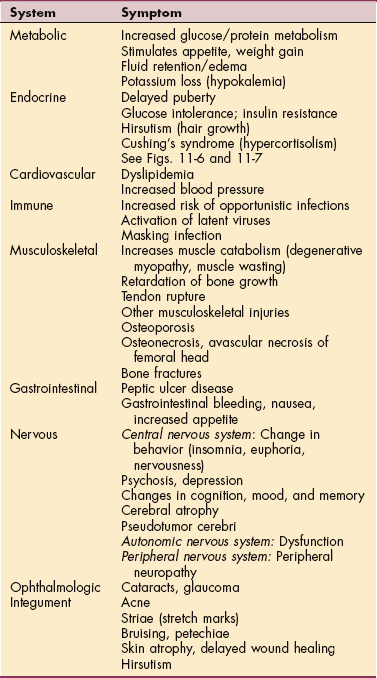
Problems Affecting Multiple Systems
Many conditions and diseases seen in the rehabilitation setting can affect multiple organs or systems (Box 5-1). With the kinds of multiple comorbidities and system impairments encountered in the health care arena, the therapist must go beyond a systems approach and use a biopsychosocial-spiritual approach to client management. Chronic diseases and multiple system impairments require such an approach because risk factors correlate with health outcome; early intervention and intervention results are correlated with improved outcome.
Individual modifying (risk) factors (IMF) such as lifestyle variables and environment affect pathology and modify how a person responds to health, illness, and disease. For example, adverse drug reactions are correlated with increasing age and obesity, whereas fitness level has a profound impact on recovery from injury, anesthesia, and illness.
Additionally, a single injury, disease, or pathologic condition can predispose a person to associated secondary illnesses. For example, the victim of a motor vehicle accident in Chapter 6 (see Special Implications for the Therapist: Cell Injury) suffered a traumatic brain injury (TBI) and concomitant pelvic fracture then developed pneumonia and pulmonary compromise, subsequently experiencing a myocardial infarction.
This type of clinical scenario involving multiple organs and comorbidities is not uncommon. Also consider the medically complex person who needs a splint. The therapist must first review laboratory values (see Chapter 40) to determine albumin levels (nutritional status) and platelet levels (potential for bleeding), perform a skin assessment (see Chapter 10), and consult with both nursing staff and the nutritionist before providing an external device that could create skin breakdown and add to an already complex case.
Although medical conditions encountered in the clinic or home health care setting are discussed individually in the appropriate chapter, the health care provider must understand the systemic and local effects of such disorders. This chapter provides a brief listing of the systemic effects of commonly encountered pathologic conditions and a basic presentation of acid-base and fluid and electrolyte imbalances. The scope of this text does not allow for an in-depth discussion of each condition or disease and its related multiple systemic effects.
The importance of using the Guide to Physical Therapist Practice6 is recognized throughout this text. However, identifying Preferred Practice Patterns in multisystem disorders depends on the presenting signs and symptoms and their influence on individual function and the rehabilitation process. For this reason, specific practice patterns are not listed with each section. The reader is referred to individual chapters discussing the underlying pathology or directly to the Guide.
SYSTEMIC EFFECTS OF PATHOLOGY
Systemic Effects of Acute Inflammation
Acute inflammation can be described as the initial response of tissue to injury, particularly bacterial infections and necrosis, involving vascular and cellular responses. Local signs of inflammation (e.g., redness, warmth, swelling, pain, and loss of function) are commonly observed in the therapy setting. Local inflammation can lead to abscesses when excessive suppuration (formation of pus) occurs.
Systemic effects of acute inflammation include fever, tachycardia, and a hypermetabolic state. These effects produce characteristic changes in the blood, such as elevated serum protein levels (C-reactive protein, serum amyloid A, complement, and coagulation factors) and an elevated white blood count (leukocytosis).79 For a complete discussion of inflammation and its effects, see Chapter 6.
Systemic Effects of Chronic Inflammation
Chronic inflammation is the result of persistent injury, repeated episodes of acute inflammation, infection, cell-mediated immune responses, and foreign body reactions. The tissue response to injury is characterized by accumulation of lymphocytes, plasma cells, and macrophages (mononuclear inflammatory cells) and production of fibrous connective tissue (fibrosis). Fibroblasts and small blood vessels, along with collagen fibers synthesized by fibroblasts, constitute fibrosis. Grossly, fibrotic tissue is light gray and has a dense, firm texture that causes contraction of the normal tissue.
The associated fibrosis may cause progressive tissue damage and loss of function. Systemic effects of chronic inflammation may include low-grade fever, malaise, weight loss, anemia, fatigue, leukocytosis, and lymphocytosis (caused by viral infection).31 Inflammation is reflected by an increased erythrocyte sedimentation rate (ESR). In general, as the disease improves, the ESR decreases.
Systemic Factors Influencing Healing
In addition to local factors that affect healing (e.g., infection, blood supply, extent of necrosis, presence of foreign bodies, protection from further trauma or movement), a variety of systemic factors influence healing as well (see Box 6-4). Systemic factors may include general nutritional status, especially protein and vitamin C; psychologic well-being; presence of cardiovascular disease, cancer, hematologic disorders (e.g., neutropenia), systemic infections, and diabetes mellitus; and whether the person is undergoing corticosteroid or immunosuppressive therapy.99
Healing in specific organs varies according to the underlying cause and site of the injury. For example, myocardial infarctions heal by scarring, and the heart may be weakened. A cerebrovascular accident (CVA), or stroke, may cause permanent disability, and healing occurs by the formation of glial tissue (e.g., astrocytes, oligodendrocytes, and microglia) rather than by collagenous scar formation; this process is called gliosis.
In other organs, effective tissue regeneration depends primarily on the site of injury. Necrosis of only parenchymal (functional visceral) cells with retention of the existing stroma (framework or structural tissue) may permit regeneration and restoration of normal anatomy, whereas necrosis that involves the mesenchymal framework (connective tissue, including blood and blood vessels) usually results in scar formation (e.g., as in hepatic cirrhosis). For further discussion, see Chapter 6.
Consequences of Immunodeficiency
Immunodeficiency diseases are caused by congenital (primary) or acquired (secondary) failure of one or more functions of the immune system, predisposing the affected individual to infections that a noncompromised immune system could resist. The therapist is more likely to encounter individuals with acquired (rather than congenital) immunodeficiency from nonspecific causes, such as those that occur with viral and other infections; malnutrition; alcoholism; aging; autoimmune diseases; diabetes mellitus; cancer, particularly myeloma, lymphoma, and leukemia; chronic diseases; steroid therapy; cancer chemotherapy; and radiation therapy.62
Predisposition to opportunistic infections, resulting in clinical manifestations of those infections, is the primary consequence of immunodeficiency. Selective B-cell deficiencies predispose an individual to bacterial infections. T-cell deficiencies predispose to viral and fungal infections. Combined deficiencies, including acquired immunodeficiency syndrome (AIDS), are particularly severe because they predispose to many kinds of viral, bacterial, and fungal infections.
Systemic Effects of Neoplasm
Malignant tumors, by their destructive nature of uncontrolled cell proliferation and spread, produce many local and systemic effects. Locally, the rapid growth of the tumor encroaches on healthy tissue, causing destruction, necrosis, ulceration, compression, obstruction, and hemorrhage.
Pain may or may not occur, depending on how close tumor cells, swelling, or hemorrhage occur to the nerve cells. This process also occurs locally at metastatic sites. Pain may occur as a late symptom as a result of infiltration, compression, or destruction of nerve tissue. Secondary infections often occur as a result of the host’s decreased immunity and can lead to death.187
The person with a malignant neoplasm often presents with systemic symptoms such as gradual or rapid weight loss, muscular weakness, anorexia, anemia, and coagulation disorders (granulocyte and platelet abnormalities). Continued spread of the cancer may lead to bone erosion or liver, gastrointestinal (GI), pulmonary, or vascular obstruction. Other vital organs may be affected; increased intracranial pressure in the brain by tumor cells can cause partial paralysis and eventual coma. Hemorrhage caused by direct invasion or necrosis in any body part leads to further anemia or even death if the necrosis is severe.
Advanced cancers produce cachexia (wasting) as a result of tissue destruction and the body’s nutrients being used by the malignant cells for further growth. Multiple mechanisms may be involved in this process, including release of cytokines such as tumor necrosis factor (also called cachectin).
Paraneoplastic syndromes (see Chapter 9) are produced by hormonal mechanisms rather than by direct tumor invasion. For example, hypercalcemia can be caused in cases of lung cancer by the secretion of a peptide with parathyroid hormone, and polycythemia can be caused by the secretion of erythropoietin by renal cell carcinoma. Neuromuscular disorders, such as Eaton-Lambert syndrome, polymyositis/dermatomyositis, and hypertrophic pulmonary osteoarthropathy, are other examples of paraneoplastic syndromes that can occur as a systemic effect of neoplasm (see Tables 9-4 and 9-5).
ADVERSE DRUG REACTIONS
Drugs were once developed through a hit or miss process in which researchers would identify a compound and test it in cells and animals to determine its effect on disease. When a compound appeared to be successful, it was often tested in humans with little knowledge of how it worked or what side effects it might have. Today, biochemists know much more about disease processes and work at the molecular level designing drugs to interact with specific molecules. Disease-modifying antirheumatic drugs (DMARDs), selective estrogen receptor modulators (SERMs), and cyclooxygenase enzyme 2 (COX-2) inhibitors are examples of such “designer drugs.”
Drugs in the future will have greater molecular specificity, possibly with the ability to accommodate for gender, age, and genetic differences between individuals. Despite these advances, adverse drug reactions still remain a significant problem in the health care industry today.
Definition and Overview
Adverse drug reactions (ADRs) are defined as unwanted and potentially harmful effect(s) produced by medications or prescription drugs. The term usually excludes nontherapeutic overdosage such as accidental exposure or attempted suicide. Side effects are usually defined as predictable pharmacologic effects that occur within therapeutic dose ranges and are undesirable in the given therapeutic situation. Overdosage toxicity is the predictable toxic effect that occurs with dosages in excess of the therapeutic range for a particular person.59
ADRs are classified as mild (no antidote, therapy, or prolongation of hospitalization necessary), moderate (change in drug therapy required, although not necessarily a cessation of therapy; may prolong hospitalization or require special treatment), severe (potentially life-threatening, requires discontinuation of the drug and specific treatment of the adverse reaction), and lethal (directly or indirectly leads to the death of the person).
Incidence
Although it is recognized that ADRs are not uncommon, the exact incidence among ambulatory or outpatient clients remains undetermined. According to one study of primary care (outpatients) ADRs were common (25%) and often preventable.71 The Centers for Disease Control and Prevention (CDC) report inappropriate medications are prescribed to older adults in about 1 out of every 12 visits (8%).76
Specific studies have been conducted to determine the rate of ADR-related admissions to hospitals, medication-dispensing errors in hospital pharmacies28 and outpatient chemotherapy clinics,70 and the rate of ADRs associated with immunizations.90 Medication changes are also common during transfer between hospital and nursing home and are a cause of adverse drug events. Most changes are discontinuations, dose changes, and class substitutions.20
The Institute of Medicine reports that at least 7000 ADR deaths occur annually in the United States and account for 2% to 3% of hospitalizations. This most likely underestimates the actual number, and efforts are being made throughout the medical system to improve reporting and monitoring complications from drug reactions.
Etiologic and Risk Factors
Definite risk factors for experiencing a serious ADR can include age (over age 75, younger for some pharmaceuticals), gender, ethnicity (occurring most often in older adult white women), concomitant alcohol consumption, new drugs, number of drugs, dosages, concomitant use of herbal compounds,34 duration of treatment, noncompliance (e.g., unintentional repeated dosage), small stature, and presence of underlying conditions (e.g., hepatic or renal insufficiency).26,29,130
Of all the risk factors, age has the most prevalent effect in the aging American population. A number of factors that affect the distribution of drugs are altered by age. A decrease in lean body mass and an increase in the proportion of body fat result in a decrease in body water. As a result, water-soluble drugs (e.g., morphine) have a lower volume of distribution that speeds up onset of action and raises peak concentration. High peak concentrations are associated with increased toxicity.
On the other hand, lipid-soluble drugs are distributed more widely, have a delayed onset of action, and accumulate with repeated dosing. Aging adults are also at risk for drug accumulation because of changes in both metabolism and elimination. With advanced age, functional liver tissue diminishes and hepatic blood flow decreases. Consequently, the capacity of the liver to break down and convert drugs and their metabolites declines. This may be exacerbated by other changes such as age-related reduction in renal mass and blood flow, the accompanying decline in glomerular filtration and tubular reabsorption rates, and other conditions such as dehydration, cancer, heart failure, and cirrhosis.
Additionally, drugs commonly prescribed for older clients, such as the calcium-channel blockers verapamil and diltiazem and the antigout drug allopurinol, further slow drug metabolism, potentially contributing to toxicity and adverse drug reactions. The drugs most commonly associated with ADRs in the aging are listed in Box 5-2. Halothane (anesthesia)–induced hepatic necrosis and anaphylactic reaction to penicillin are among the most common fatal reactions. The risk of fatal reaction is increased in older adults taking multiple drugs.
ADRs may be dose-related (predictable drug injury) or non–dose-related (unpredictable or idiosyncratic drug injury). Dose-related effects may include drug toxicity from overdose, variations in pharmaceutical preparations, preexisting liver disease, presence of comorbidities such as renal or heart failure, or drug interactions. Non–dose-related effects may occur as a result of hypersensitivity, resulting in acute anaphylaxis or delayed hypersensitivity or other nonimmunologic idiosyncratic reactions, according to individual susceptibility.
Cardiac or pulmonary toxicity may occur as a result of irradiation and immunosuppressive drugs given to prepare recipients for organ transplantation or for treatment of cancer. Some of the more common specific target organs and effects are listed in Box 5-3.
Clinical Manifestations
Rashes, fever, and jaundice are common signs of drug toxicity. Adverse skin (cutaneous) reactions include erythema, discoloration, itching, burning, urticaria, eczema, acne, alopecia, blisters, or purpura (Fig. 5-1). Onset may be within minutes to hours to days. Other signs and symptoms range from altered taste, anxiety, dizziness, headache, nasal congestion, shakiness, and brief vomiting (mild reaction) to abdominal cramps, dyspnea, hypertension or hypotension, palpitations, tachycardia, and persistent vomiting (moderate reaction) to arrhythmia, seizures, laryngeal edema, profound hypotension, pulmonary edema, and cardiopulmonary arrest (severe reaction). Arthralgias and myalgias can be part of the mild or moderate reactions.
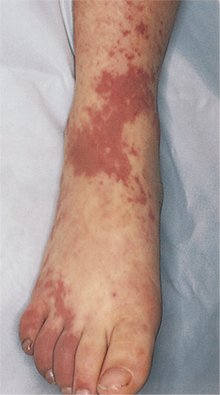
Figure 5-1 Purpura. Hemorrhaging into the tissues, particularly beneath the skin or mucous membranes, producing raised or flat ecchymoses or petechiae. Seen most often in a physical therapy practice as a result of thrombocytopenia (e.g., drug-reaction or medication-induced, especially NSAIDs, methotrexate, Coumadin or warfarin; radiation-or chemotherapy-induced); also occurs in older adults as blood leaks from capillaries in response to minor trauma. (From Hurwitz S: Clinical pediatric dermatology: a textbook of skin disorders of childhood and adolescence, ed 2, Philadelphia, 1993, Saunders.)
Older adults may develop ADRs that are clearly different from those seen in younger persons (Box 5-4).138 The therapist may observe motor tics called tardive dyskinesia, which is a neurologic syndrome caused by the long-term use of neuroleptic drugs. Neuroleptic drugs are usually prescribed for psychiatric disorders but may be used for some GI and neurologic disorders. Tardive dyskinesia is characterized by repetitive, involuntary, purposeless movements. The client may demonstrate repetitive grimacing, tongue protrusion, lip smacking, puckering and pursing, and rapid eye blinking. Rapid movements of the arms, legs, and trunk may also occur. Involuntary movements of the fingers may give the person the appearance of playing an invisible guitar or piano.
Early symptoms of salicylate intoxication include tinnitus, disequilibrium, drowsiness, and a moderate delirium.130 Digitalis toxicity is a life-threatening condition that may present with systemic (nausea, vomiting) or cardiac manifestations (see Table 12-5 and the section on Chemotherapy in this chapter).
MEDICAL MANAGEMENT
Differentiating an ADR from underlying disease requires a thorough history, especially when a symptom appears 1 to 2 months after a medication regimen has been started. Monitoring blood cell counts, liver enzymes, electrolytes, blood urea nitrogen (BUN), and creatinine is indicated for certain drugs. Digoxin and other cardiotropic drugs cause arrhythmias that require electrocardiogram (ECG) monitoring. With dose-related ADRs, dose modification is usually all that is required, whereas with non–dose-related ADRs, the drug therapy is usually stopped and reexposure avoided.
SPECIFIC DRUG CATEGORIES
Nonsteroidal Antiinflammatory Drugs
Nonsteroidal antiinflammatory drugs (NSAIDs) are a heterogeneous group of drugs that reduce inflammation, provide pain relief, and reduce fever. NSAIDs are commonly used postoperatively for discomfort; for painful musculoskeletal conditions, especially among the older adult population; and in the treatment of inflammatory rheumatic diseases.
These medications may consist of OTC preparations, such as acetylsalicylic acid (ASA) or aspirin; other salicylates; ibuprofen (e.g., Advil, Motrin, Nuprin, Medipren, Rufen), naproxen (Aleve); or prescription drugs (Table 5-1). Because of their extensive clinical uses, NSAIDs are taken by over 13 million Americans.185
Table 5-1
Nonsteroidal Antiinflammatory Drugs*

*The listing of these drugs does not imply endorsement.
†These all have “extras” in them beside aspirin but are known as aspirin products.
Modified from Goodman CC, Snyder TE: Differential diagnosis in physical therapy, ed 3, Philadelphia, 2000, WB Saunders.
Although the incidence of serious side effects from using NSAIDs is rather low, the widespread use of readily available OTC NSAIDs results in a substantial number of people being adversely affected. Risk factors associated with increased toxicities include age, higher doses, volume depletion, concurrent use of corticosteroids or anticoagulants, previous history of GI bleed or ulcer, or serious comorbidities.24,185 The use of NSAIDs is associated with a wide spectrum of potential clinical toxicities (Table 5-2), but serious side effects are most often seen with the GI tract, kidneys, and cardiovascular system.
Table 5-2
Possible Systemic Effects of Nonsteroidal Antiinflammatory Drugs

GI, Gastrointestinal; CHF, congestive heart failure; NSAIDs, nonsteroidal antiinflammatory drugs; ASA, aspirin; ACE, angiotensin-converting enzyme.
NSAIDs may cause GI symptoms ranging from mild dyspepsia to more serious complications such as GI bleeding, ulceration, and perforation. These serious side effects may occur without previous symptoms (e.g., dyspepsia) and are particularly more likely to occur in persons taking higher doses, in older adults, and with chronic use.
GI toxicity from NSAIDs is a serious problem in the United States, with over 100,000 people hospitalized because of serious complications and over 16,500 deaths each year.185,156 Medications may be used prophylactically for clients requiring NSAID treatment (e.g., osteoarthritis or rheumatoid arthritis [RA]), such as proton pump inhibitors and misoprostol, which have been shown to reduce the development of symptomatic ulcers. Misoprostol may also reduce the risk for other serious GI complications.84
NSAIDs have multiple mechanisms of action, which affect the body both locally and systemically. One of these mechanisms is to inhibit the enzymes COX-1 and-2. COX-1 is involved in synthesizing prostaglandins from arachidonic acid and felt to be a “housekeeping” enzyme. When COX-1 is inhibited, there is a reduction in prostaglandins, resulting in a decrease of the protective mucosal barrier of the GI tract.
COX-2 is thought to be produced during states of inflammation. For this reason, NSAIDs were created that selectively inhibit COX-2 but not COX-1 (Vioxx, Celebrex, and Bextra), with the hope of reducing GI side effects. Yet further scrutiny and clinical trials19,21 noted that users of Vioxx exhibited an increase in myocardial infarctions and stroke.
In September 2004, Vioxx and subsequently Bextra were removed from the market. Celebrex remains on the market with a black-box warning, although questions continue as to whether this increase in cardiovascular events is a class-wide problem.107,157 Although COX-2 inhibitors were initially designed to reduce GI bleeding, studies did not show this in people taking Celebrex long-term113 and GI bleeding was significantly reduced but remained present with Vioxx.19 COX-2 inhibitors may effectively reduce inflammation, yet they do not inhibit thromboxane, a prothrombotic enzyme, leading to continued platelet aggregation and serious cardiovascular complications.
In the kidney, COX-dependent prostaglandins are also involved with renin release, sodium excretion, and maintenance of renal blood flow, especially during times of volume contraction. To varying degrees, all NSAIDs can cause sodium retention and edema in susceptible people. Inhibition of COX enzymes leads to hyperkalemia, because of the suppression of the renin-aldosterone system; sodium retention, with resulting edema; and decreased glomerular filtration rate (GFR), resulting in edema, hypertension, and rarely acute renal insufficiency.23,24
NSAIDs are also known to interact with hypertension medications, particularly angiotensin-converting enzyme (ACE) inhibitors and β-blockers, thereby modestly increasing blood pressure in persons with hypertension.135 Papillary necrosis, nephritic syndrome, and interstitial nephritis infrequently occur as a result of NSAID treatment. Nephrotoxicity occurs especially in older clients with volume depletion, congestive heart failure (CHF), or underlying renal disorders. Careful monitoring is required of older adults taking NSAIDs in both the short-and long-term.126
NSAIDs are reversible platelet inhibitors resulting in antiplatelet activity. Aspirin is the most powerful agent because it irreversibly binds to platelets. A single dose of aspirin impairs clot formation for 5 to 7 days, and two aspirin can double bleeding time. These characteristics of ASA also make it an important drug in the treatment of coronary artery disease, myocardial infarctions, and stroke.
Immunosuppressive Agents
Immunosuppressive agents are used traditionally and most frequently in organ and bone marrow transplantation. These medications have also been found to be helpful in treating a few other diseases, but because of their significant toxicities are only indicated for serious, debilitating, and nonresponding disease (e.g., RA and psoriasis that have not responded to any other medication).
Immunosuppressive agents fall into five classes and have various mechanisms of action. The five main types of immunosuppressives include drugs that bind to small molecules (DNA or proteins in the cells), glucocorticoids, protein drugs, fusion proteins, and intravenous (IV) immune globulins.80
Cyclosporin (CSA), tacrolimus, and azathioprine are examples of small-molecule drugs. CSA and tacrolimus inhibit calcineurin, thereby inhibiting T-cell activation. Azathioprine interferes with deoxyribonucleic acid (DNA) synthesis. Newer small-molecule drugs include mycophenolate mofetil (MMF) and sirolimus (and everolimus, which is a derivative of sirolimus), which inhibit T-and B-cell proliferation and interfere with cytokine-driven T-cell proliferation, respectively.
Polyclonal antithymocyte globulin (Thymoglobulin) and muromonab-CD3 (Orthoclone OKT3) are targeted antibodies (protein drugs), which deplete or profoundly reduce lymphocytes (“depleting antibodies”). Basiliximab (Simulect) and daclizumab (Zenapax) are nondepleting antibodies, which inhibit T-cell activation. Corticosteroids are discussed in the next section.
IV immune globulins (IVIGs) have many uses; one use is for graft-versus-host disease.13 These medications are often used at different times of transplantation, depending on the medical situation such as induction or aggressive immunosuppression during the first few days after transplant, maintenance, or acute rejection.
Drugs are administered at the lowest possible doses while still maintaining adequate immunosuppression. Individual medical factors often determine the choice of immunosuppressive agent. For example, clients who have hypertension or hyperlipidemia may be given tacrolimus instead of CSA. Usually, intensive immunosuppression is required only during the first few weeks after organ transplantation or during rejection crises. Subsequently, the immune system accommodates the graft and can be maintained with relatively small doses of immunosuppressive drugs with fewer adverse effects.
Complications of immunosuppressive medications are many and can be serious. Most agents exhibit three effects: the desired immunosuppressive effect, nonimmune toxicities, and adverse effects related to immunosuppression.80 Nonimmune toxicities vary depending on the drug. Table 5-3 provides a summary of drug-related adverse effects of immunosuppressive agents.
Table 5-3
Major Immunosuppressive Agents and Adverse Effects*
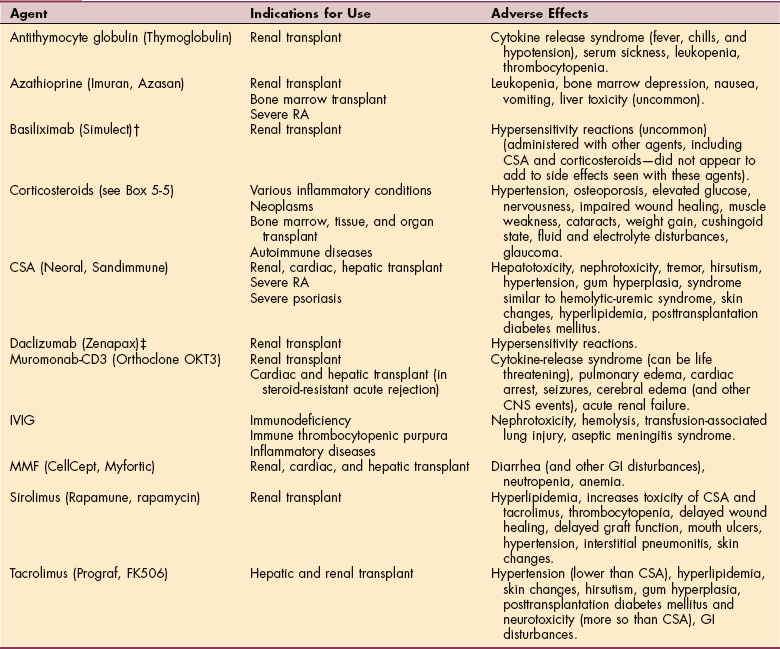
*All immunosuppressive agents (except basiliximab and daclizumab) increase the incidence of infection and may increase the potential for malignancies (posttransplant lymphoproliferative disease, skin malignancies).
†Basiliximab and daclizumab are used with CSA (or equivalent) and corticosteroids.
RA, Rheumatoid arthritis; CSA, cyclosporine; IVIG, intravenous immune globulins; MMF, mycophenolate mofetil; CNS, central nervous system; GI, gastrointestinal.
Data from Halloran PF: Immunosuppressive drugs for kidney transplantation. N Engl J Med 351(26):2715-2729, 2004; manufacturer’s inserts for health care professionals (Physicians’ Desk Reference, 2006).
Serious adverse reactions include anaphylactic reactions, renal failure, hepatotoxicity, cytokine-release syndrome, and neurotoxicities.134 Careful drug monitoring is required for CSA and tacrolimus, since toxicities are often dosage dependent. Complete blood counts are needed with azathioprine therapy and lipid monitoring is required for the drug sirolimus.
Adverse effects related to immunosuppression are often the most serious consequences of transplantation. All immunosuppressive agents, except basiliximab and daclizumab, render transplant recipients prone to infection, particularly cytomegalovirus. There is also an increased risk of developing fungal (especially Candida species) and bacterial infections. Viruses, such as herpes simplex virus and varicella zoster, may disseminate or reactivate.
An increase in certain kinds of malignancy occurs with long-term use of immunosuppressants, including lymphoma and other lymphoproliferative malignancies and nonmelanoma skin cancers.131 Host and graft survival are improving, making infection and cancer more relevant complications. Newer protocols are being developed to reduce the risk for infection and cancer.80
CSA and azathioprine may be used in the management of severe, debilitating, active RA. The dosage used to treat RA is much lower than that used to treat transplant clients. Likewise, the side effects are fewer than those associated with doses for transplantation.
Corticosteroids
Corticosteroids are naturally occurring hormones produced by the adrenal cortex and gonadal tissue. These hormones are steroid-based with similar chemical structures but quite different physiologic effects. Generally, they are divided into glucocorticoids (cortisol), which mainly affect carbohydrate and protein metabolism; mineralocorticoids (aldosterone), which regulate electrolyte and water metabolism; and androgens (testosterone), which cause masculinization. Many steroid hormones can be synthesized for clinical use. Box 5-5 contains a list of commonly prescribed synthetic corticosteroids.
Glucocorticoids are used to decrease inflammation in a broad range of local or systemic conditions (Box 5-6), for immunosuppression (see previous section on Immunosuppressive Agents), and as an essential replacement steroid for adrenal insufficiency.
Therapists most often see people who have received prolonged, systemic glucocorticoid therapy in the treatment of cancer, transplantation, autoimmune disorders (e.g., systemic lupus erythematosus), and respiratory diseases (e.g., asthma). Mineralocorticoids are given for adrenal insufficiency or type IV renal tubular acidosis (RTA), while androgens are given for deficiency states.
Generally, glucocorticoids cause fluid imbalances, and mineralocorticoids cause electrolyte imbalances. However, mineral corticosteroids are used minimally (e.g., for adrenal insufficiency or adrenogenital syndrome). Most adverse effects seen by the clinical therapist will be related to glucocorticosteroids. Adverse effects of anabolic steroids primarily occur in an athletic or sports-training setting.
Adverse Effects of Glucocorticoids
Glucocorticoids are effective antiinflammatory agents. They reduce inflammation by interacting with cell membrane receptors and activating antiinflammatory proteins (not requiring interaction with DNA). They also turn off genes involved with producing inflammatory agents (transrepression), such as cytokines, chemokines, adhesion molecules, and inflammatory enzymes, while turning on genes that produce antiinflammatory proteins (transactivation).165 But long-term use to sustain the benefits of these drugs is accompanied by an increased risk of side effects and adrenal suppression.183
Most illnesses that require glucocorticoids for treatment are used for a limited time along with disease-modifying medications, while long-term use is only necessary for adrenal insufficiency and a few other diseases (e.g., RA and inhaled steroids for asthma).
Glucocorticoids affect many functions of the body, especially in persons taking long-term steroids. Therapists should be familiar with common adverse effects such as change in sleep and mood, GI irritation, hyperglycemia, and fluid retention (Table 5-4); side effects are related to dose and duration of treatment. The most serious side effect of steroid use is increased susceptibility to infection and the masking of inflammatory symptoms from infection or intra-abdominal complications.
Most clients taking glucocorticoids notice a change in mood, behavior, or sleep. Individuals often describe a nervous or “jittery” feeling. Symptoms may range from mild anxiety to confusion or psychosis. Changes are typically noted 5 to 14 days after glucocorticoid therapy begins; improvement is seen with withdrawal of the medication.
Effects on Skin and Connective Tissue.: Effects on the skin and connective tissue include thinning of the subcutaneous tissue accompanied by splitting of elastic fibers with resultant red or purple striae (stretch marks). Ecchymoses (bruising) and petechiae are caused by decreased vascular strength.17
Glucocorticoids alter the response of connective tissue to injury by inhibiting collagen synthesis,143 which is why these agents are used to suppress manifestations of collagen diseases. Clients who are taking steroids experience delayed wound healing with decreased wound strength, inhibited tissue contraction for wound closure, and impeded epithelization.
Steroid-Induced Myopathy.: In high doses, glucocorticoids can cause muscle weakness and atrophy called steroid-induced myopathy. Enzymes of gluconeogenesis are activated, leading to the breakdown of muscle protein, resulting in muscle wasting and atrophy severe enough to interfere with daily function and activities. Histologically, there is an increased variation in size and atrophy of muscles fibers, particularly type IIb.39
Steroid-induced myopathy is often insidious, appearing as painless weakness weeks to months after the initiation of treatment. There is no special or definitive test to make the diagnosis of myopathy. EMGs are often normal and muscle biopsies are nonspecific.
Clients present with bilateral atrophy and weakness of proximal muscles; the pelvis, hips, and thighs are typically affected first.98 Upper limb muscles can be affected; occasionally distal limb muscles are involved. The diaphragm may also be involved, which results in difficulty breathing, especially in people with underlying pulmonary disease.
Recovery from chronic myopathy (with cessation of drug) is possible with reduction or discontinuation of the drug but may take from 1 to 4 months up to 1 to 2 years. Prognosis depends on the underlying diagnosis before treatment with corticosteroids (e.g., organ transplantation requiring long-term administration of glucocorticoids). Four functional classifications of muscle weakness can occur in people with steroid-induced myopathy (Table 5-5).
Table 5-5
Functional Classifications of Corticosteroid-Induced Myopathy
| Level | Function |
| Advanced | Person has difficulty climbing stairs. |
| High | Person cannot rise from a chair. |
| Intermediate | Person cannot walk without assistance. |
| Low | Person cannot elevate extremities or move in bed. |
Modified from Askari A, Vignos PJ, Moskoweitz RW: Steroid myopathy in connective tissue disease, Am J Med 61:485-492, 1976.
Effect of Steroids on Growth.: Long-term use of glucocorticoids in children causes apoptosis of the chondrocytes at the epiphyseal plate, leading to growth retardation. Although there is an increase in bone synthesis once the drug is discontinued, full height may not be achieved.173 In adults, prolonged use of glucocorticoids inhibits bone mineralization, induces apoptosis of osteoblasts, and encourages osteoclastic activity. There is also decreased GI calcium absorption and increased calcium excretion by the kidneys. These combined changes result in osteoporosis.143 Strategies should be in place before extended therapy of glucocorticoids (greater than 3 months) to avoid bone loss.
Long-term exposure to corticosteroids increases the risk of avascular necrosis, which often requires orthopedic intervention (e.g., total hip replacement). Glucocorticosteroids are also associated with a fourfold to fivefold increase in the prevalence of vertebral fracture compared with individuals who are not treated with corticosteroids.149
These medications promote gluconeogenesis in the liver and lipolysis resulting in hyperglycemia. Individuals already requiring oral diabetic agents or insulin frequently need an increase in their dosage. Persons at risk for diabetes (e.g., glucose intolerance) may require a diabetic agent. Glucose monitoring is essential.
For clients with asthma, long-term treatment with inhaled glucocorticoids is common. Glucocorticoids decrease inflammation and aid in counteracting the vasodilatation caused by β2 agonists. Researchers initially hoped that the inhaled delivery of the glucocorticoids would eliminate or significantly reduce the side effects of the glucocorticoids; but bone loss and other adverse effects remain problematic in people with asthma using inhaled steroids.91
Animal studies suggest that glucocorticoids may play a role in the development of diaphragm dysfunction in anyone with pulmonary impairment. Consistent with the previous discussion of glucocorticoids causing muscle weakness and atrophy, these drugs can cause a decrease in the force generation of the diaphragm. Physical therapy intervention may be helpful in counteracting this glucocorticoid-induced muscle dysfunction.53
The GI effects of steroids are fewer than NSAIDS, yet they are known to cause gastritis, esophageal irritation, GI bleeding, and less commonly, peptic ulcers. Many clients take both glucocorticoids and NSAIDS, increasing their risk for adverse GI events (e.g., ulcer with perforation). For these individuals, a GI protective agent (e.g., proton pump inhibitor or misoprostol) may be beneficial.
Glucocorticoids are also known to cause cataracts, both cortical and posterior subcapsular. Cataract formation is dependent on dose and duration of use. They typically develop bilaterally but slowly. Clients with a history of glaucoma and taking glucocorticoids long-term may have an increase in pressure while taking glucocorticoids, making pressure checks advisable.
Because glucocorticoids cause adrenal suppression, withdrawal must be slow and tapered to allow for endogenous hormones to be produced by the adrenal cortex. Severe adrenal insufficiency may follow sudden withdrawal of the medication, particularly in the presence of infection or other stress. The person may experience vomiting, orthostatic hypotension, hypoglycemia, restlessness, arthralgia, anorexia, malaise, and fatigue. These symptoms should be reported to the physician.
Designer Glucocorticoids
As discussed previously, classic glucocorticoids bring about their antiinflammatory function by modulating gene transcription and inhibiting the production of proinflammatory factors (transrepression). Their adverse metabolic, cardiovascular, and behavioral effects, however, are believed to be related to the activation of genes and enzymes (transactivation).
New glucocorticoids are in development that would selectively enhance the antiinflammatory aspects of the drug while selectively activating transcription to decrease or avoid the adverse side effects, although there are many overlapping factors. These glucocorticoids are referred to as selective glucocorticoid receptor agonists (SEGRAs).17,150 Studies and further trials will ultimately verify the use of these new agents.
Anabolic-Androgenic Steroids
Anabolic-androgenic steroids (AASs), anabolic steroids, or “roids” are synthetic derivatives of the hormone testosterone. They are most commonly used in a nonmedical setting to develop secondary male characteristics (androgenic function) and to build muscle tissue (anabolic function).14-16 The use of anabolic steroids to enhance physical performance by athletes has been declared illegal by all national and international athletic committees. Even so, an estimated one million individuals in the United States alone are current or past nonmedical users of AASs. Administration of these compounds can be orally, intramuscularly, or by injection.
Recently 500 AAS users who frequented AAS internet sites were questioned about their habits. Ninety-nine percent stated they most frequently injected the steroids and 13% used unsafe needle practices.128 One survey reports the average age at first-time use is 14 years with a significant number of children (15%) taking an AAS before the age of 10.160
Studies indicate that adolescent AAS users are significantly more likely to be males and to use other illicit drugs, alcohol, and tobacco.8 Previously, more athletes were found to use AASs than nonathletes to enhance their sport.10 However, questions are being raised as to the percentage who now use AASs for cosmetic reasons alone.128 The goal is to advance to a more mature body build and enhance the masculine appearance.
The use of this type of steroid is illegal and potentially unsafe, unless given under the direction of a licensed physician; most of these drugs cannot even be prescribed legally but are still obtained from other athletes, physicians, and coaches.160
Athletes tend to take doses that are 10, 100, or even 1000 times larger than the doses prescribed for medical purposes. They cycle the drugs before competition, a technique known as stacking, alternately tapering the dosage upward and downward before a competitive event. Human growth hormone has been used alone and in combination with anabolic steroids to further enhance athletic performance.
Nearly all users of AASs report side effects. The most common include an increase in sexual drive, acne vulgaris (Fig. 5-2), increased body hair, and an increase in aggressive behavior. Individuals may also exhibit an increase in low-density lipoproteins and a decrease in high-density lipoproteins, complicating atherosclerosis and coronary artery disease.81
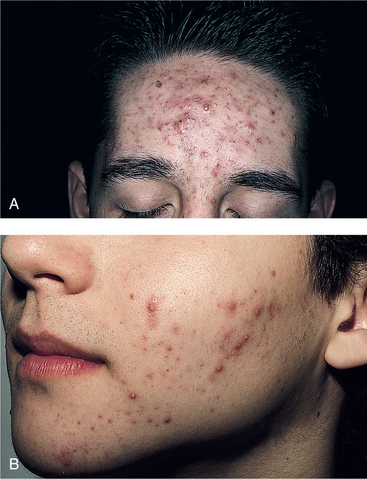
Figure 5-2 Acne vulgaris on the forehead and lower face associated with the use of anabolic steroids. It is considered an abnormal response to normal levels of the male hormone testosterone. The face, chest, back, shoulders, and upper arms are especially affected. There are many other causes of this form of acne; its presence does not necessarily mean the individual is using anabolic steroids. (From Callen JP: Color atlas of dermatology, ed 2, Philadelphia, 2000, WB Saunders.)
Misuse of supraphysiologic doses of AASs for nonmedical reasons has been linked with serious side effects such as hypertension, left ventricular hypertrophy, myocardial ischemia, peliosis hepatis (liver tissue is replaced by hemorrhagic cysts), and sudden and premature death.148,163
Users of anabolic steroids may experience an increased susceptibility to tendon strains and injuries, especially biceps and patellar tendons, because muscle size and strength increase at a rate far greater than tendon and connective tissue strength. Adolescent steroid use may lead to accelerated maturation and premature epiphyseal closure.162
Homicides, suicides, poisonings, and other accidental deaths associated with AAS use have been attributed to impulsive, disinhibited behavior characterized by violent rages, mood swings, and/or uncontrolled drug intake.133,167 Shared use of multidose vials, dividing drugs using syringes, and increased sexual risk-taking behavior are risk factors associated with AAS use and are potential routes for HIV and hepatitis infection.106
There are, however, legitimate medical uses for anabolic steroids that have come about as a result of physiologic evidence that anabolic steroids prevent loss of lean body mass. Oxandrolone, a synthetically derived testosterone, is approved as an adjuvant therapy to promote weight gain after weight loss secondary to chronic infections (HIV wasting), severe trauma (severe burns), and extensive surgery and to relieve bone pain associated with osteoporosis.42-44,60
Another anabolic steroid with a practical use is Anadrol. It is indicated for the adjuvant treatment of anemia secondary to a lack of red blood cell (RBC) production such as occurs in acquired and congenital aplastic anemia and myelofibrosis. Because of the side effects and drug interactions, these agents should be used with caution.
Other areas of testosterone use include in premenopausal women with a loss of libido and in aging men to prevent loss in muscle mass and strength.16,61,75 The adverse effects of these steroids, however, make long-term use inadvisable, particularly in the doses often required for efficacy.
RADIATION INJURIES
Definition and Overview
Radiation therapy, or radiotherapy, is the treatment of disease (usually cancer) by delivery of radiation to a particular area of the body. Radiation therapy is one of the major treatment modalities for cancer used in approximately 60% of all cases of cancer (see Chapter 9). Radiotherapy is used in the local control phase of treatment but has both direct and indirect toxicities associated with its use. Radiation reactions and injuries are the harmful effects (acute, delayed, or chronic) to body tissues of exposure to ionizing radiation.
Today, a pencil-thin beam of radiation can be targeted to deliver extremely high doses of radiation to within a millimeter of a cancer site. Advanced computer technology creates a three-dimensional model of the tumor to allow target mapping. Careful preplanning and delivery of targeted, modulated radiation doses have contributed to a reduced number of radiation side effects.
Etiologic and Risk Factors
Risk factors for developing radiation toxicities arising from therapeutic radiation are often multifactorial, depending on the organ radiated, individual variations and tolerance, tumor type, volume radiated, and fraction size (Table 5-6).
Table 5-6
Factors Contributing to Radiation Toxicity
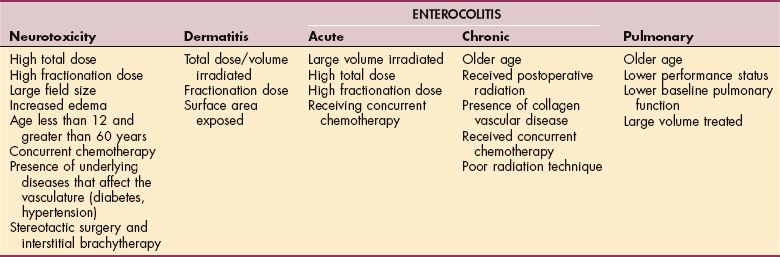
Data from Cross NE, Glantz MJ: Neurologic complications of radiation therapy, Neurol Clin 21(1):249-277, 2003; Hymes SR, Strom EA, Fife C: Radiation dermatitis: clinical presentation, pathophysiology, and treatment 2006, J Am Acad Dermatol 54(1):28-46, 2006; Nguyen NP, Antoine JE: Radiation enteritis. In Feldman M, Friedman LA, Sleisenger MH, editors: Sleisenger & Fordtran’s gastrointestinal and liver disease: pathophysiology, diagnosis, management, ed 7, Philadelphia, 2002, WB Saunders; Machtay M: Pulmonary complications of anticancer treatment. In Abeloff MD, Armitage JO, Niederhuber JE, et al, editors: Clinical oncology, ed 3, New York, 2004, Churchill-Livingstone.
People may also be exposed to radiation found in the environment, such as radon in their homes, or when rare nuclear events release large amounts of radioactivity, exposing people to total body irradiation (TBI). Bone marrow transplant clients may receive TBI as a preparative regimen.
According to the seventh report on the Biological Effects of Ionizing Radiation (BEIR VII) issued by the National Academy of Science, exposure to even low-dose imaging radiology (including computed tomography [CT] scans) can result in the development of malignancy. Exposure to medical x-rays is linked with leukemia, thyroid cancer, and breast cancer. There is a 1 in 1000 chance of developing cancer from a single CT scan of the chest, abdomen, or pelvis. The latency period for leukemias is 2 to 5 years and 10 to 30 years for solid tumors.114
Pathogenesis
Radiation therapy uses high-energy ionizing radiation to kill cancer cells. Irradiation is an effective treatment for cancer because it directly destroys hydrogen bonds between DNA strands within cancer cells. This prevents ongoing cellular replication. Although cells in all phases of the cell cycle can be damaged by radiation, cells in G2 and M phases (see Fig. 9-3) have the greatest sensitivity to radiation, making rapidly dividing cells most likely to be damaged.
Ionizing radiation interacts with nuclear DNA directly or indirectly to inhibit replicating capacity, resulting in apoptosis (programmed cellular death) or cellular necrosis. The radiation causes the breakage of one or both strands of the DNA molecule inside the cells, thereby preventing their ability to grow and divide.
Ionizing radiation also causes the production of free radicals (see discussion of free radicals Chapter 6), which leads to membrane damage and breakdown of structural and enzymatic proteins, resulting in cell death. Often arterioles supplying oxygenated blood are damaged, resulting in inadequate nutritional supply, leading to ischemia and death of the irradiated tissues. The damage to nucleic acids may result in gene mutations, possibly leading to neoplasia years later (see the section on Mechanisms of Cell Injury in Chapter 6).
Clinical Manifestations and Medical Management
The clinical manifestations of radiation, similar to risk factors associated with radiation therapy, depend on individual variations, location and type of tumor, radiation volume and fraction dose, and organ system involved. Although newer techniques allow for organ shielding and lower volumes and fraction doses, radiation therapy continues to cause symptoms and injuries.
Each organ has its own tolerance to radiation, therefore injuries vary between organ systems. Yet there are some general principles that encompass radiation therapy injuries. Most organ systems exhibit both acute injuries that occur within 30 days of irradiation and delayed injuries that occur more than 30 days later (Table 5-7). Acute injuries are frequently self-limiting, whereas delayed effects are often irreversible and difficult to treat. Acute symptoms may delay further radiation treatments because of damage to GI mucosa, bone marrow, and other vital tissues.
Table 5-7
Immediate and Delayed Effects of Ionizing Radiation*
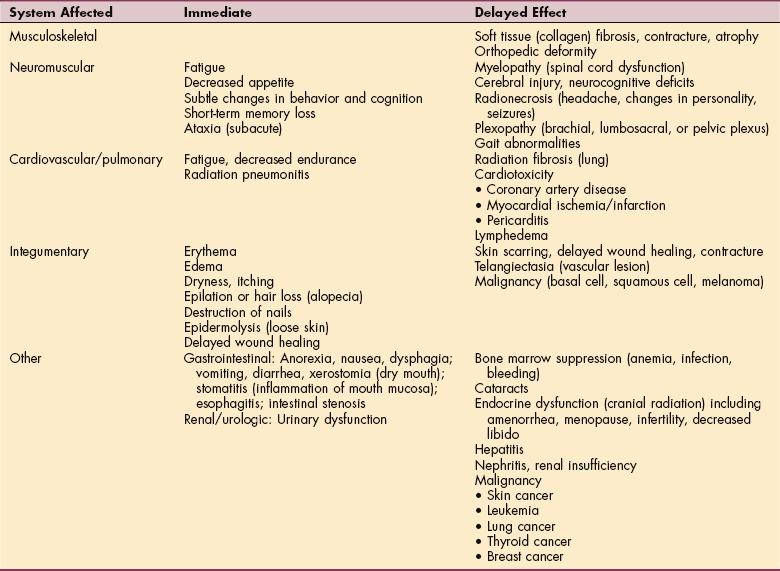
*Some of the delayed effects of radiation (e.g., cerebral injury, pericarditis, pulmonary fibrosis, hepatitis, nephritis, gastrointestinal disturbances) may be signs of recurring cancer. The physician should be notified of any new symptoms, change in symptoms, or increase in symptoms.
Management of acute injuries is often treated symptomatically with RBCs and platelet transfusions, antibiotics, fluid and electrolyte maintenance, and other supportive medical measures as needed. With prognosis poor in many cases of delayed radiation complications, more effort is being placed on prevention. Clinicians have been attempting to optimize total dose, fractionation size, and total volume being radiated.55,102
Modifications are made when chemotherapy is used in conjunction with radiation and prophylactic medications are under investigation to prevent damage or complications.86,140,170,189 Researchers are also developing more targeted therapy, which would increase tumor sensitivity while reducing damage to adjacent, normal tissue.4
Because there are unique or specific injuries to different organ systems, the following section specifies clinical manifestations, treatment, and research pertaining to different organ systems.
Radiation Esophagitis and Enterocolitis.: The esophagus is a centrally located organ in the mediastinum and is often involved in the radiation fields under treatment for lung cancer. An acute reaction may occur within 2 to 3 weeks after the initiation of radiation therapy manifested by abnormal peristalsis activity, odynophagia (pain with swallowing), and dysphagia (difficulty swallowing).
Although uncommon with radiation alone (1.3% for lung cancer), clients are more likely to develop severe esophagitis after the use of a chemosensitizer (14% to 49%),180 requiring hospitalization for tube feeding, pain control, and hydration. The medication amifostine, presumed to be a free radical scavenger, may be helpful in reducing the frequency and/or severity of esophagitis.181 Resolution of symptoms typically occurs 1 to 3 weeks after completion of radiotherapy. Late esophagitis is a result of inflammation and fibrosis of tissue, causing stricture and fistula formation. Dilation and surgical repair may be necessary.182
As in other organs receiving radiation, the intestines exhibit both acute and chronic symptoms of radiation treatment. Acutely, the rapidly dividing stem cells located in the crypts of Lieberkühn are induced into apoptosis, or programmed cellular death. This increased rate of stem cell loss contributes to acute radiation enteritis, reducing the surface area required for nutrient absorption and leading to dehydration and malnutrition.
Intestinal motility also changes, causing diarrhea, abdominal cramping, and nausea. If the terminal ileum is included in the radiation field, there may be a reduction in the absorption of B12 and bile acids leading to B12 deficiency and steatorrhea, respectively.118 Abnormal intestinal motility can occur after the first treatment, but symptoms become most pronounced around the third week of treatment.
Dehydration may require hospitalization and a break in the radiation schedule but is usually not life-threatening. Concurrent chemotherapy causes an increase in cellular damage when compared to radiation alone, with resulting neutropenia leading to serious infections and sepsis.
The incidence of chronic radiation enteritis is unknown and probably underreported but may occur in up to 15% of cases involving the intestines. Symptoms are insidious, occurring 6 months to 25 years after treatment.118 Unlike acute radiation symptoms, chronic symptoms often require treatment or surgery with more serious outcomes.
Radiation frequently causes fibrosis of tissues that may lead to strictures in the intestines, bowel obstruction, fistulas with abscess formation, ulceration with bleeding, and malabsorption. Intracavitary implants can also cause rectal damage.
Preventive strategies under investigation include free radical scavengers, antioxidants, and use of cytoprotective agents.
Radiation Heart Disease.: Radiation to the chest can cause pericarditis, coronary heart disease, and myocardial disease. People at greatest risk for radiation-induced heart disease include those who received mediastinal radiotherapy for breast cancer, testicular cancer, or Hodgkin’s disease before associated risks were known and radiation regimens were changed. Radiation-induced aortic valve disease has been reported in people who had radiation of the mediastinum for Hodgkin’s disease 20 to 30 years ago.82,88 Studies from survivors of atomic bombings suggest that the risk of mortality from radiation-induced heart disease is greatest when the dose is between 0 and 4 Gy and usually occurs at least a decade after irradiation.166,172
Radiation Lung Disease.: The lung is a radiosensitive organ that can be affected by external beam radiation therapy. Pulmonary toxicity is fairly uncommon and determined by the volume of lung radiated, the dose and fraction rate of therapy, concurrent chemotherapy, older age, lower baseline pulmonary function, and lower performance status.103
The two syndromes of pulmonary response to radiation are an acute phase (radiation pneumonitis) and a chronic phase (radiation fibrosis). Radiation pneumonitis is caused by significant interstitial inflammation creating a reduction of gas exchange. The hallmark of this toxicity is symptoms out of proportion to other findings, including appearance on radiographs. It usually occurs 2 to 3 months (range, 1 to 6 months) after completion of radiotherapy and typically resolves within 6 to 12 months.
Symptoms range from a dry cough with dyspnea on exertion to severe cough and dyspnea at rest.103 Rarely, clients may develop acute respiratory distress requiring intubation and ventilation. The National Cancer Institute has developed a grading system called Common Terminology Criteria for Adverse Events (CTCAE), version 3 (Grades 0 to 5), which does not recognize acute versus chronic symptoms but rather grades events such as aspiration or dyspnea. (This grading system is available on-line at http://ctep.cancer.gov/forms/CTCAEv3.pdf.)
Diagnosis of radiation pneumonitis can prove difficult if underlying disease, such as COPD, is present. Clients with grade 1 or 2 radiation pneumonitis respond well to corticosteroids, although their use can increase the risk for serious infection. Grades 3 and 4 radiation pneumonitis typically have a poor outcome.
Pulmonary radiation fibrosis may occur months after radiation therapy. Radiation fibrosis is progressive and symptoms may develop slowly. Only supportive therapy is available, such as oxygen supplementation, bronchodilators, and treating infection. Corticosteroids have no value, but studies are ongoing to find preventive measures.7,109,145
Other radiation-induced pulmonary problems include formation of bronchopleural fistulas, pneumothorax, hemoptysis, and bronchial stenosis.
Radiation Dermatitis.: Damage to the skin is one of the more common side effects of radiation since it is involved in most therapies, despite tumor location. While most injury to the skin is reversible, severe reactions can cause delay in therapy or a change in dosing. The cutaneous affects of radiation can be separated into acute, consequential-late, and chronic.
The National Cancer Institute has provided guidelines for grading acute cutaneous damage to the skin after radiation. Grade 1 reactions resemble sunburn and are accompanied by hair loss, dry desquamation, pruritus, dyspigmentation, and scaling (Fig. 5-3). These changes are secondary to damage of the hair follicles and sebaceous glands. The term desquamation is preferred to “radiation burn”; the latter term is unacceptable and no longer used in clinical practice.
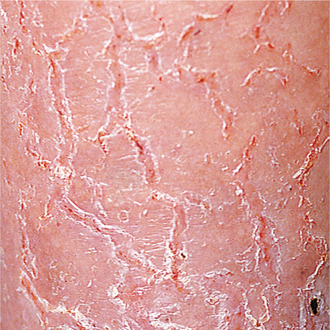
Figure 5-3 Dry desquamation with scaling associated with radiation. (From Habif TP: Clinical dermatology, ed 4, Edinburgh, 2004, Mosby.)
Grade 2 reactions produce persistent erythema or patchy moist desquamation in the folds and creases of the skin, often associated with pain and edema. Bullae may form, rupture, and become superinfected. These changes present 4 to 5 weeks into therapy and peak 1 to 2 weeks after treatment completion. Complete healing requires 1 to 3 months.89
Confluent, moist desquamation of the skin with pitting edema characterizes grade 3 reactions. Compared to grade 2 reactions, the edematous erythema of grade 3 is not confined to the skin folds. Grade 4 reactions (rare) are severe with skin necrosis or ulceration of full-dermis thickness associated with bleeding. Infrequently, grade 4 reactions do not heal and progress into consequential late effects, eliciting fibrosis with breakdown of necrotic tissue, ulceration, and exposure of underlying structures such as bone.51 These injuries are difficult to heal, since much of the tissue is avascular secondary to radiation.
While consequential late effects are persistent acute changes, chronic radiation-induced effects develop months to years after treatment. Repeated doses of radiation without sufficient time between doses to repair can lead to significant cutaneous injury. This injury is often manifested by atrophic skin, telangiectasia, hyperpigmentation, and hypopigmentation.
Sebaceous glands, hair follicles, and nails may be permanently affected. Fibrosis of the dermis accompanied by absorption of collagen creates contracted, atrophic skin, which is susceptible to tearing and ulceration (Fig. 5-4). An abnormal proliferation of arteriole cells may occur, causing thrombosis of the vessels, which combined with fibrosis, inhibits healing and predisposes ulcers to infection. These complex ulcers are painful and difficult to heal.
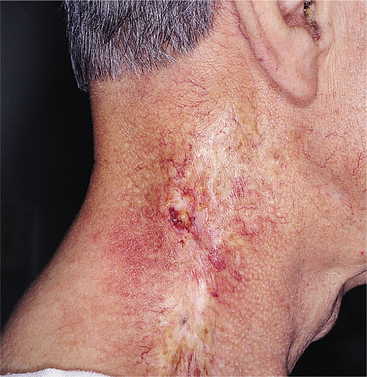
Figure 5-4 Radiation dermatitis. Acute or chronic inflammation of the skin caused by exposure to ionizing radiation (radiation therapy for cancer). Symptoms may include redness, blistering, and sloughing of the skin. The condition can progress to scarring, fibrosis, and atrophy as shown here. (From Callen JP: Color atlas of dermatology, ed 2, Philadelphia, 2000, WB Saunders.)
Treatment of acute cutaneous injury is typically symptomatic. For grade 1 and 2 reactions, washing with water or a gentle, low pH agent is sufficient to keep the skin clean and reduce bacterial load. Antiperspirants and talcum powders should be avoided in the radiation field. Ointments and creams can often benefit irritated and dry skin after radiotherapy.
Treatment of ulcers and erosions from radiation does not require specific therapy, but the same general principles of wound care apply (such as providing a moist environment, pain control, removing necrotic tissue, and protection against infection).89 Various dressings may be utilized, depending on the wound type (e.g., burn pads or foam dressings can be used for exudative wounds).
Other modalities that have been used include biosynthetic, artificial, and bioengineered skin; lasers151; and recombinant platelet-derived growth factor (PDGF).186 Diligence is required to keep fibrotic tissue intact. Active and passive range-of-motion exercises are important to retain mobility and reduce contractures.
The drug pentoxifylline41 and hyperbaric oxygen therapy (HBOT)58 have been used to enhance healing, and studies exploring prophylactic uses are ongoing. HBOT administers 100% oxygen under higher-than-atmospheric pressure, helping the body recognize damaged tissue and restoring up to 80% of the preinjury vascular density. High levels of oxygen in the tissues support healing by facilitating angiogenesis.
Another type of radiation-induced reaction is radiation recall. Radiation recall reactions (Fig. 5-5) are inflammatory reactions that occur in a previously irradiated site after the administration of certain chemotherapeutic drugs (e.g., dactinomycin, doxorubicin, bleomycin, methotrexate, gemcitabine, and paclitaxel) or antibiotics.96
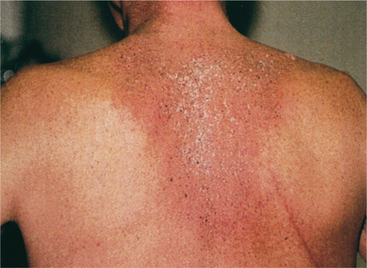
Figure 5-5 Radiation recall. This person had small cell cancer of the lung treated with radiation. Cytoxan treatment some months later elicited erythema and desquamation within the portal of radiation. This lesion is in the healing phase. (From Abeloff MD: Clinical oncology, ed 3, Philadelphia, 2004, Churchill Livingstone.)
Exposure to ultraviolet rays (e.g., tanning booths or outdoor exposure) is also a risk factor for radiation recall. Anyone undergoing radiation therapy is advised to use sunscreen and avoid exposure to ultraviolet A or B rays (which enhance the effect of radiation therapy) to prevent radiation recall.
Recall may occur in the skin, mucous membranes, lungs, central nervous system (CNS), esophagus, and GI tract, although the skin is most frequently involved.146 Months and even years may pass from the time of the initial radiation therapy to the onset of this reaction. A more immediate reaction (within 2 to 3 days) often occurs after the initiation of chemotherapy and is usually characterized by a mild, sunburned appearance. The skin may itch or burn; the reaction can last hours to days.
More significant reactions exhibit moist desquamation with blister formation and may even progress to full dermis necrosis and ulceration. Radiation recall involving other organs can be problematic but may respond to supportive measures, corticosteroids, or antiinflammatory medications.92
Effects of Radiation on Connective Tissue.: Radiation therapy, especially teletherapy, is well known to cause significant long-term or chronic effects on the connective tissue. Acute irradiation toxicity is less likely because connective tissue has a slower turnover or reproductive rate and striated muscle tolerates relatively high doses of radiation.
Late changes, such as fibrosis, atrophy, and contraction of tissue, can occur to any area irradiated but especially to collagen tissue. In growing bones and limbs, irradiation can cause profound and irreversible changes resulting in limb-length discrepancies and scoliosis requiring orthopedic surgical correction. Weakness of the bone may lead to pathologic fractures.
Fibrosis of connective tissue can result in edema, decreased range of motion, and functional impairment. Radiation of the pelvic cavity often causes dense pelvic adhesions that may cause painful motion restrictions and more rarely, plexopathy. Subsequently, these effects lead to soft tissue fibrosis, resulting in decreased range of motion, pain, and in some cases, lymphedema.
The fibrotic effect of radiation on the circulatory and lymphatic system is typically seen in a loss of elasticity and contractility of the irradiated vessels that are required to transport the blood, lymph, and waste products from the area of the body being exposed.9
Although lymphatic vessels maintain their structural integrity after being irradiated, fibrosis occurs in the surrounding tissue. This effect can inhibit normal growth of lymphatic vessels into healing tissues and delay lymphatic proliferation in response to inflammation.105a These types of effects can be minimized by sparing lymphatics from the radiation portal, but presently, up to 30% of breast cancer survivors in the United States develop lymphedema sometime in their lifetime.
It is important to remember that lymphedema may not be a side effect of radiation but rather a sign of advanced progressive metastases associated with cancer recurrence. Lymphedema can develop when lymphatic overload contributes to systemic congestion; a medical differential diagnosis is required.
Currently, physical therapy and supportive measures are the mainstay of therapy, although newer modalities and medications are under investigation.124
Effects of Radiation on the Nervous System.: As radiation therapy is used more frequently and aggressively in treating malignancies, toxicities to the nervous system increase. The incidence of nervous system toxicity related to radiation increases as the volume of nervous tissue being irradiated increases and the total dose and fraction size increase.32
Despite knowing risk factors for nervous system damage, toxicities continue to occur because individual reactions vary, safe thresholds are not known in all cases, and excessive doses may be used in an attempt to cure a malignancy. Studies determining the incidence of long-term effects of radiation are lacking but high total dose, high fractionation dose, and concomitant chemotherapy probably increase the risk.
Clinical manifestations of nervous system radiation toxicity can be separated into three categories: acute, subacute, and delayed. Neurologic symptoms relating to acute and subacute complications are most often self-limiting, requiring only supportive measures. The chronic or delayed complications are more often severe and progressive. Therapies for these complications are often palliative at best, although hyperbaric oxygen and anticoagulation have demonstrated questionable improvement.74,100,139,190
Acute Symptoms.: Acute symptoms generally occur during the period of treatment. The most common symptom is progressive and sometimes debilitating fatigue. Other clinical manifestations of cranial irradiation may include lethargy, short-term memory difficulties, and subtle changes in behavior and cognition. General symptoms that may occur during brain irradiation include decreased appetite, dry skin, hearing loss, hair loss, and decreased salivation.
Cranial radiation can often cause changes in short-term memory, cognition, and personality, ultimately leading to radiation-related encephalopathy or frank dementia.33 Clients infrequently demonstrate hydrocephalus accompanied by ataxia and incontinence.38,168
Acute radiation encephalopathy, probably related to edema, is an uncommon reaction secondary to brain irradiation, causing headache, nausea and vomiting, lethargy, seizures, new focal deficits, and mental status changes. Because of careful planning and use of the drug dexamethasone, the incidence has significantly decreased, although it can be life-threatening in clients that do develop this complication.
Subacute Symptoms.: Subacute symptoms (early delayed), noted 1 to 4 months after the completion of therapy, are fairly uncommon. If treatment included the cervical spine (and to a lesser degree the thoracic), clients may experience subacute myelopathy, a tingling, shocklike sensation passing down the arm or trunk when the neck is flexed (Lhermitte’s sign). This sign occurs in up to 15% of clients receiving mantle radiation therapy for Hodgkin’s disease.
Symptoms are usually self-limiting and peak 4 to 6 months after treatment. Irradiation of the brainstem may cause ataxia, nystagmus and dysarthria. Occasionally, a transient brachial plexopathy occurs, causing paresthesias and muscle weakness, which improves over time.
Delayed Complications.: While many of the acute and subacute complications of radiation are self-limiting or mild, late or delayed complications (late delayed) can be more serious and do not appear for months to years after therapy. For example, when exposed to radiation, cerebral vasculature, as well as other arteries, such as the carotid and coronary arteries, may be damaged, leading to coronary artery disease, transient ischemic attacks, stroke, or myocardial infarction.87 Other late effects are described as follows and in Table 5-7.
RADIONECROSIS.: One of the best-described complications of whole-brain radiotherapy is delayed cerebral radionecrosis. Symptoms occur in approximately 5% of persons who receive over 5000 cGy of cranial radiation and include headache, changes in cognition and personality, focal neurologic deficits, and seizures. Another serious long-term complication of radiotherapy of the brain is the development of tumors, including meningiomas, gliomas, lymphomas, fibrosarcomas, and malignant schwannomas. These tumors are often aggressive and difficult to cure.
Radiation of the brain may affect the hypothalamic system. Abnormalities in growth hormone, gonadotropins, thyrotropin, and corticotropin may be seen. Hyperprolactinemia commonly occurs and can resolve spontaneously.32 Hyperprolactinemia is a condition of elevated serum prolactin, which is an amino acid protein produced in the anterior pituitary gland. Its primary function is to enhance breast development during pregnancy and to induce lactation. Women present with changes in menstruation and infertility; men present with visual disturbances or headache.
MYELOPATHY.: Radiotherapy of the spinal cord may cause a radiation-induced myelopathy. This can present as the Brown-Séquard syndrome or as a motor neuron syndrome. The Brown-Séquard syndrome displays muscle weakness that ultimately leads to paraparesis or quadriparesis. Motor neuron syndrome is uncommonly seen after pelvic radiation (testicular cancer). Clients develop muscle weakness with atrophy of muscles, fasciculations, and areflexia. Sensory examination remains unchanged. This syndrome can progress over years before reaching a plateau.
PLEXOPATHY.: The brachial and lumbar plexuses may also be damaged after treatment. Clinical manifestations of radiation-induced brachial plexopathy include paresthesias with progressive motor deficits, lymphedema, and pain. Many clients lose hand function or develop arm paralysis.11,57
The incidence of brachial plexopathy after radiation therapy has been reduced significantly with improved treatment, but women who were given large daily fractions of postoperative telecobalt therapy to the axillary, supraclavicular, and parasternal lymph node regions years ago have shown a progression of both prevalence and severity of the late effects many years later, including arm paralysis.93
Today, with improved irradiation techniques (e.g., matching fields, maintaining the client’s position between fields, and avoiding overlapping fields that can cause hot spots), the overall incidence is about 0.5% of all cases of irradiated breast cancer. Plexopathies can be caused by cancer recurrence or new cancer onset rather than the effects of irradiation and must be differentially diagnosed by a physician.
Lumbar plexopathy is also possible when the pelvic area is irradiated. Clinical manifestations of radiation-induced plexopathy appear to be a result of fibrosis around the nerve trunks and include paresthesias, hypesthesia, progressive weakness, decreased reflexes, and pain.
Currently, no curative treatment is available for either brachial or lumbar plexopathies, although therapeutic interventions can achieve significant pain control and improve strength and function in the affected limb.78 Comprehensive strategies may be required in cases of lymphedema (see Chapter 13).
Pregnancy.: The fetus is very sensitive to radiation. Pregnant women or those who suspect they may be pregnant must avoid all possible exposure to sources of radiation. Congenital anomalies that develop after intrauterine exposure, especially if it occurs during early pregnancy or 2 to 12 weeks after conception during organ development, may include microcephaly, growth and mental retardation, hydrocephalus, spina bifida, blindness, cleft palate, and clubfoot. Later development of cancer, especially leukemia and thyroid cancer, is most often reported when the fetus is exposed to a source of radiation.175