1 Soft tissue distress
Somatic dysfunction
The musculoskeletal system – sometimes referred to as the locomotor system – is the means whereby we act out and express our human existence – ‘The primary machinery of life’ is what one of osteopathy’s greatest researchers, Irwin Korr (1970), called it. While, medically speaking, the musculoskeletal system may lack the glamour and fascination of vital organs and systems, the fact is that the cardiovascular and neuroendocrine and digestive (and other) systems and organs exist only to service this great biomechanical structure through which we live and function. It is by means of our musculoskeletal system (not our kidneys or livers) that we perform tasks, play games, make love, impart treatment, perform on musical instruments, paint and, in these and a multitude of other ways, interact with one another and the planet.
The musculoskeletal system – comprising mainly bones, muscle and connective tissue structures, such as ligaments, gives shape and stability, and allows movement, to the human body, while also protecting the vital organs (Mooar 2007).
The musculoskeletal system is also by far the greatest energy user in the body, as well as being one of our primary sources of pain, discomfort and disability, whether localized or general, referred or reflex, acute or chronic.
The term ‘somatic dysfunction’ offers an accurate and a comprehensive term to describe all lesions of the musculoskeletal system – osseous and soft tissue (Licciardone 2005).
Somatic dysfunction can be defined as any impairment, or altered function, of related components of the somatic system (body framework), i.e. skeletal, arthrodial and myofascial structures, as well as related vascular, lymphatic and neural elements. This general expression (somatic dysfunction) obviously requires specific definition in any given situation, which should include identification of the particular structure, tissue or area involved.
Sometimes somatic dysfunction is the primary cause of pain, however sometimes it generates reflexive symptoms, or may act as a perpetuating feature. Palpation may provide clues to the true nature of an injury (Stanton 1996). A process of assessment and examinations may help to identify structural factors associated with pain and dysfunction, often associated with repeated injury or cumulative microtrauma involving postural, habitual or occupational ergonomic stresses.
Identifying sources of pain
Different dysfunctional tissues produce different qualities of symptoms (Kuchera 2005). For example: sclerotomal tissues (skeletal, arthrodial, and ligamentous generators) typically relate to dull, aching pain – which may be experienced some distance from the actual source; myotomal (muscle) pain is also poorly localized, possibly also at a distance from the generating site. Trigger point activity in hypertonic tissues might be involved. The pain is typically described as involving stiffness, aching and sometimes having a cramp-like quality. Note: Examples of sclerotomes and myotomes are illustrated in Chapter 5, while the palpatory characteristics (such as tissue texture changes) of dysfunctional tissues that generate pain locally, or at a distance, are to be found in Chapter 3.
The objectives, if not the methods we are discussing, are not new. Carl McConnell, a major force in early 20th century osteopathy, discussed the soft tissues as follows (McConnell 1962):
A pathological point of prime importance, for example, is that osseous malalignment is sustained by ligamentous rigidity. This rigidity is incepted by way of muscular fascial and tendinous tensions and stresses. Every case portrays a uniqueness in accordance with location, architectural plan and laws, tissue texture, regional and strength ratios, resident properties, environmental settings, resolution of forces etc. Remember I am speaking of the solid biological background of individual pathogenesis, the veritable soil of prediseased conditions. The lack of either sufficient, or efficient, soft-tissue work, is one reason for mediocre technique and recurrence of lesions. The same is evident in the correction of postural defects.
The causes and the results of local and general somatic dysfunction, whether traumatic, functional, postural, pathological or psychological in origin, require a brief overview as we explore different aspects of the issues and the tissues involved, so that some of their possible solutions might become clearer.
Coherent and incoherent patterns
In Chapter 3 we will examine one of the major causes of somatic pain and dysfunction, myofascial trigger points, and the causes of this widespread phenomenon. It will then become clear that, while many forms of (referred) pain follow predictable and neurologically coherent pathways, there also exist patterns of pain and dysfunction that do not.
In this chapter our task is to evaluate a variety of influences on the evolution of soft tissue dysfunction, which follow a chronological sequence – the ways in which what is happening in an acute setting differs from what is taking place in a chronic situation, where an initial alarm state progressively gives way to a degree of organization, adaptation, compensation and (if not prevented) decompensation and dysfunction. It will also be explained just why not all muscles respond to stressors in quite the same way.
Reporting stations
The reporting mechanisms in the soft tissues and joints (Travell & Simons 1983, 1992, Wall & Melzack 1991) may be thought of as providing answers to a number of basic questions that the central nervous system (CNS) requires answering.
These questions were expressed by Keith Buzzell (1970) as follows: ‘What is happening in the peripheral machinery with respect to three questions? What is the present position? If there is motion, where is it taking us? And, third, how fast is it taking us there?’
The various neural reporting organs provide a constant information feedback to the CNS and higher centres as to the current state of tone, tension, movement, etc. of the tissues housing them. Such sensory information can be modulated and modified both by the influence of the mind, and by changes in blood chemistry, to which the sympathetic nervous system is sensitive. A variety of inputs of information will give the answers to these important questions so that the body can provide appropriate responses to the demands and adaptations constantly called for by varying situations. Some important structures involved in this internal information highway are summarized in Box 1.1.
Ruffini end-organs
These are found within the joint capsule, around the joint, so that each is responsible for describing what is happening over an angle of approximately 15°, with a degree of overlap between it and the adjacent end-organ. These organs are not easily fatigued, and are recruited progressively as the joint moves, so that movement is smooth and not jerky. The prime concern of Ruffini end-organs is to maintain a steady position. They are also, to some extent, concerned with reporting the direction of movement.
Golgi end-organs
These, too, adapt slowly, and continue to discharge over a lengthy period. They are found in the ligaments associated with the joint. Unlike the Ruffini end-organs, which respond to muscular contraction that alters tension in the joint capsule, Golgi end-organs are not thus affected, and can deliver information independently of the state of muscular contraction. This helps the body to know just where the joint is at any given moment, irrespective of muscular activity.
Pacinian corpuscle
This is found in periarticular connective tissue, and adapts rapidly. It triggers discharges, and then ceases reporting in a very short space of time. These messages occur successively, during motion, and the CNS can therefore be aware of the rate of acceleration of movement taking place in the area. It is sometimes called an acceleration receptor.
There are other end-organs, but these three can be seen to provide information regarding the present position, direction and rate of movement of any joint.
Muscle spindle
This receptor is sensitive and complex. It detects, evaluates, reports and adjusts the length of the muscle in which it lies, setting its tone. Acting with the Golgi tendon organ, most of the information as to muscle tone and movement is reported. The spindles lie parallel to the muscle fibres, and are attached either to skeletal muscle or to the tendinous portion of the muscle. Inside the spindle are fibres that may be one of two types. One is described as a ‘nuclear bag’ fibre, and the other as a chain fibre. In different muscles the ratios of these internal spindle fibres differ. In the centre of the spindle is a receptor called the annulospiral receptor (or primary ending), and on each side of this lies a ‘flower spray receptor’ (secondary ending). The primary ending discharges rapidly, and this occurs in response to even small changes in muscle length. The secondary ending compensates for this, because it fires messages only when larger changes in muscle length have occurred.
The spindle is a ‘length comparator’, and may discharge for long periods at a time. Within the spindle there are fine, intrafusal fibres that alter the sensitivity of the spindle. These can be altered without any actual change taking place in the length of the muscle itself, via an independent γ-efferent supply to the intrafusal fibres. This has implications in a variety of acute and chronic problems.
The proprioceptive role of muscles of the suboccipital region is directly related to the number of spindles per gram of muscle. There are an average of 36 spindles per gram in some of the suboccipital muscles, such as rectus capitis posterior minor, and 30.5 spindles per gram in rectus capitis posterior major, compared, for example, with 7.6 spindles per gram in splenius capitis and just 0.8 spindles per gram in gluteus maximus (Peck et al 1984). McPartland & Brodeur (1999) suggest that ‘The high density of muscle spindles found in the RCPM muscles suggests a value … [which] lies not in their motor function, but in their role as “proprioceptive monitors” of the cervical spine and head.’
Buzzell (1970) describes the neural connections with the CNS thus:
The central connections of the spindle receptors are important. The annulospiral fibre has the only known monosynaptic relationship in the body. As the fibre passes to the cord, and through the dorsal horn, it goes without synapse, directly to the anterior horn cells that serve the muscle fibres in the vicinity of the spindle. This is the basis of the so called ‘tendon reflex’, which actually is not a tendon reflex, but simply a spindle response to a sudden elongation of the muscle.
In contrast, the secondary fibres have various synapses in their central connection which can be traced to higher cortical centres. Conscious activity may, therefore, provide a modifying influence, via these structures, on muscle tone. The activities of the spindle appear to provide information regarding length, velocity of contraction and changes in velocity. How long is the muscle? How quickly is it changing length? And what is happening to this rate of change of length? (Gray 1977).
Golgi tendon receptors
These structures indicate how hard the muscle is working, they reflect the tension of the muscle, rather than its length, as does the spindle. If the tendon organ detects excessive overload it may cause cessation of function of the muscle, to prevent damage. This produces relaxation.
There are various ways of ‘manipulating’ the neural reporting stations to produce physiological modifications in soft tissues – notably of the Golgi tendon organ in muscle energy techniques (METs) and of the spindle in various positional release (PR) techniques, such as strain/counterstrain (SCS) (Jones 1980, Stiles 1984).
Effect of contradictory information
Korr’s words regarding the nature of the information that these, and other, reporting stations are providing to the CNS are worth recording (Korr 1976). He reminds us:
The spinal cord is the keyboard on which the brain plays when it calls for activity or for change in activity. But each ‘key’ in the console sounds, not an individual ‘tone’, such as the contraction of a particular group of muscle fibres, but a whole ‘melody’ of activity, even a ‘symphony’ of motion. In other words, built into the cord is a large repertoire of patterns of activity, each involving the complex, harmonious, delicately balanced orchestration of the contractions and relaxations of many muscles. The brain ‘thinks’ in terms of whole motions, not individual muscles. It calls selectively, for the preprogrammed patterns in the cord and brain stem, modifying them in countless ways and combining them in an infinite variety of still more complex patterns. Each activity is also subject to further modulation, refinement, and adjustment by the afferent feedback continually streaming in from the participating muscles, tendons, and joints.
This means that the pattern of information fed back to the CNS and brain from neural reporting stations (proprioceptors, mechanoreceptors, nociceptors, etc.) reflects, at any given time, the steady state of joints, the direction as well as speed of alteration in position of joints, together with data on the length of muscle fibres, the degree of load that is being borne, along with the tension this involves. This total input is what occurs, rather than individual pieces of information, as outlined above, from particular reporting stations.
Contradictory gibberish?
But what if any of the mass of information being constantly received should be contradictory, and actually conflict with the other information being received?
It is possible, for example, for the excessive force exerted by external trauma to induce such hyperactivity of the joint and muscle receptors that the reports from that area become gibberish.
Should conflicting reports reach the cord from a variety of sources simultaneously, no discernible pattern may be recognized by the CNS. In such a case no adequate response would be forthcoming, and it is probable that activity would be stopped. Spasm, or splinting, could therefore result.
When somatosensory, vestibular or visual afferent systems provide conflicting information, a variety of symptoms may result. Somatosensory afferent systems depend on coherent input, from the soles of the feet, the neck and lumbar spine (Gagey 1986).
Neural ‘cross-talk’
Korr (1976) discussed a variety of insults that could result in increased neural excitability: the triggering of a barrage of supernumerary impulses, to and from the cord, and also what he terms ‘cross-talk’, in which axons may overload and pass impulses to one another directly; muscle contraction disturbances, vasomotion, pain impulses, reflex mechanisms, disturbances in sympathetic activity, all may result from such activity, due to what might be relatively slight tissue changes, for example in the intervertebral foramina.
In addition, Korr reported that when any tissue is disturbed, whether bone, joint, ligament or muscle, the local stresses feed constant information to the cord, and effectively jam normal patterned transmission from the periphery. These factors, combined with any mechanical alterations in the tissues, are the background to much somatic dysfunction.
He summarized thus (Korr 1976):
These are the somatic insults, the sources of incoherent, and meaningless feedback, that causes the spinal cord to halt normal operations and to freeze the status quo in the offending and offended tissues. It is these phenomena that are detectable at the body surface, and are reflected in disorders of muscle tension, tissue texture, visceral and circulatory function, and even secretory function; the elements that are so much a part of osteopathic diagnosis.
These are the palpable changes that we will be evaluating in later chapters that can allow us the opportunity to ‘read’ dysfunction, and to potentially choose therapeutic measures – such as neuromuscular technique – to assist in restoration of normal function.
Mechanisms that alter proprioception
• Ischaemic or inflammatory events at the receptor site may produce diminished proprioceptive sensitivity due to metabolic byproduct build-up stimulating groups III and IV, mainly pain afferents (this also occurs in muscle fatigue).
• Physical trauma can directly affect receptor axons (articular receptors, muscle spindles and their innervations).
• In direct trauma to muscle, spindle damage can lead to denervation and atrophy (e.g. following whiplash) (Hallgren et al 1993).
• Structural changes in parent tissue lead to atrophy and loss of sensitivity in detecting movement, plus altered firing rate (e.g. during stretching).
• Loss of muscle force (and possibly wasting) may result when a reduced afferent pattern leads to central reflexogenic inhibition of motor neurons supplying affected muscle.
• Psychomotor influences (e.g. feeling of insecurity) can alter patterns of muscle recruitment at the local level, and may result in disuse muscle weakness.
Trophic neural influences
Setting aside for the moment the obviously important feature of nerves, and their message-carrying functions, we need to consider the less understood role they play in transporting substances – proteins, phospholipids, glycoproteins, neurotransmitters, enzymes, mitochondria and more (Canals et al 2004).
Transportation takes place, at a rate of anything from 1 mm/24 h to several hundred millimetres per 24 h, depending on what is being transported and the presence, or absence, of interfering factors. Movement occurs in both directions along nerves, with retrograde (returning from the target tissues towards the CNS) transportation seemingly ‘a fundamental means of communication between neurons and between neurons and non-neuronal cells’ which strongly influences the ‘plasticity of the nervous system’, according to Korr’s research (Korr 1981).
Patterson & Wurster (1997) note that substances known as nerve growth factors (NGFs) are supplied to the neural structures by the target (end) organ to which neurotrophic substances are being transported. ‘If the end organ does not supply the NGF, the synaptic contact is lost.’ They continue: ‘Complete withdrawal of NGF or of the material delivered by the nerve to its end organ may result in loss of function … The occurrence of the tissue tensions and fluid flow disturbances often associated with somatic dysfunctions can be factors in altering axoplasmic flows.’
Butler (1991) reports that there are two speeds of delivery of trophic substances via the nerves:
The fast transport moves at approximately 400 mm per day and the substances carried, such as neurotransmitters and transmitter vesicles, are for use in transmission of impulses at the synapse. This transport depends on an uninterrupted supply of energy from the blood. Various toxic substances and deprivation of blood will slow or block the transport.
In the slow antegrade (delivery) transport (1–6 mm per day), cytoskeletal material is carried. The return transportation (retrograde) along the nerve carries recycled transmitter vesicles and extracellular material. Butler suggests that ‘It also seems likely that the retrograde flow carries “trophic messages” about the status of the axon, the synapse and the general environment around the synapse including the target tissues.’
Very significantly, Butler states: ‘If the retrograde flow is altered by physical constriction, or from loss of blood flow, nerve cell body reactions are induced.’
Korr (1981) demonstrated that red (postural) and white (phasic) muscle fibres, which differ morphologically, functionally, chemically and, as we will discover later in this chapter, in their response to stress, can have all of their characteristic differences reversed if their innervation is ‘crossed’, so that red muscles receive white muscle innervation and vice versa. ‘This means, in effect, that the nerve instructs the muscle what kind of muscle to be, and is an expression of a neurally mediated genetic influence’, according to Korr (1981).
Neural influences on gene expression
Korr’s research (1981) therefore suggests that it is the nervous system that largely determines which genes in a muscle will be suppressed, and which expressed, and this information is carried in the material being transported along the axons. (See Box 1.2 for additional influences on gene expression.)
Korr (1981) demonstrated that obstruction of axonal transport modifies gene expression. Additional modulation of gene expression is now known to derive from biomechanical influences, specifically the status of minute structures – integrins – that penetrate the cell surface, acting as a communicating mechanism between the extracellular and intracellular environment.
‘Integrin molecules carry tension from the extracellular matrix, across the cell surface to the cytoskeleton which behaves as a tensegrity matrix’ (Wang et al 1993).
‘Of particular interest are the roles of the integrins in the migration of cells that defend the body against disease and repair injuries’ (Horwitz 1997, Hynes 1992).
The precise effects of tense, distorted, contracted, fibrosed or otherwise dysfunctional tissues on the integrins of the cells in these tissues, and subsequent gene expression, are topics for research, as are the potentially beneficial influences of appropriate bodywork designed to normalize such tissues (Oschman 2000).
Moving beyond neural and mechanical influences on gene expression, Martin (2001) interviewed functional medicine expert Jeffrey Bland, who observed:
Functional genomics derived out of the human genome project, in which it was thought that by dissecting the code of life in our 23 pairs of chromosomes, people would be able to understand how they were going to die. They would see locked in their genes heart disease, cancer, diabetes, arthritis, whatever it might be, and they would tell from these genetic imperfections what day, and what disease, they would finally fall prey to … Mendelian determinism … said that locked into our genes, when the sperm met the egg, were these strengths and weaknesses that we call the recessive and dominant characteristics of inheritance, … [and that] basically if we had the genes for cancer we would die of cancer, [and] if we had the genes for heart disease we would die of heart disease. It turns out that the human genome project has discovered that the genes that we thought were hard-wired to produce these diseases, are not hard-wired at all. Within our genes are multiple messages, and the message that is expressed at any moment – that’s in our phenotype – is a consequence of the environmental messages including diet, lifestyle, environment, that wash over our genes to give rise to different expression paths of the genes … some may be healthy, some may be unhealthy … what we’re really seeing is that the major determinants for the expression of genetic patterns, over decades of living, are the decisions that we make, either consciously or subconsciously, every day. How we exercise, how we work, what our stress patterns are.
A picture emerges that suggests structural features, neural function, stress and emotion, as well as environmental factors including diet, all determine the way in which genetic predispositions are either modified, modulated, contained or expressed.
When a muscle loses contact with its nerve (as in anterior poliomyelitis, for example) atrophy occurs, not as a result of disuse but because of loss of the integrity of the connection between nerve cells and muscle cells at the myoneural junction, where nutrient exchange occurs irrespective of whether or not impulses are being transmitted.
Korr (1981) also describes just how vulnerable these highways of nutrition are:
Any factor which causes derangement of transport mechanisms in the axon, or that chronically alters the quality or quantity of the axonally transported substances, could cause the trophic influences to become detrimental. This alteration in turn would produce aberrations of structure, function and metabolism, thereby contributing to dysfunction and disease.
What could cause such neurotrophic interference?
Korr specifies: ‘Deformations of nerves and roots, such as compression, stretching, angulation and torsion’, especially, he tells us, ‘in their passage over highly mobile joints, through bony canals, intervertebral foramina, fascial layers and tonically contracted muscles’.
Mention by Korr, Butler and others of the changes that can have a negative influence on neurotrophic flow include altered circulatory status as well as hypertonicity. Trigger point activity, as we will see in later chapters, should be capable of directly producing just such changes. Normalization of trigger point activity, and the aetiological factors that produced them, utilizing NMT for example, should therefore be at least one way of assisting more normal neurotrophic function.
More general normalization of somatic dysfunctions, which include not just trigger points but the entire range of shortened, fibrotic, hypertonic, oedematous, inflamed, restricted or otherwise compromised soft and osseous structures, can therefore be seen to offer benefit in restoration of neurotrophic function, and therefore of body functions generally.
Maitland and Butler: ‘abnormal neural movement’
Butler & Gifford (1989), building on the original work of Maitland (1986), have shown how what they term ‘adverse tension’ in the nervous system can impair its mobility and elasticity, and how many painful problems can result from this. Butler and Gifford’s detailed analysis of the diagnosis and treatment of such restrictions and tensions is highly recommended to manual therapists. The tissues that surround neural structures are known as the mechanical interface (MI). These adjacent tissues are those that can move independently of the nervous system; for example, supinator muscle is the MI to the radial nerve, as it passes through the radial tunnel.
There is no general agreement as to the terminology that should be used in describing such biomechanical changes in the neural environment. Maitland et al (2001), for example, suggest that ‘abnormal neural movement’ is a more accurate description than ‘neural tension’.
Whatever we term the dysfunctional pattern, Butler and Gifford’s focus on those ‘adverse mechanical’ changes that negatively influence neural function, and that cause a multitude of symptoms, including pain, has been an important contribution to our understanding of some aspects of pain and dysfunction.
Any pathology in the MI can produce abnormalities in nerve movement, resulting in tension on the neural structure with unpredictable ramifications. Examples of MI pathology include nerve impingement by disc protrusion, or osteophyte contact, and carpal tunnel constriction. These problems would be regarded as mechanical in origin as far as the nerve restriction is concerned. Any symptoms resulting from mechanical impingement on neural structures will be more readily provoked in tests that involve movement rather than pure (passive) tension (Alshami & Hodges 2006).
Chemical or inflammatory causes of neural tension can also occur, resulting in ‘interneural fibrosis’, which leads to reduced elasticity and increased ‘tension’, and would become obvious with tension testing of these structures.
Pathophysiological changes resulting from inflammation, or from chemical damage (i.e. toxicity), are noted by Butler & Gifford (1989) as commonly leading on to internal mechanical restrictions of neural structures in a different manner to mechanical causes such as those, for example, imposed by a disc lesion.
Korr (1970) states:
To appreciate the vulnerability of the segmental nervous system to somatic insults it must be understood that much of the pathway taken by nerves as they emerge from the cord is actually through skeletal muscle. The great contractile forces of skeletal muscles with the accompanying chemical changes exert profound influences on the metabolism and excitability of neurons. In this environment the neurons are subject to quite considerable mechanical and chemical influences of various kinds, compression and torsion and many others … slight mechanical stresses may, over a period of time, produce adhesions, constrictions and angulations imposed by protective layers.
Adverse mechanical tension changes do not necessarily affect nerve conduction, according to Butler and Gifford; however, Korr’s research indicates that axonal transport may be affected.
From the perspective of the manual therapist, this knowledge is extremely important.
The dysfunctional tissues and patients being treated by massage therapists, neuromuscular therapists, physiotherapists, Rolfers, Heller Workers, osteopathic and chiropractic practitioners, and those using movement therapies (Pilates, Feldenkrais, etc.), all have the potential to involve the mechanically interfacing structures in which neural tissues lie, and where normal mobility should be present (and often is not).
The role of neuromuscular techniques in management of adaptive overload
This book has, as a primary focus, the use of neuromuscular techniques (NMTs) in assessing and treating somatic dysfunction. The objectives of neuromuscular technique (NMT as practised in Europe) and neuromuscular therapy (NMT as practised in the USA) are summarized in Box 1.3.
Box 1.3 NMT: European (Lief’s) neuromuscular technique and American neuromuscular therapy (Chaitow & Delaney 2000)
Neuromuscular technique, as the term is used in this book, refers to the manual application of specialized (usually) digital pressure and strokes, most commonly applied by finger or thumb contact. These digital contacts can have either a diagnostic (assessment) or therapeutic objective, and the degree of pressure employed varies considerably between these two modes of application. There are subtle differences between the European and American versions of NMT. Detailed descriptions of Lief’s NMT will be found in Chapter 6, while the American NMT version is outlined by Judith DeLany in Chapter 10.
Additional complementary manual methods to NMT (both versions) include massage, muscle energy technique (MET), positional release technique (PRT), myofascial release technique (MRT) and variations of these soft tissue manipulation approaches.
Aims of NMT
Therapeutically, NMT aims to produce modifications in dysfunctional tissue, encouraging a restoration of functional normality, with a particular focus of deactivating focal points of reflexogenic activity, such as myofascial trigger points.
An alternative objective of NMT application is toward normalizing imbalances in hypertonic and/or fibrotic tissues, either as an end in itself or as a precursor to joint mobilization or manipulation.
• deactivate myofascial trigger points
• prepare for other therapeutic methods such as exercise or manipulation
• relax and normalize tense, fibrotic muscular tissue
• enhance lymphatic and general circulation and drainage
• simultaneously offer the practitioner diagnostic information.
Neuromuscular therapy (USA) utilizes similar manual methods, as well as re-education, rehabilitation and home-care approaches, to those utilized in European methodology.
NMT attempts to address a number of features that are all commonly involved in causing or intensifying pain and dysfunction (Chaitow & DeLany 2000) including, among others:
• biochemical features – nutritional imbalances and deficiencies, toxicity (exogenous and endogenous), endocrine imbalances (e.g. thyroid deficiency), ischaemia, inflammation
• pyschosocial factors – stress, anxiety, depression, etc.
• biomechanical factors – posture, including patterns of use, hyperventilation tendencies, as well as locally dysfunctional states such as hypertonia, trigger points, neural compression or entrapment.
NMT sees its role as attempting to normalize or modulate whichever of these (or additional) influences on musculoskeletal pain and dysfunction can be identified in order to remove or modify as many aetiological and perpetuating influences as possible (Simons et al 1999), without creating further distress or requirement for excessive adaptation.
To understand the context for application of such approaches, we need to appreciate time-related influences on the evolution of dysfunction: how, over time, a progression occurs that alters acute responses, as the tissues locally, or the body as a whole, modify, accommodate, compensate and adapt to the demands, stresses and insults of daily life.
General adaptation syndrome (GAS) and local adaptation syndrome (LAS), and connective tissue
Selye (1976) called stress the non-specific element in disease production. In describing the relationship between the general adaptation syndrome (GAS) – i.e. alarm reaction, resistance (adaptation) phase followed by the exhaustion phase (when adaptation finally fails), which affects the organism as a whole – and the local adaptation syndrome (LAS), which affects a specific stressed area of the body, Selye also emphasized the importance of connective tissue. He demonstrated that stress results in a pattern of adaptation, individual to each organism. He further showed that, when an individual is acutely alarmed, stressed or aroused, homeostatic (self-normalizing) mechanisms are activated – this is the alarm reaction of Selye’s general (and local) adaptation syndromes.
If the alarm status is prolonged or repetitive, defensive adaptation processes commence and produce long-term – chronic – changes. In assessing (palpating) the patient, these neuromusculoskeletal changes represent a record of the attempts on the part of the body to adapt and adjust to the stresses imposed upon it as time passes. The results of repeated postural and traumatic insults of a lifetime, combined with changes of emotional and psychological origin, will often present a confusing pattern of tense, contracted, bunched, fatigued and ultimately fibrous tissue (Chaitow 1989).
The minutiae of the process are not, for the moment, at issue. What is important is the realization that, due to prolonged stress of a postural, psychic or mechanical type, discrete areas of the body become so altered by the efforts to compensate and adapt that structural and, eventually, pathological changes become apparent. Researchers have shown that the type of stress involved can be entirely physical in nature (Wall & Melzack 1991) (e.g. a single injury or repetitive postural strain) or purely psychic in nature (Latey 1983) (e.g. chronically repressed anger). An example of localized emotional stress influences on muscle tissue is given in Box 1.4. Wider biomechanical responses to emotional stress are discussed later in this chapter.
Box 1.4 Selective motor unit involvement
The effect of psychological influences on muscles seems to be more complex than a simplistic ‘whole’ muscle or regional involvement. It has been demonstrated that a small number of motor units in particular muscles may display almost constant, or repeated, activity when influenced psychogenically (Waersted et al 1993). The reaction time taken to perform tasks was evaluated in normal individuals, so creating a ‘time pressure’ anxiety. Researchers were able to demonstrate low-amplitude levels of activity (using surface EMG in trapezius muscles) even when the muscle was not being employed. It seems that, in spite of low total activity level of the muscle, a small pool of low-threshold motor units may be under considerable load for prolonged periods of time.
Such a recruitment pattern would be in agreement with the ‘size principle’ first proposed by Henneman (1957), which states that motor units are recruited according to their size. Motor units with type I (postural) fibres are predominant among the small, low-threshold units. If tension-provoking factors (e.g. anxiety) are frequently present, and the subject repeatedly recruits the same motor units, overload may follow, possibly resulting in a metabolic crisis and the appearance of type I fibres with an abnormally large diameter, or ‘ragged-red’ fibres, which are interpreted as a sign of mitochondrial overload (Edwards 1988, Larsson et al 1990). The implications of this information are profound, because they suggest that emotional stress can selectively involve postural muscle fibres that have a tendency to shorten over time when stressed (Janda 1983).
The possible ‘metabolic crisis’ suggested by this research has strong parallels with the evolution of myofascial trigger points as suggested by Simons, a topic that is discussed in greater detail in later chapters (Wolfe & Simons 1992).
More often than not a combination of emotional and physical stresses will so alter neuromusculoskeletal structures as to create a series of identifiable physical changes, which will themselves generate further stress, such as pain, joint restriction, general discomfort and fatigue.
As described in this and later chapters, predictable chain reactions of compensating changes will evolve in the soft tissues in most instances of chronic adaptation to biomechanical and psychogenic stress (Lewit 1992). Such adaptation is almost always at the expense of optimal function, as well as being an ongoing source of further physiological embarrassment.
A biomechanical stress response sequence
(Basmajian 1974, Dvorak & Dvorak 1984, Janda 1982, 1983, Korr 1978, Lewit 1999, Travell & Simons 1983, 1992, Liebenson 2006, Key 2007, Vleeming et al 2007)
When the musculoskeletal system is ‘stressed’, a sequence of events occurs which can be summarized as follows:
• ‘Something’ (see Causes of soft tissue dysfunction below) occurs that leads to increased muscular tone.
• Increased tone, if anything but short-term, leads to a retention of metabolic wastes.
• Increased tone simultaneously leads to a degree of localized oxygen lack (relative to the efforts being demanded of the tissues) – encouraging ischaemia.
• Increased tone might also lead to a degree of oedema.
• These factors (retention of wastes, ischaemia, oedema) result in discomfort or pain.
• Discomfort or pain leads to increased or maintained hypertonicity.
• Inflammation, or at least chronic irritation, may be a result.
• Neurological reporting stations in hypertonic tissues will feed the CNS with information regarding their status, leading to a degree of sensitization of neural structures, and the evolution of facilitation – hyperreactivity (see Ch. 2).
• Macrophages are activated, as is increased vascularity and fibroblastic activity.
• Connective tissue production increases, with cross-linkage, leading to shortened fascia.
• As all fascia/connective tissue is continuous throughout the body, any distortions that develop in one region can potentially create distortions elsewhere, so having a negative influence on structures that are supported by, or attached to, the fascia, including nerves, muscles, lymph structures and blood vessels.
• Changes occur in the elastic (muscle) tissues, leading to chronic hypertonicity and, ultimately, to fibrotic changes.
• Hypertonicity in a muscle will produce inhibition of its antagonist muscles.
• Chain reactions evolve in which some muscles (postural – type I) shorten, while others (phasic – type II) lengthen.
• Because of sustained increased muscle tension, ischaemia in tendinous structures occurs, as it does in localized areas of muscles, and periosteal pain areas develop.
• Abnormal biomechanical effects occur, involving malcoordination of movement – with antagonist muscle groups becoming hypertonic (e.g. erector spinae) or inhibited (e.g. the rectus abdominis group).
• The firing sequences of antagonistic and synergistic muscles alter.
• Joint restrictions and/or imbalances as well as fascial shortenings develop.
• Progressive evolution of localized areas of hyperreactivity of neural structures occurs (facilitated areas) in paraspinal regions or within muscles (trigger points).
• The degree of energy wastage due to unnecessarily maintained hypertonicity leads to generalized fatigue.
• More widespread functional changes develop – for example affecting respiratory function – with repercussions on the total economy of the body.
Unsustainable without consequences
In the presence of a constant neurological feedback of impulses to the CNS/brain from neural reporting stations indicating heightened arousal (a hypertonic muscle status is the alarm reaction of the flight/fight alarm response), there will be increased levels of psychological arousal and an inability to relax adequately with consequent increase in hypertonicity. Functional patterns of use, of a biologically unsustainable nature, will emerge, commonly involving chronic musculoskeletal problems and pain.
At this stage, restoration of normal function requires therapeutic input which addresses the multiple changes that have occurred as well as the need to re-educate the individual as to how to use their body – to breathe, to carry and to use themselves – in less stressful ways.
The chronic adaptive changes that develop in such a scenario lead to the increased likelihood of future acute exacerbations, as the progressively chronic, less supple and resilient, biomechanical structures attempt to cope with new stress factors resulting from the normal demands of modern living.
Causes of soft tissue dysfunction
The many elements that can contribute to the ‘stress’ sequence, leading to musculoskeletal pain include:
1. Congenital factors (short or long leg, small hemipelvis, fascial, cranial and other distortions, hypermobility tendencies) (Gofton & Trueman 1971, Schamberger 2002)
2. Acquired malalignment, asymmetry, resulting from overuse, misuse and abuse (and disuse) factors (such as injury or inappropriate patterns of use involved in work, sport or regular activities) (Schamberger 2002)
3. Postural stresses (Key 2007, Vleeming et al 2007)
4. Chronic negative emotional states (anxiety, etc.) – see Box 1.4 (Bendtsen et al 1996)
5. Reflexive factors (trigger points, facilitated spinal regions) (Simons, Travell & Simons 1999)
As a result of the processes described above – most of which affect each and every one of us to some degree – acute and painful problems, overlaid on chronic soft tissue changes, become the norm – the raw material on which bodywork therapies focus.
Some major soft tissue stress categories
Physiological factors
These might involve an overall increase of muscle hypertonicity/tension or localized soft tissue changes due to habitual patterns of use or from patterns of overuse. Occupational, sporting, leisure and general activities are all potential producers of such repetitive or constant stress involving the soft tissues (Janda 1982, 1988).
Emotional factors
All emotional changes are mirrored in muscular changes. Emotional attitudes such as anger or fear, as well as moods such as excitement, anxiety or depression, are known to produce altered muscular postures and patterns. There is a close relationship between habitual tension patterns and posture, and psychological attitudes and conflicts. The use of the body as a metaphor for emotional feelings (‘pain in the neck’) is well documented (Boadella 1978).
Reich’s ‘Armouring’
Reich (1949) outlined his understanding of the postures and defensive armouring produced by neurotic patients. He believed that such individuals often behaved as though they were ‘half-dead’ and that their normal functioning, on all levels, was diminished and restricted. He described an all too frequently seen pattern: ‘They were disturbed sexually, they were disturbed in their work function, their bodily processes lacked rhythm, their breathing was uncoordinated’ (Boadella 1978).
Reich and his followers demonstrated how emotions can ‘mobilize’ or ‘paralyse’ the body, with continued and repeated stress producing ‘blockages’ and restrictions which, if unreleased, become self-perpetuating and are themselves the source of pain and further stress. The ability to relax is lost, and the drain on nervous energy is profound in such situations. The bioenergetic answer to this problem is to aid in the release of these tensions by a complex set of exercises, including facial expressions and body positions, accompanied by breathing techniques.
Behavioural factors
All movement requires muscular activity. Certain patterns of use establish themselves. Often, individual awareness of the pattern of use is diminished, and habitual, repetitive actions leading to muscular hypertonicity developing (Feldenkrais 1977). Reforming habitual use patterns is far more difficult than altering the resultant soft tissue changes, in the short term. Breathing patterns that are habitual, such as hyperventilation, should be included in this category of stress factors impacting the musculoskeletal system (Chaitow et al 2002, Timmons 1994).
Structural factors
Over and above inborn features, such as a short leg, acquired structural changes make further demands on the adaptive capacity of the body. Depending upon the mechanical and structural loads it bears and responds to, muscle tissue will change in texture, chemistry, tone, etc., and will also modify and alter the framework of the body, warping and cramping its potential for normal use. It’s worth remembering that bone also remodels when loaded. Wolff’s law states: ‘bone in a healthy person or animal will adapt to the loads it is placed under’.
In other words the body will be bent and distorted to meet the stresses imposed from without and within. Barlow (1959), whose work follows that of Alexander (1957), suggested that there is a self-regulating tendency in the way muscles behave in response to stress.
Function in this example can be seen to remodel structure. This is the mirror opposite of the response of the body to structural modification – which leads inevitably to functional changes.
These ‘laws’ reflect a basic rule, usefully summarized by the acronym SAID – specific adaptation to imposed demand … and of course this is used as the basis for many rehabilitation programmes.
Mobilization before rehabilitation?
Rehabilitation of normal pain-free function, following soft tissue changes such as those described above – whether from behavioural, emotional or structural causes – requires an appropriate sequence, so that functionality can be restored.
In other words, before an individual can learn to breathe more normally, or stand upright, or walk more normally, there is a need to modify the soft tissue and joint changes that have occurred.
Dommerholt (2000), points out:
In general, assessment and treatment of individual muscles must precede restoration of normal posture and normal patterns of movement. Claims that muscle imbalances would dissolve, following lessons in the Alexander technique are not substantiated in the scientific literature (Rosenthal 1987). Instead muscle imbalances must be corrected through very specific strengthening and flexibility exercises, since generic exercise programs tend to perpetuate the compensatory muscle patterns. Myofascial trigger points must be inactivated using either invasive or non-invasive treatment techniques. Associated joint dysfunction, especially of the cervical and thoracic spine must be corrected with joint mobilizations. Once the musculoskeletal conditions for ‘good posture’ have been met, postural retraining [Alexander or other methods], can proceed.
Hypermobility
Clearly hypermobility tendencies may be inborn; however, acquired hypermobility may result from trauma, or over-zealous manipulation of particular joints (Protopapas & Cymet 2002).
Kappler (1997) cautions: ‘A normal physiological reaction to a painful hypermobile joint is for muscles surrounding the joint to splint the joint, and protect it from excess motion. Physical examination reveals restriction of motion. Underneath the protective muscle splinting is the unstable joint.’
It may be useful to reflect that one way in which the body might maintain excess tone in a muscle offering such protective support would be for it to evolve trigger point activity. These distressed supporting muscles (and their associated trigger points) might best be left untreated until underlying use patterns can be modified. Kappler (1997) suggests: ‘Management [of hypermobile structures] involves modifying patient activity that contributes to instability, mobilizing adjacent hypomobile joints, and prescribing active rehabilitation exercises.’
Understanding pain
Research into the mechanisms involved in muscular pain has evolved rapidly over the past 50 years.
Barlow’s (1959) suggestions that in the absence of other pathology muscular pain results from one of the following:
• The muscle itself, through noxious metabolic product, such as ‘factor P’ (Lewis 1942), or an interference in blood circulation due to spasm, resulting in relative ischaemia. Butler & Mosely (2003) note that when pain sensors (nociceptors), which are scattered throughout the body, are sufficiently stimulated they transmit messages towards the spinal cord that produce a release of excitatory chemicals into the synapse. When the synapse is sufficiently stimulated the pain message is sent to the brain.
• Acidic build-up within muscles (Issberner et al 1996) and inflammatory responses, involving what Butler & Mosely (2003) call ‘inflammatory soup’, add to the irritation of nociceptors – but of course are part of the internal repair system that allows healing to take place. See more on this topic below, under the sub-heading ‘Medical acupuncture model’.
• Muscular insertions into the periosteum, may be just such sites of nociceptor irritation, due to excessive or repetitive, muscular activity leading, for example, to ‘tennis elbow’ and periosteal pain points – as described in Chapter 3 (Lewit 1992).
• Joints, which can become restricted and over-approximated may also be sources of muscular pain. In advanced cases, osteoarthritic changes can result from the regular microtrauma of repeated muscular misuse. Over-approximation of joint surfaces, due to soft tissue shortening, can also lead to uneven wear and tear, as, for example, when the tensor fascia lata structure shortens and crowds both the hip and lateral knee joint structures.
• Neural irritation, which can be produced spinally or along the course of the nerve as a result of chronic muscular contractions may produce pain. These can involve disc and general spinal mechanical faults (Korr 1976), as well as altered neural status in which there is abnormal tension between nerves and the mechanical interface they have with their surrounding nerve sheaths (Butler & Gifford 1991, Maitland et al 2001).
• Variations in pain threshold, largely to do with perception, but also with gender, with fibromyalgia occurring in a female:male ratio of approximately 4:1 (Mense & Simons 2001) as well as ethnicity, modify the interpretation of pain messages in the brain – making them more or less significant (Wall & Melzack 1991). The brain processes pain messages and ascribes value to these based on whether danger is perceived, and this perception of danger has cultural and personal belief aspects (Butler & Mosely 2003).
• Gender differences relate largely to ‘a higher sensitivity of the pain system in females’.
Other models of pain genesis
Medical acupuncture model and trigger points
Baldry (1993) described the progression from muscle in a normal state, to muscle in painful chronic distress, as commonly involving initial or repetitive trauma (strain or excessive use), resulting in the release of chemical substances such as bradykinin, prostaglandins, histamine, serotonin and potassium ions. Sensitization of A-d and C (group IV) sensory nerve fibres may follow, with involvement of the brain (limbic system and the frontal lobe).
Trigger points (see Ch. 3), which evolve from such a progression, themselves become the source of new problems in their own locality, as well as at distant sites, as their sarcoplasmic reticulum is damaged and free calcium ions are released, leading to the formation of localized taut bands of tissue (involving the actin–myosin contractile mechanisms in the muscle sarcomeres).
If free calcium and energy-producing adenosine triphosphate (ATP) is present, this becomes a self-perpetuating feature, compounded by the relative (compared with surrounding tissues) ischaemia that has been identified in such chronically contracted tissues (Simons 1987). Local myofascial changes are considered in Chapter 3.
Much of this description by Baldry has been confirmed by research which assessed the chemicals in the tissues surrounding trigger points (Shah et al 2008a).
Shah & Gilliams (2008b) report:
Painful MTrPs activate muscle nociceptors that, upon sustained noxious stimulation, initiate motor and sensory changes in the peripheral and central nervous systems, [resulting in] sensitisation. In order to investigate the peripheral factors that influence the sensitisation process, a microdialysis technique was developed to quantitatively measure the biochemical milieu of skeletal muscle. Differences were found between active and latent MTrPs, as well as in comparison with healthy muscle tissue.
Note: Where pain has been produced by repetitive habits, postural and otherwise, with emotional and psychological overtones, the task of the therapist is complex because hypertonicity can often be only partially released or relaxed without resolving the underlying pattern of use. If repeated recurrence of painful episodes is to be minimized, a state of relative equilibrium of body structure and function is needed, which calls for treatment of structural restrictions as well as re-education regarding posture and use.
Stoddard’s osteopathic perspective
Stoddard (1969), in discussing contracted musculature, describes its ‘stringy’ feel, resulting from the continuous contraction of some muscle fibres, and ascribes the cause to the underlying joint dysfunction. The resulting ache and pain, he believes, is usually a result of circulatory embarrassment as metabolic wastes build up due to sustained muscular contraction. Muscular guarding is always seen to indicate deeper pathological changes (e.g. tuberculosis of the spine, osteomyelitis, disc herniation).
Stoddard sees the metabolic wastes, which may result from a degree of stasis, as causing a vicious cycle in perpetuating muscular contraction, leading eventually to fibrous changes. There is no indication that Stoddard considers such changes to be of primary importance in his treatment programme. He does stress the importance of exercises (to strengthen muscle groups) and of correct posture, but does not indicate any great interest in treatment of the soft tissues themselves.
While release of muscular restrictions and shortening could be considered a desirable step in the restoration of normality, it is worth emphasizing that once adaptive fibrotic changes have taken place in the soft tissues (whether in response to emotional stress or anything else) these changes are no longer under purely neurological control and therefore cannot simply be ‘released’ (by exercises or anything else): they require a physical input that alters, stretches and effectively breaks down concretions such as fibrotic tissue.
Modern pain concepts
The complexity of muscular pain mechanisms becomes clearer as the diligent research and reporting of Mense & Simons (2001) is examined. Interestingly, many of the ideas promoted half a century ago, by Barlow and others, are confirmed by modern research, although far more detail is now available with regard to pain mechanisms.
A brief summary of the key elements described by Mense and Simons is offered below; however, this captures no more than a superficial glimpse of the material in their book.
Common subgroups of muscular pain are identified:
• myofascial pain resulting from trigger points (often also used to describe regional pain syndromes)
• fibromyalgia (also used to describe myoglossis, muscular induration, non-articular rheumatism and tendomyopathy)
Mense and Simons consider that most other terms used to describe muscular pain can be subsumed into these three terms.
Sensitization
Sensitization of nociceptors by vasoneuroreactive substances (such as bradykinin and prostaglandins) leads to long-lasting tenderness of trigger points (Fig. 1.1). This is discussed further in Chapter 3.
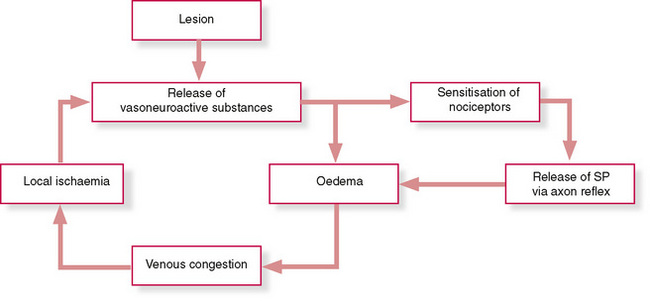
Figure 1.1 Schematic diagram showing hypothesized mechanism for sustained tenderness in trigger points. (SP = substance P)
Redrawn from Mense S & Simons D, Muscle pain, Williams & Wilkins, 2001, with permission.
The process of sensitization involves local oedema, release of neuropeptides such as substance P, subsequent compression of venous vessels, venous congestion (which reduces blood supply) and therefore local ischaemia. Ischaemic conditions lead to the release of more bradykinin, ensuring a vicious cycle, which engenders sensitization of pain receptors.
In skeletal muscle, ischaemia results in interference with normal energy (ATP) production, which leads to disturbance of normal calcium pump activity, preventing actin and myosin filaments from releasing their contractured state. This is the hypothesized cause of ‘taut bands’, which are a key feature of myofascial trigger points (see Ch. 3).
Mense & Simons (2001) suggest that when inflammation occurs in muscle for a period as short as 12 days, the concentration of thin nerve fibres containing neuropeptides increases markedly, leading to a greater reporting of pain sensations (Reinert & Mense 1993).
The widely held belief that a pain–spasm–pain cycle exists is questioned by Mense & Simons (2001). Their arguments are too complex to summarize without losing accuracy, but the following remarks offer a glimpse:
The [pain–spasm–pain] cycle is assumed to start with a muscle lesion that excites nociceptors. The small-diameter group III and IV muscle afferent fibers from the muscle nociceptors excite interneurons in the dorsal horn, which in turn activate α-motor neurons. Via their efferent fibers (α-motor axons) these motor neurons activate the neuromuscular endplate and cause spasm of the muscle. A longer-lasting spasm is painful and further activates muscle nociceptors.
Mense and Simons argue that although excitation of dorsal horn neurons by muscle nociceptors is established there is no proof that homonymous [alpha]-motor neurons are similarly activated: ‘An acute noxious stimulus to a muscle is likely to inhibit rather than excite homonymous motor neurons if the muscle is an extensor.’
They believe that because flexor muscle motor neurons display only short-lasting excitation (if any), and painful muscles frequently show little or no electrical activity when at rest, ‘the postulated [pain–spasm–pain] reflex is not functional in every muscle and cannot explain long-lasting spasm.’
Apart from the idea that spasm results via reflex arcs, as outlined above, a variety of other working hypotheses exists to explain the phenomenon of spasm, none of which is proven. These include the possibility that joint nociceptors act as initiators resulting in reflex stabilization via chronic muscle spasm.
Mense and Simons reject this, saying it cannot be universally valid, because ‘in many cases of painful joint lesions…neighbouring muscles are reflexly inhibited.’
Neuroplastic sensitization involves a process in which nociceptive input to the cord or brainstem leads to synaptic sensitization in the dorsal horn neurons. ‘Following such an input, the neurons are thought to increase their excitability and exhibit enhanced responses to both pathologic and normal afferent inflow.’
Central sensitization is then thought to perpetuate nociceptive activity.
This is a concept close to facilitation mechanisms, as hypothesized by osteopathic medicine, and discussed in Chapter 3.
There may be a malfunctioning of the descending antinociceptive system, in which central damping down of pain messages fails to be effective. Mense and Simons state: ‘It is conceivable that a malfunction of the descending antinociceptive system, which might occur spontaneously, or following a central or peripheral lesion, leads to chronic pain sensations from deep tissues, even in the absence of a peripheral lesion.’
Fibromyalgic pain may result from such a process.
Psychological factors modulate pain perception; however, controversially, Mense & Simons (2001) contend that whilst ‘psychological stress can be a potent aggravating factor … as we learn more about the pathophysiology concerning the origin of muscle pain, the less psychogenic and more somatic it becomes.’
A variety of mechanical (e.g. poor posture, asymmetry) and systemic (e.g. anaemia, low thyroid function, vitamin B deficiency) factors may interact to aggravate and perpetuate painful conditions.
Mense and Simons insist that it is essential to be aware of, and able to distinguish between, pain and tenderness that is local, projected (caused by peripheral nerve irritation), referred and of central origin – and of overlaps between these.
Additional insights from Mense and Simons will be introduced in later chapters.
At this stage it is useful to observe that pain is complex, confusing and demanding. The task of uncovering the mechanisms involved in any given patient’s painful muscular condition calls for the ability to evaluate muscular and articular status, and neurological features, as well as identification of myofascial trigger points and fibromyalgia characteristics.
One requirement for such a process is the ability to distinguish between different muscular responses to stress: overuse, misuse, disuse and abuse.
Different responses in postural and phasic muscles
It is not within the scope of this book to provide detailed physiological analysis; however, it is vital that the ways in which different muscle fibre types respond to stress are understood. To be sure there are several models in which different muscle groups are designated according to their main functions and characteristics, and an attempt is made in Box 1.5 to summarize these.
Box 1.5 Categorization of muscles
Postural and phasic muscles (sometimes called type I and type II)
Muscles have a mixture of fibre types, although there is usually a predominance of one type or another. There are those that contract slowly (‘slow twitch fibres’ or ‘slow white fibres’), which are classified as type I. These have very low stores of energy-supplying glycogen but carry high concentrations of myoglobulin and mitochondria. These fibres fatigue slowly and are mainly involved in postural and stabilizing tasks (Engel et al 1986, Woo et al 1987).
There are also several phasic/active type II fibre forms, notably:
• Type IIa fibres (‘fast twitch’ or ‘fast red’ fibres), which contract more speedily than type I and are moderately resistant to fatigue, with relatively high concentrations of mitochondria and myoglobulin.
• Type IIb fibres (‘fast twitch/glycolytic fibres’ or ‘fast white fibres’), which are less fatigue-resistant and depend more on glycolytic sources of energy, with low levels of mitochondria and myoglobulin.
• Type IIm (‘superfast’ fibres), found mainly in the jaw muscles, which depend on a unique myosin structure which, along with a high glycogen content, differentiates this from the other type II fibres (Rowlerson 1981).
The implications of the effects of prolonged stress on these different muscle types cannot be emphasized too strongly, because long-term stress involving type I muscles indicates that they will shorten, whereas type II fibres undergoing similar stress will weaken without shortening over their whole length (they may, however, develop shortened areas within the muscle). It is important to emphasize that shortness or tightness of a postural muscle does not imply strength. Such muscles may test as strong or weak. However, a weak phasic muscle will not shorten overall and will always test as weak (Janda 1982).
Fibre type is not totally fixed, in that evidence exists as to the potential for adaptability of muscles, so that committed muscle fibres can be transformed from slow-twitch to fast-twitch and vice versa (Lin et al 1994).
An example of this potential, which is of profound clinical significance, involves the scalene muscles, which Lewit (1999) confirms can be classified as either postural or phasic. If the largely phasic scalene muscles, which are dedicated to movement, have postural functions thrust upon them (as in an asthmatic condition in which they will attempt to maintain the upper ribs in elevation to enhance lung capacity) and if, owing to the laboured breathing of such an individual, they are thoroughly and regularly stressed, their fibre type will alter and they will shorten, becoming postural muscles (Lin et al 1994). A list of postural and phasic muscles is given later in this chapter.
Stabilizers and mobilizers
Norris (1995a, b, c, d, e, 1998) designates muscles according to their major functions, i.e. as ‘stabilizers’ or ‘mobilizers’. According to Norris, research has shown that muscles that are inhibited or weak may lengthen, adding to the instability of the region in which they operate. It is the ‘stabilizer’ muscles that have this tendency: if they are inhibited because of deconditioning they become unable adequately to perform the role of stabilizing joints in their ‘neutral posture’.
‘Stabilizer’ muscles, which are more deeply situated, slow twitch, and have a tendency to weaken and lengthen if deconditioned, include: transverse abdominis, multifidus, internal obliques, medial fibres of external oblique, quadratus lumborum, deep neck flexors, serratus anterior, lower trapezius, gluteus maximus and medius. These muscles can be correlated to a large extent (apart from quadratus lumborum) with muscles designated by Lewit (1999) and Janda (1982, 1983) as ‘phasic’.
The more superficial, fast-twitch muscles which have a tendency to shortening (i.e. ‘mobilizers’ in Norris’s terminology) include: suboccipital group, sternocleidomastoid, upper trapezius, levator scapulae, iliopsoas and hamstrings. These fall into the category of ‘postural’ muscles as described by Lewit (1992), Janda (1982) and Liebenson (1996).
Norris calls these ‘mobilizers’ because they cross more than one joint. This redefining of ‘postural’ as ‘mobilizer’ appears to be confusing, and many prefer to refer to these muscles simply as ‘having a tendency to shortening’ (Liebenson 1999).
Examples of patterns of imbalance that emerge as some muscles weaken and lengthen and their synergists become overworked, while their antagonists shorten, can be summarized as follows:
| Underactive stabilizer | Overactive synergist | Shortened antagonist |
|---|---|---|
| Gluteus medius | Tensor fascia lata, quadratus lumborum, piriformis | Thigh adductors |
| Gluteus maximus | Iliocostalis lumborum, hamstrings | Iliopsoas, rectus femoris |
| Transversus abdominis | Rectus abdominis | Iliocostalis lumborum |
| Lower trapezius | Levator scapulae, upper trapezius | Pectoralis major |
| Deep neck flexors | Sternocleidomastoid | Suboccipitals |
| Serratus anterior | Pectoralis major/minor | Rhomboids |
| Diaphragm | Scalenes, pectoralis major |
Multijoint or monoarticular muscles
Richardson et al (1999, 2000) have argued for the use of the terms multijoint muscles (also described as ‘global’ or ‘deep’ – slow twitch) and monoarticular muscles (also described as ‘local’ or ‘superficial’ – fast twitch). Richardson (2000) states:
For many years traditional exercise therapy was mainly focussed on building strength or endurance of whole muscle groups e.g. rotators of the trunk, extensors of the knee, internal rotators of the shoulder. Those involved in rehabilitative exercise gradually realized that people with injury not only needed general strength and endurance of whole muscle groups to perform an activity e.g. lifting a load, but also needed more specifically directed exercise.
More specifically exercise regimes were necessary to take into account that some individual muscles of a synergistic group:
(1) have distinct and different individual functions
(2) react in different ways to injury of the associated joint (reflex inhibition and excitation)
(3) react in different ways to lack of use or lack of gravitational load
(4) react in different ways to specific patterns of use (e.g. ballistic, repetitive activity)
In each of the above it can be easily predicted that individual muscles would fall consistently into basically two groups:
[Note: These are the ‘phasic’, stabilizer’, multijoint (deep, global) muscles.]
(1) more linked with joint stabilization
(2) more likely to undergo reflex inhibition with injury to associated joint
(3) more likely to atrophy quickly due to lack of use or lack of gravitational load
(4) more likely to decrease activity and change their function when exposed to ballistic repetitive exercise
[Note: These are the ‘postural’, ‘mobilizer’, monoarticular (superficial/local) muscles.]
(1) more linked with efficient movement of joints
(2) more likely to undergo reflex excitation with injury to associated joint
(3) not prone to atrophy quickly due to lack of use or lack of gravitational load
(4) more likely to become more active (and tighten) when exposed to ballistic repetitive exercise.
To minimize confusion, this book will follow the Janda/Lewit/Liebenson categorizations of postural and phasic muscles.
In this book the categorizations ‘postural’ and ‘phasic’ (Janda 1982, 1996, Lewit 1992, Liebenson 2006) are used, as these have been found by the principal author to be the most useful in clinical practice. What has been demonstrated is that, when stressed (overuse, misuse, abuse, disuse, etc.), postural muscles have a propensity to shorten, whereas phasic muscles undergoing similar ‘stress’ are inhibited, become weaker, and possibly lengthen (although localized contractures may be present).
Fetal position influences
Kolar (1999) reports that the muscles that tend to hypertonicity and shortening (i.e. ‘postural’ muscles) include most muscles shortened in the fetal position. These include finger, hand and wrist flexors; shoulder internal rotators and adductors; shoulder girdle elevators, as well as ankle plantar flexors and inverters; hip flexors, internal rotators and adductors.
The antagonists to these tend to become reciprocally inhibited (i.e. ‘phasic’ muscles). Janda (1996) suggests that these are the muscles whose neurodevelopment brings about the upright posture.
Role of the muscles in low back problems
If we examine the role of the muscular component of musculoskeletal structures, we find strong evidence for its involvement in many acute and chronic conditions.
Jokl (1984) tells us that disuse muscular atrophy, following back injury, is a major factor in the progression from an acute back problem to a chronic one. Changes take place that are observable, histologically and biochemically, in the muscle fibres, and that are translated into functional changes. The effects of these changes involve decreased endurance and weakened muscles, as well as spasm. We should remind ourselves of the basic anatomy of the low back, which includes the division of the musculature into:
1. The deep muscles, connecting the adjacent spinous processes (interspinales), adjacent transverse processes (intertransversari) and the rotatores, connecting the transverse process below to the laminae above.
2. The intermediate muscles include multifidus, which connects the transverse processes to the spinous processes of the vertebra above.
3. The superficial group includes iliocostalis, longissimus and spinalis (erector spinae). The origin of this is on the ischium, and the insertion on the sixth and 12th ribs. Together with the psoas (major and minor) and the quadratus, these greatly influence spinal stability.
4. The prevertebral muscles, which further stabilize and support the spine, are those that encircle the abdomen, such as the internal and external oblique, and rectus abdominus muscles.
Muscle types (see Box 1.5)
Any, or all, of these muscles can have a major influence on the onset of low back problems and pain. The division of muscles with regard to their different postural and phasic (volitional movement) types is worthy of re-emphasis. As mentioned earlier, muscle fibres may be differentiated into types by virtue of their role, as well as their main energy source.
• For example, type I muscles require stamina, rather than speed of action, and derive their energy via oxidative phosphorylation.
• This is in contrast to type II muscles, which produce power and speed, and derive energy from carbohydrate sources via glycolytic breakdown.
The muscles that support the spine are mainly type I, endurance and stamina muscles. Their activities are in the main related to static, antigravity efforts, which require prolonged contraction, and these muscles are far more susceptible to disuse atrophy and shortening. The strength of such muscles may not indicate much change, even after a period of disuse, but the endurance factor could be greatly affected. This necessitates a certain degree of caution when interpreting muscle strength tests involving the paravertebral musculature.
Jokl (1984) points out that electromyographic (EMG) studies indicate that paraspinal muscles show marked fatigue in individuals with low back pain. This fatigue factor may play a major part in worsening, or accentuating, an already demonstrable degree of dysfunction in this region. When such a situation exists (pain and easy fatigue of supporting musculature), it may be assumed that an increasing number of muscle fibres has been recruited in order to maintain spinal stability, which in turn results in increased muscular pressure. Jokl tells us that normal muscle can work for long periods without any EMG evidence of fatigue. As muscles become weaker, they work at an increased percentage of their maximum voluntary contraction. Ultimately this leads to muscle spasm, which allows ischaemia to develop, and pain to result.
The cycle of increased effort, local spasm and ischaemia, leading to pain, may ultimately result in paraspinal spasm and splinting.
Therapeutic choices
The use of both neuromuscular technique (NMT) and muscle energy technique (MET), are indicated in such a situation, as a means of disrupting the cycle and, initially, relaxing the contracted muscles. NMT methods have a combined effect, both relaxing the tissues as well as increasing the vascularity and mobility of these structures. They become more ‘extensible’, to use Grieve’s phrase (Grieve 1985). He enlarges on this aspect thus:
There is new evidence to support the view that suppleness and flexibility of muscle and connective tissue, are of prime importance. Long and continued occupational and postural stress, asymmetrically imposed upon the soft tissues, tends to cause fibroblasts to multiply more rapidly and produce more collagen. Besides occupying more space within the connective tissue element of the muscle, the extra fibres encroach on the space normally occupied by nerves and vessels. Because of this trespass, the tissue loses elasticity and may become painful when the muscle is required to do work in coordination with others. In the long term collagen would replace the active fibres of the muscle and since collagen is fairly resistant to enzyme breakdown these changes tend to be irreversible.
The explanations of Jokl, Korr, Patterson and Grieve, as discussed above, help us to gain a clearer picture of the structural changes that take place as stress factors, operating over a period of time, impact on the soft tissues.
Postural and phasic muscle lists
Type I postural muscles are prone to loss of endurance capabilities when disused or subject to pathological influences and become shortened or tighter, whereas type II phasic muscles, when abused or disused, become weak (Janda 1982, Lewit 1992, Liebenson 2006).
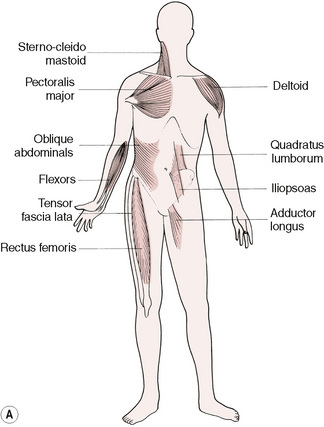
Postural muscles that become hypertonic and shorten in response to dysfunction include (Fig. 1.2):
• Trapezius (upper), sternocleidomastoid, levator scapulae and upper aspects of pectoralis major, in the upper trunk; and the flexors of the arms. Quadratus lumborum, erector spinae, oblique abdominals and iliopsoas, in the lower trunk. Tensor fascia lata, rectus femoris, biceps femoris, adductors (longus brevis and magnus) piriformis, hamstrings, semitendinosus.
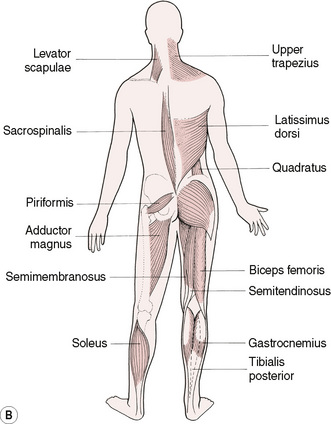
Phasic muscles, which weaken (i.e. are inhibited), and may lengthen, in response to dysfunction, include:
• The paravertebral muscles (not erector spinae) and scaleni, the extensors of the upper extremity (flexors are primarily postural), the abdominal aspects of pectoralis major; middle and inferior aspects of trapezius; the rhomboids, serratus anterior, rectus abdominus; the internal and external obliques, gluteals, the peroneal muscles and the extensors of the arms.
Muscle groups such as the scaleni are equivocal: they start out as phasic muscles but can end up as postural.
Note: Lewit (1999) does not subscribe to the theory that phasic and postural muscles can be differentiated by virtue of their fibre type, as do Grieve and Jokl, but certainly subscribes to their differences in all other regards.
Fibrositis
Having evolved from ‘muscular rheumatism’, via fibrositis, to the currently favoured term ‘fibromyalgia’, generalized muscular pain is a manifestation of multiple causative influences. The great British orthopaedic physician and writer James Cyriax (1962) believed, like Stoddard, that all primary ‘fibrositic’ conditions were a result of articular lesions (dysfunctions).
A secondary librotic change could result, he stated, from traumatic injury to soft tissues (e.g. capsular adhesion at the shoulder after injury) which he called fibrosis, not fibrositis. He saw fibrosis as scar tissue formation. Other secondary fibrositic conditions were said to result from rheumatoid disease, infection (such as epidemic myalgia) and parasitic infection (Trichinella spiralis). All other muscular and soft tissue dysfunctions, Cyriax regarded as one result of joint dysfunction, which then produced muscular protective spasm, muscular wasting, pain, etc., and which could be normalized only by correction of the joint lesion.
NMT theory and practice holds an almost precisely opposing view to that held by Cyriax – maintaining that appropriate normalization of the soft tissues can, more often than not, achieve joint normalization, when restriction exists, without active manipulation of the joint, since most joint problems appear to be the direct result of myofascial dysfunction (Cantu & Grodin 1992, Janda 1988, Stiles 1984).
Cyriax did point out that fibrous tissue is capable of maintaining inflammation, originally traumatic, almost as a matter of habit. In such cases he opted for hydrocortisone injections as the appropriate measure to ‘break’ this habit. Such treatment can work, but such an approach all too often fails if underlying fibrotic changes have not been normalized or habits of use modified.
Fibrositis changes its name to fibromyalgia
In Chapter 3, in which the phenomenon of local soft tissue dysfunction, most notably myofascial trigger point activity, is analysed in detail, the evolution of thinking regarding ‘fibrositis’ is also examined. Fibromyalgia (the new incarnation of fibrositis), with its causes, associated conditions and diagnostic criteria, requires discussion in that context because of the confusion that currently exists regarding the overlap between fibromyalgia syndrome (FMS), myofascial pain syndrome (MPS) and chronic fatigue syndrome (CFS). The fascia of the body has profound influence, and it is to this focus that we now turn.
The fascial network
Of major significance in understanding musculoskeletal function and dysfunction is the fact that the fascia comprises one connected network – from the fascia attached to the inner aspects of the skull to the fascia in the soles of the feet, there exists just one fascial structure.
If any part of this is deformed or distorted, negative stresses may be imposed on distant aspects, and on the structures that it divides, envelops, enmeshes and supports, and with which it connects. There is ample evidence that Wolff’s law applies, in that fascia accommodates to chronic stress patterns and deforms itself – something that often precedes deformity of osseous and cartilaginous structures in chronic diseases.
Note: Wolff’s law states that biological systems (including soft and hard tissues) deform in relation to the lines of force imposed on them.
The musculoskeletal system, the mechanical component of the human machine, comprising as it does 60% of the mass of the body, exists in a state of structural and functional continuity between all of its hard and soft tissues, and fascia is the ubiquitous elastic–plastic, gluey, component that invests, supports and separates, connects and divides, wraps and gives cohesion to the rest of the body – the fascial, connective tissue network. It is the connective tissues/fascia that provide most of that continuity. Any tendency to think of a local ‘lesion’ as existing in isolation should be discouraged as we try to visualize a complex, interrelated, symbiotically functioning assortment of tissues, comprising skin, muscles, ligaments, tendons and bones, as well as the neural structures, blood and lymph channels, and vessels that bisect and invest these tissues – all given shape, cohesion and functional ability by the fascia (connective tissue).
One of the major researchers into the role of connective tissue in the economy of the body is Donald Ingber. He has summarized aspects of this work, that are pertinent to manual therapists, by saying:
[The study of] mechanotransduction has revealed that molecules, cells, tissues, organs, and our entire bodies use “tensegrity” architecture to mechanically stabilize their shape, and to seamlessly integrate structure and function at all size scales. Through use of this tension-dependent building system, mechanical forces applied at the macroscale produce changes in biochemistry and gene expression within individual living cells. This structure-based system provides a mechanistic basis to explain how application of physical therapies might influence cell and tissue physiology (Ingber 2008).
Apart from its immense role in the support, structural organization and motion of the body, fascia is involved in numerous complex biochemical activities:
• Connective tissue provides a supporting matrix for more highly organized structures and attaches extensively to muscles, offering support and strength (Huijing & Baan 2007).
• Individual muscle fibres are enveloped by endomysium, which is connected to the stronger perimysium which surrounds the fasciculi. The perimysium’s fibres attach to the even stronger epimysium, which surrounds the muscle as a whole and attaches to fascial tissues nearby.
• Because it contains mesenchymal cells of an embryonic type, connective tissue provides a generalized tissue capable of giving rise, under certain circumstances, to more specialized elements.
• It provides, by its fascial planes, pathways for nerves, blood and lymphatic vessels, and structures.
• Many of the neural structures in fascia are sensory in nature.
• It supplies restraining mechanisms by the differentiation of retention bands, fibrous pulleys and check ligaments, as well as assisting in the harmonious production and control of movement.
• Where connective tissue is loose in texture, it allows movement between adjacent structures and, by the formation of bursal sacs, reduces the effects of pressure and friction.
• Deep fascia ensheathes and preserves the characteristic contour of the limbs, and promotes the circulation in the veins and lymphatic vessels.
• The superficial fascia, which forms the panniculus adiposus, allows for the storage of fat and also provides a surface covering which aids in the conservation of body heat.
• By virtue of its fibroblastic activity, connective tissue aids in the repair of injuries by the deposition of collagenous fibres (scar tissue).
• The ensheathing layer of deep fascia, as well as intermuscular septa and interosseous membranes, provides vast surface areas for muscular attachment.
• The meshes of loose connective tissue contain the ‘tissue fluid’ and provide an essential medium through which the cellular elements of other tissues are brought into functional relation with blood and lymph. Connective tissue has a nutritive function and houses nearly a quarter of all body fluids (Dicke 1978).
• The histiocytes of connective tissue comprise part of an important defence mechanism against bacterial invasion by their phagocytic activity. They also play a part as scavengers in removing cell debris and foreign material.
• Connective tissue represents an important ‘neutralizer’ or detoxicator to both endogenous toxins (those produced under physiological conditions) and exogenous toxins (those introduced from outside the organism). The mechanical barrier presented by connective tissue has important defensive functions in cases of infection and toxaemia.
• Recent research has shown evidence of the presence in connective tissue of contractile smooth muscle cells, indicating the potential for fascial tone to be actively modified (Schleip et al 2006).
Fascia, then, is not just a background structure with little function apart from its obvious supporting role, but is a ubiquitous, tenacious, connective tissue that is involved deeply in almost all of the fundamental processes of the body’s structure, function and metabolism.
In therapeutic terms there can be little logic in trying to consider muscle as a separate structure from fascia, because they are so intimately related. Remove connective tissue from the scene and any muscle left would be a jelly-like structure without form or functional ability.
See Box 1.6 for definitions relating to the characteristic features of connective tissue/fascia.
Box 1.6 Biomechanical terms relating to fascia
Creep
Continued deformation (increasing strain) of a viscoelastic material with time under constant load (traction, compression, twist). ‘Creep’ can occur during the process of myofascial release or in response to postural or functional patterns of use.
Hysteresis
Process of energy loss due to friction when tissues are loaded and unloaded, which may occur during treatment using stretching or other manual methods.
Stress
Force (load) normalized over the area on which it acts (all tissues exhibit stress–strain responses).
Thixotropy
A quality of colloids in which the more rapidly force is applied (load), the more rigid the tissue response, highlighting the need for sustained and slowly applied force when attempting to modify fascial status.
Functional fascial continuities
Myers (2001) has created a model in which myofascial linkages are described in terms of railway junctions and tracks, which he has termed ‘myofascial meridians’. While the interconnectedness of fascia throughout the body has long been understood, Myers’ descriptions highlight specific linkages. The myofascial chains, which he describes as ‘long functional continuities’, are of important clinical value when attempting to identify functional connections between anatomically distant structures, for example the plantar fascia and fascia of the scalp, attaching to the brow ridge, as described in his superficial back line (Fig. 1.3), or the anterior compartment of the periosteum of the tibia and the sternocleidomastoid attachment to the temporal bone, as described in his superficial front line (Fig. 1.4). These fascial bridges are also of potential clinical importance in application of the progressive inhibition of neuromuscular structures (PINS) method, described by Dennis Dowling in Chapter 11 of this book.
Soft tissue changes – energy and fascial considerations
Taylor (1958) has postulated that tissue changes, apparent to the trained palpating hand, often result from changes in thermodynamic equilibrium. He states that the body is a thermodynamic system and, as such, alterations in the extracellular fluids, viscosity, pH, electrophoretic changes, colloidal osmotic pressure, etc., are subject to thermodynamic laws. One of these laws states that the total energy of such a system and its surroundings must remain constant, although the energy may be changed from one form to another as a result of alterations of the stresses imposed.
For example, through postural stress and gravitational effects, particular changes in the fascia involved would result in energy loss and therefore stasis and stagnation. One of the characteristics of thermodynamics is that of thixotropy, in which gels become more solid with energy loss and more fluid with energy input. Such changes are certainly palpable in soft tissues, before and after neuromuscular and other forms of manual treatment (Mikova et al 2008).
• Taylor (1958) has stated that manipulative pressure and stretching are the most effective ways of modifying energy potentials of abnormal soft tissues.
• Little (1969) believes that an additional beneficial effect results from the interaction of the bioenergy mechanism of the practitioner with the bioenergy field of the patient.
Over and above the important factors of posture, functional ability and pain, the musculoskeletal component of the body plays a vital role in the conservation – wastage – of energy and in the attainment, or otherwise, of a truly relaxed mind. Whilst the origins of musculoskeletal dysfunction can be either psychic or physical, the constant interaction of the soma with the mind ensures a degree of psychic stress occurring as a feedback from physically caused chronic muscular tensions.
Rolf (1962) suggests that the human organism, as an energy mass, is subject to gravitational law. As a plastic medium, capable of change, Rolfing attempts to reorganize and balance the body in relation to gravitational forces. This is done by using pressure and stretch techniques on the fascial tissues in a precise sequence of body areas. The beneficial effects are claimed to be physical, emotional, postural and behavioural.
Rolf (1962) states:
Our ignorance of the role and significance of fascia is profound. Therefore even in theory it is easy to overlook the possibility that far-reaching changes may be made not only in structural contour, but also in functional manifestation, through better organisation of the layer of superficial fascia which enwraps the body. Experiments demonstrate that drastic (beneficial) changes may be made in the body, solely by stretching, separating and relaxing superficial fascia in an appropriate manner.
Osteopaths have observed and recorded the extent to which all degenerative change in the body – whether muscular, nervous, circulatory or organic – reflects in the superficial fascia. Any degree of degeneration, however minor, changes the bulk of the fascia, modifies its thickness and draws it into ridges in areas overlying deeper tensions and rigidities. Conversely, as this elastic envelope is stretched, manipulative mechanical energy is added to it, and the fascial colloid becomes more ‘sol’ and less ‘gel’ (Greenman 1989). Without minimizing the clinical importance of the structural and biomechanical aspects of the connective tissues, Oschman (2000) has examined and evaluated research, which suggests that far more may be taking place during manual treatment than current understanding can fully explain. See Box 1.7 for a brief summary of aspects of these concepts.
Box 1.7 Oschman’s energy concepts: connective tissue, crystallinity and continuity
Peters (2001) discusses Oschman’s research into energy phenomena as this relates to health enhancement. He reports that Oschman (2000) makes the point that the intricate traffic of biochemical, structural and electrical information is modulated not only through an electromagnetic flux, and ‘clouds’ of neuroreceptor traffic, but also as mechanical and rhythmic impulses of sound, heat, gravity, elasticity and pressure.
Oschman (2000) describes the processes:
What we refer to as health is when all the systems, both known and unknown, are functioning collectively, co-operatively … The solid state, electronic, photonic and vibratory properties, of this living matrix continuum, play key roles in the integration of function, including injury repair and defence against disease. A debate about whether there is such a thing as ‘healing energy’ or ‘life energy’ has been replaced with study of the interaction between biological energy fields, structures and functions … There are now instruments sensitive enough to detect the bio-magnetic fields produced by the different organs … photometers and thermographs of parallel sensitivity allow us to detect almost infinitesimal variations in light and heat emanating from the body. Previous images of the organism – as being built up of parts – have concealed the most significant attribute of living matter – its continuity.
Oschman (2000) goes on:
The major structural and functional domains of the body are the connective tissues, the cells within them, and the cytoskeletons, nuclei and genetic material within the cells … [this] assembly is best described by a single word, continuum … Structural and functional continuity has now been confirmed and appreciated by science.
Continuity in living systems is simultaneously mechanical, structural, regulatory, and energetic. A second key to the emerging concepts has come about from recognition of the crystalline properties of living tissues … Crystals have important vibratory characteristics that arise as collective properties of the whole system. When a crystal is broken into its constituents, these unique vibratory phenomena disappear. This is why collective properties such as functional organization and consciousness have been elusive for those who study the system’s components piece-by-piece. Molecular arrays or crystals are the dominant structural feature of living matter. Crystallographic techniques such as X-ray diffraction have been essential for determining the structure of nerves, muscles, cell membranes, and connective tissues. From the biophysical perspective, molecular arrays or crystals cannot be described in terms of their constituents alone. (See Fig. 1.5.)
British biophysicist Mae-Wan Ho has developed an elegant quantum theory which describes the organism as a vibrant sentient whole (Ho 1993). Key to Ho’s theory (Ho 1997) is the role of the connective tissue as a liquid crystalline material constituting a noiseless excitable vibratory continuum for rapid intercommunication and energy flow, permeating the entire organism and enabling it to function and perceive as a coherent whole.
Oschman (2000) attempts to describe a unitary concept:
The quantum coherence phenomenon, described by biophysicists, may be the origin of Sheldrake’s morphogenetic field (Sheldrake 1995). Quantum coherence is a source of measurable light emissions from living systems (Popp et al 1992). Quantum coherence in the living matrix provides a basis for a unitary theory. Water and vibrations of the crystalline molecular lattices play key roles in energy and information storage, transfer, and release.
The conclusion reached by Oschman (2000) is that, almost irrespective of the mode of treatment, a primary interaction occurs between the therapist and the patient, which may involve communication between their energy fields.
Figure 1.5 Crystalline arrays in living systems. Crystalline arrangements are the rule and not the exception in living systems. (A) Arrays of phospholipid molecules from cell membranes. (B) Collagen arrays form connective tissue. (C) Arrays of chlorophyll molecules in the leaf. (D) The myelin sheath of nerves. (E) The contractile array in muscle is composed of actin and myosin molecules organized around one another. (F) The array of sensory endings in the retina. (G) Arrays of microtubules, microfilaments and other fibrous components of the cytoskeleton occur in nerves and other kinds of cells. The example shown here is the cilia of sensory organs such as those responsible for detecting odours and sound.
Reproduced from Oschman (2000) (illustration (D) is reproduced with permission of Arnold).
Fascial stress responses and therapeutic opportunities
Changes in the fascia can result from passive congestion, which results in fibrous infiltration and a more ‘sol’-like consistency than is the norm. Under healthy conditions a ‘gel’-like ground substance follows the laws of fluid mechanics. Clearly, the more resistive drag there is in a colloidal substance, the greater will be the difficulty in normalizing this.
Scariati (1991) points out that colloids are not rigid: they conform to the shape of their container, and respond to pressure even though they are not compressible. The amount of resistance they offer increases proportionally to the velocity of motion applied to them, which makes a gentle touch a fundamental requirement if viscous drag and resistance are to be avoided when attempting to produce a release.
When stressful forces (undesirable or therapeutic) are applied to fascia, there is a first reaction in which a degree of slack is allowed to be taken up, followed by what is colloquially referred to as ‘creep’ – a variable degree of resistance (depending on the state of the tissues).
Creep is an honest term that accurately describes the slow, delayed yet continuous, stretch that occurs in response to a continuously applied load, as long as this is gentle enough to not provoke the resistance of colloidal ‘drag’.
As the fascia comprises a single structure, the implications for body-wide repercussions of distortions in that structure are clear (Fig. 1.6). An example of one possible negative influence of this sort is to be found in the fascial divisions within the cranium, the tentorium cerebelli and falx cerebri, which are commonly warped during birthing difficulties (too long or too short a time in the birth canal, forceps delivery, etc.) and which are noted in craniosacral therapy as affecting total body mechanics via their influence on fascia (and therefore the musculature) throughout the body (Brookes 1984).
Figure 1.6 Balanced posture (A) compared with two patterns of musculoskeletal imbalance that involve fascial, and general, tissue and joint adaptations.
Cantu & Grodin (1992) describe what they see as the ‘unique’ feature of connective tissue as its ‘deformation characteristics’. This refers to a combined viscous (permanent) deformation characteristic as well as an elastic (temporary) deformation characteristic. This leads to the clinically important manner in which connective tissue responds to applied mechanical force by first changing in length, followed by some of this change being lost while some remains. The implications of this phenomenon can be seen in the application of stretching techniques to such tissues, as well as in the way they respond to postural and other repetitive insults.
Such changes are not, however, permanent, because collagen (the raw material of fascia/connective tissue) has a limited (300–500 days) half-life and, just as bone adapts to stresses imposed upon it, so does fascia. Therefore, if negative stresses (posture, use, etc.) are modified for the better and/or positive ‘stresses’ are imposed (e.g. manipulation and/or exercise), dysfunctional connective tissue can usually be improved over time (Neuberger et al 1953).
Cantu & Grodin (1992), in their evaluation of the myofascial complex, concluded that therapeutic approaches that sequence their treatment protocols to involve the superficial tissues (involving autonomic responses) as well as deeper tissues (influencing the mechanical components of the musculoskeletal system), and which also address the factor of mobility (movement), are in tune with the requirements of the body when dysfunctional. NMT, as it will be presented here, does take this comprehensive approach, and much that it offers seems to be similar to the myofascial release methods currently receiving so much attention in the USA.
Cathie (1974) maintains that the contractile phase of fascial activity supersedes all its other qualities. The attachments of fascia, he states, have a tendency to shorten after periods of marked activity that are followed by periods of inactivity, and the ligaments become tighter and thicker with advancing age. The properties of fascia (connective tissue) that he regards as being important to therapeutic consideration are:
1. It is richly endowed with nerve endings.
2. It has the ability to contract and to stretch elastically.
3. It gives extensive muscular attachment.
4. It supports and stabilizes, thus enhancing the postural balance of the body.
5. It is vitally involved in all aspects of motion.
6. It aids in circulatory economy, especially of venous and lymphatic fluids.
7. Fascial change will precede many chronic degenerative diseases.
8. Fascial changes predispose towards chronic tissue congestion.
9. Such chronic passive congestion precedes the formation of fibrous tissue, which then proceeds to an increase in hydrogen ion concentration of articular and periarticular structures.
10. Fascial specializations produce definite stress bands.
11. Sudden stress (trauma) on fascial tissue will often result in a burning type of pain.
12. Fascia is a major arena of inflammatory processes.
13. Fluids and infectious processes often travel along fascial planes.
14. The CNS is surrounded by fascial tissue (dura mater) that, in the skull, attaches to bone, so that dysfunction in these tissues can have profound and widespread effects.
Greenman (1989) describes how fascia responds to loads and stress in both a plastic and an elastic manner, its response depending upon the type, duration and amount of the load. The responses to either acute injury or repetitive microtrauma (e.g. resulting from a short leg imbalance) are, according to Greenman, likely to follow a sequence of inflammation which subsequently leads to absorption of inflammatory fluids into the superficial fascia, as well as into tight compartmentalized areas in the deep fascia – with this latter event being both palpable and detrimental.
Another variable (apart from the nature of the stress load) that influences the way in which fascia responds to stress, and what the individual feels of the process, relates to the number of collagen and elastic fibres contained in any given region.
Neural receptors within the fascia report to the CNS as part of any adaptation process, with the Pacinian corpuscles being particularly important in terms of their involvement in reflex responses.
Ligaments and muscle tone
Other neural input into the pool of activity, and responses to biomechanical stress, involve specialized fascial structures such as tendons and ligaments that contain highly specialized and sensitive mechanoreceptors and proprioceptive reporting stations.
Solomonow (2009) summarizes the role of ligaments as follows:
As joints go through their range of motion, with or without external load, the ligaments ensure that the bones associated with the joint travel in their prescribed anatomical tracks, keep full and even contact pressure of the articular surfaces, prevent separation of the bones from each other by increasing their tension, as may be necessary, and ensuring stable motion. Joint stability, therefore, is the general role of ligaments without which the joint may subluxate, cause damage to the capsule, cartilage, tendons, nearby nerves and blood vessels, discs (if considering spinal joints) and to the ligaments themselves. Such injury may debilitate the individual by preventing or limiting his/her use of the joint and the loss of function. Unstable joints are also known to drastically modify the intra-articular pressure and the muscular activity pattern about the joint, resulting in early onset of osteoarthritis, pain, disability and eventually the need for joint replacement surgery.
Solomonow adds some important information regarding the link between muscle behaviour and ligamentous function:
Ligaments are also sensory organs and have significant input to sensation and reflexive/synergistic activation of muscles. The muscles associated with any given joint, therefore, also have a significant role as restraints.
For example muscular activity associated with the reflex from the ACL acts to prevent distraction of the joint, while simultaneously reducing the strain in the ACL. Evidence also supports the possibility that ligamento-muscular reflexes may also have inhibitory effects on muscles associated with that joint – inhibiting muscles that destabilize the joint or increasing antagonist co-activation to stabilize the joint.
A strong clue relative to the potential therapeutic use of ligament behaviour, in for example positional release methodology (see Ch. 8), can be gained from Solomonow’s (2009) observation that:
Ligaments are functional (effective) under tension, or when stretched, and completely non-functional in compression or when shortened below their resting length.
Having briefly scanned the influences of stress, both short and long term, on the musculoskeletal system, and of the varying ways in which the soft tissues respond – acutely, chronically and with variations based on their structure – and before looking more closely at reflex phenomena a brief introduction to neuromuscular technique (see Ch. 2) will provide an indication of its potential for dealing with soft tissue distress.
Alexander F.M. The use of the self. London: Educational Publications, 1957.
Alshami C.M., Hodges A. An experimental pain model to investigate the specificity of the neurodynamic test for the median nerve in the differential diagnosis of hand symptoms. Arch Phys Med Rehabil. 2006;87(10):1412-1417.
Baldry P. Acupuncture, trigger points and musculoskeletal pain. Edinburgh: Churchill Livingstone, 1993.
Barlow W. Anxiety and muscle tension pain. Br J Clin Prac. 1959;3(5):339-350.
Basmajian J. Muscles alive. Baltimore: Williams & Wilkins, 1974.
Bendtsen L., Jensen R., Olesen J. Qualitatively altered nociception in chronic myofascial pain. Pain. 1996;65:259-264.
Boadella D. The language of the body in bioenergetic therapy. Journal of the Research Society for Natural Therapeutics. 1978.
Brookes D. Cranial osteopathy. London: Thorsons, 1984.
Butler D. Mobilisation of the nervous system. Edinburgh: Churchill Livingstone, 1991.
Butler D., Gifford L. Adverse mechanical tensions in the nervous system. Physiotherapy. 1989;75:622-629.
Buzzell K. The physiological basis of osteopathic medicine. New York: Postgraduate Institute of Osteopathic Medicine and Surgery, 1970.
Canals J., Pineda J., Torres-Peraza J., et al. Brain-derived neurotrophic factor regulates the onset and severity of motor dysfunction associated with enkephalinergic neuronal degeneration in Huntington’s disease. J Neurosci. 2004;24:7727-7739.
Cantu R., Grodin A. Myofascial manipulation. Gaithersburg, Maryland: Aspen Publications, 1992.
Cathie A. Selected writings. Academy of Applied Osteopathy Yearbook 1974. Colorado Springs: Academy of Applied Osteopathy, 1974.
Chaitow L. Soft tissue manipulation. London: Thorsons, 1989.
Chaitow L., DeLany J. Clinical applications of neuromuscular technique. Edinburgh: Churchill Livingstone, 2000.
Chaitow L., Bradley D., Gilbert C. Interdisciplinary approaches to breathing pattern disorders. Edinburgh: Churchill Livingstone, 2002.
Cyriax J. Textbook of orthopaedic medicine. London: Cassell, 1962.
Dicke E. A manual of reflexive therapy. Scarsdale, New York: Sydney Simons, 1978.
DiGiovanna E. An osteopathic approach to diagnosis and treatment. London: Lippincott, 1991.
Dommerholt J. Posture. In: Tubiana R., Camadio P., editors. Medical problems of the instrumentalist musician. London: Martin Dunitz, 2000.
Dvorak J., Dvorak V. Manual medicine – diagnostics. Stuttgart: Georg Thiem, 1984.
Edwards R. Hypotheses of peripheral and central mechanisms underlying occupational muscle pain and injury. Eur J Appl Physiol. 1988;57:275-281.
Feldenkrais M. Awareness through movement. New York: Harper & Row, 1977.
Frankel V. Basic biomechanics of the skeletal system. Philadelphia: Lea & Febiger, 1980.
Gagey P.M. Postural disorders among workers in building sites. In: Bles W., Brandt T., editors. Disorders of posture and gait. New York: Elsevier, 1986.
Gofton J., Trueman G. Studies in osteoarthritis of the hip: Part II. Osteoarthritis of the hip and leg-length disparity. CMA Journal. 1971;104:791-799.
Gray H. Gray’s anatomy, ed 35. Edinburgh: Churchill Livingstone, 1977.
Greenman P. Principles of manual medicine. Baltimore: Williams & Wilkins, 1989.
Grieve G. Mobilisation of the spine. Edinburgh: Churchill Livingstone, 1985.
Hallgren R., Greenman P., Rechtien J. MRI of normal and atrophic muscles of the upper cervical spine. J Clin Eng. 1993;18(5):433-439.
Henneman E. Relation between size of neurons and their susceptibility to discharge. Science. 1957;126:1345-1347.
Ho M.W. The rainbow and the worm. The physics of organisms. Singapore: World Scientific, 1993.
Ho M.W. Quantum coherence and conscious experience. Kybernetes. 1997;26:265-276.
Horwitz A. Integrins and health. Sci Am. 1997;276:68-75.
Huijing P., Baan. Epimuscular myofascial force transmission between antagonistic and synergistic muscles can explain movement limitation in spastic paresis. J Electromyogr Kinesiol. 2007(6):708-724.
Hynes R. Integrins: versatility, modulation and signalling. Cell. 1992;69:11-26.
Ingber D. Tensegrity and mechanotransduction. Jnl Bodywork and Movement Therapies. 2008;12(3):198-200.
Issberner U., Reeh P., Steen K. Pain due to tissue acidosis: a mechanism for inflammatory and ischemic myalgia? Neurosci Lett. 1996;208(3):191-194.
Janda V. Introduction to functional pathology of the motor system. Proceedings of the VIIth Commonwealth and International Conference on Sport. Physiotherapy in Sport. 1982;3:39.
Janda V. Muscle function testing. London: Butterworths, 1983.
Janda V. Grant R., editor. Physical therapy of the cervical and thoracic spine. Edinburgh: Churchill Livingstone, 1988.
Janda V. Evaluation of muscle imbalances. In: Liebenson C., editor. Rehabilitation of the spine. Baltimore: Williams & Wilkins, 1996.
Jokl P. Muscle and low back pain. J Am Osteopath Assoc. 1984;84(1):64-65.
Jones L. Strain and counterstrain. Colorado Springs: Academy of Applied Osteopathy, 1980.
Kappler R. Thrust techniques. In: Ward R., editor. Foundations for Osteopathic Medicine. Baltimore: Williams & Wilkins, 1997.
Key J., et al. A model of movement dysfunction provides a classification system guiding diagnosis and therapeutic care in spinal pain and related musculoskeletal syndromes: a paradigm shift. Part 2. Journal of Bodywork & Movement Therapies. 2007;12(2):105-120.
Kolar P. Sensomotor nature of postural functions. J Orthop Med. 1999;212:40-45.
Korr I. The physiological basis of osteopathic medicine. New York: Postgraduate Institute of Osteopathic Medicine, 1970.
Korr I. Spinal cord as organiser of disease process. Colorado Springs: Academy of Applied Osteopathy Yearbook, 1976.
Korr I. Neurologic mechanisms in manipulative therapy. New York: Plenum Press, 1978.
Korr I. Axonal transport and neurotrophic functions in spinal cord as organiser of disease processes (part 4). Academy of Applied Osteopathy. 1981:451-458.
Kuchera M. Osteopathic Manipulative Medicine Considerations in Patients With Chronic Pain. J Am Osteopath Assoc. 2005;4(105):29-36.
Larsson S.E., Bodegard L., Henriksson K.G., et al. Chronic trapezius myalgia. Morphology and blood flow studied in 17 patients. Acta Orthop Scand. 1990;61:394-398.
Latey P. Muscular manifesto. London: P Latey, 1983.
Lederman E. Fundamentals of manual therapy. Edinburgh: Churchill Livingstone, 1997.
Lewis Sir T. Pain. New York: Macmillan, 1942.
Lewit K. Manipulation in rehabilitation of the locomotor system, ed 2. London: Butterworths, 1992.
Lewit K. Manipulation in rehabilitation of the locomotor system, ed 3. London: Butterworths, 1999.
Licciardone, et al. Osteopathic manipulative treatment for low back pain: a systematic review and meta-analysis of randomized controlled trials. BMC Musculoskelet Disord. 2005;6:43.
Liebenson C., editor. Rehabilitation of the spine. Baltimore: Williams & Wilkins, 1996.
Liebenson C. Rehabilitation of the spine, ed 2. Baltimore: Williams & Wilkins, 2006.
Liebenson C. Muscular imbalance – an update. Dynamic Chiropractic. 1999. Online. Available: http://www.chiroweb.com/dynamic
Lin J.P., Brown J.K., Walsh E.G. Physiological maturation of muscles in childhood. Lancet. 1994;343:1386-1389.
Little K.E. Toward more effective manipulative management of chronic myofascial strain and stress syndromes. J Am Osteopath Assoc. 1969;68:675-685.
McConnell C. Osteopathic Institute of Applied Technique Yearbook 1962. 1962. London
McPartland J., Brodeur R. Rectus capitis posterior minor. Journal of Bodywork and Movement Therapies. 1999;3(1):30-35.
Maitland G. Vertebral manipulation. London: Butterworths, 1986.
Maitland G., Hengeveld E., Banks K., et al. Maitland’s vertebral manipulation, ed 6. London: Butterworth Heinemann, 2001.
Martin S. Interview with Jeff Bland: lessons from the genome. CAM Magazine. 2001. November
Mense S., Simons D. Muscle pain. Philadelphia: Williams & Wilkins, 2001.
Miková M., Krobot A., Janura M., et al. Viscoelastic properties of fibrous connective tissue and manual treatment. Rehabilitace a Fyzikalni Lekarstvi. 2008;15(1):3-10.
Mooar P. Introduction. Merck Manual. 2007. http://www.merck.com/mmhe/sec05/ch058/ch058a.html. Retrieved 2008-11-12
Myers T. Anatomy trains. Edinburgh: Churchill Livingstone, 2001.
Neuberger A., et al. Metabolism of collagen. Biochem. J.. 1953;53:47-52.
Norris C.M. Spinal stabilisation. 1. Active lumbar stabilisation – concepts. Physiotherapy. 1995;81(2):61-64.
Norris C.M. Spinal stabilisation. 2. Limiting factors to end-range motion in the lumbar spine. Physiotherapy. 1995;81(2):64-72.
Norris C.M. Spinal stabilisation. 3. Stabilisation mechanisms of the lumbar spine. Physiotherapy. 1995;81(2):72-79.
Norris C.M. Spinal stabilisation. 4. Muscle imbalance and the low back. Physiotherapy. 1995;81(3):127-138.
Norris C.M. Spinal stabilisation. 5. An exercise program to enhance lumbar stabilisation. Physiotherapy. 1995;81(3):138-146.
Norris C.M. Sports injuries, diagnosis and management, ed 2. London: Butterworths, 1998.
Oschman J. Energy medicine. Edinburgh: Churchill Livingstone, 2000.
Patterson M., Wurster R. Neurophysiologic system: integration and disintegration. In: Ward R., editor. Foundations for osteopathic medicine. Baltimore: Williams & Wilkins, 1997.
Peck D., Buxton D., Nitz A. A comparison of spindle concentrations in large and small muscles acting in parallel combinations. J Morphol. 1984;180:243-252.
Peters D. Vitalism revisited. Journal of Bodywork and Movement Therapies. 2001;5(3):198-204.
Popp F.A., Li K.H., Gu Q. Recent advances in biophoton research. Singapore: World Scientific, 1992.
Protopapas M., Cymet T. Joint cracking and popping: Understanding noises that accompany articular release. J Am Osteopath Assoc. 2002;102(5):283-287.
Reich W. Character analysis. Vision Press, 1949.
Reinert A., Mense S. Inflammatory influence on the density of CGRP- and SP-immunoreactive nerve endings in rat skeletal muscle. Neuropeptides. 1993;24:204-205.
Richardson C. The muscle designation debate. Journal of Bodywork and Movement Therapies. 2000;4(4):235-236.
Richardson C., Jull G., Hodges P., et al. Therapeutic exercise for spinal segment stabilisation in low back pain. Edinburgh: Churchill Livingstone, 1999.
Rolf I. Structural dynamics. British Academy of Applied Osteopathy Yearbook 1962. New York: BAAO, 1962.
Rosenthal E. The Alexander technique and how it works. Medical Problems in the Performing Arts. 1987;2:53-57.
Rowlerson A. A novel myosin. J Muscle Res Cell Motil. 1981;2:415-438.
Scariati P. DiGiovanna E., editor. An osteopathic approach to diagnosis and treatment. London: Lippincott, 1991.
Schamberger W. The malalignment syndrome. Edinburgh: Churchill Livingstone, 2002.
Schleip R., Naylor I., Ursu D., et al. Passive muscle stiffness may be influenced by active contractility of intramuscular connective tissue. Med Hypotheses. 2006;66(1):71.
Selye H. The stress of life. Philadelphia: McGraw Hill, 1976.
Shah J., Danoff J., Desai M., et al. Biochemicals associated with pain and inflammation are elevated in sites near to and remote from active myofascial trigger points. Arch Phys Med Rehabil. 2008;89:16-23.
Shah J., Gilliams E. Uncovering the biochemical milieu of myofascial trigger points using in vivo microdialysis: An application of muscle pain concepts to myofascial pain syndrome. Journal of Bodywork and Movement Therapies. 2008;12(4):371-384.
Sheldrake R. A new science of life, The hypothesis of morphic resonance. Rochester, Vermont: Park Street Press, 1995.
Simons D. Myofascial pain due to trigger points. International Rehabilitation Medicine Association Monograph (Series 1). Ohio: Rademaker, 1987.
Simons D., Travell J., Simons L. Myofascial pain and dysfunction: the trigger point manual. vol. 1: Upper half of body, ed 2. Baltimore: Williams & Wilkins, 1999.
Solomonow M. Ligaments: A source of musculoskeletal disorders. Journal Bodywork and Movement Therapies. 2009;13(2):136-154.
Stanton D.F., Dutes J.C. Chronic pain and the chronic pain syndrome: the usefulness of manipulation and behavioural interventions. Phys Med Rehabil Clin North Am. 1996;7:863-875.
Stiles E. Patient Care. 1984. 15 May:16–97 & 15 August:117–164
Stoddard A. Manual of osteopathic practice. London: Hutchinson, 1969.
Taylor R.B. Bioenergetics of man. Academy of Applied Osteopathic Yearbook 1958. California: Carmel, 1958.
Timmons B. Behavioral and psychological approaches to breathing disorders. New York: Plenum Press, 1994.
Travell J., Simons D. Myofascial pain and dysfunction: the trigger point manual. vol. 1. Philadelphia: Williams & Wilkins; 1983.
Travell J., Simons D. Myofascial pain and dysfunction: the trigger point manual. vol. 2. Philadelphia: Williams & Wilkins; 1992.
Vleeming A., Mooney V., Stoeckart R., editors. Movement stability & lumbopelvic pain - Integration of research and therapy, ed 2, Edinburgh: Churchill Livingstone, 2007.
Waersted M., Eken T., Westgaard R. Psychogenic motor unit activity – a possible muscle injury mechanism studied in a healthy subject. Journal of Musculoskeletal Pain. 1993;1(3/4):185-190.
Wall P., Melzack R. Textbook of pain. Edinburgh: Churchill Livingstone, 1991.
Wang N., Butler J.P., Ingbes D.E., et al. Mechanotransduction across the cell surface and through the cytoskeleton. Science. 1993;260:1124-1127.
Wolfe F., Simons D. Fibromyalgia and myofascial pain syndromes. J Rheumatol. 1992;19(6):944-995.
Woo SL-Y., et al. Injury and repair of musculoskeletal soft tissues. Savannah, Georgia: American Academy of Orthopedic Surgeons Symposium, 1987.