CHAPTER 4 Head and neck; salivary glands
Clinical aspects
The proximity of tissues of various types and the wide range of primary and metastatic neoplasms are responsible for this site being among the most interesting and challenging in FNAC diagnosis. Close cooperation with the clinician and the radiologist is necessary to clarify the anatomical relations of the target lesion, the nature of any previous lesion, and the details of any prior therapy.
FNB, as a minimally invasive technique, is particularly suitable in this sensitive area where an incisional biopsy can cause problems. A cytological diagnosis of a non-neoplastic lesion, or confirming suspected metastatic or recurrent tumor can obviate the need for surgery. In other cases, categorisation of disease to guide clinical management including further investigation, appropriate referral or rational planning of surgery can be offered. A type-specific cytological diagnosis is often possible but may require special experience and the use of ancillary laboratory techniques, and is often better deferred to the histological examination of paraffin blocks.
A discussion and review of the usefulness, indications and techniques of FNAC of tumors in the head and neck, with general guidelines for the UK, appeared recently in Cytopathology.1
Head and neck
Lesions of the salivary glands are presented in a separate section of this chapter, cervical lymph nodes in Chapter 5 and lesions of the thyroid in Chapter 6.
The place of FNA in the investigative sequence
The investigation of suspected local recurrence or nodal metastasis of previously diagnosed cancer is a common indication for FNB in the head and neck. It is of considerable clinical value in the management of these patients since therapeutic decisions can be made without delay and without the need for further diagnostic surgery. It is usually not difficult to distinguish between tumor recurrence, on the one hand, and inflammation or scarring, on the other. However, it may be difficult to locate a small recurrence in an area of post-radiation edema and fibrosis and to obtain a representative sample. The difficulties in distinguishing radiation-induced cellular atypia from recurrent tumor are well known.
The most common primary tumors in the head and neck are squamous cell carcinoma of the lip, tongue, oral cavity, larynx, etc. Adenoma, adenocarcinoma, lymphoma and sarcoma are also encountered in many of these sites. Tumors involving a mucous membrane of the upper digestive or respiratory tracts are usually diagnosed by conventional surgical or endoscopic biopsy or by cytological examination of brush or scrape smears. Lesions that do not involve a mucous membrane are accessible to preoperative FNB, which can be performed directly under visual control or with radiological guidance. This applies to numerous different sites: scalp, eyelids, pinna of ear, nose, oral cavity, nasal sinuses, floor of mouth, tongue, palate, tonsils, nasopharynx, pharynx and parapharyngeal space.2 Branchial and thyroglossal cysts are easily sampled. A variety of orbital and intraocular tumors, e.g. lacrimal gland tumors, lymphoma, retinoblastoma and melanoma, have been successfully diagnosed by FNB.
FNAC has been shown to be helpful also in intraoperative assessment of head and neck masses.3 The application of FNAC in the investigation of head and neck tumors in children has been studied by Rapkiewicz et al.4
Single examples of serious complications have been reported following FNB of carotid body and glomus jugulare tumors.5 Confirmation by radiological investigation is therefore preferable to needle biopsy. However, since the diagnosis may not be suspected clinically and paragangliomas can occur in unexpected sites, the pathologist must be familiar with the cytological features of these tumors.
Accuracy of diagnosis
The diagnostic accuracy of FNAC in suspected recurrent or metastatic tumors is generally high. Small nodal metastases can be missed and very well-differentiated squamous cell carcinoma can be misinterpreted as benign. Diagnosis can be difficult in irradiated tissues, with some risk of a false-positive cytological diagnosis. Samples must be quantitatively and qualitatively satisfactory, and unequivocal criteria of malignancy must be met for a positive diagnosis.
In primary diagnosis, accuracy varies with the size and site of the lesion, the tissue of origin and the nature of the process.6-8 The potential for cytological diagnosis of all kinds of lesions in the head and neck has been confirmed by numerous case reports and small series of cases, but relatively few large series of specific entities have been analyzed statistically. In any case, even if a definitive, type-specific diagnosis is not possible, FNB can provide cytological categorization of the disease process with a list of differential diagnoses to guide further investigations.
Cystic lesions in the neck are common and constitute an important problem in FNAC. For example, the distinction between inflamed branchial cyst and nodal metastasis of well-differentiated squamous cell carcinoma with liquefactive necrosis can be quite difficult. False-negative diagnoses are common and false-positive diagnoses have also been reported. This problem is discussed below.
Technical considerations
Non-aspiration sampling with a 27–25-gauge needle is recommended for superficial, easily accessible lesions. Careful and gentle needling is minimally traumatic, admixture with blood is less and the operator gets a better feel of the consistency of the tissues than with the conventional technique. A syringe in a pistol grip can be used for lesions in the oral cavity or pharynx to provide sufficient operating length to reach the target. A spray surface anesthetic is useful in transmucosal biopsies. CT or US guidance is needed for non-palpable and deep-sited targets, and to obtain representative samples from small, heterogeneous or partly cystic lesions. Both air-dried and alcohol-fixed smears should be made routinely, and spare slides kept for special stains or immune markers. Cell blocks or liquid-based preparations can be very useful for voluminous aspirates of mainly fluid or blood. Selective, cost-effective use of ancillary techniques is best achieved if the pathologist is present at the biopsy and can assess samples immediately. In lesions of a suspected infectious nature, material should be collected specifically for microbiological investigation.
Salivary glands
Salivary gland tumors are generally not subjected to incisional or core needle biopsy because of the possible risk of causing a fistula or disruption of the capsule with seeding of tumor cells and subsequent recurrence. There is no evidence that FNB causes either of these complications.
The place of FNA in the investigative sequence
The 1991 WHO classification of salivary gland tumors lists nine types of primary benign tumors (adenomas) and 18 types of malignant tumors (carcinomas), some with subtypes. In addition, there are non-epithelial tumors, malignant lymphoma, secondary tumors and a number of tumor-like conditions.9 Faced with this extraordinary variety of entities, a precise diagnosis by FNAC may seem an impossible task. However, the aim of FNAC combined with clinical and radiological findings is to provide a preliminary assessment on which management decisions can be based, not necessarily a definitive, type-specific diagnosis. Is surgery indicated or can the lesion be watched? How urgent is the surgery and how extensive is it likely to be?
We recommend a stepwise approach to the cytological diagnosis of salivary gland lesions:
The potential cost-savings achievable by preoperative FNB of salivary gland tumors have been analyzed by Layfield et al.12
Accuracy of diagnosis
A number of papers documenting the diagnostic accuracy of FNB in large numbers of cases of salivary gland neoplasms of several types were published from the Karolinska Hospital in Sweden in the 1960s.13-17 Over 90% of neoplasms were recognized, over 90% of pleomorphic adenomas were correctly typed, and most malignant tumors were diagnosed as such. The accuracy increased with increasing experience. Many other studies of large series of cases have since followed.18-20 A review of the literature in 1994 found that the diagnostic sensitivity varied between 81% and 100%, specificity was 94–100% and the accuracy of tumor typing was 61–80%.21 In a more recent study by Klijanienko et al. the sensitivity was 94%, specificity was 97% and the accuracy was 95%.22
Using the criteria developed by the Karolinska group reinforced by later authors, the diagnosis of pleomorphic adenoma and of Warthin’s tumor is reliable in most cases. Adenoid cystic and acinic cell carcinomas also have distinctive cytological features. However, there are many pitfalls. These may be due to sampling problems, for example false-negative diagnoses in cystic tumors. Pleomorphic adenoma, Warthin’s tumor, low-grade mucoepidermoid carcinoma and acinic cell carcinoma can all occasionally be predominantly cystic. The limitation to accuracy due to the small size and selective character of FNB samples has been mentioned. Other examples of diagnostic difficulties are regenerative epithelial hyperplasia and squamous metaplasia in sialadenitis or Warthin’s tumor, and epithelial atypia and high cellularity in pleomorphic adenoma. Overlapping cytological features between different tumor types are common, for example hyaline stromal globules initially regarded as distinctive of adenoid cystic carcinoma can occur in other tumors. The distinction between primary and metastatic cancer may be very difficult in poorly differentiated malignancies.
Problems and pitfalls in FNAC diagnosis of salivary gland lesions have attracted considerable interest and a number of papers on this topic can be found in the literature.23-27
Complications
Needling of non-neoplastic lesions, particularly in the submandibular gland, is often moderately painful and may cause local bleeding. Blood may seep into the mouth causing patient concern, and may cause some post-biopsy swelling. Tumor implantation in the needle track has not been reported post-FNB, nor has serious damage to adjacent structures such as the facial nerve.
Infarction or hemorrhage of salivary gland tumors post-FNB occasionally occurs.28 Necrosis and subsequent reactive changes and repair can cause difficulties in histological diagnosis.29,30 A gentle biopsy technique using thin needles reduces the incidence of this complication.
Technical considerations
Biopsy without aspiration using a 27–25-gauge needle is recommended. Heavily bloodstained samples may contain tissue fragments, which can be recovered in a cell block. Liquid-based preparations can be useful as a supplement to conventional smears.31 Fluid aspirated from both non-neoplastic and neoplastic cystic lesions is often very poor in cells. Material obtained from the cyst wall is more likely to be diagnostic. If this is not possible, follow-up should be recommended and the biopsy repeated with US guidance. Most cystic lesions are multilocular so that complete emptying is not possible except in the rare simple cysts.
We recommend the use of both air-dried (MGG, Diff-Quik) and wet-fixed (Pap, H&E) smears in parallel. A mucin stain can be helpful, and cell blocks in selected cases.
FNB material can be used for various ancillary techniques for which cell blocks or liquid-based preparations are particularly suitable. Immunocytochemistry has a limited role in FNAC of salivary gland lesions. Antibodies directed against S-100 protein, keratins, GFAP, smooth muscle actin, desmin and p63 may be useful to differentiate pleomorphic adenoma, myoepithelioma, myoepithelial carcinoma, rhabdomyosarcoma, sarcoma and epithelial-myoepithelial carcinoma. Cytogenetic rearrangements have been found in pleomorphic adenoma and mucoepidermoid carcinoma. Loss of heterozygosity has been described in pleomorphic adenoma, adenoid cystic carcinoma, carcinoma ex pleomorphic adenoma and mucoepidermoid carcinoma. HPV DNA is commonly seen in head and neck squamous carcinomas and its identification by molecular techniques is a valuable guide to diagnosis in some cases (see below cystic squamous lesions, p. 41).
Cytological findings
Non-neoplastic lesions
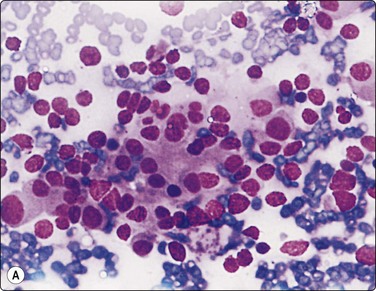
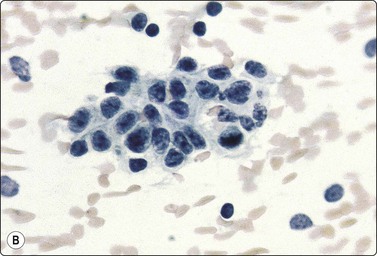
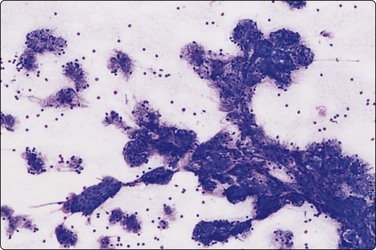
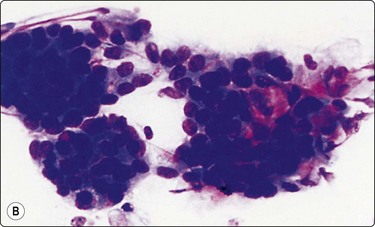
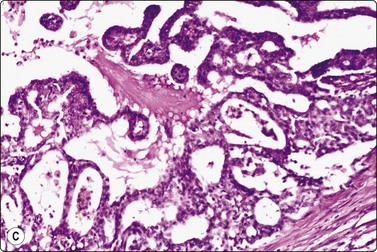
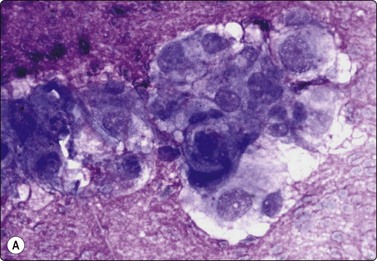
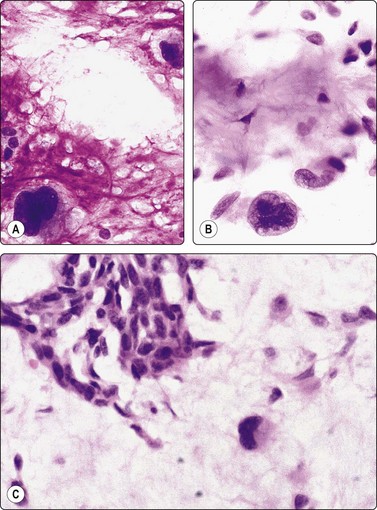
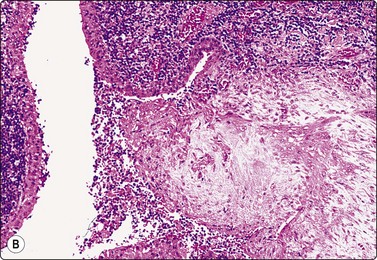
Branchial cyst (Fig. 4.1)32,33
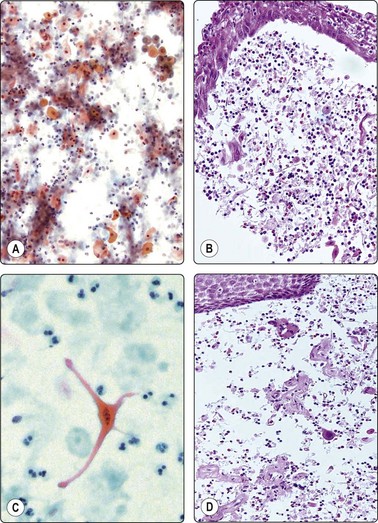
(A) FNB smear. Neutrophils, debris and mature squamous cells including degenerate forms; (B) Corresponding tissue (C, D) Other case Single highly atypical squamous cells in FNB smear (C) and in tissue section (D).
A branchial cyst can develop relatively rapidly as a firm mass of significant size in the lateral neck. It is most often seen in young adults but may become clinically apparent at any age. The sudden appearance of a mass may cause both the patient and the doctor considerable anxiety. A malignant cervical lymph node or a thyroid tumor may be suspected, or it may result in a useless course of antibiotics. An instant diagnosis by FNB is therefore of clinical value.
The aspiration of fluid causes the mass to decrease in size but it rarely disappears completely. The aspirate resembles pus also in non-inflamed cysts but the fluid is usually sterile. Sometimes the fluid contains large numbers of acute inflammatory cells. Multinucleate giant cells representing a granulomatous reaction in the wall of the cyst are sometimes seen. Lymphoid tissue, although evident in tissue sections, is not often represented in smears.
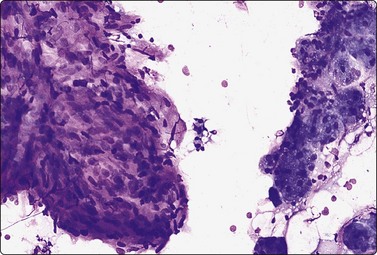
Nodal metastasis of well-differentiated SCC with liquefactive necrosis is a common, important and often difficult differential diagnosis (Figs. 4.2 and 4.3).34,35 Although 75% of branchial cysts occur in the age group 20–40 and metastatic squamous carcinoma mainly in patients over 40, there is a considerable overlap in the age group 40–60.32 We have seen several patients in whom a branchial cyst first presented clinically at the age of 60 or even later, and on the other hand, metastatic SCC can occur in young adults. The sensitivity of FNB in diagnosing malignancy in lateral cervical cysts varies widely (35–75%). In a review of the literature by Sheahan et al.36 4–24% of cases initially diagnosed as branchial cysts had unsuspected squamous cell carcinoma on histological examination. Many therefore recommend biopsy for histology even when FNB is negative, especially in patients over 40.
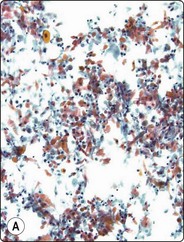
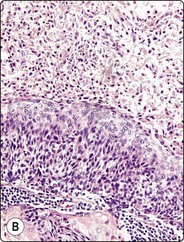
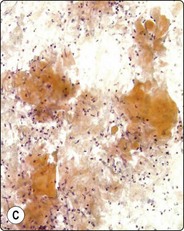
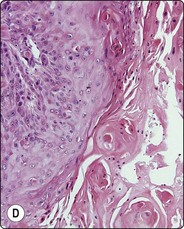
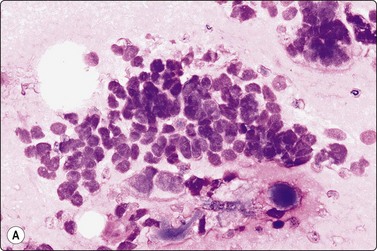
Fig. 4.2 Cystically degenerate metastasis of well-differentiated squamous carcinoma
(A, B) Exfoliating atypical cells of ‘parakeratotic’ type in FNB smear and corresponding tissue section; (C, D) Other example with predominance of anucleate keratinized cells and clumps of keratin in the cyst lumen.
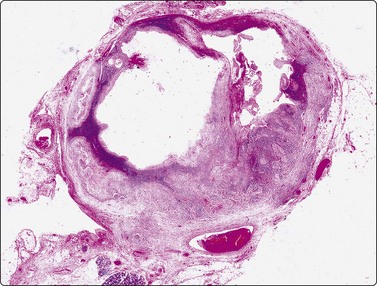
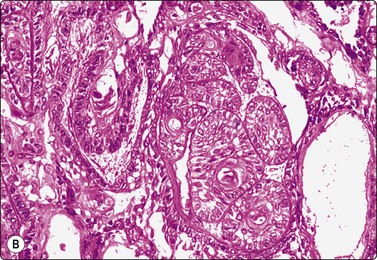
Fig. 4.3 Cystic metastasis of squamous carcinoma
Whole section of cervical lymph node containing metastatic deposit of squamous carcinoma with central cystic degeneration. Note similarity to branchial cyst (HE).
The diagnostic difficulties are due to the fact that squamous epithelial cells aspirated from a cystic metastasis of well-differentiated SCC are often anucleate or of parakeratotic type with a mature cytoplasm and a small pyknotic nucleus appearing cytologically bland, while inflammation of a benign cyst can result in immature squamous metaplasia and worrying cytological atypia. Figures 4.1 and 4.2 compare cells exfoliating from the lining of inflamed branchial cysts with those from cystic SCCs seen in histological sections. Helpful clues are that material sampled from a cystic SCC is more obviously necrotic than inflammatory, and a careful search usually reveals a few squamous epithelial cells with malignant nuclear features or abnormal keratinised cells with bizarre, globoid shapes and dense orangeophilic (Pap) cytoplasm. The nuclear atypia and hyperchromasia seen in squamous cells from a benign cyst is of degenerative type. But the distinction is not always easy (see Figs 4.1C and D). In some cases, the FNB can only be reported as indeterminate. The only ancillary test we have found useful in this setting is HPV DNA sequencing. Occult tonsillar carcinomas and other oral cavity carcinomas with cystic lymph node metastases are a common clinical problem. Many such carcinomas contain HPV DNA as evaluated by PCR or other molecular testing and a positive result in an FNA sample is strong evidence that a lesion is metastatic carcinoma rather than a branchial cleft cyst or other benign cyst.
Other non-neoplastic cysts
The content of a thyroglossal cyst can be cytologically indistinguishable from that of a branchial cyst. The differential diagnosis is mainly based on the anatomical site of the lesion. The content is sometimes mucinous and mucin-secreting and/or ciliated columnar epithelial cells may be found in the smears. Thyroid epithelial cells are rarely present (see p. 122).37
Mucocele of the lips, oral mucosa, tongue,38 and occasionally of the paranasal sinuses may be referred to FNB to exclude neoplasia or infection. The aspirated mucinous material contains mainly mucinophages and some inflammatory cells. Mucinophages sometimes appear atypical, especially in MGG-stained smears, and a suspicion of well-differentiated mucinous adenocarcinoma can arise (see Fig. 4.23).
Amyloid tumor (Fig. 4.4)
Solitary deposits of amyloid, so-called amyloid tumors, are occasionally found submucosally in the hypopharynx, the larynx and other parts of the upper respiratory tract. Amyloid stains an intense magenta color with MGG, less specific yellowish-green with Papanicolaou. It has a fairly dense amorphous texture with a finely fibrillar rather than hyaline structure discernible in high power. In FNB smears, the amyloid may be associated with histiocytic giant cells, lymphocytes or epithelial and/or mesenchymal cells from surrounding tissues (see also Chapter 14).
Problems and differential diagnosis
The appearances of amyloid in cytological smears are not always characteristic enough to be diagnostic. It can be confused with dense thyroid colloid or hyalinised fibrous stroma.39 Its nature should therefore be confirmed by staining with Congo red and polarisation. In the head and neck, the possibility of origin from medullary thyroid carcinoma, primary or metastatic, must always be considered, and immunostaining for calcitonin performed.
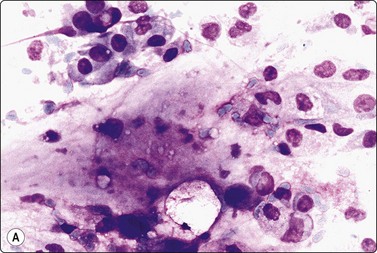
Inflammatory conditions
The cytology of lymphadenitis is described in Chapter 5. Special attention has been given to the diagnosis of sarcoid and of tuberculous lymphadenitis in the head and neck region.40 We have seen examples of actinomycosis of the parotid region and of the pharynx, clinically suspected of neoplasia due to the induration of the tissues. Sulphur granules were not seen macroscopically but microscopically a few clumps of finely filamentous microorganisms surrounded by polymorphs suggested the correct diagnosis, subsequently confirmed by culture of the aspirate (Fig. 4.5). (See also Chapter 18.)
Neoplasms
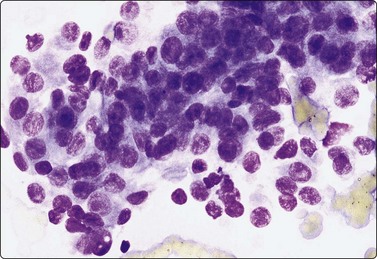
Squamous cell carcinoma (Figs 4.2 and 4.6)
Squamous cell carcinoma (SCC) is by far the commonest type of carcinoma encountered in the head and neck. Diagnostic criteria are listed in Chapter 8. Lymph node metastases of well-differentiated squamous carcinoma, particularly those arising in the Waldeyer’s ring, have a tendency to undergo liquefactive degeneration (see Fig. 4.3).41 The existence of primary SCC arising in a pre-existing branchial cyst has been doubted and is, in any case, an extremely rare event.42 The distinction from non-neoplastic cysts, mainly branchial cysts has been discussed above. Non-keratinizing squamous cell carcinoma may be represented in smears mainly by small basaloid cells in which case the differential diagnosis includes basal cell carcinoma, pilomatrixoma, poorly differentiated adenoid cystic carcinoma and other small cell tumors. Cells from a poorly differentiated squamous cell carcinoma have large vesicular nuclei and macronucleoli and resemble other anaplastic tumors such as melanoma and large cell lymphoma (Fig. 4.6).
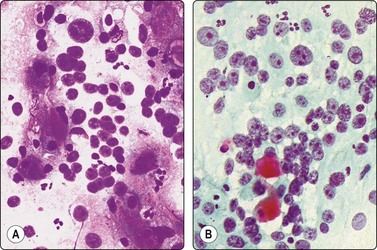
Fig. 4.6 Squamous cell carcinoma
Mainly poorly differentiated malignant cells with large vesicular nuclei and large nucleoli; a few squamous and keratinized cells. FNB smears of cervical lymph node metastasis from squamous carcinoma of larynx (A, MGG; B, Pap, HP).
Basaloid squamous carcinoma (Fig. 4.7) is a rare distinct variant of squamous cell carcinoma of the head and neck, which is clinically aggressive and has a predilection for the hypopharynx and the tongue. The smear findings are of squamous cell carcinoma without specific features, but a predominance of basal cells may make the distinction from the solid variant of adenoid cystic carcinoma difficult.43
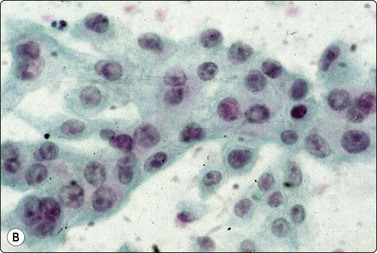
Nasopharyngeal carcinoma (NPC) (Figs 4.8, 4.9, and 5.59)44-47
Fig. 4.8 Nasopharyngeal carcinoma (squamous cell carcinoma, WHO type II)
Epithelial fragment of spindly and basaloid squamous epithelial cells with no evidence of keratinization (Pap, HP).
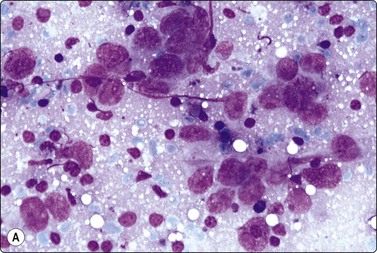
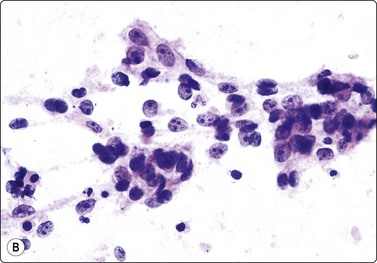
Fig. 4.9 Nasopharyngeal carcinoma (undifferentiated, lymphoepithelial type, WHO type III)
Loose clusters of undifferentiated epithelial cells with vesicular nuclei, prominent nucleoli and pale fragile cytoplasm. Background of lymphocytes. (A, MGG, HP; B, H&E, HP).
Criteria for diagnosis (undifferentiated carcinoma nasopharyngeal type (UCNT)/WHO type III/lymphoepithelial carcinoma)
Nasopharyngeal carcinoma (NPC) is a clinicopathologic entity different from other squamous cell carcinomata of the head and neck. It is distinguished by its particular histology, geographic distribution, relationship to Epstein-Barr virus, and the absence of an alcohol or tobacco etiological relationship. A proportion of NPCs show squamous differentiation and the cytological pattern of non-keratinizing squamous cell carcinoma (squamous cell carcinoma/WHO type II) (Fig. 4.8). Keratinized cells (WHO type I) are uncommonly found. The majority of NPC are poorly differentiated or undifferentiated. Cells from undifferentiated NPC (UCNT, WHO type III) form loose clusters with no specific microarchitectural pattern, and are usually mixed with lymphoid cells. In the ‘lymphoepitheliomatous’ type (Schmincke-Regaud) the cells tend to be less cohesive, resembling Hodgkin’s disease or large cell non-Hodgkin lymphoma. However, in NPC, the malignant cells are still clustered and have more abundant pale cytoplasm contrasting with the lymphoid cells in the background (Fig. 4.9). Plasma cells are frequently found among the lymphoid cells. Immunostaining for cytokeratin and a pan-lymphocyte marker is helpful. Epstein-Barr virus-associated nuclear antigen is demonstrable by anticomplement immunofluorescence in undifferentiated tumors. Other patterns of growth may occur and may cause diagnostic problems; for example, spindle cell forms may be difficult to recognize as carcinoma.
NPC frequently presents to the cytologist as a lymph node metastasis in the neck without a known primary. Cytological recognition is important since the primary is often clinically occult.
Carcinoma of sinonasal tract
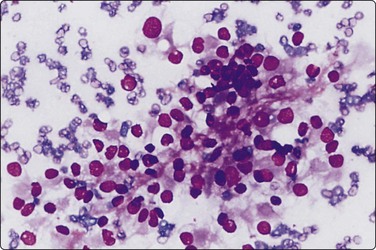
Primary or secondary sinonasal tract malignancies are rare and demonstrate a wide range of cytologic patterns. An accurate and definitive diagnosis can be made in many tumors, especially in carcinomas similar to transitional and squamous cell carcinoma, carcinoma with specific differentiation, sarcoma or melanoma.48 Poorly differentiated nasal sinus carcinomas of transitional cell type yield clusters of tightly packed cells and single cells with obvious malignant nuclear features and scanty cytoplasm.49 Smears of the intestinal type of adenocarcinoma of paranasal sinuses show aggregates of well-differentiated adenocarcinoma cells, including columnar cells and goblet cells, with a background of abundant mucus. In a case of mucinous adenocarcinoma of maxillary sinus, FNB smears from a regional lymph node metastasis showed dispersed cells with abundant cytoplasm distended by mucus and small relatively bland nuclei. A background of abundant mucus contributes to a close resemblance to a mucocele (Fig. 4.10).
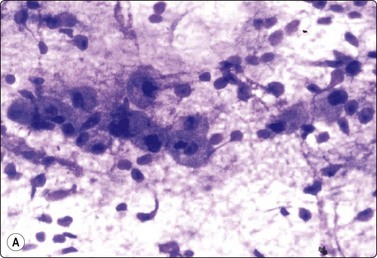
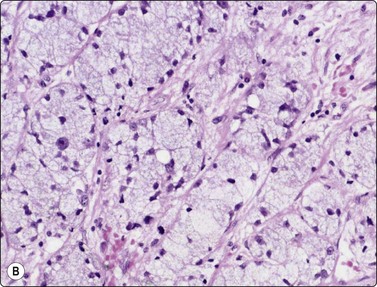
Fig. 4.10 Mucinous adenocarcinoma of maxillary sinus
(A) Poorly cohesive round cells with abundant vacuolated cytoplasm and relatively bland nuclei resembling mucinophages in a background of mucus; FNB of cervical lymph node metastasis (Diff-Quik, HP); (B) Corresponding tissue section of primary tumor (H&E; IP).
Paraganglioma (carotid body and glomus jugulare tumors) (Figs 4.11-4.13)5,50-52
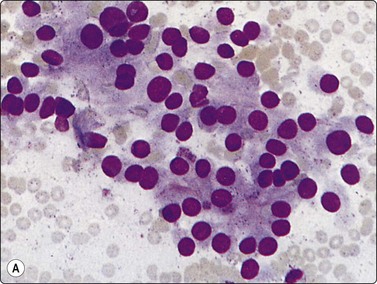
Loosely clustered cells; suggestion of follicular arrangement resembling thyroid epithelium; anisokaryosis and ‘speckled’ chromatin (Pap) typical of neuroendocrine tumors. Very fine eosinophilic cytoplasmic granules visible under the microscope but not in photograph; (A, MGG, HP; B, Pap, HP).
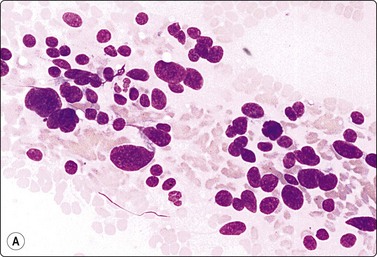
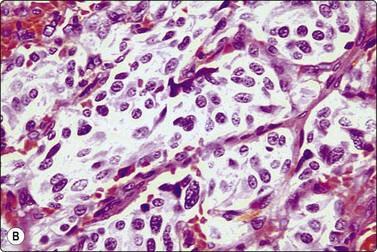
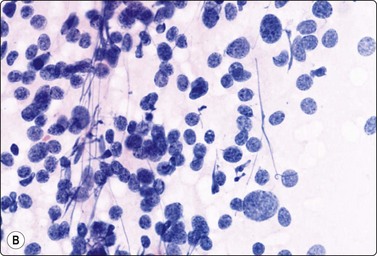
Fig. 4.13 Paraganglioma (atypical)
(A) Smear showing prominent anisokaryosis but a uniformly bland chromatin pattern (MGG, HP); (B) Tissue section of the same case (H&E, IP).
Criteria for diagnosis
Problems and differential diagnosis
The cytological pattern is suggestive of an endocrine neoplasm and, given the anatomical site, the main differential diagnosis is a thyroid tumor. A follicular arrangement of the tumor cells may suggest a follicular carcinoma, but the fine red cytoplasmic granulation, the characteristic anisokaryosis and the presence of spindle cells closely resemble medullary carcinoma, and this is the main differential diagnosis (Fig. 4.12). Immune markers are helpful. Cells of paraganglioma stain positively for neuroendocrine markers. Staining for calcitonin is negative in most cases, but can occasionally be positive. Cytokeratin, thyroglobulin and TTF1 are negative. Intranuclear cytoplasmic inclusions as in papillary and some other carcinomas of the thyroid can be found in some paragangliomas.50 Knowledge of the exact anatomical site is obviously important. However, paraganglioma can occur in atypical locations including, although rarely, the thyroid. For example, one of our cases diagnosed by FNB had a tumor in the tonsillar region, clinically thought to be a deep parotid tumor (Fig. 4.11); another had a supraclavicular mass diagnosed clinically as lymphadenopathy. Both were histologically confirmed as paragangliomas.
Paraganglioma with a spindle cell pattern can mimic other spindle cell tumors in the neck such as spindle cell medullary carcinoma of thyroid and soft tissue tumors. Nuclear pleomorphism can sometimes be prominent enough to suggest malignancy (Fig. 4.13).5 As in other endocrine tumors, pleomorphism is not a reliable indicator of malignancy, mitotic rate and evidence of necrosis are better related to clinical behavior, and metastasis is the only definitive proof.
Paragangliomas are extremely vascular lesions and the aspirate often appears to be pure blood. If this is the case, smears may be non-diagnostic but diagnostic tissue fragments can sometimes be found in a cell block.
Malignant lymphoma
Malignant lymphoma can involve several sites in the head and neck including tonsils, salivary glands, orbit, scalp and cervical lymph nodes. Cytological criteria are given in Chapter 5. The distinction from reactive lymphoproliferative lesions can be difficult. A mixed population of lymphoid cells dominated by lymphocytes and including germinal center material favors a reactive process. Immune marker studies by flow cytometry to demonstrate monoclonality versus polyclonality is generally indispensable in this situation.
Meningioma (Fig. 4.14)53-55
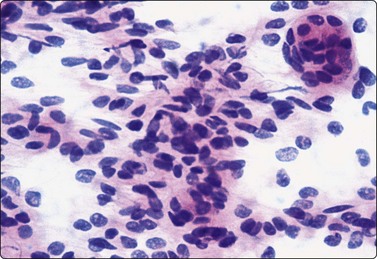
Loose cluster and a tight whorl of ovoid or spindle cells with a bland, finely granular nuclear chromatin (H&E, HP).
Extracranial meningiomas occur in relation to the base of the skull, the scalp, the orbit, the nasal cavity, the paranasal sinuses and the middle ear. In one of our cases, a meningioma extended from the base of the skull to present as a maxillary gingival lump which was correctly diagnosed by FNB. Although uncommon, meningioma should be remembered in the differential diagnosis of any tumor in the head and neck. The exact anatomical location shown by CT, the presence of characteristic whorls and the bland nuclear morphology are pointers to the correct diagnosis.
Olfactory neuroblastoma (Fig. 4.15)49,56,57
This rare tumor occurs in the upper nasal cavity and may cause nasal obstruction. It may also present as lymph node secondaries in the neck. Several cases diagnosed by FNB have been reported. The pseudorosettes formed by the tumor cells may be mistaken for microacini of an adenocarcinoma, but the nuclear morphology is relatively bland, and finely fibrillar material may be seen in the center of the rosettes, similar to the common neuroblastoma. Immunostaining for neuroendocrine and epithelial markers is helpful in the differential diagnosis.
Tumors of the orbit
The orbital region contains a variety of anatomical structures including eyelid, conjunctiva, caruncle, uvea, retina, lacrimal gland, lacrimal drainage system, skin, skin adnexa and soft tissues, and is surrounded by bone and cartilage. This complexity of adjacent, highly specialized tissues may give rise to a variety of inflammatory, benign and malignant conditions. Furthermore, the rich vascularity of the orbital region is responsible for the common occurrence of metastases from various organs, mainly from the breast and the lung.
Palpable orbital tumors can be successfully diagnosed using the FNB technique. A comparative cytological/histological study of 286 aspirates of palpable orbital and eyelid tumors showed that a concordant diagnosis of malignancy and of tumor type was achieved in 87% of cases. A false-positive diagnosis was made in 1.6% and a false-negative diagnosis in 1.8% of cases.58 Image-guided FNB is recommended for non-palpable lesions.59
Lymphoproliferative processes constitute the main problem in the orbit. A cytological diagnosis can be difficult, but FNB is useful and is a valuable addition to ultrasound, CT and MRI. The technique is reliable and safe. The combination with ancillary techniques, mainly immunocytochemistry, flow cytometry and genomic techniques, provides a reliable basis for accurate typing,60-64 and this tool may help to avoid a traumatic surgical intervention.
Extramedullary erythropoiesis/myeloid metaplasia can also give rise to an orbital mass, for example in patients with myelofibrosis. Erythroblasts, megakaryocytes and granulocytic precursors are found in the aspirate, usually easily recognized in MGG-stained smears (see p. 286). As mentioned above, metastatic malignancy is also relatively common in this site. Orbital tumors such as retinoblastoma, melanoma and metastases usually exhibit specific cytologic morphology allowing an accurate diagnosis.
Tumors of the lacrimal gland are mainly similar to primary salivary gland tumors and the same diagnostic criteria (see below) apply.65 The question whether the tumor is primary or metastatic in this site should be investigated.66 The commonest lacrimal gland tumors are pleomorphic adenoma, carcinoma ex pleomorphic adenoma and salivary duct carcinoma. An example of FNB of a well-differentiated mucinous adenocarcinoma primary in the lacrimal gland is illustrated in Figure 4.16. Malignant lymphoma of lacrimal gland has been reported.
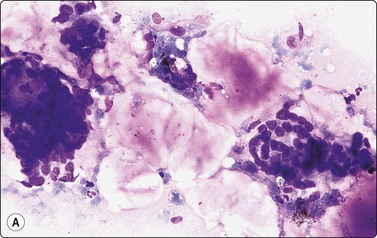
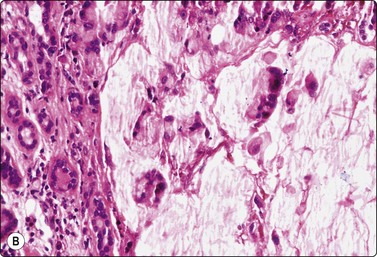
Fig. 4.16 Mucinous adenocarcinoma, lacrimal gland
Clusters of small, relatively bland glandular epithelial cells, some columnar or goblet cells, background of abundant mucin (A, smear, MGG, IP; B, tissue section, H&E, IP).
A variety of inflammatory conditions of the orbit can also be diagnosed by FNB. These may present as masses, cysts, abscesses, discharging sinuses and dermal plaques and nodules. Granulomatous inflammations are most common, such as chalazion, tuberculosis, Cysticercus cellulosae and ruptured epidermal cysts.67
Intraocular tumors
Fine needle biopsy of intraocular tumors, performed in theater, has not gained wide acceptance among ophthalmologists because of the (negligible) risk of tumor cell dissemination, intraocular complications and unfamiliarity with the technique. It offers a means of distinguishing – in the exceptional case when this is a problem for clinical diagnosis and management – between a primary and a metastatic intraocular malignancy. In our experience, the only complication has been vitreous hematoma. Indications for intraocular FNB are: (1) the patient refuses enucleation, (2) a definitive diagnosis cannot be made by conventional and ancillary ophthalmologic techniques, (3) metastatic tumor is suspected in the absence of a known primary site, and (4) genomic analysis of intraocular melanoma.68 Malignant melanoma, retinoblastoma, medulloepithelioma and metastatic tumors are the commonest intraocular tumors.
FNB smears of intraocular melanoma are similar to those of melanomas from other sites. Uveal melanomas are usually rich in pigment. The cells are often relatively bland, monomorphous spindle cells. Immunostaining for S-100 and HMB-45 provides a means of confirmation if melanin is not visible.69 For a detailed description of the cytology of melanoma, see Chapter 14.
Retinoblastoma has a characteristic age distribution, family history, radiologic findings and fundus semiology. Cells of retinoblastoma may be found in fluid aspirated from the anterior chamber of the eye. The cytology is similar to other malignant small round cell tumors (Fig. 4.17).70,71 The cells are small with hyperchromatic nuclei and scant basophilic cytoplasm. Rosette-like structures, nuclear molding and necrosis are frequently seen.
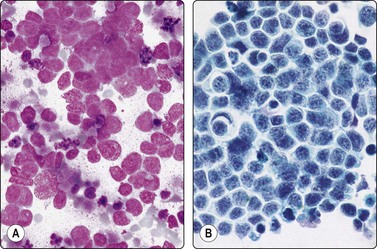
Smear pattern of a malignant small round cell tumor, some clustering but no distinctive microarchitectural features (A, MGG, HP; B, Pap, HP).
Metastatic intraocular tumors can derive from any organ but are most commonly from the lungs and breast.
Malignant lymphoma may rarely involve the uveal tract and can be diagnosed by cytological examination of fluid aspirated from the vitreous.72
Intracranial tumors
We have little experience of FNB of intracranial tumors and the reader is referred to the literature, for example the review by Willems.73 The cytology of intracranial tumors applied to intraoperative diagnosis was beautifully described 60 years ago by Russel.74 This application is of great clinical value in view of the technical difficulties with frozen sections.75-77 Rarely, intracranial tumors involve extracranial sites in the head and neck. Meningioma is the most common example. FNB of extracranial metastasis of glioblastoma multiforme has been reported.78 Tumors of the base of the skull such as pituitary tumors are accessible to FNB through the nose.
Tumors of soft tissues and bone
Soft tissue tumors such as spindle cell lipoma, nerve sheath tumors and malignant fibrous histiocytoma are not uncommon in the head and neck and can occur in sites where they may be clinically mistaken for lymphadenopathy, salivary gland tumor, etc. The tongue is a site of predilection for granular cell tumor.79 Distinction between this tumor elsewhere in the neck and adult rhabdomyoma may be difficult.80 A not infrequent pitfall is proliferative non-neoplastic lesions in the neck, mainly nodular and proliferative fasciitis, which can be mistaken for a malignant soft tissue tumor. Rhabdomyosarcoma, usually of the embryonal type, is among the most common malignant head and neck tumors in children. Cytological criteria and differential diagnosis for soft tissue tumors are presented in Chapter 15.
Chordoma may present as an orbital, nasal or posterior pharyngeal mass accessible to FNB through the oral cavity (Fig. 4.18). Of bone tumors affecting the skull, eosinophilic granuloma, multiple myeloma and metastatic carcinoma lend themselves to cytological diagnosis. Cytomorphological criteria for bone tumors are given in Chapter 16.
Physalipherous cells embedded in chondromyxoid stroma. The characteristic vacuolated cytoplasm and the chondromyxoid stroma are much less obvious in the Pap-stained smear. This tumor presented clinically as a retropharyngeal mass (A, MGG, HP; B, Pap, HP).
FNB has not been extensively applied to odontogenic tumors and cysts. A few reports of FNB of lesions in the jawbones81,82 and of ameloblastoma83,84 and ameloblastic carcinoma85 have appeared in the literature. Cells of ameloblastoma are basaloid, often spindle or rounded, and occur in clusters or pseudopapillary projections. Squamoid and keratinized cells without prominent atypia are usually present (Fig. 4.19).
Metastatic tumors
Metastatic tumors are common in this region. For example, tumors of the scalp have been shown to be predominantly metastatic.86 Guidelines for the identification of the primary tumor are given elsewhere.
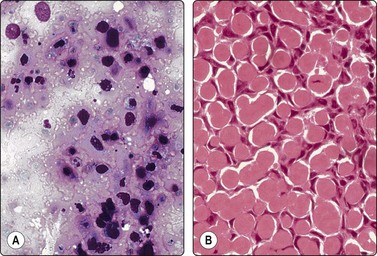
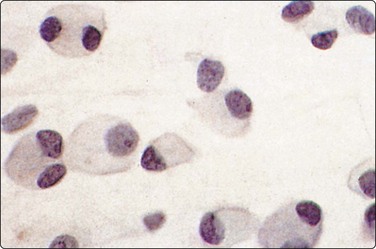
Normal structures (Fig. 4.20)
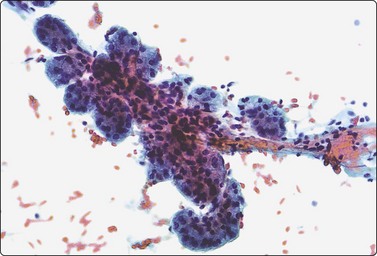
Fig. 4.20 Non-neoplastic salivary acinar and ductal cells
Normal salivary gland tissue in a FNB smear. Tissue fragment of uniform, well-formed acini along a small duct (Pap, IP).
Smears from normal or near-normal glands are usually poor in cells and heavily blood stained. Sometimes a surprisingly large number of acinar cells and tissue fragments are obtained, which can occasionally raise a suspicion of neoplasia (well-differentiated acinic cell carcinoma). The acinar cells form cohesive tissue fragments of regular spherical acini delineated by their basement membrane, often joined by small ductular structures and held together by a small amount of fibrovascular stroma, resembling a bunch of grapes. Serous acinar cells have abundant, finely vacuolated bubbly cytoplasm, an eccentric small round, dark nucleus at the base of the cell, and a small nucleolus. Scattered, stripped acinar cell nuclei may be present in the background. These must not be mistaken for lymphocytes. Ductal epithelial cells may also be found forming small cohesive flat sheets or tubules. The cells are smaller, the cytoplasm is dense, sometimes squamoid, and the nuclei are round or oval. Other common findings are adipose and loose fibrous tissue and strands of endothelial cells. Lymphoid cells may be present, derived from adjacent or intraglandular lymph nodes.
Non-neoplastic lesions
Sialadenosis (Fig. 4.21)87,88
Sialadenosis is a non-neoplastic, non-inflammatory enlargement of salivary glands, mainly the parotid. It presents clinically as soft, often bilateral and recurrent mumps-like swelling of the gland. It is usually associated with certain systemic disorders. FNB yields plenty of acinar epithelial cells, which appear normal or slightly increased in size. The microarchitectural pattern of regular acini joined by small ducts and fibrovascular stroma is the same as of normal salivary gland tissue. Smears are therefore unlikely to be mistaken for low-grade acinic cell carcinoma in spite of the cellular yield. There are no inflammatory cells.
Cysts (Figs 4.22-4.24)
Non-neoplastic cysts are relatively uncommon in the major salivary glands. There are several types: retention cysts, which may be associated with sialolithiasis; salivary duct cysts; and lymphoepithelial cysts. Aspirated fluid is poor in cells but there may be a variable number of histiocytes and inflammatory cells, and a few degenerate epithelial cells.89 Sometimes, the fluid contains numerous crystalloids (non-tyrosine, Fig. 4.22A).90-92 These have been linked with an oncocytic epithelial lining and can be found also in neoplastic cysts but their presence favors a benign lesion. Lymphoepithelial cysts are most commonly seen in HIV-infected patients.93,94 Cytological findings are similar to other non-neoplastic cysts, but lymphoid cells from subepithelial lymphoid tissue are often present. Fluid from retention cysts tends to be mucinous with a more prominent inflammatory component. Metaplastic squamous epithelial cells derived from duct epithelium in a mucous background may cause a suspicion of cystic, low-grade mucoepidermoid tumor. If the background is non-specific acellular debris, atypical metaplastic squamous epithelial cells could raise a suspicion of cystically degenerate squamous cell carcinoma (Fig 4.22B).
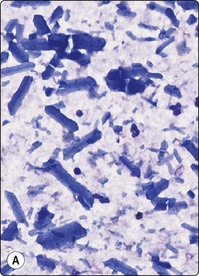
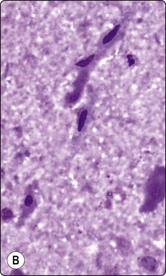
Fig. 4.22 Non-neoplastic salivary gland cysts
(A) Cyst fluid containing numerous non-tyrosine crystalloids and a few inflammatory cells (MGG, IP); (B) Fluid from a lymphoepithelial cyst with a few degenerate spindle squamous metaplastic cells, resembling cystically degenerate squamous cell carcinoma (MGG, HP).
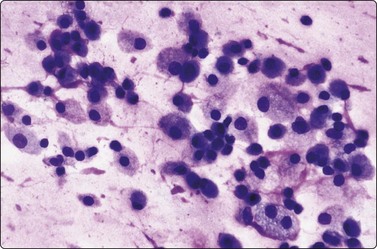
Fig. 4.23 Non-neoplastic salivary gland cyst
Numerous mucinophages, some clustered, in aspirate from mucocele of lip; compare Fig. 4.10 (MGG, HP).
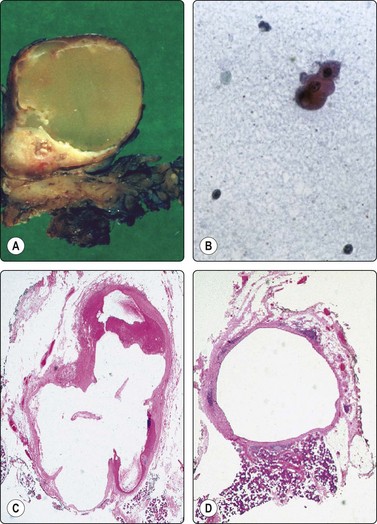
Fig. 4.24 Cystic salivary gland neoplasms
(A) Cystic Warthin’s tumor (macro); (B) Cyst fluid from same lesion with a single small group of bland oncocytic epithelial cells (Pap, IP); (C) Predominantly cystic pleomorphic adenoma (tissue section, H&E); (D) Cystic mucoepidermoid carcinoma with small tumor nodules in the cyst wall (tissue section, H&E). The fluids aspirated from (C) and (D) were practically acellular.
Mucocele is a pseudocyst of extravasated mucinous secretion, which commonly develops in minor salivary glands, particularly on the lips but also in other sites of the oral cavity and tongue. FNB yields mucus with a variable number of mucinophages and inflammatory cells. Well-preserved epithelial cells are not seen, but mucinophages may cluster and may appear atypical, particularly in MGG smears, resembling mucinous adenocarcinoma (Fig. 4.23).
Cystic neoplasm is the most important differential diagnosis of non-neoplastic salivary gland cyst. The majority of cysts occurring in the major salivary glands are, in fact, associated with neoplasms, which may be benign or malignant. Warthin’s tumor and low-grade mucoepidermoid carcinoma are the commonest, but pleomorphic adenoma, cystadenoma, acinic cell carcinoma and other tumors may also be predominantly or partly cystic (Fig. 4.24). Aspirated fluid from a cystic neoplasm is often poor in cells and indistinguishable from fluid from a non-neoplastic cyst. If the lesion disappears completely after evacuation of the fluid, it is most likely of non-neoplastic nature. However, any remaining solid part must be biopsied. US guidance is helpful in this situation, particularly because the cyst may refill with blood following the initial FNB, rendering the solid portion impalpable. Clinical follow-up of cystic salivary gland lesions is essential if a specific diagnosis cannot be made.
Sialadenitis (Figs 4.25, 4.26)
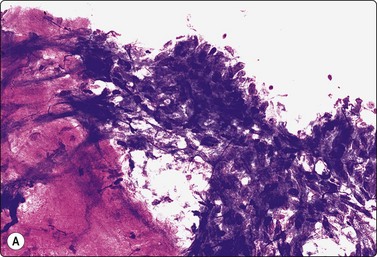
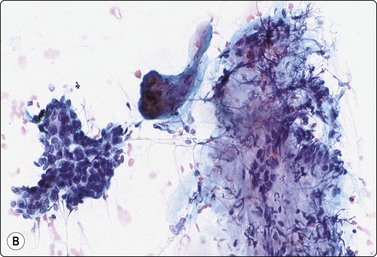
Fig. 4.25 Chronic sialadenitis
Fragments of epithelium mainly of ductal origin showing mild reactive atypia and some squamous metaplasia; fragments of fibrous stroma; relatively few chronic inflammatory cells. (A, MGG, IP; (B, Pap, IP).
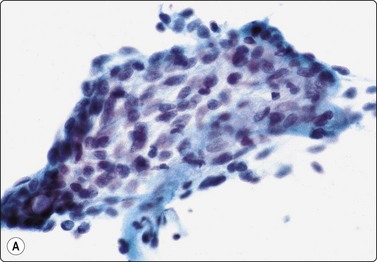
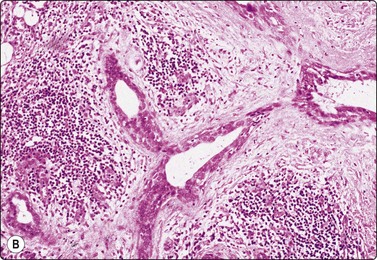
Fig. 4.26 Chronic sialadenitis
(A) Sheet of ductal epithelium showing squamous metaplasia. This could be mistaken for low-grade mucoepidermoid carcinoma or other low-grade neoplasm (Pap, HP); (B) Corresponding histology, most acinar epithelium replaced by fibrous tissue with patchy inflammatory cell infiltration, and prominent ducts showing mild reactive atypia and squamous metaplasia (H&E, IP).
Purulent material aspirated from a tender, swollen gland suggests infective sialadenitis. Smears contain a mixed population of numerous neutrophils, foamy degenerate cells and endothelial cells. The swelling should subside after antiinflammatory treatment.
Most cases of chronic sialadenitis referred for FNB are in a late stage when interstitial fibrosis and atrophy of acinar tissue have taken place. The inflammatory cell infiltration may have subsided and may be sparse and patchy. FNB smears are therefore often scanty, mainly of ductal epithelial cells associated with only few acinar cells and inconspicuous inflammatory cells. Fragments of fibrous stroma are often present (Fig. 4.25). Crystalloids may be present in the aspirate.95 Regenerating ductal epithelium in chronic sialadenitis may undergo squamous metaplasia and may appear atypical (Fig. 4.26). Mucus-like material from dilated ducts may be present. This may be suggestive of a neoplastic lesion or even of malignancy, mainly low-grade mucoepidermoid tumor. Multiple sampling and clinical correlation usually solves the problem.
FNB diagnosis of sialadenitis and other salilvary gland lesions in patients infected with HIV has been reported by several authors.93,94,96,97
Granulomatous inflammation in FNB samples from major salivary glands most likely represents granulomatous lymphadenitis related to intraparenchymal lymph nodes. The usual range of differential diagnoses must be considered (see Chapter 5). Granulomatous clusters of epithelioid cells and multinucleated giant cells without evidence of necrosis, associated with normal salivary gland components, suggest sarcoidosis involving the salivary gland directly.
Stripped nuclei of dispersed non-neoplastic acinar epithelial cells in smears of normal salivary gland tissue are of similar size and shape as lymphocytes. This must not be misinterpreted as chronic sialadenitis.
Epithelial atypia and squamous metaplasia are particularly prominent in necrotising sialometaplasia. This is a self-healing inflammatory condition of unknown etiology, possibly related to previous surgery, radiotherapy or infarction, which mainly affects minor salivary glands.98 Cellular smears of squamous metaplastic cells showing regenerative atypia and degenerative changes with necrotic material in the background can closely mimic well-differentiated squamous cell carcinoma (Fig. 4.27). Worrysome squamous epithelial cell atypia can be found in a wide spectrum of salivary gland lesions, causing diagnostic difficulties in FNB.99
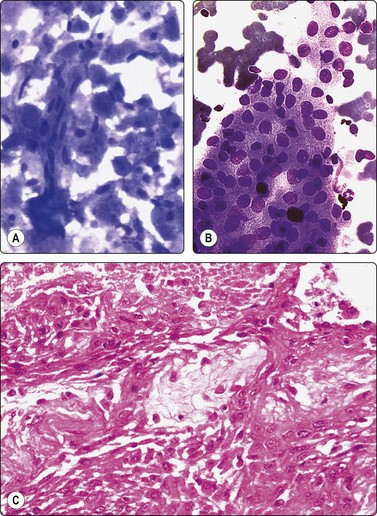
Fig. 4.27 Necrotising sialometaplasia
(A) Seminecrotic material including degenerate squamous epithelial cells with pyknotic spindle nuclei resembling cells of cystic squamous carcinoma; (B) Metaplastic squamous epithelial cells showing mild reactive atypia (MGG, HP); (C) Corresponding tissue section (H&E, IP).
Chronic sialadenitis can involve a gland focally and produce a firm nodule, which may be clinically mistaken for neoplasm. Such ‘pseudotumors’ are most common in the submandibular gland (Küttner’s tumor)100,101 and may be either the result of obstruction or a manifestation of the immunopathic so-called ‘IgG4 disease’ in cases where calculus can be reasonably excluded. Histologically, the nodule shows similar features as described in chronic sialadenitis: acinar cell atrophy, ductal and ductular hyperplasia often with squamous metaplasia and a fibrous or myxoid stroma. Chronic inflammatory cell infiltration is of variable degree and may be mild. The aggregates of ductal epithelial cells associated with myxoid stromal fragments can be mistaken for pleomorphic adenoma, low-grade mucoepidermoid carcinoma or other neoplasms in FNB smears.
Inflammatory pseudotumor caused by a proliferation of myofibrohistiocytic cells can occur in several sites such as lung, liver and soft tissues and have also been described in major salivary glands.102 Another non-neoplastic lesion that can clinically mimic a neoplasm is adenomatoid hyperplasia of the small glands of the palate.103 This lesion is of simple hyperplastic nature, forming a focal increase in the amount of acinar tissue.
Benign lymphoepithelial lesion/myoepithelial sialadenitis
Benign lymphoepithelial lesions are swellings of salivary glands caused by a reactive lymphoid infiltrate with follicular hyperplasia, which obliterates the acinar glandular tissue and causes proliferation and disruption of ductal epithelium. It may clinically manifest as Sjögren’s syndrome. Smears from a benign lymphoepithelial lesion are characterized by small clusters of ductal epithelial cells associated with lymphocytes and with a background of lymphoid cells (Fig. 4.28). The smear pattern is reminiscent of autoimmune thyroiditis. The condition is associated with HIV infection.97
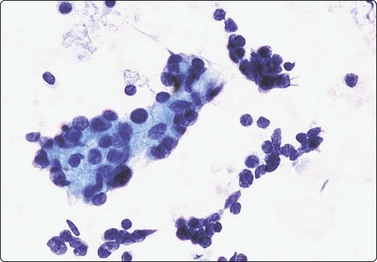
Fig. 4.28 Benign lymphoepithelial lesion
Aggregate of ductal epithelial cells associated with many lymphoid cells (Pap, HP).
The most important differential diagnosis is lymphoma, mainly MALT lymphoma. This often requires immunological studies, most conveniently by flow cytometry of aspirated material. Branchial cyst in which only the lymphoid component has been sampled should also be considered.
Benign neoplasms
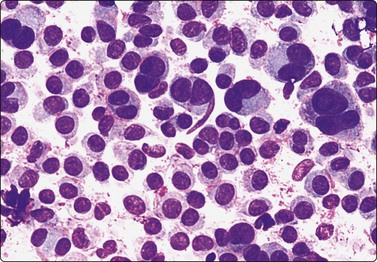
Pleomorphic adenoma (PA) (Figs 4.29-4.35)14,104,105
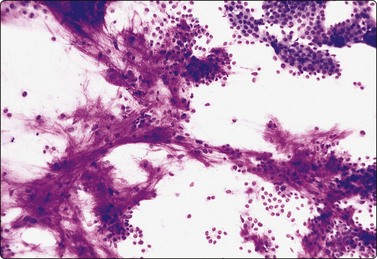
Typical low-power pattern of poorly cohesive epithelial-like cells associated with fibrillar fibromyxoid stroma staining brightly red/magenta (MGG, LP).
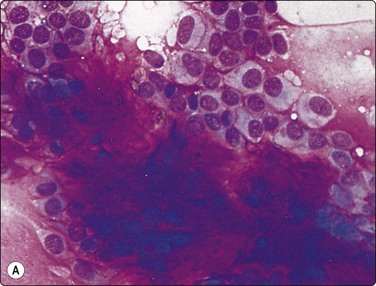
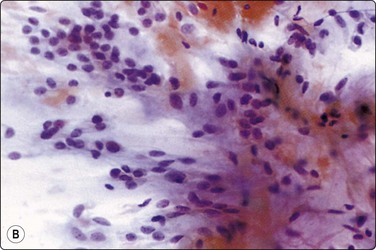
High-power view showing myoepithelial cells with abundant pale cytoplasm and bland nuclei; fibrillar fibromyxoid stroma including single oval and spindle cells (A, MGG, HP; B, Pap, IP).
Epithelial cell predominance, cellular smear; prominent anisokaryosis should not suggest malignancy (MGG, HP).
An example of multiple hyaline stromal globules in pleomorphic adenoma. Note bland appearance of epithelial cell nuclei (MGG, IP).
Single large atypical cells with bizarre nuclei and a background of usual elements of pleomorphic adenoma; no histological evidence of malignancy (A, MGG HP; B, H&E, HP; C, tissue section, H&E, IP).
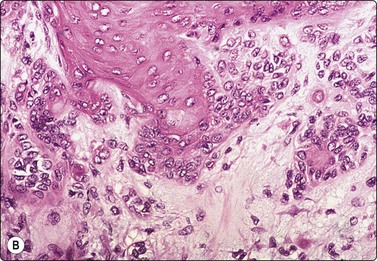
Prominent component of squamous epithelial cells (left and center); some myoepithelial cells and myxoid stroma at upper right reveal the nature of the lesion (A, Pap, HP; B, tissue section, H&E, IP).
Criteria for diagnosis
The chondromyxoid matrix is particularly characteristic in MGG smears, distinctly fibrillar and staining intensely red to purple (Figs. 4.29 and 4.30A). Staining may be so intense that it obscures the cellular component in tissue fragments. Cell detail is therefore better seen in alcohol-fixed Pap smears. With Pap staining, the ground substance is gray–green to orange in color and appears relatively amorphous or finely fibrillar (Fig. 4.30B). Spindle or rounded cells are present within the stromal fragments. The cellular component consists of relatively uniform oval, plasmacytoid or spindle cells. Nuclei are round or oval, eccentric, and have a bland, finely granular chromatin and inconspicuous nucleoli. Moderate anisokaryosis is a common feature. The cytoplasm is pale but well defined with distinct cell borders. Stripped, naked nuclei are not a feature. Often, the cells are strikingly plasmacytoid with abundant cytoplasm and eccentric nuclei (Fig. 4.31). They are generally poorly cohesive and dispersed but also form aggregates with no specific microarchitectural pattern. Red-staining intercellular material (MGG) is present within the aggregates. Tyrosine crystals have been noted in some cases.106
The majority of cells of PA are myoepithelial but a proportion of the cells may be epithelial of basaloid type. If present, they are not clearly distinguishable from the myoepithelial cells in routine stained smears. A study using immune markers for epithelial cells showed them to be present in 9 of 20 Pas, so examined.107
The cytological diagnosis of PA is not difficult in typical cases. However, the pattern can vary considerably between different parts of the same tumor. This can cause diagnostic difficulties due to the limited and often selective sampling by the thin needle.108,109 One particular feature present only focally in tissue sections may dominate the smears to the extent that the true nature of the tumor is not recognised. For example, samples of chondromyxoid matrix with few or no cells can mimic cartilage, whereas highly cellular areas with scant stroma can be mistaken for basal cell adenoma or adenoid cystic carcinoma. Aspiration of mucoid paucicellular fluid may suggest low-grade mucoepidermoid carcinoma or mucoepidermoid carcinoma arising in pleomorphic adenoma.110 PA can be predominantly cystic (see Fig. 4.24C).111 Multiple sampling is important to overcome the problems due to selective sampling.
The distinction of PA from well-differentiated adenoid cystic carcinoma is clinically important. Both tumors have relatively uniform epithelial-like cells and both may have a fibrillar myxoid stromal component. Hyaline stromal globules resembling those characteristic of adenoid cystic carcinoma, or a beaded hyaline stroma, sometimes occur also in PA (Fig. 4.33).25,112 The differential diagnosis must not be based solely on the stromal component, but cytological detail must also be closley studied. A well-defined cytoplasm, no or few stripped nuclei, and a bland, finely granular nuclear chromatin favor PA; scanty cytoplasm, a high N : C ratio, naked nuclei, nuclear molding, and nuclear hyperchromasia and coarseness favor adenoid cystic carcinoma. Multiple sampling and well-prepared smears, both MGG and Pap, reduce the likelihood of error.
If a stromal component is scanty or missing and smears are highly cellular, the distinction from basal cell adenoma and myoepithelial adenoma can be difficult or impossible. However, the distinction is not of clinical significance since these tumors are managed in the same way.
Worrisome cytological atypia can occur in PA in several forms. The myoepithelial cells of a common benign PA occasionally display prominent anisokaryosis that may cause suspicion of malignancy (Fig. 4.32). However, if the nuclear chromatin is bland, if there is no mitotic activity or necrosis, and if the anisokaryosis is randomly distributed, the pattern is consistent with a benign PA. In other cases, one may find single scattered stromal cells with considerably enlarged, irregular and multilobated, even bizarre nuclei, seen both in smears and in tissue sections of histologically benign tumors (Fig. 4.34).25 Such atypical cells are probably degenerative in nature and may be equivalent to the nuclear atypia seen in ancient schwannoma.113 Aggregates of atypical epithelial cells showing nuclear enlargement, abnormal nuclear chromatin and nucleolar prominence, coexistent with bland epithelial cells and fibromyxoid stroma typical of PA, suggest carcinoma arising in pleomorphic adenoma (see Figs 4.69, 4.70).25,114 This entity is further discussed in relation to malignant tumors.
Epithelial metaplasia, mainly squamous and oncocytic, is often seen in PA. Goblet cells are sometimes present and squamous metaplasia can be a prominent feature. If a squamous component is selectively sampled by FNB and if the metaplastic cells appear atypical, the possibility of low-grade mucoepidermoid tumor may be considered (Fig. 4.35). Sebaceous metaplasia is less frequent. The presence of groups of bland epithelial cells typical of PA and a few fragments of myxoid stroma suggest the correct diagnosis.
Tumor-like nodules caused by focal chronic inflammation have been mentioned in the section on chronic sialadenitis. The presence of epithelial cell aggregates associated with fibrillar fibrous stroma could be mistaken for PA, but the fragments of ductal epithelium – with or without squamous metaplasia – are cohesive and the stroma is not chondromyxoid. FNB samples of intraparotid schwannoma can also include tissue fragments resembling the fibromyxoid stroma of PA, but the cellular component is clearly different from the myoepithelial cells of PA.115
In summary, the hallmark of PA is the combination of bland, mainly myoepithelial cells and fragments of chondromyxoid stroma with spindle cells. In the presence of any such stromal fragments and bland epithelium, pleomorphic adenoma should be included in the differential diagnosis even when other features dominate the smears. In difficult cases, positive immunostaining for intermediate filaments such as GFAP and negative staining of the majority of cells for cytokeratin can be helpful.116
Basal cell and canalicular adenoma (Figs 4.36-4.40)117,118
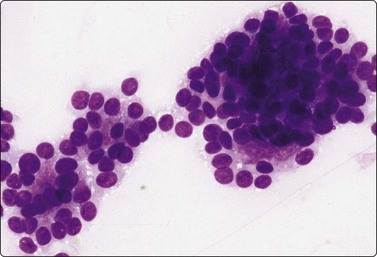
Fig. 4.36 Basal cell adenoma, solid variant
Clusters of small basaloid epithelial cells with scanty fragile cytoplasm and bland rounded nuclei; inconspicuous stroma (MGG, HP).
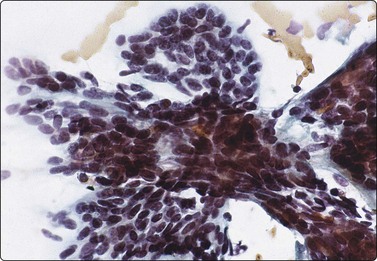
Fig. 4.37 Basal cell adenoma trabecular variant
Cohesive small epithelial cells forming a trabecular microarchitectural pattern (Pap, HP).
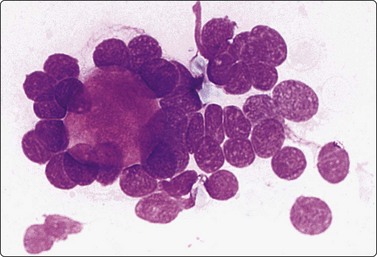
Fig. 4.38 Basal cell adenoma trabecular variant
Hyaline stromal globule surrounded by small epithelial cells with bland granular nuclear chromatin (MGG, HP oil).
(Courtesy Dr K. Lindholm, Malmö General Hospital).
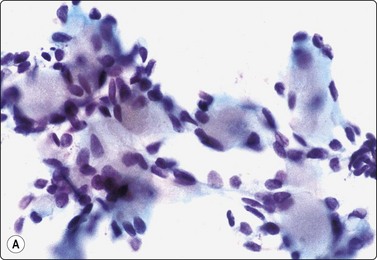
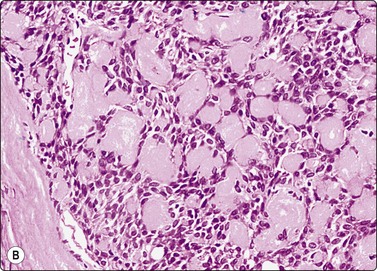
Fig. 4.39 Basal cell adenoma trabecular variant
(A) Multiple small rounded hyaline stromal globules surrounded by small bland epithelial cells (Pap, HP); (B) Corresponding tissue section (H&E, IP).
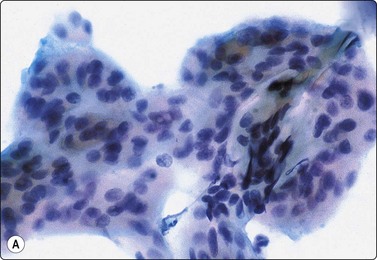
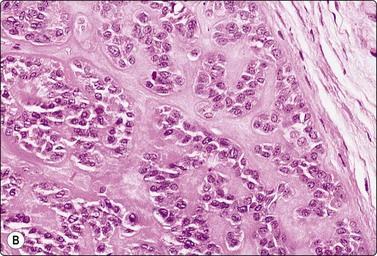
Fig. 4.40 Basal cell adenoma, membranous type
(A) Tissue fragment of small, uniform epithelial cells adherent to a background sheet of hyaline basement membrane material (Pap, HP); (B) Corresponding tissue section (H&E, IP).
Criteria for diagnosis
Basal cell adenoma (BCA) and canalicular adenoma have overlapping cytomorphologic features. Several subtypes of BCA are distinguished by the architectural patterns: solid, trabecular, tubular and membranous. These subtypes cannot always be distinguished in cytological smears. BCA is mainly seen in the major salivary glands and usually in elderly patients; canalicular adenoma mainly occur in small glands of the oral cavity.119
The cells of BCA are of basaloid epithelial type and lack the abundant cytoplasm and distinct cell borders of myoepithelial cells. Many cells present as naked nuclei. Stromal material is scanty and non-characteristic in most tumors, but basement membrane material may be prominent, delineating groups of cells or forming a background to the cells (membranous variant).
Adenoid cystic carcinoma is the most important differential diagnosis, given its malignant nature. Smears from the trabecular variant of BCA and from canalicular adenoma may contain hyaline globules resembling those seen in adenoid cystic carcinoma (Figs 4.38, 4.39).25,118 However, the globules of monomorphic adenoma are smaller, of more uniform size and have a less hyaline texture. Other tumors in which hyaline globules occur such as polymorphous low-grade adenocarcinoma, epithelial-myoepithelial carcinoma and pleomorphic adenoma may also enter the differential diagnosis. The epithelial cells of basal cell adenoma are small with scanty cytoplasm. On close scrutiny in high magnification, the nuclear chromatin is finely and evenly granular and nucleoli are inconspicuous. The cells of adenoid cystic carcinoma are similarly small with a high N : C ratio, but nuclei are less regular, hyperchromatic, with a coarsely granular chromatin, and nucleoli are more prominent. Nuclear molding is a common finding in adenoid cystic carcinoma but is not seen in BCA.
The membranous variant (dermal analogue tumor) resembles cutaneous cylindroma. Smears may show large aggregates or cohesive sheets of small basaloid cells enveloped in a rim of hyaline basement membrane material or stuck to a sheet of such material. It stains variably with both Papanicolaou and MGG (Fig. 4.40) and may or may not be metachromatic.120 The microarchitectural pattern is most striking at low magnification and is unlike that seen in either adenoid cystic carcinoma or pleomorphic adenoma. Nevertheless, hyaline basement membrane globules, as mentioned above, can cause diagnostic difficulties also in this tumor.
Distinction from cellular pleomorphic adenoma with scanty stroma is not always possible. Spindle-shaped or plasmacytoid cells with well-defined cytoplasm is against basal cell adenoma, a high N : C ratio and naked nuclei in favor. There is some overlap between these two tumors but the distinction is not of great significance clinically.
The distinction from basal cell adenocarcinoma is obviously more important and can be equally difficult. However, a review of the literature showed that FNB was accurate in all reported cases. Mitotic figures, nuclear atypia, and evidence of necrosis indicate malignancy.118
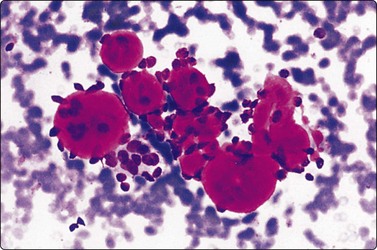
Warthin’s tumor (Figs 4.41-4.44)1,13
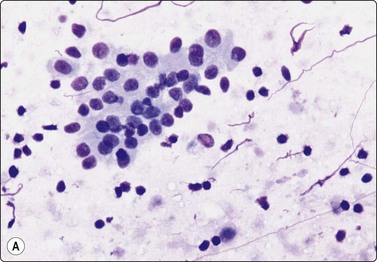
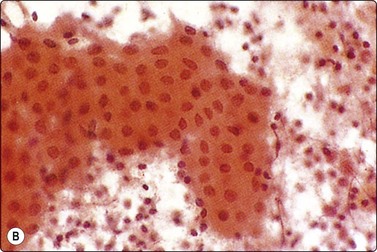
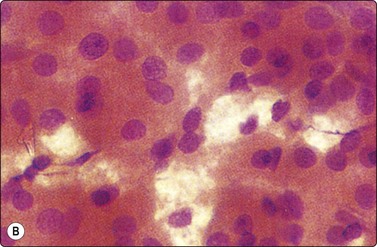
Diagnostic triad of monolayered sheets of uniform oncocytic epithelial cells with small bland nuclei, lymphocytes and proteinaceous material representing cyst fluid (A, MGG, HP; B, Pap, HP).
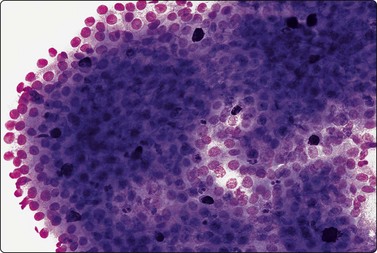
Large sheet of bland oncocytic epithelium; note scattered single mast cells staining dark blue (MGG, HP).
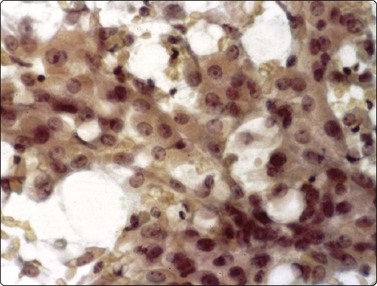
Aggregates of squamous metaplastic cells showing mild atypia and a few cells with intracytoplasmic vacuoles. This could be mistaken for low-grade mucoepidermoid tumor but there were typical features of Warthin’s tumor in other parts of the smear (Pap, HP).
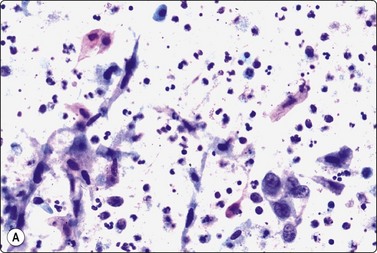
(A) Degenerating spindle metaplastic squamous epithelial cells with nuclear pyknosis and cells showing regenerative atypia mimic cystic well-differentiated squamous carcinoma (Pap, HP); (B) The corresponding tissue section shows infarction, repair and squamous metaplasia in a Warthin’s tumor (H&E, IP).
The amorphous and granular debris representing cyst contents aspirated from Warthin’s tumor (WT) has a mucoid appearance and stains blue with MGG. The oncocytes form flat, monolayered sheets with an irregular outline. They have plentiful cytoplasm and uniformly small, round, central nuclei with a bland chromatin and inconspicuous nucleoli. With Pap staining, the cytoplasm is dense, green to orangeophilic and may be finely granular; with MGG it is gray–blue and appears more homogeneous (Fig. 4.41). In MGG smears, oncocytes can be mistaken for other types of cells such as ductal epithelial cells or metaplastic squamous cells. They are more easily identified in Pap preparations. Mast cells are often seen scattered among the oncocytes (Fig. 4.42).
Obtaining diagnostic material may be difficult in a predominantly cystic WT. Both oncocytic and lymphoid cells must be identified for a definitive diagnosis, but either component may be sparse, absent or obscured by mucoid debris. The mucoid fluid from a WT with flakes of homogeneous and granular debris is characteristic but not specific. Similar hypocellular material may be obtained from low-grade mucoepidermoid carcinoma and other tumors. If the overall smear pattern is suggestive of WT but the cells appear atypical and lack a distinctly oxyphil cytoplasm, the alternative of a mucoepidermoid tumor should be considered. The problem is enhanced by the occasional finding of goblet cells in WT, and special staining for mucin may not be helpful (Fig. 4.43).
If oncocytes dominate the smears and the lymphoid and cystic component is inconspicuous, distinction from oncocytoma is difficult. The oncocytic cells of oncocytoma form multilayered aggregates rather than flat sheets as in WT.
WT may become inflamed or infarcted, spontaneously or post previous FNB. Repair results in more or less extensive squamous metaplasia. Smears contain metaplastic squamous cells showing regenerative atypia and degenerating cells, which may closely resemble malignant squamous epithelial cells of ‘fiber-cell’ type (Fig. 4.44). False-positive or suspicious diagnosis of squamous cell carcinoma with liquefaction necrosis is not uncommon.89,121,122 However, the glassy refractile nature of true keratinization is absent in degenerate metaplastic squamous cells or oncocytes. As mentioned above, mucin-secreting goblet cells can be present in the metaplastic epithelium and may suggest a low-grade mucoepidermoid tumor.
In some acinic cell tumors the cytoplasm of tumor cells may be oncocyte-like, dense and relatively homogeneous and not pale and vacuolated/bubbly or granular as in typical acinic cell carcinoma (see Fig. 4.49). Acinic cell nuclei are generally larger and more variable in size than those of oncocytes and the cells are more fragile, as shown by large numbers of bare nuclei. A microacinar architectural pattern is usually discernible in smears of acinic cell tumors. The not uncommon presence of a lymphoid stroma in acinic cell tumors can make the distinction from WT very difficult at times.
Malignant Warthin’s tumor is extremely rare and has not yet been defined cytologically.
Oncocytoma (Fig. 4.45)13,22,123
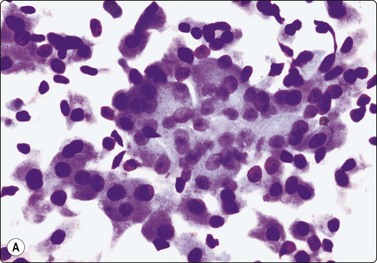
Multilayered aggregates of cohesive oxyphil cells showing some nuclear enlargement and anisokaryosis but bland nuclear chromatin (A, MGG, HP; B, Pap, HP).
Clear cell and eosinophilic variants of oncocytoma have been described.124
Cells with abundant cytoplasm from non-oncocytic tumors can resemble oncocytes in MGG-stained preparations. Acinic cell carcinoma has been mentioned above. Cells from mucoepidermoid tumors and from adenocarcinoma sometimes also have this appearance.
Oncocytomas may be cystic and their relationship to Warthin’s tumors is then uncertain. In general, cyst fluid with debris, oncocytes and lymphoid cells indicate a Warthin’s tumor, especially if the oncocytes lie in flat sheets.
Multifocal oncocytic hyperplasia of salivary gland may suggest oncocytoma in FNB smears.125
The cytologic findings in malignant oncocytic neoplasms have been described in a small number of cases.22 See also Oncocytic salivary duct carcinoma, page 70.
Other benign neoplasms
Myoepithelial adenoma22,126,127 may be of spindle cell (Fig. 4.46A), plasmacytoid (Fig. 4.46B) or epithelioid type. This tumor may not be distinguishable from a cellular pleomorphic adenoma in which a solid focus of spindle or plasmacytoid cells without specific stroma has been selectively sampled. The spindle cell type can also be confused with a benign soft tissue tumor. Distinction of the plasmacytoid type from malignant myoepithelioma can be difficult since anisokaryosis and mild nuclear atypia can occur. Mitotic figures and necrosis suggest malignancy. Positive nuclear staining for p63 supports a diagnosis of myoepithelial adenoma (Fig. 4.46C).
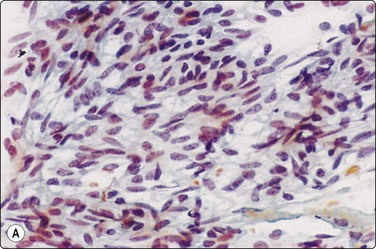
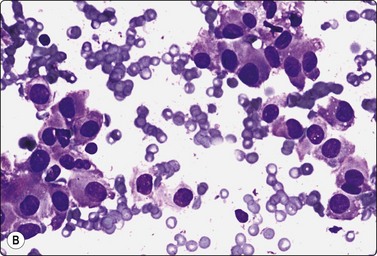
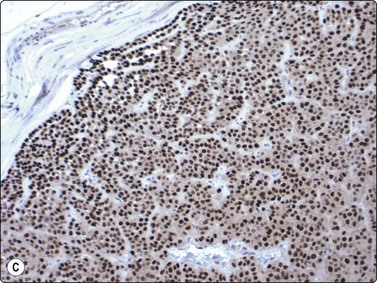
Fig. 4.46 Myoepithelial adenoma
(A) Spindle cell type; the pattern of bland spindle cells could be mistaken for a benign soft tissue tumor (Pap, HP); (B) Plasmacytoid type; poorly cohesive cells with abundant cytoplasm and eccentric nuclei. The nuclear atypica in this case caused some concern (MGG, HP); (C) Tissue section corresponding to (B), immunostaining for p63 (IP).
Sebaceous adenoma, ductal papilloma and cystadenoma are rare and few cases with cytology have been reported.22
Benign mesenchymal tumors, most commonly lipoma and schwannoma, occur in or adjacent to salivary glands, particularly the parotid. Schwannoma can be mistaken for pleomorphic adenoma or basal cell adenoma if smears are suboptimal (Fig. 4.47).128
Malignant neoplasms
Acinic cell carcinoma (Figs 4.48-4.50)129,130
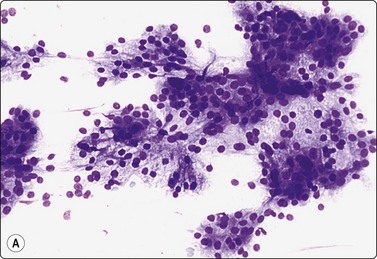
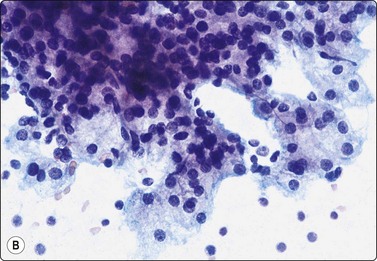
Fig. 4.48 Acinic cell carcinoma
Epithelial fragments composed of cells with abundant vacuolated cytoplasm and relatively bland nuclei, resembling normal acinar cells; many naked nuclei; scanty, thin fibrovascular stroma. Note absence of well-formed acinar structures (A, MGG, IP; B, Pap, HP).
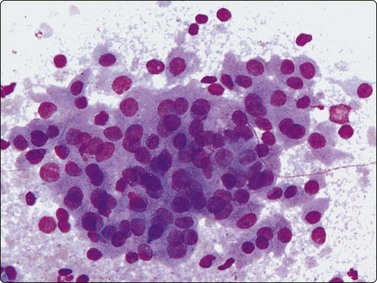
Fig. 4.49 Acinic cell carcinoma
Cells with oncocyte-like cytoplasm, distinction from oncocytoma difficult (MGG, HP).
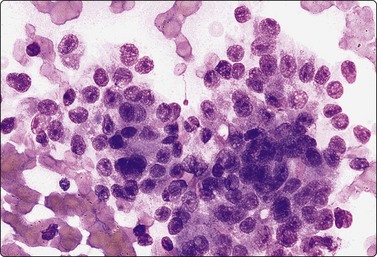
Fig. 4.50 Acinic cell carcinoma
A less well-differentiated tumor may be difficult to type as acinic cell carcinoma. Ancillary techniques such as EM may help (MGG. HP).
Acinic cell carcinoma (AcCC) is relatively common in our population. It not infrequently occurs in children and adolescents. The cells of well-differentiated AcCC resemble normal acinar epithelial cells but do not form discrete round acini defined by a basement membrane and are not associated with small ducts as in non-neoplastic salivary gland tissue (Fig. 4.48). The cells are relatively uniform, cohesive, with abundant vacuolated, foamy or bubbly cytoplasm of variable density. A clear cell appearance is sometimes seen and in some tumors the cells have a dense grayish (MGG) oncocyte-like cytoplasm (Fig. 4.49). The cytoplasm is fragile, leaving many nuclei stripped. Nuclei are round with a bland chromatin. The cells are less uniform and the nuclei are larger and less evenly distributed than those of normal acinar cells. The nuclear : cytoplasmic ratio is higher, particularly in less well-differentiated tumors. Stroma is overall scant and inconspicuous. The adherence of tumor cells to thin strands of fibrovascular stroma occasionally produces a pseudopapillary appearance. In some acinic cell tumors, the stroma contains a prominent lymphoid component.
The similarity between cells of low-grade AcCC and non-neoplastic acinar cells has been mentioned. Smears from sialadenosis can be quite cellular, but less so than samples from an acinic cell tumor. The microarchitectural patterns are distinctly different.
A clear cell pattern of large cells with abundant fragile and vacuolated cytoplasm may be seen in several other tumors, for example epithelial-myoepithelial carcinoma and low-grade mucoepidermoid carcinoma. The cytology of these tumors is described in detail below. Intracellular mucin vacuoles are not found in AcCC. Renal cell carcinoma can metastasize to the parotid gland and must be remembered in the differential diagnosis. The characteristic vascular pattern of renal cell carcinoma is a clue, and nuclear atypia is usually more prominent.
As mentioned above in relation to WT and oncocytoma, the distinction from oncocytic tumors can sometimes be difficult. This is due to the resemblance of the neoplastic cells to oncocytes in some AcCC and to the not infrequent presence of infiltrates of lymphoid cells in the stroma. Cells with vacuolated cytoplasm and many naked nuclei favor AcCC.
Less well-differentiated AcCC has a less characteristic cytological appearance which merges with adenocarcinoma of no special type (Fig. 4.50). A type-specific diagnosis may not be possible without ancillary tests such as EM.
AcCC, particularly the papillary cystic variant, can be predominantly cystic and FNB may yield only hypocellular fluid with no diagnostic cells.131,132 A false-negative diagnosis of simple cyst is possible. US guidance may solve the problem.
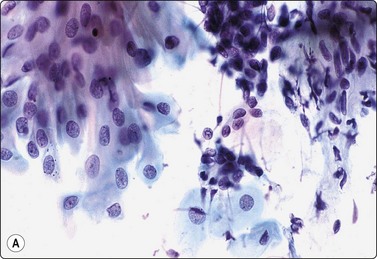
Mucoepidermoid carcinoma (Figs 4.51-4.56)17,133-135
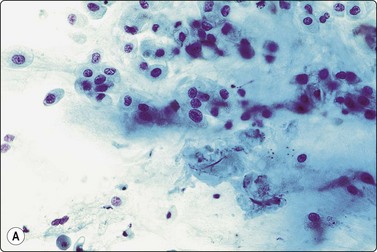
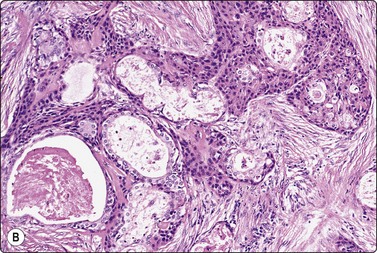
Fig. 4.51 Low-grade mucoepidermoid carcinoma
(A) Moderately cellular smear of scattered intermediate cells resembling squamous metaplasia with a ‘dirty’ background of mucus and some inflammatory cells (Pap, HP); (B) Corresponding tissue section (H&E, IP).
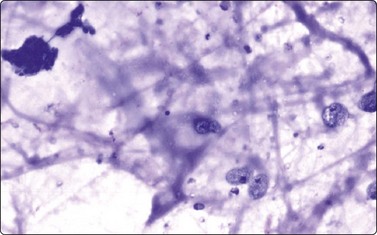
Fig. 4.52 Cystic low-grade mucoepidermoid carcinoma
Aspirated fluid typically has a ‘dirty’ appearance of mucus, debris, inflammatory cells and macrophages; a couple of small aggregates of small bland epithelial cells upper left (MGG, IP).
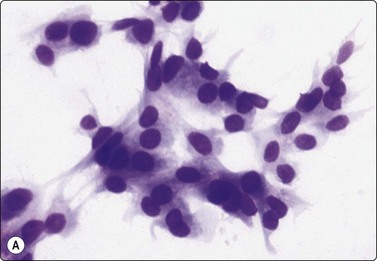
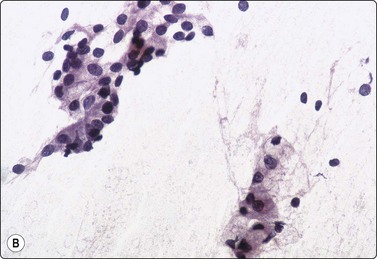
Fig. 4.53 Low-grade mucoepidermoid carcinoma
( A) Poorly cohesive non-characteristic bland epithelial cells resembling squamous metaplasia represent intermediate cells (MGG, HP); (B) In this example, the group of cells at upper left has a similar non-characteristic metaplastic appearance, the group at lower right show cytoplasmic vacuolation and a goblet cell (Pap, IP).
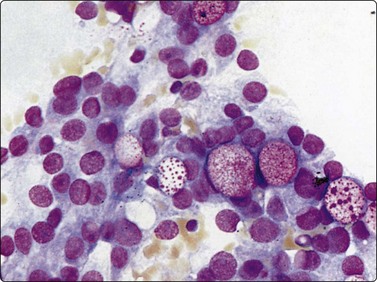
Fig. 4.54 Low-grade mucoepidermoid carcinoma
Intermediate cells with pale vacuolated cytoplasm and relatively bland nuclei; several intracytoplasmic mucin vacuoles highlighted by the Giemsa staining (MGG, HP).
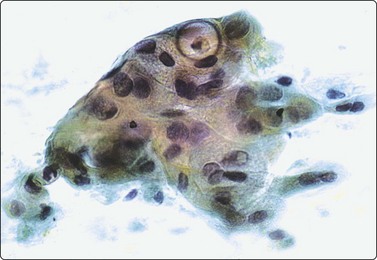
Fig. 4.55 Low-grade mucoepidermoid carcinoma
Epithelial fragment of intermediate cells, squamoid cells and a cell with an intracytoplasmic mucin vacuole, diagnostic of mucoepidermoid carcinoma (Pap, HP).
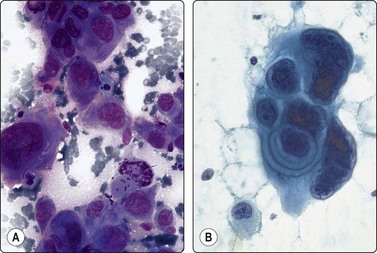
Fig. 4.56 High-grade mucoepidermoid carcinoma
Pleomorphic, clearly malignant cells, some with squamous differentiation; mitotic figures; distinction from squamous carcinoma not possible (A, MGG, HP; B, Pap, HP).
Criteria for diagnosis (low-grade tumors)
Low-grade mucoepidermoid carcinoma (MEC) is one of the commoner malignant salivary gland tumors, occurring in all age groups including children and adolescents. The clinical presentation may be innocuous. In FNB smears, the background mucus and debris stain blue–violet with MGG and may obscure the cellular component, resembling Warthin’s tumor. Cell detail is more evident in alcohol-fixed material. Scattered small clusters of intermediate cells in a mucoid background are suggestive of MEC (Figs 4.51, 4.52). The intermediate cells resemble the squamous metaplastic cells seen in cervical Pap smears. They are relatively cohesive, have a well-defined cytoplasm and mainly bland nuclei. True squamous differentiation and keratinization are uncommon in low-grade tumors. Some of the cells have abundant, finely vacuolated cytoplasm and are difficult to distinguish from macrophages. Other cells contain intracellular mucin vacuoles staining metachromatically with MGG and may have the appearance of goblet cells (Figs 4.53B-4.54). Nucleoli may be prominent, but nuclear chromatin is generally bland in low-grade tumors. Lymphoid cells are sometimes present.
A definitive diagnosis of MEC requires the coexistence in smears of cells showing squamous differentiation and of mucin-secreting cells (Fig. 4.55). Unequivocal evidence of both is not always found, particularly in cystic tumors. In such cases, only a tentative or differential diagnosis can be offered, prompting further investigation.
Smears of high-grade mucoepidermoid carcinoma contain obviously malignant squamous epithelial cells (Fig. 4.56). Mucin-secreting cells can be difficult to find, and it may be difficult or impossible to distinguish primary high-grade MEC from metastatic squamous cell carcinoma.
Contrary to the high-grade variant, low-grade MEC is difficult to diagnose as malignant cytologically and is one of the most common sources of false-negative FNB diagnoses. The main reason is that many tumors are partly or predominantly cystic (Fig. 4.24D). The aspirated material is often hypocellular and non-characteristic, consisting mainly of mucoid secretion, debris and some inflammatory cells. Cohesive epithelial cell clusters in such a background should raise a suspicion of low-grade MEC even if the cells appear bland, and should cause a diligent search for more diagnostic elements.25,136 Occasional goblet cells support the suspicion. Smears from non-neoplastic cysts such as retention cysts and lymphoepithelial cysts may also show mucus, debris, metaplastic squamous cells and glandular cells in combination, mimicking low-grade MEC. Warthin’s tumor is another benign cystic lesion that can cause differential diagnostic difficulty.136,137 Multiple sampling with US guidance is often rewarding.
The mucinous background with macrophages and inflammatory cells and the similarity of the intermediate cells of MEC to regenerating, metaplastic ductal epithelial cells in chronic sialadenitis may cause an erroneous diagnosis either way. A clinical suspicion of neoplasia (Küttner’s tumor) may add to the difficulty, but history and clinical findings are more often helpful. Similar difficulties can occur in relation to Warthin’s tumor. Cells of uniformly oncocytic type and a lymphoid cell population rather than inflammatory cells favor Warthin’s tumor.
High-grade, poorly differentiated MEC may not be distinguishable from primary or metastatic squamous carcinoma unless an obvious mucinous component is demonstrated, although obvious keratinization identified cytologically effectively excludes MEC.
Polymorphous low-grade adenocarcinoma (Figs 4.57-4.59)138-140
Usual findings
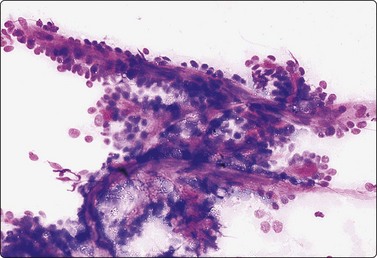
Fig. 4.57 Polymorphous low-grade adenocarcinoma
Tissue fragment with a trabecular (‘pseudopapillary’) microarchitectural pattern; small basaloid cells adhere to anastomosing strands of fibrovascular stroma (MGG, IP).
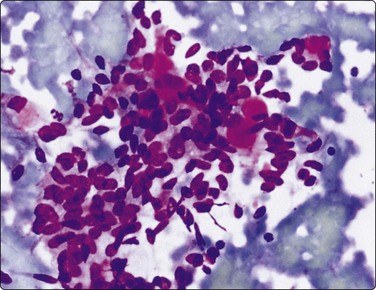
Fig. 4.58 Polymorphous low-grade adenocarcinoma
Cluster of small cells with oval, mildly irregular nuclei; bland nuclear chromatin; a few small hyaline stromal globules; some resemblance to adenoid cystic carcinoma (MGG, HP).
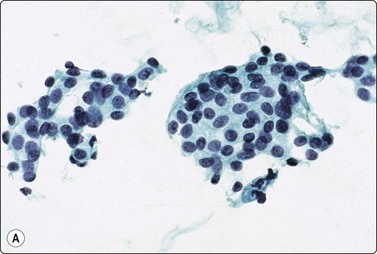
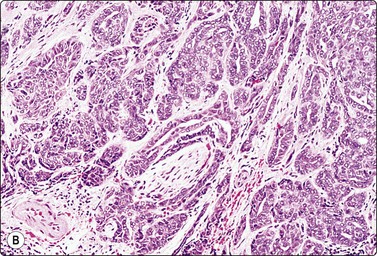
Fig. 4.59 Polymorphous low-grade adenocarcinoma
(A) Smears from this tumor of the palate were relatively scanty of small sheets of bland epithelial cells resembling squamous metaplasia (Pap, IP); (B) The corresponding tissue section shows a malignant infiltrative pattern with prominent perineural invasion (H&E, IP).
Polymorphous low-grade (terminal duct) adenocarcinoma is an uncommon tumor of low-grade malignancy occurring in minor salivary glands, mainly the palate. It is extremely rare in the parotid. Not many cases with FNB findings have been reported. The cells are mainly clustered or in epithelial fragments, which often have a trabecular/pseudopapillary structure with a fibrous stromal core (Fig. 4.57). The pattern may resemble adenoid cystic carcinoma, especially since small hyaline stromal globules are commonly seen (Fig. 4.58). The nuclei may appear deceptively bland even in tumors showing locally aggressive growth (Fig. 4.59), and false-negative reports are not uncommon.139
Epithelial-myoepithelial carcinoma (Figs 4.60-4.62)113,141-144
Usual findings
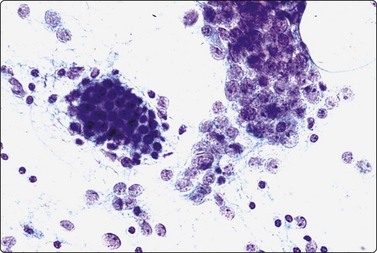
Fig. 4.60 Epithelial-myoepithelial carcinoma
Obvious biphasic pattern of clustered small epithelial cells (left), and less cohesive cells with pale fragile cytoplasm and large vesicular nuclei; no distinctly ‘clear’ cells (Pap, HP).
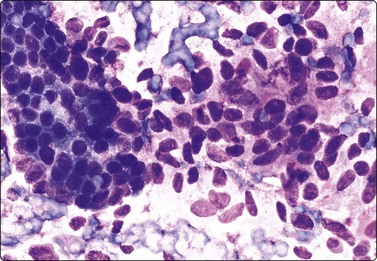
Fig. 4.61 Epithelial-myoepithelial carcinoma
Vaguely biphasic pattern of cohesive epithelial cells with rounded nuclei (left) and larger cells with indistinct cytoplasm and oval, mildly atypical nuclei. This case presented with pulmonary metastases (MGG, HP).
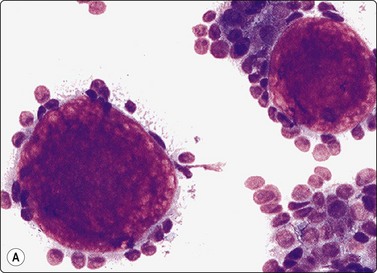
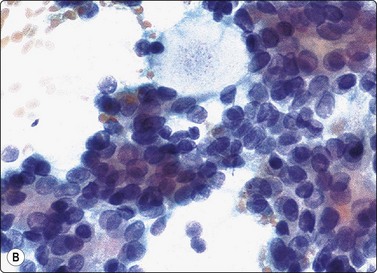
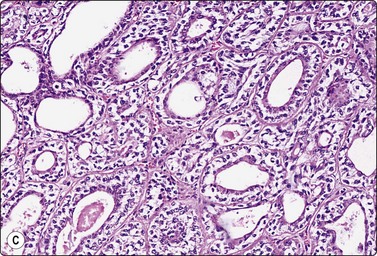
Fig. 4.62 Epithelial-myoepithelial carcinoma
In this case there were numerous hyaline stromal globules suggestive of adenoid cystic carcinoma. Note, however, fragile pale cytoplasm and bland nuclear chromatin; (A, MGG, IP; B, Pap, HP), (C) Tissue section, hyaline globules present only focally (H&E, IP)
(Courtesy Dr. J. Wright, Gribbles Pathology, Adelaide).
Epithelial-myoepithelial carcinoma is an uncommon tumor of low to intermediate malignancy, which mainly occurs in the parotid gland. The tumor has a metastatic potential. One of our cases initially presented with multiple tumor metastases in the lung from an unknown primary, another as a carcinoma ex pleomorphic adenoma.145 The cytological diagnosis is difficult for two reasons: a biphasic pattern is not often discernible, and the myoepithelial cells are not easily recognized as clear cells in cytological smears. The pale and indistinct cytoplasm is so fragile that it disperses in the background and most of the cells appear as naked nuclei, dispersed or clustered. The nuclei are mildly atypical, showing moderate enlargement and variation in size and shape, but have a pale chromatin and discrete central nucleoli. If present, epithelial cells are small and uniform and form tight cohesive clusters. Hyaline stromal material is often present, sometimes in the form of hyaline stromal globules similar to those of adenoid cystic carcinoma (Fig. 4.62). The composite population of both epithelial and myoepithelial cells can be confirmed by immunostaining.
A number of different neoplasms occurring in the salivary glands have a prominent clear cell component,146 clear cell carcinoma NOS, mainly epithelial-myoepithelial carcinoma, clear cell carcinoma NOS, sebaceous adenoma and metastatic renal cell carcinoma. A clear cell pattern can be present focally or uniformly in acinic cell and low-grade mucoepidermoid carcinoma and occasionally in oncocytoma, pleomorphic adenoma and basal cell adenoma. Although the clear cell pattern is striking in tissue sections, it is generally not well reproduced in cytological smears. The cytoplasm is never truly clear. At best it is abundant, pale and vacuolated with visible cell membranes, but it is often fragile and disperses in the background leaving the nuclei bare. Renal cell carcinoma not uncommonly metastasises to the parotid. The cells of renal cell carcinoma have abundant vacuolated pale cytoplasm with visible cell membranes. Nuclei vary in size and show variable but obvious atypia and often prominent nucleoli. Typically, the tumor cells adhere to strands of endothelial cells or vascular basement membrane material (see Fig. 4.77 and Chapter 12).
Adenoid cystic carcinoma (Figs 4.63-4.68)15,147,148
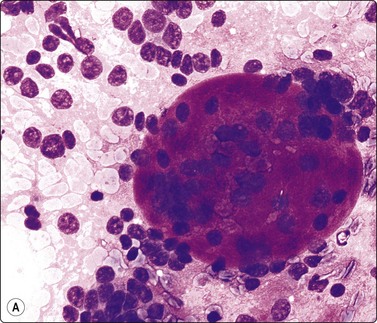
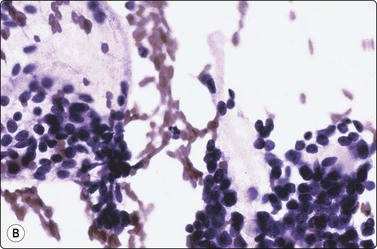
Fig. 4.63 Adenoid cystic carcinoma
(A) Small uniform epithelial cells with hyperchromatic nuclei and coarse chromatin, dispersed and adhering to a large, hyaline stromal globule (MGG, HP); (B) The hyaline stromal globules are less striking in Pap-stained smears and are pale, almost transparent (Pap, HP).
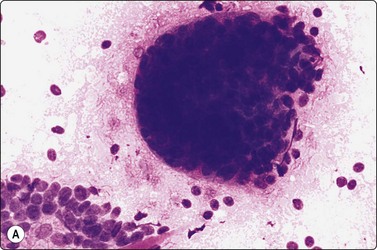
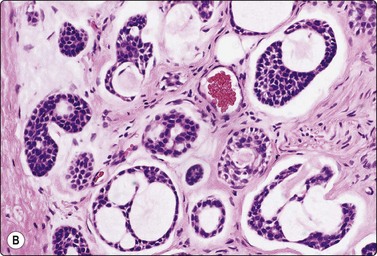
Fig. 4.64 Adenoid cystic carcinoma
Cellular epithelial tissue fragments with a characteristic cup shape open at one end; (A, MGG, IP; B, corresponding tissue section, H&E, IP).
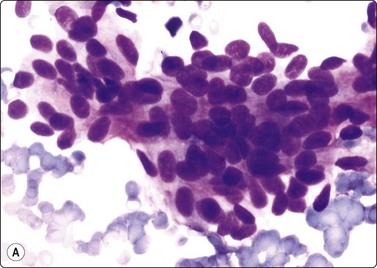
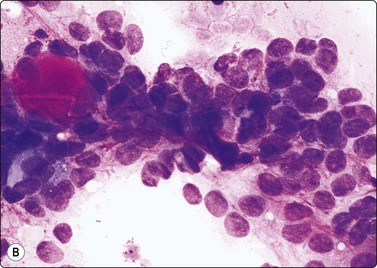
Fig. 4.65 Adenoid cystic carcinoma vs pleomorphic adenoma
(A) Pleomorphic adenoma; (B) Adenoid cystic carcinoma. Note bland nuclei of pleomorphic adenoma, coarse irregular chromatin and some nuclear molding in adenoid cystic carcinoma (MGG, HP).
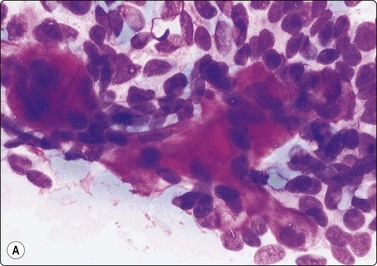
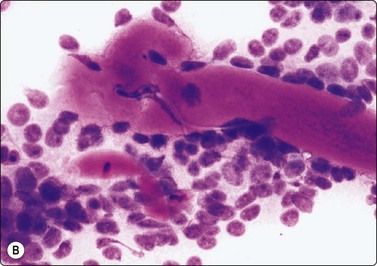
Fig. 4.66 Adenoid cystic carcinoma vs pleomorphic adenoma
(A) Pleomorphic adenoma; (B) Adenoid cystic carcinoma. Similar finger-like cords of hyaline stroma in both (MGG, HP).
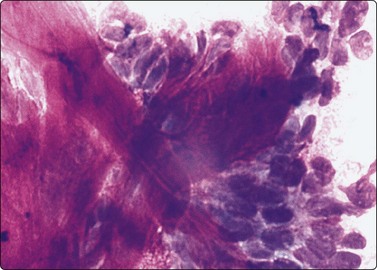
Fig. 4.67 Adenoid cystic carcinoma vs pleomorphic adenoma
Smear of adenoid cystic carcinoma showing a fibrillar fibromyxoid stroma similar to pleomorphic adenoma, but atypical nuclear chromatin (MGG, HP).
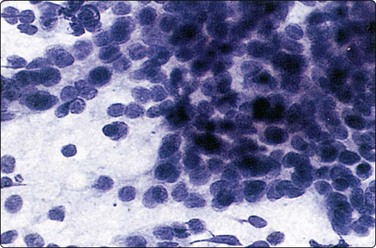
Fig. 4.68 Adenoid cystic carcinoma, poorly differentiated
Numerous small epithelial cells, single and in dense clusters; hyperchromatic nuclei with coarse chromatin, stromal elements absent (Pap, HP).
Adenoid cystic carcinoma (AdCC) is a common malignancy in the minor salivary glands and occasionally occurs in unusual sites such as the airways, lacrimal glands and external auditory canal.149 The hyaline stromal globules are the most striking feature of this neoplasm, but are not diagnostic thereof. They occur in several other entities (basal cell adenoma, canalicular adenoma, basal cell adenocarcinoma pleomorphic adenoma, polymorphous low-grade adenocarcinoma, epithelial-myoepithelial carcinoma). The globules vary considerably in size, stain bright red or purple and appear dense and homogeneous in MGG-stained smears. With Pap or H&E, the globules are pale, semi-translucent and less conspicuous (Fig. 4.63A,B). Finger-like or beaded strands or cords of stromal material have similar staining properties and hyaline texture (Fig. 4.66). They are characteristic but again not specific to AdCC. The tumor cells are both in multilayered, dense aggregates and dispersed as single cells and often adhere to the stromal material. The nuclei are rather uniform in size but have an irregular shape. They are hyperchromatic with coarsely granular chromatin and visible nucleoli. The cytoplasm is scanty, the N : C ratio is high and more nuclear molding is common. The tumor cells often form cup-shaped epithelial fragments with an open end reproducing the typical histology (Fig. 4.64).147 Malignant nuclear features are more obvious in poorly differentiated tumors with tight clusters of basaloid cells and little or no stroma (Fig. 4.68).
Not infrequently, FNB of an AdCC may cause intense pain, perhaps in some way related to the tendency for this tumor to infiltrate along nerve fibers. This can be a useful hint to the nature of the lesion.
Distinction from basal cell adenoma can be a problem (see basal cell adenoma). This mainly concerns the membranous and trabecular variants, which can contain prominent hyaline stromal globules.25,117,118 Marked variation in size and a dense, truly hyaline texture of the globules suggest AdCC. The nuclear morphology is an important distinguishing feature. The nuclei of AdCC are similarly small and relatively monomorphic but are hyperchromatic with a coarse chromatin, best seen in Pap-stained smears. Nucleoli may be prominent and nuclear membranes irregular and thickened. Other tumors that may contain hyaline stromal globules are pleomorphic adenoma, epithelial-myoepithelial carcinoma and polymorphous low-grade adenocarcinoma. Distinction from highly cellular pleomorphic adenoma is not always easy. Hyaline stromal globules may be seen in pleomorphic adenoma and, conversely, the stroma may focally appear fibromyxoid in AdCC (Figs 4.65-4.67). However, truly chondromyxoid matrix with spindle cells is specific to pleomorphic adenoma. In summary, a diagnosis of AdCC must not be based solely on the presence of hyaline globules, but requires a close scrutiny of cellular and nuclear features.
Some dermal neoplasms can closely resemble AdCC cytologically, and this may cause a problem if the tumor is situated superficially in the parotid region. For example, distinction from cutaneous cylindroma and basal cell carcinoma may be difficult.150,151 In a case from our files of recurrent cutaneous basal cell carcinoma deeply invading the parotid gland, smears were indistinguishable from poorly differentiated AdCC. Similar cases have been reported.152
A specific diagnosis of AdCC may be difficult in poorly differentiated tumors with a solid growth pattern due to the absence of characteristic stroma.153 Basaloid squamous carcinoma of the head and neck (see p. 43) is a rare tumor that should also be considered. The differential diagnosis depends on the identification of cells with obvious squamous differentiation. Neuroendocrine carcinoma and poorly differentiated metastatic carcinoma also enter the differential diagnosis.
Carcinoma ex pleomorphic adenoma (Figs 4.69, 4.70)110,114,154
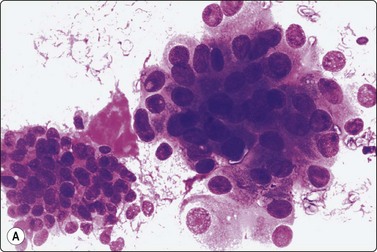
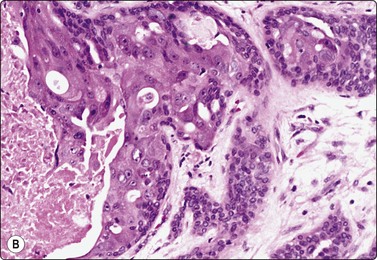
Fig. 4.69 Carcinoma ex pleomorphic adenoma
(A) The epithelial cell cluster to the right shows prominent nuclear enlargement and atypia; the cluster to the left is of benign cells associated with a fragment of myxoid stroma (MGG, HP); (B) Corresponding tissue section showing high-grade carcinoma of salivary duct carcinoma type (H&E, IP).
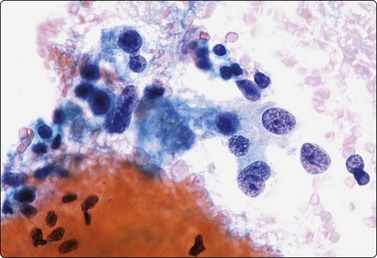
Fig. 4.70 Carcinoma ex pleomorphic adenoma
Poorly cohesive malignant epithelial cells in a bloody background; the cells at lower left appear bland with uniform oval nuclei. Histology showed a poorly differentiated carcinoma and some foci of residual benign pleomorphic adenoma (Pap, HP).
This is an uncommon event said to occur in 3–4% of pleomorphic adenomas.98 Clinically, a sudden increase in size of a tumor present for years signals the possibility of a malignant change. Most often, carcinoma ex pleomorphic adenoma is a salivary duct carcinoma or a poorly differentiated carcinoma of no specific type, but mucoepidermoid and squamous cell carcinoma, epithelial-myoepithelial carcinoma and acinic cell carcinoma have also been observed arising in PA.110,155,156
The main problem is to obtain representative samples from this type of tumor. The malignant component can be missed or overlooked. The occasional finding of worrying cytological atypia in benign pleomorphic adenoma has been mentioned. Carcinoma ex pleomorphic adenoma has the highest false-negative rate (35.3%) for FNB of all malignant salivary gland tumors.114 On the other hand, if no benign components are found in the sample, an unqualified diagnosis of malignancy is likely given. Since the prognosis and management of carcinoma ex pleomorphic adenoma are the same as for benign pleomorphic adenoma as long as the cancer remains confined within the capsule, an unqualified malignant diagnosis may result in unnecessarily extensive surgery. Clinical correlation is therefore essential.
Salivary duct carcinoma (Figs 4.71-4.73)157-161
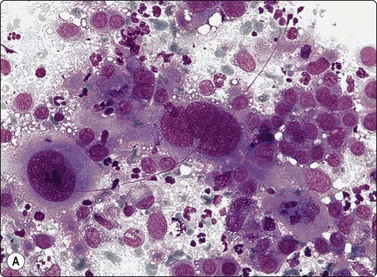
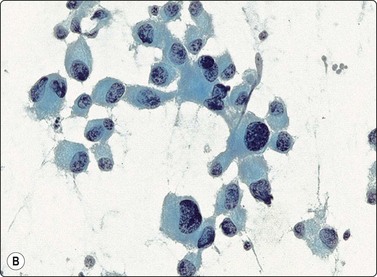
Fig. 4.71 Salivary duct carcinoma
Poorly cohesive, obviously malignant epithelial cells; large pleomorphic and hyperchromatic nuclei, abundant cytoplasm, no microarchitectural pattern, necrotic debris and inflammatory cells (A, MGG, HP; B, Pap, HP).
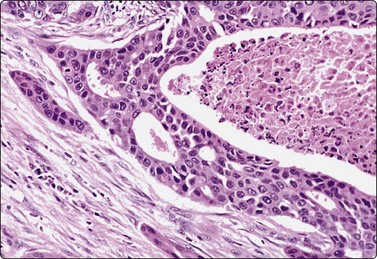
Fig. 4.72 Salivary duct carcinoma
Tissue section from similar case as Fig. 4.70. Note resemblance to high-grade ductal carcinoma in situ of breast (H& E, IP).
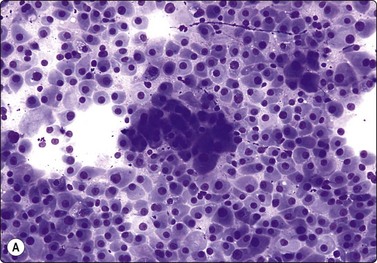
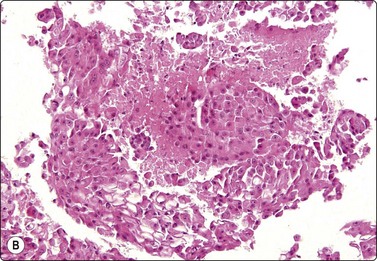
Fig. 4.73 Salivary duct carcinoma oncocytic type
(A) Smear showing numerous dispersed strikingly plasmacytoid cells with abundant dense cytoplasm, eccentric nuclei, anisokaryosis and prominent nucleoli (Diff-Quik, HP); (B) Cell button of same sample showing obvious tumor necrosis (H&E, IP).
Most salivary duct carcinomas are of high malignancy grade. They resemble high-grade ductal carcinoma in situ (comedocarcinoma) of the breast both histologically and cytologically. The neoplastic cells are obviously malignant with abundant cytoplasm and large, pleomorphic nuclei. Necrotic debris is usually present as in comedocarcinoma. The differential diagnosis is with other high-grade carcinomas such as high-grade mucoepidermoid and squamous carcinoma, and also includes metastatic breast carcinoma. In some cases, the cells are predominantly of or oncocytic type, suggesting malignant oncocytoma. Smears from one of our cases initially diagnosed as myoepithelial or oncocytic carcinoma showed prominently plasmacytoid cells with abundant dense cytoplasm and eccentric nuclei. The presence of focal tumor necrosis in a cell block pointed to salivary duct carcinoma, subsequently confirmed (Fig. 4.73).
Immune markers may be helpful in the distinction from other tumors with squamoid cells.162
Adenocarcinoma of no special type (Figs 4.74, 4.75)163
Usual findings
Primary adenocarcinoma of no special type is rare, mainly seen in minor salivary glands, or arising in pre-existing pleomorphic adenoma. There is a spectrum of patterns from relatively well-differentiated papillary cystadenocarcinoma, mucinous adenocarcinoma and ‘adenopapillary’ carcinoma (Fig. 4.74) to pleomorphic poorly differentiated carcinoma. It may represent a poorly differentiated component of a specific type such as acinic cell carcinoma. Metastatic origin can be difficult to exclude. Clinical examination and history are therefore essential and definitive type-specific diagnosis may have to await histological examination.
Squamous cell carcinoma164
The criteria for diagnosis are as for other sites. The common presence of lymph nodes within the parotid gland and adjacent to other salivary glands makes it difficult to decide if a squamous carcinoma is primary or metastatic by FNB. Distinction from poorly differentiated mucoepidermoid carcinoma may also be impossible.
Atypical metaplastic and degenerate squamous epithelial cells in some benign conditions such as necrotizing sialometaplasia, Warthin’s tumor and pleomorphic adenoma with squamous metaplasia can occasionally be mistaken for well-differentiated squamous cell carcinoma. Multiple sampling and clinical correlation are essential.
Other malignant neoplasms
Small cell anaplastic (neuroendocrine) carcinoma (Fig. 4.76) rarely occurs as a primary tumor in the salivary glands. The cytological pattern is the same as for neuroendocrine carcinomas in other sites, and a primary elsewhere, particularly in the lung, must be excluded before making this diagnosis.165 The smear pattern can be mistaken for malignant lymphoma, but there is usually a greater tendency to clustering of cells and nuclear molding. The diagnosis should be supported by immunostaining for neuroendocrine and pan-lymphocyte markers or by ultrastructural examination.
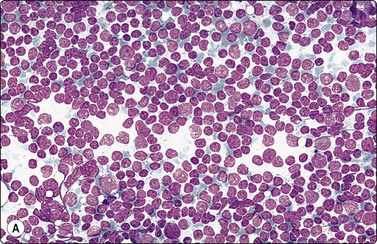
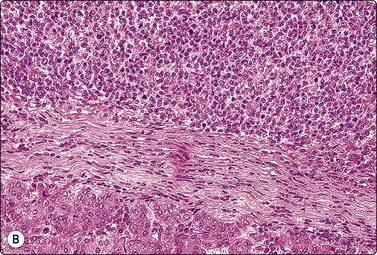
Fig. 4.76 Small cell carcinoma
Small cell neuroendocrine carcinoma primary in the parotid. Dispersed population of small cells with round nuclei and scanty cytoplasm resembling malignant lymphoma; neuroendocrine markers positive (A, MGG, HP; B, tissue section, H&E, IP).
Malignant myoepithelioma,166-168 malignant oncocytoma,169,170 carcinoma ex Warthin’s tumor,22,171 and sebaceous carcinoma172 are rare malignant tumors of the salivary glands, which have not yet been well characterized cytologically. The reader is referred to single case reports and small series for descriptions of these entities. Undifferentiated carcinoma of the parotid gland has been reported in elderly patients. Lymphoepithelial carcinoma of nasopharyngeal type has also been described in the salivary glands.173,174
Metastatic carcinoma, melanoma and lymphoma175-177 may involve either the salivary glands or lymph nodes adjacent to or within the gland. The commonest primary tumor is cutaneous squamous cell carcinoma of the head and neck. Metastatic renal cell carcinoma (Fig. 4.77) is to be distinguished from primary clear cell tumors, as mentioned above. The possibility of metastatic origin must be kept in mind when examining salivary gland aspirates.
1 Kocjan G, Ramsay A, Beale T, O’Flynn P. Head and neck cancer in the UK. What is expected of cytopathology Cytopathol. 2009;20:69-95.
2 Oliai BR, Sheth S, Burroughs FH, Ali SZ. ‘Parapharyngeal space’ tumors. A cytopathologic study of 24 cases on fine-needle aspiration. Diagn Cytopathol. 2005;32:11-15.
3 Arabi H, Yousef N, Bandyopadhyay S, et al. Fine needle aspiration of head and neck masses in the operating room: accuracy and potential benefits. Diagn Cytopathol. 2008;36:369-374.
4 Rapkiewicz A, Le BT, Simsir A, et al. Spectrum of H & N lesions diagnosed by F-NAC in the paediatric population. Cancer Cytopathol. 2007;111:242-251.
5 Engzell U, Franzen S, Zajicek J. Aspiration biopsy of tumours of the neck. II. Cytologic findings in 13 cases of carotid body tumour. Acta Cytol. 1971;15:25-30.
6 Daskalopoulou D, Rapidis AD, Maounis N, Markidou S. Fine-needle aspiration cytology in tumors and tumor-like conditions of the oral and maxillofacial region. Diagnostic reliability and limitations. Cancer (Cancer Cytopathol). 1997;81:238-252.
7 Bardales RH, Baker SJ, Mukunyadzi P. Fine-needle aspiration cytology findings in 214 cases of nonparotid lesions in the head. Diagn Cytopathol. 2000;22:211-217.
8 Layfield L. Fine-needle aspiration in the diagnosis of head and neck lesions: a review and discussion of problems in differential diagnosis. Diagn Cytopathol. 2007;35:798-805.
9 Seifert G, Sobin LH. The World Health Organization’s histological classification of salivary gland tumors. A commentary on the second edition. Cancer. 1992;70:379-385.
10 Chan M, McGuire L. Cytodiagnosis of lesions presenting as salivary gland swellings. A report of seven cases. Diagn Cytopathol. 1992;8:439-443.
11 Nettle WJ, Orell SR. Fine needle aspiration in the diagnosis of salivary gland lesions. Aust NZ J Surg. 1989;59:47-51.
12 Layfield L, Gopez E, Hirschowitz S. Cost efficiency analysis for fine-needle aspiration in the workup of parotid and submandibular gland nodules. Diagn Cytopathol. 2006;34:734-738.
13 Eneroth CM, Zajicek J. Aspiration biopsy of salivary gland tumours. II. Morphologic studies on smears and histologic sections from oncocytic tumours. Acta Cytol. 1965;9:355-361.
14 Eneroth CM, Zajicek J. Aspiration biopsy of salivary gland tumours. III. Morphologic studies on smears and histologic sections from 368 mixed tumours. Acta Cytol. 1966;10:440-454.
15 Eneroth CM, Zajicek J. Aspiration biopsy of salivary gland tumours. IV. Morphologic studies on smears and histologic sections from 45 cases of adenoid cystic carcinoma. Acta Cytol. 1969;13:59-63.
16 Zajicek J, Eneroth CM. Cytological diagnosis of salivary gland carcinomata from aspiration biopsy smears. Acta Otolaryng. 1970;263(Suppl.):183-185.
17 Zajicek J, Eneroth CM, Jakobson P. Aspiration biopsy of salivary gland tumors. VI. Morphologic studies on smears and histologic sections from mucoepidermoid carcinoma. Acta Cytol.. 1976;20:35-41.
18 Atuta T, Grenman R, Laippala P, Klemi P-J. Fine-needle aspiration biopsy in the diagnosis of parotid gland lesions: evaluation of 438 biopsies. Diagn Cytopathol. 1996;15:185-190.
19 Al-Khafaji B, Nestok BR, Katz RL. Fine-needle aspiration of 154 parotid masses with histologic correlation. Ten-year experience at the University of Texas M.D.Anderson Cancer Center. Cancer (Cancer Cytopathol). 1998;84:153-159.
20 Stewart CJR, MacKenzie K, McGarry GW, Mowat A. Fine-needle aspiration cytology of salivary gland: a review of 341 cases. Diagn Cytopathol. 2000;22:139-146.
21 Young JA. Diagnostic problems in fine needle aspiration cytopathology of the salivary glands. J Clin Pathol. 1994;47:193-198.
22 Klijanienko J, Vielh P, Batsakis JD, et al. Monographs in Clinical Cytology. Volume 15. Salivary Gland Tumours. Basel, Switzerland: Karger; 2000.
23 Cramer H, Layfield L, Lampe H. Fine needle aspiration of salivary gland lesions. In: Schmidt WA, editor. Cytopathology annual. Baltimore: Williams and Wilkins; 1993:181-206.
24 MacLeod CB, Frable WJ. Fine needle aspiration biopsy cytology of the salivary glands: problem cases. Diagn Cytopathol. 1993;9:216-225.
25 Orell SR. Diagnostic difficulties in the interpretation of fine needle aspirates of salivary gland lesions: the problem revisited. Cytopathol. 1995;6:285-300.
26 Raywanshi A, Gupta K, Gupta N, et al. Fine needle aspiration cytology of salivary glands. Diagnostic pitfalls revisited. Diagn Cytopathol. 2006;34:580-584.
27 Daneshbod Y, Daneshbod K, Khaderi B. Diagnostic difficulties in the interpretation of fine needle aspirate samples in salivary lestions. Diagnostic pitfalls revisited. Acta Cytol. 2009;53:53-70.
28 Mukunyadzi P, Bardales RH, Palmer HE, Stanley MW. Tissue effects of salivary gland fine-needle aspiration. Does this procedure preclude accurate histologic diagnosis? Am J Clin Pathol. 2000;114:741-745.
29 Batsakis JG, Sneige N, El-Naggar AK. Fine-needle aspiration of salivary glands: its utility and tissue effects. Ann Otol Rhinol Laryngol. 1992;101:185-188.
30 Li S, Baloch ZW, Tomaszewski JE, LiVolsi VA. Worrisome histologic alterations following fine-needle aspiration of benign parotid lesions. Arch Pathol Lab Med. 2000;124:87-91.
31 Parfitt JR, McLachlin M, Weir MM. Comparison of Thin Prep and conventional smears in salivary gland fine-needle aspiration biopsies. Cancer. 2007;111:123-129.
32 Engzell U, Zajicek J. Aspiration biopsy of tumours of the neck. I. Aspiration biopsy and cytologic findings in 100 cases of congenital cysts. Acta Cytol. 1970;14:51-57.
33 Frable WJ, Frable MAS. Thin-needle aspiration biopsy: the diagnosis of head and neck tumours revisited. Cancer. 1979;43:1541-1548.
34 Burgess KL, Hartwick RWJ, Bedard YC. Metastatic squamous carcinoma presenting as a neck cyst. Differential diagnosis from inflamed branchial cleft cyst in fine needle aspirates. Acta Cytol. 1993;37:494-498.
35 Üstün M, Risberg B, Davidson B, Berner A. Cystic change in metastatic lymph nodes: a common diagnostic pitfall in fine-needle aspiration cytology. Diagn Cytopathol. 2002;27:387-392.
36 Sheahan P, O’Leary G, Lee G, Fitzgibbon J. Cystic cervical metastases: incidence and diagnosis using FNAB. Otolaryngol Head Neck Surg. 2002;127:294-298.
37 Shakin A, Burroughs FH, Kirby JP, Ali SZ. Thyroglossal duct cyst: a cytopathologic study of 26 cases. Diagn Cytopathol. 2005;33:365-369.
38 De Las Casas L, Bardales RH. Fine-needle aspiration cytology of mucous retention cyst of the tongue: distinction from other cystic lesions of the tongue. Diagn Cytopathol. 2000;22:308-312.
39 Michael CW, Naylor B. Amyloid in cytologic specimens. Differential diagnosis and diagnostic pitfalls. Acta Cytol. 1999;43:746-755.
40 Frable MA, Frable WJ. Fine needle aspiration biopsy in the diagnosis of sarcoid of the head and neck. Acta Cytol. 1984;28:175-177.
41 Micheau C, Klijanienko J, Luboinski B, Richard J. So-called branchiogenic carcinoma is actually cystic metastases in the neck from a tonsillar primary. Laryngoscope. 1990;100:878-883.
42 Bhanote M, Yang GCH. Malignant first branchial cleft cysts presenting as submandibular abscesses in fine-needle aspiration: report of three cases and review of the literature. Diagn Cytopathol. 2008;36:876-881.
43 Klijanienko J, El-Naggar A, Ponzio-Prion A, et al. Basaloid squamous carcinoma of head and neck. Immunohistochemical comparison with adenoid cystic carcinoma and squamous cell carcinoma. Arch Otolaryngol Head Neck Surg. 1993;119:887-890.
44 Micheau C. What’s new in histological classification and recognition of naso-pharyngeal carcinoma (N.P.C.). Path Res Pract. 1986;181:249-253.
45 Jayaram G, Swain M, Khanijow V, Jalaludin MA. Fine-needle aspiration cytology of metastatic nasopharyngeal carcinoma. Diagn Cytopathol. 1998;19:168-172.
46 Kollur SM, El Hag IA. Fine-needle aspiration cytology of metastatic nasopharyngeal carcinoma in cervical lymph nodes: comparison with metastatic squamous cell carcinoma, and Hodgkin’s and non-Hodgkin’s lymphoma. Diagn Cytopathol. 2003;28:18-22.
47 Viguer JM, Jimenez-Hefferman JA, Lopez-Ferrer P, et al. Fine-needle aspiration cytology of metastatic nasopharyngeal carcinoma. Diagn Cytopathol. 2005;32:233-237.
48 Helsel JC, Bardales RH, Mukunyadzi P. Fine-needle aspiration biopsy cytology of malignant neoplasms of the sinonasal tract. A review of 22 primary and metastatic tumors. Cancer (Cancer Cytopathol). 2003;99:105-112.
49 Bellizzi AM, Bourne TD, Mills SE, Stelow EB. The cytologic features of sinonasal undifferentiated carcinoma and olfactory neuroblastoma. Am J Clin Pathol. 2008;129:367-376.
50 Lack EE, Cubilla AL, Woodruff JM. Paragangliomas of the head and neck region. A pathologic study of tumours from 71 patients. Human Pathol. 1979;10:191-218.
51 Fleming MV, Oertel YC, Rodrigues ER. Fine-needle aspiration of six carotid body paragangliomas. Diagn Cytopathol. 1993;9:510-515.
52 Verma K, et al. Cytomorphological spectrum in paraganglioma. Acta Cytol. 2008;52:549-556.
53 Solares J, Lacruz C. Fine needle aspiration cytology diagnosis of an extracranial meningioma presenting as a cervical mass. Acta Cytol. 1987;31:502-504.
54 Tan LHC. Meningioma presenting as a parapharyngeal tumor. Report of a case with fine needle aspiration cytology. Acta Cytol. 2001;45:1053-1059.
55 Hameed A, Gokden M, Hanna EY. Fine-needle aspiration cytology of a primary ectopic meningioma. Diagn Cytopathol. 2002;26:297-300.
56 Collins BT, Cramer HM, Hearn SA. Fine needle aspiration cytology of metastatic olfactory neuroblastoma. Acta Cytol. 1997;41:802-810.
57 Logrono R, Futoran RM, Hartig G, Inhorn SL. Olfactory neuroblastoma (esthesioneuroblastoma): appearance on fine-needle aspiration: report of a case. Diagn Cytopathol. 1997;17:205-208.
58 Zajdela A, Vielh P, Schlinger P, Haye C. Fine-needle cytology of 292 palpable orbital and eyelid tumors. Am J Clin Pathol. 1990;93:100-104.
59 Gupta S, Sood B, Gulati M, et al. Orbital mass lesions: US-guided fine-needle aspiration biopsy – experience in 37 patients. Radiology. 1999;213:568-572.
60 Krzystolik Z, Roslawska A, Bedner E. The cytological, immunocytochemical and molecular genetic analysis in diagnosis of the neoplasms of the eye, eye adnexa and orbit. Doc Ophtalmol. 1994;88:155-163.
61 Nassar DL, Raab SS, Silverman JF, et al. Fine-needle aspiration for the diagnosis of orbital hematolymphoid lesions. Diagn Cytopathol. 2000;23:314-317.
62 Rastogi A, Jain S. Fine needle aspiration biopsy in orbital lesions. Orbit. 2001;20:11-23.
63 Wolska-Szmidt E, Jakkubowska A, et al. Fine needle aspiration biopsy and molecular analysis in differential diagnosis of lymphoproliferative diseases of the orbit and eye adnexa. Pol J Pathol. 2004;55:51-57.
64 Tani E, Seregard S, Rupp G, et al. Fine-needle aspiration cytology and immunocytochemistry of orbital masses. Diagn Cytopathol. 2006;34:1-5.
65 Sturgis CD, Silverman JF, Kennerdell JS, Raab SS. Fine-needle aspiration for the diagnosis of primary epithelial tumors of the lacrymal gland and ocular adnexa. Diagn Cytopathol. 2001;24:86-89.
66 Klijanienko J, El-Naggar AK, Servois V, et al. Histologically similar synchronous or metachronous lacrimal salivary-type and parotid gland tumors: A series of 11 cases. Head Neck. 1999;21:512-516.
67 Dhaliwal U, Arora VK, Singh N, Bhatia A. Clinical and cytopathologic correlation in chronic inflammations of the orbit and ocular adnexa: a review of 55 cases. Orbit. 2004;23:219-225.
68 Trolet J, Hupé P, Huon I, et al. Genomic profiling and identification of high-risk uveal melanoma by array CGH analysis of primary tumors and liver metastases. Invest Ophthalmol Vis Sci. 2009;50:2572-2580. Epub 2009 Jan 17
69 Chan DH, Miller TR, Ljung BM, et al. Fine needle aspiration biopsy in uveal melanoma. Acta Cytol. 1989;33:599-605.
70 Sen S, Singha U, Kumar H, et al. Diagnostic intraocular fine-needle aspiration biopsy – an experience in three cases of retinoblastoma. Diagn Cytopathol. 1999;21:331-334.
71 Karcioglu ZA. Fine needle aspiration biopsy (FNAB) for retinoblastoma. Retina. 2002;22:707-710.
72 Ljung B-M, Chan D, Miller TR, et al. Intraocular lymphoma. Cytologic diagnosis and the role of immunologic markers. Acta Cytol. 1988;32:840-847.
73 Willems J. Aspiration biopsy cytology of tumors of the central nervous system and the base of the skull. In: Linsk JA, Franzen S, editors. Clinical aspiration cytology. Philadelphia: Lippincott; 1983:361-370.
74 Russel D. The wet-film technique in neurosurgery. In: Recent advances in clinical pathology. London: Churchill; 1947.
75 Smith AR, Elsheik TM, Silverman JF. Intraoperative cytologic diagnosis of suprasellar and sellar cystic lesions. Diagn Cytopathol. 1999;20:137-147.
76 Bleggi-Torres LF, de Noronha L, Schneider Gugelmin E, et al. Accuracy of the smear technique in the cytological diagnosis of 650 lesions of the central nervous system. Diagn Cytopathol. 2001;24:293-295.
77 Roessler K, Dietrich W, Kitz K. High diagnostic accuracy of cytologic smears of central nervous system tumors. A 15-year experience based on 4,172 patients. Acta Cytol. 2002;46:667-674.
78 Vural G, Hagmar B, Walaas L. Extracranial metastasis of glioblastoma multiforme diagnosed by fine needle aspiration: a report of two cases and a review of the literature. Diagn Cytopathol. 1996;15:60-65.
79 Domanski H, Akerman M. Fine-needle aspiration cytology of tongue swellings. A study of 75 cases. Diagn Cytopathol. 1998;18:387-392.
80 Jin B, Saleh H. Pitfalls in the diagnosis of adult rhabdomyoma by fine needle aspiration: report of a case and a brief literature review. Diagn Cytopathol. 2009;37:483-486.
81 Günhan Ö, Dogan N, Celasun B, et al. Fine needle aspiration cytology of oral cavity and jaw bone lesions. A report of 102 cases. Acta Cytol. 1993;37:135-141.
82 Vargas PA, da Cruz-Perez DE, Mata GM, et al. Fine needle aspiration cytology as an additional tool in the diagnosis of odontogenic keratocyst. Cytopathol. 2007;18:361-366.
83 Ramzy I, Aufdemorte TB, Duncan OL. Diagnosis of radiolucent lesions of the jaw by fine needle aspiration biopsy. Acta Cytol. 1985;29:419-424.
84 Mathew S, Rappaport K, Ali SZ, et al. Ameloblastoma. Cytologic findings and literature review. Acta Cytol. 1997;41:955-960.
85 Ingram EA, Evans ML, Zitsch RP. Fine needle aspiration cytology of ameloblastic carcinoma of the maxilla: a rare tumor. Diagn Cytopathol. 1996;14:249-252.
86 Spitz DJ, Reddy V, Selvaggi SM, et al. Fine-needle aspiration of scalp lesions. Diagn Cytopathol. 2000;23:35-38.
87 Henry-Stanley MJ, Bencke J, Bardales RH, Stanley MW. Fine-needle aspiration of normal tissue from enlarged salivary glands: sialosis or missed target? Diagn Cytopathol. 1995;13:300-303.
88 Gupta S, Sodhani P. Sialadenosis of parotid gland. A cytomorphologic and morphometric study of four cases. Analyt. Quant. Cytol. Histol. 1998;20:225-228.
89 Layfield LJ, Gopez EV. Cystic lesions of the salivary glands: cytologic features in fine-needle aspiration biopsies. Diagn Cytopathol. 2002;27:197-204.
90 Jayaram G, Khurana N, Basu S. Crystalloids in a cystic lesion of parotid salivary gland. Diagn Cytopathol. 1993;9:70-71.
91 Nasuti JF, Gupta PK, Fleisher SR, LiVolsi VA. Nontyrosine crystalloids in salivary gland lesions: report of seven cases with fine-needle aspiration cytology and follow-up surgical pathology. Diagn Cytopathol. 2000;22:167-171.
92 Pantanowitz L, Goulart RA, Cao QJ. Salivary gland crystalloids. Diagn Cytopathol. 2006;34:749.
93 Elliott JN, Oertel YC. Lymphoepithelial cysts of the salivary glands. Histological and cytologic features. Am J Clin Pathol. 1990;93:39-43.
94 Chhieng DC, Argosino R, McKenna B, et al. Utility of fine-needle aspiration in the diagnosis of salivary gland lesions in patients infected with human immunodeficiency virus. Diagn Cytopathol. 1999;21:260-264.
95 Johnson FB, Oertel YC, Ammann K. Sialadenitis with crystalloid formation: a report of six cases diagnosed by fine-needle aspiration. Diagn Cytopathol. 1995;12:76-80.
96 Wax TD, Layfield LJ, Zaleski S, et al. Cytomegalovirus sialadenitis in patients with the acquired immunodeficiency syndrome: a potential diagnostic pitfall with fine-needle aspiration cytology. Diagn Cytopathol. 1994;10:169-174.
97 Casiano RR, Cooper JD, Gould E, et al. Value of needle biopsy in directing management of parotid lesions in HIV-positive patients. Head Neck. 1991;13:411-414.
98 Ellis GL, Auclair PL, Gnepp DR. Surgical pathology of the salivary glands. Philadelphia: Saunders; 1991.
99 Mooney EE, Dodd LG, Layfield LJ. Squamous cells in fine-needle aspiration biopsies of salivary gland lesions: Potential pitfalls in cytologic diagnosis. Diagn Cytopathol. 1996;15:447-452.
100 Cheuk W, Chan JKC. Kuttner tumor of the submandibular gland. Fine-needle aspiration cytologic findings of seven cases. Am J Clin Pathol. 2002;117:103-108.
101 Kaba S, Kojima M, Matsuda H, et al. Kuttner’s tumor of the submandibular glands: report of five cases with fine-needle aspiration cytology. Diagn Cytopathol. 2006;34:631-635.
102 Williams SB, Foss RD, Ellis GL. Inflammatory pseudotumors of the major salivary glands. Clinicopathologic and immunohistochemical analysis of six cases. Am J Surg Pathol. 1992;16:896-902.
103 Aufdemorte TB, Ramzy I, Holt GR, et al. Focal adenomatoid hyperplasia of salivary glands. A differential diagnostic problem in fine needle aspiration biopsy. Acta Cytol. 1985;29:23-28.
104 Klijanienko J, Vielh P. Fine-needle sampling of salivary gland lesions I. Cytology and histology correlation of 412 cases of pleomorphic adenoma. Diagn Cytopathol. 1996;14:195-200.
105 Viguer JM, Vicandi B, Jiménez-Hefferman JA, et al. Fine needle aspiration cytology of pleomorphic adenoma. An anlysis of 212 cases. Acta Cytol. 1997;41:786-794.
106 Bottles K, Ferrell LD, Miller TR. Tyrosine crystals in fine needle aspirates of a pleomorphic adenoma of the parotid gland. Acta Cytol. 1984;28:490-492.
107 Kawahara A, Harada H, Kage M, et al. Characterization of the epithelial components in pleomorphic adenoma of salivary gland. Acta Cytol. 2002;46:1095-1100.
108 Stanley MW. Selected problems in fine needle aspiration of head and neck masses. Mod Pathol. 2002;15:342-350.
109 Handa U, Dhingra N, Chopra R, Mohan H. Pleomorphic adenoma: cytologic variants and potential diagnostic pitfalls. Diagn Cytopathol. 2009;37:11-15.
110 Klijanienko J, El-Naggar AK, Servois V, et al. Mucoepidermoid carcinoma ex pleomorphic adenoma. Nonspecific preoperative cytologic findings in six cases. Cancer. 1998;84:231-234.
111 Siddaraju N, Murugam P, Basu D, Verma SK. Preoperative diagnosis of cystic pleomorphic adenoma with squamous metaplasia and cholesterol crystals. Acta Cytol. 2009;53:101.
112 Layfield LJ, Glasgow BJ. Aspiration cytology of clear-cell lesions of the parotid gland: morphologic features and differential diagnosis. Diagn. Cytopathol. 1993;9:705-712.
113 Wax T, Layfield L. Epithelial-myoepithelial cell carcinoma of the parotid gland: a case report and comparison of cytologic features with other stromal, epithelial, and myoepithelial cell containing lesions of the salivary glands. Diagn Cytopathol. 1996;14:298-304.
114 Klijanienko J, El-Naggar AK, Vielh P. Fine-needle findings in 26 carcinoma ex pleomorphic adenomas: diagnostic pitfalls and clinical considerations. Diagn Cytopathol. 1999;21:163-166.
115 Kapila K, Mathur S, Verma K. Schwannomas: a pitfall in the diagnosis of pleomorphic adenomas on fine-needle aspiration cytology. Diagn Cytopathol. 2002;27:53-59.
116 Domagala W, Halczy-Kowalik L, Welen K, Osborn M. Coexpression of glial fibrillary acid protein, keratin and vimentin. A unique feature useful in the diagnosis of pleomorphic adenoma in the salivary gland in fine needle aspiration biopsy smears. Acta Cytol. 1988;32:403-408.
117 Stanley MW, Horwitz CA, Rollins SD, et al. Basal cell (monomorphic) and minimally pleomorphic adenomas of the salivary glands. Distinction from the solid (anaplastic) type of adenoid cystic carcinoma in fine-needle aspiration. Am. J. Clin. Pathol. 1996;106:35-41.
118 Klijanienko J, El-Naggar AK, Vielh P. Comparative cytological and histological study of 15 salivary basal cell tumors; diagnostic and differential diagnostic considerations. Diagn Cytopathol. 1999;21:30-34.
119 Fregnani ER, Gerhard R, da Cruz-Perez DE, et al. Cytological features of intraoral tumors. Cytopathol. 2006;17:205-207.
120 Sparrow SA, Frost FA. Salivary monomorphic adenoma of dermal analogue type: report of two cases. Diagn Cytopathol. 1993;9:300-303.
121 Klijanienko J, Vielh P. Fine needle sampling of salivary gland lesions II. Cytology and histology correlation of 71 cases of Warthin’s tumor (adenolymphoma). Diagn Cytopathol. 1997;16:221-225.
122 Ballo MS, Shin HJC, Sneige N. Sources of diagnostic error in the fine needle aspiration diagnosis of Warthin’s tumor and clues to a correct diagnosis. Diagn Cytopathol. 1997;17:230-234.
123 Parwani AV, Ali SZ. Diagnostic accuracy and pitfalls in fine-needle aspiration interpretation of Warthin’s tumor. Cancer (Cancer Cytopathol). 2003;99:166-171.
124 O’Dwyer P, Farrar WB, James AG, et al. Needle aspiration biopsy of major salivary gland tumors: Its value. Cancer. 1986;57:554-557.
125 Sagi A, Giorgadze TA, Eleazar J, et al. Clear cell and eosinophilic oncocytomas of salivary gland: cytological variants or parallels? Diagn Cytopathol. 2007;35:158-163.
126 Goyal R, Ahuja A, Gupta N, et al. Multifocal nodular oncocytic hyperplasia in parotid gland. A case report. Acta Cytol. 2007;51:621-623.
127 Dodd LG, Caraway NP, Luna MA, Byers RM. Myoepithelioma of the parotid. Report of a case initially examined by fine needle aspiration biopsy. Acta Cytol. 1994;38:417-421.
128 Siddaraju N, Badhe BA, Goneppanavar M, Mishra MM. Preoperative fine needle aspiration cytologic diagnosis of spindle cell myoepithelioma of the parotid gland. A case report. Acta Cytol. 2008;52:495-499.
129 Chhieng DC, Cohen J-M, Cangiarella JF. Fine-needle aspiration of spindle cell and mesenchymal lesions of the salivary glands. Diagn Cytopathol. 2000;23:253-259.
130 Klijanienko J, Vielh P. Fine needle sampling of salivary gland lesions V. Cytology of 22 cases of acinic cell carcinoma with histologic correlation. Diagn Cytopathol. 1997;17:347-352.
131 Nagel H, Laskawi R, Büter JJ, et al. Cytologic diagnosis of acinic-cell carcinoma of salivary glands. Diagn Cytopathol. 1997;16:402-412.
132 Shet T, Ghodke R, Kane S, Chinoy R. Cytomorphologic patterns in papillary cystic variant of acinic cell carcinoma of the salivary gland. Acta Cytol. 2006;50:388-392.
133 Mosunjac MB, Siddiqui MT, Tadros T. Acinic cell carcinoma – papillary cystic variant. Pitfalls of fine needle aspiration diagnosis: study of five cases and review of literature. Cytopathol. 2009;20:96-102.
134 Cohen MB, Fisher PE, Holly EA, et al. Fine needle aspiration biopsy diagnosis of mucoepidermoid carcinoma. Acta Cytol. 1990;34:43-49.
135 Kumar N, Kapila K, Verma K. Fine needle aspiration cytology of mucoepidermoid carcinoma: a diagnostic problem. Acta Cytol. 1991;35:357-359.
136 Klijanienko J, Vielh P. Fine needle sampling of salivary gland lesions IV. Review of 50 cases of mucoepidermoid carcinoma with histologic correlation. Diagn Cytopathol. 1997;17:92-98.
137 Goonewardene SA, Nasuti JF. Value of mucin detection in distinguishing MEC from Warthin’s tumor on fine needle aspiration. Acta Cytol. 2002;46:704-708.
138 Edwards PC, Wasserman P. Evaluation of cystic salivary gland lesions by FNA. Acta Cytol. 2005;49:489-494.
139 Frierson HFJr, Covell JL, Mills SE. Fine needle aspiration cytology of terminal duct carcinoma of minor salivary gland. Diagn Cytopathol. 1987;3:159-162.
140 Klijanienko J, Vielh P. Salivary carcinomas with papillae: cytology and histology analysis of polymorphous low-grade adenocarcinoma and papillary cystadenocarcinoma. Diagnostic Cytopathology. 1998;19:244-249.
141 Gibbons D, Saboorian MH, Vuitch F, et al. Fine-needle aspiration findings in patients with polymorphous low grade adenocarcinoma of the salivary glands. Cancer (Cancer Cytopathology). 1999;87:31-36.
142 Kocjan G, Milroy C, Fisher EW, Eveson JW. Cytologic features of epithelial-myoepithelial carcinoma of salivary gland: potential pitfalls in diagnosis. Cytopathol. 1993;4:173-180.
143 Klijanienko J, Vielh P. Fine needle sampling of salivary gland lesions VII. Cytology and histology correlation of 5 cases of epithelial-myoepithelial carcinoma. Diagn Cytopathol. 1998;19:405-409.
144 Ng WK, Choy C, Ip P, et al. Fine needle aspiration cytology of epithelial-myoepithelial carcinoma of salivary glands. A report of three cases. Acta Cytol. 1999;43:675-680.
145 Miliauskas J, Orell SR. Fine-needle aspiration cytological findings in five cases of epithelial/myoepithelial carcinoma of salivary glands. Diagn Cytopathol. 2003;28:163-167.
146 Seifert G. Classification and differential diagnosis of clear and basal cell tumors of the salivary glands. Semin Diagn Pathol. 1996;13:95-103.
147 Klijanienko J, Vielh P. Fine needle sampling of salivary gland lesions III. Cytology and histology correlation of 75 cases of adenoid cystic carcinoma. Review and experience at the Institut Curie with emphasis on cytologic pitfalls. Diagn Cytopathol. 1997;17:36-41.
148 Nagel H, Hotze HJ, Laskawi R, et al. Cytologic diagnosis of adenoid cystic carcinoma of salivary glands. Diagn Cytopathol. 1999;20:358-366.
149 Mohan H, Handa U, Amanjit, et al. Adenoid cystic carcinoma of the external auditory canal. A case report with diagnosis by fine needle aspiration. Acta Cytol. 2003;47:792-794.
150 Bondeson L, Lindholm K, Thorstenson S. Benign dermal eccrine cylindroma. A pitfall in the cytologic diagnosis of adenoid cystic carcinoma. Acta Cytol. 1983;27:326-328.
151 Yang GCH, Waisman J. Distinguishing adenoid cystic carcinoma from cylindromatous adenomas in salivary gland fine-needle aspirates: the cytologic clues and their ultrastructural basis. Diagn Cytopathol. 2006;34:284-288.
152 Stanley MW, Hurwitz CA, Bardales RH, et al. Basal cell carcinoma metastatic to the salivary glands: differential diagnosis in fine-needle aspiration cytology. Diagn Cytopathol. 1997;16:247-252.
153 Yu GH, Caraway NP. Poorly-differentiated adenoid cystic carcinoma: cytologic appearance in fine-needle aspirates of distant metastases. Diagn Cytopathol. 1996;15:296-300.
154 Smith-Frable MA, Frable WJ. Fine-needle aspiration biopsy of salivary glands. Laryngoscope. 1991;101:245-249.
155 Jacobs JC. Low grade mucoepidermoid carcinoma ex pleomorphic adenoma. A diagnostic problem in fine needle aspiration biopsy. Acta Cytol. 1994;38:93-97.
156 Miliauskas J, Orell SR. Acinic cell carcinoma arising in pleomorphic adenoma of parotid gland. Cytopathology. 2000;11:356-359.
157 Elsheikh TM, Bernachi EG, Pisharodi L. Fine needle aspiration cytology of salivary duct carcinoma. Diagn Cytopathol. 1994;11:47-51.
158 Fyrat P, Cramer H, Feczko J, et al. Fine needle aspiration biopsy of salivary duct carcinoma: report of five cases. Diagn Cytopathol. 1997;16:526-530.
159 Khurana KK, Pitman MB, Powers CN, et al. Diagnostic pitfalls of aspiration cytology of salivary duct carcinoma. Cancer (Cancer Cytopathol). 1997;81:373-378.
160 Klijanienko J, Vielh P. Cytologic characteristics and histomorphologic correlations of 21 salivary duct carcinomas. Diagn Cytopathol. 1998;19:333-337.
161 Garcia-Bonafé M, Catala I, Tarragona J, Tallada N. Cytologic diagnosis of salivary duct carcinoma: a review of seven cases. Diagn Cytopathol. 1998;19:120-123.
162 Kawakara A, Harada H, Akiba J, Kage M. Salivary duct carcinoma cytologically diagnosed distinctly from salivary gland carcinomas with squamous differentiation. Diagn Cytopathol. 2008;36:485-493.
163 Batsakis JG, El-Naggar AK, Luna MA. ‘Adenocarcinoma, not otherwise specified’: a diminishing group of salivary carcinomas. Ann. Otol Rhinol Laryngol. 1992;101:102-104.
164 Klijanienko J, Vielh P. Fine needle sampling of salivary gland lesions VI. Cytological review of 44 cases of primary salivary squamous-cell carcinoma with histologic correlation. Diagn Cytopathol. 1998;18:174-178.
165 Klijanienko J, Lagacé R, Servois V, et al. Fine-needle sampling of primary neuroendocrine carcinomas of salivary glands: cyto-histological correlations and clinical analysis. Diagn Cytopathol. 2001;24:163-166.
166 Torlakovic E, Ames ED, Manivel JC, Stanley MW. Benign and malignant neoplasms of myoepithelial cells: cytologic findings. Diagn. Cytopathol. 1993;9:655-660.
167 Dipalma S, Alasio L, Pilotti S. Fine needle aspiration appearances of malignant myoepithelioma of the parotid gland. Cytopathology. 1996;7:357-365.
168 Chhieng DC, Paulino AF. Cytology of myoepithelial carcinoma of the salivary gland. A study of four cases. Cancer (Cancer Cytopathol). 2002;96:32-36.
169 Harrison RF, Smallman LA, Young JA, Watkinson JC. Oncocytic carcinoma of the parotid gland: a problem in fine needle aspiration diagnosis. Cytopathology. 1995;6:54-58.
170 Wu HHJ, Silvernagel SW. Fine-needle aspiration cytology of an oncocytic carcinoma of the submandibular gland. Diagn. Cytopathol. 1998;19:186-189.
171 Croce A, Moretti A, Bianchedi M, et al. Carcinoma in ectopic Warthin’s tumor: a case study and review of the literature. Acta Otorhinolaryngol Ital. 1996;16:543-549.
172 Mandreker S, Pinto RW, Usgaonkar U. Sebaceous carcinoma of the eyelid with metastasis to the parotid region. Diagnosis by fine needle aspiration cytology. Acta Cytol. 1997;41:1636-1637.
173 Moore JG, Bocklage T. Fine-needle aspiration biopsy of large-cell undifferentiated carcinoma of the salivary glands: presentation of two cases, literature review, and differential cytodiagnosis of high-grade salivary gland malignancies. Diagn Cytopathol. 1998;19:44-50.
174 Safneck JR, Ravinsky E, Yadzi HM, et al. Fine needle aspiration biopsy findings in lymphoepithelial carcinoma of salivary gland. Acta Cytol. 1997;41:1023-1030.
175 Lussier C, Klijanienko J, Vielh P. Fine-needle sampling of metastatic non-lymphomatous tumors to the major salivary glands. A clinico-pathologic study of 40 cases cytologically diagnosed and histologically correlated. Cancer. 2000;90:350-356.
176 Zhang C, Cohen J-M, Cangiarella JF, et al. Fine-needle aspiration of secondary neoplasms involving the salivary glands. A report of 36 cases. Am J Clin Pathol. 2000;113:21-28.
177 Chhieng DC, Cangiarella JF, Cohen J-M. Fine-needle aspiration cytology of lymphoproliferative lesions involving the major salivary glands. Am J Clin Pathol. 2000;13:563-571.