or negative pressure in the dialysate compartment or both will increase the rate of fluid removal from the blood. Positive pressure is created in the blood compartment by increasing the resistance to flow on the venous side of the blood circuit. This increase in resistance usually is accomplished by placement of an adjustable clamp on the venous blood line, and the clamp is tightened until the desired pressure in the blood compartment is reached.
Negative pressure can be applied across the filter. This negative pressure draws free water and small particles from the blood, across the semipermeable membrane, and into the ultrafiltrate.
The dialysis nurse and the bedside nurse will be responsible for continuously evaluating the effect of fluid removal on the patient's systemic perfusion. If the patient's clinical condition deteriorates, some adjustment in the rate of fluid removal is often required.
When hemodialysis is initiated, a small amount of heparin is injected into the dialysis catheters and into the dialysis circuit to prevent clot formation in the filter and tubing. Heparin will then be administered at 30-minute to 1-hour intervals or by continuous infusion. The rate of infusion is adjusted based on the activated clotting time (ACT) or the Lee-White clotting time. Heparin dose and adjustment will be determined by dialysis unit policy and procedure. Citrate may be the anticoagulant of choice in hemodialysis therapy, and it is useful in the prevention of heparin-induced thrombocytopenia. Refer to protocols published on-line17 and in print69 for additional information.
Complications
Hemodialysis is efficient, but it is extremely expensive and can produce complications that do not develop during PD or hemofiltration. These complications are related largely to hypovolemia and resultant hypotension, fluid shifts (also known as dysequilibrium), hypervolemia, bleeding, anemia, infection, or malfunction of the vascular access site. Each of these is discussed in the following sections.
Hypotension and Hypovolemia
Hypotension can develop from removal of a large amount of intravascular water, resulting in hypovolemia, or from circulatory instability. The patients most at risk for developing hypotensive crises during dialysis are patients with vasomotor instability (including patients with paraplegia or quadriplegia), low cardiac output or myocardial dysfunction, those receiving vasodilators, or those with a history of hypotensive episodes during dialysis.
If the child develops hypotension during dialysis, the dialysis nurse will reduce any transmembrane pressure created across the filter, because this pressure gradient enhances fluid removal from the blood. In addition, the bedside nurse may be required to administer albumin or other volume expanders (per unit policy or physician or on-call provider order), place the patient in modified Trendelenburg position (head flat, feet elevated), or initiate cardiopulmonary resuscitation (as needed).
To avoid hypotension, any existing hypovolemia should be corrected before dialysis is begun. In addition, the patient's blood should be drawn slowly into the circuit so the patient does not experience an acute loss of intravascular volume. If excess intravascular fluid is to be removed during dialysis, venous positive pressure or negative pressure across the filter will be applied very slowly. The dialysis nurse and the bedside nurse are both responsible for monitoring the child's systemic arterial blood pressure and systemic perfusion. Deterioration in clinical status should be reported immediately to a physician or appropriate on-call provider.
Fluid Shifts and Dysequilibrium
If many osmotically active particles such as sodium or urea are removed rapidly from the patient's blood, the patient's serum osmolality will fall quickly. As a result, free water may shift from the intravascular and interstitial spaces to the intracellular compartment, producing cerebral edema. This edema following dialysis has been called dialysis dysequilibrium syndrome. The child may complain of severe headaches or may demonstrate nausea, vomiting, confusion, irritability, or seizures.
To reduce the risk of dysequilibrium, solute removal from the blood must be gradual; peritoneal dialysis may initially be performed to gradually reduce the BUN concentration. The efficiency of the hemodialysis can be reduced: the blood flow through the filter can be slowed, the direction of dialysate flow can be changed to the same direction as the blood, or the duration of the dialysis treatment can be shortened.
Intravenous mannitol can be administered slowly to increase serum osmolality and slow the removal of water by dialysis. Mannitol should be administered if evidence of dysequilibrium develops.64
Hypervolemia
If too much fluid is administered to the patient during dialysis or if an excessive volume of fluid and blood is transfused to the patient from the circuit at the end of dialysis, the child can develop hypervolemia. This condition can produce significant cardiovascular problems, particularly if the patient has preexisting cardiac disease.
The child can rapidly develop signs of congestive heart failure, including tachycardia, peripheral vasoconstriction, hepatomegaly, periorbital edema, elevated CVP, tachypnea, and increased respiratory effort. If severe hypervolemia is present, the child can develop pulmonary edema or hypertension.
Bleeding and Anemia
Because the child's blood must be anticoagulated during dialysis, bleeding can occur. The child can bleed from wounds or puncture sites or into the brain, pericardium, or abdomen. To reduce the risk of bleeding, regional heparinization may be performed. The heparin is injected into the arterial (or inflow) tubing that carries blood from the patient to the filter. To prevent large heparin infusion to the patient, protamine sulfate is administered into the tubing that is returning blood from the filter to the patient. Protamine sulfate neutralizes heparin, but can produce a coagulopathy or hypotension if it is administered separate from or in excess of heparin, or if the patient inadvertently receives a bolus of protamine.
Bleeding also can occur in patients with renal failure, because uremia is associated with depression of platelet function. If active bleeding is present in a patient with uremia, the most common treatment is the use of desmopressin or l-deamino-8-arginine vasopressin (DDAVP). Although the exact mechanism of action is unknown, desmopressin has been shown to increase release of factor VIII from storage sites, increasing the concentration of factor VIII and minimizing the effects of uremic dysfunctional von Willebrand factor. The dose of DDAVP to treat bleeding with renal failure is approximately 10-fold higher than the dose used for treatment of diabetes insipidus; it ranges from 0.3 to 0.4 mcg/kg intravenously and will improve platelet function within 1 hour after infusion. The effects disappear within 24 hours.51 DDAVP also can be administered before surgical procedures to reduce the risk of bleeding in patients with uremia.
Patients with renal failure often have anemia. Anemia can result from loss of blood within the dialysis system (through blood leaks, loose connections, clot formation, frequent blood sampling, or dilution of blood with dialysis tubing prime), from hemorrhage, or from the effects of uremia. Red cell lysis also can occur if blood is exposed to a dialysate of significantly higher osmolality. Levels of erythropoietin are low among uremic patients; therefore RBC production and survival are both reduced.
To prevent anemia, blood sampling should be minimized. Whenever blood is drawn, the amount should be recorded in the child's record of intake and output; blood replacement should be considered whenever the blood loss totals 5% to 7% of the child's circulating blood volume (circulating blood volume is approximately 70 to 80 mL/kg in infants and children and 70 mL/kg in adolescents). Because hemodialysis often is accomplished through the use of an arterial access catheter, laboratory sampling of blood can be performed during dialysis. This procedure reduces the number of venipunctures required and allows immediate replacement of the sample amount through the dialysis circuit.
Packed RBCs usually are administered to replace lost blood. Iron therapy will not be effective in the treatment of uremia-induced anemia and can contribute to the development of iron toxicity. Anemia can complicate the care of children with associated cardiovascular disease, because cardiac output must increase to maintain oxygen delivery.
Infection and Febrile Reactions
Patients with renal disease have increased risk of infection resulting from loss of immune proteins in nephrosis, multiple invasive catheters and cannulae, compromised nutritional status, frequent handling by hospital personnel, and frequent transfusions. The risk of infection can be minimized if all healthcare personnel practice strict hand-washing technique and strict asepsis, if the child receives adequate nutrition and if hepatitis and HIV screening is performed by the blood bank (for further discussion of hepatitis see Chapter 14).
The nurse should assess all of the patient's wounds and vascular access sites daily and report any areas of inflammation to an on-call provider. All wounds should be dressed according to unit policy or physician or on-call provider order.
Patients receiving hemodialysis may experience a sudden increase in temperature, known as a febrile reaction. This fever may result from an allergic reaction to the filter, from a reaction to blood administered during dialysis, or from systemic seeding from an infected shunt or access site. A preexisting fever suddenly may become manifest when the patient's serum urea, which can act as an antipyretic, is lowered.
When fever develops, the on-call provider can request that blood cultures be obtained from two different collection points; one set of cultures is typically collected from the dialysis tubing by the dialysis nurse after a 3-min povidone-iodine (Betadine) scrub. The second culture usually is obtained from a peripheral vein, although a second culture can be obtained from the dialysis circuit if at least 30 min elapse between samples. Cultures also can be collected from the ultrafiltrate.
If a transfusion reaction is suspected, specimens are collected from the transfusion bag and patient, and they are sent to the laboratory for hemolysis and incompatibility checks (see Chapter 15). If hemolysis is present, the child's serum potassium concentration should be monitored closely, because hyperkalemia can develop. A hematocrit also should be obtained, and a serum sample should be checked for evidence of hemolysis.
Hemodialysis Access
The establishment and maintenance of vascular access in small children is one of the most challenging aspects of pediatric hemodialysis. Dependable, double lumen, cuffed venous catheters are now available and they are the acute and long-term dialysis catheters of choice. If these catheters are placed surgically in the right atrium and infused appropriately with heparin, they can be maintained for a long time. When vascular access is needed, the injection port of the catheter can be used and no skin punctures are required. The care of these catheters is identical to the care of any long-term central venous catheter; meticulous care is required to ensure catheter longevity and minimize the risk of infection. Refer to hospital policies and procedures regarding central venous catheter care (also, Box 22-6).
Arterial access can be provided by a graft. Grafts consist of tubes made of Teflon or polytetrafluoroethylene. The tube is attached to an artery and then looped subcutaneously and connected to a parallel large vein.15 After surgical placement, time is allowed for the graft to become coated with the patient's endothelial cells (i.e., the graft endothelializes) before it is used for dialysis. Vascular access is achieved by piercing the graft with standard large-bore needles.
Arteriovenous fistulae can still be created in older children to provide vascular access for hemodialysis. The fistula is surgically created in the nondominant arm by connection of the radial artery to the cephalic vein.15 Soon after the fistula is created, the vein distends and is punctured easily to obtain vascular access. The risk of infection in the fistula is low because no prosthetic material is involved in its construction. However, any arteriovenous shunt will increase venous return to the heart and may precipitate or worsen congestive heart failure.
Continued Problems of Uremia
The patient requiring hemodialysis is still susceptible to complications of uremia. Although dialysis may provide temporary relief of some fluid and electrolyte or acid-base imbalances, anemia, hypertension, infection, osteodystrophy, endocrine imbalance, pruritus, anorexia, nausea, vomiting, fatigue, ulcers, and depression can persist (see section, Chronic Kidney Disease).
Throughout the care of the child with renal failure, the nurse should consult support personnel to assist in the psychosocial care of the child and family. Frequent multidisciplinary conferences and meetings with the social worker, dietician, financial counselor, physicians, and primary nurses will help the family to be aware of the support systems available to them and ensure that communication is optimal among team members and the family. As appropriate, the bedside nurse or case manager should begin to plan for the child's discharge to home or to another unit.
Whenever the critically ill child requires dialysis, the bedside nurse remains responsible for coordinating the care of the child and family. Although a dialysis nurse may be present and responsible for the dialysis procedure and circuit, the bedside nurse still must assess and document the child's fluid balance and systemic perfusion. The dialysis nurse and the bedside nurse should coordinate efforts. It is important to time the administration of medications, blood products, and fluids based on the timing and effectiveness of the dialysis. Both the dialysis nurse and the bedside nurse must provide the child and family with skilled support and compassion.
Hemoperfusion
Definition
Hemoperfusion is a treatment which exposes blood or plasma to an adsorbent material for the removal of toxins, solutes, or other materials. Adsorbent materials used can include charcoal, resin, protein A, synthetic materials, and monoclonal antibodies.21
Although hemodialysis is highly efficient at removing water-soluble drugs with low molecular weights (e.g., salicylates, ethanol, methanol, lithium) from the blood, it does not remove protein and lipid-bound substances; therefore it is not effective in the treatment of hypercholesterolemia, hyperbilirubinemia, toxicity associated with fulminant hepatic failure, or ingested toxins. Hemoperfusion is extremely effective in the treatment of these problems.21 Large molecules such as β2-microglobulin have been successfully removed by hemoperfusion with lower cardiovascular mortality,87 and clinical trials are currently underway with new adsorbents that are effective in removing β2-microglobulin.21
Hemoperfusion uses a hemodialysis circuit; however, the blood is passed through a cartridge containing the adsorbent surface, and no dialysate is present. Life-threatening poisonings from theophylline, carbamazepine, phenobarbital, and phenytoin may be an indication for charcoal hemoperfusion.74 With the use of activated charcoal, substances normally bound by lipid or protein are quickly and effectively removed from the intravascular space. Because hemoperfusion will not remove urea, it is not the treatment of choice for uremia.
Method
In the following discussion, a charcoal filter or cartridge will be used as the example of the adsorbent surface. As noted previously, the vascular access and circuit tubing used for hemoperfusion are often identical to those required for hemodialysis.
The charcoal filter is prepared, and the filter is given a glucose-heparin rinse. Because most commercially available cartridges require a priming volume of 50 to 300 mL, the total hemoperfusion circuit is primed with the equivalent of more than 10% of the circulating volume of a small child. As a result, the circuit usually is primed with blood before initiation of hemoperfusion.24
Some anticoagulation must be provided to prevent clot formation in the cartridge. Heparin usually is infused into the inflow tubing, between the patient and the cartridge. To minimize the risk of bleeding, protamine sulfate usually is added to the blood in the circuit beyond the cartridge (i.e., between the cartridge and the patient) to bind the heparin before the blood is returned to the patient.
Approximately 3 hours are required for hemoperfusion. The activated charcoal quickly will become saturated with the drug or substance removed from the blood. Frequently, several cartridges are required to complete one treatment. To determine the amount of toxin binding provided by the cartridge, blood levels of the toxic substance are drawn from the circuit immediately proximal to and distal from the cartridge. If these levels become nearly identical or if clotting is observed in the cartridge, the cartridge should be changed.
Complications
The activated charcoal binds lipid or protein-bound toxins. In addition, it binds glucose, calcium, and platelets; therefore serum concentrations of these substances must be monitored closely during hemoperfusion. Severe thrombocytopenia is the most common complication observed during this procedure.24
Occasionally, hemoperfusion effectively removes the toxins from the blood, but rebound toxicity develops several hours later as tissue-bound toxins move into the vascular space. For this reason, serum levels of the toxin should be monitored during the hemoperfusion and at regular intervals for several hours following hemoperfusion.
Bleeding can result from thrombocytopenia or heparinization. Because the half-life of protamine sulfate is shorter than the therapeutic effect of heparin, it may be necessary to administer additional protamine sulfate several hours after hemoperfusion is performed. During and after the procedure, the child should be observed closely for any evidence of bleeding, and all body secretions should be checked for the presence of blood.
Continuous Renal Replacement Therapy
The history of pediatric continuous renal replacement therapy (pCRRT) is rich with innovation, challenges, and change. In the late 1970s and early 1980s, the care of pediatric patients with AKI requiring renal replacement therapy involved improvised arteriovenous systems. Control of ultrafiltration rates in these systems required vigilant monitoring and frequent adjustment on the effluent side of the extracorporeal system.
Today, continuous renal replacement therapy (CRRT) devices are primarily venovenous and provide compact, efficient, and controlled management of fluid and electrolytes in pediatric patients with AKI. The Prospective Pediatric Continuous Renal Replacement Therapy Registry (ppCRRT Registry) was designed to evaluate clinical and therapeutic aspects of pCRRT in a prospective, observational manner.39 The ppCRRT Registry is a voluntary, multicenter, collaborative effort designed to evaluate clinical and therapeutic aspects of pCRRT and gather information about best pCCRT practices.38
The First Acute Dialysis Quality Initiative Conference examined evidence-based research and published consensus practice guidelines for CRRT in 2002.61 Complete guidelines are available online at http://www.ccm.pitt.edu/adqi/.1
Definition
CRRT performs the work of the kidney through an extracorporeal system designed to run continuously. The patient's blood flows through a filter to remove excess fluid, waste products, and toxins and to establish electrolyte and pH balance before it is returned to the patient. CRRT is similar to hemodialysis, but treatment is continuous rather than intermittent. The continuous aspect of CRRT provides more gradual fluid and electrolyte shifts and enables more tightly calibrated fluid management. CRRT as provided by current technology enables minute-to-minute adjustments in therapy titrated to patient need.
Indications for Continuous Renal Replacement Therapy
In the setting of AKI, the indications for renal replacement therapy are fluid and solute removal. Similar to PD, CRRT provides a gradual fluid and solute removal, but CRRT allows more sensitive fluid control than PD.101 Comparable to HD, CRRT can treat hypervolemia and electrolyte imbalance but instead of correcting these imbalances over 3 to 4 hours as with HD, CRRT will correct them over 1 day or more. For these reasons, CRRT is a valuable treatment choice in critically ill pediatric patients with AKI who are hemodynamically unstable with signs of multiorgan dysfunction syndrome. In data from the ppCRRT Registry, the most common acute diagnoses in patients receiving CRRT were sepsis and cardiogenic shock, and the most common comorbid conditions were bone marrow transplant and solid organ transplants—primarily the liver.38,88
In data from the ppCRRT Registry, immediate indications for CRRT include: fluid overload and electrolyte imbalance, fluid overload only, electrolyte imbalance only, prevention of fluid overload to allow intake, or other indications such as intoxications and inborn errors of metabolism. The combination of fluid overload and electrolyte imbalance was the most frequently encountered indication and had the lowest survival.
Recent experience supports the use of CRRT as a treatment modality for sepsis, not only for the management of fluids and electrolytes but also for the removal of inflammatory mediators of sepsis.26,28,33 CRRT has been effective after stem cell transplant, for treatment of drug toxicities, in acute solute removal in tumor lysis syndrome with associated hyperkalemia and hyperuricemia and to treat inborn error of metabolism.
Continuous Renal Replacement Therapy Modalities
CRRT is a broad term that encompasses treatment modalities involving an extracorporeal system and water removal by filtration. The treatment modalities include continuous arteriovenous hemofiltration (CAVH) and a variety of modalities provided by continuous venovenous hemofiltration.
The mechanism of solute transport is the distinguishing feature of the CRRT modalities. Solutes are removed by diffusion, convection, or both.
When solute is removed by diffusion, the process is similar to that described in the hemodialysis portion of this chapter. Solutes are removed from the blood through the filter's semipermeable membrane as the result of concentration gradients. Solutes move from the area of high concentration (blood) through the semipermeable membrane into an area of low concentration (i.e., fluid on the opposite side of the membrane).
The second mechanism of solute transport is convection. Solutes in solution are carried with the water across the semipermeable membrane in response to a transmembrane pressure gradient. Convection has been described as “solvent drag”: the bulk water flow literally drags the solutes through the semipermeable membrane. In venovenous CCRT, the transmembrane pressure gradient is generated by the speed of the blood pump or blood flow rate (BFR), which transmits a positive hydrostatic pressure inside the blood tubule of the filter. The rate of the effluent pump, or effluent and dialysate pump if dialysis is used, creates a negative pressure in the space surrounding the blood tubules, therefore increasing the transmembrane pressure gradient.
Continuous Arteriovenous Hemofiltration
Definition
CAVH uses an extracorporeal circuit and a small filter that is highly permeable to water and small solutes, but is impermeable to proteins and formed elements of the blood. The filter system is joined to both an arterial and a venous catheter. Passage of arterial blood through the filter results in the formation of an ultrafiltrate of plasma that consists of water and nonprotein bound solutes. The filtered blood is then returned to the patient through the venous catheter.
Because the filter used for CAVH contains no dialysate there are no concentration gradients established across the filter. The volume and content of the ultrafiltrate is determined by the rate of blood flow through the filter (essentially determined by the patient's blood pressure), the permeability of the filter, and the transmembrane pressure. The transmembrane pressure is created largely by the patient's arterial pressure, but it also can be augmented by elevating the patient above the fluid drainage bag. The transmembrane pressure also will be enhanced by the oncotic pressure difference between the patient blood and the ultrafiltrate, which is protein-free with an oncotic pressure of zero.
The volume of ultrafiltrate can also be adjusted through the use of negative pressure generated on one side of the filter by the use of a volume-controlled infusion pump that draws ultrafiltrate at a set hourly rate. The faster the rate set for the volume-controlled pump, the more ultrafiltrate drawn from the blood through the filter every hour. The infusion pump can create resistance to flow if it is adjusted to a relatively slow hourly rate. As a result, resistance to ultrafiltrate production will be created by slow flow through the infusion pump circuit. The use of infusion pumps in this setting is not optimal for neonates and small infants, because the pumps may be inaccurate and might remove fluid at an unreliable rate. For example, the amount of ultrafiltrate could actually be greater than the hourly rate set on the infusion pump.
CAVH can provide effective therapy for the treatment of AKI complicated by hypervolemia or electrolyte or acid-base disturbances. It is particularly useful in patients with extremely limited venous access, in neonates or small infants, and in patients with unstable cardiovascular function or multisystem organ failure who are likely to be intolerant of hemodialysis. This form of renal replacement is not recommended for the rapid treatment of hyperkalemia, because the rate of ultrafiltration is low and potassium removal is slower than with hemodialysis.19
CAVH may be used to remove a small but predictable volume of fluid from the vascular space. This procedure is helpful in the management of chronically ill patients with oliguria and hypervolemia (e.g., those with severe congestive heart failure), because excess fluid can be removed and replaced with parenteral fluid that has high nutrient value.
With the appropriate equipment, CAVH systems can be assembled and implemented relatively quickly. CAVH also can provide a useful option when resources are scarce. However, in many critical care areas CAVH has been replaced by venovenous hemofiltration machines with more precise technology to provide safer and more predictable blood flow and precise ultrafiltation.12,69
Contraindications
There are few contraindications to the use of CAVH. Active bleeding is a relative contraindication; PD is preferred in such patients. A severe coagulopathy is not a contraindication to the use of CAVH, although the risk of bleeding at the access site is increased.
Method
Preparation for Continuous Arteriovenous Hemofiltration
To begin CAVH, arterial and venous access must be achieved. It is extremely important to catheterize an artery large enough to allow sufficient flow into the filter; the femoral artery is used most frequently, although the umbilical artery may be catheterized in neonates. The femoral and external jugular veins are used most frequently for venous access.
The circuit tubing should contain multiple sampling ports, and it should be primed before use. A heparin infusion system (with appropriate infusion pump) should be joined to the arterial side of the circuit, and stopcocks and tubing should be placed in the circuit to enable bypass of the filter; these stopcocks will be used when the filters are changed. The filter and circuit are prepared, and the prime fluid should be warmed to no more than 37° C.
Many CAVH filters are commercially available, and most use a hollow fiber design. It is preferable to use small filters (20-mL prime volume) designed for use in small children to keep the volume in the circuit as low as possible during treatment. The ideal filter has a short fiber length, a large surface area, and a small priming volume. If the entire extracorporeal circuit must be primed with a volume exceeding 10% of the patient's blood volume, fresh bank blood should be used to prime the circuit.
As noted previously, a volume-controlled infusion pump may be required to enhance or limit the formation of ultrafiltrate. Occasionally, an additional fluid infusion pump and infusion system are prepared to administer fluid to the patient to enable replacement of ultrafiltrate with fluid that differs in solute or nutritional content (Fig. 13-10).
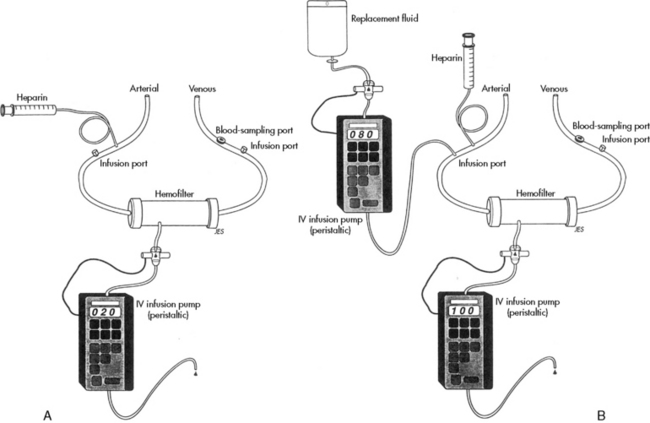
Fig. 13-10 Continuous arteriovenous hemofiltration. A, The rate of fluid removal can be controlled if a pediatric volume infusion pump is joined to the hemofilter. The variable resistance generated by the infusion pump will control the hemofilter transmembrane pressure and the filtration rate. B, Continuous hemofiltration generally requires infusion of replacement fluid to prevent volume depletion and to control the electrolytes lost through hemofiltration. Use of two pediatric infusion pumps enables controlled removal of fluid and controlled replacement of fluid and electrolytes.
(From Stark JE, Hammed J: Continuous hemofiltration and dialysis. In Blumer J, editor: A practical guide to pediatric intensive care, ed 3. St Louis, 1990, Mosby-Year Book.)
Initiation of Continuous Arteriovenous Hemofiltration
To initiate hemofiltration, the arterial and venous catheters are joined to the filter and drainage system. Clamps are placed on each limb of the circuit. The arterial and venous limbs are unclamped first, while the bypass tubing and the drainage limb remain clamped. The heparin infusion should commence when the vascular lines are unclamped. Once the area in the filter surrounding the blood tubules is filled with the patient's ultrafiltrate, the tubing of the drainage line can be unclamped.
During CAVH, the nurse is responsible for monitoring the volume of ultrafiltrate, which can be regulated with a volume-controlled infusion pump. Periodic sampling of the ultrafiltrate will be performed to monitor the solute content of the filtrate, so that appropriate replacement therapy can be provided.
Regular monitoring of the patient's activated clotting time (ACT) is required during CAVH. The heparin infusion is titrated to maintain an ACT that is approximately 1.5-fold of baseline (maximum, 200 s). The arterial ACT should be no greater than 10% above baseline to prevent bleeding.2
The filter should be examined frequently for the presence of clot formation. If clots are observed or ultrafiltrate formation decreases significantly, a physician or on-call provider should be notified and the filter probably should be changed. Filters can be expected to last approximately 12 hours, although the duration varies widely with patient condition.
Hemoglobin and hematocrit and serum chemistries should be monitored on a regular basis per unit protocol. The patient should be weighed once or twice every 24 hours.
Complications
The most common complications during CAVH are bleeding, thromboembolic events, and fluid balance problems. Bleeding occurs when heparinization is excessive, and it should be prevented with careful monitoring of arterial and venous ACTs. Anemia can result from excessive clot formation in the filters; therefore the child's hematocrit should be monitored frequently.
Air is trapped in air chambers, but thrombi entering the system or leaving the filter can embolize to the central venous system. These thrombi can be prevented by scrupulous examination of the filter and circuit and removal of any air or clots.
Hypervolemia can develop or be exacerbated by inadequate ultrafiltrate formation. Alternatively, hypovolemia can develop if an excessive volume of ultrafiltrate is removed. Electrolyte imbalances can be created by inappropriate priming fluid or excessive solute loss in the ultrafiltrate and will be detected with regular blood sampling. If electrolyte imbalances develop, adjustments are needed in the volume and solute content of the fluid replacement infusion or in the rate of ultrafiltrate removal.
Continuous Venovenous Hemofiltration Modalities
Slow continuous ultrafiltration uses an extracorporeal circuit with a hemofilter and can be set as arteriovenous or venovenous. Slow continuous ultrafiltration removes free water and small amounts of solute.
Continuous venovenous hemofiltration (CVVH) uses an extracorporeal circuit with a hemofilter. Blood is pumped from a large vein into the circuit; it flows through the filter, which removes fluid and solute, and then is pumped back into the patient through the venous line and catheter. If a double-lumen catheter is used, the blood is pulled from and returned to the same vein.
CVVH uses replacement fluid and therefore transports solutes primarily through convective transport. Replacement fluid is a physiologic solution infused into the blood component of the circuit either just before the filter (predilution) or immediately after the filter (postdilution), but before it is returned to the patient. The replacement fluid is used to replace large volumes of ultrafiltrate to help maintain the patient's fluid and electrolyte balance. The machine weighs the ultrafiltrate bag and calculates the volume of fluid removed (ultrafiltrate).
Continuous venovenous hemodialysis (CVVHD) uses an extracorporeal circuit with a filter. Blood is pumped from a large vein into the circuit, through the filter, which removes fluid and solute, and then the blood is pumped back into the patient's venous circulation. Dialysate fluid is pumped into the space surrounding the blood tubules in the filter in the direction opposite the blood flow (countercurrent flow).
Solute removal in CVVHD results from diffusion that is caused by concentration gradients between the blood and dialysate. The ultrafiltration fluid and the dialysate fluid are recovered into the same large effluent bag for disposal. The machine weighs and calculates the actual amount of fluid removed.
Continuous venovenous hemodiafiltration (CVVHDF) uses an extracorporeal system with a filter. Blood is pumped from a large vein into the circuit and through the filter, which removes fluid and solute, and then the blood is pumped back into the patient's venous circulation. CVVHDF uses dialysis fluid and replacement fluid as described previously; therefore solute transport occurs through both diffusion and convective transport mechanisms. In CVVHDF the ultrafiltrate, which includes the replacement fluid volume, and the dialysate are recovered in the same effluent bag. The machine is designed to subtract the dialysate and replacement fluid volumes from the ultrafiltrate volume to calculate the actual volume of fluid removed from the patient. Table 13-7 compares characteristics of different CRRT modalities, including the driving force of blood flow and the mechanism of solute transport.
Pediatric Considerations
Access issues are extremely important in all patients who need renal replacement therapy, but these challenges become much more difficult in small, critically ill patients. Access is the most important consideration contributing to the effectiveness of therapy, and constant problems can arise when access is inadequate.100
Data from the ppCRRT Registry indicate that femoral access is used most often.88 Care must be taken with femoral catheters in neonates and young children to prevent thrombosis and bleeding at the site. Femoral catheters are subject to kinking and are positional, and high negative pressures may lodge the catheter tip against the wall of the vessel, impeding flow. Femoral catheters may also have draw and inflow problems caused by increased intraabdominal pressure. These issues can be alleviated by careful positioning and switching the outflow (arterial) port with the inflow (venous) port of the catheter.
Catheter placement and size are based on the optimal site determined for the patient based on the risk of thrombosis and infection, ease of placement, and adequacy of function. Most pediatric practitioners recommend short, large-bore catheters in the right internal jugular vein or access in the femoral vein using the shortest possible catheter that will reach the inferior vena cava.16
Circuit volume must be considered. The CRRT circuit volume can represent a large percentage of the total blood volume in neonates or infants, especially if the patient is hemodynamically unstable as the result of sepsis, shock, or cardiac failure. The guidelines17,61 recommend a blood prime if the circuit volume represents >10% of the patient's blood volume or if the patient is hemodynamically unstable. It is important to remember that packed RBCs from the blood bank have high potassium concentration and a hematocrit of approximately 80%; therefore they should be diluted by 50% using either normal saline or albumin before the circuit prime.
Blood priming can cause early clotting and in some filters may cause acute hypotension that is thought to be caused by bradykinin release.17 If a blood prime is required, several procedures may help maintain hemodynamic stability while connecting the patient to the circuit.
The first procedure prepares the bank blood for infusion into the patient. This procedure, called zero balance ultrafiltration,46 circulates the bank blood through the CRRT circuit to normalize electrolytes and improve the acid-base status of the blood before connecting the circuit to the patient.
A second procedure uses a priming mixture of 75 mL of PRBCs, 75 mL of sodium bicarbonate, and 300 mg of calcium gluconate to be transfused post hemofilter.17 In a third option, called the bypass maneuver, RBCs are administered to the patient at the same rate that the patient's blood is entering the circuit. The heparinized priming solution is discarded as it is displaced by the patient's blood. When the circuit is completely primed with the patient's blood, the venous line of the circuit is attached to the patient and CRRT treatment is begun.35
Hypothermia is a significant problem in pediatrics, especially in neonates or small infants, whenever an extracorporeal system is used. Heat loss is related to the blood flow rate (BFR) and the volume of blood in the circuit. Heat loss can be problematic even in children with higher BFR and smaller circuit volume-to-blood volume ratios. All possible measures should be instituted to maintain normothermia. Newer machines incorporate blood warmers. However, if a warmer is not included in the circuit, the nurse should institute some method of providing external warming. Blood warmers can be attached to the patient return line of the circuit to warm the blood just before it is returned to the patient. Potential external warming devices include radiant warmers, body warmers, heating mattresses, and solution heaters.
Most commercially available systems have membrane choices that will meet the patient's needs and the prescribed therapy that is based on those needs. For a list of access catheters see Table 13-8. Table 13-9 lists characteristics of CRRT hemofilters used for children, including priming volume, and Table 13-10 lists machines.
Table 13-8 Recommended Catheter Sizes and Sites for Pediatric Continuous Renal Replacement Therapy
| Patient Size | Catheter Size and Location | Site of Insertion |
| Neonate <3-5 kg | Two 5-French single lumen | Femoral, internal jugular vein |
| Single 7-French double lumen, 10 cm (Medcomp) | Internal jugular, femoral vein | |
| Single 7-French double lumen, 13 cm (Cook) | Femoral vein | |
| Two 7-French umbilical venous catheters | Umbilical vein | |
| Infant or school-aged, 5-20 kg | Single 7-French triple lumen, 16 cm (Arrow) | Internal jugular, subclavian, or femoral vein |
| Single 8-French double lumen, 11-16 cm (Arrow, Kendall) | ||
| Infant or school-aged, >20-30 kg | Single 9-French double lumen, 12-15 cm (Medcomp) | Internal jugular, subclavian, or femoral vein |
| Single 10-French double lumen, 12-19.5 cm (Mahurkar) | ||
| Pediatric, 30-70 kg | Single 11.5-French double lumen, 12-20 cm (Medcomp, Mahurkar) | Internal jugular, subclavian, or femoral vein |
| Single 11.5- or 12-French triple lumen, 12-20 cm (Medcomp, Mahurkar, respectively) |
Data supplied by Cook Critical Care, Bloomington, IN.; Mahurkar catheters are marketed by Kendall Company, Mansfield, MA; Medcomp, Harleysville, PA.; The Kendall Company, Mansfield, MA. Adapted from McBryde KD, Bunchman TE: Continuous renal replacement therapy. In Wheeler DS, Wong HR and Shanley TP, editors: Pediatric critical care medicine basic science and clinical evidence, London, 2007, Springer-Verlag.
Table 13-10 Hemofiltration Machines
| Company | Machine | Lines |
| Edwards Lifesciences |
Aquarius | Adult Peds |
| Gambro | Prismaflex Prisma* |
Adult Peds |
| Baxter | BM 11† BM 11a† BM 25 Accura |
Adult Peds |
| Fresenius | 2008 | Adult Peds |
| B. Braun | Diapact | Adult Peds |
Baxter, Baxter Healthcare Corporation, Deerfield, IL; Braun, Braun Medical Incorporated, Renal Therapies Division, Bethleham, PA; Edwards Lifesciences, Draper UT: Fresenius, Fresdius Medical Care North America, Waltham, MA; Gambro, Gambro USA, Lakewood, CO; Hospal, Meyzieu, France.
* Prisma system no longer available, but supported by Gambro.
† Blood pump only; need the addition of the BM 14 to make the total of a BM 25 for the blood and ultrafiltration monitor together.
From Brophy PD, Bunchman TE: References and overview for hemofiltration in pediatrics and adolescents. Available at http://www.pcrrt.com/Protocols%20PCRRT%20Zurich.pdf, Accessed January 14, 2010.
Nutritional considerations in the pediatric patient during CRRT are focused on protein requirements. In critically ill children or in children during the postoperative period, provision of adequate dietary protein is the most important nutritional intervention. Recommended daily amounts of protein to be administered to these children range from 1.5 g/kg for patients with AKI who are not receiving dialysis to 3 to 4 g/kg for patients receiving CRRT.17 When critical illness is complicated by renal injury or failure, protein loss increases and it is imperative to consider this loss in planning the child's nutritional support. Protein loss in this setting results from increased muscle protein catabolism from insulin resistance, acidosis, and the presence of catabolic hormones and mediators.68
CRRT prescription variables include selecting the CRRT modality, the blood flow rate (BFR), the ultrafiltration rate, the dialysate rate, and the replacement fluid rate appropriate for the patient's clinical condition. In pediatric CVVH, a BFR of 4 to 6 mL/kg per minute is used, while attempting to keep the venous return pressure below 200 mm Hg. Dialysate and replacement fluid rates at 2000 mL/1.73 m2 per hour have historically been used with a minimum rate of 35 mL/kg per hour.17,100 The available data are insufficient to guide dialysate or replacement fluid rates, and wide variations exist worldwide in dialysis dosing.78,81,100 Because of the many options available, prescription decisions should be tailored to the needs of the patient.100
Contraindications and Indications
As with CAVH, there are few contraindications to venovenous CRRT. It is often the treatment of choice for critically ill pediatric patients who are hemodynamically unstable as a result of shock, sepsis, or poor cardiac function. It is also used in patients after transplant (e.g., liver, bone marrow, stem cell transplant) who are fragile and have a metabolic milieu that does not promote optimal organ function. Even bleeding is not a contraindication to venovenous CRRT.19 The patient must always be observed for bleeding at catheter sites, and vigilance must be heightened if the patient exhibits severe coagulopathy.
Method
Preparation for Venovenous Continuous Renal Replacement Therapy
When venovenous CRRT is ordered, the first step is to gather all the equipment: the machine, solutions, medications, and the circuit containing the hemofilter (cassette). With the new technology, all steps for inserting the cassette and priming the circuit are illustrated on the screen of the hemofiltration machine. It is imperative to follow each step in sequence. Do not jump to an “obvious” next step, because this can cause errors in preparation with omission of small but important details.
Initiation of Venovenous Continuous Renal Replacement Therapy
When priming of the circuit is complete, the patient is connected to the circuit. If the priming solution will be infused into the patient upon initiation of treatment, the patient's outflow catheter hub is connected to the red arterial outflow tubing of the circuit, and the venous return tubing is attached to the inflow hub of the patient's catheter.
To avoid infusing the priming solution into the patient when treatment is initiated, connect the outflow (arterial) line to the outflow hub of the patient's catheter, but do not connect the venous return tubing to the patient. As the patient's blood fills the circuit, begin the anticoagulation therapy (per physician order). Collect the priming solution in a sterile bag as it is flushed out of the circuit by the patient's blood. Once the patient's blood fills the circuit up to the venous return line and all of the prime solution has been displaced, stop the machine and attach the venous return tubing to the inflow hub of the patient's catheter, and restart the machine. This method discards the priming solution instead of infusing it into the patient, and prevents infusion of heparin and excess volume into the patient. This technique is especially suited for small patients, patients with poor cardiovascular function, and patients who are unable to handle the excess volume.
When considering priming methods, it is important to determine whether the patient can tolerate initial blood loss into the machine. To mitigate the effects of volume displacement, fluids (normal saline, albumin, packed RBCs) can be administered to the patient via the venous inflow catheter as priming takes place (similar to the bypass procedure described previously).
When the therapy is started, only the BFR and the anticoagulation therapy should be running. The other flow rates may be kept at zero to allow time to assess the patient's response to the extracorporeal treatment. Assess vital signs (heart rate, respiratory rate, and blood pressure) and systemic perfusion. If the patient is tolerating the procedure well, the flow rates can be adjusted to the prescribed settings. After a patient has received venovenous CRRT for a period of time, the critical care nurse becomes familiar with the patient's tolerance to treatment and can work with the healthcare team to address specific patient needs.
Throughout venovenous CRRT, the nurse assesses the patient's vital signs (including temperature) and systemic perfusion. The catheter insertion site is assessed frequently for bleeding and kinks or any obstruction to flow from the patient and back to the patient. Access for medications, calcium infusion (if using citrate as the anticoagulant), and maintenance fluid must be functional and frequently assessed. If possible, a triple lumen central venous catheter is ideal for performing venovenous CRRT and delivering calcium. The third lumen is used for intravenous administration of medications and fluid.69
It is essential to prevent hypothermia in pediatric patients; the patient should be kept normothermic. Cover the child with sufficient blankets, use warming pads or warming mattresses, and use a blood warmer placed as close to the patient's venous return site as possible. Many of the newer machines incorporate blood warmers in the system and they will be needed for most patients. The CRRT treatment may mask the development of fever; fever should be suspected if a patient no longer requires external warming procedures to maintain body temperature.
During the venovenous CRRT procedure the nurse is responsible for monitoring the patient, the machine and the entire circuit. This monitoring includes the catheter insertion site for bleeding, inadvertent clamping of the catheter, or kinks; the pressures of the pull (arterial) side, the filter and the venous return side; the flow rates; the character of the effluent (ultrafiltrate); the blood leak detector; the air chamber or detection site; and the circuit tubing for leaks, kinks, and to ensure proper attachment to the solution or effluent bags. The nurse must respond to each alarm and perform a thorough assessment and a step-by-step troubleshooting procedure to resolve the issue associated with the alarm.
Blood sampling and laboratory studies must be performed as needed (per protocol) to maintain appropriate anticoagulation. If heparin is used, ACT levels are drawn from a blood draw site distal to the filter. The ACT range is generally maintained in the range of 180 to 200 s or approximately 1.5-fold the baseline. If citrate anticoagulation is used, the ionized calcium for the machine should be drawn distal to the filter, and the patient's ionized calcium should be drawn from a separate site, such as the arterial line. Available protocols provide guidelines for the rate of citrate infusion into the outflow tubing and the proportional calcium infusion into the patient. The goal of the protocol is to maintain the patient's ionized calcium at 1.1 to 1.3 mmol/L and the machine's ionized calcium at 0.35 to 0.45 mmol/L.17,69
Patient management during CRRT focuses on the effectiveness of the treatment as assessed by the patient's perfusion parameters, fluid status, and serum laboratory values. The monitored laboratory values include serum electrolytes, glucose, ionized and total Ca2+, Mg2+, hemoglobin and hematocrit, pH, albumin, prothrombin time, activated partial thromboplastin time, BUN, and creatinine. Depending on the indication for CRRT, additional laboratory values may be monitored.
Complications
Complications associated with venovenous CRRT in general include bleeding, problems with fluid balance (hypervolemia or hypovolemia), hypothermia, and anemia. Bleeding is a consequence of the severity of illness and the need for optimal CRRT circuit function, which in turn relies on safe and reliable anticoagulation. Anticoagulation must be maintained within narrow parameters.
Careful patient assessment is needed. Support of optimal fluid balance requires monitoring of the patient's perfusion, CVP, meticulous records of intake and output, and close attention to the ultrafiltration rate and serial patient weights. The patient's clinical status is the best guide to treatment adjustments. Anemia can develop from clots formed in the filter or from discontinuing treatment without reinfusing the blood from the circuit back into the patient. The patient's hematocrit should be followed closely.
Anticoagulation therapy can cause specific complications in addition to those listed previously. Heparin therapy can cause systemic bleeding including epistaxis; intracranial, pulmonary, gastrointestinal, bladder and vaginal hemorrhage; and heparin-induced thrombocytopenia. These developments may require a change in anticoagulation therapy or discontinuation of all anticoagulation.18 Citrate anticoagulation can induce metabolic alkalosis (from the conversion of citrate to bicarbonate) and citrate lock, which is most common in patients with hepatic dysfunction or failure. Citrate lock occurs when citrate is not being cleared either in the filter or by the patient's liver. It is characterized by high serum total calcium and low serum ionized calcium. Treatment of this complication is to stop the citrate and calcium infusion for a designated period of time according to protocol (usually 30 min) and restarting the citrate at 70% of the prior rate.17
Brophy et al. published a multicenter evaluation of anticoagulation from ppCRRT Registry data comparing anticoagulation methods, filter life span, and patient complications. They concluded that when both heparin and citrate regional heparinization methods were commonly used, neither resulted in a longer filter life span, and citrate anticoagulation may result a in lower incidence of life-threatening complications.18