CHAPTER 14 Skin and subcutis
Clinical aspects
The place of FNAC in the investigative sequence
Fine needle biopsy (FNB) and cytodiagnosis has found only limited application in primary tumors of the skin and subcutis due to the ease of surgical biopsy and of excision. The main indications for FNB are rapid, non-invasive investigation of suspected metastatic malignancy, and distinction between neoplasia and a reactive process likely to resolve spontaneously or respond to conservative treatment.1,2 In patients with known malignancy, the nature of any nodules or thickenings related to surgical scars or elsewhere in the skin or subcutis the distinction can easily be made between suture granuloma, infection or other reactive process and recurrent or metastatic tumor. The possibility of a second primary or of de-differentiation of the original tumor can also be decided by cytology.
Some knowledge of the cytological features of skin adnexal tumors is needed to avoid mistaking these for metastatic malignancies. With some exceptions, a type-specific diagnosis of primary adnexal tumors by FNB is difficult or impossible but is not often of practical importance. From a clinical point of view, the information sought is mainly if the lesion is benign or malignant, and if malignant whether it is primary or metastatic. Cytology as a supplement to history and clinical findings can be useful in the management of patients with skin tumors.
Incisional biopsy or punch biopsy of pigmented lesions is generally contraindicated in view of the risk of spreading melanoma and of interfering with the histological diagnosis and staging on subsequent excision. Although not proven, FNB could have similar adverse effects and is therefore not recommended for localized pigmented skin lesions. However, since pigmented lesions are occasionally examined by FNB, cytopathologists must be familiar with the cytomorphology of melanocytic skin lesions (see below).
Scrape smears from tumors involving the epidermis or a mucous membrane is an alternative to biopsy in some cases, e.g. to confirm suspected squamous cell carcinoma, basal cell carcinoma or Paget’s disease. Scrape smears can also be used to confirm advanced ulcerated malignant melanoma. Dermatologists seeking an alternative treatment to surgical excision of basal cell carcinoma have shown some interest in cytological confirmation of this diagnosis.
Subcutaneous lipoma is one of the commonest ‘tumors’ seen in a community-based cytology practice. It may seem unnecessary to subject these clinically characteristic lesions to biopsy, but the patient and sometimes the referring doctor are often concerned and demand immediate reassurance. We have had some surprise findings in such cases and feel that FNB is generally justified. One example of several: a small, discrete, soft nodule was incidentally felt in the lower anterior abdominal wall of a 45-year-old woman. The clinical diagnosis was lipoma, but cytology showed a highly cellular, sarcomatous tumor, histologically confirmed as synovial sarcoma.
In general, FNB is not useful in the investigation of inflammatory skin disease except to provide material for microbiology, for example in tuberculosis and leprosy.
Accuracy of diagnosis
Only a few series of FNB of primary skin tumors with histologic correlation have been reported.3-8 In one of the largest series, 89% of primary skin tumors were correctly diagnosed as benign or malignant, and specific typing was possible in 81% of cases.4 Some skin adnexal tumors, in particular pilomatrixoma, have been reported as diagnostic pitfalls and a possible cause of false-positive cytological diagnosis.9 Clinical correlation is essential.
Complications
The hypothetical risk of tumor cell seeding and of compromising subsequent histological examination as a result of FNB has been mentioned. Other significant complications have not been reported.
Technical considerations
We generally use 25- or 27-gauge needles and the non-aspiration technique for skin tumors, 23–25-gauge for subcutaneous lesions. Local anesthesia is rarely necessary. Two to four passes are usually required to obtain sufficient material and to compensate for possible tumor heterogeneity. Alcohol-fixed H&E- or Pap-stained smears and air-dried smears stained with Diff-Quik or MGG should be used in parallel. FNB of skin lesions often yield cohesive tissue fragments. Alcohol-fixation makes tissue fragments transparent whereas cell detail is poorly seen in fragments in air-dried smears. Squamous differentiation is more easily recognized in Pap-stained smears. On the other hand, air-dried MGG smears provide more information on secretory products and stromal components.
Thin superficial skin lesions, particularly if eroded or ulcerated, are best sampled by scraping cells off the surface. Keratin, crust and inflammatory exudate must be removed to ensure that intact, well-preserved cells are obtained from as deeply into the lesion as possible. Scraping with a sterile scalpel blade held at a blunt angle until the lesion bleeds slightly is recommended.10 Wooden spatulas or other soft materials are less suitable since they tend to absorb the fluid component. FNB can be successful in some thin lesions using a 25–27-gauge needle inserted tangentially.
Cytological findings
Inflammatory processes
Purulent material aspirated from suppurative inflammation or abscess can be used for microbiological studies. The presence of many mature and keratinized squamous epithelial cells suggests an inflamed epidermoid cyst or suppurative hidradenitis, hair shafts indicate a dermoid cyst or a pilonidal sinus. Demodex may be identified in scrape smears of infectious folliculitis.11 A malignancy, mainly squamous cell carcinoma can be obscured by inflammatory exudate and smears should be examined carefully for single neoplastic cells.
FNB samples from a non-suppurative chronic inflammatory process usually contain fragments of inflammatory/reparative granulation tissue composed of clustered, relatively cohesive reactive fibroblasts and histiocytes, strands of endothelial cells representing capillary vessels, and various inflammatory cells. Granulation tissue can be quite cellular in smears, macrophages and fibroblasts may look atypical and mitotic figures are not uncommon, but the overall findings in the appropriate clinical context are characteristic.
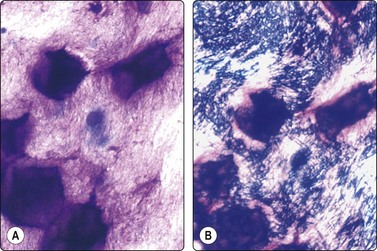
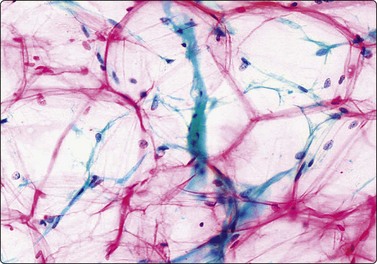
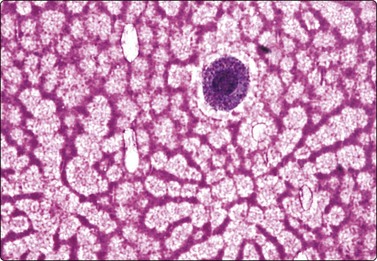
Inclusion bodies may be found in scrape smears of viral skin lesions. The cytological features of herpes virus infection are well known from gynecological cytology. Molluscum bodies of molluscum contagiosum look similar in smears and in histological sections (Fig. 14.1).
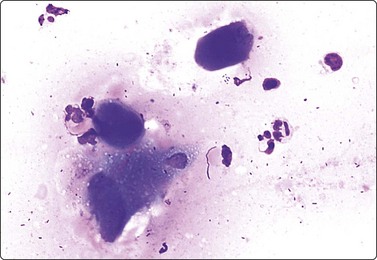
Fig. 14.1 Molluscum contagiosum
Three darkly staining molluscum bodies and some inflammatory cells (MGG, HP).
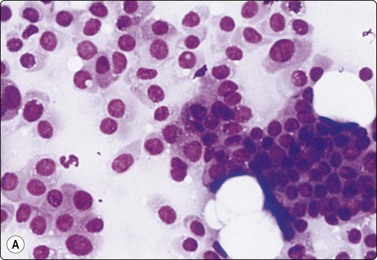
Clusters of epithelioid histiocytes with poorly defined cytoplasm and pale nuclei of a banana-, bean- or footprint-like shape, associated with multinucleated histiocytic giant cells are consistent with granulomatous inflammation. The differential diagnosis includes a number of conditions: foreign body granuloma, tuberculosis, leprosy, sarcoid, fungal infections, etc.12 Multinucleated histiocytic giant cells are prominent in foreign body granuloma, and the presence of birefringent particles, suture material, etc. supports the diagnosis (Fig. 14.2). Caseous necrosis suggests tuberculosis, to be confirmed by staining for acid-fast bacilli; fibrosis with no evidence of necrosis is more in keeping with sarcoid. The cytological patterns found in leprosy have been described.13,14 The etiology of granulomatous inflammation cannot be decided on the basis of cytomorphology alone and must be pursued by staining for microorganisms, bacteriological culture, serological tests, and other investigations.
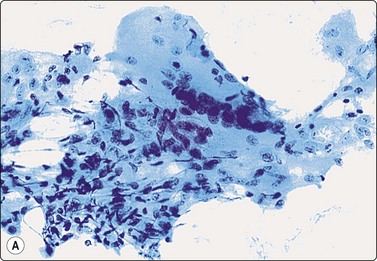
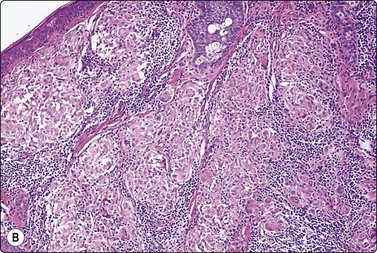
Fig. 14.2 Sarcoid-like foreign body granuloma
(A) Large multinucleated giant cells, clustered histiocytes and some inflammatory cells. Birefringent foreign material present but not seen in this microphotograph (Pap, IP); (B) Corresponding tissue section. Sarcoid-like cluster of many discrete granulomata of epithelioid histiocytes and some giant cells; no evidence of caseation; foreign bodies seen in polarized light (H&E, LP).
A vaguely granulomatous chronic inflammatory pattern with a background of fat and evidence of fat necrosis seen in FNB smears of a subcutaneous induration on the leg suggests a panniculitic process, such as erythema nodosum.15 The findings are not diagnostic, only suggestive, and necessitate a surgical biopsy. Cytological findings in fat necrosis are described in Chapter 7 (see p. 167).
Cysts and other non-neoplastic lesions
Aspirates from epidermal or dermoid cysts consist of thick, greasy, foul-smelling material. Smears show mature squamous epithelial cells, a high proportion of which are keratinized cells or ghost cells, and a background of debris and often inflammatory cells. Foreign body giant cells and calcium granules may be present (Fig. 14.3). The presence of hair shafts suggests a dermoid cyst.16 Reactive epithelial atypia in inflamed cysts can look worrisome.4 On the other hand, cells of partly necrotic and degenerate, well-differentiated metastatic squamous cell carcinoma can look deceptively bland in FNB samples. The content of trichilemmal cysts is similar, but clumps of structureless keratin are more prominent, there are no granular or parakeratotic cells and cholesterol crystals are often seen. Cytological findings in proliferating trichilemmal cyst and malignant proliferating trichilemmal tumor have been described.17,18
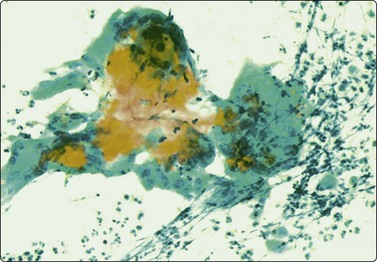
Clumps of keratin and squamous cells, multinucleate histiocytes, inflammatory cells and debris (Pap, IP).
A postoperative lymphocele yields clear, yellowish, mucoid fluid on aspiration which contains a variable number of mature lymphocytes. Smears from a hematoma show altered blood, hemosiderin-containing macrophages, amorphous material with cholesterol crystals, or fragments of reparative granulation tissue with hemosiderin pigment, depending on the age of the process.
The material aspirated from a ganglion is thick, colorless, glassy and jelly-like. It is so characteristic that the diagnosis is already obvious from the macroscopic appearance of the aspirate in the appropriate clinical setting. Smears show a small number of single cells with abundant cytoplasm and small oval nuclei and a background of abundant myxoid material, which may show interesting drying artifacts (Fig. 14.4).19 FNB of bursal cysts has been described as similar to that of ganglion.20
A single histiocyte-like cell; background of myxoid material showing peculiar drying artifact (MGG, HP).
A few cases of rheumatoid nodules with FNB findings have been reported.21,22 Samples are scanty of amorphous granular acidophilic material with a variable number of fibroblasts and/or histiocytes. Small multinucleated histiocytic giant cells may be seen. Gouty tophi are sometimes subjected to FNB if the clinical diagnosis is doubtful. Thick, putty-like material is aspirated. Smears show clumps of non-cellular material of birefringent needle-shaped crystals that are better preserved than in formalin-fixed tissue, and a few histiocytes and giant cells (Fig. 14.5).21,23
Calcinosis of subcutaneous or soft tissue may present as a mass lesion. It is usually of dystrophic or metabolic etiology but there is also a rare, primary inherited form called tumoral calcinosis.24 FNB yields amorphous calcified material, laminated concretions and variable numbers of histiocytes, lymphocytes and osteoclast-like giant cells.
Endometriosis can present as a poorly defined, tumor-like induration in the subcutaneous tissue or in relation to a scar of the anterior abdominal wall in a premenopausal woman. Cytological findings are described in Chapter 13 (see page 357).25
Amyloid tumor of subcutaneous tissue may also present as a mass lesion. FNB smears display fragments of acellular amorphous matrix, scattered histiocytic cells and occasionally small calcifications. FNB of periumbilical adipose tissue of the anterior abdominal wall can be of value in the diagnosis of secondary systemic amyloidosis although sensitivity is low.26,27 Rings of amyloid around fat cells and amyloid deposits in vessel walls can be demonstrated by Congo red staining and polarization.
The cytology of benign primary epithelial skin tumors has mainly been described in single case reports, and criteria are not well established. A wide range of tumors occur in this site and specific entities are difficult or impossible to identify in smears. Clinically, the distinction between benign and malignant tumors, particularly those of metastatic nature, is the most important, and this is possible by FNB with a high level of accuracy. In general, cell-rich smears of more or less cohesive small basaloid cells mixed with a variable number of squamoid cells and sometimes glandular elements suggest a primary cutaneous neoplasm, and close study of the nuclear cytomorphology is likely to predict malignancy. The microarchitectural pattern and certain stromal components can provide clues to a type-specific diagnosis in some cases.
Our limited experience does not permit a systematic description of the cytology of skin adnexal tumors. Some examples selected from our files are briefly described and illustrated and references are provided to relevant case reports and small series published in the literature.
Adenomatous tumors of sweat gland origin
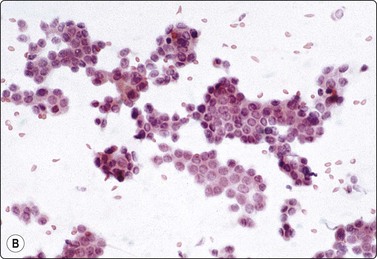
Smears of cutaneous cylindroma (’turban tumor’) are usually cell-rich, of pseudopapillary fragments of cohesive basaloid epithelial cells and hyaline stromal material often seen as globules. The cells are small, crowded with a high nuclear : cytoplasmic ratio and relatively uniform round or ovoid hyperchromatic nuclei. The pattern can closely resemble adenoid cystic carcinoma but the nuclear chromatin is bland (Figs 14.6 and 14.7). Cutaneous cylindroma should be remembered in the differential diagnosis of tumors in the head and neck.28
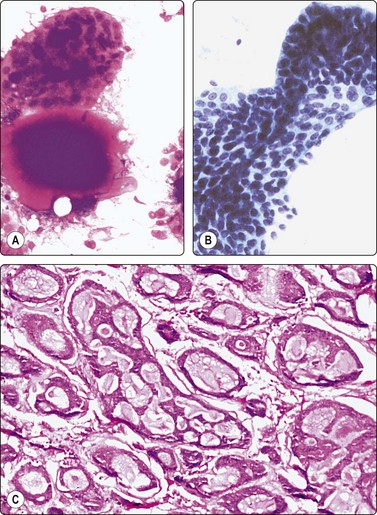
Fig. 14.6 Cutaneous cylindroma
Pseudopapillary tissue fragments of cohesive bland basaloid cells; hyaline stromal globule (A, MGG; B, Pap, IP); (C) Corresponding tissue section (H&E, IP)
(Courtesy Dr J. Klijanienko, Inst. Curie, Paris).
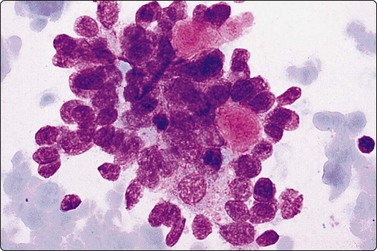
Fig. 14.7 Cutaneous cylindroma
Clustered basaloid epithelial cells; mild nuclear atypia; hyaline stromal globules; note similarity to adenoid cystic carcinoma (MGG, HP)
(Courtesy Dr K. Lindholm, Malmo, Sweden).
Spiradenoma is closely related to and resembles cylindroma. Smears are highly cellular, of clustered, variably cohesive small basaloid epithelial cells with uniform oval dark nuclei and a homogeneous chromatin. A more or less obvious acinar/tubular arrangement of the cells is discernible, but a dual population of small dark and larger pale epithelial cells as seen in histological sections is difficult to appreciate in smears. Globules of hyaline stromal material are characteristic but were scant in the case illustrated here (Fig. 14.8). The main differential diagnosis is adenoid cystic carcinoma.29
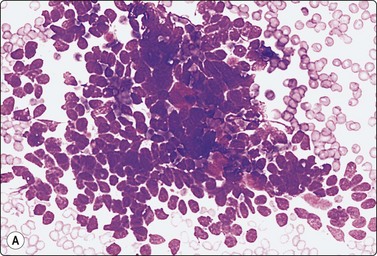
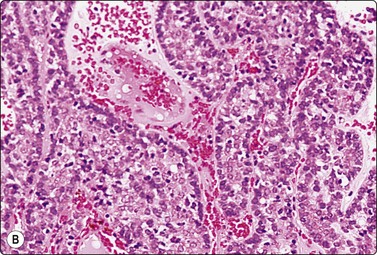
(A) Clustered and dissociated small basaloid cells with relatively uniform dark nuclei; suggestion of acinar/tubular grouping; minimal amount of hyaline stroma (MGG, IP); (B) Corresponding tissue section (H&E, IP).
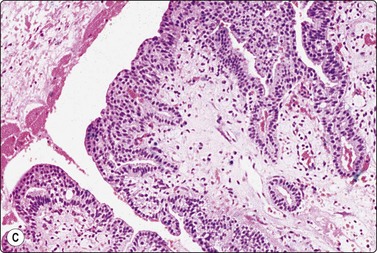
The cytology of nodular hidradenoma30-32 and of clear cell hidradenoma33,34 has been described in single cases. A benign skin adnexal tumor from our files, histologically reported as cystic eccrine hidradenoma, is shown in Figure 14.9. The FNB sample was of mucoid fluid which contained clusters of variably cohesive uniform epithelial cells with a moderate amount of cytoplasm and small dark ovoid nuclei. A dual population was not discernible. The cytology was reported as a benign skin adnexal tumor without further specification.
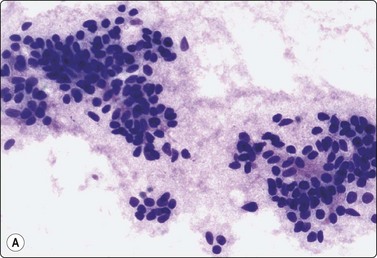
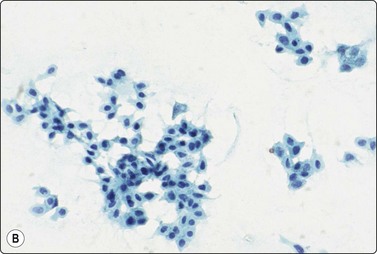
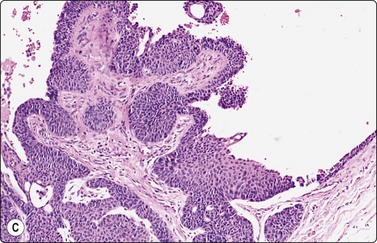
Fig. 14.9 Cystic eccrine hidradenoma
Loose clusters of basaloid cells with small, dark, uniform nuclei in a background of proteinaceous fluid (A, MGG; B, Pap, IP); (C) Corresponding tissue section (H&E, LP).
Benign skin adnexal tumors located to the breast or axilla can be mistaken for primary or metastatic breast cancer.33,35 Knowledge of the exact localization of the lesion and its relation to the skin is essential when examining FNBs, as illustrated by the following case from our files:
FNB findings in vulval papillary hidradenoma have also been reported.37
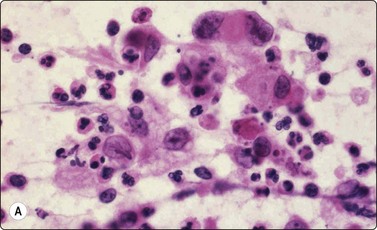
Pilomatricoma (calcifying epithelioma of Malherbe) (Fig. 14.11)37-40
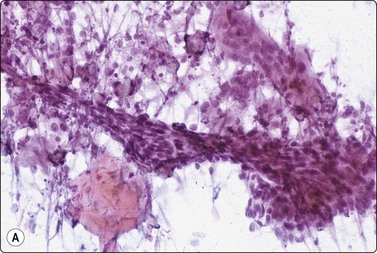
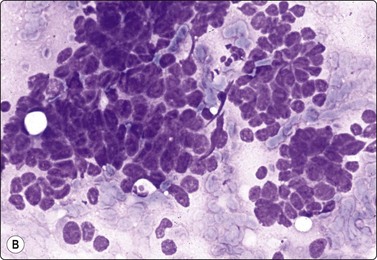
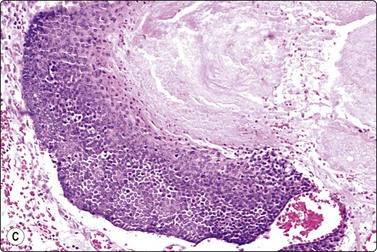
(A) Clump of anucleate keratinized squamous cells (‘ghost cells’), small basaloid cells, large multinucleate histiocytic giant cell, inflammatory cells, calcium granules and debris (Pap, IP); (B) Clusters of small, tightly packed basaloid cells; may be mistaken for malignant if the complex smear pattern is lacking and basaloid cells dominate (MGG, HP); (C) Corresponding tissue section. The basaloid cells may be selectively sampled by FNB (H&E, IP).
Pilomatrixoma has attracted considerable interest in the cytological literature as the commonest cause of false-positive diagnosis in this area. Large numbers of tightly clustered and some single basaloid cells with scanty cytoplasm and medium-sized, hyperchromatic, mildly irregular nuclei with small nucleoli may dominate the FNB samples. Mitotic figures and apoptotic bodies may be found, as well as moderate nuclear pleomorphism and atypical cells arranged in a whorled fashion more often seen in smears from squamous cell carcinoma (Fig. 14.12). The cells resemble those of basal cell carcinoma, basaloid squamous cell carcinoma or small cell anaplastic carcinoma. The ‘dirty’ background may be misinterpreted as tumor necrosis, but is rich in granular calcium and characteristically includes histiocytic multinucleated giant cells. The ‘ghost cells’ seen in tissue sections appear as clumps of keratinous material or degenerate mature squamous epithelial cells in smears. There is a tendency for the basaloid cells to be overrepresented in smears, probably because they are more easily detached than the other components.
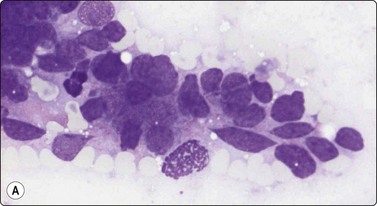
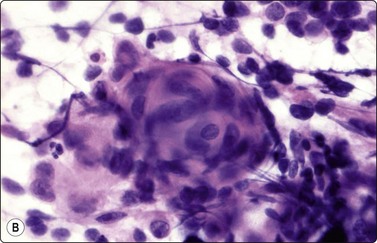
(A) Moderate nuclear pleomorphism and a mitotic figure (MGG, HP); (B) Atypical cells arranged in a whorled cluster resembling squamous cell carcinoma (H&E, HP).
There is a risk of making a false-positive cytological diagnosis if clustered basaloid cells dominate the smears and the characteristic ghost cells, background calcification and giant cells are scant or overlooked. Clinical findings, age, size and history are important clues to the correct diagnosis
Squamous cell carcinoma (Fig. 14.13)
Scrape smears are a simple way of diagnosing squamous cell carcinoma of the skin, particularly ulcerated lesions. Inflammatory crust and any surface keratinous layers must first be carefully removed to obtain diagnostic cells from the deeper layers. The technique is described above. Nodular and deeply invasive tumors and recurrences are better examined by FNB.
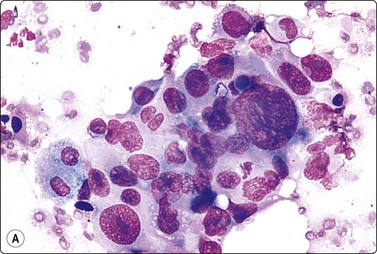
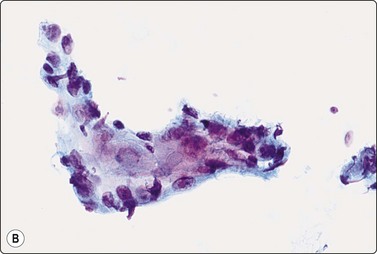
Fig. 14.13 Squamous cell carcinoma
(A) Aggregate of pleomorphic, obviously malignant cells with dense squamoid cytoplasm and distinct cell borders; no obvious keratinization in this field; FNB smear from ulcerated 10–15-mm tumor lower lip (MGG, HP); (B) Cohesive tissue fragment of malignant squamous epithelial cells; scrape smear from ulcerated skin tumor (Pap, HP).
Cytological criteria of squamous cell carcinoma are described in several other chapters, mainly in Chapter 8. The differential diagnosis between well-differentiated squamous carcinoma with cystic degeneration and branchial and other benign cysts lined by squamous epithelium can cause problems due to subtle squamous cell atypia in the former and reactive atypia and metaplasia in the latter (see Chapter 4). Distinction between keratotic basal cell carcinoma and basaloid squamous cell carcinoma can be difficult.4 Cytological criteria for basal cell carcinoma are listed below. The cytomorphology of adenoid (acantholytic) squamous cell carcinoma can cause problems.44 Distinction between in situ (dysplastic solar keratosis, Bowen’s disease) and superficially invasive carcinoma is not possible in smears.
Basal cell carcinoma (Fig. 14.14)8,45-48
Most basal cell carcinomas (BCC) are diagnosed clinically. Scrape smears from the surface can provide rapid pretreatment confirmation, of particular importance if non-surgical treatment is considered. If the lesion is ulcerated, any inflammatory debris should be removed prior to vigorous scrapings.8 Non-ulcerated, deeply invasive tumors are suitable for FNB sampling using a thin 25–27-gauge needle.
The most characteristic feature of BCC in smears is the strong cohesiveness of the cells, which remain in well-defined tissue fragments of tightly packed small cells with palisading of nuclei along the edges. The fragments resemble tumor buds seen in tissue sections (Fig. 14.14A). The same pattern was seen in FNB smears from a rare case of distant metastasis to bone from of a large, deeply invasive BCC on the back of an elderly patient (Fig. 14.14B). The cells are small with very scanty cytoplasm and overlapping, ovoid, relatively uniform hyperchromatic nuclei. The chromatin is evenly distributed and nucleoli are inconspicuous. Subtyping of BCC is generally not possible, but stromal material can be prominent in smears from desmoplastic, infiltrating basal cell carcinoma, suggesting a differential diagnosis of chondroid syringoma (Fig. 14.14D).
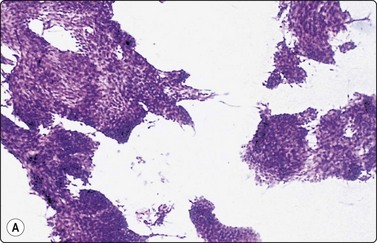
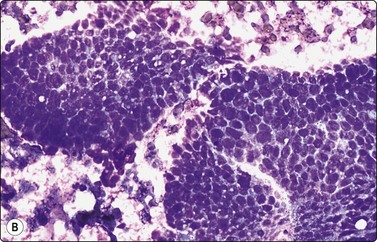
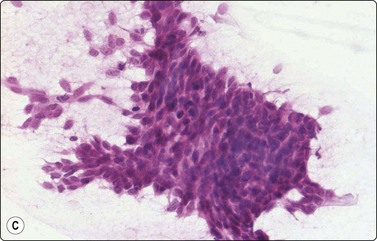
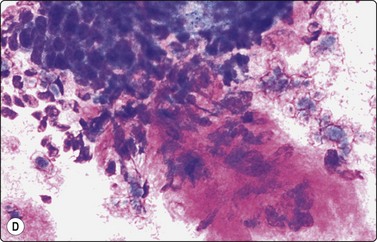
Fig. 14.14 Basal cell carcinoma
(A) Cohesive epithelial fragments with alternating sharp and irregular borders (H&E, IP); (B) Tissue fragments of very cohesive basaloid cells. The fragments have well-defined borders of cells with a tendency to palisading. This smear is a FNB sample from a bone deposit of metastasizing basal cell carcinoma (MGG, HP); (C) Tumor fragment of small, tightly packed cells, FNB smear (H&E, MP); (D) Basal cell carcinoma, infiltrating type; tumor fragment of closely packed small, uniform basal cells and adherent fibrillar fibrous stroma (MGG, LP).
In clinical practice, the main differential is BCC versus actinic keratosis. Christensen et al., in a study of 78 cases, found cytological diagnosis to be highly accurate if samples are adequate.48 In general, the microarchitecture and the predominance of typical basal cells are characteristic of BCC. Cells of actinic keratosis are less cohesive, and show features of dysplastic squamous epithelial cells with more abundant cytoplasm. Cells of non-keratinizing, basaloid squamous cell carcinoma are also less cohesive, larger, some with dense squamoid cytoplasm, some with prominent nucleoli.
The distinction from benign and malignant skin adnexal tumors is a less common but more difficult problem. Smears of adnexal tumors are similarly dominated by dense aggregates of small basaloid epithelial cells, but the pattern is usually more mixed and includes squamoid cells, glandular (tubulo-acinar) structures or small round cells, and stromal material.7
Pigmented basal cell carcinoma may be misdiagnosed as melanoma and correct diagnosis may require immunostaining.49
Cytologic findings in several cases of sebaceous carcinoma have been reported.3,4,51,52 The tumor cells have vacuolated, bubbly cytoplasm and malignant nuclear morphology including mitotic figures and prominent nucleoli. There may be an admixture with basaloid and squamous cells and evidence of necrosis (Fig. 14.15).
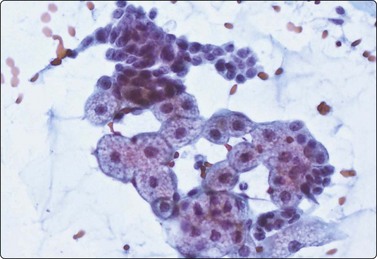
Fig. 14.15 Sebaceous carcinoma
Clusters of cells with abundant bubbly cytoplasm and central nuclei, and of small tightly cohesive basaloid epithelial cells (Pap, HP).
Single cases of malignant sweat gland tumors have been reported.53-55 A case from our files, initially diagnosed as infiltrating basal cell carcinoma both on FNB and on surgical biopsy, is illustrated in Figure 14.16. Later recurrences were histologically diagnosed as eccrine (microcystic) carcinoma The small duct-like clusters of cells showing squamous differentiation were initially overlooked in a predominantly basal cell population.
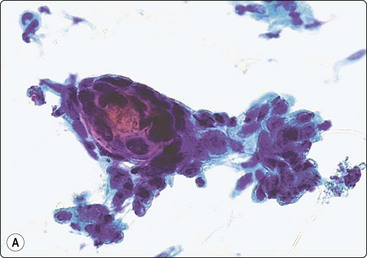
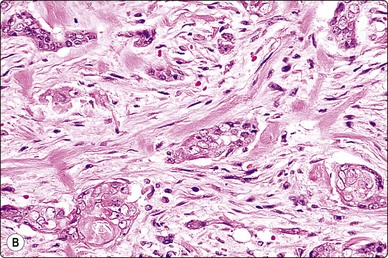
Fig. 14.16 Eccrine (microcystic) carcinoma
(A) Tight clusters of basaloid cells and a microtubular structure with squamous differentiation (Pap, HP); (B) Corresponding tissue section (H&E, IP).
The cytology of some uncommon malignant adnexal tumors has been described in single case reports, e.g. apocrine sweat gland carcinoma,56 malignant spiradenoma,57 primary mucinous carcinoma of skin,58,59 and malignant chondroid syringoma.60
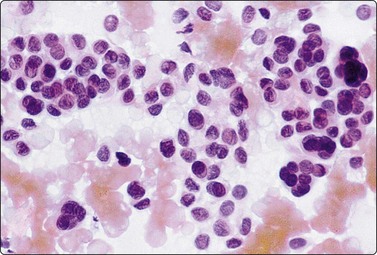
Merkel cell carcinoma (neuroendocrine carcinoma of skin) (Fig. 14.17, and see Fig. 5.23)61,62
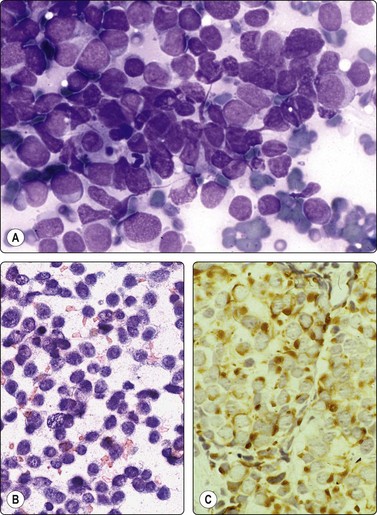
Fig. 14.17 Merkel cell carcinoma
(A) Poorly cohesive cells; nuclear pleomorphism and some molding, scanty blue cytoplasm resembling lymphoid cells (MGG, HP); (B) Cellular smear of dispersed small cells with round, dark nuclei (Pap, HP); (C) Dot-like positive staining with CAM5.2 (immunostaining, HP).
Primary neuroendocrine carcinoma of skin is an uncommon neoplasm, mainly seen in elderly patients and most often in the head and neck. It is locally aggressive and often metastasizes to regional lymph nodes. Clinically and cytologically, Merkel cell tumor can be difficult to distinguish from non-Hodgkin lymphoma when it presents as lymphadenopathy without an obvious primary, as is often the case (see Chapter 5).
Smears are usually highly cellular. The cells are mainly dispersed but some are clustered or form single files with nuclear molding and sometimes rosette-like groups. The main differential diagnoses are lymphoma and metastatic small cell carcinoma. Amelanotic melanoma may also be considered. The absence of lymphoid globules in the background and subtle differences in nuclear chromatin help in this distinction. Staining for CAM5.2, CK7 and CK20 demonstrates characteristic dot-like intracytoplasmic deposits in the tumor cells (Fig. 14.17C), and staining for neuroendocrine markers is usually positive. Electron microscopy shows well-demarcated whorls of cytoplasmic filaments.
Subcutaneous lipoma (Fig. 14.18)
As mentioned in the clinical section, subcutaneous lipoma is one of the commonest targets of FNB anywhere in the body. Samples are mainly acellular fat but usually also include fragments of mature adipose tissue and some vascular elements. Lipomatous lesions are described in detail in Chapter 15.
Histiocytoma/dermatofibroma (Figs 14.19 and 14.20)
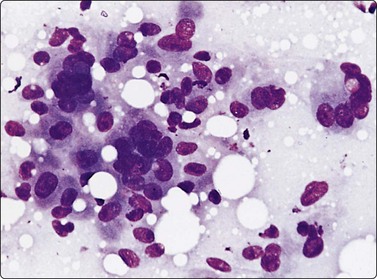
Fig. 14.19 Cutaneous histiocytoma
Loose cell cluster; ovoid nuclei, abundant granular and vacuolated cytoplasm; indistinct cell borders; note multinucleated giant cells (MGG, HP).
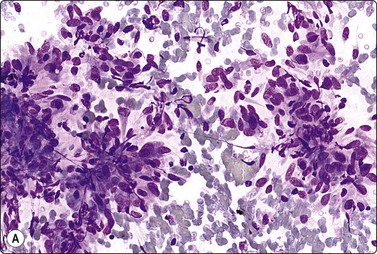
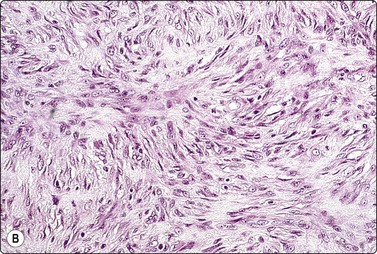
Fig. 14.20 Cutaneous fibrous histiocytoma
This example is highly cellular, predominantly of spindle cells; bland nuclei; storiform histologic pattern (A, MGG, IP; B, tissue section H&E, IP).
Criteria for diagnosis
This is a common intracutaneous lesion presenting as a small, often pigmented, poorly circumscribed induration, which can occur anywhere but most often on the leg. The diagnosis is usually clinically obvious, but larger and more deep-sited tumors may cause clinical concern. Adequate material can usually be obtained with a 25–27-gauge needle. There is a spectrum of patterns (cellular histiocytoma, fibrous histiocytoma, dermatofibroma) which is reflected in the number of cells in the smear and in the proportion of histiocytes, fibroblasts and collagen. Multinucleated histiocytic giant cells, often of the Touton type, xanthomatous histiocytes with foamy cytoplasm and iron pigment are commonly present in smears. The pattern can resemble that of other spindle cell lesions such as nodular fasciitis and dermatofibrosarcoma protuberans, but the clinical findings combined with the cytology allow a specific diagnosis in most cases.
The cytological findings in two cases of deep juvenile xanthogranuloma were reported by Grenko et al.63 Regarding atypical fibroxanthoma, see Chapter 15, p. 392.
Glomus tumor (Figs 14.21 and 14.22)
The tumor illustrated in Figure 14.21 presented as an asymptomatic subcutaneous nodule on the leg of a middle-aged woman. The FNB sample was heavily admixed with blood, but also contained cellular tissue fragments and single cells. The cells were mainly plump spindle cells with abundant eosinophilic cytoplasm but some cells appeared epithelioid. Nuclei were bland and oval. The FNB was reported as a benign skin adnexal tumor, not further specified. Immunostaining for smooth muscle actin was not done. Cytological findings have been reported in a few cases.64,65
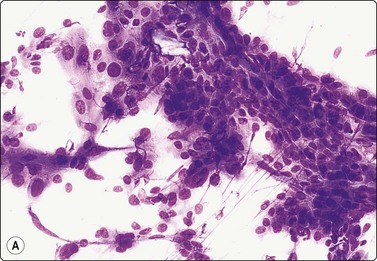
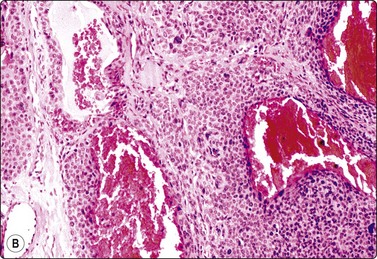
(A) Tissue fragment and dispersed cells; relatively abundant and dense eosinophilic cytoplasm; bland oval uniform nuclei; vascular structures not obvious (MGG, HP); (B) Corresponding tissue section (H&E, IP).
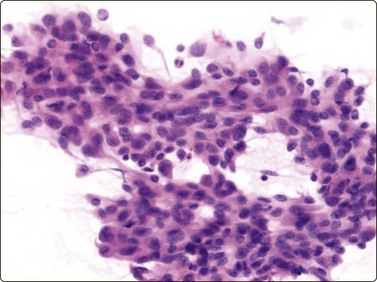
Fig. 14.22 Atypical glomus tumor
FNB of atypical (infiltrative) glomus tumor; cytomorphology resembles regular glomus tumor but shows increased cellularity and nuclear and cellular pleomorphism (H&E, HP).
There is a subset of glomus tumor termed atypical glomus tumor and rare cases of malignant glomus tumor occur. Smears from such lesions are highly cellular, showing nuclear and cellular pleomorphism (Fig. 14.22), but the morphology and immunoprofile otherwise resembles ususal glomus tumor.
The cytological findings in granular cell tumor are described in Chapters 7 and 15. These tumors are frequently located to the skin and subcutaneous tissues accessible to FNB. Immunostaining for protein S-100 easily confirms the FNB diagnosis (see Figs 7.51 and 15.49). Kalfa et al.66 reported two cases of primary cutaneous meningioma with FNB cytology.
Malignant non-epithelial tumors
Malignant melanoma (Figs 14.23-14.28)
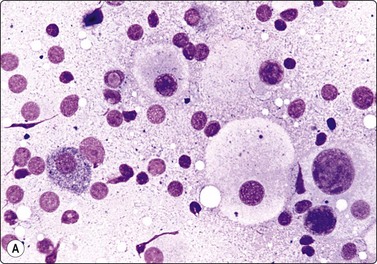
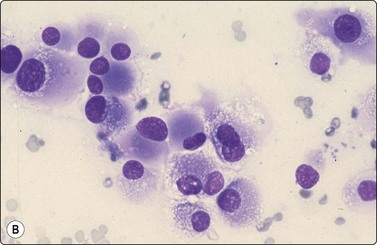
Fig. 14.23 Malignant melanoma, classic type
(A) Dispersed cells with fragile cytoplasm; marked anisokaryosis, one cell with intact cytoplasm and typical dust-like pigment; pigment granules dispersed in background; intranuclear vacuole (MGG, HP); (B) Dispersed plasmacytoid cells with paranuclear cytoplasmic condensation but no pigment (Diff-Quik, HP)
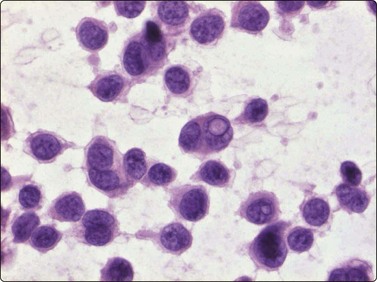
Fig.14.24 Malignant melanoma, classic type
FNB smear from ulcerated malignant melanoma. Diagnostic pattern of mainly dispersed malignant cells with scanty cytoplasm; note intranuclear vacuole (H&E, HP).
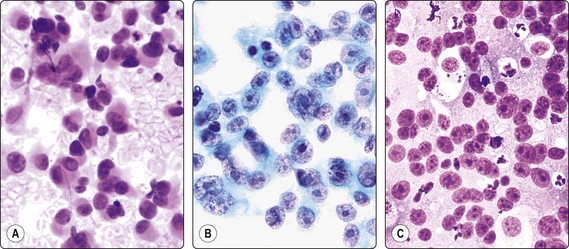
Fig. 14.25 Malignant melanoma, amelanotic, classic type
Mainly dispersed plasmacytoid malignant cells, vesicular nuclei, large nucleoli, no pigment (A, H&E; B, IP, Pap, HP); (C) Poorly differentiated squamous carcinoma, cytology resembling melanoma (H&E, HP).
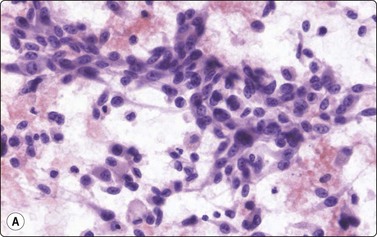
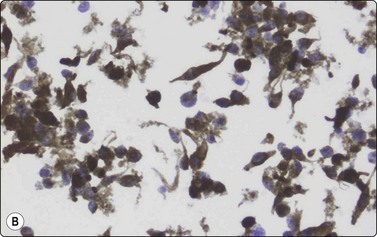
Fig. 14.26 Malignant melanoma, spindle cell type
(A) Highly cellular smear of poorly cohesive spindle cells with oval, moderately pleomorphic nuclei; no pigment seen (H&E, HP, Pap, IP); (B) Same lesion; immunostaining for HMB-45, liquid based preparation.
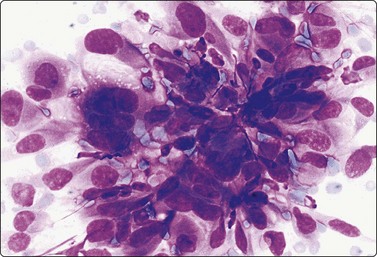
Fig. 14.27 Malignant melanoma, sarcoma-like
The cytological pattern in this local recurrence is indistinguishable from pleomorphic soft tissue sarcoma; cells were S-100 positive (MGG, HP).
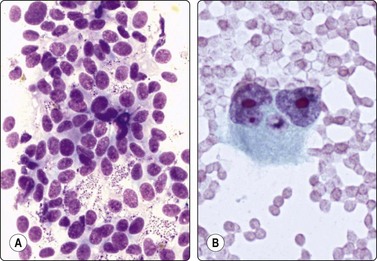
Fig. 14.28 Malignant melanoma, unusual patterns
(A) Bizarre binucleate tumor giant cell with large nucleoli (Pap, HP); (B) Poorly cohesive cells with a vaguely follicular pattern and relatively uniform nuclei resembling a neuroendocrine tumor (Diff-Quik, HP).
Criteria for diagnosis
FNB of primary malignant melanoma is not recommended, except for rapid confirmation of advanced tumors. Checking suspicious local recurrence or regional lymph nodes for metastases by FNB is of great value in the clinical management of melanoma.67-69 Correct identification of melanoma in other metastatic sites is another important application.
Typically, the cells have abundant cytoplasm and an eccentric nucleus and appear plasmacytoid. The cytoplasm is relatively dense but may be vacuolated, sometimes fragile, leaving some nuclei stripped. A darker condensation of the paranuclear cytoplasm (MGG, see Figs 5.60 and 14.23B) may be a clue in amelanotic melanoma. Nuclear hyperchromasia is often relatively uniform; it varies more from cell to cell in anaplastic carcinoma. It can be difficult to distinguish melanin pigment from lipofuscin and hemosiderin in routine stained smears, and special stains (formalin-induced fluorescence, Masson-Fontana) may be required. A fine dust-like intracytoplasmic pigment in tumor cells is characteristic of melanoma. In the large study by Murali et al.,70 melanin was demonstrated in only 27.6% of metastatic melanomas. The diagnosis can be confirmed by immunostaining (S-100 protein, Melan-A, HMB-45) or by the demonstration of pre-melanosomes by EM.
The criteria listed above apply mainly to the epithelioid or plasmacytoid, classic type of melanoma. However, the cytological pattern is extremely variable and melanoma can mimic many other tumors. Nasiell et al.71 described several subtypes: classic, carcinoma-like, spindle cell, lymphoma-like, undifferentiated, myxoid and clear cell. A small cell type could be added, resembling small cell carcinoma. Some of the subtypes are illustrated in Figures 14.23 to 14.28. Poorly differentiated amelanotic melanoma can closely resemble either anaplastic carcinoma or anaplastic lymphoma. Apoptotic cells and necrotic debris is much more a feature of poorly differentiated carcinoma than of melanoma.69 Spindle cell melanomas often have smaller, elongated, more uniform, and relatively bland nuclei (Fig. 14.26), but some are highly pleomorphic, mimicking high-grade pleomorphic sarcoma of soft tissues (Fig. 14.27). A vaguely follicular pattern with uniform cells may mimic a neuroendocrine tumor (Fig. 14.28A). Smears from malignant melanoma with a myxoid stromal reaction may be misinterpreted as a myxomatous soft tissue tumor. Desmoplastic melanoma presents special diagnostic problems in FNB samples.72
Access to immunocytochemistry is often necessary in the evaluation of unusual morphologic subtypes of malignant melanoma. A battery of markers including cytokeratin, EMA, LCA, S-100, Melan-A and HMB-45 (Fig. 14.26B) may be required to confirm the diagnosis.
Benign nevocellular tumors are rarely subjected to FNB. Figure 14.29 is of a postoperative scrape smear from a benign nevus. The cytology would be very difficult to distinguish from a nevoid melanoma.73
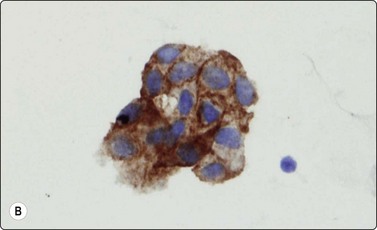
Dermatofibrosarcoma protuberans (DFSP) (Fig. 14.30)
The cytological findings in DFSP have been reported by Domanski and Gustafson.74 The yield is abundant of both tissue fragments of variably cohesive spindle cells and dispersed single cells and stripped nuclei. There is moderate anisokaryosis, but the nuclear chromatin is granular and nucleoli are small. A streaming arrangement that vaguely suggests a storiform pattern may be seen. The tumor cells stain positively for vimentin and CD34. The main differential diagnoses are low-grade fibrosarcoma and monophasic fibrosarcoma-like synovial sarcoma. The clinical presentation is an important clue to diagnosis
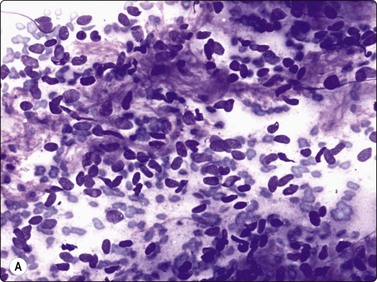
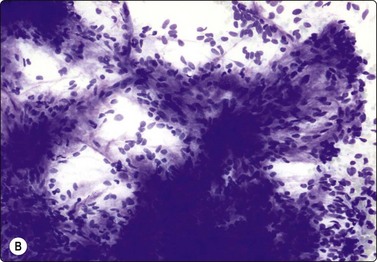
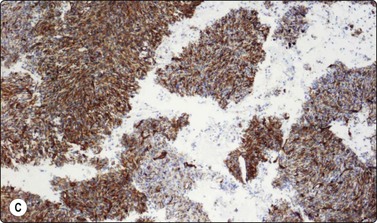
Fig. 14.30 Dermatofibrosarcoma protuberans (DFSP)
(A) Dispersed single cells and stripped nuclei with moderate anisokaryosis, but granular nuclear chromatin (MGG, MP); (B) A streaming arrangement that vaguely suggests a storiform pattern may be seen (MGG, MP); (C) The tumor cells stain positively for CD34 (cell block preparation, CD34).
Kaposi’s sarcoma is mainly seen in the skin but also occurs in other sites in HIV-positive patients. The cytological findings are described in Chapter 5 and in Chapter 18, p. 459. The cytology of a few other types of cutaneous vascular tumors has also been reported.3,75,76 Ancillary techniques, especially immunocytochemistry may be necessary to establish a cytologic diagnosis of malignant vascular tumor of the skin, as such tumors display a wide spectrum of cytomorphologic patterns (Fig. 14.31).
Metastatic malignancy; lymphoma
The skin, particularly of the scalp, is a common site for metastatic malignancy.1,2 A summary of the cytological findings in metastatic malignancy and guidelines for the identification of the primary site are given in Chapter 5. Clinical history, access to histological sections of the primary tumor, and immunostaining are the most important aids in diagnosis.
A mass in the skin or soft tissues may be the first manifestation of malignant lymphoma or it may be the first sign of recurrence of treated lymphoma. The cell patterns of cutaneous lymphoma have been beautifully illustrated in tissue sections by Isaacson and Norton.77 Cytological criteria for different types of lymphoma are listed in Chapter 5. Subtyping of lymphoma and distinction from cutaneous pseudolymphoma requires immune marker studies. The FNB findings in cutaneous T-cell lymphoma have been described in a few cases.6,78
1 Reyes CV, Thompson KS, Jensen JD, et al. Metastasis of unknown origin: the role of fine-needle aspiration cytology. Diagn Cytopathol. 1998;18:319-322.
2 Spitz DJ, Reddy V, Selvaggi SM, et al. Fine-needle aspiration of scalp lesions. Diagn Cytopathol. 2000;23:35-38.
3 Daskalopoulou D, Maounis N, Kokalis G, et al. The role of fine needle aspiration cytology in the diagnosis of primary skin tumors. Arch Anat Cytol Path. 1993;41:75-81.
4 Layfield LJ, Glasgow BJ. Aspiration biopsy cytology of primary cutaneous tumors. Acta Cytol. 1993;37:679-688.
5 Dey P, Das A, Radhika S, Nijhawan R. Cytology of primary skin tumors. Acta Cytol. 1996;40:708-713. 1996
6 Daskalopoulou D, Galanopoulou A, Statiropoulou P, et al. Cytologically interesting cases of primary skin tumors and tumor-like conditions indentified by fine-needle aspiration biopsy. Diagn Cytopathol. 1998;19:17-28.
7 Rege J, Shet T. Aspiration cytology in the diagnosis of primary tumors of skin adnexa. Acta Cytol. 2001;45:715-722.
8 Prayaga AK, Loya AC, Gottimukkala SR, et al. Cytologic features of primary malignant tumors of skin and adnexae. Acta Cytol. 2008;52:702-709.
9 Solano JG, Acosta-Cortega J, Perez-Guillermo M. Pilomatrixoma: never lower your guard. Diagn Cytopathol. 2007;35:457-458.
10 Gordon LA, Orell SR. Evaluation of cytodiagnosis of cutaneous basal cell carcinoma. J Am Acad Dermatol. 1984;11:1082-1086.
11 Dong H, Duncan LD. Cytologic findings in Demodex folliculitis. Diagn Cytopathol. 2006;34:232-234.
12 Bhatia A, Singh N, Arora VK, et al. Diagnosing granulomatous inflammation of the skin. A cytomorphologic approach based on evaluation of cellular reaction patterns. Acta Cytol. 1999;43:761-766.
13 Malik A, Bhatia A, Singh N, et al. Fine needle aspiration cytology of reactions in leprosy. Acta Cytol. 1999;43:771-776.
14 Rao IS, Singh MK, Gupta SD, et al. Utility of fine-needle aspiration cytology in the classification of leprosy. Diagn Cytopathol. 2001;24:317-321.
15 Garcia-Solano J, Garcia-Rojo B, Sanchez-Sanchez C, et al. Utility if fine-needle aspiration in the diagnosis of panniculitis. Diagn Cytopathol. 1998;18:425-430.
16 Handa U, Chhabra S, Mohan H. Epidermal inclusion cyst: cytomorphological features and differential diagnosis. Diagn Cytopathol. 2008;36:861-863.
17 Shet T, Rege J, Naik I. Cytodiagnosis of simple and proliferating tricholemmal cysts. Acta Cytol. 2001;45:582-588.
18 Kini jR, Kini H. Fine-needle aspiration cytology in the diagnosis of malignant proliferating trichilemmal tumor: Report of a case and review of the literature. Diagn Cytopathol. 2009;37:744-747.
19 Dodd LG, Layfield LJ. Fine-needle aspiration cytology of ganglion cysts. Diagn Cytopathol. 1996;15:377-381.
20 Punia RS, Gupta S, Handa U, et al. Fine needle aspiration cytology of bursal cyst. Acta Cytol. 2002;46:690-692.
21 Dodd LG, Major NM. Fine-needle aspiration cytology of articular and periarticular lesions. Cancer (Cancer Cytopathol). 2002;96:157-165.
22 Filho JSR, Soares MF, Wal R, et al. Fine-needle aspiration cytology of pulmonary rheumatoid nodule: case report and review of the major cytologic features. Diagn Cytopathol. 2002;26:150-153.
23 Bhadani PP, Sah SP, Sen R, et al. Diagnostic value of fine needle aspiration cytology in gouty tophi. A report of 7 cases. Acta Cytol. 2006;50:101-104.
24 Saleh HA, Baker H. Aspiration biopsy cytology of tumoral calcinosis. A case report. Acta Cytol. 2009;53:323-326.
25 Ashfaq R, Molberg KH, Vuitch F. Cutaneous endometriosis as a diagnostic pitfall of fine-needle aspiration cytology. A report of three cases. Acta Cytol. 1994;38:577-581.
26 Westermark P, Stenqvist B. A new method for the diagnosis of systemic amyloidosis. Arch Intern Med. 1973;132:522-523.
27 Guy CD, Jones CK. Abdominal fat pad aspiration biopsy for tissue confirmation of systemic amyloidosis: specificity, positive predictive value, and diagnostic pitfalls. Diagn Cytopathol. 2001;24:181-185.
28 Bondeson L, Lindholm K, Thorstenson S. Benign dermal eccrine cylindroma. A pitfall in the diagnosis of adenoid cystic carcinoma. Acta Cytol. 1983;27:326-328.
29 Kolda TF, Ardaman T-D, Schwartz MR. Eccrine spiradenoma mimicking adenoid cystic carcinoma on fine needle aspiration. A case report. Acta Cytol. 1997;41:852-858.
30 Mannion E, McLaren K, Al-Nafussi AI. Cytologic features of a cystic nodular hidradenoma: potential pitfalls in diagnosis. Cytopathology. 1995;6:100-103.
31 Gottschalk-Sabag S, Glick T. Fine-needle aspiration of nodular hidradenoma: a case report. Diagn Cytopathol. 1996;15:395-397.
32 Dubb M, Michelow P. Cytologic features of hidradenoma in fine needle aspiration biopsies. Acta Cytol. 2009;53:179-182.
33 Kumar N, Verma K. Clear cell hidradenoma simulating breast carcinoma: a diagnostic pitfall in fine-needle aspiration of breast. Diagn Cytopathol. 1996;15:70-72.
34 Gupta R, Singh S, Gupta K, Kudesia M. Clear-cell hidradenoma in a child: a diagnostic dilemma for the cytopathologist. Diagn Cytopathol. 2009;37:531-533.
35 Shimazaki H, Anzai M, Aida S, Endo H, et al. Trichoblastoma of the skin occurring in the breast. A case report. Acta Cytol. 2001;45:435-440.
36 Srinivasan R, Rajwanshi A, Padmanabhan V, et al. Fine needle aspiration cytology of chondroid syringoma and syringocystadenoma papilliferum. A report of two cases. Acta Cytol. 1993;37:535-538.
37 Rollins SD. Fine-needle aspiration diagnosis of a vulvar papillary hidradenoma. A case report. Diagn Cytopathol. 1994;10:60-61.
38 Kumar N, Verma K. Fine needle aspiration cytology of pilomatrixoma. Cytopathology. 1996;7:125-131.
39 Sánchez CS, Bascunana AG, Quirante FAP, et al. Mimics of pilomatrixomas in fine-needle aspirates. Diagn Cytopathol. 1996;14:75-83.
40 Lemos MM, Kindblom L-G, Meis-Kindblom JM, et al. Fine-needle aspiration features of pilomatrixoma. Cancer (Cancer Cytopathol). 2001;93:252-256.
41 Wang J, Cobb CJ, Martin SE, et al. Pilomatrixoma: clinicopathologic study of 51 cases with emphasis on cytologic features. Diagn Cytopathol. 2002;27:167-172.
42 Punia RS, Handa U, Mohan H. Fine needle aspiration cytology of eccrine acrospiroma. Acta Cytol. 2001;45:1083-1085.
43 Gottshalk-Sabag S, Glick T. Chondroid syringoma diagnosed by fine-needle aspiration. A case report. Diagn Cytopathol. 1994;10:152-155.
44 Rege J, Shet T. Aspiration cytology in the diagnosis of primary tumors of skin adnexa. Acta Cytol. 2001;45:715-722.
45 Dodd LG. Fine-needle aspiration cytology of adenoid (acantholytic) squamous-cell carcinoma. Diagn Cytopathol. 1995;12:168-172.
46 Malberger E, Tilinger R, Lichtig C. Diagnosis of basal-cell carcinoma with aspiration cytology. Acta Cytol. 1984;23:301-305.
47 Garcia-Solano J, Garcia-Rojo B, Sanchez-Sanchez C, et al. Basal-cell carcinoma: cytologic and immunocytochemical findings in fine-needle aspirates. Diagn Cytopathol. 1998;18:403-408.
48 Christensen E, Bofin A, Gudmundsdottir I, et al. Cytologic diagnosis of basal cell carcinoma and actinic keratosis using Papanicolaou and May-Grunwald-Giemsa stained cutaneous tissue smear. Cytopathol. 2008;19:316-322.
49 Henke AC, Wiemerslage SJ, Cohen MB. Cytology of metastatic cutaneous basal cell carcinoma. Diagn Cytopathol. 1998;19:113-115.
50 Calder CJ, Reynolds GM, Young FI, et al. Cytological features of pigmented BCC – a potential diagnostic pitfall. Cytopathology. 1996;7:132-135.
51 Masukawa T, Friedrich EGJr. Cytopathology of Paget’s disease of the vulva. Diagnostic abrasive cytology. Acta Cytol. 1978;22:476-478.
52 Sadhegi S, Pitman MB, Weir MM. Cytologic features of metastatic sebaceous carcinoma: report of two cases with comparison to three cases of basal cell carcinoma. Diagn Cytopathol. 1999;21:340-345.
53 Stern RC, Liu K, Dodd LG. Cytomorphologic features of sebaceous carcinoma on fine needle aspiration. Acta Cytol. 2000;44:760-764.
54 Gottschalk-Sabag S, Glick T. Sweat gland cancer diagnosed by fine needle aspiration. Cytopathology. 1996;7:66-69.
55 Bonadio J, Armstrong W, Gu M. Eccrine porocarcinoma. Report of a case with fine needle aspiration cytology, histopathology and immunohistochemistry. Acta Cytol. 2006;50:476-480.
56 Gangane N, Joshi D, Sharma SM. Cytomorphological diagnosis of malignant eccrine tumors: Report of two cases. Diagn Cytopathol. 2008;36:801-804.
57 Pai RR, Kini JR, Achar C, et al. Apocrine (cutaneous) sweat gland carcinoma of axilla with signet ring cells: a diagnostic dilemma on fine-needle aspiration cytology. Diagn Cytopathol. 2008;36:739-741.
58 Varsa E, Jordan SW. Fine needle aspiration cytology of malignant spiradenoma arising in congenital eccrine spiradenoma. Acta Cytol. 1990;34:275-277.
59 Reid-Nicholson M, Iyengar P, Friedlander MA, et al. Fine needle aspiration biopsy of primary mucinous carcinoma of the skin. A case report. Acta Cytol. 2006;50:317-322.
60 Kotru M, Manucha V, Singh UR. Cytologic and histologic features of primary mucinous adenocarcinoma of skin in the axilla. A case report. Acta Cytol. 2007;51:571-574.
61 Mishra K, Agarval S. Fine needle aspiration cytology of malignant chondroid syringoma. A case report. Acta Cytol. 1998;42:1155-1158.
62 Skoog L, Schmitt F, Tani E. Neuroendocrine (Merkel cell) carcinoma of the skin: immunocytochemical and cytomorphologic analysis on fine needle aspirates. Diagn Cytopathol. 1990;6:53-57.
63 Collins BT, Elmberger PG, Tani EM, et al. Fine-needle aspiration of Merkel cell carcinoma of the skin with cytomorphology and immunocytochemical correlation. Diagn Cytopathol. 1998;18:251-257.
64 Grenko RT, Sickel JZ, Abendroth CS, et al. Cytologic features of deep juvenile xanthogranuloma. Diagn Cytopathol. 1996;15:329-333.
65 Holck S, Bredesen JL. Solid glomus tumor presenting as an axillary mass. Report of a case with morphologic study, including cytologic characteristics. Acta Cytol. 1996;40:555-562.
66 Handa U, Palta A, Mohan H, Punia RPS. Aspiration cytology of glomus tumor. A case report. Acta Cytol. 2001;45:1073-1076.
67 Kalfa M, Daskalopoulou D, Markidou S. Fine needle aspiration (FNB) biopsy of primary cutaneous meningioma: report of two cases. Cytopathol. 1999;10:54-60.
68 Cangiarella JF, Symmans WF, Shapiro RL, et al. Aspiration biopsy and the clinical management of patients with malignant melanoma and palpable regional lymph nodes. Cancer (Cancer Cytopathol). 2000;90:162-166.
69 Saqi A, McGrath CM, Skovronsky D, Yu GH. Cytomorphologic features of fine-needle aspiration of metastatic and recurrent melanoma. Diagn Cytopathol. 2002;27:286-290.
70 Murali R, Doubrovsky A, Watson GF, et al. Diagnosis of metastatic melanoma by fine-needle biopsy: analysis of 2,204 cases. Am J Clin Pathol. 2007;127:385-397.
71 Nasiell K, Tani E, Skoog L. Fine needle aspiration cytology and immunocytochemistry of metastatic melanoma. Cytopathology. 1991;3:137-147.
72 Murali R, Loughman NT, McKenzie PR, et al. Cytologic features of metastatic and recurrent melanoma in patients with primary cutaneous desmoplastic melanoma. Am J Clin Pathol. 2008;130:715-723.
73 Rojo BG, Solano JG, Sánches CS, et al. On the limited value of fine-needle aspiration for the diagnosis of benign melanocytic proliferations of the skin. Diagn Cytopathol. 1998;19:441-445.
74 Domanski HA, Gustafson P. Cytologic features of primary, recurrent and metastatic dermatofibrosarcoma protuberans. Cancer. 2002;96:351-361.
75 Pérez-Guillermo M, Sola Pérez J, Garcia Rojo B, et al. Fine needle aspiration cytology of cutaneous vascular tumors. Cytopathology. 1993;3:213-244.
76 Ng WK, Collins RJ, Law D, et al. Cutaneous epithelioid angiosarcoma: a potential diagnostic trap for cytopathologists. Diagn Cytopathol. 1997;16:160-167.
77 Isaacson PG, Norton AJ. Extranodal lymphomas. Edinburgh: Churchill Livingstone; 1994. p. 131–91
78 Basce D, Kumar S, Skome DK, et al. Cutaneous T-cell lymphoma diagnosed by fine-needle aspiration. A case report with clinical, cytological and immunophenotypic features. Diagn Cytopathol. 1994;11:174-177.