The Female Genital/Reproductive System
The female genital or reproductive system is made up of the ovaries, fallopian tubes, uterus, vagina, and external genitalia (Figs. 20-1 and 20-2). The primary functions of this system are related to preparing the woman for conception and gestation (pregnancy) and for gestation itself. The female hormonal system, including the ovarian hormones, estrogen and progesterone, plays a key role in these functions. Malfunctioning of this system can result in a multitude of local disorders, some benign and others life-threatening, as well as widespread systemic changes via the hormonal influences.
The diseases discussed in this chapter have several implications for therapists. Some conditions, for example, pelvic floor disorders or postsurgical rehabilitation after mastectomy, may be the primary reason the woman is seeing the therapist. Other conditions, such as endometriosis, may be manifested solely by pain, and the woman presents to the therapist with back or pelvic pain of unknown cause.
The incidence of some of the disorders discussed in this chapter is quite high. For example, endometriosis is present in approximately 50% of women receiving treatment for infertility and in 10% to 15% of all premenopausal women. Breast cancer accounts for approximately one-third of all female cancers. Therapists frequently encounter disorders of this system.
The presence of some of these conditions places the woman at higher risk for developing other diseases. For example, a history of endometriosis increases the risk of ectopic pregnancy, a potentially life-threatening disorder. Menopause is known to contribute to disorders of aging such as osteoporosis, cardiovascular diseases, and cancer.
Therapists are now involved in the field of urogynecology with specific evaluation and assessment tools, treatment modalities, and therapeutic interventions. Increasingly, therapists are involved in the management of conditions such as vaginismus, dyspareunia, vulvodynia, sexual dysfunction, pelvic pain, levator ani syndrome, endometriosis, organ prolapse, and incontinence.
Therapists’ participation in women’s health issues reflects the medical profession’s changing view of women. The traditional allopathic emphasis on women’s breasts and reproductive system (the “bikini view”) is shifting focus to a more holistic view of women as unique individuals during their reproductive years and beyond, both in health and in disease.
AGING AND THE FEMALE REPRODUCTIVE SYSTEM
The most significant age-related change associated with the female reproductive system is menopause, which is the permanent cessation of menses. The average age at which this event occurs is between 45 to 50 years, but changes begin as early as mid-30s for many women. The period of time leading up to complete menopause is referred to as perimenopause.
Perimenopause
Perimenopause may be referred to as “the change before the change.” Changes in the menstrual cycle; sleep disturbances; increased body temperature; anxiety, depression, or other mood changes; fatigue; and difficulty concentrating are just a few of the sporadic symptoms women may notice. Perimenopause varies greatly from one woman to the next. Some experience symptoms as early as 10 years before the cessation of the menstrual cycle, marking the period known as menopause. For others, it can occur just a few months before menopause. Most women report a 3-or 4-year period of time when symptoms gradually escalate.
Perimenopause is the physiologic reverse of puberty. Levels of reproductive hormones start to decline gradually with sudden fluctuations on a day-to-day basis, which accounts for some of the common symptoms of perimenopause. Not all women experience or notice symptoms during this time. Women commonly transition from having regular menstrual cycles to having irregular cycles before the final menstrual period. In late perimenopause, anovulation becomes more common, leading to skipped menstrual cycles. Vaginal dryness is a common symptom late in perimenopause.313
Menopause
Menopause signifies the permanent cessation of ovarian function and the end of a woman’s reproductive potential. The process of reproductive aging unfolds as a continuum from birth through ovarian senescence to the perimenopausal and menopausal transitions into the postmenopausal phase.45
The average women experiences menopause by the time she is in her early fifties, but there is great variability in this time frame. Premenopausal women who have a hysterectomy experience surgical menopause. About 1% of women experience premature menopause known as premature ovarian failure, otherwise defined as cessation of menstruation before age 40 years.
The gradual cessation of ovarian function is accompanied by reduced estrogen levels. During the reproductive years the primordial ovarian follicles, from which ova are expelled, steadily decrease in number. By middle age, the ovaries, which each held about 300,000 eggs at puberty, have resorbed or shed nearly all of them.267 Ovarian production of estrogen and progesterone decreases significantly when the number of these follicles approaches zero. The reduced level of hormones results in a cascade of events altering the physiology of multiple systems leading to reproductive senescence.67
Symptomatically, women may experience hot flashes, vaginal dryness, fatigue, anxiety, sleep disturbances, memory loss, reduced libido, mood swings, and irritability (Box 20-1). Changes in vaginal pH that accompany estrogen decline can also contribute to an increased number of bladder and vaginal infections in some women.
Hot flashes are unpredictable flushes of heat and excessive perspiration that frequently begin several years before menopause. They are thought to be caused by fluctuating estrogen levels but can also occur in association with thyroid disorders and infection or as a side effect of some medications such as raloxifene (Evista), which is prescribed for osteoporosis prevention. Hot flashes with profuse perspiration at night are called night sweats.
One or more of these symptoms occur during menopause and last into the postmenopausal years for some women. In addition, there is evidence of increased risk for developing depression, even among women who never had depressive symptoms before.55,56 The prevalence of urinary incontinence increases as women age, but the exact role of menopause remains unclear in this transition.313
Menopause is a significant transition period that has been associated with adverse changes in body composition and fat distribution, including loss of lean mass, increases in fat mass, and redistribution of fat from the periphery to the center.292
Estrogen has effects on various target organs in the body—not just the organs of reproduction. In fact, estrogen receptor sites are found throughout the body and in all organs of the body, including skin, blood vessels, bone, brain, heart, intestinal tract, and urinary bladder. Physiologic changes associated with declining estrogen include endometrial, vaginal, and breast atrophy; decreased thyroid function; hyperparathyroidism; and decreased renal function, insulin release, and response to catecholamines. A decline in estrogen triggers a decrease in serotonin. Since serotonin helps maintain sleep and decrease anxiety, a drop in serotonin adds to the episodes of anxiety and sleep disturbance and aggravates feelings of irritability, tension, palpitations, and chest discomfort.
Local changes of the reproductive organs also occur secondary to decreased estrogen levels. The myometrium, endometrium, cervix, vagina, and labia all become atrophic to some degree. In addition, the pelvic floor musculature loses strength and tone, which can contribute to the development of cystocele, rectocele, uterine prolapse, and stress urinary incontinence.
Although vaginal bleeding should stop with menopause, 80% of all gynecologic problems in women over age 60 years are related to postmenopausal bleeding (vaginal bleeding that occurs 1 year or more after the last period). Such bleeding can be a symptom of a life-threatening problem (e.g., cancer) or a benign condition (e.g., polyps) but always requires a medical evaluation.
Women receiving continuous combined hormone replacement therapy (ccHRT), which is estrogen in combination with progestin taken without a break, are likely to experience irregular spotting until the endometrium (vaginal lining) atrophies, which takes about 6 months. An evaluation is required only if bleeding persists or suddenly appears after 6 months. Those women taking sequential HRT (estrogen taken daily for 25 days each month with progestin taken for 10 or 12 days) normally bleed lightly each time the progestin is temporarily stopped.
Even though estrogen levels decline as women age, the ovaries continue to produce significant amounts of testosterone and androstenedione after menopause. As a result, levels of testosterone may not decline sharply at menopause for women whose ovaries are intact. Testosterone is a natural means of preserving bone and muscle mass and of alleviating menopausal symptoms, particularly the loss of libido.
Menopause and Hormone Replacement Therapy
Much has been written and debated about menopausal women and the need for HRT. With an estimated 1.5 million women each year (incidence) and a total of nearly 80 million women (prevalence) currently approaching or going through menopause, this issue will remain in the forefront of health care. Therapists do not evaluate the need for HRT or prescribe these medications, but clients frequently ask questions and seek additional educational information about this topic. Therapists themselves are often faced with making the decision about HRT use, either personally or in relation to a close family member. Although an exhaustive discussion of this issue is not the focus of this text, a brief summary is warranted in today’s health care environment.
The decision to use (or not use) HRT after menopause should be based on a thorough understanding of the risks and benefits of HRT and how that understanding applies to each woman based on her individual risks. Much remains unknown about the long-term effects of ccHRT, but studies have been ongoing and new information is available daily.278,327 At one time, health experts were convinced that estrogen would be essential for reducing heart disease in women; clinicians routinely offered it in HRT as part of a prevention plan. Data from the Women’s Health Initiative (WHI) resulted in a halt to the routine use of estrogen and progestin in combination (Prempro) in 2002 and estrogen alone (Premarin) in 2004. When compared with a placebo group, it was clear that hormone users were experiencing more breast cancer, heart disease, stroke, and blood clots. Estrogen replacement showed some benefit, but it was not enough to outweigh the risks.333
No longer proved effective in reducing cardiovascular disease or preserving cognitive function, HRT is prescribed most often for short-term relief of menopausal-related symptoms such as hot flashes and vaginal dryness (Table 20-1). Current best practice suggests hormone therapy should be prescribed at the lowest effective dose for the shortest possible time.255 More recently, new findings from the Nurses’ Health Study, which has followed more than 120,000 female nurses since 1976, suggest that starting hormone therapy earlier (within 4 years of menopause) may have a coronary benefit.115,189
Table 20-1
Advantages and Disadvantages of Hormone Replacement Therapy (HRT)
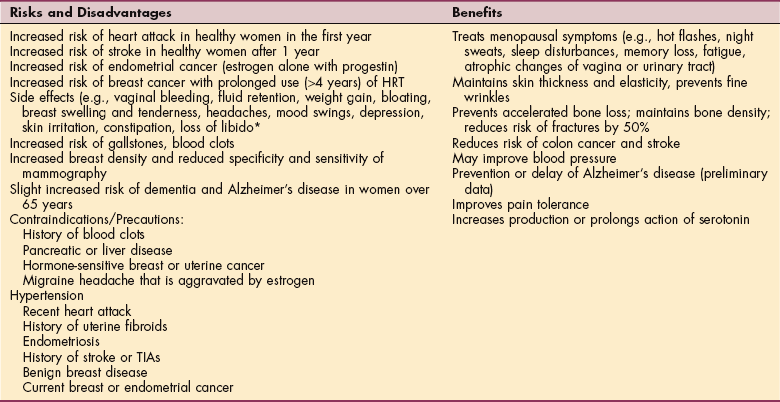
TIA, Transient ischemic attack.
*Some side effects can be managed with a reduced dose, different schedule, or changing brands. Any woman experiencing intolerable side effects should see the prescribing physician for evaluation.
Estrogen remains beneficial in preventing osteoporosis. Scientists are trying to determine the lowest dose possible to protect bone in a safe and effective manner. It is not clear yet whether observed improvements in bone mineral density actually translate into a reduced rate of fracture. Studies continue investigation to find out more about the long-term effects of HRT (both unopposed estrogen and combined estrogen/progestin) using different preparations and different ways of administration (e.g., dermal patches, oral, or vaginal) at different ages (early menopause versus late postmenopause).
The Kronos Early Estrogen Prevention Study (KEEPS), a 5-year randomized trial, will evaluate the effectiveness of HRT in preventing the progression of atherosclerosis in recently postmenopausal women.121 The National Institutes of Health (NIH) has funded another study, the early versus late intervention trial with estradiol (ELITE), comparing the effects of estrogen started early after menopause compared with estrogen initiated 10 or more years after menopause.203
Whatever decision a woman makes should be in conjunction with her health care provider, keeping in mind that any decision made must be individualized for each women and can be changed as new information becomes available.16 For the woman who cannot or prefers not to take long-term HRT, there are alternatives for preventing osteoporosis and heart disease and for modifying the transient and temporary symptoms of menopause. Drugs called selective estrogen-receptor modulators (SERMs) are being tested to determine their effects in preventing bone loss, improving serum lipids, and reducing breast cancer risk. Tamoxifen is the most widely used SERM in the treatment and prevention of breast cancer; raloxifene is currently used in the prevention and treatment of osteoporosis in postmenopausal women. These pharmaceuticals stimulate estrogen receptors in some tissues but not in others. SERMs have no effect on symptoms associated with menopause. Alternately, antidepressants can help some women with severe hot flashes. The importance of lifestyle measures, such as exercising; maintaining a low-fat, calcium-rich diet; and not smoking, has been clearly demonstrated.
Natural substances called phytoestrogens, such as black cohosh, dong quai, rose hips, soy products, flaxseed, and ginseng used in conjunction with alternative approaches (e.g., yoga, meditation, and acupuncture), are becoming increasingly popular, but research results remain limited at this time.
Menopause and the Musculoskeletal System250
Estrogen suppresses inflammatory responses in many tissues, including skeletal muscle. In animal studies, females have less muscle damage after exercise and in association with muscle diseases such as muscular dystrophy. There is also a greater activation of repair processes in female animals. Muscle injury and slower rates of repair in postmenopausal women may be linked to lower levels of estrogen.308,309
The musculoskeletal system is the site of several painful conditions that occur with increased incidence both perimenopausally and postmenopausally. These include Colles’ fracture, carpal tunnel syndrome (CTS), osteoarthritis of the basilar joint of the thumb, impingement and other rotator cuff diagnoses, and adhesive capsulitis.
Although menopause coincides with the appearance or worsening of fibromyalgia238 and many of the common arthritic conditions, it is also associated with the lessening of other conditions such as systemic lupus erythematosus, endometriosis, and migraine headaches. Hormonal changes associated with menopause may modulate these diseases and have either a beneficial or detrimental effect on the incidence and activity of several common problems.331
Menopause and Connective Tissue.: The question of why there is an increased incidence in the connective tissue disorders in perimenopausal and postmenopausal women has not been answered. In many research articles and clinical commentaries, the authors include brief thoughts or questions, usually in the discussion sections. The following are examples of some statements in research and clinical reviews found in the literature.
• Do fluctuations in female hormones play a role in the development of adhesive capsulitis, or is there a subtle, unrecognized autoimmune component of this syndrome?120
• Significant hormonal and physical attributes specific to women render them more susceptible (to CTS), but the specific causation is not yet clear.231
• The probable pathophysiology of this condition (i.e., deterioration of the palmar beak ligament) is known at least in part. The etiology of this deteriora tion, however, is not known. Is it pattern of use? Are there hormonal or lifestyle factors?231
• Differences in physiology between men and women, including hormonal effects on the connective tissues and decreased total muscle cross-sectional area, may play a role (in orthopedic conditions).195
• The association between the estrogen status and the collagen content of various structures is poorly defined at present. It is recognized that estrogen deficiency causes a loss of collagen from the skin, which results in skin thinning. Loss of collagen from around the urethra and within the trigone of the bladder may predispose to urinary dysfunction. The musculoskeletal aches and pains of which many postmenopausal women complain may be related to a loss of collagen from ligaments and other soft tissues.328
• Decreasing levels of anabolic hormones may be associated with musculoskeletal atrophy and decrease in function that are observed in older women.58
From the literature to date, it is clear is that the reason for the increased incidence of some musculoskeletal disorders in women is not clear. When one is faced with an unanswered question, the first step is to seek clues from what is known. For example, it is known that tendons, ligaments, and bones are composed of type I collagen and that type II collagen is a major structural component of cartilage. Cartilage contains estrogen receptors, and estrogen can influence inflammatory diseases by altering cell turnover, metabolism, and cytokine release.212
Other scientists have found decreased amounts of type II collagen in postmenopausal women with lumbar spine degeneration97 and decreased type I collagen in the first layer of endopelvic fascia of the pelvic floor (i.e., the arcus tendineus fascia pelvis [ATFP]) in premenopausal and postmenopausal women.210 There is an increase in degradation products of type II collagen in postmenopausal women when compared to age-matched premenopausal women.216 Additionally, the degradation products were lower in women placed on HRT. When the values for degradation products are lower, it signifies that the collagen is not being degraded or decreased in amount. The end result is that the collagen is preserved. The muscle fiber membranes (sarcolemma) may also be protected from damage associated with trauma or exercise by estrogen receptors located in the membranes.309 It follows then that the loss of estrogen may result in the loss of protection against muscle damage.
There may be a correlation between the somatic changes that occur with aging and a decrease in the activity of the hypothalamic-growth hormone (GH) axis.131 The decrease in muscle and bone mass in older individuals may indeed result from this GH deficiency.264
The implication of hormonal involvement in musculoskeletal disorders is supported by several investigations.210,242 The decrease in type I collagen found in the ATFP of women is not present when the women are on HRT. The decrease in force per unit of a cross-sectional area of the adductor pollicis muscle that occurs around the time of menopause did not occur in a sample of women taking HRT.242
Both quadriceps and hand-grip strength decreased 10% in women not taking HRT.113 In other words, women going through menopause had a decrease in collagen; those with exogenous hormones were not experiencing the same loss of collagen or loss of strength as those women who did not take HRT.
Colles’ Fracture.: Colles’ fractures, which are fractures of the distal radius, are seven times more likely to occur in women than men.236,280 The increased incidence of Colles’ fracture in women is explained by the decreased estrogen level that is a principal cause of low bone mineral density associated with postmenopausal osteoporosis.231,236 Low bone mineral density in premenopausal women is also a common finding in premenopausal women with Colles’ fracture.140,155
It has been reported that approximately two-thirds of the risk for all osteoporotic fractures in postmenopausal women is determined by the premenopausal peak bone mass.89 This fact stresses the importance of education of young women regarding methods to ensure the development of optimal bone mass. Additional factors include decreased balance, vision impairment, and regular walking, presumably reflecting the increased exposure to risk of falling.59,280
Carpal Tunnel Syndrome.: Medical diseases and physical conditions capable of causing CTS are common. For example, CTS has been found to be associated with menopause, hysterectomy, pregnancy, obesity, physical inactivity, and decreased physical fitness. Anything that can decrease wrist depth-to-width ratio can contribute to the development of this condition.
Women have smaller wrists (on average) compared to men so that any changes (e.g., ganglion, fibrous hypertrophy, or increased pressure) can result in compression of the nerve. Additional correlates of CTS include a prior Colles’ fracture, inflammatory arthritis, hypothyroidism, diabetes mellitus, and use of corticosteroids and estrogens100,157,222,223 (see Box 39-1). In the general population, approximately 3% of women and 2% of men will have the symptomatic consequences of CTS, with the greatest occurrence in women older than 55 years of age.15
Explanations of the proposed pathophysiologic dysfunctions of the syndrome have been outlined in the literature and include increased fluid in the tunnel space, square wrists,211 higher pressures within the carpal canal, the presence of a ganglion or a lipoma, and fibrous hypertrophy of the tenosynovium.164,222 Increased pressure in the carpal tunnel results in ischemia of the median nerve, which impairs nerve conduction and causes pain and paresthesia (see further discussion in section on Carpal Tunnel Syndrome in Chapter 39).
As a possible explanation for the trend of increased CTS incidence in women, there are now more women in the workplace and their occupations may require prolonged repetitive motions of the small muscles of the hand. However, studies show physical inactivity and being overweight are more reliable risk factors than job-related factors.27,222,224 The effect of workplace demands as a predictor of CTS is uncertain and remains under investigation.221
Participation in aerobic activity may relieve symptoms of CTS such as pain, clumsiness, and tightness.222 Improve ments of aerobic capacity and decreased body fat associated with improved circulation and oxygen delivery may (or may not) have the potential to prevent or reduce suffering from CTS.225, 220 Further study is needed to define the role of aerobic exercise and physical conditioning in the prevention and treatment of CTS.
Osteoarthritis of the Basilar Joint of the Thumb.: The risk of developing osteoarthritis of the basilar joint of the thumb has been reported to be 10 to 20 times higher for women compared to men.240 There is a well-established relationship between CTS and basal joint arthritis. Scientific evidence to explain the link between these two musculoskeletal conditions is lacking. Both disease processes primarily affect postmenopausal women. This may be a coincidence or there may be an as yet unknown explanation. An anatomic basis is also possible because the carpal tunnel is the narrowest at the level of the trapezium. Any changes, such as basal joint osteophytes protruding into the carpal tunnel, could contribute to CTS.105
Stability of this joint is required for a strong pinch and using a computer key board, and is also the basis for the increased fine motor control of this joint.231 Complete destruction of the joint examined postmortem has been reported in approximately 50% of postmenopausal Caucasian women.240 Proposed etiologies include both hypermobility and the deterioration of the palmer beak ligament, a major stabilizer of the joint.166,240
Adhesive Capsulitis.: Seventy percent of all cases of adhesive capsulitis are reportedly women. Additional risk factors include age greater than 40 years,120 trauma,253 diabetes,13 prolonged immobilization,114 thyroid disease,332 stroke or myocardial infarction,114 and the comorbidity of autoimmune disease.34
Menopause is often cited as a cause of adhesive capsulitis in women, but an earlier classic study seems to have ruled this out by demonstrating age to be the principal predictor. Women with earlier menopause do not experience adhesive capsulitis any earlier than their counterparts who experience menopause later.61,183
Radiographs are found to be normal and laboratory tests do not show typical inflammatory or autoimmune indicators. However, it is currently believed that this condition results from both inflammation and fibrosis of the synovium and subsynovium.312 Pathologic studies confirm the presence of an active process of hyperplastic fibroplasia and excessive type III collagen secretion that leads to soft tissue contractures of the surrounding structures (coracohumeral ligament, rotator interval soft tissues, subscapularis muscle, and subacromial bursae).
It has been shown that shrinkage of the axillary area of the capsule, a thickening of the glenohumeral ligaments, and a thick and restricted glenohumeral joint capsule occurs with adhesive capsulitis.202 These changes may occur in reaction to the abnormal presence of cytokines or lymphocytes.271
Impingement and Rotator Cuff Tendonitis/Tears.: Impingement of the cuff and biceps tendon on the acromion is more common in older women195 and can be a source of irritation and pain that may progress to rotator cuff tendonitis or tears of the rotator cuff.
Normally, the muscles of the rotator cuff serve as stabilizers of the glenohumeral joint. If the musculotendinous units of the rotator cuff lose the ability to depress and stabilize the humerus during movement, the contraction of the deltoid muscle may cause the humeral head to migrate in the superior direction and compress or impinge subacromial tissues.
Risk factors include degenerative processes at the acromioclavicular joint and structural abnormalities of the acromion that result in decreased subacromial space.195 Additionally, ligamentous laxity has been implicated in shoulder disorders.
EXERCISE AND THE REPRODUCTIVE SYSTEM
Exercise increases cardiovascular fitness, reduces adiposity, and aids in maintaining muscle mass and bone density in women of all ages from adolescents to frail older people. On the other hand, too much exercise can have negative effects on the reproductive and skeletal systems, including primary and secondary amenorrhea (absence of menstruation) caused by low body weight and improper nutrition (see further discussion in the section on Eating Disorders in Chapter 3).
The female athlete is at particular risk of reproductive system disruption with strenuous exercise. Hypothalamic dysfunction can result in delayed menarche (first menstruation) and disruption or cessation of menstrual cyclicity. When energy expenditure exceeds dietary intake, gonadotropin-releasing hormone (Gn-RH) is suppressed, resulting in hypoestrogenism and subsequent compromised bone density and infertility. Failure to attain peak bone mass combined with bone loss predisposes female athletes to osteopenia, osteoporosis, and stress reactions or fractures. Increasing caloric intake to offset high-energy demand may be sufficient to reverse menstrual dysfunction and stimulate bone growth, but many of these young women restrict calories.321,322
There is a known difference in the rate of male and female athletic injuries such as anterior cruciate ligaments injuries. No definitive explanation has been found. It is natural to assume the hormonal differences (including changes during the menstrual cycle and the use of hormonal contraceptives by women) between men and women is the most likely source for this difference in injury rate.4 Other factors under investigation include type of sport (e.g., soccer, basketball, or volleyball), contact versus noncontact, training, and anatomic and biochemical differences.3,12
Obesity also has significant consequences for the reproductive system and contributes to menstrual disorders, infertility, miscarriage, poor pregnancy outcome, impaired fetal well-being, and diabetes mellitus. Weight loss has marked effects on improving the menstrual cycle, promoting ovulation, and fertility. Fertility is improved through exercise and balanced nutrition possibly through changes in sensitivity to insulin.233
Mood disorders, including depression and postpartum depression, anxiety, and vulnerability to autoimmune and inflammatory diseases seem to follow estradiol (one of three estrogen compounds present in the body) fluctuations.54 Exercise has been shown effective in elevating mood, decreasing risk of cancer, and ameliorating some of these symptoms and conditions for most people.
Physical activity and exercise may be an effective strategy to offset some of the negative consequences of estrogen depletion on muscle mass and tissue distribution associated with menopause. Results of the Erlangen Fitness Osteoporosis Prevention Study (EFOPS) in early postmenopausal women suggests a program of mixed high-intensity exercise effectively compensates for most negative changes related to the menopausal transition.159
The results of this study showed that exercise can stop early postmenopausal bone loss, stabilize bone mineral density, increase physical fitness, and decrease coronary heart disease risk factors. Exercise had positive effects on the body composition of the women involved in the study and most importantly, the effects of exercise can be maintained with regular exercise over time.159
SEXUAL DYSFUNCTION
The participation of a physical therapist in the management of sexual dysfunction is an increasing phenomenon. More and more adults (men and women) are experiencing altered sexual function and sexuality as a result of medical treatment and medications. For example, being able to enjoy sex after cancer treatment is a significant and necessary part of the recovery process. Altered body image and hormones affect both men and women after treatment (surgery, drugs, radiation) for cancer of the reproductive organs. Menopause may be triggered prematurely in some women. An excellent resource about the side effects of radiation and chemotherapy on sexual desire and performance along with helpful suggestions is available.273
Other medical problems, such as diabetes, arthritis, depression, pelvic floor disorders, spinal cord injuries and other causes of neurologic impairment, menopause, prostate problems, and cardiovascular disease, can result in sexual dysfunction. Addressing these issues can have a positive effect on sexuality and quality of life. Most of these conditions can be managed with medications, lifestyle changes, and exercise. The therapist can be very instrumental in helping clients discuss sexual problems more readily and with less embarrassment and in helping them obtain the appropriate medical intervention, often including physical therapy intervention.
People who have been sexually victimized or traumatized are also seeking professional help for the first time. Sexual abuse occurs in approximately 1 of every 3 girls younger than 14 years and 1 of every 7 boys; only a small proportion of these cases are ever reported. Sexual abuse, marital (or partner) rape, and assault occur in the adult population; the prevalence of these events remains unknown, but social scientists estimate that 20% to 25% of the U.S. population is affected. Women with disabilities are at increased risk of abuse, including sexual violence.
There is an increased incidence of obesity, incontinence (urinary and fecal), fibromyalgia, infertility, pelvic pain, and behavioral components (e.g., eating disorders, obsessive-compulsive disorders, or learning difficulties) associated with sexual abuse.
DISORDERS OF THE UTERUS AND FALLOPIAN TUBES
Overview
Endometriosis is an estrogen-dependent disorder defined by the presence of endometrial tissue (lining of the uterus) outside of the uterus. The disorder becomes apparent in the early teen years after menses have begun, and its symptoms continue until menopause. Each month as the woman’s body prepares for a fertilized egg, the uterus becomes engorged with blood, providing a fertile place for the egg to attach and begin growing. If and when the unfertilized egg passes out of the body, the uterus sloughs off the lining of blood and the woman has a flow of menstrual blood for about 3 to 5 days.
Endometriosis occurs when the uterus sheds this blood up into the body, rather than down and out through the vagina. Endometrial tissue found outside of the uterus on other organs or structures within the pelvic cavity and the body responds each month the same way as the endometrium during the menstrual cycle. The misplaced tissue engorges with blood just as it would when lining the uterus. The blood cannot drain out of the body and the result is lesions, or “chocolate cysts,” wherever the endometrial tissue is located with subsequent swelling, bleeding, and scarring.70 These pockets of blood can be deposited anywhere in the body. The most common sites of ectopic implantation include the ovaries, fallopian tubes, broad ligaments, pouch of Douglas, bladder, pelvic musculature, perineum, vulva, vagina, or intestines (Fig. 20-3).
Although less common, endometrial tissue can also be found in the abdominal cavity, implanted on the kidneys, small bowel, appendix, diaphragm, pleura, and bony elements of the spine. Whereas it was once thought that the blood just reached the pelvic and abdominal cavities, coating the viscera contained within, it is clear now that endometrial tissue migrates throughout the body. It has been recovered from bone, lungs, and even the brain.103 Rarely, ectopic tissue has been found in joints, the nose, and the lungs.315
Wherever this tissue migrates, it is biochemically and endocrine active, behaving as if it were still under the control of the hormonal system. Every month during the menses, a woman with endometriosis develops a host of symptoms that depend on where the uterine tissue resides. During menstruation, the dislocated tissue is responding just as the uterine lining, but since it cannot shed as the endometrium does, it remains where it is, eventually forming scar tissue and irritating the affected area.
The American Society of Reproductive Medicine (ASRM) has classified endometriosis as I (minimal), II (mild), III (moderate), IV (severe), or V (extensive). Despite this classification system, a woman can have severe disease without symptoms or severe symptoms with minimal disease. This is most likely determined by the type of endometriosis and how biochemically active are the implants.
Incidence
The incidence of endometriosis has increased in the past 40 to 50 years in Western countries. Reports of incidence vary from as low as 7% to as high as 40% to 60% of all women. Endometriosis is found in up to 50% of all infertile women. Endometriosis affects women of all ethnic origins, socioeconomic backgrounds, and geographic locations.315
Etiologic and Risk Factors
Any woman of childbearing age is at risk of developing endometriosis, but it is more common in those who have postponed pregnancy. In addition, other risk factors include early menarche; regular menstruation but 27-day or shorter cycles; and menstrual periods lasting 7 days or longer. The cause of endometriosis is unknown, although the high prevalence among family members suggests a genetic predisposition. A daughter with a maternal history of endometriosis has twice the chance of developing endometriosis herself.
The most widely espoused theory suggests that endometrial cells are flushed into the pelvic cavity through retrograde menstruation, a condition in which some of the menstrual flow backs up the fallopian tubes into the pelvic cavity. This retrograde menstruation has been shown to occur in up to 90% of women, but it is unclear why endometrial cells implant in some women and not in others. Since the fimbrial openings of the fallopian tubes are in the posterior aspect of the pelvis, more of the endometrial implants are found on posterior structures, sometimes giving rise to low back pain.
Another strongly held hypothesis is a dysregulation or dysfunction of the immune system that allows these cells to locate and survive where they do not belong. The cells from the uterine lining are resistant to the body’s normal defense mechanisms and are not readily cleared away when they happen to stray outside the organ.
Other theories include the (1) dissemination of endometrial cells through the lymphatics or vascular system (explains presence of tissue in lungs); (2) metaplasia of the mesothelium (Meyer’s theory), that is, endometrial cells change from one type of cell to another, whereby the endothelium undergoes transformation able to produce the same reproductive hormones (explains presence of tissue in joints); (3) intraoperative implantation associated with procedures such as hysterectomy and episiotomy; and (4) abnormal differentiation of precursor epithelial cells during early embryology, whereby these cells are seeded before birth.
Pathogenesis
Once endometrial cells migrate to other parts of the body, they can form pockets of tissue referred to as implants. These implants swell in response to the cyclic surge of estrogen and progesterone-forming cysts on the underlying organs that contain a dark, syrupy fluid composed of old blood and menstrual debris called chocolate cysts.
There are three primary pathologic types of endometriosis: (1) red or petechial implants are the most active with the greatest capacity to produce prostaglandins (inflammatory mediators) and also capable of producing endometrial protein and hormones; (2) brown or intermediate implants are moderately active and precursors to powder burns; and (3) black or brown powder-burn implants are inactive with little cellular material but associated with adhesions that stretch organs and cause direct nerve damage through devitalization and ischemia. The powder-burn implants adhere structures together, contributing to infertility, and are sometimes referred to as a frozen pelvis. The severity of the disease depends on which one of these three types is present.
Clinical Manifestations
The symptoms and signs associated with endometriosis depend on the location of the implants, but pain and infertility are the two major symptoms. Abdominal pain, fatigue, and mood changes are common beginning 1 or 2 days before the onset of the menstrual flow and continuing for the duration. The therapist may hear reports of intermittent, cyclical, or constant pelvic and/or low back pain (unilateral or bilateral).
Dysmenorrhea (painful menstruation) will be the chief complaint if the implants are on the uterosacral ligaments. These lesions swell immediately before or during menstruation, resulting in pelvic pain. Dyspareunia (painful intercourse) is also associated with this condition because penile penetration during intercourse can aggravate the local adhesions.
Pain during defecation can occur when there are adhesions on the large bowel. The fecal material moves through the intestine, stretching and aggravating the scar tissue. Surprisingly, the extent of the disease does not always correlate with the intensity of the symptoms. A woman with widespread lesions may be asymptomatic, whereas a woman with few implants may have considerable pain.
Other symptoms can include low-grade fever; diarrhea; constipation; rectal bleeding; and referred pain to the low back/sacral, groin, posterior leg, upper abdomen, or lower abdominal/suprapubic areas. Bleeding from anywhere else (e.g., nose bleeds, coughing up blood, or blood in urine or stools) is less common but still possible.
MEDICAL MANAGEMENT
Although the classic triad of dysmenorrhea, dyspareunia, and infertility strongly suggests the presence of endometriosis, accurate diagnosis requires direct visual examination by laparoscopy or laparotomy. One advantage of laparoscopy is that the technique is also therapeutic in that lesions can be removed immediately. Ultrasound and magnetic resonance imaging (MRI) are generally used to examine the pelvis, but MRI is more sensitive in detecting the implants. Researchers are trying to develop a radioimmune assay to measure endocrine protein asso ciated or present with this disease toward the eventual development of a blood test.
TREATMENT.
There is no cure for endometriosis; the goals of medical treatment are preservation of fertility (if fertility is an issue) and pain relief. Pregnancy does appear to suppress the disease, and in animal studies, the implants disappear during pregnancy.315 Assisted reproduction may be recommended to stimulate ovulation and perform in vitro fertilization with transplantation of the embryo into the uterus.
Nonsteroidal antiinflammatory drugs (NSAIDs) may sufficiently relieve the pain, or other analgesics can be administered before or during menstruation. Other medications are used to inhibit ovulation and lower hormone levels to prevent the cyclic stimulation of the endometrial implants. Eventually, the implants will decrease in size. These medications include danazol, a combination estrogen-progesterone acetate; leuprolide (Lupron), which is injectable once per month into the muscle; goserelin, which is injectable under the skin, and nafarelin nasal spray, a Gn-RH (these analogs act on the hypothalamus-to-pituitary interface to shut down the ovaries by blocking the ability to produce gonadotropins such as follicle-stimulating hormone [FSH] and luteinizing hormone [LH]).
Danazol is a synthetic male hormone that inhibits the monthly surge of LH, reduces estrogen production, and influences the way estrogen affects endometriosis. Primary adverse side effects include weight gain, edema, decreased breast size, acne, oily skin, headache, muscle cramps, and deepening of the voice. It can also adversely affect lipid metabolism and raise blood pressure.
Birth control pills may be used to reduce painful symptoms and inhibit menstrual periods, which stop the growth of endometriotic implants, but these do not cause complete regression of implants already present. Once the woman goes off the pill, these implants become active once again, sometimes with a rebound effect (symptoms are much worse).
Surgical intervention is another approach that is used less commonly than even 10 years ago because the etiology remains unchanged and regrowth occurs rather quickly. If the endometriosis is mild without extensive adhesions, laparoscopic cauterization or laser surgery may be indicated. If the woman is over 35 to 40 years of age, disabled by the pain, and childbearing is completed, a total hysterectomy, bilateral salpingooophorectomy (removal of ovaries and fallopian tubes), and implant removal are considered.
Nontraditional therapies, such as yoga, aromatherapy, reflexology, naturopathic medicine, and homeopathy, may be useful adjuncts to allopathic medicine. Many women are using this type of alternative/complementary intervention combined with diet and nutrition to self-treat without medications. Numerous resources are now available in this area.18,68,83,206
The future treatment of endometriosis may be based on the development of remodeling enzymes that work to remodel tissue at the cellular level. Understanding the mechanisms of growth factors for the growth and development of epithelial cells, the immune system, and implant physiology will help researchers develop more specific intervention techniques.
PROGNOSIS.
As mentioned, there is no cure for endometriosis, although pregnancy and menopause appear to arrest its continued development. Endometriosis has been linked with reproductive cancers and melanoma.136,291 The link between endometriosis and these diseases remains unclear, although a genetic predisposition or shared exposures to environmental toxins (especially dioxins) have been suggested, but the findings are inconsistent and inconclusive.275,341
Uterine Fibroids
Uterine fibroids (benign tumors of the uterus) forming on the outer surface of the uterus or within the walls or lining of the uterus are common, presenting in up to one-quarter of all women of childbearing age and constituting the primary reason women have hysterectomies.
Clinical Manifestations
Usually, uterine fibroids are asymptomatic, but in 10% to 20% of women with these tumors, pain and abnormally heavy bleeding during or between menstrual periods are common. Often these women become anemic, experiencing fatigue and weakness that contribute to an impaired lifestyle.
Fibroids can and often do grow to the size of a grapefruit or larger. Growth is related to estrogen and possibly progesterone; fibroids often regress after menopause. Fibroids can place pressure on the bladder resulting in constipation, urinary frequency, and nocturia. Pressure on spinal nerves can also cause low back pain (Fig. 20-4).
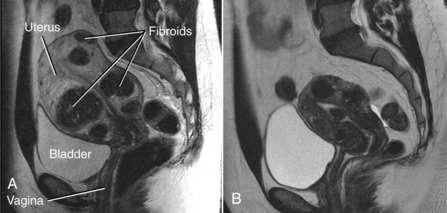
Figure 20-4 Uterine fibroids. A, MRI image of uterine fibroids. Note the position in relation to the sacrum, bladder, and pubic bone. Pressure on nerves and soft tissue in this area can cause painful pelvic, abdominal, low back, and sacral pain. B, MRI image of fibroids after uterine fibroid embolization (same patient). (Courtesy Robert L Vogelzang, MD, Chicago.)
MEDICAL MANAGEMENT
Pharmacotherapy to control fibroid-related symptoms may include NSAIDs for pain control or hormonal agents that lighten or stop the period. Other options may include surgical removal through a procedure called myomectomy. One way to perform this operation is to pass a fiberoptic scope through the vagina into the uterus (called hysteroscopy) to remove the tumors. Fibroids embedded in the uterine wall usually require a laparoscopy or more invasive open abdominal surgery. Hysterectomy (removal of the entire uterus) may be needed.
A new, less invasive technique, called uterine fibroid embolization (UFE), is performed with the woman under local anesthesia and mild sedation and involves the radiologist inserting a catheter into the femoral artery through a small incision in the groin and then snaking the catheter into the uterus. Tiny plastic or small sponge particles (polyvinyl alcohol) are injected that block off the blood supply to the smaller arteries supplying the fibroids, causing them to shrink and die. Another alternative to hysterectomy is endometrial (or balloon) ablation in which the uterine lining is destroyed (but not the uterus) through electrical energy or heat from a balloon-tipped catheter inserted into the vagina, through the cervix, into the uterus. The balloon is then filled with a sterile solution until it conforms to the shape of the uterus and heated until the heat destroys the endometrial tissue.
Eliminating red meat and ham from the diet and eating green vegetables, fruit, and fish appear to have a protective effect. Presumably, diet influences levels of the estrogen hormone, which is known to affect fibroid growth.53
Endometrial Carcinoma (Uterine Cancer)
Endometrial carcinoma is commonly known as uterine cancer, but technically the term uterine cancer refers to all cancers found in the uterus body and the cervix. Cancer of the lining of the uterus (endometrium) is the fourth most common cancer in women and the most common cancer of the female reproductive organs, accounting for approximately 7400 deaths per year in the United States.148 There is no apparent genetic component to endometrial cancer, but rather, environmental, social, and lifestyle factors are the most important.302
Risk Factors
Endometrial carcinoma is most common in women who are older (average age 60 years), white, affluent, obese, and of low parity. In fact, 75% of cases occur in postmenopausal women; the remaining 25% occur in premenopausal women, including 5% in women younger than 40 years. Hypertension and diabetes mellitus are also predisposing factors.
Any condition that increases exposure to estrogen unopposed by progesterone is a risk factor for uterine cancer. For example, obesity, polycystic ovary syndrome (PCOS), estrogen therapy, and some hormonal contraceptive formulations increase a woman’s exposure to unopposed estrogen and therefore increase the risk of endometrial cancer.
Tamoxifen (Nolvadex) therapy, estrogen replacement therapy without progestin, and the presence of estrogen-secreting tumors are also risk factors. Although tamoxifen is used as an antiestrogen treatment for breast cancer, in postmenopausal women with an intact uterus, it can enhance rather than suppress the action of estrogen. This action causes endometrial overgrowth, resulting in an increased incidence of uterine cancer in this population.
A great deal of attention has been paid to the possible induction of endometrial cancer by the antiestrogen tamoxifen, which has led to the development of new SERMs. The National Cancer Institute’s Study of Tamoxifen And Raloxifene (STAR) trials to compare these two drugs and their side effects are ongoing. The current data on raloxifene after 10 years continue to show a preventive benefit for breast cancer with less risk of uterine cancer compared to tamoxifen.
Cigarette smoking, physical activity and exercise, and the use of hormonal contraceptives appear to decrease the risk. In fact, women who exercise are 80% less likely to develop endometrial cancer than women who do not exercise at all.302 There is a strong link between obesity and endometrial cancer. Risk factors vary based on premenopausal versus postmenopausal status.
Pathogenesis
Epidemiologic and clinicopathologic evidence points to two separate types of endometrial cancer. Type I (low grade) is the most common type. It is hormonally related, associated with hyperplasia, and tends to have a better prognosis. Most of the risk factors listed refer to type I endometrial carcinoma.8 Type II endometrial cancer (high grade) accounts for approximately 10% of all uterine cancer. It is not hormonally related, is associated with endometrial atrophy, and has a worse prognosis.8
Clinical Manifestations
Unlike ovarian cancer, endometrial cancer has a major identifiable symptom in its early stages: abnormal bleeding (present in 80% of all cases). Irregular bleeding is a normal consequence of menopause, but the woman who is at least 12 months past menopause (cessation of menses) and now presenting with abnormal vaginal bleeding is the most typical presentation of endometrial cancer. Metastases to the lymphatic system can result in abdominal or lower extremity swelling.
MEDICAL MANAGEMENT
At the present time, the best prevention plan is to maintain a healthy weight through diet and regular physical activity. A plant-based diet rich in vegetables, whole grains, and beans is advised.173
DIAGNOSIS.
Abnormal bleeding in any woman of any age must be medically evaluated. Women with increased risk and those with postmenopausal bleeding or vaginal discharge should be screened for endometrial cancer. When metastatic spread occurs, the most common sites are lymph nodes, lung, or liver. More rarely, bone metastases with isolated lesions to the femur, tibia, fibula, and calcaneus may occur.186
Endometrial sampling is currently the most accurate and widely used screening technique, but ultrasonographic measurement of endometrial thickness and hysteroscopy have also been used. Staging has changed significantly over the last 25 years and is now determined surgically using the International Federation of Gynecology and Obstetrics (FIGO) classification (Box 20-2). For complete TNM and FIGO staging, see the National Comprehensive Cancer Network (NCCN) Clinical Practice Guidelines in Oncology: Uterine Cancer, available at http://www.nccn.org/professionals/physician_gls/PDF/uterine.pdf
Other less invasive methods of staging are under consideration such as laparoscopic-assisted vaginal hysterectomy with lymphadenectomy.19 Contrast-enhanced MRI may help decrease the number of unnecessary lymph node dissections.94
TREATMENT.
Endometrial cancer is usually treated surgically with total abdominal hysterectomy and bilateral salpingo-oophorectomy. The plane of excision lies outside the pubocervical fascia and does not require unroofing of the ureters.19
Progestin therapy may be used in those women who decline surgical intervention,39 and hormonal therapy (including progestin and antiestrogen tamoxifen for some women) has been used with recurrent disease. Most of these cancers are detected at an early stage when they are highly curable.
Women with advanced stages of endometrial cancer may not be candidates for operative intervention in the presence of tumor fixation or deeply invasive cancer. Medically inoperable cases may be treated with radiotherapy alone (external-beam pelvic radiotherapy or vaginal brachytherapy). Radiation may be used when the tumor spreads outside the uterus or in the case of advanced or recurrent disease after failed hormonal therapy. Cytotoxic combination chemotherapy also has been used with varying results.
PROGNOSIS.
Early detection makes this disease curable, but recurrences can occur; most recurrences occur within the first 3 years after surgery. Endometrial lining involvement of less than 50% is associated with 100% survival, but this drops precipitously when tumor growth involves more than one-half of the endometrium, especially with local or distant metastases.99
Cervical Cancer
Every year in the United States, approximately 11,000 women are diagnosed with cervical cancer and 3700 women die of this disease (288,000 worldwide deaths).148 Mortality has declined dramatically since the 1930s when the Papanicolaou (Pap) smear was introduced. It is now largely a preventable disease with preventive sexual practices, regular screening, and intervention at the precancerous stage. Twenty-five percent of new cases of cervical cancer develop in women 65 years and older.
Etiologic and Risk Factors
Clinical studies have confirmed that the transfer of human papillomavirus (HPV), also known as papillomas or genital warts, during unprotected sexual intercourse is the primary cause of cervical cancer. HPV is the most common sexually transmitted disease (STD) in the United States affecting more than 50% of sexually active adults. The CDC estimates that 7.5 million Americans become infected with genital HPV each year. A new study suggests HPV infection rates are higher than previously thought, and perhaps as many as one-third of all American women are infected.78
More than 70 types of HPV have been identified; 23 of these infect the cervix, and 13 types are associated with cancer. Infection with one of these viruses does not necessarily predict cancer, but the risk of cancer is increased significantly, and a link between HPV infection and female cervical cancer and male anal cancer has been demonstrated.128
Other risk factors include maternal use of diethylstilbestrol (DES), smoking (even passive smoking), hormonal contraceptive use, high parity (number of births), low socioeconomic status, ethnic background (black women experience a 72% higher incidence compared with whites),251 young age at first intercourse (17 years or younger), multiple sexual partners (5 or more),50 and the presence of other STDs.148
Alcohol and other drugs are additional risky behaviors that may play a role in young age of first sexual intercourse and more then five sexual partners. Impaired judgment from alcohol and other drug use can lead to unsafe sexual practices and with risky partners (partners more likely to have sexually transmitted infections [STIs]) contributing to HPV infection.151
Women infected with human immunodeficiency virus (HIV) are at increased risk for cervical intraepithelial lesions (the precursors to invasive cervical cancer), presumably associated with a high rate of persistent HPV infection.80 Other immunocompromised women (e.g., organ transplant recipients and women receiving immunosuppressants) are also at increased risk.
Pathogenesis
The common unifying oncogenic feature of the vast majority of cervical cancers is the presence of HPV. More than 99% of cervical cancers contain at least one high-risk HPV type (16, 18, 31, 45); approximately 70% contain HPV types 16 or 18.152 The molecular basis for oncogenesis in cervical carcinoma can be explained to a large degree by the regulation and function of the two viral oncogenes E6 and E7. The ability of HPV to target the function of tumor suppressors is typical of DNA tumor viruses. The E6 gene product binds to the p53 tumor suppressor gene and induces p53 degradation. E7 targets another tumor suppressor that functions like p53 in cell cycle control and inactivates it.147
As a result of these molecular disruptions, dysplastic changes occur in the thin layer of cells known as the epithelium that covers the cervix. The cells found covering the outer surface of the cervix are squamous (flat and scaly), whereas the cells lining the endocervical canal are columnar (columnlike).
Greater understanding of the behavioral and biologic mechanisms accounting for early age of first sexual intercourse and subsequent HPV infection in adolescents may help direct primary prevention of HPV infection and HPV-related disease. In addition to the risky behaviors discussed previously, it is now known that the cervix is particularly vulnerable to HPV during adolescence and especially early puberty.151 There are a number of potential reasons for increased vulnerability of the cervix. Cervical immaturity in the young girl and adolescent female is marked by inadequate cervical mucus, which acts as a protective barrier against infectious agents. Two other examples include cervical ectopy, which is characterized by rapid physiologic changes in the cervical epithelium, or immature immune response to HPV infection.151
Cervical ectopy refers to the condition in which a small ring of cells extend beyond the normal border of the endometrium (the inner wall of the uterus) to the cervical os (the neck of the uterus). Cervical ectopy is a normal physiologic phenomenon in women under hormonal influence, such as during puberty, but may increase cervical susceptibility to infection with STIs, including HPV.
The development of a protective cervical mucus is progressive through adolescence to full maturity. Until cervical maturity is reached, the woman remains at increased risk for HPV infection (and other STIs). Cervical immaturity is a risk factor for women who engage in sexual intercourse at a young age, including those who are sexually abused.151
Clinical Manifestations
Most people never know they have had HPV because there are no symptoms and a healthy immune system clears the body of infection. When it persists, HPV can cause lesions in the cervix, vagina, or vulva. Left untreated, these lesions can progress to cancer. Early-stage cervical cancer, especially in the preinvasive stage, is usually asymptomatic. Often women have advanced disease before abnormal bleeding occurs. This can present as spotting between menstrual periods, longer and heavier periods, bleeding after menopause, or bleeding after sexual intercourse. Pelvic or low back pain can occur, but this is uncommon.
More advanced stages of cervical cancer may cause bowel and bladder problems because of pressure on the rectum or bladder or sexual difficulties because of the growth in the upper vagina, causing discomfort. Ureter blockage can lead to death because of uremia (the inability of the body to excrete waste), which causes uremic poisoning. The progression to this type of advanced cancer is relatively rare in developed countries.
The physical effects of cervical cancer after treatment are actually more significant. Women who have the conization procedure or loop electrosurgical excision procedure (LEEP) may experience cramping, bleeding, or a watery discharge. Hysterectomy, radiation therapy, surgery alone or combined with radiation, and/or chemotherapy may all cause significant side effects, which should improve over time with proper intervention.
The emotional effects of cervical cancer are often significant as well. Women treated with radiation almost always lose the benefits of estrogen because the ovaries are extremely sensitive to radiation. HRT is usually prescribed, and without this intervention, the emotional effects of cervical cancer can be compounded by hypoestrogenism and its emotional side effects.
Sexuality after hysterectomy or other interventions for this cancer can be impaired; women may experience depression from no longer being able to have children; and some women may feel guilt and shame associated with feeling “unclean” because of genital tract disease.
MEDICAL MANAGEMENT
Risk of HPV and HPV-related cases of cervical cancer can be reduced and/or prevented using barrier contraceptives, engaging in monogamous sex with a likewise monogamous partner, or practicing sexual abstinence (see the section on Sexually Transmitted Disease in Chapter 8).84
Although not preventive, studies show that consistent condom use can speed the regression of the HPV-related lesions on the cervix and on the penis and shorten the time it takes to clear HPV infections.29,132 Women with five or fewer lifetime sexual partners have higher regression rates compared with women who have had more than five partners.50
The possibility of HPV infection among women who have sex with women (WSW) has been reported based on limited data.191,192 STDs can be spread between female sex partners, probably through the exchange of cervicovaginal fluid and direct mucosal contact.193
EARLY DETECTION AND SCREENING.
Early detection is the key to a 100% cure rate for cervical cancer. Routine cervical screening is recommended for all women regardless of sexual orientation or practices beginning approximately 3 years after the onset of vaginal intercourse but no later than age 21 years (Table 20-2).
Table 20-2
Cervical Cancer Screening Guidelines*
| American Cancer Society† | American College of Obstetricians and Gynecologists‡ | |
| When to start cervical screening | Approximately 3 years after beginning sexual intercourse, but no later than 21 years of age | Same |
| Frequency of Screening | ||
| Conventional Pap test | Annually; every 2-3 years for women older than 30 years with 3 negative cytology tests | Same |
| If liquid-based cytology | Every 2 years; every 2-3 years for women older than 30 years with 3 negative cytology tests | Annually; every 2-3 years for women older than 30 years with 3 negative cytology tests |
| If HPV testing used§ | Every 3 years if HPV is negative, cytology negative | Every 3 years if HPV negative, cytology negative |
| When to stop screening | Women older than 70 years with more than 3 recent, consecutive negative tests and no abnormal test in prior 10 years | Inconclusive evidence to establish upper age limit |
*Exceptions exist for women who are immunocompromised, have human immunodeficiency virus (HIV), or were exposed prenatally to DES.
†Data from Saslow D, Runowicx CD, Solomon D, et al: American Cancer Society Guideline for the early detection of cervical neoplasia and cancer, CA Cancer J Clin 52:342-362, 2002.
‡Data from Cervical Cytology Screening: AGOG Practice Bulletin No. 45, Obstet Gynecol 102:417-427, 2003.
§Wright TC, Schiffman M, Solomon D, et al. Interim guidance for the use of human papillomavirus DNA testing as an adjunct to cervical cytology for screening, Obstet Gynecol 103(2):304-309, 2004.
WSW should receive Pap smear screening according to the current guidelines. It should not be assumed that testing is not needed for those who use condoms consistently or for women who have not been in a sexual relationship with men.191,192 WSW should be educated about preventive measures including washing hands, using rubber gloves, and cleaning sex toys or using a protective barrier, such as a condom, especially when partners share such devices.193
Until age 30 years, annual screening is recommended with conventional cervical cytology. For women who have had three consecutive normal or negative cytology results, screening may be reduced to every 2 or 3 years. Average risk women aged 70 and older with an intact cervix may chose to stop cervical cancer screening if they have had no abnormal or positive cytology tests within the 10 years before age 70 years.285
Some experts advise that Pap screening can be discontinued in women who have had the cervix removed (e.g., in conjunction with hysterectomy). Others support annual testing for most women regardless of age and to detect vaginal cancer in women who have had the cervix removed.
Women with known HIV infection, HPV, or other STDs must especially be screened for cervical cancer.80 Women who have a history of cervical cancer or in utero exposure to DES should continue screening after age 30 years using the same protocol as for women before age 30 years.285
VACCINE.
The first cervical cancer vaccine (Gardasil) is now available; other HPV vaccines may be on the market soon (e.g., Cervarix). Gardasil blocks viruses that cause cervical cancer and protects against two strains of HPV believed responsible for 70% of cervical cancer cases and 2 viruses that cause 90% of genital wart cases.317 Ongoing studies are investigating the effectiveness of Gardasil in boys and men ages 16 to 26 years and in mid-adult women ages 24 to 45 years.
Currently, the vaccine is administered intramuscularly in a series over 6 months and costs between $300 and $400, which may make its use prohibitive as a routine vaccination. The vaccination is advised for females before becoming sexually active, but sexually active females can also benefit. Even if the woman is already infected with one or more of the four HPV strains covered by the vaccine, the vaccine will help protect her from the remaining strains.121a The vaccine can be given to females between ages 9 and 26.
The Advisory Committee on Immunization Practices (ACIP), which is part of the Centers for Disease Control and Prevention (CDC), has issued its first summary statement about recommendations for HPV vaccination.213 The interested reader can read the full summary at http://www.cdc.gov/mmwr/preview/mmwrhtml/rr56e312a1.htm?s_cid=rr56e312a1_e. The ACIP recommends routine immunization of girls aged 11 to 12. This is a controversial social issue because safe sex practices can prevent HPV and its associated complications, making universal vaccination unnecessary. Both the ACIP and the National Women’s Health Network advise that the vaccine should not replace routine cervical screening or education regarding sexual practices.7 Guidelines from the American Cancer Society (ACS) suggest routine vaccination for females aged 11 to 12 years, although girls as young as 9 years of age may receive the HPV vaccination. Universal vaccination for females aged 19 to 26 years is not advised.268
Each woman should make an individual decision regarding HPV vaccination based on her own risk and the potential benefit from the vaccination. Ideally, the vaccine should be administered before exposure to genital HPV through sexual intercourse because the potential benefit is likely to diminish with an increasing number of sexual partners.268
Vaccination is more imperative for women who do not have access to cervical cancer screening services. The protective effect of vaccination that is successfully provided to adolescent and young women who are unlikely to undergo regular Pap screening will yield greater positive outcomes than vaccination provided to women who will seek regular screening regardless.268
Vaccine trials in males are ongoing with results expected about the time this text is published. If efficacy is found among males, then vaccination may be recommended for males. It is not clear yet that vaccinating both males and females will provide any additional benefit in reducing HPV-cervical disease, and it may not be cost-effective.268
DIAGNOSIS.
Information is gathered from examinations and diagnostic tests to determine the size of the tumor, how deeply the tumor has invaded tissues within and around the cervix, and the metastases to lymph nodes or distant organs. Staging to find out how far the cancer has spread is an important process and the key factor in selecting the right treatment plan.
Cervical cancer is detected using a Pap test, and the Pap test is credited with reducing the incidence of this cancer in the United States by 75% over the past 50 years. This test is used to detect changes in the cells of the cervix that may indicate a precancerous or cancerous condition. However, Pap tests have a 15% to 25% false-negative rate for detecting cervical dysplasia279 and can be inconclusive, requiring further testing, including HPV testing, colposcopy, conization (cone-shaped biopsy), or LEEP.
New automated screening systems (PAPNET or AutoNet) have been approved by the U.S. Food and Drug Administration (FDA) for computer review of negative Pap smears and for primary screening. These systems have the ability to reduce human error, but their cost-effectiveness is under intense scrutiny. Numerous other cervical biomarkers are under investigation for possible diagnostic use.147
A new laparoscopic assessment of the sentinel lymph node in early-stage cervical cancer is under investigation with excellent preliminary results.66,187 Computed tomography (CT) scanning may be used in women who are not candidates for surgical.
STAGING.
There are four stages of cervical cancer with intermediate steps within each stage (Box 20-3). Staging of cervical cancer is based on clinical staging rather than surgical staging. This means that the extent of disease is evaluated by the physician’s physical examination and in some cases, a few other tests that are done such as cystoscopy and proctoscopy. Surgery may determine that the cancer has spread more than initially assessed. This new information may change the treatment plan, but it does not change the woman’s FIGO stage.
Stage 0 is the precancerous stage, and there are no gross lesions; carcinoma is limited to the mucosa and is referred to as carcinoma in situ (CIS) or cervical intraepithelial neoplasia (CIN) grade 1, grade 2, or grade 3. For premalignant dysplastic changes, the CIN grading is used for Stage 0. Grade 1 is most likely to regress naturally and does not require treatment; grade 3 is most likely to advance to cancer. Grades 2 and 3 both require treatment.
Stage I is strictly confined to the cervix, and lesions are measured as less than or greater than 4 cm in size. In stage II, the cancer extends beyond the cervix but has not extended to the pelvic wall. The vagina is minimally involved, and there may or may not be parametrial involvement.
In stage III the carcinoma has extended to the pelvic wall and involves the lower one-third of the vagina and there may be kidney involvement (spread via the ureters). Stage IV is characterized by carcinoma that has extended beyond the true pelvis or has infiltrated adjacent organs (e.g., mucosa of the bladder or rectum). There may be metastatic spread of the growth to distant organs.148
TREATMENT.
There is a concern that precancerous stages (CIN 2 or 3) may progress to invasive cancer so these are treated with cryotherapy, laser vaporization, excision (e.g., LEEP), cone biopsies, and possibly indoles (phytochemicals found in cruciferous vegetables, such as broccoli, cauliflower, and cabbage, when taken as a supplement).22,337
Some of these treatment methods can interfere with a woman’s ability to have children because weakening of the cervix makes carrying the child to full-term difficult. New findings suggest that some women with CIN 2 or 3 may not need treatment right away as the abnormality may go away on its own. The best candidates are women who have had five or fewer lifetime sexual partners who do not have HPV infection. Regression rates for CIN 2 and 3 in this group are over 60%.50
Concurrent cisplatin-based chemotherapy and radiation have shown significant survival improvement for women with locally advanced cervical cancer. The use of hypoxic cell radiosensitizers and monoclonal antibodies that inhibit cell growth increase numbers of malignant cells killed.259 More advanced cases are managed by surgery (i.e., hysterectomy) followed by radiation or chemoradiation therapy for high-risk or advanced stages.147 In addition to a vaccine and chemopreventive agents, biologic response modifiers are under investigation for future treatment options.
PROGNOSIS.
Cervical cancer is a slow-growing neoplasm with a good response rate to intervention. Almost all women with preinvasive cancer are cured. Reconstructive surgery and ovary preservation may be able to preserve childbearing status in younger women. The majority of treated women who develop recurrences do so in the first 2 years after primary therapy. Women with more advanced-stage disease or with lymph node involvement have a significantly less favorable prognosis.
Ectopic Pregnancy
Ectopic pregnancy, also known as tubal pregnancy, is marked by the implantation of a fertilized ovum outside the uterine cavity (Fig. 20-5). The fallopian tube is the most common site of ectopic pregnancy, with approximately 95% implanting there, but extrauterine pregnancies can occur anywhere outside the uterus (e.g., ovary, abdomen, or pelvic peritoneum). Ectopic pregnancy is a true gynecologic emergency, since accompanying complications are one of two primary causes of maternal death in the United States.
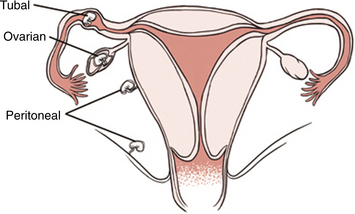
Figure 20-5 Ectopic pregnancy (outside the uterus) with implantation inside the fallopian tube (tubal pregnancy), abdomen (peritoneal or abdominal), or ovary. The majority of ectopic pregnancies (98%) are implanted inside the fallopian tube. (From Goodman CC, Snyder TE: Differential diagnosis for the physical therapist: screening for referral, ed 4, Philadelphia, 2007, WB Saunders.)
Incidence and Risk Factors
The incidence of ectopic pregnancy is increasing in the United States and worldwide with approximately 20 per 1000 ectopic pregnancies each year. This represents a fourfold increase globally over the last 20 years.171 The reasons for this rise are unclear, although several risk factors have been identified such as any condition that causes damage to the fallopian tubes that could impair transport of the ovum or impede the migration of the fertilized ovum to the uterus.
Three risk factors that have been traditionally associated with an increased risk of ectopic pregnancy are STDs (especially chlamydia and gonorrhea), prior tubal surgery, and current intrauterine contraceptive device (IUD, or IUCD). Other risk factors include ruptured appendix, endometriosis, pelvic inflammatory disease, douching,160 and previous ectopic pregnancy. In addition, a history of infertility and the use of clomiphene citrate to induce ovulation are associated with an increased risk of this condition.
Etiologic Factors and Pathogenesis
Ectopic pregnancy is caused by delayed ovum transport secondary to decreased fallopian tube motility or distorted tubule anatomy. Advancements in diagnostic technology have revealed a number of etiologic factors, the most common being salpingitis. Salpingitis is an infection and inflammation of the fallopian tubes.
Three to four days are typically required for the ovum to travel through the fallopian tube to the uterus. The ovum is rapidly dividing and growing throughout the journey. During ectopic pregnancy, fertilization does not occur in the uterus. The sperm fertilizes the ovum soon after the ovum enters the ampulla of the fallopian tube. If the journey is slowed sufficiently (tubule motility), the ovum becomes too large to complete the passage through the tubule. If the tubule anatomy has been affected by recurrent infection or endometriosis, the same problem occurs. The trophoblasts that cover the surface of the ovum easily penetrate the mucosa and wall of the tubule, and implantation occurs.
Bleeding occurs during implantation with leakage into the pelvis and abdominal cavity. Vaginal bleeding that may be perceived as menstruation may occur. The pregnancy will typically outgrow its blood supply, terminating the pregnancy. If the pregnancy does not terminate, the thin-walled tubule will no longer support the growing fetus, and rupture can occur by the twelfth week of gestation. Tubal rupture is life threatening because rapid intraabdominal hemorrhage can occur.
Clinical Manifestations
The classic presentation of ectopic pregnancy is marked by amenorrhea or irregular bleeding and spotting, nonspecific lower abdominal quadrant or back pain, and a pelvic mass. The woman may believe she had a menstrual period but when questioned will report the period was atypical for her.
The pain reported can be diffuse and aching or localized and will progress to a sharper, lancinating acute type of pain. The pain can be sudden in onset and intermittent and may be accompanied by hemorrhage. The pain is thought to be primarily a result of the leakage of blood into the pelvic and abdominal cavity. Pain referred to the shoulder area can occur if the blood comes in contact with the kidney or diaphragm.
Since the woman is pregnant, signs and symptoms associated with normal pregnancy may also be present. These findings include fatigue, nausea, breast tenderness, and urinary frequency.
MEDICAL MANAGEMENT
Physical examination reveals a pelvic mass in approximately 50% of the cases. Pelvic ultrasound studies can reliably reveal a gestational sac by 5 to 6 weeks into the pregnancy. An empty uterine cavity with elevated (slight) human chorionic gonadotropin-beta subunit (hCG-β) and symptoms strongly implies an extrauterine pregnancy. Blood studies may show anemia, and serum pregnancy tests (hCG-β hormonal levels) will be positive but show levels lower than expected in the presence of a normal pregnancy (lack of doubling over 2 days). Definitive diagnosis requires laparoscopy.
TREATMENT.
Surgical intervention consisting of a noninvasive laparoscopic salpingostomy to remove the ectopic pregnancy is performed if the fallopian tube has not ruptured. Laparotomy is indicated if there is internal bleeding or if the ectopic site cannot be adequately visualized with the laparoscope.
A chemotherapeutic agent, methotrexate, can be administered to remove residual ectopic tissue after laparoscopy. This drug is also used when the pregnancy is intact and surgery is contraindicated or the diagnosis is made early enough that the condition is not life threatening and preserving fertility is desirable.
DISORDERS OF THE OVARIES
Overview
Ovarian cysts, most of which are benign, are the most common form of ovarian tumor. The different types of cysts include functional cysts (follicular cysts and luteal cysts), endometrial cysts, neoplastic cysts, and cysts that result from PCOS.
The follicle and corpus luteum are the source of most symptomatic ovarian cysts in premenopausal women. These cysts are called functional cysts and rarely produce symptoms (unless they rupture or hemorrhage) because they develop in the course of normal ovarian activity. A follicular cyst develops when an egg matures but does not erupt from the follicle but rather continues to swell as it fills with fluid. A luteal cyst develops from a corpus luteum when the tissue that is left after the egg has been expelled fills with blood or other fluid.
Endometrial cysts develop when endometrial tissue migrates to the ovaries, forming blood-filled cysts called endometriomas, or chocolate cysts, because of the dark-brown color of the contents. Endometrial cysts can grow large enough to impair ovulation and cause pain and cramping during menstrual periods.
Neoplastic cysts are “new growths” considered benign in the majority of cases; only a small percentage of neoplastic cysts are cancerous or have the capacity to invade neighboring tissues and metastasize to distant sites. Cystadenomas are the most common type of neoplastic cyst.
A disorder marked by the presence of multiple cysts is called polycystic ovary syndrome (PCOS). This polycystic disorder is a hormonal disorder affecting premenopausal women and one of the most common causes of infertility. The disease gets its name from the many small cysts that build up inside the ovaries.
Incidence and Etiologic and Risk Factors
Ovarian cysts are one of the most common endocrine disorders in women, affecting 3% to 7% of women of childbearing age. The exact etiology of ovarian cysts remains unknown, but intrinsic ovarian defect combined with factors outside the ovaries is suspected.
In the case of PCOS, a series of central and peripheral mechanisms related to insulin resistance occurs that is determined genetically and inherited; PCOS is now considered a systemic metabolic disease.77,169 About 20% of women in the United States have this disorder, and more than one-half are obese. Among those women who seek treatment for infertility, more than 75% have some degree of PCOS.
Pathogenesis
Two types of ovarian cysts (the follicle and corpus luteum) are a normal part of the reproductive cycle. At least one follicle (a sac containing an egg and fluid) matures in an ovary during each cycle. During ovulation, the follicle ruptures to release the egg. The follicular remnant, or corpus luteum, is a smaller sac containing a viscous yellow liquid. The corpus luteum releases progesterone, which promotes the development of the uterine lining in preparation for the implantation of a fertilized egg.
Ovarian cysts develop when excess circulating androgens are converted to estrone in the peripheral adipose tissue. The elevated levels of circulating estrogens stimulate the release of Gn-RH by the hypothalamus and inhibit the secretion of FSH by the pituitary. Gn-RH stimulates the pituitary, which produces LH. The increased secretion of LH stimulates the ovary to produce and secrete more androgens. This self-perpetuating cycle results in abnormal maturation of the ovarian follicles, the development of multiple follicular cysts, and a persistent anovulatory state.
Several genes have been implicated in the pathogenesis of PCOS. Researchers have discovered that many women are resistant to their own insulin. To counter this resistance, the pancreas makes extra insulin; over many years this may exhaust the pancreas’ ability to make insulin and thus lead to diabetes and subsequent cardiovascular complications. In addition, high insulin levels boost the production of androgens that may induce muscular changes, leading to reduced insulin-mediated glucose uptake creating a repeated cycle and worsening condition.135
Clinical Manifestations
The likelihood of symptoms developing often has more to do with the size and location than the type of ovarian cyst. As cysts grow, their weight often pulls the ovary out of its customary position, sometimes cutting off the blood supply to the ovary. Pressure from the ovary in its new position against the uterus, bladder, intestine, or vagina may result in a variety of symptoms such as abdominal pressure; pain; abdominal bloating; or discomfort during urination, bowel movements, or sexual intercourse. Large cysts can impair ovarian function reducing ovulation and causing irregular periods or infertility in premenopausal women.
Depending on the type of cyst, if a cyst ruptures, the contents of the sac are usually absorbed by the body. When endometriomas rupture, the contents may be distributed on the uterus, bladder, and intestines. As the immune system moves in to clean up the debris, scar tissue develops, forming adhesions with resultant chronic pelvic pain. In the case of neoplastic cyst rupture, the more toxic contents can result in peritonitis.
Pain can be a manifestation of cysts. A dull, aching sensation experienced in the lower abdominal, groin, low back, or buttock areas can occur. The sensation may also be described as a heaviness. This pain is associated with bleeding into the cyst or with quick enlargement of the cyst. Sudden or sharp pain can indicate a cyst rupturing or hemorrhaging or a torsion occurring.
PCOS is characterized by physical and metabolic changes such as obesity, prominent facial or body hair, severe acne, thinning hair, infertility, and menstrual problems. Fifty percent of these women have amenorrhea, and another 30% have abnormal uterine bleeding. PCOS is associated with endometrial cancer because high levels of androgen interfere with ovulation so women with PCOS do not regularly shed the endometrium. Impaired glucose tolerance, a major risk factor for type 2 diabetes present in 40% of women with PCOS, and the subsequent risk for heart disease have been documented.194 Other symptoms or conditions associated with PCOS include obstructive sleep apnea and daytime sleepiness316 and benign breast disease (formerly fibrocystic breast disease).62
MEDICAL MANAGEMENT
The history and pelvic examination lead to suspicion of cystic disease. Confirmation is made by ultrasonography or laparoscopy. Ultrasound may be transvaginal (inserting a tampon-sized transducer into the vagina) or abdominal (moving a transducer across the lower abdomen) and can help identify the type of cyst and whether a cyst contains solid or liquid material. Laboratory tests include a complete blood count to identify infection or anemia (heavy bleeding) and a carcinoembryonic antigen-125 (CA-125) test for ovarian cancer. All women with PCOS should be screened for glucose intolerance.177
The treatment of ovarian cysts depends on the results of the diagnostic tests and the age of the woman (preserving childbearing status). In premenopausal women, the decision whether to drain or remove the cyst depends on the problems the cyst is causing (e.g., follicular or luteal cysts resolve without treatment, and endometriomas and neoplastic cysts may be removed surgically).
The treatment of PCOS is primarily hormonal with the goal being an interruption of the persistent elevated levels of androgens. Clomiphene citrate (Clomid) is often administered to induce ovulation. If medication is not effective, laser surgery can be instituted to puncture the multiple follicles.
It is now known that the application of diabetes management techniques aimed at reducing insulin resistance and hyperinsulinemia (e.g., weight reduction, oral hypoglycemic agents, and exercise) can reverse testosterone and LH abnormalities and infertility, as well as improve glucose, insulin, and lipid profiles.
Ovarian Cancer
Ovarian cancer is estimated to be the second most common female urogenital cancer and the most lethal of these cancers. The term extraovarian primary peritoneal carcinoma (EOPPC) is sometimes used interchangeably with the term ovarian cancer. This has been identified as a relatively newly defined disease that develops only in women, accounting for approximately 10% of cases with a presumed diagnosis of ovarian cancer. Characterized by abdominal carcinomatosis, uninvolved or minimally involved ovaries, and no identifiable primary form of cancer, EOPPC has been reported after bilateral oophorectomy performed for benign disease or prophylactically for ovarian cancer.82
The occurrence of EOPPC may be explained by the common origin of the peritoneum and the ovaries from the coelomic epithelium. The various histologic differences of ovarian and peritoneal lesions are under investigation.
Incidence
An estimated 23,300 women in the United States developed ovarian cancer in 2007 with 15,280 deaths from ovarian cancer.148 The poor outcome is based on the difficulty of diagnosing the disease, which results in 60% to 70% of the women having metastatic disease at the time of diagnosis. Although there are a number of types of ovarian cancers, epithelial tumors make up approximately 90% of the cases and are the leading cause of death from gynecologic cancer in the United States. The incidence of epithelial tumors peaks in women during their fifties and sixties; it is rare before puberty.
In the United States, white and Hawaiian women have the highest incidence of ovarian cancer, whereas Native American and black women have the lowest incidence.
Etiologic and Risk Factors
The etiology of ovarian cancer is not well understood. No single cause of ovarian cancer has been discovered, but hormonal, environmental, and genetic factors appear to influence the development of the disease (Box 20-4). None is as important as a family history of ovarian or breast cancer.
The average woman has less than a 2% chance of developing ovarian cancer in her lifetime (1 in 57 versus 1 in 8 for breast cancer), whereas a woman with first-degree relatives with ovarian cancer or who has the BRCA1 (BRCA stands for breast cancer) mutation has about a 45% lifetime chance, and for BRCA2, the risk is approximately 25%.263
Loss of two tumor suppressor genes (p53 and BRCA1) has been shown to occur early in ovarian carcinogenesis in women who are BRCA1 mutation carriers.326 Overall, more than 90% of all cases occur sporadically; only 10% of all women with ovarian cancer have a hereditary predisposition.
Nulliparous women are at increased risk of developing ovarian cancer. This factor may be related to the repeated epithelial surface disruption that occurs with cyclic ovulation. Since epithelial tumors make up approximately 90% of ovarian cancers, many of the risk factors described relate to this entity.
A history of breast feeding also is important, since women who breast feed are at decreased risk of developing this condition compared to nulliparous women and parous women who have not breast fed.
At the present time, there are no established nutritional risk factors for ovarian cancer. The association of milk/dairy products or calcium consumption with ovarian cancer has not been proved. Moderate alcohol consumption may reduce the risk; the role of obesity and physical activity in this cancer is unclear.173
Pathogenesis
The development of ovarian cancer seems to correlate with the number of times a woman ovulates during her lifetime. Every time an egg is released, it ruptures the surface of one of the ovaries. Cells have to replicate to repair the damage, and the more times they do this, the greater the chances that a cancer-causing mutation will occur. This is why anything that interferes with ovulation (e.g., pregnancy, breast feeding, or hormonal contraceptives) diminishes the risk of developing ovarian cancer.
The classification of ovarian tumors is based on the tissue of origin. The most common tumors arise from the surface epithelium or serosa of the ovary. As the ovary develops, the epithelium extends into the stroma of the ovary, forming glands and cysts. In certain cases, these inclusions become neoplastic. Other categories of tumor include germ cell tumors, sex cord and stromal tumors, and steroid cell tumors. Once present, ovarian cancer spreads to the pelvis, abdominal cavity, and bladder, whereas lymphatic metastasis carries the disease to the paraaortic lymph nodes and to a lesser extent the inguinal or external iliac lymph nodes. Hematogenous spread of the cancer can result in liver and lung involvement.
Clinical Manifestations
Most ovarian cancers are asymptomatic or present with symptoms so vague that the disease is advanced in many cases by the time the woman seeks care. The vague complaints include abdominal bloating, flatulence, fatigue and malaise, gastritis, or general abdominal discomfort. Abnormal vaginal bleeding, leg pain, pelvic mass, and low back pain are less common symptoms. Local pelvic pain also occurs late in the disease.
Symptoms associated with metastatic spread of the disease include unexplained weight loss, weakness, pleurisy, ascites, and cachexia (general feebleness and wasting). Ascites is an accumulation of fluid within the peritoneal cavity. This can occur when there is marked increased pressure within the liver sinusoids or portal hypertension that results in serum exuding through the superficial capillaries into the peritoneal cavity (see Fig. 16-4).
Paraneoplastic cerebellar degeneration (PCD) is a type of paraneoplastic syndrome that primarily affects women with gynecologic cancers. Symptoms typically include ataxic gait, truncal and appendicular ataxia, nystagmus, and speech impairment (dysarthria).181
MEDICAL MANAGEMENT
Women at high risk for ovarian cancer (e.g., those with a family history of ovarian cancer in a mother, sister, or daughter) and any woman with a personal history of breast, colon, or uterine cancer should receive annual screening with a combination of the CA-125 blood test, physical examination, and vaginal sonography.
There is no reliable screening test to detect ovarian cancer in its early, most curable stages. Two diagnostic tests are used but both lack sensitivity and specificity. The CA-125 blood test (carcinoembryonic antigen, a biologic marker produced by ovarian cancer cells) is elevated in about half the women with early-stage disease and about 80% of those with advanced disease (normal range is between 0 and 35). This test is not adequate as a screening tool because it does not detect the disease early in women who are asymptomatic and it can be elevated in other conditions, such as pelvic infections, fibroids, or endometriosis, and even during ovulation.
Researchers are continuing to evaluate CA-125 as a screening tool (e.g., rapidly rising CA-125 may be more predictive than elevation on a single test) and also investigating other substances, such as lysophosphatidic acid (LPA), a growth factor for ovarian cancer cells measured in the blood.254
Researchers at the University of Washington School of Medicine in Seattle have been able to accurately predict ovarian cancer based on the duration and frequency of key signs and symptoms, including pelvic/abdominal pain, urinary urgency or frequency, increased abdominal size and bloating, and difficulty eating/feeling full (early satiety). This symptom index is considered positive for ovarian cancer if any of these signs and symptoms occur more than 12 times a month for less than 12 months.
Routine ultrasound imaging is also under investigation as an effective screening tool when used to identify enlarged ovaries.270 Transvaginal ultrasonography helps determine whether an existing ovarian growth is benign or cancerous. Because the early-stage symptoms are quite nonspecific, most women do not seek medical attention until the disease is advanced.
Some women with a positive immediate family history of ovarian cancer choose to have prophylactic oophorectomies after completing childbearing to prevent the development of this disease. This intervention is not 100% protective because the lining of the peritoneal cavity comprises the same cells as the lining of the ovaries and development of primary peritoneal cancer after prophylactic oophorectomy can occur.263
A history of one or more full-term pregnancies, a history of breast feeding, and the use of hormonal contraceptives reduce the risk of ovarian cancer. Hormonal contraceptives (whether in pill form, injectable, or patch form) are recommended as chemoprevention for women with a family history of ovarian cancer, especially if the BRCA mutation is present. The mechanism of protection is unclear, but it is probably a result of the inhibition of ovulation; there is a potential increased risk of cervical cancer with this treatment.219,263
As a result of analgesics reducing the risk for colorectal cancer, studies of the effect of similar pharmacologic effects on ovarian cancer have been conducted. Regular use of acetaminophen (but not aspirin) may be associated with lower risk of ovarian cancer.217
DIAGNOSIS AND STAGING.
Despite ovarian cancer’s reputation as a silent killer, more than 90% of women with ovarian cancer (whether early or advanced) reported experiencing symptoms long before diagnosis.126 However, these are often nonspecific and vague and frequently are misdiagnosed as irritable bowel syndrome or some other nongynecologic condition. A pelvic mass with ascites is usually indicative of ovarian cancer that is then confirmed by ultrasonography. A cervical smear may reveal malignant cells, and a biopsy will reveal whether the mass is benign or cancerous.
Staging of the disease is as follows: stage I—disease limited to the ovaries; stage II—extension to other pelvic organs; stage III—intraperitoneal metastasis (spread to other abdominal organs but not the liver); and stage IV—distant metastasis (spread to the liver or organs outside the abdominal cavity). The cancer is considered advanced at stages II to IV.
For a more comprehensive breakdown of the American Joint Committee on Cancer (AJCC) TNM and FIGO staging systems for ovarian cancer, see the NCCN Clinical Practice Guidelines.226
TREATMENT.
Treatment of ovarian cancer depends on the specific tumor type but usually consists of cytoreductive surgery that includes total abdominal hysterectomy (TAH), bilateral salpingo-oophorectomy (BSO), omentectomy (removal of supportive tissue attached to organs in the abdominal cavity), and lymphadenectomy followed by adjuvant combination chemotherapy.82
Fertility-sparing surgery and comprehensive staging is done for any woman who desires to maintain her fertility.226 Anyone (men and women) interested in more information about preserving fertility after cancer should talk to their physicians about future fertility. The ACS offers an excellent review of this topic.281
Giving intraperitoneal (IP) chemotherapy along with intravenous (IV) chemotherapy may improve survival of women with Stage III ovarian cancer. IP chemotherapy allows higher doses and more frequent administration of drugs and appears more effective at killing cancer cells in the peritoneal cavity where ovarian cancer is likely to spread or recur first. Successful surgery to remove the bulk of the tumor is required first.14,325
New cancer drugs are being developed to treat resistant or recurrent disease. Ovarian growth is angiogenesis dependent, causing researchers to investigate the use of antiangiogenic treatment (e.g., angiostatin or endostatin) to inhibit tumor growth in this type of cancer.336 Other new interventions and approaches under investigation include the use of monoclonal antibodies (laboratory-produced substances that find and bind to cancer cells, delivering tumor-killing agents without harming normal cells), genetic techniques (e.g., gene therapy to supply a working copy of the tumor-suppressing gene p53), vaccines to boost a woman’s immune response to ovarian tumor cells that emerge after treatment, and new combinations and sequences of currently used drug interventions.
PROGNOSIS.
Ovarian cancer has a very poor prognosis because it is difficult to detect early and usually presents with advanced metastases (70% present in an advanced stage). The cancer is generally responsive to treatment if found early, with a 90% 5-year survival rate. In most cases, clinical response to treatment is approximately 80%, but tumor recurrence within 3 years after treatment occurs in most women.82 Return of the tumor within 6 months of therapy is a poor prognosticator because the cancer cells are often resistant to drug treatment. Five-year survival without recurrence is a good prognostic indicator.
Ovarian Varices
Incompetent and dilated ovarian veins (as well as other uterine and pelvic veins) are a known cause of abdominal and pelvic pain and contribute to pelvic congestion syndrome (Fig. 20-6). This is only one of many possible causes of chronic pelvic pain and pelvic congestion syndrome (see the section on Pelvic Floor Disorders in this chapter). Ovarian varices may occur unilaterally or bilaterally, most often in women who have had children but occasionally in nonparous women.262
Figure 20-6 Varicose veins (varicosities) of the ovary. Ovarian varicosities associated with pelvic congestion syndrome are the cause of chronic pelvic pain for women. This form of venous insufficiency is often accompanied by prominent varicose veins elsewhere in the lower quadrant (buttocks, thighs, calves). Men may have similar varicosities of the scrotum (not shown). (From Goodman CC, Snyder TE: Differential diagnosis for the physical therapist: screening for referral, ed 4, Philadelphia, 2007, WB Saunders.)
Reported symptoms include pain that worsens toward the end of the day or after standing for a long time, pain that occurs premenstrually and after intercourse, sensations of heaviness in the pelvis, and prominent varicose veins elsewhere on the body. This type of pelvic pain arises when blood pools in a distended ovarian vein rather than flowing back toward the heart and is more common among women with low blood pressure. This distention and pooling form a varicocele, a term traditionally applied to men to describe varicose veins in the testicles, but varicoceles can also occur in the female counterparts of those organs. In fact, 10% of men experience pelvic varices of the gonadal veins presenting as varicoceles similar to uteroovarian varices seen in women.40
Ovarian vein incompetence may be suspected from the presence of vulvar varicosities and can be diagnostically visualized with CT scanning, venogram, or transvaginal ultrasound. If observed during pregnancy, these may disappear after delivery but become more prominent with subsequent pregnancies.
Treatment with embolization of the ovarian veins (see discussion of this technique in the section on Uterine Fibroids in this chapter) is relatively new but reportedly safe and effective in alleviating pain and symptoms, improving sexual functioning, and reducing anxiety and depression with subsequent improved quality of life reported.185
PELVIC FLOOR DISORDERS
Overview and Incidence
Pelvic inflammatory disease (PID), the infection and inflammation of the female upper genital tract, is made up of a variety of conditions (i.e., it is not a single entity), including endometritis, salpingitis, tubo-ovarian abscess, and pelvic peritonitis. Any inflammatory condition affecting the female reproductive organs (uterus, fallopian tubes, ovaries, and cervix) may come under the diagnostic label of PID. It is a common cause of infertility, chronic pain, and ectopic pregnancy.119 Approximately 1 million women are affected each year; 75% occur in women under the age of 25 years and 100,000 become infertile.
Etiology and Risk Factors
PID occurs as a result of multimicrobial bacteria, such as Neisseria gonorrhoeae, Chlamydia trachomatis, and anaerobic and mycoplasmal bacteria. Either gonorrhea or chlamydia (two common STIs) acquired through vaginal, oral, or anal intercourse is the most likely cause. Infection can occur when the uterus is traumatized. Infection can be introduced from the skin, vagina, or GI tract. It can be an acute, one-time episode or chronic with multiple recurrences.
PID is often associated with STIs/STDs or develops after birth or after a surgical procedure involving the reproductive tract such as an abortion or a dilatation and curettage (D&C). D&C is a procedure to scrape and collect the tissue (endometrium) from inside the uterus. Dilatation (D) is a widening of the cervix to allow instruments into the uterus. Curettage (C) is the scraping of the contents of the uterus.
Early age at first vaginal intercourse (higher prevalence for age less than 15 years) and number of male sex partners are two major risk factors for PID.51 The more partners the woman has, the greater the risk of PID. PID occurs if chlamydia is not treated and even if treated, damage to the pelvic cavity cannot be reversed. Other risk factors include sexual activity without a condom, a sexual partner who reports symptoms or has a known history of chlamydia or gonorrhea, and previous pelvic infection(s).
Clinical Manifestations
Signs and symptoms of PID vary widely, making the medical diagnosis difficult. It is often asymptomatic but can present with vaginal bleeding and discharge and burning during urination. Constitutional symptoms associated with infection, such as fever or chills, and sometimes nausea and vomiting may be reported.
Painful intercourse (dyspareunia), painful menstruation, and back or pelvic pain are commonly reported. Pelvic pain does not occur until chlamydia leads to PID. Moderate (dull aching) to severe back, abdominal, and/or pelvic pain are possible. If the condition progresses to PID, scarring in the pelvic organs, including the ovaries, fallopian tubes, bowel and bladder, can cause chronic pain. The women can be left infertile because of damage and scarring to the fallopian tubes.
After one episode of PID, a woman’s risk of ectopic pregnancy increases sevenfold compared with the risk for women who have no history of PID.48,49 Ectopic pregnancy can occur when a partially blocked or slightly damaged fallopian tube causes an egg to get stuck in the fallopian tube where it is then fertilized.
MEDICAL MANAGEMENT
PID can be prevented by making safer choices to avoid STDs (e.g., always using barrier methods during intercourse, limiting number of sexual partners and with frequent testing for treatment of STDs, choosing a partner who does not have a current or previous history of STD, and abstaining from sexual activity until infected partner has completed treatment). All medication prescribed must be taken to prevent reinfection.
DIAGNOSIS, TREATMENT, AND PROGNOSIS.
PID is diagnosed on the basis of clinical presentation, a physical and pelvic examination, and laboratory tests. A vaginal swab or urine sample will be taken and sent to the laboratory. Ultrasound may be used to visualize the fallopian tubes or screen for pelvic abscess. Laparoscopic examination (thin, flexible tube with a light at the end is inserted through a small incision in the lower abdomen) allows the surgeon to view the internal organs and take tissue samples for diagnostic purposes.
PID is curable with antibiotics; prompt treatment does not reverse any damage already done. PID may require hospitalization and can be life threatening. Complications of PID include chronic pelvic pain, infertility (inability or difficulty getting pregnant), and ectopic or tubal pregnancy.
The CDC recommends that all sexually active teens and young adult women be screened annually for STIs. Any woman who has had a new sexual partner (male or female) and those with multiple sexual partners should be screened regularly. Signs of infection or recurrence of infection should be investigated immediately and treated appropriately48,49
Pelvic Floor Dysfunction
Overview and Etiologic Factors
Many diagnoses and symptoms that result in dysfunction of the pelvic floor musculature or chronic pelvic pain are included under the heading of pelvic floor disorders (Box 20-5). Although men and women can both be affected by pelvic floor dysfunction, women are more often treated in a physical therapy practice. Only general concepts related to the multitude of causes and symptoms can be covered in this text. For more specific information related to each of these conditions, the reader is referred elsewhere.289,329
Chronic pelvic pain (continuous or intermittent pelvic pain lasting for 6 months or more) is a significant part of pelvic floor disorders. Any of the conditions listed in Box 20-5 can result in chronic pelvic pain, and in turn, chronic pelvic pain can contribute to or result from hypertonus dysfunction of the pelvic floor muscles.
Many of these conditions fall into several categories and are not strictly classified as one entity. For example, vulvodynia and vulvar vestibulitis are both hypertonus dysfunctions, as well as neuropathic pain syndromes resulting in chronic pelvic pain (a third classification). Overall, there is considerable overlap of conditions in the categories listed.
Incidence
The prevalence of pelvic floor dysfunction remains unknown, but it is considered a common problem among women of reproductive age, many of whom have never been diagnosed. The lack of a consensus on the definition of chronic pelvic pain and lack of a classification scheme hinder epidemiologic studies. Although the majority of these conditions affect women, men can also be affected.
Pathogenesis
The pelvic floor muscles are a band of muscles sometimes referred to as the pubococcygeal (PC) muscles. These are actually made up of several muscle groups stretching like a sling from the pubic bone to the coccyx at the base of the spine that work together as a whole to support the internal and pelvic organs (Fig. 20-7). The pelvic floor muscles are voluntary, internal muscles surrounding the vagina, urethra, and rectum and also function to help close off the urethra and rectum to maintain continence (Fig. 20-8).
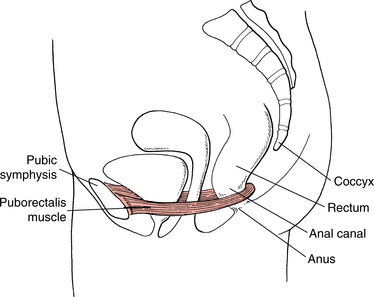
Figure 20-7 Pelvic floor sling. The first layer of the pelvic floor is made up of the endopelvic fascia, one continuous body of connective tissue surrounding and supporting the pelvic organs (not pictured). The levator ani muscles (pelvic floor muscles) make up the second layer, forming a sling across the pelvic cavity from the pubis to the coccyx with openings to allow passage of the urethra, lower vagina, and anus. The puborectalis muscle (part of the levator ani muscle) works together with the pubococcygeus muscle (not shown, also part of the levator ani muscle) to support the pelvic viscera in both the male and female. (From Myers RS: Saunders manual of physical therapy, Philadelphia, 1995, Saunders.)
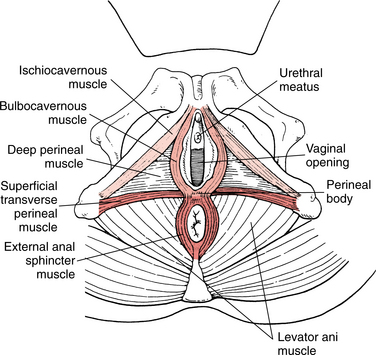
Figure 20-8 Pelvic floor muscles. Third layer of the pelvic floor, sometimes referred to as the urogenital diaphragm or deep perineum. The deep transverse perineal muscle, external urethral sphincter, and enveloping fascia of the third layer are overlaid by the fourth (superficial) layer (perineum), including the bulbocavernous, ischiocavernous, superficial transverse perineal muscles and the external anal sphincter and muscle. (From Myers RS: Saunders manual of physical therapy, Philadelphia, 1995, Saunders.)
Weakness or laxity in the endopelvic fascia or other structures of the pelvic floor results in partial or total prolapse of the organs it supports as described in the next section. Hypertonus dysfunction comprises a large portion of pelvic floor dysfunctions and is characterized by an increase in pelvic floor muscle tension or active spasm causing musculoskeletal pain or dysfunction of the urogenital and/or colorectal system.320 The pathogenesis of chronic pelvic pain remains poorly understood, and often, laparoscopic investigation reveals no obvious cause for pain.
Clinical Manifestations
Clinical manifestations of pelvic floor disorders are determined by the underlying etiologic factors and pathologic findings. For example, the primary presentation of someone with hypertonus is a pain, pressure, or ache, usually poorly localized in the perivaginal, perirectal, and lower abdominal quadrants and pelvis (suprapubic or coccyx regions) and sometimes radiating down the posterior aspect of the thigh. Symptoms are often reproduced by manual palpation and examination of the pelvic floor muscles.
Vulvar vestibulitis is characterized by a telltale patch of skin at the vaginal opening that is extremely sensitive to the gentlest tap with a cotton swab; and vulvodynia is marked by persistent pain and burning in the external genitalia, making it impossible for a woman to wear closely fitting pants or jeans, engage in sexual intercourse comfortably, or even sit comfortably.
Signs and symptoms associated with prolapsed structures, ovarian or pelvic vein varices, and endometriosis are presented elsewhere in this chapter. STDs, hernia, and GI disorders, including hemorrhoids, are presented in other chapters in this text. Other symptoms of pelvic floor dysfunction may include low back pain that is intermittent and unpredictable, changing locations often and difficult to reproduce; groin pain with hip and knee flexion; sharp rectal pain; painful intercourse with penetration or inability to penetrate; extreme rectal pressure during intercourse; urinary or bowel incontinence; abnormal vaginal discharge; and pubic bone pain or tenderness.
MEDICAL MANAGEMENT
Diagnosis and diagnostic testing depend on history and clinical presentation. Ultrasonography is being used more often now that the technology has advanced. Previously, laparoscopic examination was used with poor results in more than one half of all cases. Specific medical intervention can be employed in cases of known and treatable causes, but more often, medical management has been limited to palliative use of pharmacologic and hormonal agents and surgical intervention with variable results.
Physical therapy intervention is quickly becoming the first-line therapy of choice for many causes of pelvic floor dysfunction. Working with a counselor or other skilled professional is recommended when treating someone with a past (or current) history of abuse.
Cystocele, Rectocele, and Uterine Prolapse
The three types of pelvic floor disorders discussed here are cystocele, rectocele, and uterine prolapse. A cystocele is a herniation of the urinary bladder into the vagina (Fig. 20-9, A and B). A rectocele is a herniation of the rectum into the vagina (Fig. 20-9, C and D) in which part of the rectum protrudes into the posterior wall of the vagina, forming a pouch in the intestine. A uterine prolapse is the bulging of the uterus into the vagina (Fig. 20-10).
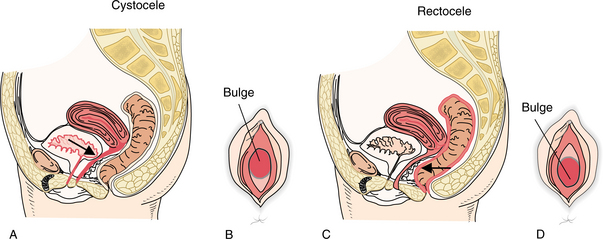
Figure 20-9 A, Cystocele (sagittal view). Note the bulging of the anterior vaginal wall. The urinary bladder is displaced downward. B, Lithotomy view: the bladder pushes the anterior vaginal wall downward into the vagina. C, Rectocele (sagittal view). D, Note the bulging of the posterior vaginal wall associated with rectocele (lithotomy view).
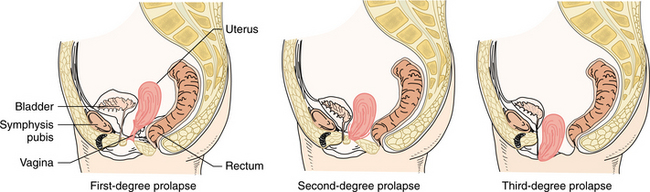
Figure 20-10 Stages of uterine prolapse. Herniation of the uterus through the pelvic floor resulting in protrusion into the vagina. First-degree: the cervix remains in the vagina. Second-degree: the cervix appears at the perineum or protrudes on straining. Third-degree: the entire uterus protrudes outside the body, and there is total inversion of the vagina.
Other types of pelvic floor prolapse may include cystourethrocele (bladder and urethra prolapse into the vagina), urethrocele (bladder neck prolapses into the vagina), enterocele (part of the intestine and peritoneum prolapses into the vagina), and vaginal vault prolapse (the apex of the vagina prolapses, occurring sometimes after a hysterectomy).
Etiologic and Risk Factors
Cystocele, rectocele, and uterine prolapse are a result of pelvic floor relaxation or structural overstretching of the pelvic musculature or ligamentous structures. Multiple pregnancies and deliveries combined with obesity increase the risk of these disorders developing. Prolonged labor, bearing down before full dilation, and forceful delivery of the placenta are possible causes of prolapse. Trauma to the pudendal or sacral nerves during birth and delivery is an additional risk factor.
Decreased muscle tone because of aging, complications of pelvic surgery, or excessive straining during bowel movements may also result in prolapse of some or all of these structures. Pelvic tumors and neurologic conditions, such as spina bifida and diabetic neuropathy, which interrupt the innervation of pelvic muscles, can also increase the risk.
Pathogenesis
The uterus and other pelvic structures are maintained in their proper position by the uterosacral, round, broad, and cardinal ligaments. The pelvic floor musculature forms a slinglike structure that supports the uterus, vagina, urinary bladder, and rectum (see Fig. 20-7). Multiple pregnancies and deliveries progressively stretch and potentially weaken or damage these important structures.
Cystocele occurs when the muscle support for the bladder is impaired. This allows the bladder to drop below the uterus. Over a period of time the vaginal wall will stretch and bulge downward. Eventually the bladder can herniate through the anterior vaginal wall and form a cystocele. A rectocele occurs when the posterior vaginal wall and underlying rectum bulge forward. Eventually protrusion occurs through the introitus as the supporting structures continue to weaken. Uterine prolapse occurs when the supporting ligaments become overstretched.
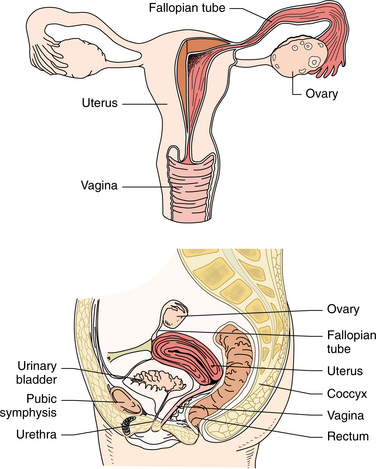
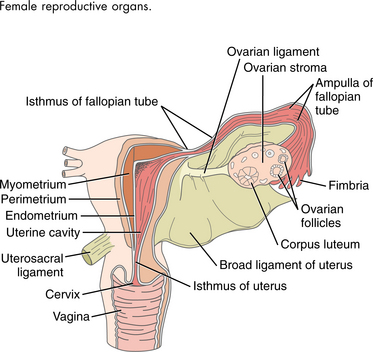
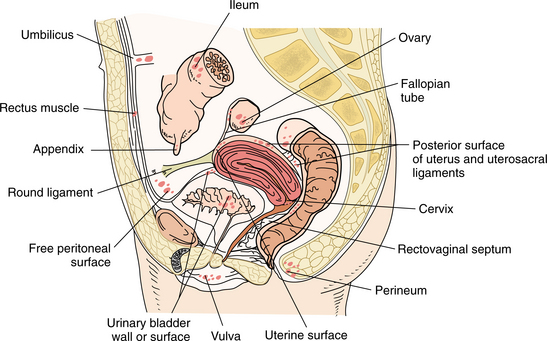
 -inch) deep and less than 7 mm (¼-inch) wide
-inch) deep and less than 7 mm (¼-inch) wide