MEDICAL MANAGEMENT
See the sections on Atherosclerosis: Prevention and Angina Pectoris: Prevention and Treatment.
DIAGNOSIS.
Diagnosis of acute MI and determination of the site and extent of necrosis rely on the clinical history, interpretation of the ECG, and measurement of serum levels of cardiac enzymes. Diagnostic uncertainty frequently arises because of a variety of factors.
Many people with acute MI have atypical symptoms, and one half of all people with typical symptoms do not have acute MI. One half of the people with acute MI have nondiagnostic ECGs, and some people are unable to provide a history. Biochemical markers of cardiac injury are commonly relied on to diagnose or exclude acute MI. These laboratory tests dramatically reduce the cost of treating heart attacks by allowing physicians to quickly discharge people with noncardiac chest pain.
Newer biochemical markers of myocardial injury, such as cardiac troponin I (TnI) and cardiac troponin T (TnT) (regulatory proteins that help the heart muscle contract), are now being used instead of or along with the standard markers, such as the myocardial isoenzyme of creatine kinase (CK-MB). TnT is quite specific for myocardial ischemia and necrosis. It remains elevated 5 to 7 days after an MI and is a predictor of cardiovascular mortality.
TnI is a better cardiac marker than CK-MB for MI because it is more sensitive and more specific to myocardial injury; TnT is a better predictor of cardiovascular mortality (as well as all-cause mortality). Both TnI and TnT are useful markers for myocardial injury that help determine the prognosis in people who have unstable angina but no evidence of CK-MB elevation. (See also the section on Cardiac Enzymes and Markers in Chapter 40; see Tables 40-15 and 40-16.)
Researchers are continuing to investigate other hemostatic markers based on the knowledge that coronary thrombosis involves both coagulation and fibrinolysis cascades. For example, increases of fibrinogen and D-dimer, a circulating marker of fibrin turnover, are significantly higher in people with acute ischemic events such as MI and unstable angina than in nonischemic individuals, but it has not been determined to what extent this is causal. Other tests may include nuclear scanning, coronary angiography, echocardiography, CT, cardiac magnetic resonance (CMR) stress testing, and MRA. Serum cholesterol must be determined because of its importance as a modifiable risk factor. See the previous sections on Angina Pectoris: Diagnosis and Atherosclerosis: Diagnosis.
Other cardiac markers include homocysteine, Lp(a), and CRP. Although these have not become “standard” laboratory values, they can be used as independent predictors of future coronary events in apparently healthy men and women. For example, elevated plasma total homocysteine is a risk factor for atherosclerosis and endothelial dysfunction, and CRP may be used as a marker of subclinical atherosclerosis and cardiovascular risk specifically linked to MI and sudden death.
Infarcted tissue is electrically silent and does not contribute to the ECG. Most clients with acute infarction have ECG changes, although this test provides only a crude estimate of the magnitude of infarction. When diagnosis by ECG and enzymes is not possible (e.g., when people seek medical attention after MI), scintigraphic studies (radionuclide imaging) can show areas of necrotic myocardium and diminished perfusion. These tests, which use radiotracers, do not distinguish old damage from recent infarction, and false-positive results can occur.
Other test procedures may include echocardiography, which is useful in assessing the ability of the heart walls to contract and relax, and transesophageal echocardiography (TEE), an ultrasonic technique that provides a clearer image of the heart, including the posterior wall, valvular anatomy, and thoracic aortic structure, providing identification of structural heart diseases. Newer technology, such as RT-3D imaging, has the potential to improve evaluation of heart function (especially ventricular) with TEE.
Magnetic resonance imaging (MRI) to evaluate structural defects of the heart and positron emission tomography (PET) to evaluate cardiac physiology and metabolism and assess tissue perfusion have contributed significantly to the understanding of the pathophysiology of the ischemic heart.
Another new technique being investigated is the use of a contrast agent called EchoGen, used in conjunction with an ultrasound procedure. This agent infiltrates healthy heart muscle but not muscle that has been deprived of blood or oxygen. Existing contrast agents only image the heart chambers, which provides information about the flow of blood through the chamber but not about the structure of the heart muscle itself.
TREATMENT.
The goal of treatment is reestablishing the flow of blood in blocked coronary arteries. Pharmacologic intervention is used to provide pain relief (essential since angina is evidence of ongoing ischemia), limit infarction size, reduce vasoconstriction, prevent thrombus formation, and augment repair. MI caused by intracoronary thrombi can be relieved by infusion of thrombolytic agents (e.g., streptokinase, urokinase, t-PA) that dissolve clots, promote vasodilation, and reduce infarct size.
PAI-1 is a naturally occurring substance that inhibits another natural substance, t-PA; t-PA is an enzyme released endogenously as part of the body’s defense against thrombosis; it promotes degradation of fibrin leading to dissolving of blood clots. The effect of PAI-1 on t-PA is to prevent clot destruction in the bloodstream.
Tissue plasminogen activator, a naturally occurring enzyme that promotes dissolving of blood clots, is now a genetically engineered drug used in thrombolytic therapy. However, a single dose of recombinant t-PA (rt-PA) costs about $1000, whereas other drugs are less expensive (e.g., streptokinase costs about $300).
This intervention initiated within 70 minutes of symptom onset is associated with improved outcome.348 After a thrombolytic agent is administered, intravenous (IV) heparin therapy is usually given with adjunctive drug therapy during and after MI, because platelet inhibitors and other cardiovascular medications (see Table 12-5) are known to further reduce mortality when administered during the acute phase.
Right now, only 5% of heart attack victims receive reperfusion therapy within that crucial first hour after symptom onset, primarily because people delay (sometimes by hours) coming to the emergency department. This points out the extreme importance of early intervention and education of the general population (and especially for those with known risk factors, such as hypertension, previous heart attack, diabetes, smoking, or hyperlipidemia) as to the importance of getting to an emergency department at the earliest sign of heart attack. Educating the public about the less common or atypical warning signs and symptoms is essential. Information about public education, reducing delays at home or at work, and the National Heart Attack Alert Program is available.88,237
Other treatment interventions, including identification and modification of risk factors, angioplasty, stenting, atherectomy, angiogenesis, tissue engineering, gene therapy, stem cell transplantation, and cardiac rehabilitation utilizing exercise programs, have been previously discussed in detail (see the sections on Atherosclerosis: Medical Management and Hypertension: Medical Management).
A study to determine whether early, rapid use of cholesterol-lowering therapy can reduce recurrent ischemic events in acute coronary syndromes is under way through the MIRACL (Myocardial Ischemia Reduction with Aggressive Cholesterol Lowering) program. The study showed that lipid-lowering therapy with 80 mg/day of atorvastatin, initiated during acute coronary syndrome, reduces recurrent ischemic events in patients in the first 16 weeks.295,364
Exercise has been recommended as a means of increasing pain tolerance, increasing the threshold of the stimulus required to induce angina, alleviating depression, reducing anxiety, and inducing collateral circulation. Increasing evidence suggests that combining a low-fat diet and intensive exercise training can improve myocardial perfusion by regression of coronary atherosclerosis. Exercise training may be contraindicated for some people (Box 12-9; see also Box 12-4). Medical clearance must be obtained for entry into an exercise training program.
Exercise testing is the most useful tool to establish guidelines for exercise training in apparently healthy adults and is mandatory for people with known or suspected cardiovascular disease.108,192 The majority of exercise testing can be done within 3 days of MI with a very low incidence of complications. Criteria for testing usually include clients who are off IV nitroglycerin with no angina at rest, uncontrolled cardiac failure, or arrhythmias. Early testing can lead to early triage and potential cost savings.
PROGNOSIS.
The size and anatomic location of the infarction, together with the amount of damage from previous infarctions, determine the acute clinical picture, the early complications, and the long-term prognosis. The first 24 hours after onset of symptoms is the time of highest risk for sudden death. The sooner someone reaches the hospital, the better the prognosis. Eighty percent of those experiencing an acute MI survive the initial attack when transported to a coronary care unit (CCU). Substantial reductions in post-MI death have occurred over the last five decades because of improved intervention.
Factors negatively affecting prognosis include age (clients older than 80 years have a 60% mortality); evidence of other cardiovascular diseases, respiratory diseases, or uncontrolled diabetes mellitus; anterior location of MI (30% mortality rate); and hypotension (clients whose systolic blood pressure is less than 55 mm Hg have a 60% mortality rate). The risk of reinfarction is increased in women, people with elevated blood pressure, and people with elevated serum cholesterol. As MI survivors with long-standing hypertension live longer, cardiac failure has become an increasingly important long-term sequela of MI.
Prognostic testing predictive of cardiac events includes standard exercise testing such as functional capacity and heart rate recovery108 and imaging using SPECT with contrast agents (e.g., thallium Tl 201, technetium Tc 99m sestamibi). In the imaging studies, a radioisotope is taken up by adequately perfused tissue, allowing detection of myocardial perfusion defects at rest and during exercise (areas of infarction appear as regions of diminished isotope activity or no activity, referred to as cold spots).
Study of the prognostic value of treadmill exercise testing in older persons has shown that workload (measured in metabolic equivalents) is the only treadmill exercise testing predictive of death both in younger persons and in adults over 65 years of age.130 An abnormal exercise test result is a more powerful predictor of risk in those people with conventional risk factors than in those without such risk factors.
Congestive Heart Failure
CHF is a condition in which the heart is unable to pump sufficient blood to supply the body’s needs. Backup of blood into the pulmonary veins and high pressure in the pulmonary capillaries lead to subsequent pulmonary congestion and pulmonary hypertension. Failure may occur on both sides of the heart or may predominantly affect the right or left side. Heart failure is not a disease but rather represents a group of clinical manifestations caused by inadequate pump performance from either the cardiac valves or the myocardium. It may be chronic over many years, requiring management by oral medications, or it may be acute and life-threatening, requiring more dramatic medical management to maintain an adequate cardiac output.
Four distinct types of CHF have been recognized: (1) systolic heart failure (caused by contractile failure of the myocardium), (2) diastolic failure (occurs when increased filling pressures are required to maintain adequate cardiac output despite normal contractile function), (3) left-sided heart failure (occurs when the left ventricle can no longer maintain a normal cardiac output), and (4) right-sided heart failure (right-sided ventricular dysfunction secondary to either left-sided heart failure or to pulmonary disease).
Strictly classified, left ventricular failure is referred to as CHF; acute right ventricular failure, seen almost exclu- sively in association with massive pulmonary embolism, is labeled cor pulmonale. Cor pulmonale is heart disease, but it arises from an underlying pulmonary pathologic condition; therefore it is discussed in Chapter 15. Right-sided heart dysfunction secondary to left-sided heart failure, vascular dysfunction, or congenital heart disease is excluded in the definition of cor pulmonale (see the section on Cor Pulmonale in Chapter 15).
Incidence
CHF is a common complication of ischemic and hypertensive heart disease, occurring most often in the older adult and, in its chronic form, referred to as a cardiogeriatric syndrome. Because the heart muscle is damaged during a heart attack, many heart attack survivors develop CHF. In the United States, heart failure develops in an estimated 500,000 individuals annually: it is the most common cause for hospitalization in people older than 65 years, with an estimated 5 million men and women living with CHF in the United States today. This condition is on the increase as the population ages and more people survive heart attacks.
Etiologic and Risk Factors
Many cardiac conditions predispose individuals to CHF, but hypertension is one of the most prevalent (Table 12-11). People with preexisting heart disease are at greatest risk for the development of CHF, because when the heart is stressed, compensatory mechanisms may be inadequate. For example, a faster redistribution of blood volume and increased demand for oxygen by the myocardium occur with increased activity, such as exercise, resulting in heart failure.
Table 12-11
Etiologic and Risk Factors Associated with Congestive Heart Failure
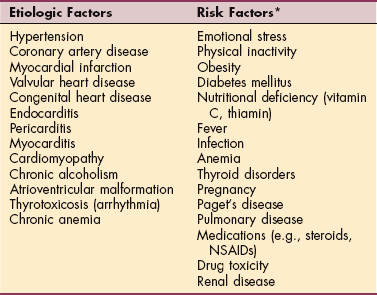
NSAIDs, Nonsteroidal antiinflammatory drugs.
*Risk factors for new onset or exacerbation of previous congestive heart failure.
Pulse pressure appears to be the best single measure of blood pressure for predicting mortality in older people and helps explain apparently discrepant results for low diastolic blood pressure. Pulse pressure is more predictive than even systolic blood pressure alone. Each elevation of 10 mm Hg between systolic and diastolic blood pressure increases the risk of CHF by 14%.70,127,252 Although the literature supports the use of pulse pressure as a significant prognostic indicator, day-to-day clinical use is not common.
CHF occurring during middle age as distinguished from CHF at advanced age includes an increasing proportion of women, a shift from CHD to hypertension as the most common etiology, and intact left ventricular systolic function.276 Women tend to have more risk factors and concurrent medical problems, such as hypertension, diabetes, or renal insufficiency. In addition, there may be other gender differences contributing to the development of CHF in women, such as differences in myocardial distensibility (the degree to which muscle fibers stretch) or hormonal differences as yet undetermined.
Paget’s disease causes vascular proliferation in the bones. When the disease involves over one third of the skeleton, a high cardiac output state exists and may tax the compromised heart. Medications such as steroids or NSAIDs and drug toxicity are also risk factors. For the person with chronic, stable heart failure, acute exacerbations may occur caused by alterations in therapy, client noncompliance with therapy, excessive salt and fluid intake, arrhythmias, excessive activity, PEs, infection, or progression of the underlying disease.
Pathogenesis and Clinical Manifestations
Over the last 15 years, major advances have occurred in our understanding of heart failure, involving the complex interactions that take place among the adrenergic nervous system, the renin-angiotensin axis, the immune system, the peripheral circulation, and other vasoactive substances in response to impaired cardiac function.
The pathophysiology involves structural changes such as loss of myofilaments, apoptosis (programmed cell death), disturbances in calcium homeostasis, and alteration in receptor density, signal transduction, and collagen synthesis. A neurohormonal hypothesis has replaced the hemodynamic model focusing on the neuroendocrine activation of a progressive disorder of left ventricular remodeling. This cascade of events occurs as a result of a cardiac event (e.g., MI) that develops into a clinical syndrome characterized by impaired cardiac function and circulatory congestion.113
CHF is a complex event involving one or both ventricles. This discussion is based on left ventricular failure. See the section on Cor Pulmonale in Chapter 15 for a complete discussion of right-sided heart failure. When the heart fails to propel blood forward normally (such as occurs with left ventricular failure), the body utilizes three neurohormonal compensatory mechanisms; these are effective for a short time but eventually become insufficient to meet the oxygen needs of the body.
First, the failing heart attempts to maintain a normal output of blood by enlarging its pumping chambers so that they can hold a greater volume of blood. This lengthening of the muscle fibers, called ventricular dilation, increases the amount of blood ejected from the heart. This compensatory mechanism has limits, because contractility of ventricular muscle fibers ceases to increase when they are stretched beyond a certain point.
During this first compensatory phase, the right ventricle continues to pump more blood into the lungs. Congestion occurs in the pulmonary circulation with accumulation of blood in the lungs. The immediate result is shortness of breath (most common symptom), and if the process continues, actual flooding of the air spaces of the lungs occurs, with fluid seeping from the distended blood vessels; this is called pulmonary congestion or pulmonary edema. Congestion in the vascular system interferes with the movement of body fluids in and out of the various fluid compartments, resulting in fluid accumulation in the tissue spaces and progressive edema.
During the second compensatory phase, the sympathetic nervous system responds to increase the stimulation of the heart muscle, causing it to pump more often. In response to failing contractility of the myocardial cells, the sympathetic nervous system activates adaptive processes that increase the heart rate and increase its muscle mass to strengthen the force of its contractions. This results in ventricular hypertrophy and a need for more oxygen.
Eventually, the coronary arteries cannot meet the oxygen demands of the enlarged myocardium, and the person may experience angina pectoris owing to ischemia. Secondary compensatory mechanisms activate the sympathetic nervous system and release endothelin from vascular linings, vasopressin (antidiuretic hormone [ADH]) from the pituitary gland, and atrial natriuretic hormone from the heart.
The third compensatory phase involves activation of the renin-angiotensin-aldosterone system. With less blood coming from the heart, less blood passes through the kidneys. The kidneys respond by retaining water and sodium in an effort to increase blood volume, which further exacerbates tissue edema. The expanded blood volume increases the load on an already compromised heart. These mechanisms are responsible for the symptoms of diaphoresis, cool skin, tachycardia, cardiac arrhythmias, and oliguria (reduced urine excretion).
When the combined efforts of these three compensatory mechanisms achieve a normal level of cardiac output, the client is said to have compensated CHF. Ultimately, however, the body’s efforts to compensate may backfire and produce higher blood volume, higher blood pressure, and more stress on the already weakened heart. The heart’s ongoing failure to supply the body with blood compels the body to keep compensating in ways that further burden the heart, and the cycle perpetuates itself. When these mechanisms are no longer effective and the disease progresses to the final stage of impaired heart function, the client has decompensated CHF.
Decompensated CHF ranges from mild congestion with few symptoms to life-threatening fluid overload and total heart failure (Table 12-12). Symptoms usually develop very gradually so that many people do not recognize or report signals of serious disease. The older adult in particular may wrongly associate early symptoms with a lack of fitness or consider them a sign of aging. Confusion and impaired thinking can characterize heart failure in older adults.
Left-Sided Heart Failure.: Failure of the left ventricle (Fig. 12-12) prevents the heart from pumping enough blood through the arterial system to meet the body’s metabolic needs and causes either pulmonary edema or a disturbance in the respiratory control mechanisms. The degree of respiratory distress varies with the client’s position, activity, and level of emotional or physical stress, but any of the symptoms listed under Pulmonary Edema in Chapter 15 may occur.
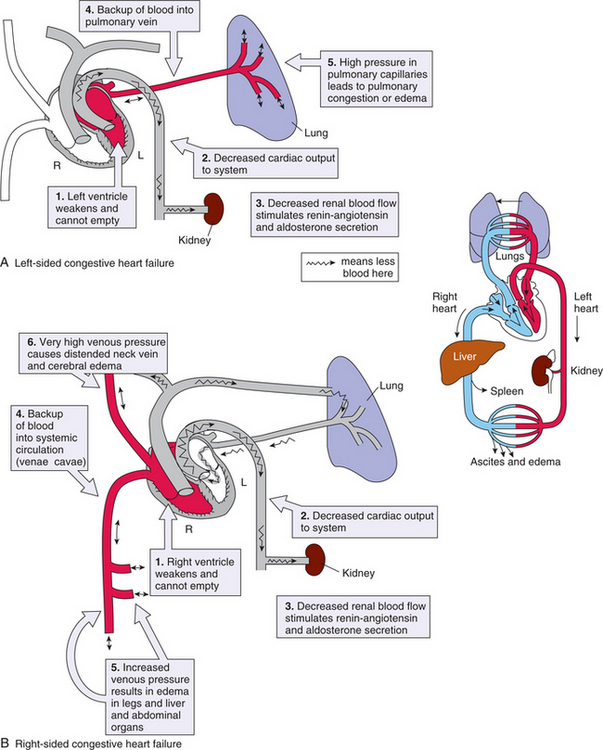
Figure 12-12 Pathophysiologic mechanisms of congestive heart failure. A, Left-sided heart failure leads to pulmonary edema (see text description). B, Right ventricular failure causes peripheral edema that is most prominent in the lower extremities. Inset, Integration of the pulmonary and systemic circulation. When the heart contracts normally, it pumps blood simultaneously into both loops, but pump failure causes circulatory or pulmonary problems, depending on the underlying pathologic mechanism. (A and B from Gould B: Pathophysiology for the health professions, ed 2, Philadelphia, 2002, Saunders, p 286; inset from Damjanov I: Pathology for the health-related professions, ed 3, Philadelphia, 2006, Saunders.)
Dyspnea is subjective and does not always correlate with the extent of heart failure; exertional dyspnea occurs in all clients to some degree. Time for dyspnea to subside is an indication of progress or deterioration in a client’s status, and it can be measured for documentation. Paroxysmal nocturnal dyspnea resembles the frightening sensation of awakening with suffocation. Once the client is in the upright position, relief from the attack may not occur for 30 minutes or longer. The client often assumes a three-point position, sitting up with both hands on the knees and leaning forward. In severe heart failure, the client may resort to sleeping upright in a chair or recliner. Other sleep disturbances may occur from central sleep apnea present in approximately 40% of all adults with heart failure.
Fatigue and muscular weakness are often associated with left ventricular failure, since dyspnea develops along with weight gain and a faster resting heart rate, which decrease the person’s ability to exercise. Inadequate cardiac output leads to decreased peripheral blood flow and blood flow to skeletal muscle. The resultant tissue hypoxia and slowed removal of metabolic wastes cause the person to tire easily. Disturbances in sleep and rest patterns may aggravate fatigue; muscle atrophy is common in advanced CHF.
Renal changes can occur in both right-and left-sided heart failure, but they are more evident with left-sided failure. During the day, the client is upright, decreased cardiac output reduces blood flow to the kidneys, and the formation of urine is reduced (oliguria). Sodium and water not excreted in the urine are retained in the vascular system, adding to the blood volume.
Diminished blood supply to the renal system causes the kidney to secrete renin, stimulating production of angiotensin, which causes vasoconstriction, thereby causing an increase in peripheral vascular resistance, increasing blood pressure and cardiac work, and resulting in worse heart failure. Renin secretion also indirectly stimulates the secretion of aldosterone from the adrenal gland. Aldosterone acts on the renal tubules, causing them to increase reabsorption of sodium and water, further increasing fluid volume. At night, urine formation increases with the recumbent position as blood flow to the kidney improves. Nocturia may interfere with effective sleep patterns, which contributes to fatigue as mentioned.
Right-Sided Heart Failure.: Failure of the right ventricle (see Fig. 12-12) to adequately pump blood to the lungs results in peripheral edema and venous congestion of the organs. Symptoms result from congestion in the heart’s right side and throughout the venous system (see Table 12-12) (see also the section on Cor Pulmonale in Chapter 15).
Dependent edema is one of the early signs of right ventricular failure, although significant CHF can be present in the absence of peripheral edema. In CHF, fluid is retained because the baroreceptors of the body sense a decreased volume of blood as a result of the heart’s inability to pump an adequate amount of blood. The receptors subsequently relay a message to the kidneys to retain fluid so that a greater volume of blood can be ejected from the heart to the peripheral tissues. Unfortunately this compounds the problem and makes the heart work even harder, which further decreases its pumping ability, causing a sense of weakness and fatigue.
The retained fluid commonly accumulates in the extracellular spaces of the periphery. The resultant edema is usually symmetric and occurs in the dependent parts of the body, where venous pressure is the highest. In ambulatory persons, edema begins in the feet and ankles and ascends up the lower legs (pretibial areas). It is most noticeable at the end of a day and often decreases after a night’s rest. In the recumbent person, pitting edema may develop in the presacral area and, as it worsens, progress to the medial thighs and genital area.
Jugular venous distention also results from fluid overload. The jugular veins empty unoxygenated blood directly into the superior vena cava. Since no cardiac valve exists to separate the superior vena cava from the right atrium, the jugular veins give information about activity on the right side of the heart. As fluid is retained and the heart’s ability to pump is further compromised, the retained fluid backs up into both the lungs and the venous system, and the jugular veins reveal this. Jugular venous pulsations are examined by inspecting the silhouette of the neck with the person reclining at a 45-degree angle (Fig. 12-13). The right internal jugular vein is recommended because the left internal jugular may be falsely elevated in some people.
As the liver becomes congested with venous blood it becomes enlarged, and abdominal pain occurs. If this occurs rapidly, stretching of the capsule surrounding the liver causes severe discomfort, and the person may notice either a constant aching or a sharp right upper quadrant pain. In chronic CHF, longstanding congestion of the liver with venous blood and anoxia can lead to ascites (see Fig. 17-5) and jaundice, which are symptoms of liver damage. Anorexia, nausea, and bloating develop secondary to venous congestion of the GI tract. Anorexia and nausea may also result from digitalis toxicity, which is a common problem since digitalis is usually prescribed for CHF.
Cyanosis of the nail beds appears as venous congestion reduces peripheral blood flow. Clients with CHF often feel anxious, frightened, and depressed. Fears may be expressed as frightening nightmares, insomnia, acute anxiety states, depression, or withdrawal from reality.
MEDICAL MANAGEMENT
Diagnosis is based on the clinical picture and depends on where symptoms are on the continuum of mild to severe. Because the two sides of the heart serve different functions, distinguishing the symptoms of left-sided heart failure from those of right-sided heart failure is critical in both diagnosis and treatment. Equally important is consideration of systolic and diastolic dysfunction, both of which indicate a functional or structural defect in the ventricles.
An echocardiogram is the main diagnostic tool; noninvasive cardiac tests such as ECG and chest radiography are secondary tools that can determine left ventricular size and function well enough to confirm the diagnosis. Cardiac catheterization is not routinely performed, but it may be useful in certain cases (e.g., atherosclerotic heart disease, which is potentially correctable). Arterial blood gases are measured to evaluate oxygen saturation. Liver enzymes (e.g., aspartate transaminase [AST], alkaline phosphatase) are often elevated (see Tables 40-5 and 40-18); liver involvement with hyperbilirubinemia commonly occurs, resulting in jaundice.
A new screening tool for individuals with suspected left ventricular dysfunction has been introduced. Measuring B-type natriuretic peptide, a protein secreted from the cardiac ventricles in response to wall tension and pressure overload, can reliably predict the presence or absence of heart failure, even helping to identify when dyspnea is associated with heart failure or some other underlying pathologic condition.208
PREVENTION AND TREATMENT.
Managing heart failure begins with treatment of the underlying cause whenever possible. Nonpharmacologic interventions such as diet and exercise that alter interactions between the heart and the periphery are now accepted therapeutic approaches.
Alterations in lifestyle reduce symptoms and the need for additional medication. There is an urgent need to develop more effective strategies for the prevention and treatment of this increasingly common disorder. Multiple comorbidities in older clients require a multidisciplinary approach to management. Persons with CHF are placed on a sodium-restricted diet, sometimes with limited fluid intake. Emotional and physical rest during the initial phases of intervention is also important in diminishing the workload of the heart.
Activity and Exercise.
Traditionally, the diagnosis of CHF was a contraindication for participation in exercise training because of concerns that further decline in cardiac function would occur. It is now clear that activity restriction is no longer appropriate, since exercise programs have proved to quantitatively achieve results similar to those attained with most effective drug therapies. These findings have shifted attention away from treating the heart toward exercising the muscles.
Whenever possible, physical activity and exercise are prescribed per client tolerance. Physical training for clients with CHF results in an increase in muscular strength and better adaptation to effort owing to the effect of training on skeletal muscles (e.g., decreased vascular resistance in the muscles, delay in the onset of anaerobic metabolism). Exercise training has also been shown to improve exercise capacity, reduce symptoms, improve psychosocial status, and improve functional capacity.159,266 Recently, resistance training combined with short or long bouts of aerobic exercise was found beneficial for patients with CHF.60
Pharmacotherapy.
Pharmacologic therapy is now responsive to the updated understanding of CHF as a cascade of neurohormonal events centered on ventricular remodeling. Pharmacologic agents are used to reduce the heart’s workload, increase muscle strength and contraction, and inhibit neuroendocrine responses to heart failure (see Tables 12-5 and 12-6).
ACE inhibitors have become standard therapy for heart failure because of their ability to block the renin-angiotensin-aldosterone system, increasing renal blood flow and decreasing renal vascular resistance, thereby enhancing diuresis. ACE inhibitors reduce left ventricular filling pressure and moderately increase cardiac output. Vasodilator therapy in combination with ACE inhibitors prolongs life in persons with moderate to severe heart failure.
Diuretics are used to control fluid buildup and prevent congestion, and digoxin may be added to stimulate the heart’s pumping action if symptoms persist despite treatment with ACE inhibitors and diuretics. Angiotensin II receptor antagonists have been added to function as an antihypertensive and enhance the clearance of sodium and water.
The β-blockers, once rarely considered in the treatment of CHF, have been shown effective in reducing symptoms, improving clinical status, reducing hospitalizations, and reducing the risk of death. Combining β-blockers with ACE inhibitors can produce additive effects on two neurohormonal systems (renin-angiotensin system and sympathetic nervous system).
A new drug, nesiritide (human recombinant B-type natriuretic peptide), has been introduced as a first-line medication for decompensated CHF that inhibits sympathetic activity and dilates arterial and venous vessels. Nesiritide binds to receptors in the vasculature, kidney, and other organs to mimic the vasodilatory and diuretic actions of endogenous natriuretic peptides.82
Surgery.
Surgical intervention may include CABG (see Fig. 12-4) for underlying myocardial ischemia and infarction; reconstruction of incompetent heart valves; ventricular remodeling or heart reduction (e.g., Batista procedure, in which a piece of the heart tissue is removed and the heart muscle is sutured back together, making a smaller, tauter heart with a stronger heartbeat); internal counterpulsation (Fig. 12-14) or external counterpulsation, which uses an external pump or balloon to adjust the aortic blood pressure; temporary ventricular assistive devices for people unable to come off bypass (see Chapter 21); and use of an artificial heart or cardiac transplantation.
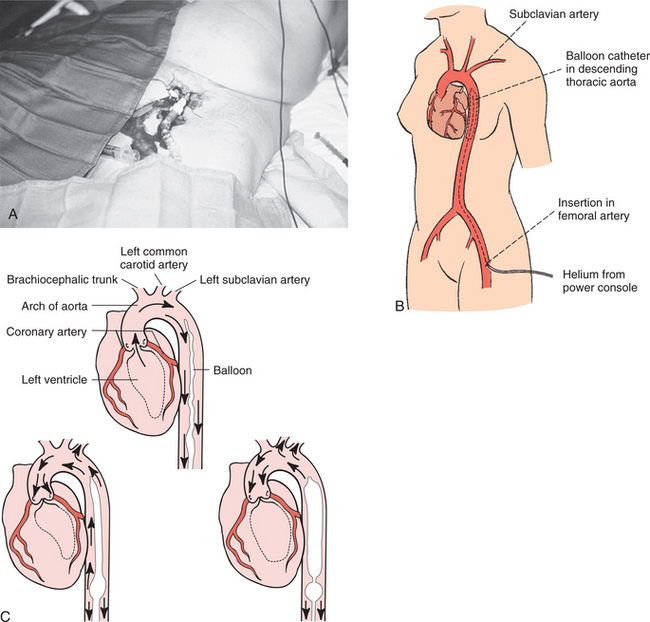
Figure 12-14 The intraaortic balloon pump (IABP) is a common type of cardiac assist device that is used to improve myocardial oxygen supply-demand for individuals with deteriorating hemodynamics or ongoing ischemia, as evidenced by rest pain or electrocardiographic changes in the region of the infarct. The primary functions of balloon counterpulsation are to reperfuse the coronary arteries at the end of systole and reduce the left ventricular afterload (the amount of work the ventricle must do), thereby decreasing myocardial oxygen consumption and improving cardiac output. These intravascular catheter-mounted counterpulsation devices are traditionally used for cases of cardiogenic shock following cardiac surgery or an acute myocardial infarction as well as for people who have chronic end-stage heart failure and who are not candidates for long-term ventricular assistive device (VAD) support. The rationale for IABP counterpulsation in this latter situation is to maintain systemic perfusion and preserve end-organ function until cardiac transplantation occurs. A, The catheter is usually placed through the femoral artery, and the balloon is moved up the iliac artery to the descending aorta, where it is then placed, B, above the renal arteries and below the subclavian artery. This position is critical in order to prevent ischemia to the upper extremities or kidneys. C, When the heart contracts (systole), the balloon is deflated, creating a decline in aortic pressure. After the heart contracts (during diastole), the balloon is filled with air, causing the blood to regurgitate back toward the root of the aorta, thereby perfusing the coronary arteries. When the left ventricle is ready to pump, the balloon is deflated (cardiac systole again), reducing ventricular afterload. (A, courtesy Chris Wells, PT, MS, PhD, University of Pittsburgh Medical Center, 2001. B, from Black JM, Hawks JH, Keene AM: Medical-surgical nursing: clinical management for positive outcomes, ed 7, Philadelphia, 2005, Saunders. C, from Lewis SL, Heitkemper MM, Dirksen SR: Medical surgical nursing: assessment and management of surgical problems, ed 7, St Louis, 2007, Mosby.)
The implantation of skeletal muscle (removed from the individual’s thigh and multiplied in the laboratory) injected into the postinfarction scar after infarction in the case of severe ischemic heart failure has been shown experimentally to improve heart function.219 A review of surgical innovations for chronic heart failure in the context of cardiopulmonary rehabilitation for the therapist is available.158 See also the sections on Atherosclerosis: Treatment in this chapter and Heart Transplantation in Chapter 21.
Cardiac transplantation is now more common for treatment of heart failure. Transplantation is successful for selected individuals, usually those who are treated early in the course of heart failure, before advanced symptoms develop. Reform of the selection process is recommended to identify people who, although not critically ill, will not survive without early transplantation. See further discussion in Chapter 21.
A pacemaker-like device designed to deliver electrical stimulation to the ventricles (biventricular pacing) in an effort to improve the heart’s overall cardiac efficiency by coordinating the heart’s contractions (both ventricles pump at the same time, making the heart pump more forcefully) has been approved by the U.S. Food and Drug Administration (FDA). This technique, referred to as cardiac resynchronization therapy, is available on a limited basis for selected individuals with severe heart failure. The results are promising for people who because of age criteria or lack of donor hearts are not able to undergo cardiac transplantation.
Other similar devices are under continued investigation and development, as is the combined use of resynchronization therapy with pharmacologic therapy and/or a cardioverter-defibrillator as adjunct treatment for CHF.
PROGNOSIS.
Treatment of CHF remains difficult, and the prognosis is poor, even with recent advances in pharmacologic therapy. Annual mortality rates range from 10% in stable clients with mild symptoms to over 50% in people with advanced, progressive symptoms. About 40% to 50% of clients with heart failure die suddenly, probably owing to ventricular arrhythmias.
To achieve the maximal benefit from drug therapy, symptoms must be recognized as early as possible and intervention initiated. Because this condition often develops gradually, intervention is delayed, full resolution is not usually possible, and CHF becomes a chronic disorder.
Exercise capacity was the most powerful predictor of survival in CHF, but a new test that measures swings in heart rate during the day has been developed that can identify individuals who are at the highest risk of dying from CHF. The test measures the amount by which the heart rate changes from slow rates to fast rates in one 24-hour period. The less the heart rate varies over 24 hours, the more likely a person is to die of CHF.244
Other signs of poor prognosis include severe left ventricular dysfunction, severe symptoms and limitation of exercise capacity, secondary renal insufficiency, and elevated plasma catecholamine levels.
Orthostatic (Postural) Hypotension
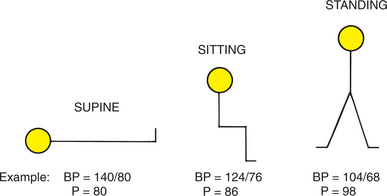
The term orthostatic (postural) hypotension signifies a decrease of 20 mm Hg or greater in systolic blood pressure or a drop of 10 mm Hg or more in both systolic and diastolic arterial blood pressure with a concomitant pulse increase of 15 beats/min or more on standing from a supine or sitting position.
Orthostatic hypotension may be acute and temporary or chronic. Orthostatic hypotension occurs frequently in older adults and occurs in more than one half of all frail, older adults, contributing significantly to morbidity from syncope, falls, vital organ ischemia (e.g., MI, transient ischemic attacks), and mortality among older adults with diabetic hypertension. It is highly variable over time but most prevalent in the morning when supine blood pressure is highest and on first arising.
Etiologic Factors
Orthostatic hypotension is recognized in all groups as a cardinal feature of autonomic nervous dysfunction as well as other nonneurogenic etiologies (Box 12-11). In young adults, orthostatic intolerance and tachycardia may be associated with norepinephrine transporter deficiency. A single gene coding a protein that clears norepinephrine does not function in some individuals, pointing to a genetic etiology.
Postural reflexes are slowed as part of the aging process for some, but not all, persons. Normal aging is associated with various changes that may lead to postural hypotension. Cardiac output falls with age; in the older adult with hypertension, it is even lower. When older subjects (more than 65 years) are put under passive postural stress (60-degree upright tilt), their stroke volume falls even further. These normal changes obviously predispose the aging adult to postural hypotension from any process that further reduces fluid volume or vascular integrity. For example, pooling of blood after eating may lead to profound hypotension, called postprandial hypotension.
In addition, as systolic pressure rises from atherosclerosis, baroreceptor sensitivity and vascular compliance are reduced further, increasing the likelihood of postural hypotension. In the older adult with hypertension and cardiovascular disease receiving vasoactive drugs, the circulatory adjustments to maintain blood pressure are disturbed, leaving the person vulnerable to postural hypotension.178
Drugs are a major cause of orthostatic hypotension in the aging adult. Many have effects on the autonomic nervous system, both centrally and peripherally, and on fluid balance. Diuretics, calcium channel blockers, nitrates, and L-dopa have hypotensive effects. Antidepressants are a common, overlooked cause of orthostasis, even though this is a known side effect of these medications. A general result of treatment for hypertension may be hypotension. In addition, many older adults with systolic hypertension have postural hypotension that may require management before the hypertension is addressed.
Chronic orthostatic hypotension may occur secondary to a specific disease, such as endocrine disorders, metabolic disorders, nephropathy, or neurogenic disorders affecting the autonomic or central nervous systems. Alcohol and drugs such as vincristine used in the treatment of cancer can cause autonomic neuropathy.
Pathogenesis
Orthostasis is a physiologic stress related to upright posture. When a normal individual stands up, the gravitational changes on the circulation are compensated for by several mechanisms, including the circulatory and autonomic nervous systems. On standing, the force of gravity in the vertical axis causes venous pooling in the lower limbs, a sharp decline in venous return, and reduction in filling pressure of the heart, which increase further on prolonged standing because of shifting of water to interstitial spaces and hemoconcentration.
These mechanical events can cause a marked reduction in cardiac output and consequent fall in arterial blood pressure. In healthy people, cardiac output and blood pressure regulation are maintained by powerful compensatory mechanisms involving a rise in heart rate. Blood pressure is maintained by a rise in peripheral resistance. These compensatory mechanisms are initiated by the baroreceptors located in the aortic arch and carotid bifurcation. Orthostatic hypotension results from failure of the arterial baroreflex, most commonly because of disorders of the autonomic nervous system.178
In people with autonomic failure or dysreflexia (e.g., Parkinson’s disease, aging, diabetes, fibromyalgia), orthostatic hypotension results from an impaired capacity to increase vascular resistance during standing. This dysfunction leads to increased downward pooling of venous blood and a consequent reduction in stroke volume and cardiac output that exaggerates the orthostatic fall in blood pressure.
Approximately 80% of the blood pooled in the lower limb is contained in the upper leg (thighs, buttocks) with less pooling in the calf and foot. The location of the additional venous pooling has not been clearly identified, but present data suggest the abdominal compartment and perhaps leg skin vasculature. The pooled blood in the veins of the feet and calves is arterial in origin in that it arises as a result of decreased venous drainage of that region.
In contrast, the blood pooled in the thighs, buttocks, pelvis, and abdomen arises primarily from venous reflux. The pooled blood is not actually stagnant; its mean circulatory time through the dependent region is merely increased by changes in the pressure gradient across the vascular bed and by increases in venous volume. The identification of venous pooling may offer insights for intervention techniques in the future.
Clinical Manifestations
Orthostatic hypotension is often accompanied by dizziness, blurring or loss of vision, and syncope or fainting. There are three main modes of presentation in the older adult: (1) falls or mobility problems, (2) acute or chronic mental confusion, and (3) cardiac symptoms.
A common clinical picture is the person whose legs give way when attempting to stand, usually after prolonged recumbency, after physical exertion, or in a warm environment. These episodes may be accompanied by confusion, pallor, tremor, and unsteadiness. Loss of consciousness may cause frequent falls and additional injuries that can be quite serious. Ischemic neck pain in the suboccipital and paracervical region is often reported by individuals with autonomic failure and orthostatic hypotension.41
Other reported ischemic symptoms of orthostatic hypotension are nonspecific, such as lethargy, weakness, low backache, calf claudication, and angina. Some older adults may experience unexpected and unexplained falls associated with orthostatic hypotension. The cause of such falls may be circulatory impairment that results in a drop in blood pressure on standing upright quickly. Orthostatic hypotension may be an early sign of some other illness or the effects of medication.
Medical Management
There are several general and specific approaches to the management of orthostatic hypotension but no curative intervention for orthostatic hypotension of unknown cause. Prevention is important, and whenever the underlying disorder causing hypotension is corrected, symptoms cease. Nonneurogenic causes, such as diminished intravascular volume, are treated specifically. In orthostatic hypotension caused by autonomic failure there are considerable difficulties in reestablishing sympathetic or parasympathetic efferent activity.
Tilt study or tilt-table testing may be used to assess hypotension by monitoring blood pressure and pulse while tilting a person from horizontal supine to 60 degrees upright. This test has proved very valuable in determining the cause of dizziness or syncope and can reveal irregularities in the vascular regulating system. A combination of general measures and pharmacologic measures is needed in the management of neurogenic postural hypotension.134
Myocardial Disease
Myocarditis is a relatively uncommon acute or chronic inflammatory condition of the muscular walls of the heart (myocardium). It has now been reclassified by the American Heart Association as an acquired (inflammatory) cardiomyopathy.212
It is most often a result of bacterial or viral infection, but it also includes those inflammatory processes related to infectious and noninfectious causes of ischemic heart disease. Other possible causes of myocarditis include chest radiation for treatment of malignancy, sarcoidosis, and drugs, such as lithium, interleukin-2, and cocaine.
The therapist is most likely to treat the person with systemic lupus erythematosus (SLE) (see Chapter 7) who may have a type of myocarditis called lupus carditis (see also the section on The Heart in Collagen Vascular Diseases: Lupus Carditis in this chapter). SLE is a multisystem autoimmune disease characterized by a release of autoantibodies into the circulation, with a subsequent inflammatory process that can target the heart and vasculature.
Myocarditis typically evolves through active, healing, and healed stages that are characterized by inflammatory cell infiltrates leading to interstitial edema and focal myocyte necrosis with replacement fibrosis over time. Ventricular tachyarrhythmias develop as a result of the pathologic changes’ creating an electrically unstable environment.212
Clinical evidence of cardiac involvement is found in up to one half of all people with SLE. Clinical manifestations may include mild continuous chest pain or soreness in the epigastric region or under the sternum, palpitations, fatigue, and dyspnea; and onset may follow a viral upper respiratory tract illness in the population at large as well as in persons with SLE. Complications include heart failure, arrhythmias, dilated (congestive) cardiomyopathy (see next section), and sudden death.
Myocarditis usually resolves with treatment of the underlying condition or cause; specific antimicrobial therapy is prescribed if an infectious agent can be identified. Viral myocarditis is treated with medications that improve cardiac output and reduce arrhythmias, if present. Management of myocarditis in SLE is usually with corticosteroids, but immunosuppressive agents may be required. Myocarditis that progresses to dilated cardiomyopathy with heart failure is frequently fatal without heart transplantation.
Cardiomyopathy
Definition and Overview.: Cardiomyopathy is actually part of a group of conditions affecting the heart muscle itself, so that contraction and relaxation of myocardial muscle fibers are impaired. The original definition of cardiomyopathy stated that this condition was not caused by other heart or systemic disease, which excluded structural and functional abnormalities caused by valvular disorders, CAD, hypertension, congenital defects, and pulmonary vascular disorders.
The American Heart Association 2006 expert consensus panel proposed the following definition for cardiomyopathy, which reflects the idea that many cardiomyopathies have an underlying etiology. Ischemia from CAD is probably the most common.
Cardiomyopathies are a heterogeneous group of diseases of the myocardium associated with mechanical and/or electrical dysfunction that usually (but not invariably) exhibit inappropriate ventricular hypertrophy or dilatation and are due to a variety of causes that frequently are genetic. Cardiomyopathies either are confined to the heart or are part of generalized systemic disorders, often leading to cardiovascular death or progressive heart failure–related disability.212
The classification of cardiomyopathies is problematic. There was much confusion using the former classification of dilated, hypertrophic, and restrictive categories because of overlap when the same disease could appear in two different categories. And sometimes cardiomyopathy progresses from one category to another during the natural history of the disease, making classification difficult. As new knowledge of the pathogenesis of cardiomyopathy has unfolded and as new cardiomyopathies have been defined, the old classification scheme has been replaced with a new (but probably not final) classification.212
Cardiomyopathies are classified as primary and secondary, based on predominant organ involvement. Primary cardiomyopathies include genetic, mixed (genetic and nongenetic), and acquired (Table 12-14). They are confined to the heart muscle.
Table 12-14
American Heart Association Classification of Cardiomyopathies
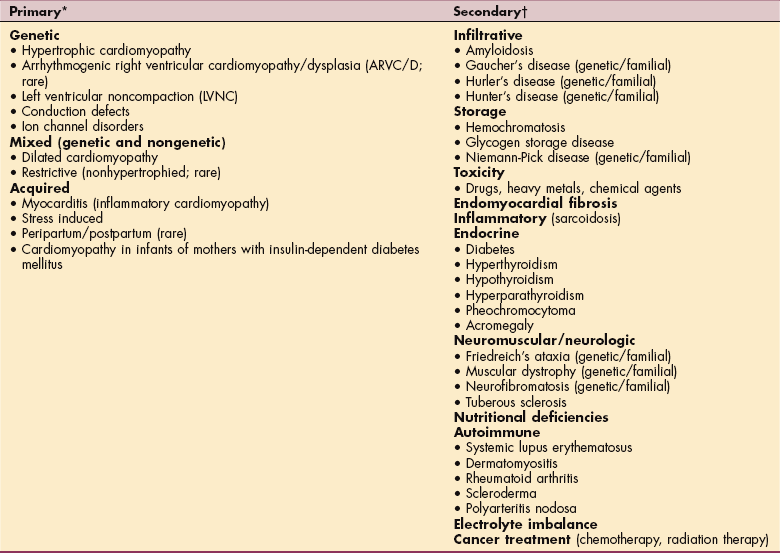
*Predominantly involves the heart.
†Myocardial changes occur as part of a generalized systemic disorder affecting many organs; previously referred to as specific cardiomyopathies. Only the most common diseases associated with cardiomyopathies are listed.
Data from Maron BJ, Towbin JA, Thiene G, et al: Contemporary definitions and classification of the cardiomyopathies: an American Heart Association scientific statement from the Council on Clinical Cardiology, Heart Failure and Transplantation Committee; Quality of Care and Outcomes Research and Functional Genomics and Translational Biology Interdisciplinary Working Groups; and Council on Epidemiology and Prevention, Circulation 113(14):1807-1816, April 11, 2006.
Genetic cardiomyopathies include hypertrophic and arrhythmogenic right ventricular cardiomyopathies, left ventricular noncompaction, conduction system disease, and ion channelopathies. In general, these congenital or familial types of cardiomyopathies are fairly uncommon individually, but a growing number of different types caused by mutations in genetic encoding have been identified.
Mixed cardiomyopathies included dilated and primary restrictive nonhypertrophied cardiomyopathies. An example of an acquired cardiomyopathy is myocarditis. Considerable overlap can occur among the primary classifications within the same person (see Pathogenesis).
Secondary cardiomyopathies involve myocardial pathology as part of a large number and variety of generalized systemic disorders that affect the heart along with other organs at the same time.
Incidence and Risk Factors.: Cardiomyopathy can affect any age group and is often seen in young adults in the second and third decades. The actual incidence is unknown, but the disease may be more common than was previously realized.
This increase in incidence may be attributed to two important variables: (1) improved technology, which has allowed for more accurate evaluation of ventricular dimensions and ventricular wall movement; and (2) an increased incidence of myocarditis, an important precursor to cardiomyopathy, as a result of a wide variety of pathogens, toxins, and autoimmune reactions.
Delayed-onset cardiotoxic effects of chemotherapeutic agents may appear as chronic cardiomyopathy. Risk factors for the development of this type of cardiomyopathy include increasing doses of chemotherapeutic agents and previous mediastinal radiation.345 Doxorubicin hydrochloride (Adriamycin) and daunorubicin hydrochloride (Cerubidine) are the two agents recognized most often in association with dilated cardiomyopathy.
Dilated cardiomyopathy occurs most often in black men between the ages of 40 and 60 years. About one half of the cases of dilated cardiomyopathy are idiopathic, and the remainder result from some known disease process (e.g., rheumatic fever, myasthenia gravis, progressive muscular dystrophy, hemochromatosis, amyloidosis, sarcoidosis). Risk factors for dilated cardiomyopathy may include obesity, long-term alcohol abuse, systemic hypertension, cigarette smoking, infections, and pregnancy.
Peripartum cardiomyopathy is a rare but very serious disease that results in heart failure. It may appear for no apparent reason during the last month of pregnancy or shortly after delivery; incidence is higher among multiparous women older than 30 years, particularly those with malnutrition or preeclampsia. Estimates vary, but the occurrence may be 1 in every 1300 to 4000 deliveries. Maternal death from CHF, blood clots, and infection, and stillbirth can occur. Symptoms of orthopnea, cough, palpitations, and high blood pressure may not occur until several weeks after delivery.
Hypertrophic cardiomyopathy appears to be genetically transmitted as an autosomal dominant trait on chromosome 14; currently 11 mutant genes have been linked with hypertrophic cardiomyopathy. It is still the most frequently occurring cardiomyopathy and the most common cause of sudden cardiac death in the young (including trained athletes).212
Restrictive cardiomyopathy occurs as a result of myocardial fibrosis (e.g., amyloidosis, sarcoidosis, hemochromatosis), hypertrophy, infiltration, or defect in myocardial relaxation.
Pathogenesis.: The exact pathogenesis of cardiomyopathy is unknown; the risk factors mentioned previously seem to lower the threshold for the development of cardiomyopathy. For example, heavy consumption of alcohol is thought to cause dilated cardiomyopathy through three mechanisms: direct toxic effect of alcohol or of its metabolites; effects of nutritional deficiencies, especially thiamine deficiency; and toxic effects of beverage additives, such as cobalt.
Obesity produces an increase in total blood volume and cardiac output because of the high metabolic activity of excessive fat. In moderate to severe cases of obesity, this may lead to left ventricular dilation, increased left ventricular wall stress, and left ventricular diastolic dysfunction.
Regardless of the underlying cause, dilated cardiomyopathy results from extensively damaged myocardial muscle fibers and is characterized by cardiac enlargement. The heart ejects blood less efficiently than normal, so that a large volume of blood remains in the left ventricle after systole, which results in ventricular dilation with enlargement and dilation of all four chambers and eventually leads to CHF (Figs. 12-16 and 12-17).
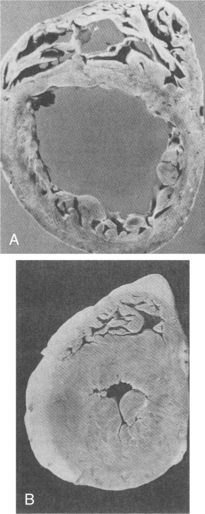
Figure 12-16 A, Cross-sectional view of dilated cardiomyopathy. B, Hypertrophied heart. (From Kinney M: Comprehensive cardiac care, ed 7, St Louis, 1991, Mosby, pp 346, 349.)
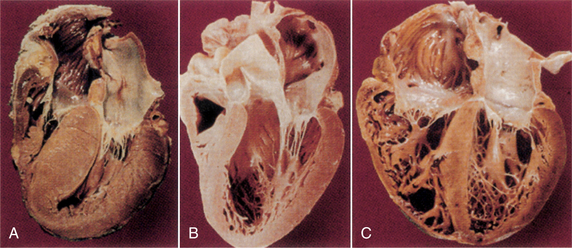
Figure 12-17 Gross pathologic specimens of the cardiomyopathies. A, Hypertrophic cardiomyopathy, showing a marked increase in myocardial mass and preferential hypertrophy of the interventricular septum. B, Normal heart, with normal left ventricular dimensions and thickness. C, Dilated cardiomyopathy, showing marked increase in chamber size. Atrial enlargement is also evident in both cardiomyopathies (A and C). (From Seidman JG, Seidman C: The genetic basis for cardiomyopathy: from mutation identification to mechanistic paradigms, Cell 104:557, 2001.)
Hypertrophic cardiomyopathy is distinguished by inappropriate and excessive left ventricular hypertrophy (thickening of the interventricular septum) and normal or even enhanced cardiac muscle contractile function. Over time, the overgrowth of the wall leads to rigidity in the myocardium. The result is decreased diastolic functioning, since the rigid myocardium cannot relax during the diastolic phase, reducing the amount of blood flowing into the ventricles. Restrictive cardiomyopathy is the least common form; it is identified by marked endocardial scarring (fibrosis) of the ventricles, and the resulting rigidity impairs diastolic filling.
Clinical Manifestations.: Generally, the symptoms of cardiomyopathy are the same as for heart failure (e.g., dyspnea, orthopnea, tachycardia, palpitations, peripheral edema, distended jugular vein).
Dilated cardiomyopathy is characterized by fatigue and weakness; chest pain (unlike angina) may occur. Blood pressure is usually normal or low.
Hypertrophic cardiomyopathy is frequently asymptomatic, sudden death being the presenting sign; in fact, hypertrophic cardiomyopathy is the most common cause of sudden death in young competitive athletes. The most common symptom is dyspnea caused by high pulmonary pressures produced by the elevated left ventricular diastolic pressure; symptoms are often exacerbated during strenuous exercise.
Restrictive cardiomyopathy causes clinical manifestations related to decreasing cardiac output. As cardiac output falls and intraventricular pressures rise, signs of CHF appear. The earliest manifestations may include exercise intolerance, fatigue, and shortness of breath followed by other symptoms such as peripheral edema and ascites.
MEDICAL MANAGEMENT
Diagnosis requires exclusion of other causes of cardiac dysfunction, especially causes of CHF and arrhythmias. Catheterization to assess arteries and valves, echocardiography, chest radiography, blood chemistries, deoxyribonucleic acid (DNA) analysis (for hypertrophic cardiomyopathy), and ECG are specific tests performed. Researchers continue to investigate ways to monitor people with heart failure and to devise noninvasive diagnostic techniques.
The specific treatment of cardiomyopathy is determined by the underlying cause and may include physical, dietary, or pharmacologic interventions; mechanical circulatory support; or surgical intervention, including transplantation. Cardiac resynchronization therapy, the use of a pacemaker-like device to electrically stimulate both ventricles simultaneously (biventricular pacing), has been approved for use in CHF and is under investigation for use with dilated cardiomyopathy.
Alternatively, a cardiac support device called a “heart jacket” is under investigation for use in the United States for cardiomyopathy. This specially designed polyester material is stitched into place around the heart to prevent diseased heart muscle from further enlargement. Clinical safety trials are under way at the University of Pennsylvania.
Idiopathic dilated cardiomyopathy has no known cause; therefore there is no specific therapy. In contrast to the other forms of cardiomyopathy, the progression of myocardial dysfunction in dilated cardiomyopathy may be stopped or reversed if alcohol consumption is reduced or stopped early in the course of the disease.
The β-blockers have an important immunoregulatory role in modifying the dysregulated cytokine network and reducing myocardial contractility and workload.245 Calcium channel blocking agents (see Table 12-5) may be used to relieve symptoms and reduce exercise intolerance. Restrictive cardiomyopathy has no specific treatment interventions. The goal is to control CHF through the use of diuretics, vasodilators, and salt restriction.
PROGNOSIS.
Seventy-five percent of persons diagnosed with idiopathic dilated cardiomyopathy die within 5 years after the onset of symptoms, because diagnosis does not usually occur until advanced stages. Persons with hypertrophic cardiomyopathy can lead long, relatively asymptomatic lives; some people have a history of gradually progressive symptoms, but others experience sudden death, especially during exercise, as the initial diagnostic event. Restrictive cardiomyopathy may cause sudden death as a result of arrhythmia, or a more progressive course may occur, with eventual heart failure. Intervention rarely results in long-term improvement.
Many persons with various types of cardiomyopathy experience stabilization or even an improvement in symptoms, but the end result of cardiomyopathy is sudden death or a fatal progression toward heart failure. No cure exists, outside of cardiac transplantation. Heart transplantation shows a 1-year survival rate of over 80% and a 3-year survival rate of 70% for dilated cardiomyopathy. The 1-year survival rate without transplant is 5%.
Trauma
Any blunt chest trauma, which is especially common in steering wheel impact from an automobile accident, may produce myocardial contusion, resulting in myocardial hemorrhage with little if any myocardial scar once healing is complete. Large contusions may lead to myocardial scars, cardiac rupture, CHF, or formation of aneurysms.
The chest pain of myocardial contusion is similar to that of MI and is often confused with musculoskeletal pain from soft tissue consequences of chest trauma. Myocardial contusion is usually treated similarly to MI, with initial monitoring and subsequent progressive ambulation and cardiac rehabilitation (see Special Implications for the Therapist: Myocardial Infarction).
Penetrating
Penetrating cardiac injuries are most often due to external objects, such as bullets or knives, and sometimes from bony fragments secondary to chest injury. Iatrogenic causes of cardiac penetrating injury include perforation of the heart during catheterization and cardiac trauma from cardiopulmonary resuscitation. Complications include arrhythmias, aneurysm formation, death from infection (e.g., bacterial endocarditis or infection from a retained foreign body), a form of pericarditis associated with this type of injury, ventricular septal defects, and foreign body embolus.
Myocardial Neoplasm
Primary cardiac tumors are rare, with an autopsy frequency of 0.001% to 0.030%.56 Malignant cardiac tumors account for 25% of primary cardiac tumors, with 95% of these tumors being sarcomas arising from connective tissue (e.g., angiosarcoma, rhabdomyosarcoma, mesothelioma, fibrosarcoma) and the remaining 5% being lymphomas.
Some of these sarcomas are limited to the myocardium, replacing functional cardiac tissue with cancerous cells without any intracavity extension. These tumors may produce no cardiac symptoms or may present with arrhythmias and conduction disturbances.
Tumors projecting into a cardiac cavity may present with progressive CHF, precordial pain, pericardial effusion tamponade, arrhythmias, conduction disturbances, and sudden death. Because these tumors occur more frequently in the right side of the heart, right-sided heart failure is more common (jugular venous distention, ascites, systemic edema). People with sarcomas face a rapid functional decline, with death occurring from a few weeks to 2 years after onset of symptoms. These tumors proliferate rapidly, invading and damaging not only the myocardium but contiguous structures such as the venae cavae and tricuspid valve as well.56
Benign primary cardiac tumors occur approximately three times more often than malignant primary tumors, with myxomas accounting for nearly 50% of these primary benign tumors. Myxomas arise most often from the endothelial surface of the left atrium, causing mechanical interference with cardiac function including intracardiac obstruction.47 Tumors located in other cardiac chambers account for 10% of myxomas.56 Other benign cardiac tumors (also rare) include lipoma, papilloma, fibroelastoma, rhabdomyoma, and fibroma.
Signs of obstruction can include right-sided heart failure, pulmonary edema, orthopnea, and dyspnea. Constitutional symptoms include fatigue, fever, weight loss, arthralgia, and myalgia. Embolization caused by fragments from the tumor can also occur in these individuals. If the tumor is in the left side of the heart, the emboli result in infarction damage to the viscera, including the heart, limbs, kidneys, and CNS.274 Because these tumors often lie in the atrial cavity they can (if large enough) cause damage to the mitral valve or even block the orifice of this valve, leading to sudden death. Tumors found in the right side of the heart infrequently lead to pulmonary hypertension and PEs.
Metastases to the heart and pericardium are much more common, occurring 100 to 1000 times more often than primary cardiac tumors.56,284 Melanoma has the highest frequency of metastasis to the heart, with metastases also possible from carcinomas of the lung, breast, and esophagus and malignant leukemia and lymphoma.275
Tumor may involve the heart by one of four metastatic pathways: retrograde lymphatic extension, hematogenous spread, direct contiguous extension, or transvenous extension. Metastatic involvement of the heart and pericardium may go unrecognized until autopsy. Impairment of cardiac function occurs in approximately 30% of cases and is usually attributed to pericardial effusion. The clinical presentation includes shortness of breath, cough, anterior thoracic pain, pleuritic chest pain, or peripheral edema. Cardiac neoplasms come to the attention of a therapist when (1) progressive interference with mitral valve function results in exercise intolerance or exertional dyspnea; (2) embolus causes a stroke; or (3) systemic manifestations occur, including muscle atrophy, arthralgias, malaise, or Raynaud’s phenomenon.
Diagnosis of myxomas and other cardiac neoplasms is usually made by echocardiography followed by imaging studies, with MRI being of greater value in delineating cardiac tumors.284 There are no specific physical or laboratory tests for metastatic heart disease, and diagnosis is difficult as these tumors can masquerade as other cardiac defects. ECG is nonspecific, chest radiography may reveal an enlarged cardiac silhouette, and radionuclide angiography is helpful in diagnosing intracavity tumors. Two-dimensional echocardiography is the method of choice to detect cardiac metastases.275
Treatment of choice for myxomas is usually resection of the tumor, which in most cases is curative. Cardiac rehabilitation may be required according to the individual’s postoperative cardiovascular condition. Recurrence is rare and appears to be the result of incomplete resection of the tumor or intraoperative dislocation of tumor material. The presence of cancer cells in more than one area of the myocardium (multifocal genesis) may also lead to recurrence despite treatment.274
In most cases, cardiac metastases are treated with palliative care because in most cases, advanced disease is present at the time of diagnosis. Radiation is not typically used to treat cardiac neoplasms, which means that radiation heart disease occurs secondarily to radiation therapy for tumors in the area of the heart (e.g., mediastinum, breast, head and neck, and thyroid). A history of such tumors should alert the therapist to the possibility that cardiac defects may be present.
Congenital Heart Disease
Congenital heart disease is an anatomic defect in the heart that develops in utero during the first trimester and is present at birth. Over the past three decades, major advances have been made in the diagnosis and treatment of congenital heart disease, resulting in many more children who have survived to adulthood with surgically corrected or uncorrected anomalies. Today, there are over 1 million adults with congenital heart conditions. Congenital heart disease affects about 8 of every 1000 babies born in the United States, making this the most common category of congenital structural malformation. Other than prematurity, it is the major cause of death in the first year of life. Children with congenital heart disease are also more likely to have extracardiac defects, such as tracheoesophageal fistula, diaphragmatic hernias, and renal abnormalities.
There are two categories of congenital heart disease: cyanotic and acyanotic (Table 12-15). In clinical practice this system of classification is problematic, because children with acyanotic defects may develop cyanosis and those with cyanotic defects may be pink and have more clinical signs of CHF.
Table 12-15
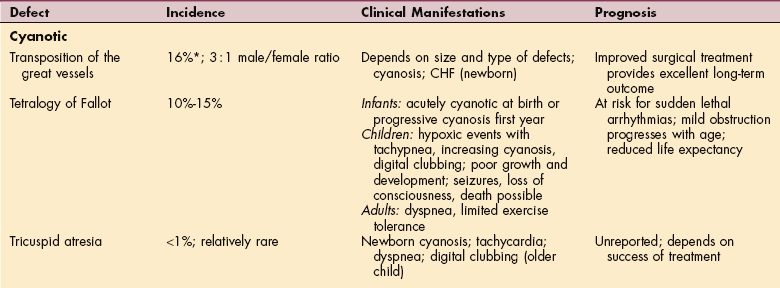

CHF, Congestive heart failure; CHD, congenital heart disease; BP, blood pressure.
*Figures represent percentage of all congenital heart disease.
Cyanotic defects result from obstruction of blood flow to the lungs or mixing of desaturated blue venous blood with fully saturated red arterial blood within the chambers of the heart. Most acyanotic defects involve primarily left-to-right shunting through an abnormal opening.
Etiologic Factors
Many congenital heart diseases have genetic causes with well-known chromosomal anomalies (e.g., trisomy 13, 18, 21; Turner’s syndrome). Approximately 10% of all congenital heart defects are known to be associated with a single identified mutant gene or chromosomal abnormalities; for the remainder, the causes are either unknown or involve multiple factors, such as diabetes, alcohol consumption, viruses, maternal rubella infection during the first trimester, and drugs such as thalidomide.
In the case of atrial septal defect, most result from spontaneous genetic mutations, although some are inherited. Patent ductus arteriosus occurs in pregnancies complicated by persistent perinatal hypoxemia or maternal rubella infection and among infants born at high altitude or prematurely.50
Pathogenesis
The heart begins to form from a tubelike structure during the fourth week after conception. As development progresses, the tube lengthens and forms chambers, septa, and valves. Anything that interferes with this developmental process during the first 8 to 10 weeks of pregnancy can result in a congenital defect (Fig. 12-18).
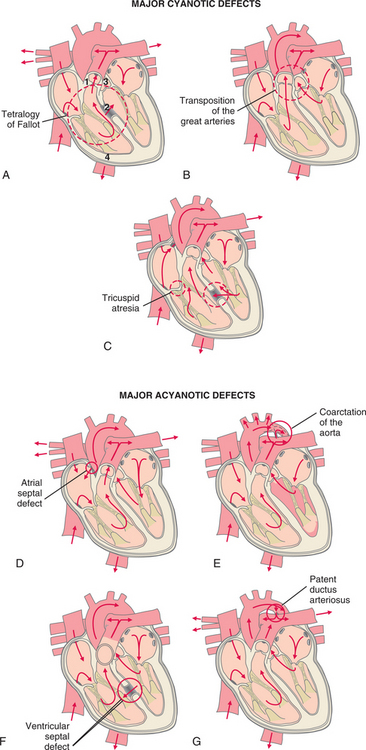
Figure 12-18 Major cyanotic defects (see Fig. 12-1 for normal structure and circulation of the heart): A, Tetralogy of Fallot has four defects: (1) pulmonary stenosis: narrowing at or just below the pulmonary valve; (2) ventricular septal defect (VSD): hole between the two bottom chambers (ventricles) of the heart; (3) aorta is positioned over the ventricular septal defect instead of in the left ventricle; (4) right ventricle is more muscular than normal. B, Transposition of the great arteries: systemic venous blood returns to the right atrium and then goes to the right ventricle and on to the aorta instead of going to the lung via the pulmonary artery. C, Tricuspid atresia: failure of the tricuspid valve to develop with a lack of communication from the right atrium to the right ventricle. Major acyanotic defects: D, Atrial septal defect: blood from the pulmonary vein enters the left atrium, and some blood crosses the atrial septal defect into the right atrium and ventricle. E, Coarctation of the aorta: severe obstruction of blood flow in the descending thoracic aorta. F, Ventricular septal defect: when the left ventricle contracts, it ejects some blood into the aorta and some across the ventricular septal defect into the right ventricle and pulmonary artery. G, Patent ductus arteriosus: some of the blood from the aorta crosses the ductus arteriosus and flows into the pulmonary artery.
Cyanotic.: In transposition of the great vessels (TGV), no communication exists between systemic and pulmonary circulations, so that the pulmonary artery leaves the left ventricle and the aorta exits from the right ventricle. In order for the infant with this condition to survive, there must be communication between the two circuits. In approximately one third of all cases, another associated defect occurs that permits intracardiac mixing (e.g., atrial septal defect, ventricular septal defect, patent ductus arteriosus), but two thirds have no other defect present and severe cyanosis develops.51
Tetralogy of Fallot consists of four classic defects: (1) pulmonary stenosis, (2) large ventricular septal defect, (3) aortic communication with both ventricles, and (4) right ventricular hypertrophy. Tricuspid atresia is a failure of the tricuspid valve to develop, with a lack of communication from the right atrium to the right ventricle. Blood flows through an atrial septal defect or a ductus arteriosus to the left side of the heart and through a ventricular septal defect to the right ventricle and out to the lungs. There is complete mixing of unoxygenated and oxygenated blood in the left side of the heart, resulting in systemic desaturation and varying amounts of pulmonary obstruction.
Acyanotic.: Ventricular septal defect is an abnormal opening between the right and left ventricles that may vary in size from a small pinhole to complete absence of the septum, resulting in a common ventricle. Atrial septal defect is an abnormal opening between the atria, allowing blood from the higher-pressure left atrium to flow into the lower-pressure right atrium.
Coarctation of the aorta is a localized narrowing near the insertion of the ductus arteriosus, resulting in increased pressure proximal to the defect (head, upper extremities) and decreased pressure distal to the obstruction (body, lower extremities).
Patent ductus arteriosus is a failure of the fetal ductus arteriosus (artery connecting the aorta and pulmonary artery) to close within the first weeks of life. The continued function of this vessel allows blood to flow from the high-pressure aorta to the low-pressure pulmonary artery, causing continuous flow from the aorta to the pulmonary artery (referred to as left-to-right shunting). A patent ductus arteriosus rarely closes spontaneously after infancy.
Aortic stenosis is discussed later in this chapter in the section on Diseases Affecting the Heart Valves.
Clinical Manifestations
The most common signs and symptoms include cyanosis and signs of CHF (e.g., dyspnea, pulmonary edema, fatigue). See Table 12-15 for clinical manifestations of each particular defect. Complications may include heart failure, pulmonary edema, pneumonia, hypoxia, and sudden death. There is often a risk of bacterial endocarditis and pulmonary vascular obstructive disease later in life.
MEDICAL MANAGEMENT
As whole genome sequencing continues to develop, identification of genetic mutations predisposing to congenital heart disease may allow preventive measures by modulation of secondary genetic or environmental factors.313 Until then, most forms of congenital heart disease can potentially be detected in utero with the routine use of ultrasonography.
The prenatal diagnosis of a major cardiac malformation requires further assessment for extracardiac and chromosomal disorders. Conversely, diagnosis of Down syndrome (prenatally or postnatally) requires early cardiologic assessment for cardiac anomalies, most commonly atrioventricular and ventricular septal defects. Prenatal knowledge of cardiac anomalies allows for optimal perinatal and postnatal management.
Prenatal screening for maternal rubella antibodies provides important information for further diagnostic testing. In cases where prenatal diagnosis does not occur and when there are no symptoms initially, cardiac anomalies can remain undetected for years and even decades. For example, a person with atrial septal defect may have normal sinus rhythm for the first three decades of life and then develop atrial fibrillation (AF) and supraventricular tachycardia (SVT).51 Clinical diagnosis begins with detection of signs and symptoms, auscultation, and detection of heart murmur. Transesophageal echocardiography, Doppler color-flow echocardiography and now RT-3D echocardiography provide a definitive diagnosis without invasive cardiac catheterization and angiography.
TREATMENT AND PROGNOSIS.
Remarkable innovations in medical and surgical approaches over the past several decades now allow for correction of major cardiac defects in children, even in early infancy. Prenatal (in utero) correction has not been accomplished as yet. Postnatally, curative or palliative (providing relief of symptoms) surgical correction is now available for more than 90% of persons with congenital heart disease.
There is a clear trend toward complete correction of malformations rather than staged procedures to obtain initial palliation and delayed correction. The risk for most surgical procedures is low (between 1% and 5%). Gene transfer to create a patent ductus arteriosus in animal studies may lead the way for additional gene transfer techniques to be successful in humans in the future.
DISEASE AFFECTING THE CARDIAC NERVOUS SYSTEM
Arrhythmias: Disturbances of Rate or Rhythm
Definition and Overview
The number of times the heart beats (rate) and the heart rhythm are generated and regulated by the sinoatrial (SA) node, the internal pacemaker located in the upper right portion of the heart. The signal from the SA node travels through the cardiac conduction system, first through the walls of the atria and then through the walls of the ventricles, causing the atrial (supraventricular) and ventricular chambers of the heart to contract and relax at regular rates necessary to maintain circulation at different levels of activity. An arrhythmia (dysrhythmia) is a disturbance of heart rate or rhythm caused by an abnormal rate of electrical impulse generation by the SA node or the abnormal conduction of impulses.
Arrhythmias can be classified according to their origin as ventricular or supraventricular (atrial), according to the pattern (fibrillation or flutter), or according to the speed or rate at which they occur (tachycardia or bradycardia).
Several types of AF are now recognized, including first-detected-episode AF (may or may not be symptomatic; may self-resolve), recurrent paroxysmal AF (two or more episodes that resolve spontaneously), persistent AF, and permanent AF. Persistent AF is sustained for more than 7 days. It can occur after a first-detected-episode AF or after recurrent paroxysmal AF. Permanent AF, also known as chronic AF, occurs when sinus rhythm cannot be sustained after cardioversion (normal heart rhythm returns spontaneously) or when the decision has been made to let AF continue without efforts to restore normal sinus rhythm.117
Arrhythmias vary in severity from mild, asymptomatic disturbances that require no intervention (e.g., sinus arrhythmia, in which the heart rate increases and decreases with respiration) to catastrophic ventricular fibrillation, which requires immediate resuscitation. The clinical significance depends on the effect on cardiac output and blood pressure, which is partially influenced by the site of origin.
Etiologic Factors and Incidence
Arrhythmias may be congenital or may result from one of several factors, including hypertrophy of heart muscle fibers secondary to hypertension, previous MI, valvular heart disease, or degeneration of conductive tissue that is necessary to maintain normal heart rhythm (called sick sinus syndrome).
Chronic alcohol use and binge drinking have been linked with disturbances in cardiac rhythm, even in individuals without underlying heart disease. Holiday heart syndrome is the term used to describe acute arrhythmia (usually SVT) triggered by excessive alcohol intake in an otherwise healthy person. The affected individual experiences intermittent or continuous palpitations with dyspnea, dizziness, or chest pain often mentioned.
The prevalence of AF doubles with each advancing decade of age beginning at age 50 to 59 years, with a statistically significant increase among men age 65 to 84 years, although this gap closes with advancing age and remains unexplained.175,221
BMI appears to correlate strongly with the risk of AF.100 With each unit increment of BMI the risk of AF increases 3%. A person who is obese has about a 34% greater risk of AF when compared to a person with normal BMI. Moreover, people in the heaviest BMI category have 2.3 times the risk. Improved cardiac care has increased the number of survivors of cardiac incidents who may experience subsequent complications, such as arrhythmias.
Cardiac arrhythmias are very common in the setting of heart failure, with atrial and ventricular arrhythmias often present in the same person. Arrhythmias can occur when a portion of the heart is temporarily deprived of oxygen, disturbing the normal pathway of the heartbeat. Toxic doses of cardioactive drugs (e.g., digoxin and other cardiac glycosides), phenylpropanolamine found in some decongestants, alcohol and caffeine consumption, high fevers, and excessive production of thyroid hormone (hyperthyroidism) may also lead to arrhythmias. In many cases, particularly in younger people, there is no known or apparent cause.
Pathogenesis and Clinical Manifestations
Rate.: The adult heart beats an average of 60 to 100 beats/min; an arrhythmia is considered to be any significant deviation from the normal range. Whether change in heart rate (number of contractions of the cardiac ventricles per period of time) produces symptoms at rest or on exertion depends on the underlying state of the cardiac muscle and its ability to alter its stroke output to compensate.
Rate arrhythmias are of two basic types: tachycardia and bradycardia. Tachycardia occurs when the heart beats too fast (more than 100 beats/min). Tachycardia develops in the presence of increased sympathetic stimulation, such as occurs with fear, pain, emotion, exertion, or exercise; or with ingestion of artificial stimulants, such as caffeine, nicotine, and amphetamines.
Tachycardia is also found in situations in which the demands for oxygen are increased, such as fever, CHF, infection, anemia, hemorrhage, myocardial injury, and hyperthyroidism. Usually the individual with tachycardia perceives no symptoms, and medical intervention is directed toward the underlying cause.
Bradycardia (less than 50 beats/min) is normal in well-trained athletes, but it is also common in individuals taking β-blockers, those who have had traumatic brain injuries or brain tumors, and those experiencing increased vagal stimulation (e.g., from suctioning or vomiting) to the physiologic pacemaker.
Organic disease of the sinus node, especially in older people and those with heart disease, can also cause sinus bradycardia. Bradycardia is usually asymptomatic, but when it is caused by a pathologic condition, the person may experience fatigue, dyspnea, syncope, dizziness, angina, or diaphoresis (profuse perspiration). Medical intervention is not usually required unless symptoms interfere with function or are drug or angina induced; atropine or a mechanical pacemaker can be used to reestablish a more normal heart rate.
Rhythm.: Arrhythmias as variations from the normal rhythm of the heart (especially the heartbeat) are detected when they become symptomatic or during monitoring for another cardiac condition. Abnormalities of cardiac rhythm and electrical conduction can be lethal (sudden cardiac death), symptomatic (syncope or near syncope, dizziness, chest pain, dyspnea, palpitations), or asymptomatic. They are dangerous because they reduce cardiac output so that perfusion of the brain or myocardium is impaired, or they tend to deteriorate into more serious arrhythmias with the same consequences.
The many different types of abnormal cardiac rhythms are usually classified according to their origin (atrial, ventricular), but only the most common ones are included here. Complete discussion of all other cardiac arrhythmias is available.150
Sinus arrhythmia is an irregularity in rhythm that may be a normal variation in athletes, children, and older people or may be caused by an alteration in vagal stimulation. Sinus arrhythmia may be respiratory (increases and decreases with respiration) or nonrespiratory and associated with infection, drug toxicity (e.g., digoxin, morphine), or fever. Treatment for the respiratory type of sinus arrhythmia is not necessary; all other sinus arrhythmias are treated by providing intervention for the underlying cause.
AF is the most common type of SVT or chronic arrhythmia. SVT is also called paroxysmal supraventricular tachycardia (PSVT) or paroxysmal atrial tachycardia (PAT). It is characterized by rapid, involuntary, irregular muscular contractions of the atrial myocardium—quivering or fluttering instead of contracting normally. Consequently, blood remains in the atria after they contract and the ventricles do not fill properly. The heart races, but blood flow may diminish, creating a drop in oxygen levels that results in symptoms of shortness of breath, palpitations, fatigue, and, more rarely, fainting. AF occurs most often as a secondary arrhythmia associated with rheumatic heart disease, dilated cardiomyopathy, atrial septal defect, hypertension, mitral valve prolapse, recurrent cardiac surgery, and hypertrophic cardiomyopathy (conditions that affect the atria).
Secondary AF can also occur in people without cardiac disease but in the presence of a systemic abnormality that predisposes the individual to arrhythmia (e.g., hyperthyroidism, medications, diabetes, obesity, pneumonia, or alcohol intoxication or withdrawal). People with AF are prone to blood clots because blood components that remain in the atria aggregate and attract other components, triggering clot formation. The effect rarely occurs before 72 hours of the first abnormal contraction. AF can result in CHF, cardiac ischemia, and arterial emboli that can result in an ischemic stroke.
Ventricular fibrillation is an electrical phenomenon that results in involuntary uncoordinated muscular contractions of the ventricular muscle; it is a frequent cause of cardiac arrest. Treatment is directed toward depolarizing the muscle, thus ending the irregular contractions and allowing the heart to resume normal regular contractions.
Heart block is a disorder of the heartbeat caused by an interruption in the passage of impulses through the heart’s electrical system. This may occur because the SA node misfires or the impulses it generates are not properly transmitted through the heart’s conduction system. Heart blocks are differentiated into three types determined by ECG testing: first-degree, second-degree, and third-degree (complete) heart block. Causes include CAD, hypertension, myocarditis, and overdose of cardiac medications (e.g., digitalis, calcium channel blockers, β-blockers). Depending on the degree of the heart block, it can cause fatigue, dizziness, or fainting. Heart block can affect people at any age, but this condition primarily affects older people. Mild cases do not require intervention; medication and pacemakers are the two primary forms of management for symptomatic cases.
Sick sinus syndrome, or brady-tachy syndrome, is a complex cardiac arrhythmia and conduction disturbance that is associated with advanced age, CAD, or drug therapy (e.g., digitalis, calcium channel blockers, β-blockers, antiarrhythmics). Sick sinus syndrome as a result of degeneration of conductive tissue necessary to maintain normal heart rhythm occurs most often among older people. A variety of other heart diseases and other conditions (e.g., cardiomyopathy, sarcoidosis, amyloidosis) also may result in sinus node dysfunction. Sick sinus syndrome is characterized by bradycardia alone, bradycardia alternating with tachycardia, or bradycardia with atrioventricular block resulting in cerebral manifestations of light-headedness, dizziness, and near or true syncope.
Sinus node dysfunction is suspected in the older adult experiencing episodes of syncope or near syncope, especially in the presence of heart palpitations. An accurate diagnosis is made with ECG, often requiring a 24-hour Holter monitor to document the arrhythmias described. Treatment for the symptomatic person varies according to the specific arrhythmia manifestations and may include antiarrhythmic agents alone or combined with a permanent-demand pacemaker or withdrawal of agents that may be responsible.
Holiday heart syndrome may occur when the heart responds to the increase in catecholamines (epinephrine, norepinephrine) brought on by excessive alcohol intake. Alcohol metabolites may also cause conduction delays. The toxic effects of alcohol can also cause a rise in the level of free fatty acids, contributing to the onset of this condition.
MEDICAL MANAGEMENT
ECG is the most common test procedure to document arrhythmias, but if the person is not experiencing symptoms, the heartbeat may look normal. Tape-recorded ambulatory ECG may be used to document arrhythmias. The individual may use continuous monitoring (external cardiac monitoring; Holter monitoring; Fig. 12-19) recording all cardiac cycles over a prescribed period of time (usually 24 to 48 hours) or cardiac event monitoring recording ECG just when symptoms are perceived.
Figure 12-19 A, External cardiac monitoring (a form of telemetry, also called ambulatory electrocardiography [ECG] or Holter monitoring) uses a tape recorder that is attached to the skin by ECG electrodes. It is able to record the heart rhythm over a 24-hour period. Any symptoms experienced while wearing the unit should be recorded by the individual wearing the device. The recording is then analyzed. It may detect changes in heart rhythm or changes in the ECG that might indicate a lack of blood supply to the heart. B, Any number of electrodes up to 12 leads can be used. The standard three-electrode system in A consists of positive electrode, negative electrode, and ground electrode. C, The unit is small and convenient and can be clipped to the belt or waistband or slipped into a pocket. (Courtesy Cardiac Science Corporation, Bothell, WA. Used with permission.)
Monitoring is especially helpful in recording sporadic arrhythmias that an office or stress test ECG might miss. Monitoring may also be used by persons recovering from MIs, receiving antiarrhythmic medications, or using pacemakers. New pocket-sized devices to allow home monitoring are available; readings may be stored, and the device can be hooked up to the physician’s ECG or diagnostic computer or transmitted over the telephone. For symptoms that occur rarely (e.g., once every 6 months), an insertable loop recorder can be used. This small device is implanted under the skin in the chest using a local anesthetic. Monitoring units do not replace an ECG and should not be used without a physician’s approval.
TEE imaging study using an ultrasonic transducer mounted on the tip of a flexible instrument is used to detect cardiac emboli before medications are initiated to control rate and rhythm. If a serious arrhythmia is suspected, an electrophysiologic study (EPS) can be performed. This test is an invasive study that uses wires placed via catheterization to electronically stimulate the heart in an attempt to reproduce the arrhythmia.
TREATMENT.
The goal of treatment is to control ventricular rate, prevent thromboembolism, and restore normal sinus rhythm if possible. Normal heart rhythm returns spontaneously (called cardioversion) almost immediately in some cases, especially if there is no underlying heart disease. When conversion to normal rate and rhythm does not occur, there are two major approaches to cardioversion: electrical and pharmacologic.
The electrical method employs the use of a device called a defibrillator and is usually most effective and may require several weeks of anticoagulant therapy (warfarin) to reduce stroke risk. Anyone who has been in AF less than 48 hours but is hemodynamically unstable with serious signs and symptoms related to AF will need immediate electrical cardioversion. Low-voltage electric shocks interrupt the irritable foci of the heart, letting the SA node resume its role as a primary pacemaker.285
Pharmacologic treatment may include agents prolonging depolarization and/or other cardiovascular medications (see Table 12-5). If successful, cardioversion restores sinus rhythm, and drug therapy is used to maintain normal heart rate and rhythm. Even with successful electrical cardioversion, long-term antiarrhythmic and anticoagulation drug therapy is used to sustain normal sinus rhythm.
Some tachycardias can be treated with radio-wave ablation, a nonsurgical but invasive technique that uses catheterization to thread wires into the heart through which radio waves can be aimed at the heart tissue where the arrhythmia originates. The catheter-delivered quick bursts of current destroy the specific areas of heart muscle that are generating the abnormal electrical signals causing the arrhythmia. One complication of this technique is the potential destruction of the conducting system (the heart’s own internal pacemaker), which necessitates surgical implantation of an artificial pacemaker for some people.
Pacemakers, implants designed to replace the heartbeat by delivering a battery-supplied electrical stimulus through leads attached to electrodes in contact with the heart, may be used in cases of bradycardia, heart block, or refractory tachycardia. Refractory tachycardia is a condition in which the heart is beating very quickly, but only a portion of those beats are functional; many more beats just echo or make a beat but without contractile force behind the blood flow. Functionally, the heartbeat is actually very slow.
Pacemakers initiate the heartbeat when the heart’s intrinsic conduction system fails or is unreliable. In the case of life-threatening arrhythmias (e.g., ventricular tachycardia, ventricular fibrillation) that do not respond to other types of intervention, a device called an implant- able cardioverter-defibrillator may be implanted (Fig. 12-20). The cardioverter-defibrillator monitors the heart rhythm, and if the heart starts beating abnormally, it generates an electric shock to restore the normal sinus (heart) rhythm.
Figure 12-20 A, Placement of an implantable cardioverter defibrillator (ICD) and epicardial lead system. The generator is placed in a subcutaneous “pocket” created in the left upper abdominal quadrant. The epicardial screw-in sensing leads monitor the heart rhythm and connect to the generator. If a life-threatening dysrhythmia is sensed, the generator can pace-terminate the dysrhythmia or deliver electrical cardioversion or defibrillation through the epicardial patches. With this system, the leads/patches must be placed during open chest surgery. B, Transvenous lead system. Open chest surgery is not needed to place this unit. The pacing/cardioversion/defibrillation functions are all contained in a lead (or leads) inserted into the right atrium and ventricle. New generators are small enough to place in the pectoral region. C, An example of a dual-chamber ICD (Medtronic Gem II DR) with tiered therapy and pacing capabilities. D, Tiered therapy is designed to use increasing levels of intensity to terminate ventricular dysrhythmias. (From Urden LD: Thelan’s critical care nursing: diagnosis and management, ed 5, St Louis, 2006, Mosby. Courtesy Medtronic Inc., Minneapolis, MN.)
For people whose arrhythmias are resistant to pharmacologic therapy, another surgical intervention is available called the maze procedure. This procedure requires open heart surgery and involves a series of mazelike cuts made in the atria and then sewn back together. The scar tissue that forms during the healing process blocks faulty circuits, preventing AF. Many people still need a pacemaker and drug therapy to maintain normal rate and rhythm. A more refined version of this procedure (catheter maze) takes a percutaneous, nonsurgical, noninvasive approach using radiofrequency ablation to destroy tissue.
A more recently developed treatment intervention called ventricular resynchronization therapy is gaining recognition for the treatment of intraventricular conduction disturbances associated with CHF. This redesigned pacemaker resynchronizes the right and left ventricles so they pump at the same time, making the heart pump more forcefully instead of pumping faster (as occurs with a typical pacemaker or in the case of CHF when the heart beats faster to compensate for a weak pumping mechanism).339
PROGNOSIS.
About half of all individuals with AF will spontaneously convert to normal sinus rhythm within 24 to 48 hours; this is less likely to occur in people whose AF has lasted more than 7 days.360
Sudden cardiac arrest (sudden death) is responsible for 300,000 deaths annually and is often preceded by fatal heart dysrhythmias in people who have no prior history of heart disease. In fact, new data from the Framingham Heart Study indicate that AF is independently associated with a substantially increased risk for death in both men and women, even after adjustment for age and associated factors, such as hypertension, CHF, and stroke.
Defibrillation within the first few minutes of cardiac arrest can save up to 50% of lives; by comparison, an estimated 5% of sudden cardiac arrest victims in the United States survive without this treatment. Early defibrillation is the key to survival, and toward that end, emergency medical teams are using portable automatic external defibrillator (AED) units that use a computer program to sense whether a defibrillatory shock is warranted and will initiate the shock.270
The most appropriate and effective drug or drug combination remains unknown, and side effects of long-term rate and rhythm control intervention (e.g., organ toxicity of the lung, liver, and thyroid; aggravation of a preexisting arrhythmia or development of a new arrhythmia instead of preventing it) may prevent long-term use of drug therapy. About 10% of affected individuals continue to have episodes despite treatment, and one half who are treated have a recurrence within 6 months.
DISEASES AFFECTING THE HEART VALVES
Heart problems that occur secondary to impairment of valves may be caused by infections such as endocarditis, congenital deformity, or disease (e.g., rheumatic fever, coronary thrombosis). Valve deformities are classified as functional (e.g., stenosis, insufficiency) or anatomic (e.g., prolapse; congenital deformities; deformities caused by rheumatic fever, trauma, infection, ischemia) (Fig. 12-21).
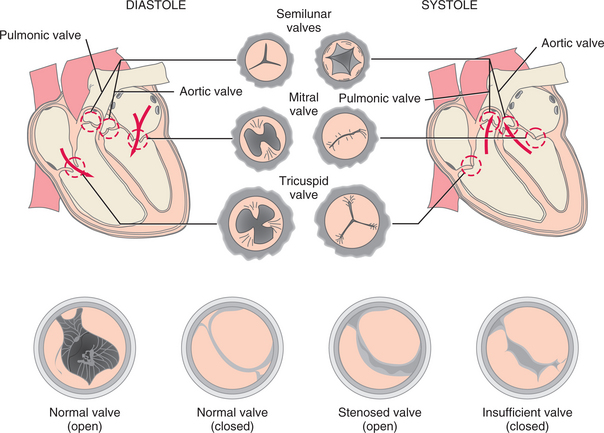
Figure 12-21 Valves of the heart. A, The pulmonic, aortic, mitral, and tricuspid valves are shown here as they appear during diastole (ventricular filling) and systole (ventricular contraction). B, Normal position of the valve leaflets, or cusps, when the valve is open and closed; fully open position of a stenosed valve; closed regurgitant valve showing abnormal opening into which blood can flow back.
Stenosis is a narrowing or constriction that prevents the valve from opening fully and may be caused by scars or abnormal deposits on the leaflets. Valvular stenosis causes obstruction to blood flow, and the chamber behind the narrow valve must produce extra work to sustain cardiac output.
Insufficiency (also referred to as regurgitation) occurs when the valve does not close properly and causes blood to flow back into the heart chamber. The heart gradually dilates in response to the increased volume of work; severe degrees of incompetence are possible in the absence of symptoms. Prolapse affects the mitral or tricuspid valve and occurs when enlarged leaflets bulge backward into the atrium.
Valve conditions increase the workload of the heart and require the heart to pump harder to force blood through a stenosed valve or to maintain adequate flow if blood is seeping back. Initially the cardiovascular system compensates for the overload and the person remains asymptomatic, but eventually as stenosis or insufficiency progresses, cardiac muscle dysfunction and accompanying symptoms of heart failure (breathlessness, dyspnea) develop.
Over the past 15 years, advances in surgical techniques and a better understanding of timing for surgical intervention have brought tremendous improvement in the clinical outcome of people with valvular heart disease, extending survival rates with less overall morbidity.63
The presence of CAD in clients with either mitral or aortic valve disease is a negative prognostic indicator; ischemic mitral regurgitation carries the worst prognosis, with higher operative mortality and lower long-term survival compared with nonischemic cases.63
Heart transplantation may be necessary when the risk of surgery is prohibitively high in some cases of valvular disease. Continued advances in noninvasive assessment (e.g., RT-3D echocardiography) and noninvasive treatment (e.g., gene therapy, valves grown from blood vessel cells, and even valve self-repair with tissue-engineering techniques) should improve the outlook for anyone with valvular heart disease in the years to come.
Mitral Stenosis
Etiologic Factors and Pathogenesis
Mitral stenosis is a sequela of rheumatic heart disease that primarily affects women. Often a history of rheumatic fever is absent. Because the mitral valve is thickened, it opens in early diastole with a snap that is audible on auscultation and then closes slowly with a resultant murmur.
The anterior and posterior leaflets are fixed like a funnel with an opening in the center, and they move together, rather than in opposite directions. When the valve has narrowed sufficiently, left atrial pressure rises to maintain normal flow across the valve and to maintain a normal cardiac output. This results in a pressure difference between the left atrium and the left ventricle during diastole.
Clinical Manifestations
In mild cases, left atrial pressure and cardiac output remain normal, and the person is asymptomatic, perhaps until pregnancy or the development of AF, when dyspnea and orthopnea develop. In moderate stenosis, dyspnea and fatigue appear as the left atrial pressure rises and mechanical obstruction of filling of the left ventricle reduces cardiac output.
With severe stenosis, left atrial pressure is high enough to produce pulmonary venous congestion at rest and reduce cardiac output, with resulting dyspnea, fatigue, and right ventricular failure. Lying down at night further increases the pulmonary blood volume, causing orthopnea and paroxysmal nocturnal dyspnea.
MEDICAL MANAGEMENT
DIAGNOSIS, TREATMENT, AND PROGNOSIS.
Echocardiography is the most valuable technique for assessing mitral valve stenosis and providing information about the condition of the valve and left atrial size. Doppler techniques (measuring blood flow using ultrasound) can be used to determine the severity of the problem.
Because mitral stenosis may be asymptomatic, intervention is delayed until symptoms develop. Mitral stenosis may be present for a lifetime with few or no symptoms, or it may become severe in a few years. The onset of AF accompanied by more severe symptoms may be treated pharmacologically (digoxin, antiarrhythmic agents, anticoagulants).
Surgery may be indicated in the presence of uncontrollable pulmonary edema, severe dyspnea limiting function, pulmonary hypertension, arrhythmia, or systemic emboli uncontrolled by anticoagulation treatment. Surgical procedures include valve repair (commissurotomy to break apart the adherent leaves), replacement with an artificial valve, or balloon valvotomy.
In many cases, balloon valvotomy provides excellent mechanical relief with prolonged benefit, in contrast to the poor results in aortic stenosis.63 Operative mortality rates are low; problems associated with prosthetic valves may occur because of thrombosis, leaking, endocarditis, or degenerative changes in tissue valves.
Mitral Regurgitation
Etiologic Factors and Pathogenesis
Mitral regurgitation has many possible causes, but involvement of the mitral valve from ischemic heart disease accounts for approximately one half of all cases. Other secondary causes include infective endocarditis (valve perforation), dilated cardiomyopathy, rheumatic disease, collagen vascular disease, rupture of the chordae tendineae, and, rarely, cardiac tumors. It is independently associated with female gender, lower BMI, and older age. Evidence suggesting that mitral regurgitation may be induced by appetite-suppressant medications has resulted in new research investigating the relationship of mitral regurgitation to obesity.168
During left ventricular systole, the mitral leaflets do not close normally, and blood is ejected into the left atrium as well as through the aortic valve. In acute regurgitation, left atrial pressure rises abruptly, possibly leading to pulmonary edema. When regurgitation is a chronic condition, the left atrium enlarges progressively; the degree of enlargement usually reflects the severity of regurgitation.
Clinical Manifestations
Unfortunately, people with mitral regurgitation lack early warning signs and may remain asymptomatic until severe and often irreversible left ventricular dysfunction occurs. For many years the left ventricular end-diastolic pressure and the cardiac output may be normal at rest, even with considerable increase in left ventricular volume. Eventually, left ventricular overload may lead to left ventricular failure. People with mitral regurgitation experience exertional dyspnea (because of increased left atrial pressure) and exercise-induced fatigue (because of reduced cardiac output). AF may also develop.
MEDICAL MANAGEMENT
DIAGNOSIS, TREATMENT, AND PROGNOSIS.
The diagnosis is primarily clinical (auscultation), but it can be confirmed and quantified by color Doppler echocardiography. Other testing procedures may include cardiac catheterization to assess the regurgitation, left ventricular function, and pulmonary artery pressure; coronary arteriography to determine the cause of the lesion and for preoperative evaluation; and nuclear medicine techniques to measure left ventricular function and estimate the severity of regurgitation.
Persons with chronic lesions who are asymptomatic require careful monitoring for left ventricular function and may require surgery even if no symptoms are present. Unlike stenosis, regurgitant lesions may progress insidiously, causing left ventricular damage before symptoms have developed.
Surgical intervention may be recommended if left ventricular function is impaired or when activity becomes severely limited. Mitral valve repair has a lower operative mortality and a better late outcome than mitral valve replacement. Acute mitral regurgitation secondary to MI often requires emergency surgery, but the surgical risk is high and the outcome poor. Acute non–MI-related mitral regurgitation has a much better prognosis with higher postoperative survival after well-timed mitral valve repair. Indicators of poorer prognosis include mitral valve replacement, age more than 75 years, and the presence of CAD.63
Mitral Valve Prolapse
Incidence and Etiologic Factors
Mitral valve prolapse (MVP) has been described as a common disease with frequent complications. There is some dispute about the incidence of MVP. According to data from the Framingham Heart Study, MVP is not as prevalent as previously reported (2.4% compared to previously reported 10% or higher).
The American Heart Association and other sources report that MVP occurs in about 2% to 6% of “normal” adults, especially young women, detected most often during pregnancy.13,65 Other researchers report that MVP is equally common in men and women, although men seem to have a higher incidence of complications.144
MVP is characterized by a slight variation in the shape or structure of the mitral (left atrioventricular) valve. This structural variation has many other names, including floppy valve syndrome, Barlow’s syndrome, myxomatous mitral valve syndrome, and click-murmur syndrome. Barlow’s syndrome is a controversial clinical syndrome that may have as its only manifestation MVP without regurgitation.
The cause remains unknown, although there may be a genetic component involving the angiotensin receptor gene resulting in autonomic or neuroendocrine dysfunction.323 Results of family studies of people with MVP favor an autosomal dominant pattern of transmission for primary MVP with nearly 100% gene expression by females.97 This condition usually occurs in isolation; however, it can be associated with a number of other conditions, such as Marfan’s syndrome, rheumatic fever, endocarditis, myocarditis, atherosclerosis, SLE, muscular dystrophy, acromegaly, adult polycystic kidney disease, and cardiac sarcoidosis.
Pathogenesis
MVP is a pathologic, anatomic, and physiologic abnormality of the mitral valve apparatus affecting mitral valve leaflet motion. Normally, when the lower part of the heart contracts, the mitral valve remains firm and prevents blood from leaking back into the upper chambers. In MVP, the slight variation in shape of the mitral valve allows one part of the valve, the leaflet, to billow back into the left atrium during contraction of the ventricle. One or both of the valve leaflets may bulge into the left atrium during ventricular systole. Usually the amount of blood that leaks back into the left atrium is not significant, but in a small number of people, it develops into mitral regurgitation. MVP is the most common cause of isolated mitral regurgitation.
The presence of symptoms linked to neuroendocrine dysfunctions or to the autonomic nervous system has led to the recognition of a pathologic condition known as mitral valve prolapse syndrome (MVPS). Usually diagnosed by chance in asymptomatic individuals during routine tests, MVPS (prolapse with or without mitral regurgitation) has a high clinical incidence of neuropsychiatric symptoms (e.g., anxiety disorder, panic attacks, depression), as well as symptoms of autonomic dysfunction (e.g., postural hypotension, palpitations, cold hands and feet, shortness of breath, chest pain).
As the autonomic nervous system is being formed in utero, the mitral valve is also being formed. If there is a slight variation in the structure of the heart valve, there is also a slight variation in the function or balance of the autonomic nervous system. The importance of recognizing that MVP may occur as an isolated disorder or with other coincident findings has led to the use of both terms.
Clinical Manifestations
More than 50% of all people with MVP are asymptomatic, another 40% experience occasional symptoms that are mildly to moderately uncomfortable, and only 1% suffer severe symptoms and lifestyle restrictions. Although the malformation occurs during gestation, it usually remains unnoticed until young adulthood. The person usually becomes aware of symptoms suddenly, and there does not appear to be any correlation between the severity of symptoms and the severity of the prolapse.
The most common triad of symptoms associated with MVP is profound fatigue that cannot be correlated with exercise or stress, palpitations, and dyspnea. Fatigue may not be related to exertion, but deconditioning from prolonged inactivity may develop, further complicating the picture.
The therapist is more likely to see the individual with MVP associated with connective tissue disorders or the MVPS with autonomic dysfunction. Frequently occurring musculoskeletal findings in clients with MVPS include joint hypermobility, temporomandibular joint (TMJ) syndrome, pectus excavatum, mild scoliosis, straight thoracic spine, and myalgias. The increased joint mobility that has been identified in a small proportion of persons with MVP does not appear to lead to either severe arthritis or frequent joint dislocations.98
Other symptoms associated with MVPS may include tremors, swelling of the extremities, sleep disturbances, low back pain, irritable bowel syndrome, excessive perspiration or inability to perspire, rashes, muscular fasciculations, visual changes or disturbances, difficulty in concentrating, memory lapses, and dizziness.
Chest pain or discomfort may occur as a result of autonomic nervous system dysfunction (dysautonomia). The autonomic nervous system imbalance results in inadequate relaxation between respirations and eventually causes the chest wall muscles to go into spasm. The chest pain is sharp, lasts several seconds, and is usually felt to the left of the sternum. It is intermittent pain that may occur frequently for a few weeks and then disappear completely, only to return again some weeks later.
MEDICAL MANAGEMENT
DIAGNOSIS, TREATMENT, AND PROGNOSIS.
MVP is often discovered during routine cardiac auscultation or when echocardiography is performed for another reason. It is characterized by a symptomatic clinical presentation and clicking noise on auscultation in late systole, with or without sounds of valvular leak (murmur).
The mitral valve begins to prolapse when the reduction of left ventricular volume during systole reaches a critical point at which the valve leaflets no longer coapt (edges approximate together); at that instant, the click occurs and the murmur begins. Complete diagnostic (major and minor) criteria have been outlined elsewhere.48 Echocardiography may be used to confirm the diagnosis, and ECG, event, or Holter monitoring (see Fig. 12-19) to show arrhythmias may be used.
Management includes reassurance; β-blockers to control arrhythmias; an exercise program to improve overall cardiovascular function; counseling to eliminate caffeine, alcohol, and cigarette use; and administration of antibiotics before any invasive procedure (including dental work, sigmoidoscopy) as prophylaxis against endocarditis.
Rarely, surgical replacement of the valve may be recommended when severe structural problems are present that contribute to reduced activity or deterioration of left ventricular function from progression of MVP to mitral regurgitation.
MVP or MVPS is a benign condition in the vast majority of people. It is not life-threatening and only rarely does it result in complications or significantly alter a person’s lifestyle. Progressive mitral regurgitation with gradual increase in left atrial and left ventricular size, AF, pulmonary hypertension, and the development of CHF occur in 10% to 15% of people with both murmurs and clicks. Men older than 50 years are most often affected.48
According to new data available, people with MVP or MVPS are not at greater risk for heart failure, other forms of heart disease, or early death from stroke as was once thought.
Aortic Stenosis
Etiologic Factors and Pathogenesis
Aortic stenosis is a disease of aging that is likely to become more prevalent as the proportion of older people in our population increases. It is most commonly caused by progressive valvular calcification either superimposed on a congenitally bicuspid valve or, in the older adult, involving a previously normal valve following rheumatic fever.
Other risk factors for aortic stenosis are the same as those for heart disease and include obesity, a sedentary lifestyle, smoking, and high cholesterol. Factors affecting the progression of the disease remain uncertain. Over 80% of affected persons are men, and when women are affected, differences are noted (e.g., women with aortic stenosis have thicker ventricular walls reducing wall stress and higher ejection fractions) that require different postoperative management (e.g., low cardiac output requiring volume expansion rather than the use of pressor agents).63 Ejection fraction is the amount of blood the ventricle ejects; the normal ejection fraction is about 60% to 75%. A decreased ejection fraction is a hallmark finding of ventricular failure.
Although the deformed valve is not stenotic at birth, it is subjected to abnormal hemodynamic stress, which may lead to thickening and calcification of the leaflets with reduced mobility. The orifice of the aortic valve narrows, causing increased resistance to blood flow from the left ventricle into the aorta.
Outflow obstruction increases pressure within the left ventricle as it tries to eject blood through the narrow opening, causing decreased cardiac output, left ventricular hypertrophy, and pulmonary vascular congestion. Preschool and school-aged children are more likely to have a bicuspid valve; teenagers and young adults present with three leaflets, but these are partially fused.
Clinical Manifestations
In adults, aortic stenosis is usually asymptomatic until the sixth (or later) decade. Characteristic sounds may be heard on auscultation, but cardiac output is maintained until the stenosis is severe and left ventricular failure, angina pectoris, or exertional syncope develops. The origin of exertional syncope in aortic stenosis remains controversial; it is perhaps caused by an exercise-induced decrease in total peripheral resistance, which is uncompensated because cardiac output is restricted by the stenotic valve. The most common sign of aortic stenosis is a systolic ejection murmur radiating to the neck (usually heard best in the aortic area). Sudden death may occur, even in previously asymptomatic individuals.
MEDICAL MANAGEMENT
DIAGNOSIS, TREATMENT, AND PROGNOSIS.
The clinical assessment of aortic stenosis can be difficult, especially in the older person. Echo Doppler (echocardiography with Doppler ultrasonography) is diagnostic in most cases. ECG may show left ventricular hypertrophy, and x-ray or fluoroscopy may show a calcified aortic valve. Coronary angiography may be necessary in older adults at risk for coronary disease before valve replacement.
Pharmacologic therapy has limited use in this condition. Surgical intervention is usually required for the symptomatic person and should be strongly considered for the asymptomatic person because of the risk of sudden death.
Surgical procedures may include valve replacement with a mechanical prosthesis or bioprosthesis (made with biologic material) or use of the pulmonary valve in place of the aortic valve and replacement of the pulmonary valve with a homograft (Ross procedure). Homografts have been shown to have a superior durability compared to xenogenic biologic prostheses. Approximately 40% were still in place 20 years after implantation in the aortic position. Their low rate of thromboembolic events made life-time anticoagulative therapy unnecessary, and their hemodynamics may be superior to that of all other heart valve prostheses.137
A less invasive alternative to aortic valve replacement, balloon valvuloplasty (splitting the stenotic valve with a balloon-tipped catheter that is introduced into the valve and inflated), has been used in treatment for aortic stenosis. Limited long-term success with less reliable and less durable outcomes has resulted in this procedure’s being considered palliative rather than curative. It may be useful in alleviating symptoms in people who are not candidates for aortic valve replacement because of other medical problems.
Adults with aortic stenosis who are asymptomatic have a normal life expectancy; they should receive prophylactic antibiotics against infective endocarditis. Once symptoms appear, the prognosis is poor without surgery but excellent with valve replacement even in the older adult, especially in the absence of coexisting illnesses.63
The onset of angina, exercise-induced syncope, or cardiac failure indicates a poor prognostic outcome resulting in death. Mortality rises to 10% after age 80 years. Bioprostheses may develop degenerative changes, requiring replacement in 2 to 20 years. This is quite variable and depends on the person’s age at the time of implantation.
Aortic Regurgitation (Insufficiency)
Etiologic Factors and Pathogenesis
In the past, aortic regurgitation occurred secondary to rheumatic fever, but antibiotics have reduced the number of rheumatic fever–related cases. Nonrheumatic causes account for most cases today, including congenitally bicuspid valves, infective endocarditis (valve destruction by bacteria), and hypertension. Aortic regurgitation may also occur secondary to aortic dissection with or without aortic aneurysm (see Fig. 12-27), ankylosing spondylitis, Reiter’s syndrome, collagen vascular disease, syphilis, and Marfan’s syndrome.
Figure 12-27 Dissecting aneurysm. Cross-section of the aorta with dissecting aneurysm showing true aortic lumen (above and right) compressed by dissecting column of blood that separates the media and creates a false lumen. (From Kissane JM, ed: Anderson’s pathology, St Louis, 1990, Mosby.)
When cardiac systole ends, the aortic valve should completely prevent the flow of aortic blood back into the left ventricle. A leakage during diastole is referred to as aortic regurgitation or aortic insufficiency. When aortic regurgitation develops gradually, the left ventricle compensates by both dilation and enough hypertrophy to maintain a normal wall thickness/cavity ratio, thereby preventing development of symptoms. Eventually the left ventricle fails to stand up under the chronic overload, and symptoms develop.
Clinical Manifestations
Longstanding aortic regurgitation may remain asymptomatic even as the deformity increases, causing enlargement of the left ventricle. The large total stroke volume in aortic regurgitation produces a wide pulse pressure and systolic hypertension, resulting in exertional dyspnea, fatigue, and excessive perspiration with exercise as the most frequent symptoms; paroxysmal nocturnal dyspnea and pulmonary edema may also occur. Angina pectoris or atypical chest pain may be present, but this is uncommon in the absence of CAD.
MEDICAL MANAGEMENT
DIAGNOSIS, TREATMENT, AND PROGNOSIS.
Once aortic regurgitation is suspected on physical examination, echocardiography with Doppler examination of the aortic valve can help estimate its severity. Aortography during catheterization helps confirm the severity of the disease. Scintigraphic studies can quantify left ventricular function and functional reserve during exercise and provide a useful predictor of prognosis.
Acute aortic regurgitation may lead to left ventricular failure; surgical reconstruction or replacement of the valve (Ross procedure; see the section on Aortic Stenosis) is advisable before onset of permanent left ventricular damage (usually before ejection fraction falls below 55%), even in asymptomatic cases. Chronic regurgitation carries a poor prognosis without surgery when significant symptoms develop. Medical therapy may include vasodilators to reduce the severity of regurgitation and diuretics and digoxin to stabilize or improve symptoms.
Tricuspid Stenosis and Regurgitation
Tricuspid stenosis may be congenital or rheumatic in origin and is uncommon. Exercise testing and rehabilitation do not occur until after valve surgery. Tricuspid regurgitation may occur secondary to carcinoid syndrome, SLE, or infective endocarditis among injection drug users, and in the presence of mitral valve disease. Surgical repair is more common than valvular replacement for tricuspid valve disease.
Infective Endocarditis
Infective, or bacterial, endocarditis is an infection of the endocardium, the lining inside the heart, including the heart valves; it most commonly damages the mitral valve, followed by the aortic, tricuspid, and pulmonic valves. Bacterial endocarditis may involve normal valves but more often affects valves that have been damaged by some other previous pathologic process (e.g., rheumatic disease, congenital defects, cardiac surgery).
Endocarditis is categorized as either acute or subacute, depending on the clinical course, organisms, and condition of the valves. Endocarditis can occur at any age but rarely occurs in children; one half of all clients diagnosed are older than 60 years. Older adults may be at greater risk of endocarditis because valvular endocardial disruption is more common, immunity is impaired, and nutrition is poor. Endocarditis is more prevalent among men than women.
Etiologic and Risk Factors
Endocarditis is frequently caused by bacteria (particularly streptococci or staphylococci) normally present in the mouth, respiratory system, or GI tract or as a result of abnormal growths on the closure lines of previously damaged valves (e.g., rheumatic disease).
In addition to those with previous valvular damage, persons with prosthetic heart valves, injection drug users, immunocompromised clients (including individuals receiving treatment for cancer), women who have had a suction abortion or pelvic infection related to intrauterine contraceptive devices, and postcardiac surgical clients are at high risk for developing endocarditis. Congenital heart disease and degenerative heart disease, such as calcific aortic stenosis, may also cause endocarditis.
Hospital-acquired infective endocarditis has become more common as a result of iatrogenic endocardial damage produced by surgery, intracardiac pressure-monitoring catheters, ventriculoatrial shunts, and hyperalimentation lines that reach the right atrium. Portals of entry for microorganisms are also provided by wounds, biopsy sites, pacemakers, IV and arterial catheters, indwelling urinary catheters, and intratracheal airways.
Pathogenesis
As an infection, endocarditis causes inflammation of the cardiac endothelium with destruction of the connective tissue. As these bloodborne microorganisms adhere to the endocardial surface, destruction of the connective tissue occurs as a result of the action of bacterial lytic enzymes. The surface endocardium becomes covered with fibrin and platelet thrombi that attract even more thrombogenic material.
The result is the formation of wartlike growths called vegetations. These vegetations, consisting of fibrin and platelets, can break off from the valve, embolize, and cause septic infarction in the myocardium, kidney, brain, spleen, abdomen, or extremities. These thromboemboli contain bacteria that not only cause ischemic infarcts but also form new sites of infection transforming into microabscesses. Bacteria may further invade the valves, causing intravalvular inflammation, destroying portions of the valves, and causing valve deformities.
Splinter hemorrhages of the nail beds may be caused by distal vasospasm, embolic events, or other local factors promoting engorgement and bleeding of the capillaries that lie right below the nail. The cause of digital clubbing is unclear, but perhaps platelet clumps lodge in the nail bed capillaries of the fingers and toes and release platelet-derived growth factor, resulting in the pathologic changes of clubbed digits.
Petechiae (small, red, nonblanching macules on the conjunctivae, palate, buccal mucosa, heels, shoulders, arms, legs, and upper chest) are thought to involve microemboli, but some have also suggested that immune complex vasculitis is the primary process.173 Infective endocarditis of the right-side heart valves occurs commonly in injection drug users. Although a variety of hypotheses have been put forward to explain this phenomenon, no single explanation has been proven.
Clinical Manifestations
Endocarditis can develop insidiously, with symptoms remaining undetected for months, or it can cause symptoms immediately, as in the case of acute bacterial endocarditis. Clinical manifestations can be divided into many groups (Table 12-16). It causes varying degrees of valvular dysfunction and may be associated with manifestations involving any number of organ systems, including lungs, eyes, kidneys, bones, joints, and CNS. The mitral, aortic, tricuspid, and pulmonic valves can be affected (descending order); more than one valve can be infected at the same time. Neurologic signs and symptoms are predominant in about one third of all cases in those people over 60 years. The classic findings of fever, cardiac murmur, and petechial lesions of the skin, conjunctivae, and oral mucosa are not always present.
Up to 50% of people with infective endocarditis initially have musculoskeletal symptoms, including arthralgia (most common), arthritis, low back pain, and myalgias. One half of these people will have only musculoskeletal symptoms without other manifestations of endocarditis. The early onset of joint pain and myalgia as the first sign of endocarditis is more likely if the person is older and has had a previously diagnosed heart murmur.
Proximal joints are most often affected, especially the shoulder, followed by knee, hip, wrist, ankle, metatarsophalangeal and metacarpophalangeal joints, and acromioclavicular joints (order of declining incidence). Most often one or two joints are painful, and symptoms begin suddenly, accompanied by warmth, tenderness, and redness. Symmetric arthralgia in the knees or ankles may lead to a diagnosis of rheumatoid arthritis, but as a rule, morning stiffness is not as prevalent in clients with endocarditis as in those with rheumatoid arthritis or polymyalgia rheumatica.
Bone and joint infections are particularly common among injection drug users. The most common sites of osteoarticular infections are the vertebrae, wrist, and sternoclavicular and sacroiliac joints, often with multiple joint involvement.290
Almost one third of clients with endocarditis have low back pain, which may be the primary symptom reported. Back pain is accompanied by decreased range of motion and spinal tenderness. Pain may affect only one side, and it may be limited to the paraspinal muscles. Endocarditis-induced back pain may be very similar to that associated with a herniated lumbar disk, since it radiates to the leg and may be accentuated by raising the leg or by sneezing, coughing, or laughing; however, neurologic deficits are usually absent in persons with endocarditis.
Endocarditis may produce destructive changes in the sacroiliac joint characterized by pain localized over the sacroiliac, probably as a result of seeding of the joint by septic emboli. Widespread diffuse myalgia may occur during periods of fever, but this is not appreciably different from the general myalgia seen in clients with other febrile illnesses. More commonly, myalgia is restricted to the calf or thigh. Bilateral or unilateral leg myalgias occur in approximately 10% to 15% of all persons with endocarditis.
The cause of back pain and leg myalgia associated with endocarditis has not been determined. Concurrent aseptic meningitis is a possible hypothesis; a role for emboli that break off from the infected cardiac valves is supported by biopsy evidence of muscle necrosis or vasculitis in clients with endocarditis. Rarely, other musculoskeletal symptoms, such as osteomyelitis, tendinitis, hypertrophic osteoarthropathy, bone infarcts, and ischemic bone necrosis, may occur.
MEDICAL MANAGEMENT
DIAGNOSIS, TREATMENT, AND PROGNOSIS.
Infective endocarditis is often difficult to diagnose, since it can present with a wide array of signs and symptoms, as well as a confusing clinical picture. Blood cultures to identify specific pathogens in the presence of septicemia are required to determine appropriate antibiotic therapy, which is the primary medical intervention.
Other laboratory test results indicative of infectious endocarditis include elevated erythrocyte sedimentation rate, proteinuria, and hematuria. Echocardiography may be used to confirm the diagnosis and is useful in showing underlying valvular lesions and quantifying their severity. This test is not as useful in older adults, because it is common to find echogenic areas around and on degenerative valves that are impossible to distinguish from the infective vegetations seen in infective endocarditis. Large masses on valves are much more diagnostic.
Although it is easily prevented (for the at-risk person) by taking antibiotics before and after procedures such as dental cleaning, genitourinary instrumentation, and open cardiovascular surgery, endocarditis is difficult to treat and can result in serious heart damage or death. Potential complications are many, including CHF and arterial, systemic, or PEs. Therapy with antibiotics may be prolonged, and without complete treatment, relapse can occur up to 2 or more weeks after medical intervention. Surgical valve replacement may be necessary, depending on the response to treatment, sites of infection, recurrent infection, or infection of a prosthetic valve.
Rheumatic Fever and Heart Disease
Overview, Incidence, and Etiologic Factors
Rheumatic fever is one form of endocarditis (infection), caused by streptococcal group A bacteria, that can be fatal or may lead to rheumatic heart disease (10% of cases), a chronic condition caused by scarring and deformity of the heart valves (Figs. 12-22 and 12-23). It is called rheumatic fever because two of the most common symptoms are fever and joint pain.
Figure 12-22 Cardiac valvular disease caused by rheumatic fever. A, Inflammation of the membrane over the mitral (and aortic) valves may cause edema and accumulation of fibrin and platelets on the chordae tendineae. B, This accumulation of inflammatory materials produces rheumatic vegetations that affect the support provided by the chordae tendineae to the atrioventricular valves. C, In this view, the mitral valve leaflets have become thickened with scar tissue and calcified. The chordae tendineae often fuse. D, As a result, the scarred valve fails to close tightly (mitral stenosis) and regurgitation or backflow of blood into the atrium develops. Prolonged, severe stenosis with mitral regurgitation leads to symptoms of congestive heart failure. (Modified from Goodman CC, Snyder TE: Differential diagnosis in physical therapy, ed 3, Philadelphia, 2000, Saunders, p 110.)
Figure 12-23 A, Chest radiograph of a 15-year-old boy who had multiple occurrences of acute rheumatic fever, showing gross cardiac enlargement and failure. He had mitral regurgitation and stenosis, and aortic regurgitation and stenosis. B, Postmortem cardiac examination of the same boy, showing thickened, shortened mitral valve cusps with calcific vegetation and thickened chordae tendineae. Chordae are the tendinous cords connecting the two atrioventricular (AV) valves (the tricuspid valve between the right atrium and right ventricle and the mitral valve between the left atrium and left ventricle) to the appropriate papillary muscles in the heart ventricles; the chordae tendineae in effect anchor the valve leaflets. This support to the AV valves during ventricular systole helps prevent prolapse of the valve into the atrium. (From Cohen J, Powderly WG: Infectious diseases, ed 2, St Louis, 2004, Mosby. Courtesy Professor Bart Currie, Darwin, NT, Australia.)
The infection generally starts with strep throat in children between ages 5 and 15 years and damages the heart in approximately 50% of cases. The aggressive use of specific antibiotics in the United States had effectively reduced the incidence of rheumatic fever to around 0.5 cases per 100,000 school-age children and removed it as the primary cause of valvular damage.
However, between 1985 and 1987, a series of epidemics of rheumatic fever were reported in several widely diverse geographic regions of the continental United States, affecting children, young adults aged 18 to 30 years, and, occasionally, middle-aged persons. Currently, the prevalence and incidence of cases have not approximated the 1985 record, but they have remained above previous levels.
Pathogenesis
The exact pathogenesis is unclear, but rheumatic fever produces a diffuse, proliferative, and exudative inflammatory process in the connective tissue of certain struc- tures. The bacteria adhere to the oral and pharyngeal cells and then release their degradation products. Antigens to streptococcal cells bind to receptors on the heart, brain cells, muscles, and joints, which begins the autoimmune response; thus rheumatic fever is classified as an autoimmune disease. In the case of the heart valves, the inflammatory products cross-react with cardiac proteins, affecting cardiac valve tissue and myocardium.
All layers of the heart (epicardium, endocardium, myocardium, pericardium) (see Fig. 12-1) may be involved, including the valves. Endocardial inflammation causes swelling of the valve leaflets, with secondary erosion along the lines of leaflet contact. Small, beadlike clumps of vegetation containing platelets and fibrin are deposited on eroded valvular tissue and on the chordae tendineae; the mitral and aortic valves are most commonly affected.
Chordae are the tendinous cords connecting the two atrioventricular valves (the tricuspid valve between the right atrium and right ventricle and the mitral valve between the left atrium and left ventricle) to the appropriate papillary muscles in the heart ventricles; the chordae tendineae in effect anchor the valve leaflets. This support to the atrioventricular valves during ventricular systole helps prevent prolapse of the valve into the atrium.
Over time, scarring and shortening of the involved structures occur, and the leaflets adhere to each other as the valves lose their elasticity. As many as 25% of clients will have mitral valvular disease 25 to 30 years later, with fibrosis and calcification of valves, fusion of commissures (union or junction between adjacent cusps of the heart valves) and chordae tendineae, and mitral stenosis with fish-mouth deformity (Fig. 12-24).
Clinical Manifestations
Although strep throat is the most common manifestation of the streptococcal virus, streptococcal infections can also affect the skin and, less commonly, the lungs. In some cases of strep throat the initial triggering sore throat or pharyngitis does not cause extreme illness, if any discomfort at all.
However, the major manifestations of acute rheumatic fever are usually carditis, acute migratory polyarthritis, and chorea, which may occur singly or in combination. In the acute, full-blown sequelae, shortness of breath and increasing nocturnal cough also occur. A ring-or crescent-shaped rash with clear centers on the skin of the limbs or trunk (erythema marginatum) is present in fewer than 2% of persons in an acute episode. Subcutaneous nodules may occur over bony prominences and along the extensor surfaces of the arms, heels, knees, or back of the head, but these do not interfere with joint function.
Carditis is most likely to occur in children and adolescents. Mitral or aortic semilunar valve dysfunction (see Pathogenesis) may result in a previously undetected murmur. Chest pain caused by pericardial inflammation and characteristic heart sounds may occur. Polyarthritis may develop in a child or young adult with acute rheumatic fever 2 to 3 weeks after an initial cold or sore throat. Sudden or gradual onset of painful migratory joint symptoms in knees, shoulders, feet, ankles, elbows, fingers, or neck; fever (99° to 103° F [37.2° to 39.4° C]); palpitations; and fatigue are present. Malaise, weakness, weight loss, and anorexia may accompany the fever.
The migratory arthralgias usually involve two or more joints simultaneously or in succession and may last only 24 hours, or they may persist for several weeks. In adults, only a single joint may be affected. Joints that are sore and hot and contain fluid completely resolve, followed by acute synovitis, heat, synovial space tenderness, swelling, and effusion present in a different area the next day. The persistence of swelling, heat, and synovitis in a single joint or joints for more than 2 to 3 weeks is extremely unusual in acute rheumatic fever.
Rheumatic chorea (also called Sydenham’s chorea or St. Vitus’ dance) occurs in 3% of cases 1 to 3 months after the streptococcal infection and is always preceded by polyarthritis. Chorea in a child, teenager, or young adult is almost always a manifestation of acute rheumatic fever. Other causes of chorea are SLE, thyrotoxicosis, and cerebrovascular accident, but these are uncommon and unlikely in a child.
The chorea develops as rapid, purposeless, nonrepetitive movements that may involve all muscles except the eyes. This pattern of movement may last for 1 week, several months, or even several years without permanent impairment of the CNS.
MEDICAL MANAGEMENT
Late diagnosis can have serious consequences requiring immediate antibiotic and antiinflammatory treatment. Jones criteria are used as the basis for diagnosis (Table 12-17), and results of throat culture for group A streptococci are usually positive. Echocardiography combined with Doppler technology provides reliable hemodynamic and anatomic data in the assessment of rheumatic heart disease.
Table 12-17
Jones Criteria for Diagnosis of Rheumatic Fever

From Dajani AS, Ayoub A, Burman FZ, et al: American Heart Association medical/scientific statement: guidelines for the diagnosis of rheumatic fever: Jones criteria, 1992 update, Circulation 87:302-307, 1993. The Jones criteria have been reviewed and remain valid per the Jones Criteria Working Group. Source: Ferrieri P: Proceedings of the Jones Criteria Workshop, Circulation 106:2521-2523, 2002.
Aspirin may be used to treat the joint manifestations and as a general antiinflammatory agent. Corticosteroids are used when there is clear evidence of rheumatic carditis. Children with acute chorea are generally treated with some form of CNS depressant, such as phenobarbital. Commissurotomy and prosthetic valve replacement may be necessary for valvular dysfunction associated with chronic rheumatic disease.
PROGNOSIS.
Initial episodes of rheumatic fever last weeks to months, but 20% of children affected have recurrences within 5 years; relapses increase the risk of heart damage that leads to rheumatic heart disease, with mitral or aortic stenosis or insufficiency caused by progressive valve scarring. Mortality for acute rheumatic fever is low (1% to 2%), but persistent rheumatic activity with complications (enlarged heart, AF, arterial embolism, heart failure, pericarditis) is associated with long-term morbidity and mortality.
DISEASES AFFECTING THE PERICARDIUM
The pericardium consists of two layers: the inner visceral layer, which is attached to the epicardium; and an outer parietal layer (see Fig. 12-1). The pericardium stabilizes the heart in its anatomic position despite changes in body position and reduces excess friction between the heart and surrounding structures. It is composed of fibrous tissue that is loose enough to permit moderate changes in cardiac size but that cannot stretch fast enough to accommodate rapid dilation or accumulation of fluid without increasing intracardiac pressure.
The pericardium may be a primary site of disease and is often involved by processes that affect the heart; it may also be affected by diseases of the adjacent tissues. Pericardial diseases are common and have multiple causes. Three conditions primarily affect the pericardium: acute pericarditis, constrictive pericarditis, and pericardial effusion. These three diseases are grouped together for ease of understanding in the following section.
Pericarditis
Pericarditis or inflammation of the pericardium, the double-layer membrane surrounding the heart, may be a primary condition or may be secondary to a number of diseases and circumstances (Box 12-12). It may occur as a single acute event, or it may recur and become a chronic condition called constrictive pericarditis (uncommon).
Incidence and Etiologic Factors
The most common types of pericarditis encountered by the therapist will be drug induced or those present in association with autoimmune diseases (e.g., connective tissue disorders such as SLE, rheumatoid arthritis), after MI, in conjunction with renal failure, after open heart surgery, and after radiation therapy.
Other types encountered less often include viral pericarditis (e.g., Epstein-Barr, hepatitis, human immunodeficiency virus [HIV]) and neoplastic pericarditis (from spread to the pericardium of adjacent lung cancer or invasion by breast cancer, leukemia, Hodgkin’s disease, or lymphoma). Isolated cases of constrictive pericarditis as a manifestation of chronic graft-versus-host disease after peripheral stem cell transplantation have been reported.309