MEDICAL MANAGEMENT
Although there is no specific way to prevent bladder cancer, modification of risk factors may help. Smoking cessation is the number one prevention strategy for bladder cancer. Reducing exposure to industrial or occupational carcinogens would also lower the incidence of this type of malignancy. Large total fluid intake may reduce the risk of bladder cancer by reducing the time of contact between carcinogens and the bladder epithelium.21,129
Vitamins and increasing consumption of fruits and vegetables initially showed benefit in reducing the risk for bladder cancer, but larger studies have not supported this relationship.85,130
Currently, screening of the general population for bladder cancer is not recommended, principally due to a lack of evidence for its effectiveness (large studies have not been continued for more than 10 years). But there is evidence to suggest that screening people at high risk (i.e., smokers, people with an occupational exposure) may be beneficial.127 Early detection of bladder cancer when still superficial can reduce mortality.
The best specific tests to use to screen for bladder cancer have not been determined, but in high-risk individuals, a urine dipstick test, evaluating for the presence of hematuria, and urine cytology are economical and noninvasive. Even with the presence of intermittent symptoms, these screening tests may miss many tumors. Some experts suggest cystoscopy, which visualizes the bladder and has an increased ability to detect tumors that are intermittently symptomatic. Cystoscopy, however, is more invasive and expensive. Further research is needed to determine the most appropriate candidates and tests to screen for bladder cancer.
The majority of bladder cancers are low-grade, superficial carcinomas that do not tend to metastasize. However, 50% to 90% of bladder cancers will recur, depending on the grade and stage. With recurrence, 10% to 50% will progress in stage or grade. Regular follow-up for early detection of cancer recurrence is important for anyone with a previous history of bladder cancer.
DIAGNOSIS.
Bladder cancer is seldom recognized in its preclinical stage but rather is detected once symptoms present, usually hematuria. Younger people with hematuria most often have a benign cause, such as a UTI or kidney or bladder stones.
Evaluation usually consists of a history, physical examination, urinalysis, and urine culture. If the cause is not determined with these measures, further evaluation should be done. For people over the age of 50 with hematuria (gross or microscopic), a history, physical examination, urinalysis, urine cytology, and cytoscopy should be performed. It is important to note risk factors the client may have for bladder cancer.
Cystoscopy allows the urologist to view the bladder for tumor and take a biopsy and cytology washings for evaluation. The cytology samples improve the ability to detect smaller tumors (especially if they are flat), since they are difficult to distinguish from normal bladder tissue.
If tumor is noted in the biopsy specimen, depth of involvement can be determined, which aids in determining staging and treatment. Staging of the tumor may involve ultrasound, CT scan, bone scan, or other tests. A number of urine-based markers, including telomerase and nuclear matrix protein 22 (NMP22), are under investigation for their potential usefulness in diagnosing transitional cell cancer and in monitoring for recurrence.77,107,169
STAGING.
The TNM staging system is a staging scheme based on the progressive depth of invasion of tumor into the bladder wall that has been used to assign treatment, assess outcomes, and predict prognosis. Cancer cells that are present along the surface of the bladder mucosa but have not yet invaded, also called in situ carcinomas, do not yet have the potential for metastasis and are classified as Tis.
Tumors that have penetrated the basement membrane and invaded into the submucosa/lamina propria but not the muscularis propria are categorized as T1. T2 tumors invade the muscularis propria, while T3 tumors invade through the wall into perivesical tissue. T4 tumors invade other organs and structures, such as the prostate, uterus, vagina, pelvic wall, or abdominal wall (Fig. 18-8).
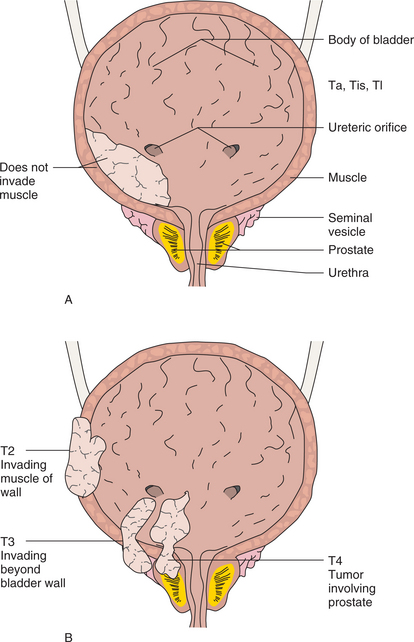
Figure 18-8 Bladder cancer staging using the TNM method. A, In Ta, Tis, and T1 tumors, cells do not invade muscle. B, If the muscle is involved, the tumor is staged as T2. T3 tumors invade beyond the bladder wall but do not involve other organs. T4 tumors are locally invasive to outside structures such as the prostate as shown or systemically with distant metastases (not shown).
Once tumor is invasive, it has the capacity to metastasize and commonly first reaches lymph nodes near the bladder. The presence of lymph node involvement, number of lymph nodes affected, and distance of involved lymph nodes from the bladder determine the N categorization (N0 for no involvement, N1 for nodes near the bladder, and N2 for nodes further away). Distant sites of metastasis include the liver, lung, and bone. The stage is determined by combining the T, N, and M status (e.g., stage 1 is T1, N0, M0). About 74% of bladder cancer is diagnosed as T1 or T2 tumors; 19% is T3; and 3% is T4.
TREATMENT.
Treatment of bladder cancer is determined on the basis of the stage of the tumor and the person’s general health. Surgery is the principal treatment for bladder cancer. Transurethral resection (TUR), surgery completed through the urethra using a rigid cystoscope called a resectoscope, is performed for early and superficial tumors. After the removal of the lesion, the tumor bed is treated either with a high-beam laser or fulguration (electric current used to destroy tumor tissue). Most clients can return home the same day or the next day. Complications include bleeding and discomfort. Long-term effects of repeated TUR include fibrosis of the bladder and loss of continence.
Cystectomy is performed for invasive bladder cancer. If the tumor is small, a partial cystectomy may be performed in order to salvage functioning bladder tissue.103 This approach is controversial, with some urologists preferring cystectomy even for selected clients with small tumor mass. A radical cystectomy is the surgery of choice for larger, invasive tumors or multiple tumors. This procedure removes the bladder and adjacent lymph nodes. The prostate is removed in men, and the uterus, ovaries, and a portion of the vagina are removed in women.
Following cystectomy, reconstructive surgery is performed to create a urine drainage system to compensate for the loss of the bladder. A urostomy procedure allows drainage of urine into a bag outside the abdomen. This is the least preferred long-term method of draining urine. The surgery that uses a short piece of small or large intestine to create a pouch or conduit from the ureters to the outside of the body is called an ileal conduit procedure.
Another surgical option is the creation of a continent diversion. In this procedure, the reserve pouch (intestine) has a valve. This valve allows urine to be stored until a catheter is placed to drain the urine. Newer reconstructive methods create a “neobladder” from intestine, which is then attached to the urethra. This surgery allows clients the ability to urinate normally, using a Valsalva maneuver (increasing intraabdominal pressure). The surgical complications from these types of procedures include infection, urine leakage, and obstruction. Sexual side effects are common, particularly for men. Impotence has been an issue with radical cystectomies in the past, but newer techniques have reduced the risk of nerve damage. If impotence does occur, function can improve with time. Generally, younger men (under the age of 60) are more likely to regain function than older men.
Chemotherapy is administered in two ways: intravesically or systemically. Intravesical chemotherapy is given through a catheter into the bladder, directly affecting the lining of the bladder. Chemotherapy agents are not absorbed into the deep layers of the bladder; therefore, intravesical chemotherapy is effective only for superficial cancers. Systemic chemotherapy is usually given in combination for invasive cancer (such as methotrexate, vinblastine, doxorubicin, and cisplatin). Radiotherapy in conjunction with concurrent chemotherapy is recommended less frequently because of the long-term consequences, although it is an option for treating clients unable to tolerate surgery because of health issues.
Another therapy option for treating low-stage bladder cancer is intravesical immunotherapy with bacillus Calmette-Guérin (BCG; a bacterium sometimes used to immunize people against tuberculosis). BCG is administered through a catheter into the bladder. The immune system responds to the BCG and becomes activated. These activated cells are then believed to recognize the cancer cells as foreign and destroy them. BCG is typically administered on a weekly basis for 6 weeks. BCG may reduce the risk of recurrence by as much as 50%. Side effects include flulike symptoms and a burning sensation in the bladder. Rarely, the bacterium can spread into the bloodstream, causing sepsis.
Stage 0 and 1 tumors are treated with TUR followed by intravesical BCG. If the tumor does not respond to the BCG, intravesical chemotherapy is administered, although either BCG or intravesical chemotherapy can be used first.68 However, over half of the people with stage 1 tumors will have a recurrence, and 20% to 30% will have a cancer that is invasive.
Stage 2 is treated with a radical cystectomy, with or without lymph node removal. Selected people may undergo a partial cystectomy with fulguration. Systemic chemotherapy may be given prior to or after surgery to treat any small micrometastases not seen during the staging process. A radical cystectomy is also the treatment for stage 3.
Chemotherapy may be added prior to surgery (neoadjuvant) to improve survival, but further investigations are needed to determine the appropriate timing for chemotherapy.94 Stage 4 therapy focuses on quality of life and slowing tumor growth. A radical cystectomy may be performed with chemotherapy if there are no distant metastases. Various combinations of therapy may be employed when distant metastases are present.
PROGNOSIS.
Despite the continued increase in the number of new cases occurring each year, the mortality attributed to bladder cancer has remained fairly stable. Stage and grade of the tumor are prognostic indicators for local failure. The 5-year survival rates with treatment are 84% for white males, 71% for black males, 76% for white females, and 51% for black females (most likely reflecting delay in diagnosis and inadequate care due to socioeconomic issues). Individuals with T1 tumors have a 90% 5-year survival, while for those with muscleinvasive tumors survival at 5 years is 50%; those with deep muscle invasion will go on to have metastatic disease within 2 years.5
Bladder cancer is sensitive to chemotherapy and immunotherapy but also has a high incidence of local recurrence, usually within the first 2 years. In approximately 30% of cases metastasis develops during the course of the disease, and 50% of individuals with muscle-invasive disease at the time of diagnosis already have distant metastases. Although rare, long-term survival with recurrent cancer can be achieved in some individuals. Continued improvements in the management of bladder cancer will improve the prognosis in the future.
Neurogenic Bladder Disorders
Voiding dysfunction associated with neurologic pathology is termed a neurogenic bladder disorder. There are many types of voiding dysfunction that can interfere with normal urine storage and coordinated, voluntary release.
Voiding dysfunction associated with neurologic pathology can be classified using one of many descriptive systems available. Each categorization scheme has advantages and disadvantages. For example, the Bors-Comarr Classification is well suited for clients with voiding dysfunction secondary to spinal cord injury but not as useful for clients with other problems.
Urodynamic classification correlates urodynamic findings and symptoms, while the International Continence Society Classification separates storage and voiding abnormalities and expands many of the urodynamic classification categories. The Lapides Classification is well known and helpful to nonurologists, so will be used as the classification system for this section. It correlates cystometry findings with clinical symptoms.
This classification system separates voiding dysfunction into five categories: (1) sensory neurogenic bladder, (2) motor paralytic bladder or motor neurogenic bladder, (3) uninhibited neurogenic bladder, (4) reflex neurogenic bladder, and (5) autonomous neurogenic bladder. This system provides a framework for understanding neurogenic bladder disorders, particularly for the nonurologist, but applies only to those disorders with a neurologic basis for pathology. Many clients may demonstrate a mixture of sensory and motor abnormalities, and symptoms may overlap between categories.
Incidence
Voiding dysfunction is a common problem associated with many types of neurologic diseases. In the United States, over 91,000 people are released from the hospital each year with a neurologic disease or spinal cord injury.30 Voiding dysfunction is costly and leads to significantly decreased quality of life, particularly in the older adult when long-term care is often considered for this problem in an otherwise healthy adult.
Etiologic Factors
The common disorders that can result in neurogenic bladder dysfunction include cerebrovascular accident, dementia, Parkinson’s disease, multiple sclerosis, and brain tumors. Neurogenic bladder dysfunction can also occur secondary to spinal cord lesions such as spinal cord injury, herniated intervertebral disc, vascular lesions, spinal cord tumors, and myelitis.
Local pelvic irritation can result in spasm of the external bladder sphincter, impairing urinary function. The local irritation can occur in the presence of vaginitis, perineal inflammation, urethral inflammation, and chronic prostatitis. Hypotonic (flaccid) bladder dysfunction can be secondary to meningomyelocele, spina bifida, and diabetes mellitus.
Pathogenesis
The process of micturition involves a complex interplay of nerves. Proper functioning of these nerves is needed for voiding to occur. The normal structures involved in micturition include the brain, the spinal cord, and nerves to the bladder. The type of voiding dysfunction that occurs is dependent on the underlying cause and which nerves are affected (Box 18-4).
The micturition reflex center is located in the brainstem. The efferent (exiting) neurons travel down the spinal cord in the reticulospinal tract to the detrusor muscle of the bladder. Parasympathetic nerves originate in the spinal cord at the level of S2, S3, and S4 and innervate the bladder wall via the pelvic nerve.
Preganglionic sympathetic nerves have their origin in the spinal cord at the levels of T10 through L2 and travel through the sympathetic chain ganglion to the bladder neck and fundus. The bladder neck contains the internal urethral sphincter (involuntary muscle). The external urethral sphincter (voluntary muscle) is innervated by the pudendal nerves, which originate in the spinal cord at the level of S2 through S4.
Damage to nerves involved in micturition can result in different types of voiding dysfunction. Cerebral injury (above the micturition reflex center) leads to loss of voluntary inhibition of voiding and a hyperreflexic bladder, but coordinated sphincter function is retained. This results in the ability to completely void, but because of the hyperreflexic bladder, incontinence occurs. This can be seen in brain tumors, cerebral palsy, cerebrovascular accidents, dementia, Parkinson’s disease, pernicious anemia, and Shy-Drager syndrome.
Lesions in the region of the micturition center to S2 result in loss of voluntary inhibition and coordinated sphincter activity. Since the sphincters are unable to coordinate their activity (the sphincter remains closed although bladder contractions occur), high pressure along with ureteral reflux result, termed dyssynergic sphincter function (detrusor-sphincter dyssynergia). This leads to urinary retention and incomplete voiding. Diseases that can result in this type of voiding problem include anterior spinal cord lesions, ischemia, multiple sclerosis, myelodysplasia, and trauma.
Complete spinal cord injury above T5 or T6 leads to autonomic dysreflexia (loss of sensation of bladder distension results in overdistension) and detrusor-sphincter dyssynergia. This type of injury also involves the sympathetic nerves and loss of sympathetic inhibition, leading to systemic sympathetic symptoms such as hypertension, facial flushing, perspiration, and headache. Since the vagal nerve is intact, bradycardia accompanies this syndrome.
Spinal cord lesions at the level of S2 and below lead to bladder areflexia and dysfunction of the external sphincter. Since parasympathetic nerves are not affected (ganglia are in or near the bladder wall), bladder tone is preserved, but bladder compliance decreases with time (secondary to repeated infection, fibrosis, and changes in innervation).
The external sphincter maintains some tone but the bladder neck does not relax, leading to obstructive problems when voiding (overflow incontinence). Acute transverse myelitis, diabetes, Guillain-Barré syndrome, herniated intervertebral disc, myelodysplasia, pelvic surgery, tabes dorsalis (syphilis), and trauma can cause this type of neurogenic bladder.
Diabetic bladder neuropathy occurs in 43% to 87% of people with type 1 diabetes mellitus and 60% to 75% of people with type 2 diabetes.63,104 The actual neurologic damage and symptoms vary among clients with diabetes and include diabetic cystopathy (impaired bladder sensation, increased postvoid residuals, increased bladder capacity, and decreased bladder contractility), detrusor overactivity, bladder outlet obstruction (seen in men), and urge and stress incontinence. Diabetic bladder neuropathy occurs when other diabetic complications are apparent (e.g., diabetic retinopathy, microalbuminuria).