CHAPTER 10 Liver and spleen
Clinical aspects
Liver
The place of FNA in the investigative sequence
Malignancy in the liver, primary or metastatic, is usually inoperable at the time of diagnosis and, as such, portends an ominous prognosis. A diagnostic modality such as FNA, which offers accuracy without significant complications and which requires minimal intervention at low cost, warrants consideration early in the investigative sequence. Portal vein FNA sampling is feasible in patients with hepatocellular carcinoma (HCC) and may be considered as a staging option.1-3 FNA may become useful after local ablative therapy of liver tumors to determine residual viable tumor.4,5 In this era of liver transplantation there is some reluctance in performing FNA in patients who are being worked up for a transplant because of the risk of needle track implantation in the setting of immunosuppression, although this is disputed by some.6-10 Single or multiple focal abnormalities demonstrated by palpation, ultrasonography (US), computed tomography (CT) or nuclear scan constitute the main indications for FNA of the liver. In industrialized countries, metastatic tumor deposits are the most common cause of focal abnormalities. In developing countries, however, hepatocellular carcinoma (HCC) is a major health problem, and is more likely to be encountered in FNA. Approximately 80% of the annual global burden of 560 000 new cases of HCC occurs in Africa and Asia. In these areas, etiology is frequently the hepatitis B virus, with superadded aflatoxin ingestion. HCC in developed countries arises in a background of cirrhosis consequent on chronic alcohol abuse or, more recently, hepatitis C virus.11
The differential diagnosis of hepatic mass lesions includes primary liver tumors, benign or malignant, metastatic deposits, congenital and acquired cysts, abscesses and granulomas. Single radiolucencies in the liver are not infrequently found at US scanning of asymptomatic patients. The question raised is that of metastatic cancer versus a primary liver lesion such as hemangioma.
Some workers have advocated the use of FNA in diffuse parenchymal liver disease.12-14 Most physicians feel that cytological methods lack the precision necessary to be of diagnostic value in processes such as cirrhosis, hepatitis and drug-induced effects.15,16 The pathologist must nevertheless be familiar with the hepatic changes which may be present as a local reaction adjacent to space-occupying lesions, and with the severe parenchymal abnormality which may represent the cirrhotic background of HCC. The cytological features of normal liver, and various changes associated with diffuse chronic liver disease have been described.12-14,16-19
FNA can diagnose certain diffuse processes such as hemosiderosis, amyloidosis and myeloid metaplasia. Metastatic melanoma or carcinoma, particularly of breast and lung, and malignant lymphoma can occasionally cause hepatomegaly by diffuse infiltration, without demonstrable focal abnormalities. These infiltrates may be sampled and can be diagnosed by FNA techniques. FNA has also been used for monitoring liver transplants.20-25
Advantages of FNA over conventional core biopsy are: it is less invasive and therefore leads to fewer complications, sampling can be performed over a wide area, either superficial or deep lesions can be targeted, small nodules high in the dome of the right lobe can be sampled, multiple sites including both left and right lobes can be sampled, immediate triage for adequacy can be performed limiting the number of needle passes, and hospitalization is not required. The disadvantages are: the material is limited with less architectural information, and a skilled radiologist and a skilled cytopathologist are required for optimal results. Small focal abnormalities can be missed by either method. Morphologic features must therefore always be correlated with clinical presentation and radiologic findings to determine whether the liver lesion has been fully explained by either modality.
Accuracy of diagnosis
Most studies comparing core needle biopsy and FNA favor the fine needle for focal liver disease,26-28 although not all,29 and generally a combination of the two yields the best results.28,30 Diagnostic sensitivity of FNA in malignancy is usually to around 90% (range 67–100%).26,28,30-36 Cell typing can sometimes be problematic in smears and many centers therefore advocate the additional use of cell blocks for histological assessment and providing multiple sections for immunocytochemistry.32,37 The use of cell blocks improved reported accuracy of hepatocellular carcinoma typing from around 85%38,39 to 95%.40,41 Certain groups report the combined use of aspirates and tissue cores for more definitive diagnosis of focal liver lesions.30 Recent use of the endoscopic ultrasound guided approach to FNA of the liver appears not only to maintain the high sensitivity and specificity of liver FNA, but also to identify and target lesions too small to be detected by conventional transabdominal ultrasound and CT.42-44 Whichever modality is used for sampling, we strongly advise the on-site participation of personnel from cytopathology to ensure optimal handling of the sample, rapid interpretation, and triage of the specimen as required.
There are pitfalls which the cytopathologist must be aware of. These include: the clinical history or imaging appearance may be misleading, the sample may be of the liver adjacent to the lesion and therefore non-representative, contamination from structures traversed on the way to the lesion, e.g. stomach, bowel or mesothelium, may predominate.
Complications
No deaths and only one significant complication, an intrahepatic hematoma, occurred in an early report of 2611 cases.45 A later summary of 7500 FNAs from 11 series recorded no deaths.46 In an extensive literature review and questionnaire study,47 Smith collated fatalities following abdominal percutaneous FNA. Of 21 deaths involving liver FNA, he noted that 17 were due to hemorrhage. A needle larger than 0.8 mm (21 gauge) was used in seven. Three of the other 10 followed FNA of vascular tumors (hemangioma 1, angiosarcoma 2). Fatalities due to other causes were rare (sepsis 2, carcinoid crisis 1, uncertain 1). More recently, two deaths due to hemorrhage from among 1750 US guided FNA has been reported.48
Allegations of needle track spread of tumor have always dogged FNA, and the liver is no exception in this regard. Of recent concern has been a number of literature reports of subcutaneous seeding of needle tracks from otherwise operable HCCs.47,49-51 Whereas in some of these reports the procedures comply in every respect with the usual safety precautions advised (0.8-mm/21-gauge needles or smaller, few needle passes, traversal of normal liver parenchyma between abdominal wall and tumor),52 other reports often included in discussion of this issue have used 18-gauge needles,53 have traversed one lobe to reach a lesion in the other lobe and have not defined needle size,54 or are series in which cutting55 or Trucut56 needles have been used. Cytopathologists and radiologists performing hepatic FNA should discuss and be very clear about the caliber of needles used.
In those cases in which potentially resectable or transplantable HCC is being investigated, local opinion might preclude preoperative sampling.9 This is despite studies that show FNA has no significant adverse effect on operability, extrahepatic metastases or long-term survival.6,8,57 Experience with cases of needle track spread has shown that local treatment by resection has been successful for isolated subcutaneous tumor deposits.47,49,53-56 In the vast majority of hepatic aspiration procedures, physicians and patients can be assured of very low complication rates (hemorrhage 0.006–0.031%, needle track spread 0.007%).47
Contraindications
Hemorrhagic diathesis and anticoagulation are contraindications, and the prothrombin time and the platelet count should be checked before the FNA. The patient should be kept under observation for a few hours after biopsy to monitor pulse, blood pressure and abdominal pain. Hydatid cyst was formerly considered to be a relative contraindication in view of the risk of an anaphylactic reaction.58 However, no allergic reactions have been observed in two series of 11 hydatid cysts aspirated inadvertently.59,60 The single documented case of anaphylactic shock, which was reversed, occurred when an 18-gauge needle was used.58 The risk of hemorrhage by needling of vasoformative tumors exists, but is low if correct techniques are used. This is no longer considered an absolute contraindication.61-64
Technical considerations
Whenever possible, the biopsy should be directed rather than blind. If a mass cannot be clearly felt, the biopsy must be guided by radiological imaging. US is the most commonly used modality.31,32 More recently, clinicians and radiologists have been using endoscopic ultrasound to target some liver lesions, in particular those involving the hilum and left lobe. Great technical expertise is required for good results, and is acquired after significant experience.43,44,65-67
Ideally, a cytopathologist or cytologist attends the FNA procedure for rapid review to confirm diagnostic material and to triage the material.68 Rapid staining (Diff-Quik or H&E) allows immediate screening of samples. The parallel use of MGG and Pap staining is strongly recommended for permanent smears of liver aspirates. Most adjunctive cytochemical or immunocytochemical tests are more easily performed on cell blocks, but some may be useful in smears or in cytocentrifuged and thin-layer preparations. Additionally, demonstration of architectural patterns in sections of tissue fragments increases diagnostic accuracy significantly.37,40,69-72 If immediate assessment suggests an acute, chronic or granulomatous inflammatory process, material for microbiological assessment should be collected in a sterile container. If a lymphoproliferative disorder is suggested, a separate needle pass should be collected into RPMI or normal saline for flow cytometry. Cytogenetic FISH testing, e.g. for specific translocations, can be performed on cytospin preparations and molecular studies, e.g. for lymphoma clonality, can be conducted on material from cell blocks. Electron microscopy may also contribute to type-specific tumor diagnosis.70,73 These adjunctive techniques ensure that liver aspirates are highly suited to sophisticated cytohistological interpretation. A core biopsy should occasionally be requested, particularly if architectural assessment is crucial to the diagnosis e.g. hepatic adenoma versus hepatocellular carcinoma. If radiology staff are to prepare the sample they should be trained, appropriate consumables should be supplied and collection of material into fluid for a cell block or cytospin preparations should be encouraged.
Cytological findings
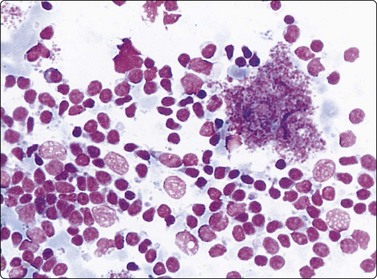
Normal structures (Fig. 10.1)
Cytologic features
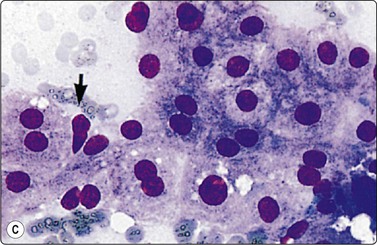
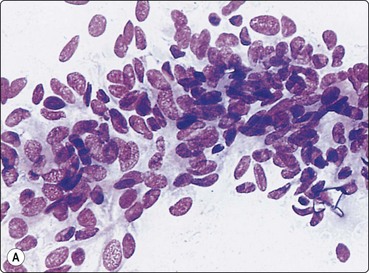
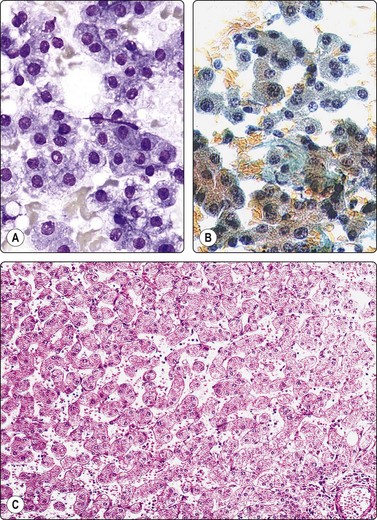
(A) Irregular cohesive sheet showing a trabecular arrangement (Pap, IP); (B) Cohesive groups of polygonal cells with abundant sometimes vacuolated granular cytoplasm (Pap, HP); (C) Cohesive cells with central round nuclei, granular cytoplasm and indistinct cell borders; note the small wedge-shaped Kupffer cell (arrow) (MGG, HP).
Although the aspirate may appear to consist mainly of blood, it usually contains many liver cells. Cell cohesion is particularly strong, and when the needle content is placed on a slide one can often see small, firm, semitranslucent tissue fragments which are difficult to spread. In comparison, tumor tissue fragments are usually soft, fragile and easily smeared.
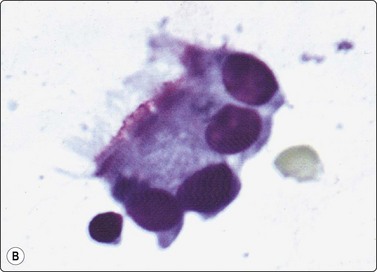
In smears, normal hepatocytes form irregular, cohesive sheets and narrow cords 1–2 cells thick (Fig. 10.1A). Single cells are infrequent. The cells are large, round or distinctly polygonal in outline with abundant granular cytoplasm, sometimes vacuolated, staining eosinophilic with H&E, blue with Papanicolaou or gray–blue with MGG (Fig. 10.1B). Nuclei are centrally located, round, with finely granular chromatin which is evenly distributed. Binucleate cells are common. Nucleoli are central and usually small although prominent. The cell membrane is indistinct. Normal hepatocytes may display mild to moderate anisokaryosis due to polyploidy, increasing with the patient’s age. Occasional nuclei may be very large in non-neoplastic liver tissue. Coarse, granular intracytoplasmic pigment is commonly present in hepatocytes. It stains green–black with MGG, brown with H&E and is probably lipofuscin rather than bile. Kupffer cells can usually be found and appear as single, bare, comma-shaped nuclei between the hepatocytes. Bile duct epithelial cells form small, regular monolayered sheets. They are palisaded when seen from the side. The cytoplasm is minimal, nuclear chromatin is granular and nucleoli are inconspicuous. These are few in number in samples from normal liver tissue.
Diffuse parenchymal disease (Figs 10.2-10.9)
Cytologic features
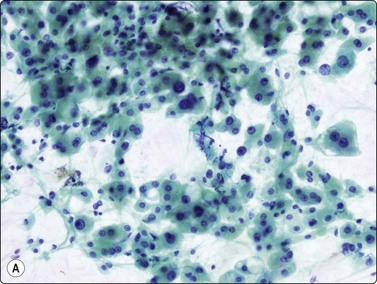
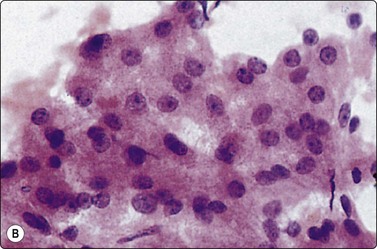
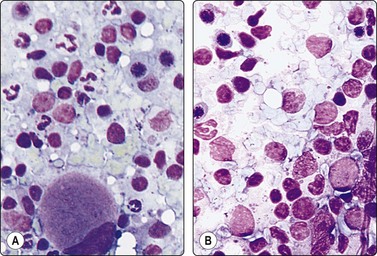
Fig. 10.2 Hepatocyte reactive changes
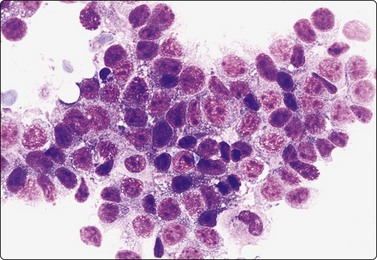
(A) Cells showing decreased cohesion, anisokaryosis and well-defined cell borders; note the scattered Kuppfer cells and lymphocytes (Pap, IP); (B) A sheet of cells showing mild disorganization and prominent nucleoli (H&E, HP).
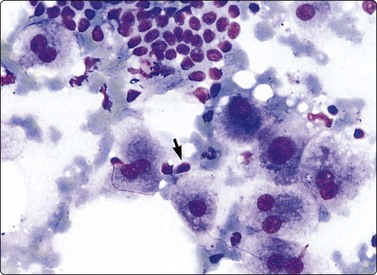
Poorly cohesive hepatocytes with degenerative/regenerative features; a sheet of small cohesive bile duct cells; note several Kupffer cells (arrow) (MGG, HP).
As already stated, FNA is not primarily suited to diagnosis of non-neoplastic, parenchymal liver disease.15 However, it is essential that the cytologist recognizes these changes, as they may occur in the region of space-occupying lesions and may themselves mimic neoplasia.
The presence of parenchymal disease is indicated by decreased cohesion of hepatocytes. Smears may be hypercellular and appear quite pleomorphic due to anisocytosis. The hepatocytes are swollen to a variable degree, and the cytoplasm stains less uniformly than usual, paler at the periphery of the cell. Cell membranes which are indistinct in normal hepatocytes become more clearly visible (Fig. 10.2A). Fatty change is commonly seen as intracytoplasmic vacuoles of variable size displacing the nucleus to the periphery. Despite nuclear pleomorphism and binucleation, the chromatin pattern remains finely granular. Intranuclear cytoplasmic invaginations are seen in some nuclei, large prominent nucleoli in others (Fig. 10.2B). There are no atypical bare hepatocyte nuclei in the background. Liver cell necrosis may be obvious in severe disease. Acidophilic bodies and alcoholic hyaline are difficult to identify in cytological preparations.12,75 The number of lymphocytes and of Kupffer cells is increased. The Kupffer cells often appear swollen and may cluster. The number of sheets of bile duct epithelium is often increased, particularly in cirrhosis (Fig. 10.3).
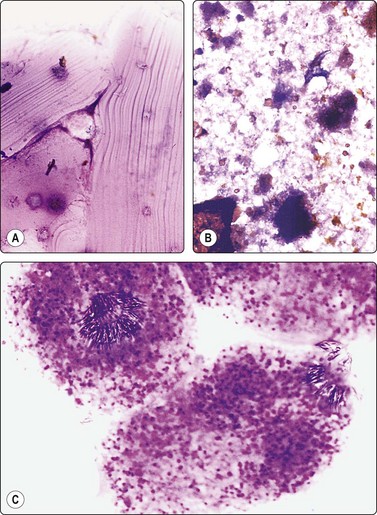
The degree of parenchymal abnormality varies widely. The reactive change commonly seen adjacent to a neoplasm is an example of mild change, including some dissociation and swelling of hepatocytes and mild to moderate anisokaryosis. In cirrhosis, which may mimic neoplasia radiologically as well as cytologically,76 there is a marked increase in the number of Kupffer cells and of bile duct epithelial cells, fatty change, and dissociation of hepatocytes. Bile stasis is easily recognized as casts of dense material between hepatocytes, staining black–green with MGG (Fig. 10.4), yellow–green with Pap or H&E. Nuclear changes and nucleolar enlargement may be quite prominent. In so-called liver cell dysplasia associated with cirrhosis, there is a dominant background of normal hepatocytes among which are scattered atypical cells with considerably enlarged nuclei. The fibrosis of cirrhosis is not often evident on routinely stained smears, but can be demonstrated on smears or on cell block material using a connective tissue stain. Normal liver will demonstrate a fine regular network of reticulin. Cirrhosis is characterized by a disorganized but still intact network (Fig. 10.5). HCC typically shows fragmentation or a complete absence of reticulin framework.77,78
Hemosiderosis is recognized by the demonstration of coarse, refractile hemosiderin pigment – golden brown in Papanicolaou, black in Romanowsky stains – in the hepatocytes (Fig. 10.6) and in the Kupffer cells. It is not always easy to distinguish hemosiderin from lipofuscin or even from bile in routinely stained smears; a suitable special stain such as Prussian blue should be used if hemosiderosis is suspected.14,79 Further, with malignant transformation, iron pigment is lost. Prussian blue negative clusters can be regarded with suspicion in the setting of a mass arising in a background of siderosis.80 Amyloidosis is not common but may cause massive hepatomegaly and is diagnosable cytologically. Amyloid appears in smears as a dense amorphous, waxy material, which stains pink with H&E (Fig. 10.7), magenta with MGG and orangeophilic with Papanicolaou stain. Its presence should be confirmed with special stains such as Congo red. The hepatocytes often appear atrophic.81-84
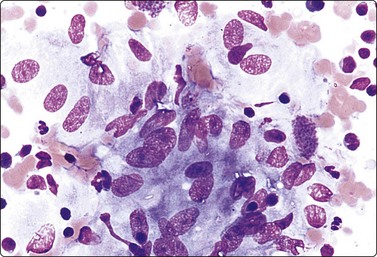
The finding of granulomatous inflammation may either be incidental or represent the lesion being aspirated but does not permit a specific diagnosis.85 Cytological features include loose aggregates of epithelioid histiocytes which have large elongated plump nuclei, finely granular chromatin, small distinct nucleoli, abundant, pale-staining cytoplasm, Langhans and/or foreign body type giant cells and non-specific inflammation (Fig. 10.8). Caseous necrosis may be seen. The etiology most likely to present as a mass, particularly in the Third World, is infection such as mycobacteria.86 Material should be collected appropriately for culture and special stains to demonstrate the acid-fast bacilli.12 Failing this, PCR for mycobacteria can be performed on fresh or fixed material. The list of differential diagnoses for granulomata in the liver is long and their demonstration in FNA smears requires clinical correlation and further investigation to determine their exact etiology.
Non-neoplastic focal lesions
Cysts
Congenital (developmental) cysts
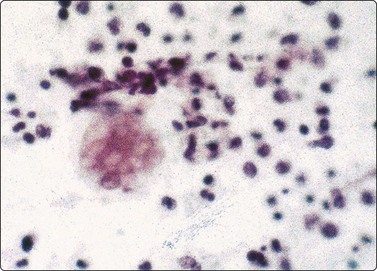
An aspirate from a congenital cyst, in reality a dilatation of a biliary duct, consists of thin, clear fluid but may be bloody or bile stained. A few uniform, cuboidal epithelial cells, single or in monolayered sheets, and some macrophages with vacuolated cytoplasm may be found in the smears (Fig. 10.9A).87 The columnar cells may balloon if degenerate. The ciliated hepatic foregut cyst can be diagnosed cytologically by observing clusters of ciliated columnar cells in the cyst milieu (Fig. 10.9B).88,89
Criteria for diagnosis
Hydatid cyst
Routine aspiration of a hydatid cyst is not recommended because of the theoretical risk of anaphylaxis should the cyst contents spill into the abdomen.62,90
Criteria for diagnosis59,90-92
Abscess (Fig. 10.11)
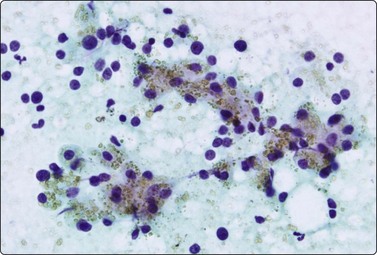
Purulent material aspirated from a focal lesion should always be subjected to microbiological investigation. Routine smears should also be prepared and screened for neoplastic cells, as tumor metastases, for example from a primary bowel adenocarcinoma, can undergo central necrosis, simulating an abscess. Pyogenic abscess demonstrates a marked neutrophil infiltrate, with necrotic debris.93 Hepatocytes from the periphery of the abscess may show considerable atypia.76 The predominant organism in pyogenic abscesses is Klebsiella.93 Amebic abscess due to Entamoeba histolytica, with contents likened to anchovy paste (thick reddish-brown semi-fluid material), shows abundant necrosis with fewer inflammatory cells. Trophozoites are not found in the central necrotic area; they should be sought in the viable periphery of the abscess (Fig. 10.11).93,94 They are globular organisms, with an eccentric spherical nucleus showing a central clear zone with a dot-like karyosome and margination of chromatin. The cytoplasm is vacuolated and contains ingested red blood cells. Amebae stain positive with PAS. They resemble foamy macrophages and may be missed if not suspected. The diagnosis of hepatic actinomycosis by FNB has been reported.95,96 It occurs in a background of systemic actinomycosis, secondary to pulmonary or uterine primary sources of infection.
Focal nodular hyperplasia
(FNH) is a solitary tumor-like lesion of uncertain histogenesis but benign prognosis. It occurs at all ages, predominantly in middle life and in women. Most FNH are detected incidentally in otherwise normal livers. Radiologically and macroscopically, the lesion shows a central radiating scar separating nodules of hepatocytes.
Distinction from low-grade hepatocellular tumors such as adenoma and well-differentiated hepatocellular carcinoma can be problematic and cell block or core biopsy with a reticulin stain may provide useful architectural information (see section on Hepatocellular carcinoma: Problems and differential diagnoses).
Other non-neoplastic lesions
Focal fatty change in the liver can simulate neoplasia on radiologic imaging, and diagnosis by FNA is of practical importance.97,98 It is less common than the diffuse form and the etiology is uncertain. The hepatocytes have normal nuclei and the majority contain multiple or single clear vacuoles, bile duct epithelium may be present and the reticulin framework may be distorted by the fat droplets.
Inflammatory pseudotumor is a distinct entity increasingly being identified radiologically and thus entering the ambit of cytodiagnosis. The liver is probably the second most common site after lung for its occurrence.99 It is a localized lesion of uncertain etiology, representing an atypical immune response or a hypersensitivity reaction to antigen. Histological variants include inflamed granulation tissue, xanthogranuloma, hyaline sclerosing variant and plasma cell granuloma. FNAC can be expected to be equally variable. Hepatocytes and bile duct cells, which can show atypia and plasma cells, lymphocytes, neutrophils ± eosinophils and/or xanthoma cells in a background of fibroblasts, create a mixed picture which should not be overdiagnosed as malignant. Single large abnormal cells with lobulated vesicular nuclei resembling Reed-Sternberg cells have been reported.100-102
Nodular extramedullary hemopoiesis occurs typically in the setting of myeloproliferative disease and is recognized by the criteria of the triple cell lines – normoblasts, promyelocytes and megakaryocytes.103-105 The last mentioned could be mistaken for bizarre malignant cells.
Primary hepatic tumors of epithelial origin
Hepatobiliary cystadenoma
These solitary multiloculated lesions may reach sizes over 20 cm. They are encountered almost exclusively in females, generally of middle age.107,108 They are composed of a bland, cuboidal to columnar, nonciliated mucin-secreting epithelium, overlying dense ovarian-like stroma. The cyst contents are mucoid and may show raised CEA and CA 19-9 levels on biochemical testing.87,107,109
Criteria for diagnosis108
Tumors have a definite malignant potential and any solid, nodular or polypoid region seen on imaging should be sampled.110-114
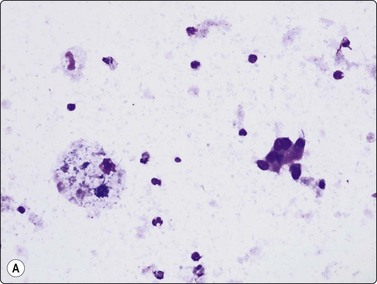
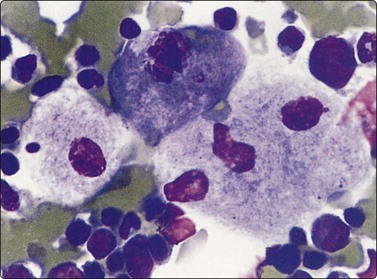
Liver cell adenoma (Fig. 10.13)
Cytodiagnosis of this entity can hardly be made with confidence by cytology alone and a core biopsy can provide additional architectural information. The cytological pattern must be correlated with clinical presentation and radiological findings.115 A solitary liver mass found in a young woman with a history of oral contraceptive usage is most likely a hepatocellular adenoma, whereas a mass in an older patient with cirrhosis is more likely either a regenerative nodule or a hepatocellular carcinoma.
Poorly cohesive epithelial cells resembling hepatocytes, bland nuclei, Kupffer cells but no bile duct epithelium (A, H&E, IP; B, Pap, HP); (C) Corresponding tissue section (H&E, IP).
Criteria for diagnosis46
Problems and differential diagnoses
Adenoma is difficult to differentiate from other low grade hepatocellular lesions, ie FNH and well differentiated hepatocellular carcinoma.
The differences in cytological patterns between adenoma and well-differentiated carcinoma can be subtle and will be discussed in the section on hepatocellular carcinoma. The cytoplasm is more abundant, less fragile and better defined with more distinct cell borders in adenoma, the nuclear : cytoplasmic ratio is consistently low, and single, bare neoplastic nuclei are less apparent (Fig. 10.13). Marked anisokaryosis is not a feature. Adenoma and FNH both comprise benign hepatocytes. In the absence of abundant bile duct epithelium, which favors FNH, distinction between them is not possible.41,106
Hepatocellular carcinoma (Figs 10.14-10.27)
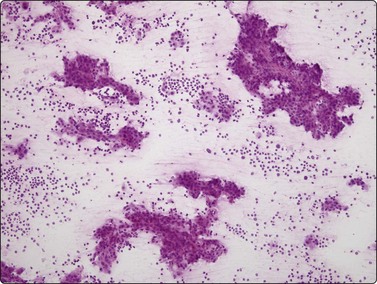
Fig. 10.14 Hepatocellular carcinoma
Cellular smears with irregular large fragments, clusters and dispersed cells (H&E, LP).
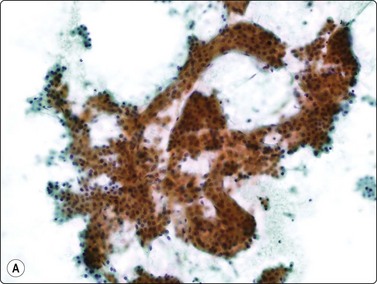
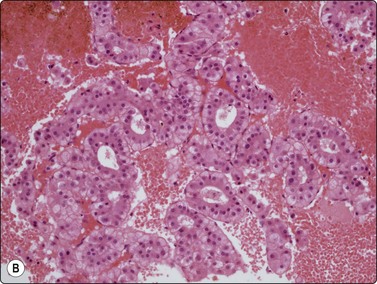
Fig. 10.15 Hepatocellular carcinoma
(A) Cells are typically arranged in widened trabeculae (Pap, IP); (B) Less commonly in acinar arrangements (cell block H&E, IP).
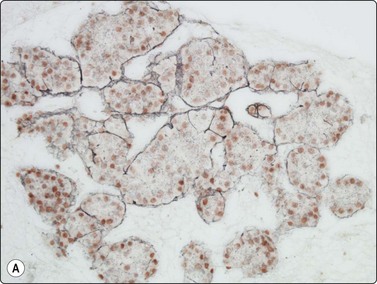
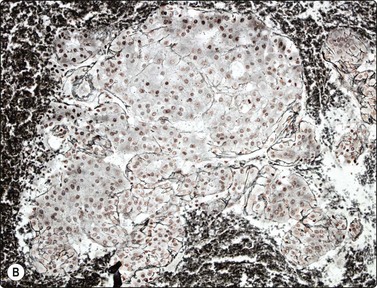
Fig. 10.16 Hepatocellular carcinoma
A reticulin stain on cell block sections may highlight (A) widened trabeculae and/or acinar structures (Reticulin, HP); (B) reduced or absent reticulin (cell block reticulin, HP).
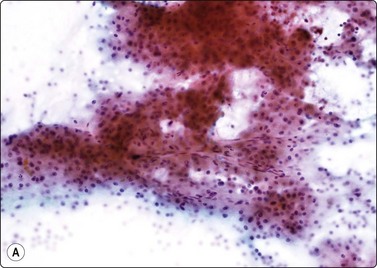
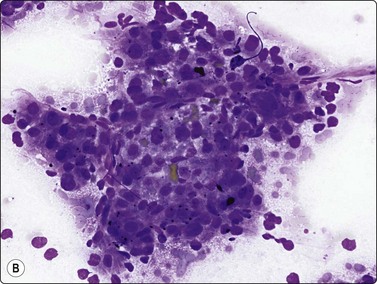
Fig. 10.17 Hepatocellular carcinoma
Tissue fragment of malignant cells traversed by spindle endothelial cells representing capillary blood vessels (A, Pap, IP; B, MGG, HP).
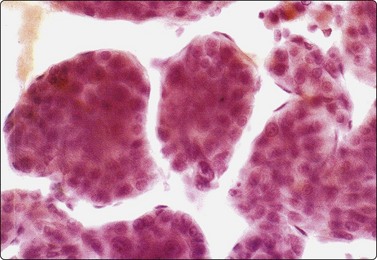
Fig. 10.18 Hepatocellular carcinoma
Endothelial cell envelopes seen as thin spindle nuclei surrounding tumor fragments (H&E, HP).
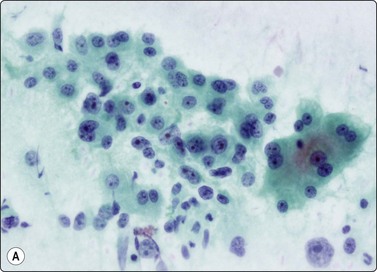
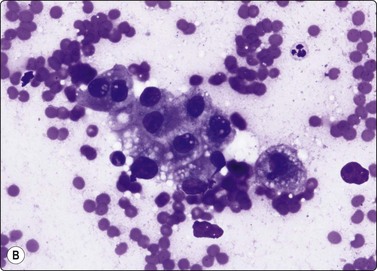
Fig. 10.19 Hepatocellular carcinoma
The cells resemble hepatocytes, but are larger with increased nucleocytoplasmic ratios; tumor cells have round nuclei and central large nucleoli; note cluster of benign hepatocytes (to right in A) (A, Pap, HP; B, MGG, HP).
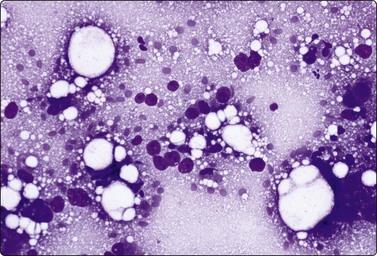
Fig. 10.20 Hepatocellular carcinoma
Multiple large round atypical nuclei stripped of their cytoplasm; one or multiple macronucleoli (MGG, HP).
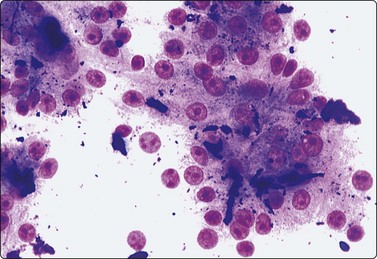
Fig. 10.21 Hepatocellular carcinoma
Moderately differentiated tumor with characteristic nuclear morphology; numerous green/black bile thrombi between tumor cells (MGG, HP).
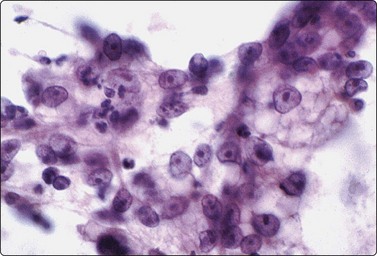
Fig. 10.22 Hepatocellular carcinoma
‘Clear cell’ variant; large cells with abundant pale vacuolated cytoplasm, large vesicular nuclei, large nucleoli (Pap, HP).
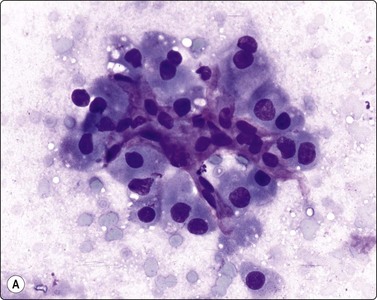
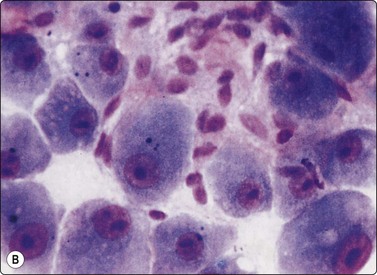
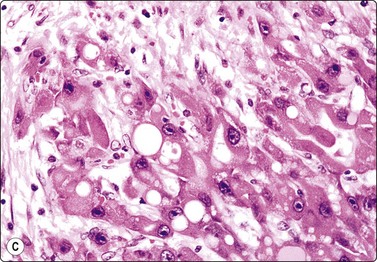
Fig. 10.23 Hepatocellular carcinoma, fibrolamellar type
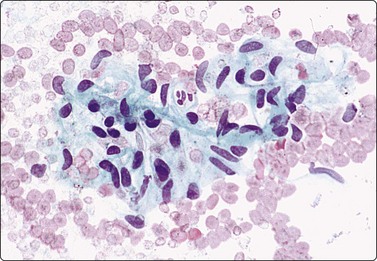
(A) Neoplastic cells adherent to fragments of lamellar collagen (MGG, HP); (B) Very large, malignant hepatocytes with prominent nucleoli and abundant granular cytoplasm (MGG, HP oil); (C) Corresponding tissue section (H&E, IP).
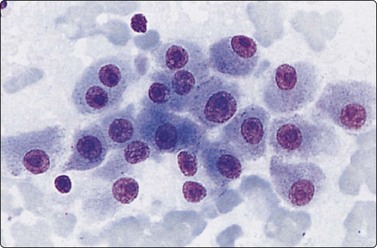
Fig. 10.24 Hepatocellular carcinoma
Well-differentiated tumor composed of poorly cohesive hepatocytic cells with mild anisokaryosis but large nucleoli; differentiation from adenoma difficult (MGG, HP).
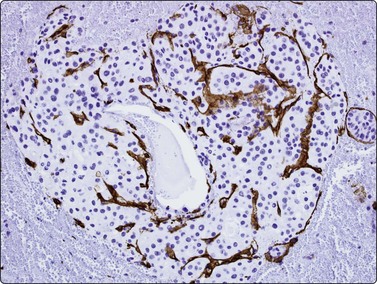
Fig. 10.25 Hepatocellular carcinoma
CD34 immunostaining demonstrates the classic endothelial staining pattern which is absent in reactive liver cell block (CD34, HP).
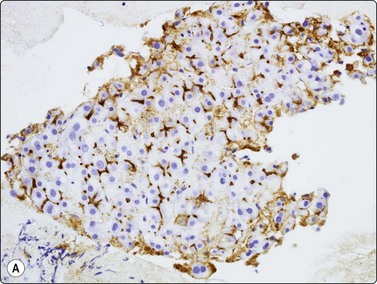
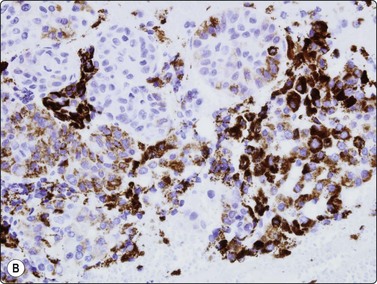
Fig. 10.26 Hepatocellular carcinoma
(A) pCEA immunostaining shows cytoplasmic staining along canalicular luminal edges only (pCEA, HP); (B) HepPar-1 produces diffuse granular cytoplasmic staining in distinction to adenocarcinoma, which is negative (HepPar-1, HP).
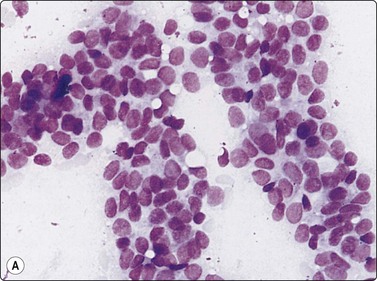
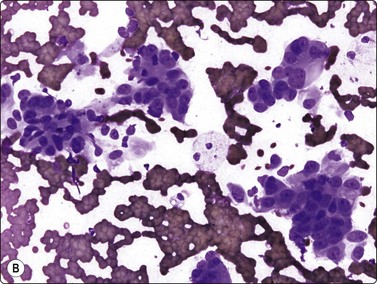
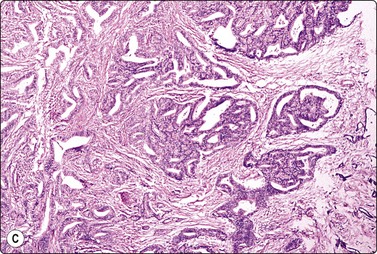
(A) Disorganized clusters of irregular but not very pleomorphic tumor cells with pale cytoplasm and relatively small nuclei (MGG, HP); (B) Disorganized cluster of mildly pleomorphic epithelial cells with hyperchromatic nuclei and prominent nucleoli (MGG, HP); (C) Corresponding tissue section (H&E, IP).
Criteria for diagnosis
The characteristic findings relate to (1) cell groupings, (2) relationship to endothelium and (3) cell morphology.39,41,72,79,116-132
The features in any individual FNA sample are extremely variable and largely dictated by tumor differentiation.116,117,127,132 As the degree of differentiation decreases, the cells become more obviously malignant and their resemblance to hepatocytes decreases.
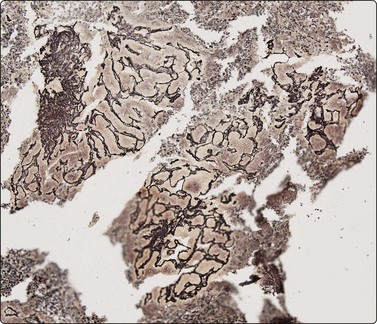
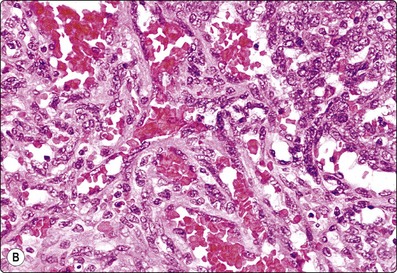
Smears are typically cellular with large fragments, clusters and dispersed cells (Fig. 10.14). Cell groupings are classically trabecular (Fig. 10.15A), particularly in better-differentiated tumors. Acinar arrangements may be seen in up to 40% of HCC (Fig. 10.15B).72 With decreasing differentiation, smaller sheets and single-lying cells become more frequent. A reticulin stain on smears or cell block material may highlight the widened trabeculae and/or acinar structures or the reduced or absent reticulin (Fig. 10.16). Endothelial relationships to HCC cell groups are an integral part of the diagnosis. Endothelial cells of sinusoidal capillaries may traverse (Fig. 10.17) or enclose (Fig. 10.18) trabeculae or separate tumor cell groupings.128 This important diagnostic criterion is diminished and then lost with decreasing differentiation.
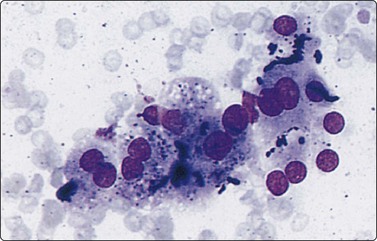
The cells resemble hepatocytes, thus demonstrating polygonal outlines and dense, granular cytoplasm, but with increased nucleocytoplasmic ratios over normal counterparts (Fig. 10.19). Centrally placed nuclei, characterized by granular to coarse chromatin, show cytoplasmic invaginations (intranuclear inclusions) in up to 70% of cases and large central nucleoli in up to 60% of cases. One of the most outstanding cytological appearances in HCC is the presence of many stripped malignant nuclei with identical characteristics, lying free in the background between cell groups (Fig. 10.20).126 The presence among malignant cells of occasional tumor giant cells or of bizarre hepatocytes would seem to favor primary liver carcinoma as they are rare in metastatic carcinoma.72,131 Bile pigment within the cytoplasm and between the cells proves the hepatocellular origin of the tumor, but is demonstrable in only 25% of cases (Fig. 10.21). Fouchet’s reagent counterstained with hematoxylin/Sirius red stains bile intensely green (Fig. 2.19). Contaminant benign hepatocytes and bile duct epithelium may be collected from the surrounding non-neoplastic liver, particularly in the setting of cirrhosis. They usually lie apart from the tumor cells.
Cytoplasmic vacuolation is pronounced in clear cell variants of HCC (Fig. 10.22).133 The clear cytoplasm is due to accumulation of glycogen or fat, and mucin stains are usually negative.134,135 This is not a defined subtype but rather a morphological pattern usually associated with a component of ‘conventional’ HCC. Its importance lies in distinguishing it from metastatic clear cell malignancies including carcinomas of renal, adrenal, pancreatic and ovarian origin, and sarcomas.133
The fibrolamellar subtype of HCC occurs as a distinct clinicopathologic variant in the second and third decades of life, without associated parenchymal liver disease and is therefore more often resectable. The neoplastic cells are intermingled with bundles of spindle-shaped fibroblasts and fragments of lamellar collagen (Fig. 10.23A). The tumor cells, readily recognized as of hepatocellular origin, are very large and may be pleomorphic. They have a polygonal shape, abundant, dense, granular cytoplasm, large vesicular nuclei, and large, single, central nucleoli (Fig. 10.23B). Trabeculae and peripheral endothelial wrapping are not observed in this otherwise well-differentiated variant, as the cell population is dispersed or adherent to the fibrous tissue. Intranuclear cytoplasmic inclusions are plentiful and there may be intracytoplasmic hyaline inclusions and pale bodies.136-138
Problems and differential diagnoses
Problems in HCC diagnosis occur at both ends of the differentiation spectrum; well differentiated HCC from benign hepatocellular lesions and poorly differentiated HCC from other primary and secondary malignancies.
The differential diagnosis of HCC depends on the clinical setting. In a patient with known chronic liver disease the major differential diagnosis is macroregenerative nodule (MRN). In a young female patient with a non-cirrhotic liver it is either focal nodular hyperplasia (FNH) or benign hepatocellular adenoma (HA). In an older patient, who may or may not have a past history of a primary malignancy elsewhere, it is metastatic malignancy.
The problem of distinguishing well-differentiated HCC (Fig. 10.24) from a benign regenerative nodule, focal nodular hyperplasia or adenoma has been mentioned. Clinical data such as age and sex, presence or absence of cirrhosis, exposure to steroidal hormones and radiological appearances may help to distinguish between these entities. There is considerable risk of overdiagnosing malignancy in regenerative nodules because nuclear enlargement, anisokaryosis, macronucleoli and dissociation may be prominent. Total absence of bile duct epithelium in a cellular smear favors a neoplasm, benign or malignant. The subtle cytological changes may be insufficient to reach a diagnosis of malignancy, and architectural abnormalities are of great importance. If there are endothelial-lined trabeculae or acini, capillary blood vessels which intersect groups of tumor cells, raised nucleocytoplasmic ratios, macronucleoli, many naked neoplastic nuclei, poorly defined cell borders, overall tumor cell monotony with sporadic tumor giant cells, and mitoses, the lesion is probably HCC.39-41,72,79,116-132
Prior to embarking on costly immunotests, a reticulin stain can highlight loss of a normal sinusoidal pattern and shows either widened trabeculae, rounded islands and pseudoglands or just a reduction or total absence of reticulin and distinguish benign from malignant liver lesions (see Figs 10.6 and 10.16).77-79120
CD34 and factor VIII immunostaining have been used in demonstrating the classic endothelial staining pattern of HCC (Fig. 10.25), absent in reactive liver and metastatic carcinoma,139 but are limited by partial staining of adenoma and focal nodular hyperplasia. Indeed, they provide little more information than the less expensive reticulin.131,139-141 Over-expression of Glypican-3, a heparan sulphate proteoglycan that plays an important role in cell growth and differentiation, has been observed in malignant hepatocytes. In several studies on cytological smears, positive staining for Glypican-3 showed a high sensitivity (80–90%) and specificity (> 95%) in HCC versus metastatic tumors and benign liver lesions.142-144
Image analysis or flow cytometric studies may permit prognostication on the basis of ploidy analysis. Most HCCs are aneuploid, with the remainder euploid (either diploid or polyploid), whereas all benign samples are euploid.145-147
Poorly differentiated HCC loses many classic criteria and is difficult to distinguish from metastatic adenocarcinoma, or other undifferentiated malignancies. Most HCC show a spectrum of differentiation and the diagnosis can be reached when the better-differentiated cells show recognisable hepatocellular features. Various cytological features have been identified as useful in discriminating between poorly differentiated HCC and metastatic adenocarcinoma; the presence of trabeculae versus true acinar structures, endothelial cells lining cell groups or small capillaries transgressing clusters of tumor cells, dissociation and atypical stripped nuclei, polygonal cells with central nuclei rather than columnar or cuboidal cells, round nuclei with one or more large nucleoli or intranuclear cytoplasmic inclusions, eosinophilic granular cytoplasm versus cytoplasmic vacuolation positive staining for glycogen, particularly if metastatic renal cell and adrenocortical carcinoma can be excluded, the presence of cytoplasmic bile, lipid, hyaline globules or Mallory bodies.72,117,119,148-150
Along with immunotests, a mucin stain may separate HCC from adenocarcinoma.
Various antibodies have been used to differentiate between hepatocellular carcinoma, cholangiocarcinoma and metastatic malignancies of various types.151,152 Positive immunoperoxidase staining for alpha fetoprotein favors primary cancer, but is seen in only around 50% of HCCs.129,153,154 It has good specificity despite also staining some germ cell tumors and occasional hepatoid adenocarcinomas of the gastrointestinal tract.
The demonstration of a characteristic bile canalicular staining pattern in HCC by means of polyclonal carcinoembryonic antigen (pCEA) is useful although it decreases with increasing anaplasia. Whereas metastatic adenocarcinoma displays full cytoplasmic immunostaining, HCC will show staining only along canalicular luminal membrane (Fig. 10.26A).73,116,129,154-158 Reported sensitivity ranges from 47% to 90%. It is essential that the polyclonal antibody be used; the same results will not be observed with the monoclonal antibody. Staining for CD10 gives the same pattern as CEA but is negative in adenocarcinoma.159,160
HepPar-1 is an antibody which recognises mitochondria in both benign and malignant hepatocytes. It has recently proved highly sensitive and specific in the distinction between adenocarcinoma and HCC, producing diffuse granular cytoplasmic staining in the latter (Fig. 10.26B).156,161,162
Glypican-3 antigen has also been described as useful in distinguishing HCC from metastatic malignancy. In one study on cytological material, positive staining for Glypican-3 showed a sensitivity of 90% and specificity of 100% for HCC versus metastatic malignancy.142,144,163
Cytokeratins 7 and 20 are most often negative in HCC, and may thus be employed as useful adenocarcinoma stains in the liver,164,165 as may Glut-1 and CA 15-3.166
More recently, cytoplasmic staining in HCC for TTF-1 (as opposed to the nuclear staining it produces in pulmonary adenocarcinomas) has been used in the successful discrimination of HCC from metastatic adenocarcinoma.167,168
In situ hybridisation for the detection of albumin mRNA has shown positivity in the majority of HCCs.129,169,170
Cholangiocarcinoma
Intrahepatic cholangiocarcinoma (CC) is associated with etiological factors such as liver fluke infestation (common in parts of South-east Asia), the primary sclerosing cholangitis/ulcerative colitis complex, congenital conditions such as Caroli’s disease and intrahepatic lithiasis. Tumors typically occur at the hilum where they present early with obstructive jaundice. They may be difficult to diagnose although the use of EUS-FNA has reported good results.66,171
Criteria for diagnosis (Fig. 10.27)172
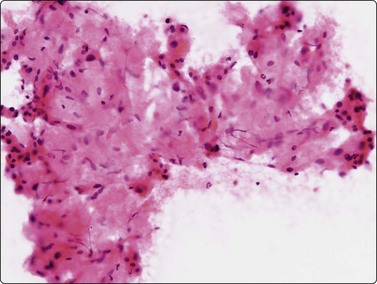
Cholangiocarcinomas often have abundant, desmoplastic stroma and may prove difficult to sample by FNA, or CNB for that matter. Although cells from a well-differentiated cholangiocarcinoma may not appear too different from bile duct epithelium, hepatocytes are absent and the number and size of epithelial sheets are larger than are usually obtained from non-neoplastic liver tissue (Fig. 10.27A). The application of P53, bcl-2 and Ki-67 has been reported as useful in discriminating between them.173 Less well-differentiated carcinomas are indistinguishable from metastatic adenocarcinoma (Fig. 10.27B), particularly those of pancreatic origin. Unfortunately, they are both generally positive for CK7 and CK19 and negative for CK20.174 Distinction from HCC relies on adenocarcinoma showing positivity for mucin and diffuse cytoplasmic staining for CK7, CK19 and pCEA and HCC staining for alpha fetoprotein and HepPar-1 and others as described earlier in the section on HCC.
Diagnosis depends not only on cytological assessment but on correlation of the clinical and radiological findings, in particularly excluding an extrahepatic malignancy.
The cytology of combined hepatocellular–cholangiocarcinomas has been reported.175,176 They show classic HCC cells, pure glandular cells, and hybrid or intermediate cells with features of both components. A histochemical stain for mucin will confirm intracytoplasmic, brush border or intraluminal mucin. Immunohistochemistry is helpful in confirming the two components. pCEA highlights the canalicular areas of the HCC component and the brush border of the glandular areas.
Hepatoblastoma177-181
Hepatoblastoma is a rare, highly malignant tumor of infants and young children, which metastasizes early. It is not associated with chronic liver disease. The alpha fetoprotein level is significantly raised in approximately 90% of cases. Patients can have a good response to surgery and chemotherapy. There is a spectrum of subtypes from anaplastic through embryonal and fetal to macrotrabecular, based on growth pattern and increasing resemblance of component cells from anaplastic towards hepatocyte morphology. A mixed epithelial and mesenchymal pattern includes primitive mesenchyme. The cytological appearance is that of a small round cell childhood tumor, (Fig. 10.28, and also Figs 17.18–17.20), with clusters, ribbons and rosettes of embryonal cells, which are small cells with a high N : C ratio, oval to spindled nuclei, coarse chromatin, prominent, often multiple, nucleoli, and scant to moderate cytoplasm. Fetal epithelial cells, are slightly smaller than hepatocytes with round regular nuclei, fine chromatin, prominent central nucleoli and abundant granular or clear cytoplasm which may contain fat, bile or glycogen. Fragments of mesenchyme may be observed. Foci of extramedullary hemopoiesis are often present. The differential diagnosis is that of other small round cell childhood tumors metastatic to the liver or of HCC, which may occur in older children. HepPar-1 staining of hepatoblastoma can be useful in this regard.182
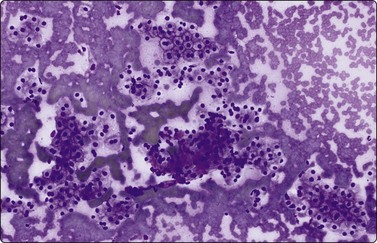
Malignant lymphoma183,184
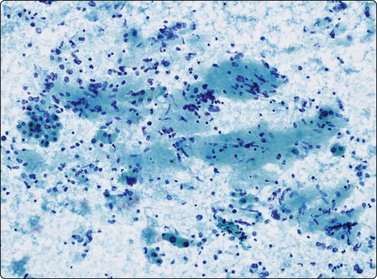
Liver involvement in stage IV malignant lymphoma portends a poor prognosis and has been found in 25% of random FNA biopsies in patients with lymphoma.185 Primary involvement of the liver is relatively rare. It is often patchy and microscopic and may be missed by either FNA or CNB. The liver tumor may be solitary, multiple or diffuse. Aspirates are usually markedly cellular with dispersed cells and scattered small aggregates without true cohesion, and lymphoglandular bodies are present in the background. Non-Hodgkin lymphoma of diffuse large B cell type (high grade) is the most common type. If on-site rapid evaluation detects a lymphoid lesion, a further needle pass should be performed to obtain cells for phenotyping by immunocytochemistry or flow cytometry, for cytogenetics and for molecular gene rearrangement studies. For detailed cytological criteria for the diagnosis of lymphoma, see Chapter 5. The differential diagnosis of a post-transplant lymphoproliferative disorder (PTLD) should be considered where there is a history of organ transplantation. The smears may be either polymorphous with a mixture of lymphocytes, plasma cells and histiocytes, or monomorphous with large atypical lymphocytes. The diagnosis is supported by identifying B-cell monoclonality and the presence of EBV DNA.183,186,187
Vascular tumors
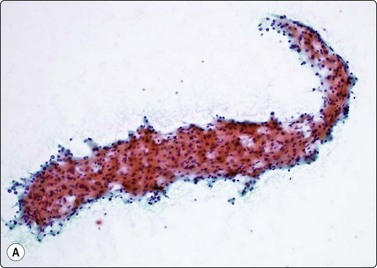
Cavernous hemangioma is the most common benign solid lesion in the liver and is not infrequently found by US or by CT in the routine investigation of cancer patients. The diagnosis can often be made by imaging, but in some cases with an atypical appearance a needle biopsy is necessary to confidently exclude metastatic tumor or adenoma.61,63,64,188,189 In our experience, clusters of benign spindle cells recognizable as endothelial cells are infrequently seen in FNA smears (Fig. 10.29). The diagnosis is based on appropriate clinico-radiological history and aspiration of profuse blood as from a venepuncture, provided the correct positioning of the needle can be ascertained. When diagnostic cells are present, they assume three-dimensional swathes of endothelial cells surrounding cavernous spaces, into which the cell nuclei protrude. Connective tissue fragments may be present. A definitive cytological diagnosis of hemangioma has been made in 27–96% of cases reported in the literature.61,63,64,188,189
Epithelioid hemangioendothelioma is a solid vascular-derived tumor with a clinical course intermediate between that of hemangioma and angiosarcoma. Females are more commonly affected than males. The tumor is multifocal and increasingly sclerotic, often pre-empting good FNA yield. The cells are spindled or epithelioid, showing pleomorphism with scattered bizarre tumor giant cells. Intracytoplasmic lumina may be identified.190 The differential diagnosis includes pleomorphic HCC. Diagnosis is more easily made on cell blocks than on smears and immunohistochemistry for vascular markers is usually positive.191,192
Angiosarcoma is a very rare primary hepatic tumor, but reports have appeared in the cytological literature,191,193,194 in which the FNA findings indicated a malignant spindle-cell neoplasm. Again, diagnosis can be facilitated by the morphology of tissue fragments in cell blocks, which also allow appropriate immunocytochemistry including factor VIII, CD31, CD34 and Ulex europaeus.195 The cytomorphology shows consistently abundant blood, variable cellularity, spindle-shaped tumor cells arranged in anastomosing vessels, papillae or solid groups showing variable pleomorphism, and vasoformative features such as erythrophagocytosis and hemosiderin. As indicated in the preceding section on complications, massive bleeding and death have been reported after FNA of vascular lesions.
Other mesenchymal tumors
Angiomyolipoma of the liver is a rare benign tumor, important not because of its frequency but because of its mimicry of malignancy. As in other sites, it comprises vascular tissue, fat cells, and immature smooth muscle cells with fibrillar cytoplasm, in varying proportions (Fig. 10.30). The muscle cells can be spindled or epithelioid and show pleomorphism with giant cells, but no mitoses or necrosis should be present. Transgressing endothelium may be seen, but no peripheral endothelial wrapping occurs.196 The diagnosis rests on identifying fat cells as an integral part of the tumor fascicles and not regarding them as hepatocytes distended with fat vacuoles. Extramedullary hematopoiesis is a consistent finding. Immunostaining with HMB-45 is confirmatory. Smooth muscle and vascular markers are also positive, as is S-100 in fat cells. In the more pleomorphic forms without fat tissue, absence of keratin staining is an important criterion.196-198
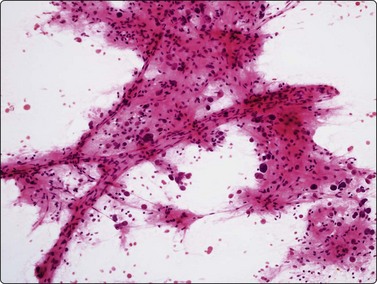
Well-defined blood vessels associated with a mixture of epithelioid and spindle cells showing marked nuclear pleomorphism. Fat cells are not obvious (H&E, IP).
Mesenchymal hamartoma is a benign multicystic tumor which may reach very considerable size. It occurs predominantly in male children under the age of 2 years,199,200 but adult variants have been reported.201 The mass, which may mimic other paediatric neoplasms clinically and radiologically, yields a biphasic population of small bland rounded epithelial cells of hepatocytic or bile duct origin, admixed with single or aggregated bland spindle cells, often in a myxoid stromal or mucoid background. The spindled component may predominate.200 Excision is curative. The cytological differential diagnosis includes hepatoblastoma of mixed epithelial and mesenchymal type.
Gastrointestinal stromal tumors (GISTs) are the most frequently encountered sarcoma in the liver (see also Chapter 11). Most are metastatic from the stomach. Diagnosis is accomplished without difficulty if there is an available history, if the morphology is spindled (Fig. 10.31) or if cell blocks are made. Great difficulty arises, however, with the epithelioid variant, which is characterized by small round dissociated cells with clear cytoplasm. The differential diagnosis in these cases is that of metastatic carcinoma. Immunocytochemical positivity for CD117 is diagnostic and CD34 may also be positive.202-204
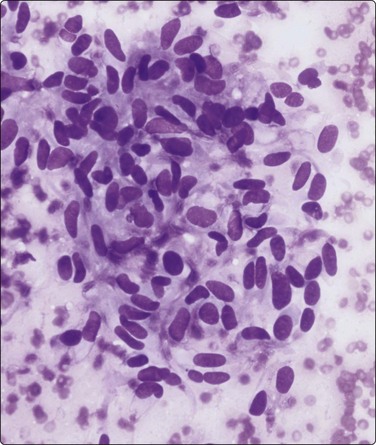
Fig. 10.31 Liver metastasis of gastrointestinal stromal tumor
Cohesive cluster of haphazardly arranged spindle cells showing mild anisonucleosis and tapered cytoplasm (MGG, HP).
Leiomyosarcoma may arise from the inferior vena cava but is more often a metastatic deposit. It presents as clusters and fascicles of spindle cells with ill-defined bipolar cytoplasm. Pleomorphism, necrosis and mitoses can occur in higher-grade tumors. Immunocytochemistry shows positivity for vimentin, desmin, and smooth muscle actin.205,206
Undifferentiated (embryonal) sarcoma is a rare malignant mesenchymal hepatic tumor, occurring predominantly in children under the age of 15 years. Tumor cells occur singly or in clusters. They are polygonal and spindle with large, pleomorphic nuclei, one or several nucleoli and variable poorly defined cytoplasm containing vacuoles and eosinophilic globules which are PAS/D positive. Multinucleated tumor cells are not uncommon and myxoid matrix may be present. Distinction from other paediatric liver tumors is reliant on vimentin, α1-antichymotrypsin and α1-antitrypsin positivity.207-210 Regarding liver tumors in childhood, see also Chapter 17.
Metastatic malignancy (Figs 10.31 and 10.32)
The liver is a common site of metastatic disease because it acts as a filter. Differentiation between primary and secondary tumors on imaging can be problematic. The differential diagnosis between primary hepatocellular carcinoma and metastatic malignancy and the use of immunostaining to discriminate between them has been previously discussed in the section on HCC. Metastatic tumors may be single, multiple or diffusely infiltrating. The metastatic deposit usually shows the morphological characteristics of the primary tumor. Clinical history is very important in making the diagnosis. Review of any previous histology or cytology is recommended.
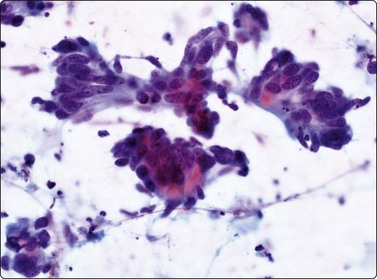
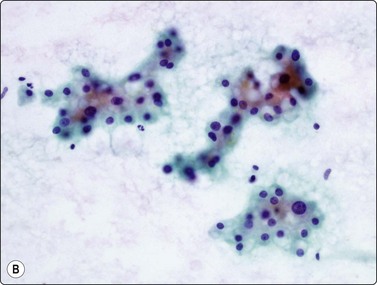
Fig. 10.32 Liver metastasis of colorectal carcinoma
Columnar cells arranged in glandular formation on a background of granular debris (Pap, HP).
The clinician needs to know whether liver involvement is present, either from a primary tumor which has already manifested or one which is still occult. The result of FNA will often dictate management such as chemotherapy.211 Cell typing is thus desirable in most instances. We regard it as particularly important to identify accurately those metastases which are potentially chemosensitive or hormonally manipulable. It is useful to run through a mental checklist to ensure that one is not missing germ cell tumors, neuroendocrine tumors (including small cell undifferentiated carcinomas) or carcinomas of breast, prostatic, endometrium, thyroid or even nasopharyngeal origin.
The macroscopic appearance of the aspirate is usually quite different from that of normal liver tissue. There is frequently less admixture with blood and often a high cell content. The aspirate is easily smeared into a grayish film. Necrosis may be pre-eminent, sometimes to the extent that preserved, diagnostic cancer cells are hard to find. A necrotizing metastatic tumor deposit may present as a cyst.87 If hepatocytes are found amongst the neoplastic cells, they provide a useful baseline for the evaluation of cell and nuclear size.
The cytologic patterns of metastatic carcinomas and clues to the identification of the primary site of origin are described in Chapter 5 and will not be repeated at length here. Adequate sampling is the key. Colonic adenocarcinoma is probably the most common source of liver metastases. The cytological pattern is characteristic, showing malignant columnar epithelial cells in palisaded rows or microglandular groups with a background of necrotic debris (Fig. 10.32), often with evidence of mucin secretion.35 Small cell undifferentiated carcinoma of pulmonary or colonic derivation often involves the liver diffusely. Hepatocytes may be numerous and evenly dispersed between tumor cell groups in smears. Hepatic FNA in these cases has staging value in anticipation of chemotherapy.212 Carcinoid tumors of bronchogenic, pancreatic or colonic origin are distinguishable from metastatic adenocarcinoma by their endocrine appearance. The nuclei have a rounded shape with ‘speckled’ granular, hyperchromatic chromatin. Red (MGG) cytoplasmic granularity may be visible. The diagnosis can be confirmed by immunoperoxidase staining for neuroendocrine markers.213
Immunohistochemistry can aid in determining the primary site of the metastases.214,215 Adenocarcinoma of unknown origin remains a diagnostic problem because of lack of specific immunohistochemical markers. It can arise from many different sites, e.g. bronchus, colon, pancreas, breast, stomach, etc. A limited but useful immunoperoxidase panel to determine site of origin of metastatic adenocarcinoma, not otherwise specified in patients in whom clinical history is nondirective, consists of cytokeratins 7 and 20, TTF-1, CDX2 and additional ER/PR in female patients or PSA in male patients.
Clinical aspects
The place of FNAC in the investigative sequence
Fine needle biopsy of the spleen is not a commonly performed procedure, although there are increasing reports in the cytological literature, partly due to the advent of endoscopic ultrasound FNA.216-226 The possible risk of hemorrhagic complications has probably been a discouraging factor and it is uncommon for a patient to present with isolated splenomegaly or a localized splenic lesion which cannot be explained using other diagnostic pathways.
The main purpose of FNA is to diagnose or confirm hematologic or metastatic malignancy.216,219,226 However, a number of non-neoplastic lesions and disorders involving the spleen can be diagnosed by FNA.227 Examples are: cysts,228 abscesses,223-229 a variety of infectious and granulomatous processes notably leishmaniasis, tuberculosis and sarcoid,224,227,229-233 myeloid metaplasia,234 inflammatory pseudotumor,219,235 amyloidosis236 and, in combination with biochemical investigations, lipid storage disease.237 The cytology is ‘normal’ in a proportion of cases of isolated splenomegaly and gives no clue to the possible etiology (e.g. in portal hypertension).
Malignant lymphoma most often involves the spleen diffusely, but can occur as solitary or multiple circumscribed deposits.238 CT scanning or ultrasonography can thus be helpful in selecting the best site for a biopsy. Splenic metastases are not uncommon in disseminated carcinoma, but rarely constitute a clinical problem. FNA can replace splenectomy for diagnosis of solitary splenic metastases.239 Splenic hamartoma, hemangioma and angiosarcoma are examples of the rare primary tumors of the spleen.240,241
Accuracy of diagnosis
Diagnostic sensitivity for neoplasia was 86.4% and specificity 97.5% based on 78 cases with histological correlation in the series by Zeppa et al.226 Friedlander et al. reported a sensitivity of 94% with a specificity of 84% and a positive predictive value of 79%. They reported four false positives although these included cases they had categorized as ‘supicious of malignancy’.218 In the series from M.D. Anderson Cancer Center there were no false positives and only one false-negative diagnosis in 50 cases, only 10 of whom had subsequent splenectomy. Six biopsies were non-diagnostic.216 Other series reported accuracy rates of between 80% and 90%.220,229,242,243
Diagnostic pitfalls
As FNB of the spleen is not commonly performed there are a number of pitfalls.218 These include:
Most of these are not unique to the spleen; however, increased experience with splenic FNA should go some way to minimizing errors related to these pitfalls.
Complications
The risk of a needle biopsy causing rupture and major hemorrhage appears to be small.218,220 Söderström had no complications in over 1000 FNAs225 and Selroos et al. had none in 557 cases.232 However, a few cases of hemorrhagic complications leading to splenectomy have been reported.224,226,243 Hemorrhagic diathesis and low platelet count are a contraindication. Söderström regarded glandular fever and polycythemia as relative contraindications.225 Core needle biopsy of the spleen has been advocated by some as it has a greater accuracy, particularly in the diagnosis of of lymphoma,243-245 but the risk of complications is likely to be greater.243-247
Technical considerations
Most FNA samples are obtained using imaging guidance; either percutaneously, or more recently using endoscopic ultrasound FNA.217,218,242 Direct FNA can be performed into a diffusely enlarged spleen or into a palpable localized mass if no imaging is available. Most splenic aspirates may appear to consist mainly of blood but are, nevertheless, usually quite rich in cells. The material should be smeared thinly, and air-dried MGG-stained smears are essential to allow comparison with blood films and bone marrow aspirates. Immunostaining can be performed on spare slides, cytospins or cell block material, and a cell suspension for flow cytometry should be obtained if lymphoma is considered in the differential diagnosis.
Cytological findings
Non-specific findings in splenomegaly (Fig. 10.33)
It is unlikely that normal spleens are ever subjected to FNA and the normal cytological pattern is therefore not well known. The above features are seen in cases of splenomegaly related to portal hypertension, hemolytic anemia, etc. FNA is performed mainly to exclude hematological malignancy, and does not contribute to a specific diagnosis in these conditions.
Lymphoid cells predominate in FNB samples from the spleen. There is invariably a background of abundant blood and platelet aggregates. The lymphoid cells are more often seen in tissue fragments, trapped in a meshwork of endothelial cells, than as evenly spread, isolated cells. Blast cells constitute a relatively higher proportion of the cell population than in smears from reactive lymph nodes. Diagnostic criteria applied in lymph node cytology should be used with caution when smears from the spleen are examined.
Non-neoplastic processes225,227
Myeloid metaplasia (Fig. 10.34)
The findings in myeloid metaplasia include normoblasts, myelocytes and megakaryocytes intermingled with the cells of normal splenic tissue. Normoblasts are easily recognized by their relatively small, round, hyperchromatic nuclei and their homogeneous, dense eosinophilic or amphophilic cytoplasm. Megakaryocytes may be mistaken for malignant neoplastic cells but should be recognizable by the giant, lobulated nucleus and the abundant, granular cytoplasm. Myelocytes have specific cytoplasmic granules. All these features are best seen in MGG-stained smears.234
Granulomatous processes
Histiocytes forming granulomatous clusters may be found in several unrelated conditions and do not permit a specific diagnosis when found in splenic aspirates. Well-formed granulomata of epithelioid histiocytes, with or without Langhans giant cells, suggest the main differential diagnoses of sarcoidosis and tuberculosis (Fig. 10.35).232,233 Caseous necrosis is characteristic of tuberculosis but is not always seen in smears. The demonstration of AFBs is only possible in some cases of tuberculosis involving the spleen.231 Material should always be sent for culture where possible. Noncaseating granulomas may also be found in splenic aspirates in cases of malignant lymphoma, particularly Hodgkin’s lymphoma, and the background lymphoid cell population needs to be studied closely.
Others
Conspicuous large histiocytes with foamy cytoplasm indicate lipid storage disease. Histiocytes in Gaucher’s disease have a characteristic striated cytoplasmic appearance (Fig. 10.36).237 The foamy histiocytes seen in Niemann-Pick disease are less characteristic and the specific diagnosis rests on biochemical analyses. The cytology of histiocytosis X is described in Chapter 16 and amyloidosis236 in Chapter 4. Other examples of non-neoplastic lesions in the spleen diagnosed by FNB are splenic abscess223,229,242 and splenic epidermoid cyst.227,248
Neoplasms
Malignant lymphoma220,222,226,238
The findings in malignant lymphoma in splenic aspirates are the same as in other tissues (see Chapter 5). The abnormal lymphoid cells are more dispersed, distributed as single cells, and tissue fragments are less conspicuous than in smears of non-neoplastic splenic tissue. Non-Hodgkin lymphoma of mixed cell type is particularly difficult to diagnose with confidence in splenic aspirates and immune marker studies by flow cytometry or immunohistochemistry are indispensable.222,249,250 In Hodgkin lymphoma, the diagnosis rests on the demonstration of Reed-Sternberg cells.251
Hairy cell leukemia (Fig. 10.37)
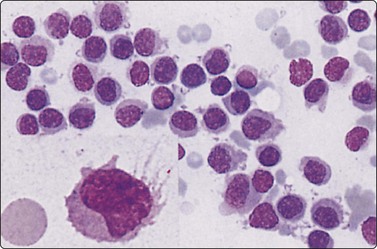
Fig. 10.37 Hairy cell leukemia
Uniform lymphoid cells, larger and paler than small lymphocytes and with slightly reniform nuclei (MGG, HP). Inset: fine hair-like cytoplasmic processes which may, however, be artifactually produced in other lymphoid cells (MGG, HP oil).
Splenomegaly is one of the main symptoms in hairy cell leukemia, whereas lymph node involvement appears late in the disease and is less conspicuous. The diagnosis is usually made on examination of a bone marrow aspirate or of peripheral blood but can also be made on a splenic aspirate.252,253
Vascular tumors
Very few cases of splenic hemangioma,254 littoral cell angioma255 and of splenic hamartoma (splenoma)241,255,256 with FNB have been reported. Splenic hamartoma (Fig. 10.38) is a hamartomatous tumor of the spleen consisting exclusively of red pulp tissue. We have seen one example in which the smears were dominated by numerous spindle cells of endothelial type. The nuclei were elongated and uniform, with a bland, finely granular chromatin. FNB was reported as an angiomatous tumor. Distinction between splenic hamartoma and angioma may not be possible in FNA smears.
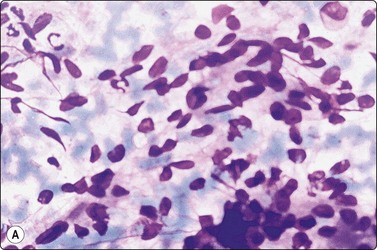
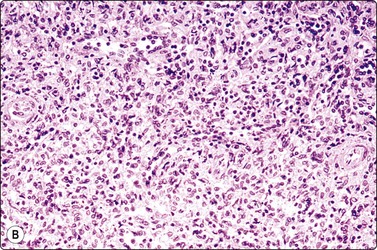
(A) Smear showing many spindle cells of endothelial type with bland nuclei and background of blood (MGG, HP); (B) Corresponding tissue section (H&E, IP).
Angiosarcoma occasionally presents as a primary tumor of the spleen.240 A case from our files, in which the diagnosis was correctly made by FNA, showed smears which were rich in cells. These were spindly and had elongated, mildly pleomorphic, hyperchromatic nuclei. Most of the cells formed highly cellular tissue fragments which showed a distinctly vasoformative pattern with bunches of radiating spindle cells (Fig. 10.39).
Metastases
Metastases may be solitary or multiple and a history of a primary malignancy elsewhere is not always available. Splenic FNA has value in diagnosis and staging to provide prognostic information and directing further management, e.g. chemotherapy. The more common primary sites include breast, lung, colorectal and ovarian carcinoma and melanoma.239,257 The cytologic patterns of metastatic malignancies and clues to the identification of the primary site of origin are described in Chapter 5 and will not be repeated here.
1 Dusenbery D, Dodd GD3rd, Carr BI. Percutaneous fine-needle aspiration of portal vein thrombi as a staging technique for hepatocellular carcinoma. Cytologic findings of 46 patients. Cancer. 1995 Apr 15;75(8):2057-2062.
2 Tarantino L, Francica G, Sordelli I, et al. Diagnosis of benign and malignant portal vein thrombosis in cirrhotic patients with hepatocellular carcinoma: color Doppler US, contrast-enhanced US, and fine-needle biopsy. Abdom Imaging. 2006 Sep–Oct;31(5):537-544.
3 Yang L, Lin LW, Lin XY, et al. Ultrasound-guided fine needle aspiration biopsy in differential diagnosis of portal vein tumor thrombosis. Hepatobiliary Pancreat Dis Int. 2005 May;4(2):234-238.
4 Lin SM, Kuo SH, Lin DY, et al. Cytologic changes in hepatocellular carcinoma after percutaneous acetic acid injection. Correlation with helical computed tomography findings. Acta Cytol. 2000 Jan–Feb;44(1):1-6.
5 Moreland WS, Zagoria RJ, Geisinger KR. Use of fine needle aspiration biopsy in radiofrequency ablation. Acta Cytol. 2002 Sep–Oct;46(5):819-822.
6 Bialecki ES, Ezenekwe AM, Brunt EM, et al. Comparison of liver biopsy and noninvasive methods for diagnosis of hepatocellular carcinoma. Clin Gastroenterol Hepatol. 2006 Mar;4(3):361-368.
7 Forner A, Vilana R, Ayuso C, et al. Diagnosis of hepatic nodules 20 mm or smaller in cirrhosis: Prospective validation of the noninvasive diagnostic criteria for hepatocellular carcinoma. Hepatology. 2008 Jan;47(1):97-104.
8 Ng KK, Poon RT, Lo CML, et al. Impact of preoperative fine-needle aspiration cytologic examination on clinical outcome in patients with hepatocellular carcinoma in a tertiary referral center. Arch Surg. 2004 Feb;139(2):193-200.
9 Ohlsson B, Nilsson J, Stenram U, et al. Percutaneous fine-needle aspiration cytology in the diagnosis and management of liver tumours. Br J Surg. 2002 Jun;89(6):757-762.
10 Torzilli G, Olivari N, Del Fabbro D, et al. Indication and contraindication for hepatic resection for liver tumors without fine-needle biopsy: validation and extension of an Eastern approach in a Western community hospital. Liver Transpl. 2004 Feb;10(2 Suppl 1):S30-S33.
11 Stewart BW, Kleihues P. World cancer report. Lyon: IARC Press; 2003.
12 Brits CJ. Liver aspiration cytology. S Afr Med J. 1974 Nov 2;48(53):2207-2214.
13 Linsk JA, Franzen S. Clinical aspiration cytology, 2nd ed. Philadelphia: Lippincott; 1989.
14 Perry MD, Johnston WW. Needle biopsy of the liver for the diagnosis of nonneoplastic liver diseases. Acta Cytol. 1985 May–Jun;29(3):385-390.
15 Brunetti E, Silini E, Pistorio A, et al. Coarse vs. fine needle aspiration biopsy for the assessment of diffuse liver disease from hepatitis C virus-related chronic hepatitis. J Hepatol. 2004 Mar;40(3):501-506.
16 Wasastjerna C. Liver. In: Zajicek J, editor. Aspiration biopsy cytology Part 2 Cytology of infradiaphragmatic organs. Basel: Karger; 1979:167-193.
17 Dominis M, Cerlek S, Solter D. Cytology of diffuse liver disorders. Acta Cytol. 1973 May–Jun;17(3):205-208.
18 Henriques UV, Hasselstrom K. Evaluation of jaundice: fine-needle aspiration liver cytology as a discriminating tool. Dan Med Bull. 1977 Jun;24(3):104-108.
19 Wasastjerna C, Reissell P, Karjalainen J, Ekelund P. Fatty liver in diabetes. A cytological study. Acta Med Scand. 1972 Mar;191(3):225-228.
20 Carbonnel F, Samuel D, Reynes M, et al. Fine-needle aspiration biopsy of human liver allografts. Correlation with liver histology for the diagnosis of acute rejection. Transplantation. 1990 Oct;50(4):704-707.
21 Hockerstedt K, Lautenschlager I, Ahonen J, et al. Diagnosis of acute rejection in liver transplantation. J Hepatol. 1988 Apr;6(2):217-221.
22 Kirby RM, Young JA, Hubscher SG, et al. The accuracy of aspiration cytology in the diagnosis of rejection following orthotopic liver transplantation. Transpl Int. 1988 Oct;1(3):119-126.
23 Kubota K, Ericzon BG, Reinholt FP. Comparison of fine-needle aspiration biopsy and histology in human liver transplants. Transplantation. 1991 May;51(5):1010-1013.
24 Lautenschlager I, Hockerstedt K, Hayry P. Fine-needle aspiration biopsy in the monitoring of liver allografts. Transpl Int. 1991 Apr;4(1):54-61.
25 Their M, Lautenschlager I, von Willebrand E, et al. The use of fine-needle aspiration biopsy in detection of acute rejection in children after liver transplantation. Transpl Int. 2002 May;15(5):240-247.
26 Babb RR, Jackman RJ. Needle biopsy of the liver. A critique of four currently available methods. West J Med. 1989 Jan;150(1):39-42.
27 Jacobsen GK, Gammelgaard J, Fuglo M. Coarse needle biopsy versus fine needle aspiration biopsy in the diagnosis of focal lesions of the liver. Ultrasonically guided needle biopsy in suspected hepatic malignancy. Acta Cytol. 1983 Mar–Apr;27(2):152-156.
28 Stewart CJ, Coldewey J, Stewart IS. Comparison of fine needle aspiration cytology and needle core biopsy in the diagnosis of radiologically detected abdominal lesions. J Clin Pathol. 2002 Feb;55(2):93-97.
29 O’Connell AM, Keeling F, Given M, et al. Fine-needle trucut biopsy versus fine-needle aspiration cytology with ultrasound guidance in the abdomen. J Med Imaging Radiat Oncol. 2008 Jun;52(3):231-236.
30 Franca AV, Valerio HM, Trevisan M, et al. Fine needle aspiration biopsy for improving the diagnostic accuracy of cut needle biopsy of focal liver lesions. Acta Cytol. 2003 May–Jun;47(3):332-336.
31 Fornari F, Civardi G, Cavanna L, et al. Ultrasonically guided fine-needle aspiration biopsy: a highly diagnostic procedure for hepatic tumors. Am J Gastroenterol. 1990 Aug;85(8):1009-1013.
32 Guo Z, Kurtycz DF, Salem R, et al. Radiologically guided percutaneous fine-needle aspiration biopsy of the liver: retrospective study of 119 cases evaluating diagnostic effectiveness and clinical complications. Diagn Cytopathol. 2002 May;26(5):283-289.
33 Leiman G, Leibowitz CB, Dunbar F. Fine-needle aspiration of the liver: out of the ivory tower and into the community. Diagn Cytopathol. 1989;5(1):35-39.
34 Nazir RT, Sharif MA, Iqbal M, Amin MS. Diagnostic accuracy of fine needle aspiration cytology in hepatic tumours. J Coll Physicians Surg Pak June;20(6):373–376.
35 Samaratunga H, Wright G. Value of fine needle aspiration biopsy cytology in the diagnosis of discrete hepatic lesions suspicious for malignancy. Aust N Z J Surg. 1992 Jul;62(7):540-544.
36 Wang P, Liu LM, Meng ZQ, et al. [Evaluation of the results of fine-needle aspiration liver biopsies and the complications in 2528 cases.]. Zhonghua Gan Zang Bing Za Zhi. 2007 Oct;15(10):758-762.
37 Bell DA, Carr CP, Szyfelbein WM. Fine needle aspiration cytology of focal liver lesions. Results obtained with examination of both cytologic and histologic preparations. Acta Cytol. 1986 Jul–Aug;30(4):397-402.
38 Bret PM, Labadie M, Bretagnolle M, et al. Hepatocellular carcinoma: diagnosis by percutaneous fine needle biopsy. Gastrointest Radiol. 1988 Jul;13(3):253-255.
39 Sole M, Calvet X, Cuberes T, et al. Value and limitations of cytologic criteria for the diagnosis of hepatocellular carcinoma by fine needle aspiration biopsy. Acta Cytol. 1993 May–Jun;37(3):309-316.
40 Sangalli G, Livraghi T, Giordano F. Fine needle biopsy of hepatocellular carcinoma: improvement in diagnosis by microhistology. Gastroenterology. 1989 Feb;96(2 Pt 1):524-526.
41 Zainol H, Sumithran E. Combined cytological and histological diagnosis of hepatocellular carcinoma in ultrasonically guided fine needle biopsy specimens. Histopathology. 1993 Jun;22(6):581-586.
42 Awad SS, Fagan S, Abudayyeh S, et al. Preoperative evaluation of hepatic lesions for the staging of hepatocellular and metastatic liver carcinoma using endoscopic ultrasonography. Am J Surg. 2002 Dec;184(6):601-604. discussion 4–5
43 Nguyen P, Feng JC, Chang KJ. Endoscopic ultrasound (EUS) and EUS-guided fine-needle aspiration (FNA) of liver lesions. Gastrointest Endosc. 1999 Sep;50(3):357-361.
44 tenBerge J, Hoffman BJ, Hawes RH, et al. EUS-guided fine needle aspiration of the liver: indications, yield, and safety based on an international survey of 167 cases. Gastrointest Endosc. 2002 Jun;55(7):859-862.
45 Lundquist A. Liver biopsy with a needle of 0.7 MM outer diameter. Safety and quantitative yield. Acta Med Scand. 1970 Dec;188(6):471-474.
46 Nguyen GK. Fine-needle aspiration biopsy cytology of hepatic tumors in adults. Pathol Annu. 1986;21(Pt 1)):321-349.
47 Smith EH. Complications of percutaneous abdominal fine-needle biopsy. Review. Radiology. 1991 Jan;178(1):253-258.
48 Drinkovic I, Brkljacic B. Two cases of lethal complications following ultrasound-guided percutaneous fine-needle biopsy of the liver. Cardiovasc Intervent Radiol. 1996 Sep–Oct;19(5):360-363.
49 Lundstedt C, Stridbeck H, Andersson R, et al. Tumor seeding occurring after fine-needle biopsy of abdominal malignancies. Acta Radiol. 1991 Nov;32(6):518-520.
50 Saborido BP, Diaz JC, de Los Galanes SJ, et al. Does preoperative fine needle aspiration-biopsy produce tumor recurrence in patients following liver transplantation for hepatocellular carcinoma? Transplant Proc. 2005 Nov;37(9):3874-3877.
51 Tung WC, Huang YJ, Leung SW, et al. Incidence of needle tract seeding and responses of soft tissue metastasis by hepatocellular carcinoma postradiotherapy. Liver Int. 2007 Mar;27(2):192-200.
52 Roussel F, Dalion J, Benozio M. The risk of tumoral seeding in needle biopsies. Acta Cytol. 1989 Nov–Dec;33(6):936-939.
53 Navarro F, Taourel P, Michel J, et al. Diaphragmatic and subcutaneous seeding of hepatocellular carcinoma following fine-needle aspiration biopsy. Liver. 1998 Aug;18(4):251-254.
54 Jourdan JL, Stubbs RS. Percutaneous biopsy of operable liver lesions: is it necessary or advisable? N Z Med J. 1996 Dec 13;109(1035):469-470.
55 Huang CJ, Pitt HA, Lipsett PA, et al. Pyogenic hepatic abscess. Changing trends over 42 years. Ann Surg. 1996 May;223(5):600-607. discussion 7–9
56 John TG, Garden OJ. Needle track seeding of primary and secondary liver carcinoma after percutaneous liver biopsy. HPB Surg. 1993;6(3):199-203. discussion 4
57 Regimbeau JM, Fargas O, Vilgrain V, et al. What is the local risk of recurrence after percutaneous biopsy of hepatocellular carcinoma? Hepato-Gastroenterol. 1995;42:601-606.
58 Agarwal PK, Husain N, Singh BN. Cytologic findings in aspirated hydatid fluid. Acta Cytol. 1989 Sep–Oct;33(5):652-654.
59 Das DK, Bhambhani S, Pant CS. Ultrasound guided fine-needle aspiration cytology: diagnosis of hydatid disease of the abdomen and thorax. Diagn Cytopathol. 1995 Mar;12(2):173-176.
60 Pogacnik A, Pohar-Marinsek Z, Us-Krasovec M. Fine needle aspiration biopsy in the diagnosis of liver echinococcosis. Acta Cytol. 1990 Sep–Oct;34(5):765-766.
61 Caturelli E, Rapaccini GL, Sabelli C, et al. Ultrasound-guided fine-needle aspiration biopsy in the diagnosis of hepatic hemangioma. Liver. 1986 Dec;6(6):326-330.
62 Langlois SL. Fine-needle biopsy of hepatic hydatids and haemangiomas: an overstated hazard. Australas Radiol. 1989 May;33(2):144-149.
63 Nakaizumi A, Iishi H, Yamamoto R, et al. Diagnosis of hepatic cavernous hemangioma by fine needle aspiration biopsy under ultrasonic guidance. Gastrointest Radiol. 1990 Winter;15(1):39-42.
64 Solbiati L, Livraghi T, De Pra L, et al. Fine-needle biopsy of hepatic hemangioma with sonographic guidance. AJR Am J Roentgenol. 1985 Mar;144(3):471-474.
65 Crowe DR, Eloubeidi MA, Chhieng DC, et al. Fine-needle aspiration biopsy of hepatic lesions: computerized tomographic-guided versus endoscopic ultrasound-guided FNA. Cancer. 2006 Jun 25;108(3):180-185.
66 Fritscher-Ravens A, Broering DC, Sriram PV, et al. EUS-guided fine-needle aspiration cytodiagnosis of hilar cholangiocarcinoma: a case series. Gastrointest Endosc. 2000 Oct;52(4):534-540.
67 Singh P, Erickson RA, Mukhopadhyay P, et al. EUS for detection of the hepatocellular carcinoma: results of a prospective study. Gastrointest Endosc. 2007 Aug;66(2):265-273.
68 Miller DA, Carrasco CH, Katz RL, et al. Fine needle aspiration biopsy: the role of immediate cytologic assessment. AJR Am J Roentgenol. 1986 Jul;147(1):155-158.
69 Hall-Craggs MA, Lees WR. Fine needle biopsy: cytology, histology or both? Gut. 1987 Mar;28(3):233-236.
70 Pinto MM, Avila NA, Heller CI, Criscuolo EM. Fine needle aspiration of the liver. Acta Cytol. 1988 Jan–Feb;32(1):15-21.
71 Tatsuta M, Yamamoto R, Kasugai H, et al. Cytohistologic diagnosis of neoplasms of the liver by ultrasonically guided fine-needle aspiration biopsy. Cancer. 1984 Oct 15;54(8):1682-1686.
72 Wee A, Nilsson B, Tan LK, Yap I. Fine needle aspiration biopsy of hepatocellular carcinoma. Diagnostic dilemma at the ends of the spectrum. Acta Cytol. 1994 May–Jun;38(3):347-354.
73 Silverman JF, Geisinger KR. Ancillary studies in FNA of liver and pancreas. Diagn Cytopathol. 1995 Dec;13(5):396-410.
74 Lundquist A, Akerman M. Fine-needle aspiration biopsy in acute hepatitis and liver cirrhosis. Ann Clin Res. 1970 Sep;2(3):197-203.
75 Tao LC. Liver and pancreas. In: Bibbo M, editor. Comprehensive Cytopathology. Philadelphia: Saunders; 1991:822-859.
76 Servoll E, Viste A, Skaarland E, et al. Fine-needle aspiration cytology of focal liver lesions. Advantages and limitations. Acta Chir Scand. 1988 Jan;154(1):61-63.
77 Bergman S, Graeme-Cook F, Pitman MB. The usefulness of the reticulin stain in the differential diagnosis of liver nodules on fine-needle aspiration biopsy cell block preparations. Mod Pathol. 1997 Dec;10(12):1258-1264.
78 Gagliano EF. Reticulin stain in the fine needle aspiration differential diagnosis of liver nodules. Acta Cytol. 1995 May–Jun;39(3):596-598.
79 Bottles K, Cohen MB. An approach to fine-needle aspiration biopsy diagnosis of hepatic masses. Diagn Cytopathol. 1991;7(2):204-210.
80 Terada T, Nakanuma Y. Iron-negative foci in siderotic macroregenerative nodules in human cirrhotic liver. A marker of incipient neoplastic lesions. Arch Pathol Lab Med. 1989 Aug;113(8):916-920.
81 Bose S, Kapila K, Verma K. Amyloidosis of the liver diagnosed by fine needle aspiration cytology. Acta Cytol. 1989 Nov–Dec;33(6):935-936.
82 Gangane N, Anshu, Shivkumar VB, Sharta S. Cytodiagnosis of hepatic amyloidosis by fine needle aspiration cytology: a case report. Acta Cytol. 2006 Sep-Oct;50(5):574-576.
83 Michael CW, Naylor B. Amyloid in cytologic specimens. Differential diagnosis and diagnostic pitfalls. Acta Cytol. 1999 Sep–Oct;43(5):746-755.
84 Srinivasan R, Nijhawan R, Gautam U, Bambery P. Potassium permanganate resistant amyloid in fine-needle aspirate of the liver. Diagn Cytopathol. 1994;10(4):383-384.
85 Stormby N, Akerman M. Aspiration cytology in the diagnosis of granulomatous liver lesions. Acta Cytol. 1973 May–Jun;17(3):200-204.
86 Wee A, Nilsson B, Wang TL, et al. Tuberculous pseudotumor causing biliary obstruction. Report of a case with diagnosis by fine needle aspiration biopsy and bile cytology. Acta Cytol. 1995 May–Jun;39(3):559-562.
87 Pinto MM, Kaye AD. Fine needle aspiration of cystic liver lesions. Cytologic examination and carcinoembryonic antigen assay of cyst contents. Acta Cytol. 1989 Nov–Dec;33(6):852-856.
88 De J, Rossman L, Kott MM, Deavers MT. Cytologic diagnosis of ciliated hepatic foregut cyst. Diagn Cytopathol. 2006 Dec;34(12):846-849.
89 Hornstein A, Batts KP, Linz LJ, et al. Fine needle aspiration diagnosis of ciliated hepatic foregut cysts: a report of three cases. Acta Cytol. 1996 May–Jun;40(3):576-580.
90 von Sinner WN, Nyman R, Linjawi T, Ali AM. Fine needle aspiration biopsy of hydatid cysts. Acta Radiol. 1995 Mar;36(2):168-172.
91 Singh A, Singh Y, Sharma VK, et al. Diagnosis of hydatid disease of abdomen and thorax by ultrasound guided fine needle aspiration cytology. Indian J Pathol Microbiol. 1999 Apr;42(2):155-156.
92 Vercelli-Retta J, Manana G, Reissenweber NJ. The cytologic diagnosis of hydatid disease. Acta Cytol. 1982 Mar–Apr;26(2):159-168.
93 Wee A, Nilsson B, Yap I, Chong SM. Aspiration cytology of liver abscesses. With an emphasis on diagnostic pitfalls. Acta Cytol. 1995 May–Jun;39(3):453-462.
94 Bhambhani S, Kashyap V. Amoebiasis: diagnosis by aspiration and exfoliative cytology. Cytopathology. 2001 Oct;12(5):329-333.
95 Granger JK, Houn HY. Diagnosis of hepatic actinomycosis by fine-needle aspiration. Diagn Cytopathol. 1991;7(1):95-97.
96 Shurbaji MS, Gupta PK, Newman MM. Hepatic actinomycosis diagnosed by fine needle aspiration. A case report. Acta Cytol. 1987 Nov–Dec;31(6):751-755.
97 Caturelli E, Rapaccini GL, Sabelli C, et al. Ultrasonography and echo-guided fine-needle biopsy in the diagnosis of focal fatty liver change. Hepatogastroenterology. 1987 Aug;34(4):137-140.
98 Layfield LJ. Focal fatty change of the liver: cytologic findings in a radiographic mimic of metastases. Diagn Cytopathol. 1994 Dec;11(4):385-387.
99 Anthony PP, Telesinghe PU. Inflammatory pseudotumour of the liver. J Clin Pathol. 1986 Jul;39(7):761-768.
100 Isobe H, Nishi Y, Fukutomi T, et al. Inflammatory pseudotumor of the liver associated with acute myelomonocytic leukemia. Am J Gastroenterol. 1991 Feb;86(2):238-240.
101 Lupovitch A, Chen R, Mishra S. Inflammatory pseudotumor of the liver. Report of the fine needle aspiration cytologic findings in a case initially misdiagnosed as malignant. Acta Cytol. 1989 Mar–Apr;33(2):259-262.
102 Malhotra V, Gondal R, Tatke M, Sarin SK. Fine needle aspiration cytologic appearance of inflammatory pseudotumor of the liver. A case report. Acta Cytol. 1997 Jul–Aug;41(4 Suppl):1325-1328.
103 Dardi LE, Marzano M, Froula E. Fine needle aspiration cytologic diagnosis of focal intrahepatic extramedullary hematopoiesis. Acta Cytol. 1990 Jul–Aug;34(4):567-569.
104 Lemos LB, Baliga M, Benghuzzi HA, Cason Z. Nodular hematopoiesis of the liver diagnosed by fine-needle aspiration cytology. Diagn Cytopathol. 1997 Jan;16(1):51-54.
105 Raab SS, Silverman JF, McLeod DL, Geisinger KR. Fine-needle aspiration cytology of extramedullary hematopoiesis (myeloid metaplasia). Diagn Cytopathol. 1993 Oct;9(5):522-526.
106 Ruschenburg I, Droese M. Fine needle aspiration cytology of focal nodular hyperplasia of the liver. Acta Cytol. 1989 Nov–Dec;33(6):857-860.
107 Adam YG, Nonas CJ. Hepatobiliary cystadenoma. South Med J. 1995 Nov;88(11):1140-1143.
108 Logrono R, Rampy BA, Adegboyega PA. Fine needle aspiration cytology of hepatobiliary cystadenoma with mesenchymal stroma. Cancer. 2002 Feb 25;96(1):37-42.
109 Filippi de la Palavesa MM, Vasilescu C, et al. Biliary cystadenocarcinoma: sonographic and cytologic findings. J Clin Ultrasound. 1999 May;27(4):210-212.
110 Del Poggio P, Jamoletti C, Forloni B, et al. Malignant transformation of biliary cystadenoma: a difficult diagnosis. Dig Liver Dis. 2000 Nov;32(8):733-736.
111 Iemoto Y, Kondo Y, Nakano T, et al. Biliary cystadenocarcinoma diagnosed by liver biopsy performed under ultrasonographic guidance. Gastroenterology. 1983 Feb;84(2):399-403.
112 Ishak KG, Willis GW, Cummins SD, Bullock AA. Biliary cystadenoma and cystadenocarcinoma: report of 14 cases and review of the literature. Cancer. 1977 Jan;39(1):322-338.
113 Wee A, Nilsson B, Kang JY, et al. Biliary cystadenocarcinoma arising in a cystadenoma. Report of a case diagnosed by fine needle aspiration cytology. Acta Cytol. 1993 Nov–Dec;37(6):966-970.
114 Woods GL. Biliary cystadenocarcinoma: Case report of hepatic malignancy originating in benign cystadenoma. Cancer. 1981 Jun 15;47(12):2936-2940.
115 Tao LC. Are oral contraceptive-associated liver cell adenomas premalignant? Acta Cytol. 1992 May-Jun;36(3):338-344.
116 Ali MA, Akhtar M, Mattingly RC. Morphologic spectrum of hepatocellular carcinoma in fine needle aspiration biopsies. Acta Cytol. 1986 May–Jun;30(3):294-302.
117 Bottles K, Cohen MB, Holly EA, et al. A step-wise logistic regression analysis of hepatocellular carcinoma. An aspiration biopsy study. Cancer. 1988 Aug 1;62(3):558-563.
118 Cohen MB, Haber MM, Holly EA, et al. Cytologic criteria to distinguish hepatocellular carcinoma from nonneoplastic liver. Am J Clin Pathol. 1991 Feb;95(2):125-130.
119 Das DK. Cytodiagnosis of hepatocellular carcinoma in fine-needle aspirates of the liver: its differentiation from reactive hepatocytes and metastatic adenocarcinoma. Diagn Cytopathol. 1999 Dec;21(6):370-377.
120 de Boer WB, Segal A, Frost FA, Sterrett GF. Cytodiagnosis of well differentiated hepatocellular carcinoma: can indeterminate diagnoses be reduced? Cancer. 1999 Oct 25;87(5):270-277.
121 Granados R, Aramburu JA, Murillo N, et al. Fine-needle aspiration biopsy of liver masses: diagnostic value and reproducibility of cytological criteria. Diagn Cytopathol. 2001 Dec;25(6):365-375.
122 Greene CL, Fehrman I, Nery D, et al. Liver transplant aspiration cytology (TAC) at three weeks and one year in healthy recipients with grafts free of histologic abnormality. Transplant Proc. 1989 Feb;21(1 Pt 2):2211-2212.
123 Kung IT, Chan SK, Fung KH. Fine-needle aspiration in hepatocellular carcinoma. Combined cytologic and histologic approach. Cancer. 1991 Feb 1;67(3):673-680.
124 Longchampt E, Patriarche C, Fabre M. Accuracy of cytology vs. microbiopsy for the diagnosis of well-differentiated hepatocellular carcinoma and macroregenerative nodule. Definition of standardized criteria from a study of 100 cases. Acta Cytol. 2000 Jul–Aug;44(4):515-523.
125 Noguchi S, Yamamoto R, Tatsuta M, et al. Cell features and patterns in fine-needle aspirates of hepatocellular carcinoma. Cancer. 1986 Jul 15;58(2):321-328.
126 Pedio G, Landolt U, Zobeli L, Gut D. Fine needle aspiration of the liver. Significance of hepatocytic naked nuclei in the diagnosis of hepatocellular carcinoma. Acta Cytol. 1988 Jul–Aug;32(4):437-442.
127 Pisharodi LR, Lavoie R, Bedrossian CW. Differential diagnostic dilemmas in malignant fine-needle aspirates of liver: a practical approach to final diagnosis. Diagn Cytopathol. 1995 Jun;12(4):364-370. discussion 70–1
128 Pitman MB, Szyfelbein WM. Significance of endothelium in the fine-needle aspiration biopsy diagnosis of hepatocellular carcinoma. Diagn Cytopathol. 1995 May;12(3):208-214.
129 Salomao DR, Lloyd RV, Goellner JR. Hepatocellular carcinoma: needle biopsy findings in 74 cases. Diagn Cytopathol. 1997 Jan;16(1):8-13.
130 Takenaka A, Kaji I, Kasugai H, et al. Usefulness of diagnostic criteria for aspiration cytology of hepatocellular carcinoma. Acta Cytol. 1999 Jul–Aug;43(4):610-616.
131 Wee A, Nilsson B. Highly well differentiated hepatocellular carcinoma and benign hepatocellular lesions. Can they be distinguished on fine needle aspiration biopsy? Acta Cytol. 2003 Jan–Feb;47(1):16-26.
132 Wee A, Nilsson B, Chan-Wilde C, et al. Cytological diagnosis from fine needle aspiration biopsy of the liver. Ann Acad Med Singapore. 1991 Mar;20(2):208-214.
133 Singh HK, Silverman JF, Geisinger KR. Fine-needle aspiration cytomorphology of clear-cell hepatocellular carcinoma. Diagn Cytopathol. 1997 Oct;17(4):306-310.
134 Buchanan TFJr, Huvos AG. Clear-cell carcinoma of the liver. A clinicopathologic study of 13 patients. Am J Clin Pathol. 1974 Apr;61(4):529-539.
135 Mathew T, Affandi MZ. Fine needle aspiration biopsy of a hepatic mass. An example of a near error. Acta Cytol. 1989 Nov–Dec;33(6):861-864.
136 Davenport RD. Cytologic diagnosis of fibrolamellar carcinoma of the liver by fine-needle aspiration. Diagn Cytopathol. 1990;6(4):275-279.
137 Perez Gil MA, Ruiz Recuento J, Relanzon Molinero S, et al. [Focal liver lesions in multiple myeloma: ecography, computed tomography, and magnetic resonance findings. A case report]. Radiologia. 2006 Jul–Aug;48(4):251-254.
138 Suen KC, Magee JF, Halparin LS, et al. Fine needle aspiration cytology of fibrolamellar hepatocellular carcinoma. Acta Cytol. 1985 Sep–Oct;29(5):867-872.
139 Gottschalk-Sabag S, Ron N, Glick T. Use of CD34 and factor VIII to diagnose hepatocellular carcinoma on fine needle aspirates. Acta Cytol. 1998 May–Jun;42(3):691-696.
140 de Boer WB, Segal A, Frost FA, Sterrett GF. Can CD34 discriminate between benign and malignant hepatocytic lesions in fine-needle aspirates and thin core biopsies? Cancer. 2000 Oct 25;90(5):273-278.
141 Kong CS, Appenzeller M, Ferrell LD. Utility of CD34 reactivity in evaluating focal nodular hepatocellular lesions sampled by fine needle aspiration biopsy. Acta Cytol. 2000 Mar–Apr;44(2):218-222.
142 Kandil D, Leiman G, Allegretta M, et al. Glypican-3 immunocytochemistry in liver fine-needle aspirates : a novel stain to assist in the differentiation of benign and malignant liver lesions. Cancer. 2007 Oct 25;111(5):316-322.
143 Ligato S, Mandich D, Cartun RW. Utility of glypican-3 in differentiating hepatocellular carcinoma from other primary and metastatic lesions in FNA of the liver: an immunocytochemical study. Mod Pathol. 2008 Feb 8.
144 Nassar A, Cohen C, Siddiqui MT. Utility of glypican-3 and survivin in differentiating hepatocellular carcinoma from benign and preneoplastic hepatic lesions and metastatic carcinomas in liver fine-needle aspiration biopsies. Diagn Cytopathol. 2009 Sep;37(9):629-635.
145 Cottier M, Jouffre C, Maubon I, et al. Prospective flow cytometric DNA analysis of hepatocellular carcinoma specimens collected by ultrasound-guided fine needle aspiration. Cancer. 1994 Jul 15;74(2):599-605.
146 Ng IO, Lai EC, Ho JC, et al. Flow cytometric analysis of DNA ploidy in hepatocellular carcinoma. Am J Clin Pathol. 1994 Jul;102(1):80-86.
147 Russo A, Bazan V, Plaja S, et al. Flow cytometric DNA analysis of hepatic tumours on ultrasound-guided fine-needle aspirates. J Surg Oncol. 1992 Sep;51(1):26-32.
148 Pitman MB. Fine needle aspiration biopsy of the liver. Principal diagnostic challenges. Clin Lab Med. 1998 Sep;18(3):483-506.
149 Renshaw AA, Haja J, Wilbur DC, Miller TR. Fine-needle aspirates of adenocarcinoma/metastatic carcinoma that resemble hepatocellular carcinoma: correlating cytologic features and performance in the College of American Pathologists Nongynecologic Cytology Program. Arch Pathol Lab Med. 2005 Oct;129(10):1217-1221.
150 Renshaw AA, Haja J, Wilbur DC, Miller TR. Fine-needle aspirates of hepatocellular carcinoma that are misclassified as adenocarcinoma: correlating cytologic features and performance in the College of American Pathologists Nongynecologic Cytology Program. Arch Pathol Lab Med. 2006 Jan;130(1):19-22.
151 Kakar S, Gown AM, Goodman ZD, Ferrell LD. Best practices in diagnostic immunohistochemistry: hepatocellular carcinoma versus metastatic neoplasms. Arch Pathol Lab Med. 2007 Nov;131(11):1648-1654.
152 Saleh HA, Aulicino M, Zaidi SY, et al. Discriminating hepatocellular carcinoma from metastatic carcinoma on fine-needle aspiration biopsy of the liver: the utility of immunocytochemical panel. Diagn Cytopathol. 2009 Mar;37(3):184-190.
153 Guindi M, Yazdi HM, Gilliatt MA. Fine needle aspiration biopsy of hepatocellular carcinoma. Value of immunocytochemical and ultrastructural studies. Acta Cytol. 1994 May–Jun;38(3):385-391.
154 Johnson DE, Powers CN, Rupp G, Frable WJ. Immunocytochemical staining of fine-needle aspiration biopsies of the liver as a diagnostic tool for hepatocellular carcinoma. Mod Pathol. 1992 Mar;5(2):117-123.
155 Rishi M, Kovatich A, Ehya H. Utility of polyclonal and monoclonal antibodies against carcinoembryonic antigen in hepatic fine-needle aspirates. Diagn Cytopathol. 1994 Dec;11(4):358-361. discussion 61–2
156 Wang L, Vuolo M, Suhrland MJ, Schlesinger K. HepPar1, MOC-31, pCEA, mCEA and CD10 for distinguishing hepatocellular carcinoma vs. metastatic adenocarcinoma in liver fine needle aspirates. Acta Cytol. 2006 May–Jun;50(3):257-262.
157 Wee A, Nilsson B. pCEA canalicular immunostaining in fine needle aspiration biopsy diagnosis of hepatocellular carcinoma. Acta Cytol. 1997 Jul–Aug;41(4):1147-1155.
158 Wolber RA, Greene CA, Dupuis BA. Polyclonal carcinoembryonic antigen staining in the cytologic differential diagnosis of primary and metastatic hepatic malignancies. Acta Cytol. 1991 Mar–Apr;35(2):215-220.
159 Ahuja A, Gupta N, Kalra N, et al. Role of CD10 immunochemistry in differentiating hepatocellular carcinoma from metastatic carcinoma of the liver. Cytopathology. 2008 Aug;19(4):229-235.
160 Lin F, Abdallah H, Meschter S. Diagnostic utility of CD10 in differentiating hepatocellular carcinoma from metastatic carcinoma in fine-needle aspiration biopsy (FNAB) of the liver. Diagn Cytopathol. 2004 Feb;30(2):92-97.
161 Siddiqui MT, Saboorian MH, Gokaslan ST, Ashfaq R. Diagnostic utility of the HepPar1 antibody to differentiate hepatocellular carcinoma from metastatic carcinoma in fine-needle aspiration samples. Cancer. 2002 Feb 25;96(1):49-52.
162 Zimmerman RL, Burke MA, Young NA, et al. Diagnostic value of hepatocyte paraffin 1 antibody to discriminate hepatocellular carcinoma from metastatic carcinoma in fine-needle aspiration biopsies of the liver. Cancer. 2001 Aug 25;93(4):288-291.
163 Kandil DH, Cooper K. Glypican-3: a novel diagnostic marker for hepatocellular carcinoma and more. Adv Anat Pathol. 2009 Mar;16(2):125-129.
164 Centeno BA. Pathology of liver metastases. Cancer Control. 2006 Jan;13(1):13-26.
165 Maeda T, Kajiyama K, Adachi E, et al. The expression of cytokeratins 7, 19, and 20 in primary and metastatic carcinomas of the liver. Mod Pathol. 1996 Sep;9(9):901-909.
166 Zimmerman RL, Burke M, Young NA, et al. Diagnostic utility of Glut-1 and CA 15-3 in discriminating adenocarcinoma from hepatocellular carcinoma in liver tumors biopsied by fine-needle aspiration. Cancer. 2002 Feb 25;96(1):53-57.
167 Lei JY, Bourne PA, diSant’Agnese PA, Huang J. Cytoplasmic staining of TTF-1 in the differential diagnosis of hepatocellular carcinoma vs cholangiocarcinoma and metastatic carcinoma of the liver. Am J Clin Pathol. 2006 Apr;125(4):519-525.
168 Wieczorek TJ, Pinkus JL, Glickman JN, Pinkus GS. Comparison of thyroid transcription factor-1 and hepatocyte antigen immunohistochemical analysis in the differential diagnosis of hepatocellular carcinoma, metastatic adenocarcinoma, renal cell carcinoma, and adrenal cortical carcinoma. Am J Clin Pathol. 2002 Dec;118(6):911-921.
169 Papotti M, Pacchioni D, Negro F, et al. Albumin gene expression in liver tumors: diagnostic interest in fine needle aspiration biopsies. Mod Pathol. 1994 Apr;7(3):271-275.
170 Stephen MR, Oien K, Ferrier RK, Burnett RA. Effusion cytology of hepatocellular carcinoma with in situ hybridisation for human albumin. J Clin Pathol. 1997 May;50(5):442-444.
171 Eloubeidi MA, Chen VK, Jhala NC, et al. Endoscopic ultrasound-guided fine needle aspiration biopsy of suspected cholangiocarcinoma. Clin Gastroenterol Hepatol. 2004 Mar;2(3):209-213.
172 Jain M, Ahluwalia C, Agarwal K, Pathania OP. Cytological diagnosis of cholangiocarcinoma with rib metastasis in a young female–a case report. Indian J Pathol Microbiol. 2004 Jul;47(3):417-420.
173 Tan G, Yilmaz A, De Young BR, et al. Immunohistochemical analysis of biliary tract lesions. Appl Immunohistochem Mol Morphol. 2004 Sep;12(3):193-197.
174 Chaudhary HB, Bhanot P, Logrono R. Phenotypic diversity of intrahepatic and extrahepatic cholangiocarcinoma on aspiration cytology and core needle biopsy: case series and review of the literature. Cancer. 2005 Aug 25;105(4):220-228.
175 Gibbons D, de las Morenas A. Fine needle aspiration diagnosis of combined hepatocellular carcinoma and cholangiocarcinoma. A case report. Acta Cytol. 1997 Jul–Aug;41(4 Suppl):1269-1272.
176 Wee A, Nilsson B. Combined hepatocellular-cholangiocarcinoma. Diagnostic challenge in hepatic fine needle aspiration biopsy. Acta Cytol. 1999 Mar–Apr;43(2):131-138.
177 Dekmezian R, Sneige N, Popok S, Ordonez NG. Fine-needle aspiration cytology of paediatric patients with primary hepatic tumors: a comparative study of two hepatoblastomas and a liver-cell carcinoma. Diagn Cytopathol. 1988;4(2):162-168.
178 Iyer VK, Kapila K, Agarwala S, Verma K. Fine needle aspiration cytology of hepatoblastoma. Recognition of subtypes on cytomorphology. Acta Cytol. 2005 Jul–Aug;49(4):355-364.
179 Parikh B, Jojo A, Shah B, et al. Fine needle aspiration cytology of hepatoblastoma: a study of 20 cases. Indian J Pathol Microbiol. 2005 Jul;48(3):331-336.
180 Sola Perez J, Perez-Guillermo M, Bas Bernal AB, Mercader JM. Hepatoblastoma. An attempt to apply histologic classification to aspirates obtained by fine needle aspiration cytology. Acta Cytol. 1994 Mar–Apr;38(2):175-182.
181 Wakely PEJr, Silverman JF, Geisinger KR, Frable WJ. Fine needle aspiration biopsy cytology of hepatoblastoma. Mod Pathol. 1990 Nov;3(6):688-693.
182 Fasano M, Theise ND, Nalesnik M, et al. Immunohistochemical evaluation of hepatoblastomas with use of the hepatocyte-specific marker, hepatocyte paraffin 1, and the polyclonal anti-carcinoembryonic antigen. Mod Pathol. 1998 Oct;11(10):934-938.
183 Collins KA, Geisinger KR, Raab SS, Silverman JF. Fine needle aspiration biopsy of hepatic lymphomas: cytomorphology and ancillary studies. Acta Cytol. 1996 Mar–Apr;40(2):257-262.
184 Rappaport KM, DiGiuseppe JA, Busseniers AE. Primary hepatic lymphoma: report of two cases diagnosed by fine-needle aspiration. Diagn Cytopathol. 1995 Aug;13(2):142-145.
185 Jansson SE, Bondestam S, Heinonen E, et al. Value of liver and spleen aspiration biopsy in malignant diseases when these organs show no signs of involvement in sonography. Acta Med Scand. 1983;213(4):279-281.
186 Gattuso P, Castelli MJ, Peng Y, Reddy VB. Posttransplant lymphoproliferative disorders: a fine-needle aspiration biopsy study. Diagn Cytopathol. 1997 May;16(5):392-395.
187 Raymond E, Tricottet V, Samuel D, et al. Epstein-Barr virus-related localized hepatic lymphoproliferative disorders after liver transplantation. Cancer. 1995 Oct 15;76(8):1344-1351.
188 Layfield LJ, Mooney EE, Dodd LG. Not by blood alone: diagnosis of hemangiomas by fine-needle aspiration. Diagn Cytopathol. 1998 Oct;19(4):250-254.
189 Taavitsainen M, Airaksinen T, Kreula J, Paivansalo M. Fine-needle aspiration biopsy of liver hemangioma. Acta Radiol. 1990 Jan;31(1):69-71.
190 Manucha V, Sun CC. Cytologic findings and differential diagnosis in hepatic Epithelioid hemangioendothelioma: a case report. Acta Cytol. 2008 Nov–Dec;52(6):713-717.
191 Cho NH, Lee KG, Jeong MG. Cytologic evaluation of primary malignant vascular tumors of the liver. One case each of angiosarcoma and epithelioid hemangioendothelioma. Acta Cytol. 1997 Sep–Oct;41(5):1468-1476.
192 Soslow RA, Yin P, Steinberg CR, Yang GC. Cytopathologic features of hepatic epithelioid hemangioendothelioma. Diagn Cytopathol. 1997 Jul;17(1):50-53.
193 Boucher LD, Swanson PE, Stanley MW, et al. Cytology of angiosarcoma. Findings in fourteen fine-needle aspiration biopsy specimens and one pleural fluid specimen. Am J Clin Pathol. 2000 Aug;114(2):210-219.
194 Liu K, Layfield LJ. Cytomorphologic features of angiosarcoma on fine needle aspiration biopsy. Acta Cytol. 1999 May–Jun;43(3):407-415.
195 Wong JW, Bedard YC. Fine-needle aspiration biopsy of hepatic angiosarcoma: report of a case with immunocytochemical findings. Diagn Cytopathol. 1992;8(4):380-383.
196 Cha I, Cartwright D, Guis M, et al. Angiomyolipoma of the liver in fine-needle aspiration biopsies: its distinction from hepatocellular carcinoma. Cancer. 1999 Feb 25;87(1):25-30.
197 Ma TK, Tse MK, Tsui WM, Yuen KT. Fine needle aspiration diagnosis of angiomyolipoma of the liver using a cell block with immunohistochemical study. A case report. Acta Cytol. 1994 Mar–Apr;38(2):257-260.
198 Sawai H, Manabe T, Yamanaka Y, et al. Angiomyolipoma of the liver: case report and collective review of cases diagnosed from fine needle aspiration biopsy specimens. J Hepatobiliary Pancreat Surg. 1998;5(3):333-338.
199 al-Rikabi AC, Buckai A, al-Sumayer S, et al. Fine needle aspiration cytology of mesenchymal hamartoma of the liver. A case report. Acta Cytol. 2000 May–Jun;44(3):449-453.
200 Jimenez-Heffernan JA, Vicandi B, Lopez-Ferrer P, et al. Fine-needle aspiration cytology of mesenchymal hamartoma of the liver. Diagn Cytopathol. 2000 Apr;22(4):250-253.
201 Drachenberg CB, Papadimitriou JC, Rivero MA, Wood C. Distinctive case. Adult mesenchymal hamartoma of the liver: report of a case with light microscopic, FNA cytology, immunohistochemistry, and ultrastructural studies and review of the literature. Mod Pathol. 1991 May;4(3):392-395.
202 Cheuk W, Lee KC, Chan JK. c-kit immunocytochemical staining in the cytologic diagnosis of metastatic gastrointestinal stromal tumor. A report of two cases. Acta Cytol. 2000 Jul–Aug;44(4):679-685.
203 Padilla C, Saez A, Vidal A, et al. Fine-needle aspiration cytology diagnosis of metastatic gastrointestinal stromal tumor in the liver: a report of three cases. Diagn Cytopathol. 2002 Nov;27(5):298-302.
204 Wieczorek TJ, Faquin WC, Rubin BP, Cibas ES. Cytologic diagnosis of gastrointestinal stromal tumor with emphasis on the differential diagnosis with leiomyosarcoma. Cancer. 2001 Aug 25;93(4):276-287.
205 Smith MB, Silverman JF, Raab SS, et al. Fine-needle aspiration cytology of hepatic leiomyosarcoma. Diagn Cytopathol. 1994 Dec;11(4):321-327.
206 Wee A, Nilsson B. Fine needle aspiration biopsy of hepatic leiomyosarcoma. An unusual epithelioid variant posing a potential diagnostic pitfall in a hepatocellular carcinoma-prevalent population. Acta Cytol. 1997 May–Jun;41(3):737-743.
207 Garcia-Bonafe M, Allende H, Fantova MJ, Tarragona J. Fine needle aspiration cytology of undifferentiated (embryonal) sarcoma of the liver. A case report. Acta Cytol. 1997 Jul–Aug;41(4 Suppl):1273-1278.
208 Gupta C, Iyer VK, Kaushal S, et al. Fine needle aspiration cytology of undifferentiated embryonal sarcoma of the liver. Cytopathology Jan 22.
209 Kaur J, Dey P, Das A. Fine needle aspiration cytology of undifferentiated (embryonal) sarcoma of liver in an adult male. Diagn Cytopathol Jul;38(7):547–548.
210 Sola-Perez J, Perez-Guillermo M, Gimenez-Bascunana A, Garre-Sanchez C. Cytopathology of undifferentiated (embryonal) sarcoma of the liver. Diagn Cytopathol. 1995 Jul;13(1):44-51.
211 Khalbuss WE, Grigorian S, Bui MM, Elhosseiny A. Small-cell tumors of the liver: a cytological study of 91 cases and a review of the literature. Diagn Cytopathol. 2005 Jul;33(1):8-14.
212 Miralles TG, Gosabez F, de Lera J, et al. Percutaneous fine needle aspiration biopsy cytology of the liver for staging small cell lung carcinoma. Comparison with other methods. Acta Cytol. 1993 Jul-Aug;37(4):499-502.
213 Prosser JM, Dusenbery D. Histocytologic diagnosis of neuroendocrine tumors in the liver: a retrospective study of 23 cases. Diagn Cytopathol. 1997 May;16(5):383-391.
214 Bocking A, Pomjansky N, Buckstegge B, Onofre A. [Immunocytochemical identification of carcinomas of unknown primaries on fine-needle-aspiration-biopsies]. Pathologe. 2009 Dec;30(Suppl 2):158-160.
215 Onofre AS, Pomjanski N, Buckstegge B, Bocking A. Immunocytochemical diagnosis of hepatocellular carcinoma and identification of carcinomas of unknown primary metastatic to the liver on fine-needle aspiration cytologies. Cancer. 2007 Aug 25;111(4):259-268.
216 Caraway NP, Fanning CV. Use of fine-needle aspiration biopsy in the evaluation of splenic lesions in a cancer center. Diagn Cytopathol. 1997 Apr;16(4):312-316.
217 Eloubeidi MA, Varadarajulu S, Eltoum I, et al. Transgastric endoscopic ultrasound-guided fine-needle aspiration biopsy and flow cytometry of suspected lymphoma of the spleen. Endoscopy. 2006 Jun;38(6):617-620.
218 Friedlander MA, Wei XJ, Iyengar P, Moreira AL. Diagnostic pitfalls in fine needle aspiration biopsy of the spleen. Diagn Cytopathol. 2008 Feb;36(2):69-75.
219 Iwashita T, Yasuda I, Tsurumi H, et al. Endoscopic ultrasound-guided fine needle aspiration biopsy for splenic tumor: a case series. Endoscopy. 2009 Feb;41(2):179-182.
220 Lal A, Ariga R, Gattuso P, et al. Splenic fine needle aspiration and core biopsy. A review of 49 cases. Acta Cytol. 2003 Nov–Dec;47(6):951-959.
221 Lishner M, Lang R, Hamlet Y, et al. Fine needle aspiration biopsy in patients with diffusely enlarged spleens. Acta Cytol. 1996 Mar–Apr;40(2):196-198.
222 Ramdall RB, Cai G, Alasio TM, Levine P. Fine-needle aspiration biopsy for the primary diagnosis of lymphoproliferative disorders involving the spleen: one institution’s experience and review of the literature. Diagn Cytopathol. 2006 Dec;34(12):812-817.
223 Schwerk WB, Maroske D, Roth S, Arnold R. [Ultrasound-guided fine-needle puncture in the diagnosis and therapy of liver and spleen abscesses]. Dtsch Med Wochenschr. 1986 May 30;111(22):847-853.
224 Silverman JF, Geisinger KR, Raab SS, Stanley MW. Fine needle aspiration biopsy of the spleen in the evaluation of neoplastic disorders. Acta Cytol. 1993 Mar–Apr;37(2):158-162.
225 Soderstrom N. How to use cytodiagnostic spleen puncture. Acta Med Scand. 1976;199(1–2):1-5.
226 Zeppa P, Vetrani A, Luciano L, et al. Fine needle aspiration biopsy of the spleen. A useful procedure in the diagnosis of splenomegaly. Acta Cytol. 1994 May–Jun;38(3):299-309.
227 Kumar PV, Monabati A, Raseki AR, et al. Splenic lesions: FNA findings in 48 cases. Cytopathology. 2007 Jun;18(3):151-156.
228 Nerlich A, Permanetter W. Fine needle aspiration cytodiagnosis of epidermoid cysts of the spleen. Report of two cases. Acta Cytol. 1991 Sep–Oct;35(5):567-569.
229 Kang M, Kalra N, Gulati M, et al. Image guided percutaneous splenic interventions. Eur J Radiol. 2007 Oct;64(1):140-146.
230 Haque I, Haque MZ, Krishnani N, et al. Fine needle aspiration cytology of the spleen in visceral leishmaniasis. Acta Cytol. 1993 Jan–Feb;37(1):73-76.
231 Rajwanshi A, Gupta D, Kapoor S, et al. Fine needle aspiration biopsy of the spleen in pyrexia of unknown origin. Cytopathology. 1999 Jun;10(3):195-200.
232 Selroos O, Koivunen E. Usefulness of fine-needle aspiration biopsy of spleen in diagnosis of sarcoidosis. Chest. 1983 Feb;83(2):193-195.
233 Taavitsainen M, Koivuniemi A, Helminen J, et al. Aspiration biopsy of the spleen in patients with sarcoidosis. Acta Radiol. 1987 Nov–Dec;28(6):723-725.
234 Sen R, Bhadani PP, Singh H, et al. Myeloid metaplasia in aspirates from enlarged spleens: a clue in the absence of peripheral blood findings characteristic of myelofibrosis. Acta Cytol. 2006 Jul–Aug;50(4):379-383.
235 Colovic R, Micev M, Grubor N, et al. [Inflammatory pseudotumours of spleen]. Srp Arh Celok Lek. 2009 Mar–Apr;137(3–4):189-193.
236 Pasternack A. Fine-needle aspiration biopsy of spleen in diagnosis of generalized amyloidosis. Br Med J. 1974 Apr 6;2(5909):20-22.
237 Domanski H, Dejmek A, Ljung R. Gaucher’s disease in an infant diagnosed by fine needle aspiration of the liver and spleen. A case report. Acta Cytol. 1992 May–Jun;36(3):410-412.
238 Moriarty AT, Schwenk GRJr, Chua G. Splenic fine needle aspiration biopsy in the diagnosis of lymphoreticular diseases. A report of four cases. Acta Cytol. 1993 Mar–Apr;37(2):191-196.
239 Comperat E, Bardier-Dupas A, Camparo P, et al. Splenic metastases: clinicopathologic presentation, differential diagnosis, and pathogenesis. Arch Pathol Lab Med. 2007 Jun;131(6):965-969.
240 Delacruz V, Jorda M, Gomez-Fernandez C, et al. Fine-needle aspiration diagnosis of angiosarcoma of the spleen: a case report and review of the literature. Arch Pathol Lab Med. 2005 Aug;129(8):1054-1056.
241 Lee SH. Fine-needle aspiration cytology of splenic hamartoma. Diagn Cytopathol. 2003 Feb;28(2):82-85.
242 Fritscher-Ravens A, Mylonaki M, Pantes A, et al. Endoscopic ultrasound-guided biopsy for the diagnosis of focal lesions of the spleen. Am J Gastroenterol. 2003 May;98(5):1022-1027.
243 Gomez-Rubio M, Lopez-Cano A, Rendon P, et al. Safety and diagnostic accuracy of percutaneous ultrasound-guided biopsy of the spleen: a multicenter study. J Clin Ultrasound. 2009 Oct;37(8):445-450.
244 Cavanna L, Artioli F, Vallisa D, et al. Primary lymphoma of the spleen. Report of a case with diagnosis by fine-needle guided biopsy. Haematologica. 1995 May–Jun;80(3):241-243.
245 Civardi G, Vallisa D, Berte R, et al. Ultrasound-guided fine needle biopsy of the spleen: high clinical efficacy and low risk in a multicenter Italian study. Am J Hematol. 2001 Jun;67(2):93-99.
246 Lindgren PG, Hagberg H, Eriksson B, et al. Excision biopsy of the spleen by ultrasonic guidance. Br J Radiol. 1985 Sep;58(693):853-857.
247 Lopez JI, Del Cura JL, De Larrinoa AF, et al. Role of ultrasound-guided core biopsy in the evaluation of spleen pathology. APMIS. 2006 Jul–Aug;114(7–8):492-499.
248 Goldfinger M, Cohen MM, Steinhardt MI, et al. Sonography and percutaneous aspiration of splenic epidermoid cyst. J Clin Ultrasound. 1986 Feb;14(2):147-149.
249 Bonifacio A, Goldberg RE, Patterson BJ, Haider M. Flow-cytometry-enhanced fine-needle aspiration biopsy of the spleen. Can Assoc Radiol J. 2000 Jun;51(3):158-162.
250 Zeppa P, Picardi M, Marino G, Troncone G, et al. Fine-needle aspiration biopsy and flow cytometry immunophenotyping of lymphoid and myeloproliferative disorders of the spleen. Cancer. 2003 Apr 25;99(2):118-127.
251 Gupta R, Jain P, Bakshi S, Sharma MC. Primary Hodgkin’s disease of spleen – a case report. Indian J Pathol Microbiol. 2006 Jul;49(3):435-437.
252 Meara RS, Reddy V, Arnoletti JP, et al. Hairy cell leukemia: a diagnosis by endoscopic ultrasound guided fine needle aspiration. Cytojournal. 2006;3:1.
253 Pinto RG, Rocha PD, Vernekar JA. Fine needle aspiration of the spleen in hairy cell leukemia. A case report. Acta Cytol. 1995 Jul–Aug;39(4):777-780.
254 Barbazza R, De Martini A, Mognol M, et al. Fine needle aspiration biopsy of a splenic hemangioma. A case report with review of the literature. Haematologica. 1990 May–Jun;75(3):278-281.
255 Ramdall RB, Alasio TM, Cai G, Yang GC. Primary vascular neoplasms unique to the spleen: littoral cell angioma and splenic hamartoma diagnosis by fine-needle aspiration biopsy. Diagn Cytopathol. 2007 Mar;35(3):137-142.
256 Conlon PJ, Procop GW, Fowler V, et al. Predictors of prognosis and risk of acute renal failure in patients with Rocky Mountain spotted fever. Am J Med. 1996 Dec;101(6):621-626.
257 Cavanna L, Lazzaro A, Vallisa D, et al. Role of image-guided fine-needle aspiration biopsy in the management of patients with splenic metastasis. World J Surg Oncol. 2007;5:13.