CHAPTER 12 Kidney, adrenal and retroperitoneum proper
Clinical aspects
The place of FNAC in the investigative sequence
By the 1960s, Scandinavian workers had already demonstrated the value of FNAC in the diagnosis of solid renal masses.1-3 Since then, several series of kidney tumors investigated by FNAC have been published, reporting good results.4-8 Two recent developments have had an important influence on the practice of FNAC in this area. Advances in radiological imaging technique, particularly computed tomography (CT), have considerably increased diagnostic accuracy, reducing the need for a preoperative tissue diagnosis by needle biopsy. On the other hand, the revised classification of renal tumors and the definition of new subtypes with different behavior and prognosis have produced a greater need for specific preoperative tumor typing to allow more conservative surgical treatment in selected cases.9 Immunophenotyping and cytogenetics play an increasing role in this context, and FNB is an ideal, minimally invasive way of providing cell material for such studies. The most rational approach to diagnosis seems to be the combination of radiological imaging and FNB in the same session.10
Generally accepted indications for FNB of solid renal masses are:
Puncture and aspiration through a thin needle has been the standard procedure to confirm simple renal cysts. Today, the diagnostic accuracy of ultrasound (US) examination has rendered cytological confirmation of simple cysts unnecessary, whereas FNB still has a place in the investigation of complex cysts such as multicystic nephroma and cystic renal cell carcinoma. Material from a renal abscess for microbiological investigation can be obtained by FNB.
For a review of the indications and usefulness of FNAC of lesions in the kidney based on current experience, see Puttaswamy et al.15
With regard to the adrenal, FNAC has mainly been used to investigate lesions detected by abdominal CT in the preoperative work-up of potentially resectable lung tumors. Mass lesions found incidentally by upper abdominal CT examination, so-called ‘incidentalomas’, may be primary adrenal tumors, metastatic malignancies or non-neoplastic lesions, with an approximately equal probability of neoplasms being metastatic or primary.16,17 The efficacy of image-guided FNB in the diagnosis of adrenal lesions has been demonstrated in several studies.18-21 Appropriate radiological and biochemical investigations may reduce the need for biopsy.13
For tumors of the retroperitoneum proper, percutaneous image-guided FNB is a valuable supplement to preoperative radiological investigations. For example, the distinction between tumor recurrence and retroperitoneal fibrosis as the cause of ureteric obstruction in patients treated for cancer of this region is of great clinical importance.22,23 In advanced inoperable disease, a cytological diagnosis may be a sufficient basis for palliative radiotherapy or chemotherapy without the need for a formal surgical biopsy. Cytological diagnosis of primary soft tissue tumors of the retroperitoneum is difficult and a type-specific diagnosis may not be possible, but the exclusion of metastatic malignancy or lymphoma is of clinical value. If smears prove to be non-diagnostic on immediate checking, a core needle biopsy can be performed in the same session.
Abdominal and retroperitoneal lymphadenopathy are a common targets for image-guided FNB, to distinguish metastatic malignancy, malignant lymphoma and reactive lymphadenopathy.24,25 In metastatic malignancy the cytology often suggests the site and type of the primary tumor. FNB is a valuable supplement to lymphangiography or CT in the preoperative staging of urogenital cancer.26,27
FNAC in the investigation of renal, adrenal and retroperitoneal lesions in the paediatric age group is presented in Chapter 17.
Accuracy of diagnosis
As in other sites, diagnostic accuracy is highly dependent on representative and adequate samples and on expertly prepared smears. Diagnostic criteria are well established for the commonly occurring tumors, less so for unusual entities. If a type-specific diagnosis is not possible, a categorization of disease with a differential diagnosis as a guide to further investigation and management is still of clinical value. Histochemical, immunohistochemical, ultrastructural and cytogenetic examination of FNB samples can provide more precise information of the histogenesis and phenotype of the tumor cells. However, expensive supplementary techniques should be used selectively in order to avoid nonproductive increase in costs.
In a literature review of 1585 cystic and solid renal and adrenal masses, the false-positive rate was 2.3% if cases reported as suspicious of malignancy were included. The diagnostic sensitivity was 86% for 603 malignant tumors, specificity was 98% and the predictive value of a positive result was 96%.4 More recent series recorded similar levels of accuracy.5-1028 The commonest causes of false-negative diagnosis are insufficient diagnostic cells in cystic tumors, small tumors, or large tumors with extensive necrosis and hemorrhage. False-positive diagnoses have been recorded in angiomyolipoma, in inflammatory processes such as pyelonephritis with regenerative epithelial atypia, and in infarcts.29,30
Large series of FNB of adrenal lesions have been reported, but histologic correlation is often lacking. Only two incorrect diagnoses were made in 81 primary adrenal lesions reported from M. D. Anderson Cancer Center.17 Others have reported similar results and a 100% specificity for the diagnosis of malignancy.18-2031 Tumor size is an important parameter in the diagnosis of adrenal tumors, underlining the importance of clinical and radiological correlation.32
The accuracy of image-guided FNB of abnormal retroperitoneal lymph nodes has been analyzed in the context of staging urogenital cancer.23,33 FNB may reveal metastatic involvement of nodes that appear radiologically normal.
Complications and contraindications
The complications of puncture of renal cysts were analyzed in a large number of cases accumulated from several institutions in the USA by Lang in 1977.34 Hemorrhage, infection and pneumothorax did occur after puncture, but the rate was low and was reduced by experience and technical modifications. Complications have been recorded in about 0.4% of FNB of solid renal masses.4,27 A few cases of tumor seeding in the needle track following FNB of renal cell carcinoma have been reported.28,35 The risk is considered higher for transitional cell carcinoma.28,36 Needle size is not always specified in the reports and some involved core needle biopsy.37 This serious complication is extremely rare but should not be ignored.38 Infarction of renal cell carcinoma following FNA has been reported.39
There is a documented risk of causing a hypertensive crisis by needling adrenal pheochromocytoma.40,41 The expertise and facilities to deal with such an event must therefore be immediately available at the procedure, which should only be performed in major hospitals.42
Aortic or other arterial aneurysm must obviously be excluded before an abdominal/retroperitoneal mass is subjected to FNB. An aneurysm may be occluded by organizing thrombus, may not pulsate and may appear solid on US examination.
Technical considerations
The biopsy technique is described in detail in Chapters 2 and 3. Image guidance is the routine, even in palpable masses, to ensure representative samples avoiding necrosis, cystic change, hemorrhage and major vessels. The parallel use of air-dried, MGG/Diff-Quik-stained and wet-fixed Pap-stained smears is recommended. Cell blocks are often of great value and are better suited for immunohistochemical studies than smears. The pros and cons of FNB and core needle biopsy (CNB) in renal tumors have been discussed in a review by Volpe et al.,28 concluding that the methods are complementary. We feel that FNB, correctly carried out, is less traumatic and less costly than CNB and should be the first line of investigation. Immediate checking of samples during the procedure allows the selective use of CNB.
Cytological findings
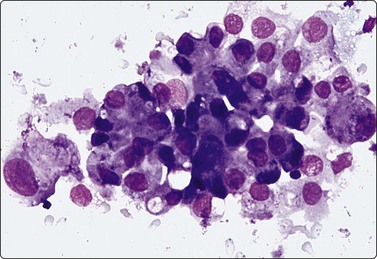
Normal structures; cortical pseudotumor (Figs 12.1 and 12.2)3
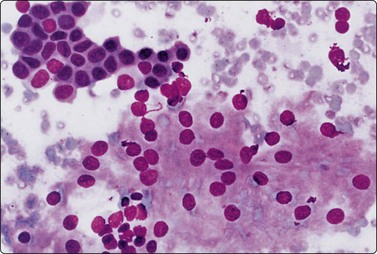
Fig. 12.1 Kidney, normal cortical tissue
Sheet of large cells with abundant granular cytoplasm, invisible cell borders and small dark nuclei, representing proximal convoluted tubule, and sheet of smaller epithelial cells with dense cytoplasm from distal tubule (upper left) (MGG, HP).
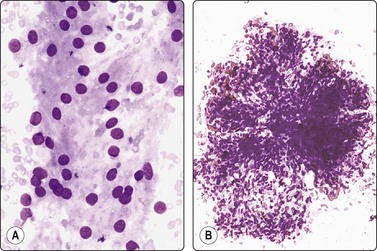
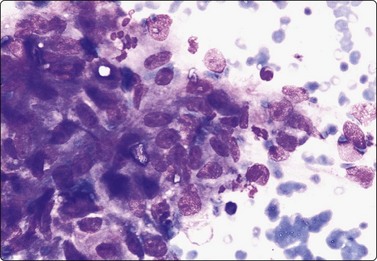
Fig. 12.2 Kidney, normal cortical tissue
(A) Monolayered sheet of cells from proximal convoluted tubule (MGG, HP); (B) Glomerulus with some separation of lobules (MGG, LP).
Non-neoplastic epithelial cells and other components of renal cortical tissue may be sampled by FNB in three different ways: (1) The needle may inadvertently sample normal tissue adjacent to the target; (2) focal hyperplasia of renal cortical tissue, either as a mass at the convexity (cortical pseudotumor) or as a rounded expansion of a medullary pyramid (inversion of renal lobule or lobular dysmorphism), may mimic an avascular solid tumor radiologically; (3) an ectopic kidney may be radiologically mistaken for a neoplasm.43 Smears from non-neoplastic renal tissue may be surprisingly cellular, suggestive of a low-grade renal cell tumor, a possible pitfall for the inexperienced observer. The hallmark of non-neoplastic renal cortical tissue is the coexistence in smears of tubular epithelial cells from different parts of the nephron looking distinctly different. Glomeruli are not always found.
Large epithelial cells from the proximal convoluted tubules usually dominate the smears. The cytoplasm is abundant, pale eosinophilic (gray–violet in MGG), finely granular with indistinct cell borders. Nuclei are round, central, relatively small and uniform, with small indistinct nucleoli. Cytoplasmic vacuolation is not a feature of normal tubular epithelial cells. Single cells and stripped nuclei may be seen but most of the cells form monolayered sheets. The nuclei are often arranged in rows. Cells of intermediate size, from the distal convoluted tubules, also lack distinct cell borders. The smaller tubular epithelial cells from the loop of Henle and from the collecting tubules have scanty, dense cytoplasm and distinct cell borders and form monolayered sheets or short tubular segments (Fig. 12.1). Some of the cells contain coarse, dark, cytoplasmic granules, probably lipofuscin. Aggregates of tubular epithelial cells may include strands of pink (MGG) hyaline material, contributing to the resemblance to renal cell tumors. Glomeruli are seen as rounded tissue fragments of tightly cohesive, small, spindly endothelial cells with indistinct cytoplasm and strands of stroma. They have a lobulated shape similar to glomeruli in tissue sections, but more or less distorted by smearing (Fig. 12.2).44
Benign and inflammatory conditions
Renal cysts (Fig. 12.3)
The diagnosis of simple renal cyst is usually made by US examination alone and cytological confirmation is not often necessary, but the aspirated fluid is often routinely submitted for cytological examination. The fluid is typically thin, clear, containing small numbers of degenerate epithelial cells and macrophages, but the number of cells can occasionally be surprisingly large. Macrophages in air-dried smears may appear atypical with moderately enlarged irregular nuclei, and may be clustered (Fig. 12.3).45 This could raise a suspicion of a cystic tumor, but if the radiological features are typical of a cyst and if the aspirated fluid is clear, there is no cause for concern. On the other hand, if the aspirate contains old blood and/or necrotic debris, a cystic neoplasm has to be excluded by further investigations, even if no tumor cells are found in the smears. Carcinoma within the wall of an apparently solitary simple cyst is a rare but well-recognized event in which the malignant component can be missed by needle biopsy.46,47 Accurate radiological guidance is essential.
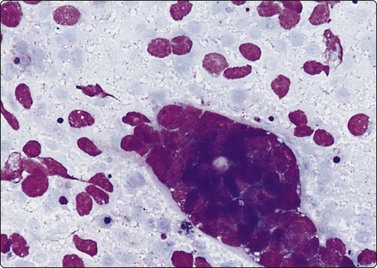
Cystic nephroma (multilocular renal cyst) (Figs.12.4, 12.5)
Cystic nephroma is most common in children and is related to Wilms’ tumor, but also occurs in adults. In adults, tumors of this type are now often reclassified as other entities unrelated to nephroblastoma. It is included here as a cystic lesion of the kidney. Cytological findings have been reported in a number of single cases.48-51 Aspiration yields mainly cyst fluid. The cell content of the fluid is fairly low, but the cells often appear atypical and may be mistaken for cystic renal cell carcinoma. The atypical cells, which are both clustered and single, represent the hobnail type of epithelial cells lining the cystic spaces (Fig. 12.4). In one reported paediatric case, the cytology showed features mimicking a malignant small round cell tumor.52 The cystic spaces are separated by septa of solid connective tissue, which is usually densely fibrous with no blastema and which does not contribute cells to the smears.
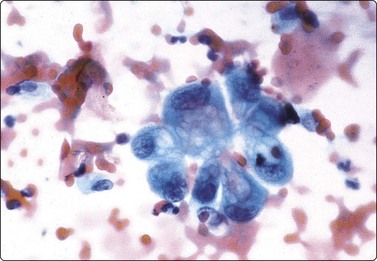
Small cluster of histiocyte-like cells with large, eccentric, pleomorphic nuclei representing the ‘hob-nail’ epithelial cells lining cystic spaces (Pap, HP)
(Courtesy of Dr. B. H. Coombes, Southport, Queensland).
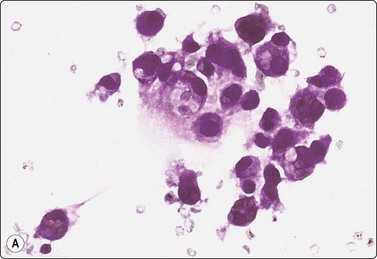
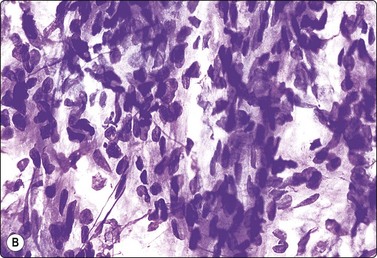
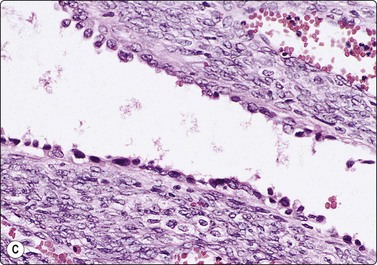
(A) Loose cluster of cells, which could be either macrophages or atypical, degenerate epithelial cells; cyst fluid; (B) Cellular tissue fragment of spindle cells with bland nuclei; smear from a solid part of the same lesion (MGG, HP); (C) Corresponding tissue section; spaces lined by ‘hob-nail’ epithelial cells and septae of cellular mesenchymal tissue (H&E, IP).
In one of our adult cases, smears contained highly cellular tissue fragments of spindle cells in addition to epithelial cells of hobnail type (Fig. 12.5B). The histological counterpart was multifocal thickening of cyst walls and septa by cellular stroma of spindle cells reminiscent of ovarian cortex. The possibility of a mixed epithelial and stromal tumor of the kidney,53 a recently defined entity that was previously regarded as adult mesoblastic nephroma or as a variant of cystic nephroma with cellular stroma, was therefore considered. However, this lesion should also contain an epithelial component of immature tubules, which was not present in our case. Morgan and Greenberg reported a similar case with spindle cells of smooth muscle type, incorrectly diagnosed as angiomyolipoma.50
Abscess
The aspirate is purulent and smears show mainly degenerating polymorphs and macrophages. A few normal or degenerating tubular epithelial cells may be present. Part of the aspirate should be submitted for culture.
Xanthogranulomatous pyelonephritis3,54
Macrophages in FNB smears from xanthogranulomatous pyelonephritis can be numerous and look quite atypical, and can be mistaken for malignancy. They have a vacuolated cytoplasm and the nuclei appear enlarged and irregular, particularly in air-dried smears. Chronic inflammatory processes involving adipose tissue in other sites in the retroperitoneum can show a similar cytological pattern (Fig. 12.6). The clinical and radiological findings, the generally inflammatory character of the smear, and the recognition of the ‘atypical’ cells as histiocytes (easier in alcohol-fixed smears) point to the correct diagnosis.
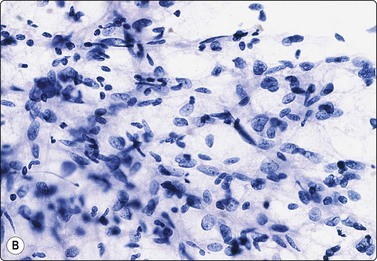
Angiomyolipoma of kidney (Figs 12.7-12.11)55-58
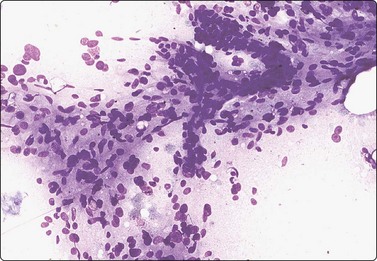
Tissue fragment of spindle cells with abundant cytoplasm and indistinct cell borders; a small branching blood vessel; fat droplets (MGG, IP).
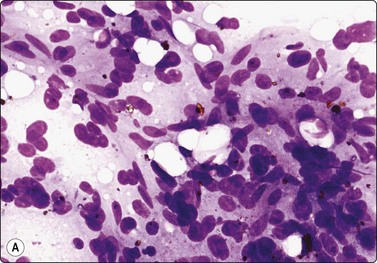
(A) Loosely clustered spindle cells with abundant cytoplasm and indistinct cell borders; plump spindled or rounded nuclei; some anisokaryosis but bland chromatin; many fat droplets (MGG, HP); (B) Similar appearances in Pap-stained smear (HP).
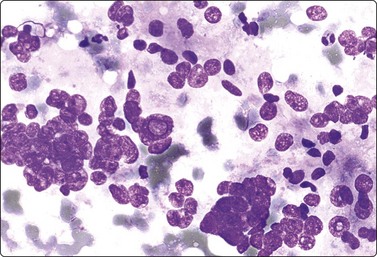
Fig. 12.9 Angiomyolipoma (atypical)
Cellular smear of clustered cells with mainly round, moderately enlarged and pleomorphic nuclei; some intranuclear inclusions; a small number of usual bland spindle cells (MGG, HP). Conservative surgical resection confirmed benign angiomyolipoma.
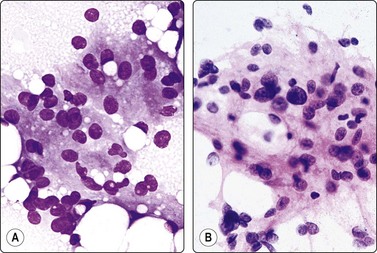
Fig. 12.10 Angiomyolipoma (round-cell epithelioid pattern)
The round nuclei and the abundant cytoplasm with many vacuoles convey a resemblance to renal cell carcinoma, but the cytoplasm is syncytial, eosinophilic, non-granular, there are many large lipid droplets, nuclei are bland and some are spindle-shaped (A, MGG; B, H&E, HP).
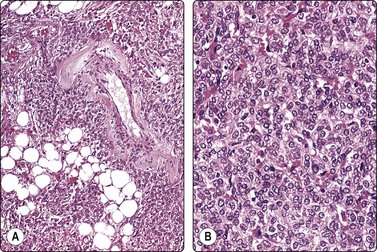
Tissue sections; same case as Fig. 12.10. (A) Typical pattern (H&E, IP); (B) Round cell pattern (H&E, HP).
Conservative surgical intervention may be required to control hematuria in angiomyolipoma, but nephrectomy is generally not indicated. A definitive diagnosis by FNB may therefore be important.
The cytological findings are fairly characteristic. Aspirates tend to be bloody but contain a variable number of tissue fragments, syncytial cell clusters and single cells. The cells have elongated or spindle-shaped nuclei, some with truncated ends. Their size and shape are moderately variable. Nuclear chromatin is bland and mitoses are rare. The abundant fragile eosinophilic cytoplasm appears as a background to the nuclei, and cell borders are generally not visible (Fig. 12.8). Large fat vacuoles and some adipocytes are intimately associated with the spindle cells. Strands of endothelial cells and short segments of small vessels are often, but not always present in smears (Fig. 12.7). Tallada et al. found vascular structures in only one of four cases.56
Some angiomyolipomas include highly cellular areas composed predominantly of epithelioid cells of leiomyoblastic type with rounded nuclei (Figs 12.10 and 12.11).59 Nuclear enlargement, anisokaryosis and hyperchromasia of moderate degree may be present and be mistaken for a low-grade renal cell tumor. Several false-positive diagnoses have been reported in the literature.4,5,30,55 However, the cytoplasm of smooth muscle cells is not granular or vacuolated but pale eosinophilic, and the nuclear chromatin is bland. Some typical spindle-shaped nuclei can usually be found also in predominantly round cell areas. A background of fat droplets and adipocytes is usually seen in angiomyolipoma, whereas strands of basement membrane material with adhering tumor cells are characteristic of renal cell tumors. Prominent nuclear atypia and pleomorphism can occur also in angiomyolipoma of the usual spindle cell type raising a suspicion of malignancy (Fig. 12.9). Correlation with clinical and radiological findings and immunostaining (EMA, SMA, HMB-45) is helpful in atypical cases.55
Heavy admixture with blood is common and can make a cytological diagnosis difficult. In such cases, a cell block preparation may contain diagnostic tissue fragments including a vascular component.60 CNB has been successfully used in some cases to confirm the diagnosis.
Renal cell tumors4,5,9,18,61,62
The current classification of renal cell tumors (WHO 2004) includes the following entities:
Sarcomatoid change can occur in any of the types and is not classified as a separate entity. The granular type is included with the clear cell tumors.
Clear cell renal cell carcinoma (Figs. 12.12-12.16)
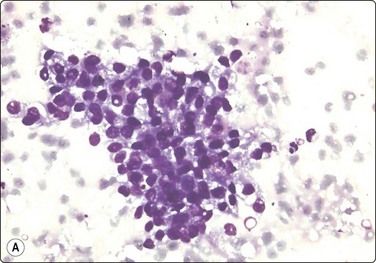
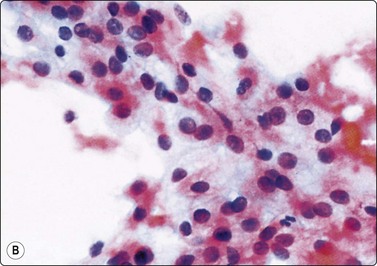
Fig. 12.12 Low-grade renal cell carcinoma, clear cell type
Loosely cohesive cell clusters; abundant fragile, vacuolated cytoplasm, relatively uniform small nuclei; inconspicuous nucleoli; stromal material not obvious in this example but there are a few endothelial cells (A, DQ, HP; B, Pap, HP).
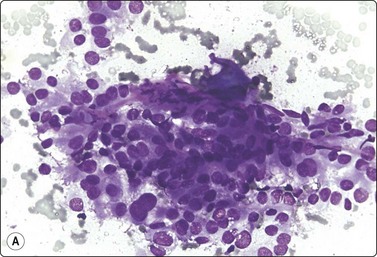
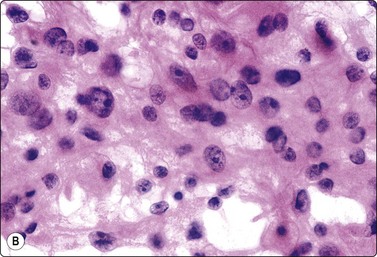
Fig. 12.13 Intermediate-grade renal cell carcinoma, clear cell type
Poorly cohesive cells with abundant fragile vacuolated and granular cytoplasm; indistinct cell borders; strands of stroma and some endothelial cells; moderate nuclear enlargement and anisokaryosis. Nucleolar prominence best seen in H&E (A, DQ; B, H&E, HP).
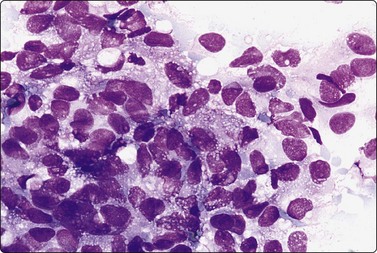
Fig. 12.14 High-grade renal cell carcinoma, clear cell type
Tissue fragment of pleomorphic malignant cells; variable N:C ratio; some cells with abundant finely vacuolated cytoplasm (MGG, HP).
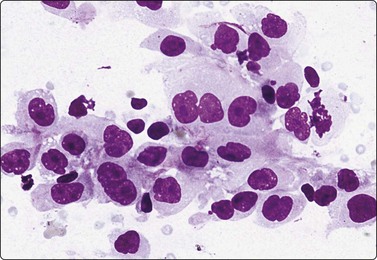
Fig. 12.15 Metastatic, high-grade renal cell carcinoma
Cluster of poorly cohesive large cells; abundant pale cytoplasm with some vacuoles; relatively distinct cell borders; large pleomorphic nuclei; prominent nucleoli; mitotic figure (MGG, HP).
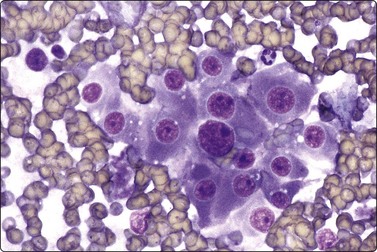
Fig. 12.16 Renal cell carcinoma, granular cell pattern
Cluster of epithelial cells with abundant, dense, finely granular cytoplasm, malignant nuclear features (MGG, HP).
Criteria for diagnosis
About three-quarters of renal cell tumors (RCC) are histologically of the classic clear cell type. Although the clear cell appearance is not well reproduced in cytological preparations, the findings in low- and intermediate-grade tumors are fairly characteristic. Cells have abundant, pale eosinophilic, granular and vacuolated cytoplasm, and cell borders are generally distinct. The cells are relatively cohesive, forming sheets and solid, trabecular or pseudopapillary aggregates. There are variable numbers of single cells and stripped nuclei due to cytoplasmic fragility. Cell cohesion is reduced and single cells are more common in less well-differentiated tumors. The cells are polygonal and have a low nuclear:cytoplasmic ratio. Nuclei are rounded, relatively small and uniform in low-grade tumors (Fig. 12.12), and are enlarged and of variable size, shape and chromatin pattern in intermediate and high-grade tumors (Fig. 12.13). Nucleoli are hardly visible in low-grade tumors, and are large to very large in high-grade tumors. The pattern is less characteristic at the poorly differentiated end of the spectrum, but some cells with abundant pale vacuolated cytoplasm can usually be found (Fig. 12.14). A characteristic feature in smears is tumor cells adhering to strands of stromal material staining pink with MGG, probably derived from the wall of sinusoidal blood vessels (Fig. 12.13). This pseudopapillary pattern must not be mistaken for true papillae. Intranuclear cytoplasmic inclusions can be found in about one-third of RCCs. Intracytoplasmic hyaline eosinophilic globules have been observed but can be found also in large cell carcinomas of other sites.63 Finally, fresh and altered blood, necrotic material and foamy or hemosiderin-containing macrophages are commonly present in the background.
The cytology of RCC, at least of low or intermediate grade, is usually characteristic enough to be recognized also in FNB samples from metastatic sites (Fig. 12.15).64,65 Co-expression of cytokeratin and vimentin by the tumor cells, and positive staining for RCC antigen and CD10, although not specific, can be helpful in the identification of unsuspected metastatic RCC.66,67 Higher specificity has been claimed for two new markers, PAX-2 and H2AX.68
Malignant cells of granular cell type are most commonly seen focally in clear cell carcinoma but occur also in other types of RCC. A separate granular cell variant is no longer recognized. Cells of this type usually have high-grade nuclear morphology. The cytoplasm is abundant, but eosinophilic and finely granular rather than vacuolated (Fig. 12.16).69
Multilocular cystic renal cell carcinoma is a rare subtype of clear cell carcinoma with a generally very good prognosis.70 Conservative, nephron-sparing surgery may be suitable for this tumor, but to our knowledge the potential of preoperative diagnosis by needle biopsy has not yet been documented.
Papillary renal cell carcinoma
Criteria for diagnosis
Ten to fifteen percent of RCCs are of the papillary type (Figs 12.17 and 12.18).71,72 The cells of the type 1 variant are small with a small amount of dense cytoplasm showing no obvious vacuolation or granularity. Nuclei are small, uniform and bland-looking, and nucleoli are inconspicuous. Intranuclear vacuoles occur. True papillary fragments are often prominent in smears. Psammoma bodies are commonly found. Numerous vacuolated macrophages are a characteristic component when present and intracytoplasmic hemosiderin is often seen.73,74 Distinction of type 1 from renal cortical adenoma is mainly based on size. Metastases have been observed from tumors as small as 1 cm75 and tumors larger than 5 mm are now classified as RCC. A conservative approach and radiological follow-up may be an option in very small, incidentally discovered tumors, but if FNB findings are of a clear cell pattern or high nuclear grade the tumor should probably be regarded as RCC regardless of size. Metanephric adenoma also enters the differential diagnosis (see below).
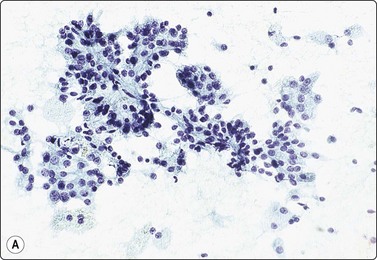
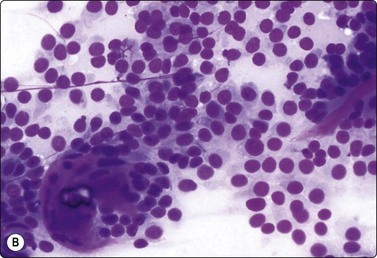
Fig. 12.17 Renal cell carcinoma, papillary type
(A) Cell clusters and papillary fragments; small uniform nuclei; moderate amount of pale cytoplasm; some of the single cells are foamy macrophages (Pap, IP); (B) Poorly cohesive small cells with round uniform nuclei; stromal fragments from disrupted papillae; psammoma body lower left (DQ, HP).
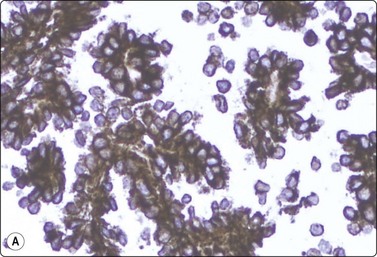
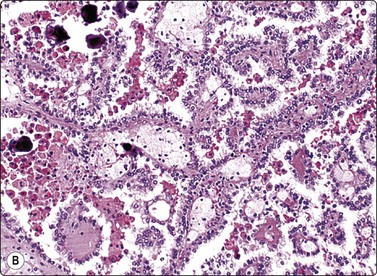
Fig. 12.18 Renal cell carcinoma, papillary type
(A) Tumor cells strongly positive for CK7; same case as Fig. 12.17 (immunostaining, HP); (B) Tissue section from same case. Papillary pattern; interstitial foam cells; psammoma bodies (H&E, LP).
Papillary RCC type 2 can be mistaken for clear cell carcinoma due to higher nuclear grade and more abundant eosinophilic cytoplasm. It is CK7+ in only a small proportion of cases whereas most type 1 tumors are strongly positive. EMA is usually negative in type 2, positive in type 1 and in clear cell RCC. Prognosis is generally less favorable than for type 1 but varies with tumor size and nuclear grade.
Chromophobe renal cell carcinoma
Criteria for diagnosis
Five percent of RCCs are of the chromophobe type (Figs 12.19 and 12.20). Smears of chromophobe RCC are cellular, showing both single cells and small sheets. There is a conspicuous variation in the size of the cells. Most cells are large with abundant, vacuolated cytoplasm of a flocculent texture. The cytoplasmic density varies; it is dense at the periphery and pale in the center, like a perinuclear halo. Cell membranes are thick. There are also groups of smaller cells with dense cytoplasm, giving an impression of a dual population. There is moderate anisokaryosis, but the nuclear:cytoplasmic ratio is consistently low. Nuclei are moderately hyperchromatic and nucleoli are small. Nuclear grade is mainly 2. The nuclear membranes have been described as irregular and ‘raisinoid’. Binucleation is a common feature, and small nuclear inclusions occur.76-78
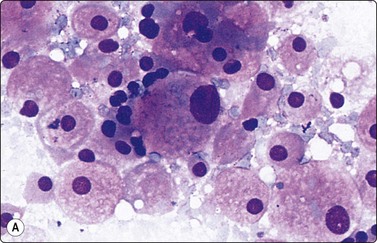
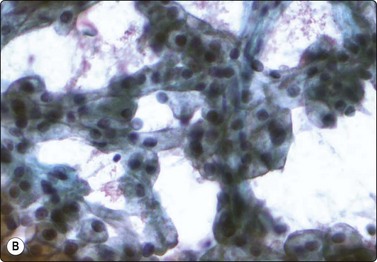
Fig. 12.19 Renal cell carcinoma, chromophobe type
Clustered cells, majority with abundant granular and vacuolated cytoplasm, some much smaller cells; moderate anisokaryosis, some cells with small nucleoli. Cytoplasm condensed peripherally, perinuclear pale area, best seen in Pap (A, MGG; B, Pap, HP).
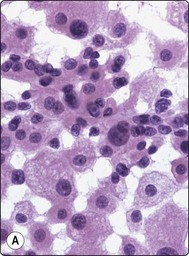
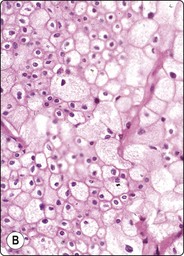
Fig. 12.20 Renal cell carcinoma, chromophobe type
(A) A dual population of large and smaller cells more obvious in this example (H&E, HP); (B) Corresponding tissue section (H&E, IP).
Renal oncocytoma is the main differential diagnosis in FNB smears. Cells of oncocytoma have dense granular eosinophilic cytoplasm and small bland nuclei of uniform size.79 Immune markers are helpful in this distinction.80,81
Uncommon variants of renal cell carcinoma
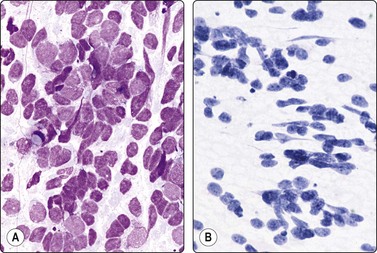
Sarcomatoid renal cell carcinoma is no longer distinguished as a subtype since this pattern can occur focally in any type of RCC. The tumor tissue is poorly differentiated of highly pleomorphic, anaplastic and often with spindle-shaped cells (see Fig. 12.21). This pattern, which resembles a pleomorphic sarcoma, is seen focally in 1.5% of RCC and suggests a poor prognosis.82,83
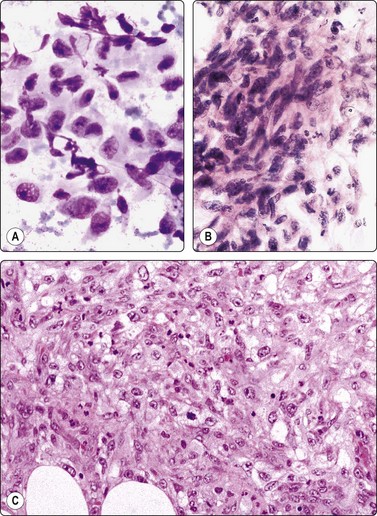
Fig. 12.21 Renal cell carcinoma, sarcomatoid type
Moderately cohesive, pleomorphic spindle cells with malignant nuclear features (A, MGG; B, H&E; HP); (C) Corresponding tissue section (H&E, IP).
The collecting duct type of renal carcinoma is rare. Smears show small groups and single cells of glandular or intermediate type with malignant nuclear features. A tubular or papillary arrangement may be present. Fragments of fibrous stroma may reflect desmoplasia seen in tissue sections (Fig. 12.22). Metastatic carcinoma, papillary RCC and TCC should be included in the differential diagnosis.84-86
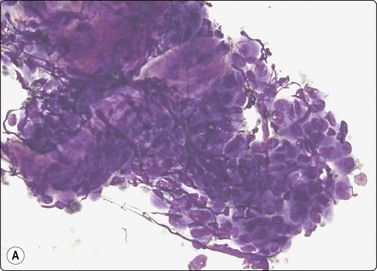
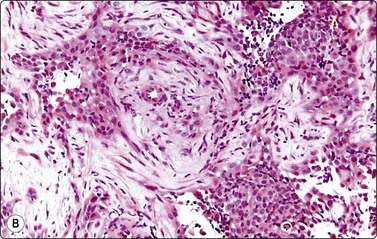
Fig. 12.22 Collecting duct RCC
(A) Tissue fragment of obviously malignant epithelial cells resembling TCC; note stromal fragments representing desmoplasia (DQ, HP); (B) Corresponding tissue section (CNB, H&E, IP).
A few cases of FNB of the rare medullary carcinoma of the kidney have been reported. Distinction from other types of high-grade carcinoma including RCC, TCC and collecting duct carcinoma is difficult by cytology and clinical correlation is important.87 The tumor occurs almost exclusively in patients with sickle cell hemoglobinopathy (trait).
Another entity, Xp11.2 translocation carcinoma, has recently been separated from clear cell RCC on the basis of cytogenetic findings. It is most common in children but also occurs in adult patients and has a very aggressive behavior.88 The histological features are a mixed pattern of clear cell and papillary components, prominent vascularity and in some cases hyaline globules and psammoma bodies. Positive nuclear staining for TFE3 has been reported. These features may be reflected in FNB smears as shown by a few single case reports.89
Mucinous tubular and spindle cell carcinoma is also a recently defined variant of RCC but of low grade. A preoperative diagnosis may be important in view of the good prognosis. Only single case reports of the cytology are available and criteria have yet to be defined.90,91
Grading of renal cell carcinoma
Histological grading of all variants of RCC based on nuclear and nucleolar size, nuclear irregularity, chromatin pattern and mitotic activity has been found to be a useful indicator of prognosis.92 Cytological nuclear grading on similar principles correlates well with histological grade as demonstrated in several reported series.1,62,93,94 There may be a tendency for the cytological grade to be lower than the histological, probably because of limited sampling.
Renal oncocytoma (Fig. 12.23)
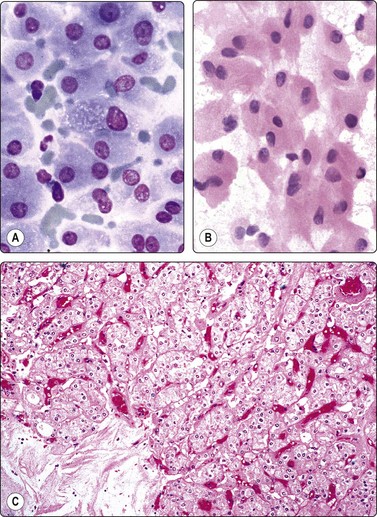
Uniform population of epithelial cells with bland nuclei and abundant, eosinophilic, finely granular cytoplasm; distinct cell borders (A, MGG; B, H&E, HP); (C) Corresponding tissue section. Note central scar lower left (H&E, IP).
Renal oncocytoma should be separated from low-grade RCC because of its favorable prognosis.95 The main differential diagnosis is low-grade clear cell carcinoma and chromophobe carcinoma. The cells of oncocytoma are uniformly of oncocytic type and have abundant dense, finely granular, eosinophilic cytoplasm and small bland nuclei with inconspicuous nucleoli. Cell borders are distinct.96,97 An oncocytic pattern may occur focally in RCC, and selective sampling of a heterogenous tumor may result in an erroneous diagnosis. A confident diagnosis of renal oncocytoma should therefore probably not be based on cytology alone, but should take clinical and radiological features such as a well-circumscribed lesion, a central scar and low vascularity into account. Immunocytochemistry is helpful,80,81,98 although reported results are somewhat inconsistent. Vimentin, RCC antigen and CK7 are negative in most cases, CD117 is positive. Hemorrhage, necrosis, mitotic figures and obvious nuclear atypia are not seen in oncocytoma and, if present, favor renal cell carcinoma.
Diagnostic cell material can be difficult to obtain in cystic tumors and in large tumors with extensive necrosis or hemorrhage. Necrotic material aspirated from a renal mass should raise a suspicion of carcinoma even in the absence of intact neoplastic cells. Small tumors can be missed, and cells aspirated from adjacent non-neoplastic cortical tissue can be confusing (see p. 316).
Histiocytes in xanthogranulomatous pyelonephritis and other chronic inflammatory conditions can appear disturbingly atypical, particularly in air-dried MGG smears (see p. 317). Foamy histiocytes may be prominent in smears from some tumors, particularly papillary RCC. Silverman et al.29 have drawn attention to the regenerative epithelial atypia that may be seen in aspirates from renal infarcts.
The distinction from cellular angiomyolipoma with a predominance of round epithelioid cells can be difficult. The differential diagnosis is briefly discussed in the section on angiomyolipoma. The cytological pattern of renal oncocytoma has been described above. Metanephric adenoma is mentioned on page 325. The distinction of these benign tumors from low-grade RCC is obviously essential in view of the differences in prognosis and clinical management.
The cytological features of RCC may in some cases resemble endocrine tumors: poorly cohesive cells with abundant fragile cytoplasm and uniformly round nuclei, cells arranged in curved rows or loose follicles, prominent vascularity (Fig. 12.24). Adrenal cortical neoplasms are the main problem in view of the anatomical proximity to the kidney, but other tumors such as paraganglioma may also be considered. Immune marker studies (Inhibin, Melan A, RCC antigen, CD10, etc.) are helpful in this situation.
Other tumors of the kidney
Metanephric adenoma99-102
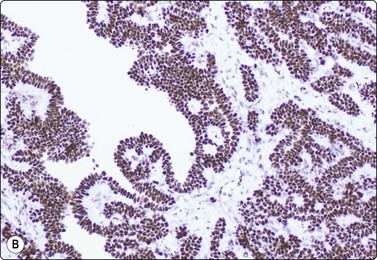
Metanephric (embryonal) adenoma is a rare benign renal tumor arising from metanephric blastema, most often seen in middle-aged women. A few cases with cytology have been reported. Smears are relatively cellular of monomorphous cells with small oval overlapping nuclei, bland chromatin and scanty cytoplasm. The cells are arranged in micropapillary or trabecular clusters and rosette-like, small tubular or microfollicular groups. The cell groups are associated with scanty stromal, basement membrane-like material sometimes forming a thin central core in small rounded clusters (Fig. 12.25). Figure 12.26 is of similar FNB findings from a solitary, well-circumscribed renal lesion in a 70-year-old woman, radiologically thought to be a cyst. It had been present for 15 years, showing no change. The cytology in conjunction with the clinical/radiological findings was consistent with metanephric adenoma, and surgery was not performed.
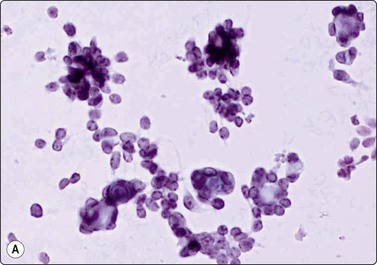
(A) Single and clustered cells forming small trabecular and follicular groups, some with hyaline stromal material in the center (Pap; HP); (B) Tissue section, immunostaining for WT1 (IP)
(Courtesy Dr. L. Francis, Brisbane).
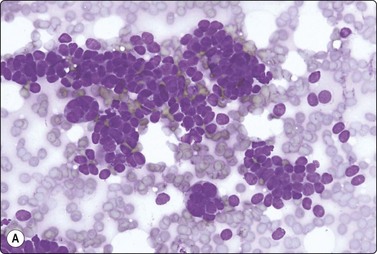
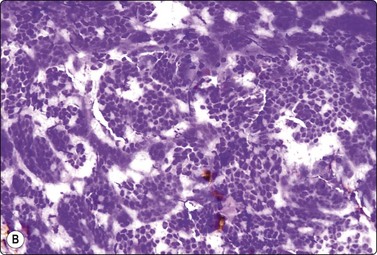
(A) Cellular smear of clusters of small cells with uniform oval bland nuclei and scanty cytoplasm, some cells forming small round tubular groups; (B) The same smear contained tissue fragments almost like a cell block, showing microarchitectural features of trabecular or tubular groups separated by stroma (A, DQ, HP; B, IP).
The main differential diagnoses are well-differentiated Wilms’ tumor and papillary RCC. Patel et al.100 propose a battery of immune markers (WT1, CD57, CD56, AMACR, CK7) to help in this distinction.
Wilms’ tumor (Figs 12.27 and 12.28)103-105
Wilms’ tumor (nephroblastoma) is predominantly a tumor of childhood and is dealt with in more detail in Chapter 17. However, it can also occur in adults. Smears are usually dominated by small, undifferentiated malignant cells representing blastema. A specific diagnosis requires in addition evidence of epithelial and mesenchymal differentiation, but this may only be obvious in the better-differentiated tumors. Individual cells generally appear undifferentiated. Epithelial differentiation is suggested by microarchitectural patterns such as rosettes, tubules or cords (Fig. 12.27), which are not always present in smears. Correlation with clinical and radiological findings is important. The diagnosis may be supported by ancillary studies. The results of immunostaining are variable, depending on differentiation. Useful markers are vimentin, LMWCK, EMA and WT1. Heterogeneous cytogenetic abnormalities can be demonstrated in a proportion of tumors.
The differential diagnosis with other malignant small round cell tumors of the abdominal organs, primary and metastatic, is also discussed in detail in Chapter 17. Ancillary techniques are usually necessary since distinguishing features in routine smears may be subtle. Nuclear molding is prominent in classic small cell anaplastic carcinoma, cytoplasm is minimal and nucleoli are absent. Tight cell clusters are not a common feature in lymphoma, lymphoid globules (round cytoplasmic fragments) are present in the background, and cells with more abundant pale-blue cytoplasm and a perinuclear pale zone are usually found. Rhabdomyoblasts with eccentric nuclei and a ‘tail’ of dense eosinophilic cytoplasm are indicative of rhabdomyosarcoma. Tumor cell rosettes with finely fibrillar material in the center (neuropil) suggest neuroblastoma. Ultrastructural characteristics are particularly helpful in this group of tumors and FNB samples can and should be used for EM examination, immunocytochemistry and cytogenetics to reach a confident, type-specific diagnosis.
Transitional cell carcinoma (Figs 12.29-12.31)106,107
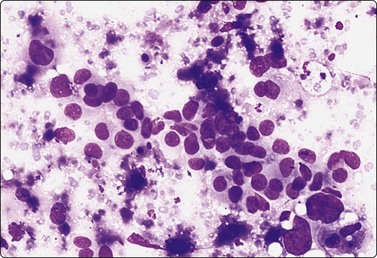
Fig. 12.29 Transitional cell carcinoma of renal pelvis
Loosely clustered malignant epithelial cells; pleomorphic hyperchromatic nuclei; moderate amount of dense cytoplasm; necrotic debris (MGG, HP).
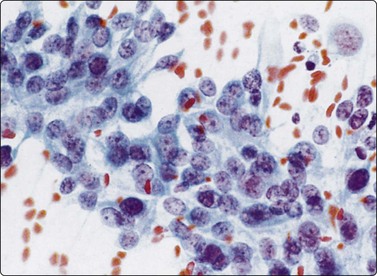
Fig. 12.30 Transitional cell carcinoma of renal pelvis
Poorly cohesive malignant cells with relatively high N:C ratio; pale cytoplasm; vesicular nuclei; prominent nucleoli (Pap, HP).
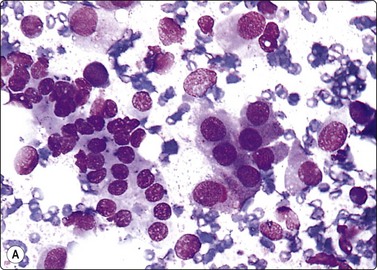
Fig. 12.31 Transitional cell carcinoma
(A) Mixed population of non-neoplastic tubular epithelium and malignant cells with dense cytoplasm and eccentric nuclei (MGG, HP); (B) Corresponding tissue section; carcinoma cells infiltrating between normal renal structures (H&E, IP).
Transitional cell carcinoma (TCC) developing from the epithelial lining of the renal pelvis involves the parenchyma by invasion and the malignant cells may therefore be intimately mixed with non-neoplastic tubular epithelial cells in FNB smears (Fig. 12.29). Most of the tumors subjected to FNB are solid and high grade, and true papillary structures are not often found. A tendency to squamous differentiation is not uncommon, some tumors have a spindle cell pattern and some are highly anaplastic and cannot easily be identified as of transitional cell origin. So-called ‘cercariform’ cells, which have long unipolar cytoplasmic processes with a thickening at the tail, are regarded as a clue to the recognition of TCC in smears.108,109
The distinction between RCC and TCC is not always easy. TCC sometimes has a clear cell appearance in tissue sections and correspondingly a pale vacuolated cytoplasm in smears. This is usually a focal phenomenon; the nuclear:cytoplasmic ratio is still relatively high and the nuclei are hyperchromatic with a coarse, irregular chromatin. Tumor cells are positive for CK7, usually also for CK20, high molecular weight cytokeratin and thrombomodulin, negative for RCC antigen and CD10.
Some uncommon primary renal tumors
Carcinoid tumors and small cell carcinoma of neuroendocrine type primary in the kidney are a rare occurrence, which has been associated with horseshoe kidney in several cases.110,111 A single case with FNB cytology has been reported.112 Juxtaglomerular cell tumors are diagnosed by clinical (hypertension) and radiological findings. A variety of mesenchymal tumors can also arise in the kidney or renal pelvis. The cytological findings in leiomyosarcoma of the kidney have been reported.113,114
Metastatic carcinoma and malignant lymphoma
Metastatic carcinoma is not uncommon in the kidney and may be solitary mimicking a primary tumor. This possibility should be kept in mind if there is a past history of extrarenal malignancy and must be considered if the cytological findings are not typical of a primary renal tumor. In the series reported by Gattuso et al. 21% of malignant renal tumors were metastatic.115 Giashuddin et al.116 reported eleven cases, all accurately diagnosed by FNB as metastatic to the kidney. A correct diagnosis is essential in view of the different management. Clinical and radiological data are usually helpful.
In the reverse situation, a patient presenting with metastatic deposits form an unknown primary, RCC is usually not difficult to recognize in FNB samples.64-67 This may be of clinical importance since metastatic RCC is often a solitary lesion that may be considered for surgical excision. Tumors of the thyroid, salivary glands and breast showing a clear cell or oncocytic pattern can cause differential diagnostic problems. Large pleomorphic nuclei with macronucleoli in cells with relatively abundant cytoplasm are seen in both high-grade RCC and in adrenal cortical carcinoma, hepatocellular carcinoma and large cell anaplastic carcinoma of lung or pancreas. Immunocytochemistry is helpful in this situation.68
Malignant lymphoma may involve the kidneys and may cause bilateral diffuse enlargement. It is rarely the first manifestation of systemic lymphoma but can occasionally be an unexpected finding in the investigation of a mass in the kidney by FNB.117
Non-neoplastic lesions
Adrenal hemorrhage is not always symptomatic and an old hematoma is sometimes discovered incidentally as a mass on abdominal CT scan. An adrenal hematoma can be either spontaneous or secondary to a neoplasm. This distinction may not be possible by FNB. Macrophages in a background of altered blood and debris can be difficult to distinguish from single degenerate neoplastic cells.
Fluid aspirated from adrenal cysts, most of which lack an epithelial lining, is usually acellular or contains only a few macrophages.118 Tuberculosis is a possible cause of adrenal enlargement. Epithelioid histiocytes, giant cells and granular necrotic material in smears suggest this diagnosis, to be confirmed by the demonstration of acid-fast bacilli.
Smears from myelolipoma, adrenal and extra-adrenal, show a characteristic mixture of fat droplets, adipocytes and hematopoietic cells. The combination of these features in this site is diagnostic.119 Single megakaryocytes can be mistaken for pleomorphic malignant cells but are the hallmark of hematopoietic tissue in any site.
Metastatic carcinoma
The confirmation or exclusion of metastatic carcinoma is the main indication for FNB of the adrenal (see p. 314). In this context, the primary is usually known and is most commonly in the lung. The cytological findings in metastatic tumors and clues to their origin are described elsewhere.
Primary adrenal cortical tumors17-19,120,121
Criteria for diagnosis
Most cortical adenomas subjected to FNB are asymptomatic lesions detected by abdominal CT in the preoperative investigation of patients with potentially resectable lung tumors, or incidentally by abdominal CT for other reasons (‘incidentaloma’). The main purpose of FNB is to exclude metastatic cancer. Smears are moderately cellular. The cells are poorly cohesive and appear single, in groups and in loose monolayered sheets without a distinctive architectural pattern. There are numerous stripped nuclei with a background of lipid droplets, but some cells have an abundant granular and vacuolated cytoplasm (Fig. 12.32). Lipofuscin pigment may be seen. The nuclei are generally small and uniformly round and have one or several small nucleoli, but anisokaryosis can be prominent in some tumors. Nuclear chromatin is granular and evenly distributed. Saboorian et al.17 emphasize the importance of precise positioning of the needle at biopsy since cells of an adenoma are not easily distinguished from those of normal adrenal cortex.
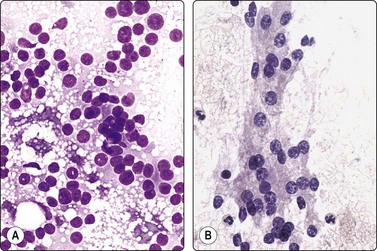
Fig. 12.32 Adrenal cortical adenoma
Sheets and single cells; small, round uniform nuclei; numerous small cytoplasmic lipid vacuoles (A, MGG; B, H&E, HP).
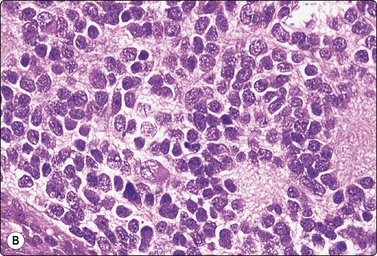
As in other endocrine tumors, the cytological pattern is not a good predictor of biological behavior and features overlap between adenoma and adrenal cortical carcinoma. Tumor size is an important parameter. In general, adenomas are less than 3–4 cm in diameter, carcinomas over 5 cm.21,121 Cells of well-differentiated adrenal cortical carcinoma to a variable degree resemble those of cortical adenoma, whereas cells of poorly differentiated carcinomas display severe nuclear pleomorphism, abnormal nuclear chromatin and very large nucleoli (Figs 12.34 and 12.35). Ren et al.,121 in an analysis of 20 cases, found high cellularity, necrosis, nuclear pleomorphism, mitoses and prominent nucleoli to be the most useful indicators of malignancy. Clinical, radiological and immunocytochemical findings must be correlated with the cytology.
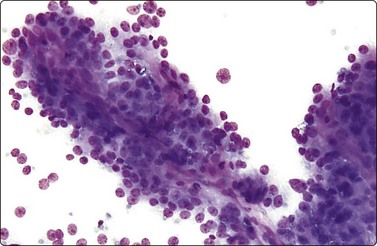
Fig. 12.34 Adrenal cortical carcinoma
Well-differentiated tumor; trabecular tissue fragment with prominent fibrovascular stromal cores; crowded nuclei; moderate anisokaryosis, some nucleolar prominence (DQ, IP).
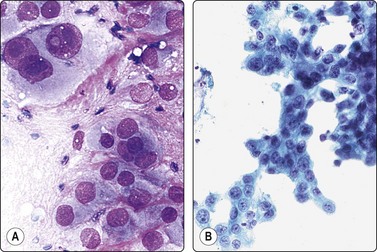
Fig. 12.35 Adrenal cortical carcinoma
(A) Poorly differentiated tumor. Obviously malignant cells with large pleomorphic nuclei and large nucleoli; resemblance to high-grade renal cell carcinoma or hepatocellular carcinoma (DQ, HP); (B) Moderately differentiated tumor; trabecular aggregates; moderate anisokaryosis, some nucleolar prominence (Pap, IP).
FNB findings in oncocytic variants of both adrenal cortical adenoma and adrenocortical carcinoma have been described.124,125 We have seen one example of oncocytic adrenal cortical adenoma, a large tumor of uncertain malignant potential, which was originally diagnosed by FNB as a renal cell tumor with a predominantly granular cell pattern (Fig. 12.33).
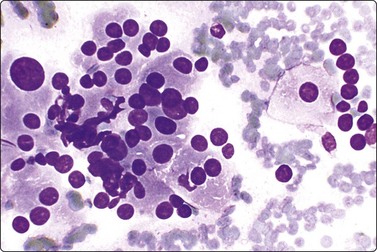
Fig. 12.33 Adrenal oncocytic adenoma
Loose clusters of epithelial cells with a suggestion of acinar groupings and rows of cells; prominent anisokaryosis; bland chromatin; abundant relatively dense oxyphil cytoplasm. Smears are from a large tumor of uncertain malignant potential. FNB initially reported as low-grade renal cell tumor (MGG, HP).
Distinction between adrenal cortical adenoma or low-grade carcinoma and low-grade renal cell tumor can be a problem.126 Both are composed of cells with abundant, vacuolated cytoplasm, a low nuclear:cytoplasmic ratio and small, round, bland nuclei. Vascular structures are prominent in both, but are of sinusoidal type in RCC, more like a network of capillaries with adherent tumor cells in adrenal tumors. Dispersal of cells, predominance of naked nuclei and discrete small lipid vacuoles favor cortical adenoma. Positive staining for Melan A (A103) is a good marker for adrenal cortical cells, both normal and neoplastic, and staining for inhibin and CD10 may also be helpful.122,123,127 Investigation of the vascular supply of the tumor by angiography, and histochemical and ultrastructural examination of the aspirate may be helpful in establishing a correct diagnosis.
Anaplastic carcinomas, both primary and metastatic, display severe nuclear pleomorphism, abnormal nuclear chromatin and very large nucleoli. Malignancy is obvious, but the origin of the tumor can be difficult to decide. Pheochromocytoma, metastatic melanoma, hepatocellular carcinoma and, of course, renal cell carcinoma need to be considered in the differential diagnosis. Some tumors have a spindle cell pattern that could be suggestive of a mesenchymal tumor.128
Distinction from pheochromocytoma and other neuroendocrine tumors is discussed below.
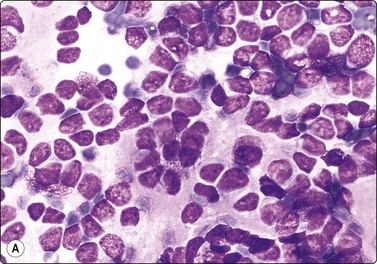
Pheochromocytoma (Figs 12.36, 12.37)128-131
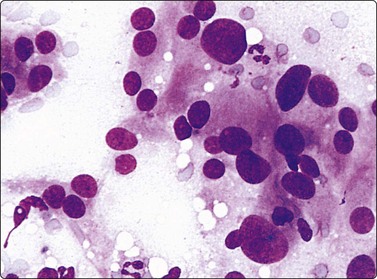
Loosely clustered cells with prominent anisokaryosis; nuclear chromatin uniformly granular; some large nucleoli; fragile, finely granular cytoplasm (MGG, HP).
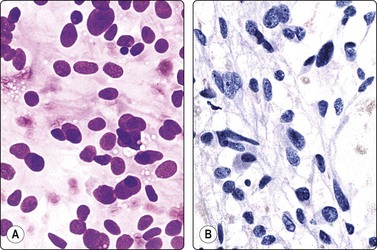
Mainly dispersed cells, rounded or spindled; anisokaryosis; bland chromatin, abundant fragile cytoplasm with anastomosing strands (A, MGG; B, Pap, IP).
Criteria for diagnosis
Needle biopsy of suspected pheochromocytoma is generally discouraged in view of the risk of precipitating a hypertensive crisis, but is not an absolute contraindication if necessary precautions are taken. In any case, since pheochromocytoma may be an unexpected finding and may occur in extra-adrenal sites, the cytopathologist must be familiar with its cytological pattern.
Smears are usually bloody but relatively cellular. Cells are poorly cohesive but may be arranged in a loosely acinar or trabecular pattern, or in semicircular rows, and associated with vascular elements. The cytoplasm is abundant and fragile, syncytial, without distinct cell borders, often with anastomosing strands creating a web-like background. There is a fine, red cytoplasmic granulation (MGG) in a proportion of cells. Nuclei vary in size and shape, round, oval or often spindled, sometimes bizarre. Anisokaryosis is typically prominent, seen as scattered very large nuclei in an otherwise relatively uniform population. Bi- or multinucleated cells are common. Nuclear chromatin is granular, evenly distributed and relatively uniform. Nucleoli are inconspicuous to prominent, single or multiple. Intranuclear inclusions are not uncommon, and intracytoplasmic hyaline globules (H&E) are found in some cases.
The pattern closely resembles that of medullary carcinoma of the thyroid, non-chromaffin paraganglioma and other neuroendocrine tumors. A case cytologically mimicking small cell anaplastic carcinoma has been reported.130 Immunostaining for neuroendocrine markers is helpful. A bloody aspirate can successfully be processed as a cell block to be used for this purpose.
Neuroblastoma (see Chapter 17)
The cytology of neuroblastoma, ganglioneuroblastoma and ganglioneuroma seen in FNB smears is described in Chapter 17, where several references are also given. The pattern is basically of a malignant small round cell tumor common to a range of neoplasms including small cell anaplastic carcinoma, Wilms’ tumor, embryonal rhabdomyosarcoma, etc. Characteristic rosettes of tumor cells, in the center of which there is finely fibrillar, pink staining (MGG) material (neuropil), are often found but are not specific (Fig. 12.38). As with other malignant small round cell tumors, a type-specific diagnosis should be supported by immunocytochemical, ultrastructural or other ancillary studies, which can be carried out on FNB samples (see p. 428).
The soft tissues of the retroperitoneum can give rise to a great variety of neoplasms, both benign and malignant. Liposarcoma, malignant fibrous histiocytoma and leiomyosarcoma are the most frequent malignant soft tissue tumors encountered in the retroperitoneum. Rhabdomyosarcoma is mainly seen in children and is most often of the embryonal type. Fibrosarcoma and neurogenic sarcoma are rare in this site. Among benign soft tissue tumors are the fibromatoses, xanthogranuloma, lipoma (much less common than liposarcoma), leiomyoma (rare), neurinoma, neurofibroma and angiomatous tumors. The cytological findings in soft tissue tumors are described and illustrated in Chapter 15. However, a few entities, which we have encountered mainly in the retroperitoneum, are included in this section.
Aneurysm
Needle biopsy is obviously contraindicated in case of arterial aneurysm. However, an aneurysm occluded by organizing old thrombus can be mistaken for a solid tumor clinically and radiologically and subjected to FNB. Smears of the semisolid aspirated material show altered blood, amorphous debris and a few macrophages with intracytoplasmic lipid droplets and blood pigment, similar to material aspirated from an old hematoma in any site. Degenerate cells and nuclei of uncertain nature may raise a suspicion of necrotic tumor. Further noninvasive investigations should be recommended.
Hemangiopericytoma (see also Chapter 15)
The cytology of hemangiopericytoma (most adult hemangiopericytomas may now be reclassified as solitary fibrous tumor) seen in FNB smears has been described. One example is illustrated in Figure 12.39 from a case presenting with a retroperitoneal mass.
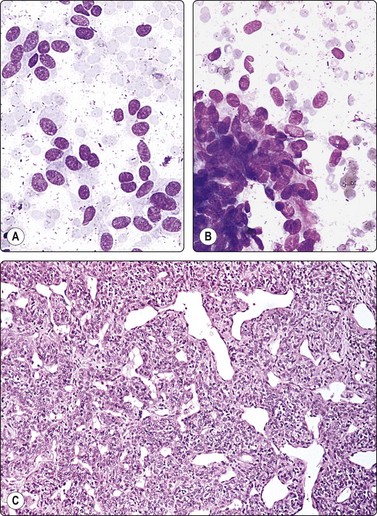
Fig. 12.39 Malignant haemangiopericytoma
(A) Dispersed plump spindle cells; uniform bland nuclei and indistinct cytoplasm; (B) Cohesive tissue fragment of spindle cells fanning out from a stromal core (MGG, IP); (C) Corresponding tissue section; classic pattern (H&E, LP).
A 62-year-old man had some months’ history of increasing pelvic pain on defecation. A firm mass was felt between the sacrum and the posterior rectal wall. Transrectal FNB yielded abundant blood and numerous neoplastic cells, some single but mainly clusters and small tissue fragments (Fig. 12.39). The cells were elongated or spindle shaped with a moderate amount of pale fragile cytoplasm. Some were attached to strands of basement membrane-like material. The nuclei were relatively uniform, ovoid or elongated and measured on average 10 × 30 µm. Nucleoli were inconspicuous. Even though the nuclear morphology appeared bland and no mitoses were found, the high cellularity was considered suspicious of low-grade malignancy. Histologically, the tumor was reported as a hemangiopericytoma, probably malignant on the basis of its large size, the presence of necrosis (not seen in the FNB samples) and its focally infiltrating borders. Some months after surgery, a solitary lung metastasis appeared.
Predominantly solid variants of hemangiopericytoma may not show any suggestion of a vasoformative pattern, and are more likely to be reported as spindle cell sarcoma. Variants with larger cells may be mistaken for epithelial papillary tumors. EM and/or immunostaining of FNB samples can render a type-specific diagnosis possible.132
Extra-adrenal paraganglioma (Fig. 12.40)133,134
Extra-adrenal paraganglioma of the sympathetic system of the retroperitoneum down to the pelvic floor is a rare tumor. It is often functional and often malignant. Paragangliomas of the parasympathetic system of the head and neck with a similar cytomorphology are described in Chapter 4. A case of malignant metastasizing paraganglioma of the retroperitoneum is illustrated in Figure 12.40. Intranuclear inclusions are sometimes a feature of this tumor.133
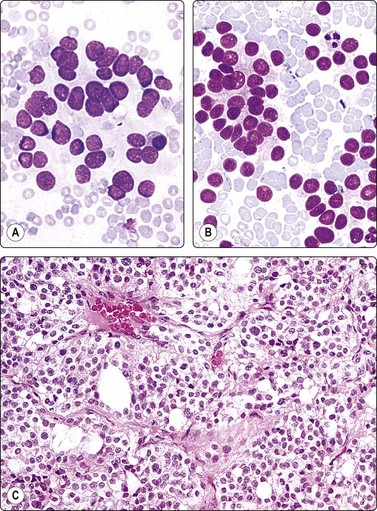
Fig. 12.40 Paraganglioma; malignant, comparison with thyroid carcinoma
(A) Paraganglioma; loose cluster of cells with microfollicular groupings; small round dark relatively uniform nuclei; indistinct cytoplasm; fine cytoplasmic granulation not evident on photograph; (B) Metastasis of thyroid follicular carcinoma to retroperitoneal lymph node (MGG, HP); (C) Tissue section from the same case as in (A) (H&E, IP).
A 40-year-old man presented with a large palpable retroperitoneal mass. CT-guided FNB was done as one of the initial investigations and smears showed moderate numbers of cells, single and in loose acinar/follicular clusters resembling thyroid follicular epithelial cells. Nuclei were uniformly round and relatively small and showed moderate anisokaryosis. The nuclear chromatin was granular and evenly distributed; nucleoli were indistinct. The cytoplasm was pale, without distinct cell borders. Many cells showed a fine red cytoplasmic granularity (MGG). The overall pattern suggested a neuroendocrine tumor (Fig. 12.40A). The differential diagnosis included paraganglioma as well as other neuroendocrine tumors such as carcinoid and pheochromocytoma, but also metastasis of follicular thyroid carcinoma and of low-grade renal cell carcinoma. Surgical biopsy confirmed the diagnosis of paraganglioma. Serum catecholamines were not significantly raised, but abundant neurosecretory granules were demonstrated by EM. There were widespread metastases, and the patient died 4 months later.
The striking similarity between the cytology of thyroid carcinoma and that of paraganglioma is illustrated by an FNB smear of a para-aortic lymph node metastasis in a case of follicular carcinoma of the thyroid (Fig. 12.40B). The distinction between paraganglioma and medullary or follicular carcinoma of the thyroid, whether in a primary site in the neck or metastatic to other sites, requires immune marker studies.
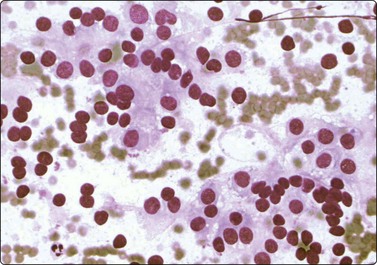
Cystic lymphangioma135
Smears of fluid aspirated from a cystic lymphangioma contain only lymphoid cells, mainly small lymphocytes, in variable numbers. A case from our files is illustrated in Figure 12.41.
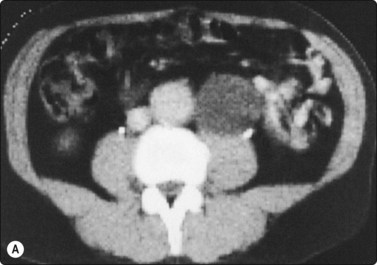
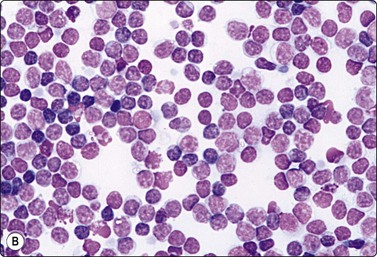
Fig. 12.41 Cystic lymphangioma, retroperitoneum
(A) Abdominal CT scan; low-density round lesion left of aorta; (B) Smear of cyst fluid; numerous mainly small lymphocytes (MGG, HP).
A 66-year-old man was investigated for suspected abdominal aortic aneurysm. A CT scan demonstrated a round mass below the left kidney between the aorta and the psoas muscle which was clearly separated from the aorta (Fig. 12.41A). The mass appeared to be cystic on ultrasonography. FNB yielded milky fluid and the mass decreased considerably in size. Smears of the fluid showed numerous normal lymphocytes. A diagnosis of cystic lymphangioma was made, there was no surgical intervention and follow-up was uneventful.
Idiopathic retroperitoneal fibrosis136,137
Fine needle biopsy may be called upon to urgently investigate the cause of ureteric obstruction, whether it is due to tumor recurrence or postradiation fibrosis.22,23 This problem occurs mainly in patients with a history of surgery and/or radiotherapy for urogenital cancer. Idiopathic fibrosis or metastases from other sites or lymphoma may also enter the differential diagnosis. Smears from idiopathic or postradiation fibrosis are likely to be hypocellular and only a positive finding of neoplastic cells is diagnostic.
Cancers of the urogenital tract, large bowel and pancreas are the main sources of metastases to retroperitoneal nodes. The cytological findings in metastatic carcinoma in lymph nodes from different sources and of malignant lymphoma are described in Chapter 5.
The possibility of a germ cell tumor, primary or metastatic, should be considered in cases without a known primary.138 A primary in the testis may be small and clinically undetected, but may be revealed by radiological imaging. Germ cell tumors have a fairly characteristic cytological pattern and the diagnosis can be supported by immunostaining (see Chapter 13). The most common germ cell tumors in the paediatric age group are teratoma, embryonal carcinoma and yolk sac tumor; in the adult, all types can occur in this site.
The tendency to fibrosis shown by some malignant lymphomas of retroperitoneal nodes can make it difficult to obtain sufficient numbers of cells by FNB, or can mimic a chronic inflammatory process. Core needle biopsy may be necessary to obtain diagnostic material and tissue for immune marker studies.
1 Von Schreeb T, Franzén S, Ljungqvist A. Renal adenocarcinoma. Evaluation of malignancy on a cytologic basis: a comparative cytologic and histologic study. Scand J Urol Nephrol. 1967;1:265-269.
2 Kristensen JK, Holm HH, Rasmussen SN, et al. Ultrasonically guided percutaneous puncture of renal masses. Scand J Urol Nephrol. 1972;6(Suppl 15):49-56.
3 Zajicek J. Aspiration biopsy cytology. Part 2. Cytology of infradiaphragmatic organs. Basel: Karger; 1979.
4 Orell SR. The diagnosis of solid renal and adrenal masses by aspiration cytology. In: Luciani L, Piscioli F, editors. Aspiration cytology in the staging of urological cancer. Berlin: Springer; 1988:215-223.
5 Leiman G. Audit of fine needle aspiration cytology of 120 renal lesions. Cytopathology. 1990;1:65-72.
6 Kelley CM, Cohen MB, Raab SS. Utility of fine-needle aspiration biopsy in solid renal masses. Diagn Cytopathol. 1996;14:60-63.
7 Renshaw AA, Granter SR, Cibas ES. Fine-needle aspiration of the adult kidney. Cancer (Cancer Cytopathol). 1997;81:71-88.
8 Truong LD, Todd TD, Dhurandhar B, et al. Fine-needle aspiration of renal masses in adults. Analysis of results and diagnostic problems in 108 cases. Diagn Cytopathol. 1999;20:339-349.
9 Renshaw AA, Lee KR, Madge R, et al. Accuracy of fine needle aspiration in distinguishing subtypes of renal cell carcinoma. Acta Cytol. 1997;41:987-994.
10 Garcia-Solano J, Acosta-Ortega J, Perez-Guillermo M, et al. Solid renal masses in adults: Image-guided fine-needle aspiration cytology and imagaing techniques – ‘Two heads better than one?’. Diagn Cytopathol. 2008;36:8-12.
11 Niceforo JR, Coughlin BF. Diagnosis of renal cell carcinoma: value of fine-needle aspiration cytology in patients with metastases or contraindications to nephrectomy. AJR. 1993;161:1303-1305.
12 Rybicki FJ, Shu KM, Cibas ES, et al. Percutaneous biopsy of renal masses: sensitivity and negative predictive value stratified by clinical setting and size of masses. A J R. 2003;180:1281-1287.
13 Wood BJ, Khan MA, McGovern F, et al. Imaging guided biopsy of renal masses: indications, accuracy and impact on clinical management. J Urol. 1999;161:1470-1474.
14 DeRoche T, Walker E, Magi-Galluzzi C, et al. Pathologic characteristics of solitary small renal masses. Can they be predicted by preoperative clinical parameters? Am J Clin pathol. 2008;130:560-564.
15 Puttaswamy S, Bagby J, Turbat-Herrera EA. Pathology Case Reviews. 2006;11:161-168.
16 Gross MD, Shapiro B. Clinically silent adrenal masses. J Clin Endocrinol Metab. 1993;77:885-888.
17 Saboorian MH, Katz RL, Chamsangavej C. Fine needle aspiration cytology of primary and metastatic lesions of the adrenal gland. A series of 188 biopsies with radiologic correlation. Acta Cytol. 1995;39:843-851.
18 Nguyen GK. Percutaneous fine-needle aspiration biopsy cytology of the kidney and adrenal. Pathol Annu. 1987;22(pt 1):163-191.
19 Wadih GE, Nance KV, Silverman JF. Fine needle aspiration cytology of the adrenal gland. Fifty biopsies in 48 patients. Arch Pathol Lab Med. 1992;116:841-846.
20 De Agustin P, López-Ríos F, Alberti N, et al. Fine-needle aspiration biopsy of the adrenal glands: a ten-year experience. Diagn Cytopathol. 1999;21:92-97.
21 Lumachi F, Borsato S, Brandes AA, et al. Fine-needle aspiration cytology of adrenal masses in noncancer patients. Clinicoradiologic and histologic correlations in functioning and nonfunctioning tumors. Cancer (Cancer Cytopathol). 2001;93:323-329.
22 Freiman DB, Ring EJ, Oleaga JA, et al. Thin needle biopsy in the diagnosis of ureteral obstruction with malignancy. Cancer. 1978;42:714-716.
23 Barbaric ZL, MacIntosh PK. Periureteral thin-needle aspiration biopsy. Urol Radiol. 1981;2:181-185.
24 Al-Mofleh IA. Ultrasound-guided fine needle aspiration of retroperitoneal, abdominal and pelvic lymph nodes. Diagnostic reliability. Acta Cytol. 1992;36:413-415.
25 Memel DS, Dodd GD, Esola CC. Efficacy of sonography as a guidance technique for biopsy of abdominal, pelvic and retroperitoneal lymph nodes. AJR. 1996;167:957-962.
26 Luciani L, Piscioli F. Aspiration cytology in the staging of urological cancer. Berlin: Springer; 1988.
27 Chagnon S, Cochand-Priollet B, Gzaeil M, et al. Pelvic cancers: staging of 139 cases with lymphography and fine-needle aspiration biopsy. Radiology. 1989;173:103-106.
28 Volpe A, Kachura JR, Geddie WR, et al. Techniques, safety and accuracy of sampling of renal tumors by fine needle aspiration and core biopsy. J Urol. 2007;178:379-386.
29 Silverman JF, Gurley M, Harris JP, et al. Fine needle aspiration cytology of renal infarcts: cytomorphologic findings and potential diagnostic pitfalls in two cases. Acta Cytol. 1991;35:736-741.
30 Zardawi IM. Renal fine needle aspiration cytology. Acta Cytol. 1999;43:184-190.
31 Fassina S, Borsato S, Fedeli U. Fine needle aspiration cytology (FNAC) of adrenal masses. Cytopathol. 2000;11:302-311.
32 Candel AG, Gattuso P, Reyes CV, et al. Fine-needle aspiration biopsy of adrenal masses in patients with extraadrenal malignancy. Surgery. 1993;114:1132-1136.
33 Wajsman Z, Gamarra M, Park JJ, et al. Transabdominal fine needle aspiration of retroperitoneal lymph nodes in staging of genito-urinary tract cancer. J Urol. 1982;128:1238-1240.
34 Lang EK. Renal cyst puncture and aspiration: a survey of complications. AJR. 1977;128:723-727.
35 Von Schreeb T, Arner O, Skovsted G, et al. Renal adenocarcinoma. Is there a risk of spreading tumour cells in diagnostic puncture? Scand J Urol Nephrol. 1967;1:270-276.
36 Slywotzky C, Maya M. Needle tract seeding of transitional cell carcinoma following fine needle aspiration of a renal mass. Abdom Imaging. 1994;19:174-176.
37 Lee IS, Nguyen S, Shanberg AM. Needle tract seeding after percutaneous biopsy of Wilms tumor. J Urol. 1995;153:1074-1076.
38 Powers CN. Complications of fine needle aspiration biopsy: the reality behind the myths. In: Schmidt WA, editor. Cytopathology. Chicago: ASCP Press; 1996:69-91.
39 Malatskey A, Fields S, Shapiro A. Complete hemorrhagic necrosis of renal adenocarcinoma following percutaneous biopsy. Urology. 1989;33:125-126.
40 Heaston DK, Handel DB, Ashton PR, et al. Narrow gauge needle aspiration of solid adrenal masses. AJR. 1982;138:1143-1148.
41 Cassola G, Nicolet V, van Sonnenberg E, et al. Unsuspected pheochromocytoma: risk of blood pressure alterations during percutaneous adrenal biopsy. Radiology. 1986;159:733-735.
42 Bernardino ME, Walther MM, Phillips VM, et al. CT guided adrenal biopsy; accuracy, safety and indications. AJR. 1985;144:67-69.
43 Goreagonkar R, Viswanathan S, Merchant NH, et al. Diagnostic dilemmas in fine needle aspiration cytology of an ectopic kidney. A case report. Acta Cytol. 2009;53:83-85.
44 Pasternack A, Helin H, Törnroth T, et al. Aspiration biopsy of the kidney with a new fine needle: a way to obtain glomeruli for morphologic study. Clin Nephrol. 1978;10:79-84.
45 Horwitz CA, Manivel JC, Inampudi S, et al. Diagnostic difficulties in the interpretation of needle aspiration material from large renal cysts. Diagn Cytopathol. 1994;11:380-384.
46 Khorsand D. Carcinoma within solitary renal cysts. J Urol. 1965;93:440-444.
47 Todd TD, Dhurandhar B, Mody DM, et al. Fine-needle aspiration of cystic lesions of the kidney. Morphologic spectrum and diagnostic problems in 41 cases. Am J Clin Pathol. 1999;111:317-328.
48 Clark SP, Kung IT, Tang SK. Fine-needle aspiration of cystic nephroma (multilocular cyst of the kidney). Diagn Cytopathol. 1992;8:349-351.
49 Drut R. Cystic nephroma: cytologic findings in fine-needle aspiration cytology. Diagn Cytopathol. 1992;8:593-595.
50 Morgan C, Greenberg ML. Multilocular renal cyst: a diagnostic pitfall on fine-needle aspiration cytology: case report. Diagn Cytopathol. 1995;13:66-70.
51 Hughes JH, Niemann TH, Thomas PA. Multicystic nephroma: report of a case with fine-needle aspiration findings. Diagn Cytopathol. 1996;14:60-63.
52 Gupta R, Dhingra K, Singh S, et al. Multicystic nephroma. A case report. Acta Cytol. 2007;51:651-653.
53 Adsay NV, Eble JN, Strigley JR, et al. Mixed epithelial and stromal tumor of kidney. Am J Surg Pathol. 2000;24:958-970.
54 Sease WC, Elyanderani MK, Belis JA. Ultrasonography and needle aspiration in diagnosis of xanthogranulomatous pyelonephritis. Urology. 1987;29:231-235.
55 Bonzanini M, Pea M, Marignoni G, et al. Preoperative diagnosis of renal angiomyolipoma: fine needle aspiration cytology and immunocytochemical characterization. Pathology. 1994;26:170-175.
56 Tallada N, Martinez S, Raventos A. Cytologic study of renal angiomyolipoma by fine-needle aspiration biopsy: report of four cases. Diagn Cytopathol. 1994;10:37-40.
57 Wadih GE, Raab SS, Silverman JF. Fine needle aspiration cytology of renal and retroperitoneal angiomyolipoma. Report of two cases with cytologic findings and clinicopathologic pitfalls in diagnosis. Acta Cytol. 1995;39:945-950.
58 Crapanzano JP. Fine-needle aspiration of renal angiomyolipoma: Cytological findings and diagnostic pitfalls in a series of five cases. Diagn Cytopathol. 2005;32:53-57.
59 Mojica WD, Jovanoska S, Bernacki EG. Epithelioid angiomyolipoma: appearance on fine-needle aspiration – report of a case. Diagn Cytopathol. 2000;23:192-195.
60 Handa U, Nanda A, Mohan H. Fine-needle aspiration of renal angiomyolipoma: a report of four cases. Cytopathol. 2007;18:250-254.
61 Franzén S, Brehmer-Andersson E. Cytologic diagnosis of renal cell carcinoma. Prog Clin Biol Res. 1982;100:425-432.
62 Cajulis RS, Katz RL, Dekmezian R, et al. Fine needle aspiration biopsy of renal cell carcinoma. Cytologic parameters and their concordance with histology and flow cytometric data. Acta Cytol. 1993;37:367-372.
63 Unger P, Hague K, Klein G, et al. Fine needle aspiration of a renal cell carcinoma with eosinophilic globules. Acta Cytol. 1993;39:201-204.
64 Linsk JA, Franzén S. Aspiration cytology of metastatic hypernephroma. Acta Cytol. 1984;28:250-260.
65 Nguyen GK. Fine needle aspiration biopsy cytology of metastatic renal cell carcinoma. Acta Cytol. 1988;32:409-414.
66 Saleh H, Masood S, Wynn G, et al. Unsuspected metastatic renal cell carcinoma diagnosed by fine needle aspiration biopsy. A report of four cases with immunocytochemical contributions. Acta Cytol. 1994;38:554-561.
67 Hughes JH, Jensen CS, Donnelly AD, et al. The role of fine-needle aspiration cytology in the evaluation of metastatic clear cell tumors. Cancer (Cancer Cytopathol). 1999;87:380-389.
68 Wasco MJ, Pu RT. Comparison of PAX-2, RCC antigen and antiphosphorylated H2AX antibody in diagnosing metastatic renal cell carcinoma by fine needle aspiration. Diagn Cytopathol. 2008;36:568-573.
69 Tickoo SK, Amin MB, Linden DM, et al. Antimitochondrial antibody (113–1) in the differential diagnosis of granular renal cell tumors. Am J Surg Pathol. 1997;21:929-930.
70 Suzigan S, Lopez-Beltran A, Montironi R, et al. Multilocular cystic renal cell carcinoma: a report of 45 cases of a kidney tumor of low malignant potential. Am J Clin Pathol. 2006;125:217-222.
71 Flint A, Cookingham C. Cytologic diagnosis of the papillary variant of renal-cell carcinoma. Acta Cytol. 1987;31:325-329.
72 Dekmezian R, Sneige N, Shabb N. Papillary renal-cell carcinoma: fine-needle aspiration of 15 cases. Diagn Cytopathol. 1991;7:198-203.
73 Granter SR, Perez-Atayde AR, Renshaw AA. Cytologic analysis of papillary renal cell carcinoma. Cancer (Cancer Cytopathol). 1998;84:303-308.
74 Wang S, Filipowicz EA, Schnadig VJ. Abundant intracytoplasmic hemosiderin in both histiocytes and neoplastic cells: a diagnostic pitfall in fine-needle aspiration of cystic papillary renal-cell carcinoma. Diagn Cytopathol. 2001;24:82-85.
75 Ligato S, Ro JY, Tamboli P, et al. Benign tumors and tumor-like lesions of the adult kidney. Part I: benign renal epithelial neoplasms. Advances in Anatomic Pathology. 1999;6:1-11.
76 Granter SR, Renshaw AA. Fine-needle aspiration of chromophobe renal cell carcinoma. Analysis of six cases. Cancer (Cancer Cytopathol). 1997;81:122-128.
77 Salamanca J, Alberti N, Lopez-Rios F, et al. Fine needle aspiration of chromophobe renal cell carcinoma. Acta Cytol. 2007;51:9-15.
78 Tejerina E, Gonzalez-Peramato P, Jimenez-Heffernan JA. Cytological features of chromophobe renal cell carcinoma, classic type. A report of nine cases. Cytopathol. 2009;20:44-49.
79 Wiatrowska BA, Zakowski MF. Fine-needle aspiration biopsy of chromophobe renal cell carcinoma and oncocytoma. Comparison of cytomorphologic features. Cancer (Cancer Cytopathol). 1999;87:161-167.
80 Garcia E, Li M. Caveolin-1 immunohistochemical analysis in differentiating chromophobe renal cell carcinoma from renal oncocytoma. Am J Clin Pathol. 2006;125:392-398.
81 Memeo L, Jhang J, Assaad AM, et al. Immunohistochemical analysis for cytokeratin 7, KIT, and PAX2. Value in the differential diagnosis of chromophobe cell carcinoma. Am J Clin Pathol. 2007;127:225-229.
82 Delahunt B. Sarcomatoid renal cell carcinoma: the final common dedifferentiation pathway of renal epithelial malignancies. Pathology. 1999;31:185-190.
83 Auger M, Katz RL, Sella A, et al. Fine-needle aspiration cytology of sarcomatoid renal cell carcinoma; a morphologic and immunocytochemical study of 15 cases. Diagn Cytopathol. 1993;9:46-51.
84 Layfield LJ. Fine-needle aspiration biopsy of renal collecting duct carcinoma. Diagn Cytopathol. 1994;11:74-78.
85 Caraway NP, Wojcik EM, Katz RL, et al. Cytologic findings of collecting duct carcinoma of the kidney. Diagn Cytopathol. 1995;13:304-309.
86 Bejar J, Szvalb S, Maly B, et al. Collecting duct carcinoma of kidney: a cytologic study and case report. Diagn Cytopathol. 1996;15:136-138.
87 Assad L, Resetkova E, Oliveira VL, et al. Cytologic features of renal medullary carcinoma. Cancer (Cancer Cytopathol). 2005;105:28-34.
88 Meyer PN, Clark JI, Flanigan RC, et al. Xp11.2 translocation renal cell carcinoma with very aggressive course in five adults. Am J Clin Pathol. 2007;128:70-79.
89 Schinstine M, Filie AC, Torres-Cabala C, et al. Fine-needle aspiration of a Xp11.2 translocation/TFE3 fusion renal cell carcinoma metastatic to the lung: report of a case and review of the literature. Diagn Cytopathol. 2006;34:751-756.
90 Otani M, Shimizu T, Serizawa H, et al. Mucinous tubular and spindle cell carcinoma of the kidney. Report of a case with imprint cytology features. Acta Cytol. 2006;50:680-682.
91 Owens CL, Argani P, Ali SZ. Mucinous tubular and spindle cell carcinoma of the kidney. Cytopathologic findings. Diagn Cytopathol. 2007;35:593-596.
92 Fuhrman S, Lasky L, Lims C. Prognostic significance of morphologic parameters in renal cell carcinoma. Am J Surg Pathol. 1982;6:655-663.
93 Kelley CM, Cohen MB, Raab SS. Utility of fine-needle aspiration biopsy in solid renal masses. Diagn Cytopathol. 1996;14:14-19.
94 Al Nazer M, Mourad WA. Successful grading of renal-cell carcinoma in fine-needle aspirates. Diagn Cytopathol. 2000;22:223-226.
95 Hartwick RWJ, El-Naggar AK, Ro JY, et al. Renal oncocytoma and granular renal cell carcinoma. A comparative clinicopathologic and DNA flow cytometric study. Am J Clin Pathol. 1992;98:587-593.
96 Nguyen GK, Amy RW, Tsang S. Fine needle aspiration biopsy cytology of renal oncocytoma. Acta Cytol. 1985;29:33-36.
97 Caputo V, Repetti ML, Bordoni V, et al. Preoperative diagnosis of renal oncocytoma by fine needle aspiration. Cytopathology. 1996;7:366-371.
98 Liu J, Fanning CV. Can renal oncocytomas be distinguished from renal cell carcinoma on fine-needle aspiration specimens? A study of conventional smears in conjunction with ancillary studies. Cancer (Cancer Cytopathol). 2001;93:390-397.
99 Zafar N, Spencer D, Berry AD. Embryonal adenoma of the kidney: a report of two cases. Diagn Cytopathol. 1997;16:42-46.
100 Khayyata S, Grignon DJ, Aulicino MR, et al. Metanephric adenoma vs. Wilms’ tumor. A report of 2 cases with diagnosis by fine needle aspiration and cytologic comparisons. Acta Cytol. 2007;51:464-467.
101 Francis LP, James DT. Pitfalls in the cytological diagnosis of metanephric adenoma. Cytopathol. 19(suppl 1), 2008. abstract 135
102 Patel NP, Geisinger KR, Zagoria RJ, et al. Fine needle aspiration biopsy of metanephric adenoma. A case report. Acta Cytol. 2009;53:327-331.
103 Hazarika D, Narasimhamurthy KN, Rao CR, et al. Fine needle aspiration cytology of Wilms’ tumor. A study of 17 cases. Acta Cytol. 1994;38:355-360.
104 Ellison DA, Silverman JF, Strausbauch PH, et al. Role of immunocytochemistry, electron microscopy and DNA analysis in fine-needle aspiration biopsy diagnosis of Wilms’ tumor. Diagn Cytopathol. 1996;14:101-107.
105 Li P, Perle MA, Scholes JV, et al. Wilm’s tumor in adults: aspiration cytology and cytogenetics. Diagn Cytopathol. 2002;26:99-103.
106 Nguyen G-K, Schumann GB. Needle aspiration cytology of low-grade transitional cell carcinoma of the renal pelvis. Diagn Cytopathol. 1997;16:437-441.
107 Ho CC, Nguyen G-K, Schumann GB. Needle aspiration cytology of metastatic high-grade transitional-cell carcinomas of the urinary tract. Diagn Cytopathol. 1998;18:409-415.
108 Powers CN, Elbadawi A. ‘Cercariform’ cells: a clue to the cytodiagnosis of transitional cell origin of metastatic neoplasms. Diagn Cytopathol. 1995;13:15-21.
109 Dey P, Amir T, Jogai S, et al. Fine-needle aspiration cytology of metastatic transitional cell carcinoma. Diagn Cytopathol. 2005;32:226-228.
110 Têtu B, Ro JY, Ayala AG, et al. Small cell carcinoma of the kidney. A clinicopathologic, immunohistochemical and ultrastructural study. Cancer. 1987;60:1809-1814.
111 Quinchon JF, Aubert S, Biserte J, et al. Primary atypical carcinoid of the kidney: a classification is needed. Pathology. 2003;35:353-355.
112 Bhalla R, Popp A, Nassar A. Case report: metastatic renal carcinoid to the thyroid diagnosed by fine needle aspiration biopsy. Diagn Cytopathol. 2007;35:597-600.
113 Tsun-Cheung Chow L, Chan S-K, Chow W-H. Fine needle aspiration cytodiagnosis of leiomyosarcoma of the renal pelvis. A case report with immunohistochemical study. Acta Cytol. 1994;38:759-763.
114 Villaneuva RR, Nguyen-Ho P, Nguyen G-K. Leiomyosarcoma of the kidney. Report of a case diagnosed by fine needle aspiration and electron microscopy. Acta Cytol. 1994;38:568-572.
115 Gattuso P, Ramzy I, Truong LD, et al. Utilization of fine-needle aspiration in the diagnois of metastatic tumors to the kidney. Diagn Cytopathol. 1999;21:35-38.
116 Giashuddin S, Cangiarella J, Elgert P, et al. Metastases to the kidney: eleven cases diagnosed by aspiration biopsy with histological correlation. Diagn Cytopathol. 2005;32:325-329.
117 Truong LD, Caraway N, Ngo T, et al. Renal lymphoma. The diagnostic and therapeutic roles of fine-needle aspiration. Am J Clin Pathol. 2001;115:18-31.
118 Scheible W, Coel M, Siemers PT, et al. Percutaneous aspiration of adrenal cysts. AJR. 1977;128:1013-1016.
119 Dunphy CH. Computed tomography-guided fine needle aspiration biopsy of adrenal myelolipoma. Case report and review of the literature. Acta Cytol. 1991;35:353-356.
120 Her-Juing H, Cramer HM, Kho J, et al. Fine needle aspiration cytology of benign adrenal cortical nodules. A comparison of cytologic findings with those of primary and metastatic adrenal malignancies. Acta Cytol. 1998;42:1352-1358.
121 Ren R, Guo M, Sneige N, et al. Fine-needle aspiration of adrenal cortical carcinoma. Cytologic spectrum and diagnostic challenges. Am J Clin Pathol. 2006;126:389-398.
122 Fetsch PA, Powers CN, Zakowski MF, et al. Anti-alpha-Inhibin. Marker of choice for the consistent distinction between adrenocortical carcinoma and renal cell carcinoma in fine-needle aspiration. Cancer (Cancer Cytopathol). 1999;87:168-172.
123 Shin SJ, Hoda RS, Ying L, et al. Diagnostic utility of the monoclonal antibody A103 in fine-needle aspiration biopsies of the adrenal. Am J Clin Pathol. 2000;113:295-302.
124 Wragg T, Nguyen G-K. Cytopathology of adrenal cortical oncocytoma. Diagn Cytopathol. 2001;24:222-223.
125 Krishnamurthy S, Ordonez NG, Shelton TO, et al. Fine-needle aspiration cytology of a case of oncocytic adrenocortical carcinoma. Diagn Cytopathol. 2000;22:299-303.
126 Sharma S, Singh R, Verma K. Cytomorphology of adrenocortical carcinoma and comparison with renal cell carcinoma. Acta Cytol. 1997;41:385-392.
127 Yang B, Ali SZ, Rosenthal DL. CD10 facilitates the diagnosis of metastatic renal cell carcinoma from primary adrenal cortical neoplasm in adrenal fine-needle aspiration. Diagn Cytopathol. 2002;27:149-152.
128 Nance KV, McLeod DL, Silverman JF. Fine-needle aspiration biopsy of spindle cell neoplasms of the adrenal gland. Diagn Cytopathol. 1992;8:235-241.
129 Nguyen GK. Cytopathologic aspects of adrenal pheochromocytoma in a fine needle aspiration biopsy. A case report. Acta Cytol. 1982;26:354-358.
130 Deodhare S, Chalvardjian A, Lata A, et al. Adrenal pheochromocytoma mimicking small cell carcinoma on fine needle aspiration biopsy. A case report. Acta Cytol. 1996;40:1003-1006.
131 Shidham VB, Galindo LM. Pheochromocytoma. Cytologic findings on intraoperative scrape smears in five cases. Acta Cytol. 1999;43:207-213.
132 Geisinger KR, Silverman JF, Cappellari JO, et al. Fine-needle aspiration cytology of malignant hemangiopericytomas with ultrastructural and flow cytometric analyses. Arch Pathol Lab Med. 1990;114:705-710.
133 Vera-Alvarez J, Marigil-Gómez M, Abascal-Agorreta M, et al. Malignant retroperitoneal paraganglioma with intranuclear vacuoles in a fine needle aspirate. Acta Cytol. 1993;37:229-233.
134 Jimenez-Hefferman JA, Vicandi B, Lopez-Ferrer P, et al. Cytologic features of Pheochromocytoma and retroperitoneal paraganglioma. A morphologic and immnuohistochemical study of 13 cases. Acta Cytol. 2006;50:372-378.
135 Sarno RC, Carter BL, Bankoff MS. Cystic lymphangiomas: CT diagnosis and thin needle aspiration. Br J Radiol. 1984;57:424-426.
136 Stein AL, Bardawil RG, Silverman SG, et al. Fine needle aspiration biopsy of idiopathic retroperitoneal fibrosis. Acta Cytol. 1997;41:461-466.
137 Dash RC, Liu K, Sheafor DH, et al. Fine-needle aspiration findings in idiopathic retroperitoneal fibrosis. Diagn Cytopathol. 1999;21:22-26.
138 Collins KA, Geisinger KR, Wakely PE, et al. Extragonadal germ cell tumours: A fine-needle aspiration biopsy study. Diagn Cytopathol. 1995;12:223-229.