The Male Genital/Reproductive System
The male genital or reproductive system is made up of the testes, epididymis, vas deferens, seminal vesicles, prostate gland, and penis (Fig. 19-1). These structures are susceptible to inflammatory disorders, neoplasms, and structural defects. Unless treating individuals with urinary incontinence, therapists do not typically treat people for primary reproductive system disease, but because of the incidence and nature of these disorders, an understanding of their clinical presentation is essential.
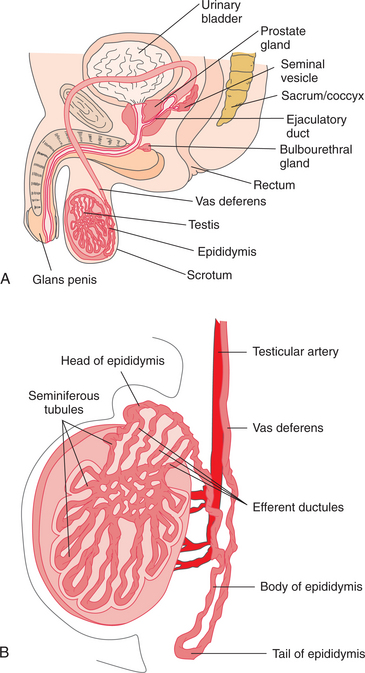
Figure 19-1 A, The male reproductive system. B, Internal structure of the testis and relationship of the testis to the epididymis. (A, modified from Bloom V, Fawcett DW: Textbook of histology, ed 10, Philadelphia, 1975, WB Saunders; B, redrawn from Guyton AC: Anatomy and physiology, Philadelphia, 1985, Saunders College Publishing.)
Prostate cancer is the most common cancer in males in the United States, and testicular cancer, although relatively rare, is on the rise and the most common cancer in males age 15 to 35 years. Benign prostatic hyperplasia (BPH) is one of the most common disorders of the aging male population. Because of the high incidence of these diseases, therapists will see clients with such a history, and the disorder or prescribed medical treatment could have a profound effect on the client’s clinical presentation and response to treatment.
The initial presenting symptom for some of these disorders could be back pain, a condition for which physical therapy care is often sought. An awareness of other symptoms besides pain and signs associated with urogenital system diseases may alert the therapist to other origins of the back pain. The presence of such symptoms warrants communication with a physician regarding the client’s status. Therapists have long been taught to ask clients with back pain questions regarding sexual function, the concern being the possible presence of cauda equina syndrome. An awareness of the more probable causes of sexual dysfunction helps the therapist determine the relevance and potentially urgent nature of a client’s complaints. The disorders discussed in this chapter are those of the highest incidence or of greatest implications for therapists.
AGING AND THE MALE REPRODUCTIVE SYSTEM
The reproductive system undergoes degenerative changes associated with aging that can affect sexual function. The testes become smaller, with thickening of the seminiferous tubules impeding sperm production; the prostate gland enlarges, potentially affecting urine outflow; and sclerotic changes occur in the local blood vessels, possibly resulting in sexual dysfunction (erectile dysfunction [ED]/impotence).
The age-related decrease in male sex hormone levels (androgen deficiency) also has significant local and systemic effects. Protein synthesis, salt and water balance, bone growth, and homeostasis and cardiovascular function are all under the influence of these hormones. A decline in bioavailable testosterone has been clearly correlated with age-related memory changes; decreasing sexual interest; and physical changes, including decreased strength, body mass, and bone density.98,108,165
Arteriosclerosis of the blood vessels resulting in peripheral vascular disease can also affect vessels supplying the penis. Sexual (erectile) dysfunction can be an indicator of ischemic heart disease and should not be ignored as a symptom requiring medical evaluation.
DISORDERS OF THE PROSTATE
Overview
Clinically, the diagnosis of “prostatitis” refers to multiple disorders that cause pelvic pain and discomfort ranging from acute bacterial infection to complex conditions that may not necessarily be caused by prostatic inflammation. Because the traditional etiologic-based classification system did not always correlate symptoms with what worked in treatment, the National Institutes of Health (NIH) proposed a new classification of prostatitis.81 Inflammation of the prostate gland can be acute bacterial, chronic bacterial, chronic prostatitis/chronic pelvic pain syndrome, or asymptomatic inflammatory prostatitis.
Acute bacterial prostatitis (category I) is the least common of the four types but also the easiest to diagnose and treat effectively. Men with this disease often have chills, fever, pain in the lower back and genital area, urinary frequency and urgency often at night, burning or painful urination, body aches, and a demonstrable infection of the urinary tract as evidenced by white blood cells and bacteria in the urine. The treatment is an appropriate antibiotic.109
Chronic bacterial prostatitis (category II), also relatively uncommon, is acute prostatitis associated with an underlying defect in the prostate, which becomes a focal point for bacterial persistence in the urinary tract. Effective treatment usually requires identifying and removing the defect and then treating the infection with antibiotics. However, antibiotics often do not cure this condition.109
Chronic prostatitis/chronic pelvic pain syndrome (CP/CPPS), previously known as nonbacterial prostatitis, is classified as category III and the most common (more than 90% of cases) but least understood form of prostatitis. It is found in men of any age, its symptoms go away and then return without warning, and it may be inflammatory or noninflammatory. In the inflammatory form (category IIIA), urine, semen, and other fluids from the prostate show no evidence of a known infecting organism but do contain the kinds of cells the body usually produces to fight infection.
In the noninflammatory form (category IIIB), no evidence of inflammation, including white blood cells, is present in the semen.109 Because there appears to be no correlation between the presence of leukocytes and symptoms, classification into categories IIIA and IIIB is controversial. An α-blocker may be used to relax the muscle tissue in the prostate. No single solution works for everyone with this condition.109
Asymptomatic inflammatory prostatitis (category IV) is the diagnosis when the man does not complain of pain or discomfort but has white blood cells in his semen. Doctors usually find this form of prostatitis when looking for causes of infertility or when testing for prostate cancer.109
These conditions are typically preceded by lower urinary tract infections (UTIs) (see Chapter 18). Therapists are least likely to encounter problems associated with acute prostatitis because the symptoms are usually severe enough that physician contact is initiated by the client (or family), and rehabilitation is typically placed on hold until the antibiotic therapy is successful.
On the other hand, therapists are likely to encounter chronic bacterial prostatitis, which is a much more subtle disorder, and complete resolution with treatment is often difficult to obtain. CPPS is common and may benefit by pelvic floor reeducation with a physical therapist.12
Incidence and Risk Factors
Prostatitis affects millions of men, and about half of all men have at least one episode during their lifetime. The prevalence is highest among men in their forties; older men especially prone to UTI develop prostatitis, but this condition can affect men of all ages. UTIs are among the more common infections, afflicting the male population secondary to bladder outlet obstruction associated with BPH. As the infection ascends through the urogenital system, the prostate can become involved. UTIs generally occur with much higher frequency in women than men, but men can still be affected and should not view UTI as a “woman’s disease.”
Besides a history of UTI and BPH, recent urethral catheterization or instrumentation and multiple sexual partners increase the risk of developing prostatitis. Some men find that stress, emotional factors, alcohol, spicy foods, or caffeine triggers episodes. Poorly controlled diabetes mellitus increases the risk of UTI and prostatitis developing because the increased urine glucose provides the substrate for bacterial growth.
Etiologic Factors and Pathogenesis
The etiology of prostatitis appears to be multifactorial. The etiology of the most common form of prostatitis, CP/CPPS, is poorly understood. Despite the assumption that there is an infectious or inflammatory process, this theory has not been proven. Autoimmunity may play a role in chronic prostatitis, since up to one-third of men with prostatitis have elevated levels of specific molecules that regulate the inflammatory response. Another hypothesis is that many of the men affected have a pelvic floor spasm associated with undiagnosed pelvic floor disorders mimicking prostatitis. The NIH is funding several prostatitis studies to examine this further.
The most commonly found pathogens associated with chronic bacterial infections are the gram-negative enterobacteria such as Escherichia coli, Proteus mirabilis, Klebsiella pneumoniae, and Pseudomonas aeruginosa. The pathogens associated with acute prostatitis include E. coli, pseudomonads, staphylococci, and streptococci. Although controversial, other infectious agents, such as gonococci, Ureaplasma species, chlamydiae, and mycoplasmata, are possible etiologic factors.
Clinical Manifestations
Table 19-1 contains a summary of symptoms associated with the four categories of prostatitis. Acute prostatitis occurs suddenly with severe symptoms. Fever, chills, and UTI are typical, along with pelvic discomfort. Blood in the urine, urinary retention, and urinary blockage are frequent, and there may be a steep rise in prostate-specific antigen (PSA), a marker for prostate cancer.
Table 19-1
Clinical Manifestations of Prostatitis
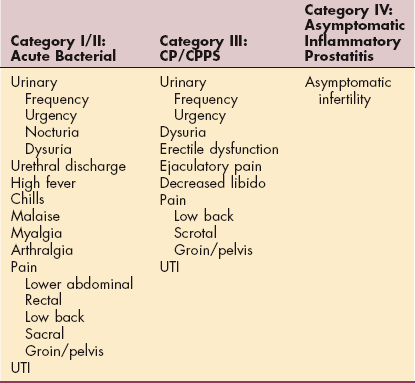
CP/CPPS, Chronic prostatitis/chronic pelvic pain syndrome; UTI, urinary tract infection.
If not treated, acute prostatitis may become chronic. Men who experience chronic prostatitis are plagued by persistent low-grade symptoms with flare-ups of pelvic pain, voiding problems, and sexual dysfunction (e.g., erectile dysfunction, ejaculatory pain, and a decline in emotional well-being).130 The pelvic pain is usually located behind the scrotum or in the perineum, the area between the rectum and testicles. This pain is usually made worse by sitting down and may be relieved by ejaculation. Pain in the tip of the penis may be experienced.
MEDICAL MANAGEMENT
Urinalysis, analysis of an expressed prostatic specimen, and a digital rectal examination (DRE), in which the physician palpates manually for prostate changes or enlargement, are used to establish a diagnosis of prostatitis. The DRE may reveal a swollen, tender, and warm prostate. Computed tomography (CT) and transrectal ultrasonography (TRUS) provide anatomic details often needed in the evaluation of these individuals. Urodynamic testing may help identify voiding dysfunction, especially in men with pelvic or perineal pain and voiding symptoms.59
Prostatitis is differentiated in part from BPH and prostate cancer by the presence of pain (rarely present in BPH or cancer) and by age (more than half of all men with prostatitis are younger than 45 years of age). Current NIH guidelines, which stratify prostatitis into the four categories of prostatitis, advise using the NIH Chronic Prostatitis Symptom Index (NIH-CPSI) to assess symptoms and plan treatment. The CPSI provides scores for pain, urinary symptoms, and quality of life.12,88,110
TREATMENT.
Treatment depends on the type of prostatitis present. Acute prostatitis (category I) is treated with antibiotics. For men who are unable to empty their bladders, suprapubic drainage is preferred over an indwelling urethral catheter.
Treatment of category II (chronic bacterial) prostatitis also involves antibiotics to eliminate the organism producing the infection. Men with frequent recurrences may be placed on antibiotic prophylaxis for 3 to 6 months and have their clinical course reassessed. Treatment of bladder outlet obstruction, which may impair bladder emptying, is also important.
Optimal treatment for CP/CPPS is unknown. Treatment with antibiotics and antiinflammatories often fail. Multimodal treatment may be best with biofeedback, pelvic floor reeducation, and α-blocker therapy (to relax smooth muscle of the prostate and at the base of the bladder). Newly diagnosed cases or those who have not been treated before are more likely to respond to α-blocker therapy compared to chronic refractory cases. Longer courses of treatment (4 to 6 months) seem to work better than shorter courses.82
Because the cause of nonbacterial prostatitis is unknown, treatment is often given to simply provide symptom control and relief. Antiinflammatory medications are administered. Antibiotics, fluoroquinolone, and antifungal agents may also be given. For unknown reasons, about half of men with nonbacterial prostatitis respond to antibiotics; antibiotics are used to treat bacterial prostatitis.
Treatment of chronic bacterial prostatitis can be equally as difficult, as the antibiotic agents have difficulty penetrating the chronically inflamed prostate. Drug therapy of 4 to 6 months’ duration may be used in an attempt to treat this infection. Transurethral prostatectomy (TURP) may be indicated if the disease is not cured with medications.
Other treatments for CP/CPPS range from medications to treat neuropathic pain, anticholinergic medications, phytotherapies (e.g., herbal treatment with quercetin or bee pollen), physical therapy, and in rare cases, surgery to treat bladder neck obstruction. For category IV prostatitis, no treatment is recommended.
Benign Prostatic Hyperplasia
Benign prostatic hyperplasia (BPH) is an age-related nonmalignant enlargement of the prostate gland.
Incidence and Risk Factors
Of men age 50 years and older, 75% experience symptoms of prostate enlargement.139 The disease is rarely noted in men under age 40 and tends to become symptomatic after age 50 years. Besides age, geography and ethnicity are important factors in the incidence of this disease. BPH is found most often in the United States and western Europe and least often in the Far East. The incidence of BPH is also higher in blacks than in whites. Drinking moderate amounts (one or two alcoholic drinks per day) is associated with a reduced risk of BPH. Cigarette smoking increases the risk for BPH-like symptoms, possibly because bladder irritation caused by cigarette smoke may heighten the urgency and frequency of urination.
Dietary risk factors are under investigation. Milk and dairy products have been related to an increase in BPH but not all studies confirm this association; fruit has been reported to have a protective effect against this condition. Other food groups, including meat and vegetables, have been analyzed in relation to BPH, but the results have been inconsistent. More frequent consumption of prepared cereals, bread, eggs, and poultry may be a risk factor.18
Pathogenesis
The prostate gland, a muscular part of the male reproductive system, is normally about the size and shape of a walnut, normally weighing about 20 g. It is located in front of the rectum and just below the bladder (see Fig. 19-1). It consists of five lobes that surround the urethra and produces seminal fluid to nourish and transport semen. Throughout life, the body constantly replaces old, dying prostate cells with new ones. For reasons still not completely clear, as men age, the ratio of new prostate cells to old prostate cells shifts in favor of lower cell death. By age 70 years, the hypertrophic prostate can weigh up to 200 g, resulting in significant urethral obstruction, decreased urine storage capability, and difficulty emptying the bladder.
Although the cause is unknown, changes in hormone balance associated with aging may be responsible for the development of BPH. The condition is commonly referred to as benign prostatic hypertrophy, but the pathologic changes are marked by hyperplasia, not hypertrophy. Multiple prostatic nodules develop, resulting from the proliferation of epithelial cells, smooth muscle cells, and stromal fibroblasts of the gland. These nodules initially develop in the periurethral region of the prostate as opposed to the periphery of the gland (Fig. 19-2). The lumen of the urethra becomes progressively narrowed.
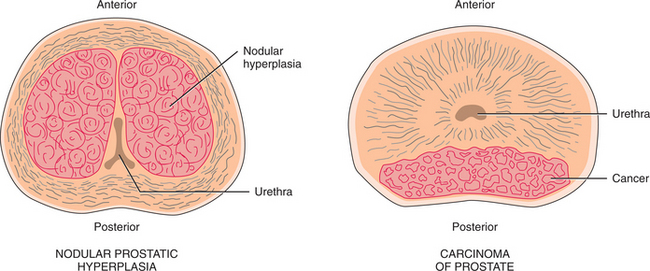
Figure 19-2 In benign prostatic hyperplasia (BPH), the nodules initially develop in the periurethral region, compressing the urethra. Cancer of the prostate typically develops initially in the periphery of the gland.
Both androgens and estrogens contribute to the hyperplasia of this condition. Dihydrotestosterone (DHT), the biologically active metabolite of testosterone, is thought to be the primary mediator of hyperplasia, whereas estrogens sensitize the prostatic tissue to the growth-producing effects of DHT. The increased levels of estrogen that occur with aging may enhance the action of androgens at this point in the life cycle.
Clinical Manifestations
The clinical presentation of BPH is related to secondary involvement of the urethra. As men age, the prostate slowly grows larger. If it becomes too large, urine flow can become restricted. The nodular hyperplasia results in a narrowing of the urethra, producing urinary outflow obstruction. The client may note a decreased caliber and force of the urine stream and difficulty initiating or continuing the urine stream so that only small amounts of urine are released.
In addition, residual urine in the bladder results in urinary frequency, which is particularly troublesome at night (nocturia). Quality of life and emotional function can be adversely affected by fatigue, micturition problems, and sleeping disturbances.70 As the urethral obstruction progresses, the risk of developing UTIs, marked bladder distention with destructive bladder wall changes, hydroureter, and hydronephrosis increases (Fig. 19-3).
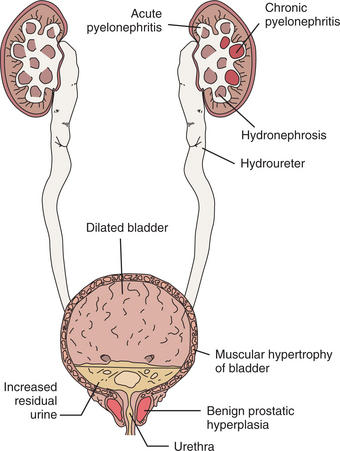
Figure 19-3 A cascade of destructive events potentially associated with advanced benign prostatic hyperplasia (BPH). Viewing Fig. 19-1, A, the relationship of the prostate located at the base of the bladder, surrounding a part of the urethra can be visualized. As the prostate enlarges, the urethra becomes obstructed, interfering with the normal flow of urine.
Hydroureter refers to the ureteral wall as it becomes severely stretched secondary to the bladder outflow obstruction, which increases the pressure in a retrograde direction. The ureteral wall can become stretched to the point where it loses the ability to undergo peristaltic contractions. With hydronephrosis, urine-filled dilation of the renal pelvis and calices occurs; destruction of renal tissue may occur.
Ultimately, renal failure and death may occur if treatment is not initiated. Other urinary symptoms associated with the later stages of the disease include urge incontinence, terminal urinary dribbling, urgency, hematuria, and dysuria.
MEDICAL MANAGEMENT
There has been some controversy over the use of saw palmetto, lycopene, and tomato products as possible antioxidant prevention of BPH. The general consensus is that these natural products will not do any harm but they may not help either.
DIAGNOSIS.
Correlation of the history, palpation findings (DRE), and urodynamic test results (flow rate and force of stream) typically give rise to the diagnosis of BPH and indicate the choice of treatment. Regarding palpation, the bladder may be palpable as urinary retention progresses. In addition, a smooth, rubbery enlargement of the prostate may be noted during DRE, although the perceived size of the prostate does not always correlate with the degree of urethral compression.
PSA is an additional test that helps predict the natural course of BPH. Higher levels of PSA are linked with a greater risk of future prostate growth and subsequent complications. Urinalysis is usually done to check for hematuria and UTI. The most commonly employed uro- dynamic test for the assessment of BPH is uroflowmetry. The urine flow rate and the force of the urine stream are measured. It is generally agreed that a peak urine flow rate of less than 10 ml/sec is suggestive of obstruction.
Uroflowmetry by itself is a screening modality, not diagnostic, because the urinary obstruction could be occurring at sites other than at the prostate gland. In addition, diagnostic ultrasound, magnetic resonance imaging (MRI), and abdominal radiographs may be used to evaluate the size and length of the urethra, the size and configuration of the prostate, and the bladder capacity (Fig. 19-4). The American Urological Association has also developed a self-administered screening tool used to determine the frequency and severity of urinary symptoms.
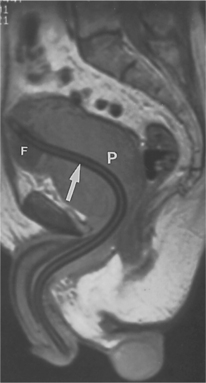
Figure 19-4 Magnetic resonance image (MRI) demonstrating benign nodular hyperplasia with enlargement of the prostate gland (P). The arrow is pointing to a Foley catheter in place. The inflated Foley balloon (F) indicates the level of the bladder neck. (From Grainger RG: Grainger & Allison’s diagnostic radiology: a textbook of medical imaging, ed 4, Philadelphia, 2001, Churchill Livingston.)
TREATMENT.
If symptoms related to BPH are mild, the condition is often just monitored because the clinical status of the disorder may stabilize or even improve. Aggressive treatment is indicated, however, if the more severe symptoms of obstruction develop.
Symptoms suggesting advanced disease include urine retention, incontinence, hematuria, and chronic UTIs. The goals of treatment include providing client comfort and avoiding serious renal damage. For those with moderate-to-severe symptoms, the two medical treatment approaches for BPH are pharmacologic and surgical.
Medication.
In most cases, symptoms associated with BPH can be controlled with medications. One trial, known as Medical Therapy Of Prostatic Symptoms (MTOPS), found that combining medications produces better results than administering only one drug (monotherapy).75
Pharmacologic agents are used to treat BPH by shrinking glandular tissue (5-α-reductase inhibitors) and relaxing smooth muscle tissue of the prostate, bladder neck, and urethra (α-adrenergic blockers). The 5-α-reductase inhibitors address the hormonal causes of BPH by preventing the enzyme 5-α-reductase from converting testosterone into DHT (DHT prompts glandular tissue to develop). As a result, the prostate shrinks by as much as 20%. This is a gradual process that can take up to a year to obtain maximum benefit.
The α-adrenergic receptors are located in the muscle fibers of the adenoma and prostate capsule. The resultant smooth muscle relaxation decreases pressure on the urethra, enhancing urinary flow. Phytopharmaceuticals (e.g., substrates from the saw palmetto plant) used for the management of BPH represent up to 80% of all prescriptions in many European countries.77 Earlier studies in the United States showed significant effects of saw palmetto on epithelial contraction, urinary flow rates, and improved symptoms compared with placebos,51,94 but more recent studies have not shown similar benefits.7,15,58
Androgen suppression drug therapy can also be used to block the synthesis and action of testosterone, including DHT. A 20% to 30% reduction in prostate volume can be noted. This can result in a lessening of the severity of symptoms and improvement in the objective criteria related to urinary obstruction although this can take 3 to 6 months.
Surgery.
Not everyone with BPH responds adequately to medications. In such cases, BPH can progress to the point where surgery is required. The goal of surgical intervention is to alleviate the obstruction to urine flow. TURP is considered the gold standard in surgical treatments for BPH. A long tube with a miniature camera on the tip is threaded up the urethra into the bladder, allowing visual inspection of both of these structures. When the excess prostate tissue is identified, a surgical instrument is threaded through this tube, and resection of excess prostate tissue is performed to enlarge the urinary channel.
TURP is very successful in relieving symptoms and improving quality of life, but some drawbacks are evident (e.g., requires a general anesthetic and hospital stay and may be accompanied by side effects such as bleeding, incontinence, ED/impotence, or retrograde ejaculation in which an ejaculation goes back into the bladder). An alternative technique is transurethral incision of the prostate (TUIP). In the case of TUIP, incisions in the muscle wall of the prostate and bladder neck allow for an outward expansion of the gland, relieving some of the pressure on the urethra. TURP is preferable when there is a large gland, severe and recurrent gross hematuria, and prostatitis; the goal is to remove infected tissue and calculi.
Thermotherapy by either transurethral microwave therapy (TUMT), such as Prostatron, or laser surgery through laser-induced thermotherapy, such as Green Light laser or indigo laser, has been developed over the past 10 years to provide effective alternatives to surgical management. Lasers are used for rapid incision and vaporization of the prostate with minimal bleeding. This treatment procedure is indicated in the case of small-size BPH and in the presence of risk factors, such as heart disease or anticoagulation therapy, or for men who do not tolerate medications well. This modality restores spontaneous urine flow by destroying diseased prostate tissue.
Results of long-term studies are not available at this time, but short-term results demonstrate that laser therapy is at least as safe and effective in relieving BPH symptoms as TURP and may provide reduced morbidity.49 The potential advantages of these procedures over TURP and TUIP are shorter operative time, usually without hospitalization; minimal bleeding; and decreased incidence of postoperative retrograde ejaculation and bladder neck contracture.
Potential disadvantages of laser treatment include postoperative urinary retention, no tissue being available for histologic study, and less than optimal prospects for treating large lesions. This technique may be associated with a high recurrence rate requiring additional treatment.160 In the case of laser, the larger the lesion, the more passes are required, increasing the associated risks.164 The introduction of an integrated system of computer, robotics, and laser technology for prostate resection may be the intervention of the future.65
Other.
Other procedures include water-induced thermotherapy. Heated water is injected into a balloon inserted into the urethra. The heat destroys excess prostate tissue. The procedure is done on an outpatient basis, but a catheter must be worn for 1 to 3 weeks afterward. Water-induced thermotherapy is not advised for men who have had previous prostate, pelvic, or rectal surgery. A past history of pelvic radiation for prostate cancer is another exclusion factor.
Transurethral ethanol ablation of the prostate (TEAP) involves injecting ethanol (a type of alcohol) through the urethra into the prostate to destroy excess prostate tissue. Both of these procedures are designed to improve peak flow rate, but long-term data are not yet available.
Botulinum toxin (BTX), or Botox, has been used successfully in the treatment of BPH. Botox injected directly into the prostate has been shown to reduce lower urinary tract symptoms and improve erectile function. The mechanism by which BTX can reduce prostate volume and intravesicular resistance remains unclear.6 Promising preliminary results have been reported in studies combining medications with BTX.24,152 The use of botulinum neurotoxin in the prostate is currently considered Food and Drug Administration (FDA) off-label use.
PROGNOSIS.
BPH can contribute to chronic problems with lower urinary tract symptoms, ED, and decreased quality of life. Treatment is reserved for symptomatic presentation, but there is considerable risk of progression with possible development of prostate cancer.
Prostate Cancer
Adenocarcinoma of the prostate arises from the glandular cells and accounts for 98% of primary prostatic tumors. Ductal and transitional cell carcinomas make up the remainder of the tumors. Prostate cancer usually starts in the outer portion of the prostate and spreads inwardly and then beyond the gland with metastases in more advanced stages.
Incidence
Prostate cancer, the most frequently diagnosed visceral malignancy in American men, is the second most common cause of male death from cancer. One in five American men will develop prostate cancer. Approximately 218,890 men were newly diagnosed in 2007 (up from 189,000 in 2002).71
The number of new cases of prostate cancer in the last decade represents an increase of 200%.72 However, the evidence suggests the use of PSA screening as a means of early detection may be the reason for the increased incidence. Widespread implementation of prostate cancer screening in the United States has led to more cancers being detected but at an earlier stage with fewer cases of metastases at diagnosis and a decrease in mortality rate.151
Autopsy studies indicate that occult prostate cancer is present in 25% of men age 60 to 69 years, in 40% of men age 70 to 79 years, and in more than 50% of men age 80 years and older.131 Men less than 50 years of age make up less than 1% of those with prostate cancer. Prostate cancer incidence and mortality vary strikingly among ethnic and national groups with a particular propensity for African Americans.42
In the United States the incidence of prostate cancer is approximately 60% higher in African American men than in European American men; the mortality rate from the disease is more than twice as high among African Americans.124 Screening rates for African American men with a positive family history of prostate cancer are significantly lower than in Caucasians, especially in older men ages 60 to 70 years.169
Risk Factors
Box 19-1 lists risk factors related to prostate cancer. Prostate cancer is a disease of the aging male, but evidence exists that genetic, environmental, and social factors jointly and in combination contribute to observed differences in various populations. Geographically, the highest frequencies of prostate cancer are found in the United States and northwestern Europe (Scandinavian countries), whereas the lowest are found in Mexico, Greece, and Japan. As mentioned, African Americans have twice the risk of non-Hispanic whites, which is attributed to traditional socioeconomic, clinical, and pathologic factors.66
Although no genetic marker has been found, a familial history of prostate cancer is a risk factor. A significant increase in risk has been estimated for men who have both a first-and second-degree relative with the disease.141 If a close relative has prostate cancer, a man’s risk of the disease doubles; with two relatives, his risk increases fivefold; and with three close relatives, the risk is about 97%.111 In addition, mortality from prostate cancer is believed to be three times greater in relatives of men with prostate cancer compared with those without such a family history.
Exposure to cadmium through welding, electroplating, alkaline battery production, farming, typesetting, and ship fitting places men at higher risk of prostate cancer. High dietary fat intake has been correlated with prostate cancer,54,114 with a protective effect from higher intake of tomato sauce and physical activity.55,56 Other risk factors still under investigation include having a vasectomy, eating red meat, vitamin deficiency (vitamins D and E), and frequency of ejaculations. The role of frequency of ejaculations (either through sexual intercourse or masturbation) in developing prostate cancer has been questioned but results of a study involving 30,000 men suggest that ejaculation frequency is not related to increased risk of prostate cancer.83
Etiologic Factors
Although the precise cause of prostate cancer is unknown, a strong endocrine system link is theorized. Androgens in particular have been implicated based on the androgenic control of normal growth and development of the prostate and the fact that males castrated before puberty do not develop prostate cancer or BPH. In addition, the responsiveness of prostate cancer to surgical castration and estrogen therapy supports this theory.125 The higher incidence of cancer in African Americans is correlated with a 15% higher serum testosterone level in this same population.39
The development of prostate cancer reflects a complex sequence of biologic and molecular events. Even though it is difficult to identify genes that predispose to prostate cancer because of late age at diagnosis, several inheritable and somatic genetic changes have been identified, including a prostate cancer susceptibility locus with linkage to a locus on chromosome 17p.163 However, this gene is only responsible for between 2% and 5% of all prostate cancers, suggesting that additional genes contribute to this disease.113,148 The EphB2 gene was recently implicated as a prostate cancer tumor suppressor gene, with somatic inactivating mutations occurring in approximately 10% of sporadic tumors. This may be an important factor in African American men with a positive family history of prostate cancer.76
A complementary deoxyribonucleic acid (cDNA) fragment (C13) has been identified that is down-regulated in malignant prostate tissues, suggesting that this gene encodes a protein that may have a tumor-or metastasis-suppressing function in prostate tissue.79,132
A newly identified virus, tentatively called XMRV, may be associated with the development of prostate cancer in genetically susceptible men. XMRV is closely related to a virus that causes leukemia in mice and is a newly identified infectious agent in humans. Researchers theorize that XMRV could be sexually transmitted, leading to chronic inflammation and cancer similar to how human papillomavirus triggers cervical cancer.32
Pathogenesis
Most prostatic adenocarcinomas are characterized by small-to moderate-size disorganized glands that infiltrate the stroma of the prostate. The tumors are more likely to develop initially in the periphery of the prostate, unlike BPH, in which the pathologic changes typically originate close to the urethra (see Fig. 19-2). The cancer invades adjacent local structures, such as the seminal vesicles and urinary bladder, and spreads to the musculoskeletal system, particularly the axial skeleton, and lungs. Lymphatic metastasis may involve the obturator, iliac, and periaortic lymph nodes, extending up through the thoracic duct (see Fig. 13-8).
The mechanism underlying the organ-specific metastasis of prostate cancer cells to the bone is still poorly understood. Whether the cells only invade the bone and proliferate there or whether they invade many tissues but survive mainly in the bone is unclear; this concept is referred to as seed and soil.
Research suggests that osteonectin, a small protein found in bone marrow, attracts prostate cancer cells to bone and once attracted, stimulates the cells to invade bone. These findings suggest that antibodies to osteonectin could reduce the invasiveness of prostate cancer (and breast cancer) and offer a potential way of preventing the spread of prostate cancer to the bone.68
Clinical Manifestations
The clinical presentation of prostate cancer is extremely variable and may be completely asymptomatic until the disease is advanced. In many men the disease is noted incidentally on DRE or discovered in fragments of prostatic tissue removed through TURP for BPH. Depending on the size and location of the lesion, the initial presenting symptom could be related to urinary obstruction, onset of pain, or constitutional symptoms such as fatigue and weight loss.
The urinary obstruction symptoms associated with cancer are similar to those associated with BPH but typically present in later stages of the disease compared with BPH. Cancer originating in the subcapsular region of the prostate as opposed to the periurethral area would account for this difference. The obstructive symptoms include urinary urgency, frequency, hesitancy, dysuria, hematuria, difficulty initiating or continuing the urine stream, and decreased urine stream. Blood in the ejaculate may also be noted.
Bony metastasis occurs via lymphatics to adjacent structures and pelvic nodes in the majority of people with metastatic disease. Of the primary tumors that metastasize to the spine, prostate lesions rank fourth behind breast, lung, and myeloma. The axial skeleton is affected more than the appendicular skeleton, especially the spine, ribs, sternum, femur, and pelvis. Prostate cancer is unique in that bone is often the only clinically detectable site of metastasis, and the resulting tumors tend to be osteoblastic (bone forming) rather than being osteolytic (bone lysing).89
Prostate cancer can also metastasize to the lungs and liver. The Gleason score is used to grade prostate cancer cells on how they appear under a microscope (Fig. 19-5) and predict the likelihood of metastases (Box 19-2).
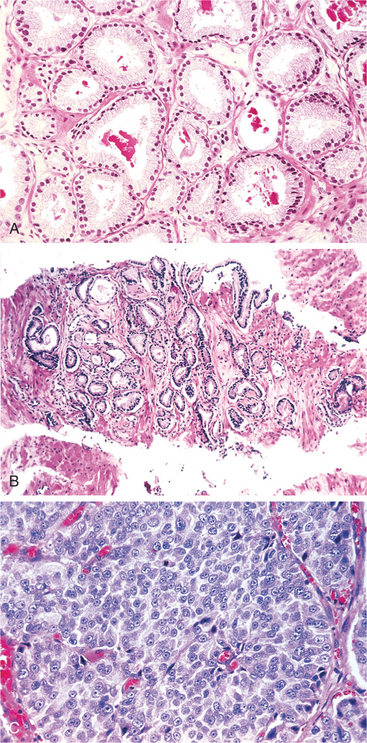
Figure 19-5 A, Low-grade (Gleason score 2) prostate cancer consisting of backto-back, uniformly sized, well-differentiated (resembling normal cells) malignant glands. B, Variably sized, more widely dispersed glands of moderately differentiated adenocarcinoma (Gleason score 6). The higher the Gleason score, the more abnormal and poorly differentiated the cells, the more aggressive the tumor is likely to be. C, Poorly differentiated adenocarcinoma composed of sheets of malignant cells (Gleason score 10). (From Kumar V: Robbins and Cotran: pathologic basis of disease, ed 7, Philadelphia, 2005, WB Saunders.)
Pain complaints associated with prostate cancer can vary tremendously. A dull, vague ache may be noted in the rectal, sacral, or lumbar spine region, and the individual may have difficulty walking. The sacral and lumbar pain is typically associated with bony metastasis. Pain may be noted in the thoracic and shoulder girdle areas secondary to lymphatic spread of the disease or again secondary to local bony metastasis. Symptoms, such as fatigue, weight loss, anemia, and dyspnea, have all been attributed to metastatic spread of the disease.
MEDICAL MANAGEMENT
Prostate cancer has a long latency period between appearance of premalignant lesions and clinically evident cancer, making it a type of cancer susceptible to chemoprevention (the use of agents to slow progression of, reverse, or inhibit carcinogenesis). There is some evidence that this cancer may be preventable with a nontoxic oral agent such as vitamin E, selenium, soy,112,167 or other dietary or nutritional supplement.44,155
In 2003, the Prostate Cancer Prevention Trial (PCPT) became the first phase III clinical trial of prostate cancer prevention. Started in 1993, this landmark study ended early because of the 24.8% reduction of prostate cancer prevalence over a 7-year period in men taking finasteride (Proscar), a 5-α-reductase inhibitor. Other ongoing phase III clinical trials of prostate cancer chemoprevention include the REDUCE study using dutasteride (Avodart) and the SELECT study using vitamin E and selenium.155
An inverse relationship has been observed between dietary intake of lycopene and the risk of prostate cancer. Vitamin and lycopene supplementation (and other antioxidants) may be another form of effective chemoprevention currently under investigation.78,90
Prostate cancer growth is known to depend on androgen, making this another area of study in chemoprevention of this particular type of cancer. Data support the use of antiandrogens to block androgens at the cellular level in the prevention of prostate cancer, although the toxicity of these agents (gynecomastia and gastrointestinal disturbance) poses concerns for the current application in all men.17 Studies of predictive factors, biomarkers for chemoprevention, or agents, such as calcitriol, that can interrupt the development of prostate cancer are under investigation.73,158
A review of the literature suggests a strong link between physical activity and exercise and decreased prostate cancer risk.55,56 Average risk reduction from physical activity and exercise is between 10% and 30%. Exercise may modulate hormone levels and can prevent obesity while enhancing immune function and reducing oxidative stress as possible mechanisms for risk reduction.157
SCREENING.
The American Urological Society recommends annual PSA screening and DRE for men ages 50 years and older if they have more than a 10-year life expectancy, usually defined as having a greater than 50% probability of surviving 10 years. The PSA is a blood test that measures an enzymatic protein manufactured only in the prostate gland. An elevated value (usually 4.0 ng/ml or higher) may be caused by BPH, prostatitis, or cancer. Some medications (e.g., finasteride [Proscar] for BPH and Propecia for hair loss) can depress PSA levels.
PSA may be age-specific, but the exact values for each age remain under investigation. PSA normally rises with age but remains in the 0 to 4 ng/ml range. The level should not rise more than 0.75 ng/dl per year. A level 4 to 10 increases the risk of prostate cancer by 25%; a level greater than 10 ng/ml has a 50% higher risk of prostate cancer.
The optimal upper limit of the normal range for PSA is unknown; approximately 15% of men with PSA levels of 4 ng/ml or less have prostate cancer (some even aggressive tumors).154 However, lowering the PSA threshold below 4.0 ng/ml would dramatically increase the number of men subjected to prostate biopsy without any evidence that the mortality rate would be lowered.
Men who have a high risk of prostate cancer should start testing at age 45 years; this includes African American men and men with a first-degree relative who had prostate cancer before age 65 years. Testing may be advised even earlier (age 40 years) for men with several first-degree relatives who have had prostate cancer at an early age. There is no conclusive evidence that PSA screening reduces prostate mortality at any age or life expectancy, and benefit is unlikely for men older than 75 years.166
Some questions have been raised about how long to continue PSA screening in older men with limited life expectancy. There is some evidence that the potential immediate harms (e.g., false positive tests leading to additional procedures, psychologic distress, and morbidity associated with treating clinically insignificant prostate cancer) outweigh the potential benefits.166
DIAGNOSIS.
The diagnosis of prostate cancer is made by a variety of tests, including DRE, transrectal ultrasound, serum PSA assay, and radiographic imaging modalities. Tissue biopsy is performed to confirm the diagnosis, usually when PSA is elevated for unexplained reasons. The DRE may reveal a hardened, fixed structure or a diffusely undulated gland. A limitation to this procedure is that typically only the posterior and lateral aspects of the prostate can be palpated. An ultrasound probe (TRUS) can be performed by inserting a small ultrasound probe into the rectum. The sound waves emitted by the probe produce a video or photographic image of the prostate. These images help guide a thin needle through the rectum to the gland for a biopsy and staging (TRUS-guided biopsy). The use of positron emission tomography (PET) as a noninvasive means of detecting prostate cancer is under investigation.20
The PSA assay is an important part of the screening process and offers some diagnostic information, but prostate cancer cannot be diagnosed by PSA alone. The average prostatic cancer produces approximately 10 times the PSA a normal prostate gland does. However, PSA values bounce around on a daily basis and an elevated PSA assay (between 4 and 10 ng/ml) may also be related to prostatitis, BPH, prostatic infarcts, and prostatic biopsy and surgery. Therefore an elevated PSA is organ-specific and has some predictive value but is not diagnostic by itself. The PSA may be false negative in up to 30% of cases with localized prostate cancer.
Radiographic imaging modalities are also utilized for staging of the disease. MRI allows for evaluation of the prostate gland and regional lymph nodes. Lymph node involvement must typically be advanced (nodes larger than 1 cm) to be demonstrable. Radiographs may detect metastatic lesions, but the radionuclide bone scan, although not diagnostic, is a more sensitive modality for detection of a metabolically active lesion.
A relatively new testing method, known as reverse transcription-polymerase chain reaction (RT-PCR), is now available and is capable of testing tiny markers that cancer cells carry when they spread to lymph nodes. This test is able to detect cancer previously undetected with standard tests and may also be able to assist in the detection of cancer cells in the surgical margins after radical prostatectomy.74,144
STAGING.
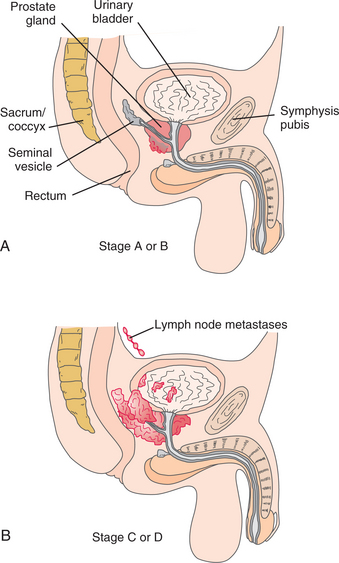
The Whitmore-Jewett staging system is a commonly used method of staging for prostate cancer. With this system, the spread of prostate cancer has been divided into four stages: A through D (Fig. 19-6). The stage of the disease at the time of the diagnosis helps dictate the course of treatment. Alternately, the Gleason score, which rates the tumor from 1 (slow growing) to 6 (very fast growing), may be used to determine the aggressiveness of the cancer tumor and the suitability of treatment (see Box 19-2).
TREATMENT.
The medical treatment of prostate cancer depends on the man’s age and health, the stage of the disease, speed at which the PSA is doubling, and personal preferences. Many uncertainties surround the issue of treating prostate cancer because of the inability to differentiate tumors that will progress from those that will remain unchanged. More men appear to die with prostate cancer than from it.63
No randomized controlled trials have demonstrated the superiority of one treatment over another in terms of survival, although differences in complications vary. The American Urologic Association advocates a presentation of all options for the individual to consider. Once the cancer treatment has begun, most clients are reevaluated every 3 to 6 months. At follow-up visits, symptoms of urinary obstruction and pain are investigated. DRE and a PSA assay are carried out, and prostatic acid phosphatase levels are measured. Other tests may be routinely ordered depending on the individual’s status.
The most commonly used effective treatments include observation; radical prostatectomy; radiation (external-beam or brachytherapy); and hormonal therapy. Unless relatively young, men with early stage A disease may be simply monitored because prostate cancer can be an indolent disease. These cancers rarely progress during the initial 5 years after diagnosis but do progress in 10% to 25% of clients 10 years after diagnosis.13,41 Generally, however, men with a life expectancy of 10 years or more and with disease diagnosed in later stage A or B undergo radical prostatectomy and possibly radiation therapy.
Surgery.
Radical prostatectomy involves removal of the entire prostate, seminal vesicles, a portion of the bladder neck, and regional lymph nodes and ensures that the original source of the cancer is removed. The conventional surgical approach can be retropubic or perineal, but using a laparoscopic approach provides a significantly magnified televised view of the surgical area and minimizes the potential nerve damage more likely with traditional prostatectomy.52,61
The retropubic approach involves an incision from the pubic symphysis to the umbilicus and allows for staging pelvic lymphadenectomy. The primary postoperative complications with conventional surgery are infection; urinary incontinence (e.g., detrusor instability with small bladder capacity or pudendal nerve denervation); ED/impotence, excessive bleeding; and rectal injury with fecal incontinence. Potency may return gradually over the course of a year; however, after these procedures, as many as one-third of men have incontinence or urinary frequency beyond 1 year and an even greater number continue to experience ED.
Radiation.
Radiation therapy may be used to treat localized prostatic lesions as an adjunctive treatment after radical prostatectomy or for palliative effects in the presence of widespread disease. Relief of pain and improvement in symptoms associated with urethral obstruction are possible benefits of this treatment modality. Potential side effects of radiation therapy include urinary frequency, burning with urination, rectal pain or burning, and diarrhea. Some men never experience these problems, but if they do, they are usually transient and clear up within days to weeks after therapy.
Sexual dysfunction (erectile dysfunction or impotence) is a possible long-term complication and occurs in up to 50% of men treated with radiation. Age and comorbidities, such as diabetes or heart disease, can also increase this risk. Pharmacologic treatment for ED in this population is usually successful.
Palliative radiation therapy may be used for sites of pain secondary to bone lesions from bone metastases in men who have failed hormonal therapy. Metastases to the spine with or without spinal cord compression can be treated with external-beam radiation. Benefit has been demonstrated even for men with neurologic changes. Surgery may be combined with radiation for spinal cord compression.123
Radiation therapy is most often used with stage C disease because many men have occult pelvic lymph node metastases. The radiation therapy can be administered daily by external-beam radiation over a number of weeks (usually up to 2 months). External-beam radiation makes it easier to focus and shape the radiation beam (intensity modulation) so that it is less likely to hit other organs, thereby reducing rectal and bladder toxicity.
Examples of external-beam radiation include three-dimensional conformal radiation therapy (3DCRT), which conforms to the shape of the tumor, or intensity-modulated external-beam radiation therapy [IMRT] that directs thousands of pencil-thin radiation beams of varying intensities at the tumor from many different angles. IMRT beam intensity is highest in the thicker, middle portion of the prostate and much lower in thinner, outer parts of the gland, which minimizes the risk of postradiation bladder and bowel problems.
For men with small, nonaggressive tumors, brachytherapy may be the preferred treatment. Brachytherapy is also indicated for men with medical problems that put them at increased risk for any type of surgery or for men who do not feel comfortable with the idea of losing their prostate if there is another option. Small tumors in men with a Gleason score of 6 or less and a pretreatment PSA level of less than 10 may be the best candidates for this type of treatment. Difficulty with urination may preclude brachytherapy since the procedure increases the risk of developing a sudden inability to urinate after undergoing the procedure. Brachytherapy is the delivery of tiny, rice-sized radioactive seeds by a single injection directly into the prostate. This treatment provides a constant, highly concentrated, yet confined dose of radiation to the tissue in and immediately around the tumor. Once in place, the seeds deliver radiation 24 hours a day for several months; they remain in place after they lose their radioactivity.
The prostate absorbs almost all of the radiation so that nerves and other tissues of the surrounding structures of the rectum, bladder, and urethra are spared with fewer complications with ED or incontinence.120 With technologic advances, brachytherapy now has similar cure rates for localized cancer as radiation and radical prostatectomy.
Hormone Therapy.
Hormone therapy is the treatment of choice in the presence of stage D disease; the goal of treatment is androgen deprivation. Testosterone stimulates prostate cancer growth, so by blocking its production, hormone therapy shrinks or slows the progression of prostate tumors. Since the testes produce 95% of the circulating testosterone, orchiectomy is the primary method of manipulating hormone levels.
In recent years, there has been an increase in the use of hormone therapy at all stages of prostate cancer. The extensive use of androgen-deprivation therapy (ADT) has raised concerns about potential adverse effects such as hot flashes, osteoporosis, sexual dysfunction (e.g., loss of libido or ED/impotence), and psychologic effects (e.g., depression, mood swings, or memory loss).57
A second option, estrogen therapy, is used less often because of adverse effects, including gynecomastia, loss of libido and ED/impotence, bloating, and pedal edema. Hormone therapy is not usually advocated for newly diagnosed, locally contained prostate cancer because of its adverse effects (e.g., bone deterioration, decreased libido, mood changes, and anemia). Additionally, serious cardiovascular disease, including stroke, heart attack, and deep venous thrombosis, can be associated with estrogen therapy in anyone at high risk.
Chemotherapy.
Chemotherapy may be advised for men with androgen-independent prostate cancer (AIPC), also known as hormone refractory prostate cancer, whose PSA doubling time (PSADT) is less than 6 months, especially if there is a positive bone scan. These men have advanced, metastatic prostate cancer and have relapsed after initial treatment with hormone therapy. In the majority of men with prostate cancer, AIPC occurs after a median time of 18 months of hormone deprivation.133 They are resistant to further hormone manipulation.104
Docetaxel chemotherapy is now the standard of care for men with metastatic hormone-resistant prostate cancer (HRPC); however, the benefit of this treatment is limited. Optimal treatment timing, dose, and duration have not yet been established.93,106,123
Some men are treated indefinitely until unacceptable toxicity or disease progression occurs. Others are treated using an intermittent approach with chemotherapy interrupted after the initial response.87 Research is focused on improving the efficacy of docetaxel by combining it with novel biologic agents; studies combining docetaxel with angiogenesis inhibitors are underway. Other studies are investigating second-line treatments of HRPC with new cytotoxic agents (e.g., satraplatin and ixabepilone).31
Immunotherapy.
Targeted immunotherapy for control of metastatic prostate cancer is focused on designing an antibody that discriminates between normal and adenocarcinoma cells and targets specific components within tumor cells (e.g., mucins such as MUC1) with toxins or radionuclides. Overexpression of MUC1 plays an important role in prostate cancer progression. Targeting this particular glycosylated protein may help prevent and/or control micrometastases and hormone refractory disease. It may even be possible to develop a vaccine targeting tumor–associated MUC1 antigen.85
Vaccine.
Clinical trials are under way to investigate the development of a vaccine that fights prostate cancer by a variety of immunomodulation techniques.97,156 For example, a dendritic cell–based cancer vaccine for prostate cancer has moved from the laboratory to human clinical trials. Dendritic cells are antigen-presenting (immune system) cells. They initiate and shape an adaptive immune response by capturing, processing, and presenting antigen material to T and B cells. Early results of dendritic cell vaccinations are being reported.103,153 Research continues to focus on improving patient selection, vaccine delivery methods, immune monitoring, and vaccine manufacturing.
In other areas of research, scientists are able to trick the human immune system into recognizing cancerous prostate cells as foreign invaders by genetically engineering the individual’s own cells and injecting them back into the body. This is done by harvesting prostate cancer cells, irradiating them, treating them with autologous granulocyte-macrophage colony–stimulating factor (GM-CSF), and reintroducing them into the body.10 Results so far indicate that both T-cell and B-cell immune responses to human prostate cancer can be generated.46,137
Researchers are also combining vaccines with cytokines or immune modulators, which can foster dramatic antitumor responses. A variety of immunotherapeutic approaches have been followed through to phase III trials for other types of cancer. There is hope that this type of immune therapy can be developed for prostate cancer treatment as well.138
PROGNOSIS.
Multiple sources of data show that prostate cancer incidence rates rose after the introduction of PSA testing. The average age at diagnosis has fallen, the proportion of advanced stage tumors has declined, the proportion of moderately differentiated tumors has increased, and a decline in mortality began in the United States in 1991.102 Data show that 93% of men diagnosed with prostate cancer survive at least 10 years and 77% survive at least 15 years, but it is still the second most common cause of cancer deaths among American men. Earlier detection has given men more choices and better treatment resulting in improved morbidity and mortality rates.5
Men with small or nonaggressive tumors are more likely to be cancer free than men with large or aggressive tumors. Seed implants are more successful for men whose pretreatment level of PSA is below 10 ng/ml. Without any treatment at all, men with the least aggressive prostate cancers face only a 4% to 7% risk of dying from prostate cancer within 15 years of diagnosis.4
The rate of relapse in men with pathologic evidence of pelvic lymph node involvement is high (more than 50%, although some studies report as high as 90%) within 5 years of local treatment. Lymph node involvement occurs in 15% to 20% of men with localized prostate cancer that exhibits high-risk features (as defined by tumor size, serum PSA level, and Gleason score).43
Prostate cancer is sometimes referred to as a “two-decade disease” because it often returns 10 to 20 years after successful local therapy, which is one reason why some physicians advocate radical prostatectomy. Even with radical prostatectomy, recurrence is reported in up to 40% of patients. One-third of those men progress to incurable metastatic disease.153 Metastatic prostate cancer remains a lethal disease, although new treatment strategies discussed earlier are being developed and incorporated into clinical trials that may improve survival rates.
Men with advanced, metastatic prostate cancer who have relapsed after hormone therapy often have disease extending to the skeleton, which is associated with severe pain and disability. The prognosis for this group of individuals is poor at this time (median survival is between 10 and 20 months)133 but improved overall survival demonstrated in studies with chemotherapy for these individuals shows promising results.104
Postoperatively, after radical prostatectomy, incontinence is 100% when the catheter is removed, but urinary control is regained for many men within the first 6 to 8 weeks. Men can speed recovery of urinary function and reduce the likelihood of remaining incontinent with pelvic floor exercises.161,162 Any incontinence 12 months or more after surgery and rehabilitation may require additional medical intervention such as collagen injection (a sclerosing agent to “tighten up” the sphincter) or placement of an artificial urinary sphincter (AUS).
Besides the complications of radiation therapy already discussed, rectal cancer is another potential serious complication, especially for men treated with older forms of radiation therapy 10 years ago before the development of more modern tissue-sparing techniques. Men who have had radiation therapy for prostate cancer should be followed and screened regularly for rectal cancer.
DISORDERS OF THE TESTES
Overview and Etiologic Factors
Orchitis is inflammation of the testis, can be acute or chronic, and is often associated with epididymitis. Gram-negative bacteria and Chlamydia trachomatis are the usual infectious agents.
Incidence and Risk Factors
Anyone with primary infections of the genitourinary tract or infections in other body regions (e.g., pneumonia or scarlet fever) is at risk of developing orchitis. Sexually active males with multiple partners are at higher risk of developing genitourinary infections. Males 10 years of age and older who have parotitis (mumps) are also at risk of developing orchitis. The incidence of orchitis in this group can be as high as 25% to 33%.53 Men with indwelling catheters are also at higher risk of developing orchitis secondary to genitourinary tract infection.
Pathogenesis and Clinical Manifestations
Orchitis is often secondary to UTIs. The bloodstream and lymphatics are then the route of spread from other body areas to the testes. Systemic infections, such as pneumonia, scarlet fever, and parotitis, use the same routes to spread to the testes. Orchitis is marked by testicular pain and swelling. Pain may also be noted in the lower abdominal area. Fever and malaise can be present, but symptoms of urinary dysfunction are usually absent.
MEDICAL MANAGEMENT
Physical examination reveals a tender and swollen testicle. Laboratory tests revealing an elevated white blood cell (WBC) count and urinalysis are an important component of the diagnostic process.
TREATMENT.
See the section on Epididymitis and Special Implications for the Therapist: Epididymitis and Testicular Torsion in this chapter.
Epididymitis
Epididymitis is an inflammation of the epididymis. The two primary types of epididymitis are sexually and nonsexually transmitted infections. Males typically under the age of 40 years who are sexually active with multiple partners are at higher risk of developing genitourinary disease that can lead to epididymitis. Iatrogenic sources of infection include cystoscopy and indwelling catheters. In the older male population, this condition can be precipitated by prostatitis and UTIs.
Pathogenesis and Etiologic Factors
Epididymitis is typically caused by bacterial pathogens. The sexually transmitted infections leading to epididymitis are associated with urethritis. In the nonsexually transmitted infections, urine containing pathogens may be forced up the ejaculatory ducts, through the vas deferens, and into the epididymis. Pressure associated with voiding and physical strain can force the urine from the urethra. Infection originating elsewhere in the body can spread to the epididymis via the lymphatics of the spermatic cord. Congenital urinary tract abnormalities can lead to epididymitis in children.
Clinical Manifestations
Epididymitis may be associated with pain, urinary dysfunction, fever, and urethral discharge. Unilateral scrotal pain is common, but pain may also be noted in the lower abdominal, groin, or hip adductor muscle areas. When bacteriuria is present, urinary frequency and urgency and dysuria can occur. When the epididymitis is related to sexually transmitted disease, urethritis and urethral discharge are present concurrently. Local scrotal erythema and swelling are associated with epididymitis.
MEDICAL MANAGEMENT
In addition to the findings noted under the section on Clinical Manifestations, urinalysis, urethral smear, and urine culture are important for the diagnosis of epididymitis. An elevated WBC count is also usually present.
TREATMENT.
During the acute phase, treatment includes bed rest; scrotal elevation and support; nonsteroidal antiinflammatory drugs (NSAIDs); or antibiotics. Hospitalization may be necessary if signs of sepsis or abscess formation are present or if the pain is severe. Once treatment has been initiated, a significant decrease in pain should be noted within a week, but the scrotal edema may be present for 2 to 3 months. (See the sections on Epididymitis and Testicular Torsion.)
Testicular Torsion
Testicular torsion is an abnormal twisting of the spermatic cord as the testis rotates within the tunica vaginalis. The torsion can occur intravaginally or extravaginally, but intravaginal torsion is the more common. This condition is a surgical emergency. Early diagnosis and treatment is imperative to save the testis.
Etiologic Factors
Torsion of the spermatic cord is often associated with congenital abnormalities. These include absence of the scrotal ligaments, incomplete descent of the testis, and a high attachment of the tunica vaginalis. Increased mobility of the testis and epididymis within the tunica vaginalis facilitate twisting of the spermatic cord. Testicular torsion can also occur after heavy physical activity.
Incidence and Risk Factors
Intravaginal torsion most often occurs in males 8 to 18 years of age. The condition is rarely seen after age 30 years. The extravaginal torsions occur primarily in neonates. A firm, painless scrotal mass is discovered shortly after birth.
Pathogenesis
The spermatic cord contains the vas deferens and the nerve and blood supply for the scrotal contents. If the torsion is severe enough to occlude the arterial supply, infarction of testicular germ cells can quickly occur. Intravaginal torsions are often precipitated by congenital abnormalities. These anomalies allow for rotation of the testis around the spermatic cord, or the torsion may occur between the testis and epididymis. Extravaginal torsion occurs most often during the fetal descent of the testes, before the tunica adheres to the scrotal wall. This allows the testis and fascial tunica to rotate around the spermatic cord above the level of the tunica vaginalis.
MEDICAL MANAGEMENT
Diagnosis can be difficult and misdiagnosis of epididymitis can delay treatment, resulting in testicular loss.96 Physical examination reveals a firm, tender testis that is often positioned high in the scrotum. Erythema and scrotal edema may be present. The cremasteric reflex is often absent. Color and power Doppler sonography make it possible to identify the absence of perfusion in the affected testis. Scintigraphy (radionuclide scanning) remains an acceptable diagnostic alternative in evaluation testicular torsion when color Doppler is unavailable or inadequate.119 A urinalysis is performed to help rule out infection.
TREATMENT.
Testicular torsion is considered a urologic emergency. Once the diagnosis of testicular torsion is made, emergency surgery is performed. The procedure includes detorsion, and if the testis is deemed nonviable, then an orchiectomy is performed. The duration of the torsion is critical regarding salvage of the testis. If the surgery is performed within 3 hours of onset, a greater than 80% salvage rate occurs. The salvage rate drops to 20% if more than 12 hours passes before the surgery.
Testicular Cancer
Testicular cancer occurs when cells in one or both testicles become malignant. Primary testicular tumors are divided into two histogenetic categories: seminoma and nonseminoma. Germ cell tumors (origin in the primordial germ cells) make up more than 95% of testicular tumors, whereas the remaining neoplasms are mostly tumors of stromal or sex cord origin. Metastatic tumors to the testis from primary neoplasm elsewhere in the body are uncommon, although involvement by lymphoma may occur in older men.
Incidence
Although relatively rare, testicular cancer is the most common solid organ tumor in young men. There are considerable geographic and ethnic variations in the global incidence of testicular cancer. The disease mainly affects Western populations, with an increasing incidence since the middle of the twentieth century.50 Average rates in developed areas of the world are six times higher than those in developing areas.19
Testicular cancer is the most common cancer in the 15-to 35-year-old male age group and second most common malignancy from age 35 to 39 years, with a white-to-black incidence ratio of 5 to 1. Approximately 79,200 new cases of testicular cancer were estimated to occur in the United States in 2007.71
Etiologic and Risk Factors
The etiology of testicular cancer is not well understood. Although the cause is unknown, hormonal balance at various life stages appear to be related. Congenital and acquired factors have been associated with the development of testicular cancer; familial clustering has been observed particularly among siblings.33,47
The first susceptibility gene for testicular cancer has been located and named TGCT1 on the long arm of chromosome X, inherited from the mother; its presence increases a man’s risk of testicular cancer by up to 50 times.126 There is some evidence that cancer stem cells are derived from normal stem cells that have gradually accumulated various genetic defects; a group of tumor-specific antigens known as cancer/testis antigens (CTAs) may be expressed at early stages during embryogenesis in germ cell precursors eventually leading to testicular cancer.29
The most significant factor in testicular cancer is the association of cryptorchidism (the testes not descending into the scrotum). The incidence of testicular cancer is 35 times higher in males with a cryptoid testis. There is now strong evidence that prenatal or postnatal environmental estrogen exposures (e.g., endocrine-mimicking environmental pollutants, pesticides, industrial chemicals, or chemical contaminants in drinking water) contribute to testicular cancer; this remains under further investigation.62,143
Other risk factors still under investigation may include mothers who took exogenous estrogen during pregnancy (diethylstilbestrol)145,146; scrotal trauma101; exposure to high levels of radiofrequency/microwave radiation in radar technicians127; first-born sons170; and nonidentical twins.
Men with Klinefelter’s syndrome (a sex chromosome disorder characterized by low levels of male hormones, sterility, breast enlargement, and small testes) are also at greater risk of developing testicular cancer. Lifestyle and occupational exposures occurring later in life may play a role in promoting the disease, but they are not likely involved in the initiation of the cancer development.50
A previous history of testicular cancer and history of infertility and poor semen quality or infection have been associated with an increased risk of developing testicular cancer. A causal relationship has not been established between infertility and infection and testicular cancer.92
Pathogenesis
Since germ cell tumors make up the vast majority of testicular cancers, the following discussion focuses solely on such tumors. Carcinoma in situ usually becomes an invasive germ cell tumor in a median period of approximately 5 years.16 The neoplastic transformation of a germ cell results in either a seminoma, an undifferentiated tumor, or a nonseminomatous tumor composed of embryonal carcinoma teratoma, choriocarcinoma, or yolk-sac carcinoma (endodermal-sinus tumor). Seminomas are the most common testicular cancers, accounting for approximately 40% to 50% of germ cell tumors and most often appearing in the fourth decade of life. Seminomas appear as a solid, grey-white growth and are rarely necrotic or cystic. The entire testis can be replaced by the tumor.
The incidence of nonseminomas peaks in the third decade of life, and hemorrhage, necrosis, or cystic changes are more common. Yolk-sac tumors are the most common germ cell tumors of infants. These tumors result in enlarged testes, which appear grossly as poorly defined lobulated masses. Focal areas of hemorrhage are common.
Clinical Manifestations
The most common initial sign of testicular cancer is enlargement of the testis with diffuse testicular pain, swelling, hardness, or some combination of these findings. The condition may go undetected if no pain is experienced and the male is not periodically performing testicular self-examination. The enlargement may be accompanied by an ache in the abdomen or scrotum or a sensation of heaviness in the scrotum. Metastasis, although with little or no local change, is noted in the scrotum.
Retroperitoneal primary tumors may present with back pain and/or groin or pelvic pain (psoas muscle invasion). Metastatic testicular cancer can present as back pain (may be the primary presenting complaint), abdominal mass, hemoptysis, or neck or supraclavicular adenopathy. Up to 21% of men with testicular germ cell cancer have back pain as the primary presenting symptoms.22 A significant delay occurs in diagnosis of testicular cancer in these men compared with men who also had symptomatic unilateral testicular enlargement.
Pain may be the sole presenting symptom in metastasis to the retroperitoneal, cervical, and supraclavicular lymphatic chains. Since the testis embryologically originates in the genital ridge and descends during fetal life through the abdomen and inguinal canal into the scrotum, the primary lymphatic and vascular drainage of the testis is to the retroperitoneal lymph nodes and the renal or great vessels, respectively. Approximately 20% of all cases have involved lymph nodes.16 Bone metastasis is a late event, often combined with metastasis to the retroperitoneal lymph nodes, lung, and liver. Symptomatic, solitary bone lesions are responsive to chemotherapy and local radiation therapy.
Occurring during the prime of life for most men and potentially affecting sexual and reproductive capabilities, testicular cancer has a major emotional impact and can affect overall quality of life.
MEDICAL MANAGEMENT
No known preventive strategies exist but teaching and promoting testicular self-examination as a technique for early detection of this disease is recommended.26 Since survival is dependent on early detection, men should be encouraged to practice testicular self-examination at least every 6 months. For optimal effect, health education programs need to take into account complexities such as cultural diversity, if men are to heed vital and in some cases, life-sustaining advice.
DIAGNOSIS.
A thorough urologic history and physical examination are the basis for making a diagnosis of testicular cancer. A painless testicular mass is highly suggestive of testicular cancer. Transillumination of the scrotum may also reveal a testicular mass. Serum tumor markers (e.g., alpha-fetoprotein [AFP], human chorionic gonadotropin [hCG], and lactate dehydrogenase [LDH]) are increased in 40% to 60% of all cases and may be used to guide treatment and follow-up.
Testicular ultrasound is used to differentiate a variety of scrotal disorders besides cancer (e.g., epididymitis, orchitis, hydrocele, or hematocele). The modalities used to assess metastatic spread include CT scans and MRI. MR lymphography may replace the currently used tomography scanning or MRI used to noninvasively stage retroperitoneal lymph nodes.11 Biopsy with microscopic examination of testicular tissue by a pathologist is the best way to make a definitive diagnosis.
STAGING.
Clinical staging is based on the TNM classification system: stage I, or tumor confined to the testis, epididymis, or spermatic cord; stage II, which has spread to the retroperitoneal lymph nodes and divided into A, B, and C according to the size of the nodes; and stage III, which is distant metastasis or high serum tumor–marker values.
TREATMENT.
Management of testicular cancer has changed substantially in the last 20 years, primarily because of the ability of cisplatin-containing combination chemotherapy to cure advanced disease. Even over the last 5 years, the management of stage I testis cancer has changed tremendously.3
Organ-sparing surgery has become an accepted approach to treat malignant and nonmalignant tumors of a single testis. Combined with adjuvant radiotherapy to the retroperitoneal and ipsilateral pelvic lymph nodes, this approach has proven as effective as orchidectomy. Nerve-sparing retroperitoneal lymph node dissection is an integral part of testis cancer management strategies for both early and advanced-stage disease.68 Nerve-sparing techniques help preserve sexual function and prevent incontinence. As many as 50% of men with clinical stage I testis cancer can be treated with close surveillance instead of immediate adjuvant treatment.3
Testicular-sparing management of testicular masses is an alternative to radical orchiectomy and allows for preservation of sperm and hormonal function without endangering survival rates.117 Sperm collection and cryopreservation before the initiation of therapy is an available reproductive technology. Only a few sperm are needed for successful in vitro fertilization.173
Chemotherapy is used in cases of relapse after radiation therapy or when there is a high risk for occult metastatic disease. The recommendation of which therapies to include is based on pathologic findings from the orchiectomy and results of the CT and MRI procedures. Second-line conventional-dose or high-dose chemotherapy with stem cell rescue may cure 25% to 50% of men with recurrent testicular tumors. New chemotherapeutic agents, including the taxanes gemcitabine and oxaliplatin, may also be used.140
PROGNOSIS.
With early detection, more than 95% of men with newly diagnosed germ-cell tumors are cured. It is estimated that with adequate diagnostics and treatment, 100% of men with stage I testis cancer will survive in the future.3 Delay in diagnosis correlates with a higher stage at presentation for treatment.16 One confounding factor related to the diagnosis being delayed is the potential lack of perceptible testicular changes while the disease spreads. Men who have had testicular cancer have an increased likelihood of developing cancer in the remaining testicle.
Long-term sequelae of cisplatin use may include leukemia, cardiovascular events, and reports that circulating cisplatin can be detected in the plasma as long as 20 years after treatment.99,116 The most serious long-term complications of chemotherapy or radiotherapy are cardiovascular toxicity and second malignancies; each has a 25-year risk of approximately 16%.40
Chemotherapy-related cardiovascular toxicity may be the result of both direct endothelial damage induced by cisplatin and indirect hormonal and metabolic changes. There is an increased incidence of metabolic syndrome in long-term survivors that is most likely caused by the lower testosterone levels.40
Current therapeutic regimens have significantly improved survival but often adversely affect fertility117; cisplatin-based chemotherapy results in 30% to 50% infertility rates in men with testicular cancer who are treated with chemotherapy.80 Options to maintain fertility after testicular cancer and its treatment are discussed in detail elsewhere.136
Other chemotherapy-induced complications include chronic neurotoxicity (50%), permanent ototoxicity and renal function impairment (30%), pulmonary fibrosis (5% to 10% when treated with bleomycin); and late relapse.40
Erectile Dysfunction
Erectile dysfunction (ED), also referred to as impotence, is a general term that refers to the inability to achieve, keep, or sustain an erection sufficient for satisfactory sexual performance for both partners and may include a problem with libido, penile erection, ejaculation, or orgasm. The degree of erectile dysfunction may be graded according to the number of satisfactory attempts out of 10 (mild: 7 to 8, moderate 4 to 6, and severe 0 to 3).
Incidence and Risk Factors
The prevalence of ED in various community studies has varied from as low of 7% to as high as 52% and has a definite correlation with increasing age. There are an estimated 30 million men in the United States and 152 million men worldwide with ED.2 Epidemiologic studies of ethnic groups in the United States are not available at this time.
Risk factors for ED include age-related testosterone deficiency; certain medications; chronic diseases, particularly coronary artery disease, neurologic conditions, and diabetes mellitus; alcohol use; mental or emotional problems; and smoking84,136,150 (Box 19-4). Being overweight or underweight is a risk factor; physical activity can reduce the risk of ED, but only in men who are obese.23 Treatment for prostate cancer such as prostatectomy, external radiation therapy, and brachytherapy can contribute to the development of ED.
Etiologic Factors
The underlying causes of erectile dysfunction are commonly classified as neurogenic, arteriogenic, venogenic, or psychogenic. Approximately 50% to 80% of men seeking treatment for sexual dysfunction have an organic lesion. The ratio of organic to psychogenic lesions is directly proportional to age. In 70% of men less than 35 years old, the cause is psychogenic. The psychogenic factors include anxiety, fear, depression, stress, fatigue, and so on.
In 85% of men over the age of 50 years, the cause is organic. Conditions that can impede blood flow (e.g., atherosclerosis or medications) or impair nerve transmission (e.g., diabetes or surgery) are the most common organic causes.95,100 Neurogenic or neurovascular ED can result from surgical or traumatic injury to the autonomic or somatic penile innervation or neurovascular mechanisms that initiate erections.
Autonomic denervation of the pudendal nerve and pelvic or perineal trauma from radical pelvic surgeries (e.g., for colon or prostate cancer) result in poor smooth muscle relaxation, arterial insufficiency, and venous inadequacy, thus preventing adequate erection.91
Both surgery and radiation therapy can cause this type of autonomic dysfunction. Radiation may also produce ED by accelerating microvascular angiopathy, causing cavernosal fibrosis or stenosis of the pelvic arteries, and by accelerating existing arteriosclerosis, leading to vascular impotence.149
Another possible neurologic mechanism is microscopic neuropathy with breakdown of cholinergic, adrenergic, noncholinergic, or nonadrenergic neurotransmission. Individuals with diabetes mellitus experience change in the ultrastructure of the cavernous nerves arising from the S2 to S4 spinal nerves. These changes include diffuse thickening of the Schwann cells and perineural basement membranes.
ED can be a hemodynamic event and can warn of ischemic heart disease in some men. Conditions resulting in arterial insufficiency can also result in local penile changes. Researchers refer to ED as penis angina14 and a penile stress test that can be as predictive as a treadmill exercise stress test for atherosclerosis and subsequent peripheral vascular disease.115
Pathogenesis
Despite great strides in research into ED, knowledge and understanding of the pathophysiologic mechanisms responsible for ED are still not entirely clear. Numerous factors and structures can influence the process of sexual function and dysfunction. Coordinated interaction between regulatory centers in the diencephalon, brainstem, and spinal cord are required for sexual function. Penile erection is a neurovascular event modulated by neurotransmitters and hormonal status.
The penis is innervated by autonomic and somatic nerves. In the pelvis, the sympathetic and parasympathetic nerves merge to form the cavernous nerves, which enter the corpora cavernosa to regulate blood flow during erection and ejaculation. The corpus cavernosum is one of two chambers in the penis that run lengthwise and are surrounded by a membrane called tunica albuginea. A spongy tissue filling the chambers contains smooth muscles, fibrous tissues, spaces, veins, and arteries. The urethra along the underside of the corpus cavernosum is surrounded by the corpus spongiosum.149
The parasympathetic visceral efferent fibers arise from sacral roots 2 to 4 to supply the pelvic plexus located on the lateral wall of the rectum. The cavernous nerves leave the pelvic plexus and travel in the lateral pelvic fascia on he posterolateral surface of the prostate gland to supply the corpora cavernosa of the penis. The somatic component, the pudendal nerve, is responsible for penile sensation.149
During sexual stimulation, impulses from the brain and local nerves trigger the arteries in the penis to widen and the smooth muscle to relax. Blood moves into the spongy smooth muscle while the penile veins constrict to keep the penis engorged and rigid. The tunica albuginea helps trap the blood in the corpus cavernosum, sustaining the erection.168
Clinical Manifestations
Manifestations of sexual dysfunction include inability to have or maintain penile erection, inability to achieve orgasm, infertility, and dyspareunia. Most men (and their partners) experience significant feelings of emotional distress and/or depression associated with sexual dysfunction and subsequent infertility. Quality-of-life issues are an important aspect of this disorder.
MEDICAL MANAGEMENT
Without a better understanding of the pathophysiology behind ED, treatment rather than prevention is the main focus. Preventing coronary artery disease and diabetes mellitus may aid in preventing this condition. Anyone with ED should be counseled to avoid recreational drugs and excessive alcohol. Hypertension should be treated, and for anyone with diabetes, glucose levels must be monitored carefully and kept under control.
Regular exercise, keeping cholesterol levels below recommended levels, maintaining an ideal body weight, and quitting smoking are all healthy lifestyle choices that may help prevent ED.
DIAGNOSIS.
Because of the sheer number of possible etiologic factors, a definitive diagnosis related to ED can be difficult. Differentiating between an organic versus psychogenic cause is the initial challenge. The nocturnal penile tumescence test, which monitors the incidence of nocturnal erections, helps with this distinction. Men with psychogenic ED/impotence will have nocturnal erections, whereas those with an organic lesion will not.
Physical examination includes an abdominal and genital inspection and palpation, assessment of peripheral pulses and blood pressure, and DRE to evaluate prostate size. Laboratory values, including serum chemistries, complete blood cell count, fasting lipid levels, thyroid function tests, and hormone levels may help rule out systemic diseases.
As a potential marker of cardiovascular risk, ED is no longer considered a benign result of aging. Its documented association with many chronic diseases and high-risk situations extends the significance of ED beyond the purely sexual domain, providing insight into general and cardiovascular health.14 Arteriography is the standard test for vascular diagnostic testing, but ultrasonography is also used to assess the vascular integrity of the genital area.
TREATMENT.
The goals of treatment for ED are to improve self-esteem, confidence, and overall sexual and relationship satisfaction. Management of ED can help restore men to their full erectile potential. Optimal treatment improves erection hardness and is designed to improve outcomes in overall sexual experience.107
Medical treatment for ED varies, depending on the cause. Pharmacologic treatment with phosphodiesterase type 5 (PDE-5) inhibitors, such as sildenafil (Viagra), vardenafil (Levitra), and tadalafil (Cialis), to increase blood flow to the penis by inhibiting a particular enzyme has become the new first-line treatment for anyone without heart disease. PDE-5 inhibitors can be used cautiously with α-blockers but are contraindicated in men who take any form of nitrate or nitrate because these drug combinations can cause serious hypotension.168
Other products are available for select individuals. For example, alprostadil (applied as a topical gel to the head of the penis or used as a suppository inserted into the urethra), previously only available as an injection into the penile tissue, can be used by anyone who is not hypersensitive to the drug, who does not have a history of venous thrombosis, or who does not have sickle cell anemia or sickle cell trait, thrombocytopenia, polycythemia, or multiple myeloma. An erection occurs within 10 minutes and will last for 30 to 60 minutes.
Testosterone injections are an option for men with documented androgen deficiency. Injection of vasoactive substances directly into the penis may also be used. These drugs relax the arterioles and smooth muscle of the corpus cavernosum and increase cavernous arterial blood flow, causing an erection. Penile prosthetic devices can be implanted surgically, and men with arterial and venous deficiencies are candidates for vascular reconstruction procedures. Erections can be achieved mechanically with a vacuum pump device.
Anyone with arteriogenic ED should be counseled regarding risk-factor modification, including lifestyle interventions such as exercise and weight loss and pharmacologic management of hypertension and hyperlipidemia.128 Psychologic counseling and/or behavioral therapy is indicated for those with a psychogenic basis for the sexual dysfunction.
PROGNOSIS.
ED occurs immediately after radical prostatectomy with variable return of function. Preservation of sexual function can be difficult even among men who have been treated with nerve-sparing surgery. Preservation of the nerves responsible for erection may depend on experience of the surgeon and preoperative potency. Younger, healthier men also may have better outcomes after surgery. In the case of radiation-induced ED, years may elapse before clinically significant ED develops.149
References
1. Albaugh, J. Insights into the management of erectile dysfunction. Part I. Urol Nurs. 1999;19(4):241–245.
2. Albaugh, J. Insights into the management of erectile dysfunction. Part II. Urol Nurs. 2000;20(1):29–36.
3. Albers, P. Management of stage I testis cancer. Eur Urol. 2007;51(1):34–43.
4. Albertsen, P.C., et al. Competing risk analysis of men aged 55 to 74 years at diagnosis managed conservatively for clinically localized prostate cancer. JAMA. 1998;280:975–980.
5. American Cancer Society (ACS): Prostate cancer (2006). Available at http://documents.cancer.org/117.00/117.00.pdf Accessed June 15, 2007.
6. Antunes, A.A. Botulinum toxin for the treatment of lower urinary tract symptoms due to benign prostatic hyperplasia. Nat Clin Pract Urol. 2007;4(3):155–160.
7. Avins, A.L. Saw palmetto and lower urinary tract symptoms: what is the latest evidence? Curr Urol Rep. 2006;7(4):260–265.
8. Bales, G.T. Effect of preoperative biofeedback/pelvic floor training on continence in men undergoing radical prostatectomy. Urology. 2000;56:627–630.
9. Ballard, D.J. Treatment of erectile dysfunction: can pelvic muscle exercises improve sexual function? J Wound Ostomy Continence Nurs. 1997;34(5):255–264.
10. Basler, M. Advances in prostate cancer immunotherapies. Drugs Aging. 2007;24(3):197–221.
11. Bellin, M.F. Magnetic resonance lymphography. Curr Opin Urol. 2007;17(1):65–69.
12. Bergman, J. Prostatitis and chronic prostatitis/chronic pelvic pain syndrome. Expert Rev Neurother. 2007;7(3):301–307.
13. Blute, M.L., Zincke, H., Farrow, G.M. Long term follow-up of young patients with stage A adenocarcinoma of the prostate. J Urol. 1986;136:840–843.
14. Bondil, P. “Penis angina” or the current revolution in erectile dysfunction (ED). Prog Urol. 2005;15(6):1030–1034.
15. Bonnar-Pizzorno, R.M. Saw palmetto supplement use and prostate cancer risk. Nutr Cancer. 2006;55(1):21–27.
16. Bosl, G.J., Motzer, R.J. Testicular germ-cell cancer. N Engl J Med. 1997;337(4):242–253.
17. Bostwick, D.G., Qian, J. Effect of androgen deprivation therapy on prostatic intraepithelial neoplasia. Urology. 2001;58(suppl 1):91–93.
18. Bravi, F. Food groups and risk of benign prostatic hypertrophy. Urology. 2006;67(1):73–79.
19. Bray, F. Interpreting the international trends in testicular seminoma and nonseminoma incidence. Nat Clin Pract Urol. 2006;3(10):532–543.
20. Brush, J.P. Positron emission tomography in urological malignancy. Curr Opin Urol. 2001;11(2):175–179.
21. Burgio, K.L. Preoperative biofeedback assisted behavioral training to decrease post-prostatectomy incontinence: a randomized, controlled trial. J Urol. 2006;175(1):196–201.
22. Cantwell, B., Mannix, K.A., Harris, A.L. Back pain: a presentation of metastatic testicular germ cell tumors. Lancet. 1987;1:262–264.
23. Cheng, J.Y. Body mass index, physical activity and erectile dysfunction: a U-shaped relationship from population-based study. Int J Obes (Lond). 2007;31(10):1571–1578.
24. Chuang, Y.C. The application of botulinum toxin in the prostate. J Urol. 2006;176(Pt 1):2375–2382.
25. Claes, H. Pelvic floor exercise versus surgery in the treatment of impotence. Br J Urol. 1993;71(1):52–57.
26. Cook, R. Teaching and promoting testicular self-examination. Nurs Stand. 2000;14(24):48–51.
27. Cornel, E.B. Evaluation of early pelvic floor physiotherapy on the duration and degree of urinary incontinence after radical retropubic prostatectomy in a non-teaching hospital. World J Urol. 2005;23(5):353–355.
28. Cornel, E.B. The effect of biofeedback physical therapy in men with chronic pelvic pain syndrome type III. Eur Urol. 2005;47(5):607–611.
29. Costa, F.F. Concise review: cancer/testis antigens, stem cells, and cancer. Stem Cells. 2007;25(3):707–711.
30. Cotthoff, A., Goff, K., Kelly, D.G. The incidence of lymphedema following treatment for prostate cancer: a retrospective study (abstract). Phys Ther. 2001;8(5):A23.
31. Di Lorenzo, G. Hormone-refractory prostate cancer: where are we going? Drugs. 2007;67(8):1109–1124.
32. Dong, B. An infectious retrovirus susceptible to an IFN antiviral pathway from human prostate tumors. Proc Natl Acad. 2007;104(5):1655–1660.
33. Dong, C., Hemminiki, K. Modification of cancer risks in offspring by sibling and parental cancers from 2,112,616 nuclear families. Int J Cancer. 2001;92(1):144–150.
34. Dorey, G. Pelvic floor exercises for erectile dysfunction. BJU Int. 2005;96(4):595–597.
35. Dorey, G. Pelvic floor exercises for treating post-micturition dribble in men with erectile dysfunction: a randomized controlled trial. Urol Nurs. 2004;24(6):490–497. [512].
36. Dorey, G. Pelvic floor muscle exercises and manometric biofeedback for erectile dysfunction and postmicturition dribble. J Wound Ostomy Continence Nurs. 2003;30(1):44–51.
37. Dorey, G. Randomised controlled trial of pelvic floor muscle exercises and manometric biofeedback for erectile dysfunction. Br J Gen Pract. 2004;54(508):819–825.
38. Dorey, G. Restoring pelvic floor function in men: review of random controlled trials. Br J Nurs. 2005;14(19):1014–1021.
39. Edelstein, R., Babayan, R. Managing prostate cancer. I. Localized disease. Hosp Pract. 1993;28(4):61–78.
40. Efstathiou, E. Review of late complications and treatment and late relapse in testicular cancer. J Natl Compr Canc Netw. 2007;4(10):1059–1070.
41. Epstein, J.I., Paull, G., Eggleston, J.C., Walsh, P.C. Prognosis of untreated stage A1 prostatic carcinoma: a study of 94 cases with extended follow-up. J Urol. 1986;136:837–839.
42. Farkas, A., Marcella, S., Rhoads, G.G. Ethnic and racial differences in prostate cancer incidence and mortality. Ethn Dis. 2000;10(1):69–75.
43. Ferrari, A.C., et al. Prospective analysis of prostate-antigen specific markers in pelvic lymph nodes of patients with high-risk prostate cancer. J Natl Cancer Inst. 1997;89(20):1498–1504.
44. Fleshner, N.E., Kucuk, O. Antioxidant dietary supplements: rationale and current status as chemopreventive agents for prostate cancer. Urology. 2001;57(suppl 1):90–94.
45. Floratos, D.L. Biofeedback vs. verbal feedback as learning tools for pelvic muscle exercises in the early management of urinary incontinence after radical prostatectomy. BJU Int. 2002;89(7):714–719.
46. Fong, L. Immunotherapy for prostate cancer. Curr Oncol Rep. 2007;9(3):226–233.
47. Forman, D., et al. Familial testicular cancer: a report of the UK family register, estimation of risk and an HLA class 1 sib-pair analysis. Br J Cancer. 1992;65:255–262.
48. Franke, J.J. Early post-prostatectomy pelvic floor biofeedback. J Urol. 2000;163(1):191–193.
49. Fried, N.M. New laser treatment approaches for benign prostatic hyperplasia. Curr Urol Rep. 2007;8(1):47–52.
50. Garner, M.J. Epidemiology of testicular cancer: an overview. Int J Cancer. 2005;116(3):331–339.
51. Gerber, G.S. Saw palmetto for the treatment of men with lower urinary tract symptoms. J Urol. 2000;163(5):1408–1412.
52. Gill, I.S., Zippe, C.D. Laparoscopic radical prostatectomy: technique. Urol Clin North Am. 2001;28(2):423–436.
53. Gillenwater, J.Y., et al. Adult and pediatric urology, ed 2. St Louis: Mosby, 1991.
54. Giovannucci, E. A prospective study of dietary fat and risk of prostate cancer. J Natl Cancer Inst. 1994;271:498.
55. Giovannucci, E. A prospective study of physical activity and incident and fatal prostate cancer. Arch Intern Med. 2005;165:1005–1010.
56. Giovannucci, E., Liu, Y., Platz, E.A., et al. Risk factors for prostate cancer incidence and progression in the health professionals follow-up study. Int J Cancer. 2007;121(7):1571–1578.
57. Gomella, L.G. Contemporary use of hormonal therapy in prostate cancer: managing complications and addressing quality-of-life issues. BJU Int. 2007;99(suppl 1):25–29.
58. Gong, E.M. Saw palmetto and benign prostatic hyperplasia. Am J Chin Med. 2004;32(3):331–338.
59. Gonzalez, R.R. Is there a role for urodynamics in chronic nonbacterial prostatitis? Curr Urol Rep. 2006;7(4):335–338.
60. Goodman, C.C., Snyder, T.E. Differential diagnosis for the physical therapist: screening for referral, ed 4. Philadelphia: WB Saunders, 2007.
61. Guillonneau, B., et al. Laparoscopic radical prostatectomy: assessment after 240 procedures. Urol Clin North Am. 2001;28(1):189–202.
62. Habert, R. Effects of estrogens on the development of the testis during fetal and neonatal life. Gynecol Obstet Fertil. 2006;34(10):970–977.
63. Hamdy, F.C. Prognostic and predictive factors in prostate cancer. Cancer Treat Rev. 2001;27(3):143–151.
64. Hamilton, A.S., et al. Health outcomes after external-beam radiation therapy for clinically localized prostate cancer: results from the Prostate Cancer Outcomes Study. J Clin Oncol. 2001;19(9):2517–2526.
65. Ho, G., et al. Experimental study of transurethral robotic laser resection of the prostate using the LaserTrode light guide. J Biomed Opt. 2001;6(2):244–251.
66. Hoffman, R.M., et al. Racial and ethnic differences in advanced-stage prostate cancer: the Prostate Cancer Outcomes Study. J Natl Cancer Inst. 2001;93(5):388–395.
67. Holmes-Walker, D.J. Maintaining bone health in patients with prostate cancer. Med J Austr. 2006;184(4):176–179.
68. Jacob, K., et al. Osteonectin promotes prostate cancer cell migration and invasion: a possible mechanism for metastasis to bone. Cancer Res. 1999;59(17):4453–4457.
69. Jacobsen, N.E. Retroperitoneal lymph node dissection in testicular cancer. Surg Oncol Clin. 2007;16(1):199–220.
70. Jakobsson, L. Micturition problems in relation to quality of life in men with prostate cancer or benign prostatic hyperplasia. Cancer Nurs. 2004;27(3):218–229.
71. Jemal, A. Cancer statistics, 2007. CA Cancer J Clin. 2007;57(1):43–66.
72. Jemal, A., et al. Cancer statistics, 2002. CA Cancer J Clin. 2002;2:23–47.
73. Johnson, C.S. The antitumor efficacy of calcitriol: preclinical studies. Anticancer Res. 2006;26(4A):2543–2549.
74. Kantoff, P.W., et al. Prognostic significance of reverse transcriptase polymerase chain reaction for prostate-specific antigen in men with hormone-refractory prostate cancer. J Clin Oncol. 2001;19(12):3025–3028.
75. Kaplan, S.A. Combination therapy with doxazosin and finasteride for benign prostatic hyperplasia in patients with lower urinary tract symptoms and a baseline total prostate volume of 25 ml or greater. J Urol. 2006;175(1):217–220.
76. Kittles, R.A. A common nonsense mutation in EphB2 is associated with prostate cancer risk in African American men with a positive family history. J Med Genet. 2006;43(6):507–511.
77. Koch, E. Extracts from fruits of saw palmetto and roots of stinging nettle: viable alternatives in the medical treatment of benign prostatic hyperplasia and associated lower urinary tract symptoms. Planta Med. 2001;67(6):489–500.
78. Kucuk, O., et al. Phase II randomized clinical trial of lycopene supplementation before radical prostatectomy. Cancer Epidemiol Biomarkers Prev. 2001;10(8):861–868.
79. Kunze, D. Functional analyses of C13orf19/P38IP in prostate cell lines. Oncol Rep. 2006;15(6):1599–1604.
80. Lambert, S.M. Infertility and testis cancer. Urol Clin North Am. 2007;34(2):269–277.
81. Langer, J.E. Inflammatory disorders of the prostate and the distal genital tract. Radiol Clin North Am. 2006;44(5):665–677.
82. Lee, S.W. Chronic prostatitis/chronic pelvic pain syndrome: role of alpha blocker therapy. Urol Int. 2007;78(2):97–105.
83. Leitzmann, M.F. Ejaculation frequency and subsequent risk of prostate cancer. JAMA. 2004;291(13):1578–1586.
84. Lewis, R.W. Epidemiology of erectile dysfunction. Urol Clin North Am. 2001;28(2):209–216.
85. Li, Y. MU1 is a promising therapeutic target for prostate cancer therapy. Curr Cancer Drug Targets. 2007;7(3):259–271.
86. Lilli, P. Impact of preoperative biofeedback on incontinence in cancer patients undergoing radical prostatectomy. Arch Ital Urol Androl. 2006;78(3):92–96.
87. Lin, A.M. Intermittent chemotherapy for metastatic hormone refractory prostate cancer. Crit Rev Oncol Hematol. 2007;61(3):243–254.
88. Litwin, M.S. Chronic Prostatitis Symptom Index: development and validation of a new outcome measure. J Urol. 1999;162(2):369–375.
89. Logothetis, C.J. Osteoblasts in prostate cancer metastasis to bone. Nat Rev Cancer. 2005;5(1):21–28.
90. Lu, Q.Y., et al. Inverse associations between plasma lycopene and other carotenoids and prostate cancer. Cancer Epidemiol Biomarkers Prev. 2001;10(7):749–756.
91. Lue, T.F. Erectile dysfunction. N Engl J Med. 2000;342:1802–2113.
92. Mancini, M. High prevalence of testicular cancer in azoospermic men without spermatogenesis. Human Reprod. 2007;22(4):1042–1046.
93. Mancuso, A. Effective chemotherapy for hormone-refractory prostate cancer (HRPC): present status and perspectives with taxane-based treatments. Crit Rev Oncol Hematol. 2007;61(2):176–185.
94. Marks, L.S., et al. Effects of saw palmetto herbal blend in men with symptomatic benign prostatic hyperplasia. J Urol. 2000;163(5):1451–1456.
95. Masters, W.H. Sex and aging: expectations and reality. Hosp Pract. 1986:175–198.
96. Matteson, J.R., et al. Medicolegal aspects of testicular torsion. Urology. 2001;57(4):783–786.
97. McNeel, D.G. Prostate cancer immunotherapy. Curr Opin Urol. 2007;17(3):175–181.
98. Meacham, R.B. Androgen replacement in the aging man. Infect Urol. 2001;14(2):30.
99. Meinardi, M.T., et al. Cardiovascular morbidity in long-term survivors of metastatic testicular cancer. J Clin Oncol. 2000;18(8):1725–1732.
100. Mellinger, B.C., Weiss, J. Sexual dysfunction in the elderly male. Am Urol Assoc Update Ser. 1992;11:146–152.
101. Merzenich, H., et al. Sorting the hype from the facts in testicular cancer: is testicular cancer related to trauma? J Urol. 2000;164(6):2143–2144.
102. Mettlin, C. Impact of screening on prostate cancer rates and trends. Microsc Tes Tech. 2000;51(5):415–418.
103. Mihalyo, M.A. Dendritic cells program non-immunogenic prostate-specific T cell responses beginning at early stages of prostate tumorigenesis. Prostate. 2007;67(5):536–546.
104. Mike, S. Chemotherapy for hormone-refractory prostate cancer. Cochrane Database Syst Rev. 2006;18(4):CD005247.
105. Moore, K.N. Urinary incontinence after radical prostatectomy: a randomized controlled trial comparing pelvic muscle exercises with or without electrical stimulation. BJU Int. 1999;83:57–65.
106. Moss, R.A. Cytotoxic chemotherapy for prostate cancer: who and when? Curr Treat Options Oncol. 2006;7(5):370–377.
107. Mulhall, J. Erectile dysfunction: monitoring response to treatment in clinical practice-recommendations of an international panel. J Sex Med. 2007;4(2):448–464.
108. Myers, R.B. Androgen replacement therapy in the aging male. Rev Urol. 2003;5(4):216–226.
109. National Institute of Diabetes and Digestive and Kidney (NIDDK): Prostatitis: disorders of the prostate. Available at: http://kidney.niddk.nih.gov/kudiseases/pubs/prostatitis/index.htm Accessed June 13, 2007.
110. National Institutes of Health (NIH): NIH chronic prostatitis symptom index (NIH-CPSI). Available on-line at http://www.nih.gov Accessed June 13, 2007.
111. National Prostate Cancer Coalition: Facts and statistics: prostate cancer, 2001. Available at http://www.4npcc.org
112. Nelson, E.C. Inhibition of Akt pathways in the treatment of prostate cancer. Prostate Cancer Prostatic Dis. 2007;10(4):331–339.
113. Neuhausen, S.L., et al. Prostate cancer susceptibility locus HPC1 in Utah high-risk pedigrees. Hum Mol Genet. 1999;8(13):2437–2442.
114. Newcomer, L.M., et al. The association of fatty acids with prostate cancer risk. Prostate. 2001;47(4):262–268.
115. O’Kane, P.D., Jackson, G. Erectile dysfunction: is there silent obstructive coronary artery disease? Int J Clin Pract. 2001;55(3):219–220.
116. Oliver, R.T. Testicular cancer. Curr Opin Ocol. 2001;13(3):191–198.
117. Paduch, D.A. Testicular cancer and male infertility. Curr Opin Urol. 2006;16(6):419–427.
118. Parekh, A.R. The role of pelvic floor exercises on post-prostatectomy incontinence. J Urol. 2003;170(1):130–133.
119. Pavlica, P., Barozzi, L. Imaging of the acute scrotum. Eur Radiol. 2001;11(2):220–228.
120. Peschel, R.E. Long-term outcomes after prostate implant. Cancer J. 2007;13(2):78–79.
121. Peschel, R.E. Surgery, brachytherapy, and external-beam radiotherapy for early prostate cancer. Lancet Oncol. 2003;4(4):233–241.
122. Peyromaure, M. The management of stress urinary incontinence after radical prostatectomy. BJU Int. 2002;90:155–161.
123. Pienta, K.J. Advances in prostate cancer chemotherapy: a new era begins. Cancer J Clin. 2005;55(5):300–318.
124. Powell, I.J. Epidemiology and pathophysiology of prostate cancer in African-American men. J Urol. 2007;177(2):444–449.
125. Prezioso, D. Estrogens and aspects of prostate disease. Int J Urol. 2007;14(1):1–16.
126. Rapley, E.A., et al. Localization to Xq27 of a susceptibility gene for testicular germ-cell tumours. Nat Genet. 2000;24(2):197–200.
127. Richter, E., et al. Cancer in radar technicians exposed to radiofrequency/microwave radiation: sentinel episodes. Int J Occup Environ Health. 2000;6(3):187–193.
128. Rosen, R.C. Erectile dysfunction and cardiac disease: recommendations of the Second Princeton Conference. Curr Urol Rep. 2006;7(6):490–496.
129. Ruzaev, M.L. Rehabilitation of patients with chronic prostatitis. Vopr Kurortol Fizioter Lech Fiz Kult (Russian). 2004;1:35–37.
130. Sadeghi-Nejad, H. Sexual dysfunction and prostatitis. Curr Urol Rep. 2006;7(6):479–484.
131. Scardino, P.T. Early detection of prostate cancer. Urol Clin North Am. 1989;16:635–655.
132. Schmidt, U., Fiedler, U., Pilarsky, C.P. Identification of a novel gene on chromosome 12 between BRCA-2 and RP-1. Prostate. 2001;47(2):91–101.
133. Schrijvers, D. Androgen-independent prostate cancer. Recent Results Cancer Res. 2007;175:239–249.
134. Segal, R.J. Resistance exercise in men receiving androgen deprivation therapy for prostate cancer. J Clin Oncol. 2003;21(9):1653–1659.
135. Silvalingam, S. An overview of the diagnosis and treatment of erectile dysfunction. Drugs. 2006;66(18):2339–2355.
136. Simon, B., et al. Preserving fertility after cancer. Cancer J Clin. 2005;55(4):211–228.
137. Simons, J.W., et al. Induction of immunity to prostate cancer antigens: results of a clinical trial of vaccination with irradiated autologous prostate tumor cells engineered to secrete granulocyte-macrophage colony-stimulating factor using ex vivo gene transfer. Cancer Res. 2001;61(6):2736–2743.
138. Slovin, S.F. Emerging role of immunotherapy in the management of prostate cancer. Oncology (Williston Park). 2007;21(3):326–348.
139. Smith, R.H., Wake, R., Soloway, M.S. Benign prostatic hyperplasia: a universal problem among aging men. Postgrad Med. 1988;83(6):79.
140. Sonpavde, G. Management of recurrent testicular germ cell tumors. Oncologist. 2007;12(1):51–61.
141. Steinber, G.D., et al. The familial aggregation of prostate cancer: a case control study (abstract). J Urol. 1990;143:131A.
142. Stoch, S.A., Parker, R.A., Chen, L. Bone loss in men with prostate cancer treated with gonadotropin-releasing hormone agonists. J Clin Endocrin Metabol. 2001;86(6):2787–2791.
143. Storgaard, L. Male reproductive disorders in humans and prenatal indicators of estrogen exposure. A review of published epidemiological studies. Reprod Toxicol. 2006;21(1):4–15.
144. Straub, B., et al. Reverse transcriptase-polymerase chain reaction for prostate-specific antigen in the molecular staging of pelvic surgical margins after radical prostatectomy. Urology. 2001;57(5):1006–1011.
145. Strohsnitter, W.C., et al. Cancer risk in men exposed in utero to diethylstilbestrol. J Natl Cancer Inst. 2001;93(7):545–551.
146. Swan, S.H. Intrauterine exposure to diethylstilbestrol: long-term effects in humans. APMIS. 2000;108(12):793–804.
147. Tanvetyanon, T. Physician practices of bone density testing and drug prescribing to prevent osteoporosis during androgen deprivation therapy. Cancer. 2005;103(2):237–241.
148. Tavtigian, S.V., et al. A candidate prostate cancer susceptibility gene at chromosome 17p. Nat Genet. 2001;27(2):172–180.
149. Telöken, C. Management of erectile dysfunction secondary to treatment for localized prostate cancer. Cancer Control. 2001;8(6):540–545.
150. Tengs, T.O., Osgood, N.D. The link between smoking and impotence: two decades of evidence. Prev Med. 2001;32(6):447–452.
151. Tenke, P. Prostate cancer screening. Recent Results Cancer Res. 2007;175:65–81.
152. Thomas, C.A. Botulinum A toxin for the treatment of benign prostatic hyperplasia/lower urinary tract symptoms. Curr Urol Rep. 2006;7(4):266–271.
153. Thomas-Kaskel, A.K. Immunotherapy with dendritic cells for prostate cancer. Int J Cancer. 2007;121(3):467–473.
154. Thompson, I.M. Prevalence of prostate cancer among men with a prostate-specific antigen level less than or equal to 4.0 ng/ml. N Engl J Med. 2004;350(22):2239–2246.
155. Thorpe, J.F. A review of phase III clinical trials of prostate cancer chemoprevention. Ann R Coll Surg Engl. 2007;89(3):207–211.
156. Tjoa, B.A., et al. Dendritic cell-based immunotherapy for prostate cancer. CA Cancer J Clin. 1999;49:117–128.
157. Torti, D.C. Exercise and prostate cancer. Sports Med. 2004;34(6):363–369.
158. Trump, D.L. Vitamin D compounds: clinical development as cancer therapy and prevention. Anticancer Res. 2006;26(4A):2551–2556.
159. Trump, D.L., et al. Androgen antagonists: potential role in prostate cancer prevention. Urology. 2001;57(suppl 1):64–67.
160. Tsai, Y.S., et al. Transurethral microwave thermotherapy for symptomatic benign prostatic hyperplasia: long-term durability with postcare. Eur Urol. 2001;39(6):688–694.
161. Van Kampen, M. Treatment of erectile dysfunction by perineal exercise, electromyographic biofeedback, and electrical stimulation. Phys Ther. 2003;83(6):536–543.
162. Van Kampen, M., et al. Effect of pelvic-floor re-education on duration and degree of incontinence after radical prostatectomy: a randomized controlled trial. Lancet. 2000;355(9198):98–102.
163. Vecchione, A. Molecular genetics of prostate cancer: clinical translational opportunities. J Exp Clin Cancer Res. 2007;26(1):25–37.
164. Volpe, M.A., Fromer, D., Kaplan, S.A. Holmium and interstitial lasers for the treatment of benign prostatic hyperplasia: a laser revival. Curr Opin Urol. 2001;11(1):43–48.
165. Wald, M. Testosterone replacement therapy for older men. J Androl. 2006;27(2):126–132.
166. Walter, L.C. PSA screening among elderly men with limited life expectancies. JAMA. 2006;296:2336–2342.
167. Wang, J. Genistein chemoprevention of prostate cancer. J Carcinog. 2007;166:3.
168. Weeks, B. How new drugs help treat erectile dysfunction. Nursing2006. 2006;36(1):18–19.
169. Weinrich, S.P. Prostate cancer screening in high-risk men: African American Hereditary Prostate Cancer Study Network. Cancer. 2006;106(4):796–803.
170. Weir, H.K., et al. Pre-natal and peri-natal exposures and risk of testicular germ-cell cancer. Int J Cancer. 2000;87(3):438–443.
171. Wille, S. Pelvic floor exercises, electrical stimulation, and biofeedback after radical prostatectomy: results of a prospective randomized trial. J Urol. 2003;170(2 Pt 1):490–493.
172. Windsor, P.M. A randomized, controlled trial of aerobic exercise for treatment-related fatigue in men receiving radical external beam radiotherapy for localized prostate carcinoma. Cancer. 2004;101(3):550–557.
173. Zapalka, D.M., Redmon, J.B., Pryor, J.L. A survey of oncologists regarding sperm cryopreservation and assisted reproductive techniques for male cancer patients. Cancer. 1999;86(9):1812–1817.