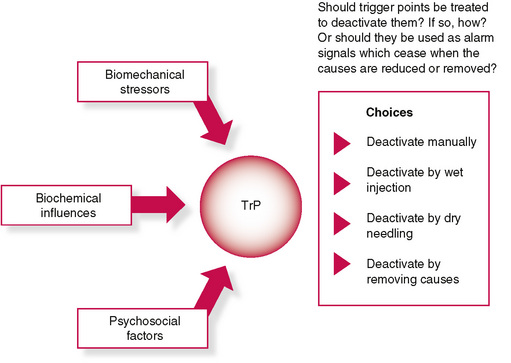
3 Myofascial trigger points and other reflex phenomena
Pain patterns
The radiating and/or referred symptoms deriving from myofascial trigger points are a major cause of sustained pain and dysfunction, according to Wall & Melzack (1989), the leading researchers into pain. Trigger points are indeed stated by them to be a part of all chronic pain conditions, often the major part. To be sure there are many other descriptions and classifications of reflex pain patterns – many of which are outlined in the following chapters – and these demand a brief review of the possible mechanisms involved.
A priority involves the need to differentiate between pain and referred symptoms that are of a spinal, nerve root, origin and those that have different aetiologies.
Difference between referred phenomena and radicular pain
Pain and other root syndrome effects, deriving from damaged or dysfunctional vertebral or intervertebral structures, need to be differentiated from the non-radicular pain and symptoms that derive from reflexogenic activity, such as myofascial trigger points.
The key characteristics of radicular dysfunction from, for example, a herniated disc, will include (Lavelle et al 2008, Fairbank 2008, Dvorak & Dvorak 1984):
• pain located in the regions supplied by the nerve roots from the segment(s) involved
• a loss of sensitivity in the appropriate dermatomes related to the segment(s) involved
• a loss of motor power – possibly to the point of paralysis – of the muscle innervated by the nerve roots involved, and possible atrophy
• disturbances of the deep tendon reflexes in the related areas. Diagnosis of the presence of such dysfunction requires expert neurological assessment; however, practitioners and therapists should be aware of these key signs, which alert them to the possibility that nerve root syndromes may be a feature of the patient’s problem.
Historical perspectives on non-radicular patterns of referred pain
Before looking at current explanations for the evolution of chronic pain, it may be useful to look at the perspectives offered by clinicians and researchers during the middle and later 20th century. These insights are valuable in understanding where modalities such as neuromuscular technique fit into the therapetic spectrum.
Five variations
Dvorak & Dvorak (1984) have described five variations on the theme of referred symptomatology, different ways of seeing the same phenomena, aside from nerve root syndromes. These are:
1. Referred pain – which includes the findings described by researchers such as Lewis (1938), Kellgren (1938), Hockaday & Whitty (1967), who demonstrated that mechanical and chemical stimulation to various spinal structures produces referred pain. This is discussed further in this chapter.
2. Myofascial trigger points, which are discussed extensively in this chapter, and which form the major focus of neuromuscular technique application in North America.
3. Pseudoradicular syndromes, which Brugger (1962) describes as being quite distinct from root syndromes and which derive from a ‘nociceptive somatomotoric blocking effect’ occurring in tissues such as joint capsules, tendon origins and other local (to joint) tissues. These painful reflex effects are noted in muscles and their tendinous junctions as well as the skin – which Brugger calls ‘tendomyosis’ – defined as, ‘the reflexogenic functional change in the muscle in the presence of concurrent functionally dependent muscle pain’. Dvorak & Dvorak (1984) include in this category of referred pain and symptoms the phenomena of viscerosomatic and somatovisceral influences, in which, for example, organ dysfunction is said to produce tendomyotic changes (Korr 1975). Some aspects of these phenomena form part of the discussion of facilitation in this chapter.
4. Tender points, as described by Jones (1981), which Dvorak & Dvorak equate with the tender points described by Kellgren (1938) (see Ch. 4 for more on Jones’ work), are seen to be spontaneously arising areas of tenderness related to acute or chronic strains, usually located in those soft tissues shortened at the time of the strain. These seem to equate with so-called ‘Ah shi’ points in traditional Chinese acupuncture methodology (i.e. points not necessarily present on acupuncture meridian maps, but occurring spontaneously in association with strain in the region).
5. Spondylogenic reflexes, knowledge of which Dvorak & Dvorak describe as being based on empirical clinical observation that demonstrates relationships between the axial skeleton and the peripheral soft tissues which ‘are not easily explained on the basis of radicular, vascular or humoral reasoning’. The effects of these reflexes include ‘demonstrable zones of irritation … which are painful swellings, tender upon pressure, located in the musculofascial tissue in topographically well-defined sites’ (Sutter 1975).
Dvorak & Dvorak (1984) suggest that these different classifications and descriptions are focusing on the same phenomena, with terminology and interpretations being the variables, rather than there being a host of different physiological patterns of response to stress and trauma.
In this context, earlier research into referred pain is worth re-evaluating, because it highlights basic issues – of which many have lost sight.
Speransky
Speransky, in his classic book A Basis for the Theory of Medicine (Speransky 1943), demonstrated clearly that ‘From any nerve point it is easy to bring into action nerve mechanisms, the functioning of which terminates at the periphery, in changes of a bio-physico-chemical character.’ And further: ‘Justification exists for the thesis that any nerve point, not excluding peripheral nerve structures, can become the originator of neurodystrophic processes, serving as the temporary nerve centre of these processes.’
Speransky continued: ‘It is obvious from this [evidence] that the irritation of any point of the complex network of the nervous system, can evoke changes, not only in the adjacent parts, but also in remote regions of the organism.’
 Kellgren
Kellgren
Following on from the pioneering work of Sir Thomas Lewis, the researcher J. H. Kellgren performed a series of studies in the late 1930s which deserve our attention since he clearly identified the character and nature of what became known as ‘myofascial trigger points’ (Kellgren 1938, 1939, Lewis 1938).
Kellgren (1938) effectively showed (using himself and volunteers) that by irritating fascia and muscle he could produce referred sensations in other structures. For example, among his early findings was evidence that a saline injection into the occipital muscles would produce a headache, while similar irritation to the masseter muscle produced a toothache.
Kellgren (1939) concluded that such distribution of pain usually followed segmental pathways, although he modified this position when he applied his studies to clinical work, at which time he not only identified localized, exquisitely painful spots that referred painful symptoms to distant areas, but also noted that the distribution of pain from such spots did not in fact always follow peripheral neural pathways. He also showed that a local anaesthetic injection into such spots could obliterate the referred pain sensations.
Kellgren (1939) stated:
Superficial fascia (of the back) and supraspinous ligaments induce local pain when stimulated, while stimulation of the superficial portions of the interspinous ligaments and superficial muscles results in diffuse pain. Deep muscles, ligaments and periostium of the apophyseal joints as well as the joints themselves can cause referred pain according to segmental innervation when stimulated [saline solution or mechanical stimulus].
Additional research at that time (1940s) indicated that there was not always a predictable pattern of pain distribution from such experiments (Feinstein et al 1954) (Fig. 3.1).
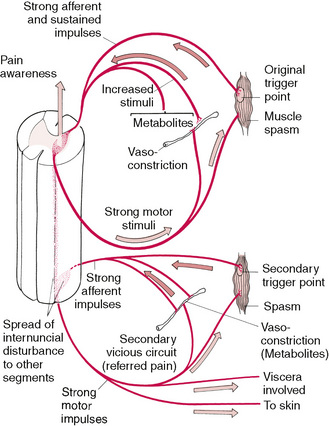
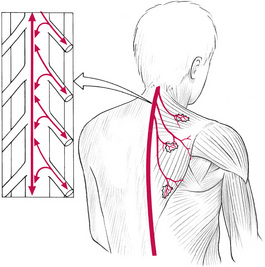
Figure 3.1 Schematic representation of the secondary spread of neurologically induced influences deriving from acute or chronic soft tissue dysfunction and involving trigger point activity and/or spasm.
Research by many others in the 1940s, continuing up to the present, has further investigated referred patterns of pain that do not seem to follow neurological pathways, or even known patterns of viscerally caused pain – or, for that matter, the common acupuncture pathways as described in traditional Chinese medicine – myofascial trigger points.
Evans
Evans, in discussing reflex pain, describes the mechanism as follows:
A prolonged bombardment of pain impulses sets up a vicious circle of reflexes spreading through a pool of many neuron connections upward, downward and even across the spinal cord and perhaps reaching as high as the thalamus itself. Depending upon the extent of the pool (internuncial pool), we detect the phenomena of pain and sympathetic disturbances a long distance from the injured (trigger) area of the body and occasionally even spread to the contralateral side.
Dittrich
Dittrich (1954) has shown a constant pattern of fibrosis of subfascial tissue, with adhesions between this and the overlying muscle fascia, in a number of distinctive and common pain patterns. For example, in what he calls the ‘mid-sacral syndrome’ which develops from sacral lesions, referred pain is almost always present in the buttock and sometimes in the thigh, leg or foot. Referred tenderness is elicited in the lower part of the buttock. The fibrous adhesions (trigger area) are found at the level of the 3rd sacral vertebra, near the spine.
In the ‘mid-lumbar syndrome’, Dittrich showed that there is referred pain and tenderness in the lower lumbar, upper sacral and sacroiliac regions. The trigger point is found over the lateral third of the sacrospinalis muscle at the level of the upper margin of the iliac crest.
The ‘latissimus dorsi syndrome’ could result from irritation of the aponeurosis of this muscle at either of the sites of injury, sacral or lumbar, mentioned above. Referred pain would develop in the sclerotonic distributions of the 6th, 7th and 8th cervical nerves.
Dittrich pointed out that pathological changes at the two sites had been discovered by operative findings. His technique was to remove these triggers surgically or to obliterate the triggers by injection of local anaesthetic. (No concern is expressed as to the mechanical, postural or other reasons that may have produced them, nor any mention made of more conservative manipulative methods of normalization.)
The presence of ‘fibrosed subfascial tissue’ supports the theories of Stanley Lief and Boris Chaitow, and their use of NMT in treating such problems (see Ch. 2). The locale of these, close to bony insertions, further supports the rationale behind neuromuscular technique application.
It seems that soft tissue lesions, characterized by fibrosis of subfascial tissue (fat, etc.) with fibrous connections between the structure and the overlying fascia, can initiate sensory irritation that produces referred pain and tenderness. In addition, autonomic nervous involvement may be activated to produce vasomotor, trophic, visceral or metabolic changes. Symptoms will disappear when the offending lesion is normalized, by whatever method. However, a system that both corrects the offending trigger and attempts to prevent its recurrence would seem to offer greater clinical benefits. Before surveying some of the other reflex patterns that are pertinent to this text, a brief summary of current thinking on the aetiology of muscle pain, as described by Mense & Simons (2001) is provided in Box 3.1.
Box 3.1 Mense & Simons perspective on muscle pain
In Chapter 1 a brief overview (see ‘Modern pain concepts’) was provided of how muscle pain evolves, as described by Mense & Simons (2001). In this section some of the major causes of muscle pain are outlined, based on the same researchers’ descriptions and discussions.
Local muscular pain
Local muscular pain is defined as deriving from local pain receptors, in the muscle, rather than via neural or central neurons. These free nerve endings (nociceptors) may be sensitized by mechanical (blows, repetitive or single strain injuries, rupture of fibres, etc.) and chemical (e.g. bradykinin) influences that either irritate or increase nociceptor sensitivity, leading to tenderness and soreness (such as occurs following unaccustomed exercise). Non-steroidal anti-inflammatory drugs decrease the degree of such sensitization. The types of situation that bring about such sensitization include trauma, inflammation and ischaemic circulatory deficit (such as that resulting from intermittent claudication or prolonged voluntary contraction, especially if this involves an already ischaemic muscle).
Local muscle pain involving inflammation may additionally lead to feelings of weakness and dysaesthesia. Local muscle pain may also be caused by the influence of a trigger point within the painful muscle, or referring to it from a distant muscle.
Metabolic causes of local muscle pain include enzyme deficiencies and hypothyroid status that ‘compromise oxidative metabolism in the muscle mitochondria’ (Mense & Simons 2001). Inflammatory process may result from injury or infection (e.g. Staphylococcus aureus) or infestation (e.g. trichinosis caused by Trichinella spiralis deriving from undercooked meat such as pork).
Local muscle pain resulting from compartment syndrome may result from development of a haematoma, oedema or infection in a muscle surrounded by a fascial envelope (such as flexor digitorum longus muscle in the deep posterior compartment). Surgical intervention may be called for to relieve the pressure before irreparable tissue damage occurs. A similar scenario may result from a plaster cast applied too tightly. Rarely toxicity-induced myalgic pain may derive from contaminated tryptophan, a sleep-inducing and pain-relieving amino acid, useful in the treatment of fibromyalgia. As pure tryptophan is no longer available because of contamination in the manufacturing process following a Japanese attempt to modify genetically the bacteria used in its production, tryptophan is now available in a plant-derived form, free of contaminants, as 5-hydroxytryptophan (Chaitow 2000).
Nerve lesion or dysfunction
Neuropathic muscle pain derived from a nerve lesion or dysfunction in the cranial (spinal) or dorsal root nerves. Causes may be mechanical (trauma) or entrapment (see notes on Maitland, Butler and Gifford in Ch. 1), or as a result of the development of a neuroma, which can lead to phantom pain. Pain from a herniated disc is an example of radicular pain. Peripheral neuropathies may occur in conditions such as diabetes. The causes of widespread pain conditions, which also involve autonomic symptoms, such as complex regional pain syndrome (previously called reflex sympathetic dystrophy), are thought to involve the sympathetic nervous system in an as-yet unknown way.
Referred pain
Pain that is referred to and from muscles can be confusing. Mense & Simons (2001) use the term ‘mislocalization of pain’, in which ‘the muscle in which the pain is felt serves only as a starting point for finding the source of pain, which is really what requires treatment’. The mechanisms are not clear, although the phenomenon is at the very heart of the pain often found to derive from distant trigger point activity. Mense & Simons indicate that there are confusing influences from trigger points when they reflexly influence other structures: ‘Why trigger points cause reflex spasm in one situation and reflex inhibition in another is not at all clear. One possibility is that the distinction between muscles prone to tightness (postural – see Ch. 1) and muscles prone to weakness (phasic – see Ch. 1) may be of fundamental physiologic significance.’ What is known is that specific dermatome and myotome distribution of sensations is not constant in relation to the symptoms produced by an active trigger point. Discussion in this chapter on the phenomena of local and segmental facilitation, and also of viscerosomatic reflex activity, covers aspects of this form of reflexogenic and referred pain.
Muscular tension
Pain deriving from muscular tension may involve biomechanical rather than neural influences. Factors such as the viscoelastic tone and the colloidal nature of soft tissues involving thixotropic qualities, as well as non-neurologically mediated contractures, may be present (see notes on fascia in Ch. 1).
This topic is complex and the reader is referred to Mense & Simons (2001, Ch. 5) for greater detail.
Aspects of the influence on muscular pain deriving from organ dysfunction, postural imbalances, the presence of active trigger points, and conditions such as fibromyalgia are all discussed in this and later chapters.
Other reflexogenic models
Gutstein
Gutstein (1956) showed that conditions such as ametropia may result from changes in the neuromuscular component of the craniocervical area, as well as from more distant conditions involving the pelvis or shoulder girdle. He states:
Myopia is the long-term effect of pressure of extra-ocular muscles in the convergence effort of accommodation involving spasm of the ciliary muscles, with resultant elongation of the eyeball. A sequential relationship has been shown between such a condition and muscular spasm of the neck.
Normalization of these muscles by manipulation relieves eye symptoms as well as fascial, dorsolumbar and abdominal tenderness. Gutstein terms these reflexes ‘myodysneuria’ and suggests that the reference phenomena of such spots or triggers would include pain, modifications of pain, itching, hypersensitivity to physiological stimuli, spasm, twitching, weakness and trembling of striated muscles; hypertonus or hypotonus of smooth muscle of blood vessels, and of internal organs; hypersecretion or hyposecretion of visceral, sebaceous and sudatory glands. Somatic manifestations may also occur in response to visceral stimuli of corresponding spinal levels (Gutstein 1944).
Many such trigger areas are dormant and asymptomatic. Gutstein’s method of treatment was the injection of an anaesthetic solution into the trigger area. He indicated, however, that, where accessible (e.g. muscular insertions in the cervical area), the chilling of these areas combined with pressure yielded good results. This is in line with Mennell’s work (1975), and fits into the field of NMT.
Amongst the patterns of vasomotor sebaceous, sudatory and gastrointestinal dysfunction mentioned by Gutstein (1944a) are the following, all of which relate to reflex trigger points or ‘myodysneuria’ (fibrositis/fibromyalgia):
1. Various patterns of vasomotor abnormality such as coldness, pallor, redness, cyanosis, etc. These variations in response to stimulation relate to the fact that most organs respond to weak stimuli by an increase in activity and to very strong stimuli by inhibiting activity. Menopausal hot flushes are one example, and these seem often to be linked to musculoskeletal pain. Gutstein found that obliteration of overt and silent triggers in the occipital, cervical, interscapular, sternal and epigastric regions was accompanied by years of alleviation of premenopausal, menopausal and late menopausal symptoms. Proponents of NMT have long emphasized the importance of normalizing these very structures.
2. Gutstein maintains that normalization of skin secretion, and therefore of hair and skin texture and appearance, may be altered for the better by the removal of active trigger areas in the cervical and interscapular areas.
3. The conditions of hyperhidrosis, hypohidrosis and anhidrosis may accompany vasomotor and sebaceous dysfunction. Gutstein noted that abolition of excessive perspiration as well as anhidrosis followed adequate treatment.
4. Gutstein quoted a number of practitioners who have achieved success in treating gastrointestinal dysfunctions by treating trigger areas. Some of these were treated by procaine injection, others by pressure techniques and massage. The abdominal wall lends itself to the latter procedure, as evidenced by the work of Cornelius (1909), whose treatment was not dissimilar to Lief’s NMT.
Among the conditions that have responded to such treatment are pylorospasm, bad breath, heartburn, regurgitation, nausea, abdominal distension, constipation and nervous diarrhoea. Gutstein (1944b) called localized functional sensory and/or motor abnormalities of musculoskeletal tissue (comprising muscle, fascia, tendon, bone and joint) myodysneuria (now known as fibromyalgia; formerly known as ‘fibrositis’ and ‘muscular rheumatism’).
He viewed the causes of such changes as multiple; among them are:
1. Acute and chronic infections which, it is postulated, stimulate sympathetic nerve activity via their toxins.
2. Excessive heat or cold, changes in atmospheric pressure, and draughts.
3. Mechanical injuries, both major and repeated minor microtraumas. Postural strain, unaccustomed exercises, etc., which may predispose towards future changes by lowering the threshold for future stimuli (facilitation).
4. Allergic and endocrine factors, which can cause imbalance in the autonomic nervous system.
5. Inherited factors that make adjustment to environmental factors difficult.
6. Arthritic changes – because muscles are the active components of the musculoskeletal system it is logical to assume that their circulatory state has influence over bones and joints. Spasm in muscle may contribute towards osteoarthritic changes, and such changes may produce further neuromuscular changes, which themselves produce new symptoms.
7. Visceral diseases may intensify and precipitate somatic symptoms in the distribution of their spinal and adjacent segments.
In these examples of Gutstein’s concept, we can see strong echoes of the facilitation hypothesis in osteopathic medicine, and it seems likely that they are describing the same set of circumstances leading to hyperreactive responses – and all that this leads to in terms of pain and dysfunction.
Diagnosis of myodysneuria was made according to some of the following criteria:
• A varying degree of muscular tension and contraction is usually present, although sometimes adjacent, apparently unaffected, tissue is more painful.
• Sensitivity to pressure or palpation of affected muscles and their adjuncts.
• Marked hypertonicity may require the application of deep pressure to demonstrate tenderness.
Travell and Bigelow
Travell & Bigelow (1947) produced evidence to support much of what Gutstein had reported. They indicated that high-intensity stimuli from active trigger areas produced, by reflex, prolonged vasoconstriction with partial ischaemia in localized areas of the brain, spinal cord or peripheral nerve structures (Fig. 3.2). A wide pattern of dysfunction might result, affecting almost any organ of the body. The phenomenon of hysteria, with symptoms as varied as disordered vision, respiration, motor power and cutaneous sensation, was often mediated by afferent neural impulses from trigger areas in skeletal muscle. These triggers, when similarly located, produced the same pattern of clinical effects, whether activated in one patient by psychogenic factors or in another by different factors (e.g. mechanical or traumatic).
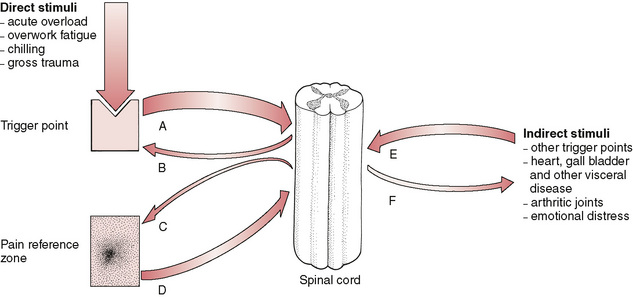
Figure 3.2 Direct stress influence can affect the hyperreactive neural structure of a myofascial trigger point, leading to increased activity (A–B) as well as referring sensations (pain, paraesthesia, increased sympathetic activity) to a target area (C–D) that feed back into the cord to increase the background stress load. Other stimuli reach the cord from distant trigger points and additional dysfunctional areas (E–F).
It is worth noting that Travell (1981) has shown that the effect of a trigger point on the muscle housing it is to produce weakness of contraction without atrophy, and that this is often accompanied by a decreased range of movement in associated joints.
Dowling
Dowling (2000) has described a variation on the use of inhibitory pressure (see Box 3.5), which allies itself closely with the methods of NMT and other soft tissue approaches utilizing the effects of compression in a physiologically useful manner. Using two points of contact, progressive, sequential pressure applications are used to inhibit pain and modify function. Dowling’s work fits well with other approaches (acupressure, strain/counterstrain, use of neurolymphatic reflexes, etc.) that depend on the underlying mechanisms that allow manual input to modify neuromuscular function and pain. See Chapter 11 for a full explanation of progressive neuromuscular inhibition technique (PINS).
Central sensitization (see Fig. 3.3)
All these historical perspectives are describing, in one way or another, what is now known as peripheral and central sensitization. In osteopathic medicine the evolution over time of hypersensitivity and reactivity in neural structures, is known as facilitation, and this is described later in this chapter (Korr 1970).
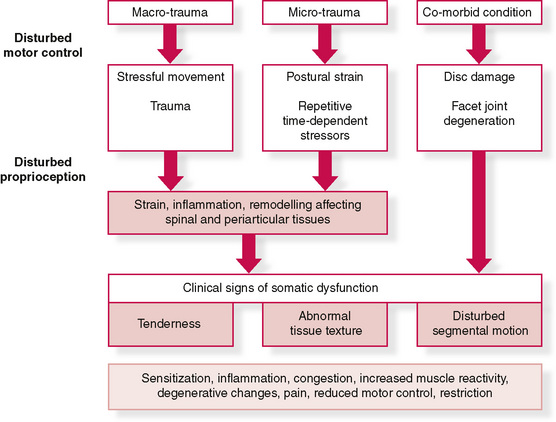
Figure 3.3 Diagram showing possible aetiological pathways to somatic dysfunction and sensitization
Modified from Fryer G, Fossum C. Possible etiology of somatic dysfunction. In: Physical Therapy for tension type and cerviocogenic headache. Fernández de las Peñas C., Arendt-Nielsen L., Gerwin R. (Eds): Jones & Bartlett, Boston, 2009.
In brief, sensitization involves what happens when a simple stimulus, that is repeated, becomes irritable, and eventually painful.
A demonstration of this process can be seen in the example of a repetitively pressed, or rubbed, or knocked area of the body. This stimulation might initially feel warm, uncomfortable and eventually painful. The pain would be the result of the progressively amplified synaptic response of the peripheral nerves producing an alarm message that the repetitive stimulation is potentially harmful. Neural structures peripherally, or in the cord, or the brain may be involved in different variations of this process.
Sensitization may underlie both adaptive as well as maladaptive learning processes for the organism. This process is also known as long-term potentiation, and involves complex biochemical changes in the tissues involved (Ji et al 2003, McEachern et al 1999).
Just how manual therapy can modify this process is another matter, and this will be explored in other chapters.
Understanding facilitation
If we are to make sense of the phenomenon of myofascial trigger points there is a need to grasp the processes that lead to hyperirritability and hyperreactivity of specific neural structures – facilitation (aka sensitization). Trigger points are localized areas of soft tissue dysfunction that negatively influence distant target areas, and that evolve in a manner similar to that observed when spinal segments become facilitated as a result of stress of one sort or another.
To understand trigger points, we need to understand spinal facilitation first. Facilitation occurs in both spinal and paraspinal tissues (segmental facilitation) as well as in discrete local areas of muscles, mainly near their origins and insertions but also close to their bellies and in areas where fascial stress occurs due to external influences and forces (myofascial trigger points). Understanding facilitation helps us to see how the different classification systems, all describing variations on the same phenomenon of referred pain – listed by Dvorak & Dvorak (1984) and described earlier in this chapter – are held together aetiologically.
Patterson (1976) explains segmental (spinal) facilitation as follows:
The concept of the facilitated segment states that because of abnormal afferent or sensory inputs to a particular area of the spinal cord, that area is kept in a state of constant increased excitation. This facilitation allows normally ineffectual or subliminal stimuli to become effective in producing efferent output from the facilitated segment, causing both skeletal and visceral organs innervated by the affected segment to be maintained in a state of overactivity. It is probable that the ‘osteopathic lesion’, or somatic dysfunction with which a facilitated segment is associated, is the direct result of the abnormal segmental activity as well as being partially responsible for the facilitation.
A facilitated segment therefore emerges from a prolonged period during which abnormal or altered inputs from a single source (or more than one source) of irritation, impinging on the spinal cord, keep the interneurons or motor neurons of that spinal segment in a constant state of excitement, thus allowing normally ineffectual inputs to produce outputs to all organs receiving innervation from the excited area. This concept implies that the spinal cord is a relatively passive mediator of the influences imposed on it and that the neural paths act as communicators of that activity (Denslow et al 1949).
Research on spinal functions seems to indicate, however, that the spinal cord, besides being the determiner of where abnormal activity is sent, by virtue of predetermined pathways, may participate actively in either controlling abnormal or unusually intense inputs, or amplifying and retaining such inputs in certain circumstances (Korr 1986).
Initially, only intensities of afferent input above a certain level would result in increased sensitivity of the spinal pathway. Inputs of lower intensity would either produce no alterations, or would cause an actual decrease in sensitivity as a protective mechanism against undue changes in homeostatic processes. It is apparent that the potential for sensitization by different types of afferent inputs may differ widely. Thus, inputs from pain receptors may sensitize the pathway at low levels because of the properties of the initial synapses between pain afferent fibres and interneurons. In this event an initially protective increase in response might occur, followed eventually by detrimental facilitation of a segment. On the other hand, inputs from joint receptors seem to have a less dramatic effect at similar input levels (Dowling 1991).
It is now known that emotional arousal would also affect the susceptibility of the pathways to sensitization (Baldry 1993). The increase in descending influences from the emotionally aroused subject would result in an increase in toxic excitement in the pathways and allow all additional inputs to produce sensitization at low intensities. Thus, highly emotional people, or those in a highly emotional situation, would be expected to show a higher incidence of facilitation of spinal pathways.
As the higher brain centres influence the tonic levels of the spinal paths, it might also be expected that physical training and mental attitudes would tend to alter the tonic excitability, reducing the person’s susceptibility to sensitization from everyday stress. Thus the athlete would be expected to withstand a comparatively high level of afferent input before experiencing the self-perpetuating results of sensitization.
A further corollary of the hypothesis is that slowly developing conditions, or slowly increasing inputs, would result in less sensitization at high levels than sudden inputs. A process known as ‘wind-up’ can be part of sensitization, with the rate of stimulus, as well as the size of stimuli, determining how rapidly the process evolves. If noxious stimuli occur rapidly, the degree of stimulus required to produce symptoms (pain mainly) decreases (Codere et al 1993, Li 1999).
Windup is just such a mechanism that is thought to operate in fibromyalgia where relatively minor stimuli can produce symptoms that appear to be proportionately excessive (Staud 2006).
The slow development of a chronic source of increased sensory input initially causes habituation, resulting in resistance to sensitization until the input level is abnormally high. On the other hand, sudden increases in input, such as a sudden mechanical stress, would be expected to rapidly produce sensitization of the neural pathways.
Korr
The premier researcher into facilitation over the past half-century has been Irwin Korr (Korr 1970, 1976). In early studies he demonstrated, for example, that if readings were taken of resistance to electricity in the paraspinal skin of an individual there were often marked differences, with one side showing normal resistance and the other showing reduced resistance (facilitated area).
When ‘stress’ was applied elsewhere in the body and the two areas of the spine were monitored, it was the area of facilitation, where electrical resistance was reduced, that showed a dramatic rise in electrical (i.e. neurological) activity. In one experiment, volunteers had pins inserted into one calf muscle in order to gauge the effect in the paraspinal areas under investigation – with the spinal areas being monitored for electrical activity. Almost no increase occurred in the normal region, but the facilitated area showed enormously enhanced neurological activity after 60 seconds (Korr 1977) (Fig. 3.4).
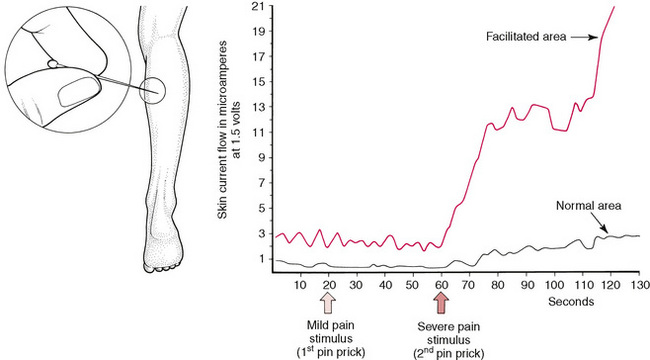
Figure 3.4 Pain stimuli produce a marked reaction in the facilitated area (red line) and little reaction in the normal area (black line).
The implications of this and hundreds of similar studies are that any form of stress impacting the individual, be it climatic, chemical, emotional, physical or anything else, would produce just such a rise in neurological output from facilitated areas.
In many instances involving spinal segmental facilitation there is a chronic degree of neurological bombardment resulting from internal organ dysfunction. For example, it is almost always possible to predict that cardiovascular disease is present (or will soon be present) when two or more segments of the spine in the region of T2, T3 and T4 display tense, rigid, ‘board-like’ characteristics on palpation, especially if these tissues do not respond to normal efforts to reduce their hypertonicity (Beal 1983).
Tension headache as an example of facilitation
Danish research (Bendtsen 2000) suggests that there exists:
… a pathophysiological model for tension-type headache [resulting from] central sensitization [facilitation] at the level of the spinal dorsal horn/trigeminal nucleus due to prolonged nociceptive inputs from pericranial myofascial tissues. The increased nociceptive input to supraspinal structures may in turn result in supraspinal sensitization. The central neuroplastic changes may affect the regulation of peripheral mechanisms and thereby lead to, for example, increased pericranial muscle activity, or sensitization may be maintained even after the initial eliciting factors have been normalized, resulting in conversion of episodic into chronic tension-type headache.
This research demonstrates the need to understand how, over time, a reversible problem may become entrenched and chronic. It is knowledge of facilitation and sensitization that also explains how multiple stress elements affecting the person as a whole (climatic, emotional, postural, nutritional, etc.) might feed into the sensitized structures, exacerbating symptoms, despite apparently not directly impacting the involved structures.
Viscerosomatic reflexes
Many of the various systems involving reflexively active points described in this and the next chapter, such as connective tissue zones, Chapman’s reflexes and Bennett’s reflexes, as well as trigger points, may involve viscerosomatic reflex activity.
Beal
Myron Beal (1985) has described this phenomenon as resulting from afferent stimuli, arising from dysfunction of a visceral nature. The reflex is initiated by afferent impulses arising from visceral receptors, which are transmitted to the dorsal horn of the spinal cord, where they synapse with interconnecting neurons. The stimuli are then conveyed to sympathetic and motor efferents, resulting in changes in the somatic tissues, such as skeletal muscle, skin and blood vessels.
Abnormal stimulation of the visceral efferent neurons may result in hyperaesthesia of the skin, and associated vasomotor, pilomotor and sudomotor changes. Similar stimuli of the ventral horn cells may result in reflex rigidity of the somatic musculature. Pain may result from such changes.
The degree of stimulus required, in any given case, to produce such changes will differ, because factors such as prior facilitation of the particular segment, as well as the response of higher centres, will differ from person to person.
In many cases it is suggested, by Korr and others, that viscerosomatic reflex activity may be noted before any symptoms of visceral change are evident, and that this phenomenon is therefore of potential diagnostic and prognostic value.
The first signs of viscerosomatic reflexive influences are vasomotor reactions (increased skin temperature), sudomotor (increased moisture of the skin), skin textural changes (e.g. thickening), increased subcutaneous fluid and increased contraction of muscle. The value of light skin palpation in identifying areas of facilitation cannot be too strongly emphasized (Lewit 1992).
These signs disappear if the visceral cause improves. When such changes become chronic, however, trophic alterations are noted, with increased thickening of the skin and subcutaneous tissue, and localized muscular contraction. Deep musculature may become hard, tense and hypersensitive. This may result in deep splinting contractions, involving two or more segments of the spine, with associated restriction of spinal motion. The costotransverse articulations may be significantly involved in such changes.
Patterns of somatic response will be found to differ from person to person, and to be unique, in terms of location, the number of segments involved, and whether or not the pattern is unilateral or bilateral. The degree of intensity will also differ, and is related to the degree of acuteness of the visceral condition (Hix 1976). Research involving animals, as well as observations in humans, using regional nerve blocks, has helped to define site locations of response, in various forms of visceral dysfunction. Beal (1985) notes that, when the voluminous research into segmental associations with organ dysfunction is compounded, three distinct groups of visceral involvement are found in respect of particular sites:
2. T5–T10: oesophagus, stomach, small intestine, liver, gall bladder, spleen, pancreas and adrenal cortex
3. T10–L2: large bowel, appendix, kidney, ureter, adrenal medulla, testes, ovaries, urinary bladder, prostate gland, uterus.
There appears to be a consensus as to sidedness being apparent, in reflexes of unpaired organs. Thus, left-sidedness is noted in conditions involving the small intestine and heart, and right-sidedness for gall bladder disease and appendix. The stomach may produce reflex activity on either, or both, sides. Many studies have been concerned with the identification of viscerosomatic reflexes. One 5-year study involved more than 5000 hospitalized patients (Beal 1985), and concluded that most visceral disease appeared to influence more than one spinal region, and that the number of spinal segments involved seemed to be related to the duration of the disease. Kelso noted in this study that there was an increase in the number of palpatory findings in the cervical region, related to patients with sinusitis, tonsillitis, diseases of the oesophagus and liver complaints. Soft tissue changes were noted in patients with gastritis, duodenal ulceration, pyelonephritis, chronic appendicitis and cholecystitis, in the region of T5–T12.
 Palpating facilitated spinal tissues
Palpating facilitated spinal tissues
Somatic dysfunction is usually assessed by means of palpatory investigation, and Beal (1983) insists that investigation should pay attention to the various soft tissue layers:
The skin for changes in texture, temperature and moisture; the subcutaneous tissue for changes in consistency and fluid; the superficial and deep musculature for tone, irritability, consistency, viscoelastic properties, and fluid content; and the deep fascial layers for textural changes.
He advises that ‘Special attention [should] be given to the examination of the costotransverse area, where it is felt that autonomic nerve effects are predominant’, and notes that tests for the quality and range of joints have not been found to differentiate between visceral reflexes and somatic changes, which confirms the importance of the soft tissue assessment in order to elicit such information.
Beal (1983) notes that the supine position is ideal for assessment of paraspinal tissues – the hand being gently inserted under the region, and pressure, or springing techniques applied (Fig. 3.5). He has not investigated the use of patient examination in the prone position, as he suggests that this position is precluded in acutely ill patients.
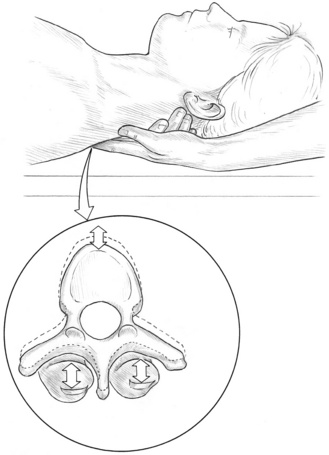
Figure 3.5 Beal’s ‘springing’ assessment for paraspinal facilitation rigidity associated with segmental facilitation.
Nevertheless, since Beal notes the difficulty of applying diagnostic measures with the patient supine when the mid to lower thoracic area is under review, a prone position is suggested, unless the patient cannot manage this. The availability of a couch with a split head-piece would make this more comfortable. As we will see, the methods employed by those using connective tissue massage involve the patient being seated. This helps in assessing skin and superficial tissue status, but is not really suitable for deeper penetration.
Beal suggests that the diagnosis of a paraspinal viscerosomatic reflex be based on two or more adjacent spinal segments showing evidence of somatic dysfunction, and being located within the specific autonomic reflex area. There should be deep confluent spinal muscle splinting, and resistance to segmental joint motion. Skin and subcutaneous tissue changes that are consistent with the acuteness or chronicity of the reflex should be noted.
Specific identification of the origin of the reflex is, he suggests, difficult.
The usefulness of understanding the nature of such reflexes often involves clinical frustration when, for instance, localized soft tissue dysfunction fails to respond to treatment. Suspicion may then be alerted to possible visceral activity maintaining the muscular or joint dysfunction.
According to Beal, treatment of the acute stage should be aimed primarily at breaking into the reflex arc. In cases of serious illness the treatment may consist of gentle digital pressure, of short duration, to effect a local change in superficial tissues. When relaxation has been accomplished in the subcutaneous and superficial paraspinal musculature, the deep muscle contraction can be addressed. The duration of treatment is dependent upon the patient’s condition and perceived energy level. Beal suggests that acute conditions that are not life threatening may be addressed in a more aggressive manner (asthma is given as an example).
It is suggested that NMT is also an ideal method of addressing soft tissue manifestations of such reflex activity, because it offers a diagnostic as well as a therapeutic opportunity to address both superficial and deep tissues. Viscerosomatic reflex changes are just one of the many reasons for altered tissue findings, which may be noted in the general NMT assessment. Awareness of the possibility that what is being noted is of reflex origin adds to the potential for accurate diagnosis. In Chapter 4 a variety of other reflex systems are evaluated.
Causes of local facilitation
Melzack & Wall (1989), in their exhaustive investigation of pain, are clear in their statement that all chronic pain has myofascial trigger point activity as at least a part of its aetiology, and that in many instances trigger points are the major contributors to the pain.
A trigger point is a localized, palpable area of soft tissue that is painful on pressure and that refers symptoms, usually including pain, to a predictable target area some distance from itself. It is an area of local facilitation that has developed following a very similar aetiological pathway to that occurring in segmental (spinal) facilitated areas.
Facilitation paraspinally and in general muscle tissue can be the result of repetitive minor or single major traumatic influences or stress factors (as described in Ch. 1). The form of facilitation that is our main focus in this chapter is a localized area of hyperirritability – the trigger point.
Travell and Simons
Much research and clinical work has been done in recent years in this field by Janet Travell (Travell 1957, Travell & Simons 1983, 1992, Travell, Simons & Travell 1999), who is on record as stating that if a pain is severe enough to cause a patient to seek professional advice (in the absence of organic disease) referred pain is likely to be a factor, and therefore a trigger area is probably involved. She maintains that patterns of referred pain are constant in distribution in all people; only the intensity of referred symptoms or referred pain will vary.
Among the effects of an active trigger point, apart from pain, there may be numbness, tingling, weakness, lack of normal range of movement. The aetiological myofascial trigger point for a particular pain pattern is always located in a particular part of a particular muscle (Webber 1973). While eradication of the trigger, by whatever appropriate means, can remove all symptoms, treatment of the target or reference area is useless.
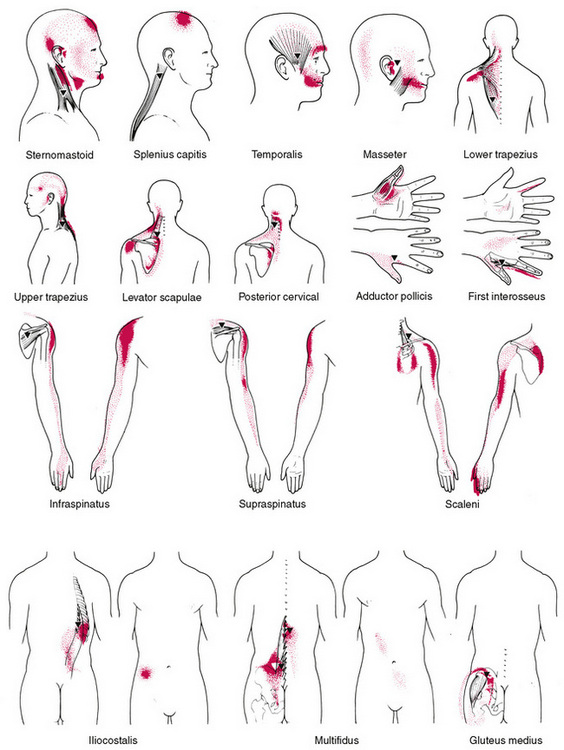
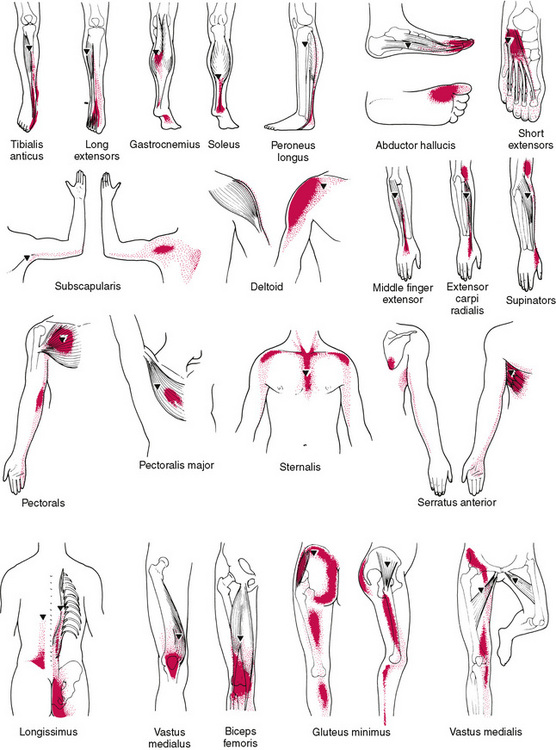
 Trigger points (Fig. 3.6)
Trigger points (Fig. 3.6)
Trigger points are localized areas of deep tenderness and increased resistance, and digital pressure on such a trigger will often produce twitching and fasciculation (see Fig. 3.6). Pressure maintained on such a point will produce referred pain in a predictable area. If there are a number of active trigger points, the reference areas may overlap.
What is distinctive about active trigger points (myofascial trigger points) is that, when active, they also refer sensations or symptoms to a distinct target area, and this target area is more or less reproducible in other individuals, when trigger points are located in similar positions. No other soft tissue dysfunction has this particular attribute.
Before an active trigger point exists there needs to be a period of evolution towards that unhappy state. This involves the development of soft tissue changes that are palpable and probably sensitive, or painful, but that, until sufficient localized stress has been involved, will not refer symptoms onwards. In other words, many localized muscular areas of sensitivity or pain, which do not refer pain or other symptoms, may be considered to be embryonic or evolutionary trigger points. A single trigger may refer pain to several reference sites and can give rise to embryonic, or satellite, triggers in those target areas. Travell (1981) describes, for example, how a trigger in the distal areas of the sternomastoid muscle can give rise to new triggers in the sternalis muscle, the pectoral muscle and/or serratus anterior.
Travell’s description of a trigger point is:
It lies in skeletal muscle, and is identified by localised deep tenderness, in a palpable firm band of muscle (muscle hardening); and at the point of maximum deep hyperalgesia, by a positive ‘jump sign’, a visible shortening of the part of the muscle which contains the band. To elicit the jump sign most effectively, one must place the relaxed muscle under moderate passive tension, and snap the band briskly with the palpating finger.
The trigger point must also refer symptoms or sensations, which are familiar to the patient, to a target area. Otherwise, rather than being active, it may be a latent trigger point, which could be activated by stress or strain on the tissues in which it lies (Box 3.2). The difference between most other areas of discrete palpable soft tissue dysfunction and an active trigger is this quality of referring symptoms. All other points may be prospective triggers, but are not active (Fig. 3.7).
Box 3.2 Active and latent trigger points defined (see Fig. 3.6)
• An active trigger point, when mechanically stimulated by compression, needling, stretch or other means, will refer to, or intensify, a target zone (referral pattern, usually of pain) which the person recognizes as being part of their current symptom picture.
• When a latent trigger point is stimulated, it refers a pattern that may be unfamiliar to the person, or that is an old pattern which they used to have but have not had for a while (previously active, reverted to latent).
All the same characteristics that denote an active trigger point may be present in the latent trigger point, with the exception of the person’s recognition of their active pain pattern. The same signs as described for segmental facilitation, such as increased hydrosis, a sense of ‘drag’ on the skin, loss of elasticity, etc., can be observed and palpated in these localized areas as well.
Travell & Simons (1983, 1992) use the term ‘essential pain zone’ to describe a referral pattern that is present in almost every person when a particular trigger point is active. Some trigger points may also produce a ‘spill-over pain zone’, beyond the essential zone or in place of it, where the referral pattern is usually less intense. These target zones should be examined, and ideally palpated, for changes in tissue ‘density’, temperature, hydrosis and other characteristics associated with satellite trigger point formation. Any appropriate manual treatment, movement or exercise programme that encourages normal circulatory function is likely to modulate these negative effects and reduce trigger point activity.
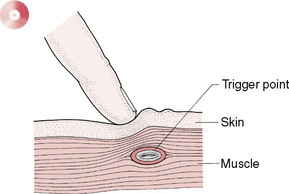
Figure 3.7 Trigger points are areas of local facilitation that can be housed in any soft tissue structure, most usually muscle and/or fascia. Palpation from the skin or at depth may be required to localize them.
 STAR or TART
STAR or TART
In osteopathic medicine an acronym ‘STAR’ is used as a reminder of the characteristics of somatic dysfunction, which would include those relating to myofascial trigger points:
The acronym is modified in some texts to ‘TART’ (Tenderness – Asymmetry – Range of movement modified – Tissue texture change).
While the ‘tenderness’ and range of motion characteristics, as listed in the STAR acronym, hold true for trigger points, the following refinements have been detailed by Simons et al (1999):
• The soft tissues housing the trigger point will demonstrate a painful limit to stretch range of motion.
• A palpable taut band with exquisitely tender nodule should be located.
• Pressure on the tender spot elicits pain familiar to the patient and often a pain response (‘jump sign’).
Baldry (1993) noted that the commonest sites for trigger points are close to:
• muscular origins and insertions
• body tissues other than muscle, including skin, fascia, ligament, joint capsule and periosteum, and also in scar tissue.
Simons et al (1999), in their latest research, place great emphasis on the distinct differences in treatment protocols for so-called ‘attachment’ and ‘central’ trigger points (see Box 3.3).
Box 3.3 Understanding and treating central and attachment trigger points (Chaitow & DeLany 2000, Simons et al 1999)
Simons, Travell and Simons’ (1999) model of central trigger points
• Central trigger points form almost directly in the centre of the muscle’s fibres, close to the motor endplate (neuromuscular junction).
• This formation is probably due to dysfunctional endplate activity.
• A metabolic crisis occurs and acetylcholine (ACh) is excessively released at the synapse, usually associated with overuse or strain, leading to release of calcium.
• The resulting ischaemia in the area creates an oxygen/nutrient deficit and a local energy crisis.
• Without available adenosine triphosphate (ATP), the calcium ions, which are keeping the gates open for ACh to keep flowing, cannot be removed.
• A chemically sustained contracture occurs. This is involuntary without motor potentials, and should be distinguished from a contraction, which is voluntary and involves motor potentials, and spasm, which is involuntary with motor potentials.
• As the endplate keeps producing this ACh flow, the actin/myosin filaments shorten and bunch in the immediate area of the motor endplate (centre of fibre).
• A contracture ‘knot’ forms the characteristic trigger point nodule.
• The remainder of the sarcomeres of that fibre are stretched, creating the equally characteristic taut band, which is usually palpable.
• When massage, stretch applications, injection or other modalities are applied, which disturbs the sarcomeres, alters the chemistry or possibly damages the endplate, the cycle is disrupted and the tissue relaxes, often in seconds – often permanently.
Simons, Travell and Simons’ model of attachment trigger points
• Attachment trigger points form at junctures of myofascial and tendinous or periosteal tissues.
• Tension from taut bands on periosteal or connective tissues can lead to enthesopathy or enthesitis, as recurring concentrations of muscular stress may provoke inflammation, with a strong tendency towards subsequent fibrosis and calcific deposition.
Processes
• Central and attachment trigger points can both lead to referred and/or radiating pain; however, the local processes seem to be different and the two types of trigger point seem to respond to different therapeutic approaches.
• Until they have been examined thoroughly and tissue reactions noted, attachment trigger points should be addressed, with their tendency towards inflammation in mind.
• Central trigger points should be addressed with their contracted central sarcomeres and local ischaemia in mind.
• As the end of that taut band is likely to create enthesopathy, stretching the muscle before releasing its central trigger point might further inflame the attachments.
• Techniques should first be applied to relax the taut fibres before manual stretching (such as gliding strokes or myofascial release) is attempted.
• Stretches, particularly those involving active range of motion, should be applied gently until reaction is noted, to avoid further tissue insult.
• When passive stretching is applied, care should be taken to assess for tendinous or periosteal inflammation, in order to avoid placing more tension on already distressed connective tissue attachments.
Treatment choices for central trigger points
• Elongation of the tissue to its full length is, according to Simons et al (1999), critical to abolish the mechanism sustaining a trigger point.
• This may be achieved manually by application of gliding strokes from the centre of the fibres out to the attachment sites, stretching the tissue passively through its range of motion, or by having the person perform active range of motion, both during the treatment session and at home.
• Proprioceptive neuromuscular facilitation (PNF) techniques can be used to override the mechanisms causing the condition.
• Reciprocal inhibition (RI) and post-isometric relaxation (PIR) can accompany or precede range of motion movements and stretches to augment the benefits (see MET notes in Ch. 8).
• Central trigger points seem to respond well to heat.
• When compression techniques are used, local chemistry can change as a result of induced ischaemia, followed by flushing of the tissues when the compression is released (see Box 3.5).
• The effects of thermal or other applications (skin irritants such as capsaicin, moxibustion, dry or wet needling, etc.) may induce the contracture to release.
• Contrast hydrotherapy, employing alternating ice and heat, can be effective via its circulatory and/or neurological (reflex) influences.
Treatment choices for attachment trigger points
• Attachment trigger points seem to respond to ice applications rather than heat.
• Gliding techniques should be applied from the centre of the fibres out towards the attachments, unless contraindicated (as in some extremity tissues).
• By elongating the tissue toward the attachments, sarcomeres that are shortened at the centre of the fibre will be lengthened, and those that are overstretched near the attachment sites will have their tension released.
Trigger points: symptoms other than pain
While pain is the commonest symptom arising from the activity of trigger points, other symptoms may be noted, including lymphatic stasis and reduced mobility of joints.
Lymphatic stasis
Travell & Simons (1983, 1992) have identified triggers that impede lymphatic function.
The scalenes (in particular anticus) can entrap structures passing through the thoracic inlet. This is aggravated by the 1st rib (and clavicular) restriction, which can be caused by triggers in anterior and middle scalenes. Scalene trigger points have been shown reflexly to suppress lymphatic duct peristaltic contractions in the affected extremity, whereas triggers in the posterior axillary folds (subscapularis, teres major, latissimus dorsi) influence lymphatic drainage affecting the upper extremities and breasts. Similarly, triggers in the anterior axillary fold (pectoralis minor) can be implicated in lymphatic dysfunction affecting the breasts (Kuchera 1997).
Clinical symptoms other than pain
• Vasoconstriction and headache
• Proprioceptive disturbance, dizziness
• Excessive maxillary sinus secretion
• Cardiac arrhythmias (especially pectoralis major triggers)
• Ptosis, excessive lacrimation
• Interstitial cystitis (Weiss 2001).
Linking symptoms to specific trigger points
• Tension-type headache may derive from active triggers located in sternocleidomastoid, upper trapezius, posterior cervical musculature or temporalis.
• Frozen shoulder symptoms may be associated with trigger points in subscapularis, supraspinatus, pectoralis major and minor, and in the deltoid muscles.
• Epicondylitis may derive from trigger points present in the extensors of the fingers and hand, supinator and triceps brachii.
• Carpal tunnel syndrome may be associated with triggers in the scaleni and finger extensors.
• Atypical angina pectoris pain may be associated with active trigger points in left side pectoralis major or in the intercostals.
• Low back pain may involve trigger points in quadratus lumborum, iliopsoas, thoracolumbar paraspinals, rectus abdominis, piriformis, gluteus maximus and/or medius.
Reduced mobility
Joint restrictions have been noted as deriving from trigger point activity; for example, Kuchera (1997) has listed trigger points associated with shoulder restriction as shown in Table 3.1.
Table 3.1 Trigger points associated with shoulder restriction (Kuchera 1997)
| Restricted motion | Muscle housing trigger point |
|---|---|
| Flexion | Triceps |
| Abduction | Subscapularis |
| Infraspinatus | |
| Supraspinatus | |
| Teres major | |
| Levator scapulae | |
| Internal rotation | Teres major |
| Infraspinatus | |
| External rotation | Subscapularis |
| Pectoralis minor |
Production of trigger points
The causes of trigger point presence can relate to any combination of physical or psychic stress factors that result in alterations in normal tone in muscles, fascia and other soft tissues which, in turn, can effect changes in joint play, breathing, posture, etc. (Fig. 3.8). The progression from hypertonicity to retained metabolic wastes and relative ischaemia, in muscles affected by, for example, poor posture, has been discussed previously (see Ch. 1). The feature of ischaemia and prolonged stress seems to be a major predisposing condition in the production of trigger points and their referred pain and dysfunction. Travell’s research has shown that a series of embryonic trigger points develops in target/referred areas so that, in time, a chain reaction of triggers may be present.
Identifying trigger points
Kuchera & McPartland (1997) have described the major clinical characteristics of trigger points, based largely on the work of Travell and Simons. They report that, due to an apparent metabolic crisis involving impaired circulation and neural hyperreactivity (facilitation), a number of palpable signs are usually apparent:
• Altered cutaneous temperature (increased or decreased) (see Box 3.4)
• Altered cutaneous humidity (usually increased)
• Small nodular or spindle-shaped thickening representing trigger point locality
• A ‘jump’ sign (or exclamation !) may accompany palpation because of extreme sensitivity
• A local twitch response may be elicited by stroking across the long axis of taut band housing trigger
• Local trophic changes or ‘goose-flesh’ may be evident overlying the trigger site.
Box 3.4 Thermography and trigger points (Baldry 1993)
Various forms of thermography have been used to identify trigger point activity, including infrared electrical or liquid crystal.
Swerdlow & Dieter (1992) found, after examining 365 patients with clinically demonstrable trigger points in the upper back, that ‘although thermographic “hot-spots” are present in the majority, the sites are not necessarily where the trigger points are located.’
Is it possible that ‘old’ triggers lie in ischaemic tissue leading to ‘cold spots’ being noted?
Thermal examination of reference zones (target areas) usually shows that skin temperature is raised – but not always. Simons (1987) attributes this anomaly to different effects of triggers on the autonomic nervous system.
Barrall (1996) has shown that manual–thermal diagnosis is accurate with respect to ‘heat’ only 70% of the time. Apparently, when heat is noted during off-the-body scanning as when the hand moves over a truly warm area and then over a cooler area, the difference between the two areas provokes a sensation of ‘heat’, even though the area being scanned when this is noted may be appreciably cooler than surrounding tissues. This remains a valuable tool, however, as ‘different’ – whether hot or cold – suggests direct palpation of the area to be a useful next step.
Palpation might therefore involve (see Ch. 5 for a fuller presentation of assessment methods when using skin characteristics as a feature):
1. Off-body scan (manual thermal diagnosis), which may offer evidence of variations in local circulation; trigger point activity is more likely in areas of greatest ‘difference’ (see Box 3.4).
2. Movement of skin on fascia – resistance indicates the general locality of reflexogenic activity – a ‘hyperalgesic skin zone’ such as a trigger point (Lewit 1992).
3. Local loss of skin elasticity – refines definition of the location (Lewit 1992).
4. Light stroke, seeking ‘drag’ sensation (increased hydrosis), offers pinpoint accuracy of location (Lewit 1992).
5. Digital pressure (angled rather than perpendicular) into tissues seeks confirmation of active trigger (recognizable referred symptoms) or inactive, latent, trigger (local pain or unfamiliar referral pattern).
Obstacles to direct palpation of myofascial trigger points
Using skin characteristics (see Ch. 5) and thermal assessments (see Box 3.4) it is usually possible to identify the location of soft tissue somatic dysfunction. To establish whether the form that dysfunction takes involves a myofascial trigger point, palpatory pressure into the tissues is required (as in assessment mode NMT; see Chs 5 and 6 in particular). Apart from undeveloped, or unrefined, palpation skills, Simons (2002) suggests various reasons for failing to achieve direct palpation of myofascial trigger points, including:
Trigger point features
The pathways that allow particular triggers to produce symptoms in target areas do not follow known neurological patterns, nor do they precisely mimic the pathways of traditional Chinese medicine meridians – although there is some overlap. Wall & Melzack (1989) have shown that approximately 80% of major trigger point sites are on established acupuncture points.
Travell & Simons (1983) have described the trigger point as follows:
In the core of the trigger lies a muscle spindle that is in trouble for some reason. Visualise a spindle like a strand of yarn in a knitted sweater … a metabolic crisis takes place which increases the temperature locally in the trigger point, shortens a minute part of the muscle (sarcomere) – like a snag in a sweater, and reduces the supply of oxygen and nutrients into the trigger point. During this disturbed episode an influx of calcium occurs and the muscle spindle does not have enough energy to pump the calcium outside the cell where it belongs. Thus a vicious cycle is maintained and the muscle spindle can’t seem to loosen up and the affected muscle can’t relax.
Simons (1994) has reviewed the work of others who have tested this concept and found that at the centre of the trigger points there is indeed a lack of oxygen compared with that in the muscles surrounding it (Simons 1994). Using sophisticated microdialysis equipment Shah & Gilliams (2008) have identified extreme ischaemia, hypoxia and acidity, as features of the local tissues in which trigger points are found. Distinct differences were noted between these conditions in active trigger points, latent trigger points and normal muscle.
Travell & Simons (1983, 1992) have confirmed that the following factors can all help to maintain and enhance trigger point activity:
• nutritional deficiency, especially vitamins C, B complex and iron
• hormonal imbalances (low thyroid hormone production, menopausal or premenstrual situations)
• infections (bacteria, viruses or yeast)
• allergies (wheat and dairy in particular)
• low oxygenation of tissues (aggravated by tension, stress, inactivity, poor respiration).
Trigger point deactivation
A number of methods exist for the deactivation of such trigger points, ranging from use of pharmacological agents such as novocaine or xylocaine, to coolant sprays and acupuncture techniques. It is noteworthy that direct digital pressure techniques can also effectively deactivate trigger points – if only temporarily in many instances. Clinical experience has shown that an absolute requirement for trigger point deactivation (apart from removal of the causes) involves the need to restore the muscle in which the trigger point lies to its normal resting length.
Trigger point deactivation may be achieved by (Chaitow & DeLany 2000, Kuchera 1997, Travell & Simons 1992):
• inhibitory compression as used in osteopathic soft tissue manipulation, neuromuscular therapy and massage (see Boxes 3.5 and 3.6)
• chilling techniques (spray, ice, possibly combined with stretching – ‘spray & stretch’)
• acupuncture, injection, etc. (Baldry 1993)
• positional release methods such as strain/counterstrain (Chaitow & Delany 2000, Jones 1981, Ibáñez-García 2009)
• muscle energy (stretch) techniques (Simons et al 1999)
• myofascial release methods (Barnes 1997)
• combination sequences such as integrated neuromuscular inhibition technique (INIT) (Chaitow 1994)
• correction of associated somatic dysfunction possibly involving high-velocity thrust (HVT) adjustments and/or osteopathic or chiropractic mobilization method (Liebenson 1996)
• education and correction of contributory and perpetuating factors (posture, diet, stress, breathing habits, etc.) (Bradley 1999)
• self-help strategies (stretching, etc.) (Simons et al 1999)
• microcurrent applications (McMakin 1998)
• ultrasound (Lowe & Honeyman-Lowe 1999).
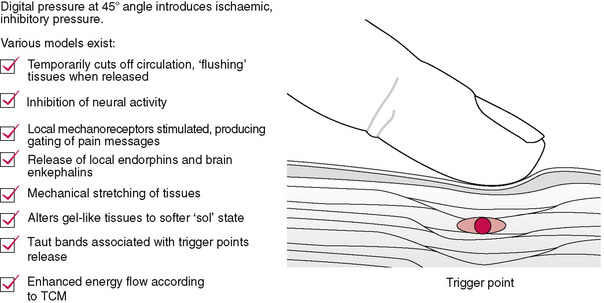
Box 3.5 Compression: method and effects (see Fig. 3.9)
 Method 1
Method 1
1. Apply firm digital compression to the trigger point sufficient to produce localized discomfort or pain, as well as symptoms in the target area.
2. Maintain this compression for 5 seconds.
4. Reapply pressure (same level) and keep repeating ‘5 seconds on, 2–3 seconds off’ until the patient reports a reduction in local or referred pain or an increase in pain (which is rare) or until 2 minutes have passed with no change in the pain level.
5. If using the INIT sequence (see Ch. 9), the next phase involves the positional release component.
Method 2
1. Apply firm digital pressure to the trigger point, sufficient to produce localized discomfort or pain, as well as symptoms in the target area.
2. Maintain the pressure for approximately 10 seconds.
3. Increase the degree of pressure slightly and maintain for a further 10 seconds.
4. Increase the degree of pressure once more and maintain for approximately 10 more seconds.
5. Slowly release pressure and, if utilizing the INIT sequence, the positional release component follows (see Ch. 9).
Manual contacts with trigger points
Tendons should be ignored when locating central trigger points. Only the actual length of the fiber is considered; for instance, the long tendon of either biceps brachii head is not included when assessing for central trigger points in this muscle; only the length of its belly is taken into account.
Central trigger points are usually palpable, with either flat palpation (against underlying structures), flat compression (between thumb and fingers like a clothes-peg) or pincer compression (between thumb and fingers like a C-clamp) (see upper trapezius treatment in Chapter 10).
A general thickening in the central portion of a muscle’s belly will usually soften when a broad general pressure is applied by using a flat compression. Compressions may be applied wherever the tissue may be lifted without compressing neurovascular bundles.
A precise compression of individual fibres is possible by using the more specific pincer palpation or flat palpation, both of which capture specific bands of tissue.
Underlying structures, including neurovascular courses which might be impinged or compressed, and sharp surfaces such as foraminal gutters, determine whether pincer or flat palpation is more appropriate.
Compression techniques, between fingers and thumb, have the advantage of offering information from two or more of the examiner’s digits simultaneously, whereas flat palpation against underlying tissues offers a more solid and stable background against which to assess the tissue.
Effects
The following effects are probable or possible during the application of sustained or intermittent digital compression of tissues:
• Ischaemia, which is reversed when pressure is released (Simons et al 1999).
• ‘Neurological inhibition’ results from sustained efferent barrage (Ward 1997). See Chapter 12 for details of the use of inhibitory pressure in a sequential manner when treating localized pain.
• A degree of mechanical stretching occurs as ‘creep’ of connective tissue commences (Cantu & Grodin 1992).
• Piezoelectric effects modify the ‘gel’ state of tissues to a more solute (‘sol’) state (Barnes 1997).
• Rapid mechanoreceptor impulses interfere with slower pain messages (‘gate theory’) (Wall & Melzack 1989).
• Pain-relieving endorphin and enkephalin release occurs (Baldry 2001), as does up-regulation of analgesic endocannabinoids (Klein 2005, McPartland & Simons 2007)
• Taut bands associated with trigger points release spontaneously (Simons et al 1999).
• Traditional Chinese medicine suggests modification of energy flow through tissues following pressure application (Zhao-pu 1991).
Box 3.6 Algometrics: using appropriate pressure
When applying digital pressure to a tender point in order to ascertain its status (‘Does it hurt?’, ‘Does it refer?’, etc.), or when treating a trigger point, it is important to have some way of knowing that the pressure being applied is uniform.
![]() It has been shown that, using a simple technology (bathroom scales), physical therapy students can be taught accurately to produce specific degrees of pressure on request. The students were tested applying posteroanterior pressure force to lumbar tissues. After training, using bathroom scales to evaluate pressure levels, the students showed significantly reduced error, both immediately after training and 1 month later (Keating et al 1993).
It has been shown that, using a simple technology (bathroom scales), physical therapy students can be taught accurately to produce specific degrees of pressure on request. The students were tested applying posteroanterior pressure force to lumbar tissues. After training, using bathroom scales to evaluate pressure levels, the students showed significantly reduced error, both immediately after training and 1 month later (Keating et al 1993).
The term ‘pressure threshold’ is used to describe the least amount of pressure required to produce a report of pain and/or referred symptoms (Hong et al 1996). It is obviously useful to know how much pressure is required to produce pain and/or referred symptoms, and whether this degree of pressure is different before and after treatment, or at a subsequent clinical encounter. Without a measuring device such as an algometer there would be no means of standardized pressure application.
![]() Use of an algometer is not really practical in everyday clinical work, but it becomes an important tool in research, as an objective measurement of a change in the degree of pressure required to produce symptoms. An algometer is also a useful tool for a practitioner to use in training themselves to apply a standardized degree of pressure when treating, and to ‘know’ how hard they are pressing.
Use of an algometer is not really practical in everyday clinical work, but it becomes an important tool in research, as an objective measurement of a change in the degree of pressure required to produce symptoms. An algometer is also a useful tool for a practitioner to use in training themselves to apply a standardized degree of pressure when treating, and to ‘know’ how hard they are pressing.
An algometer is a hand-held, spring-loaded, rubber-tipped, pressure-measuring device, which offers a means of achieving standardized pressure application. Using an algometer, sufficient pressure to produce pain is applied to points, at a precise 90° angle to the skin. The measurement is taken when pain is reported.
Baldry (referring to research by Fischer) discusses algometer use (he calls it a ‘pressure threshold meter’) and suggests it should be employed to measure the degree of pressure required to produce symptoms ‘before and after deactivation of a trigger point, for when this is successful, the pressure threshold over the trigger point increases by about 4 kg’ (Baldry 1993, Fischer 1988).
Stretching
Clinical experience suggests that failure to restore the muscle containing the trigger point to its normal resting length means that treatment is likely to provide only short-term relief.
It is vital to remember that a trigger point is self-perpetuating, unless it is treated correctly and sufficiently. This means that, once symptoms have been relieved, the muscle containing the trigger should be stretched gently to its longest resting length. Failing this, symptoms will return, irrespective of the technique used (chilling, pressure, injection, acupuncture, etc.).
Such stretching should be gradual and gentle, and the recommendation of Lewit (1992) and Travell & Simons (1992) is that muscle energy technique (MET), in which gentle isometric contractions followed by stretch are employed (see Ch. 8), is the method of choice to achieve that stretching. Lewit (1992) suggests that, in many instances, stretching is adequate in itself for deactivating trigger point activity.
Other views on trigger points
In order to gain further understanding of the significance of these widespread noxious entities, it is essential to be aware of the process of facilitation, as well as Selye’s general and local adaptation syndromes (see Chs 1 and 9 on clinical applications of NMT). We need, also, to be aware of views of others who have tried to make sense of the myriad systems that have identified patterns of reflex activity in surface tissues (as discussed in Ch. 4).
Mennell
Mennell (1975) agrees that a muscle that can attain and maintain its normal resting length is a pain-free muscle. One that cannot (a muscle in spasm) is usually a source of pain, regardless of whether the source of the spasm is in that muscle or not. Whatever the means used to ‘block’ the trigger activity, and whatever the neuropathological routes involved, the critical factor in the restoration of pain-free normality is that, during any relief from the state of spasm or contraction, the affected muscle should have its normal resting length restored by stretching. Mennell defines trigger points as localized palpable spots of deep hypersensitivity from which noxious impulses bombard the central nervous system to give rise to referred pain. Mennell favours chilling the trigger area by vapocoolant or ice-massage – an approach supported by Travell and Simons, who now both advocate MET as well. Details of their recommended methods are given in Chapter 8.
Chaitow
Chaitow (1994) has proposed that a sequence of treatment to achieve trigger point deactivation – commencing with palpation/identification utilizing NMT, followed by ischaemic compression (also NMT), followed by adoption of a positional release posture (such as is used in osteopathic functional technique or strain/counterstrain; see Ch. 8) – should be followed by a stretching of the tissues housing the trigger point. The stretching in this sequence can follow a focused (to activate the fibres involved) isometric contraction, or be applied at the same time as the contraction – introducing an isolytic muscle energy approach into the methodology (see Ch. 8).
This sequence has been dubbed ‘integrated neuromuscular inhibitor technique’ (INIT) and is considered further in the chapters that deal with treatment.
Chaitow and Delany
Chaitow & Delany (2001) have hypothesized that there are times when the activity of a trigger point might be regarded as serving a useful physiological role, and that in such circumstances deactivation would be counterproductive. Treatment of trigger points may then best be left until after correction of the adaptational mechanisms that have caused their formation. An example involves the stabilization of the sacroiliac joint during the gait cycle. As the right leg swings forward, the right ilium rotates backwards in relation to the sacrum (Greenman 1996). Simultaneously, sacrotuberous and interosseous ligamentous tension increases to brace the sacroiliac joint in preparation for heel strike. Just before heel strike, the ipsilateral hamstrings are activated, thereby tightening the sacrotuberous ligament (into which they merge) to further stabilize the sacroiliac joint. In the case of an unstable or dysfunctional sacroiliac joint, a persistent bracing mechanism, involving a hypertonic biceps femoris, possibly including trigger point activity, might represent a potentially useful attempt to maintain stability in the joint. Deactivation of the trigger point, and/or release of the tight hamstring, could in such circumstances reduce stability, with undesirable results (van Wingerden et al 1997).
Bradley
New Zealand physiotherapist Dinah Bradley epitomizes those practitioners who use trigger points as monitors, rather than treating them. Bradley, an expert in breathing rehabilitation, palpates and identifies key trigger points in the intercostals and upper trapezius as a rule, at the outset of the patient’s course of breathing rehabilitation. She asks them to ascribe a value out of 10 to the trigger point, while it is under digital pressure, before they commence their exercise and treatment programme (no direct treatment is given to the trigger points themselves), and periodically during the rehabilitation course, as well as at the time of discharge.
Bradley (1999) states:
I use trigger point testing as an objective measurement. Part of [the patient’s] recovery is a reduction in musculoskeletal pain in these overused muscles. I use a numeric scale to quantify this. Patients themselves feel the reduction in tension and pain [over time], a useful subjective marker for them, and an excellent motivator.
This use of trigger points, in which they are not directly deactivated but are used as monitors of improved breathing function, highlights several key points (Fig. 3.10).
It is possible to draw various conclusions from this approach:
• As function and oxygenation improve, trigger points become less reactive and painful.
• Enhanced breathing function represents a reduction in overall stress, reinforcing the concepts associated with facilitation: as stress (of whatever kind) reduces, trigger points react less actively.
• Direct deactivation tactics are not the only way to reduce trigger point activity.
• Trigger points can be seen to be acting as ‘alarm’ signals, virtually quantifying the current levels of adaptive demand being imposed on the individual, or on the specific tissues.
The stress burden and trigger points
One of Selye’s most important findings is commonly overlooked when the concurrent impact of multiple stressors on the system is being considered (Selye 1974).
Shealy (1984) summarizes as follows:
Selye has emphasized the fact that any systemic stress elicits an essentially generalized reaction, with release of adrenaline and glucocorticoids, in addition to any specific damage such stressor may cause. During the stage of resistance (adaptation), a given stressor may trigger less of an alarm; however, Selye insists that adaptation to one agent is acquired at the expense of resistance to other agents. That is, as one accommodates to a given stressor, other stressors may require lower thresholds for eliciting the alarm reaction. Of considerable importance is Selye’s observation that concomitant exposure to several stressors elicits an alarm reaction at stress levels that individually are sub-threshold. That is, one-third the dose of histamine, one-third the dose of cold, one-third the dose of formaldehyde, elicit an alarm reaction equal to a full dose of any one agent.
In short, therefore, as adaptation to life’s stresses and stressors continues, pain thresholds drop, and a lesser stress load is required to produce responses (pain, etc.) from facilitated structures, whether paraspinal or myofascial.
Hyperventilation: an example of compound stress influences
There are few more complex functional stress influences than those that arise from altered breathing patterns, such as are manifest when upper chest breathing and/or hyperventilation is habitual. Chaitow et al (2002) have expressed this as follows:
Consider someone who is habitually breathing in an upper chest mode (for postural, habitual, emotional or other reasons), the stress of which will place adaptive demands on the accessory breathing muscles, with consequent hypertonicity, shortness, reciprocal inhibition, malcoordination, stiffness and pain, almost certainly involving trigger point activity, as well as joint dysfunction.
• The biomechanical changes which evolve from this functional chaos will modify the structure of the breathing apparatus to the extent that normal breathing function becomes difficult or impossible.
• In addition the individual will probably display evidence of anxiety as a direct result of respiratory alkalosis, deriving from CO2 imbalance … caused by the breathing pattern. Or the breathing pattern may have been created because of a predisposing anxiety (Timmons 1994).
• This pattern of breathing, and the anxiety it encourages, feeds back into a cycle of aggravated upper chest breathing, reinforcing the pattern.
• What may have started as an emotional state will have evolved into a chronic biochemical imbalance and a state of relative biomechanical rigidity.
It is easy to see how [therapeutic] interventions which focus on the anxiety would be helpful, as would focus on the biomechanical/structural imbalances: Interventions which reduce anxiety will help all associated symptoms. Such interventions might also involve biochemical modification (medication etc.), stress coping approaches or psychotherapy.
Interventions which improve breathing function, probably involving easing of soft tissue distress (including deactivation of trigger points) and/or joint restrictions, should also help to reduce the symptoms by allowing, with retraining, a more normal breathing pattern to be practiced.
The most appropriate approach will be the one which most closely deals with causes rather than effects, and which allows for long term changes that will reduce the likelihood of recurrence. Biochemistry, biomechanics and the mind are seen, in this example, to be inextricably bonded with each other.
The lesson to be learned from this all-too-common example is that treating pain and dysfunction in the neck, shoulders and thorax of such an upper chest breather, with trigger points as major features, might usefully involve manual methods, or relaxation/psychotherapy (if appropriate), or rebreathing retraining, or a combination of methods.
Pathophysiology of fibromyalgia/fibrositis/myodysneuria
The changes that occur in tissue involved in the onset of myodysneuria/fibromyalgia, according to Gutstein (see earlier in this chapter), are thought to be initiated by localized sympathetic predominance, associated with changes in the hydrogen ion concentration and calcium and sodium balance in the tissue fluids (Petersen 1934). This is associated with vasoconstriction and hypoxia/ischaemia. Pain results, it is thought, from these alterations affecting the pain sensors and proprioceptors. Muscle spasm and hard nodular localized tetanic contractions of muscle bundles (Bayer 1950), together with vasomotor and musculomotor stimulation, intensify each other, creating a vicious cycle of self-perpetuating impulses. There are varied and complex patterns of referred symptoms that may result from such ‘trigger’ areas, as well as local pain and minor disturbances.
Such sensations as aching, soreness, tenderness, heaviness and tiredness may all be manifest, as may modification of muscular activity due to contraction resulting in tightness, stiffness, swelling, etc.
Recent research has resulted in strict guidelines for a diagnosis of fibromyalgia from the American College of Rheumatology (Wolfe et al 1990):
Pain is considered widespread when all of the following are present: pain in the left side of the body, pain in the right side of the body, pain above the waist and pain below the waist. In addition there should be pain in the spine or the neck or front of the chest, or thoracic spine or low back.
There should be pain on pressure (around 4 kg of pressure maximum) on not fewer than 11 of the following sites (Fig. 3.11):
• Either side of the base of the skull where the suboccipital muscles insert
• Either side of the side of the neck between the 5th and 7th cervical vertebrae – technically described as between the ‘anterior aspects of inter-transverse spaces’
• Either side of the body on the midpoint of the muscle that runs from the neck to the shoulder (upper trapezius)
• Either side of the body on the origin of the supraspinatus muscle that runs along the upper border of the shoulder blade
• Either side, on the upper surface of the rib, where the 2nd rib meets the breast bone, in the pectoral muscle
• On the outer aspect of either elbow just below the prominence (epicondyle)
• In the large buttock muscles, either side, on the upper outer aspect in the fold in front of the muscle (gluteus medius)
• Just behind the large prominence of either hip joint in the muscular insertion of piriformis muscle
• On either knee in the fatty pad just above the inner aspect of the joint.
Figure 3.11 Sites of the 18 fibromyalgia tender points as defined by the American College of Rheumatologists.
A puzzle
Questions often asked include:
• Is fibromyalgia the same as myofascial pain syndrome (pain problems in which trigger points are clearly involved)?
The condition called myofascial pain syndrome (MPS) – a disorder in which pain of a persistent aching type is referred to a number of target areas by triggers lying some distance away – has long been recognized as a cause of severe and chronic pain. Since many experts insist that the ‘tender’ points that are palpated when diagnosing fibromyalgia need to refer pain elsewhere if they are to be taken seriously in the diagnosis (making them trigger points by definition), the question needs to be asked whether MPS is not the same thing as fibromyalgia syndrome (FMS)?
Scandinavian researchers showed in 1986 that around 65% of people with fibromyalgia had identifiable trigger points, and it is clear, therefore, that there is an overlap between FMS and MPS.
D. P. Baldry, a leading British physician/acupuncturist, has summarized the similarities and differences between these two conditions (Baldry 1993) as follows. The two conditions are similar or identical in that both fibromyalgia and MPS:
• are affected by cold weather
• may involve increased sympathetic nerve activity and may involve conditions such as Raynaud’s phenomenon
• have tension headaches and paraesthesia as a major associated symptom
• are unaffected by anti-inflammatory pain-killing medication, whether of the cortisone type or standard formulations.
Fibromyalgia and MPS are different in that:
• MPS affects males and females equally; fibromyalgia mainly involves females.
• MPS is usually local to an area such as the neck and shoulders, or low back and legs, although it can affect a number of parts of the body at the same time, whereas fibromyalgia is a generalized problem, often involving all four ‘corners’ of the body at the same time.
• Muscles containing areas that feel ‘like a tight rubber band’ are found in around 30% of people with MPS, but in more than 60% of people with FMS.
• People with FMS have poorer muscle endurance than people with MPS.
• MPS can sometimes be bad enough to cause disturbed sleep, whereas in fibromyalgia the sleep disturbance has a more causative role, and is a pronounced feature of the condition.
• MPS produces no morning stiffness whereas fibromyalgia does.
• Fatigue is not usually associated with MPS, while it is common in fibromyalgia.
• MPS can sometimes lead to depression (reactive) and anxiety, whereas, in a small proportion of fibromyalgia cases, depression/anxiety can be the trigger for the start of FMS.
• Conditions such as irritable bowel syndrome, dysmenorrhoea and a feeling of swollen joints are noted in fibromyalgia, but seldom in MPS.
• Low-dosage tricyclic antidepressant drugs are helpful in dealing with the sleep problems, and many of the symptoms of fibromyalgia, but not of MPS.
• Exercise programmes (cardiovascular fitness) can help some patients with fibromyalgia, according to experts, but this is not a useful approach in MPS.
• The outlook for people with MPS is excellent because the trigger points usually respond quickly to massage, manipulative or acupuncture techniques, whereas the outlook for those with fibromyalgia is less positive, with a lengthy treatment and recovery phase being the norm.
Schneider (1995) reports:
It is now clear that there are several important differences between fibromyalgia (FM) and myofascial pain syndrome (MFPS) … local treatment applied to tender points [in FM] is ineffective, yet specific treatment of trigger points [in MFPS] is dramatically effective.
Simons (2002) has summarized the main differences between MPS and fibromyalgia (Table 3.2).
Table 3.2 Differences between MPS and fibromyalgia (Simons 2002)
| Myofascial trigger points | Fibromyalgia |
|---|---|
| Muscular origin | Systemic or central nervous system origin |
| Female:male ratio 1:1 | Female:male ratio 4–9:1 |
| Local or regional pain | Widespread, general pain |
| Focal tenderness | Widespread tenderness of most of body |
| Muscle feels tense (taut bands) | Muscle feels soft and doughy |
| Restricted stretch range of motion | Commonly hypermobile |
| Examination for trigger points anywhere | Examination for prescribed tender points |
| Immediate response to trigger point injection | Delayed response to trigger point injection |
| May also have fibromyalgia | Nearly all also have myofascial trigger points |
| All myofascial trigger points are tender | Not all tender points are myofascial trigger points |
Conclusion and hypothesis
We have seen how hyperreactive local (trigger point) and spinal areas can arise and be maintained and/or aggravated as a result of repetitive and continuous stress of one sort or another.
What we see in fibromyalgia is that areas of the brain behave in a facilitated manner, and that this hyperreactive brain activity could well be another version of this same phenomenon (facilitation). If so, we can learn much about fibromyalgia from our experience of handling localized facilitation processes such as trigger points.
Korr’s studies, discussed previously, show that facilitated areas act as ‘neurological lenses’, focusing whatever stress impacts on the person as a whole through these sensitized tissues.
If that ‘tissue’ happens to be (a part of) the brain, we have a situation in which it becomes imperative for stress of all sorts (climatic, emotional, structural/postural, nutritional, toxic, infective, allergenic, etc.) to be minimized, and this includes tailoring therapeutic interventions to be as non-invasive as possible (deep massage causes increased muscle pain in fibromyalgia, whereas light massage does not).
Constitutional therapeutic approaches such as deep relaxation, non-stressful hydrotherapy, wellness massage and similar methods are more likely to be helpful than anything that makes adaptive demands on an already compromised individual.
Summary
Trigger points are certainly often part – in some cases the major part – of the pain suffered by people with fibromyalgia. When they are present (as they certainly are if pressure on the ‘tender point’ produces pain somewhere else in the body), we need to know more about them and how they can be treated successfully.
In the next chapter an overview is presented of other reflex systems, awareness of which should add to our comprehensive understanding of myofascial trigger points.
Baldry P. Acupuncture trigger points and musculoskeletal pain. Edinburgh: Churchill Livingstone, 1993.
Baldry P. Myofascial pain and fibromyalgia syndromes. Edinburgh: Churchill Livingstone, 2001.
Barnes M. The basic science of myofascial release. Journal of Bodywork and Movement Therapies. 1997;1(4):231-238.
Barrall J.P. Manual–thermal diagnosis. Seattle: Eastland Press, 1996.
Bayer H. Pathophysiology of muscular rheumatism. Zeitschrift für Raeumaforschung. 1950;9:210.
Beal M. Palpatory testing of somatic dysfunction in patients with cardiovascular disease. J Am Osteopath Assoc. 1983;82(11):73-82.
Beal M.C. Viscerosomatic reflexes: a review. J Am Osteopath Assoc. 1985;85(12):786-801.
Benatsen L. Central sensitization in tension-type headache – possible pathophysiological mechanisms. Cephalalgia. 2000;20(6):603-610.
Bradley D. Breathing retraining advice from three therapists. Journal of Bodywork and Movement Therapies. 1999;3(3):159-167.
Brugger A. Pseudoradikulare syndrome. Acta Rheumatol. 1962;19:1.
Cantu R., Grodin A. Myofascial manipulation. Gaithersburg, Maryland: Aspen Publications, 1992.
Chaitow L. Integrated neuro-muscular inhibition technique in treatment of pain and trigger points. British Osteopathic Journal. 1994;XIII:17-21.
Chaitow L. Fibromyalgia syndrome – a practitioner’s guide to treatment. Edinburgh: Churchill Livingstone, 2000.
Chaitow L., DeLany J. Clinical applications of neuromuscular technique. vol. 2. Edinburgh: Churchill Livingstone; 2000. Lower body
Chaitow L., Bradley D., Gilbert C. Multidisciplinary approaches to breathing pattern disorders. Edinburgh: Churchill Livingstone, 2002.
Codere T., Katz J., Vaccarino A., et al. Contributions of central neuroplasticity to pathological pain: review of clinical and experimental evidence. Pain. 1993;52:259.
Cornelius A. Die neurepunkt lehre. vol. 2. Leipzig: George Thins; 1909.
Denslow J., Korr I., et al. Quantitative studies of chronic facilitation. Am J Physiol. 1949;150:229-238.
Dittrich R.J. Somatic pain and autonomic concomitants. Am J Surg. 1954.
Dowling D. DiGiovanna E., editor. An osteopathic approach to diagnosis and treatment. Philadelphia: Lippincott, 1991.
Dowling D. Progressive neuromuscular inhibition technique (PINS). J Am Osteopath Assoc. 2000;100(5):285-298.
Dvorak J., Dvorak V. Manual medicine diagnostics. Stuttgart: Georg Thieme, 1984.
Fairbank J. Prolapsed intervertebral disc. BMJ. 2008;336(7657):1317-1318.
Feinstein B., Longton J., Jameson R., et al. Experiments on pain referred from deep somatic tissues. J Bone Joint Surg. 1954;36A:981.
Fischer A. Documentation of muscle pain and soft tissue pathology. In: Kraus H., editor. Diagnosis and treatment of muscle pain. Chicago: Quintessence, 1988.
Greenman P.E. Principles of manual medicine, ed 2. Baltimore: Williams & Wilkins, 1996.
Gutstein R. A review of myodysneuria (fibrositis). Am Pract Dig Treat. 1944;6(4):114-124.
Gutstein R. The role of abdominal fibrositis in functional indigestion. Miss Valley Med J. 1944;66:114-124.
Gutstein R. The role of craniocervical myodysneuria in functional ocular disorders. Am Pract Dig Treat November. 1956.
Hix E. Reflex viscerosomatic reference phenomena. Osteopathic Annals. 1976;4(12):496-503.
Hockaday M., Whitty C.W. Patterns of referred pain in the normal subject. Brain. 1967;90:481-496.
Hong C.Z., Chen Y.N., Twehouse D., et al. Pressure threshold for referred pain by compression on trigger point and adjacent area. Journal of Musculoskeletal Pain. 1996;4(3):61-79.
Ibáñez-García J., et al. Changes in masseter muscle trigger points following strain-counterstrain or neuro-muscular technique. Journal of Bodywork and Movement Therapies. 2009;13(1):2-10.
Ji R.R., Kohno T., Moore K.A., et al. Central sensitization and LTP: Do pain and memory share similar mechanisms? Trends Neurosci. 2003;26(12):696-705.
Jones L. Strain and counterstrain. Colorado Springs: Academy of Applied Osteopathy, 1981.
Keating J., Matuyas T., Bach T. The effect of training on physical therapists’ ability to apply specified forces of palpation. Phys Ther. 1993;73(1):38-46.
Kellgren J. Observations on referred pain coming from muscle. Clin Sci. 1938;3:175-190.
Kellgren J. On the distribution of pain arising from deep somatic structures. Clin Sci. 1939;4:35-46.
Klein T. Cannabinoid-based drugs as anti-inflammatory therapeutics. Nat Rev Immunol. 2005;5(5):400-411.
Korr I. Physiological basis of osteopathic medicine. New York: Postgraduate Institute of Osteopathic Medicine and Surgery, 1970.
Korr I. Proprioceptors and somatic dysfunction. J Am Osteopath Assoc. 1975;74:638.
Korr I. Spinal cord as organiser of disease process. Academy of Applied Osteopathy Yearbook. 1976.
Korr I., editor. Neurobiological mechanisms in manipulation. New York: Plenum Press, 1977.
Korr I. Somatic dysfunction. J Am Osteopath Assoc. 1986;86(2):109-114.
Kuchera M. Travell & Simons myofascial trigger points. In: Ward R., editor. Foundations of Osteopathic Medicine. Philadelphia: Williams & Wilkins, 1997.
Kuchera M., McPartland J. Myofascial trigger points. In: Ward R., editor. Foundations of Osteopathic Medicine. Philadelphia: Williams & Wilkins, 1997.
Lavelle W., Lavelle E., Smith H. Interventional Techniques for Back Pain. Clin Geriatr Med. 2008;24(2):345-368.
Lewis T. Suggestions relating to the study of somatic pain. Br Med J. 1938;1:321-325.
Lewit K. Manipulative therapy in rehabilitation of the locomotor system. London: Butterworths, 1992.
Li J., Simone D., Larson A. Windup leads to characteristics of central sensitization. Pain. 1999;79:75-82.
Liebenson C. Rehabilitation of the spine. Baltimore: Williams & Wilkins, 1996.
Lowe J.C., Honeyman-Lowe G. Ultrasound treatment of trigger points. Medecine du Sud-est. 1999;2:12-15. April–May–June
McEachern J.C., Shaw C.A. The plasticity-pathology continuum: Defining a role for the LTP phenomenon. J Neurosci Res. 1999;58(1):42-61.
McMakin C. Microcurrent treatment of myofascial pain in head, neck and face. Topics in Clinical Chiropractic. 1998;5(1):29-35.
McPartland J., Simons D. Myofascial trigger points: translating molecular theory into manual therapy. Journal Manual and Manipulative Therapies. 2007;14:232-239.
Melzack R., Wall P. Textbook of pain, ed 2. London: Churchill Livingstone, 1989.
Mennell J. The therapeutic use of cold. J Am Osteopath Assoc. 1975;74:1146-1158.
Mense S., Simons D. Muscle pain. Philadelphia: Williams & Wilkins, 2001.
Patterson M. Model mechanism for spinal segmental facilitation. 1976. Academy of Applied Osteopathy Yearbook,
Petersen W. The patient and the weather: autonomic disintegration. Ann Arbor: Edward Bros, 1934.
Schneider M. Tender points – fibromyalgia vs. trigger points – myofascial pain syndrome. J Manipulative Physiol Ther. 1995;18(6):398-406.
Selye H. Stress without distress. Philadelphia: Lippincott, 1974.
Shah J., Gilliams E. Uncovering the biochemical milieu of myofascial trigger points using in vivo microdialysis: An application of muscle pain concepts to myofascial pain syndrome. Journal of Bodywork and Movement Therapies. 2008;12(4):371-384.
Shealy C.N. Total life stress and symptomatology. Journal of Holistic Medicine. 1984;6(2):112-129.
Simons D. Myofascial pain due to trigger points. International Rehabilitation Medicine Association. 1987. Monograph 1,
Simons D. Myofascial pain syndromes. Journal of Musculoskeletal Pain. 1994;2(2):113-121.
Simons D. Understanding effective treatments of myofascial trigger points. Journal of Bodywork and Movement Therapies. 2002;6(2):81-88.
Simons D., Travell J., Simons L. ed 2. Myofascial pain and dysfunction: the trigger point manual. vol. 1. Baltimore: Williams & Wilkins; 1999. Upper half of body
Speransky A. A basis for the theory of medicine. New York: International Publishers, 1943.
Staud R. Biology and therapy of fibromyalgia: pain in fibromyalgia syndrome. Arthritis Res Ther. 2006;8:208.
Sutter M. Versuch einer Wesensbestimmung pseudoradikularer Syndrome. Schweiz Rundsch Med Prax. 1975;63:42.
Swerdlow B., Dieter N. Evaluation of thermography. Pain. 1992;48:205-213.
Timmons B. Behavioral and psychological approaches to breathing disorders. New York: Plenum Press, 1994.
Travell J. Symposium on mechanism and management of pain syndromes. 1957. Proceedings of the Rudolph Virchow Medical Society
Travell J. Identification of myofascial trigger point syndromes. Arch Phys Med. 1981;62:100.
Travell J., Bigelow N. Role of somatic trigger areas in the patterns of hysteria. Psychosom Med. 1947;9(6):353-363.
Travell J., Simons D. ed 1. Myofascial pain and dysfunction: the trigger point manual. vol. 1. Baltimore: Williams & Wilkins; 1983. Upper half of body
Travell J., Simons D. Myofascial pain and dysfunction: the trigger point manual. vol. 2. Baltimore: Williams & Wilkins; 1992. The lower extremities
Travell J., Simons D., Travell L. ed 2. Myofascial pain and dysfunction: the trigger point manual. vol. 1. Baltimore: Williams & Wilkins; 1999. The upper extremities
Van Wingerden J-P., Vleeming A., Kleinvensink G., Stoekart R. The role of the hamstrings in pelvic and spinal function. A. Vleeming, V. Mooney, T. Dorman. 1997.
Wall P., Melzack R. Textbook of pain. Edinburgh: Churchill Livingstone, 1989.
Ward R. Foundations of osteopathic medicine. Philadelphia: Williams & Wilkins, 1997.
Webber T.D. Diagnosis and modification of headache and shoulder-arm-hand syndrome. J Am Osteopath Assoc. 1973;72:697-710.
Weiss J. Pelvic floor myofascial trigger points: manual therapy for interstitial cystitis and the urgency-frequency syndrome. J Urol. 2001;166(6):2226-2231.
Wolfe F., Smythe H.A., Yunus M.B., et al. The American College of Rheumatology 1990 Criteria for the Classification of Fibromyalgia. Arthritis Rheum. 1990;33:160-172.
Zhao-Pu W. Acupressure therapy. Edinburgh: Churchill Livingstone, 1991.