The Renal and Urologic Systems
The structures associated with the excretion of urine are (1) the kidneys and ureters, comprising the upper urinary tract, and (2) the bladder and urethra of the lower urinary tract (Fig. 18-1). The kidneys serve as both an endocrine organ and a target of endocrine action, with the aim of controlling mineral and water balance. The kidneys’ main function is to filter waste products and remove excess fluid from the blood. Every day the kidneys filter 200 qt of fluid; about 2 qt leave the body in the form of urine, and the remainder is retained in the body.
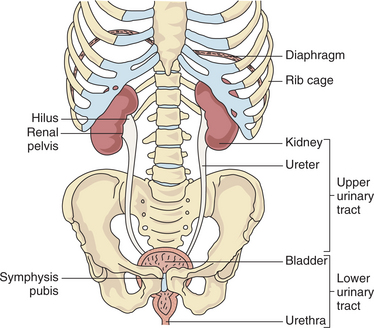
Figure 18-1 Structure and function of the renal and urologic systems. The kidneys are located in the posterior upper abdominal cavity in the retroperitoneal space (behind the peritoneum) at the vertebral level of T12 to L2. The upper portion of the kidney is in contact with the diaphragm and moves with respiration. The lower urinary tract consists of the bladder and urethra. The bladder, a membranous, muscular sac, is located directly behind the symphysis pubis and is used for storage and excretion of urine. From the renal pelvis, urine is moved by peristalsis to the ureters and into the bladder. The urethra serves as a channel through which urine is passed from the bladder to the outside of the body. (From Goodman CC, Snyder TE: Differential diagnosis in physical therapy, ed 3, Philadelphia, 2000, Saunders.)
These filtration and storage functions associated with excretion expose the kidney and bladder to carcinogens for extended periods, increasing the risk of cancer’s developing in these organs compared with the other urinary tract structures. In addition, the urethra of females lies close to the vaginal and rectal openings, allowing for relative ease of bacterial transport and increased risk of infection. The shorter urethra in females also contributes to the increased incidence of urinary tract infections (UTIs) in females.
Therapists have an important role on the medical team for primary intervention for a number of renal/urinary tract disorders such as urinary incontinence (UI) and for those on dialysis or having a renal transplant. UI afflicts a significant percentage of the geriatric population, and UTIs rank second only to upper respiratory tract infections in incidence of bacterial infections. Therapists encounter these two disorders often as common comorbidities in the clinical arena.
The presence of a UTI increases the risk of infections developing elsewhere. This could occur while the therapist is treating someone for a knee injury or after cerebral vascular accident. Recognizing clinical signs and symptoms of renal/urologic problems (Box 18-1) will facilitate medical referral. Understanding how these diseases and the prescribed medical treatment can influence rehabilitative efforts is essential to help ensure a positive functional outcome.
AGING AND THE RENAL AND UROLOGIC SYSTEMS
Aging is accompanied by a gradual reduction of blood flow to the kidneys, coupled with a reduction in nephrons (the units that extract wastes from the blood and concentrate them in the urine; see Fig. 18-6). As a result the kidneys become less efficient at removing waste from the blood, and the volume of urine increases somewhat with age. A tendency toward greater renal vasoconstriction in the older adult is evident, as compared with young individuals. This occurs as a result of mental stress, in physiologic circumstances such as physical exercise, or in disease manifestations such as the effective circulatory volume depletion that develops in heart failure.191
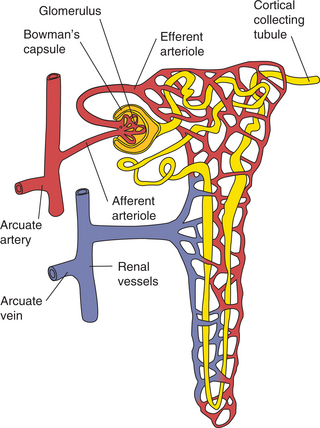
Figure 18-6 Components of the nephron. The afferent arteriole carries blood to the glomerulus for filtration through Bowman’s capsule and the renal tubular system.
Renal system changes that occur with aging cause alterations in the functional balance of fluid and electrolytes so that sodium regulation is not as effective. Older people are at greater risk for developing hyponatremia (reduced sodium in the blood), affecting the musculoskeletal system. Changes typical of the aging kidney are also accelerated when hypertension overlaps the physiologic renal process, because both aging and hypertension affect the same structure (i.e., the glomeruli).
A reduction in bladder capacity increases the number of times an individual urinates in a day, and the urinary timetable also changes. Although the kidneys produce most of the urine during the day in young people, a shift to night production over time makes one or two nocturnal trips to the bathroom commonplace after age 60.
Although specific age-related anatomic changes have not been associated with urinary tract disease, certain age groups (e.g., older adults) are at significant risk of developing a variety of disorders. Hormonal changes in women, combined with the aging properties of the connective tissues, fascia, or collagen fibers, contribute to pelvic floor disorders. Transient ischemic attacks (TIAs) and strokes may result in mild to severe deficits or fluctuations in muscle tone affecting the pelvic floor muscles.
The effect of multiple medications, conditions such as benign prostatic hyperplasia and pelvic floor disorders, and the incidence of pelvic surgeries and catheterization in the older population all increase the risk of developing urinary tract disease. A large number of adults over age 60 are incontinent, and many older adults require dependent living situations because of this disorder. Considering the percentage of the rehabilitation population made up by the older adult, therapists will continue to be involved in the treatment of renal/urologic disorders.
INFECTIONS
UTIs are very common, affecting men, women, and children. Any portion of the urinary tract can become infected, although the bladder (cystitis) and urethra (urethritis) are usually involved. Bacteria may also spread to the upper portion of the urinary tract and involve the kidneys, causing a more serious infection referred to as pyelonephritis.
UTIs can be defined as either uncomplicated or complicated, and relapsed or recurrent. Complicated infections develop in persons with factors that can make diagnosis and treatment difficult—including diabetes, history of stroke, pregnancy, immunosuppression, structural abnormalities, or functional abnormalities of the urinary tract. The presence of such complications often requires longer treatment and further testing.183
Generally, UTIs in men, pregnant women, children, and clients who are hospitalized or in a long-term care setting can be considered complicated. Uncomplicated UTIs lack these factors and are more easily diagnosed and treated. UTIs may relapse or, more commonly, reoccur. Relapsed infections are infections that persist with the original organism without completely clearing. Reoccurrence of UTIs is considered a different infection that occurs after successful treatment of the initial infection, although it may be with the same organism due to repeated contamination.183
Incidence and Prevalence
UTIs frequently occur in the general population, although women and older adults comprise the majority of cases. UTIs affect over 11.3 million women per year,59 or up to 5% of all females. Five percent to thirty percent of the older adult population is also affected.60 By age 24, one third of women will have had at least one physician-diagnosed UTI that is treated with prescription medication.
For those living in skilled nursing facilities, assisted living arrangements, or extended care facilities, the prevalence of infection is even higher: 25% for women and 20% for men.7 UTIs affect children, involving approximately 7% of girls and 2% of boys before the age of 6 years.125 Recurrent UTIs can be problematic for many people, occurring in 3% to 5% of women87 and 80% of children who previously experienced an uncomplicated infection. The cost is substantial at over $1.6 billion per year (diagnosis, treatment, and management cost). UTIs also result in restrictions in daily activities and lost days of work.
Etiologic and Risk Factors
Most UTIs occur in adult women. The urethra in females is shorter, compared to that in males, and also close to the entrances to the vagina and rectum. The bacteria that result in most UTIs are acquired from the large bowel (fecal flora). The urethral meatus is close to the fecal reservoir and rectum.
Young, sexually active women are at higher risk of developing UTIs because it is thought that sexual intercourse can influence the movement of bacteria in the direction of the bladder. This again is due to the proximity of the urethral meatus and vagina. There are numerous risk factors for UTIs (Box 18-2) that depend upon the characteristics of the person affected. For example, risk factors for an acute, uncomplicated UTI in a premenopausal woman may be different than those for a postmenopausal woman in a long-term care setting.
For young women, the most common risk factors include a history of a previous UTI, frequent or recent sexual activity, or the use of a spermicidal agent.53,61 UTIs are also more common during pregnancy. The increased risk is due to dilation of the upper urinary system, reduction of the peristaltic activity of the ureters, and displacement of the urinary bladder, which moves to a more abdominal position, thus further affecting the ureteral position.
In older women who are in a long-term care setting, the most frequently noted risk factors are advancing age and debilitation associated with conditions that impair voiding or cause poor perineal hygiene, such as dementia or stroke.52 Healthy, community-dwelling postmenopausal women share risk factors seen in both young and older women. Sexual activity and a previous history of UTI are common risk factors for both young and postmenopausal women, while incontinence is an additional risk factor in these older women.89 The effects of estrogen decline (dry mucosa and vaginitis) may contribute to increased risk of infection because of the change in vaginal flora, but study findings remain unclear.
People with diabetes receiving treatment are also more prone to UTIs due to immunologic impairments; the presence of glycosuria, which provides a fertile medium for bacterial growth; and voiding difficulties resulting from diabetic neuropathy (detrusor paresis).15,73
Another significant and common risk factor for UTI is catheterization. Placement of a urinary catheter is a leading cause of infection in the hospital setting, accounting for 40% of nosocomial infections. The reasons are clearly related to the introduction of a foreign body that provides a direct pathway for bacteria to travel from the perineum to the bladder. Less commonly, a client may display a structural or functional abnormality that leads to a UTI. Contributing structural problems may be kidney stones, cystocele, or prostatic hyperplasia. Examples of functional problems include reflux of urine from the bladder to the kidney and neurogenic bladder from diabetes, spinal cord injury, or multiple sclerosis.
Pathogenesis
The bacteria most often responsible for UTI are fecal-associated gram-negative organisms, with Escherichia coli accounting for about 80% of urinary tract pathogens. Staphylococcus saprophyticus causes 5% to 15% of UTIs, while Enterococci, Klebsiella, and Proteus make up the remaining common organisms.
Hospitalized clients are more likely to become infected with Enterobacter, Klebsiella, Proteus, Pseudomonas, enterococci, and staphylococci bacteria than outpatients with UTI. Candida species can be seen in persons who have undergone invasive instrumental investigations or catheterizations and in children with urogenital abnormalities.
These common urinary tract pathogens are able to adhere to the urinary tract mucosa, colonize, and cause infection. Several subtypes of bacteria contain genes that allow for greater virulence and ability to colonize urothelium than other organisms, making them uropathogenic. The most common route of entry of bacteria into the urinary tract is ascending up the urethra into the bladder. Although infrequent in occurrence, infections may be bloodborne (bacteria in the bloodstream) or acquired via the lymphatic system.
Clinical Manifestations
Classic features of UTIs are evident in older children and adults and include frequency, urgency, dysuria, nocturia, and, in children, enuresis. Fever, chills, and malaise may also be present. The individual may notice cloudy, bloody, or foul-smelling urine and a burning or painful sensation during urination or intercourse. Pain may be noted in the suprapubic, lower abdominal, groin, or flank areas, depending on the location of the infection.
In the case of kidney involvement, the diaphragm may become irritated, resulting in ipsilateral shoulder or lumbar back pain. The clinical manifestations in frail, older adults can be varied, often with malaise, anorexia, and mental status changes (especially confusion or increased confusion) as the most prominent features. Flank pain, fever, and chills often indicate an upper UTI or pyelonephritis.
MEDICAL MANAGEMENT
UTIs can be prevented in some cases by drinking at least eight 8-oz glasses of water each day; urinating soon after sexual intercourse; for females, wiping from the front to back after urination so that bacteria from the anal area are not pushed into the urethra; changing sanitary pads often during menstruation; and washing the genital area with warm water before sexual activity to minimize the chance that bacteria can be introduced. The use of spermicidal agents with a diaphragm has been associated with an increased risk for UTIs. The use of another form of birth control may be warranted if repeated UTIs become problematic.
Certain foods may also be preventative. Berry juices and products containing fermented milk may be helpful in reducing the occurrence of UTIs, although further studies are needed to verify this relationship.109 The use of cranberry juice in the prevention and treatment of UTIs has been controversial, with different studies showing positive and negative effects of cranberry juice as a preventive or therapeutic agent.6,31 More studies are needed to determine efficacy, dose, and appropriate candidates for this dietary treatment. The use of probiotics to increase normal vaginal flora may be of benefit. Preliminary studies are encouraging, although only certain types of Lactobacillus have had promising results.49
There has also been debate as to whether hormone therapy can prevent UTIs in postmenopausal women. Recent studies indicate that oral as well as vaginal hormone therapy is not preventative and may be detrimental to heart health.17,95,158
Instrumentation and particularly placement of urinary catheters frequently lead to UTI. However, if a catheter is needed, a condom catheter may have a reduced risk of causing UTI compared to indwelling catheters.163 Preliminary studies also suggest that use of catheters coated with an antimicrobial agent may reduce the risk, but further investigations are needed.98
DIAGNOSIS AND TREATMENT.
The diagnosis of a UTI is typically made based on history and urinalysis results. A bacterial count of greater than 100,000 organisms per milliliter of urine is a commonly accepted criterion for diagnosis. Besides the bacterial count, the urine leukocyte count (more than 10 leukocytes per cubic millimeter of urine collected midstream), and presence of leukocyte esterase, nitrates, and protein are also helpful.
Many people demonstrate pyuria (leukocytes in the urine) without infection, and corroborating clinical and laboratory information must be evident to diagnose an infection. In clients who are healthy and without complicating features, empiric treatment with antibiotics is effective and urine cultures are not required.
Acute UTIs in healthy, nonpregnant clients are typically treated with antibiotics, particularly trimethoprim-sulfamethoxazole (TMP/SMX) or a fluoroquinolone, as recommended by the Infectious Diseases Society of America guidelines.198 Yet over the past decade there has been a significant rise in resistance to TMP/SMX, with rates varying regionally. Initial treatment must take into account local resistance patterns as well as the health of the client. For individuals with complicating features, treatment failure could lead to severe morbidity and mortality, and in areas where TMP/SMX resistance is common, a fluoroquinolone may be more appropriate initial treatment until culture results are available.
Women who experience recurrent infections have several options for treatment depending upon the clinical situation and compliance of the client. They may take antibiotics prophylactically (typically as a daily dose), they may self-treat as they recognize typical symptoms,79 or in the case of sexual intercourse as a precipitating factor, women may be advised to take antibiotics just after sex. Increased fluid intake may also help relieve symptoms and signs and is often used as an adjunct to pharmacologic treatment. Lactobacillus acidophilus, a probiotic supplement of live, active organisms, may be recommended for anyone taking antibiotics to replace the naturally occurring bacteria in the intestines and to prevent candidiasis (yeast growth). A vaccine to prevent recurrent urinary infections of the bladder has proved successful in mice and is currently being tested in clinical trials.
For the minority of clients who develop UTIs as a result of structural or functional problems, further testing is needed to correct the abnormality. Ultrasound, radiographs, computed tomographic (CT) scans, and renal scans may be used to identify contributing factors such as obstruction. Postvoid residual and more complex tests such as voiding cystourethrography (VCUG) (upper urinary system) and urodynamic testing with and without fluoroscopy determination may be recommended for anyone at risk for urinary retention. Whenever possible, ultrasound assessment, rather than urinary catheterization, is recommended for measuring the postvoid residual.
Pyelonephritis
Pyelonephritis can be either an infectious process involving the kidneys (acute pyelonephritis) or a chronic inflammatory disease involving the kidney parenchyma and renal pelvis (chronic pyelonephritis). Acute pyelonephritis occurs in over 250,000 people per year, causing over 100,000 hospitalizations. The direct and indirect costs are estimated at $2.14 billion.20 It typically results from bacteria ascending from the bladder to infect the kidneys.88 Similar to UTIs, acute pyelonephritis occurs more frequently in women than men, although men have a higher complication rate.62
Chronic pyelonephritis is a tubulointerstitial disorder characterized by specific changes in the kidney (cortical scarring and deformation of the calices). These alterations can be a result of several diseases that can lead to renal insufficiency. Chronic pyelonephritis may be responsible for up to 25% of the population with end-stage renal disease (ESRD).
Etiologic and Risk Factors
The majority of acute pyelonephritis cases are associated with ascending UTIs (see Box 18-2) and are caused most commonly by E. coli (up to 85%).173 A smaller proportion are caused by other gram-negative organisms such as Proteus, Klebsiella, Enterobacter, and Pseudomonas species.
Risk factors associated with increased risk for pyelonephritis in healthy, nonpregnant women include frequent sexual activity, recent UTI, recent spermicide use, diabetes, and recent incontinence.173 In other cases, pyelonephritis can stem from bloodborne pathogens associated with infection elsewhere. People with bacterial endocarditis and miliary tuberculosis are susceptible to kidney involvement. In addition, immunocompromised people are at risk for bacterial and fungal seeding of the kidney with subsequent abscess formation.
Chronic pyelonephritis is the term that describes specific, abnormal renal findings. Several diseases or processes can lead to chronic pyelonephritis, such as vesicoureteral reflux (urine is forced from the urinary bladder into the ureters and kidneys), urinary obstruction, analgesic nephropathy, or bacterial infection superimposed on a structural/functional abnormality. The most common cause of chronic pyelonephritis is vesicoureteral reflux, although the renal insufficiency associated with this is most often referred to as reflux nephropathy.
Pathogenesis
Although urine is typically sterile, the distal end of the urethra is commonly colonized by bacterial flora. As described under Etiologic and Risk Factors, bacteria can be transported to the urinary bladder in many ways. After urination, the subsequent passage of sterile urine from the kidneys to the bladder dilutes any bacteria that may have entered the bladder. If the residual urine volume is increased, as with an atonic bladder, an accumulation of insufficiently diluted bacteria can occur. Bacteria in the bladder urine typically do not gain access to the ureters for a variety of anatomic reasons. However, people with an abnormally short passage of the ureter within the bladder muscle wall and an angle of ureter insertion into the bladder wall that is more perpendicular are at risk for reflux of urine into the ureter itself. This reflux can be of sufficient force to carry the urine and the accompanying bacteria into the renal pelvis and calices.
Chronic pyelonephritis is defined by scarring with deformity of the calices. Processes that continually cause inflammation in the kidney can lead to chronic changes. Only a few processes can cause these changes, and they can be divided into three main groups: reflux, obstruction, and idiopathic.
Clinical Manifestations
The onset of symptoms and signs associated with acute pyelonephritis is usually abrupt. The complaints may include fever, chills, malaise, headache, and flank pain. The person may also complain of tenderness over the costovertebral angle (Murphy’s sign). Symptoms of bladder irritation may be present (including dysuria, urinary frequency, and urgency) but are not required for the diagnosis.
Symptoms associated with chronic pyelonephritis vary depending upon the causative process; however, symptoms may not be present. The diagnosis is made more often by laboratory detection of kidney function changes.
MEDICAL MANAGEMENT
The presence of suggestive symptoms for acute pyelonephritis warrants laboratory testing and treatment. Urinalysis typically reveals pyuria, bacteriuria, and varying degrees of hematuria. A urine culture should always be obtained and often will result in the growth of the offending bacteria or fungus. In addition, the blood count usually demonstrates leukocytosis.
If the infection is severe enough or if complicating factors are present, hospital admission may be required for intravenous antibiotics and hydration. Typically, however, the condition is treated with an appropriate antibiotic medication. Symptoms typically begin disappearing within several days. If the person does not show improvement within 48 to 72 hours, contact with the physician is warranted.
If the process associated with chronic pyelonephritis continues to progress, creating worsening scarring, the result may be ESRD requiring dialysis or transplantation.
RENAL DISORDERS
Renal Cell Carcinoma
Overview and Incidence.: Adult kidney neoplasms account for approximately 3% to 4% of all cancers.96 During the past two decades, the incidence of these cancers has increased by approximately 2% each year.161
Renal cell carcinoma (RCC) is the most common adult renal neoplasm, accounting for more than 90% of renal tumors, and its incidence is rising (although the death rate is not). RCC occurs more frequently in males than females (about a 1.6: 1 ratio), with a peak incidence between 60 and 70 years.
RCC is a heterogeneous group of cancers that are separated into four main types according to the cell type of origin: clear cell, papillary, chromophobe, and collecting duct RCC. Clear cell constitutes the majority of cases (80%), papillary is the next most common at 10% to 15% of RCCs, while chromophobe RCC and collecting duct RCC account for only 4% and 1%, respectively.
Etiologic and Risk Factors.: RCC is linked to several hereditary diseases, including von Hippel-Lindau disease (a rare autosomal dominant familial cancer syndrome related to clear cell RCC), hereditary papillary renal carcinoma (an autosomal dominant disorder related to papillary RCC), and the Birt-Hogg-Dubé syndrome (a rare autosomal dominant disorder related to chromophobe RCCs or mixed chromophobe RCCs–oncocytomas). These hereditary disorders are rare and only account for a small percentage of RCCs. Risk factors that can lead to the development of sporadic RCC include tobacco smoking, obesity, hypertension (diuretics), occupational exposure to substances such as organic solvents and asbestos, and acquired cystic kidney disease associated with ESRD.28,170
Pathogenesis.: As for other cancers, genetic mechanisms are now being discovered for RCC, which better explain the causes and aid in the treatment. Because RCC is seen in a few hereditary diseases, scientists have been able to locate specific genetic abnormalities. Von Hippel-Lindau disease has several characteristic abnormalities, including the development of RCC (clear cell type). The von Hippel-Lindau tumor suppressor gene (VHL) is located on chromosome 3.
Of interest, this same abnormality has been detected in 60% to 80% of people with sporadic clear cell RCC. The product of the VHL gene normally suppresses genes that, in the presence of hypoxia, cause endothelial growth, cell growth, and glucose uptake, and affect acid-base balance.32 When this gene is altered, cell proliferation occurs unchecked, despite the absence of hypoxia. Factors other than VHL mutations are involved in this process and most likely account for the remaining percentage of sporadic clear cell RCC occurrences.
Another gene linked to RCC is the MET protooncogene. This abnormal gene, located on chromosome 7, is duplicated in approximately 75% of sporadic papillary RCC cases. If the FH gene (which encodes for the Krebs cycle enzyme fumarate hydratase) is inactivated, the result is another hereditary disorder known as hereditary leiomyomatosis and renal cell cancer syndrome. Chromophobe RCC may develop in clients with mutations to the BHD gene, whose product is suspected to suppress tumors.32
Clinical Manifestations.: The classic triad of symptoms related to RCC is flank pain, hematuria, and a palpable abdominal mass. Yet about half of all cases are discovered incidentally on a radiographic examination, such as a CT scan.32 Kidney cancers are generally silent, particularly in the early stages, although nonspecific symptoms may develop such as malaise, anemia, or unexplained weight loss. Hematuria is the single most common presenting finding, occurring in up to 50% of cases, yet it is frequently intermittent and microscopic.
Symptoms associated with metastasis can be the initial manifestation; about 25% to 30% of clients have metastatic disease at the time of diagnosis.116 Metastases most often occur in the lungs (75%), regional lymph nodes (65%), bones (40%), and liver (40%).210 The client may develop a cough or bone pain secondary to metastasis to the lungs or bone, respectively.
Potentially confusing the clinical presentation of this condition is the fact that RCCs are associated with ectopic hormone production and paraneoplastic symptoms, including fever, hypertension, hepatic dysfunction, and hypercalcemia. Hormones produced by the tumors include parathyroid-like hormone, gonadotropins, renin, erythropoietin, glucagon, and insulin.
As discussed earlier, several hereditary syndromes predispose to the development of RCC. Each of these disorders has its own unique clinical manifestations other than RCC. For example, von Hippel-Lindau disease is associated with retinal angiomas, hemangioblastomas of the central nervous system, pheochromocytomas, and clear cell RCCs. These distinguishing features lead to the diagnosis and allow regular monitoring and surveillance for RCC.
MEDICAL MANAGEMENT
The primary feature of RCC is the renal parenchymal mass, which can be detected by a variety of imaging modalities (Fig. 18-2). The widespread availability of abdominal ultrasound (Fig. 18-3), magnetic resonance imaging (MRI), and CT scanning has increased the diagnosis of incidental renal tumors.
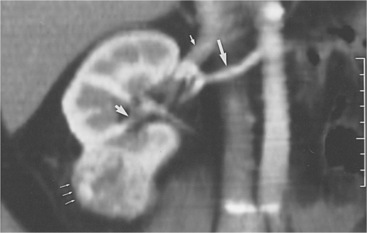
Figure 18-2 Renal cell carcinoma on computed tomographic scan. A reconstructed image in the coronal plane of section shows vascular anatomy by demonstration of the renal artery (large arrow), renal vein (small arrow), excretion into the pelvicaliceal system, which is not obstructed (arrowhead); and renal cell carcinoma arising from the lower pole of the right kidney (thin arrows). The cancer is confined to the cortex; therefore, partial nephrectomy can be performed. (From Brenner BM: Brenner and Rector’s The kidney, ed 7, Philadelphia, 2004, Saunders.)
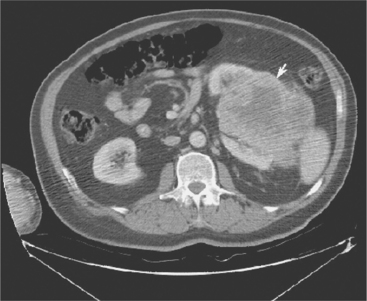
Figure 18-3 Computed tomographic scan of abdomen demonstrating a large left renal mass (arrow) consistent with renal cell carcinoma. (From Townsend CM: Sabiston textbook of surgery, ed 17, Philadelphia, 2004, Saunders.)
Determining whether the mass is benign or malignant can be difficult, often requiring surgical removal before a definitive diagnosis can be made. If hematuria is present, intravenous pyelography (IVP) may be the initial procedure to identify renal abnormalities. IVP is a radiographic test that allows for evaluation of the kidneys, ureters, and bladder. A dye injected into the bloodstream is filtered and secreted by the renal tubules. The IVP provides infor- mation including renal size, function, position, the presence of calculi, masses, and congenital variants.
Ultrasonography can be used to further evaluate the renal parenchyma and detect small tumors (less than 1 cm). The advantage of the CT scan is that the greatest renal anatomic detail is obtained and details of neighboring organs such as the liver, colon, spleen, and lymphatics will also be available.
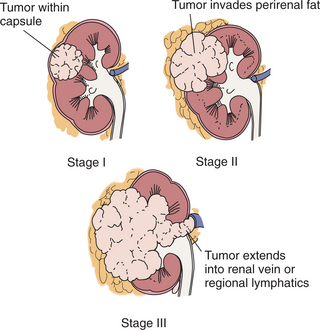
STAGING.
The American Joint Committee on Cancer (AJCC) and the University of California, Los Angeles (UCLA) Integrated Staging Systems are currently used to stage this disease. The AJCC utilizes the TNM (tumor, node, metastasis) system defining stages I through IV (Fig. 18-4). The UCLA Integrated Staging System employs the Eastern Cooperative Oncology Group (ECOG) performance status scale and the Fuhrman nuclear grade not only to determine stage but also prognosis. Clients are classified into low-, intermediate-, and high-risk groups. This staging system also provides risk categories for those with metastatic disease.
TREATMENT.
Surgery is the principal treatment for RCC. Traditionally, a radical nephrectomy (removal of the kidney; Gerota’s fascia, a fibroareolar tissue surrounding the kidney and perirenal fat; the adrenal gland; and regional lymph nodes) has been preferred, although less aggressive methods are being investigated. Nephrectomy may also be beneficial for clients with metastatic disease, since combined surgical and medical therapy has slightly improved survival. Metastatic lesions may be removed at the time of surgery; however, this has not been shown to improve survival.
A partial nephrectomy may be appropriate for clients with a small mass (less than 4 cm), a solitary kidney, masses in both kidneys, renal insufficiency, or the presence of a hereditary disorder related to RCC. This approach, however, carries a 3% to 6% risk of tumor recurrence.138 Laparoscopic nephrectomy has gained acceptance as a method for reducing hospital stay and postoperative pain, and providing a faster recovery. Partial nephrectomy is challenging and often requires an open procedure.
Newer, less invasive methods for treating RCC include percutaneous thermal ablation using radiofrequency heat ablation or cryoablation. These types of procedures are best for small tumors (less than 3 cm) and in clients with comorbidities that can increase surgical risk. Further investigations are required, although the complication rates appear to be low.32
Medical treatment, adjuvant to surgery, is offered for more locally advanced or metastatic disease. Response rates to standard medical modalities have not been good. Only 4% to 6% of clients respond to chemotherapy207; clear cell and papillary RCC may express a protein that transports the drug out of the cells, leading to chemotherapy resistance. Collecting duct RCC may demonstrate a higher response rate to chemotherapy.
Other chemotherapy agents are currently being tested to determine if they provide a better response rate. Immunomodulatory agents have been used with some success. Interferon-alfa has provided a 14% response rate alone in clients with metastatic clear cell RCC, which is apparent for an average of 6 months. When this drug is used in conjunction with nephrectomy, the survival rate has increased by 3 to 10 months.56
High-dose interleukin-2 is another agent that has shown some benefit in clients with metastatic disease; it is currently the only Food and Drug Administration (FDA)–approved agent for advanced RCC. High-dose interleukin-2 has demonstrated a 21% response rate, but the side effects (including capillary leak syndrome) are often too severe to continue treatment. For those persons able to tolerate the high dose, response rates are reported to be 54 months and perhaps longer.67
Research has shown that allogeneic stem cell transplantation following a nonmyeloablative regimen can successfully create a graft-versus-tumor effect, which in initial studies demonstrated a 44% response (although continued studies have been less successful).27 Two difficulties encountered with transplantation include the life-threatening problem of graft-versushost disease and the requirement of a matched sibling donor.
Other areas of ongoing research include tumor vaccines and tumor-specific targeting agents (i.e., targeting specific pathways with an antibody or blocking blood vessel formation). One angiogenic agent, sunitinib, a tyrosine kinase inhibitor, is able to block the receptors of vascular endothelial growth factor and platelet-derived growth factor, decreasing the blood supply to the tumor.135 Preliminary studies are encouraging, and other targeted agents are in trials.
PROGNOSIS.
The prognosis of RCC depends upon the type and staging. Sporadic papillary RCC has a 5-year survival rate of almost 90% and is known to have a lower incidence of metastasis compared to clear cell RCC. However, individuals with metastatic papillary RCC have a lower survival rate than those with metastatic clear cell RCC.
Most chromophobe RCCs will have a favorable postsurgical course providing the tumor size is small and the grade low. Collecting duct RCC is an aggressive tumor with a poorer prognosis. The survival rates according to the AJCC system of tumor size demonstrate a 5-and 10-year survival of 95% and 91%, respectively, for T1-stage tumors; of 80% and 70% for T2 tumors; and of 66% and 53% for T3a tumors.
Utilizing the UCLA Integrated Staging System, the 5-year survival rate for the low-risk group is 91%, while the intermediate-and high-risk groups have an 80% and 55% survival rate, respectively. Unfortunately, 40% of locally removed renal cell tumors return.146
Factors that portend a poor prognosis for metastatic RCC include a low performance score (Karnofsky performance status score), a high lactate dehydrogenase level, a low hemoglobin level, and a high serum calcium level. Metastatic disease has a much worse prognosis than localized tumor, with a 5-year survival of 0% to 7% for clients with multiple metastatic lesions.72
Wilms’ Tumor
Overview, Incidence, and Risk Factors.: Wilms’ tumor, or nephroblastoma, is the most common malignant kidney neoplasm in children. Approximately 500 new cases are reported annually in the United States. Age is the primary risk factor. The disease most commonly occurs during the first 6 years of life, with approximately 75% of cases occurring in children under the age of 5. The peak incidence is between the ages of 3 and 4, and the tumor occurs slightly more often in African Americans and girls. Wilms’ tumors occur bilaterally in 5% of cases.
There are several hereditary syndromes that can predispose children to the development of Wilms’ tumor. The three most common include the WAGR syndrome (Wilms, aniridia, genitourinary malformation, mental retardation), the Beckwith-Wiedemann syndrome, and the Denys-Drash syndrome.
Etiologic Factors and Pathogenesis.: Although the majority of cases are sporadic, about 1% to 3% have a family history of Wilms’ tumor, and up to 10% are seen in hereditary syndromes. Molecular genetics plays an important role in the cause of Wilms’ tumor. The biologic signaling pathways determining the origin of Wilms’ tumor are complex, and several genes at several loci may be involved.
The most well-known and studied gene of Wilms’ tumor is the WT1 suppressor gene, a complex protein that is an essential regulator of kidney development; mutations in this gene result in the formation of tumors in about 10% of Wilms’ tumor cases.121 These genes have also been implicated in the formation of many other cancers.
Other genetic alterations such as WT2 and familial genetic alterations termed FWT1 and FWT2 have also been located. Significant investigations are ongoing to determine how these genes and their products interact in order to understand the mechanisms in the development of Wilms’ tumor and provide improved therapy.
Clinical Manifestations.: Wilms’ tumors can be difficult to discover early because the tumor can grow to a large size before causing symptoms. Fortunately, despite large tumor size, most Wilms’ tumors do not metastasize. An abdominal mass, most often detected by the parents, is the most common presenting sign. Up to 30% of children may complain of abdominal pain, malaise, loss of appetite, or nausea/vomiting. Hematuria may occur in up to 30% of cases and hypertension in up to 25% of affected children. Congenital abnormalities may be present, particularly those associated with hereditary syndromes with a predilection for Wilms’ tumor (13% to 28%).
MEDICAL MANAGEMENT
Staging is performed according to the National Wilms Tumor Study Group (NWTSG) staging system. Tumors are staged into five groups (stages I to V), depending on tumor size and growth into surrounding structures. Histologic features are also important. Histologically, tumors can be classified as having favorable histology or unfavorable histology (anaplastic features). About 40% of tumors are discovered while in stage I, while only 5% of cases are at stage V.
Abdominal ultrasonography helps define the cystic or solid nature of the mass and helps determine whether the renal vein or vena cava is involved. CT scan of the abdomen is helpful in determining the extent of the tumor but can be difficult to perform with small children. A chest radiograph and CT of the chest are used to determine the presence of metastases to the lung. MRI may also be beneficial in determining the extent of the disease.
TREATMENT.
Surgical resection of the tumor is the primary treatment regardless of the stage of the disease. A radical nephrectomy is the most common procedure, although a nephron-sparing procedure may be performed in clients who have lesions in both kidneys. Regional lymphadenectomy may be carried out, as lymph node involvement strongly affects the prognosis. Chemotherapy is also used for all stages of the disease, sometimes preoperatively, with radiation therapy being added to the treatment regimen for stage III and IV disease and tumors with unfavorable histologic findings. (See Chapter 9 for the side effects associated with chemotherapy and radiation therapy.)
PROGNOSIS.
Prognosis depends on the histologic appearance of the lesion, stage of the disease, and age of the child. But the overall 5-year survival is very good at 92%. Individuals with stage I tumors with favorable histology have a 92% 4-year event-free survival rate and an overall survival rate of 98%. Those with stage I tumors with unfavorable histology have only a 70% 4-year event-free survival rate and an overall survival rate of 83%.42 Even those with stage V tumors with favorable histology have a 4-year event-free survival rate of 81%. Those with stage V tumors with unfavorable histology exhibit only a 17% 4-year event-free survival rate.
With the development of successful treatment, emphasis is now being placed on limiting significant long-term side effects while maintaining the high cure rate in tumors with favorable histology.99 Further treatment options are needed for advanced tumors with unfavorable histology. More than 80% of people with Wilms’ tumor can be cured using multimodal therapy.34 Wilms’ tumor may recur years after the initial diagnosis.
Renal Cystic Disease
A renal cyst is a cavity filled with fluid or renal tubular elements making up a semisolid material. The presence of these cysts can lead to degeneration of renal tissue and obstruction of tubular flow. Renal cysts vary considerably in size, ranging from microscopic to several centimeters in diameter, and can be single or multiple, unilateral or bilateral.
Cysts in the kidney are rather common and can be classified into six categories of cystic diseases: (1) polycystic kidney disease (PKD), (2) cystic diseases of the renal medulla, (3) acquired cystic disease, (4) single cysts, (5) cystic renal dysplasia, and (6) miscellaneous renal cystic disorders.
The formation of simple cysts is the most common cystic disorder of the kidney. Simple cysts are usually less than 1 cm in diameter and do not often produce symptoms or compromise renal function. Acquired cysts may develop secondary to dialysis, diabetes mellitus, or glomerulonephritis. PKD is a leading cause of ESRD, frequently requiring dialysis and renal transplantation. Because of the seriousness and fairly common occurrence of PKD, this section will principally discuss PKD. The remaining disorders constitute less common causes of renal cysts.
Incidence
PKD is manifested as either an autosomal dominant (ADPKD) or autosomal recessive (ARPKD) disorder. Although PKD can occur spontaneously, most cases are hereditary. ADPKD is one of the most common hereditary disorders in the United States, affecting more than 600,000 Americans (about 1 in every 500 to 1 in every 1000 persons). ARPKD is rare.
Persons with ADPKD may not manifest symptoms until the third or fourth decade of life, while ARPKD is evident at birth and can cause death early in life. ADPKD affects people from all races and ethnic groups. Most people with ADPKD will exhibit evidence of the disease by the age of 80, but only half progress to ESRD. ADPKD is the fourth leading cause of ESRD and accounts for 10% of all cases of ESRD.
Risk Factors
While there is currently no way of determining which people with ADPKD will develop ESRD, there are a few risk factors that have been linked to a more rapid progression. These factors include hypertension, multiple pregnancies, male gender, and the expression of the genetic mutation PKD1.
Etiology and Pathogenesis
Most renal cysts form from the epithelium of a preexisting renal tubule. These epithelial cells typically exhibit a reabsorptive function, but have secretory capabilities. In the case of cyst formation, epithelial cells with genetic mutations begin to secrete fluid into the tubule once stimulated by endocrine, paracrine, and autocrine regulating proteins. Such proteins may also play a role in the size and rate of growth of the cyst.
As a cyst grows, it detaches from the nephron (about 75% detach completely). The epithelial cells then continue to proliferate and fibrosis develops. With time, the pressure created by the expanding, multiple cysts interrupts the function of neighboring nephrons, leading to apoptosis of noncystic nephrons. Although normal nephrons enlarge in an attempt to compensate for the loss of nephrons, this is unsuccessful in half of people with PKD.
In ADPKD, there have been several genes linked to the development of cysts. These abnormalities are located on chromosomes 16 and 4 and are called PKD1 and PKD2. Other genes are likely involved as well. These genes code for proteins that function in transferring signals from the extracellular matrix into the cell to promote cellular proliferation and differentiation. Both genes need to be affected before there is the resultant development of disease.
About 85% of persons with ADPKD express a mutation in PKD1, while only 15% demonstrate a mutation in PKD2. A small percentage has a mutation in another gene. Although clinical manifestations are similar for people with PKD1 and PKD2, clients who exhibit the PKD2 mutation progress to end-stage renal failure about 10 years later than those with the PKD1 mutation.
Mutations to the gene coding for a large protein called fibrocystin on chromosome 6 lead to ARPKD. Further studies are needed in order to clearly define the role of these genes and their products in the formation of PKD.
Clinical Manifestations
Although PKD is a hereditary disorder, only 60% of people are able to give a familial history of PKD, suggesting that spontaneous mutations occur frequently. For those families with a history of PKD, individuals can be monitored. In people who lack a familial history, cysts often are asymptomatic and found incidentally on routine urographic examination.
Symptoms associated with autosomal dominant disease may include pain, hematuria, fever, and hypertension. Abdominal or flank pain is the most common symptom in ADPKD. It can be associated with bleeding, growth of cysts, stones, infection, or, rarely, tumor. Most of these clients will have significantly enlarged kidneys that are palpable abdominally.
Associated hematuria may be gross or microscopic. Rupture of a cyst usually accounts for incidents of gross hematuria. Fever can be related to an infected cyst secondary to pyelonephritis. Hypertension is hypothesized to occur as a result of sodium and water retention because of damage to the tubules.118,157 Hypertension also hastens the development of fibrosis and is linked with accelerated progression to ESRD.
Liver cysts are also common in clients with ADPKD; about half have liver cysts at diagnosis. Unlike the kidney cysts, liver cysts rarely lead to problems such as liver failure or portal hypertension. People with ADPKD may also be affected with other genetic abnormalities, such as thoracic and abdominal aortic aneurysms, cerebral aneurysms, mitral and aortic valve prolapse, colonic diverticular disease, and pancreatic cysts.
MEDICAL MANAGEMENT
Ultrasonography is used to screen for PKD. People less than 30 years of age should demonstrate at least two cysts in one kidney in order for PKD to be diagnosed. Persons between the ages of 30 and 59 should have at least two cysts in each kidney, while people older than 60 years should demonstrate four cysts per kidney. Genetic tests can be performed to corroborate radiographic information. Simple cysts are uncommon in clients with PKD; the simultaneous presence of both large and small cysts is the norm.
CT is a useful radiographic test, distinguishing between solid and fluid-filled masses and displaying the presence of cysts of varied sizes. CT can also reveal the presence of hepatic cysts, making the diagnosis of PKD more likely. Prognostic information can also be obtained from the contrast-enhancing portion of a CT. Only the normal renal tubules will have contrast in them, revealing the degree of functioning nephrons.
MRI is often a better choice, especially for children or persons with renal dysfunction, or in the early stages of the disease. Urinalysis may detect hematuria and proteinuria, or the clinical examination may reveal enlarged, palpable kidneys. As appropriate, other causes of renal cysts should be addressed. Occasionally, tissue biopsy or surgical exploration is necessary to make the definitive diagnosis.
TREATMENT.
Because hypertension is a known risk factor for progression to ESRD, blood pressure should be monitored and controlled, particularly if a family history is present and the disease is diagnosed in young adults. Stimulation of the renin-angiotensin system was thought to be the cause of hypertension in ADPKD, but this has been questioned and other causes postulated. For this reason, angiotensin-converting enzyme (ACE) inhibitors and angiotensin receptor blockers (ARBs) have not been more successful than other blood pressure medications in preventing the progression of ADPKD.126 Studies have suggested that by keeping blood pressure at a normotensive level there is a slowing of progression to ESRD.174
Pain can be controlled through analgesics or treatment of the underlying cause (i.e., treating infections with antibiotics). Percutaneous aspiration of cystic fluid followed by injection of a sclerosing agent has helped some clients with pain from expanding cysts. Surgery and laparoscopic surgery can be performed to remove or unroof large cysts for pain relief. Infections can be difficult to treat, since the infection may be isolated in the cyst or an abscess may form. Possible associated findings, such as cerebral aneurysms, should be screened for and monitored.
Renal Calculi
Urinary stone disease, or nephrolithiasis, is the third most common urinary tract disorder, exceeded only by infections and prostate disease. A majority of the stones develop in the kidneys. Once the stones move out of the kidney into the ureter, they are referred to as ureteral stones (bladder stones are considered a separate disorder).
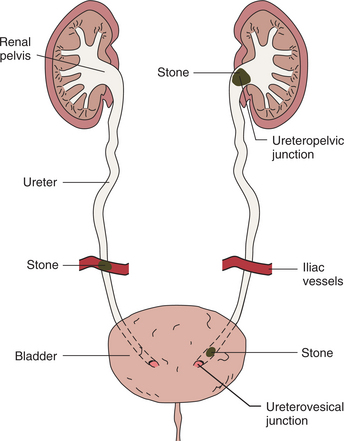
The stones, or calculi, are crystalline, ranging from popcorn kernel shapes to jagged starbursts, and can cause urinary obstruction and severe pain. Urinary obstruction typically occurs at one of the following three sites: (1) the ureteropelvic junction, (2) where the ureter crosses over the iliac vessels, and (3) at the ureterovesical junction (Fig. 18-5). The four basic types of stones are calcium (oxalate and phosphate), struvite, uric acid, and cystine.
Calcium stones are by the far the most common (70% to 85%). Struvite stones are related to recurrent bacterial UTIs with organisms that produce urease. Uric acid stones (5% to 10% of nephrolithiasis cases) occur due to an increased level of urate in the blood and uric acid crystals in the urine, which is common in persons with gout. Cystine stones are uncommon (accounting for about 1% of all cases of nephrolithiasis) and caused by a hereditary disorder, cystinuria. Affected persons are unable to absorb cystine, and large amounts are excreted in the urine.
Incidence
Nephrolithiasis occurs in about 5% of adults, with men being affected more frequently than women (6% versus 4%, respectively).182 Because of the severe and debilitating pain associated with kidney stones, cost is significant, including doctor visits, hospitalizations, and lost work. In the year 2000, over 2 million doctor visits and 177,496 hospital stays resulted from kidney stones, costing $2.07 billion.151 The primary age span for the initial presentation of the disease is 30 to 60 years of age for men, and 20 to 30 years for women. A higher incidence of renal calculi occurs in industrialized countries and areas noted for high temperatures and humidity. The incidence of this disease is highest in the hot summer months.
Etiologic and Risk Factors
With an understanding of the factors and mechanisms that lead to stone formation, risk factors are more evident and modifiable. Disorders that lead to an overexcretion and hypersaturation of calcium or oxalate can lead to stone formation. These include illnesses such as idiopathic hypercalciuria, renal tubular acidosis (RTA), primary hyperparathyroidism, and hyperoxaluria.
It is also known that low quantities of citrate (which typically binds calcium, thereby acting as an inhibitor to stone formation) can lead to nephrolithiasis. Uric acid crystals are sensitive to urine pH, coming out of solution in an acidic pH; thus an acidic urine pH can lead to uric acid stones.180 Gout is a disorder in which excess urate is excreted into the urine, leading to a supersaturation of uric acid crystals. Chronic dehydration can lead to stone formation due to a decreased fluid content compared to crystals.
Other risk factors have been identified by epidemiologic studies but the mechanism remains unclear. For example, among persons with recurrent stone formation, it is often on one side only (unilateral). Some investigators have hypothesized that sleep posture (i.e., consistently sleeping on one side) may promote stone formation on the one side.179 Obesity is associated with an increased incidence of urinary stone episodes in women but not in men.154
Excess intake of supplemental calcium, sodium, sucrose, and animal protein have been dietary risk factors implicated in stone formation. A lack of sufficient calcium and potassium in the diet can also increase a person’s risk for kidney stones.36
Pathogenesis
Several factors lead to the formation of stones—saturation, nucleation, crystal growth and aggregation, and cell/crystal interactions. Saturation refers to the amount of dissolved crystal (such as calcium oxalate or calcium phosphate) in the urine compared to volume.
Crystals are able to stay dissolved in the urine until it becomes oversaturated. Factors such as the amount of calcium, oxalate, and water excretion determine saturation. With oversaturation, crystals come out of solution (or out of the urine) into a solid and begin to grow around a particle, or nucleus. It is uncommon for urine to become so oversaturated that a new nucleus of calcium oxalate or calcium phosphate is formed. Most often, a small particle, bacteria (small nanobacteria), or other crystal already present in the urine acts as a nucleus for the crystals to grow around (i.e., stones may have a nucleus of one crystal type, but surrounded by another crystal type).
Crystals then grow at a rate depending on the saturation of the urine. The more supersaturated, the more quickly larger stones form. These stones also require enough time to enlarge, since normal flow is often sufficient to move small stones through the urinary tract. It is proposed that the growing crystal aggregate becomes attached to the urinary tract epithelium and transported into the cell membrane. Here, the cell membrane may also act as a nucleus. Investigations are ongoing to determine the significance of cell/crystal interactions.
Clinical Manifestations
Clinical symptoms are the same for the various types of stones. The classic presentation of a kidney stone is acute “colicky” flank pain radiating to the groin or perineal areas (including the scrotum in males and labia in females) with hematuria. The pain is severe; most people are unable to find a comfortable position.
The location of the pain may vary depending on where the stone is lodged in the ureter. Abdominal pain with radiation to the groin may be more pronounced if the stone is higher in the abdomen, while a stone at the ureterovesical junction may give rise to lower quadrant abdominal pain radiating to the tip of the urethra.
Symptoms consistent with a UTI such as urinary urgency and frequency and dysuria are often present. Hematuria is present in over 90% of cases (although the absence of hematuria does not mean nephrolithiasis is not the diagnosis).14 Nausea and vomiting may also be manifested.
MEDICAL MANAGEMENT
Recurrence of calculi is common in up to 50% of people within 5 years if preventive steps are not taken. A metabolic evaluation should be preformed in clients who are willing to comply with tests and prophylactic methods (only 36% to 70% of people with recurrent stones comply long term with recommended measures).148,193 Tests and appropriate preventive measures vary depending on the type of stone passed. Common tests include 24-hour urine collection for calcium, oxalate, uric acid, phosphate, citrate, pH, potassium, and creatinine; serum calcium; serum blood urea and creatinine; and parathyroid hormone (PTH). If a specific disorder, such as hyperparathyroidism, is identified, treatment reduces the risk for further stones.
Adequate fluid intake is essential to the prevention of stones and recurrences by reducing the saturation of stone-forming crystals. Clients should be encouraged to drink enough fluid to maintain clear-colored urine. Other dietary modifications are made according to stone type. Urinary uric acid can be reduced by decreasing the amount of protein ingested. Urine citrate can be increased by consuming more fruits and vegetables and decreasing the amount of acid-producing food (such as animal protein).36 A restriction of calcium is not recommended and may be harmful.13,38
Medications can be helpful in certain situations if dietary means alone are not sufficient in preventing stone formation. Thiazide diuretics (which increase calcium excretion); alkali, such as potassium citrate (beneficial in increasing urine citrate excretion); and allopurinol (prevents the precipitation of uric acid crystals) may be useful. Further research is needed to determine the efficacy of specific dietary changes and the best means to prevent kidney stones.
DIAGNOSIS.
A variety of tests are used to diagnose this disease. Noncontrast helical CT scanning is the first-line imaging test for renal colic. Once a stone has been visualized on CT, a plain radiograph can help determine the type of stone. Approximately 90% of calculi are radiopaque, making them visible on an abdominal radiograph. These types of stones are composed of calcium or other minerals, while uric acid stones are not visible on a plain radiograph. Other traditional imaging tests (e.g., ultrasonography and IVP) can also help in the management of stone disease. Hematuria, infection, the presence of stone-forming crystals, and urine pH can be determined on urinalysis.
TREATMENT.
The mainstay of treatment for acute nephrolithiasis includes intravenous fluids and medications to relieve nausea/vomiting and pain (narcotics or nonsteroidal antiinflammatory drugs [NSAIDs]). The α-blockers and calcium channel blockers are commonly used to treat hypertension, but both drugs also appear to flush out kidney stones by relaxing the ureter and increasing liquid pressure. Some physicians prefer α-blockers because they have fewer side effects. Most clients can be watched; however, some require immediate intervention to remove the stone.
Characteristics requiring urgent care include the presence of high-grade obstruction, anuria (no passage of urine), obstruction plus infection proximal to the stone, impending renal function deterioration, unresponsive pain or vomiting, or a solitary or transplanted kidney.187 In these situations, percutaneous nephrostomy or ureteral stenting can be performed. Intravenous antibiotics are given for infection; E. coli is the most common organism.
A majority of stones less than 5 mm in diameter (a little smaller than the width of a pencil eraser) will pass spontaneously; two thirds that do pass on their own do so within 4 weeks. The urine should be strained in order to retrieve any stones; these can be analyzed for crystal content.
Persons waiting for stones to pass should continue drinking fluid (enough to produce about 2 L/day of urine or keep the urine clear-colored instead of yellow). Fluids with sodium should be avoided; lemonade may be helpful since it increases urinary citrate and decreases calcium oxalate supersaturation.177 A follow-up CT 3 to 4 weeks after the initial episode will verify the passage of the stone or the need for intervention if the stone is unmoved.
Clients who have kidney stones of less than 1 cm in the proximal ureter can receive shock-wave lithotripsy. Shock-wave lithotripsy uses the transmission of shock waves (a type of sound wave) to break the calculi into fragments. Since the soft tissues of the body have similar densities, the shock waves pass through these structures with low attenuation. When the shock wave encounters a boundary between substances of differing acoustic density (i.e., a calculus in the ureter), high compressive forces are generated, causing a breakdown of the stone. The goal is to reduce the diameter to the point where spontaneous passage of the stone occurs.
Stones greater than 1 cm (and in the proximal ureter) benefit from ureteroscopy. Ureteroscopy involves passing a scope through the urethra and bladder into the ureter until the stone is reached. Then a laser (holmium: yttrium-aluminum-garnet [YAG]) is passed through the scope, the tip of the laser is placed on the stone, and the laser is discharged, producing photothermal lithotripsy.195
Stones located in the distal portion of the ureter can be treated with either method. Ureteroscopy is less expensive than shock-wave lithotripsy but requires greater technical expertise.122 Some studies show that clients slightly prefer shock-wave lithotripsy to ureteroscopy.113 Both methods produce excellent results.112 Guidelines from the Ureteral Stones Clinical Guidelines Panel are available.176
Uric acid stones are unique in their treatment. Since these stones dissolve in acid, the urine of affected clients can be acidified with potassium citrate or sodium citrate to increase the urine pH to at least 6.5. Stones are frequently not composed purely of uric acid, and further intervention may be required.
Chronic Kidney Disease
Chronic kidney disease (CKD) is defined as the alteration of kidney function or structure for greater than or equal to 3 months’ duration.46 CKD can be attributed to a variety of conditions that lead to a loss of kidney function. The three most common causes are diabetes (44%), hypertension (27%), and glomerulonephritis (8%). Cystic kidney disease and other urologic diseases account for about 5%, while other conditions (such as excessive aspirin or acetaminophen use) account for the remaining few percent.
ESRD is the final stage of CKD, with the loss of kidney function accompanied by symptoms requiring either dialysis or kidney transplant. The loss of kidney function is devastating, resulting in significant systemic effects, reduced quality of life, increased morbidity and mortality, and costing over $23 billion per year.159
Incidence
The incidence of CKD continues to increase in the United States, with more than 20 million people currently affected. The number of persons with ESRD also continues to rise, with a prevalence of over 400,000. Recent data show a significant leap in the number of people with ESRD requiring treatment (dialysis or transplant) from 1994 to 2004. In 1994, the number of people who started dialysis or received a renal transplant was 68,757; in 2004 this number had increased to 102,356.70 Much of this increase was reported in persons with diabetes, with 26,848 cases reported in 1994 and 44,953 cases in 2004.
The incidence of ESRD from diabetes, hypertension, and glomerulonephritis is higher in African Americans compared to the general population. The rate of ESRD caused by hypertension was three times higher in African Americans compared to the general population. Encouraging data from 2004, however, showed that the incidence of CKD decreased in the Native American population, although prevalence has tripled among Hispanic Americans.137
ESRD carries a high mortality rate, particularly in the older population. In 2004, people over the age of 65 who received dialysis had a mortality rate seven times that of equivalent-aged people not on dialysis.70 People over the age of 60 who start dialysis have a life expectancy of only 5 years, while a 60-year-old without ESRD can expect to live 20 more years.
Etiologic and Risk Factors
The presence of a number of diseases can account for destruction of nephrons, but diabetes mellitus (principally type 2 causes diabetic nephropathy), high blood pressure, and glomerulonephritis are the leading causes of CKD. Other disorders contributing to the development of kidney failure include PKD, urinary tract obstruction, repeated infection, hereditary defects of the kidneys, toxicities, and systemic lupus erythematosus.
An increased risk of renal damage and ESRD is also associated with excessive over-the-counter (OTC) analgesic drug use, called analgesic nephropathy. This association was first noted with phenacetin-containing analgesics,76 but is also noted with the drugs acetaminophen, aspirin, and combination analgesics (i.e., combining analgesics with codeine or caffeine).58,84
Heavy average intake or high cumulative intake of analgesics may increase the likelihood of developing ESRD, particularly in older people with a disorder already affecting the kidneys.74 NSAIDs, both selective and nonselective, have significant short-term effects on the kidney, yet data have been inconsistent with regard to the risk for ESRD from NSAID use.37,175 More research is needed to answer this question, but it may be that moderate use of NSAIDS in healthy individuals does not put them at significant increased risk for ESRD.160
Pathogenesis
The basic functioning unit of the kidney is the nephron. It is composed of the glomerulus, the renal tubules, and the collecting duct (Fig. 18-6). The glomerulus is a small bundle of capillaries, surrounded by a capsule, that allows fluid and electrolytes to pass through the membrane and into the tubules. The renal tubules transport electrolytes or create a gradient for fluid and electrolytes to become balanced, while the collecting tubule is responsible for the final regulation of electrolytes and water under the influence of the hormone aldosterone.
As discussed earlier, many disease processes can result in CKD. Diabetes, for example, induces kidney damage through hyperglycemia. Angiotensin II is also released, which causes vasoconstriction of the arterioles and arteries (both in the glomerulus and systemically) in an attempt to keep the pressure adequate for filtration.
Angiotensin II release also leads to the attraction of inflammatory cells, which release cytokines and growth factors (which change the structure of the glomerulus). These changes result in mesangial expansion (a layer of cells around the glomerular capillaries), enlargement of the glomerulus, and ultimately interstitial fibrosis and glomerular sclerosis.
These processes slowly reduce the amount of surface area available for filtration to occur, thereby reducing the glomerular filtration rate (GFR). With the gradual loss of nephron function, the kidneys are unable to adequately regulate fluid, electrolytes, and pH balance or remove metabolic waste products from the blood.
The rate of nephron destruction can vary considerably, depending on the disease process. Typically, five stages mark the progression of chronic renal failure; each stage is defined by the level of the GFR (measured in milliliters per minute): (1) kidney damage with normal or increased GFR (90 ml/min or more), (2) kidney damage with mildly decreased GFR (60 to 89 ml/min), (3) moderately decreased GFR (between 30 and 59 ml/min), (4) severely decreased GFR (15 to 29 ml/min), and (5) kidney failure (ESRD; GFR of less than 30 ml/min). Systemic complications typically develop once stage 4 CKD is reached, when the GFR is less than 30 ml/min.
Clinical Manifestations
In stage 1 of CKD, the GFR is normal or increased (hyperfiltration). No overt symptoms of impaired renal function are typically evident. Depending on the health of the person, the kidneys have tremendous adaptive and compensatory capabilities, accounting for the delay in symptoms. The unaffected nephrons undergo structural and physiologic hypertrophy in an attempt to make up for those nephrons that are no longer functioning. Results of blood tests such as blood urea nitrogen (BUN) and creatinine, which are indicative of kidney function, are typically normal.
The onset of symptoms is usually very gradual and subtle with the continued loss of nephrons and reduction in GFR, often resulting in a delay in diagnosis. Early clinical manifestations include hypertension and anemia. Abnormalities in laboratory values include an increase in BUN and creatinine, or protein detected in the urine. Stage 1 is reversible for some people (e.g., those with diabetes who benefit from early detection and proper glycemic control). Some individuals remain in stage 1 indefinitely, while others progress.
During stage 2, the damaged capillaries allow small amounts of albumin to be excreted in the urine. Individuals may remain in this stage for several years with proper control of hypertension and blood glucose levels. Stage 3 is more noticeable as albumin levels increase in the urine and decrease in the blood, resulting in noticeable edema. During this stage levels of creatinine and BUN increase, resulting in an accumulation of waste products in the blood called azotemia.48
In the final stages of CKD (stages 4 and 5, with stage 5 being ESRD), the kidney is unable to function and a multitude of complications appear with accompanying symptoms and signs. Proteinuria is the hallmark of this stage; the kidneys are no longer able to excrete toxins, so there is a progressive increase in BUN and creatinine levels. Most people in this stage are hypertensive because of an increased production of renin. Hypertension accelerates the progression to stage 5 (ESRD) when the kidneys fail to function.
Stage 5 or ESRD is heralded by a cluster of symptoms referred to as uremia. The kidneys cannot excrete toxins; maintain fluid, pH, and electrolyte balances; or secrete important hormones (e.g., renin, vitamin D, erythropoietin). Uremia develops when poorly identified toxins are not removed from the blood. Uremia is characterized by nausea, vomiting, anorexia, lethargy, pruritus (itching), sensory and motor neuropathy, pericarditis, impaired heart function, asterixis, and seizures. Asterixis is an intermittent inability to sustain a posture, often noted when holding up the hand with the wrist flexed, creating a small “flapping-like” motion.
Dialysis or kidney transplant improves these symptoms. Hematologic, cardiovascular, gastrointestinal, musculoskeletal, and neurologic complications become more common in stages 4 and 5. Table 18-1 summarizes the systemic effects associated with CKD and ESRD.
Table 18-1
Systemic Manifestations of Kidney Failure


From Goodman CC, Snyder TE: Differential diagnosis in physical therapy, ed 3, Philadelphia, 2000, Saunders.
Hematologic.: Anemia is a significant hematologic problem associated with CKD. The hormone erythropoietin, primarily produced by the interstitial cells of the kidneys, has the principal function of controlling the production of red blood cells in the bone marrow. CKD leads to decreased erythropoietin production, reduced red blood cell lifespan, and reduced iron absorption, resulting in a subsequent decrease in red blood cells and anemia.
Anemia associated with CKD occurs most frequently in clients with a GFR of less than 60 ml/min and in persons 75 years of age or older.3 However, a lack of erythropoietin is not the only factor causing anemia in clients with CKD. Other causes such as gastrointestinal bleeding, iron or folate deficiency, or hemolysis may play a role and should be evaluated.
Anemia in CKD can cause significant fatigue and reduced quality of life. Another important result of anemia is the stress it places on the heart. Anemia is an independent risk factor for cardiovascular disease. Because of this, anemia should be aggressively treated (see Chapter 14 for additional information regarding anemia).190 ESRD also leads to white blood cell dysfunction and bleeding problems due to impaired platelet function.
Cardiovascular.: Cardiovascular diseases often occur in people with CKD and are the leading cause of death in persons with ESRD. As the GFR decreases, the risk for cardiovascular disease increases in a graded fashion.71 Many of the risk factors that cause CKD, such as diabetes and hypertension, also contribute to cardiovascular disease. Diseases common in clients with CKD include coronary artery disease, left ventricular hypertrophy, and congestive heart failure. Persons with CKD often have hyperlipidemia, another risk factor for coronary artery disease.
Symptoms may include chest pain (although many often have atypical or no pain), nausea, shortness of breath, and sweating. Excess fluid volume, sodium retention, and anemia associated with ESRD lead to left ventricular hypertrophy, a thickening of the left ventricle of the heart, which predisposes to congestive heart failure.
Associated clinical features include lower extremity edema and shortness of breath. Hypertension often appears early in the course of this disease because of increased angiotensin II production. Clients in stage 3 of CKD often need two or more medications to control blood pressure. Clients with CKD also have a higher incidence of stroke, peripheral vascular disease, arrhythmias, pericarditis, and heart valve abnormalities.
Gastrointestinal.: Gastrointestinal system complaints occur often once the later stages of ESRD are reached. Azotemia (high levels of urea and other toxins in the blood) causes nausea, vomiting, and anorexia. The resultant depressed appetite contributes to malnutrition, fatigue, weakness, and malaise. Malnutrition has a high prevalence in those with advanced kidney disease, partly as a result of the therapeutic restriction on calories and proteins but also because of the metabolic reactions typical with this disease. Other clinical manifestations include gastritis, duodenitis, pancreatitis, hiccups (often difficult to control), and ascites.
Musculoskeletal.: The skeletal changes associated with CKD are common and can occur early in the disease secondary to abnormalities in calcium, phosphate, and vitamin D metabolism. With the impairment of GFR, the body is unable to excrete phosphate or synthesize 1,25-dihydroxyvitamin D (a vitamin D derivative called calcitriol). Higher blood levels of phosphate lead to low ionized calcium levels. Calcitriol normally regulates calcium absorption from the gut and inhibits the parathyroid gland. But with low levels of calcitriol, hypocalcemia and increased PTH secretion result. A drop in serum calcium signals a cascade of events starting with the release of PTH, which signals the body to increase calcium resorption from bone to make up for the perceived loss (Fig. 18-7).
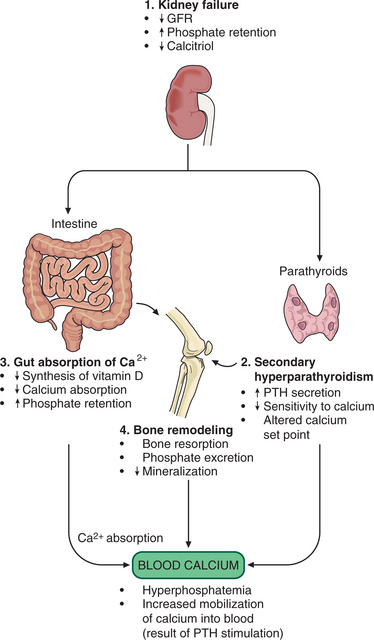
Figure 18-7 Mechanisms of altered bone turnover. (1) Renal osteodystrophy can be viewed as the result of a vicious cycle that begins with moderate to severe renal failure. As the glomerular filtration rate (GFR) decreases, phosphate excretion decreases, and calcium elimination increases. (2) The body attempts to compensate for the loss of calcitriol and reduced calcium absorption by increasing parathyroid hormone (PTH) secretion. PTH mobilizes calcium from the bones (bone reabsorption) and facilitates phosphate excretion. This release of calcium and phosphate into the blood results in hyperphosphatemia and hypercalcemia. (3) As kidney failure progresses, the damaged kidneys can no longer convert vitamin D to its active form and without active vitamin D, calcium absorption in the intestines is decreased and paradoxically facilitates phosphate retention. (4) Thus the normal process of bone mineralization with calcium and phosphate is impaired. Demineralization of the bone frees more calcium and phosphorus into the blood. As the disease progresses even more, the parathyroid gland may become unresponsive to the normal feedback system and continue to produce PTH, causing acceleration of renal osteodystrophy.
Several skeletal abnormalities result from varying types of bone turnover, collectively referred to as renal osteodystrophy. Renal osteodystrophy is a type of rickets formerly called renal rickets and sometimes referred to as azotemic osteodystrophy. Although uncommon at one time, the incidence has risen with increased survival of people with renal disease on dialysis.
Renal osteodystrophy is characterized by varying degrees of osteomalacia, osteitis fibrosa, and adynamic bone disease. Osteomalacia occurs secondary to low bone turnover due to aluminum deposition in the bones with resultant increased nonmineralized bone matrix formation. Osteitis fibrosa results in inflammation and fibrosis of bone because of high bone turnover, while adynamic bone disease is a low bone turnover state, which may be related to excessive PTH suppression from therapy.47
Clients with renal osteodystrophy may present with bone pain, especially in the spine, hips, knees, or lower extremities, and fractures.47 The pain is worse with exercise and other weight-bearing activities; the fractures occur most often in the vertebrae and long bones.48 Increased secretion of PTH and a decreased secretion of calcitriol appear to be the cause. Metabolic acidosis may also play a role, either by increasing osteoclastic activity or by increasing the effects of PTH.200
Osteopenia and pseudofractures are seen most frequently in people with osteomalacia. Osteitis fibrosa is evident on radiographic films, particularly in the phalanges, skull, and distal clavicles, where there is subperiosteal bone resorption.
In addition to bone demineralization, calcification of vessels and soft tissues occurs. This calcification may be related to bone turnover and occurs during both high and low bone turnover. It is postulated that deposition of minerals may occur in extraskeletal sites because the bone is unable to incorporate them. If bone turnover is high, minerals are removed from bone and are deposited in extraskeletal sites. If bone turnover is low, minerals are also deposited in extraskeletal sites because the bone is forming abnormally or at a slow rate.
The most common sites for extraskeletal calcification include the coronary arteries, lungs, skin, peripheral arteries, joints, and cornea. Calcifications of the coronary arteries are common and may be the reason for the high death rate in CKD clients. Intraarterial calcifications can occlude vessels, leading to ischemia and gangrene.
Deposition of minerals in the skin can lead to intense pruritus (itching), and occlusion of an arteriole can cause necrosis of skin, termed calciphylaxis. This occurs most commonly in the lower extremities, trunk, or buttocks. Articular cartilage and joints can become calcified, causing pseudogout and arthritis symptoms.
Calcification of a tendon may lead to spontaneous rupture with minimal stress. The quadriceps tendon may rupture simply by walking, tripping, or going down stairs. Tendon ruptures can lead to pain, deformity, and disability. Other areas of commonly observed ruptures in this population include the triceps and extensor tendons of the fingers.48 Hyperparathyroidism and metabolic acidosis are responsible for the abnormal collagen that results in weak tendons.29,200
Calcifications are frequently found on the cornea and conjunctiva but are typically asymptomatic. Clients who have kidney disease characterized by a slow decline in function may have more severe cases of both renal osteodystrophy and calcification of vessels and soft tissue. (Although cardiovascular manifestations have already been discussed earlier in this section, in view of the information here on calcification, it should be noted that vascular calcification of the arteries resulting in vascular insufficiency has been observed in nearly all individuals with ESRD by age 50 years. In fact, most people with CKD actually die mainly from cardiovascular disease, rather than progress to the final stages of ESRD.100,156)
Myopathy can occur with proximal muscle weakness affecting the muscles of the shoulder and pelvic girdles, leading to functional disabilities. The gluteus medius, hamstring, and psoas muscles are affected first and most severely, resulting in gait impairments and difficulty rising from low seats or accomplishing functional activities such as getting in and out of a bathtub. As the condition progresses, activities of daily living (ADLs) such as combing the hair, brushing the teeth, or household tasks become more difficult to manage independently.
Neurologic.: Alteration of central nervous system and peripheral nervous system function often occurs in association with ESRD. Early central nervous system changes include sleeping disturbances (both getting to sleep and staying asleep) followed by daytime sleepiness and personality changes.
Uremic encephalopathy occurs when the GRF is less than 10 ml/min and is manifested by recent memory loss, inability to concentrate, perceptual errors, confusion, asterixis, and decreased alertness. These symptoms abate once dialysis is instituted. Without treatment, seizures, lethargy, obtundation, and coma develop.
The peripheral neuropathy associated with uremic toxins is characterized by a dying back of the axons of both sensory and motor nerves. These neurologic changes are typically symmetrical (stocking-glove distribution) and similar to those of any other polyneuropathy with a stocking-glove distribution. Clients can also exhibit restless leg syndrome, which occurs in up to 40% of uremic clients.78 Like central nervous system manifestations of uremia, peripheral nervous system symptoms improve with dialysis.
MEDICAL MANAGEMENT
Everyone should be screened for hypertension and diabetes with early intervention when present. The ultimate goal and mission of the National Kidney Foundation is the eradication of diseases of the kidney. Toward that end, Healthy People 2010 has identified several goals related to kidney failure, including (1) reduce kidney failure due to diabetes; (2) increase the proportion of people with chronic kidney failure who receive a transplant within 3 years of registration on the waiting list; (3) increase the proportion of people with chronic kidney failure who receive counseling on nutrition, treatment choices, and cardiovascular care 12 months before the start of renal replacement therapy; (4) reduce deaths from cardiovascular disease in persons with chronic kidney failure; and (5) reduce the rate of new cases of ESRD.
Since diabetic nephropathy is the leading cause of kidney failure in the United States, prevention strategies and risk factor modification related to improving glycemic control, preventing hypertension, preventing coronary artery disease, increasing physical activity, and reducing or eliminating tobacco-related behaviors (e.g., smoking) are a large part of the prevention of renal disease. In the future, pancreas transplantation may become a part of diabetes prevention and therefore ESRD prevention.
DIAGNOSIS.
As discussed earlier, the early stages of CKD are often without symptoms. Because of the significant morbidity and mortality associated with CKD, early detection is emphasized, with the goal of slowing progression or reversing the disease if possible. People with diabetes, hypertension, or a family history of kidney disease are at high risk for kidney disease and should be monitored for early indications of kidney disease.
Because there are many causes for CKD, multiple tests may be required to determine the cause and severity. Several laboratory tests are helpful in detecting and following progression of CKD and ESRD. Persons with diabetes should routinely be tested for microprotein in the urine (an indication of early kidney disease). GFR, BUN, and creatinine are blood tests that can indicate kidney dysfunction (GFR is actually calculated using laboratory results).
In the presence of CKD, GFR will be the first laboratory indicator of renal damage, followed by increases in BUN and creatinine with continued kidney damage. GFR not only characterizes the stage of kidney disease but also demonstrates disease progression. Urine tests may show protein or casts (abnormal protein “castings” of the tubules). For those persons with known CKD or ESRD, intact PTH (or N-terminal PTH molecule) levels should be monitored in an attempt to avoid renal osteodystrophy.
Imaging modalities may demonstrate obstruction, masses, or bilateral small kidneys (which is consistent with ESRD from a chronic disease). A kidney biopsy may be needed to confirm specific kidney pathology (such as a glomerulonephritis).
TREATMENT.
Following the diagnosis of CKD, a referral to a nephrologist should be made. Although best outcomes occur when a referral is made early in the course of CKD, up to 40% are not referred until 3 months prior to initiating dialysis.192 Delayed referrals lead to an increase in morbidity and mortality.
The goals of treating CKD include treating the underlying disease, modifying risk factors for cardiovascular disease, and preventing further loss of kidney function. For some diseases, treating the principal disease may involve immunosuppressive agents, such as with the treatment of membranous nephropathy. Renal artery stenosis may require angioplasty with stenting. Diabetes requires tight glycemic control.
Cardiovascular complications are common and require risk factor modification and treatment. Clients with CKD should lose weight, exercise, eliminate smoking, and modify their diet. Dyslipidemia treatment involves lifestyle modifications, and medication may be needed. Blood pressure should be strictly controlled in order to reduce cardiovascular risk and prevent further loss of renal function (although the exact range is disputed, a systolic blood pressure between 120 mm Hg and 130 mm Hg should be the goal).120
Measures that help prevent further loss of kidney function include avoidance of nephrotoxic drugs and radiocontrast agents; treatment of anemia; strict blood pressure control; and the use of ACE inhibitors or ARBs.120
For the predialysis stage, data are lacking to establish the optimal hemoglobin level to begin erythropoietin therapy. But a current recommendation suggests initiation of subcutaneous erythropoietin when the hemoglobin drops below 10 g/dl, after verification of adequate iron stores and exclusion of other causes of anemia. Studies have demonstrated that the use of an ACE inhibitor or ARB is renoprotective beyond the ability of just lowering blood pressure.
The treatment of clients who develop ESRD continues with many of the same measures used to treat the other stages of CKD, such as blood pressure control and anemia treatment. Dietary modifications are necessary, including low-potassium, low-sodium, and low-protein diets. Excess protein can increase urea levels, and clients should not consume more than 1 g/kg/day of protein. Dietitian referrals are important, since malnutrition may occur with strict dietary regulation. Fluid intake is restricted, and diuretics are needed to maintain fluid balance.
Prevention of renal osteodystrophy necessitates monitoring of calcium/phosphate balance and PTH levels. The goal is to maintain calcium at less than 9.5 mg/dl and phosphate at less than 5.5 mg/dl. This has typically been accomplished using calcium-binding agents and vitamin D sterols.
New questions have been raised, however, concerning calcification of vessels (principally the coronary arteries) and calcium/phosphate metabolism. The traditional methods of lowering calcium and phosphate may still leave levels too high to reduce calcification of the coronary arteries. A new calcimimetic agent, recently approved by the FDA, called cinacalcet, appears to treat secondary hyperparathyroidism and maintain normal calcium and phosphate levels. Further use of this drug in a larger population will determine efficacy in preventing renal osteodystrophy and cardiovascular complications.
Renal replacement therapy (dialysis or transplantation) is the treatment of choice for ESRD. The decision is based on the client’s age, general health, donor availability, and personal preference. Dialysis can take the form of hemodialysis (HD) or peritoneal dialysis (PD) (also referred to as continuous ambulatory peritoneal dialysis [CAPD] and continuous cycling peritoneal dialysis [CCPD]). The main difference between these methods of dialysis is in their exchange schedules (daytime versus nighttime, length of time required).
HD typically requires three sessions per week for 3 to 4 hours per session. The blood flows from an artery through the dialysis machine chambers and then back to the body’s venous system, with the waste products and excess fluid and electrolytes diffusing into the dialyzing solution. HD can be done at home, although this requires specialized training, or the client can travel to a renal center. During the procedure, the person remains relatively immobile throughout the session. Only 1% of dialysis clients receive home HD, while 85% receive in-center treatment.
Approximately 10% of dialysis clients use PD. PD relies on the same principles as HD but can be done independently at home. A catheter is implanted in the peritoneal cavity, and sterile dialyzing solution is instilled and then drained over a specific period. This process is completed four times daily. With CAPD/CCPD, fewer dietary restrictions and fewer of the dramatic symptom swings associated with HD occur. Potential complications of PD include infection, catheter malfunction, dehydration, hyperglycemia, and hernia. Peritonitis occurs about once every 3 years in those undergoing PD and is the most serious potential complication. The majority of affected people are successfully treated with intraperitoneal antibiotics.
The steady improvement in the outcome of renal allografts has made kidney transplantation the treatment of choice for many people with ESRD. Transplantation is less expensive than long-term dialysis, but donor availability limits the number of transplants performed. The current contraindications for transplant include active substance abuse or noncompliance, metastatic cancer, severe arterial disease involving the iliac arteries, active infection, active ischemic cardiac and cerebrovascular disease, advanced dementia, and debility.
In adults, the renal graft is placed extraperitoneally in the iliac fossa through an oblique lower abdominal incision. In small children, the graft is located retroperitoneally with a midline abdominal incision. Complications of the procedures include renal artery thrombosis, urinary leak, and lymphocele.
PROGNOSIS.
Despite significant advances in technologic and pharmacologic interventions, the current annual mortality rate of people with ESRD in the United States is about 24%. Cardiovascular diseases remain the number one cause of death in those with all categories of renal disease, including persons with CKD, those with ESRD on dialysis, and renal transplant recipients. This is most likely due to the presence of multiple cardiovascular risk factors (e.g., hypertension, abnormal lipids, smoking, dietary factors).123 Individuals with diabetes and ESRD have higher morbidity and mortality rates than individuals with ESRD only.136